Abstract
Introduction
Following lockdown periods and restricting public health measures in response to the COVID-19 pandemic, respiratory tract infections (RTIs) rose significantly worldwide. This led to an increased burden on children’s hospitals compromising medical care of acutely and chronically ill children. We characterized changes in the epidemiological pattern of circulating respiratory viral infections.
Methods
We assessed the number of patients with RTIs and the annual distribution of virus detections between 2019 and 2022 based on 4809 clinical samples (4131 patients) from a German pediatric tertiary care-center. We investigated the impact of lockdown periods on spectra of circulating respiratory viruses, pattern of coinfections, age, and seasonality of infections.
Results
A fourfold increase in the number of respiratory virus detections was observed in 2022 vs 2019 with numbers doubling in 2022 (vs 2021). In 2022, seasonal patterns of circulating virus, particularly Adeno and seasonal Coronavirus were far less pronounced compared to previous years, in fact almost disappeared for Rhinoviruses.”. SARS-CoV-2, Parainfluenza- and human Metapneumovirus detections increased significantly in 2022 (2019 vs 2022, p < 0.01). Coinfections with multiple viruses occurred more frequently since 2021 compared to pre-pandemic years, especially in younger children (2019 vs 2022, p < 0.01).
Conclusion
Compared to pre-pandemic years, we observed a dramatic increase in pediatric RTIs with an incrementing spectrum of viruses and a predominance in Rhino/Enterovirus infections – leading to a high rate of hospital admissions, particularly in conjunction with other viruses. This caused an acute shortage in medical care and may also be followed by an increase of virus-triggered secondary chronic respiratory diseases like asthma—rendering a burden on the health system.
Supplementary Information
The online version contains supplementary material available at 10.1007/s15010-023-02085-w.
Keywords: Respiratory tract infection, Pandemic, Epidemiology, Rhinovirus, Respiratory viruses
Introduction
Respiratory tract infections (RTIs) are among the most common childhood illnesses resulting in a high number of physician contacts and hospitalizations. RTIs contribute decisively to morbidity and mortality in the early years of life [1]. Infections with respiratory syncytial virus (RSV) and Rhinovirus type A and C are associated with chronic lung disease, like asthma, later in life [2–4]. RTIs are characterized by an epidemic occurrence at virus-specific seasonal times [5, 6]. Climatic factors such as temperature and humidity affect respiratory virus stability and transmission rates, and appear to affect the host’s intrinsic, innate, and adaptive immune responses to RTIs. This may explain why most viral infections were observed in the fall and winter seasons [7, 8]. However, human behavioral patterns are a crucial factor in the spread of viruses influencing contact rates between infected individuals and susceptible hosts. With the onset of the COVID-19 pandemic, contact rates were significantly reduced to contain the spread of SARS-CoV-2. In addition, NPI such as the use of masks and hand hygiene significantly reduced the transmission of viruses. This led not only to containment of the pandemic but also to a significant decrease in other viral infections, such as Influenza A/B and RSV [9, 10]. After easing of pandemic-related measures, a rising and persistent wave of non-COVID-19 RTIs in children was observed in various countries since June 2021 [11–14].
After initially assuming that respiratory viruses would only occur outside of their typical season, it quickly became apparent that the number of infected children was also increasing [12]. The actual impact on epidemiological changes that occurred by the end of 2022, the consequences and potential causes of these changes are not well-understood [15].
At the beginning of the pandemic, infections with SARS-CoV-2 played only a minor role in children. From autumn 2021, with the beginning of the circulation of the omicron variant, younger patients got increasingly infected [16]. Initially, only mild acute infections occurred, with admissions to children’s hospitals increasing from September 2021. Agathis et al. described that patients with co-detection of other respiratory viruses other than RSV and Rhino/Enterovirus (RV), showed a more severe course of disease [17].
Although several European countries reported an increase in RTIs in 2022, there is hardly any data as to whether the frequency and clinical impact of coinfections differed compared to the pre-pandemic period. It remains unclear whether this development will only remain a short-term phenomenon. It is equally unclear whether children with severe courses of acute infection will have to fear long-term consequences. We know that infections with RV or RSV, even if they take a mild course, significantly increase the risk of respiratory complications and chronic respiratory illness [4].
In this study, we aimed to illustrate the spectra of circulating viruses, patterns of coinfections and age and seasonality of RTIs in children from 2019 to 2022. We set out to provide a better understanding of pandemic-related changes in the epidemiology and the interaction of viral respiratory diseases—whose impact has since increased drastically on the population and healthcare system.
Methods
At the Hauner Children’s Hospital (Munich), a large tertiary care university hospital, the number of detected viral infections in patients between January 2019 and November 2022 was investigated. We analyzed 4702 (2019 with n = 550, 2020 with n = 735, 2021 with n = 1243, and 2022 with n = 2243) positive viral results from routine clinical specimens of both outpatients and inpatients (n = 4131). We included all patients presenting as outpatients or inpatients in the defined study period that had a positive test result at the in-house microbiology laboratory for at least one of the following viruses: Influenza A/B, RSV, Parainfluenza virus, human Metapneumovirus, Adenovirus, seasonal Coronaviruses (OC43, NL63, E229, HKU1) SARS-CoV-2, and Rhino/Enterovirus. Testing was performed at the discretion of the attending physician when an acute respiratory illness was diagnosed requiring either inpatient admission or outpatient drug treatment. Particular criteria were: reduction in oxygen saturation below 92%, wheezing, fever, poor general condition or young age of the patient. During the COVID-19 pandemic, multiplex-PCR testing (including RSV, Influenza A/B, and SARS-CoV-2) was performed on patients regardless of specific respiratory symptoms in case of hospital admission. Samples were taken by nasal or pharyngeal. All samples were tested by immunofluorescence assay (Quidel Sofia 1) [18] or using of a Mulitplex PCR system; (BioMerieux Biofire, Askim, SWEDEN [19]; Extraction by Nimbus and subsequent analysis by BioRad CFX, Seegene PCR-Kit) [20].
