Abstract
The plethora of newly discovered vaccines implies that, in the future, many vaccines will have to be administered simultaneously to infants. We examined the potential interference with the immune response of several coadministered vaccines containing the same protein component, namely, tetanus toxoid (TT). Infants simultaneously receiving a tetravalent pneumococcal vaccine conjugated to TT (PncT) and a diphtheria-tetanus-pertussis–poliovirus–Haemophilus influenzae type b-tetanus conjugate vaccine showed significantly lower anti-H. influenzae type b polysaccharide (polyribosylribitol phosphate [PRP]) antibody concentrations than those receiving either a tetravalent pneumococcal vaccine conjugated to diphtheria toxoid or placebo. A dose range study showed that anti-PRP antibody concentrations were inversely related to the TT content of the PncT vaccines administered in infancy. Postimmunization antitetanus antibody concentrations were also affected adversely as the TT content of the coadministered vaccines was increased. This phenomenon, which we believe derives from interference by a common protein carrier, should be taken into account when the introduction of an immunization program including multiple conjugate vaccines is considered.
Recently licensed vaccines include the Haemophilus influenzae type b (Hib) conjugate, varicella-zoster, acellular pertussis, and hepatitis A vaccines. Many additional vaccines are being tested in clinical studies. For practical reasons, if included in childhood vaccination programs, they ought to be administered simultaneously at separate sites or as combined vaccines (6). Possible interactions between the vaccines thus become important from both theoretical and practical points of view (7).
The first conjugate vaccines were those against Hib, in which a polysaccharide or oligosaccharide derived from the Hib capsule (polyribosylribitol phosphate [PRP]) was covalently conjugated to a protein carrier (24, 29). The same technology is now used to widen the range of conjugate vaccines against invasive organisms such as pneumococci and other encapsulated organisms (28). Multiple vaccines based on the same protein carrier and thus having common antigenic epitopes might be available soon, and the possibility of their interactions must be considered. The simultaneous administration of several conjugate vaccines sharing the same protein carrier and the carrier itself may be associated with the suppression of the response to polysaccharides through various mechanisms. Examples of such theoretical mechanisms are competition for antigen capture and presentation between B cells with surface immunoglobulins specific for epitopes on the carrier and B cells specific for the polysaccharide; prevention of the binding of the conjugate vaccines to polysaccharide-specific B cells by the free protein carrier; and suppression of the response to polysaccharides by expansion of the number of carrier-specific B cells induced by previous injection of the carrier, thus directing the conjugate away from polysaccharide-specific B cells (17).
We recently studied the immunogenicity of two newly developed tetravalent pneumococcal conjugate vaccines (2, 8, 9). Both of these vaccines contained polysaccharide antigens of four pneumococcal serotypes (6B, 14, 19F, and 23F) conjugated either to tetanus toxoid (TT) (PncT vaccine) or to diphtheria toxoid (PncD vaccine). These pneumococcal vaccines were administered simultaneously with two other vaccines, diphtheria-tetanus-pertussis (DTP) and Hib polysaccharide-TT conjugate (PRP-T). The purpose of this study was to examine if the simultaneous administration of PncT adversely affects the immunologic response to the two other vaccines also containing TT, namely, DTP and PRP-T.
MATERIALS AND METHODS
Study design.
Two parallel studies on the safety and immunogenicity of new tetravalent pneumococcal conjugate vaccines were conducted, one in Israel and one in Finland. Both studies were double blinded, randomized, and controlled. Each study was approved by the relevant ethics committees, and written informed consent was obtained from the parents or legal guardians before enrollment at both study sites.
Vaccines. (i) Pneumococcal conjugate vaccines and placebo.
PncT vaccine (manufactured by Pasteur Mérieux Connaught, Lyon, France; lot S2840) was a mixture of four purified capsular polysaccharides from Streptococcus pneumoniae serotypes 6B, 14, 19F, and 23F conjugated to TT. The ratios of TT to polysaccharide in the bulk (individual batches) were 1.6 for type 6B, 2.2 for type 14, 1.4 for type 19F, and 2.2 for type 23F. PncD vaccine (manufactured by Pasteur Mérieux Connaught, Swiftwater, Pa.; lot 930095) was a mixture of the same four pneumococcal polysaccharides conjugated to diphtheria toxoid. The respective ratios of diphtheria toxoid to polysaccharide were 2.0, 2.7, 3.1, and 2.8. The placebo used in the study consisted of phosphate-buffered saline.
All of these vaccines were contained in single-dose, ready-to-use glass syringes indistinguishable in appearance. The vaccines were administered as a 0.5-ml intramuscular injection into the upper part of the anterolateral thigh.
(ii) Other vaccines.
