Abstract
Systemic emboli are not uncommon in patients with advanced non-small cell lung cancer. The present study describes a rare case of long-term control in a patient with lung adenocarcinoma, nonbacterial thrombotic endocarditis and multiple systemic emboli. Briefly, a 56-year-old man was diagnosed with metastatic lung adenocarcinoma and was treated with pembrolizumab, which was discontinued due to the appearance of a pulmonary immune-related adverse event. During the clinical course, the patient developed pseudo-progression of a brain tumor, repeated thromboembolism in multiple organs and a small vegetation attached to the aortic valve. These lesions were controlled with apixaban after heparin therapy for >3 years. Lung cancer was subsequently treated with pemetrexed and bevacizumab; however, this treatment was terminated due to a complete response and the patient's request to discontinue treatment. More than 3 years have passed since the diagnosis of lung adenocarcinoma, and the patient has been followed up at the hospital without signs of cancer recurrence. Although unusual, the patient's course may provide useful suggestions for the treatment of other patients with a similar evolution.
Keywords: lung adenocarcinoma, nonbacterial thrombotic endocarditis, systemic emboli, immune checkpoint inhibitor, pembrolizmab
Introduction
Cancer patients have a high incidence of thromboembolism (1). Thrombosis associated with cancer or cancer therapy is collectively referred to as ‘cancer-associated thrombosis (CAT)’ (1). CAT includes venous thromboembolism (VTE), arterial thromboembolism, nonbacterial thrombotic endocarditis (2-4), and disseminated intravascular coagulation. Among CAT, VTE is most common and includes deep vein thrombosis and pulmonary embolism. The development of CAT can be triggered by immune-related adverse events of immune checkpoint inhibitor (ICI), which has brought about a major revolution in cancer treatment in recent years (5-9). Regardless of the path of onset, the presence of CAT complicates subsequent cancer treatment because it worsens the patient's general condition and dysfunction of important organs. As a result, long-term survival may be thwarted. For the treatment of CAT, low molecular heparin and direct oral anticoagulants (DOACs), which selectively inhibit thrombin or blood coagulation factor Xa (FXa), have proven useful (10-12).
We show herein a case of metastatic lung adenocarcinoma with nonbacterial thrombotic endocarditis and repeated arterial thromboembolism at multiple sites after ICI treatment. This patient was treated with the DOAC, ‘apixaban’ following heparin treatment. Apixaban is a DOAC that strongly and reversibly directly inhibits FXa, and exerts antithrombotic effects by suppressing thrombin production through inhibition of FXa. The CAT was considered an immune-related adverse event, and more than 3-year control was achieved by administration of apixaban, a DOAC, after heparin therapy.
Case report
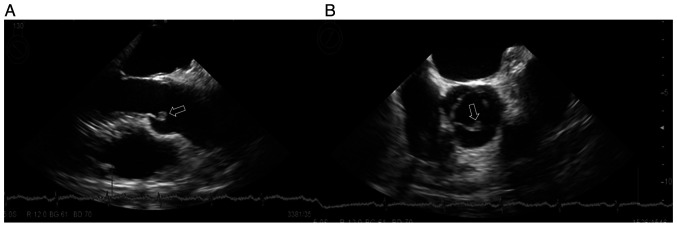
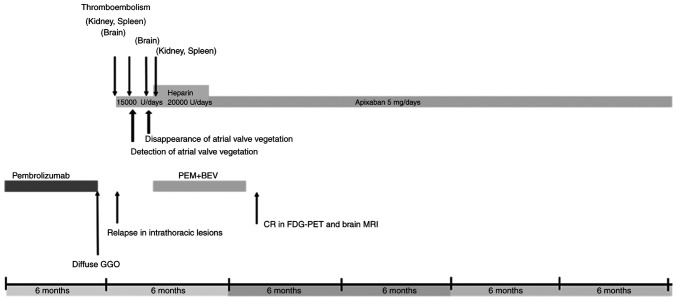
A 56-year-old man was referred to Mito Kyodo General Hospital (Mito, Japan) in August 2020 because of an abnormal opacity on a chest radiograph taken during annual mass screening. A chest CT image showed a nodule in the right lung and ipsilateral hilar lymphadenopathy (Fig. 1A). Although he was diagnosed with bronchial asthma at the age of 29, he had no previous history of arrhythmia, heart disease, or diseases that caused thromboembolism. There was no family history of genetic diseases. The patient had a 27 pack-year smoking history. At the time of admission, his body temperature was 36.8 