Abstract
We developed an efficient electrotransformation system for the pathogenic fungus Histoplasma capsulatum and used it to examine the effects of features of the transforming DNA on transformation efficiency and fate of the transforming DNA and to demonstrate fungal expression of two recombinant Escherichia coli genes, hph and lacZ. Linearized DNA and plasmids containing Histoplasma telomeric sequences showed the greatest transformation efficiencies, while the plasmid vector had no significant effect, nor did the derivation of the selectable URA5 marker (native Histoplasma gene or a heterologous Podospora anserina gene). Electrotransformation resulted in more frequent multimerization, other modification, or possibly chromosomal integration of transforming telomeric plasmids when saturating amounts of DNA were used, but this effect was not observed with smaller amounts of transforming DNA. We developed another selection system using a hygromycin B resistance marker from plasmid pAN7-1, consisting of the E. coli hph gene flanked by Aspergillus nidulans promoter and terminator sequences. Much of the heterologous fungal sequences could be removed without compromising function in H. capsulatum, allowing construction of a substantially smaller effective marker fragment. Transformation efficiency increased when nonselective conditions were maintained for a time after electrotransformation before selection with the protein synthesis inhibitor hygromycin B was imposed. Finally, we constructed a readily detectable and quantifiable reporter gene by fusing Histoplasma URA5 with E. coli lacZ, resulting in expression of functional β-galactosidase in H. capsulatum. Demonstration of expression of bacterial genes as effective selectable markers and reporters, together with a highly efficient electrotransformation system, provide valuable approaches for molecular genetic analysis and manipulation of H. capsulatum, which have proven useful for examination of targeted gene disruption, regulated gene expression, and potential virulence determinants in this fungus.
Histoplasma capsulatum is a dimorphic pathogenic fungus that has worldwide distribution and is a major cause of respiratory and systemic mycosis in healthy and especially immunocompromised people. Histoplasmosis is the most common cause of fungal respiratory infection in the world and of systemic mycosis in the United States, where the Mississippi and Ohio River valleys are the major areas of endemicity. The organism has two morphotypes, growing as a saprophytic mold in the soil and at room temperature in the laboratory (infectious form) and as a yeast in the mammalian host and at 37°C in the laboratory (pathogenic form). After inhalation of conidia or mycelial fragments by mammals, the organisms enter pulmonary macrophages and convert to yeasts, which survive and proliferate within phagolysosomes. Infection with H. capsulatum is extremely common, but severe disease (e.g., disseminated, fulminant, life threatening) is much less common, because of limitation of the infection by a T-lymphocyte-mediated immune response in immunocompetent hosts. Even in these cases, the organism may not be eradicated, and lifelong latent infection may be reactivated later, e.g., during periods of immunocompromise that occur in patients with AIDS or cancer or people receiving immunosuppressive therapy (8, 9, 22).
Molecular genetic experimental approaches have recently been applied to H. capsulatum, as to a range of other fungi pathogenic for humans, animals, and/or plants (4, 6, 12, 13, 15, 19, 21). In earlier work, we used the Podospora anserina URA5 gene (PaURA5) and lithium acetate-polyethylene glycol-mediated DNA delivery to transform uracil-auxotrophic ura5 mutants of H. capsulatum and to characterize the fate of the transforming DNA (24, 25, 30). Transformants, which occur at a relatively low efficiency with the chemical transformation system, display the transforming DNA either on de novo-generated modified linear plasmids or integrated apparently randomly into chromosomal DNA; both processes frequently involve multimerization and may involve rearrangement or other modification of the transforming DNA (24, 30). One invariant modification in the case of linear plasmid formation is the appearance of H. capsulatum telomeric sequences at the termini of the molecules, which are present in multiple copies (24). We exploited this phenomenon to rescue the telomeric sequences and to generate autonomously replicating Escherichia coli - H. capsulatum shuttle plasmids that can be propagated in circular form in E. coli and unmodified linear form in H. capsulatum (25).
For this study, we developed a highly efficient electrotransformation system for H. capsulatum, initially using a telomeric plasmid containing the H. capsulatum URA5 gene (HcURA5) as a selectable marker (27). After optimizing electrotransformation parameters with this plasmid, we examined the effects of various characteristics of the transforming DNA on electrotransformation efficiency and on the fate of the transforming DNA. Finally, we engineered expression in H. capsulatum of two E. coli genes, hph and lacZ, for use as selectable and detectable/quantifiable markers, respectively. In the former case, we adapted a hygromycin B resistance marker used in other pathogenic fungi (13, 15, 16, 21) for transformation of H. capsulatum, providing a second selectable marker system and enabling transformation of potentially any strain, relieving the previous restriction to ura5 mutant strains. In the latter case, we constructed an in-frame fusion gene between lacZ and the 5′ end of the H. capsulatum URA5 gene in order to provide appropriate fungal expression sequences and showed production of enzymatically active β-galactosidase. These results represent, to our knowledge, the first demonstration of expression of bacterial genes in H. capsulatum. Additionally, these methods and reagents have provided the basis for preliminary work in targeted gene disruption (26, 27) and examination of gene expression (14, 17) in H. capsulatum.
MATERIALS AND METHODS
Fungal strains.
H. capsulatum G184AS, G184ASura5-11, G184A-HTE, G186AS, G217B, and G217Bura5-21 have been described previously (10, 24, 29). The parental strains G184A and G186A are clinical isolates (ATCC 26027 and ATCC 26029, respectively) of restriction fragment length polymorphism (RFLP) class 3. G217B is a clinical isolate (ATCC 26032) of RFLP class 2. G184ASura5-11 and G217Bura5-21 were isolated after UV mutagenesis and selection using 5-fluoroorotic acid (24, 29). Unless otherwise indicated, experiments were done with H. capsulatum grown as yeast cells at 37°C; exceptions in which the fungus was grown as mycelia at room temperature are noted.
Bacterial strains.
Plasmids were propagated in E. coli HB101 (supE44 hsdS20 recA13 ara-14 proA2 lacY1 galK2 rpsL20 xyl-5 mtl-1), SURE (e14− [mcrA]Δ[mcrCB-hsdSMR-mrr]171 endA1 supE44 thi-1 gyrA96 relA1 lac recB recJ sbsC umuC::Tn5 uvrC [F′ proAB lacIqZΔM15Tn10]) (obtained from Stratagene Cloning Systems, La Jolla, Calif.), and a tetracycline-sensitive variant of SURE (lacking Tn10 on the F′ plasmid, used when selection for tetracycline resistance was necessary; kindly provided by Alan Greener, Stratagene). Circular plasmids containing telomeric sequences are stable in these strains and also in the recA+ derivative of HB101, RR1, but we have noted instability of telomeric plasmids in strain DH5 (supE44 hsdR17 recA1 endA1 gyrA96 thi-1 relA1); this instability is manifested by variable deletion of plasmid sequences involving areas separate from the telomeric regions (23).
Media.
H. capsulatum was grown in broth in HMM, supplemented with uracil (100 μg/ml) for nonselective growth of ura5 strains, as previously described (24). HMM consists of F-12 nutrient mixture with l-glutamine, with phenol red, without sodium bicarbonate (Gibco BRL, Gaithersburg, Md.), supplemented with (per liter) 18.2 g of glucose, 1.0 g of glutamic acid, 84 mg of cystine, and 5.96 g of HEPES, adjusted to pH 7.5 (28). Solid HMM medium also contained 0.5% (wt/vol) agarose (SeaKem LE agarose, lot no. 634592; FMC BioProducts, Rockland, Maine) and was supplemented with 10 μM FeSO4 in addition to the 3 μM FeSO4 included in HMM. In some experiments focused on antibiotic resistance, hygromycin B (Boehringer Mannheim Biochemicals, Indianapolis, Ind.) was added to solid or liquid HMM at a concentation of 50, 100, 200, or 500 μg/ml. In some experiments focused on β-galactosidase expression, the colorimetric indicator X-Gal (5-bromo-4-chloro-3-indolyl-β-d-galactopyranoside) was incorporated in the HMM-agarose at a concentration of 10, 20, 40, or 100 μg/ml in the solid medium, or the X-Gal solution was directly added to existing colonies on solid medium. Penicillin-streptomycin was added to HMM broth, and gentamicin was added to HMM-agarose. E. coli was grown in LB broth (3, 18) or on LB–1.5% agarose solid medium, supplemented with appropriate antibiotics.
Chemicals and molecular biology reagents and methods.
