Abstract
An 18-month-old neutered male domestic shorthair cat, domiciled in the southwest of France, was first presented having suffered for a few days from dysorexia and vomiting. Abdominal palpation revealed lymph node enlargement. Cytological examinations of a fine needle aspirate demonstrated granulomatous inflammation with many non-staining elements consistent with mycobacteria. Diagnosis was confirmed by culture and polymerase chain reaction and Mycobacterium avium subspecies was isolated. Treatment was initiated with marbofloxacin, rifampicin and cefoxitin. There was a rapid clinical improvement. The cat suddenly died 2 months later. The main hypothesis is the administration of an inappropriate combination therapy that leads to the development of mycobacterial resistance. A volvulus and acute peritonitis secondary to the significant enlargement of a mesenteric lymph node were present at necropsy. Histopathological analysis of mesenteric lymph node, liver and spleen revealed multicentric granulomatous and severely necrotic lesions with numerous Ziehl–Neelsen positive intracytoplasmic elements.
The patient was an 18-month-old neutered male domestic shorthair cat, with indoor and outdoor access, which was apparently in good condition with a body weight of 4 kg. The cat was presented having suffered for a few days from inappetence and vomiting. The clinical examination of abnormalities was limited to the presence of abdominal masses on palpation, which could be consistent with abdominal lymph nodes enlargement.
Cell blood count, serum chemistry, urine analysis and thoracic radiographs were unremarkable. The results of enzyme-linked immunosorbent assays for feline leukaemia virus (FeLV) antigen and feline immunodeficiency virus (FIV) antibodies on blood were negative. These results were later confirmed by Real-Time polymerase chain reaction (RT-PCR) on a mesenteric lymph node biopsy.
Ultrasonographic examination of the abdomen confirmed the suspicion of mesenteric lymphadenopathy (2.3 cm in length) and revealed modifications in the digestive tract and kidneys with hyperechoic cortex.
The clinical picture suggested a disseminated neoplastic process, like a lymphoma. In order to confirm this hypothesis, fine needle tissue aspirates were collected from mesenteric lymph nodes by means of ultrasound guidance. Slides were prepared for cytological evaluation using the May–Grünwald–Giemsa stain. Preparations obtained were highly cellular; a significant granulomatous inflammation was present. At high power field (Fig 1), numerous thin, negative-staining (3–4 μm in length) rods were observed in the cytoplasm of large macrophages as well as extracellularly. Mycobacteriosis was strongly suspected. To confirm this, nucleic acid extraction, PCR amplification and sequencing were performed.
Fig 1.
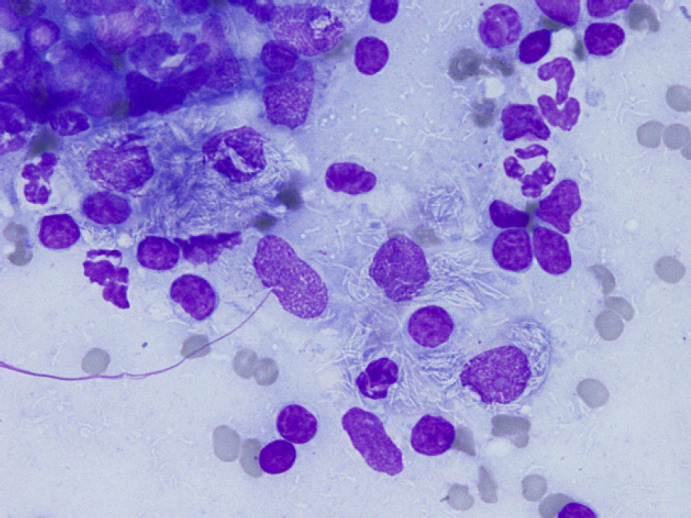
May–Grünwald–Giemsa (×1000). Numerous cells with a low nuclear-to-cytoplasmic ratio, pale blue staining and granular cytoplasm with indistinct borders, consistent with macrophages. Their cytoplasms contain phagocytised elements (debris, neutrophils) and non-staining bacterial rods consistent with the appearance of mycobacteria.
