Abstract
Vitiligo is a chronic auto-immune disease characterized by skin depigmentation due to the loss of melanocytes. The better understanding of the disease mechanisms is currently undergoing a significant dynamism, opening a new era in therapeutic development. The pathophysiology of vitiligo has attracted the attention of researchers for years and many advances have been made in clarifying the crosstalk between the cellular players involved in the development of vitiligo lesions. The understanding of the complex interactions between epidermal cells (i.e. melanocytes and keratinocytes), dermal fibroblasts, and immune cells, led to a better characterization of the signals leading to the loss of melanocytes. Recent advances highlighted the role resident T memory cells in the development and recurrence of lesions. This narrative review aims to give an overview of the mechanisms leading to melanocyte disappearance in vitiligo, with a focus on the intercellular interaction network involved in the activation of the local skin immune response.
Keywords: vitiligo, melanocytes, keratinocytes, fibroblasts, immune cells, resident memory T cells, cytokines
Introduction
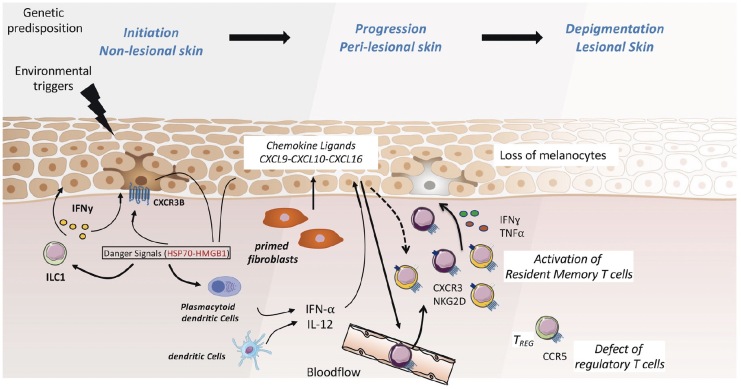
The pathophysiology of vitiligo is complex and involves combinatorial factors (genetic predisposition, environmental triggers, intrinsic melanocyte abnormalities, oxidative stress), leading to an exaggerated activation of the innate and adaptive immune response (Fig. 1) [1]. Genome wide association studies (GWAS) have improved the understanding of the genetic architecture of vitiligo. It is considered that the overall risk of vitiligo linked to genetic determinants is high, corresponding to about 80% of the risk, whereas environmental factors account for 20% [2,3]. Indeed, four large-scale GWAS, three of which were carried out on European and North American populations and one on an Asian (Chinese) populations, identified more than fifty vitiligo susceptibility genes [4–7]. Interestingly, most of these genes are associated with proteins related to innate, adaptive or regulatory immune responses. Others regulate cell apoptosis and some genes are related to proteins regulating melanocyte function. This review discusses how the crosstalk between epidermal, dermal, and immune cells leads to melanocyte disappearance in vitiligo.
Figure 1.
Pathophysiological mechanisms of vitiligo. In a genetically predisposed individual and under environmental triggers, epidermal cells (keratinocytes and melanocytes) will release danger signals (e.g.: HSP70), leading to the activation of the innate immune system. IFNγ-producing innate lymphoid cells (ILC1) or IFNα-producing plasmacytoid dendritic cells will induce the production of chemokines, such as CXCL9, CXCL10, or CXCL16 by epidermal and dermal cells. Melanocytes expressing the CXCR3B subunit can be impacted by the interaction with these ligands. Together, these events will induce local activation of resident memory T cells and the recruitment of circulating T cells expressing CXCR3 and NKG2D and producing elevated levels IFNγ and TNFα, leading to the loss of melanocytes and depigmentation.
Initiation of the Disease: Bridging Oxidative Stress, Epidermal, Dermal Cells and the Activation of the Innate Immune Response
For many years, it has been considered that melanocytes located in the non-lesional pigmented skin of vitiligo patients are more sensitive to oxidative stress by releasing elevated levels of reactive oxygen species (ROS), associated with an imbalance in the antioxidant system [8]. Keratinocytes and fibroblasts also display increased levels of ROS [9]. This accumulation of ROS leads to several intracellular events such as DNA abnormalities and premature apoptosis that induce the release of several melanocyte peptides as well as abnormal melanocyte function and melanin production [8].
