Abstract
Objectives
In the first year of the COVID-19 pandemic, health systems implemented programmes to manage outpatients with COVID-19. The goal was to expedite patients’ referral to acute care and prevent overcrowding of medical centres. We sought to evaluate the impact of such a programme, the COVID-19 Home Care Team (CHCT) programme.
Design
Retrospective cohort.
Setting
Kaiser Permanente Northern California.
Participants
Adult members before COVID-19 vaccine availability (1 February 2020–31 January 2021) with positive SARS-CoV-2 tests.
Intervention
Virtual programme to track and treat patients with ‘CHCT programme’.
Outcomes
The outcomes were (1) COVID-19-related emergency department visit, (2) COVID-19-related hospitalisation and (3) inpatient mortality or 30-day hospice referral.
Measures
We estimated the average effect comparing patients who were and were not treated by CHCT. We estimated propensity scores using an ensemble super learner (random forest, XGBoost, generalised additive model and multivariate adaptive regression splines) and augmented inverse probability weighting.
Results
There were 98 585 patients with COVID-19. The majority were followed by CHCT (n=80 067, 81.2%). Patients followed by CHCT were older (mean age 43.9 vs 41.6 years, p<0.001) and more comorbid with COmorbidity Point Score, V.2, score ≥65 (1.7% vs 1.1%, p<0.001). Unadjusted analyses showed more COVID-19-related emergency department visits (9.5% vs 8.5%, p<0.001) and hospitalisations (3.9% vs 3.2%, p<0.001) in patients followed by CHCT but lower inpatient death or 30-day hospice referral (0.3% vs 0.5%, p<0.001). After weighting, there were higher rates of COVID-19-related emergency department visits (estimated intervention effect −0.8%, 95% CI −1.4% to −0.3%) and hospitalisation (−0.5%, 95% CI −0.9% to −0.1%) but lower inpatient mortality or 30-day hospice referral (−0.5%, 95% CI −0.7% to −0.3%) in patients followed by CHCT.
Conclusions
Despite CHCT following older patients with higher comorbidity burden, there appeared to be a protective effect. Patients followed by CHCT were more likely to present to acute care and less likely to die inpatient.
Keywords: COVID-19, PUBLIC HEALTH, Public health
STRENGTHS AND LIMITATIONS OF THIS STUDY
While the programme was not randomised, a natural control group was used, when demand for the programme (number of cases) went above the capacity of the programme.
We ensured that patients included in the control group would have been eligible for the intervention had it been available and carefully defined the time period to be prior to vaccine availability.
Robust methods were used to conduct the analysis (propensity scores with ensemble super learner and augmented inverse probability weighting).
Introduction
The COVID-19 pandemic posed many operational challenges for health systems. During each pandemic wave, bed demand exceeded supply, causing strain within the system to accommodate the influx of patients.1 Units had to adapt to treat patients with acute respiratory failure outside of the intensive care unit, non-urgent outpatient procedures were delayed, and providers were needed to work additional shifts.2 3 Several studies have documented higher inpatient mortality during inpatient surge periods.4–7
Considerable attention has been given to outcomes of patients hospitalised with COVID-19.6 8–12 However, limited attention has been given to outpatient care of patients with COVID-19, including managing increased volume of secure messages and clinic visits and developing guidelines for triage to the acute care setting. Integrated health systems have the unique capability of managing patients across inpatient and outpatient settings, providing opportunities to intervene prior to reaching the acute care setting and expediting their arrival to the acute care setting when necessary. Providing care upstream can reduce emergency department overcrowding by managing patients at home or by outpatient-only touchpoints. Additionally, referring patients early to acute care centres that have the capacity, even if physically located further away, prevents overcrowding and actually expedites care. Also, having a handle on the number of referrals made to the emergency department allows providers to call in more staff to assist in triaging and managing patients in a timely way.
In the first year of the pandemic, Kaiser Permanente Northern California (KPNC) repurposed resources to accommodate the increased demands on the healthcare system to support primary care physicians, manage patients with COVID-19 in the outpatient setting as much as possible and then expedite their referral to an acute care centre that was not overcrowded. The novel intervention was the COVID-19 Home Care Team (CHCT), which provided a coordinated system to track and treat outpatients who developed COVID-19. We sought to evaluate the impact of the CHCT programme on risk of hospitalisation and death.
Methods
This is a retrospective cohort study. The work was approved, and informed consent was waived by the KPNC Institutional Review Board (#1634347). A Strengthening the Reporting of Observational Studies in Epidemiology checklist is presented in the online supplemental file 1, Supplemental Methods.
bmjopen-2023-073622supp001.pdf (135.1KB, pdf)
Setting
KPNC is an integrated healthcare delivery system that cares for 30% of the population in Northern California. Under a mutual exclusivity agreement, 9500 physicians of The Permanente Medical Group provide integrated healthcare for >4.4 million Kaiser Foundation Health Plan members at 21 hospitals owned by Kaiser Foundation Hospitals and 242 medical office buildings.
Study population
To establish our base population, we identified all records of members who were ≥18 years old as of 1 February 2020 and who had a positive SARS-CoV-2 PCR test ordered between 1 February 2020 and 31 January 2021. Prior to 13 March 2020, SARS-CoV-2 tests were performed by the Centers for Disease Control and Prevention and state/county health departments, but the results were uploaded into our electronic health record system and available in KPNC databases. If a patient had multiple positive tests, we examined the characteristics and outcomes of the first positive test. The study end date was chosen because it was prior to the widespread dissemination of vaccinations for SARS-CoV-2. We excluded patients who were not eligible for CHCT service, such as those who had the first positive test during or after a COVID-19-related emergency departments visit or hospitalisation. We also excluded patients who were hospitalised within 48 hours of their positive test, because contact with the CHCT team took up to 48 hours to initiate.