Statistical analysis
The Wilcoxon’s rank test was used to compare categorical (grouped) variables. The Pearson’s Chi-square test (using Cramer’s V) was used to evaluate for the goodness of fit between variables. The Z-test was used to compare proportions of the number of positive tests for individual virus groups across gender (males vs females). The analysis we assumed independence between the categorical (or grouped) variables. The variables included in the Chi-square test were gender (males vs females), age-groups (< 6, 6–12, and ≥ 12 years of age), virus infection mode (as binary: single vs multiple infections). Single virus infections were more prominent among children from the younger age-groups (< 6, 6–12, ≥ 12 years), so we performed a Chi-square test for independence to assess if the infection mode is independent of the age-group. Proportionally fewer subjects with coinfections (“multiple”) were observed in the pre-pandemic year (2019) compared to the pandemic years (vs 2020, vs 2021, vs 2022). The Kruskal–Wallis test was used to test for differences in the overall mean age of children across four years. In the post-hoc analysis, differences in the mean age of children between the four years (pairwise comparisons) were done using the Wilcoxon’s rank test. The data was curated in Microsoft Excel and analyzed in R software [21].
Ethics approval
The Ethics committee of the Ludwig-Maximilians University (LMU) Munich approved this study under project numbers 21-0334 and 23-0061.
Results
Prevalence of RTIs from 2019–2022
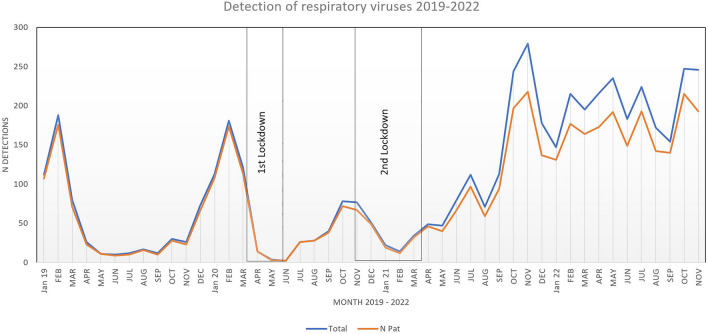
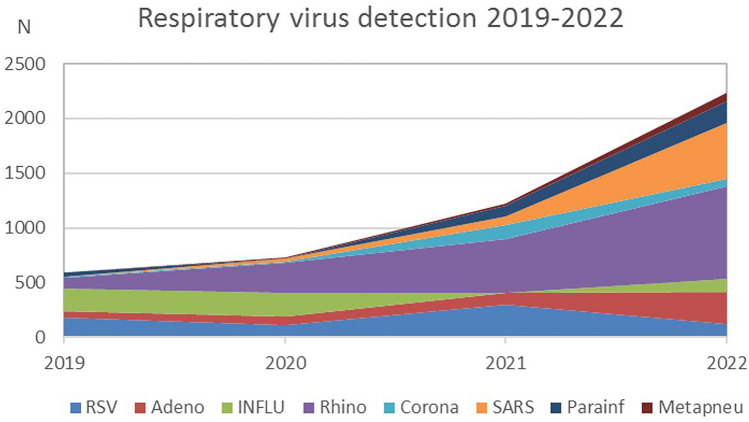
In pre-pandemic years (2017–2019), we demonstrated stable numbers of respiratory virus detections following a seasonal pattern [11]. As previously described, changes in the epidemiology of respiratory infections occurred in the context of introducing pandemic hygiene measures. These led to a significant reduction in circulating respiratory viruses, followed by a marked increase in circulating respiratory viruses after the first and second lockdowns [11]. Comparing the total numbers for respiratory virus with 2019, an increase of 23% in 2020, 100% in 2021 and 270% in 2022 was observed (Table 1). An all-time high in respiratory virus detection was observed in November 2021, with a decline to pre-pandemic year levels in December 2021 and January 2022. While both the pre-pandemic and the lockdown periods (1 & 2) showed relatively low numbers in March/April 2019, 2020 and 2021, consistently high detection rates occurred from May 2021 onwards, with significantly higher numbers across all detected viruses (Fig. 1). RV accounted for the highest proportion of infections, followed by SARS-CoV-2, Adenovirus, RSV and Influenza virus. In addition, Parainfluenza- and Metapneumovirus showed increased case numbers (Fig. 2).
Table 1.
