Abstract
By immunoelectron microscopy with a polyclonal antibody against the cytosolic glycolytic enzyme glyceraldehyde-3-phosphate dehydrogenase (GAPDH) from Candida albicans (anti-GAPDH PAb), the protein was clearly detected at the outer surface of the cell wall, particularly on blastoconidia, as well as in the cytoplasm. Intact blastoconidia were able to adhere to fibronectin and laminin immobilized on microtiter plates, and this adhesion was markedly reduced by both the anti-GAPDH PAb and soluble GAPDH from Saccharomyces cerevisiae. In addition, semiquantitative flow cytometry analysis with the anti-GAPDH PAb showed a decrease in antibody binding to cells in the presence of soluble fibronectin and laminin. Purified cytosolic C. albicans GAPDH was found to bind to fibronectin and laminin in a ligand Western blot assay. These observations suggest that the cell wall-associated form of the GAPDH in C. albicans could be involved in mediating adhesion of fungal cells to fibronectin and laminin, thus contributing to the attachment of the microorganism to host tissues and to the dissemination of Candida infection.
The dimorphic fungus Candida albicans is both a commensal and opportunistic pathogen of humans. The fungus is carried commensally by up to half of healthy individuals; however, it is also a common pathogen of the mucosal epithelial tissues of the oral and urogenital tracts (37). In the immunocompromised host, it can cause deep-seated and systemic infections that could prove fatal (3, 37). The lack of an early and effective diagnostic procedure and the toxicity displayed by the most commonly used drugs to treat infection contribute to the high mortality rates observed with this type of systemic infection (3, 33).
Adhesion of C. albicans to host tissues seems to be an essential factor for the establishment of candidiasis. Attachment may involve binding between complementary molecules on both host and parasite cell surfaces. The establishment of metastatic sites of infection throughout the body in disseminated candidiasis presumably occurs following yeast adherence to the endothelial basement membrane and/or subendothelial extracellular matrix (ECM). Adherence to ECM components therefore represents a crucial step in the development of candidiasis. The ability of C. albicans strains to bind ECM proteins correlates with the rank order of their relative pathogenicity, suggesting that adherence to ECM components is a significant virulence factor (6, 11, 24). C. albicans is known to bind different ECM proteins such as fibronectin, laminin, entactin, and collagens, and these proteins are implicated as possible target molecules when Candida dissemination occurs (5, 10, 14, 20, 24, 41). Although a number of molecules with receptor-like characteristics implicated in fibronectin and laminin binding have been described for C. albicans (4, 15, 25, 26, 30, 36, 45, 46), the molecular mechanisms involved in these adhesive interactions remain basically undefined. The understanding of the mechanisms mediating C. albicans adherence to the ECM or host cells could lead to development of antifungal agents whose mechanisms of action would be to compete with the endogenous ligands for binding to the pathogen receptors or adhesins. These inhibitors may prevent adhesion to host tissues and thereby prevent invasive infections.
In a previous study (16), we screened a C. albicans cDNA library for sequences that encode immunogenic proteins by using pooled sera from patients with a high level of anti-C. albicans antibodies, in order to identify antigens potentially useful for diagnosis of candidiasis or that may play a role in infection. Using this approach, we isolated the glyceraldehyde-3-phosphate dehydrogenase (GAPDH) whole gene and demonstrated that in addition to its cytoplasmic localization, an immunogenic, enzymatically active cell wall-associated form of the glycolytic enzyme is found at the cell surface of C. albicans. The glycolytic enzyme GAPDH has also been identified on the surface of other organisms, such as Schistosoma mansoni (18), group A streptococci (38, 39, 55) and Kluyveromyces marxianus (12, 13).
In the present paper, we report the location of the GAPDH in the cell wall of C. albicans yeast and mycelial cells by immunoelectron microscopy. We have also demonstrated that the cell wall-associated GAPDH is able to bind to fibronectin and laminin. The ability to bind to ECM proteins exhibited by the surface C. albicans GAPDH suggested that this protein may play a role in mediating attachment of the fungus to host tissues, thus playing a role in the establishment of the disease.
MATERIALS AND METHODS
Microorganism and growth conditions.
C. albicans ATCC 26555 was employed in this study. It was maintained by subculturing on 1.5% Bacto Agar slants of Sabouraud dextrose medium. Cells were propagated as blastoconidia at 28°C in a minimal (Lee) medium supplemented with amino acids (29), harvested, and stored at 4°C for 72 to 96 h in sterile water (starvation period) as reported previously (7, 9). Starved blastoconidia were inoculated (200 μg [dry weight] of cells per ml) in fresh Lee medium at 28°C to obtain cultures of blastoconidia or at 37°C for the formation of blastoconidia bearing germ tubes (7).
Immunoelectron microscopy.
Cells were fixed in 0.5% glutaraldehyde–4% formaldehyde for 120 min at room temperature and washed in 0.5 M NH4Cl for 60 min. For preembedding analysis, cells were incubated for 60 min at 37°C with the polyclonal antibody (PAb) against C. albicans GAPDH (anti-GAPDH PAb) diluted (1:200) in 20 mM Tris-HCl buffer (pH 7.4), containing 0.1% bovine serum albumin (BSA) and 0.9% NaCl (buffer A) supplemented with 1% inactivated fetal calf serum (buffer B). The cells were then washed with buffer A and incubated for 60 min at 37°C in buffer B containing 0.5% Tween 20 and a goat anti-rabbit immunoglobulin G (IgG)-gold complex (average size particle, 10 nm; 1:10 dilution). After washing in buffer A, the cells were embedded in Lowicryl K4M (43). For postembedding immunocytochemistry (35, 44), the cells were fixed as described above and processed for Lowicryl embedding. Ultrathin sections mounted on Formvar-carbon-coated nickel grids were floated on droplets containing the same solutions described for the preembedding procedure. After washing, ultrathin sections from both preembedding and postembedding assays were stained with 2% uranyl acetate and examined with a Philips EM 301 electron microscope.
Adherence assays on microtiter plates.
Wells of microtitration plates (Nunc-Immunoplate Y [A/S Nunc]) were coated with 200 μl of fibronectin or laminin solutions (50 μg/ml) in phosphate-buffered saline (PBS). Adherence of biotinylated, Extravidin-peroxidase-labelled blastoconidia to fibronectin and laminin immobilized on the wells was assessed by using the experimental protocol previously reported (40) except that 106 cells were added to each well and that surface biotinylation of blastoconidia was performed as described by Casanova et al. (8). After incubation of the wells with a chromogenic reagent (o-phenylenediamine), the intensity of the colored reaction was determined at 492 nm with an automated plate reader (Labsystems Multiskan MCC/340). Results, expressed as the optical density at 492 nm, are the means for triplicate wells with standard deviations. Statistical analysis of data was performed by means of Dunnett’s t test for multiple comparisons.