Melanocytes produce pro-inflammatory signals such as damage-associated-molecular patterns (DAMPs) and chemokines, leading to the activation of the immune response and the recruitment of immune cells. Among these danger signals, inducible heat shock protein 70 (HSP70i), calreticulin (CRT) or high mobility group B1 (HMGB1) proteins are the most evaluated in vitiligo [10–14]. These DAMPs released in the extracellular environment are likely bridging cellular stress and the autoimmune response directed against melanocytes in vitiligo, and could therefore represent interesting potential targets to prevent the initiation of disease-causing autoimmunity [15]. Indeed, HMGB1 can induce the production of chemokines, such as ligand (C-X-C motif) CXCL16 or interleukin (IL)-8 by epidermal cells (melanocytes and keratinocytes), which are important for the recruitment of immune cells [11]. CRT may induce apoptosis of melanocytes and the release of membrane debris important for immunogenicity [10]. Lastly, several studies from the group of Le Poole emphasized the role of HSP70i in vitiligo [13,16–18], and our group showed its involvement in the activation of innate immunity and the production of type I interferon (IFN) [14].
Keratinocytes also play an important role in the physiopathology of vitiligo. Structural alterations of keratinocytes have been observed in the non-lesional skin of patients, with abnormal thickening of the epidermis due to an increase in the spinous layer [19]. Alterations of basal and suprabasal keratinocytes, in particular vacuolar degeneration and spongiosis, are sometimes found [20]. At the peri-lesional site (at the edge of the lesions, where melanocytes are still present), keratinocytes have the capacity to produce several factors that contribute to the generation of a pro-inflammatory environment, such as stem cell factor (SCF), endothelin-1 (ET-1), and pro-inflammatory cytokines like IL-1β, IL-6, and tumor necrosis factor (TNF)-α. Danger signals produced locally in the skin are responsible for the activation of the pyrin domain of NOD like receptor (NLR) proteins, such as NLRP3, an inflammasome activating protein, promoting the secretion of IL-1β and IL-18 [21]. In addition, keratinocytes from vitiligo non-lesional and peri-lesional skin release chemokines involved in the recruitment of T cells in the skin, like CXCL9, CXCL10, and CXCL16. CXCL9 and CXCL10, induced by IFNγ (an important cytokine in the physiopathology of vitiligo), bind to their cognate receptor CXCR3 expressed on immune cells, and most T cells in vitiligo patients skin express CXCR3 [14,22–24]. In addition, Xu et al. recently showed in a mouse model of vitiligo that IFNγ-responsive dermal fibroblasts, through their release of chemokines, are active players in the homing of CD8 T cells to the skin [25].
Another innate inflammatory signature found increased in peri-lesional areas of vitiligo is the IFNα pathway, which contributes to the activation of antigen-presenting cells in the skin and the production of chemokines to reinforce the recruitment of other immune populations at the peri-lesional site [26,27]. IFNα production is linked to the presence of plasmacytoid dendritic cells and induces the release of chemokines such as CXCL9 and CXCL10 by epidermal cells [14,26]. In addition, the presence of innate immune lymphoid type 1 cells (ILC1) in vitiligo skin has been recently demonstrated, which respond to non-specific stimuli and produce significant amounts of IFNγ during the initiation of the disease, leading to early melanocyte apoptosis [28].
Progression Phase of the Disease: Involvement of a Cutaneous Immune Memory Response
Following these initial events, vitiligo skin is characterized by the presence of a localized T cell infiltrate close to the epidermis and residual melanocytes, mainly consisting of CD8 T cells. Recent studies highlighted the involvement of resident memory T cells (TRM) in disease pathogenesis [24,27,29–31]. TRM are long-lived memory T cells that persist in tissues like the skin and are characterized by a specific transcriptional program [32,33]. They express characteristic cell surface markers involved in their retention in the tissue, such as CD69, CD103, or CD49a, the latter marker defining a subset of TRM cells with cytotoxic properties [29,34–36]. It is now clear that the micro environment plays a crucial role in the formation and regulation of TRM. Indeed, the expression of CD103 is dependent on transforming growth factor (TGF)-β [36], and a growing number of studies reported the involvement of several pro-inflammatory cytokines involved in TRM homeostasis, such as IL-15, IL-12, IL-18, IL-33, IFNγ, TNFα [32]. The presence of TRM in vitiligo skin is undoubtedly linked to the recurrence of lesions on previously affected anatomical sites that have repigmented following treatment [37], as shown in psoriasis or atopic dermatitis [38–40]. Therefore, owing to their functional role in the pathogenesis of vitiligo, targeting TRM appears as a reliable therapeutic strategy.
