Abstract
Acrophialophora fusispora is a thermotolerant soil fungus which is very unusual in clinical samples. Here we report four cases of transient or chronic airway colonization by A. fusispora in patients with cystic fibrosis (CF). However, the prevalence of this fungus in CF patients may be underestimated due to the currently poor knowledge of this fungus in most of the medical mycology laboratories. In addition, its clinical importance regarding CF remains to be evaluated.
CASE REPORTS
Case 1.
A 5-year-old Guadeloupean boy whose cystic fibrosis (CF) was diagnosed by a positive sweat test at the age of two (Table 1) was monitored in the Pediatric Department of Necker Hospital (Paris, France) from January 2001 to July 2002. He had a history of recurrent bronchopulmonary infections, caused at first by Staphylococcus aureus and then by Pseudomonas aeruginosa, that required several admissions to the hospital for antibiotic and respiratory therapy. Aspergillus fumigatus was first isolated from bronchial secretions in April 2001, but it was not recovered in the next sputum sample 1 month later. At this time, cultures yielded another fungus with buff colonies reaching a diameter of 5 to 6 cm after 7 days at 37°C on Sabouraud dextrose agar. Initially considered Scedosporium prolificans, the fungus was referred to the Scientific Institute of Public Health (Brussels, Belgium) where it was identified as Acrophialophora fusispora. This fungus was then recovered from another sputum specimen in October 2001 with no evidence of respiratory infection.
TABLE 1.
Clinical data from CF patients colonized by A. fusispora
| Patient characteristic | Data for patient (hospital location in France)
|
|||
|---|---|---|---|---|
| 1 (Paris) | 2 (Paris) | 3 (Paris) | 4 (Giens) | |
| Date of birtha (sexb) | 01/26/1996 (M) | 10/16/1989 (M) | 02/12/1998 (M) | 10/06/1973 (F) |
| Yr of CF diagnosis | 1998 | 1990 | 1999 | 1996 |
| Relevant gene mutation | Unknown | ΔF508/ΔF508 | ΔF508/ΔF508 | 574 del A/unknown |
| Date of first isolation of: | ||||
| A. fumigatus | April 2001 | March 2000 | February 2002 | February 1998 |
| A. fusispora | May 2001 | September 2001 | August 2002 | April 1999 |
| No. of successive samples positive for A. fusispora | 2 | 1 | 4 | 1 |
| Antifungal treatment (duration) | None | Itraconazole (January 2001-June 2002) | None | Itraconazole (June-August 1999) |
Month/day/year.
M, male; F, female.
Case 2.
A 13-year-old boy living in an urban area of Paris was diagnosed with CF at the age of one (Table 1). The respiratory tract of this patient was initially infected by S. aureus and then, from December 1995, by P. aeruginosa, resulting in numerous bronchitis episodes treated with antibiotics and respiratory therapy. A. fumigatus was first isolated in March 2000, and it was then regularly recovered from the bronchial secretions of the patient. In January 2001, an acute-phase infection was diagnosed, with bilateral bronchiectasis shown by a computed tomographic scan of the thorax. At this time, A. fumigatus was isolated from sputum samples and related serum antibodies were detected, with one precipitin line, by counterimmunoelectrophoresis. Complement studies showed a high titer of total serum immunoglobulin E (IgE) (432 kU/liter) and the presence of anti-A. fumigatus IgE (grade 3 by a radioallergosorbent test). Based on these criteria, treatment with itraconazole was started, consisting initially of daily doses of 10 mg/kg of body weight. In March 2001, the persistence of A. fumigatus in the airways led to the increase of the itraconazole dose to 15 mg/kg/day for 15 months. Despite this treatment, A. fumigatus was again isolated from a sputum sample collected in September 2001 concomitantly with the isolation of a brown mold that was sent to the Scientific Institute of Public Health, where the mold was identified as A. fusispora. No deterioration of clinical and scanographic signs was seen at this time, and A. fusispora was not recovered again from this patient. A culture of a new sputum sample collected in December 2001 yielded A. fumigatus exclusively.
Case 3.