Unless otherwise indicated, chemicals were obtained from Sigma Chemical Company (St. Louis, Mo.). Restriction endonucleases, T4 DNA ligase, and polynucleotide kinase were obtained from Boehringer Mannheim Biochemicals or New England BioLabs (Beverly, Mass.) and used according to the manufacturer’s recommendations. Oligodeoxyribonucleotides were synthesized at the Washington University Protein Chemistry Laboratory or were obtained from Gibco BRL, and their sequences and uses are described in the text. Nucleotide sequencing was performed by the dideoxy-chain termination protocol (3, 18). Methods for DNA electrophoresis, staining, band purification from agarose gels, and Southern blotting have been described previously (24); radiolabeling of DNA probes was performed by random priming.
DNA preparation.
Plasmids were prepared from E. coli by using an alkaline lysis miniprep protocol (31) or Z523 columns (5 Prime→3 Prime, Boulder, Colo.) according to the manufacturer’s recommendations. The procedure for preparing genomic Histoplasma DNA by using enzymatic spheroplasting, alkaline-sodium dodecyl sulfate lysis, RNase A and proteinase K treatments, and organic extractions has been described previously (24).
Construction of URA5 plasmids.
We previously described plasmids containing PaURA5 as a selectable marker: pWU1 (24), containing PaURA5 on a 1.6-kb EcoRI fragment in pBR328, and pWU44 (25), containing adjacent inverted Histoplasma telomeric sequences added to pWU1. Restriction endonuclease digestion of pWU44 with HpaI yields a linear molecule with appropriately oriented telomeric termini, which transforms H. capsulatum with increased efficiency and is generally maintained in multiple copies without modification other than extension of the telomeric sequences (25). Plasmid pWU3 consists of a 1.5-kb EcoRI-SspI subcloned fragment containing the PaURA5 gene cloned into pBluescript SK+ digested with EcoRI and EcoRV.
The HcURA5 gene was cloned by using a degenerate PCR approach based on short peptide sequences relatively conserved among URA5 gene products from other organisms; a fragment amplified from H. capsulatum G184AS genomic DNA was used as a probe to isolate the full-length gene from the same strain. The open reading frame for this gene is about 880 bp in length and includes two introns, each about 70 bp in length; the minimal functional fragment for complementation in Histoplasma ura5 mutants is about 1.05 kb in length. Details of the cloning and characterization of this gene and its encoded product are presented elsewhere (26, 27). Following are descriptions of plasmids used to evaluate the effect on electrotransformation of the native Histoplasma gene in comparison to the heterologous Podospora gene encoding the same enzymatic function, as well as effects of other variables.
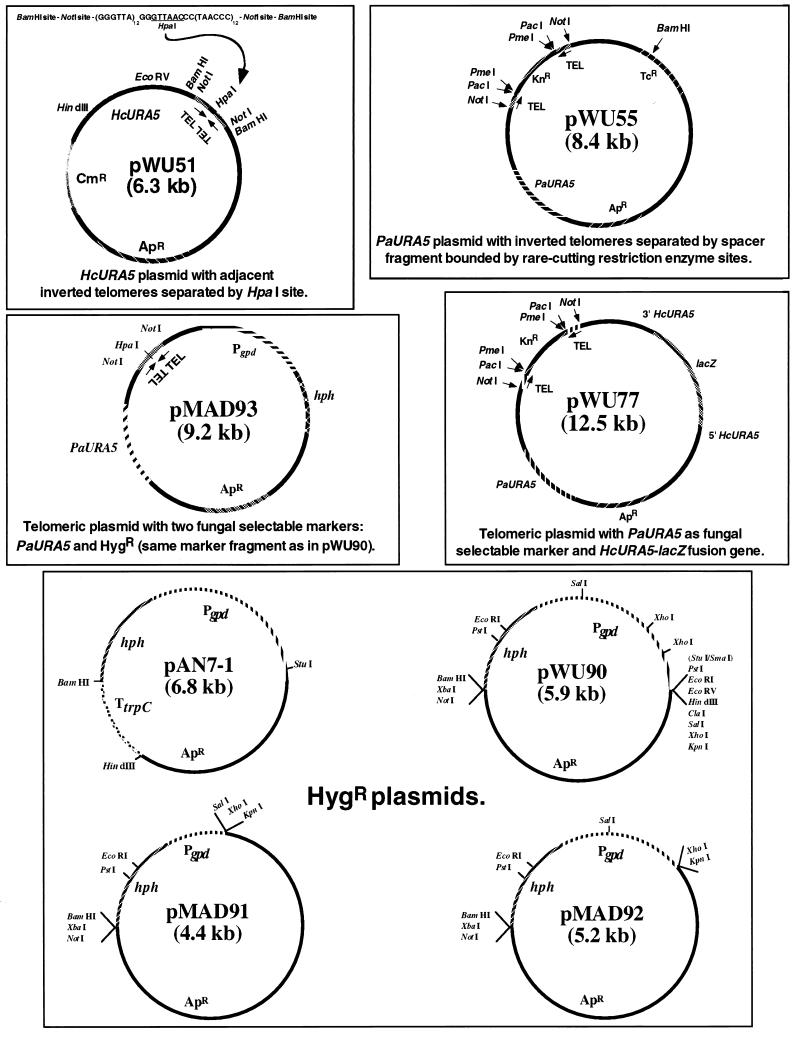
Plasmid pWU20 consists of a 2.8-kb HindIII/SalI subcloned fragment containing the HcURA5 gene cloned into the large fragment of pBR328 after digestion with HindIII and SalI. Plasmid pWU22 consists of a 1.05-kb BglII/EcoRV subcloned fragment containing the HcURA5 gene (the minimal functional fragment for complementation in Histoplasma ura5 mutants) cloned into the large fragment of pBR328 after digestion with BamHI and EcoRV. To construct a telomeric shuttle plasmid containing the HcURA5 gene, first the PaURA5 gene EcoRI fragment was removed from pWU44, resulting in plasmid pWU50, which is pBR328 containing the adjacent inverted Histoplasma telomeric sequences inserted at the BamHI site. Then, a 1.3-kb HindIII/EcoRV subcloned fragment containing the HcURA5 gene was cloned into the large fragment of pWU50 after digestion with HindIII and EcoRV to generate plasmid pWU51, which yields a linear molecule with appropriately oriented telomeric termini upon digestion with HpaI (Fig. 1).
FIG. 1.
Diagrams of selected plasmids used in this study. When a restriction enzyme is designated on a plasmid map, then all of the sites for that enzyme on that plasmid are indicated. “TEL” indicates an H. capsulatum telomeric sequence comprised of repeats of the hexamer GGGTTA. Other plasmid elements are described in the text.
Construction of hygromycin B resistance plasmids.
Plasmid pAN7-1 (16) is a pUC18 derivative containing the E. coli hygromycin phosphotransferase gene hph (∼1.1 kb) flanked by Aspergillus nidulans regulatory sequences: the glyceraldehyde phosphate dehydrogenase promoter region Pgpd (∼2.2 kb) and the trpC terminator region TtrpC (∼0.8 kb) (Fig. 1). This plasmid was kindly provided by Paul Szaniszlo and Ming Peng, University of Texas. To construct plasmid pWU90 (Fig. 1), we cloned from pAN7-1 a 2.9-kb StuI/BamHI fragment containing the hph gene and most of the Pgpd promoter region, but lacking the TtrpC terminator region, into pBluescript SK+ digested with SmaI and BamHI. To construct plasmid pMAD91 (Fig. 1), pWU90 was digested with SalI and the large fragment was self-ligated to circularize, resulting in removal of approximately 1.5 kb upstream-distal DNA from the Aspergillus promoter region, leaving a 1.4-kb marker fragment. To construct plasmid pMAD92 (Fig. 1), pWU90 was digested with XhoI and the large fragment was self-ligated to circularize, resulting in removal of approximately 0.6 kb upstream-distal DNA from the Aspergillus promoter region, leaving a 2.3-kb marker fragment. To construct a telomeric shuttle plasmid containing two selectable markers for H. capsulatum (PaURA5 and hph), we first cloned the 0.2-kb BamHI fragment containing adjacent inverted Histoplasma telomeric sequences from pWU44 into the BclI site of plasmid pWU1 to generate plasmid pWU45, which is the same as pWU44 except for the moving of the telomeric region from the BamHI site to the BclI site, restoring the tetracycline resistance gene originally in pBR328. Then we cloned from pWU90 a 2.9-kb ClaI/BamHI fragment containing the entire marker fragment plus part of the pBluescript SK+ polylinker into the large fragment of pWU45 after digestion with ClaI and BamHI to generate plasmid pMAD93, which yields a linear molecule with appropriately oriented telomeric termini upon digestion with HpaI (Fig. 1).
Construction of plasmids with separated telomeres.