Immunofluorescence and flow cytometry analysis.
An immunofluorescence assay with the anti-C. albicans GAPDH PAb was performed by the procedure described previously (16). Cells were subsequently fixed in 1% paraformaldehyde solution in PBS and analyzed by flow cytometry. Flow cytometry analyses were performed on an EPICS Elite cell sorter (Coulter Electronics Inc., Hialeah, Fla.), as previously described (40). Duplicate samples were processed in the absence of anti-GAPDH PAb as negative controls. Immunofluorescence reactions were also determined in the presence of soluble fibronectin or laminin (final concentrations, 50 and 100 μg/ml).
Immunoaffinity purification of GAPDH protein.
Anti-C. albicans GAPDH PAbs were purified from a rabbit antiserum, obtained as previously described (16), by precipitation with 50% ammonium sulfate, followed by affinity chromatography on protein A-Sepharose (Pharmacia). The purified immunoglobulins were then coupled to cyanogen bromide-activated Sepharose 4B (Pharmacia) according to the manufacturer’s guideline. The anti-GAPDH PAb–Sepharose affinity column was used to purify the GAPDH protein from a cytosolic C. albicans extract prepared by stirring protoplasts in lysis buffer (10 mM phosphate buffer [pH 7.4] containing 1 mM CaCl2, 1 mM MgCl2, 1 mM phenylmethylsulfonyl fluoride, and 0.1% sodium dodecyl sulfate [SDS]) as described before (8). The lysate was clarified by centrifugation (12,000 × g, 30 min) and then recirculated over the column previously equilibrated in lysis buffer. After extensive washing, bound proteins were eluted by a pH shift with 10 mM glycine buffer (pH 2.5). Fractions were collected, brought to neutral pH, and assayed for protein content (31); purity was determined by SDS-polyacrylamide gel electrophoresis (SDS-PAGE) (see below). The fractions containing the purified protein were pooled, freeze-dried, and dissolved and dialyzed against PBS. The enzymatic activity of the purified GAPDH was assayed as previously described (16).
SDS-PAGE and Western blotting techniques.
SDS-PAGE was performed basically as described by Laemmli (28) with minor modifications (7, 9). Electrophoretic transfer (Western blotting) to polyvinylidene difluoride (PVDF) membranes (Millipore) was carried out as described previously (7, 9, 16). Blotted proteins were assayed for fibronectin or laminin binding as follows. The PVDF membranes were incubated with 3% BSA in 10 mM Tris-HCl buffer (pH 7.4) containing 0.9% NaCl (TBS buffer) for 1 h at room temperature and then for 6 h in PBS containing fibronectin (80 μg/ml) or laminin (80 μg/ml). After washing (four times, 10 min each time) with TBS buffer containing 0.05% Tween 20 (TBST buffer), the PVDF sheets were incubated for 1 h with either rabbit antifibronectin antibody (Ab; 1:1,000 dilution) or rabbit antilaminin Ab (1:1,000 dilution) in TBST plus 1% BSA. The blots were washed with TBST and incubated with peroxidase-labelled goat anti-rabbit immunoglobulin (1:2,000 dilution in TBST plus 1% BSA). Finally, the blots were washed again, and reactive bands were developed with hydrogen peroxide and 4-chloro-1-naphthol as the chromogenic reagent.
Miscellaneous.
Human fibronectin and mouse laminin (isolated from a mouse Englebreth-Holm-Swarm sarcoma tumor) were obtained from Boehringer Mannheim. GAPDH from S. cerevisiae, rabbit antifibronectin and antilaminin Abs, fluorescein-conjugated goat anti-rabbit immunoglobulin Abs, and peroxidase-conjugated goat anti-rabbit immunoglobulin Abs were from Sigma Chemical Co. Rabbit anti-α-amylase PAbs from the yeast Lypomyces kononenkoae (43) were used as irrelevant Abs in adherence assays.
RESULTS
Detection of the GAPDH protein by immunoelectron microscopy.
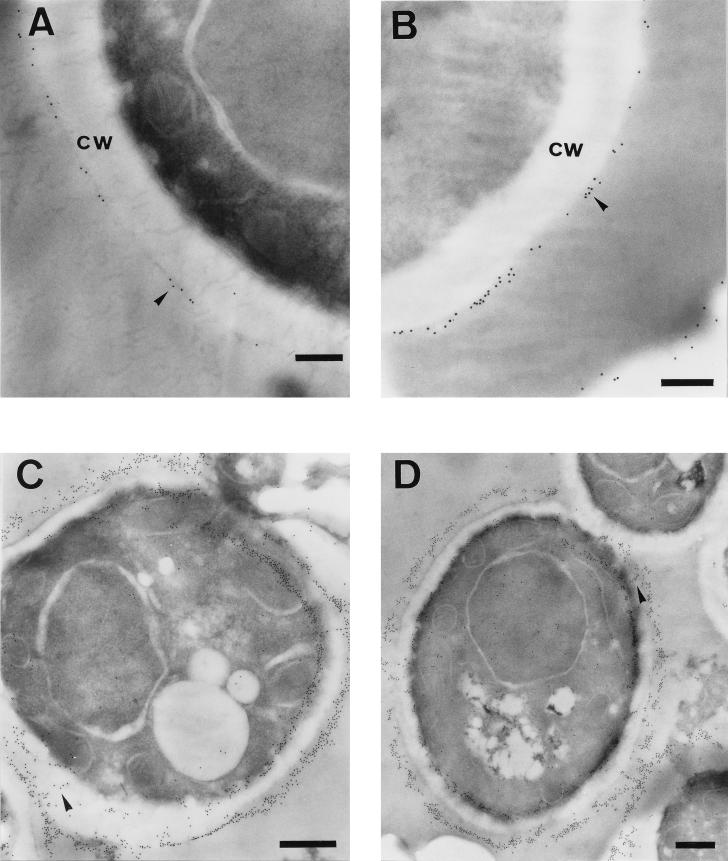
Since we have described that GAPDH is a C. albicans surface antigen (16), both intact yeast and mycelial cells were examined by immunoelectron microscopy with the anti-C. albicans GAPDH PAb to show ultrastructural evidence of cell wall localization of the GAPDH protein. In blastoconidial cells processed by the preembedding method, gold particles showed a patchy distribution over the outermost layer of the cell surface (Fig. 1A and B). Gold particles were also present at the outermost layer of the cell wall when thin sections of blastoconidial cells processed by the postembedding method were observed, although in this case the particles were also detected extending through the cell wall (Fig. 1C and D). The particles were also found distributed all over the cytoplasm, mainly located close to the plasma membrane, but not associated with any other cytoplasmic organelle (Fig. 1C and D).
