Abstract
Objectives
Air pollution has been suggested as an important risk factor for chronic obstructive pulmonary disease (COPD); however, evidence of interactive effects on COPD between different factors was sparse, especially for young adults. We aimed to assess the combined effects of ambient ozone (O3) and household air pollution on COPD in young individuals.
Methods
We conducted a population-based study of residents aged 15–50 years in the low-income and middle-income regions of western China. We used multivariable logistic regression models to examine the associations between long-term ozone exposure and COPD in young individuals.
Results
A total of 6537 young cases were identified among the participants, with a COPD prevalence rate of 7.8 (95% CI 7.2% to 8.5%), and most young COPD individuals were asymptomatic. Exposure to household air pollution was associated with COPD in young patients after adjustment for other confounding factors (OR 1.82, 95% CI 1.41 to 2.37). We also found positive associations of COPD with O3 per IQR increase of 20 ppb (OR 1.92, 95% CI 1.59 to 2.32). The individual effects of household air pollution and O3 were 1.68 (95% CI 1.18 to 2.46) and 1.55 (95% CI 0.99 to 2.43), respectively, while their joint effect was 3.28 (95% CI 2.35 to 4.69) with the relative excess risk due to interaction of 1.05 (95% CI 0.33 to 1.78).
Conclusions
This study concludes that exposure to ambient O3 and household air pollution might be important risk factors for COPD among young adults, and simultaneous exposure to high levels of the two pollutants may intensify their individual effects.
Keywords: COPD epidemiology, Health Economist
WHAT IS ALREADY KNOWN ON THIS TOPIC
Ozone and household air pollution are considered to be major risk factors for non-smoking people with chronic obstructive pulmonary disease (COPD). Early exposure to these risk factors may be failed to achieve optimal lung function trajectories, which may contribute to younger population with COPD. We aimed to estimate the prevalence of COPD for young individuals in the low-income and middle-income regions and identify the combined effect of ozone and household air pollution on COPD for these populations.
WHAT THIS STUDY ADDS
Our findings confirmed that COPD in the young population is highly prevalent but most are asymptomatic. Simultaneous exposure to ambient ozone and household air pollution could intensify their individual effects on COPD.
HOW THIS STUDY MIGHT AFFECT RESEARCH, PRACTICE OR POLICY
The prevalence of COPD in young people is no longer negligible. Enhancing prevention and control of air pollution (including ozone and household air pollution) should be considered public health priorities in low-income and middle-income regions.
Introduction
Chronic obstructive pulmonary disease (COPD) is one of the leading causes of morbidity and mortality in low-income and middle-income countries (LMICs), leading to substantial clinical, economic and societal burdens.1 2 COPD has been traditionally considered a disease of old people, self-inflicted by tobacco smoking, air pollution, tuberculosis and other environmental factors. Many studies on COPD have mainly focused on older patients aged more than 40 years with established severe disease, as these cases comprise a significant proportion of the clinical consultations.3 4 Yet, it is time for a change that attached importance to the primary prevention of COPD for young adults. It is anticipated that early diagnosis of COPD in young individuals may offer a window of opportunity for taking preventive measures and pharmacological interventions, resulting in better outcomes and slowing down disease progression.
Air pollution contributes to approximately 50% of the risk of COPD, and more so in LMICs.5 Household air pollution (HAP) is mainly caused by the burning of solid fuel for cooking or heating and is associated with a variety of respiratory diseases. Approximately half of the world’s population uses solid fuels, especially in rural areas in LMICs. The most highly exposed are women of childbearing age and young people.6 Several epidemiological studies have reported an increased risk of COPD with HAP.7–9 Ambient ozone (O3), a typical secondary air pollutant, has been ranked as the fifth largest cause of COPD in recent decades. The effects of ozone exposure on COPD include contributions to COPD mortality,10 new-onset COPD11 and an increased risk of COPD hospital admission.12 However, no evidence of the interactive effects of HAP and O3 has been reported. Given that both air pollutants have irritated effects on COPD, we thus hypothesised that HAP and O3 may have both independent and interactive associations with the prevalence of COPD among young individuals.
In this context, we first aimed to describe the general characteristics of people with COPD among young participants aged 15–50 years in the low-income and middle-income regions. In addition, we also investigated whether exposure to HAP and O3 was associated with COPD and examined their combined effects on COPD in the study population.
Methods
Study design and participants
The multistage stratified sampling was used to enrol a regionally representative sample of adults aged 15 years or older between June 2015 and August 2016, details of which have been reported elsewhere.13 14 First, 13 districts or counties (4 cities and 9 counties) were selected in both urban and rural areas of Tibet and Xinjiang using the probability proportional to size method. Then, two streets and townships were selected with the simple random sampling method from each city and county, respectively. Third, three communities or village communities were selected using simple random sampling method from each of the street or township, respectively. Finally, participants from each of the sex/age strata from communities or villages were chosen using the simple random sampling method. Only one participant was selected from each household. The proportion of samples from each gender and age group was based on the 2010 census of Chinese population. A standardised questionnaire covering sociodemographic status, living conditions, respiratory symptoms, comorbidities, and environmental and occupational factors was administered by experienced interviewers at local community health centres. Furthermore, a range of physical measurements was taken using a standard protocol, including anthropometry, blood pressure, oxygen saturation by pulse oximetry and lung function by spirometry.
Pulmonary function tests were measured by trained technicians in all qualified study participants (spirometry) with a MasterScreenTM Pneumo PC spirometer (CareFusion, Yorba Linda, California) according to the American Thoracic Society/European Respiratory Society.15 Each participant underwent the same procedure two times, before and after receiving a bronchodilator (BD) (400 µg of salbutamol through a 500 mL spacer). The forced expiratory manoeuvres were performed 3–8 times until the lung function results achieved acceptable repeatability, defined as the differences between the two highest forced expiratory volume in 1 s (FEV1) values and the two highest forced vital capacity (FVC) values within 150 mL.16 Data were uploaded daily to a database and examined for inconsistencies by the study supervisors and the principal investigator. Quality control was performed by a field supervisor at the filing centre and included analysis of flow volume curves for artefacts and appropriate techniques. We excluded those participants whose lung function results did not meet the quality control requirements.