Variable extraction
We examined patients’ electronic health records for demographic and clinical variables, including the following data elements: self-reported race and ethnicity (in order to show the delivery of CHCT across a population),13 individual comorbid conditions based on diagnosis codes and neighbourhood deprivation index, a composite index ranging from –5 to 5 with more positive values reflecting lower socioeconomic status.14 We also captured two composite indices that are assigned to adults in the KPNC system: a longitudinal comorbidity score (COmorbidity Point Score, V.2 (COPS2)) and an outpatient physiology-based severity of illness score (abbreviated Laboratory-based Acute Physiology Score (abLAPS)). Each month, all adults with a KPNC medical record number are assigned COPS2, which is based on diagnoses accrued in the preceding 12 months with higher scores associated with increasing mortality risk.15 They are also assigned a monthly abLAPS, which is based on 14 laboratory tests obtained in the preceding month; higher scores are associated with increased physiological derangement.13 16 These variables are more fully described in published studies.13 15 17 18
Exposure
Prior to the pandemic, KPNC had several existing population health programmes for non-pregnant adults with chronic conditions (eg, diabetes,19–21 cancer screening22 23 and cardiovascular disease21). In addition, a variety of follow-up processes existed to support primary care providers, such as combinations of inperson and automated outreach for the management of hypertensive patients.24 25 At the start of the pandemic, KPNC leadership used this population health management infrastructure to develop a novel outpatient population health programme, CHCT, with the goal of increasing front-line primary care provider support by repurposing non-physician staff, including nurses and nurse practitioners, as well as physicians from departments outside Adult and Family Medicine. After the state of California issued a Shelter in Place order in March 2020, non-emergent surgeries, procedures (eg, routine cervical cancer screening and colonoscopies) and routine specialty follow-up appointments were deprioritised. KPNC was, thus, able to repurpose ~450 non-AFM physicians from over 20 specialties as well as non-physician staff to assist in assessing and caring for patients with COVID-19 using standardised protocols which provided recommendations for when to triage patients to a higher level of care. All CHCT staff underwent formal training by the medical director (RD). CHCT provided individualised follow-up of patients with early COVID-19 infection including education, assessment and, if indicated, explicit directions for how to access emergency department care.
CHCT was designed based on KPNC’s 20+ years of doing population care. The framework for the programme was ‘Right patients, right clinicians, right tools, right oversight’. Starting in March 2020, patients were electronically enrolled in CHCT when they developed a positive SARS-CoV-2 test. Programme staff attempted to reach patients by phone soon after they were informed of their positive test result (usually within 24 hours after a positive result, including weekends). The population care platform that was embedded in the electronic medical record allowed CHCT to document outreach attempts so that multiple outreach attempts could be made. The platform allowed staff to easily record outreach attempts, customise follow-up intervals via electronic reminders and track multiple contact attempts per day. As most members were KP.org active for secure messaging, self-care instructions and care resource information were echoed by programme staff to eligible patients. Patients received in the mail a kit of supplies, including a pulse oximeter, to prevent them from needing to enter the public domain while contagious. Patients active on KPNC’s web portal were sent an automated personal message with links to information about available resources and advice on managing their symptoms. Examples of patients who were prioritised for outreach and follow-up had a history of organ transplant, supplemental oxygen at home and active treatment for cancer. Patients at moderate risk were those who were aged >60, suffered from uncontrolled diabetes and were sent to cardiac or emergency department within 30 days. Patients with no high-risk features were offered an e-visit first. Based on standardised protocols developed by front-line physicians, patients were escalated to video visits, inperson outpatient visits or acute care (emergency department). During the surge of COVID-19, the number of patients with positive tests exceeded CHCT bandwidth, which provided a natural control group of patients who were not followed by CHCT that could be used to compare outcomes. Those who attempted to be reached at least once were included in the intervention group. Additional information about the programme is available in the Supplemental Methods.
Outcome
The primary outcome was COVID-19-related acute care utilisation. We examined (1) COVID-19-related emergency department visit or (2) COVID-19-related hospitalisation within 2 weeks of positive test. Attribution of hospitalisations to COVID-19 was based on the International Classification of Diseases, V.10 codes, timing of test orders and non-elective status using a previously published algorithm.13 26 As a secondary outcome, we also examined the composite outcome of inpatient mortality or hospice referral within 30 days after a positive test as we and others have done in the past.6 13
Statistical analysis
We report mean with SD or median with IQR for continuous variables. We report number with per cent for categorical variables. We compare univariate values with t tests, Wilcoxon rank sum tests or χ2 tests, as appropriate.