Numbers of virus detections and patient characteristics from January 2019 to November 2022. Mean age comparison pairwise comparisons between viruses using Wilcoxon’s test
| 2019 | 2020 | p-value 2019 vs 2020 | 2021 | p-value 2019 vs 2021 | p-value 2020 vs 2021 | 2022 | p-value 2019 vs 2022 | p-value 2020 vs 2022 | p-value 2021 vs 2022 | |
|---|---|---|---|---|---|---|---|---|---|---|
| RSV, n | 184 | 113 | 301 | 124 | ||||||
| Age (mean) | 1.43 | 1.45 | 0.99 | 2.17 | 0.016 | 0.037 | 2.95 | 0.0015 | 0.0039 | 0.16 |
| Sex (% male) | 61.4 | 58.4 | 0.695 | 55.1 | 0.208 | 0.629 | 56.1 | 0.418 | 0.821 | 0.944 |
| Adeno | 55 | 75 | 106 | 298 | ||||||
| Age (mean) | 4.8 | 3.49 | 0.12 | 3.75 | 0.25 | 0.49 | 3.7 | 0.26 | 0.44 | 0.89 |
| Sex (% male) | 61.5 | 57.8 | 0.829 | 58.9 | 0.882 | 1.00 | 53 | 0.324 | 0.576 | 0.352 |
| Influenza A/B | 204 | 223 | 3 | 114 | ||||||
| Age (mean) | 4.36 | 4.7 | 0.29 | 7.28 | 0.1 | 0.19 | 7.12 | < 0.001 | < 0.001 | 0.96 |
| Sex (% male) | 51.5 | 61.9 | 0.037 | 40 | 0.957 | 0.596 | 45.6 | 0.377 | 0.006 | 1.00 |
| Rhinovirus | 101 | 273 | 488 | 846 | ||||||
| Age (mean) | 4.15 | 3.88 | 0.9 | 4.02 | 0.84 | 0.99 | 4.1 | 0.89 | 0.87 | 0.84 |
| Sex (% male) | 75.2 | 62.6 | 0.031 | 58.3 | 0.002 | 0.277 | 54.9 | < 0.001 | 0.032 | 0.225 |
| Corona | 15 | 9 | 128 | 70 | ||||||
| Age (mean) | 1.37 | 5.79 | 0.0019 | 4.18 | 0.002 | 0.13 | 4.48 | 0.0024 | 0.23 | 0.62 |
| Sex (% male) | 73.3 | 75 | 1.00 | 62.9 | 0.610 | 0.756 | 54.3 | 0.286 | 0.457 | 0.306 |
| SARS-COV-2 | 0 | 25 | 508 | |||||||
| Age (mean) | NA | 5.93 | NA | 6.35 | NA | 0.87 | 4.46 | NA | 0.12 | 0.055 |
| Sex (% male) | NA | 68 | NA | 58 | 0.166 | 0.50 | 48.4 | 0.506 | 0.088 | 0.125 |
| Parainfluenza | 35 | 4 | 194 | |||||||
| Age (mean) | 3.82 | 8.38 | 0.023 | 2.57 | 0.26 | 0.0046 | 3.98 | 0.8 | 0.022 | 0.016 |
| Sex (% male) | 88.9 | NA | NA | 50.7 | 0.070 | 1.00 | 52.8 | 0.075 | 0.474 | 0.873 |
| Metapneumo | 3 | 13 | 80 | |||||||
| Age (mean) | 1.03 | 5.42 | 0.043 | 3.21 | 0.31 | 0.069 | 2.62 | 0.3 | 0.012 | 0.99 |
| Sex (% male) | 100 | 1 | NA | 58.3 | 0.733 | 1.00 | 62.5 | 0.731 | 1.00 | 1.00 |
| Total n | 597 | 735 | 2234 | |||||||
| Age (mean) | 3.4 | 3.9 | 0.024 | 3.8 | 1 | 0.092 | 4.4 | < 0.001 | 1 | 0.005 |
| Sex (% male) | 57.9 | 61.7 | 0.901 | 57.5 | 0.158 | < 0.001 | 52.8 | 0.083 | < 0.001 | 0.01 |
| No. patients (N) | 550 | 694 | 1869 |
Mean age of all patients comparing 2019–2022 using chi-square (3 df) = 25.32, p < 0.0001, with n = 4131
Fig. 1.
Monthly distribution of respiratory viruses detected, including Influenza A/B, RSV, Parainfluenzavirus, Metapneumovirus, Adenovirus, seasonal Coronaviruses, SARS-CoV-2 and Rhinovirus (blue) and number of patients infected (orange), years 2019–2022
Fig. 2.
Cumulative numbers of respiratory virus detections (years 2019–2022)
To determine the significance of virus detections in relation to the number of hospitalized patients, admission data from 2019 to 2022 were analyzed. Patients whose primary treatment diagnosis was coded with upper respiratory tract infections (URTI) (ICD-10 J00-J06), influenza/pneumonia (ICD-10 J09-J18), and lower respiratory tract infection (LRTI)/ acute bronchitis (ICD-10 J19-J20) were included. We observed a 22% reduction in hospital admissions in 2020 vs 2019 and an increase of 11% in hospital admission comparing the post-lockdown (2022) with the pre-lockdown period (2019). In addition, an increase of upper respiratory tract infections post-lockdown vs pre-lockdown (2021 vs 2019: 17%; 2022 vs 2019: 30%) and an increase of lower respiratory tract infection post-lockdown vs pre-lockdown (2021 vs 2019: 25%; 2022 vs 2019: 7%) was seen. There was no change in influenza/pneumonia pre- vs post-lockdown; less influenza/pneumonia during lockdown was observed (Supplementary Table E1).
