Abstract
Capsaicin, which is abundant in chili peppers, exerts antioxidative, antitumor, antiulcer and analgesic effects and it has demonstrated potential as a treatment for cardiovascular, gastrointestinal, oncological and dermatological conditions. Unique among natural irritants, capsaicin initially excites neurons but then ‘calms’ them into long-lasting non-responsiveness. Capsaicin can also promote weight loss, making it potentially useful for treating obesity. Several mechanisms have been proposed to explain the therapeutic effects of capsaicin, including antioxidation, analgesia and promotion of apoptosis. Some of the mechanisms are proposed to be mediated by the capsaicin receptor (transient receptor potential cation channel subfamily V member 1), but some are proposed to be independent of that receptor. The clinical usefulness of capsaicin is limited by its short half-life. The present review provided an overview of what is known about the therapeutic effects of capsaicin and the mechanisms involved and certain studies arguing against its clinical use were mentioned.
Keywords: pharmacological, capsaicin, mechanisms, transient receptor potential cation channel subfamily V member 1
1. Introduction
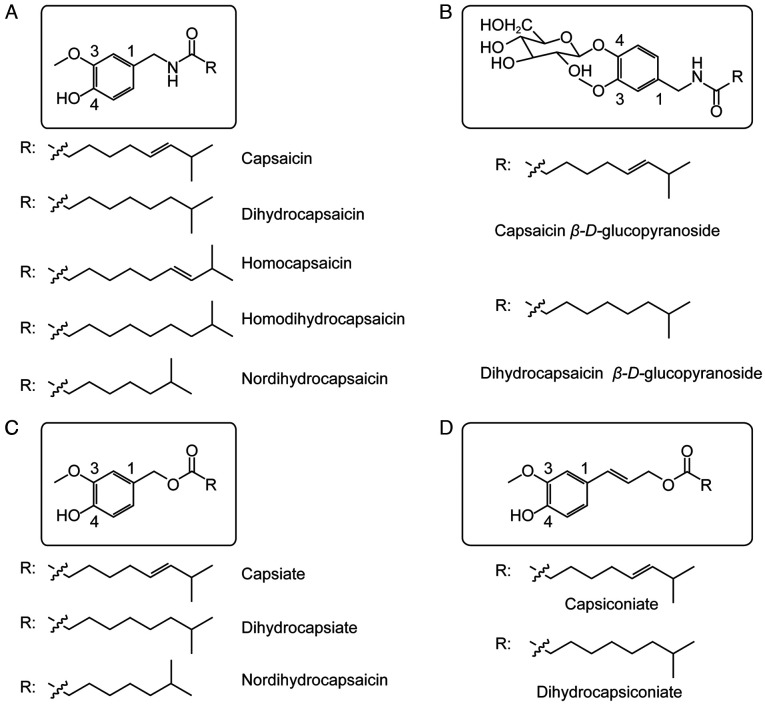
The chili pepper Capsicum annuum L., which belongs to the family Solanaceae in the class Magnoliopsida, is an annual or limited perennial herb widely used gobally as a medicinal and edible plant. The fruit is used in the traditional medicines of China and other countries for warming the body, ‘dispelling cold’ and promoting digestion. The fruit contains various active components, including capsaicin, which is the most abundant pungent compound; capsaicinoids and carotenoids (1). Capsaicin, in turn, exists as a family of compounds including capsaicin, dihydrocapsaicin, homocapsaicin, homodihydrocapsaicin, nordihydrocapsaicin, capsaicin esters, dihydrocapsaicin esters, nordihydrocapsaicin esters, capsanthin-β-d-glucoside and dihydrocapsanthin-β-d-glucoside (2) (Fig. 1). Capsaicin exerts analgesic, antioxidant, cardioprotective, anticancer and thermogenic effects, and it can promote weight loss (3). Some of these effects are mediated by the receptor called ‘transient receptor potential cation channel subfamily V member 1’ (TRPV1), to which capsaicin binds specifically. Some evidence suggests that capsaicin may inhibit signal transducer and activator of transcription 3 (STAT3), but the minimal concentration needed to inhibit STAT3 (50 M) is substantially higher than the concentration required to stimulate TRPV1 (1–5 M) (4,5).
Figure 1.
Chemical structures of capsaicins and capsaicin esters. (A) Capsaicin and its analogues; (B) capsaicin glucopyranoside and dihydrocapsaicin glucopyranoside; (C) and (D) capsiate and its analogues.
2. Structure and physicochemical properties of capsaicin
Capsaicin (trans−8-methyl-N-vanillyl-6-nonenamide, C18H27NO3) is a colorless lipophilic crystalline substance (Fig. 2). It is an amide that forms through condensation of vanillylamine and caprylic acid. It has a melting point of 65°C and a boiling point of 210–220°C and it is highly soluble in ethanol, ether, benzene and chloroform, but only slightly soluble in carbon disulfide. The capsaicin structure can be divided into an aromatic ring (Fig. 2A), amide bond (Fig. 2B) and hydrophobic side chain (Fig. 2C). The various members of the capsaicin family differ from capsaicin mainly in the substitutions on the aromatic ring and hydrophobic side chain (Fig. 1) (6,7). The substituents at positions 3 and 4 on the aromatic ring (Fig. 1) are important active groups. The phenolic hydroxyl group at position 4, for example, acts as a hydrogen bond donor or acceptor in capsaicin agonists; substitution of this hydroxyl group for a hydrophobic group can increase capsaicin activity. Whether the hydrophobic side chain is a saturated or unsaturated alkyl chain, substituted naphthyl group, or something else can influence the activity of capsaicin (8,9).
Figure 2.
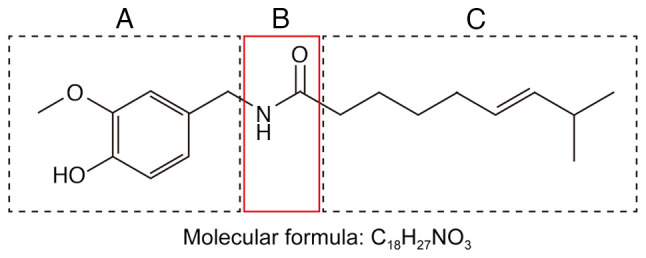
Functionally important subdivisions of the capsaicin structure: (A) Aromatic ring, (B) amide bond and (C) hydrophobic side chain.
3. Pharmacological effects of capsaicin
Antioxidant effects
Capsaicin has been revealed to inhibit lipid peroxidation in red blood cell membranes as well as in the liver and mitochondria of mice, and it can block the peroxidation of low-density lipoproteins in humans (10,11). In fact, the antioxidant activity of capsaicin exceeds that of vitamin E in some cases (12). The levels of capsaicin in food can alleviate oxidative stress and increase cellular antioxidant capacity by preventing reactive oxygen species from oxidizing glutathione (13). Capsaicin can reverse the ability of high blood cholesterol levels to inhibit the antioxidant enzymes glutathione reductase, glutathione transferase and superoxide dismutase (14,15). Capsaicin can also scavenge free radicals such as 1,1′-diphenyl-2-picrylhydrazyl (DPPH) (13). Other members of the capsaicin family, such as dihydrocapsaicin and 9-hydroxycapsaicin, appear to have antioxidant activity similar to that of capsaicin (16).
