Abstract
Murine antibody responses to soluble proteins are generally restricted to the immunoglobulin G1 (IgG1) isotype. When mice were infected with Toxoplasma gondii Beverley and concomitantly immunized with a soluble unrelated protein antigen, a modification in the isotypic distribution of antibodies directed against this nonparasite antigen was observed, with a preferential production of IgG2a. Interestingly, when mice were immunized with a soluble protein antigen during the chronic phase (day 40) of infection with T. gondii Beverley, a similar modification in the isotypic distribution of antiprotein antibodies was observed.
For mice, the current paradigm is that CD4+ T cells can be separated into subsets on the basis of the repertoire of cytokines produced and that the distinct cytokine profile observed in these cells determines their function. This model includes two major subsets: Th1 cells produce interleukin-2 (IL-2) and gamma interferon (IFN-γ) and control the production of immunoglobulin G2a (IgG2a), whereas Th2 cells produce IL-4, IL-5, and IL-10 and control the production of IgG1 and IgE (17, 27). These subsets cross-regulate each other. Which subset predominates may determine the outcome of an infection. The weakly virulent species Toxoplasma gondii triggers high levels of IgG2a in serum (8, 33, 36) and induces persistent expression of IFN-γ and IL-12 cytokines (19, 20, 22). In contrast, antibodies raised after immunization with a soluble protein predominantly belong to the IgG1 subclass (13); such soluble proteins induce IL-4 and IL-10 cytokine expression (10, 26, 37). When some viruses, like murine hepatitis virus or lactate dehydrogenase-elevating virus, are inoculated concomitantly with such an immunization, the isotypic distribution of antiprotein antibodies is biased in favor of IgG2a (12); this phenomenon could have implications for the development of autoimmune reactions (35). In the present study, we analyzed the effect of acute and chronic infections with T. gondii on the isotypic pattern of antibodies raised against nonparasitic soluble protein antigens that usually raise a Th2 response characterized by IgG1 antibodies.
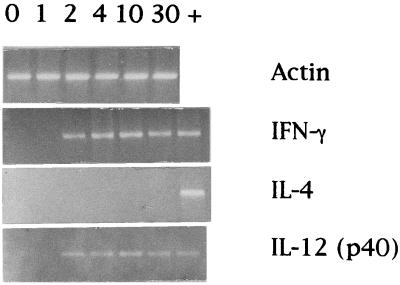
Cytokine mRNA expression by spleen cells during the different stages of T. gondii Beverley infections.
BALB/c female mice (8 to 10 weeks old) were bred in isolators at the Ludwig Institute for Cancer Research by G. Warnier and were infected intraperitoneally (i.p.) with the weakly virulent Beverley strain of T. gondii, isolated by J. K. A. Beverley (5) and kindly provided by G. Desmonts from the Institut de Puériculture, Paris, France, in 1977. Splenocytes were collected at five time points after infection, and the expression of cytokine mRNA was analyzed by reverse transcription-PCR as described previously (14). Briefly, unfractionated spleen cells were resuspended in TRIzol (Gibco) and frozen at −80°C. The cells were then homogenized and processed for RNA isolation, after separation with chloroform and precipitation with isopropanol as recommended by the manufacturer; cDNA was prepared with Moloney murine leukemia virus reverse transcriptase (Gibco) and amplified by PCR with a Gene Amp kit (Perkin-Elmer Cetus) in a Techne PHC 3 programmable Dri-block (New Brunswick Scientific, Duxford Cambridge, United Kingdom). Nucleotide sequences of primers for actin, IL-12 (p40) (35 to 40), IFN-γ (30 to 35), and IL-4 (35 to 40) were the same as those we described previously (14, 26), as were the experimentally determined optimal cycle numbers (indicated in parentheses). Nucleotide sequences of primers for actin were 5′-ATG GAT GAC GAT ATC GCT GC-3′ and 5′-GCT GGA AGG TGG ACA GTG AG-3′, those for IL-12 (p40) were 5′-CTC ACA TCT GCT GCT CCA CAA-3′ and 5′-CTC CTT CAT CTT TTC TTT CTT-3′, those for IFN-γ were 5′-GAC AAT CAG GCC ATC AGC AAC-3′ and 5′-CGC AAT CAC AGT CTT GGC TAA-3′, and those for IL-4 were 5′-ATG GGT CTC AAC CCC CAG CTA-3′ and 5′-GCA TGG TGG CTC AGT ACT ACG-3′. Analysis of cytokine messages showed increased expression of IL-12 (p40) and IFN-γ mRNAs in spleen cells as early as 2 days postinfection (p.i.) and up to 30 days p.i. (Fig. 1). These findings confirm the recent observations of Burke et al. (8) and Gazzinelli et al. (20). We could not detect expression of IL-4 (Fig. 1). Because T. gondii is a potent stimulator of IL-12 release by macrophages, the production of this cytokine early in infection could be responsible for driving the parasite-specific T-cell response in the Th1 direction. In addition, the effect may be enhanced by IFN-γ, which has been shown to be a potent inhibitor of Th2 cell proliferation (18). The observations by Gazzinelli et al. (20) on levels of IL-4 and IL-10 synthesis in anti-IL-12-treated mice support this hypothesis.