In Israel, the DTP, PRP-T, and trivalent inactivated poliovirus (IPV) vaccines were administered as a single dose after lyophilized PRP-T was reconstituted with 0.5 ml of liquid DTP-IPV to form a pentavalent DTP–IPV–PRP-T vaccine (Pasteur Mérieux Connaught, Lyon, France). This mixture contained 10 flocculation units (Lf) (approximately 30 μg) of TT, 25 Lf of diphtheria toxoid, and 10 μg of PRP conjugated to 24 μg of TT.
In Finland, DTP was produced locally (National Public Health Institute, Helsinki, Finland). The vaccine contained 5 Lf (approximately 15 μg) of purified TT and 19 Lf of purified diphtheria toxoid. PRP-T (ActHIB; Pasteur Mérieux Connaught, Lyon, France) was reconstituted with 0.5 ml of liquid DTP to form DTP–PRP-T.
Trivalent oral live poliovirus (OPV) vaccine (Pasteur Mérieux Connaught, Lyon, France) was administered in Israel at 4, 6, and 12 months of age.
In addition, Israeli children received an intramuscular dose of a 23-valent pneumococcal nonconjugate vaccine (Pneumo-23; Pasteur Mérieux Connaught, Lyon, France) as a booster vaccination at 12 months of age.
Vaccination and sampling schedule.
Infants were enrolled in the study at the age of 2 months (range, 6 to 10 weeks). They were examined by a physician at enrollment and by a physician or a public health nurse before each immunization.
In Israel, healthy infants were recruited from six Mother and Child Health Centers located in the cities of Beer-Sheva and Arad, in southern Israel. The total number of recruited infants was 75 (25 in the PncT group, 25 in the PncD group, and 25 in the placebo group). PncT (3 μg of polysaccharide for each serotype [PncT03]), PncD (3 μg of polysaccharide for each serotype), or placebo was administered at 2, 4, and 6 months of age (Table 1). At each visit, all infants received a combined DTP–IPV–PRP-T injection at the contralateral site. To comply with national vaccination program requirements, OPV vaccine also was administered at 4 and 6 months. At 12 months of age, all children received an injection of pneumococcal polysaccharide vaccine simultaneously with the fourth dose of DTP–IPV–PRP-T and a third dose of OPV vaccine.
TABLE 1.
Study designs in Israel (75 infants) and Finland (200 infants)
| Country and parameter | Regimen at mo:
|
|||||
|---|---|---|---|---|---|---|
| 2 | 4 | 6 | 7 | 12 | 13 | |
| Israel | ||||||
| Study vaccines | +a | +a | +a | +b | ||
| Other simultaneously administered vaccines | DTP–IPV–PRP-T | DTP–IPV–PRP-T, OPV | DTP–IPV–PRP-T, OPV | DTP–IPV–PRP-T, OPV | ||
| Blood samples | + | + | + | + | + | + |
| Finland | ||||||
| Study vaccines | +c | +c | +c | |||
| Other simultaneously administered vaccines | DTP–PRP-T | DTP–PRP-T | DTP–PRP-T | |||
| Blood samples | + | + | + | + | ||
PncT (n = 25), PncD (n = 25), or placebo (n = 25). PncT and PncD were given at a dose of 3 μg of polysaccharide per conjugate.
23-valent pneumococcal nonconjugate vaccine.
PncT (n = 75), PncD (n = 75), or placebo (n = 50). For PncT and PncD, each group of 75 was further divided into three groups receiving 1 μg of each component (PncT01; n = 25), 3 μg of each component (PncT03; n = 25), or 10 μg of each component (PncT10; n = 25).
In the Finnish study, children were recruited from Child Health Centers in the towns of Joensuu, Kerava, and Tampere. They were divided into three groups: 75 received PncT vaccine (25 received 1 μg of polysaccharide of each serotype per vaccine dose [PncT01], 25 received PncT03, and 25 received a 10-μg dose of each polysaccharide [PncT10]), 75 received PncD vaccine (25 each received 1, 3, or 10 μg of polysaccharide of each serotype), and 50 received placebo (since both PncD and placebo did not contain TT protein, they were both considered PncT0, implying that no TT content except that in DTP was present). These vaccines were administered at 2, 4, and 6 months of age. At each visit, all infants received a combined DTP–PRP-T injection at the contralateral site (Table 1). In Finland, no PRP-T was given simultaneously with the booster dose of pneumococcal vaccine; therefore, the follow-up period for the present analysis ended by the age of 7 months.
The total TT content administered at each visit to infants in the various regimens in Finland and Israel is presented in Table 2.
TABLE 2.
TT content administered at 2, 4, 6, and 12 months in Israel and 2, 4, and 6 months in Finlanda
| Regimen | TT content (μg) of vaccines administered
at each encounter at mo:
|
|
|---|---|---|
| 2, 4, and 6 | 12 | |
| Israel | ||
| PncT0 (placebo and PncD groups) | 54 | 54 |
| PncT03 | 78 | 54 |
| Finland | ||
| PncT0 (placebo and PncD groups) | 39 | NR |
| PncT01 | 48 | NR |
| PncT03 | 63 | NR |
| PncT10 | 111 | NR |
The TT contents in individual vaccines were as follows: DTP in Israel, 30 μg; DTP in Finland, 15 μg; PRP-T, 24 μg; PncT01, 9 μg; PncT03, 24 μg; and PncT10, 72 μg. NR, not relevant.