In capsaicin and other members of the family, the benzene ring and the substituents of the benzene ring appear to be important for antioxidant activity. The benzene ring of capsaicin may interact with the benzene ring of DPPH, while the methoxy and hydroxy substituents at the ortho position of the benzene ring can strongly influence antioxidant activity (13).
Adults who received capsaicin for 4 weeks demonstrated lower levels of oxidation of serum lipoproteins (17). In mitochondria, capsaicin can reduce lipid peroxidation and, more generally, oxidative stress. It can alleviate ischemia-reperfusion injury in myocardium and kidney. Most of these antioxidant effects appear to be mediated by TRPV1 (18). The search continues for capsaicin analogues with even stronger antioxidant activity.
Analgesic effects
TRPV1 is a Ca2+-selective member of the family of transient release potential ion channels, which sense heat. TRPV1 in the prelimbic and infralimbic cortex has also been revealed to mediate neuropathic pain. TRPV1 is broadly distributed in tissues of the brain, bladder, kidneys, intestines, epidermal keratinocytes, glial cells, liver, polymorphonuclear granulocytes, mast cells and macrophages (19).
Capsaicin is an agonist of TRPV1 that reduces its activation threshold. Uniquely, after TRPV1 has been activated by capsaicin, the receptor enters a long-lasting refractory state, in which it does not respond to mechanical pressure, pain or inflammatory agents (20). This so-called ‘defunctionalization’ results from the closing of the channel pore due to conformational changes that depend on extracellular Ca2+. To what extent this transient ‘defunctionalization’ explains the observed analgesic effects of capsaicin remains unclear (21).
When activated by capsaicin, TRPV1 mediates Ca2+ influx and glutamate release, which may damage cutaneous autonomic nerve fibers and sensory nerve endings, decreasing pain sensation. In adult rats, the capsaicin analogue resiniferatoxin damages TRPV1-expressing myelinated nerve fibers and eliminates TRPV1-expressing unmyelinated nerve fibers, reducing perception of thermal pain.
The US Food and Drug Administration has approved capsaicin as an 8% dermal analgesic patch (640 mcg/cm2, total dose 179 mg), while lower doses do not relieve pain effectively (22). The 8% patch has proven safe and effective in controlling neuropathic pain resulting from post-herpetic neuralgia, post-surgical neuralgia, post-traumatic neuropathy, polyneuropathy and mixed pain syndrome (23). In a previous trial (24), capsaicin markedly reduced pain attacks, prolonged sleep duration and improved sleep quality, while reducing dependence on opioids and antiepileptics. ~10% of patients in that trial reported adverse drug reactions, the most frequent of which were erythema and pain at the application site. In a different study including two clinical trials, it has been suggested that the 8% patch is effective against HIV-associated distal sensory polyneuropathy (25).
Adlea, a highly purified form of capsaicin, has exhibited analgesic efficacy in clinical trials involving patients with intermetatarsal neuromas, lateral epicondylitis or end-stage osteoarthritis. A trial is ongoing to assess the safety and efficacy of the drug for patients undergoing total knee arthroplasty (23,26).
N-palmitoyl-vanillamide, also called palvanil, is also present in Capsicum but at markedly lower levels than capsaicin. Palvanil has demonstrated analgesic potential while inducing smaller fluctuations in body temperature and bronchoconstriction than capsaicin. It also exerts the analgesic effects through TRPV1, activating the receptor more slowly and defunctionalizing it more completely than capsaicin does (27).
Antitumor effects
Similar to numerous other dietary phytochemicals, capsaicin shows antitumor activity. It alters the expression of several genes that arrest the cell cycle in tumor cells and promotes apoptosis. These effects have been demonstrated in colon adenocarcinoma, pancreatic cancer, hepatocellular carcinoma, prostate cancer, breast cancer and numerous other types of cancer (Table I), without damage to normal cells. The way capsaicin exerts these effects is only beginning to emerge and the mechanisms appear to involve accumulation of intracellular Ca2+, generation of reactive oxygen species, disruption of mitochondrial membrane potential and upregulation of the transcription factors NF-κB and STATS. Capsaicin has been revealed to act through TRPV1 to promote apoptosis of numerous types of cancers. Whether it also acts through other TRPVs, such as TRPV6 in prostate cancer, remains to be clarified (28,29).
Table I.
Reported antitumor effects of capsaicin in animal models.
| Capsaicin treatment | |||
|---|---|---|---|
|
|
|||
| Animal model | Dose | Regime | Results |
| BALB/cJ and BALB/cJ nu/nu mice injected with CT26 tumor cells | 100-200 µg | Intratumoral on days 5, 10 and 15 | Reduced tumor growth |
| BNX nu/nu male mice injected with PC-3 cells | 5 mg/kg | Gavage 3 days per week for 4 weeks | Reduced tumor growth |
| Athymic nude mice injected with PC-3 cells | 5 mg/kg | Subcutaneous injection every two days for 14 days | Reduced tumor growth and induced apoptosis |
| Female athymic nude mice injected subcutaneously with AsPC-1 tumor | 2.5 mg/kg | Five times a week | Reduced tumor growth without adverse effects |
| cells | 5 mg/kg | Three times a week | |
| Male athymic nu/nu mice injected with U266 cells | 1 mg/kg | Twice a week for 3 weeks | Reduced tumor growth |
| Male athymic nude mice injected subcutaneously with T24 cells | 5 mg/kg | Subcutaneous injection every 3 days for 4 weeks | Reduced tumor growth |
| Female triple deficient beige/nude/xid mice injected with MDA-MB231 cells | 5 mg/kg | Oral gavage 3 days per week for 4 weeks | Reduced tumor growth by 50% |
| Male nude mice injected subcutaneously with H69 cells | 10 mg/kg | Solid diet until tumors of control group reached 2,000 mm3 | Reduced tumor growth |
| Female BALB/c athymic nude mice injected subcutaneously with Colo 205 cells | 1 or 3 mg/kg | Intraperitoneal injection once daily for 4 weeks | Reduced tumor growth |
| Female athymic nude mice injected subcutaneously with AsPC-1 tumor cells | 2.5 mg/kg | Orally fed 5 days a week for 6 weeks | Reduced superoxide dismutase activity in tumors by 60%, while increasing the ratio of oxidized to reduced glutathione |
| Male BALB/c (nu/nu) athymic nude mice injected subcutaneously with PANC-1 cells | 5 mg/kg | Gavage 3 days per week for 4 weeks | Reduced tumor growth |
Weight-lowering effects
Capsaicin causes TRPV1 to stimulate the release of catecholamine from catecholaminergic neurons in the rostral ventrolateral medulla of the brain, thereby promoting weight loss (30). It upregulates adiponectin and other adipokines to reduce fat accumulation in obese mice. Capsaicin has been shown to decrease appetite (31). When delivered as part of a high-fat diet, it increases thermogenesis and lipid oxidation, while also reducing levels of fasting glucose and plasma triglycerides, which suggests therapeutic potential for obesity-related diseases such as insulin resistance and type 2 diabetes mellitus (32,33). Indeed, studies of various capsaicin doses in obese mice have suggested that it can partially reverse obesity-induced glucose intolerance by suppressing inflammatory responses and enhancing fatty acid oxidation in adipose tissue and liver (34).