degrees, heart rate was 84/min, blood pressure was 138/83 mmHg, and respiratory rate was 16/min. Physical examination detected no abnormalities. A biopsy specimen obtained by bronchoscopy was diagnosed histologically as lung adenocarcinoma. As two brain metastatic lesions in the right temporal and right occipital lobes were detected (Fig. 2A), his clinical stage was determined to be T2aN2M1b stage IVB. Treatment was initiated with pembrolizumab alone (200 mg at a time as an intravenous drip at 3-week intervals), considering the result of high PD-L (75%) in the biopsied cancer tissue and the patient's preference to receive ICI monotherapy. MRI taken in October 2020 revealed an increase of the occipital lobe lesion (Fig. 2B), despite a decrease of the temporal lobe lesion. The patient's general condition was good without symptoms, so he was considered to be experiencing pseudo-progression. MRI taken in November 2020 showed the shrinkage of this metastatic lesion (Fig. 2C). In an MRI taken in January 2021, complete disappearance of the lesion was confirmed (Fig. 2D). In CT taken at the same time, intrathoracic lesions also disappeared (Fig. 1B), and a complete response was achieved. However, pembrolizumab was discontinued due to the appearance of ground glass opacity in both lungs (Fig. 3A) in CT scan taken in December 2020. The patient was evaluated to have a Grade 2 (National Cancer Institute Common Terminology Criteria for Adverse Events, version 5.0) pulmonary immune-related adverse effect (irAE), and steroids were not administered. It was followed closely, and the ground glass opacity improved, but recurrence of ipsilateral lymph node lesion (Fig. 3B) in CT scan taken in February 2021, 2 months after stopping pembrolizumab. At that time, the patient developed back pain, and CT confirmed arterial thromboembolisms in the right kidney and spleen (Fig. 4). These arterial thromboembolisms occurred 4 times during the subsequent 4 months. Ultrasound examination did not show any thrombus formation in the veins of the lower extremities, but echocardiography performed at that time confirmed a 3 mm-sized vegetation attached to the aortic valve (Fig. 5). Continuous intravenous infusion of unfractionated heparin (20,000 units per day) was given for each episode of four repeated arterial thromboembolisms. After discharge, the patient underwent subcutaneous self-injection of heparin (18,000 units per day from February 2020 to July 2021) at home. Four courses of intravenous pemetrexed (500 mg/m2) and bevacizumab (15 mg/kg) repeated every 4 weeks were given for lung adenocarcinoma, and a complete response was achieved. Anticancer treatment was terminated due to the patient's request to discontinue treatment. Subcutaneous injections of heparin were administered for five months, until arterial thromboembolisms were considered controlled without recurrence. Considering side effects such as induration at the injection site and the complexity of the self-injection, subcutaneous heparin injections were changed to a DOAC, apixaban (5 mg orally twice a day) in July 2021. In an echocardiography performed in January 2022, the vegetation at the aortic valve had disappeared. The patient continued to receive apixaban at the same dose and remain in good condition with no thromboembolism or recurrence of cancer until November 2023, more than three years after the diagnosis of lung adenocarcinoma. The clinical course is shown in Fig. 6. The patient has visited the hospital once every two months so far, and will continue to visit the hospital once every two months for follow-up observation.
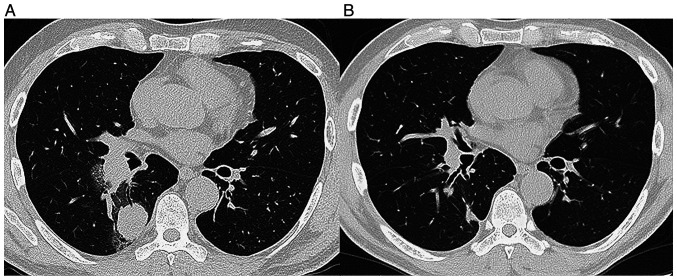
Figure 1.