Venous blood was obtained from all children at 2, 4, 6, and 7 months; additional samples in Israel were taken at 12 and 13 months. Sera were stored at −20°C until analyzed.
Serologic assays.
Type-specific pneumococcal polysaccharide immunoglobulin G was assessed for each of the four serotypes in the conjugate vaccines. Detailed data are presented elsewhere (1, 8, 9). The anti-PRP antibody concentration was measured by a radioimmunoassay (25) in two different laboratories. The sera of the Israeli infants were tested at Pasteur Mérieux Connaught in France, and the sera of the Finnish infants were tested at Pasteur Mérieux Connaught in the United States.
Methods to measure TT and diphtheria toxoid antibody concentrations differed in the two studies. In Israel, tetanus antibody concentration was measured by a solid-phase enzyme-linked immunosorbent assay (EIA) with 0.11 Lf of purified TT per ml and compared to the international standard for tetanus immunoglobulins (TE.3; Statens Seruminstitut). The lower limit of quantification was 0.006 IU/ml. Diphtheria antibody concentration was measured by a solid-phase EIA with 0.13 Lf of purified diphtheria toxoid per well and compared to the equine international standard for diphtheria antitoxin (Statens Seruminstitut).
In Finland, a modified version of an indirect alkaline phosphatase EIA was used to assay the tetanus and diphtheria antitoxin immunoglobulin G concentrations (21). Duplicates of four serum dilutions, starting with the 1:100 dilution and progressing by dilution factors of five, were incubated in EIA plate wells coated with TT (0.5 Lf/ml) or diphtheria toxoid (2.5 Lf/ml) from the Vaccine Department at the National Public Health Institute. The A405 was measured with a Multiscan MS EIA reader (Labsystems, Helsinki, Finland), and the antibody concentrations were calculated with a reference line method (Unitcalc; Unisys, Stockholm, Sweden). An immunoglobulin was used as an in-house standard and was calibrated against the World Health Organization international standards for tetanus and diphtheria antitoxins. Tetanus and diphtheria antitoxin concentrations were thus expressed in international units per milliliter. The quantification limit was 0.03 IU/ml for both types of antibodies.
The assays were done in a randomized manner, and the laboratory technicians were blinded to the group to which the subject belonged.
Statistical analysis.
In Israel, the anti-PRP response was analyzed 1 month after the third dose by use of analysis of variance (ANOVA) after logarithmic transformation for comparison of the three groups, and Dunnett’s t test was used for the comparisons of PncT versus placebo and PncD versus placebo (11). In the Finnish study, the response was analyzed by use of ANOVA for the seven groups. Duncan’s multiple-range procedure was used to classify the seven groups, in particular, PncT, PncD at 3 μg, and placebo (10).
P values of 0.05 were considered significant. Correlations between antibody response to PRP and TT or diphtheria toxoid were calculated with the Pearson coefficient.
RESULTS
Of the 75 infants recruited in Israel, 39 (52%) were males. The mean age at enrollment was 2.1 months; the mean ages at second and third injections were 4.0 and 6.1 months, respectively. Seventy-four of the 75 enrolled subjects (99%) completed the follow-up at 1 month after the third dose; 70 (93%) received the vaccination at 12 months of age and completed the follow-up at 13 months of age according to the protocol.
Two hundred infants were recruited in Finland; 102 (51%) of them were males. The mean age at enrollment was 2.5 months; the mean ages at second and third injections were 4.3 and 6.1 months, respectively. Ninety-nine percent (197 of 200) of the enrolled subjects completed the follow-up at 1 month after the third dose.
Effect of PncT on anti-PRP antibody responses.
The anti-PRP antibody response was significantly impaired when PRP-T was administered simultaneously with DTP and the tetravalent PncT (Table 3). In Israel, the geometric mean concentrations (GMC) of anti-PRP antibodies in the PncT group were 0.86 μg/ml after the second dose and 2.81 μg/ml after the third dose, versus 1.19 and 4.62 μg/ml in the PncD group and 2.95 and 6.62 μg/ml in the placebo group, respectively (ANOVA for the three groups: P = 0.0085 after the second dose and P = 0.0218 after the third dose). Dunnett’s t test revealed significant differences between PncT and placebo but not between PncD and placebo or between PncD and PncT.
TABLE 3.