Vitiligo: Mainly a Type 1 Skewed Immune Response
Analyses of vitiligo peri-lesional and lesional skin revealed upregulation of type-1 associated pathways. Vitiligo is consistently associated with an infiltration of CD8 T cells producing high levels of IFNγ and TNFα. These T cells are characterized by the expression of the receptor CXCR3 that respond CXCL9 and CXCL10, which are highly expressed in the skin of patients [24,41–44]. Targeting the CXCR3-CXCL9 and CXCL10 axis in vitiligo seems to be a promising therapeutic strategy in vitiligo [43,45]. Our group showed that natural killer group 2 member D (NKG2D) defines a subset of highly functional memory CD8 T cells in vitiligo and may represent a potential therapeutic target [46]. This type-1 immune response appears to be a key driver of melanocyte loss in vitiligo and participates in every stage of the pathogenesis. To date, IFNγ and TNFα immune pathways are the most studied in vitiligo. The binding of IFNγ to its receptor induces a signaling dependent of the Janus kinase (JAK) and signal transducer and activator of transcription (STAT) pathways, in particular the activation of JAK1/2 and STAT1. Interestingly, JAK inhibition appears to be a reliable strategy to treat patients and ruxolitinib cream (a topical JAK 1 and JAK 2 inhibitor) is now approved both in US and EU for the management of vitiligo [47–49]. IFNγ induces the production of CXCL9 and CXCL10 by keratinocytes, amplifying the inflammation and the recruitment of immune cells expressing CXCR3 which will promote the progression of vitiligo [14]. IFNγ and TNFα also have a direct impact on the function of melanocytes by decreasing the pigmentation process [50–55]. We recently demonstrated that the combined activity of these two cytokines induces the disruption of E-cadherin expression, the main protein responsible for the adhesion of melanocytes to keratinocytes, leading to their destabilization and detachment [53]. In addition, Tulic et al. showed that melanocytes are able to express the B isoform of CXCR3 in response to IFNγ, making them more susceptible to apoptosis in response to CXCL10 [28].
Additional cytokines and signaling pathways may also be involved in vitiligo pathogenesis. For example, elevated levels of IL-17 and IL-23 are found in the serum and/or skin of vitiligo patients, and IL-17 has been reported to regulate melanocyte function and survival [56]. However, targeting IL-17 in vitiligo patients did not show a major benefit [57]. Type-2-related cytokines have been reported increased in vitiligo patients’ blood, and few studies showed an inhibition of melanogenesis in response to IL-4 and IL-13 [58–62]. Our group found that vitiligo skin is also associated with a type-2 immune response and an increased production of IL-13 by T cells [27]. In addition, Jin et al. reported that IFNγ-induced dermal fibroblasts to release CCL2 and CCL8, inducing a type-2 cytokine profile in vitiligo skin lesion [63]. However, the precise role of type-2 related cytokines in the development of vitiligo remains unclear.
Defects in Immune Regulation During Vitiligo
Like any chronic inflammatory disease characterized by an exaggerated immune response, vitiligo is associated with a disruption of immune regulatory systems. Indeed, GWAS identified a polymorphism of FOXP3 gene, the main transcription factor of regulatory T cells (Tregs), in vitiligo, but also of genes involved in regulation of the immune response, such as CTLA-4 (cytotoxic T lymphocyte antigen 4), IL-10, and TGFβ [3,64,65]. However, it is still unclear whether this defect in the regulation mechanisms results from reduced migration of Tregs into the skin and/or loss of suppressive function of these cells, which would favor the exacerbated effector activity of CD8 T cells [66,67]. Chemokines and their receptors appear critical factors to replenish Tregs in the skin and ensure their immunosuppressive function. For instance, enhanced skin expression of CCL22 induced migration of Tregs into the skin and reduced depigmentation in vivo in mice [68]. In addition, Gellatly et al. showed in a vitiligo mouse model that CCR5/CCL5 axis facilitates Tregs function in the skin [69]. Hence, therapeutic strategies aiming to restore Tregs abundance and function in the skin may be promising for patients [70].
