Abstract
Importance
Atrial fibrillation (AF) is an established risk factor for ischaemic stroke. The introduction of continuous cardiac rhythm monitoring devices has enabled detection of brief and asymptomatic episodes of AF.
Observations
The search yielded 727 studies, 11 of which met the inclusion criteria. Four studies suggested a strong temporal association between episodes of AF and stroke, while seven indicated a weak relationship. The conflicting nature of the studies may be attributed to inconsistencies in ischaemic stroke verification (n=5/11), event rate and power (n=6/11) and lack of controlling for anticoagulation (n=10/11), mitigating the relationship between AF episodes and stroke.
Conclusions and relevance
The temporal relationship between AF and stroke still remains unclear due to varying study methodology, lack of control for anticoagulation and inconsistent stroke subtyping. Our review identifies limitations to the current literature and makes recommendations for future studies assessing the temporal relationship between AF episodes and cardioembolic stroke.
Keywords: STROKE, CARDIOLOGY
WHAT IS ALREADY KNOWN ON THIS TOPIC
The association between atrial fibrillation (AF) and stroke is widely known; however, knowledge on the temporal relationship is less understood.
WHAT THIS STUDY ADDS
This review provides new insight into the association of AF and stroke by critically appraising the quality of studies and comparing methodologies to assess if there is a true temporal relationship.
HOW THIS STUDY MIGHT AFFECT RESEARCH, PRACTICE OR POLICY
This review suggests that the temporal relationship remains unclear and suggests study measures such as control for anticoagulation, AF burden threshold and AF time that should be implemented for future research to ensure consistency in research. Clinicians can then tailor their treatment based on an individualised risk for AF and occurrence of stroke.
Introduction
Cardiac monitoring technology has granted clinicians the ability to view a continuous timeline of heart rhythm over months or years compared with conventional 12-lead ECGs and Holter monitors. These devices vary from pure monitoring devices such as implantable loop recorders to newer interventional devices with atrial rhythm sensing capacity This allows detection of common deadly arrhythmias such as transient episodes of atrial fibrillation (AF). The usefulness of these devices extends beyond simple AF diagnosis. We can also examine the timing of atrial events in relation to thromboembolic events, particularly thromboembolic stroke and peripheral embolism.1 The literature on this topic is, however contradictory.
It is important to identify whether there is a strong temporal relationship as it would strengthen the causality argument of AF, leading to stroke and imply that stopping the arrhythmia could reduce stroke risk.
This review aims to examine the evidence of temporal relationship between AF and thromboembolic events in population with devices capable of sensing atrial arrhythmias. We analyse the literature, discuss qualities of studies needed to elicit a true temporal relationship and finally outline future directions.
Methods
Database selection and search strategy
Two investigators (RK and VT) identified potentially eligible studies in MEDLINE from January 2011 until 31 August 2021. Key search concepts involved: atrial arrhythmia, thromboembolic events, cardiac monitoring devices and temporal information.
Study selection
Articles were uploaded in Covidence software and suitability was assessed independently by the two reviewers using inclusion and exclusion criteria.
Data extraction
Data were extracted on the following predetermined criteria, namely: sample size, type of event (ie, type of stroke and systemic emboli), number of these events, whether AF history was excluded or included, monitoring device type and author findings and conclusions.
Studies were then critically appraised based on predetermined criteria:
A precise temporal window for relationship (eg, within 5 or 30 days).
A positive AF burden (ie, how long AF has to occur before the patient is considered to have AF on that day).
Reporting and correction for patients using anticoagulation and antiplatelets.
The stroke risk of included population using an established risk stratification scheme (eg, CHADS2 or CHA2DS2-VASc).
How AF episodes were adjudicated.
How the diagnosis of ischaemic stroke was determined and adjudicated.
Article described whether any stroke subtyping had been done (ruling out other causes of embolic stroke, for example, patent foramen ovale/carotid disease).