A 4-year-old boy living in a suburban area of Paris was being monitored for CF at Necker hospital (Table 1). From the age of one, he presented with recurrent bronchopulmonary infections caused by S. aureus, Haemophilus influenzae, or P. aeruginosa. Airway colonization by A. fumigatus emerged in February 2002. During the following 6 months, it was isolated from three other sputum samples, without any biological signs of sensitization to A. fumigatus (precipitins or specific IgE). Since his clinical status remained unchanged, the patient did not receive any antifungal treatment. In August 2002, a culture of one sputum sample grew A. fumigatus and a brown mold which was identified in Brussels as A. fusispora. A. fusispora continued to be isolated until November 2002. During that period, there were three isolations out of three sputum cultures: one case of the mold in conjunction with A. fumigatus and two cases of the mold by itself. No bronchopulmonary episodes were observed at the time of airway colonization by A. fusispora.
Case 4.
A 26-year-old woman living in Corsica, France, had been monitored since 1995 at Renée Sabran Hospital in Giens in southern France (Table 1). The diagnosis of CF was established by sweat chloride determination in 1996 when the patient was age 23. She had a history of chronic sinusitis and recurrent bronchitis, with bronchiectasis in the right lower lobe that required a lobectomy in 1990. The patient was chronically colonized by P. aeruginosa when A. fumigatus was isolated from bronchial secretions in February 1998. In April 1999, a sputum culture once again yielded A. fumigatus in conjunction with A. fusispora. The patient received 3 months of treatment with itraconazole (300 mg per day). In August 1999, no fungus was isolated from sputum samples, and A. fusispora was not cultured again.
Discussion.
In CF patients, mutations in the gene encoding the CF transmembrane conductance regulator protein lead to abnormal airway conditions suitable for the growth of microorganisms (13). Bacteria such as S. aureus, P. aeruginosa, and Burkholderia cepacia are typical CF pathogens that cause recurrent exacerbations of pulmonary disease and often contribute to the mortality of the patients (10). Filamentous fungi are frequently isolated from bronchial secretions at a more advanced stage of the disease (3). A. fumigatus and, to a lesser extent, Scedosporium apiospermum, Aspergillus terreus, and Exophiala dermatitidis, inducing transient or chronic colonization of the respiratory tract, allergic bronchopulmonary disease, or pneumonia, are the main fungal species associated with CF (5, 12, 14).
A. fusispora is a soil fungus rarely isolated from human sources (7). A unique case of transient airway colonization by A. fusispora in an 11-year-old CF patient without apparent clinical symptoms has been previously reported (11). Likewise, the isolation of the fungus from single sputum samples from two of our patients could be considered a transient occurrence of fungal spores trapped in the abnormally thickened viscous mucus. In the two other cases, A. fusispora was repeatedly isolated from sputum samples for a few months, suggesting its continuous presence in the respiratory tract during this period.
We previously reported a case in which Penicillium emersonii (the anamorph state of Talaromyces emersonii), another fungus atypical in humans, chronically colonized the airways of a CF patient (4) in the same way that A. fusispora did. Both A. fusispora and P. emersonii are soil saprophytic fungi with thermophilic behaviors (9), which could explain their ability to colonize the respiratory tracts of the patients. Some host factors, however, are certainly associated in their pathophysiologies, particularly the dehydration of respiratory secretions and the defective mucociliary clearance that characterize pulmonary disease associated with CF. Likewise, the bacterial flora isolated from CF patients also comprises very atypical species that are rarely seen in clinical samples and whose pathogenic role in CF lung disease is still unclear (6).
Although the clinical statuses of the patients remained apparently unchanged, with no evidence of infectious symptoms at the times of colonization, the literature clearly establishes A. fusispora as a pathogen. This fungus has been reported as an etiologic agent of keratitis (2, 15) and pulmonary infection (D. A. Sutton, L. Sigler, K. G. Kalassian, A. W. Fothergill, and M. G. Rinaldi, Abstr. 13th Int. Soc. Hum. Anim. Mycol. Congr., abstr. 363, 1997) in nonimmunocompromised adults and of a brain abscess in a child with acute lymphoblastic leukemia (1). In addition, A. fusispora was also responsible for a multisystemic infection in a dog (16). Fungi, being opportunists, should never be considered devoid of clinical significance; in CF patients, especially with cases of long-term colonization, the fungus could contribute to the chronic inflammatory reaction which leads to slowly progressing pulmonary damage.