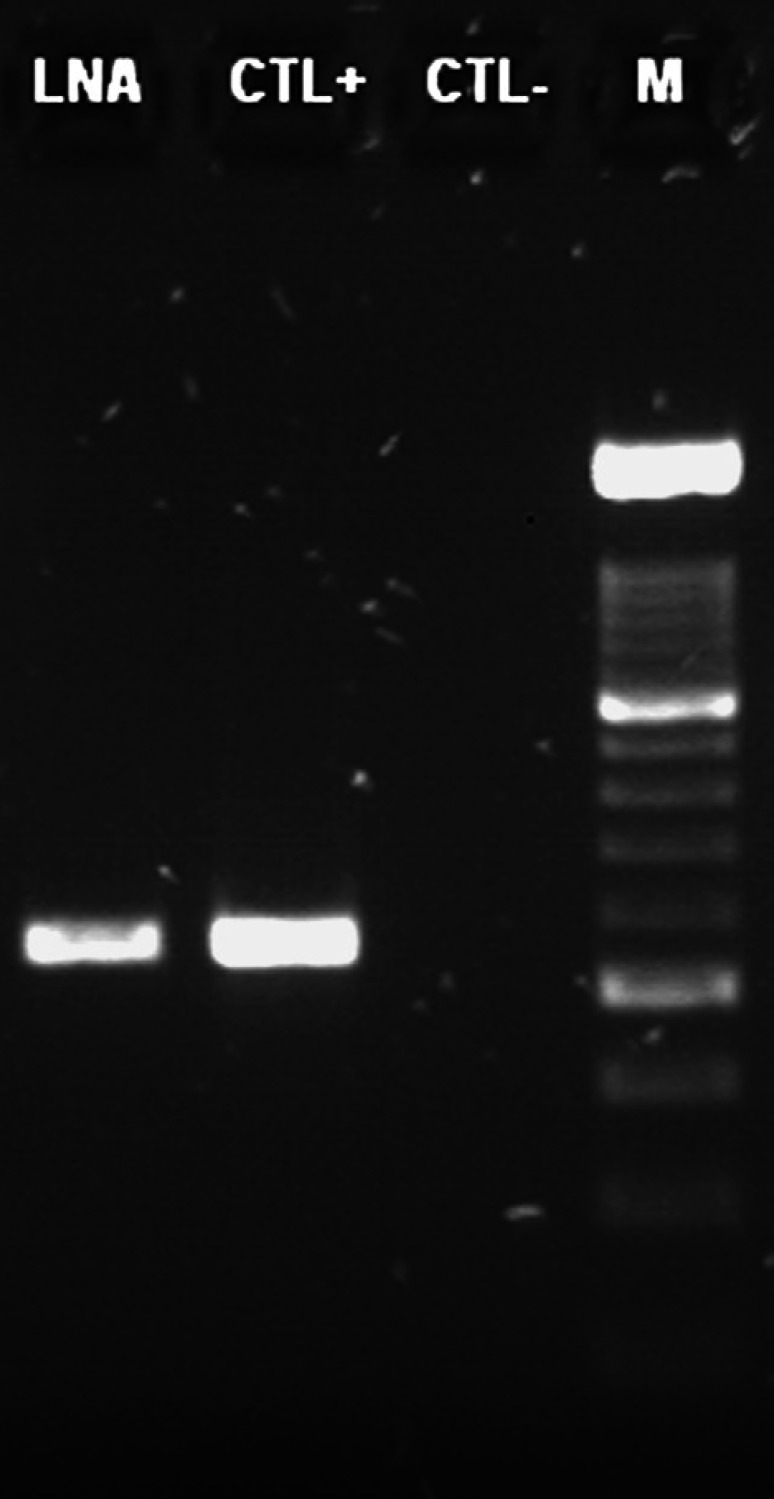
Total DNA was extracted from lymph node aspirate using a silica-based extraction kit as recommended by the supplier (Macherey–Nagel, Hoerdt, France). Sequences from the 5′ end of the 16S ribosomal gene of mycobacteria available from the Genbank database were aligned (data not shown) in order to design a set of primers (Mycoseq1F: acacatgcaagtcgaacg; Mycoseq1R tgagatttcacgaacaacg). A PCR was performed following the Platinum Taq DNA polymerase supplier recommendations (Invitrogen, Paisley, UK). Cycling conditions were as follows: 95°C 5 min/35× [94°C 15 s/55°C 20 s/72°C 1 min]/72°C 5 min. PCR products were separated by electrophoresis onto a 1.5% agarose gel (Fig 2). The set of Mycoseq1F/Mycoseq1R primers produced a 550-pb fragment. The typical band was obtained for the lymph node aspirate and the positive control (DNA from mycobacterium culture), whereas no signal appeared for the negative control (water), confirming the absence of cross-contamination (Fig 2). The 550-pb PCR product was sequenced on both strands (MilleGen, Labège, France). The sequence analysis showed 100% nucleotide identity (513/513) with Mycobacterium avium sequences from the four subspecies avium, hominissuis, sylvaticum and paratuberculosis (respective Genbank accession numbers: EF521895, EF521892, EF521891 and EF521896). The identity score fell to 98.4% (505/513) by comparison with Mycobacterium intracellulare and to 97.3% (499/513) with Mycobacterium tuberculosis (respective accession numbers: AF547939 and AF547977). The molecular identification determined Mycobacterium avium infection. This result was also confirmed by microbiological culture. Following decontamination and fluidification, the lymph node was inoculated on Löwenstein–Jensen and Coletsos medium at 37°C. A mycobacterial species was first detected after 6 weeks of incubation.
Fig 2.
PCR detection of Mycobacterium species DNA. LNA: lymph node aspirate; CTL+ = positive control; CTL− = negative control; M = molecular weight marker size.