The HpaI site separating the telomeres and used to generate linear transforming molecules with telomeric termini from plasmids such as pWU44, pWU45, and pWU51 is a 6-bp sequence that is inconvenient for a number of applications, such as when a HpaI site is present elsewhere on the plasmid (see below), or may be present in randomly cloned genomic fragments, in which it is statistically predicted to occur approximately every 4 kb. Additionally, we observed that HpaI digestion of telomeric plasmids such as pWU44, pWU45, and pWU51 was sometimes incomplete (23), which we hypothesize is due to the physical nature of the DNA in such an extended palindromic and telomeric region (5, 7). Therefore, we used the following approach to generate plasmids with telomeric repeat sequences in the appropriate orientation but separated by a spacer region to interrupt the long palindrome, and which could be exposed on the termini of linear molecules by different, rare-cutting restriction enzymes. Plasmid pWU51 was digested with HpaI, leaving blunt termini. A synthetic oligodeoxynucleotide with the palindromic sequence 5′ AATTAAGGGTTTAAACGAGCTCGTTTAAACCCTTAATT 3′ was phosphorylated and allowed to self-anneal, resulting in a double-stranded fragment containing a central SacI site, flanked by PmeI sites, flanked by PacI sites, with blunt termini, followed by digestion with SacI to bisect this fragment and generate blunt end-PacI site-PmeI site-SacI cohesive end linkers. Finally, a 1.6-kb fragment with SacI termini containing a kanamycin resistance gene was isolated from pUC4-KIXX (Pharmacia LKB Biotechnology, Piscataway, N.J.). HpaI-digested pWU51 (with blunt ends), the small blunt-SacI linker fragments, and the kanamycin resistance fragment with SacI termini were included in a quadrimolecular ligation reaction that resulted in a plasmid designated pWU54, carrying the kanamycin resistance marker, flanked by PmeI sites, flanked by PacI sites, flanked by inverted series of telomeric repeats, flanked by NotI sites, flanked by BamHI sites; the presence of the outermost restriction sites made this cassette easily movable into plasmids already carrying adjacent inverted telomeric repeats flanked by NotI and/or BamHI sites, or any plasmid with such sites or compatible sites. For instance, plasmid pWU55 (Fig. 1) was constructed by cloning the 1.9-kb BamHI fragment from pWU54 comprising the telomeric and kanamycin resistance cassette into the BclI site of pWU1. Digestion of pWU54 or pWU55 with either PacI or PmeI, which both have rare 8-bp TA-rich recognition sequences, removes the central spacer fragment and leaves a linear molecule with telomere-like termini. The presence of the spacer fragment was selectable by using kanamycin, and physical separation of the telomeric sequences with the spacer fragment was associated with complete digestion of the appropriate telomeric plasmids, such as pWU54 and pWU55, using PacI or PmeI (data not shown).
Construction of an HcURA5-lacZ fusion gene.
To develop a reporter system marker the expression of which would be readily detectable in colonies and quantifiable in cell lysates, we constructed an in-frame coding sequence fusion between a lacZ gene lacking its promoter, start codon, and distal N-terminus-coding region, and the distal N-terminus-coding region of the HcURA5 gene, including also sequence upstream of the start codon for expression in H. capsulatum (27). We used a 3.1-kb PstI fragment from plasmid pMC1871 (Pharmacia LKB Biotechnology) as the source of the 5′-truncated lacZ gene. This fragment was ligated with the nonphosphorylated oligodeoxynucleotide 5′ TCGATGCA 3′, in order to adapt the PstI overhang to an XhoI overhang. This fragment with XhoI-compatible termini was then phosphorylated with polynucleotide kinase and ligated into the unique XhoI site in a plasmid carrying a 1.2-kb BglII/AvrII fragment containing the HcURA5 gene including about 0.1 kb of upstream sequence that is sufficient for expression in H. capsulatum; this XhoI site occurs near the start codon of the HcURA5 gene, before the occurrence of any intron (27). Restriction endonuclease mapping of minipreps allowed selection of a plasmid carrying the insert in the appropriate orientation, and nucleotide sequencing was performed over the artificially manipulated region to confirm in-frame fusion of the HcURA5 upstream sequence and lacZ downstream sequence. The encoded product of the fusion gene includes 19 amino acids from the N terminus of the HcURA5 gene product, 1 amino acid resulting from the adapter linker design, and 12 amino acids from pMC1871 sequence, before the start of β-galactosidase amino acid sequence. The lacZ gene contains an HpaI site, and in order to place the fusion gene on a telomeric plasmid that can be digested to expose telomeres without using HpaI and that contains another marker for independent selection after transformation of H. capsulatum, we selected as a base plasmid pWU55, containing the intact PaURA5 gene and the cassette with telomeres separated by the kanamycin resistance marker and digestible with PacI or PmeI. The final construct, plasmid pWU77 (Fig. 1), includes the fusion gene inserted between the BamHI and NheI sites of pWU55, originally by using the compatible BglII and AvrII sites, respectively, at the termini of the HcURA5 gene fragment used to make the fusion gene.
Transformation of H. capsulatum.
Chemical transformation using treatment with lithium acetate and polyethylene glycol has been described previously (24, 30). In this study, we developed and optimized parameters for H. capsulatum electrotransformation. The following protocol is described for a single electroporation sample. Five milliliters of a log-phase broth culture of yeast cells was used; typically, a 3-day broth culture was diluted 1:50 in HMM and grown for 42 h. The culture was observed microscopically for typical appearance of dispersed budding yeasts and the absence of mycelia. The culture was centrifuged at 300 × g for 5 min at room temperature, and the pellet was washed with 5 ml of warm (37°C) electroporation solution (see Results for details of electroporation solutions), centrifuged under the same conditions, and resuspended in a final volume of 0.2 ml with electroporation solution. Following transfer of the cells to a 0.2-cm electroporation cuvette (Bio-Rad Laboratories, Hercules, Calif.) at room temperature, 1 to 10 μl of a DNA solution or TE (10 mM Tris, 1 mM EDTA [pH 8.0]) or water control was added and mixed by flicking the cuvette. Electroporation (see Results for details of conditions) was performed in a Gene Pulser II with Pulse Controller Plus (Bio-Rad Laboratories). All or a portion of the electroporated cell suspension was immediately plated on solid medium, directly on the agarose or on a Nytran (Schleicher & Schuell, Keene, N.H.) filter disc placed on the agarose surface. For URA5 transformation, plating on selective medium, i.e., HMM-agarose lacking exogenous uracil, was done immediately. In some experiments for hygromycin B selection, the Nytran filter was left on nonselective medium, i.e., lacking antibiotic, for variable times before transfer to medium containing antibiotic. Transformant colonies were typically observable microscopically within a few days, were grossly visible by 2 weeks, and continued to appear for a number of weeks; usually we incubated plates for 3 to 5 weeks. Details of our determination of optimal conditions are given in Results, but the best conditions include the following: 10% (wt/vol) = 549 mM mannitol in water, filter sterilized, as the electroporation solution; and electroporation using a capacitance of 25 μF, resistance of 600 Ω, and set voltage of 0.75 kV.
Transformation of E. coli.
Preparation of H. capsulatum cell lysates for detection and quantification of β-galactosidase activity.
A yeast culture was grown to log or late log phase, and a 1-ml aliquot was placed in a 1.5-ml microcentrifuge tube on ice. The cells were pelleted at 1,300 × g for 10 min at 4°C, washed with 1 ml of ice-cold 0.1 M sodium phosphate (0.082 M Na2HPO4, 0.018 M NaH2PO4 [pH 7.5]), and then resuspended with 0.2 ml of ice-cold 0.1 M sodium phosphate (pH 7.5). Approximately one-third volume of acid-washed 0.5-mm-diameter glass beads was added, followed by vortexing for 5 min or by agitation in a Mini-Beadbeater-8 cell disruption apparatus (Biospec Products, Bartlesville, Okla.) for three 1-min intervals at high speed, separated by cooling on ice for 1 min. The suspension of lysed cells was centrifuged at 15,000 × g for 30 min at 4°C to pellet glass beads, unbroken cells, cell fragments, and cell walls. The supernatant containing cytoplasmic contents was assayed in microtiter plate wells for protein concentration by the Bio-Rad protein assay (in comparison to a bovine serum albumin standard curve) and for β-galactosidase activity, using a standard o-nitrophenyl-β-d-thiogalactopyranoside (ONPG) assay (18), modified for microtiter plates. Comparison of the A420 values was made with a standard curve constructed by using grade VIII E. coli β-galactosidase. The β-galactosidase activity in milliunits/milliliter was normalized to the protein concentration in milligrams/milliliter to yield a value of milliunits of β-galactosidase/milligram of protein.
Image photodocumentation.
Autoradiographs or photographs were scanned with a Microtek, Umax, or Agfa scanner, and figures were constructed by using Adobe Photoshop and Canvas software.
RESULTS
Development and optimization of electrotransformation conditions for strain G184ASura5-11.