Another issue with our study is the challenge that this unusual fungus poses to medical mycology laboratories. Indeed, A. fusispora has been previously misidentified as Scopulariopsis chartarum (1, 16) or, as with patient 1, S. prolificans (J. Guarro and J. Gené, Letter, J. Clin. Microbiol. 40:3544, 2002; L. Sigler, and D. A. Sutton, Letter, J. Clin. Microbiol. 40:3544-3545, 2002). Likewise, in the case reported by Al-Mohsen et al. (1), the causative agent of the brain abscess was initially identified as Paecilomyces species. These regular misidentifications suggest that this fungus may be unrecognized by microbiologists. Regarding CF, the possibility that misdeterminations have been made in the past, in particular with S. prolificans, a fungus sometimes reported in this context, could not be excluded (8).
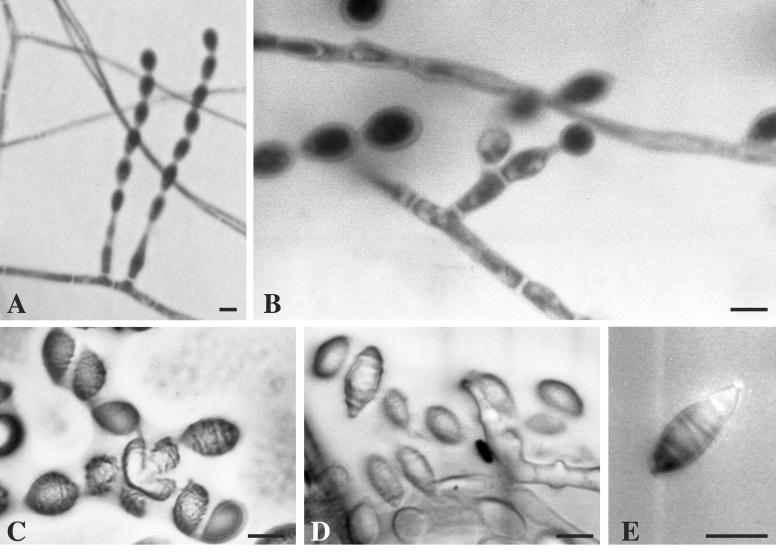
A. fusispora grows rapidly on Sabouraud dextrose agar and is characterized by colonies that are usually greyish brown on the top and dark brown on the bottom (7). However, great variations in the colonial features of this fungus have been observed, including variation in isolate pigmentation from light orange to pale gray (1), leading to difficulties in the macroscopic step of mycological identification. Therefore, all our isolates were identified on the basis of the following features: they were thermotolerant, showing good growth at 42°C; microscopically, they exhibited pale brown mycelia, 1.5 to 3.5 μm in diameter, and basally inflated phialides with elongated necks, arising mostly singly on vegetative hyphae (Fig. 1A) or along the length and near the tip of pale brown echinulate conidiophores (Fig. 1B); and they showed long chains of limoniform-to-fusiform single-celled conidia that were pale brown, 6 to 10 × 3.5 to × 5.0 μm in size, and smooth or finely echinulate with ornamentations often arranged in distinct spiral bands (Fig. 1C to E). A. fusispora resembles S. prolificans in that it forms flask-shaped conidiogenous cells, but it may be distinguished from the latter fungus by its phialides arising singly on vegetative hyphae or on conidiophores and by its production of conidia in chains. Paecilomyces spp. also produce chains of conidia, but they arise from whorls of phialides. In addition, conidia are sometimes echinulate but do not have arrangements of the ornamentations in spiral bands (7).
FIG. 1.
Microscopic morphology of A. fusispora. (A) Long chains of conidia produced by flask-shaped conidiogenous cells arising on vegetative hyphae. (B) A basally inflated phialide with an elongated neck arising on a short conidiophore. (C to E) Limoniform-to-fusiform single-celled conidia with ornamentations in spiral bands visualized by interferential phase-contrast microscopy. Bars, 5 μm.
Finally, in addition to previous reports about the isolation of very unusual bacteria and fungi in CF patients (4, 6), the present observations reinforce the conclusion that the respiratory tracts of CF patients are an ecological niche suitable for the growth of a wide variety of microorganisms not commonly encountered in humans.
Acknowledgments
We express our gratitude to I. Sermet (Service de Pédiatrie Générale, Centre Hospitalier Universitaire Necker, Paris, France), M. Le Bourgeois (Service de Pneumologie Pédiatrique, Centre Hospitalier Universitaire Necker, Paris, France), and J. P. Chazalette (Hôpital Renée Sabran de Giens, Hyères France) for their cooperation in this study.