During hospitalisation, the cat rapidly exhibited a total anorexia and diarrhoea. Triple-antibiotic therapy was initiated with marbofloxacin (Marbocyl; Vétoquinol), rifampicin (Rifadine; Sanofi Aventis France), and cefoxitin (Zeclar; Abbott France). A clinical improvement was observed 2 days after the onset of the treatment. After 1 week of triple-antibiotic therapy, the patient returned home and continued the same treatment. Seven weeks after the initial diagnosis of mycobacteriosis, it was presented to the veterinarian for emergency treatment. Resuscitation was unsuccessful and the cat died 2 h after its admission.
At necropsy, enlargement of the mesenteric lymph nodes was severe (more than 5 cm) and was associated with torsion of a small intestine loop, secondary venous infarction and focal extensive acute fibrinous peritonitis. The cut section of the lymph node showed an extensive central caseous necrosis. Tissue samples of liver, spleen, mesenteric lymph nodes and digestive tract were fixed in 10% neutral buffered formalin for histopathological examination. Granulomatous and severely necrotic inflammatory reaction was present in the mesenteric lymph node. Numerous acid-fast bacilli were detected in the cytoplasm of some macrophages in Ziehl–Neelsen stained sections of lymph nodes, liver and spleen. According to the conclusions of the necropsy, the cat probably died from the secondary complications of intestinal torsion and peritonitis rather than of the mycobacterial disease itself.
Mycobacteria are Gram-positive, aerobic, non-spore-forming bacteria. The high lipid content of their cell wall is responsible for their resistance to many antibiotics and their staining characteristics (acid-fast stains). Mycobacteria species are responsible for chronic disease inducing a granulomatous immune response.
In simple terms, mycobacteria can be divided into three groups:
Mycobacterium leprae, that causes leprosy lesions and is impossible to culture using routine laboratory methods.
Mycobacteria that are causative agents of tuberculosis (M bovis, M tuberculosis, M canettii, M microti, M pinnipedii and M africanum).
