Abstract
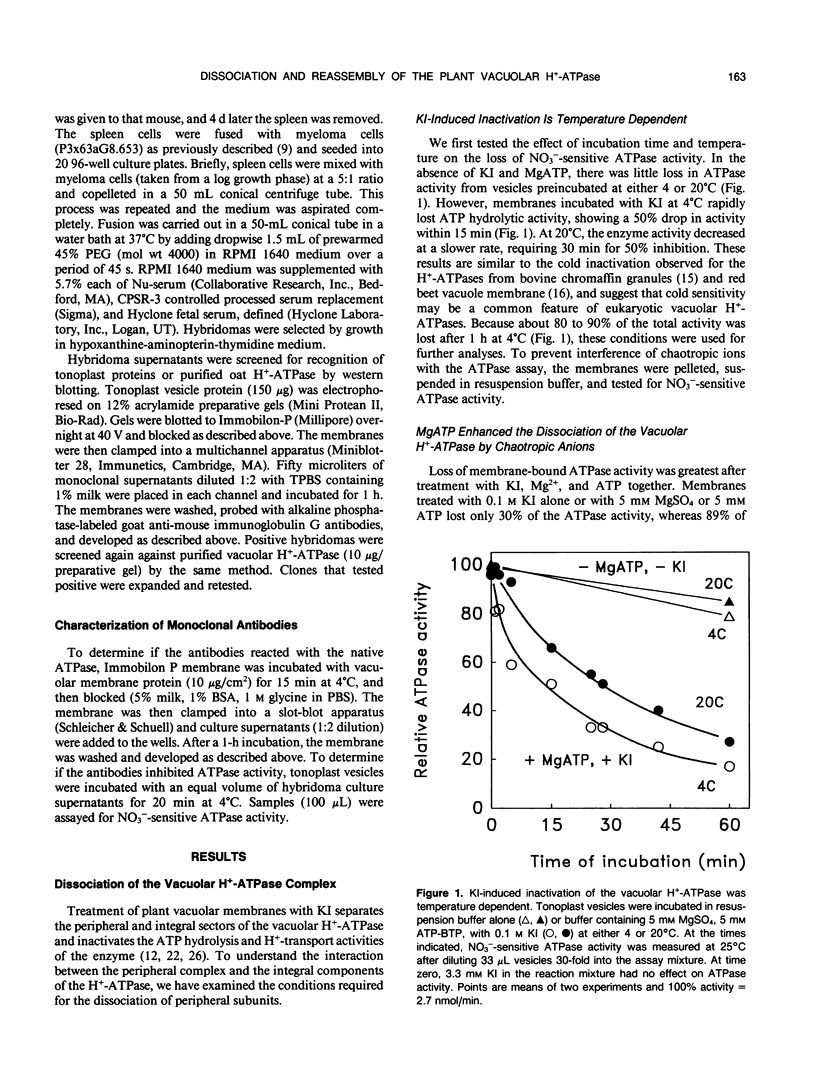
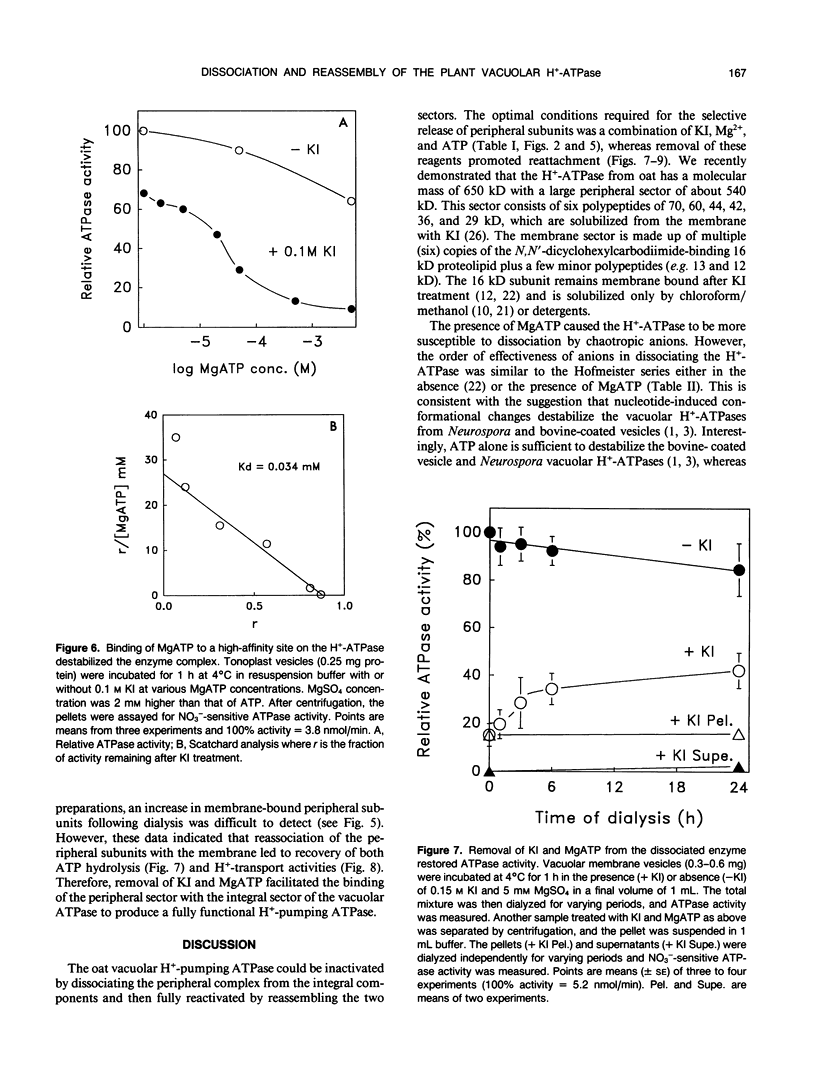
Conditions for the dissociation and reassembly of the multi-subunit vacuolar proton-translocating ATPase (H+-ATPase) from oat roots (Avena sativa var Lang) were investigated. The peripheral sector of the vacuolar H+-ATPase is dissociated from the membrane integral sector by chaotropic anions. Membranes treated with 0.5 molar KI lost 90% of membrane-bound ATP hydrolytic activity; however, in the presence of Mg2+ and ATP, only 0.1 molar KI was required for complete inactivation of ATPase and H+-pumping activities. A high-affinity binding site for MgATP (dissociation constant = 34 micromolar) was involved in this destabilization. The relative loss of ATPase activity induced by KI, KNO3, or KCl was accompanied by a corresponding increase in the peripheral subunits in the supernatant, including the nucleotide-binding polypeptides of 70 and 60 kilodaltons. The order of effectiveness of the various ions in reducing ATPase activity was: KSCN > KI > KNO3 > KBr > K-acetate > K2SO4 > KCl. The specificity of nucleotides (ATP > GTP > ITP) in dissociating the ATPase is consistent with the participation of a catalytic site in destabilizing the enzyme complex. Following KI-induced dissociation of the H+-ATPase, the removal of KI and MgATP by dialysis resulted in restoration of activity. During dialysis for 24 hours, ATP hydrolysis activity increased to about 50% of the control. Hydrolysis of ATP was coupled to H+ pumping as seen from the recovery of H+ transport following 6 hours of dialysis. Loss of the 70 and 60 kilodalton subunits from the supernatant as probed by monoclonal antibodies further confirmed that the H+-ATPase complex had reassembled during dialysis. These data demonstrate that removal of KI and MgATP resulted in reassociation of the peripheral sector with the membrane integral sector of the vacuolar H+-ATPase to form a functional H+ pump. The ability to dissociate and reassociate in vitro may have implications for the regulation, biosynthesis, and assembly of the vacuolar H+-ATPase in vivo.