Conclusion
The development of vitiligo lesions involves a complex crosstalk between epidermal, dermal, and immune cells. Such understanding of the mechanisms leading to the loss of melanocytes led to the identification of several targets and the development of future targeted therapies that will undoubtedly improve the management of the disease.
Footnotes
Competing Interests: Katia Boniface received grants from Calypso Biotech, LEO Pharma, Pfizer, Pierre Fabre, Sanofi, and has a patent on MMP9 inhibitors and uses thereof in the prevention or treatment of a depigmenting disorder and three-dimensional model of depigmenting disorder.
Funding: None.
References
- 1.Boniface K, Seneschal J, Picardo M, Taïeb A. Vitiligo: Focus on Clinical Aspects, Immunopathogenesis, and Therapy. Clin Rev Allergy Immunol. 2018;54:52–67. doi: 10.1007/s12016-017-8622-7. [DOI] [PubMed] [Google Scholar]
- 2.Spritz RA. Modern vitiligo genetics sheds new light on an ancient disease. J Dermatol. 2013;40:310–8. doi: 10.1111/1346-8138.12147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Spritz RA, Santorico SA. The Genetic Basis of Vitiligo. J Invest Dermatol. 2021;141:265–73. doi: 10.1016/j.jid.2020.06.004. [DOI] [PubMed] [Google Scholar]
- 4.Jin Y, Andersen G, Yorgov D, et al. Genome-wide association studies of autoimmune vitiligo identify 23 new risk loci and highlight key pathways and regulatory variants. Nat Genet. 2016;48:1418–24. doi: 10.1038/ng.3680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Jin Y, Mailloux CM, Gowan K, et al. NALP1 in vitiligo-associated multiple autoimmune disease. N Engl J Med. 2007;356:1216–25. doi: 10.1056/NEJMoa061592. [DOI] [PubMed] [Google Scholar]
- 6.Jin Y, Birlea SA, Fain PR, et al. Genome-wide association analyses identify 13 new susceptibility loci for generalized vitiligo. Nat Genet. 2012;44:676–80. doi: 10.1038/ng.2272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Jin Y, Birlea SA, Fain PR, et al. Genome-wide analysis identifies a quantitative trait locus in the MHC class II region associated with generalized vitiligo age of onset. J Invest Dermatol. 2011;131:1308–12. doi: 10.1038/jid.2011.12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Chang W-L, Ko C-H. The Role of Oxidative Stress in Vitiligo: An Update on Its Pathogenesis and Therapeutic Implications. Cells. 2023;12:936. doi: 10.3390/cells12060936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kovacs D, Bastonini E, Ottaviani M, et al. Vitiligo Skin: Exploring the Dermal Compartment. J Invest Dermatol. 2018;138:394–404. doi: 10.1016/j.jid.2017.06.033. [DOI] [PubMed] [Google Scholar]
- 10.Zhang Y, Liu L, Jin L, et al. Oxidative stress-induced calreticulin expression and translocation: new insights into the destruction of melanocytes. J Invest Dermatol. 2014;134:183–91. doi: 10.1038/jid.2013.268. [DOI] [PubMed] [Google Scholar]
- 11.Cui T, Zhang W, Li S, et al. Oxidative Stress-Induced HMGB1 Release from Melanocytes: A Paracrine Mechanism Underlying the Cutaneous Inflammation in Vitiligo. J Invest Dermatol. 2019;139:2174–2184.e4. doi: 10.1016/j.jid.2019.03.1148. [DOI] [PubMed] [Google Scholar]
- 12.Zhang K, Anumanthan G, Scheaffer S, Cornelius LA. HMGB1/RAGE Mediates UVB-Induced Secretory Inflammatory Response and Resistance to Apoptosis in Human Melanocytes. J Invest Dermatol. 2019;139:202–12. doi: 10.1016/j.jid.2018.05.035. [DOI] [PubMed] [Google Scholar]