Mycobacteria other than tuberculosis (MOTT), slow- and rapid-growing opportunistic Mycobacteria species; M avium subspecies belongs to the slow-growing group. 1
M avium and related species, like M intracellulare, belong to the Mycobacterium avium complex (MAC), a heterogeneous group of ubiquitous, saprophytic and resistant organisms. M avium, the main species that causes avian mycobacteriosis, is an opportunistic pathogen in immunocompromised people (human immunodeficiency virus (HIV) positive), causing disseminated disorders. The main sources of contamination remain waters and soils. The distribution of lesions reflects the route of primary exposure: pulmonary infection most probably occurs via aerosols, gastrointestinal clinical signs probably result from the ingestion of M avium complex in natural waters and cutaneous lesions are probably secondary to a breach in the integument. Pulmonary or gastrointestinal infection is highly predictive of dissemination to blood. 2,4
Only a few cases of generalised disease in cats and dogs have been reported in the last 45 years. In the study of Baral, 12 cases were described in North America and Australia between 1994 and 2005. 3 Clinical signs were similar to those in our case, with typically digestive disorders (dysorexia, weight loss and diarrhoea) associated with mesenteric lymphadenomegaly. Additional clinical features included respiratory signs and peripheral adenopathy. Moreover, this study proposed the hypothesis of a predisposition of certain lines of Abyssinian and Somali cats. They would be more sensitive to infection with slow-growing mycobacteria because of a familial immunodeficiency.
Disseminated mycobacterial disease is usually associated with defects in cell-mediated immunity. Infection with M avium complex in acquired immune deficiency syndrome (AIDS) patients seems to be closely related to a declining CD4+ cell count. 4 In cats, the hypothesis of CD4+ lymphocytopenia was also evoked by the team of Meeks. 5 Relating to our case, peripheral blood lymphocyte subset analysis performed by flow cytometry would have been interesting to identify abnormal CD4+: CD8+ T lymphocytes ratio.
The great variety of mycobacterial species and subspecies leads to significant clinical polymorphism, due to the preferential localisation of the mycobacteria and the extent of the infection (localised/disseminated). The publications relating to isolated cases describe cats affected by panniculitis, 6–8 cutaneous and/or subcutaneous nodules, 9,10 granulomatous pneumonia, 2,11 pyogranulomatous neuritis, 12 or irido-cyclitis. 13
With regard to M avium, cats can be treated with a triple-antibiotic therapy. According to the studies the surgical excision of granuloma or lymph nodes when possible would have a positive impact on therapeutic success. Unlike the localised form, for which a complete cure is possible, the disseminated form has a worse prognosis with a variable response to treatment: successful outcomes in a few cases: 7 years of survival time for one case with M xenopi. 5 Recurrence and/or death can arise after a variable period of time: from 3 months to 2 years following therapy. 3,13 Using a combination of antibiotics is very important for successful treatment and minimising development of resistance, which could potentially endanger human patients. 14 In our case, an inappropriate combination therapy was chosen according to the availability and familiarity of the drugs rather than on their real efficiency. Indeed, the contribution of cefoxitin is not certain and marbofloxacine is a fluoroquinolone that has no place in the treatment of MAC infections. Rifampicine was the only drug of the combination that was really effective. Finally, the poor therapy probably contributed to the development of resistance and the short success of the treatment.
In France, the diagnosis of mycobacteriosis in a domestic carnivore is only subject to compulsory notification if the mycobacterium belongs to the M tuberculosis complex, in which case euthanasia is recommended for public health reasons (French Ministerial Circular of 17.03.70).
The risk of transmission to human beings of mycobacteria belonging to the avium-intracellulare complex is supposed to be low and mainly concerns the immunocompromised people (HIV). 4,14,15 The risk of zoonotic transmission from pets remains controversial and unquantified. However, M avium paratuberculosis has also been incriminated as a possible causative agent in Crohn's disease, a severe granulomatous ileocolitis of humans, and in chronic gastrointestinal disorders in dogs. 16
Lymph node cytology is the first key stage in the diagnostic step, as it can eliminate the clinical hypothesis of lymphoma and suggests an infectious disease. This non-invasive complementary examination is easy to perform, rapid, inexpensive and does not require anaesthesia. A strong suspicion of mycobacteriosis enables an appropriate treatment to be administered immediately (triple therapy with specific antibiotics instead of chemotherapy), specific tests to be requested (PCR and culture) and the zoonotic risk to be taken into account.