For each outcome, we estimated the average treatment effect of the CHCT programme, comparing patients who were enrolled in CHCT with those who were not using an augmented inverse probability weighting (AIPW)27 estimator. In AIPW, models are developed for the propensity of treatment and outcome probability as a function of covariates, using all available data. AIPW has the appealing property that only one of the models needs to be specified correctly, known as ‘doubly robust’. We implemented the approach using the AIPW R package28 that employs the ensemble machine learning approach super learner (random forest, XGBoost, generalised additive model and multivariate adaptive regression splines) to estimate the probability models. The following variables were used in both the propensity score and outcome probability models: age, sex, neighbourhood deprivation index, abLAPS, COPS2, obesity, diabetes, hypertension and month of the pandemic. These three comorbidities were chosen because they are highly prevalent in patients with COVID-19 and impact mortality.29 Month of the pandemic was included because outcomes of patients have improved over time.6 11 30 We reported the standardised between-group differences in covariates before and after inverse probability weighting based on the propensity score. We also reported the AIPW adjusted outcome prevalence depending on whether patients were followed by CHCT or not and the estimated intervention effect with 95% CI, which is the average treatment effect.28 Threshold for significance was <0.05.
Patient involvement
Patients were not involved in the design or conduct of the study.
Results
There were 98 585 patients with positive tests, the majority of whom were followed by CHCT (n=80 067, 81.2%). Very few patients (n=18) died in the 4 weeks after a positive test without hospitalisation, and there was no difference in the percentage who died depending on whether they were followed by CHCT (n=16, 5.1%) or not (n=2, 4.9%, p=0.40). Patients followed by CHCT were older (mean age 43.9 compared with 41.6 years, p<0.001) and more comorbid with COPS2 score ≥65 (1.7% vs 1.1%, p<0.001, table 1). They were less likely to be male (45.0% vs 49.3%, p<0.001). Patients studied were diverse with 14.0% Asian, 43.7% Hispanic and 5.8% Black. They were also more likely to have diabetes (11.4% vs 5.7%, p<0.001), obesity (7.6% vs 4.8%, p<0.001) and hypertension (13.7% vs 8.4%, p<0.001). The majority of patients (n=69 150, 70.1%) had positive tests during the third wave of the pandemic (15 October 2020–31 January 2021). The time from positive test result to first contact with CHCT staff was median 1 day (IQR 0, 4). In the 30 days after positive test, the median time until the first ambulatory encounter was 1 day (IQR 1, 4) for patients followed by CHCT and 3 days (IQR 1, 7) for patients not followed by CHCT. The median time between positive test and presentation to acute care (emergency department) was 4 days (IQR 2, 7) for those followed by CHCT and 4 days (IQR 1, 8) for those not followed by CHCT.
Table 1.
Characteristics of patients with COVID-19+ who were and were not followed by COVID-19 Home Care Team
| All patients | Patients followed by CHCT | Patients not followed by CHCT | P value | |
| n=98 585 | n=80 067 | n=18 518 | ||
| Age in years, mean (SD) | 43.47 (15.54) | 43.90 (15.65) | 41.61 (14.91) | <0.001 |
| Sex, male (%) | 45 183 (45.8%) | 36 052 (45.0%) | 9131 (49.3%) | <0.001 |
| Race, n (%) | ||||
| Asian | 13 796 (14.0%) | 10 890 (13.6%) | 2906 (15.7%) | <0.001 |
| Black | 5721 (5.8%) | 4541 (5.7%) | 1180 (6.4%) | |
| Hispanic | 43 082 (43.7%) | 35 111 (43.9%) | 7971 (43.0%) | |
| White | 27 646 (28.0%) | 22 958 (28.7%) | 4688 (25.3%) | |
| Other/unknown race* | 8340 (8.5%) | 6567 (8.2%) | 1773 (9.6%) | |
| NDI (median, Q1–Q3)† | −0.07 (−0.61, 0.64) | −0.08 (−0.63, 0.64) | −0.03 (−0.53, 0.62) | <0.001 |
| COPS2 (median, Q1–Q3)‡ | 10.0 (10.0, 10.0) | 10.0 (10.0, 10.0) | 10.0 (10.0, 10.0) | <0.001 |
| COPS2 ≥65, n (%) | 1555 (1.6%) | 1350 (1.7%) | 205 (1.1%) | <0.001 |
| Comorbidities | ||||
| Diabetes, n (%) | 10 176 (10.3%) | 9122 (11.4%) | 1054 (5.7%) | <0.001 |
| Obesity, n (%) | 6988 (7.1%) | 6105 (7.6%) | 883 (4.8%) | <0.001 |
| Hypertension, n (%) | 12 505 (12.7%) | 10 948 (13.7%) | 1557 (8.4%) | <0.001 |
| Chronic pulmonary disease, n (%) | 7119 (7.2%) | 6142 (7.7%) | 977 (5.3%) | <0.001 |
| Congestive heart failure, n (%) | 764 (0.8%) | 665 (0.8%) | 99 (0.5%) | <0.001 |
| Cancer, n (%) | 1363 (1.4%) | 1173 (1.5%) | 190 (1.0%) | <0.001 |
| abLAPS (median, Q1–Q3)§ | 0.0 (0.0, 0.0) | 0.0 (0.0, 0.0) | 0.0 (0.0, 0.0) | <0.001 |
| abLAPS ≥4, n (%) | 3219 (3.3%) | 2753 (3.4%) | 466 (2.5%) | <0.001 |
| Wave, n (%) | <0.001 | |||
| 1 (1 February 2020–31 May 202) | 2476 (2.5%) | 1978 (2.5%) | 498 (2.7%) | <0.001 |
| 2 (1 June 2020–14 October 2020) | 26 959 (27.3%) | 21 580 (27.0%) | 5379 (29.1%) | |
| 3 (15 October 2020–31 January 2021) | 69 150 (70.1%) | 56 509 (70.6%) | 12 641 (68.3%) | |
| COVID-19-related emergency department visit, n (%) | 9165 (9.3%) | 7587 (9.5%) | 1578 (8.5%) | <0.001 |
| COVID-19-related hospitalisation, n (%) | 3703 (3.8%) | 3116 (3.9%) | 587 (3.2%) | <0.001 |
| Inpatient death or 30-day hospice referral, n (%) | 365 (0.4%) | 265 (0.3%) | 100 (0.5%) | <0.001 |
*Other races include American Indian, Alaska Native, Asian Pacific, Native Hawaiian, Pacific Islander and multiracial.