FIG. 1.
Immunoelectron microscopy detection of GAPDH protein in C. albicans yeast cells by the preembedding (A and B) and postembedding (C and D) methods. The labelling was detected at the outermost layer of the cell wall (cw) in a patchy distribution (A and B, arrowheads). The antigen also appears extending through the cell wall (C and D, arrowheads). The label was also detected in the cytoplasm (C and D). Bars, 0.25 μm (A and B) and 0.5 μm (C and D).
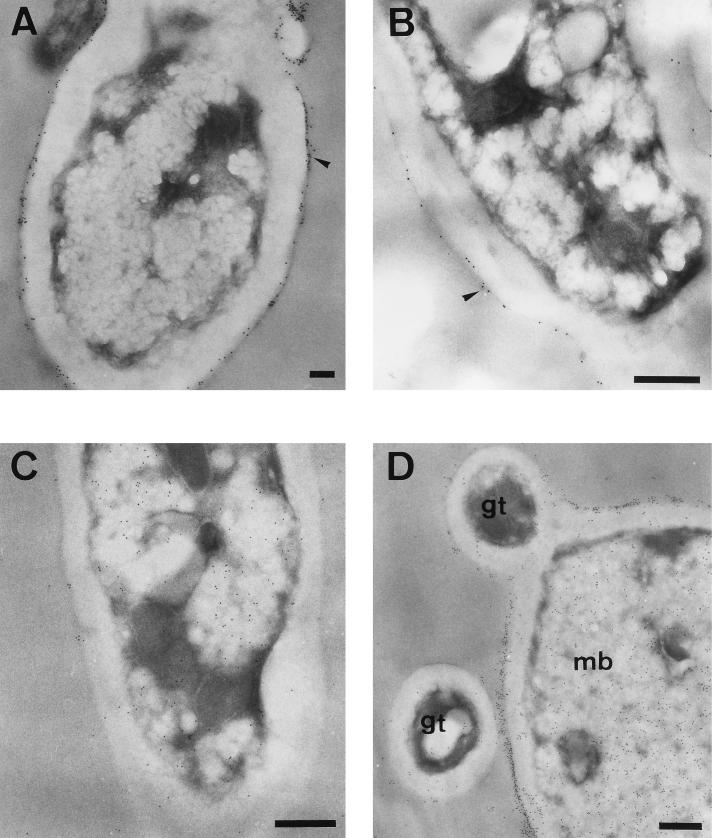
In germinated blastoconidia processed by the preembedding method, the moiety reacting with the Ab was present at the cell surface in a patchy distribution (Fig. 2A and B) as observed for blastoconidial cells. After embedding, a specific immunolabelling was detected in the cytoplasm and in the cell walls of germinated cells (Fig. 2C and D), similar to that observed in blastoconidia. Interestingly, as shown in Fig. 2D, mother blastoconidia from which germ tubes originate were more heavily labelled than hyphal filaments. Moreover, labelling intensity varied in different cells, both in the cytoplasm and in the cell wall. A similar pattern of GAPDH distribution on blastoconidial and hyphal cell surface was previously observed by immunofluorescence (16). Control samples not exposed to anti-GAPDH PAb prior to incubation with the gold-conjugated Ab were free of label (data not shown).
FIG. 2.
Immunoelectron microscopy detection of GAPDH protein in C. albicans germinated cells by the preembedding (A and B) and postembedding (C and D) methods. The labelling was present at the outermost layer of the cell wall in a patchy distribution (A and B, arrowheads). It is important to note that mother blastoconidia (mb) were more heavily labelled than a cross-section of a germ tube (gt) (D). Bars, 0.5 μm.
Role of GAPDH in the attachment of C. albicans yeast cells to fibronectin and laminin.
C. albicans has been shown to bind to fibronectin and laminin (4, 22, 23, 27, 30, 36, 42, 49), although the molecular basis of this interaction has not yet been clearly defined. On the other hand, the GAPDH on the surface of Streptococcus pyogenes displays multiple binding activities for host ligands (fibronectin, lysozyme, actin, and myosin) (38). In an attempt to investigate whether the surface-located C. albicans GAPDH could be involved in the ability of cells to adhere to host tissues, an in vitro assay was developed to study the adherence of Candida cells to fibronectin and laminin.
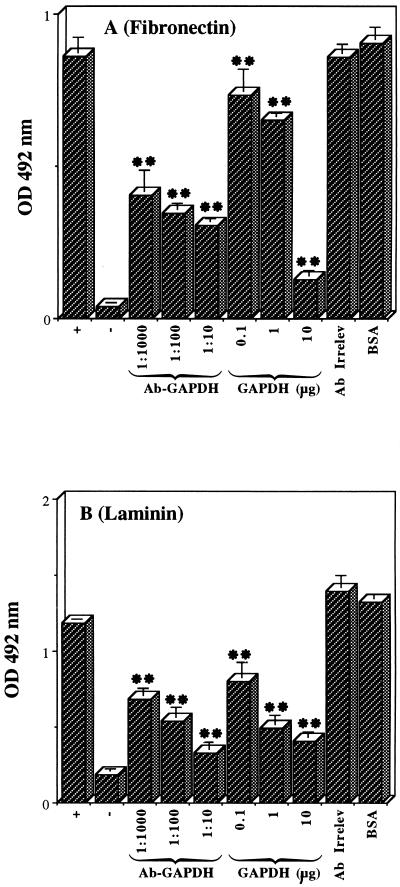
As shown in Fig. 3, biotinylated, Extravidin-peroxidase-labelled blastoconidia adhered to immobilized fibronectin (Fig. 3A) and laminin (Fig. 3B). Coating wells of microtiter plates with protein solutions of increasing concentrations resulted in an increase in the number of adherent cells (data not shown). Maximal adhesion was found at a coating fibronectin or laminin concentration of 50 μg/ml. Hence, this concentration was chosen for the subsequent experiments. Background attachment to plastic did not exceed 16% of control adhesion to fibronectin or laminin (Fig. 3, negative controls).