(A) A chest CT scan taken in August 2020 showed a nodule in the right lung and ipsilateral hilar lymphadenopathy. (B) A chest CT scan taken in January 2021 after the diagnosis of lung adenocarcinoma showed a shrinkage of the primary lesion and ipsilateral hilar lymphadenopathy.
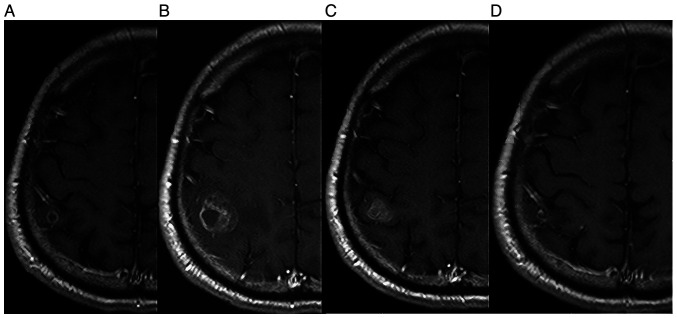
Figure 2.
(A) A brain MRI taken in August 2020 revealed a metastatic lesion in the right occipital lobe of the cerebrum. (B) A brain MRI taken in October 2020 showed an increase of the occipital lobe lesion. (C) On MRI taken in November 2020, shrinkage of the lesion was observed. (D) On MRI taken in January 2021, further shrinkage of the lesion was observed (D).
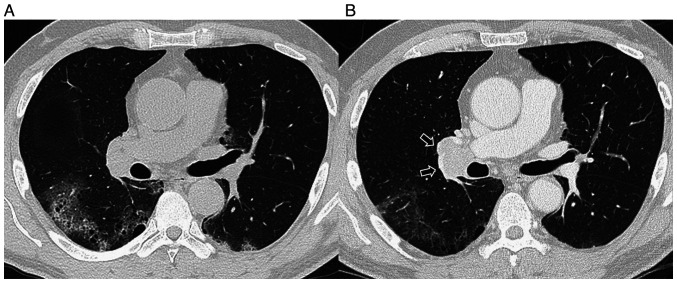
Figure 3.
(A) The appearance of ground glass opacity in both lungs in CT scan taken at December 2020. (B) The ground glass opacity in both lungs improved, but recurrence of ipsilateral lymph node lesion (arrows) in CT scan taken at February 2021.
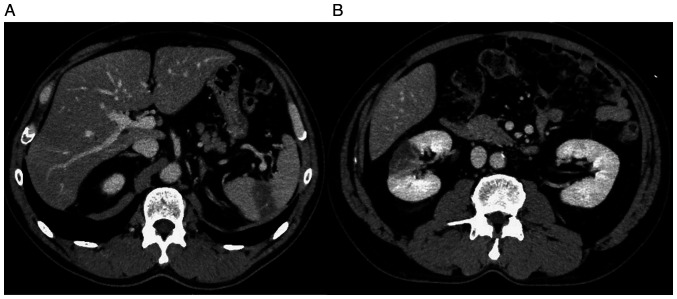
Figure 4.
An abdominal CT scan taken in February 2021, after stopping pembrolizumab showed arterial thromboembolisms in (A) spleen and (B) right kidney.
Figure 5.
Echocardiography performed in February 2021 confirmed a 3 mm-sized vegetation attached to the aortic valve (arrow). (A) Parasternal long axis view, (B) parasternal short axis view.
Figure 6.
Clinical course of the patient. PEM, pemetrexed; BEV, bevacizumab; GGO, ground glass opacity; CR, complete response; FDG-PET, 18F-Fluorodeoxyglucose positron emission tomography.
Discussion
The patient's case was characterized by several noteworthy features: i) Pseudo-progression of a brain tumor, ii) repeated thromboembolism in multiple organs and a small vegetation attached to the aortic valve, iii) long-term control of thromboembolism with apixaban after heparin therapy, iv) good response to lung cancer treatment and recurrence-free survival 36 months after diagnosis.
Cerebral pseudo-progression is rare (13-15). To date, only single case reports or series of cases of cerebral pseudo-progression have been published, and the incidence of cerebral pseudo-progression after ICI treatment has not been determined. Urban et al reported that five of twelve patients with cerebral pseudo-progression had non-small cell lung cancer (15). In this patient, ICI treatment resulted in shrinkage of one of two metastatic lesions and enlargement of the other. Careful follow-up without anticancer treatment or steroid treatment confirmed disappearance of metastatic lesions after several months. Craniotomy and tissue collection were not performed, but pseudo-progression was diagnosed.