COPD was defined as a postbronchodilator FEV1/FVC ratio of <0.70 based on Global Initiative for Chronic Obstructive Lung Disease guidelines. Young COPD was a subpopulation of the population with COPD less than 50 years of age.17 18 A postbronchodilator FEV1/FVC ratio below the lower limit of normal (LLN) was used to define young COPD for the 15–50 years in a sensitivity analysis based on the reference values on GLI lung function equations for a North East Asian population. Small airway dysfunction was diagnosed when at least two of these three indicators, MMEF, FEF at 50% of vital capacity and FEF at 75% of vital capacity, were less than 65% of predicted values.19 HAP was defined as the use of charcoal, coal, coke, wood, crop residues or dung as the primary means of cooking or heating during the previous 6 months or longer. Ever smoker, never smoker, post-tuberculosis, occupational exposure, history of asthma and respiratory symptoms have been previously reported.13 14
Ground maximum daily 8-hour average (MDA8) ozone concentrations were predicted by random forest models at the daily level and 1 km×1 km spatial resolution in 2013–2019 in mainland China. The detailed methodology was described in a previous study and summarised here.20 Random forest models were developed by combining ground ozone measurements from fixed stations, ozone simulations from the Community Multiscale Air Quality modelling system, meteorological parameters, road length, elevation and population density. Overall, the tenfold cross validation R2 and root-mean-square error values between the measured and predicted MDA8 ozone at the daily level of the random forest models were 0.80 and 20.93 μg/m3, respectively. The monthly and annual mean ozone concentrations were calculated for each grid cell at a 1 km spatial resolution. They were assigned to participants based on the geographical coordinates, which were converted by the specific address information through geocoding.
Statistical analysis
All analyses were performed with R statistical program V.4.0.3 (www.r-project. org/). The statistically significant differences were tested by analysis of variance or Student’s t-test for continuous variables and by χ2 test for categorical variables. Multivariable logistic regression models were built to explore risk factors (sex, age, body mass index (BMI), educational level, smoking status, HAP, history of tuberculosis, occupational exposure, history of asthma and O3 exposure) for young COPD. The variance inflation factor was used to detect collinearity between variables. It is generally believed that there is no multicollinearity when the variance inflation factor is less than 10.21 We then examined the concentration–response relationship between exposure to O3 and COPD in young individuals using a natural spline smoothing function.
We conducted multivariable logistic regression models to investigate associations of young COPD with ozone and risk factors (smoking status, HAP, occupational exposure, history of tuberculosis and history of asthma) with adjustment for potential confounding covariates (sex, age, education level, BMI, smoking status, HAP, occupational exposure, history of tuberculosis and history of asthma except for grouping factor). Ozone levels were categorised into quartiles according to the distribution of ozone, and the lowest quartile group of ozone combined with no exposure to related risk factors was used as the reference group in the logistic regression model. Ozone levels were divided into three groups according to the 25th and 75th percentiles of ozone exposure. Group 1 is defined as ozone lower than 58 parts per billion (ppb) (<25 percentile). And group 2 is defined as ozone between 58–77 ppb (25th–75th percentile). While group 3 is defined as ozone higher than 78 ppb (≥75th percentile).
To evaluate interaction effects between exposure variables, interactions of ozone and HAP were evaluated by using multiplicative and additive interaction terms. The multivariable logistics model fit via multiplicative interaction was assessed by including a product term between O3 and HAP. The product term between O3 and HAP is the multiplicative interaction coefficient. ‘exp(multiplicative interaction coefficient)=1’ means no interaction; ‘exp(multiplicative interaction coefficient)>1’ represents synergistic effect; ‘exp(multiplicative interaction coefficient)<1’ represents antagonism effect.22 Additive interactions were examined using three indicators: relative excess risk due to interaction (RERI), attributable proportion (AP) and synergy index (SI).23 RERI or AP more than 0 or SI greater than 1 denoted a synergetic interaction, meaning that the joint effects of O3 and HAP were larger than the sum of their individual effects. RERI or AP less than 0 or SI smaller than 1 indicated an antagonistic interaction, meaning that with simultaneous exposure to the two pollutants, one pollutant could reduce the effect of the other.24 25 We classified O3 into two levels (low and high) using the median value as the cut-point, based on which we created a new variable to represent the combination of the variable. As a categorical variable, it had four categories: (1) low O3 exposure and no exposure to HAP; (2) low O3 exposure and exposure to HAP; (3) high O3 exposure and no exposure to HAP and (4) high O3 exposure and exposure to HAP. Differences with two-sided p<0.05 were considered statistically significant.
Results
Participants were recruited between June 2015 and August 2016. Of the 12 991 subjects invited to participate in the survey, 11 747 completed the survey questionnaire. After excluding 3630 participants aged more than 50 years old, 8117 participants underwent the spirometry examination. Among them, 1056 participants were excluded from this analysis because they could not complete post-BD testing; 504 participants were excluded because they had no reliable spirometric data. The final analysis for determining COPD included 6537 individuals (3256 men and 3281 women), with an overall mean age of 33.39 (SD 10.23) years (online supplemental figure 1).
thorax-2022-219691supp001.pdf (277.6KB, pdf)
The distribution of our study population by general characteristics and risk factors is summarised in table 1. Young COPD individuals were more likely to be older, overweight, have a lower educational level and have a more frequent history of asthma. They had a higher frequency of exposure to HAP and a higher concentration level of O3. When compared with subjects without COPD, these participants had worse pre-BD and post-BD lung function parameters. Most young people with COPD had small airway dysfunction (SAD, 84.2%), but we still found that approximately 30.3% of young adults without COPD suffered from SAD. Online supplemental table 1 presents the exposures, respiratory symptoms and spirometric results of SAD participants among young participants without COPD. These subjects with SAD had lower pre-BD and post-BD spirometric values, more recurrent wheezing symptoms, a more frequent history of asthma and TB, and more exposure to environmental factors such as HAP, ambient O3 and occupational exposure compared with ‘no SAD’ participants.