- 13.Mosenson JA, Zloza A, Nieland JD, et al. Mutant HSP70 reverses autoimmune depigmentation in vitiligo. Sci Transl Med. 2013;5:174ra28. doi: 10.1126/scitranslmed.3005127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Jacquemin C, Rambert J, Guillet S, et al. Heat shock protein 70 potentiates interferon alpha production by plasmacytoid dendritic cells: relevance for cutaneous lupus and vitiligo pathogenesis. Br J Dermatol. 2017;177:1367–75. doi: 10.1111/bjd.15550. [DOI] [PubMed] [Google Scholar]
- 15.Migayron L, Boniface K, Seneschal J. Vitiligo, From Physiopathology to Emerging Treatments: A Review. Dermatol Ther. 2020;10:1185–98. doi: 10.1007/s13555-020-00447-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Henning SW, Fernandez MF, Mahon JP, et al. HSP70iQ435A-Encoding DNA Repigments Vitiligo Lesions in Sinclair Swine. J Invest Dermatol. 2018;138:2531–9. doi: 10.1016/j.jid.2018.06.186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Mosenson JA, Eby JM, Hernandez C, Le Poole IC. A central role for inducible heat-shock protein 70 in autoimmune vitiligo. Exp Dermatol. 2013;22:566–9. doi: 10.1111/exd.12183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Denman CJ, McCracken J, Hariharan V, et al. HSP70i accelerates depigmentation in a mouse model of autoimmune vitiligo. J Invest Dermatol. 2008;128:2041–8. doi: 10.1038/jid.2008.45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kovacs D, Bastonini E, Briganti S, et al. Altered epidermal proliferation, differentiation, and lipid composition: Novel key elements in the vitiligo puzzle. Sci Adv. 2022;8:eabn9299. doi: 10.1126/sciadv.abn9299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Moellmann G, Klein-Angerer S, Scollay DA, Nordlund JJ, Lerner AB. Extracellular granular material and degeneration of keratinocytes in the normally pigmented epidermis of patients with vitiligo. J Invest Dermatol. 1982;79:321–30. doi: 10.1111/1523-1747.ep12500086. [DOI] [PubMed] [Google Scholar]
- 21.Marie J, Kovacs D, Pain C, et al. Inflammasome activation and vitiligo/nonsegmental vitiligo progression. Br J Dermatol. 2014;170:816–23. doi: 10.1111/bjd.12691. [DOI] [PubMed] [Google Scholar]
- 22.Li S, Zhu G, Yang Y, et al. Oxidative stress drives CD8+ T-cell skin trafficking in patients with vitiligo through CXCL16 upregulation by activating the unfolded protein response in keratinocytes. J Allergy Clin Immunol. 2017;140:177–189.e9. doi: 10.1016/j.jaci.2016.10.013. [DOI] [PubMed] [Google Scholar]
- 23.Richmond JM, Bangari DS, Essien KI, et al. Keratinocyte-Derived Chemokines Orchestrate T-Cell Positioning in the Epidermis during Vitiligo and May Serve as Biomarkers of Disease. J Invest Dermatol. 2017;137:350–8. doi: 10.1016/j.jid.2016.09.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Boniface K, Jacquemin C, Darrigade A-S, et al. Vitiligo Skin Is Imprinted with Resident Memory CD8 T Cells Expressing CXCR3. J Invest Dermatol. 2018;138:355–64. doi: 10.1016/j.jid.2017.08.038. [DOI] [PubMed] [Google Scholar]
- 25.Xu Z, Chen D, Hu Y, et al. Anatomically distinct fibroblast subsets determine skin autoimmune patterns. Nature. 2022;601:118–24. doi: 10.1038/s41586-021-04221-8. [DOI] [PubMed] [Google Scholar]
- 26.Bertolotti A, Boniface K, Vergier B, et al. Type I interferon signature in the initiation of the immune response in vitiligo. Pigment Cell Melanoma Res. 2014;27:398–407. doi: 10.1111/pcmr.12219. [DOI] [PubMed] [Google Scholar]