Results
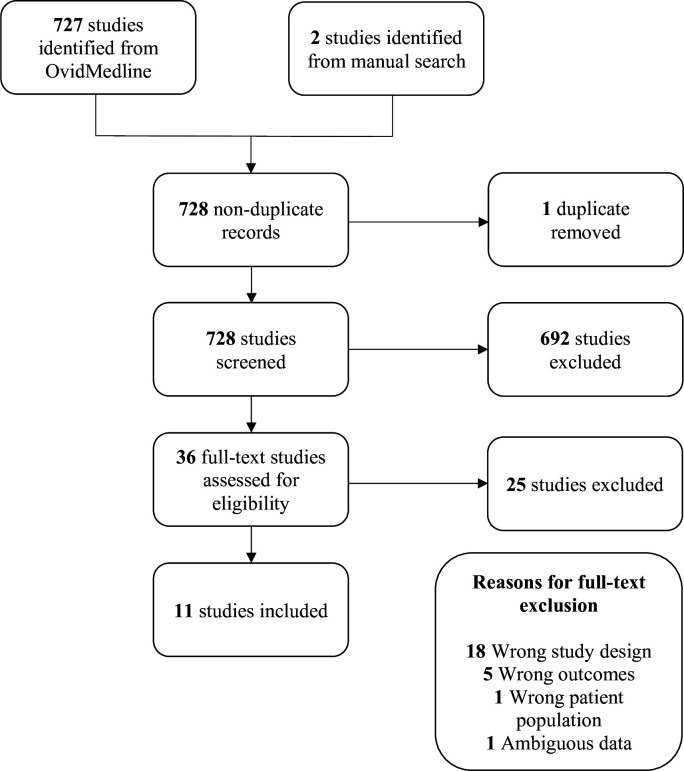
The database search yielded 727 results shown in figure 1 and an additional two studies identified through manual searching. After title/abstract screening, 36 papers were assessed for full-text review and a total of 11 studies were included in the final analysis. One paper was excluded after extraction due to ambiguous data.2 Nine of the included studies were observational and two were randomised clinical trials. Patients were included if they had at least one stroke risk factor such as age ≥ 65, hypertension or a prior stroke. Thromboembolic events of each study ranged from 6 to 891. A list of the studies appraised with the study validation criteria is outlined in table 1.
Figure 1.
PRISMA flow diagram of articles selected for inclusion. PRISMA, Preferred Reporting Items for Systematic Review and Meta-Analysis Protocols.
Table 1.
Summary of studies appraised study validation criteria
| Authors | Temporal window | AF burden | Control for antiplatelet and antithrombotic | Stroke risk stratification | AF episode adjudication | Ischaemic stroke adjudication | Ischaemic stroke subtyping |
| Moubarak et al6 | |||||||
| Turakhia et al 3 | |||||||
| Cuadrado-Godia et al5 | |||||||
| Singer et al4 | |||||||
| Dauod et al7 | |||||||
| Shanmugam et al10 | |||||||
| Brambatti et al8 | |||||||
| Martin et al11 | |||||||
| Witt et al9 | |||||||
| van Velzen et al20 | |||||||
| Svendsen et al12 |
 Studies adhered to the criteria.
Studies adhered to the criteria.
 Studies did not adhere or report variable.
Studies did not adhere or report variable.
Studies reporting a temporal relationship
Of the 11 studies identified, two were retrospective and used a case-crossover design using data from a large database.3 4 Patients were used as their own control with days 0–30 prior to the stroke event assigned as the case period, and days 60–90 used as the control period for comparison. AF was defined if a patient experienced an episode ≥ 5.5 hours. The highest OR of stroke was in the first 5 days immediately after AF at 17.4 (95% CI 5.4 to 73.1)3 and 5.00 (95% CI 2.62 to 9.55),4 respectively. The risk of AF exposure, however, levelled off after approximately 10–20 days.
A further two studies used a prospective model for ‘ultra-early monitoring’5 and retrospective interrogation of implanted pacemakers for AF.6 In the ‘ultra-early monitoring’ study, AF occurred concurrently at the time of stroke presentation in seven cases, although information relating to duration and frequency of AF episodes prior to the stroke was not provided. The second study found 14 cryptogenic stroke events in patients with pacemakers when patients with known AF or stroke of known origin were excluded. Six patients were found to have episodes of AF, almost all within 2 weeks preceding the stroke event. There was no threshold of AF burden defined in this study, and the duration of AF for each patient ranged between 1 min and 65 hours.
Studies reporting a weak or absent temporal relationship
Seven studies reported a weak or absent temporal relationship between AF and stroke. Three of these examined whether AF occurred before stroke in a predetermined time window.7–9 AF and thromboembolism were considered temporally related if they occurred within either 30 or 60 days of the thromboembolism. Any atrial event longer than ≥5 min or 6 min was considered an AF or atrial-tachycardic episode. In all three studies, atrial events were detected before stroke in less than 56%9 of stroke patients down to 35%.8
The Relationship Between Daily Atrial Tachyarrhythmia Burden from Implantable Device Diagnostics and Stroke Risk (TRENDS) was a prospective study that examined the clinical significance of device-detected atrial arrhythmias.7 The analysis demonstrated that AF burden ≥5.5 hours doubled the risk of a stroke in a 30-day window. In the substudy, however, when the AF threshold burden was at ≥6 min, no temporal relationship was found. 11 thromboembolic events exhibited AF within the 30-day window prior to stroke and 6 AF events occurred concurrently with the event (s). As many AF episodes were observed after the stroke, the authors concluded that there was no temporal relationship.