Table 1.
Characteristics of participants with chronic obstructive pulmonary disease aged less than 50 years
| Total (n=6537) | Non-COPD (n=6026) | COPD (n=511) | P value | |
| Demographic characteristics | ||||
| Gender | ||||
| Male | 3256 (49.8%) | 2993 (49.7%) | 263 (51.5%) | 0.462 |
| Female | 3281 (50.2%) | 3033 (50.3%) | 248 (48.5%) | |
| Age, years | 33.39 (10.23) | 33.07 (10.22) | 37.17 (9.47) | <0.001 |
| Education attainment | ||||
| Primary school and lower | 2271 (34.7%) | 2060 (34.2%) | 211 (41.3%) | <0.001 |
| Middle and high school | 2908 (44.5%) | 2678 (44.4%) | 230 (45.0%) | |
| College and higher | 1358 (20.8%) | 1288 (21.4%) | 70 (13.7%) | |
| BMI, kg/m2 | 23.92 (4.06) | 23.89 (4.10) | 24.28 (3.62) | 0.036 |
| City dwellers | 2301 (35.2%) | 2153 (35.7%) | 148 (29.0%) | 0.002 |
| Exposures | ||||
| Cigarette smoking | ||||
| Never-smoker | 4757 (72.8%) | 4399 (73.0%) | 358 (70.1%) | 0.161 |
| Ever-smoker | 1777 (27.2%) | 1624 (27.0%) | 153 (29.9%) | |
| History of TB | 327 (5.0%) | 294 (4.9%) | 33 (6.5%) | 0.143 |
| Household air pollution | 4551 (69.6%) | 4127 (68.5%) | 424 (83.0%) | <0.001 |
| Occupational exposure | 668 (10.6%) | 606 (10.4%) | 62 (12.6%) | 0.150 |
| O3 exposure, ppb | 69.67 (10.92) | 69.37 (10.95) | 73.27 (9.83) | <0.001 |
| Lung function parameters | ||||
| FVC pre-BD % pred | 101.86 (19.90) | 101.66 (19.83) | 104.24 (20.54) | 0.005 |
| FVC post-BD % pred | 101.64 (19.11) | 101.31 (18.75) | 105.56 (22.57) | <0.001 |
| FEV1 pre-BD % pred | 97.34 (19.86) | 98.85 (18.94) | 79.60 (21.83) | <0.001 |
| FEV1 post-BD % pred | 99.61 (20.07) | 101.66 (18.69) | 75.46 (20.02) | <0.001 |
| FEV1/FVC post-BD, % | 83.62 (9.89) | 85.60 (7.00) | 60.32 (9.18) | <0.001 |
| MMEF pre-BD % pred | 73.13 (28.75) | 75.64 (27.83) | 43.58 (22.07) | <0.001 |
| MMEF post-BD % pred | 81.46 (60.84) | 84.70 (58.40) | 40.86 (74.62) | <0.001 |
| FEF50% pre-BD % pred | 83.02 (28.06) | 85.87 (26.59) | 49.48 (22.69) | <0.001 |
| FEF50% post-BD % pred | 90.25 (29.83) | 94.20 (27.17) | 41.67 (14.44) | <0.001 |
| FEF75% pre-BD % pred | 81.42 (37.75) | 83.95 (37.05) | 51.49 (32.73) | <0.001 |
| FEF75% post-BD % pred | 91.55 (40.84) | 95.19 (39.64) | 46.77 (26.42) | <0.001 |
| Respiratory symptoms | 1618 (24.8%) | 1492 (24.8%) | 126 (24.7%) | 1.000 |
| SAD | 2233 (34.5%) | 1807 (30.3%) | 426 (84.2%) | <0.001 |
| History of asthma | 203 (3.1%) | 179 (3.0%) | 24 (4.7%) | 0.043 |
Data are expressed as the number (%) or mean (SD). The statistically significant differences were tested by analysis of variance or Student’s t test for continuous variables and by χ2 test for categorical variables.
BMI, body mass index; COPD, chronic obstructive pulmonary disease; FEF, forced expiratory flow; FEV1, forced expiratory volume in 1 s; FVC, forced vital capacity; HAP, household air pollution; MMEF, mMaximum mid-expiratory flow; O3, Ozone; post-BD, post-bronchodilator; ppb, parts per billion; pre-BD, pre-bronchodilator; SAD, small airway dysfunction; TB, tuberculosis.
The overall prevalence of COPD was 7.8% (95% CI 7.2% to 8.5%) among the young population aged less than 50 years or older (online supplemental table 2). Men had a higher prevalence (8.7%, 95% CI 7.2% to 9.1%) than women (7.6%, 95% CI 6.7% to 8.5%), but the difference for sex was not statistically significant (p=0.434). The prevalence increased with age and was 4.9% (95% CI 4.1% to 5.8%) among individuals aged 15–30 years, 7.6% (95% CI 6.5% to 8.9%) among participants aged 31–40 years and 11.8% (95% CI 10.4% to 13.3%) among those aged 41–50 years or older (p<0.001 for age difference). The overall prevalence of LLN-defined COPD was 14.0% (95% CI 13.2% to 14.9%), with 13.8% (95% CI 12.6% to 15.2%) in men and 14.3% (95% CI 13.1% to 15.5%) in women. The prevalence of LLN-defined COPD by sex and age group is shown in online supplemental figure 2. Main respiratory symptoms by COPD grades are presented in online supplemental table 3. Approximately 24.7% of COPD subjects self-reported typical respiratory symptoms, such as frequent cough, sputum, recurrent wheezing or apnoea.