FIG. 3.
Attachment of blastoconidia to immobilized fibronectin (A) and laminin (B). Biotin-Extravidin-peroxidase-labelled cells were allowed to adhere to uncoated wells (negative control) or to wells coated with fibronectin or laminin without any inhibitor (positive control) or in the presence of (i) anti-GAPDH PAb (Ab-GAPDH) at 1:1,000, 1:100, and 1:10 dilutions, (ii) 0.1, 1, and 10 μg of GAPDH from S. cerevisiae, (iii) rabbit anti-α-amylase PAb (1:10 dilution) as irrelevant antibody (Ab Irrelev), or (iv) 10 μg of BSA. The amount of adherent cells was estimated indirectly by measuring the optical density (OD) at 492 nm of the colored reaction produced by the peroxidase reaction (see Materials and Methods). The results represent triplicate determinations and are expressed as means ± standard deviations. ∗∗, P < 0.01.
Anti-C. albicans GAPDH PAb inhibited the adhesion of yeast cells to fibronectin and laminin in a dose-dependent manner (Fig. 3). Dilutions of 1:1,000, 1:100, and 1:10 of the Ab caused 53, 60, and 65% inhibition, respectively, of the adhesion to immobilized fibronectin and 42, 55, and 72% inhibition, respectively, of the adhesion to laminin. Moreover, when the adhesion experiment was performed in the presence of different concentrations (0.1, 1, and 10 μg per well) of soluble GAPDH from S. cerevisiae, the attachment to fibronectin decreased significantly (15, 25, and 85% inhibition, respectively) (Fig. 3A). The same concentrations of soluble GAPDH inhibited binding to laminin by 32, 58, and 66%, respectively (Fig. 3B). No inhibition was observed in the presence of 10 μg of BSA, in the presence of an irrelevant Ab (1:10 dilution, rabbit PAbs against an α-amylase from L. kononenkoae) (43) (Fig. 3), or in the presence of preimmune sera (data not shown). These results indicate that the surface-located GAPDH mediates adhesion of C. albicans blastoconidia to both ligands, fibronectin and laminin.
Fibronectin and laminin cause partial inhibition of GAPDH immunodetection as determined by flow cytometry analysis.
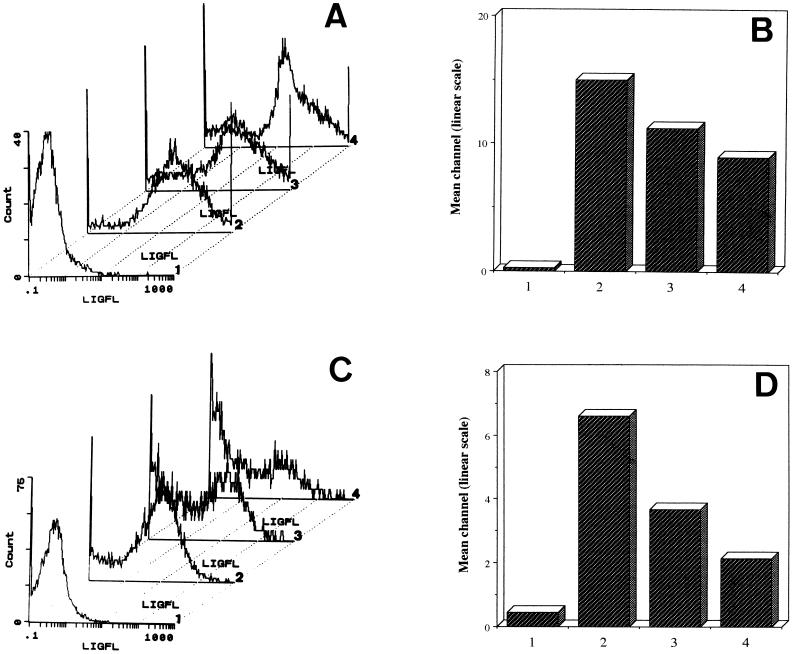
C. albicans blastoconidia were analyzed by indirect immunofluorescence and flow cytometry with the anti-GAPDH PAb. The fluorescence observed in most of the cells indicated that the GAPDH is exposed on their surfaces (Fig. 4, assays 2). Control assays performed either with preimmune sera or with irrelevant Ab (anti-α-amylase PAb from L. kononenkoae) did not result in a substantial cell surface staining (less than 1% of the positive control) (data not shown). To confirm that the surface-located GAPDH binds fibronectin and laminin, we performed the same indirect immunofluorescence assay in the presence of different concentrations of fibronectin or laminin. Binding of the anti-GAPDH PAb was reduced by about 25 and 40% by fibronectin at concentrations of 50 and 100 μg/ml, respectively (Fig. 4A and B; assays 3 and 4); binding of the Ab was reduced by about 44 and 67% by laminin at concentrations of 50 and 100 μg/ml, respectively (Fig. 4C and D; assays 3 and 4).
FIG. 4.
Flow cytometry analysis of C. albicans blastoconidia using anti-GAPDH PAb. The immunofluorescence assay was performed in the presence of 50 and 100 μg of fibronectin per ml (assays 3 and 4, respectively) (A and B) or in presence of 50 and 100 μg of laminin per ml (assays 3 and 4, respectively) (C and D) or without any inhibitor (assays 2) (A to D). Control assays were performed in the absence of anti-GAPDH PAb (negative control; assays 1) (A to D). (A and C) Representative histograms. x axis, log of fluorescence intensity (LIGFL); y axis, number of fluorescent cells. (B and D) Fluorescence mean channel represented on a linear scale. It is important to note the correlation between sample numbers in panels A and B and in panels C and D.
The purified cytoplasmic GAPDH binds fibronectin and laminin.
The GAPDH protein was purified from a cytosolic C. albicans extract by immunoaffinity chromatography using the anti-C. albicans GAPDH PAb. SDS-PAGE of the final purified preparation revealed a homogeneous protein with a molecular mass of 33 kDa (see Fig. 5A, lane 2). The yield of GAPDH purification was 0.1% (50 μg of purified protein was obtained from 45 mg of total protein). The purified GAPDH protein was enzymatically active (specific activity, 23 [expressed as micromoles of NADH per minute per milligram]).