Changes in the seasonal pattern of respiratory viruses
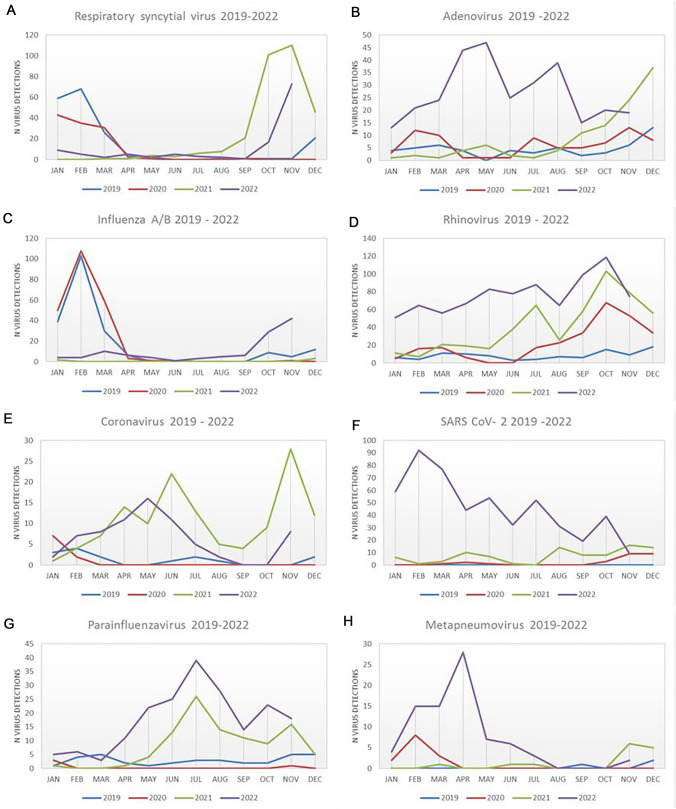
In pre-pandemic years the season for RTIs in the Munich area would start in September and end in March; however, consistently high numbers for RV, Adenovirus and Metapneumovirus were observed in summer 2022 (Figs. 1 and 3B,D,F). We observed distinct changes in the seasonal infection pattern for each virus in 2021 and 2022.
Fig. 3.
Seasonal pattern of number of respiratory virus infections A Respiratory syncytial virus, B Adenovirus, C Influenza A/B, D Rhinovirus, E Seasonal Coronavirus, F SARS-CoV-2, G Parainfluenza, H Metapneumovirus
Infections with Parainfluenza virus were barely detected outside the winter period in pre-pandemic years. However, 2021 and 2022 demonstrated rising numbers of detected viruses with peaks in July 2021/2022. A high number of human Metapneuomovirus infections occurred in April 2022, while in previous years, this virus did not play any particular role in children. Seasonal coronaviruses showed peaks in winter month and around July in pre-pandemic years. In contrast, in 2022, high numbers were observed in May, June and November. SARS-CoV-2 caused only a few relevant childhood infections in the first two pandemic years 2020/2021. In 2022, however, numerous infections occurred, peaking in January, and slowly declining over the summer months. Adenovirus infections became increasingly relevant from September 2021 onwards. The total number of infections remained significantly higher in 2022 compared to the previous years, with a maximum in July and August 2022. In 2019 and 2020, typical Influenza A/B waves were observed from January to March – which were absent in 2021. However, in autumn 2022, increasing numbers of cases occurred well before the typical season (pre-pandemic) for Influenza A/B infection in the region.
The typical infectious season for RSV spreads from November to February, as observed in pre-pandemic years. This changed significantly during the pandemic with infections occurring much earlier and with significantly higher case numbers – with cases detected as early as August 2020. A similar pattern was observed in 2022 with increasing numbers from September. Particularly impressive changes were seen for RV detections. In contrast to all other respiratory viruses, which varied seasonally, Rhino/Enterovirus infections were consistently high across all months in 2022 (Fig. 3).
Coinfections and association between age /gender and type of virus infection
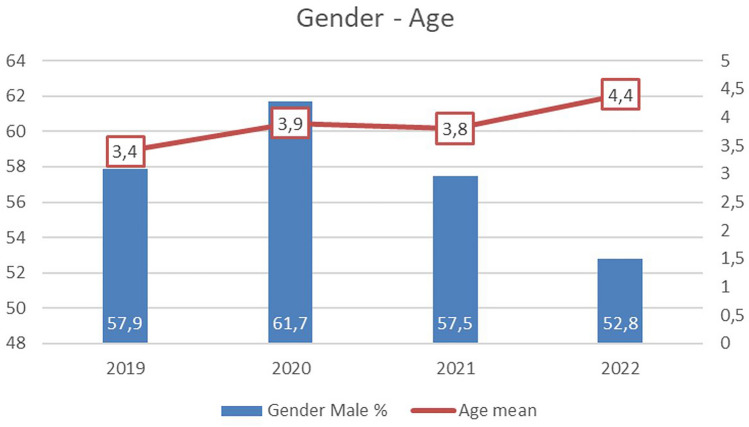
Compared with pre-pandemic years, there were significant changes in age (2019 vs 2022 p < 0.001) and sex-related association with respiratory virus infection (2020 vs 2022 and 2021 vs 2022 p < 0.001) in children, with an increase in female and older children (Fig. 4).
Fig. 4.
Percentage of male and mean age in years of patients with viral respiratory tract infections (years 2019–2022)
Children infected with RSV, seasonal Coronavirus, Influenza A/B, and Metapneumovirus predominantly drove this observation. On the other hand, the number of infections with RV was not associated to the age of affected children. The proportion of females increased for RV infections, and this was also the case for Influenza infections (Table 1). A significant increase in coinfections was observed in 2021 and 2022 (2019 vs 2021 and 2019 vs 2022 p < 0.001) compared to 2019 (Table 2).