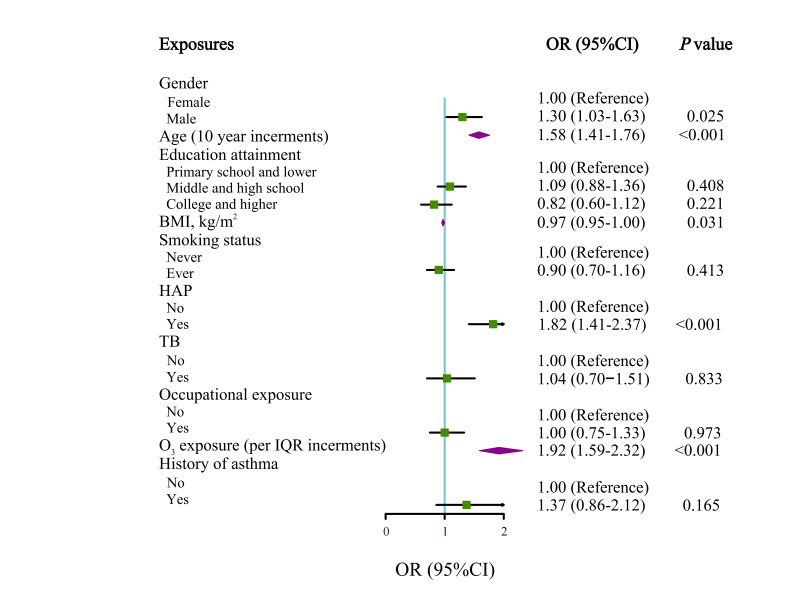
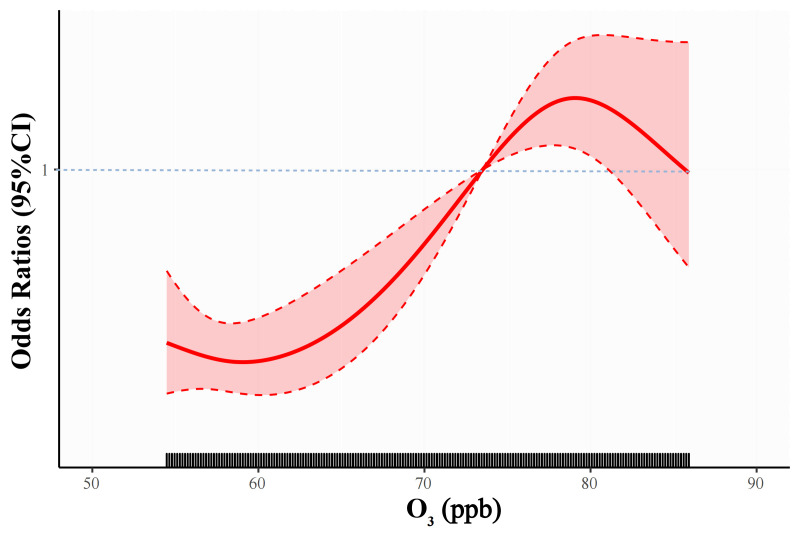
In multivariable-adjusted analyses, male sex, age, underweight (BMI<18.5 kg/m²), history of asthma, exposure to HAP and O3 were significantly associated with the risk of COPD among the young population (figure 1). In addition, we assessed the cumulative effects of O3 exposure according to the IQR group on young COPD patients (table 2). A significant interaction was noted between O3 exposure and HAP. Compared with those without exposure to HAP and exposed to less than 58 ppb O3, the risk of COPD was increased (OR 3.94, 95% CI 2.53 to 6.41) among HAP-exposed people who were exposed to 78 ppb or more O3. Figure 2 shows the concentration–response relationships of ambient O3 with COPD among the young population in the multivariable regression models.
Figure 1.
Forest plot showing OR for young COPD participants. Each square represents an OR. The horizontal lines indicate 95% CIs. Adjusted for age, sex, BMI and education plus history of asthma, history of TB and environmental exposures (ozone (O3), HAP, smoking status and occupational exposure). BMI, body mass index; COPD, chronic obstructive pulmonary disease; HAP, household air pollution; TB, tuberculosis.
Table 2.
Multiple-adjusted ORs of chronic obstructive pulmonary disease by ambient O3
| Variables* | O3 exposure, ppb | ||
| <58 | 58–77 | ≥78 | |
| HAP | |||
| No | 1.00 (Reference) | 1.52 (0.82–2.74) | 1.71 (0.98–3.02) |
| Yes | 1.70 (1.03–2.89)† | 2.74 (1.78–4.45)§ | 3.94 (2.53–6.41)§ |
| Smoking status | |||
| Never | 1.00 (Reference) | 1.43 (1.04–1.98)† | 2.13 (1.55–2.94)§ |
| Ever | 0.71 (0.33–1.37) | 1.52 (1.04–2.24)† | 1.79 (1.18–2.71)‡ |
| History of TB | |||
| No | 1.00 (Reference) | 1.53 (1.14–2.07)‡ | 2.15 (1.61–2.92)§ |
| Yes | 0.89 (0.26–2.24) | 2.04 (1.07–3.66)† | 1.99 (1.01–3.67)† |
| History of asthma | |||
| No | 1.00 (Reference) | 1.57 (1.17–2.13)‡ | 2.20 (1.64–2.97)§ |
| Yes | 1.70 (0.50–4.46) | 2.30 (1.10–4.43)† | 2.49 (1.10–5.09)† |
| Occupational exposure | |||
| No | 1.00 (Reference) | 1.64 (1.22–2.23)‡ | 2.18 (1.22–2.23)§ |
| Yes | 1.85 (0.74–4.00) | 1.87 (1.22–2.84)‡ | 1.57 (0.84–2.76) |
*First, we adjusted the common variables to be age, sex, BMI and education. Then we set grouping factor as dummy variable in combination with risk factors of COPD with adjusting for other variables. For example, when HAP as group variable, we adjusted for history of asthma, history of TB and environmental exposures (smoking status and occupational exposure).