Anti-PRP and antitetanus antibody responses of infants receiving PncT, PncD, or placebo at 2, 4, and 6 months of agea
| Country and mo (time related to dose) | Anti-PRP
antibodies
|
Antitetanus antibodies
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| GMC
(μg/ml)
|
% with concn ≥1.0 μg/ml
|
GMC
(mIU/ml)
|
% with concn ≥1.0 μg/ml
|
|||||||||
| PncT | PncD | Placebo | PncT | PncD | Placebo | PncT | PncD | Placebo | PncT | PncD | Placebo | |
| Israel | ||||||||||||
| 2 (pre) | 0.17 | 0.36 | 0.20 | 13 | 28 | 8 | 0.08 | 0.12 | 0.21 | 0 | 13 | 4 |
| 4 (post 1) | 0.18 | 0.23 | 0.30 | 8 | 14 | 26 | 0.06 | 0.07 | 0.09 | 5 | 0 | 0 |
| 6 (post 2) | 0.86b | 1.19b | 2.95b | 44 | 64 | 74 | 0.26 | 0.24 | 0.25 | 16 | 10 | 13 |
| 7 (post 3) | 2.81c | 4.62c | 6.62c | 83 | 91 | 96 | 1.85 | 2.08 | 1.75 | 61 | 74 | 71 |
| Finland | ||||||||||||
| 2 (pre) | 0.07 | 0.08 | 0.12 | 0 | 4 | 4 | 0.65 | 0.60 | 0.54 | 33 | 28 | 22 |
| 4 (post 1) | 0.09 | 0.09 | 0.11 | 0 | 4 | 6 | 0.31 | 0.29 | 0.26 | 8 | 8 | 1 |
| 6 (post 2) | 0.93 | 1.64 | 1.51 | 56 | 68 | 67 | 0.60 | 0.67 | 0.50 | 32 | 28 | 22 |
| 7 (post 3) | 7.18 | 12.50 | 11.0 | 92 | 100 | 96 | 3.49 | 3.99 | 4.27 | 96 | 96 | 96 |
| Booster (Israel) | ||||||||||||
| 12 (prebooster) | 0.55d | 1.44d | 1.37d | 24 | 64 | 71 | 0.36 | 0.42 | 0.24 | 12 | 9.5 | 0 |
| 13 (postbooster) | 10.95e | 20.13e | 20.84e | 96 | 100 | 100 | 4.37 | 6.94 | 4.95 | 100 | 100 | 96 |
Each group contained 25 subjects, except for the Finnish placebo group, which contained 50 subjects.
P = 0.0085 among the three groups.
P = 0.0218 among the three groups.
P = 0.0052 among the three groups.
P = 0.076 among the three groups.
The same trend was observed in Finland with the 3-μg dose. After the second and third doses, the GMC of anti-PRP antibodies were 0.93 and 7.18 μg/ml, respectively, in the PncT group, versus 1.64 and 12.50 μg/ml in the PncD group and 1.51 and 11.0 μg/ml, respectively, in the placebo group (nonetheless, the differences between the groups did not reach statistical significance).
When the proportions of infants with anti-PRP antibody levels of ≥1.0 μg/ml were compared, the same tendency toward a lower proportion in the PncT group (83% in Israel and 92% in Finland after the third dose) than in the PncD group (91 and 100%, respectively) or the placebo group (96% in both sites) was found.
In Israel, infants were vaccinated with DTP–IPV–PRP-T and pneumococcal polysaccharide vaccine at 12 months of age. Before this booster vaccination, both the GMC and the proportion of children having antibody levels of ≥1.0 μg/ml were significantly lower in the PncT group than in the PncD or placebo group (Table 3). One month after the booster, although all children but one had anti-PRP antibody concentrations of ≥1.0 μg/ml, the GMC in the PncT group was still only about half (10.95 μg/ml) that in the PncD (20.13 μg/ml) or placebo (20.84 μg/ml) group (ANOVA for the three groups: P = 0.076).
Dose effect of pneumococcal conjugate vaccines on anti-PRP antibody responses.
To examine whether the dosage of PncT could influence the responses to PRP and TT, we compared the antibody responses of four groups in Finland: PncT0 (placebo), PncT01 (1 μg of polysaccharide of each of the four serotypes included in the tetravalent vaccine), PncT03 (3 μg of polysaccharide of each serotype), and PncT10 (10 μg of polysaccharide of each serotype). Thus, the total TT protein loads for the vaccines administered at 2, 4, and 6 months (simultaneously with DPT–PRP-T) were 39, 48, 63, and 111 μg for PncT0 (placebo), PncT01, PncT03, and PncT10, respectively (Table 2).