Full text
PDF






Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Arai H., Pink S., Forgac M. Interaction of anions and ATP with the coated vesicle proton pump. Biochemistry. 1989 Apr 4;28(7):3075–3082. doi: 10.1021/bi00433a051. [DOI] [PubMed] [Google Scholar]
- Bowman B. J., Dschida W. J., Harris T., Bowman E. J. The vacuolar ATPase of Neurospora crassa contains an F1-like structure. J Biol Chem. 1989 Sep 15;264(26):15606–15612. [PubMed] [Google Scholar]
- Bowman E. J., Bowman B. J. Identification and properties of an ATPase in vacuolar membranes of Neurospora crassa. J Bacteriol. 1982 Sep;151(3):1326–1337. doi: 10.1128/jb.151.3.1326-1337.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bowman E. J., Mandala S., Taiz L., Bowman B. J. Structural studies of the vacuolar membrane ATPase from Neurospora crassa and comparison with the tonoplast membrane ATPase from Zea mays. Proc Natl Acad Sci U S A. 1986 Jan;83(1):48–52. doi: 10.1073/pnas.83.1.48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976 May 7;72:248–254. doi: 10.1006/abio.1976.9999. [DOI] [PubMed] [Google Scholar]
- Forgac M. Structure and function of vacuolar class of ATP-driven proton pumps. Physiol Rev. 1989 Jul;69(3):765–796. doi: 10.1152/physrev.1989.69.3.765. [DOI] [PubMed] [Google Scholar]
- Gillespie P. G., Hudspeth A. J. Chemiluminescence detection of proteins from single cells. Proc Natl Acad Sci U S A. 1991 Mar 15;88(6):2563–2567. doi: 10.1073/pnas.88.6.2563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hatefi Y., Hanstein W. G. Destabilization of membranes with chaotropic ions. Methods Enzymol. 1974;31:770–790. doi: 10.1016/0076-6879(74)31080-4. [DOI] [PubMed] [Google Scholar]
- Kaestner K. H., Randall S. K., Sze H. N,N'-dicyclohexylcarbodiimide-binding proteolipid of the vacuolar H+-ATPase from oat roots. J Biol Chem. 1988 Jan 25;263(3):1282–1287. [PubMed] [Google Scholar]
- Kasho V. N., Boyer P. D. Vacuolar ATPases, like F1,F0-ATPases, show a strong dependence of the reaction velocity on the binding of more than one ATP per enzyme. Proc Natl Acad Sci U S A. 1989 Nov;86(22):8708–8711. doi: 10.1073/pnas.86.22.8708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lai S. P., Randall S. K., Sze H. Peripheral and integral subunits of the tonoplast H+-ATPase from oat roots. J Biol Chem. 1988 Nov 15;263(32):16731–16737. [PubMed] [Google Scholar]
- Manolson M. F., Rea P. A., Poole R. J. Identification of 3-O-(4-benzoyl)benzoyladenosine 5'-triphosphate- and N,N'-dicyclohexylcarbodiimide-binding subunits of a higher plant H+-translocating tonoplast ATPase. J Biol Chem. 1985 Oct 5;260(22):12273–12279. [PubMed] [Google Scholar]
- Matsuura-Endo C., Maeshima M., Yoshida S. Subunit composition of vacuolar membrane H(+)-ATPase from mung bean. Eur J Biochem. 1990 Feb 14;187(3):745–751. doi: 10.1111/j.1432-1033.1990.tb15362.x. [DOI] [PubMed] [Google Scholar]
- Moriyama Y., Nelson N. Cold inactivation of vacuolar proton-ATPases. J Biol Chem. 1989 Feb 25;264(6):3577–3582. [PubMed] [Google Scholar]
- Parry R. V., Turner J. C., Rea P. A. High purity preparations of higher plant vacuolar H+-ATPase reveal additional subunits. Revised subunit composition. J Biol Chem. 1989 Nov 25;264(33):20025–20032. [PubMed] [Google Scholar]
- Puopolo K., Forgac M. Functional reassembly of the coated vesicle proton pump. J Biol Chem. 1990 Sep 5;265(25):14836–14841. [PubMed] [Google Scholar]
- Randall S. K., Sze H. Probing the catalytic subunit of the tonoplast H+-ATPase from oat roots. Binding of 7-chloro-4-nitrobenzo-2-oxa-1,3,-diazole to the 72-kilodalton polypeptide. J Biol Chem. 1987 May 25;262(15):7135–7141. [PubMed] [Google Scholar]
- Randall S. K., Sze H. Properties of the partially purified tonoplast H+-pumping ATPase from oat roots. J Biol Chem. 1986 Jan 25;261(3):1364–1371. [PubMed] [Google Scholar]
- Rea P. A., Griffith C. J., Sanders D. Purification of the N,N'-dicyclohexylcarbodiimide-binding proteolipid of a higher plant tonoplast H+-ATPase. J Biol Chem. 1987 Oct 25;262(30):14745–14752. [PubMed] [Google Scholar]
- Wang Y., Sze H. Similarities and differences between the tonoplast-type and the mitochondrial H+-ATPases of oat roots. J Biol Chem. 1985 Sep 5;260(19):10434–10443. [PubMed] [Google Scholar]
- Ward J. M., Sze H. Subunit Composition and Organization of the Vacuolar H-ATPase from Oat Roots. Plant Physiol. 1992 May;99(1):170–179. doi: 10.1104/pp.99.1.170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoshida S. Chilling-Induced Inactivation and Its Recovery of Tonoplast H-ATPase in Mung Bean Cell Suspension Cultures. Plant Physiol. 1991 Feb;95(2):456–460. doi: 10.1104/pp.95.2.456. [DOI] [PMC free article] [PubMed] [Google Scholar]