On the other hand, certain studies (35,36) have failed to detect any effect of capsaicin on energy expenditure or lipid oxidation. While the absence of these effects may be real, it may also be an artifact of administering too little capsaicin for a short period of time, or the thermogenic effects may be too subtle to detect in the relatively small animal groups and short measurement periods in those studies.
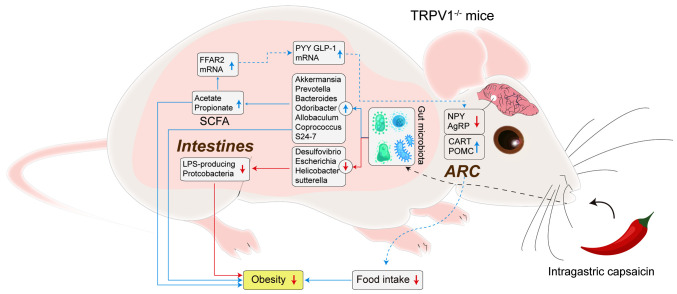
The available evidence suggests that capsaicin lowers lipid levels by altering intestinal permeability and the gut microbiome, in turn influencing the gut-brain axis (37) (Fig. 3). Future studies are needed to verify and elucidate the molecular pathways involved.
Figure 3.
Potential pathways through which capsaicin may mitigate obesity in mouse models. AgRP, agouti-related peptide; ARC, arcuate nucleus; CART, cocaine- and amphetamine-regulated transcript; GLP-1, glucagon like peptide-1; LPS, lipopolysaccharide; NPY, neuropeptide Y; POMC, pro-opiomelanocortin; PYY, peptide YY; SCFA, short-chain fatty acids.
Gastrointestinal effects
In rats and guinea pigs, TRPV1 is expressed and active within the myenteric ganglia and inter-ganglionic fiber tracts that extend throughout the gastrointestinal tract, including the muscle layers, blood vessels and mucosa within the tract (38). TRPV1 is also expressed outside the gastrointestinal nervous system, such as in gastric epithelial cells, in which it stimulates the secretion of gastrin (39).
Previous studies have attributed several positive gastrointestinal effects to capsaicin: It induces the release of calcitonin gene-related peptide, activates gastroprotective cyclooxygenase-1 and increases the absorptive surface of the small intestine by lengthening and thickening microvilli and by altering the permeability of the brush border membrane, in turn increasing zinc absorption (40–42). In non-alcoholic fatty liver disease, dietary capsaicin has been revealed to promote hepatic phosphorylated hormone-sensitive lipase, carnitine palmitoyltransferase 1 and peroxisome proliferator-activated receptor δ (43).
On the other hand, a number of studies have suggested that prolonged exposure to high doses of capsaicin can harm the gastrointestinal tract. Thus, exploring the minimum effective doses required to achieve the desired therapeutic effects and minimizing the potential side effects are quite necessary for enhancing the clinical utility of capsaicin-based treatments. TRPV1 activation induces release of substance P, which can drive gastrointestinal inflammation (44). In addition, TRPV1 is upregulated in irritable bowel syndrome and appears to contribute to the gastrointestinal hypersensitivity and pain associated with the condition (22,45).
Anti-neurodegenerative effects
Capsaicin has demonstrated therapeutic potential in several animal models of Alzheimer's disease (AD). It can partially reverse streptozotocin-induced biochemical and behavioral changes that mimic AD (46). In the APP/PS1 mouse model, capsaicin reduced the formation of amyloid fibrils from amyloid precursor protein. In a third AD model (47), capsaicin substantially ameliorated synaptic damage and tau hyperphosphorylation induced by cold water stress. Further studies should explore the therapeutic potential of dietary capsaicin for treating and possibly even preventing AD (48).
In an animal model of Parkinson's disease based on lipopolysaccharide-induced inflammation, capsaicin appeared to activate TRPV1 in M1/M2 dopaminergic neurons, which may alleviate neuro-inflammation and oxidative stress from activated glia (49). The beneficial effects of capsaicin and TRPV1 were confirmed in these studies using appropriate antagonists (50). Future studies should continue to explore the potential of capsaicin for treating Parkinson's disease and should elucidate the mechanisms involved.
Dermatological effects
TRPV1 is expressed in human keratinocytes. Although activation of epidermal TRPV1 induces the release of inflammatory factors, capsaicin downregulates hypoxia-inducible factor-1α in psoriatic epidermis, slowing epidermal proliferation (51). It also mitigates itching mediated by histamine, substance P and proteinase activated receptor-2. On the other hand, previous studies have failed to detect therapeutic effects of capsaicin against hemodialysis-induced pruritis, idiopathic intractable pruritis and notalgia paresthetica (52,53). In fact, an animal study linked capsaicin to the development of chronically relapsing pruritic dermatitis, which was associated with an elevated number of mast cells and hyperproduction of immunoglobulin E (54,55).
Cardiovascular effects
TRPV1 is expressed in the sensory nerves in cardiovascular structures, near the epicardium and in vascular endothelial cells (56). When blood flow to myocardium is reduced, such as during myocardial infarction, free oxygen radicals are produced, which activate TRPV1 (57). Myocardial injury also upregulates 12-hydroperoxyeicosatetraenoic acid, a metabolite of 12-lipooxygenase arachidonic acid that may bind to TRPV1 (58). Activation of the receptor may exert cardio-protective effects, leading to smaller infarcts and milder ischemic/reperfusion injury (59).
TRPV1 in the vasculature can promote vasoconstriction or vasodilation, depending on the situation. In the case of vasoconstriction, TRPV1 activation results in the release of substance P, which binds to neurokinin 1 (60,61). In the case of vasodilation, TRPV1 activation results in the release of calcitonin gene-related peptide or of protein kinase A and nitric oxide synthase (62,63). In both cases, TRPV1 activation leads to an increase in intracellular Ca2+ (64,65).
Capsaicin inhibits platelet aggregation, potentially by altering the fluidity of the platelet membrane. This mechanism appears to be independent of TRPV1 because the effects are not inhibited by a competitive TRPV1 inhibitor. On the other hand, capsaicin has also been shown to promote platelet aggregation through a mechanism dependent on TRPV1 (66–69). In that mechanism, TRPV1 may induce release of serotonin to drive platelet activation in response to adenosine diphosphate and thrombin (70–74).
4. Pharmacokinetics of capsaicin
Numerous studies have indicated a relatively short half-life of capsaicin in various parts of the organism, including liver, kidney, intestine, lung and blood, restricting its clinical use (75). Within the organism, most capsaicin is metabolized in the liver, where it appears to be metabolized faster in microsomes than in the S9 fraction (76,77). The most abundant metabolites produced in liver microsomes are 16-hydroxycapsaicin, followed by 16,17-dehydrocapsaicin; the most abundant metabolites produced in the S9 fraction are different but have not been definitively identified (78). A number of experiments suggested that P450 enzymes can oxidize capsaicin to generate free radical intermediates (79–81). Further studies should clarify the metabolites of capsaicin in the liver, since some of the discrepancies reported so far may reflect different dosing conditions (82,83).
Capsaicin in the body diffuses into intestinal tissues, the jejunum and serosal fluid (84). A previous study has suggested that capsaicin and dihydrocapsaicin are absorbed to a greater extent by the jejunum and ileum than by the stomach (85).