REFERENCES
- 1.Al-Mohsen, I. Z., D. A. Sutton, L. Sigler, E. Almodovar, N. Mahgoub, H. Frayha, S. Al-Hajjar, M. G. Rinaldi, and T. J. Walsh. 2000. Acrophialophora fusispora brain abscess in a child with acute lymphoblastic leukemia: review of cases and taxonomy. J. Clin. Microbiol. 38:4569-4576. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Arthur, S., L. L. Steed, D. J. Apple, Q. Peng, G. Howard, and M. Escobar-Gomez. 2001. Scedosporium prolificans keratouveitis in association with a contact lens retained intraocularly over a long term. J. Clin. Microbiol. 39:4579-4582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Cimon, B., J. Carrère, J. P. Chazalette, J. L. Giniès, P. Six, J. F. Vinatier, D. Chabasse, and J. P. Bouchara. 1995. Fungal colonization and immune response to fungi in cystic fibrosis. J. Mycol. Med. 5:211-216. [Google Scholar]
- 4.Cimon, B., J. Carrère, J. P. Chazalette, J. F. Vinatier, D. Chabasse, and J. P. Bouchara. 1999. Chronic airway colonization by Penicillium emersonii in a patient with cystic fibrosis. Med. Mycol. 37:291-293. [PubMed] [Google Scholar]
- 5.Cimon, B., J. Carrère, J. F. Vinatier, J. P. Chazalette, D. Chabasse, and J. P. Bouchara. 2000. Clinical significance of Scedosporium apiospermum in patients with cystic fibrosis. Eur. J. Clin. Microbiol. Infect. Dis. 19:53-56. [DOI] [PubMed] [Google Scholar]
- 6.Coenye, T., J. Goris, T. Spilker, P. Vandamme, and J. J. LiPuma. 2002. Characterization of unusual bacteria isolated from respiratory secretions of cystic fibrosis patients and description of Inquilinus limosus gen. nov., sp. nov. J. Clin. Microbiol. 40:2062-2069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.De Hoog, G. S., J. Guarro, J. Gené, and M. J. Figueras. 2000. Atlas of clinical fungi. Centraalbureau voor Schimmelcultures, Utrecht, The Netherlands.
- 8.Del Palacio, A., M. Garau, E. Amor, I. Martinez-Alonso, T. Calvo, A. Carrillo-Munoz, and J. Guarro. 2001. Transient colonization with Scedosporium prolificans. Report of four cases in Madrid. Mycoses 44:321-325. [PubMed] [Google Scholar]
- 9.Domsch, K. H., W. Gams, and T.-H. Anderson. 1993. Compendium of soil fungi. IHW-Verlag, Eching, Germany.
- 10.Gilligan, P. H. 1991. Microbiology of airway disease in patients with cystic fibrosis. Clin. Microbiol. Rev. 4:35-51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.González-Escalada, A., A. del Palacio, M. T. Calvo, J. Gené, and J. Guarro. 2000. A propósito de dos casos de colonización por hongos filamentosos en secreciones respiratorias y en herida traumática de cuero cabelludo. Rev. Iberoam. Micol. 17:149-151. [PubMed] [Google Scholar]
- 12.Kusenbach, G., H. Skopnik, G. Haase, F. Friedrichs, and H. Dôhmen. 1992. Exophiala dermatitidis pneumonia in cystic fibrosis. Eur. J. Pediatr. 151:344-346. [DOI] [PubMed] [Google Scholar]
- 13.Moss, R. B. 1995. Cystic fibrosis: pathogenesis, pulmonary infection and treatment. Clin. Infect. Dis. 21:839-851.8645828 [Google Scholar]
- 14.Schwartz, H. J. 1998. The association of allergic bronchopulmonary aspergillosis and cystic fibrosis. Immunol. Allergy Clin. N. Am. 18:503-508. [Google Scholar]
- 15.Shukla, P. K. 1983. Clinical and experimental keratitis caused by the Colletotrichum state of Glomerella cingulata and Acrophialophora fusispora. Sabouraudia 21:137-147. [PubMed] [Google Scholar]
- 16.Welsh, R. D., and R. W. Ely. 1999. Scopulariopsis chartarum systemic mycosis in a dog. J. Clin. Microbiol. 37:2102-2103. [DOI] [PMC free article] [PubMed] [Google Scholar]