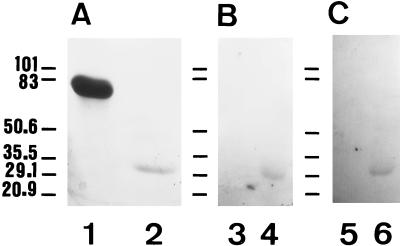
FIG. 5.
Binding activity of the purified cytosolic C. albicans GAPDH to fibronectin and laminin. Samples were subjected to SDS-PAGE on triplicate reducing 9% polyacrylamide gels. One gel was stained with Coomassie brilliant blue to visualize proteins (A). The other two gels were electroblotted onto PVDF membranes and the blots were reacted with either fibronectin, rabbit antifibronectin Abs, and peroxidase-labelled goat anti-rabbit Ig (B) or with laminin, rabbit antilaminin Abs, and peroxidase-labelled goat anti-rabbit Ig (C). Lanes: 1, 3, and 5, BSA (5 μg); 2, 4, and 6, GAPDH (0.5 μg). The positions of standard proteins with known molecular masses (expressed in kilodaltons) run in parallel are shown at the left of each panel.
The ability of the purified GAPDH to bind fibronectin and laminin was determined by a ligand Western blotting assay. The purified GAPDH (0.5 μg) and BSA (5 μg), used as a control protein, were subjected to SDS-PAGE under reducing conditions on slab gels (9% acrylamide) and then stained with Coomassie blue (Fig. 5A). Binding of fibronectin (Fig. 5B) and laminin (Fig. 5C) to the proteins transferred to PVDF membranes was only observed to the purified GAPDH and not to BSA. The absence of bands following detection with the specific antifibronectin or antilaminin Abs when previous incubation of the PVDF blots in the fibronectin or laminin solutions was omitted indicates the specificity of the ligand binding.
In addition, since soluble GAPDH enzyme from S. cerevisiae inhibited adhesion of cells to fibronectin and laminin (Fig. 3), we checked the purity of this commercial protein and its ability to bind these ligands to exclude the presence of other ECM binding components. Analyses by SDS-PAGE and Western blotting indicate that no detectable contaminant species are present in the preparation and that the S. cerevisiae GAPDH protein actually binds fibronectin and laminin (Fig. 6), as expected from the adherence assays.
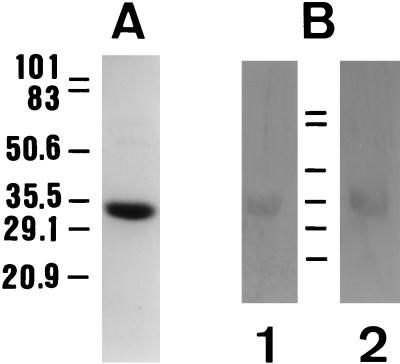
FIG. 6.
Binding activity of the S. cerevisiae GAPDH to fibronectin and laminin. Samples (2 μg) of GAPDH from a commercial source were subjected to SDS-PAGE and stained with Coomassie brilliant blue to show protein species (A) or electroblotted onto PVDF membranes (B), and the blots were reacted with either fibronectin, rabbit antifibronectin Abs, and peroxidase-labelled goat anti-rabbit Ig (lane 1) or with laminin, rabbit antilaminin Abs, and peroxidase-labelled goat anti-rabbit Ig (lane 2). The positions of standard proteins with known molecular masses (expressed in kilodaltons) run in parallel are shown.
DISCUSSION
The identification of glycolytic enzymes as immunogens during candidiasis is well documented. C. albicans enolase, phosphoglycerate kinase, alcohol dehydrogenase, pyruvate kinase, and aldolase have been described as major allergens or immunogens during candidiasis (19, 21, 32, 34, 47, 50–54). Recently, we have extended the above list of immunogenic glycolytic enzymes to GAPDH and showed that the GAPDH is located at the C. albicans cell surface (16). Immunoelectron microscopy with a anti-C. albicans GAPDH PAb confirmed that this protein is a genuine component of the cell wall of C. albicans. In addition to its intracellular location, the enzyme was detected at the outermost layer of the cell wall and also extended through the cell wall structure. Quantification of percentage of GAPDH present in the cell wall is difficult to assess since labelling intensity varied in different cells, both in the cytoplasm and in the cell wall. However, from the immunoelectron microscopy observations, it can be estimated that the cell wall-bound protein represents an important percentage of the cellular GAPDH. This is in accordance with previous quantification (20 to 35% of the enzyme is cell wall bound) estimated from the activity data (16).
The presence of glycolytic enzymes on C. albicans cell wall is not unprecedented: enolase is found in the culture supernatant and in the inner layers of the cell wall but is not exposed at the cell surface (2, 50), and phosphoglycerate kinase has been detected at the cell surface of C. albicans and also extended through the cell wall (1). However, for these two cell wall-associated enzymes, any enzymatic activities or other functions unrelated to glycolysis have not been reported. On the other hand, GAPDH proteins have been previously described as localized on the surface of other organisms. Evidence for an active GAPDH on the surface of S. mansoni associated with human resistance to schistosomiasis was reported by Goudot-Crozel et al. (18). In K. marxianus, the GAPDH is clearly induced in the cell wall of flocculent cells, supporting the hypothesis that the protein is involved in cell surface interaction or adhesion leading to flocculation (12, 13). Finally, the enzyme has been reported as a major surface and enzymatically active protein on group A streptococci (38); the protein also binds various mammalian proteins such as lysozyme, fibronectin, the cytoskeletal proteins actin and myosin (38), and plasmin (54) and displays an ADP-ribosylating activity (39). These observations and the fact that in higher eukaryotes GAPDH has been found in several subcellular locations, displaying functions unrelated to glycolysis (48), raise the question of whether GAPDH on the surface of C. albicans may also have nonglycolytic functions. Since C. albicans is known to bind to fibronectin and laminin in a typical ligand-receptor manner (4, 22, 23, 27, 36, 41, 49), we determined whether the surface-located C. albicans GAPDH is a mediator of adhesion to both ECM proteins.