Table 2.
Numbers and proportion of respiratory viral co-infections per year (years 2019 – 2022)
| 2019 | 2020 | 2021 | 2022 | Total | P-Value | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| n | % | n | % | n | % | n | % | n | % | 2019 vs 2020 | 2019 vs 2021 | 2019 vs2022 | |
| Single | 501 | 0.91 | 655 | 0.94 | 818 | 0.8 | 1559 | 0.83 | 3533 | 0.86 | 0.003 | 0.00 | 0.00 |
| Multiple | 49 | 0.09 | 39 | 0.06 | 200 | 0.2 | 310 | 0.17 | 598 | 0.14 | |||
| Total | 550 | 1 | 694 | 1 | 1018 | 1 | 1869 | 1 | 4131 | 1 |
In 2019, detection of two viruses in one sample was observed in only 8.9%, this share doubled in 2021 (19.6%) and was 16.6% in 2022 (Fig. 5).
Fig. 5.
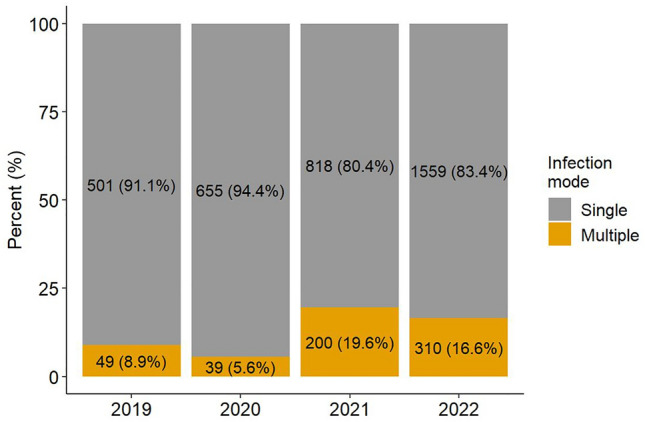
Percentage of single and multiple virus detections, years 2019–2022
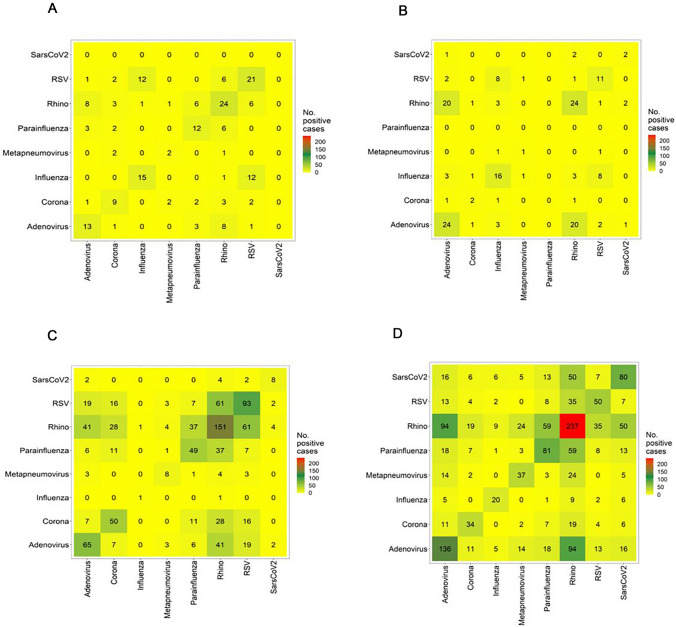
More than two viruses were observed in less than 1% in 2019 and 2020, and occurred in 5–6% of cases in 2021 and 2022. In 2021, up to four viruses per sample were detected for the first time, and in 2022 in individual cases, up to five viruses were detected in a single sample (Table E). Coinfections of RV and SARS-CoV-2 occurred significantly more frequently in younger children (age 0–2 years), and in 2022 in RV and Adenovirus infections (Figure E1, E2). Most coinfections occurred in the context of a RV infection. In 2021, RV coinfections were particularly detected frequently with Parainfluenza or RSV. Coinfections with Metapneumovirus and SARS-CoV-2 were also observed in 2022 (Fig. 6).
Fig. 6.
Heat map diagram showing respiratory viral co-infections: co-infections occurred most frequently in 2022, in combination with Rhinovirus and Parainfluenza, SARS-CoV-2 or RSV, and SARS-CoV-2 with RSV
Discussion
In this study, we observed a dramatic increase in pediatric RTIs with an incrementing spectrum of viruses and an astonishing rise in RV infections leading to a high rate of hospital admissions in 2022. Hospitalizations due to upper and lower respiratory tract infections were observed more frequently in the post-lockdown than in the pre-lockdown period. There were significant changes in the age of affected children and a notable variation in the association between virus infections with sex in 2021–2022 compared to the pre-pandemic year 2019. More female and older children were particularly affected. The annual numbers of coinfections changed markedly, especially in younger children infected with RV.