Capsaicin is metabolized only slowly on the skin, where the main metabolites are vanillylamine and vanillic acid. It penetrates the skin with first-order kinetics. These characteristics make topical administration of capsaicin effective (86,87).
Capsaicin is widely used worldwide, but there is ongoing debate about the safety. For example, epidemiological and laboratory data have suggested that capsaicin can act as a carcinogen or anticarcinogen (88–90). Capsaicin appears to interact with xenobiotic-metabolizing enzymes, particularly microsomal cytochrome P450-dependent monooxygenases, which are involved in activation as well as detoxification of various chemical carcinogens and mutagens (1,91,92). The Indian population consumes several-fold more chili than populations in other countries, yet this does not appear to adversely affect growth, organ weight, nitrogen balance or blood chemistry (93). Previous studies in animals and mammalian cell lines have not suggested any mutagenic effects of capsaicin in somatic cells or the germline (94). Capsaicin cream has been used in the clinic for numerous years to relieve various types of pain, and long-term local application of capsaicin can be effective for treating skin cancer in mice (28,50,95–98).
Although animal studies have indicated few or no side effects of capsaicin, it can irritate the skin of humans and excessive ingestion can cause nausea, vomiting, abdominal pain and burning diarrhea (99). Contact of capsaicin with eyes can cause severe tearing, pain, conjunctivitis, eyelid spasms and it can trigger mucosal irritation leading to serious gastritis and diarrhea. Loading capsaicin into nanoparticles may reduce these adverse effects while improving its efficacy by counteracting its hydrophobicity and prolonging its half-life in the circulation (100,101). Combining capsaicin with other phytochemicals may also mitigate the side effects (102–104); these compounds may include vanilloids, flavonoids, alkaloids, terpenoids, terpenyl phenols, fatty acids, cannabinoids and sulfur-containing compounds. Furthermore, different individuals have different intolerance to capsaicin, personalized treatment programs are needed. Therefore, capsaicin products should be used carefully in light of the range of pharmacological activities and potential for adverse effects; meanwhile, further research is needed to assess the safety of prolonged capsaicin exposure (105).
5. Conclusion
In addition to being widely used as a local analgesic, capsaicin has also demonstrated antioxidant, anticancer, antiobesity and gastroprotective activities. The longer half-life of capsaicin in the lungs and skin implies that it may have stronger effects in these tissues. Systemic administration of capsaicin seems unlikely to be effective because of its metabolic instability and short half-life in the circulation. Further efforts to develop capsaicin analogues and nanoparticles delivery system may succeed in prolonging half-life while also increasing efficacy, making it an effective analgesic against numerous diseases.
Acknowledgements
Not applicable.
Funding Statement
The present study was supported by the National Natural Science Foundation of China (grant no. U1804179).
Availability of data and materials
Not applicable.
Authors' contributions
XS and WZ conceived and designed the study. YZ, JF and ZF analyzed the data. XS wrote the manuscript. Data authentication is not applicable. All authors read and approved the final version of the manuscript.
Ethics approval and consent to participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Surh YJ, Lee SS. Capsaicin, a double-edged sword: Toxicity, metabolism, and chemopreventive potential. Life Sci. 1995;56:1845–1855. doi: 10.1016/0024-3205(95)00159-4. [DOI] [PubMed] [Google Scholar]
- 2.Higashiguchi F, Nakamura H, Hayashi H, Kometani T. Purification and structure determination of glucosides of capsaicin and dihydrocapsaicin from various Capsicum fruits. J Agric Food Chem. 2006;54:5948–5953. doi: 10.1021/jf0607720. [DOI] [PubMed] [Google Scholar]
- 3.Cunha MR, Tavares MT, Fernandes TB, Parise-Filho R. Peppers: A ‘hot’ natural source for antitumor compounds. Molecules. 2021;26:1521. doi: 10.3390/molecules26061521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Zhang S, Wang D, Huang J, Hu Y, Xu Y. Application of capsaicin as a potential new therapeutic drug in human cancers. J Clin Pharm Ther. 2020;45:16–28. doi: 10.1111/jcpt.13039. [DOI] [PubMed] [Google Scholar]
- 5.Popescu GDA, Scheau C, Badarau IA, Dumitrache MD, Caruntu A, Scheau AE, Costache DO, Costache RS, Constantin C, Neagu M, Caruntu C. The effects of capsaicin on gastrointestinal cancers. Molecules. 2020;26:94. doi: 10.3390/molecules26010094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Nanok K, Sansenya S. α-Glucosidase, α-amylase, and tyrosinase inhibitory potential of capsaicin and dihydrocapsaicin. J Food Biochem. 2020;44:e13099. doi: 10.1111/jfbc.13099. [DOI] [PubMed] [Google Scholar]
- 7.Katritzky AR, Xu YJ, Vakulenko AV, Wilcox AL, Bley KR. Model compounds of caged capsaicin: Design, synthesis, and photoreactivity. J Org Chem. 2003;68:9100–9104. doi: 10.1021/jo034616t. [DOI] [PubMed] [Google Scholar]
- 8.Basith S, Cui M, Hong S, Choi S. Harnessing the therapeutic potential of capsaicin and its analogues in pain and other diseases. Molecules. 2016;21:966. doi: 10.3390/molecules21080966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Walpole CS, Bevan S, Bloomfield G, Breckenridge R, James IF, Ritchie T, Szallasi A, Winter J, Wrigglesworth R. Similarities and differences in the structure-activity relationships of capsaicin and resiniferatoxin analogues. J Med Chem. 1996;39:2939–2952. doi: 10.1021/jm960139d. [DOI] [PubMed] [Google Scholar]
- 10.Srinivasan K. Antioxidant potential of spices and their active constituents. Crit Rev Food Sci Nutr. 2014;54:352–372. doi: 10.1080/10408398.2011.585525. [DOI] [PubMed] [Google Scholar]
- 11.Naidu KA, Thippeswamy NB. Inhibition of human low density lipoprotein oxidation by active principles from spices. Mol Cell Biochem. 2002;229:19–23. doi: 10.1023/A:1017930708099. [DOI] [PubMed] [Google Scholar]