†Neighbourhood deprivation index ranges from −5 to +5 with more positive values indicating lower status. See text of Messer et al14 for additional detail.
‡The COmorbidity Point Score, V.2 (COPS2), described in Escobar et al15 is a score assigned every month to all adults with a Kaiser Permanente Northern California medical record number. Range is from 0 to 1010; higher scores indicate worse mortality risk. The univariate relationship between the COPS2 and 1-year mortality is as follows: 0–39, 0.3%; 40–64, 5.3%; and 65+, 17.2%.
§The abbreviated Laboratory-based Acute Physiology Score (abLAPS) is a monthly score employing 14 laboratory tests based on the LAPS score described in Escobar et al. Range is from 0 to 256; higher scores indicate increasing physiological abnormalities in the preceding month. The univariate relationship between the abLAPS and 30-day mortality is as follows: 0–4, 0.06%; 4–9, 0.18%; and 10+, 1.32%.
abLAPS, abbreviated Laboratory-based Acute Physiology Score; CHCT, COVID-19 Home Care Team; COPS2, COmorbidity Point Score, V.2; NDI, neighbourhood deprivation index.
The overall raw outcome rates were the following: 9.3% had COVID-19-related emergency department visits, 3.8% had COVID-19-related hospitalisations and 0.4% had inpatient death or 30-day hospice referral (table 1). There were more COVID-19-related emergency department visits (9.5% vs 8.5%, p<0.001) and hospitalisations (3.9% vs 3.2%, p<0.001) in patients followed by CHCT but lower inpatient death or 30-day hospice referral (0.3% vs 0.5%, p<0.001).
Table 2 shows the standardised differences in characteristics between patients followed by CHCT and not followed by CHCT. After inverse probability weighting, there were no significant differences in standardised differences of characteristics, as expected. After AIPW, there appeared to be a protective effect from the programme. There were higher rates of COVID-19-related emergency department visits (estimated intervention effect −0.8%, 95% CI −1.4% to −0.3%) and hospitalisations (−0.5%, 95% CI −0.9% to −0.1%, table 3) and lower inpatient mortality or 30-day hospice referral (−0.5%, 95% CI −0.7% to −0.3%).
Table 2.
Selected characteristics of patients who were and were not followed by COVID-19 Home Care Team before and after weighting
| Before weighting | After weighting | |||||
| Patients followed by CHCT n=80 067 |
Patients not followed by CHCT n=18 518 |
Standardised difference |
Patients followed by CHCT |
Patients not followed by CHCT |
Standardised difference, % |
|
| Age, median (IQR) | 43 (31, 55) | 40 (30, 52) | 0.15 | 42 (31, 55) | 42 (31, 55) | 0.01 |
| Sex, male (%) | 36 052 (45.0%) | 9131 (49.3%) | −0.09 | 44 889.3 (45.7) | 42 202.6 (45.9) | 0.00 |
| NDI, median (IQR)1 | 0.08 (−0.63, 0.64) | 0.03 (−0.53, 0.62) | −0.04 | −0.07 (−0.61, 0.64) | −0.08 (−0.63, 0.63) | 0.01 |
| abLAPS, median (IQR) | 0.00 (0.00, 0.00) | 0.00 (0.00, 0.00) | 0.05 | 0.00 (0.00, 0.00) | 0.00 (0.00, 0.00) | 0.02 |
| COPS2, median (IQR) | 10.00 (10.00, 10.00) | 10.00 (10.00, 10.00) | 0.10 | 10.00 (10.00, 10.00) | 10.00 (10.00, 10.00) | 0.04 |
| Obesity, n (%) | 6105 (7.6%) | 883 (4.8%) | 0.13 | 6991.7 (7.1) | 6693.1 (7.3) | −0.01 |
| Diabetes, n (%) | 9122 (11.4%) | 1054 (5.7%) | 0.25 | 10 172.2 (10.4) | 9822.9 (10.7) | −0.01 |
| Hypertension, n (%) | 10 948 (13.7%) | 1557 (8.4%) | 0.19 | 12 520.3 (12.8) | 12 463.6 (13.6) | −0.02 |
| March 2020, n (%) | 388 (0%) | 112 (1%) | −0.02 | 498.3 (0.5) | 549.7 (0.6) | −0.01 |
| April 2020, n (%) | 719 (1%) | 199 (1%) | −0.02 | 915.4 (0.9) | 903.9 (1.0) | −0.01 |
| May 2020, n (%) | 871 (1%) | 187 (1%) | 0.01 | 1061.3 (1.1) | 1011.3 (1.1) | 0.00 |
| June 2020, n (%) | 3687 (5%) | 827 (4%) | 0.01 | 4538.3 (4.6) | 4310.7 (4.7) | 0.00 |
| July 2020, n (%) | 8316 (10%) | 2277 (12%) | −0.06 | 10 575.5 (10.8) | 10 142.7 (11.0) | −0.01 |
| August 2020, n (%) | 5087 (6%) | 1290 (7%) | −0.02 | 6348.4 (6.5) | 5979.4 (6.5) | 0.00 |
| September 2020, n (%) | 3118 (4%) | 703 (4%) | 0.01 | 3803.9 (3.9) | 3628.0 (3.9) | 0.00 |
| October 2020, n (%) | 3611 (5%) | 676 (4%) | 0.05 | 4293.8 (4.4) | 4243.5 (4.6) | −0.01 |
| November 2020, n (%) | 10 888 (14%) | 2416 (13%) | 0.02 | 13 227.8 (13.5) | 12 639.9 (13.8) | −0.01 |
| December 2020, n (%) | 26 592 (33%) | 6059 (33%) | 0.01 | 32 458.2 (33.1) | 29 917.7 (32.6) | 0.01 |
| January 2021, n (%) | 16 790 (21%) | 3772 (20%) | 0.01 | 20 416.6 (20.8) | 18 567.2 (20.2) | 0.01 |
abLAPS, Abbreviated Laboratory-based Acute Physiology Score; CHCT, COVID-19 Home Care Team; COPS2, COmorbidity Point Score, V.2; NDI, neighbourhood deprivation index.