- 27.Martins C, Migayron L, Drullion C, et al. Vitiligo Skin T Cells Are Prone to Produce Type 1 and Type 2 Cytokines to Induce Melanocyte Dysfunction and Epidermal Inflammatory Response Through Jak Signaling. J Invest Dermatol. 2022;142:1194–1205.e7. doi: 10.1016/j.jid.2021.09.015. [DOI] [PubMed] [Google Scholar]
- 28.Tulic MK, Cavazza E, Cheli Y, et al. Innate lymphocyte-induced CXCR3B-mediated melanocyte apoptosis is a potential initiator of T-cell autoreactivity in vitiligo. Nat Commun. 2019;10:2178. doi: 10.1038/s41467-019-09963-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Cheuk S, Schlums H, Gallais Sérézal I, et al. CD49a Expression Defines Tissue-Resident CD8+ T Cells Poised for Cytotoxic Function in Human Skin. Immunity. 2017;46:287–300. doi: 10.1016/j.immuni.2017.01.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Richmond JM, Strassner JP, Zapata L, et al. Antibody blockade of IL-15 signaling has the potential to durably reverse vitiligo. Sci Transl Med. 2018;10:eaam7710. doi: 10.1126/scitranslmed.aam7710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Richmond JM, Strassner JP, Rashighi M, et al. Resident Memory and Recirculating Memory T Cells Cooperate to Maintain Disease in a Mouse Model of Vitiligo. J Invest Dermatol. 2019;139:769–78. doi: 10.1016/j.jid.2018.10.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Strobl J, Haniffa M. Functional heterogeneity of human skin-resident memory T cells in health and disease. Immunol Rev. 2023;316:104–19. doi: 10.1111/imr.13213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Milner JJ, Goldrath AW. Transcriptional programming of tissue-resident memory CD8+ T cells. Curr Opin Immunol. 2018;51:162–9. doi: 10.1016/j.coi.2018.03.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Rotrosen E, Kupper TS. Assessing the generation of tissue resident memory T cells by vaccines. Nat Rev Immunol. 2023:1–11. doi: 10.1038/s41577-023-00853-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Clark RA, Chong B, Mirchandani N, et al. The vast majority of CLA+ T cells are resident in normal skin. J Immunol Baltim Md 1950. 2006;176:4431–9. doi: 10.4049/jimmunol.176.7.4431. [DOI] [PubMed] [Google Scholar]
- 36.Mackay LK, Rahimpour A, Ma JZ, et al. The developmental pathway for CD103(+)CD8+ tissue-resident memory T cells of skin. Nat Immunol. 2013;14:1294–301. doi: 10.1038/ni.2744. [DOI] [PubMed] [Google Scholar]
- 37.Nicolaidou E, Antoniou C, Stratigos AJ, Stefanaki C, Katsambas AD. Efficacy, predictors of response, and long-term follow-up in patients with vitiligo treated with narrowband UVB phototherapy. J Am Acad Dermatol. 2007;56:274–8. doi: 10.1016/j.jaad.2006.09.004. [DOI] [PubMed] [Google Scholar]
- 38.Matos TR, O’Malley JT, Lowry EL, et al. Clinically resolved psoriatic lesions contain psoriasis-specific IL-17-producing αβ T cell clones. J Clin Invest. 2017;127:4031–41. doi: 10.1172/JCI93396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Cheuk S, Wikén M, Blomqvist L, et al. Epidermal Th22 and Tc17 cells form a localized disease memory in clinically healed psoriasis. J Immunol Baltim Md 1950. 2014;192:3111–20. doi: 10.4049/jimmunol.1302313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Brunner PM, Emerson RO, Tipton C, et al. Nonlesional atopic dermatitis skin shares similar T-cell clones with lesional tissues. Allergy. 2017;72:2017–25. doi: 10.1111/all.13223. [DOI] [PubMed] [Google Scholar]
- 41.Seneschal J, Boniface K, D’Arino A, Picardo M. An update on Vitiligo pathogenesis. Pigment Cell Melanoma Res. 2021;34:236–43. doi: 10.1111/pcmr.12949. [DOI] [PubMed] [Google Scholar]