A direct demonstration that the surface GAPDH is involved in the interaction of C. albicans with fibronectin and laminin was obtained by adhesion experiments of yeast cells to immobilized ligands. The addition of anti-C. albicans GAPDH PAb and soluble GAPDH from S. cerevisiae reduced the attachment to fibronectin- and laminin-coated wells up to 70 to 85%. Adhesion to immobilized ligands was strongly reduced when yeast cells were pretreated either with trypsin or β-mercaptoethanol (β-ME) prior to the biotin labelling reaction (87 and 98% inhibition, respectively) (data not shown), indicating that these treatments also remove cell surface receptors for laminin and fibronectin other than GAPDH. In addition, soluble fibronectin or laminin prevents interaction of the specific Ab (anti-GAPDH PAb) to the cell surface as determined by immunofluorescence techniques, indicating that both ligands and Ab compete to bind to the surface GAPDH. Ligand blot analysis of the purified cytosolic GAPDH was used to confirm the multiple binding capacity of the protein. However, binding of ligands to purified protein was not very strong. In fact, analysis by ligand Western blotting of β-ME extracts containing cell wall-associated GAPDH failed to detect any 33-kDa protein (data not shown). This suggests that ligand binding detection is under the detection limit and that β-ME extraction of cell wall-associated GAPDH and SDS-PAGE under reducing conditions probably modify the native protein conformation, resulting in a decrease of its binding ability.
Different laminin binding proteins have been described in C. albicans. Bouchara et al. (4) identified germ tube-specific cell surface components (68, 62, and 60 kDa), with laminin binding activity, which appear to belong to a family of C. albicans cell wall proteins and glycoproteins exhibiting multiple affinities for laminin, fibrinogen, and C3d. Subsequently, López-Ribot et al. (30) described, in the β-ME extract obtained from nongerminated blastoconidia, the presence of a 37-kDa laminin binding protein that cross-reacted with Abs against the high-affinity human laminin receptor and that did not bind to other mammalian proteins, such as fibrinogen, fibronectin, and type IV collagen. These results point to the possibility that different cell surface receptors for laminin may be differentially expressed in C. albicans yeast or germinated cells. Several putative receptors for fibronectin on C. albicans have been identified, including homologs of mammalian integrins (45, 46) and 60- and 105-kDa glycoproteins (25). However, the identification of receptors for fibronectin have been limited by the variability of their expression depending on the strain and growth conditions (27, 36, 56) as well as by the experimental procedure used for their identification (17). Recently, a C. albicans gene (ALA1) that confers adherence properties upon S. cerevisiae for ECM proteins, including laminin and fibronectin, has been characterized (15). Our results (surface GAPDH binds fibronectin and laminin) suggest that the ECM proteins share to some extent the same receptor-like molecule, and thus a single receptor may display multiple binding activities, as described for the C. albicans laminin receptors identified by Bouchara et al. (4) and for the ALA1 gene product (15). Furthermore, the GAPDH protein on the surface of group A streptococci was found to have multiple binding ability to host proteins (38).
In this study, we have demonstrated that the C. albicans GAPDH, besides having a cytoplasmic location, is an integral protein of the cell wall, mainly exposed at the cell surface. In this location, the protein may contribute to the microorganism’s invasiveness by its ability to bind to host fibronectin and laminin.
ACKNOWLEDGMENTS
The support of grant SAF95-0595 from the CICyT (Plan Nacional de Salud y Farmacia), Ministerio de Educación y Ciencia (Spain), to J.P.M. is acknowledged.
We thank J. E. O’Connor (Departamento de Bioquímica y Biología Molecular, Universitat de València, Valencia, Spain) for his assistance with flow cytometry experiments and P. Sanz (Instituto de Agroquímica y Tecnología de Alimentos, CSIC) for the gift of the anti-α-amylase Ab.
REFERENCES
- 1.Alloush H M, López-Ribot J L, Masten B J, Chaffin W L. 3-Phosphoglycerate kinase: a glycolytic enzyme present in the cell wall of Candida albicans. Microbiology—UK. 1997;143:321–330. doi: 10.1099/00221287-143-2-321. [DOI] [PubMed] [Google Scholar]
- 2.Angiolella L, Facchin M, Stringaro A, Maras B, Simonetti N, Cassone A. Identification of a glucan-associated enolase as a main cell wall protein of Candida albicans and an indirect target of lipopeptide antimycotics. J Infect Dis. 1996;173:684–690. doi: 10.1093/infdis/173.3.684. [DOI] [PubMed] [Google Scholar]
- 3.Bodey G P. Candidiasis: pathogenesis, diagnosis, and treatment. 2nd ed. New York, N.Y: Raven Press; 1993. [Google Scholar]
- 4.Bouchara J P, Tronchin G, Annaix V, Robert R, Senet J M. Laminin receptors on Candida albicans germ tubes. Infect Immun. 1990;58:48–54. doi: 10.1128/iai.58.1.48-54.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Calderone R A. Molecular interactions at the interface of Candida albicans and host cells. Arch Med Res. 1993;24:275–279. [PubMed] [Google Scholar]