We used plasmid pWU51, containing the HcURA5 gene as a selectable marker, linearized with HpaI to expose telomeric termini, as the transforming DNA to develop an electroporation system. When delivered via chemical (lithium acetate and polyethylene glycol) treatment, this construct yielded transformants that generally contained monomeric linear plasmids, as we have reported previously for the PaURA5 marker (25), although occasionally larger plasmids or possibly chromosomal integration occurred (Fig. 2); we did not attempt to distinguish between the two fates of chromosomal integration and very large linear plasmid formation in this study. Using HpaI-linearized pWU51, we performed 51 electrotransformations by the protocol given in Materials and Methods, varying the following parameters over the values indicated (not all combinations of variables were used): amount of DNA, 0.2, 2, or 8 μg; capacitance, 3.0 or 25 μF; resistance, 100, 200, 400, 600, 800, or 1,000 Ω; set voltage, 0.25, 0.30, 0.50, 0.75, 1.00, 1.50, 2.00, and 2.50 kV. For all of these samples, the wash and electroporation solution used was 10% (wt/vol) = 549 mM mannitol. The transformation efficiency in number of transformants per microgram of transforming DNA ranged from <0.125 to 4,500. Consistently the highest efficiencies were obtained with a capacitance of 25 μF, resistance of 600 Ω, and set voltage of 0.75 kV. Time constants using these optimized conditions ranged from 6.5 to 12 ms, with greatest efficiency correlating with time constants of 8 to 12 ms. Using these optimized settings for the electroporation device and 2 μg of HpaI-linearized pWU51 as transforming DNA, we examined the effect of the solution used for washing and suspending yeasts for electroporation, testing water, mannitol at concentrations of 10% (wt/vol) = 549 mM and 270 mM = 4.9% (wt/vol), sorbitol at concentrations of 10% (wt/vol) = 549 mM and 270 mM = 4.9% (wt/vol), sucrose at concentrations of 10% (wt/vol) = 292 mM and 270 mM = 9.2% (wt/vol), and glycerol at a concentration of 270 mM = 2.5% (wt/vol). The water sample yielded only 50 transformants/μg of DNA, whereas all other sample values were >100, with 10% (wt/vol) = 549 mM mannitol (the same solution used for the initial experiments) yielding the best value of 1,185 transformants/μg of DNA.
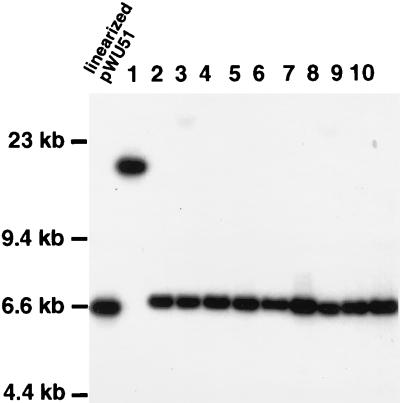
FIG. 2.
Southern blot of undigested genomic DNA from 10 strain G184ASura5-11 transformants (lanes 1 to 10) obtained after chemical transformation using HpaI-linearized pWU51, probed with 32P-labeled vector pBR328 (4.9 kb). The leftmost lane contains linearized pWU51 (6.3 kb). Positions of molecular size markers are indicated to the left. Transformants 2 to 10 carry monomeric linear plasmids. Transformant 1 carries a larger linear plasmid. Transformants 3 and 8 show faint hybridization with the broad smear of chromosomal fragments in undigested H. capsulatum genomic DNA, indicating either very large linear plasmids or the occurrence of chromosomal integration of the transforming DNA at a barely detectable level. We did not attempt to distinguish between the two fates of chromosomal integration and very large linear plasmid formation in this study.
Effect of characteristics of the transforming DNA on electrotransformation efficiency in strain G184ASura5-11, using optimized electroporation conditions.
We tested the following variables (Table 1): plasmid vector (pBR328 or pBluescript), heterologous or native source of the selectable marker (PaURA5 or HcURA5, respectively), circular or linearized physical state, and absence or presence of telomeric sequences; in the case of telomeric plasmids, the linearization was always done to yield molecules with telomeric termini. In these experiments, the following conditions were used: 4 μg of circular plasmid or 1 μg of linearized plasmid as transforming DNA; 10% (wt/vol) = 549 mM mannitol in water, filter sterilized, as the electroporation solution; and electroporation using a capacitance of 25 μF, resistance of 600 Ω, and set voltage of 0.75 kV. We used a slightly larger amount (4 μg) of transforming DNA for circular plasmids because some showed no transformants with 1 μg. In the plasmids used, the amount of DNA upstream or downstream of the coding sequence of the fungal gene varied slightly, but all of the constructs used carry sequences sufficient for expression in H. capsulatum as shown by the ability to transform, and we have not observed differences in transformation efficiency related to the slightly different amounts of flanking sequence present in these different plasmids (Table 1 and reference 23). Comparison of the transformation efficiencies for the two nontelomeric PaURA5 plasmids pWU1 (pBR328 based) and pWU3 (pBluescript based) indicated no significant effect of these two vector backbones. Comparison of the transformation efficiencies for the corresponding forms (circular or linearized, nontelomeric or telomeric) of PaURA5 with HcURA5 plasmids indicated little or no effect of the heterologous or native source of the selectable URA5 marker, with one exception. The circular form, i.e., without exposed telomeres, of the telomeric PaURA5 plasmid pWU44 showed about a 10-fold-greater transformation efficiency than the circular form of the telomeric HcURA5 plasmid pWU51 (on a log scale, 2.3 ± 0.3 versus 0.9 ± 0.2). However, the linearized forms of these plasmids with telomeric termini exposed showed nearly the same transformation efficiency (3.1 ± 0.2 versus 3.0 ± 0.4), and this result, along with other data in Table 1, is consistent with a lack of any difference in functional efficacy of these two genes; we have not pursued this difference further. Two characteristics of the different plasmids showed independent and substantial effects on transformation efficiency: the physical state of the transforming DNA (circular or linearized), and the absence or presence of telomeres. The linearized nontelomeric plasmids pWU3 and pWU22 showed a 30- to 150-fold-greater transformation efficiency than the corresponding circular plasmids, while the linearized, telomere-exposed plasmids pWU44 and pWU51 showed a 6- to 200-fold-greater transformation efficiency than the corresponding circular plasmids. Finally, comparison of the circular or linearized forms of nontelomeric pWU1 and pWU3 with corresponding forms of telomeric pWU44 (all PaURA5 plasmids) and of circular or linearized forms of nontelomeric pWU20 and pWU22 with corresponding forms of telomeric pWU51 (all HcURA5 plasmids) indicated that the presence of telomeres increased transformation efficiency 2- to 100-fold for circular molecules and 6- to 10-fold for linearized molecules.
TABLE 1.
Electrotransformation efficiencies of plasmids in H. capsulatum G184ASura5-11a
| Plasmid | Size (kb) | Vector | Fungal insert | Physical state of transforming DNA | No. of expts | Transformation efficiency (log no./μg of DNA)
|
|
|---|---|---|---|---|---|---|---|
| Mean ± SD | Range | ||||||
| pWU1 | 6.5 | pBR328 | PaURA5 + 0.5 kb upstream and 0.4 kb downstreamb | Circular | 4 | 0.2 ± 0.2 | 0.0–0.3 |
| pWU3 | 4.5 | pBLUESCRIPT | PaURA5 + 0.4 kb upstream and 0.4 kb downstreamb | Circular BamHI linearizedc |
5 4 |
0.0 ± 0.0 2.0 ± 0.2 | 0.0 1.7–2.2 |
| pWU44 | 6.7 | pBR328 | Same as pWU1 | Circulard | 5 | 2.3 ± 0.3 | 2.0–2.7 |
| HpaI linearizede | 7 | 3.1 ± 0.2 | 3.0–3.6 | ||||
| pWU20 | 7.1 | pBR328 | HcURA5 + 0.4 kb upstream and 1.5 kb downstreamb | Circular | 4 | 0.5 ± 0.4 | 0.0–1.0 |
| pWU22 | 5.8 | pBR328 | HcURA5 + 0.1 kb upstream and 0.1 kb downstreamb | Circular EcoRV linearizedf | 2 2 | 0.6 ± 0.1 2.2 ± 0.1 | 0.5–0.6 2.1–2.2 |
| pWU51 | 6.3 | pBR328 | HcURA5 + 0.4 kb upstream and 0.1 kb downstreamb | CirculardHpaI linearizede | 5 8 | 0.9 ± 0.2 3.0 ± 0.4 | 0.7–1.1 2.6–3.6 |
Electroporation conditions: 4 μg of circular plasmid or 1 μg of linearized plasmid as transforming DNA; 10% (wt/vol) mannitol in water as the electroporation solution; and electroporation with a capacitance of 25 μF, resistance of 600 Ω, and set voltage of 0.75 kV. We used a slightly larger amount (4 μg) of transforming DNA for circular plasmids because some showed no transformants with 1 μg.