- 12.Kursunluoglu G, Taskiran D, Kayali HA. The investigation of the antitumor agent toxicity and capsaicin effect on the electron transport chain enzymes, catalase activities and lipid peroxidation levels in lung, heart and brain tissues of rats. Molecules. 2018;23:3267. doi: 10.3390/molecules23123267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kogure K, Goto S, Nishimura M, Yasumoto M, Abe K, Ohiwa C, Sassa H, Kusumi T, Terada H. Mechanism of potent antiperoxidative effect of capsaicin. Biochim Biophys Acta. 2002;1573:84–92. doi: 10.1016/S0304-4165(02)00335-5. [DOI] [PubMed] [Google Scholar]
- 14.Ochi T, Takaishi Y, Kogure K, Yamauti I. Antioxidant activity of a new capsaicin derivative from Capsicum annuum. J Nat Prod. 2003;66:1094–1096. doi: 10.1021/np020465y. [DOI] [PubMed] [Google Scholar]
- 15.Kempaiah RK, Srinivasan K. Influence of dietary curcumin, capsaicin and garlic on the antioxidant status of red blood cells and the liver in high-fat-fed rats. Ann Nutr Metab. 2004;48:314–320. doi: 10.1159/000081198. [DOI] [PubMed] [Google Scholar]
- 16.Kempaiah RK, Srinivasan K. Antioxidant status of red blood cells and liver in hypercholesterolemic rats fed hypolipidemic spices. Int J Vitam Nutr Res. 2004;74:199–208. doi: 10.1024/0300-9831.74.3.199. [DOI] [PubMed] [Google Scholar]
- 17.Qin Y, Ran L, Wang J, Yu L, Lang HD, Wang XL, Mi MT, Zhu JD. Capsaicin supplementation improved risk factors of coronary heart disease in individuals with low HDL-C levels. Nutrients. 2017;9:1037. doi: 10.3390/nu9091037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Nakagawa H, Hiura A. Capsaicin, transient receptor potential (TRP) protein subfamilies and the particular relationship between capsaicin receptors and small primary sensory neurons. Anat Sci Int. 2006;81:135–155. doi: 10.1111/j.1447-073X.2006.00141.x. [DOI] [PubMed] [Google Scholar]
- 19.Ramsey IS, Delling M, Clapham DE. An introduction to TRP channels. Annu Rev Physiol. 2006;68:619–647. doi: 10.1146/annurev.physiol.68.040204.100431. [DOI] [PubMed] [Google Scholar]
- 20.Knotkova H, Pappagallo M, Szallasi A. Capsaicin (TRPV1 Agonist) therapy for pain relief: Farewell or revival? Clin J Pain. 2008;24:142–154. doi: 10.1097/AJP.0b013e318158ed9e. [DOI] [PubMed] [Google Scholar]
- 21.Aiello F, Badolato M, Pessina F, Sticozzi C, Maestrini V, Aldinucci C, Luongo L, Guida F, Ligresti A, Artese A, et al. Design and synthesis of new transient receptor potential vanilloid type-1 (TRPV1) channel modulators: Identification, molecular modeling analysis, and pharmacological characterization of the N-(4-Hydroxy-3-methoxybenzyl)-4-(thiophen-2-yl)butanamide, a small molecule endowed with agonist TRPV1 Activity and protective effects against oxidative stress. ACS Chem Neurosci. 2016;7:737–748. doi: 10.1021/acschemneuro.5b00333. [DOI] [PubMed] [Google Scholar]
- 22.Sharma SK, Vij AS, Sharma M. Mechanisms and clinical uses of capsaicin. Eur J Pharmacol. 2013;720:55–62. doi: 10.1016/j.ejphar.2013.10.053. [DOI] [PubMed] [Google Scholar]
- 23.Lo Vecchio S, Andersen HH, Elberling J, Arendt-Nielsen L. Sensory defunctionalization induced by 8% topical capsaicin treatment in a model of ultraviolet-B-induced cutaneous hyperalgesia. Exp Brain Res. 2021;239:2873–2886. doi: 10.1007/s00221-021-06170-0. [DOI] [PubMed] [Google Scholar]
- 24.Gašparini D, Ljubičić R, Mršić-Pelčić J. Capsaicin-potential solution for chronic pain treatment. Psychiatr Danub. 2020;32((Suppl 4)):S420–S428. [PubMed] [Google Scholar]
- 25.Brown S, Simpson DM, Moyle G, Brew BJ, Schifitto G, Larbalestier N, Orkin C, Fisher M, Vanhove GF, Tobias JK. NGX-4010, a capsaicin 8% patch, for the treatment of painful HIV-associated distal sensory polyneuropathy: Integrated analysis of two phase III, randomized, controlled trials. AIDS Res Ther. 2013;10:5. doi: 10.1186/1742-6405-10-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Anand P, Bley K. Topical capsaicin for pain management: Therapeutic potential and mechanisms of action of the new high-concentration capsaicin 8% patch. Br J Anaesth. 2011;107:490–502. doi: 10.1093/bja/aer260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Luongo L, Costa B, D'Agostino B, Guida F, Comelli F, Gatta L, Matteis M, Sullo N, De Petrocellis L, de Novellis V, et al. Palvanil, a non-pungent capsaicin analogue, inhibits inflammatory and neuropathic pain with little effects on bronchopulmonary function and body temperature. Pharmacol Res. 2012;66:243–250. doi: 10.1016/j.phrs.2012.05.005. [DOI] [PubMed] [Google Scholar]
- 28.Chapa-Oliver AM, Mejía-Teniente L. Capsaicin: From plants to a cancer-suppressing agent. Molecules. 2016;21:931. doi: 10.3390/molecules21080931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Merritt JC, Richbart SD, Moles EG, Cox AJ, Brown KC, Miles SL, Finch PT, Hess JA, Tirona MT, Valentovic MA, Dasgupta P. Anti-cancer activity of sustained release capsaicin formulations. Pharmacol Ther. 2022;238:108177. doi: 10.1016/j.pharmthera.2022.108177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Akabori H, Yamamoto H, Tsuchihashi H, Mori T, Fujino K, Shimizu T, Endo Y, Tani T. Transient receptor potential vanilloid 1 antagonist, capsazepine, improves survival in a rat hemorrhagic shock model. Ann Surg. 2007;245:964–970. doi: 10.1097/01.sla.0000255577.80800.e1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Yoshioka M, St-Pierre S, Suzuki M, Tremblay A. Effects of red pepper added to high-fat and high-carbohydrate meals on energy metabolism and substrate utilization in Japanese women. Br J Nutr. 1998;80:503–510. doi: 10.1017/S0007114598001597. [DOI] [PubMed] [Google Scholar]
- 32.Kang JH, Tsuyoshi G, Le Ngoc H, Kim HM, Tu TH, Noh HJ, Kim CS, Choe SY, Kawada T, Yoo H, Yu R. Dietary capsaicin attenuates metabolic dysregulation in genetically obese diabetic mice. J Med Food. 2011;14:310–315. doi: 10.1089/jmf.2010.1367. [DOI] [PubMed] [Google Scholar]
- 33.Josse AR, Sherriffs SS, Holwerda AM, Andrews R, Staples AW, Phillips SM. Effects of capsinoid ingestion on energy expenditure and lipid oxidation at rest and during exercise. Nutr Metab (Lond) 2010;7:65. doi: 10.1186/1743-7075-7-65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Lejeune MPGM, Kovacs EMR, Westerterp-Plantenga MS. Effect of capsaicin on substrate oxidation and weight maintenance after modest body-weight loss in human subjects. Br J Nutr. 2003;90:651–659. doi: 10.1079/BJN2003938. [DOI] [PubMed] [Google Scholar]