Table 3.
Estimated intervention effect of COVID-19 Home Care Team
| Adjusted outcome prevalence | Estimated intervention effect | ||
| Outcome | CHCT | No CHCT | (95% CI) |
| COVID-19-related emergency department visit | 9.3% | 10.1% | −0.8% (−1.4% to −0.3%) |
| COVID-19-related hospitalisation | 3.8% | 4.3% | −0.5% (−0.9% to −0.1%) |
| Inpatient death or 30-day hospice referral | 0.3% | 0.8% | −0.5% (−0.7% to −0.3%) |
The intervention effect being negative indicates a protective effect of the intervention.
CHCT, COVID-19 Home Care Team.
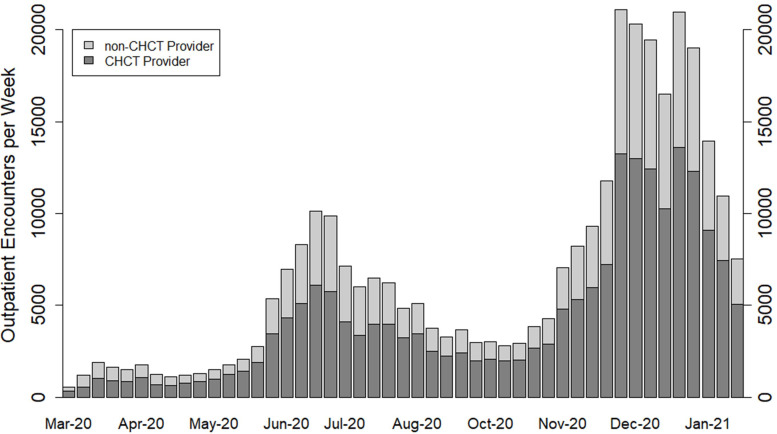
The volume of COVID-19-related outpatient visits increased dramatically during the three waves of the pandemic. The majority was conducted by CHCT providers (dark portion of stacked bar chart, figure 1), demonstrating an offloading of outpatient work to CHCT providers. In the 30 days after a positive test, patients followed by CHCT had the following encounter types (71% telephone only, 6% video only and 23% both), and patients not followed by CHCT had the following encounter types (70% telephone only, 16% video only and 12% both). In the 30 days after a positive test, patients followed by CHCT had encounters with the following clinicians (58% medical doctor (MD) only, 10% registered nurse (RN) only and 32% both), and patients not followed by CHCT had encounters with the following clinicians (96% MD only, 1% RN only and 3% both).
Figure 1.
Offloading of outpatient COVID-19-related visits to COVID-19 Home Care Team (CHCT) providers. The figure shows the number of COVID-19-related outpatient encounters per week depending on whether providers were part of CHCT (dark grey) or not (light grey). The bars shown are stacked.
Discussion
Using population-level data from an integrated health system and robust methods (AIPW), we found a protective effect of an outpatient management programme for COVID-19 which was implemented very early in the pandemic to manage patients at home and expedite their referral to acute care when needed. Patients followed by CHCT were older and had higher comorbidity burden, which argues against the possibility of cherry picking. We interpret the results to mean that patients followed by CHCT were more likely to be referred to acute care because of proactive outpatient outreach and monitoring, which likely explains the lower inpatient mortality or 30-day referral to hospice. We showed that there were drastic increases in the volume of COVID-19-related outpatient visits during the three surge periods and that much of the outpatient COVID-19-related visits were conducted by CHCT providers, demonstrating an offloading of outpatient burden by the programme. We believe these findings are important to disseminate as other health systems struggle to manage entire populations of patients through the waves of the current pandemic. The programme is scalable and generalisable, as the programme itself is delivered completely virtually.
Strain is a term that refers to the time when a clinical care team’s ability to provide high-quality care is exceeded due to high occupancy, acuity or turnover. During the pandemic, the Centers for Disease Control and Prevention estimated that if the critical care bed capacity reached 75% nationwide, 12 000 (95% CI=8623 to 17 294) excess deaths would occur nationally 2 weeks later.7 Preventing emergency room overcrowding due to unnecessary visits during periods of high transmission was critical to prevent strain-related deaths.7 11 31 32 Programmes such as CHCT attempted to streamline the management of patients with COVID-19 in the outpatient setting and to facilitate appropriate emergency room care when patients demonstrate the need for acute care.