Epidemics involving viral RTIs have always vary in regional intensity and duration—This is thought to be due to the genetic diversity of circulating viruses and their respective virulence. Climatic changes also seem to impact on virus spread [7, 22, 23]. Numerous studies have shown that NPIs reduce the circulation of SARS-CoV-2 and other respiratory pathogens [24]. While certain non-respiratory infections, such as Rotavirus, occurred less frequently in times of hard lockdowns, a rebound of Rota- and other viruses were already evident when NPIs were still in place. Consistent with our data, Engels et al. reported that Adeno- and Rhino/Enteroviruses detections increased as early as the second and third COVID-19 waves [25]. Interestingly, these are non-enveloped viruses, rendering them less sensitive to disinfection agents. Most other respiratory viruses are transmitted by aerosols. However, Rota-, Adeno- and Rhino/Enterovirus are also following a contact-transmission route and may therefore, be less preventable by the use of facemasks. This corroborates the observation in our study, that RV was highly present despite NPIs. Our results are supported by Buchholz et al.: The authors describe high numbers of RSV infection at the beginning of the year 2020, a sharp decline and low numbers during the lockdown periods and rising numbers in the end of 2021 and 2022. The observation that RV/EV was hardly affected by the hygiene measures and continued to lead to respiratory tract infections from 2020–2021 was also demonstrated. Consistent with our data, an overall increase in infections in 2022 was observed in their multicenter study, on observation that is complemented by our data covering an additional period until November 2022 [26].
We noted that the virus-specific seasonality observed in pre-pandemic years changed in the post-lockdown period. This, however, was not observed for all viruses but was predominantly prevalent for infections with RV, Adenovirus and Metapneumovirus. Host-specific properties of these viruses may also contribute to this finding. While RV is consistently detected in children, RSV is predominantly found in young children during epidemic phases [27]. Outside of epidemics, RSV was mainly detected in adults at risk (usually with COPD—chronic obstructive pulmonary disease) or children with immune deficiency [28, 29]. While RV was able to spread unhindered among children, the occurrence of RSV was long prevented by mandatory masking for adults, which reduced transmission events. This supports our observation that RSV only occurred more frequently after the lifting of the mask regulation in September 2022. Infections with RV, Parainfluenza, Metapneumovirus, and Adenovirus occurred despite the NPIs and were also detected more frequently than before the pandemic. The presence or absence of certain pathogens could have played a role.
In previous studies, virus-virus interactions could be disentangled, which caused a promoting or inhibiting effect on the pathogenicity of viruses [30, 31]. Greer et al. demonstrated that RV detection was associated with a reduced probability of detecting human Adenoviruses, Coronaviruses, Bocavirus, Metapneumovirus, respiratory syncytial virus, Parainfluenza virus and Influenza A virus [32]. A negative interaction was also observed between RV and SARS-CoV-2 by Dee et al., leading to the hypothesis that RV infections could reduce the number COVID-19 cases [33]. The presence of RV could therefore, reduce susceptibility for other RTIs. However, it remains unclear whether the absence of certain viruses influenced susceptibility and pathogenicity of the host towards RV. Contrary to this assumption, the data from our study shows an increase in co-detections, particularly in conjunction with RV. The significance of the increasing virus co-detections cannot be clarified with certainty at present. In chronically ill patients, who make up a large proportion of the inpatients in this study, RV in particular can represent a colonization rather than a trigger for the underlying complaints. Nevertheless, the significant increase in hospital admissions in 2022 in our data indicates a serious clinical impact of multiple detections of respiratory viruses in children.
In addition to the exposure to viral pathobionts, bacteria probably play an equally important role. Initial studies suggest that the reduced exposure to pathogens could have led to a change in the lung microbiome [34]. The development and stability of the microbiome of the respiratory tract are decisive factors in susceptibility to viral infections, which also determine the risk of developing chronic lung diseases such as asthma [35].
Frequent occurrence coinfections, particularly seen in young patients, were complemented by a change in the mean age of the affected children. In 2022, RSV infections were significantly more common in older children. This might be attributed to a lack of exposure to the virus in early childhood and to a lack in transfer of protective antibodies from unexposed mothers. Thus, an increase in the number of cases could be due to an accumulation of various infections in larger group of children including a wider age range who have missed out exposure to pathogens.
Finally, our data indicates that a combination of many confounding factors has caused the increase in detection rates and coinfections and changes in the seasonal pattern of viral RTIs. A limitation of this monocentric study is that it only draws a regional picture and does not allow deductive statements about the impact on the clinical course of the virus detections. This issue needs to be addressed in a follow-up study. Since test strategies have changed during the pandemic, a bias in the detection of viruses cannot be ruled out. However, while short-term consequences for the morbidity of children are already obvious, the impact on resulting chronic lung disease is unclear and remains the focus of further studies.
Conclusion
COVID-19 pandemic-related interventions caused a profound change in the epidemiology of viral RTIs in children, with an increasing spectrum of circulating viruses and an astonishing peak in RV infections leading to a high rate of hospital admissions in 2022. Compared to pre-pandemic years, infections occurred more frequently in female and older children. The annual number of coinfections changed markedly – especially in younger children, predominantly in conjunction with RV. The underlying factors associated with the increase in cases, the peak in coinfections, and changes in the seasonal pattern of viral RTIs are not completely understood and remain the focus of further studies.
Supplementary Information
Below is the link to the electronic supplementary material.
Abbreviations
- NPI
Non-pharmaceutical intervention
- LRTI
Lower respiratory tract infection
- RTI
Respiratory tract infections
- RV
Rhino/Enterovirus
- RSV
Respiratory syncytial virus
- SARS-CoV-2
Severe acute respiratory syndrome coronavirus 2
- URTI
Upper respiratory tract infection
Author contributions
NM, UvB and JO contributed to conceptualization, methodology, data analysis and presentation of the published work and wrote the manuscript. SR, JH and EvM provided scientific advice and specifically critical review, commentary or revision of the manuscript. JO performed data curation and statistical analysis. LK and MMB conducted and supervised lab measurements and data curation, provided scientific advice, specifically critical review, commentary or revision of the manuscript.