FIG. 1.
Detection of IL-12 (p40), IFN-γ, IL-4, and actin mRNAs in spleen cells of BALB/c mice infected with 20 cysts of T. gondii Beverley. Spleen cells were harvested at time zero (control animals) and at 1, 2, 4, 10, and 30 days p.i. as indicated above the lanes and then frozen in TRIzol; mRNA preparations were reverse transcribed, and specific messages were amplified by PCR. Products of PCR amplification were detected by ethidium bromide staining in agarose gels. Lane +, control for cytokine mRNAs.
IgG subclass distribution of antiparasite antibody responses during acute and chronic T. gondii Beverley infections.
NMRI female mice (6 to 8 weeks old) that were obtained from the animal facility of the Catholic University of Louvain, Brussels, Belgium, and BALB/c mice were infected i.p. with weakly virulent T. gondii Beverley (28), whereas other NMRI and BALB/c mice were kept as uninfected controls. Heart blood samples were collected from mice by cardiac puncture under anesthesia with diethyl ether. Between 100 and 1,000 μl of blood was collected in EDTA or heparin on days 21 and 56 p.i.; the IgG subclasses of anti-T gondii antibodies in individual mouse plasma samples were determined by enzyme-linked immunosorbent assay (ELISA). Briefly, microplates (Immunoplate Maxisorp F96; Nunc, Roskilde, Denmark) were coated by overnight incubation at 4°C with 100 μl of a lysate of T. gondii (6.5 μg of protein/ml) in phosphate-buffered saline (PBS) (pH 7.2). The plates were washed three times in PBS (pH 7.2). Wells were saturated with 5% fetal calf serum (Gibco) in PBS for 15 min, and then 100 μl of plasma diluted 1:50, 1:150, 1:450, or 1:1,350 in PBS containing 0.5% Tween 20 (PBS-Tween 20) was added and incubated at 22°C for 30 min. After three washings in PBS, 100 μl of anti-mouse IgG subclass rabbit antibody labeled with peroxidase (Serotec, Oxford, England), diluted 1:1,000 in PBS-Tween 20, was added and incubated for 30 min at 22°C. The plates were washed again before addition of 100 μl of chromogen (tetramethylbenzidine [27 g/liter] plus hydrogen peroxide [0.1 ml/liter]) (Sorin Biomedica, Saluggia, Italy) solution. The reaction was stopped with 1 N H2SO4. The absorbance of each sample was read at 450 nm with a Sorin spectrophotometer. Results, expressed in micrograms per milliliter, were calculated from standard curves obtained with selected anti-DNP monoclonal antibodies (11). For NMRI mice, the specific antibody concentration for each isotype could be ranked in the acute phase (21 days p.i.) as IgG2b > IgG2a > IgG3 > IgG1 and in the chronic phase (56 days p.i.) as IgG2a >> IgG2b > IgG3 > IgG1 (Table 1). For BALB/c mice, the levels of T. gondii-specific IgG antibody isotypes could be ranked in the acute phase (21 days p.i.) as IgG2b = IgG2a > IgG3 > IgG1, whereas the chronic phase (56 days p.i.) was characterized by higher levels, with IgG2a >> IgG2b > IgG3 > IgG1 (Table 1). This investigation has shown that T. gondii infection in mice induces a stable polyisotypic parasite-specific response characterized by high concentrations of IgG2a but not IgG1 antibodies. These findings confirm the observations of Burke et al. (8), Suzuki et al. (33), and Villard et al. (36). This positivity was maintained during 325 days (not shown). Little variation was observed between BALB/c and NMRI mouse strains (Table 1). However, C57BL/10 mice produce less IgG1 than BALB/c mice after infection with cysts of the RRA strain (8, 29), whereas C57BL/6 animals produce IgG3 after infection with cysts of the ME49 strain (33). The differences of the IgG subclass responses in different strains of T. gondii or strains of mice can be influenced by the susceptibility of the mice (15, 32). Different routes of infection (orally, i.p., and subcutaneously) gave no significant differences for the IgG subclass responses in mice (data not shown).