It is important to evaluate real-world programmes, such as CHCT, that can impact outcomes for a population of patients. Patients with early COVID-19 had a tremendous need for both outpatient and inpatient care. One analysis reported that patients required 5.6–9 visits in the 30 days after the diagnosis depending on whether they ultimately were hospitalised.33 Given the rapidity of the onset of the pandemic, this demand for healthcare resources strained our healthcare system. We showed that the majority of COVID-19-related outpatient visits were managed by CHCT providers, repurposed from specialties who had the capacity at certain points during the pandemic. Additionally, the majority of CHCT encounters were via telephone, which required fewer resources than video or inperson visits. In this study, we demonstrated the management of large demand upswings due to deployment and redeployment of resources which had a positive impact on patient care.
Other programmes like CHCT have been implemented and described in the literature. The direction of our results (favouring programme benefit) is consistent with the other three programmes described here, but the details of the programme and outcomes measured differed. First, the Cleveland Clinic Home Monitoring Programme included telephone outreach to 3975 patients after a positive SARS-CoV-2 test to assess patients’ symptoms and escalate their care.34 They performed a matched propensity score analysis and found lower odds of 30- and 90-day outpatient visits and hospitalisation, but not emergency department visits. In our study, which evaluated a programme implemented at scale, we report more acute care hospitalisation and improved clinical outcomes. Second, the University of Pennsylvania COVID Watch Programme was a texting intervention whereby 3488 patients received twice daily texts to inquire about symptoms.35 They performed a propensity score analysis and found a 64% relative reduction in death for enrolled patients. They found that patients were reporting to the emergency department sooner and had more frequent telemedicine encounters. Our programme was implemented on a larger scale but found similar reduction in inpatient mortality. Third, the Home Monitoring Programme at Providence Health System delivered pulse oximeters and thermometers to the home and administered surveys in English/Spanish to monitor symptoms over time.36 The authors performed propensity scores with inverse probability of treatment weighting. Of 4358 participants, the programme was associated with more outpatient and emergency department encounters and resulted in high enrollee satisfaction. This study did not report hospitalisation rates or rates of clinical outcomes, such as death.36
There are several limitations to the current analysis. We did not measure physician or patient satisfaction/experience related to the programme. We also did not measure provider burnout created or alleviated by the programme. In an effort to reach as many people as possible, the intervention was not randomised, but we used the natural control group formed when programme demand exceeded bandwidth.
There are several key takeaways and advantages to our study. We showed that patients followed by CHCT were older, more comorbid and from diverse racial backgrounds. While being older and more comorbid could have disfavoured the programme, we found higher rates of acute care utilisation even after adjusting for confounding and lower inpatient mortality or 30-day hospice referral. Additionally, we examined patients who had had at least one outreach by CHCT into the intervention group to make it harder to find a statistically significant difference between the groups. We ensured that patients included in the analysis were eligible for CHCT services, that is, they were not hospitalised at the time of receiving a positive test or within 48 hours because it took CHCT 48 hours to initiate contact. We appropriately examined the period of the pandemic prior to widespread vaccination; including the post-vaccination period would complicate the interpretation of the result given that unvaccinated patients are more likely to be hospitalised for COVID-19 and may be less likely to engage with the programme.37 38 We also demonstrated that the number of patients who died in the 4 weeks after a positive test were few and not different between whether they were followed by CHCT or not. We performed a robust analysis using AIPW and adjustment for confounding, including month of the pandemic,11 and capitalised on the natural control group that occurred when programme demand exceeded bandwidth.
In conclusion, we evaluated the KPNC CHCT programme, which was developed and implemented early in the pandemic and at scale to manage rises in outpatient care needs related to COVID-19 surges. Despite CHCT following older patients with higher comorbidity burden, there appeared to be a protective effect with a higher likelihood of presenting to acute care but a lower likelihood of inpatient mortality. We found the programme was successful in offloading outpatient clinical care onto repurposed providers during the early part of the pandemic. This type of programme is scalable for future waves of the COVID-19 pandemic or future pandemics.
Supplementary Material
Acknowledgments
We wish to thank the Kaiser Permanente Division of Research Strategic Programming Group for formatting the dataset and The Permanente Medical Group Consulting Services for consulting on data extraction and analysis.
Footnotes
Twitter: @MDqualitysafety
Contributors: LCM is the corresponding author and guarantor for the study. LCM, BLL, GJE, Y-fIC, RD, CL and VL conceptualised the project. LCM, GJE, VL, BLL, CL and Y-fIC contributed to the data curation, formal analysis and methodology. KAD provided administrative support and contributed to generating tables and figure. LCM wrote the first draft of the manuscript, with subsequent editing done by listed coauthors; following this, LCM and KAD prepared the final version which is submitted here.
Funding: This work was supported by the Permanente Medical Group, Inc. (award/grant number N/A) and Kaiser Foundation Hospitals, Inc. (award/grant number N/A). Dr Vincent Liu was also supported by NIH grant R35GM128672. The funder(s) had no involvement in the study design; in the collection, analysis and interpretation of the data; in the writing of the report; and in the decision to submit the paper for publication.