The Asymptomatic Atrial Fibrillation and Stroke Evaluation in Pacemaker Patients and the Atrial Fibrillation Reduction Atrial Pacing Trial (ASSERT) trial involved 2580 patients.8 A key difference between the TRENDS and ASSERT study was the exclusion of a prior diagnosis of AF to specifically examine ‘sub-clinical’ AF (SCAF). This trial also excluded patients using oral anticoagulants at the beginning of the study unlike TRENDS. The total number of thromboembolic events in TRENDS was 51, with 26 showing any SCAF. Only four patients had SCAF within the 30-day window prior to stroke, constituting 8% of all strokes and only one had AF during the time of stroke. Again, no strong temporal relationship was concluded.
A smaller retrospective study involved examining monitoring devices of 394 patients without history of AF9 over 2 months. Of the 27 stroke events, 15 experienced atrial high-rate episodes (likely AF), 10 (37%) of which occurred within the 2-month time window, 3 (11%) occurring at time of hospitalisation.
A small number of studies interrogated heart failure and hypertrophic cardiomyopathy patients with CRT device implantation. In patients with heart failure and CRT device implantation, it was concluded that there was no temporal association between episodes of AF and stroke.10
Further studies examined arrhythmia-monitoring-guided anticoagulation compared with conventional anticoagulation when AF was found clinically. Both concluded that no temporal relationship could be detected as an almost equal number of patients had AF detected after the thromboembolic events than before or many strokes occurred without recording AF episodes.11 12
A more recent study completed in 202112 enrolled 6004 high-risk individuals and implanted loop recorders in the intervention group, whereas the control group received usual care consisting of annual interviews with a study nurse and standard contact with the participants’ general practitioner. Temporal data on the relationship between AF episode and stroke occurrence were only provided for the intervention group, with only 17 patients diagnosed with AF before or on the day of the event, out of a total of 67 thromboembolic events.
Several factors such as duration of AF, threshold for AF burden and subtyping of strokes varied significantly within studies. Four studies investigated AF episodes within a 30-day window3 4 7 8 and one within a 60 day window.9 Most studies assigned the lowest practicable duration for AF burden threshold (≥5 min or ≥6 min episodes) to maintain AF detection sensitivity of approximately 95%.13 Subtyping into different stroke aetiologic mechanisms was only rarely performed. Only one of the reviewed studies attempted validation of the embolic nature of the stroke by excluding other sources of stroke with ultrasound of the supra-aortic arteries and trans-oesophageal echocardiograms.6 The review found only one study that controlled for anticoagulation use, interestingly this study found the presence of a temporal relationship.
Discussion
The presence of a temporal association between atrial and thromboembolic events remains unclear. However, many limitations were found in the current literature that may obscure the relationship between AF episodes and stroke.
Time window of AF before stroke
Multiple factors contribute to a potential discordance between timing of AF and stroke. First, atrial stunning may persist for days to weeks after cardioversion of AF,14 which maintains the prothrombotic state despite reversion to sinus rhythm. Furthermore, it is possible that on resumed sinus rhythm, the now effective atrial contraction dislodges the thrombus from the atrium, causing a stroke.15
Justifications for the monitoring window from studies ‘based on the assumption that AF induced cardiac thrombi >30 days are unlikely to embolise and cause stroke (because of dissolution or fibrosis)’ are less useful for assessing a temporal correlation as AF can occur transiently before a stroke event while also being a contributing factor.3 Furthermore, the AF episode may simply have occurred coincidentally at the time of stroke.5 10 Without examination of rhythm patterns in the period preceding the event or a control group, it is difficult to determine the temporal relationship.
Duration of AF
Episodes of at least 6 min appear to be significantly associated with increased risk of thromboembolic events in pacemaker patients without clinical AF.16However, this duration may be insufficient for thrombus formation. Trans-oesophageal echocardiogram of patients with AF <48 hours in duration exhibited thrombus formation in merely 1.4% of anticoagulated and 4% of non-anticoagulated patients.17 Short episodes may instead be examining underlying pathophysiological processes common to AF and ischaemic stroke.