†P<0.05.
‡P<0.01.
§P<0.001.
BMI, body mass index; COPD, chronic obstructive pulmonary disease; HAP, household air pollution; O3, ozone; ppb, parts per billion; TB, tuberculosis.
Figure 2.
Concentration response curves between ozone (O3) exposures and COPD. COPD, chronic obstructive pulmonary disease; ppb, parts per billion.
Table 3 depicts the interaction between HAP and O3 on the prevalence of COPD in young people by additive interactions analysis. Using the low O3-no HAP group as a reference, we found that the ORs in the other three groups (low-yes, high-no and high-yes) were higher than that in the reference group. In the interaction model, we found a larger joint effect than the sum of their individual effects, indicating a synergistic interaction. The individual effects of HAP and O3 were 1.68 (95% CI 1.18 to 2.46) and 1.55 (95% CI 0.99 to 2.43), respectively, while their joint effect was 3.28 (95% CI 2.35 to 4.69) with the RERI of 1.05 (95% CI 0.33 to 1.78), the AP of 0.32 (95% CI 0.10 to 0.54) and the SI of 1.85 (95% CI 0.99 to 3.46). The multiplicative interaction analysis was presented in online supplemental table 4.
Table 3.
The interactive effects between HAP and O3 on the prevalence of COPD in the study population
| Category | N | Adjusted OR* (95% CI) | P value | RERI (95% CI) | AP (95% CI) | SI (95% CI) |
| O3-HAP | 1.05 (0.33 to 1.78) | 0.32 (0.10 to 0.54) | 1.85 (0.99 to 3.46) | |||
| Low-no | 1208 | Reference | ||||
| Low-yes | 1938 | 1.68 (1.18 to 2.46) | 0.005 | |||
| High-no | 778 | 1.55 (0.99 to 2.43) | 0.057 | |||
| High-yes | 2613 | 3.28 (2.35 to 4.69) | <0.001 |
*We controlled for age, sex, BMI and education plus history of asthma, history of TB and environmental exposures (smoking status and occupational exposure).
AP, attributable proportion; BMI, body mass index; COPD, chronic obstructive pulmonary disease; HAP, household air pollution; O3, ozone; RERI, relative excess risk due to interaction; SI, synergy index.
Discussion
To the best of our knowledge, this is the first study to estimate the prevalence and burden of COPD among young participants in the low-income and middle-income regions of Western China. First, using a rigorous sampling design and stringent quality control process, we found the overall prevalence of COPD to be rather high (7.8%), considering the young age of the participants for the first time, and the prevalence of COPD has been reported among the Chinese population aged <20 years in China. Second, approximately one-third of young adults without COPD suffer from SAD. These people with SAD were exposed to high concentrations of ambient O3 and had a higher proportion of HAP. Finally, ambient O3 and HAP were identified as major preventable risk factors for COPD. In particular, there may be a synergistic interaction between O3 and HAP on the effect of COPD in young individuals in the study population.
Many epidemiological studies have reported the prevalence of COPD among young individuals. The Epidemiological Study of COPD in Spain study on the prevalence of COPD established that the global prevalence of COPD in individuals aged <50 years was 3.8% (95% CI 2.9% to 5.0%).26 The Global Burden of Disease study identifies a large geographical heterogeneity of COPD prevalence in those aged <50 years by country; prevalence is highest in males in Papua New Guinea (4.99%) and United Arab Emirates (4.35%) and in females in Papua New Guinea (6.16%) and Taiwan (6.01%).5 The LEAD study found that the prevalence of pre-BD FEV1/FVC<LLN increased with age, from 6.6%, 8.8% to 8.6% in adolescence and early adulthood (15–30 years), adulthood (30–45 years) and late adulthood (45–60 years).27 In a recent large epidemiological study in China, the prevalence of COPD in adults aged 20–49 years was 2.1% (95% CI 1.4% to 3.2%).28 However, these studies included older people aged more than 40 years, did not strictly use random sampling, or incorporated no postbronchodilator testing. Furthermore, although for the data from China, this study emphasised residents in low-income and middle-income regions. This study describes that the prevalence of COPD was 4.9% (95% CI 4.1% to 5.8%) among individuals aged 15–30 years, 7.6% (95% CI 6.5% to 8.9%) among those aged 31–40 years and 11.8% (95% CI 10.4% to 13.3%) among those aged 41–50 years in the young individuals. Thus, there was a substantial disease burden in young participants with COPD among the low-income and middle-income regions.