TABLE 1.
IgG subclass distribution of antibody responses during acute and chronic T. gondii Beverley infections
| Mice | Day p.i. | IgG antibody (μg/ml)a
|
|||
|---|---|---|---|---|---|
| IgG1 | IgG2a | IgG2b | IgG3 | ||
| NMRI | 21 | <0.1 | 3.8 ± 1.7 | 10.8 ± 3.1 | 1.6 ± 0.2 |
| BALB/c | 21 | <0.1 | 3.6 ± 0.4 | 3.6 ± 0.6 | 1.0 ± 0.1 |
| NMRI | 56 | <0.1 | 66.2 ± 16.1 | 11.7 ± 1.4 | 2.2 ± 0.2 |
| BALB/c | 56 | 0.2 ± 0.1 | 47.3 ± 6.4 | 6.5 ± 1.8 | 1.0 ± 0.2 |
Anti-T. gondii IgG subclasses were measured by ELISA. Results (means ± standard errors) are for groups of three to five animals injected i.p.
IgG subclass distribution of anti-soluble-protein antibody responses elicited concomitantly with acute and chronic T. gondii Beverley infections.
For the acute phase, the experimental protocol is shown in Table 2. Six groups of BALB/c mice were used, with five mice in each group. On day 0, three groups were immunized with tetanus toxoid obtained from the Institut Pasteur (Brussels, Belgium) or keyhole limpet hemocyanin or lactoferrin obtained from Sigma (St. Louis, Mo.), while three other groups received the same amount of antigen concomitantly with T. gondii Beverley infection. In the chronic phase, 40 days after this infection with T. gondii, the mice received one injection of lactoferrin in PBS. All mice were bled on day 21. IgG antibody subclasses in plasma were determined by ELISA as described by Coutelier et al. (11). Briefly, microplates (Immunoplate Maxisorp F96) were coated by overnight incubation at 4°C with purified proteins (10 μg/ml) and incubated with serial dilutions of sera. Binding of antibodies was measured and calculated as for the anti-T. gondii antibody ELISA. Antibodies raised in uninfected mice after primary immunization with a soluble protein predominantly belonged to the IgG1 subclass. The acute infection strongly increased the proportion of IgG2a in antibodies directed against all of the antigens tested (Table 2). Those modifications were mainly due to a dramatic increase in absolute amounts of IgG2a (19.4- to 211.6-fold) rather than to a decrease in IgG1 (1.2- to 7.4-fold). The isotypic profiles of immunoglobulins after some DNA and RNA murine virus infections, as well as parasitic diseases (malaria or trypanosomiasis), have been studied previously; these infections mainly enhance IgG2a (3, 12, 34). Nematode and cestode parasites preferentially stimulate a concomitant increase of IgG1 associated with a decrease of IgG2a levels (39). The trematode Schistosoma mansoni induces increased IgG1 without modification of IgG2a and IgG2b levels (7). Coinfection with T. gondii and murine leukemia viruses (murine AIDS) increased susceptibility to T. gondii and may alter the progression of murine AIDS (24). Coinfection with Leishmania major and T. gondii in albino mice showed that the clinical and histopathological pictures for this concomitant infection differ from those for infection with either parasite alone (1). In L. major infection, BALB/c mice immunized subcutaneously with a soluble leishmanial antigen develop a Th2-like response in which IL-4-producing cells dominate; in this model, coimmunization with recombinant IL-12 (rIL-12) induces a switch from Th2 to Th1 responses in which IFN-γ-producing cells dominate (2). IL-12 stimulates IFN-γ production by natural killer (NK) cells and T cells (9, 19, 38), which in turn can trigger IgG2a production by B lymphocytes (16, 31).