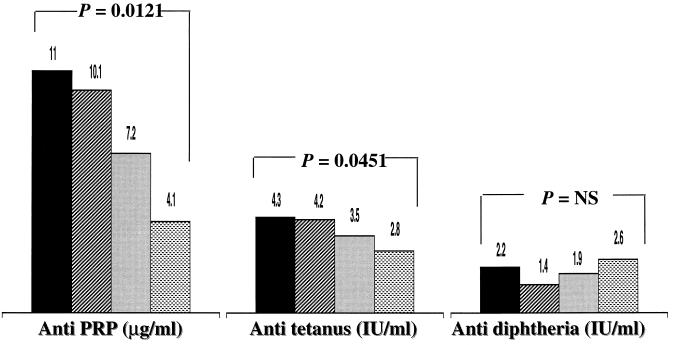
The concentrations of the anti-PRP antibodies elicited by these vaccines gradually decreased with increasing TT load (Fig. 1). After the third dose, the GMC of anti-PRP antibodies were 11.0, 10.1, 7.2, and 4.1 μg/ml for PncT0, PncT01, PncT03, and PncT10, respectively (ANOVA for the four groups: P = 0.0121). The same trend was seen even after the second dose (GMC of 1.5, 1.2, 0.9, and 0.6 μg/ml for PncT0, PncT01, PncT03, and PncT10, respectively; data not shown). In contrast, anti-PRP antibody concentrations did not decrease significantly with increasing PncD concentration. After the third dose, the GMC of anti-PRP antibodies were 11.0, 11.5, 12.5, and 7.8 μg/ml for PncD0, PncD01, PncD03, and PncD10, respectively.
FIG. 1.
Anti-PRP, antitetanus, and anti-diphtheria toxoid
antibodies after the third injection of one of the various doses of
PncT: placebo (PncT0) (▪); tetravalent PncT vaccine, 1
μg of each polysaccharide (PncT01) (▨); tetravalent
PncT vaccine, 3 μg of each polysaccharide (PncT03)
(░⃞); and
tetravalent PncT vaccine, 10 μg of each polysaccharide
(PncT10) ( ).
NS, not significant.
).
NS, not significant.
Similarly, a trend toward reduced proportions of infants with anti-PRP antibody levels of ≥1.0 μg/ml was seen with increasing PncT concentration (96, 96, 92, and 80% for PncT0, PncT01, PncT03, and PncT10, respectively). Such a trend was not seen with PncD (with which the respective proportions were 96, 92, 100, and 92%).
Effect of PncT on antitetanus antibody responses.
Although there were no significant differences in antitetanus antibody concentrations among the three study groups with regard to primary response or response to booster in either country when the 3-μg dose was used (Table 3), a significant trend toward a decrease was found with increasing doses of PncT in Finland (GMC of 4.3, 4.2, 3.5, and 2.8 IU for PncT0, PncT01, PncT03, and PncT10, respectively; P = 0.045, as determined by ANOVA for the four groups) (Fig. 1).
Since an interference effect of PncT was found for anti-PRP antibodies in infants immunized with PRP-T and DTP and a similar trend was found for antitetanus antibodies, we examined whether a correlation between anti-PRP and antitetanus antibody responses was present in the 200 Finnish vaccine recipients. A correlation was indeed found between anti-PRP and antitetanus antibody concentrations in each conjugate group (Table 4). In other words, the subjects who had low anti-PRP antibody concentrations tended to be the same ones who also had low antitetanus antibody concentrations. This linkage was not found with the anti-PRP antibody response and the anti-diphtheria toxoid antibody response.
TABLE 4.
Correlation between antibody responses to PRP and TT or diphtheria toxoid among Finnish infants receiving PncT, PncD, or placebo
| Correlation of: | PncT group (n =
75)
|
PncD group (n =
75)
|
Placebo group (n = 50)
|
|||
|---|---|---|---|---|---|---|
| r | P | r | P | r | P | |
| Anti-PRP and antitetanus antibodies | 0.395 | 0.0005 | 0.638 | ≤0.0001 | 0.608 | ≤0.0001 |
| Anti-PRP and anti-diphtheria toxoid antibodies | −0.015 | 0.897 | 0.231 | 0.046 | 0.137 | 0.347 |
DISCUSSION
New vaccines against encapsulated bacteria are based on the chemical coupling of capsular oligo- or polysaccharides to carrier proteins. For practical reasons, the future of vaccination schedules will require the simultaneous administration of these various conjugates. So far, only a few protein carriers have been used for the preparation of these vaccines. Therefore, the addition to the routinely used Hib vaccine of conjugate vaccines against pathogens such as Neisseria meningitidis and S. pneumoniae could require the simultaneous administration of several polysaccharides conjugated to the same protein carrier. It is thus of primary importance to analyze possible interference between conjugates that share the same carrier moiety. In the present study, such an issue was addressed for infants who received either the DTP or the PRP-T vaccine alone or together with a vaccine containing pneumococcal polysaccharide antigens conjugated to TT or diphtheria toxoid. Our results clearly showed that the anti-PRP antibody response decreased significantly when the PRP-T conjugate was administered together with DTP and the tetravalent PncT conjugates. This decrease was dose dependent and also affected the antitetanus antibody response. Notably, this suppression was carrier specific, since it was more accentuated with increasing load of TT and was not observed after simultaneous administration of PRP-T with pneumococcal polysaccharide conjugated to diphtheria toxoid. This last finding excludes the possibility that the suppression was due to some immunosuppressive properties of the pneumococcal polysaccharides.