- 6.Calderone R A, Braun P C. Adherence and receptor relationships of Candida albicans. Microbiol Rev. 1991;55:1–20. doi: 10.1128/mr.55.1.1-20.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Casanova M, Gil M L, Cardeñoso L, Martínez J P, Sentandreu R. Identification of wall-specific antigens synthesized during germ tube formation by Candida albicans. Infect Immun. 1989;57:262–271. doi: 10.1128/iai.57.1.262-271.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Casanova M, López-Ribot J L, Martínez J P, Sentandreu R. Characterization of cell wall proteins from yeast and mycelial cells of Candida albicans by labelling with biotin: comparison with other techniques. Infect Immun. 1992;60:4898–4906. doi: 10.1128/iai.60.11.4898-4906.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Casanova M, López-Ribot J L, Monteagudo C, Llombart-Bosch A, Sentandreu R, Martínez J P. Identification of a 58-kilodalton cell surface fibrinogen-binding mannoprotein from Candida albicans. Infect Immun. 1992;60:4221–4229. doi: 10.1128/iai.60.10.4221-4229.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Chaffin W L, López-Ribot J L, Casanova M, Gozalbo D, Martínez J P. Cell wall and secreted proteins of Candida albicans: identification, function, and expression. Microbiol Mol Biol Rev. 1997;62:130–180. doi: 10.1128/mmbr.62.1.130-180.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Cutler J E. Putative virulence factors of Candida albicans. Annu Rev Microbiol. 1991;45:187–218. doi: 10.1146/annurev.mi.45.100191.001155. [DOI] [PubMed] [Google Scholar]
- 12.Fernandes P A, Keen J N, Findlay J B C, Moradas-Ferreira P. A protein homologous to glyceraldehyde-3-phosphate dehydrogenase is induced in the cell wall of a flocculent Kluyveromyces marxianus. Biochim Biophys Acta. 1992;1159:67–73. doi: 10.1016/0167-4838(92)90076-p. [DOI] [PubMed] [Google Scholar]
- 13.Fernandes P A, Sousa M, Moradas-Ferreira P. Flocculation of Kluyveromyces marxianus is induced by a temperature upshift. Yeast. 1993;9:859–866. doi: 10.1002/yea.320090806. [DOI] [PubMed] [Google Scholar]
- 14.Fukazawa Y, Kagaya K. Molecular bases of adhesion of Candida albicans. J Med Vet Mycol. 1997;35:87–99. doi: 10.1080/02681219780000971. [DOI] [PubMed] [Google Scholar]
- 15.Gaur N K, Klotz S A. Expression, cloning, and characterization of a Candida albicans gene, ALA1, that confers adherence properties upon Saccharomyces cerevisiae for extracellular matrix proteins. Infect Immun. 1997;65:5289–5294. doi: 10.1128/iai.65.12.5289-5294.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Gil-Navarro I, Gil M L, Casanova M, O’Connor J E, Martínez J P, Gozalbo D. The glycolytic enzyme glyceraldehyde-3-phosphate dehydrogenase of Candida albicans is a surface antigen. J Bacteriol. 1997;179:4992–4999. doi: 10.1128/jb.179.16.4992-4999.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Glee P M, Masuoka J, Ozier W T, Hazen K C. Presence of multiple laminin- and fibronectin-binding proteins in cell wall extracts of Candida albicans: influence of dialysis. J Med Vet Mycol. 1996;34:57–61. doi: 10.1080/02681219680000091. [DOI] [PubMed] [Google Scholar]
- 18.Goudot-Crozel V, Caillol D, Djabali M, Dessein A J. The major parasite surface antigen associated with human resistance to schistosomiasis is a 37 kDa glyceraldehyde-3P-dehydrogenase. J Exp Med. 1989;170:2065–2080. doi: 10.1084/jem.170.6.2065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Gutiérrez J, Maroto C, Piédrola G, Martín E, Pérez J A. Circulating Candida antigens and antibodies: useful markers of candidemia. J Clin Microbiol. 1993;31:2550–2552. doi: 10.1128/jcm.31.9.2550-2552.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Hostetter M K. Adhesins and ligands involved in the interaction of Candida albicans with epithelial and endothelial surfaces. Clin Microbiol Rev. 1994;7:29–42. doi: 10.1128/cmr.7.1.29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ishiguro A, Homma M, Torii S, Tanaka K. Identification of Candida albicans antigens reactive with immunoglobulin E antibody of human sera. Infect Immun. 1992;60:1550–1557. doi: 10.1128/iai.60.4.1550-1557.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Jakab E, Paulsson M, Ascencio F, Ljungh A. Expression of vitronectin and fibronectin binding by Candida albicans yeast cells. APMIS. 1993;101:187–193. [PubMed] [Google Scholar]
- 23.Kalo A, Segal E, Sahar E, Dayan D. Interaction of Candida albicans with genital mucosal surfaces: involvement of fibronectin in adherence. J Infect Dis. 1988;157:1253–1256. doi: 10.1093/infdis/157.6.1253. [DOI] [PubMed] [Google Scholar]
- 24.Klotz S A. Plasma and extracellular matrix proteins mediate in the fate of Candida albicans in the human host. Med Hypotheses. 1994;42:328–334. doi: 10.1016/0306-9877(94)90008-6. [DOI] [PubMed] [Google Scholar]
- 25.Klotz S A, Chen R C, Smith R L, Rouse J B. The fibronectin adhesin of Candida albicans. Infect Immun. 1994;62:4679–4681. doi: 10.1128/iai.62.10.4679-4681.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Klotz S A, Rutten M J, Smith R L, Babcock S R, Cunningham M D. Adherence of Candida albicans to immobilized extracellular matrix proteins is mediated by calcium-dependent surface glycoproteins. Microb Pathog. 1993;14:133–147. doi: 10.1006/mpat.1993.1014. [DOI] [PubMed] [Google Scholar]
- 27.Klotz S A, Smith R L. A fibronectin receptor on Candida albicans mediates adherence of the fungus to extracellular matrix. J Infect Dis. 1991;163:604–610. doi: 10.1093/infdis/163.3.604. [DOI] [PubMed] [Google Scholar]
- 28.Laemmli U K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature (London) 1970;227:680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- 29.Lee K L, Buckley H R, Campbell C. An amino acid liquid synthetic medium for development of mycelial and yeast forms of Candida albicans. Sabouraudia. 1975;13:148–153. doi: 10.1080/00362177585190271. [DOI] [PubMed] [Google Scholar]
- 30.López-Ribot J L, Casanova M, Monteagudo C, Sepúlveda P, Martínez J P. Evidence for the presence of a high-affinity laminin receptor-like molecule on the surface of Candida albicans yeast cells. Infect Immun. 1994;62:742–746. doi: 10.1128/iai.62.2.742-746.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Lowry O H, Rosebrough N J, Farr A L, Randall R J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951;193:265–275. [PubMed] [Google Scholar]
- 32.Martínez J P, Gil M L, López-Ribot J L, Chaffin W L. Serologic response to cell wall mannoproteins and proteins of Candida albicans. Clin Microbiol Rev. 1998;11:121–141. doi: 10.1128/cmr.11.1.121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Meunier F. Candidiasis. Eur J Clin Microbiol Infect Dis. 1989;8:438–447. doi: 10.1007/BF01964058. [DOI] [PubMed] [Google Scholar]