Long-term O3 exposure is a main cause of some chronic disease burdens and is influenced by the following underlying mechanisms. Only a limited number of studies have examined the associations between O3 and COPD incidence. In the national UK cohort, Atkinson et al reported negative associations with COPD incidence, with hazard ratios (HRs) of 0.94 (95% CI 0.89 to 1.00) per 3 μg/m3 increase in O3 based on general practitioner records.29 Another population-based cohort study of all Ontarians found that each IQR increase in pollution exposure yielded 1.9 (1.3–2.5) excess cases of COPD per 100 000 adults for O3.11 The CPH study found that the association between long-term ozone exposure (warm-season average ozone concentrations of 42.1 ppb) and small airway dysfunction was consistently greater in the COPD population than in the non-COPD population.30 In our study, we observed positive associations of COPD among young participants with a per IQR increase in O3 (1.92, 95% CI 1.59 to 2.32) in the low-income and middle-income regions with relatively high ozone levels, suggesting that people residing in high poverty and high ozone areas have a potentially large respiratory health burden, especially COPD. Long-term exposure to ozone is associated with impaired pulmonary function,30 31 adverse respiratory symptoms, 32 33 increased hazard of COPD admissions,12 34 35 deterioration of lung disease and increased mortality due to diseases of the respiratory system, including COPD.10 36–38 Exposure to ozone is associated with reduced pulmonary function, increased airway inflammation and progressed emphysema. Li et al found that an IQR increase in ambient O3-8 hours max (80.5 mg/m3, 5 days) was associated with a 5.9% (95% CI −11.0% to −0.7%) reduction in FEV1 and a 6.2% (95% CI −10.9% to −1.5%) reduction in peak expiratory flow.31 Another study found that exposure to 0.06 ppm ozone for 6.6 hours of healthy young adults causes a significant decrement of FEV1 and an increase in neutrophilic inflammation in the airways.39 Niu et al performed the randomised, double-blind, cross-over, controlled exposure trial and revealed that inhalation of ozone could impair lung function and disturb microbiota and glucose homoeostasis in the respiratory tract.40 The cohort study showed that ambient concentrations of O3 were significantly associated with greater increases in percent emphysema per 10 years (O3: 0.13 per 3 parts per billion (95% CI 0.03 to 0.24)). Ambient O3 during follow-up was also significantly associated with greater increases in percent emphysema.41 Overall, our findings strengthen the evidence base by adding new results to support the association between long-term exposure to ozone and risk of COPD. In addition, decisions and policies aimed at lowering the long-term O3 concentration level are important in alleviating the public health burden associated with ambient O3 exposures. Studies in many countries have linked solid fuel exposure to an increased risk of COPD.7 42 HAP-related COPD showed greater small airway disease and less emphysema supported by CT scans and had more chronic bronchitis symptoms and greater bronchial hyper-responsiveness.43 In our study, we found that the proportion of subjects exposed to HAP was fairly high (83.0% for young COPD) and COPD had an increased risk of 82% (OR 1.82, 95% CI 1.41 to 2.37) of HAP, confirming the increasingly acknowledged impact of HAP on COPD.
In this study, we did not only investigate the associations between young COPD participants and their exposure to air pollution, including ambient ozone and household environments; we also observed that ambient O3 and HAP may have a synergistic interaction on COPD. Besides, our results found that ozone exposure level and HAP proportion were both higher in SAD group than in the no-SAD group among young people without COPD. The findings indicate that people exposed to HAP may be at greater risk for SAD and COPD in areas known to have higher levels of ozone exposure. Thus, addressing the threats of ambient and HAP from a range of sources will require strong public health policies such as alternatives to clean energy for heating or cooking, application of ventilation systems reducing the concentration of pollutes and avoiding exposure to multiple risk factors at the same time.
Our study findings have important public health implications. With a decline in lung function in early adulthood, a high prevalence of solid fuels, and heavy ozone pollution, the burden of COPD in young people is anticipated to continue to increase, especially in low-income and middle-income regions. Furthermore, a higher level of ozone exposure and HAP could be the most important factors for young COPD patients, as well as for SAD young individuals, providing more useful information for policymakers to consider more stringent air pollution control measures, especially in underdeveloped areas. Several limitations should be acknowledged. First, our cross-sectional research design cannot establish a causal relationship between air pollution (O3 and HAP) and COPD. The adjustment factors involved in this study were based on the adjustment factors reported in previous studies11 28 30 and statistical analysis. Indeed, adjustments to the cause-and-effect map may make the conclusions more accurate. Second, people with asthma were not excluded from the study population, which might cause an overestimation of COPD prevalence in younger age groups. Third, some analyses were limited by the design of the questionnaire, which was intended to be comprehensive and easy to administer, but in some cases prevented optimal detailed data collection. We were unable to quantify direct exposure to HAP caused by solid fuels beyond self-report questionnaires. Forth, the FEF50 predicted value was derived from the prediction equations of European Community for Steel and Coal report in 1993. The equation is more appropriate for adults aged 18–70 years, so there may be a bias for younger people.44 Finally, ozone exposure misclassification is possible, as the ozone concentrations were estimated from the spatiotemporal models and thus may not accurately reflect individual exposure indoors. However, we believe such measurement errors should be nondifferential and so would bias the results towards the null.
Conclusion
In conclusion, our data document the prevalence of COPD among young residents aged 15–50 years living in the low-income and middle-income regions. Ozone exposure and HAP are major preventable risk factors for the disease, and the impact on SAD young people without COPD should also pay more attention. Moreover, it seems that simultaneous exposure to high levels of the two pollutants enhances their individual effects.
Acknowledgments
We acknowledge the following investigators for their continuous support, assistance and cooperation: Wang Miao from Beijing Anzhen Hospital, HongSheng Zhang, Xiaomeng Li, Xiaoming Tan, and Aonan Li from Beijing Hospital, Zengwu Wang, Linfeng Zhang, and Xin Wang from Fu Wai Hospital, Yundai Chen, and Bin Feng from Chinese PLA General Hospital, Sinan Wu and Wenquan Liu form China-Japan Friendship Hospital.
Footnotes
Contributors: YG is responsible for the overall content as the guarantor. YG and TS conceived and designed the study. YG and TY supervised the study. ZX and HL did the statistical analysis. SS, XM and RC provided the information about fine particulate matter. ZX drafted the manuscript. DC, WL, YT, XY, YM, MP and JC response for data investigation. All authors contributed to acquisition, analysis or interpretation of data, revised the report and approved the final version before submission.
Funding: This work was supported by the National High Level Hospital Clinical Research Funding (BJ-2018-199); Ministry of Science and Technology of China (2018YFC1315101).
Competing interests: None declared.
Provenance and peer review: Not commissioned; externally peer reviewed.
Supplemental material: This content has been supplied by the author(s). It has not been vetted by BMJ Publishing Group Limited (BMJ) and may not have been peer-reviewed. Any opinions or recommendations discussed are solely those of the author(s) and are not endorsed by BMJ. BMJ disclaims all liability and responsibility arising from any reliance placed on the content. Where the content includes any translated material, BMJ does not warrant the accuracy and reliability of the translations (including but not limited to local regulations, clinical guidelines, terminology, drug names and drug dosages), and is not responsible for any error and/or omissions arising from translation and adaptation or otherwise.