In patients with lung cancer, as with other carcinomas, the development of thromboembolism in the brain and extracerebral organs is common (1). Since cancer-bearing patients often have abnormalities of the fibrinolytic coagulation system (1), the thromboembolisms were presumed to be related to the abnormalities. Hypercoagulability is triggered by the production of mucins by cancer cells, which react with leukocytes and platelet selectins to form platelet-rich microthrombi (1). In general, patients with CAT have a poor prognosis because CAT are often associated with advanced cancer, complicating treatment of the underlying disease (16). Moreover, nonbacterial thrombotic endocarditis with vegetations attached to the mitral or the aortic valve has been described in cancer patients (2-4). Our patient had no further risk factors for developing thromboembolism such as diabetes or arrhythmia. He had had no thromboembolism before the start of ICI treatment, and early echocardiography performed at the time of cerebral thromboembolism revealed no vegetation in the aortic valve. Therefore, it was suggested that the later vegetation was caused by CAT or was related to ICI treatment, but the exact cause was not determined.
Treatment of thromboembolism in cancer-bearing patients requires both anticoagulant therapy and treatment for the underlying malignant disease. At present, low molecular weight heparin has a lower risk of causing bleeding than unfractionated heparin, and low molecular weight heparin appears to be more effective for venous thromboembolism in cancer patients (10-12). In contrast, warfarin is not sufficiently effective against thromboembolism in cancer patients (10-12). Because low molecular weight heparin is not covered by health insurance in our country, our patient's condition was controlled by continuous intravenous infusion of unfractionated heparin, followed by self-subcutaneous injection of unfractionated heparin and later apixaban. There was no recurrence of CAT during the long-term follow-up with these treatments or after discontinuation of the treatments. In recent years, guidelines and reviews for thromboembolism in cancer-bearing patients have evaluated the administration of DOACs for thromboembolism in cancer-bearing patients, and their superiority and inferiority to conventional treatments have been discussed (10-12). Advantages of DOACs include i) no need to adjust dosage, ii) no need to draw blood to assess efficacy, iii) rapid onset of action, and iv) fewer drug interactions, and v) no need to adjust the patient's diet during treatment. In contrast, drawbacks have been pointed out: i) Administration is contraindicated in patients with severe renal impairment, ii) there is no direct Xa inhibitor reversal agent, and iii) DOACs are relatively expensive. Among DOACs, rivaroxaban and edoxaban have been considered effective, but the administration of apixaban has been shown to have a lower bleeding risk (17). Tachihara et al described two patients who developed VTE two to three years after diagnosis of lung cancer (18). One of them had asymptomatic lower extremity deep vein thrombosis, the other patient had pulmonary embolism and lower extremity deep vein thrombosis. However, neither patient experienced arterial thromboembolisms (18). Both received heparin injections and one patient received EGFR-tyrosine kinase inhibitors with long-term response (18). Moreover, Nonagase et al presented an EGFR-mutated patient with lower extremity deep vein thrombosis, who was treated with intravenous unfractionated heparin and survived for ten months after lung cancer diagnosis (19). According to a case report by Soga et al, a gastric cancer patient developed VTE recurrence and nonbacterial thrombotic endocarditis despite administration of apixaban for VTE (4). That patient underwent valve replacement, gastric cancer resection, and then subcutaneous heparin self-injection, and survived for 18 months (4). Although such cases have been reported, to the best of our knowledge, ours is the first report of a patient with multiple arterial thromboembolisms and without cancer recurrence whose condition was controlled with DOAC for more than three years.
Although good results were achieved in this patient, caution should be exercised regarding the indications for administering antineoplastic agents to cancer-bearing patients with thromboembolism. Optimal therapies and timing of treatment as well as the optimal administration of DOAC in these patients remain to be established. Whether CAT or irAE, thromboembolism in patients with non-small cell lung cancer could be controlled with an effective immune checkpoint inhibitor-combined chemotherapy in addition to appropriate anticoagulant therapy, and long-term survival of these patients could be achieved as in our patient. Although uncommon, the clinical course of this patient was considered to be suggestive for the treatment of patients with a similar course.