TABLE 2.
Effect of T. gondii Beverley infection on the subclass distribution of antisoluble protein IgG antibody responses
| Immunizationa | T. gondiib | IgG antibody, μg/ml (% of total IgG)c
|
|||
|---|---|---|---|---|---|
| IgG1 | IgG2a | IgG2b | IgG3 | ||
| Tetanus toxoid | − | 9.1 ± 4.3 (74.6) | 2.0 ± 0.5 (16.4) | 0.7 ± 0.3 (5.7) | 0.4 ± 0.1 (3.3) |
| + | 7.5 ± 3.5 (13.3) | 45.8 ± 11.8 (81.2) | 2.1 ± 0.8 (3.7) | 1.0 ± 0.5 (1.8) | |
| Keyhole limpet hemocyanin | − | 5.0 ± 2.2 (48.1) | 2.8 ± 0.9 (26.9) | 1.4 ± 0.5 (13.5) | 1.2 ± 0.8 (11.5) |
| + | 4.1 ± 1.5 (5.2) | 54.4 ± 17.1 (68.9) | 17.0 ± 8.7 (21.5) | 3.5 ± 2.1 (4.4) | |
| Lactoferrin | − | 5.2 ± 1.0 (89.6) | 0.3 ± 0.1 (5.2) | 0.2 ± 0.1 (3.4) | <0.1 (<1.8) |
| + | 0.7 ± 0.3 (1.1) | 63.5 ± 18.5 (97.1) | 0.9 ± 0.4 (1.4) | 0.3 ± 0.1 (0.4) | |
Day 0, i.p. injection of 100 μg of protein diluted in PBS.
+, infection at day 0 by i.p. injection of 20 cysts; −, no infection.
IgG subclass responses specific for each immunizing protein in sera obtained on day 21 were measured by ELISA. Results (means ± standard errors) are for groups of three to five animals.
In agreement with previous studies (13), we observed that antibody responses of uninfected mice to tetanus toxoid, keyhole limpet hemocyanin, or lactoferrin were restricted to the IgG1 subclass (shown in Table 2). Immunization with a plasmid encoding fragment C of tetanus toxin is biased in favor of IgG, and stimulated splenocytes secreted high levels of IFN-γ to protect mice against lethal challenge with tetanus toxin (4). However, when some viruses—for example, murine hepatitis virus or lactate dehydrogenase-elevating virus—are inoculated concomitantly with such a protein immunization, the isotypic distribution of antiprotein antibodies is biased in favor of IgG2a. This isotype modulation, which effects T-cell-dependent responses, is observed only when the infection occurs around the time of immunization (13). The isotypic bias induced at the time of the primary immunization persists in subsequent secondary responses. Whether such a bias in concomitant immune responses may increase the pathogenicity, for instance, of other parasitic infections or of autoimmune diseases remains to be determined.