Data availability statement
No data are available.
Ethics statements
Patient consent for publication
Consent obtained from parent(s)/guardian(s).
Ethics approval
This study involves human participants and the study protocol was approved by the Institutional Review Board and ethics committee of Beijing Hospital (2013BJYYEC042C-01). Participants gave informed consent to participate in the study before taking part.
References
- 1. Soriano JB, Abajobir AA, Abate KH, et al. Global, regional, and national deaths, prevalence, disability-adjusted life years, and years lived with disability for chronic obstructive pulmonary disease and asthma, 1990–2015: A systematic analysis for the global burden of disease study 2015. Lancet Respir Med 2017;5:691–706. 10.1016/S2213-2600(17)30293-X [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. James SL, Abate D, Abate KH. Disease and injury incidence and prevalence collaborators global, regional, and national incidence, prevalence, and years lived with disability for 354 diseases and injuries for 195 countries and territories, 1990-2017: A systematic analysis for the global burden of disease study 2017. Lancet 2018;392:1789–858. 10.1016/S0140-6736(18)32279-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Yang Y, Li W, Guo Y, et al. Early COPD risk decision for adults aged from 40 to 79 years based on lung Radiomics features. Front Med (Lausanne) 2022;9:845286. 10.3389/fmed.2022.845286 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Wen H, Xie C, Wang L, et al. Difference in long-term trends in COPD mortality between China and the U.S., 1992⁻2017: an Age⁻Period⁻Cohort analysis . Int J Environ Res Public Health 2019;16:1529. 10.3390/ijerph16091529 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. GBD Compare | IHME Viz Hub Available online, Available: http://vizhub.healthdata.org/gbd-compare [Accessed 17 May 2022].
- 6. Fuel for Life: Household Energy and Health Available online, Available: https://www.who.int/publications-detail-redirect/9789241563161 [Accessed 3 Jun 2022].
- 7. Long H, Xing Z, Chai D, et al. Solid fuel exposure and chronic obstructive pulmonary disease in never-Smokers. Front Med (Lausanne) 2021;8:757333. 10.3389/fmed.2021.757333 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Siddharthan T, Grigsby MR, Goodman D, et al. Association between household air pollution exposure and chronic obstructive pulmonary disease outcomes in 13 Low- and middle-income country settings. Am J Respir Crit Care Med 2018;197:611–20. 10.1164/rccm.201709-1861OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Camp PG, Ramirez-Venegas A, Sansores RH, et al. COPD phenotypes in Biomass Smoke- versus tobacco smoke-exposed Mexican women. Eur Respir J 2014;43:725–34. 10.1183/09031936.00206112 [DOI] [PubMed] [Google Scholar]
- 10. Lim CC, Hayes RB, Ahn J, et al. Long-term exposure to ozone and cause-specific mortality risk in the United States. Am J Respir Crit Care Med 2019;200:1022–31. 10.1164/rccm.201806-1161OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Shin S, Bai L, Burnett RT, et al. Air pollution as a risk factor for incident chronic obstructive pulmonary disease and asthma. A 15-year population-based cohort study. Am J Respir Crit Care Med 2021;203:1138–48. 10.1164/rccm.201909-1744OC [DOI] [PubMed] [Google Scholar]
- 12. Danesh Yazdi M, Wang Y, Di Q, et al. Long-term exposure to Pm2.5 and ozone and hospital admissions of Medicare participants in the southeast USA. Environ Int 2019;130:104879. 10.1016/j.envint.2019.05.073 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Xing Z, Sun T, Janssens J-P, et al. Airflow obstruction and small airway dysfunction following pulmonary tuberculosis: a cross-sectional survey. Thorax 2023;78:274–80. 10.1136/thoraxjnl-2021-218345 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Guo Y, Xing Z, Shan G, et al. Prevalence and risk factors for COPD at high altitude: A large cross-sectional survey of subjects living between 2,100-4,700 m above sea level. Front Med (Lausanne) 2020;7:581763. 10.3389/fmed.2020.581763 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Miller MR, Hankinson J, Brusasco V, et al. Standardisation of Spirometry. Eur Respir J 2005;26:319–38. 10.1183/09031936.05.00034805 [DOI] [PubMed] [Google Scholar]
- 16. Enright P. Fev1 and FVC Repeatability goals when performing Spirometry. Prim Care Respir J 2010;19:194. 10.4104/pcrj.2010.00031 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. GOLD Reports, . 2022. Available: https://goldcopd.org/2022-gold-reports-2/ [Accessed 19 May 2022].