In NSCLC, personalized medicine is making remarkable progress with the discovery of new pathological and serum biomarkers and the clinical introduction of drugs that are compatible with these biomarkers (20,21). However, biomarkers related to paraneoplastic syndromes including CAT are still in the development stage. Progress in translational research in this area is strongly desired.
Acknowledgements
Not applicable.
Funding Statement
Funding: No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the present study are available from the corresponding author on reasonable request.
Authors' contributions
TK, YS, HS and NH designed the study. TK, YS, GO, SO, SH, MT, EO and HS collected the data. TK, YS and HS analyzed the data. TK, YS,HS and NH prepared the manuscript. HS and NH supervised the study. HS and NH confirm the authenticity of all the raw data. All authors read and approved the final manuscript.
Ethics approval and consent to participate
The present study was approved by the Institutional Review Board of the Mito Medical Center, University of Tsukuba-Mito Kyodo General Hospital (approval no. 16-39).
Patient consent for publication
Written informed consent was obtained from the patient for the publication of their data.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Akinbo DB, Ajayi OI. Thrombotic pathogenesis and laboratory diagnosis in cancer patients, an update. Int J Gen Med. 2023;16:259–272. doi: 10.2147/IJGM.S385772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Benedetti M, Morroni S, Fiaschini P, Coiro S, Savino K. Nonbacterial thrombotic endocarditis with multiple systemic emboli in a patient with primary lung cancer. J Cardiovasc Echogr. 2022;32:129–131. doi: 10.4103/jcecho.jcecho_68_21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Scalia GM, Tandon AK, Robertson JA. Stroke, aortic vegetations and disseminated adenocarcinoma-a case of marantic endocarditis. Heart Lung Circ. 2012;21:234–236. doi: 10.1016/j.hlc.2011.08.003. [DOI] [PubMed] [Google Scholar]
- 4.Soga Y, Taira K, Sugimoto A, Kurosawa M, Kira H, Su T, Doi K, Nakano A, Himura Y. Mitral valve nonbacterial thrombotic endocarditis: A rare multi-surgery-tolerant survivor of Trousseau's syndrome. Surg Case Rep. 2018;4(104) doi: 10.1186/s40792-018-0513-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Moik F, Ay C. Venous and arterial thromboembolism in patients with cancer treated with targeted anti-cancer therapies. Thromb Res. 2022;213 (Suppl 1):S58–S65. doi: 10.1016/j.thromres.2022.01.004. [DOI] [PubMed] [Google Scholar]
- 6.Cheng Y, Ouyang W, Jie C, Zhang J, Yu J, Xie C. A case of late and lethal Trousseau's syndrome induced by pembrolizumab in lung adenocarcinoma. Immunotherapy. 2023;15:71–75. doi: 10.2217/imt-2022-0018. [DOI] [PubMed] [Google Scholar]
- 7.Zhao L, Zheng H, Zhu L, Jiang P. Trousseau's syndrome in a non-small cell lung cancer patient aggravated by pembrolizumab. J Oncol Pharm Pract. 2023;29:233–238. doi: 10.1177/10781552221102311. [DOI] [PubMed] [Google Scholar]
- 8.Shionoya Y, Hirohashi Y, Takahashi H, Hashimoto M, Nishiyama K, Takakuwa Y, Nakatsugawa M, Kubo T, Kanaseki T, Tsukahara T, Torigoe T. Possible pseudo-progression of non-small cell lung carcinoma in a patient with clinical hyper-progression associated with trousseau syndrome who was treated with pembrolizumab: A case report. Anticancer Res. 2021;41:3699–3706. doi: 10.21873/anticanres.15161. [DOI] [PubMed] [Google Scholar]
- 9.Horio Y, Takamatsu K, Tamanoi D, Sato R, Saruwatari K, Ikeda T, Nakane S, Nakajima M, Saeki S, Ichiyasu H, et al. Trousseau's syndrome triggered by an immune checkpoint blockade in a non-small cell lung cancer patient. Eur J Immunol. 2018;48:1764–1767. doi: 10.1002/eji.201847645. [DOI] [PubMed] [Google Scholar]