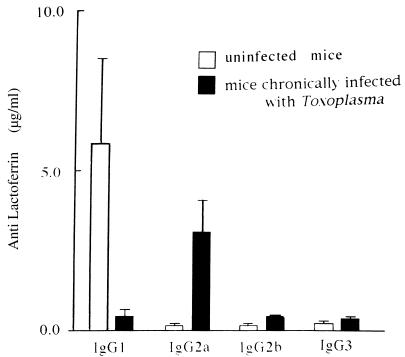
In the viral model, infection initiated 3 days before or 4 days after injection of soluble proteins does not influence the subclass proportion of antibodies reacting with the nonviral protein antigen. In contrast, and interestingly, we show here that after T. gondii inoculation, antilactoferrin IgG subclasses assayed in sera of mice immunized with this antigen during the chronic infection with the parasite display a sharp increase in IgG2a (18.9-fold) and a decrease in IgG1 (12.5-fold) (Fig. 2). The same results were observed at 40 days p.i. with NMRI mice. The difference may be due to a more important Th1 cytokine environment in chronically infected mice, preventing IL-4 secretion and IgG1 production in these animals. Experiments with mice infected with T. gondii indicate that resistance to infection is dependent upon IFN-γ during both the acute and chronic stages of infection. NK cell production of IFN-γ is necessary but not sufficient for acquired immunity to T. gondii, and T cells (CD4+ and CD8+) are necessary to maintain resistance in chronically infected mice (19, 20, 22). IFN-γ production by these T cells may also stimulate antilactoferrin IgG2a production, although IL-12 and IFN-γ produced by cells that are activated during chronic infection are probably responsible for the enhanced level of IgG2a antibodies (18.9-fold).
FIG. 2.
Isotypic distribution of antilactoferrin antibodies in uninfected BALB/c mice and in animals infected for 40 days with T. gondii Beverley. Antilactoferrin antibodies in individual sera obtained 3 weeks after lactoferrin immunization were assayed. Results are shown as means ± standard errors for groups of four or five mice. Essentially identical results were obtained in three experiments.
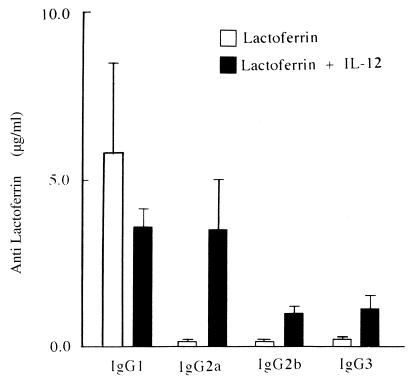
IgG subclass distribution of primary antibody responses to lactoferrin administered concomitantly with IL-12.
Two groups of BALB/c mice were used, with five mice in each group. On day 0, one group received 100 μg of lactoferrin per mouse, while the other group received the same amount of antigen with 0.05 μg of murine rIL-12 (provided by Genetics Institute, Inc., Cambridge, Mass.) diluted in sterile PBS containing 1% normal mouse serum. All mice were bled on day 21. Antibodies raised after immunization with lactoferrin alone belonged predominantly to the IgG1 subclass. Immunization with lactoferrin plus rIL-12 increased the IgG2a subclass antilactoferrin antibodies. This modification was mainly due to a dramatic increase in absolute amounts of IgG2a (20.1-fold) rather than to a decrease in IgG1 (1.6-fold) (Fig. 3). In mice immunized with protein antigens or hapten-protein conjugate, IL-12 strongly enhanced the humoral immune response by increasing the synthesis of antigen-specific antibody of the IgG2a subclass (21, 25, 30). This influence on B-cell responses persists in the memory of the immune system (6). In addition, the protein–IL-12 fusion protein was much more effective than free rIL-12 in enhancing IFN-γ synthesis (by T-helper cells) and increasing serum antiprotein IgG2a (23). In our study, rIL-12 administered together with lactoferrin similarly increased the IgG2a subclass (20.1-fold) and slightly decreased the IgG1 isotype (1.6-fold) (Fig. 3).
FIG. 3.
Isotypic distribution of antilactoferrin antibodies in BALB/c mice immunized on day 0 with 100 μg of lactoferrin per mouse alone or together with 50 ng of rIL-12. Antilactoferrin antibodies in individual sera obtained 3 weeks later were assayed by ELISA. Results are shown as means ± standard errors for groups of four or five mice.
Together, these observations suggest that T. gondii Beverley can modulate the host immune responses in the acute and chronic phases of infection.