Funding
Open Access funding enabled and organized by Projekt DEAL.
Availability of data and materials
The data of this ongoing study are currently not available for public use due to data protection reasons.
Declarations
Conflict of interest
Nicole Maison, Jimmy Omony, Ulrich von Both, Johannes Huebner, Sophia Rinderknecht, Laura Kohlberg, Melanie Meyer-Buehn have no conflict of interest. Erika von Mutius received consulting fees and funding for the research project “Impact of COVID 19 pandemic on patients with asthma and wheeze” from OM Pharma S.A.
Ethical approval
The Ethics committee of the Ludwig-Maximilians University (LMU) Munich approved this study under project numbers 21-0334 and 23-0061.
Footnotes
Nicole Maison is first author and Ulrich von Both last author.
References
- 1.De Conto F, Conversano F, Medici MC, Ferraglia F, Pinardi F, Arcangeletti MC, et al. Epidemiology of human respiratory viruses in children with acute respiratory tract infection in a 3-year hospital-based survey in Northern Italy. Diagn Microbiol Infect Dis. 2019;94:260–7. doi: 10.1016/j.diagmicrobio.2019.01.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Bergroth E, Aakula M, Elenius V, Remes S, Piippo-Savolainen E, Korppi M, et al. Rhinovirus type in severe bronchiolitis and the development of asthma. J Allergy Clin Immunol Pract. 2020;8:588–95 e4. doi: 10.1016/j.jaip.2019.08.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Kotaniemi-Syrjanen A, Vainionpaa R, Reijonen TM, Waris M, Korhonen K, Korppi M. Rhinovirus-induced wheezing in infancy–the first sign of childhood asthma? J Allergy Clin Immunol. 2003;111:66–71. doi: 10.1067/mai.2003.33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Raita Y, Camargo CA, Jr, Bochkov YA, Celedon JC, Gern JE, Mansbach JM, et al. Integrated-omics endotyping of infants with rhinovirus bronchiolitis and risk of childhood asthma. J Allergy Clin Immunol. 2021;147:2108–17. doi: 10.1016/j.jaci.2020.11.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Chadha M, Hirve S, Bancej C, Barr I, Baumeister E, Caetano B, et al. Human respiratory syncytial virus and influenza seasonality patterns-Early findings from the WHO global respiratory syncytial virus surveillance. Influenza Other Respir Viruses. 2020;14:638–46. doi: 10.1111/irv.12726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Wrotek A, Czajkowska M, Jackowska T. Seasonality of respiratory syncytial virus hospitalization. Adv Exp Med Biol. 2020;1279:93–100. doi: 10.1007/5584_2020_503. [DOI] [PubMed] [Google Scholar]
- 7.Moriyama M, Hugentobler WJ, Iwasaki A. Seasonality of respiratory viral infections. Annu Rev Virol. 2020;7:83–101. doi: 10.1146/annurev-virology-012420-022445. [DOI] [PubMed] [Google Scholar]
- 8.Zhu G, Xu D, Zhang Y, Wang T, Zhang L, Gu W, et al. Epidemiological characteristics of four common respiratory viral infections in children. Virol J. 2021;18:10. doi: 10.1186/s12985-020-01475-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Terliesner N, Unterwalder N, Edelmann A, Corman V, Knaust A, Rosenfeld L, et al. Viral infections in hospitalized children in Germany during the COVID-19 pandemic: association with non-pharmaceutical interventions. Front Pediatr. 2022;10:935483. doi: 10.3389/fped.2022.935483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Haapanen M, Renko M, Artama M, Kuitunen I. The impact of the lockdown and the re-opening of schools and day cares on the epidemiology of SARS-CoV-2 and other respiratory infections in children - A nationwide register study in Finland. EClinicalMedicine. 2021;34:100807. doi: 10.1016/j.eclinm.2021.100807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Maison N, Peck A, Illi S, Meyer-Buehn M, von Mutius E, Hubner J, et al. The rising of old foes: impact of lockdown periods on “non-SARS-CoV-2” viral respiratory and gastrointestinal infections. Infection. 2022;50:519–524. doi: 10.1007/s15010-022-01756-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Eden JS, Sikazwe C, Xie R, Deng YM, Sullivan SG, Michie A, et al. Off-season RSV epidemics in Australia after easing of COVID-19 restrictions. Nat Commun. 2022;13:2884. doi: 10.1038/s41467-022-30485-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Hodjat P, Christensen PA, Subedi S, Bernard DW, Olsen RJ, Long SW. The reemergence of seasonal respiratory viruses in Houston, Texas, after relaxing COVID-19 restrictions. Microbiol Spectr. 2021;9:e0043021. doi: 10.1128/Spectrum.00430-21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Delestrain C, Danis K, Hau I, Behillil S, Billard MN, Krajten L, et al. Impact of COVID-19 social distancing on viral infection in France: a delayed outbreak of RSV. Pediatr Pulmonol. 2021;56:3669–3673. doi: 10.1002/ppul.25644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Di Mattia G, Nenna R, Mancino E, Rizzo V, Pierangeli A, Villani A, et al. During the COVID-19 pandemic where has respiratory syncytial virus gone? Pediatr Pulmonol. 2021;56:3106–3109. doi: 10.1002/ppul.25582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Khemiri H, Ayouni K, Triki H, Haddad-Boubaker S. SARS-CoV-2 infection in pediatric population before and during the Delta (B.1.617.2) and Omicron (B.1.1.529) variants era. Virol J. 2022;19:144. doi: 10.1186/s12985-022-01873-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Agathis NT, Milucky J, Taylor CA, Whitaker M, Pham H, Anglin O, Chai SJ, Alden NB, Meek J, Anderson EJ, Weigel A, Kim S, Lynfield R, Smelser C, Muse A, Popham K, Billing LM, Sutton M, Talbot HK, George A, McMorrow M, Havers FP CDC COVID-NET Surveillance Team. Codetections of Other Respiratory Viruses Among Children Hospitalized With COVID-19. Pediatrics 2023;151:e2022059037. 10.1542/peds2022-059037.2023 [DOI] [PubMed]