Competing interests: None declared.
Patient and public involvement: Patients and/or the public were not involved in the design, or conduct, or reporting, or dissemination plans of this research.
Provenance and peer review: Not commissioned; externally peer reviewed.
Supplemental material: This content has been supplied by the author(s). It has not been vetted by BMJ Publishing Group Limited (BMJ) and may not have been peer-reviewed. Any opinions or recommendations discussed are solely those of the author(s) and are not endorsed by BMJ. BMJ disclaims all liability and responsibility arising from any reliance placed on the content. Where the content includes any translated material, BMJ does not warrant the accuracy and reliability of the translations (including but not limited to local regulations, clinical guidelines, terminology, drug names and drug dosages), and is not responsible for any error and/or omissions arising from translation and adaptation or otherwise.
Data availability statement
The datasets generated and/or analysed during the current study are not publicly available due to being the property of Kaiser Foundation Health Plan, Inc., but are available to interested collaborators in the context of a formal collaboration approved by the Kaiser Permanente Northern California Institutional Review Board for the Protection of Human Subjects.
Ethics statements
Patient consent for publication
Not applicable.
Ethics approval
This study involves human participants. The work was approved, and informed consent was waived by the Kaiser Permanente Northern California Institutional Review Board (#1634347).
References
- 1.Myers LC, Liu VX. The COVID-19 pandemic strikes again and again and again. JAMA Netw Open 2022;5:e221760. 10.1001/jamanetworkopen.2022.1760 [DOI] [PubMed] [Google Scholar]
- 2.Parodi SM, Liu VX. From containment to mitigation of COVID-19 in the US. JAMA 2020;323:1441–2. 10.1001/jama.2020.3882 [DOI] [PubMed] [Google Scholar]
- 3.Rouillard S, Liu VX, Corley DA. COVID-19 and long-term planning for procedure-based specialties during extended mitigation and suppression strategies. Gastroenterology 2021;160:4–9. 10.1053/j.gastro.2020.05.047 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Dang A, Thakker R, Li S, et al. Hospitalizations and mortality from non-SARS-Cov-2 causes among Medicare beneficiaries at US hospitals during the SARS-Cov-2 pandemic. JAMA Netw Open 2022;5:e221754. 10.1001/jamanetworkopen.2022.1754 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kadri SS, Sun J, Lawandi A, et al. Association between caseload surge and COVID-19 survival in 558 U.S. Ann Intern Med 2021;174:1240–51. 10.7326/M21-1213 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Asch DA, Sheils NE, Islam MN, et al. Variation in US hospital mortality rates for patients admitted with COVID-19 during the first 6 months of the pandemic. JAMA Intern Med 2021;181:471–8. 10.1001/jamainternmed.2020.8193 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.French G, Hulse M, Nguyen D, et al. Impact of hospital strain on excess deaths during the COVID-19 pandemic - United States. MMWR Morb Mortal Wkly Rep 2020;70:1613–6. 10.15585/mmwr.mm7046a5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Jeganathan N, Grewal S, Sathananthan M. Comparison of deaths from COVID-19 and seasonal influenza in the USA. Lung 2021;199:559–61. 10.1007/s00408-021-00468-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Piroth L, Cottenet J, Mariet A-S, et al. Comparison of the characteristics, morbidity, and mortality of COVID-19 and seasonal influenza: a nationwide, population-based retrospective cohort study. Lancet Respir Med 2021;9:251–9. 10.1016/S2213-2600(20)30527-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Horwitz LI. Trends in COVID-19 risk-adjusted mortality rates. J Hosp Med 2020. 10.1101/2020.08.11.20172775 [DOI] [PubMed] [Google Scholar]
- 11.Anesi GL, Jablonski J, Harhay MO, et al. Characteristics, outcomes, and trends of patients with COVID-19-related critical illness at a learning health system in the United States. Ann Intern Med 2021;174:613–21. 10.7326/M20-5327 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Myers LC, Parodi SM, Escobar GJ, et al. Characteristics of hospitalized adults with COVID-19 in an integrated health care system in California. JAMA 2020;323:2195–8. 10.1001/jama.2020.7202 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Escobar GJ, Adams AS, Liu VX, et al. Racial disparities in COVID-19 testing and outcomes: retrospective cohort study in an integrated health system. Ann Intern Med 2021;174:786–93. 10.7326/M20-6979 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Messer LC, Laraia BA, Kaufman JS, et al. The development of a standardized neighborhood deprivation index. J Urban Health 2006;83:1041–62. 10.1007/s11524-006-9094-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Escobar GJ, Gardner MN, Greene JD, et al. Risk-adjusting hospital mortality using a comprehensive electronic record in an integrated health care delivery system. Med Care 2013;51:446–53. 10.1097/MLR.0b013e3182881c8e [DOI] [PubMed] [Google Scholar]
- 16.Escobar GJ, Greene JD, Scheirer P, et al. Risk-adjusting hospital inpatient mortality using automated inpatient, outpatient, and laboratory databases. Med Care 2008;46:232–9. 10.1097/MLR.0b013e3181589bb6 [DOI] [PubMed] [Google Scholar]
- 17.Escobar GJ, Greene JD, Scheirer P, et al. Risk adjusting hospital inpatient mortality using automated inpatient, outpatient, and laboratory databases. Med Care 2008;46:232–9. 10.1097/MLR.0b013e3181589bb6 [DOI] [PubMed] [Google Scholar]