Additional support for the suggestion that TT overload interferes with the anti-PRP antibody response can be derived from the finding that when PncT03 (tetravalent PncT vaccine with 3 μg of polysaccharide of each serotype) was administered to Israeli and Finnish infants, the interference with the anti-PRP antibody response was more accentuated in the Israeli group (Israeli DTP contains twice the concentration of TT as Finnish DTP).
Several mechanisms can be proposed. First, the decrease in the anti-PRP antibody response seems to be related to the load of TT injected as free or conjugate proteins: the largest decrease in the anti-PRP antibody response was observed with the highest dose of PncT conjugates. The load of TT injected seems to affect the antitetanus antibody response as well, since a correlation between anti-PRP antibody and antitetanus antibody concentrations was found (in other words, vaccinees who tended to have lower anti-PRP antibody levels also had lower antitetanus antibody levels).
Second, previous studies indicated that the development of an immune response against the carrier can interfere with the response to saccharide-protein vaccines (3–5, 13, 23). Preimmunization with the protein carrier can reduce antibody responses both to the carrier and to Hib polysaccharide epitopes (5). Such a phenomenon may be related to the epitopic suppression initially described with 2,4,6-trinitrobenzenesulfonic acid hapten (TNP) or 2,4,6-dinitrophenyl hapten (DNP) (15, 16) or peptide epitopes (26). This suppression is mediated by the clonal dominance of B cells specific for carrier epitopes (20, 27). Although these conclusions were obtained with carrier-primed animals, we can now propose that similar mechanisms control the responses to simultaneously administered conjugates.
Whereas optimal doses of TT will rapidly stimulate the activation and proliferation of carrier-specific B-cell clones that will then efficiently take up the conjugate and present it to T cells, polysaccharide-specific B-cell clones are certainly much less frequent and will never reach the same frequency as carrier-specific B-cell clones. Thus, the extent of the decrease in the response against the polysaccharide will be proportional to the intensity of the immune response to the carrier (i.e., dependent upon its immunogenicity and on the dose) and will be more pronounced with saccharide epitopes for which a low frequency of B-cell precursors exists. This reasoning could explain the results of a recent study where an overload of homologous protein carrier at the site of injection reduced the immunogenicity in mice of two experimental conjugates (12). Indeed, a 12-valent conjugate Escherichia coli vaccine and an 8-valent Staphylococcus aureus vaccine consisting of polysaccharide conjugated to protein induced significantly lower antibody concentrations than each monovalent conjugate administered separately. A similar reduction in immunogenicity was seen when free protein carrier was injected together with the conjugate vaccine.
Further support for the theory of carrier suppression can be found in a recently presented work by Greenberg et al. (14). The authors coadministered to infants a heptavalent pneumococcal conjugate vaccine (which was conjugated to an outer membrane protein of N. meningitidis group B [OMPC]) with either PRP conjugated to OMPC (PRP-OMPC) or PRP conjugated to a mutant diphtheria toxin polypeptide (PRP-CRM) at 2, 4, and 6 months. After the primary immunization series (at 7 months), anti-pneumococcal polysaccharide antibody concentrations to some serotypes were significantly lower in infants receiving PRP-OMPC (sharing the same carrier as the heptavalent pneumococcal vaccine) than in those receiving PRP-CRM.
We cannot rule out the possibility that the mechanism of epitopic suppression based on the dominance of carrier-specific B cells played at least in part a role in the currently described phenomenon. We believe, however, that its role, if any, is only minor, based on the finding that the decrease in the anti-PRP antibody response was linked to the decrease in the antitetanus antibody response.
Third, an elevated concentration of antibodies to the protein carrier could also interfere with conjugate immunogenicity through antigen capture or modulation of antigen presentation. Barington et al. (3) showed that maternal antibodies to TT inhibited the response of human infants to the polysaccharide moiety of Hib conjugate vaccine. However, such an effect was not seen by others (19).
Fourth, the finding that the injection of high doses of PncT affected both anti-carrier and anti-PRP antibody responses may reflect inadequate T-helper-cell activity. Several recent studies have indicated that the attachment of oligosaccharides to peptides profoundly affects binding to major histocompatibility complex class II molecules and peptide immunogenicity (22). Moreover, it has been shown that T cells specifically recognize synthetic glycopeptides (18). It could therefore be suggested that TT modified by conjugation to Pnc stimulates a T-cell repertoire that does not cross-react with TT-specific T cells. Since both PncT and PRP-T conjugates would be processed and presented by TT-specific B cells, the processing of these two antigens could lead to different glycopeptides that may compete for major histocompatibility complex class II binding.