Acknowledgments
We thank M. El Azami El Idrissi and N. Havaux for expert technical assistance.
J.P.C. is research associate with the Fonds National de la Recherche Scientifique (FNRS). This work was supported by a grant from Sorin Biomedica, Brussels, Belgium.
REFERENCES
- 1.Abdel-Wahab R M, Morsy T A, Abdel-Rahim M I, Essa M H, Al-Alfy Y E. The histopathological picture of concomitant infection with Leishmania major and Toxoplasma gondii in albino mice. J Egypt Soc Parasitol. 1989;19:1–2. [PubMed] [Google Scholar]
- 2.Afonso L C C, Scharton T M, Vieira L Q, Wysocka M, Trichieri G, Scott P. The adjuvant effect of interleukin-12 in a vaccine against Leishmania major. Science. 1994;263:235–237. doi: 10.1126/science.7904381. [DOI] [PubMed] [Google Scholar]
- 3.Albright J W, Holmes K L, Albright J F. Fluctuations in subsets of splenocytes and isotypes of Ig in young adult and aged mice resulting from Trypanosoma musculi infections. J Immunol. 1990;144:3970–3979. [PubMed] [Google Scholar]
- 4.Anderson R, Gao X M, Papakonstantinopoulou A, Roberts M, Dougan G. Immune response in mice following immunization with DNA encoding fragment C of tetanus toxin. Infect Immun. 1996;64:3168–3173. doi: 10.1128/iai.64.8.3168-3173.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Beverley J K A. Congenital transmission of toxoplasmosis through successive generations of mice. Nature (London) 1959;183:1348–1350. doi: 10.1038/1831348a0. [DOI] [PubMed] [Google Scholar]
- 6.Bliss J, Cleave V V, Murray K, Wiencis A, Ketchum M, Maylor R, Haire T, Resmini C, Abbas A K, Wolf S F. IL-12, as an adjuvant, promotes a T helper 1 cell, but does not suppress a T helper 2 cell recall response. J Immunol. 1996;156:887–894. [PubMed] [Google Scholar]
- 7.Bout D, Rousseaux R, Carlier Y, Capron A. Kinetics of classes and subclasses of total immunoglobulins and specific antibodies to Schistosoma mansoni during murine infection. Parasitology. 1980;80:247–256. doi: 10.1017/s003118200000072x. [DOI] [PubMed] [Google Scholar]
- 8.Burke J M, Roberts C W, Hunter C A, Murray M, Alexander J. Temporal differences in the expression of mRNA for IL-10 and IFN-γ in the brains and spleens of C57BL/10 mice infected with Toxoplasma gondii. Parasite Immunol. 1994;16:305–314. doi: 10.1111/j.1365-3024.1994.tb00353.x. [DOI] [PubMed] [Google Scholar]
- 9.Chan S H, Perussia B, Gupta J W, Kobayashi M, Pospisil M, Yong H A, Wolf S F, Young D, Clark S C, Trinchieri G. Induction of interferon-γ production by natural killer cell stimulatory factor: characterization of the responder cells and synergy with other inducers. J Exp Med. 1991;173:869–879. doi: 10.1084/jem.173.4.869. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Coffman R L, Carty J. A T cell activity that enhances polyclonal IgE production and its inhibition by interferon-gamma. J Immunol. 1986;136:949–954. [PubMed] [Google Scholar]
- 11.Coutelier J P, Van Roost E, Lambotte P, Van Snick J. The murine antibody response to lactate dehydrogenase-elevating virus. J Gen Virol. 1986;67:1099–1108. doi: 10.1099/0022-1317-67-6-1099. [DOI] [PubMed] [Google Scholar]
- 12.Coutelier J P, Van Der Logt J T M, Heessen F W A, Vink A, Van Snick J. Virally induced modulation of murine IgG antibody subclasses. J Exp Med. 1988;168:2373–2378. doi: 10.1084/jem.168.6.2373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Coutelier J P, Van Der Logt J T M, Heessen F W A. IgG subclass distribution of primary and secondary immune responses concomitant with viral infection. J Immunol. 1991;147:1383–1386. [PubMed] [Google Scholar]