- 18. Martinez FJ, Agusti A, Celli BR, et al. Treatment trials in young patients with chronic obstructive pulmonary disease and pre–chronic obstructive pulmonary disease patients: time to move forward. Am J Respir Crit Care Med 2022;205:275–87. 10.1164/rccm.202107-1663SO [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Xiao D, Chen Z, Wu S, et al. Prevalence and risk factors of small airway dysfunction, and association with smoking, in China: findings from a national cross-sectional study. Lancet Respir Med 2020;8:1081–93. 10.1016/S2213-2600(20)30155-7 [DOI] [PubMed] [Google Scholar]
- 20. Meng X, Wang W, Shi S, et al. Evaluating the Spatiotemporal ozone characteristics with high-resolution predictions in Mainland China, 2013-2019. Environ Pollut 2022;299:118865. 10.1016/j.envpol.2022.118865 [DOI] [PubMed] [Google Scholar]
- 21. Kim JH. Multicollinearity and misleading statistical results. Korean J Anesthesiol 2019;72:558–69. 10.4097/kja.19087 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Rose DS, Robinson B, Kannan S, et al. Interaction between the effects of proton pump inhibitors and aspirin on gastric wall sestamibi uptake on myocardial perfusion imaging. J Nucl Cardiol 2021;28:1976–85. 10.1007/s12350-019-01951-1 [DOI] [PubMed] [Google Scholar]
- 23. Li Y, Gao Q, Li L, et al. Additive interaction of snoring and body mass index on the prevalence of metabolic syndrome among Chinese coal mine employees: A cross-sectional study. BMC Endocr Disord 2019;19:28. 10.1186/s12902-019-0352-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Andersson T, Alfredsson L, Källberg H, et al. Calculating measures of biological interaction. Eur J Epidemiol 2005;20:575–9. 10.1007/s10654-005-7835-x [DOI] [PubMed] [Google Scholar]
- 25. de Jager DJ, de Mutsert R, Jager KJ, et al. Reporting of interaction. Nephron Clin Pract 2011;119:c158–61. 10.1159/000327598 [DOI] [PubMed] [Google Scholar]
- 26. Miravitlles M, Soriano JB, García-Río F, et al. Prevalence of COPD in Spain: impact of Undiagnosed COPD on quality of life and daily life activities. Thorax 2009;64:863–8. 10.1136/thx.2009.115725 [DOI] [PubMed] [Google Scholar]
- 27. Breyer-Kohansal R, Faner R, Breyer M-K, et al. Factors associated with low lung function in different age bins in the general population. Am J Respir Crit Care Med 2020;202:292–6. 10.1164/rccm.202001-0172LE [DOI] [PubMed] [Google Scholar]
- 28. Wang C, Li L, Li W. 2019 the lancet-CAMS health conference: a call for abstracts. Lancet 2018;392:2534–5. 10.1016/S0140-6736(18)33176-3 [DOI] [PubMed] [Google Scholar]
- 29. Atkinson RW, Carey IM, Kent AJ, et al. Long-term exposure to outdoor air pollution and the incidence of chronic obstructive pulmonary disease in a national English cohort. Occup Environ Med 2015;72:42–8. 10.1136/oemed-2014-102266 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Niu Y, Yang T, Gu X, et al. Long-term ozone exposure and small airway dysfunction: the China pulmonary health (CPH) study. Am J Respir Crit Care Med 2022;205:450–8. 10.1164/rccm.202107-1599OC [DOI] [PubMed] [Google Scholar]
- 31. Li H, Wu S, Pan L, et al. Short-term effects of various ozone Metrics on cardiopulmonary function in chronic obstructive pulmonary disease patients: results from a panel study in Beijing, China. Environ Pollut 2018;232:358–66. 10.1016/j.envpol.2017.09.030 [DOI] [PubMed] [Google Scholar]
- 32. Belz DC, Woo H, Putcha N, et al. Ambient ozone effects on respiratory outcomes among Smokers modified by neighborhood poverty: an analysis of SPIROMICS AIR. Sci Total Environ 2022;829:154694. 10.1016/j.scitotenv.2022.154694 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Evangelopoulos D, Chatzidiakou L, Walton H, et al. Personal exposure to air pollution and respiratory health of COPD patients in London. Eur Respir J 2021;58:2003432. 10.1183/13993003.03432-2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Ghanbari Ghozikali M, Heibati B, Naddafi K, et al. Evaluation of chronic obstructive pulmonary disease (COPD) attributed to atmospheric O3, No2, and So2 using air Q model (2011-2012 year). Environ Res 2016;144(Pt A):99–105. 10.1016/j.envres.2015.10.030 [DOI] [PubMed] [Google Scholar]
- 35. Khaniabadi YO, Hopke PK, Goudarzi G, et al. Cardiopulmonary mortality and COPD attributed to ambient ozone. Environ Res 2017;152:336–41. 10.1016/j.envres.2016.10.008 [DOI] [PubMed] [Google Scholar]
- 36. Chen J, Shi C, Li Y, et al. Effects of short-term exposure to ambient airborne Pollutants on COPD-related mortality among the elderly residents of Chengdu city in Southwest China. Environ Health Prev Med 2021;26:7. 10.1186/s12199-020-00925-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Chen K, Fiore AM, Chen R, et al. Future ozone-related acute excess mortality under climate and population change scenarios in China: A modeling study. PLoS Med 2018;15:e1002598. 10.1371/journal.pmed.1002598 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Kazemiparkouhi F, Eum K-D, Wang B, et al. Long-term ozone exposures and cause-specific mortality in a US Medicare cohort. J Expo Sci Environ Epidemiol 2020;30:650–8. 10.1038/s41370-019-0135-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Kim CS, Alexis NE, Rappold AG, et al. Lung function and inflammatory responses in healthy young adults exposed to 0.06 ppm ozone for 6.6 hours. Am J Respir Crit Care Med 2011;183:1215–21. 10.1164/rccm.201011-1813OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Niu Y, Chen R, Wang C, et al. Ozone exposure leads to changes in airway permeability, Microbiota and Metabolome: A randomised, double-blind, crossover trial. Eur Respir J 2020;56:2000165. 10.1183/13993003.00165-2020 [DOI] [PubMed] [Google Scholar]
- 41. Wang M, Aaron CP, Madrigano J, et al. Association between long-term exposure to ambient air pollution and change in quantitatively assessed emphysema and lung function. JAMA 2019;322:546–56. 10.1001/jama.2019.10255 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Pathak U, Gupta NC, Suri JC. Risk of COPD due to indoor air pollution from Biomass cooking fuel: A systematic review and meta-analysis. Int J Environ Health Res 2020;30:75–88. 10.1080/09603123.2019.1575951 [DOI] [PubMed] [Google Scholar]
- 43. Sood A, Assad NA, Barnes PJ, et al. ERS/ATS workshop report on respiratory health effects of household air pollution. Eur Respir J 2018;51:1700698. 10.1183/13993003.00698-2017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Quanjer PH, Tammeling GJ, Cotes JE, et al. Lung volumes and forced ventilatory flows. report working party standardization of lung function tests, European Community for steel and coal. Eur Respir J Suppl 1993;16:5–40. [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
thorax-2022-219691supp001.pdf (277.6KB, pdf)
Data Availability Statement
No data are available.