- 35.Lee GR, Shin MK, Yoon DJ, Kim AR, Yu R, Park NH, Han IS. Topical application of capsaicin reduces visceral adipose fat by affecting adipokine levels in high-fat diet-induced obese mice. Obesity (Silver Spring) 2013;21:115–122. doi: 10.1002/oby.20246. [DOI] [PubMed] [Google Scholar]
- 36.Okumura T, Tsukui T, Hosokawa M, Miyashita K. Effect of caffeine and capsaicin on the blood glucose levels of obese/diabetic KK-A(y) mice. J Oleo Sci. 2012;61:515–523. doi: 10.5650/jos.61.515. [DOI] [PubMed] [Google Scholar]
- 37.Wang Y, Zhou Y, Fu J. Advances in antiobesity mechanisms of capsaicin. Curr Opin Pharmacol. 2021;61:1–5. doi: 10.1016/j.coph.2021.08.012. [DOI] [PubMed] [Google Scholar]
- 38.Ward SM, Bayguinov J, Won KJ, Grundy D, Berthoud HR. Distribution of the vanilloid receptor (VR1) in the gastrointestinal tract. J Comp Neurol. 2003;465:121–135. doi: 10.1002/cne.10801. [DOI] [PubMed] [Google Scholar]
- 39.Ericson A, Nur EM, Petersson F, Kechagias S. The effects of capsaicin on gastrin secretion in isolated human antral glands: Before and after ingestion of red chilli. Dig Dis Sci. 2009;54:491–498. doi: 10.1007/s10620-008-0400-1. [DOI] [PubMed] [Google Scholar]
- 40.Ohno T, Hattori Y, Komine R, Ae T, Mizuguchi S, Arai K, Saeki T, Suzuki T, Hosono K, Hayashi I, et al. Roles of calcitonin gene-related peptide in maintenance of gastric mucosal integrity and in enhancement of ulcer healing and angiogenesis. Gastroenterology. 2008;134:215–225. doi: 10.1053/j.gastro.2007.10.001. [DOI] [PubMed] [Google Scholar]
- 41.Prakash UNS, Srinivasan K. Beneficial influence of dietary spices on the ultrastructure and fluidity of the intestinal brush border in rats. Br J Nutr. 2010;104:31–39. doi: 10.1017/S0007114510000334. [DOI] [PubMed] [Google Scholar]
- 42.Prakash UNS, Srinivasan K. Enhanced intestinal uptake of iron, zinc and calcium in rats fed pungent spice principles-piperine, capsaicin and ginger (Zingiber officinale) J Trace Elem Med Biol. 2013;27:184–190. doi: 10.1016/j.jtemb.2012.11.003. [DOI] [PubMed] [Google Scholar]
- 43.Li Q, Li L, Wang F, Chen J, Zhao Y, Wang P, Nilius B, Liu D, Zhu Z. Dietary capsaicin prevents nonalcoholic fatty liver disease through transient receptor potential vanilloid 1-mediated peroxisome proliferator-activated receptor δ activation. Pflugers Arch. 2013;465:1303–1316. doi: 10.1007/s00424-013-1274-4. [DOI] [PubMed] [Google Scholar]
- 44.Wang L, Hu CP, Deng PY, Shen SS, Zhu HQ, Ding JS, Tan GS, Li YJ. The protective effects of rutaecarpine on gastric mucosa injury in rats. Planta Med. 2005;71:416–419. doi: 10.1055/s-2005-864135. [DOI] [PubMed] [Google Scholar]
- 45.Akbar A, Yiangou Y, Facer P, Walters JR, Anand P, Ghosh S. Increased capsaicin receptor TRPV1-expressing sensory fibres in irritable bowel syndrome and their correlation with abdominal pain. Gut. 2008;57:923–929. doi: 10.1136/gut.2007.138982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Hardy J, Selkoe DJ. The amyloid hypothesis of Alzheimer's disease: Progress and problems on the road to therapeutics. Science. 2002;297:353–356. doi: 10.1126/science.1072994. [DOI] [PubMed] [Google Scholar]
- 47.Postina R, Schroeder A, Dewachter I, Bohl J, Schmitt U, Kojro E, Prinzen C, Endres K, Hiemke C, Blessing M, et al. A disintegrin-metalloproteinase prevents amyloid plaque formation and hippocampal defects in an Alzheimer disease mouse model. J Clin Invest. 2004;113:1456–1464. doi: 10.1172/JCI20864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Wang J, Sun BL, Xiang Y, Tian DY, Zhu C, Li WW, Liu YH, Bu XL, Shen LL, Jin WS, et al. Capsaicin consumption reduces brain amyloid-beta generation and attenuates Alzheimer's disease-type pathology and cognitive deficits in APP/PS1 mice. Transl Psychiatry. 2020;10:230. doi: 10.1038/s41398-020-00918-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Shi Z, El-Obeid T, Riley M, Li M, Page A, Liu J. High chili intake and cognitive function among 4582 adults: An open cohort study over 15 years. Nutrients. 2019;11:1183. doi: 10.3390/nu11051183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Tyagi S, Shekhar N, Thakur AK. Protective role of capsaicin in neurological disorders: An overview. Neurochem Res. 2022;47:1513–1531. doi: 10.1007/s11064-022-03549-5. [DOI] [PubMed] [Google Scholar]
- 51.Li WH, Lee YM, Kim JY, Kang S, Kim S, Kim KH, Park CH, Chung JH. Transient receptor potential vanilloid-1 mediates heat-shock-induced matrix metalloproteinase-1 expression in human epidermal keratinocytes. J Invest Dermatol. 2007;127:2328–2335. doi: 10.1038/sj.jid.5700880. [DOI] [PubMed] [Google Scholar]
- 52.Yu CS. Study on HIF-1α gene translation in psoriatic epidermis with the topical treatment of capsaicin ointment. ISRN Pharm. 2011;2011:821874. doi: 10.5402/2011/821874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Sekine R, Satoh T, Takaoka A, Saeki K, Yokozeki H. Anti pruritic effects of topical crotamiton, capsaicin, and a corticosteroid on pruritogen-induced scratching behavior. Exp Dermatol. 2012;21:201–204. doi: 10.1111/j.1600-0625.2011.01433.x. [DOI] [PubMed] [Google Scholar]
- 54.Gooding SM, Canter PH, Coelho HF, Boddy K, Ernst E. Systematic review of topical capsaicin in the treatment of pruritus. Int J Dermatol. 2010;49:858–865. doi: 10.1111/j.1365-4632.2010.04537.x. [DOI] [PubMed] [Google Scholar]
- 55.Back SK, Jeong KY, Li C, Lee J, Lee SB, Na HS. Chronically relapsing pruritic dermatitis in the rats treated as neonate with capsaicin; a potential rat model of human atopic dermatitis. J Dermatol Sci. 2012;67:111–119. doi: 10.1016/j.jdermsci.2012.05.006. [DOI] [PubMed] [Google Scholar]
- 56.Zahner MR, Li DP, Chen SR, Pan HL. Cardiac vanilloid receptor 1-expressing afferent nerves and their role in the cardiogenic sympathetic reflex in rats. J Physiol. 2003;551:515–523. doi: 10.1113/jphysiol.2003.048207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Poblete IM, Orliac ML, Briones R, Adler-Graschinsky E, Huidobro-Toro JP. Anandamide elicits an acute release of nitric oxide through endothelial TRPV1 receptor activation in the rat arterial mesenteric bed. J Physiol. 2005;568:539–551. doi: 10.1113/jphysiol.2005.094292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Huang HS, Pan HL, Stahl GL, Longhurst JC. Ischemia- and reperfusion-sensitive cardiac sympathetic afferents: Influence of H2O2 and hydroxyl radicals. Am J Physiol. 1995;269:H888–H901. doi: 10.1152/ajpheart.1995.269.3.H888. [DOI] [PubMed] [Google Scholar]