- 18.Escobar GJ, Ragins A, Scheirer P, et al. Nonelective Rehospitalizations and Postdischarge mortality: predictive models suitable for use in real time. Med Care 2015;53:916–23. 10.1097/MLR.0000000000000435 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Sadur CN, Moline N, Costa M, et al. Diabetes management in a health maintenance organization. efficacy of care management using cluster visits. Diabetes Care 1999;22:2011–7. 10.2337/diacare.22.12.2011 [DOI] [PubMed] [Google Scholar]
- 20.Schmittdiel JA, Uratsu CS, Fireman BH, et al. The effectiveness of diabetes care management in managed care. Am J Manag Care 2009;15:295–301. [PubMed] [Google Scholar]
- 21.Rana JS, Karter AJ, Liu JY, et al. Improved cardiovascular risk factors control associated with a large-scale population management program among diabetes patients. Am J Med 2018;131:661–8. 10.1016/j.amjmed.2018.01.024 [DOI] [PubMed] [Google Scholar]
- 22.Doubeni CA, Corley DA, Zhao W, et al. Association between improved colorectal screening and racial disparities. N Engl J Med 2022;386:796–8. 10.1056/NEJMc2112409 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Mehta SJ, Jensen CD, Quinn VP, et al. Race/Ethnicity and adoption of a population health management approach to colorectal cancer screening in a community-based Healthcare system. J Gen Intern Med 2016;31:1323–30. 10.1007/s11606-016-3792-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Jaffe MG, Lee GA, Young JD, et al. Improved blood pressure control associated with a large-scale hypertension program. JAMA 2013;310:699–705. 10.1001/jama.2013.108769 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Jaffe MG, Young JD. The Kaiser Permanente northern California story: improving hypertension control from 44% to 90% in 13 years (2000 to 2013). J of Clinical Hypertension 2016;18:260–1. 10.1111/jch.12803 Available: https://onlinelibrary.wiley.com/toc/17517176/18/4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Myers LC, Ng K, Plimier C, et al. Trends in outpatient visits and hospital and intensive care unit admissions of adults with COVID-19 in an integrated US health care system. JAMA Netw Open 2023;6:e2253269. 10.1001/jamanetworkopen.2022.53269 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kurz CF. Augmented inverse probability weighting and the double robustness property. Med Decis Making 2022;42:156–67. 10.1177/0272989X211027181 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Zhong Y, Kennedy EH, Bodnar LM, et al. AIPW: an R package for augmented inverse probability-weighted estimation of average causal effects. Am J Epidemiol 2021;190:2690–9. 10.1093/aje/kwab207 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Ko JY, Danielson ML, Town M, et al. Risk factors for Coronavirus disease 2019 (COVID-19)-Associated hospitalization: COVID-19-associated hospitalization surveillance network and behavioral risk factor surveillance system. Clin Infect Dis 2021;72:e695–703. 10.1093/cid/ciaa1419 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Horby P, Lim WS, et al. , RECOVERY Collaborative Group . Dexamethasone in hospitalized patients with COVID-19. N Engl J Med 2021;384:693–704. 10.1056/NEJMoa2021436 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Anesi GL, Chowdhury M, Small DS, et al. Association of a novel index of hospital capacity strain with admission to intensive care units. Ann Am Thorac Soc 2020;17:1440–7. 10.1513/AnnalsATS.202003-228OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Anesi GL, Liu VX, Gabler NB, et al. Associations of intensive care unit capacity strain with disposition and outcomes of patients with sepsis presenting to the emergency Department. Ann Am Thorac Soc 2018;15:1328–35. 10.1513/AnnalsATS.201804-241OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Huang BZ, Creekmur B, Yoo MS, et al. Healthcare utilization among patients diagnosed with COVID-19 in a large integrated health system. J Gen Intern Med 2022;37:830–7. 10.1007/s11606-021-07139-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Misra-Hebert AD, Ji X, Jehi L, et al. COVID-19 home monitoring after diagnosis and health care utilization in an integrated health system. JAMA Health Forum 2021;2:e210333. 10.1001/jamahealthforum.2021.0333 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Delgado MK, Morgan AU, Asch DA, et al. Comparative effectiveness of an automated text Messaging service for monitoring COVID-19 at home. Ann Intern Med 2022;175:179–90. 10.7326/M21-2019 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Vartanian KB, Holtorf M, Cox EJ, et al. Patient experience and Healthcare utilization for a COVID-19 Telemedicine home monitoring program offered in English and Spanish. PLoS One 2022;17:e0270754. 10.1371/journal.pone.0270754 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Kipnis P, Soltesz L, Escobar GJ, et al. Evaluation of vaccination strategies to compare efficient and equitable vaccine allocation by race and Ethnicity across time. JAMA Health Forum 2021;2:e212095. 10.1001/jamahealthforum.2021.2095 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Myers LC, Kipnis P, Greene J, et al. Adults hospitalized with breakthrough COVID-19 have lower mortality than matched Unvaccinated adults. J Intern Med 2022;292:377–84. 10.1111/joim.13504 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
bmjopen-2023-073622supp001.pdf (135.1KB, pdf)
Data Availability Statement
The datasets generated and/or analysed during the current study are not publicly available due to being the property of Kaiser Foundation Health Plan, Inc., but are available to interested collaborators in the context of a formal collaboration approved by the Kaiser Permanente Northern California Institutional Review Board for the Protection of Human Subjects.