- 10.Ogata M, Ama Y, Ogata T, Hirabatake M, Yasui H, Satake H. Direct oral anticoagulants for the treatment of venous thromboembolism in patients with active cancer. In Vivo. 2021;35:2747–2753. doi: 10.21873/invivo.12559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Samaranayake CB, Anderson J, McCabe C, Zahir SF, Upham JW, Keir G. Direct oral anticoagulants for cancer-associated venous thromboembolisms: A systematic review and network meta-analysis. Intern Med J. 2022;52:272–281. doi: 10.1111/imj.15049. [DOI] [PubMed] [Google Scholar]
- 12.Frere C, Farge D, Schrag D, Prata PH, Connors JM. Direct oral anticoagulant versus low molecular weight heparin for the treatment of cancer-associated venous thromboembolism: 2022 updated systematic review and meta-analysis of randomized controlled trials. J Hematol Oncol. 2022;15(69) doi: 10.1186/s13045-022-01289-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Cohen JV, Alomari AK, Vortmeyer AO, Jilaveanu LB, Goldberg SB, Mahajan A, Chiang VL, Kluger HM. Melanoma brain metastasis pseudoprogression after pembrolizumab treatment. Cancer Immunol Res. 2016;4:179–182. doi: 10.1158/2326-6066.CIR-15-0160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Vander Jagt TA, Davis LE, Thakur MD, Franz C, Pollock JM. Pseudoprogression of CNS metastatic disease of alveolar soft part sarcoma during anti-PDL1 treatment. Radiol Case Rep. 2018;13:882–885. doi: 10.1016/j.radcr.2018.05.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Urban H, Steidl E, Hattingen E, Filipski K, Meissner M, Sebastian M, Koch A, Strzelczyk A, Forster MT, Baumgarten P, et al. Immune Checkpoint inhibitor-induced cerebral pseudoprogression: patterns and categorization. Front Immunol. 2022;12(798811) doi: 10.3389/fimmu.2021.798811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Tanimura J, Yamamoto T, Hashimoto T. Clinical features and prognostic factors in patients with cancer-associated multiple ischemic stroke: A retrospective observational study. J Stroke Cerebrovasc Dis. 2022;31(106813) doi: 10.1016/j.jstrokecerebrovasdis.2022.106813. [DOI] [PubMed] [Google Scholar]
- 17.Riaz IB, Fuentes HE, Naqvi SAA, He H, Sipra QR, Tafur AJ, Padranos L, Wysokinski WE, Marshall AL, Vandvik PO, et al. Direct oral anticoagulants compared with dalteparin for treatment of cancer-associated thrombosis: A living, interactive systematic review and network meta-analysis. Mayo Clin Proc. 2022;97:308–324. doi: 10.1016/j.mayocp.2020.10.041. [DOI] [PubMed] [Google Scholar]
- 18.Tachihara M, Nikaido T, Wang X, Sato Y, Ishii T, Saito K, Sekine S, Tanino Y, Ishida T, Munakata M. Four cases of Trousseau's syndrome associated with lung adenocarcinoma. Intern Med. 2012;51:1099–1102. doi: 10.2169/internalmedicine.51.6453. [DOI] [PubMed] [Google Scholar]
- 19.Nonagase Y, Takeda M, Tanaka K, Hayashi H, Iwasa T, Nakagawa K. Treatment of EGFR mutation-positive non-small cell lung cancer complicated by Trousseau syndrome with gefitinib followed by osimertinib: A case report. Oncotarget. 2018;9:29532–29535. doi: 10.18632/oncotarget.25687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ortega MA, Navarro F, Pekarek L, Fraile-Martínez O, García-Montero C, Saez MA, Arroyo M, Monserrat J, Alvarez-Mon M. Exploring histopathological and serum biomarkers in lung adenocarcinoma: Clinical applications and translational opportunities (review) Int J Oncol. 2022;61(154) doi: 10.3892/ijo.2022.5444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ortega MA, Pekarek L, Navarro F, Fraile-Martínez O, García-Montero C, Álvarez-Mon MÁ, Diez-Pedrero R, Boyano-Adánez MDC, Guijarro LG, Barrena-Blázquez S, et al. Updated views in targeted therapy in the patient with non-small cell lung cancer. J Pers Med. 2023;13(167) doi: 10.3390/jpm13020167. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analyzed during the present study are available from the corresponding author on reasonable request.