- 59.Schultz HD, Ustinova EE. Capsaicin receptors mediate free radical-induced activation of cardiac afferent endings. Cardiovasc Res. 1998;38:348–355. doi: 10.1016/S0008-6363(98)00031-5. [DOI] [PubMed] [Google Scholar]
- 60.Pan HL, Chen SR. Sensing tissue ischemia: Another new function for capsaicin receptors? Circulation. 2004;110:1826–1831. doi: 10.1161/01.CIR.0000142618.20278.7A. [DOI] [PubMed] [Google Scholar]
- 61.Steagall RJ, Sipe AL, Williams CA, Joyner WL, Singh K. Substance P release in response to cardiac ischemia from rat thoracic spinal dorsal horn is mediated by TRPV1. Neuroscience. 2012;214:106–119. doi: 10.1016/j.neuroscience.2012.04.023. [DOI] [PubMed] [Google Scholar]
- 62.Ide R, Saiki C, Makino M, Matsumoto S. TRPV1 receptor expression in cardiac vagal afferent neurons of infant rats. Neurosci Lett. 2012;507:67–71. doi: 10.1016/j.neulet.2011.11.055. [DOI] [PubMed] [Google Scholar]
- 63.Jones WK, Fan GC, Liao S, Zhang JM, Wang Y, Weintraub NL, Kranias EG, Schultz JE, Lorenz J, Ren X. Peripheral nociception associated with surgical incision elicits remote nonischemic cardioprotection via neurogenic activation of protein kinase C signaling. Circulation. 2009;120((11 Suppl)):S1–S9. doi: 10.1161/CIRCULATIONAHA.108.843938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Wang L, Wang DH. TRPV1 gene knockout impairs postischemic recovery in isolated perfused heart in mice. Circulation. 2005;112:3617–3623. doi: 10.1161/CIRCULATIONAHA.105.556274. [DOI] [PubMed] [Google Scholar]
- 65.Sexton A, McDonald M, Cayla C, Thiemermann C, Ahluwalia A. 12-Lipoxygenase-derived eicosanoids protect against myocardial ischemia/reperfusion injury via activation of neuronal TRPV1. FASEB J. 2007;21:2695–2703. doi: 10.1096/fj.06-7828com. [DOI] [PubMed] [Google Scholar]
- 66.Yang D, Luo Z, Ma S, Wong WT, Ma L, Zhong J, He H, Zhao Z, Cao T, Yan Z, et al. Activation of TRPV1 by dietary capsaicin improves endothelium-dependent vasorelaxation and prevents hypertension. Cell Metab. 2010;12:130–141. doi: 10.1016/j.cmet.2010.05.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Chen Q, Zhu H, Zhang Y, Zhang Y, Wang L, Zheng L. Vasodilating effect of capsaicin on rat mesenteric artery and its mechanism. Zhejiang Da Xue Xue Bao Yi Xue Ban. 2013;42:177–183. doi: 10.3785/j.issn.1008-9292.2013.02.008. (In Chinese) [DOI] [PubMed] [Google Scholar]
- 68.Adams MJ, Ahuja KD, Geraghty DP. Effect of capsaicin and dihydrocapsaicin on in vitro blood coagulation and platelet aggregation. Thromb Res. 2009;124:721–723. doi: 10.1016/j.thromres.2009.05.001. [DOI] [PubMed] [Google Scholar]
- 69.Mittelstadt SW, Nelson RA, Daanen JF, King AJ, Kort ME, Kym PR, Lubbers NL, Cox BF, Lynch JJ., III Capsaicin-induced inhibition of platelet aggregation is not mediated by transient receptor potential vanilloid type 1. Blood Coagul Fibrinolysis. 2012;23:94–97. doi: 10.1097/MBC.0b013e32834ddf18. [DOI] [PubMed] [Google Scholar]
- 70.Raghavendra RH, Naidu KA. Spice active principles as the inhibitors of human platelet aggregation and thromboxane biosynthesis. Prostaglandins Leukot Essent Fatty Acids. 2009;81:73–78. doi: 10.1016/j.plefa.2009.04.009. [DOI] [PubMed] [Google Scholar]
- 71.Sylvester DM, LaHann TR. Effects of capsaicinoids on platelet aggregation. Proc West Pharmacol Soc. 1989;32:95–100. [PubMed] [Google Scholar]
- 72.Meddings JB, Hogaboam CM, Tran K, Reynolds JD, Wallace JL. Capsaicin effects on non-neuronal plasma membranes. Biochim Biophys Acta. 1991;1070:43–50. doi: 10.1016/0005-2736(91)90144-W. [DOI] [PubMed] [Google Scholar]
- 73.Aranda FJ, Villalaín J, Gómez-Fernández JC. Capsaicin affects the structure and phase organization of phospholipid membranes. Biochim Biophys Acta. 1995;1234:225–234. doi: 10.1016/0005-2736(94)00293-X. [DOI] [PubMed] [Google Scholar]
- 74.Harper AG, Brownlow SL, Sage SO. A role for TRPV1 in agonist-evoked activation of human platelets. J Thromb Haemost. 2009;7:330–338. doi: 10.1111/j.1538-7836.2008.03231.x. [DOI] [PubMed] [Google Scholar]
- 75.Batiha GE, Alqahtani A, Ojo OA, Shaheen HM, Wasef L, Elzeiny M, Ismail M, Shalaby M, Murata T, Zaragoza-Bastida A, et al. Biological properties, bioactive constituents, and pharmacokinetics of some Capsicum spp. and capsaicinoids. Int J Mol Sci. 2020;21:5179. doi: 10.3390/ijms21155179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Jung SH, Kim HJ, Oh GS, Shen A, Lee S, Choe SK, Park R, So HS. Capsaicin ameliorates cisplatin-induced renal injury through induction of heme oxygenase-1. Mol Cells. 2014;37:234–240. doi: 10.14348/molcells.2014.2322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Valentovic MA, Ball JG, Brown JM, Terneus MV, McQuade E, Van Meter S, Hedrick HM, Roy AA, Williams T. Resveratrol attenuates cisplatin renal cortical cytotoxicity by modifying oxidative stress. Toxicol In Vitro. 2014;28:248–257. doi: 10.1016/j.tiv.2013.11.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Ito K, Nakazato T, Yamato K, Miyakawa Y, Yamada T, Hozumi N, Segawa K, Ikeda Y, Kizaki M. Induction of apoptosis in leukemic cells by homovanillic acid derivative, capsaicin, through oxidative stress: Implication of phosphorylation of p53 at Ser-15 residue by reactive oxygen species. Cancer Res. 2004;64:1071–1078. doi: 10.1158/0008-5472.CAN-03-1670. [DOI] [PubMed] [Google Scholar]
- 79.Mózsik G, Past T, Abdel Salam OM, Kuzma M, Perjési P. Interdisciplinary review for correlation between the plant origin capsaicinoids, non-steroidal antiinflammatory drugs, gastrointestinal mucosal damage and prevention in animals and human beings. Inflammopharmacology. 2009;17:113–150. doi: 10.1007/s10787-009-0002-3. [DOI] [PubMed] [Google Scholar]
- 80.Luo XJ, Peng J, Li YJ. Recent advances in the study on capsaicinoids and capsinoids. Eur J Pharmacol. 2011;650:1–7. doi: 10.1016/j.ejphar.2010.09.074. [DOI] [PubMed] [Google Scholar]