- 34.Mitsutake K, Kohno S, Miyazaki T, Miyazaki H, Maesaki S, Koga H. Detection of Candida enolase antibody in patients with candidiasis. J Clin Lab Anal. 1994;8:207–210. doi: 10.1002/jcla.1860080405. [DOI] [PubMed] [Google Scholar]
- 35.Montoliu C, Sancho-Tello M, Azorin Y, Burgal M, Vallés S, Renau-Piqueras J, Guerri C. Ethanol increases cytochrome P4502E1 and induces oxidative stress in astrocytes. J Neurochem. 1995;65:2561–2570. doi: 10.1046/j.1471-4159.1995.65062561.x. [DOI] [PubMed] [Google Scholar]
- 36.Nègre E, Vogel T, Levanon A, Guy R, Walsh T J, Roberts D D. The collagen binding domain of fibronectin contains a high affinity binding site for Candida albicans. J Biol Chem. 1994;269:22039–22045. [PubMed] [Google Scholar]
- 37.Odds F C. Candida and candidosis: a review and bibliography. London, United Kingdom: Bailliere Tindall; 1988. pp. 93–114. [Google Scholar]
- 38.Pancholi V, Fischetti V A. A major surface protein on group A streptococci is a glyceraldehyde-3-phosphate-dehydrogenase with multiple binding activity. J Exp Med. 1992;176:415–426. doi: 10.1084/jem.176.2.415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Pancholi V, Fischetti V A. Glyceraldehyde-3-phosphate dehydrogenase on the surface of group A streptococci is also an ADP-ribosylating enzyme. Proc Natl Acad Sci USA. 1993;90:8154–8158. doi: 10.1073/pnas.90.17.8154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Peñalver M C, O’Connor J E, Martínez J P, Gil M L. Binding of human fibronectin to Aspergillus fumigatus conidia. Infect Immun. 1996;64:1146–1153. doi: 10.1128/iai.64.4.1146-1153.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Pendrak M L, Klotz S A. Adherence of Candida albicans to host cells. FEMS Microbiol Lett. 1995;129:103–114. doi: 10.1111/j.1574-6968.1995.tb07566.x. [DOI] [PubMed] [Google Scholar]
- 42.Penn C, Klotz S A. Binding of plasma fibronectin to Candida albicans occurs through the cell binding domain. Microb Pathog. 1994;17:387–393. doi: 10.1006/mpat.1994.1084. [DOI] [PubMed] [Google Scholar]
- 43.Prieto J A, Bort B R, Martinez J, Randez-Gil F, Buesa C, Sanz P. Purification and characterization of a new α-amylase of intermediate thermal stability from the yeast Lipomyces kononenkoae. Biochem Cell Biol. 1995;73:41–49. doi: 10.1139/o95-005. [DOI] [PubMed] [Google Scholar]
- 44.Renau-Piqueras J, Zaragozá R, De Paz P, Báguena-Cervellera R, Megías L, Guerri C. Effects of prolonged ethanol exposure on the glial fibrillary acidic protein-containing intermediate filaments of astrocytes in primary culture: a quantitative immunofluorescence and immunogold electron microscopic study. J Histochem Cytochem. 1989;37:229–240. doi: 10.1177/37.2.2642942. [DOI] [PubMed] [Google Scholar]
- 45.Santoni G, Birarelli P, Jin-Hong L, Gamero A, Djeu J Y, Piccoli M. An α5β1-like integrin receptor mediates the binding of less pathogenic Candida species to fibronectin. J Med Microbiol. 1995;43:360–367. doi: 10.1099/00222615-43-5-360. [DOI] [PubMed] [Google Scholar]
- 46.Santoni G, Gismondi A, Liu J H, Punturieri A, Santoni A, Frati L, Piccoli M, Djeu J Y. Candida albicans expresses a fibronectin receptor antigenically related to α5β1 integrin. Microbiology. 1994;140:2971–2979. doi: 10.1099/13500872-140-11-2971. [DOI] [PubMed] [Google Scholar]
- 47.Shen H D, Choo K B, Lee H H, Hsieh J C, Lin W L, Lee W R, Han S H. The 40-kilodalton allergen of Candida albicans is an alcohol dehydrogenase: molecular cloning and immunological analysis using monoclonal antibodies. Clin Exp Allergy. 1991;21:675–681. doi: 10.1111/j.1365-2222.1991.tb03195.x. [DOI] [PubMed] [Google Scholar]
- 48.Singh R, Green M R. Sequence specific binding of transfer RNA by glyceraldehyde-3-phosphate dehydrogenase. Science. 1993;259:365–368. doi: 10.1126/science.8420004. [DOI] [PubMed] [Google Scholar]
- 49.Skerl K G, Calderone R A, Segal E, Sreevalsan T, Scheld W M. In vitro binding of Candida albicans yeast cells to human fibronectin. Can J Microbiol. 1984;30:221–227. doi: 10.1139/m84-033. [DOI] [PubMed] [Google Scholar]
- 50.Sundstrom P M, Aliaga G R. A subset of proteins found in culture supernatants of Candida albicans include the abundant, immunodominant, glycolytic enzyme enolase. J Infect Dis. 1994;169:452–456. doi: 10.1093/infdis/169.2.452. [DOI] [PubMed] [Google Scholar]
- 51.Sundstrom P M, Jensen J, Balish E. Humoral and cellular immune responses to enolase after alimentary tract colonization or intravenous immunization with Candida albicans. J Infect Dis. 1994;170:390–395. doi: 10.1093/infdis/170.2.390. [DOI] [PubMed] [Google Scholar]
- 52.Swoboda R K, Bertram G, Hollander H, Greenspan D, Greenspan J S, Gow N A R, Gooday G W, Brown A J P. Glycolytic enzymes of Candida albicans are nonubiquitous immunogens during candidiasis. Infect Immun. 1993;61:4263–4271. doi: 10.1128/iai.61.10.4263-4271.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.van Deventer A J M, van Vliet H J A, Hop W C J, Goessens W H F. Diagnostic value of anti-Candida enolase antibodies. J Clin Microbiol. 1994;32:17–23. doi: 10.1128/jcm.32.1.17-23.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Walsh T J, Hathorn J W, Sobel J D, Merz W G, Sánchez V, Maret S M, Buckley H R, Pfaller M A, Schaufele R, Sliva C, Navarro E, Lecciones P, Chandrasekar P, Lee J, Pizzo P A. Detection of circulating Candida enolase by immunoassay in patients with cancer and invasive candidiasis. N Engl J Med. 1991;324:1026–1031. doi: 10.1056/NEJM199104113241504. [DOI] [PubMed] [Google Scholar]
- 55.Winram S B, Lottenberg R. The plasmin-binding protein Plr of group A streptococci is identified as glyceraldehyde-3-phosphate dehydrogenase. Microbiology—UK. 1996;142:2311–2320. doi: 10.1099/13500872-142-8-2311. [DOI] [PubMed] [Google Scholar]
- 56.Yan S, Nègre E, Cashel J A, Guo N, Lyman C A, Walsh T J, Roberts D D. Specific induction of fibronectin binding activity by hemoglobin in Candida albicans grown in defined media. Infect Immun. 1996;64:2930–2935. doi: 10.1128/iai.64.8.2930-2935.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]