- 18.Quidelhttps://www.quidel.com/cultures-fluorescent-tests/rsv
- 19.Biofire BIOFIRE® Respiratory 2.1 plus Panelhttps://www.biomerieux-diagnostics.com/filmarrayr-respiratory-panel
- 20.Seegenehttps://www.seegene.com/assays/rp
- 21.R Core Team (2021) R: A language and environment for statistical computing. R Foundation for Statistical Computing V, Austria. https://www.R-project.org
- 22.Burbank AJ. Risk factors for respiratory viral infections: a spotlight on climate change and air pollution. J Asthma Allergy. 2023;16:183–194. doi: 10.2147/JAA.S364845. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Mirsaeidi M, Motahari H, Taghizadeh Khamesi M, Sharifi A, Campos M, Schraufnagel DE. Climate change and respiratory infections. Ann Am Thorac Soc. 2016;13:1223–1230. doi: 10.1513/AnnalsATS.201511-729PS. [DOI] [PubMed] [Google Scholar]
- 24.Baker RE, Park SW, Yang W, Vecchi GA, Metcalf CJE, Grenfell BT. The impact of COVID-19 nonpharmaceutical interventions on the future dynamics of endemic infections. Proc Natl Acad Sci. 2020;117:30547–30553. doi: 10.1073/pnas.2013182117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Engels G, Sack J, Weissbrich B, Hartmann K, Knies K, Hartel C, et al. Very low incidence of SARS-CoV-2, influenza and RSV but high incidence of Rhino-, Adeno- and endemic coronaviruses in children with acute respiratory infection in primary care pediatric practices during the second and third wave of the SARS-CoV-2 pandemic. Pediatr Infect Dis J. 2022;41:e146–e148. doi: 10.1097/INF.0000000000003460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Buchholz U, Lehfeld AS, Tolksdorf K, Cai W, Reiche J, Biere B, et al. Respiratory infections in children and adolescents in Germany during the COVID-19 pandemic. J Health Monit. 2023;8:20–38. doi: 10.25646/11437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Perez A, Lively JY, Curns A, Weinberg GA, Halasa NB, Staat MA, et al. Respiratory virus surveillance among children with acute respiratory illnesses - new vaccine surveillance network, United States, 2016–2021. MMWR Morb Mortal Wkly Rep. 2022;71:1253–1259. doi: 10.15585/mmwr.mm7140a1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Falsey AR, Walsh EE, Esser MT, Shoemaker K, Yu L, Griffin MP. Respiratory syncytial virus-associated illness in adults with advanced chronic obstructive pulmonary disease and/or congestive heart failure. J Med Virol. 2019;91:65–71. doi: 10.1002/jmv.25285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Linden D, Guo-Parke H, Coyle PV, Fairley D, McAuley DF, Taggart CC, et al. Respiratory viral infection: a potential "missing link" in the pathogenesis of COPD. Eur Respir Rev. 2019;28:180063. doi: 10.1183/16000617.0063-2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Nickbakhsh S, Mair C, Matthews L, Reeve R, Johnson PCD, Thorburn F, et al. Virus–virus interactions impact the population dynamics of influenza and the common cold. Proc Natl Acad Sci. 2019;116:27142–27150. doi: 10.1073/pnas.1911083116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Piret J, Boivin G. Viral interference between respiratory viruses. Emerg Infect Dis. 2022;28:273–281. doi: 10.3201/eid2802.211727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Greer RM, McErlean P, Arden KE, Faux CE, Nitsche A, Lambert SB, et al. Do rhinoviruses reduce the probability of viral co-detection during acute respiratory tract infections? J Clin Virol. 2009;45:10–15. doi: 10.1016/j.jcv.2009.03.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Dee K, Goldfarb DM, Haney J, Amat JAR, Herder V, Stewart M, et al. Human rhinovirus infection blocks severe acute respiratory syndrome coronavirus 2 replication within the respiratory epithelium: implications for COVID-19 epidemiology. J Infect Dis. 2021;224:31–38. doi: 10.1093/infdis/jiab147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Rocafort M, Henares D, Brotons P, Launes C, Fernandez de Sevilla M, Fumado V, et al. Impact of COVID-19 lockdown on the nasopharyngeal microbiota of children and adults self-confined at home. Viruses. 2022;14:1521. [DOI] [PMC free article] [PubMed]
- 35.Bosch A, de Steenhuijsen Piters WAA, van Houten MA, Chu M, Biesbroek G, Kool J, et al. Maturation of the infant respiratory microbiota, environmental drivers, and health consequences. A prospective cohort study. Am J Respir Crit Care Med. 2017;196:1582–90. doi: 10.1164/rccm.201703-0554OC. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The data of this ongoing study are currently not available for public use due to data protection reasons.