Although the present study indicates that the simultaneous administration of PncT and PRP-T conjugates may decrease the anti-PRP antibody response, the levels of anti-PRP antibodies induced by such combined vaccines still exceeded the concentrations needed for protection against invasive Hib disease. In addition, clear evidence for the induction of memory toward the polysaccharide antigens was obtained. While the present study analyzed the interference between PRP-T and a tetravalent pneumococcal conjugate vaccine, the licensed pneumococcal vaccine will most probably contain more than seven pneumococcal serotypes. This increase in the amount of TT used for immunization could potentially reduce the anti-PRP or even the antitetanus antibody response to an unacceptable level. Thus, although the safety and immunogenicity of TT make it suitable as a protein carrier for human vaccines, increasing its quantity in multicomponent vaccines might affect the efficiency of such vaccines. Restriction of the content of the protein carrier in conjugate vaccines is desirable, as is the development of other protein or nonprotein carriers for saccharide conjugate vaccines.
ACKNOWLEDGMENTS
This work was supported in part by a grant from Pasteur Mérieux Connaught.
Fabrice Bailleux was instrumental in the statistical analysis. Helena Käyhty and Rose Marie Ölander were responsible for the antibody assays in Finland. P. Helena Mäkelä was of great help in our discussions. Orly Zamir was instrumental in conducting the studies in Israel.
REFERENCES
- 1.Ahman, H., H. Käyhty, H. Lehtonen, O. Leroy, J. Froeschle, and J. Eskola. Streptococcus pneumoniae capsular polysaccharide-diphtheria toxoid conjugate vaccine is immunogenic in early infancy and able to induce immunologic memory. Pediatr. Infect. Dis. J., in press. [DOI] [PubMed]
- 2.Ahman H, Käyhty H, Leroy O, Froeschle J, Eskola J. Program and Abstracts of the 35th Interscience Conference on Antimicrobial Agents and Chemotherapy. Washington, D.C: American Society for Microbiology; 1995. Immunogenicity of tetravalent pneumococcal (Pnc) conjugate vaccines (PncD, PncT) in Finnish infants, abstr. G69; p. 170. [Google Scholar]
- 3.Barington T, Gyhrs A, Kristensen K, Heilmann C. Opposite effects of actively and passively acquired immunity to the carrier on responses of human infants to a Haemophilus influenzaetype b conjugate vaccine. Infect Immun. 1994;62:9–14. doi: 10.1128/iai.62.1.9-14.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Barington T, Kristensen K, Henrichsen J, Heilmann C. Influence of prevaccination immunity on the human B-lymphocyte response to a Haemophilus influenzaetype b conjugate vaccine. Infect Immun. 1991;59:1057–1064. doi: 10.1128/iai.59.3.1057-1064.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Barington T, Skettrup M, Juul L, Heilmann C. Non-epitope-specific suppression of the antibody response to Haemophilus influenzaetype b conjugate vaccines by preimmunization with vaccine components. Infect Immun. 1993;61:432–438. doi: 10.1128/iai.61.2.432-438.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bloom B. Summary: a perspective on issues relating to future vaccines. Ann N Y Acad Sci. 1995;754:388–395. doi: 10.1111/j.1749-6632.1995.tb44475.x. [DOI] [PubMed] [Google Scholar]
- 7.Clemens J, Brenner R, Rao M. Interactions between PRP-T vaccine against Haemophilus influenzae band conventional infant vaccines. Lessons for future studies of simultaneous immunization and combined vaccines. Ann N Y Acad Sci. 1995;754:255–266. doi: 10.1111/j.1749-6632.1995.tb44458.x. [DOI] [PubMed] [Google Scholar]
- 8.Dagan R, Muallem M, Melamed R, Leroy O, Yagupsky P. Reduction of pneumococcal nasopharyngeal carriage in early infancy after immunization with tetravalent pneumococcal vaccines conjugated to either tetanus toxoid or diphtheria toxoid. Pediatr Infect Dis J. 1997;16:1060–1064. doi: 10.1097/00006454-199711000-00011. [DOI] [PubMed] [Google Scholar]
- 9.Dagan R, Melamed R, Zamir O, Leroy O. Safety and immunogenicity of tetravalent pneumococcal vaccines containing 6B, 14, 19F and 23F polysaccharides conjugated to either tetanus toxoid or diphtheria toxoid in young infants and their boosterability by native polysaccharide antigens. Pediatr Infect Dis J. 1997;16:1053–1059. doi: 10.1097/00006454-199711000-00010. [DOI] [PubMed] [Google Scholar]
- 10.Duncan D B. T-tests and intervals for comparisons suggested by the data. Biometrics. 1975;31:339–359. [Google Scholar]