Sequence flanking start and stop codons.
Near downstream insert/vector junction.
Having adjacent, inverted telomeric repeat sequences.
Having telomeric termini.
At downstream insert/vector junction.
Comparison between chemical and electrotransformation in efficiency and fate of the transforming DNA in strain G184AS ura5-11.
For direct comparison of the efficiency of electrotransformation with our earlier chemical transformation protocol, we performed the latter technique with circular and HpaI-linearized pWU44 (telomeric PaURA5 plasmid), yielding mean transformation efficiencies (in number of transformants/microgram of DNA) of 1 (n = 5, range, <0.25 to 3) and 11 (n = 5, range, 1 to 24), respectively. By comparison with the analogous experiments shown in Table 1, electrotransformation was about 2 orders of magnitude more efficient than chemical transformation. Since chemical transformation has been reported to have a threshold effect, in which no transformants are obtained until a certain minimum amount of DNA is provided for transformation (30), we examined electrotransformation efficiency by using different amounts of transforming HpaI-linearized pWU44 (Table 2). In these experiments, transformation efficiencies were somewhat higher than in the experiments shown in Table 1, even when identical conditions were used. We obtained transformants using as little as 100 pg of DNA, the smallest amount tested. Over a range of amounts of transforming DNA spanning 5 orders of magnitude (100 pg to 10 μg), the transformation efficiency generally remained high. However, this close correlation between the amount of transforming DNA and number of transformants obtained was less clear with the greater amounts of DNA, for which transformation efficiency was progressively reduced, consistent with saturation of the DNA uptake or transformation capacity of the yeast cells. We also observed an effect of the amount of transforming DNA on the fate of this DNA in the resulting transformants. When a small amount of DNA, such as 1 ng, was used for electrotransformation, nearly all of the transformants carried monomeric linear plasmids as the only location of the transforming DNA, with very rare occurrence of larger plasmids or possibly chromosomal integration (Fig. 3A); this result is similar to that observed after chemical transformation (25). However, when larger amounts of DNA, such as 1 μg, were used, there was more frequent appearance of the transforming DNA on larger plasmids or possibly chromosomally integrated, although transformants with monomeric plasmids were still obtained; occasionally, monomeric plasmids were found in the same transformants with larger plasmids or with possibly chromosomally integrated transforming DNA (Fig. 3B). In addition to examining individual transformants, we pooled numerous transformants obtained by using different amounts of transforming DNA and prepared DNA from the pools to obtain a better representation of the populations of transformants. The results confirmed the findings from the individual transformants: monomeric linear plasmids predominated in pools of transformants obtained with low amounts of DNA, while there was a clear correlation between the occurrence of other fates, i.e., larger plasmids and possibly chromosomal integration, with increasing amounts of transforming DNA (Fig. 3C). We did not attempt to distinguish between the two fates of chromosomal integration and very large linear plasmid formation in this study.
TABLE 2.
Electrotransformation using HpaI-linearized pWU44 (with telomeric termini)
| Amt of transforming DNA | Transformation efficiency (no. of transformants/μg of DNA)
|
||
|---|---|---|---|
| Expt 1 | Expt 2 | Expt 3 | |
| 100 pg | 2 × 104 | NDa | ND |
| 1 ng | 4.8 × 104 | 7.9 × 104 | 2.1 × 104 |
| 10 ng | 3.3 × 104 | 9.4 × 104 | 2.6 × 104 |
| 100 ng | 3.5 × 104 | 5.0 × 104 | 1.2 × 104 |
| 1 μg | 2.6 × 104 | 2.3 × 104 | 7.3 × 103 |
| 10 μg | ND | ND | 1.4 × 103 |
ND, not done.
FIG. 3.
Effect of amount of transforming DNA on the occurrence of monomeric linear plasmids after electroporation of strain G184ASura5-11 with HpaI-linearized pWU44. Each Southern blot was probed with the 32P-labeled selectable marker PaURA5 (1.6-kb EcoRI fragment from pWU44) and contains in the leftmost lane linearized pWU44 (6.7 kb). Positions of molecular size markers are indicated on the left, as is the location of the broad smear of chromosomal fragments in undigested H. capsulatum genomic DNA on the precedent agarose gels. We did not attempt to distinguish between the two fates of chromosomal integration and very large linear plasmid formation in this study. (A) Undigested genomic DNA from 14 transformants (lanes 1 to 14) obtained by using 1 ng of transforming DNA. All transformants carry monomeric linear plasmids except for transformant 2, which shows evidence of either very large linear plasmids or chromosomal integration of the transforming DNA. (B) Undigested genomic DNA from 13 transformants (lanes 1 to 13) obtained by using 1 μg of transforming DNA. Nine of the transformants carry monomeric linear plasmids, alone or with modified linear plasmids; seven of the transformants carry modified linear plasmids, alone or with a monomeric linear plasmid; and one transformant (transformant 8) shows evidence of either very large linear plasmids or chromosomal integration of the transforming DNA. (C) Undigested genomic DNA prepared from pools of transformants. Lane 1, pool of 21 transformants obtained by using 1 ng of transforming DNA; lane 2, 265 transformants obtained by using 10 ng; lane 3, 123 transformants obtained by using 100 ng; lane 4, 780 transformants obtained by using 1 μg; lane 5, 1,512 transformants obtained by using 10 μg. Each pool shows the presence of monomeric linear plasmids, and additionally modified linear plasmids and either very large linear plasmids or chromosomal integration of the transforming DNA are shown in pools obtained by using larger amounts of transforming DNA.
Electrotransformation of H. capsulatum strains with plasmid pAN7-1.
We established conditions for transformation with a plasmid containing the hph gene flanked by Aspergillus regulatory sequences and selection using hygromycin B by plating directly on solid medium containing various concentrations of the antibiotic. Increasing the concentration of hygromycin B reduced the level of background growth and the occurrence of spontaneously resistant mutants that arose in untransformed yeasts or yeasts electroporated with control preparations (water, TE, plasmids lacking the hph gene) but also reduced the efficiency of genuine transformation to antibiotic resistance. For instance, electroporation of strain G184ASura5-11 (with inclusion of uracil in all media) resulted in thousands of colonies on plates containing 50 μg of hygromycin B per ml for both HindIII-linearized pAN7-1 and pBR328 control plasmid (∼5 μg of DNA). When higher levels of antibiotic were used, no spontaneous resistant mutants arose in the pBR328 control samples whereas genuine transformants (confirmed by Southern blotting) did appear in the HindIII-linearized pAN7-1 samples, but the numbers of transformants decreased with increasing concentrations of hygromycin B: 132 with 100 μg/ml, 43 with 200 μg/ml, and 2 with 500 μg/ml. We also occasionally have observed minor strain differences in natural resistance to hygromycin B or the occurrence of spontaneous resistance (data not shown), and we generally use a concentration of 200 μg/ml for selection to achieve relatively high transformation efficiency with negligible occurrence of spontaneous resistance.
Having demonstrated transformation to hygromycin B resistance in the same strain that we have used for most of our URA5 transformation work, G184ASura5-11, we next demonstrated this phenomenon by using several uracil-prototrophic H. capsulatum strains of two different RFLP genotypes: G184AS, G184A-HTE, G186AS, and G217B. Transformation of all of these strains with HindIII-linearized pAN7-1 or pAN7-1 digested with StuI and BamHI resulted in hygromycin B-resistant transformants. StuI/BamHI digestion of pAN7-1 physically separates the TtrpC terminator region from the hph gene, and this treatment actually resulted in a slight increase in the number of transformants relative to linearized pAN7-1 (data not shown).
Reduction in size of hygromycin B marker fragment while retaining function in H. capsulatum.
Since the TtrpC terminator region appeared dispensable in our system, we constructed plasmid pWU90 (Fig. 1), containing only the hph gene and most of the upstream flanking Pgpd promoter region, and showed that electrotransformation using BamHI-linearized pWU90 resulted in hygromycin B-resistant transformants in strains G184AS, G184A-HTE, and G217B. We then removed substantial 5′ distal parts of the Pgpd promoter region to construct plasmids pMAD91 and pMAD92 (Fig. 1) and tested the effect on H. capsulatum transformation. Electrotransformation of strain G184ASura5-11 (with inclusion of uracil in all media) by using BamHI-linearized plasmids pWU90, pMAD91, and pMAD92 yielded transformation efficiencies of 2, 7, and 12 transformants per μg of DNA, respectively, after plating directly on solid medium containing 200 μg of hygromycin B per ml. Similar results were obtained in experiments using recipient strain G184AS and allowing 48 h of nonselective expression time before exposing the transformation filters to hygromycin B (see below): transformation efficiencies of 16, 32, and 8 transformants per μg of DNA for BamHI-linearized plasmids pWU90, pMAD91, and pMAD92, respectively.