- 42.Martins C, Darrigade A-S, Jacquemin C, et al. Phenotype and function of circulating memory T cells in human vitiligo. Br J Dermatol. 2020;183:899–908. doi: 10.1111/bjd.18902. [DOI] [PubMed] [Google Scholar]
- 43.Rashighi M, Agarwal P, Richmond JM, et al. CXCL10 is critical for the progression and maintenance of depigmentation in a mouse model of vitiligo. Sci Transl Med. 2014;6:223ra23. doi: 10.1126/scitranslmed.3007811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Regazzetti C, Joly F, Marty C, et al. Transcriptional Analysis of Vitiligo Skin Reveals the Alteration of WNT Pathway: A Promising Target for Repigmenting Vitiligo Patients. J Invest Dermatol. 2015;135:3105–14. doi: 10.1038/jid.2015.335. [DOI] [PubMed] [Google Scholar]
- 45.Richmond JM, Masterjohn E, Chu R, et al. CXCR3 Depleting Antibodies Prevent and Reverse Vitiligo in Mice. J Invest Dermatol. 2017;137:982–5. doi: 10.1016/j.jid.2016.10.048. [DOI] [PubMed] [Google Scholar]
- 46.Jacquemin C, Martins C, Lucchese F, et al. NKG2D Defines a Subset of Skin Effector Memory CD8 T Cells with Proinflammatory Functions in Vitiligo. J Invest Dermatol. 2020;140:1143–1153.e5. doi: 10.1016/j.jid.2019.11.013. [DOI] [PubMed] [Google Scholar]
- 47.Qi F, Liu F, Gao L. Janus Kinase Inhibitors in the Treatment of Vitiligo: A Review. Front Immunol. 2021;12:790125. doi: 10.3389/fimmu.2021.790125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Rosmarin D, Pandya AG, Lebwohl M, et al. Ruxolitinib cream for treatment of vitiligo: a randomised, controlled, phase 2 trial. Lancet Lond Engl. 2020;396:110–20. doi: 10.1016/S0140-6736(20)30609-7. [DOI] [PubMed] [Google Scholar]
- 49.Rosmarin D, Passeron T, Pandya AG, et al. Two Phase 3, Randomized, Controlled Trials of Ruxolitinib Cream for Vitiligo. N Engl J Med. 2022;387:1445–55. doi: 10.1056/NEJMoa2118828. [DOI] [PubMed] [Google Scholar]
- 50.Englaro W, Bahadoran P, Bertolotto C, et al. Tumor necrosis factor alpha-mediated inhibition of melanogenesis is dependent on nuclear factor kappa B activation. Oncogene. 1999;18:1553–9. doi: 10.1038/sj.onc.1202446. [DOI] [PubMed] [Google Scholar]
- 51.Son J, Kim M, Jou I, Park KC, Kang HY. IFN-γ inhibits basal and α-MSH-induced melanogenesis. Pigment Cell Melanoma Res. 2014;27:201–8. doi: 10.1111/pcmr.12190. [DOI] [PubMed] [Google Scholar]
- 52.Yang L, Wei Y, Sun Y, et al. Interferon-gamma Inhibits Melanogenesis and Induces Apoptosis in Melanocytes: A Pivotal Role of CD8+ Cytotoxic T Lymphocytes in Vitiligo. Acta Derm Venereol. 2015;95:664–70. doi: 10.2340/00015555-2080. [DOI] [PubMed] [Google Scholar]
- 53.Boukhedouni N, Martins C, Darrigade A-S, et al. Type-1 cytokines regulate MMP-9 production and E-cadherin disruption to promote melanocyte loss in vitiligo. JCI Insight. 2020;5:e133772, 133772. doi: 10.1172/jci.insight.133772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Natarajan VT, Ganju P, Singh A, et al. IFN-γ signaling maintains skin pigmentation homeostasis through regulation of melanosome maturation. Proc Natl Acad Sci U S A. 2014;111:2301–6. doi: 10.1073/pnas.1304988111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Ng CY, Chan Y-P, Chiu Y-C, et al. Targeting the elevated IFN-γ in vitiligo patients by human anti- IFN-γ monoclonal antibody hampers direct cytotoxicity in melanocyte. J Dermatol Sci. 2023;110:78–88. doi: 10.1016/j.jdermsci.2023.04.006. [DOI] [PubMed] [Google Scholar]