Diagnosis of stroke and stroke subtyping
Majority of studies poorly validated the diagnosis of ischaemic stroke or subtyped into different stroke aetiologic mechanisms. In clinical practice, co-occurrence of multiple sources of stroke is very common, although some studies reviewed the imaging6 to verify the stroke, many only used administrative coding data or records of imaging or treatment consistent with stroke.4 7 11 12 A recent trial involving patients with subclinical AF lasting 6 min to 24 hours was monitored to assess the risk of stroke or systemic embolism. Strokes were assessed by experts and categorised according to mortality, stroke subtype and transient ischaemic attack with motor deficit, aphasia or a duration of more than 5 min. While the trial assessed patients with AF and the risk of stroke, it was excluded in the final analysis due to the use of anticoagulation as a primary outcome.18
Other identified issues
One key limitation common to almost all the studies was their small number of thromboembolic events, making it difficult to assess whether a true relationship exists.
Oral anticoagulation is commonly prescribed in patients in whom AF is detected on continuous monitoring, which may have obscured the temporal relationship between AF and stroke.
Recommendations for future research
Future prospective studies should include a large number of well-characterised strokes and stroke subtypes and diagnose AF episodes from adjudicated or verified rhythm strips. Additionally, a sufficiently long temporal window should be assigned to analyse AF episodes prior to the stroke event. Finally, studies should assign an adequately high threshold of AF burden to avoid correlating strokes with incidental or clinically irrelevant AF. Controlling for oral anticoagulation use, subtherapeutic levels or left atrial appendage closure will be required. Future studies could also explore alternate ways to quantify AF rather than burden, such as episodic density or patterns of daily AF burden over time.19
Limitations
Devices used in these studies lacked capacity to discern between atrial tachycardia and true AF. Although some studies excluded patients with a history of AF or AF at time of study inclusion, there was very little to no information on patients that developed permanent AF during the observation period.
Conclusion
So far, evidence of a temporal relationship between AF episodes and ischaemic stroke remains mixed. Whether this is due to a true absence or due to varying study methodology, lack of control for anticoagulation or inconsistent stroke subtyping remains to be established.
Footnotes
Contributors: RK and VT planned and carried out the initial literature search and screening. Both RK and JD contributed to the analysis of the results and to the writing of the final manuscript. VT and HL supervised the project. All authors contributed to the final manuscript. JD and VT are the guarantors of this work.
Funding: The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Competing interests: VT receives consulting fees from Medtronic and lecture and consulting fees from Boehringer Ingelheim and Bayer. HL reports having received research support from St. Jude Medical (now Abbott).
Provenance and peer review: Not commissioned; externally peer-reviewed.
Data availability statement
No data are available.
Ethics statements
Patient consent for publication
Not applicable.
Ethics approval
Not applicable.
References
- 1.Bogousslavsky J, Cachin C, Regli F, et al. Cardiac sources of embolism and cerebral infarction--clinical consequences and vascular concomitants: the lausanne stroke registry. Neurology 1991;41:855–9. 10.1212/wnl.41.6.855 [DOI] [PubMed] [Google Scholar]
- 2.Xu J, Sethi P, Biby S, et al. Predictors of atrial fibrillation detection and features of recurrent strokes in patients after cryptogenic stroke. J Stroke Cerebrovasc Dis 2020;29:104934. 10.1016/j.jstrokecerebrovasdis.2020.104934 [DOI] [PubMed] [Google Scholar]
- 3.Turakhia MP, Ziegler PD, Schmitt SK, et al. Atrial fibrillation burden and short-term risk of stroke. Circ: Arrhythmia and Electrophysiology 2015;8:1040–7. 10.1161/CIRCEP.114.003057 [DOI] [PubMed] [Google Scholar]