Relative transformation efficiencies and cotransformation efficiency for the URA5 and hygromycin B resistance markers.
We constructed plasmid pMAD93 (Fig. 1), a telomeric plasmid containing both the PaURA5 gene and the hygromycin B resistance marker from pWU90, and plasmid pWU45, a similar control plasmid lacking the hygromycin B resistance marker. These plasmids, linearized with HpaI to expose telomeric termini, were electrotransformed into strain G184ASura5-11 followed by plating directly on solid medium under conditions selective either for URA5 (no exogenous uracil) or hygromycin B (200 μg/ml) transformation or both simultaneously. HpaI-linearized pWU45 yielded ∼8,000 transformants per μg of DNA under uracil selection and, as expected, none when hygromycin B was present. HpaI-linearized pMAD93 yielded ∼10,000 transformants per μg of DNA under uracil selection, ∼2,400 transformants/μg under hygromycin B selection, and ∼300 transformants/μg when both selections were imposed simultaneously. Thus, while pMAD93 was able to transform H. capsulatum with an efficiency comparable to that of the control plasmid pWU45 under uracil selection, the efficiency was somewhat reduced when hygromycin B selection was used and greatly reduced when both selections were imposed simultaneously. One hypothesis that we considered was that despite the telomeric nature of pMAD93 and its generally high transformation efficiency, the hygromycin B resistance marker did not function effectively in H. capsulatum or was being lost from the transformants. Although we cannot universally exclude these possibilities in every case, two lines of evidence were inconsistent with this hypothesis. First, when HpaI-linearized pMAD93 transformants initially selected for one of the markers were tested under the other selective condition or both selective conditions, all or most grew under the other condition(s). Second, probing of Southern blots of uncut genomic DNA from HpaI-linearized pMAD93 transformants with the hygromycin B resistance marker invariably showed the presence of this marker in the transformants, generally on linear plasmids, analogous to results seen when URA5 was used as the selectable marker on telomeric plasmids (data not shown).
Allowing nonselective time for expression of hygromycin B resistance.
An alternate hypothesis for the different transformation efficiencies under the two selective conditions was that immediate imposition of hygromycin B selection by plating after electroporation directly on solid medium containing antibiotic was damaging the recipient cells before the hph gene product (and in the case of double selection, the URA5 gene product) could be sufficiently expressed. This idea is consistent with the hygromycin B’s mechanism of action as a protein synthesis inhibitor. Allowing nonselective expression time after transformation is a common technique with bacteria (3, 18), and so we tested the effect on electrotransformation efficiency of allowing variable amounts of time under nonselective conditions before exposure to hygromycin B (Table 3). Two uracil-auxotrophic recipient strains of different RFLP genotypes, G184ASura5-11 and G217Bura5-21, were electrotransformed with HpaI-linearized pMAD93, followed by plating on Nytran filters on HMM-agarose lacking uracil or hygromycin B; thus, uracil selection was immediately and continuously imposed in these experiments. As a control for URA5 transformation alone, one sample was left on the medium without hygromycin B. Four other filters in each experiment were transferred to HMM-agarose containing 200 μg of hygromycin B per ml at various times after transformation, from 0 h (immediate hygromycin B selection) up to 48 h of nonselective time. For both recipient strains, immediate imposition of hygromycin B selection substantially reduced transformation efficiency; for G217Bura5-21, no transformants were obtained. The transformation efficiency increased as more nonselective time was allowed. In one experiment with strain G184ASura5-11, 6 h of nonselective time was sufficient for an increase in transformation efficiency to approximately that of URA5 transformation. With strain G217Bura5-21, the transformation efficiency increased progressively as more nonselective time was allowed before transfer to hygromycin B medium, and the hygromycin B resistance transformation efficiency did not achieve the level of URA5 transformation efficiency even after 48 h. In two of the experiments in Table 3, HpaI-linearized pWU51, a telomeric plasmid carrying the HcURA5 gene, was used as a control, yielding 1,100 transformants/μg of DNA for G184AS ura5-11 experiment 1 and 390 transformants/μg of DNA for the G217Bura5-21 experiment, using only uracil selection. No colonies appeared on pWU51 transformation filters on hygromycin B plates, even with 48 h allowed before exposure to hygromycin B, consistent with this plasmid’s lack of a hygromycin B resistance marker but also indicating no occurrence of spontaneous hygromycin B-resistant mutants under these conditions. However, we have observed that allowing more than 48 h of nonselective time occasionally results in the appearance of spontaneous resistant mutants, especially when only hygromycin B transformation in uracil-prototrophic strains is used, an experimental condition that lacks the suppression of background growth imposed by uracil selection in ura5 recipient strains (data not shown). Thus, 48 h is generally the maximum amount of nonselective time that we allow, even though hygromycin B transformation efficiency might continue to increase beyond this time.
TABLE 3.
Electrotransformation of H. capsulatum with HpaI-linearized pMAD93
| Expt | Strain | Time of transfer to hygromycin B plate after electroporation (h)a | Transformation efficiency (no. of transformants/ μg of DNA) |
|---|---|---|---|
| 1 | G184ASura5-11 | — | 640 |
| 0 | 94 | ||
| 6 | 580 | ||
| 24 | 660 | ||
| 48 | 690 | ||
| 2 | G217Bura5-21 | — | 980 |
| 0 | <1 | ||
| 6 | 3 | ||
| 24 | 52 | ||
| 48 | 140 | ||
| 3 | G184ASura5-11 | — | 423 |
| 0 | 15 | ||
| 6 | 209 | ||
| 24 | 312 | ||
| 48 | 320 |
Transformed cells were continuously under uracil selection, i.e., on medium without uracil. —, never (only uracil selection).
Detection and quantitation of β-galactosidase expressed from an HcURA5-lacZ fusion gene in H. capsulatum.
We constructed telomeric plasmid pWU77 (Fig. 1), containing the PaURA5 gene as a selectable marker and an HcURA5-lacZ fusion gene as described in Materials and Methods, digested it with PacI to expose telomeric termini, and transformed strain G184ASura5-11. Uracil-prototrophic transformants were tested for the ability to hydrolyze the β-galactosidase colorimetric substrate X-Gal by plating the transformations immediately on HMM-agarose containing X-Gal, by growing the transformants first on Nytran filters on HMM-agarose and then transferring the filters to HMM-agarose containing X-Gal or to filter paper soaked with an X-Gal solution, or by adding X-Gal solution directly to colonies on solid medium. Each of these approaches resulted in a readily apparent blue color with no other manipulation required to allow X-Gal access to the presumably cytoplasmic β-galactosidase; an example of the last approach of direct addition of X-Gal solution is shown in Fig. 4. A blue color could also be observed after addition of X-Gal to pWU77 transformant mycelial colonies grown at room temperature. Additionally, pWU77 transformants grown as yeast or as mycelia showed a blue color in individual cells observed microscopically after addition of X-Gal (data not shown). Although transformants did arise on HMM-agarose containing X-Gal, there was evidence that the accumulation of the insoluble blue product of the β-galactosidase reaction was deleterious to the growth of the transformant colonies. Immediate plating on X-Gal resulted in reduced transformation efficiency and decreased size of the colonies in direct correlation with the concentration of X-Gal included in the medium (ranging from 10 to 100 μg/ml). These effects were not due to toxicity of unhydrolyzed X-Gal, since untransformed H. capsulatum or transformants obtained by using plasmids without the fusion gene were not significantly impaired in colony number or size by X-Gal in the medium (data not shown). Therefore, we routinely grew transformants in the absence of X-Gal and visually detected β-galactosidase activity by transfer of colonies on filters to X-Gal-containing agarose or filter paper or by direct addition of X-Gal solution. Several transformants confirmed by Southern blotting to carry the expected monomeric linear plasmids (data not shown) were also subjected to a cell lysis protocol as described in Materials and Methods, and the cytoplasmic preparations were assayed for β-galactosidase activity by using the colorimetric substrate ONPG (Table 4). The results indicated readily detectable and quantifiable activity resulting from expression of the fusion gene.
FIG. 4.
Colorimetric detection of HcURA5-lacZ fusion gene expression in colonies on solid medium. Strain G184ASura5-11 was electrotransformed with PacI-digested pWU77 carrying the fusion gene (top line, blue color) or with HpaI-linearized plasmid pWU44 lacking the fusion gene (bottom line, white color) and streaked in a pattern on HMM-agarose. Following growth, X-Gal solution was added to the streaks, leading to the appearance of blue color within minutes.
TABLE 4.