Cats with disseminated mycobacteriosis generally have a poor prognosis, even though successful outcomes have been described, thanks to appropriate combination therapy (clarithromycin, clofazimine). New agents, such as moxifloxacin or linezolid also seem promising.
Acknowledgement
We thank Dr A Bogey (Billiemaz laboratory, Toulon, France) for her technical support.
References
- 1.Euzéby J.P. Dictionnaire de bactériologie vétérinaire, http://www.bacterio.cict.fr/bacdico.
- 2.Foster S.F., Martin P., Alan G.S., et al. Lower respiratory tract infections in cats: 21 cases (1995–2000), J Feline Med Surg 6, 2004, 167–180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Baral R.M., Metcalfe S.S., Krockenberger M.B., et al. Disseminated Mycobacterium avium infection in young cats: overrepresentation of Abyssinian cats, J Feline Med Surg 8, 2006, 23–44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Falkinham J.O. Epidemiology of infection by non-tuberculous mycobacteria, Clin Microbiol Rev 9, 1996, 177–215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Meeks C., Levy J.K., Crawford P.C., et al. Chronic disseminated Mycobacterium xenopi infection in a cat with idiopathic CD4+ T lymphocytopenia, J Vet Intern Med 22, 2008, 1043–1047. [DOI] [PubMed] [Google Scholar]
- 6.Beccati M., Peano A., Gallo M.G. Pyogranulomatous panniculitis caused by Mycobacterium alvei in a cat, J Small Anim Pract 48, 2007, 664. [DOI] [PubMed] [Google Scholar]
- 7.Verde M. Zoonotic dermatoses in cats. Proceedings of the North American Veterinary Conference; 2005; Florida.
- 8.Malik R. Feline panniculitis due to rapidly-growing mycobacteria. Proceedings of the 28th World Small Animal Veterinary Association Congress; 2003; Bangkok, Thailand.
- 9.Henderson S.M., Baker J., William R., Gunn-Moore D.A. Opportunistic mycobacterial granuloma in a cat associated with a member of the Mycobacterium terrae complex, J Feline Med Surg 5, 2003, 37–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Elsner L., Wayne J., O’Brien C.R., et al. Localised Mycobacterium ulcerans infection in a cat in Australia, J Feline Med Surg 10, 2008, 407–412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Couto S.S., Artacho C.A. Mycobacterium fortuitum pneumonia in a cat and the role of lipid in the pathogenesis of atypical mycobacterial infections, Vet Pathol 44, 2007, 543–546. [DOI] [PubMed] [Google Scholar]
- 12.Paulsen D.B., Kern M.R., Weigand C.M. Mycobacterial neuritis in a cat, J Am Vet Med Assoc 10, 2000, 1589–1591. [DOI] [PubMed] [Google Scholar]
- 13.Dietrich U., Arnold P., Guscetti F., et al. Ocular manifestation of disseminated Mycobacterium simiae infection in a cat, J Small Anim Pract 44, 2003, 121–125. [DOI] [PubMed] [Google Scholar]
- 14.Waddel L.A., Rajic A., Sargeant J., et al. The zoonotic potential of Mycobacterium avium spp paratuberculosis: a systematic review, Can J Public Health 99, 2008, 145–155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Legrand E, Sola C, Rastogi N. Le complexe Mycobacterium avium-intracellulare: marqueurs phénotypiques et génotypiques et les bases moléculaires de la transmission inter-espèces. 3e colloque du réseau international des Instituts Pasteur et instituts associés; 1999 Oct 14–15; Paris, France.
- 16.Glanemann B., Schönenbrücher H., Bridger N., Abdulmawjood A., Neiger R., Bülte M. Detection of Mycobacterium avium subspecies paratuberculosis-specific DNA by PCR in intestinal biopsies of dogs, J Vet Intern Med 22, 2008, 1090–1094. [DOI] [PubMed] [Google Scholar]