- 14.Coutelier J P, Van Broeck J, Wolf S F. Interleukin-12 gene expression after viral infection in the mouse. J Virol. 1995;69:1955–1958. doi: 10.1128/jvi.69.3.1955-1958.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Deckert-Schluter M, Schluter D, Schmidt D, Schwendemann G, Wiestler O D, Hof H. Toxoplasma encephalitis in congenic B10 and BALB mice: impact of genetic factors on the immune response. Infect Immun. 1994;62:221–228. doi: 10.1128/iai.62.1.221-228.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Finkelman F D, Katona I M, Mosmann T R, Coffman L R. Interferon-γ regulates the isotypes of immunoglobulin secreted during in vivo humoral immune responses. J Immunol. 1988;140:1022–1027. [PubMed] [Google Scholar]
- 17.Fiorentino B F, Bond M W, Mosmann T R. Two types of mouse T helper cell. IV. Th2 clones secrete a factor that inhibits cytokine production by Th1 clones. J Exp Med. 1989;170:2081–2095. doi: 10.1084/jem.170.6.2081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Gajewski T F, Fitch F W. Anti-proliferative effect of IFN-γ in immunoregulation. I. IFN-γ inhibits the proliferation of Th2 but not Th1 murine helper T lymphocyte clones. J Immunol. 1988;140:4245–4252. [PubMed] [Google Scholar]
- 19.Gazzinelli R T, Hieny S, Wynn T, Wolf S, Sher A. IL-12 is required for the T-cell independent induction of IFN-γ by an intracellular parasite and induces resistance in T-deficient hosts. Proc Natl Acad Sci USA. 1993;90:6115–6119. doi: 10.1073/pnas.90.13.6115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Gazzinelli R T, Wysocka M, Denkers E Y, Hieny S, Caspar P, Trichieri G, Sher A. Parasite-induced IL-12 stimulates early IFN-γ synthesis and resistance during acute infection with Toxoplasma gondii. J Immunol. 1994;153:2533–2543. [PubMed] [Google Scholar]
- 21.Germann, Bongartz M, Dlugonska H, Hess H, Schmitt E, Kolbe L, Kölsch E, Podlaski F J, Gately M K, Rüde E. Interleukin-12 profoundly up-regulates the synthesis of antigen-specific complement-fixing IgG2a, IgG2b and IgG3 antibody subclasses in vivo. Eur J Immunol. 1995;25:823–829. doi: 10.1002/eji.1830250329. [DOI] [PubMed] [Google Scholar]
- 22.Khan I A, Matsuura T, Kasper L H. Interleukin-12 enhances murine survival against acute toxoplasmosis. Infect Immun. 1994;62:1639–1642. doi: 10.1128/iai.62.5.1639-1642.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Kim T S, Dekruyff R H, Rupper R, Maecker H T, Levy S, Umetsu D T. An ovalbumin-IL-12 fusion protein is more effective than ovalbumin plus free recombinant IL-12 in inducing a T helper cell type 1-dominated immune response and inhibiting antigen-specific IgE production. J Immunol. 1997;158:4137–4144. [PubMed] [Google Scholar]
- 24.Lacroix C, Levacher-Clergeot M, Chau F, Sumuyen M H, Sinet M, Pocidalo J J, Derouin F. Interactions between murine AIDS (MAIDS) and toxoplasmosis in co-infected mice. Clin Exp Immunol. 1994;98:190–195. doi: 10.1111/j.1365-2249.1994.tb06124.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.McKnight A J, Zimmer G J, Fogelman I, Wolf S F, Abbas A K. Effects of IL-12 on helper T cell-dependent immune responses in vivo. J Immunol. 1994;152:2172–2179. [PubMed] [Google Scholar]
- 26.Monteyne P, Renauld J C, van Broeck J, Dunne D W, Brombacher F, Coutelier J P. IL-4 independent regulation of in vivo IL-9 expression. J Immunol. 1997;159:2616–2623. [PubMed] [Google Scholar]