- 4.Singer DE, Ziegler PD, Koehler JL, et al. Temporal association between episodes of atrial fibrillation and risk of ischemic stroke. JAMA Cardiol 2021;6:1364–9. 10.1001/jamacardio.2021.3702 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Cuadrado-Godia E, Benito B, Ois A, et al. Ultra-early continuous cardiac monitoring improves atrial fibrillation detection and prognosis of patients with cryptogenic stroke. Eur J Neurol 2020;27:244–50. 10.1111/ene.14061 [DOI] [PubMed] [Google Scholar]
- 6.Moubarak G, Tamazyan R, Garcon P, et al. Detection of occult atrial fibrillation by pacemaker interrogation in cryptogenic stroke. J Interv Card Electrophysiol 2014;39:261–5. 10.1007/s10840-014-9879-x [DOI] [PubMed] [Google Scholar]
- 7.Daoud EG, Glotzer TV, Wyse DG, et al. Temporal relationship of atrial tachyarrhythmias, cerebrovascular events, and systemic emboli based on stored device data: a subgroup analysis of TRENDS. Heart Rhythm 2011;8:1416–23. 10.1016/j.hrthm.2011.04.022 [DOI] [PubMed] [Google Scholar]
- 8.Brambatti M, Connolly SJ, Gold MR, et al. Temporal relationship between subclinical atrial fibrillation and embolic events. Circulation 2014;129:2094–9. 10.1161/CIRCULATIONAHA.113.007825 [DOI] [PubMed] [Google Scholar]
- 9.Witt CT, Kronborg MB, Nohr EA, et al. Early detection of atrial high rate episodes predicts atrial fibrillation and thromboembolic events in patients with cardiac resynchronization therapy. Heart Rhythm 2015;12:2368–75. 10.1016/j.hrthm.2015.07.007 [DOI] [PubMed] [Google Scholar]
- 10.Shanmugam N, Boerdlein A, Proff J, et al. Detection of atrial high-rate events by continuous home monitoring: clinical significance in the heart failure-cardiac resynchronization therapy population. Europace 2012;14:230–7. 10.1093/europace/eur293 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Martin DT, Bersohn MM, Waldo AL, et al. Randomized trial of atrial arrhythmia monitoring to guide anticoagulation in patients with implanted Defibrillator and cardiac Resynchronization devices. Eur Heart J 2015;36:1660–8. 10.1093/eurheartj/ehv115 [DOI] [PubMed] [Google Scholar]
- 12.Svendsen JH, Diederichsen SZ, Højberg S, et al. Implantable loop recorder detection of atrial fibrillation to prevent stroke (the LOOP study): a randomised controlled trial. Lancet 2021;398:1507–16. 10.1016/S0140-6736(21)01698-6 [DOI] [PubMed] [Google Scholar]
- 13.Purerfellner H, Gillis AM, Holbrook R, et al. Accuracy of atrial tachyarrhythmia detection in implantable devices with arrhythmia therapies. Pacing Clin Electrophysiol 2004;27:983–92. 10.1111/j.1540-8159.2004.00569.x [DOI] [PubMed] [Google Scholar]
- 14.Sparks PB, Jayaprakash S, Mond HG, et al. Left atrial mechanical function after brief duration atrial fibrillation. J Am Coll Cardiol 1999;33:342–9. 10.1016/s0735-1097(98)00585-3 [DOI] [PubMed] [Google Scholar]
- 15.Manning WJ, Leeman DE, Gotch PJ, et al. Pulsed doppler evaluation of atrial mechanical function after electrical cardioversion of atrial fibrillation. J Am Coll Cardiol 1989;13:617–23. 10.1016/0735-1097(89)90602-5 [DOI] [PubMed] [Google Scholar]
- 16.Healey JS, Connolly SJ, Gold MR, et al. Subclinical atrial fibrillation and the risk of stroke. N Engl J Med 2012;366:120–9. 10.1056/NEJMoa1105575 [DOI] [PubMed] [Google Scholar]
- 17.Kleemann T, Becker T, Strauss M, et al. Prevalence of left atrial thrombus and dense spontaneous echo contrast in patients with short-term atrial fibrillation < 48 hours undergoing cardioversion: value of transesophageal echocardiography to guide cardioversion. J Am Soc Echocardiogr 2009;22:1403–8. 10.1016/j.echo.2009.09.015 [DOI] [PubMed] [Google Scholar]
- 18.Healey JS, Lopes RD, Granger CB, et al. Apixaban for stroke prevention in subclinical atrial fibrillation. N Engl J Med November 12, 2023. 10.1056/NEJMoa2310234 [DOI] [PubMed] [Google Scholar]
- 19.Ziegler PD. The temporal relationship between atrial fibrillation and ischemic stroke. J Atr Fibrillation 2013;5:738. 10.4022/jafib.738 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.van Velzen HG, Theuns D, Yap S-C, et al. Incidence of device-detected atrial fibrillation and long-term outcomes in patients with hypertrophic cardiomyopathy. Am J Cardiol 2017;119:100–5. 10.1016/j.amjcard.2016.08.092 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
No data are available.