- 56.Singh RK, Lee KM, Vujkovic-Cvijin I, et al. The role of IL-17 in vitiligo: A review. Autoimmun Rev. 2016;15:397–404. doi: 10.1016/j.autrev.2016.01.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Speeckaert R, Mylle S, van Geel N. IL-17A is not a treatment target in progressive vitiligo. Pigment Cell Melanoma Res. 2019;32:842–7. doi: 10.1111/pcmr.12789. [DOI] [PubMed] [Google Scholar]
- 58.Khan R, Gupta S, Sharma A. Circulatory levels of T-cell cytokines (interleukin [IL]-2, IL-4, IL-17, and transforming growth factor-β) in patients with vitiligo. J Am Acad Dermatol. 2012;66:510–1. doi: 10.1016/j.jaad.2011.07.018. [DOI] [PubMed] [Google Scholar]
- 59.Vaccaro M, Cicero F, Mannucci C, et al. IL-33 circulating serum levels are increased in patients with non-segmental generalized vitiligo. Arch Dermatol Res. 2016;308:527–30. doi: 10.1007/s00403-016-1675-2. [DOI] [PubMed] [Google Scholar]
- 60.Choi H, Choi H, Han J, et al. IL-4 inhibits the melanogenesis of normal human melanocytes through the JAK2-STAT6 signaling pathway. J Invest Dermatol. 2013;133:528–36. doi: 10.1038/jid.2012.331. [DOI] [PubMed] [Google Scholar]
- 61.Han J, Lee E, Kim E, et al. Role of epidermal γδ T-cell-derived interleukin 13 in the skin-whitening effect of Ginsenoside F1. Exp Dermatol. 2014;23:860–2. doi: 10.1111/exd.12531. [DOI] [PubMed] [Google Scholar]
- 62.Czarnowicki T, He H, Leonard A, et al. Blood endotyping distinguishes the profile of vitiligo from that of other inflammatory and autoimmune skin diseases. J Allergy Clin Immunol. 2019;143:2095–107. doi: 10.1016/j.jaci.2018.11.031. [DOI] [PubMed] [Google Scholar]
- 63.Jin R, Zhou M, Lin F, Xu W, Xu A. Pathogenic Th2 Cytokine Profile Skewing by IFN-γ-Responding Vitiligo Fibroblasts via CCL2/CCL8. Cells. 2023;12:217. doi: 10.3390/cells12020217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Birlea SA, Jin Y, Bennett DC, et al. Comprehensive association analysis of candidate genes for generalized vitiligo supports XBP1, FOXP3, and TSLP. J Invest Dermatol. 2011;131:371–81. doi: 10.1038/jid.2010.337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Aydıngöz IE, Kanmaz-Özer M, Gedikbaşi A, et al. The combination of tumour necrosis factor-α −308A and interleukin-10 −1082G gene polymorphisms and increased serum levels of related cytokines: susceptibility to vitiligo. Clin Exp Dermatol. 2015;40:71–7. doi: 10.1111/ced.12446. [DOI] [PubMed] [Google Scholar]
- 66.Dwivedi M, Kemp EH, Laddha NC, et al. Regulatory T cells in vitiligo: Implications for pathogenesis and therapeutics. Autoimmun Rev. 2015;14:49–56. doi: 10.1016/j.autrev.2014.10.002. [DOI] [PubMed] [Google Scholar]
- 67.Giri PS, Mistry J, Dwivedi M. Meta-Analysis of Alterations in Regulatory T Cells’ Frequency and Suppressive Capacity in Patients with Vitiligo. J Immunol Res. 2022;2022:6952299. doi: 10.1155/2022/6952299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Eby JM, Kang H-K, Tully ST, et al. CCL22 to Activate Treg Migration and Suppress Depigmentation in Vitiligo. J Invest Dermatol. 2015;135:1574–80. doi: 10.1038/jid.2015.26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Gellatly KJ, Strassner JP, Essien K, et al. scRNA-seq of human vitiligo reveals complex networks of subclinical immune activation and a role for CCR5 in Treg function. Sci Transl Med. 2021;13:eabd8995. doi: 10.1126/scitranslmed.abd8995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Le Poole IC, Mehrotra S. Replenishing Regulatory T Cells to Halt Depigmentation in Vitiligo. J Investig Dermatol Symp Proc. 2017;18:S38–45. doi: 10.1016/j.jisp.2016.10.023. [DOI] [PubMed] [Google Scholar]