- 27.Mosmann T R, Cherwinski H, Bond M W. Two types of murine helper T cell clone. I. Definition according to profiles of lymphokine activities and secreted proteins. J Immunol. 1986;136:2348–2357. [PubMed] [Google Scholar]
- 28.Nguyen T D, De Kesel M, Bigaignon G, Hoet P, Pazzaglia G, Lammens M, Delmée M. Toxoplasma gondii tachyzoites and bradyzoites, detection in blood, urine and brain of infected mice. Clin Diagn Lab Immunol. 1996;3:635–639. doi: 10.1128/cdli.3.6.635-639.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Parker S J, Roberts C W, Alexander J. CD8+ T cells are the major lymphocyte subpopulation involved in the protective immune response to Toxoplasma gondii in mice. Clin Exp Immunol. 1991;84:207–212. doi: 10.1111/j.1365-2249.1991.tb08150.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Rempel J D, Wang M D, Hayglass K T. In vivo IL-12 administration induces profound but transient commitment to T helper cell type 1-associated patterns of cytokine and antibody production. J Immunol. 1997;159:1490–1496. [PubMed] [Google Scholar]
- 31.Snapper C M, Mond J J. Towards a comprehensive view of immunoglobulin class switching. Immunol Today. 1993;14:15–17. doi: 10.1016/0167-5699(93)90318-F. [DOI] [PubMed] [Google Scholar]
- 32.Suzuki Y, Joh K. Effect of the strain of Toxoplasma gondii on the development of toxoplasmic encephalitis in mice treated with antibody to interferon-gamma. Parasitol Res. 1994;80:125–130. doi: 10.1007/BF00933779. [DOI] [PubMed] [Google Scholar]
- 33.Suzuki Y, Yang Q, Yang S, Nguyen N, Lim S, Liesenfeld O, Kojima T, Remington J S. IL-4 is protective against development of toxoplasmic encephalitis. J Immunol. 1996;157:2564–2569. [PubMed] [Google Scholar]
- 34.Taylor D W, Pacheco E, Evans C B, Asofsky R. Inbred mice infected with Plasmodium yoelii differ in their antimalarial immunoglobulin isotype response. Parasite Immunol. 1988;10:33–46. doi: 10.1111/j.1365-3024.1988.tb00201.x. [DOI] [PubMed] [Google Scholar]
- 35.Vella A T, Pfau C J. The presence of an antierythrocyte autoantibody in C3HeB/FeJ mice after lymphocytic choriomeningitis virus infection. Autoimmunity. 1991;9:319–329. doi: 10.3109/08916939108997134. [DOI] [PubMed] [Google Scholar]
- 36.Villard O, Candolfi E, Despringre J L, Derouin F, Marcellin L, Viville S, Kien T. Protective effect of low doses of an anti-IL-4 monoclonal antibody in a murine model of acute toxoplasmosis. Parasite Immunol. 1995;17:233–236. doi: 10.1111/j.1365-3024.1995.tb01020.x. [DOI] [PubMed] [Google Scholar]
- 37.Vitetta E S, Ohara J, Myers C D, Layton J E, Krammer P H, Paul W E. Serological, biochemical, and functional identity of B cell stimulatory factor 1 and B cell differentiation factor for IgG1. J Exp Med. 1985;162:1726–1731. doi: 10.1084/jem.162.5.1726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Wolf S F, Temple P A, Kobayashi M, Young D, Dicig M, Lowe L, Dzialo R, Fitz L, Ferenz C, Hewick R M, Kelleher K, Herrmann S H, Clark S C, Azzoni L, Chan S H, Trinchieri G, Perussia B. Cloning of cDNA for natural killer cell stimulatory factor, a heterodimeric cytokine with multiple biologic effects on T and natural killer cells. J Immunol. 1991;146:3074–3081. [PubMed] [Google Scholar]
- 39.Zakroff S G H, Beck L, Platzer E G, Spielberg H L. The IgE and IgG subclass responses of mice to four helminth parasites. Cell Immunol. 1989;119:193–201. doi: 10.1016/0008-8749(89)90235-9. [DOI] [PubMed] [Google Scholar]