- 11.Dunnett C W. A multiple comparisons procedure for comparing several treatments with a control. J Am Stat Assoc. 1955;50:1096–1121. [Google Scholar]
- 12.Fattom A, Cho Y H, Fuller S, Naso R. Program and Abstracts of the 36th Interscience Conference on Antimicrobial Agents and Chemotherapy. Washington, D.C: American Society for Microbiology; 1996. Overload of homologous carrier protein at the site of injection might cause a reduced immunogenicity of capsular polysaccharide conjugate vaccines, abstr. G93; p. 160. [Google Scholar]
- 13.Granoff, D. M., M. H. Rathore, S. J. Holmes, and P. D. Granoff. 1993. Effect of immunity to the carrier protein on antibody responses to Haemophilus influenzae type b conjugate vaccines. Vaccine 11(Suppl. 1):S46–S51. [DOI] [PubMed]
- 14.Greenberg D P, Zangwill K M, Partridge S, Chang S-J, Wong V K, Curry E S, Ward J I. Factors influencing the immunogenicity of a pneumococcal conjugate vaccine in infants. Pediatr Res. 1997;41:121A. . (Abstract 709.) [Google Scholar]
- 15.Herzenberg C A, Tokuhisha T, Herzenberg L A. Carrier-priming leads to hapten-specific suppression. Nature. 1980;285:664–667. doi: 10.1038/285664a0. [DOI] [PubMed] [Google Scholar]
- 16.Herzenberg L A, Tokuhisha T. Epitope-specific regulation. I. Carrier-specific induction of suppression for IgG anti-hapten antibody responses. J Exp Med. 1982;155:1730–1740. doi: 10.1084/jem.155.6.1730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Insel R A. Potential alterations in immunogenicity by combining or simultaneously administering vaccine components. Ann N Y Acad Sci. 1995;754:35–67. doi: 10.1111/j.1749-6632.1995.tb44436.x. [DOI] [PubMed] [Google Scholar]
- 18.Jensen T, Galli-Stampino L, Mouritsen S, Frische K, Peters S, Meldal M, Werdelin O. T cell recognition of Tn-glycosylated peptide antigens. Eur J Immunol. 1996;26:1342–1349. doi: 10.1002/eji.1830260625. [DOI] [PubMed] [Google Scholar]
- 19.Kurikka, S., R.-M. Olander, and H. Käyhty. Priming with diphtheria-tetanus-pertussis vaccine enhances the response to the Haemophilus influenzae type b tetanus conjugate vaccine in infancy. Vaccine, in press. [DOI] [PubMed]
- 20.Leclerc C, Schutze M-P, Deriaud E, Przewlocki G. The in vivo elimination of CD4+T cells prevents the induction but not the expression of carrier-induced epitopic suppression. J Immunol. 1990;145:1343–1349. [PubMed] [Google Scholar]
- 21.Melville-Smith M. Diphtheria and tetanus antitoxins. In: Wreghitt T G, Morgan-Capner P, editors. ELISA in the clinical microbiology laboratory. London, England: Public Health Laboratory Service; 1990. pp. 136–147. [Google Scholar]
- 22.Mouritsen S, Meldal M, Christiansen-Brams I, Elsner H, Werdelin O. Attachment of oligosaccharides to peptide antigen profoundly affects binding to major histocompatibility complex class II molecules and peptide immunogenicity. Eur J Immunol. 1994;24:1066–1072. doi: 10.1002/eji.1830240509. [DOI] [PubMed] [Google Scholar]
- 23.Peeters C C, Tenbergen-Meekes A-M, Poolman J T, Beurret M, Zegers B J, Rijkers G T. Effect of carrier priming on immunogenicity of saccharide-protein conjugate vaccines. Infect Immun. 1991;59:3504–3510. doi: 10.1128/iai.59.10.3504-3510.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Robbins J B, Schneerson R. Polysaccharide-protein conjugates: a new generation of vaccines. J Infect Dis. 1990;161:821–832. doi: 10.1093/infdis/161.5.821. [DOI] [PubMed] [Google Scholar]
- 25.Robbins J B, Parke J C, Jr, Schneerson R, Whisnant J K. Quantitative measurement of “natural” and immunization induced Haemophilus influenzaetype b capsular polysaccharide antibodies. Pediatr Res. 1973;7:103–110. doi: 10.1203/00006450-197303000-00001. [DOI] [PubMed] [Google Scholar]
- 26.Schutze M-P, Leclerc C, Jolivet M, Audibert F, Chedid L. Carrier-induced epitopic suppression, a major issue for future synthetic vaccines. J Immunol. 1985;135:2319–2322. [PubMed] [Google Scholar]
- 27.Schutze M-P, Deriaud E, Przewlocki G, Leclerc C. Carrier-induced epitopic suppression is initiated through clonal dominance. J Immunol. 1989;142:2635–2640. [PubMed] [Google Scholar]
- 28.Siber G R. Pneumococcal disease: prospects for a new generation of vaccines. Science. 1994;265:1385–1387. doi: 10.1126/science.8073278. [DOI] [PubMed] [Google Scholar]
- 29.Weinberg G A, Granoff D M. Polysaccharide-protein conjugate vaccines for the prevention of Haemophilus influenzaetype b disease. J Pediatr. 1988;113:621–631. doi: 10.1016/s0022-3476(88)80369-x. [DOI] [PubMed] [Google Scholar]