ONPG assays of cytoplasmic preparations
| Strain | β-Galactosidase activitya (mU/mg of protein) |
|---|---|
| Parental G184AS | 0 ± 0 |
| Recipient G184ASura5-11 | 8 ± 14 |
| Transformants | |
| pWU77/Pac-1 | 578 ± 68 |
| pWU77/Pac-2 | 708 ± 90 |
| pWU77/Pac-3 | 673 ± 99 |
| pWU77/Pac-4 | 741 ± 57 |
Mean ± standard deviation for three cultures of each strain, triplicate wells for each culture.
DISCUSSION
We previously demonstrated genetic transformation of H. capsulatum by a method using the PaURA5 marker and DNA delivery by a chemical treatment protocol, but the relatively low efficiency of this method and the existence of only a single marker, itself useful only in ura5 mutant strains, have restricted the ability of investigators to perform crucial elements of molecular genetic technology in this medically important fungus. Such previously inaccessible approaches to studying biology and pathogenesis in this organism include (i) transformation and gene disruption in wild-type strains and (ii) facile detection and quantitation of gene expression that may be environmentally regulated or associated with virulence. In this work, we have developed important tools and techniques for molecular genetic manipulation and analysis of H. capsulatum, elucidated aspects of its basic molecular biology such as handling of transforming DNA and expression of selectable markers, and demonstrated H. capsulatum expression of bacterial genes for the first time, to our knowledge.
The limited efficiency of the previously described chemical transformation system is adequate for delivery of specific cloned genes by transformation but is ill suited, for example, to any random cloning approach, such as screening random libraries of cloned Histoplasma DNA for phenotypic complementation in the native organism. Therefore, we developed an electrotransformation system that is far more efficient and has the added advantages of better reproducibility, greater experimental ease, and reduced manipulation of the recipient yeasts. With this more reproducible protocol, we were able to address definitively characteristics of the transforming DNA in regard to their effect on transformation efficiency and the fate of the transforming DNA. While the plasmid vector and the source of the selectable URA5 gene did not show any effect, in vitro restriction enzyme-mediated linearization of the transforming plasmid and the presence of telomeres on the transforming circular or linearized DNA each independently resulted in about 1 to 2 orders of magnitude enhancement of transformation efficiency relative to circular or to nontelomeric plasmids. Linear plasmids with telomeric termini showed transformation efficiencies of >103 to nearly 105 transformants per μg of transforming DNA, comparable to efficiencies observed with model organisms such as Saccharomyces cerevisiae and potentially enabling the construction of random genomic libraries in telomeric plasmids to approximate representation of the H. capsulatum genome. We are currently pursuing such a quest in order to achieve complementation cloning of Histoplasma genes in the native genetic background.
While gratified with the increased efficiency of electrotransformation, we were initially puzzled by deviation in the fate of the transforming DNA from the nearly universal occurrence of monomeric linear plasmids observed after chemical transformation (Fig. 2; and reference 25). However, we determined that the more frequent occurrence of modified linear plasmids and possibly chromosomal integration of transforming DNA after electroporation was correlated with the amount of transforming DNA used and hypothesize that the phenomenon may be an effect of the very efficiency of the system for introducing DNA into competent cells. The telomeric sequences included on our plasmids are 77 to 90 bp in length, consisting of 12 exact repeats of the hexameric subunit GGGTTA and some other telomere-like sequence, and are thus shorter than native fungal telomeres; we have previously shown that these sequences are extended after transformation to a length of 150 to 250 bp or longer, presumably by the action of telomerase, and the resulting plasmids are stable and autonomously replicating (25). Thus, these regions can efficiently serve as a core for telomere development but are not sufficient to function as telomeres without further extension. We hypothesize that the efficiency of electroporation enables the entry of more than one transforming DNA molecule into the same recipient cell and that these molecules then face a number of possible fates, including telomere extension to stabilize monomeric linear plasmids (as desired by the investigators) but also interaction with other transforming molecules by ligation or recombination leading to multimerization and/or interaction with chromosomal DNA by integration. Our results that are consistent with this hypothesis include the decrease in transformation efficiency with the largest amounts of transforming DNA, as would occur if the DNA uptake or transformation mechanisms were approaching saturation, and the correlation of the occurrence of these alternate fates with increasing amounts of transforming DNA. We have no direct evidence for such interaction of two or more transforming molecules entering the same cell in this work, but there are precedents for recombination between transforming DNA molecules in other systems (2). Additionally, in other work (26, 27), we have observed direct in vivo ligation at the termini of transforming DNA molecules introduced by electroporation to regenerate a selectable marker gene.
Genetic transformation of fungi, including the well-studied model organism S. cerevisiae, has most frequently involved genes encoding metabolic enzymes as selectable markers that complement auxotrophic mutations in recipient strains rather than antibiotic resistance genes used most frequently as markers for transformation of bacteria (11, 20). Attractions to this approach include in some cases the stability of the untransformed auxotrophic phenotype (lack of spontaneous mutation to prototrophy) and the ability to impose selection for the transforming DNA immediately after transformation, without consideration of expression time for the selectable marker. However, a major drawback is the requirement for specific auxotrophic mutants as transformation recipients, and so molecular genetic analyses can be performed only with well-characterized mutant laboratory strains or specific auxotrophic mutants must be isolated for a new strain, frequently by mutagenic techniques. With H. capsulatum, we have been confined to specific ura5 mutants of very few strains, and these mutants were constructed by untargeted and random UV mutagenesis. Dominant selectable markers, such as genes encoding resistance to antibiotics to which the organism is susceptible, may potentially be used in any strain, including wild-type prototrophs and fresh clinical isolates, without the need for mutagenesis or isolation of specific auxotrophs. Our work with the hygromycin B resistance marker demonstrates the utility of such an approach for nonmutagenized prototrophic strains of H. capsulatum, as well as providing a second selectable marker for ura5 mutant strains.
The 4.1-kb size and concomitant plethora of restriction sites in the fungal selectable marker present on the oft-used plasmid pAN7-1 are inconvenient in plasmid construction, and we have shown that the marker can be substantially reduced in size and still provide effective function in H. capsulatum. The TtrpC terminator region was omitted from nearly all of our transforming constructs, and much of the Pgpd promoter region was removed to generate the smallest marker, of 1.4 kb, shown to function in H. capsulatum. Finally, we demonstrated the utility of allowing nonselective time after transformation to maximize transformation efficiency using hygromycin B selection, presumably by enhancing expression of the hph gene product before exposure to the protein synthesis inhibitor hygromycin B. In spite of its utility and clear advantages in some situations, e.g., when two markers are required or when a uracil-prototrophic transformation recipient is required, antibiotic as opposed to uracil selection does have clear drawbacks for which compromises must be made. One example is that increasing the concentration of hygromycin B reduces the occurrence of spontaneous resistance but also reduces the transformation efficiency. A second example is that increasing the nonselective expression time allowed after electroporation before exposure to hygromycin B increases the transformation efficiency but also increases background growth and the occurrence of spontaneous resistance.
The hygromycin B resistance marker, coupled with the increased transformation efficiency afforded by electroporation, has proved essential in allowing us to achieve homologous gene targeting by allelic replacement at the URA5 locus in H. capsulatum. In this case, a URA5 marker was obviously inappropriate for disrupting URA5 in the uracil-prototrophic strain G184AS, and so we used the hygromycin B resistance marker to replace an internal fragment of the HcURA5 gene, leaving both 5′ and 3′ flanking sequences. Allelic replacement of the native locus by crossover in the flanking sequences following electrotransformation with the linearized construct occurred with a frequency of about one homologously targeted event per 103 hygromycin-resistant transformants (26, 27). Such relatively rare homologous targeting is similar to that found in some other pathogenic fungi (6) and very different than the facility of allelic replacement in S. cerevisiae. This point emphasizes the necessity for an efficient transformation system capable of readily generating many transformants, such as the electroporation protocol described here, and for optimization of the transformation system, such as by the allowance of nonselective expression time for the hygromycin B resistance marker, as described here.
There have been several examples of heterologous genes or sequences functioning for transformation selection in H. capsulatum, including the P. anserina URA5 gene (24, 25, 30) and now the E. coli hph coding sequence with an A. nidulans Pgpd promoter region (this work). We have also demonstrated here the utility of an E. coli lacZ coding sequence when fused to the 5′ region of the HcURA5 gene to encode functional β-galactosidase. Our results indicate that the relatively long fusion gene is effectively transcribed and translated, that the encoded protein undergoes the tetramerization necessary for enzymatic function, that β-galactosidase activity can be detected and quantified in cytoplasmic preparations using ONPG, and that the colorimetric substrate X-Gal can be hydrolyzed in intact cells, presumably after gaining access to the cytoplasm without the need for any artificial permeabilization techniques as have been used for other organisms (1). The latter point is indicated by the occurrence of blue color from X-Gal hydrolysis in colonies on solid medium and in individual yeast or mycelial cells viewed microscopically and by the deleterious biological effects (reduced colony number and size) for fusion gene transformants on X-Gal medium. We are currently using this marker as a reporter for native H. capsulatum promoters other than that of the HcURA5 gene and as a means of detecting differential and regulated gene expression, including expression of genes induced during in vivo infection (17) and of the CBP gene encoding a Histoplasma calcium-binding protein (14).