- 81.Kang JY, Yeoh KG, Chia HP, Lee HP, Chia YW, Guan R, Yap I. Chili-protective factor against peptic ulcer? Dig Dis Sci. 1995;40:576–579. doi: 10.1007/BF02064373. [DOI] [PubMed] [Google Scholar]
- 82.Chanda S, Bashir M, Babbar S, Koganti A, Bley K. In vitro hepatic and skin metabolism of capsaicin. Drug Metab Dispos. 2008;36:670–675. doi: 10.1124/dmd.107.019240. [DOI] [PubMed] [Google Scholar]
- 83.Reilly CA, Ehlhardt WJ, Jackson DA, Kulanthaivel P, Mutlib AE, Espina RJ, Moody DE, Crouch DJ, Yost GS. Metabolism of capsaicin by cytochrome P450 produces novel dehydrogenated metabolites and decreases cytotoxicity to lung and liver cells. Chem Res Toxicol. 2003;16:336–349. doi: 10.1021/tx025599q. [DOI] [PubMed] [Google Scholar]
- 84.Kawada T, Suzuki T, Takahashi M, Iwai K. Gastrointestinal absorption and metabolism of capsaicin and dihydrocapsaicin in rats. Toxicol Appl Pharmacol. 1984;72:449–456. doi: 10.1016/0041-008X(84)90121-2. [DOI] [PubMed] [Google Scholar]
- 85.Wang YY, Hong CT, Chiu WT, Fang JY. In vitro and in vivo evaluations of topically applied capsaicin and nonivamide from hydrogels. Int J Pharm. 2001;224:89–104. doi: 10.1016/S0378-5173(01)00755-4. [DOI] [PubMed] [Google Scholar]
- 86.O'Neill J, Brock C, Olesen AE, Andresen T, Nilsson M, Dickenson AH. Unravelling the mystery of capsaicin: A tool to understand and treat pain. Pharmacol Rev. 2012;64:939–971. doi: 10.1124/pr.112.006163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Suresh D, Srinivasan K. Tissue distribution & elimination of capsaicin, piperine & curcumin following oral intake in rats. Indian J Med Res. 2010;131:682–691. [PubMed] [Google Scholar]
- 88.Rollyson WD, Stover CA, Brown KC, Perry HE, Stevenson CD, McNees CA, Ball JG, Valentovic MA, Dasgupta P. Bioavailability of capsaicin and its implications for drug delivery. J Control Release. 2014;196:96–105. doi: 10.1016/j.jconrel.2014.09.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Thornton T, Mills D, Bliss E. Capsaicin: A potential treatment to improve cerebrovascular function and cognition in obesity and ageing. Nutrients. 2023;15:1537. doi: 10.3390/nu15061537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Petroianu GA, Aloum L, Adem A. Neuropathic pain: Mechanisms and therapeutic strategies. Front Cell Dev Biol. 2023;11:1072629. doi: 10.3389/fcell.2023.1072629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Erin N, Szallasi A. Carcinogenesis and metastasis: Focus on TRPV1-positive neurons and immune cells. Biomolecules. 2023;13:983. doi: 10.3390/biom13060983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Fernández-Carvajal A, Fernández-Ballester G, Ferrer-Montiel A. TRPV1 in chronic pruritus and pain: Soft modulation as a therapeutic strategy. Front Mol Neurosci. 2022;15:930964. doi: 10.3389/fnmol.2022.930964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Zhang L, Angst E, Park JL, Moro A, Dawson DW, Reber HA, Eibl G, Hines OJ, Go VL, Lu QY. Quercetin aglycone is bioavailable in murine pancreas and pancreatic xenografts. J Agric Food Chem. 2010;58:7252–7257. doi: 10.1021/jf101192k. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Santos VAM, Bressiani PA, Zanotto AW, Almeida IV, Berti AP, Lunkes AM, Vicentini VEP, Düsman E. Cytotoxicity of capsaicin and its analogs in vitro. Braz J Biol. 2023;83:e268941. doi: 10.1590/1519-6984.268941. [DOI] [PubMed] [Google Scholar]
- 95.Chaiyasit K, Khovidhunkit W, Wittayalertpanya S. Pharmacokinetic and the effect of capsaicin in Capsicum frutescens on decreasing plasma glucose level. J Med Assoc Thai. 2009;92:108–113. [PubMed] [Google Scholar]
- 96.Braga Ferreira LG, Faria JV, Dos Santos JPS, Faria RX. Capsaicin: TRPV1-independent mechanisms and novel therapeutic possibilities. Eur J Pharmacol. 2020;887:173356. doi: 10.1016/j.ejphar.2020.173356. [DOI] [PubMed] [Google Scholar]
- 97.Liu T, Wan Y, Meng Y, Zhou Q, Li B, Chen Y, Wang L. Capsaicin: A novel approach to the treatment of functional dyspepsia. Mol Nutr Food Res. 2023;10:e2200793. doi: 10.1002/mnfr.202200793. [DOI] [PubMed] [Google Scholar]
- 98.Szallasi A. Capsaicin for weight control: ‘Exercise in a pill’ (or just another fad)? Pharmaceuticals (Basel) 2022;15:851. doi: 10.3390/ph15070851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Huang Z, Sharma M, Dave A, Yang Y, Chen ZS, Radhakrishnan R. The antifibrotic and the anticarcinogenic activity of capsaicin in hot chili pepper in relation to oral submucous fibrosis. Front Pharmacol. 2022;13:888280. doi: 10.3389/fphar.2022.888280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Malewicz NM, Rattray Z, Oeck S, Jung S, Escamilla-Rivera V, Chen Z, Tang X, Zhou J, LaMotte RH. Topical capsaicin in Poly(lactic-co-glycolic)acid (PLGA) nanoparticles decreases acute itch and heat pain. Int J Mol Sci. 2022;23:5275. doi: 10.3390/ijms23095275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Yue WWS, Yuan L, Braz JM, Basbaum AI, Julius D. TRPV1 drugs alter core body temperature via central projections of primary afferent sensory neurons. Elife. 2022;11:e80139. doi: 10.7554/eLife.80139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Abbas MA. Modulation of TRPV1 channel function by natural products in the treatment of pain. Chem Biol Interact. 2020;330:109178. doi: 10.1016/j.cbi.2020.109178. [DOI] [PubMed] [Google Scholar]
- 103.Yeon KY, Kim SA, Kim YH, Lee MK, Ahn DK, Kim HJ, Kim JS, Jung SJ, Oh SB. Curcumin produces an antihyperalgesic effect via antagonism of TRPV1. J Dent Res. 2010;89:170–174. doi: 10.1177/0022034509356169. [DOI] [PubMed] [Google Scholar]
- 104.Sui F, Zhang CB, Yang N, Li LF, Guo SY, Huo HR, Jiang TL. Anti-nociceptive mechanism of baicalin involved in intervention of TRPV1 in DRG neurons in vitro. J Ethnopharmacol. 2010;129:361–366. doi: 10.1016/j.jep.2010.03.039. [DOI] [PubMed] [Google Scholar]
- 105.Dludla PV, Nkambule BB, Cirilli I, Marcheggiani F, Mabhida SE, Ziqubu K, Ntamo Y, Jack B, Nyambuya TM, Hanser S, Mazibuko-Mbeje SE. Capsaicin, its clinical significance in patients with painful diabetic neuropathy. Biomed Pharmacother. 2022;153:113439. doi: 10.1016/j.biopha.2022.113439. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.