ACKNOWLEDGMENTS
This work was supported by Public Health Service grants HL55949 from the National Heart, Lung, and Blood Institute to J.P.W. and AI25584 from the National Institute of Allergy and Infectious Diseases to W.E.G. J.P.W. is a Lucille P. Markey Scholar, and this work was supported in part by Scholar Award 94-21 from the Lucille P. Markey Charitable Trust. W.E.G. is a recipient of the Burroughs Wellcome Fund Scholar Award in Pathogenic Mycology.
We thank Paul Szaniszlo and Ming Peng, University of Texas, for supplying plasmid pAN7-1; Alan Greener, Stratagene, for the tetracycline-sensitive variant of SURE; and members of our laboratories for discussion and assistance.
REFERENCES
- 1.Aguirre J. Spatial and temporal controls of the Aspergillus brlA developmental regulatory gene. Mol Microbiol. 1993;8:211–218. doi: 10.1111/j.1365-2958.1993.tb01565.x. [DOI] [PubMed] [Google Scholar]
- 2.Ahn B-Y, Dornfeld K J, Fagrelius T J, Livingston D M. Effect of limited homology on gene conversion in a Saccharomyces cerevisiae plasmid recombination system. Mol Cell Biol. 1988;8:2442–2448. doi: 10.1128/mcb.8.6.2442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Ausubel F M, Brent R, Kingston R E, Moore D D, Seidman J G, Smith J A, Struhl K, editors. Current protocols in molecular biology. New York, N.Y: John Wiley & Sons, Inc.; 1994. . (CD-ROM version.) [Google Scholar]
- 4.Brown D H, Jr, Slobodkin I V, Kumamoto C A. Stable transformation and regulated expression of an inducible reporter construct in Candida albicans using restriction enzyme-mediated integration. Mol Gen Genet. 1996;251:75–80. doi: 10.1007/BF02174347. [DOI] [PubMed] [Google Scholar]
- 5.Budarf M, Blackburn E. S1 nuclease sensitivity of a double-stranded telomeric DNA sequence. Nucleic Acids Res. 1987;15:6273–6292. doi: 10.1093/nar/15.15.6273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Chang Y C, Penoyer L A, Kwon-Chung K J. The second capsule gene of Cryptococcus neoformans, CAP64, is essential for virulence. Infect Immun. 1996;64:1977–1983. doi: 10.1128/iai.64.6.1977-1983.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Collins J. Instability of palindromic DNA in Escherichia coli. Cold Spring Harbor Symp Quant Biol. 1981;45:409–416. doi: 10.1101/sqb.1981.045.01.055. [DOI] [PubMed] [Google Scholar]
- 8.Eissenberg L G, Goldman W E. Histoplasma variation and adaptive strategies for parasitism: new perspectives on histoplasmosis. Clin Microbiol Rev. 1991;4:411–421. doi: 10.1128/cmr.4.4.411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Eissenberg L G, Goldman W E. The interplay between Histoplasma capsulatum and its host cells. Baillière’s Clin Infect Dis. 1994;1:265–283. [Google Scholar]
- 10.Eissenberg L G, West J L, Woods J P, Goldman W E. Infection of P388D1 macrophages and respiratory epithelial cells by Histoplasma capsulatum: selection of avirulent variants and their potential role in persistent histoplasmosis. Infect Immun. 1991;59:1639–1646. doi: 10.1128/iai.59.5.1639-1646.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Fincham J R S. Transformation in fungi. Microbiol Rev. 1989;53:148–170. doi: 10.1128/mr.53.1.148-170.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Goldman W E. Molecular genetic technology transfer to pathogenic fungi. Arch Med Res. 1995;26:437–440. [PubMed] [Google Scholar]
- 13.Hogan L H, Klein B S. Transforming DNA integrates at multiple sites in the dimorphic fungal pathogen Blastomyces dermatitidis. Gene. 1997;186:219–226. doi: 10.1016/s0378-1119(96)00713-5. [DOI] [PubMed] [Google Scholar]
- 14.Patel, J. B., J. W. Batanghari, and W. E. Goldman. Probing the yeast-phase specific expression of the CBP1 gene in Histoplasma capsulatum. Submitted for publication. [DOI] [PMC free article] [PubMed]
- 15.Peng M, Cooper C R, Jr, Szaniszlo P J. Genetic transformation of the pathogenic fungus Wangiella dermatitidis. Appl Microbiol Biotechnol. 1995;44:444–450. doi: 10.1007/BF00169942. [DOI] [PubMed] [Google Scholar]
- 16.Punt P J, Oliver R P, Dingemanse M A, Pouwels P H, van den Hondel C A M J J. Transformation of Aspergillus based on the hygromycin B resistance marker from Escherichia coli. Gene. 1987;56:117–124. doi: 10.1016/0378-1119(87)90164-8. [DOI] [PubMed] [Google Scholar]
- 17.Retallack D M, Heinecke E L, Woods J P. Abstracts of the 13th Congress of the International Society for Human and Animal Mycology. Parma, Italy: International Society for Human and Animal Mycology and University of Parma; 1997. Development of a method to identify Histoplasma capsulatum genes expressed exclusively in vivo, abstr. P268; p. 145. [Google Scholar]
- 18.Sambrook J, Fritsch E F, Maniatis T. Molecular cloning: a laboratory manual. 2nd ed. Cold Spring Harbor, N.Y: Cold Spring Harbor Laboratory Press; 1989. [Google Scholar]
- 19.Smith J M, Tang C M, Noorden S V, Holden D W. Virulence of Aspergillus fumigatus double mutants lacking restriction and an alkaline protease in a low-dose model of invasive pulmonary aspergillosis. Infect Immun. 1994;62:5247–5254. doi: 10.1128/iai.62.12.5247-5254.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Timberlake W E, Marshall M A. Genetic engineering of filamentous fungi. Science. 1989;244:1313–1317. doi: 10.1126/science.2525275. [DOI] [PubMed] [Google Scholar]
- 21.Voisard C, Wang J, McEvoy J L, Xu P, Leong S A. urbs1, a gene regulating siderophore biosynthesis in Ustilago maydis, encodes a protein similar to the erythroid transcription factor GATA-1. Mol Cell Biol. 1993;13:7091–7100. doi: 10.1128/mcb.13.11.7091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Wheat L J. Histoplasmosis. Infect Dis Clin North Am. 1988;2:841–859. [PubMed] [Google Scholar]
- 23.Woods, J. P., and W. E. Goldman. Unpublished data.
- 24.Woods J P, Goldman W E. In vivo generation of linear plasmids with addition of telomeric sequences by Histoplasma capsulatum. Mol Microbiol. 1992;6:3603–3610. doi: 10.1111/j.1365-2958.1992.tb01796.x. [DOI] [PubMed] [Google Scholar]
- 25.Woods J P, Goldman W E. Autonomous replication of foreign DNA in Histoplasma capsulatum: role of native telomeric sequences. J Bacteriol. 1993;175:636–641. doi: 10.1128/jb.175.3.636-641.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Woods J P, Heinecke E L. Abstracts of the 96th General Meeting of the American Society for Microbiology 1996. Washington, D.C: American Society for Microbiology; 1996. Homologous gene targeting in Histoplasma capsulatum: URA5 gene disruption using a hygromycin resistance marker, abstr. F-116; p. 94. [Google Scholar]
- 27.Woods, J. P., D. M. Retallack, E. L. Heinecke, and W. E. Goldman. Unpublished data.
- 28.Worsham P L, Goldman W E. Quantitative plating of Histoplasma capsulatum without addition of conditioned medium or siderophores. J Med Vet Mycol. 1988;26:137–143. [PubMed] [Google Scholar]
- 29.Worsham P L, Goldman W E. Selection and characterization of ura5 mutants of Histoplasma capsulatum. Mol Gen Genet. 1988;214:348–352. doi: 10.1007/BF00337734. [DOI] [PubMed] [Google Scholar]
- 30.Worsham P L, Goldman W E. Development of a genetic transformation system for Histoplasma capsulatum: complementation of uracil auxotrophy. Mol Gen Genet. 1990;221:358–362. doi: 10.1007/BF00259400. [DOI] [PubMed] [Google Scholar]
- 31.Zhou C, Yang Y, Jong A Y. Mini-prep in ten minutes. BioTechniques. 1990;8:172–173. [PubMed] [Google Scholar]