Abstract
Nitric oxide (NO) generated by gamma interferon (IFN-γ) activation of macrophages mediates the killing of many intracellular pathogens. IFN-γ is essential to innate resistance to Shigella flexneri infection. We demonstrate that NO is produced following S. flexneri infection both in mice and in activated cells in vitro and that while it is able to kill S. flexneri in a cell-free system, it is not required for clearance of S. flexneri in either infected mice or in activated cells in vitro.
Shigella spp. are the primary cause of bacillary dysentery worldwide (14). Pathogenic Shigella flexneri induces host cell uptake and escape from the endocytic vacuole into the cytoplasm, where cell-to-cell spread of bacteria is mediated through recruitment of host cell actin. In vitro, both macrophage and epithelial cell lines can be infected with S. flexneri. Infected mouse and human macrophage cells and HeLa cells foster intracellular replication and eventually die by apoptosis, oncosis, or necrosis (9, 19, 32). We have recently demonstrated that gamma interferon (IFN-γ) activation of macrophage or fibroblast cells induces host cell killing of intracellular S. flexneri (30), although the specific IFN-γ-induced mediators responsible for killing remain undefined.
Nitric oxide (NO) and other reactive nitrogen intermediates (RNIs) are the primary mediators of host cell defense against many intracellular and extracellular bacterial, parasitic, and fungal pathogens (8, 13, 17). Experiments implicating NO and RNIs in killing have largely been based on three complementary approaches. First, many pathogens, including Mycobacterium tuberculosis and Salmonella typhimurium, are susceptible to NO generated in cell-free systems (3, 5, 24). Second, inhibitors of nitric oxide synthase (NOS) inhibit activated cell killing of M. tuberculosis, Listeria monocytogenes, or S. typhimurium in vitro (1, 3, 28) and increase the susceptibility of mice to these same pathogens (2, 4, 5). Finally, mice with a targeted deletion in the inducible NOS gene (NOS2) demonstrate increased susceptibility to infection with M. tuberculosis, L. monocytogenes, Toxoplasma gondii, or Leishmania major (15, 16, 26, 31). In this report, we examine the contribution of NO or other RNIs in mediating killing of S. flexneri during infection.
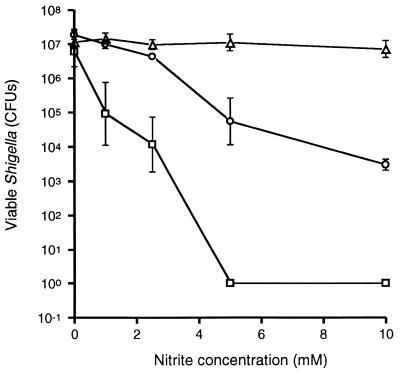
Under acidic conditions, NO and other RNIs can be generated from nitrite through a nitrous acid intermediate (27). To test whether NO generated in cell-free systems can exert bactericidal effects on S. flexneri, bacterial survival was measured in tryptic soy broth, at pH 4.5, 5.0, or 5.5, supplemented with sodium nitrite to final concentrations of 1 to 10 mM (Fig. 1). At pH 4.5 and 5.0, increasing bactericidal effects were observed with increasing nitrite concentrations, while no significant killing was observed at pH 5.5 at any nitrite concentration. Furthermore, no killing was observed at any pH without the addition of nitrite, demonstrating that the killing observed at pH 4.5 and 5.0 was not due solely to lower pH.
FIG. 1.
Numbers of viable wild-type S. flexneri strain 2457T organisms after 2 h of incubation in different concentrations of sodium nitrite in tryptic soy broth at pH 4.5 (squares), 5.0 (circles), or 5.5 (triangles). Each data point represents the mean of three independent determinations. Error bars, ±1 standard deviation. Where error bars are not seen, the error bar was smaller than the symbol.
IFN-γ in combination with either tumor necrosis factor alpha or lipopolysaccharide (LPS) has been shown to maximally stimulate NO production in mouse macrophages (3, 6). To define optimal concentrations for the production of NO in J774 and L2 cells, levels of nitrite (a stable end product of NO production) were assayed for the culture supernatants of cells treated overnight with medium containing no additive, recombinant mouse (J774 cells) or rat (L2 cells) IFN-γ (100 U/ml), LPS (1 μg/ml), or a combination of these factors. Only baseline levels of nitrite were found for J774 cells or L2 fibroblast cells not activated or activated with IFN-γ (100 U/ml) alone (Table 1). J774 cells activated overnight with IFN-γ and LPS produced 31.3 ± 12.5 μM (mean ± standard deviation) nitrite; increased nitrite concentrations were completely inhibited with aminoguanidine (2 mM). L2 cell monolayers activated in the same manner failed to produced nitrite after activation overnight or for 48 h (Table 1).
TABLE 1.
Nitrite concentrations in culture supernatants of uninfected J774 or L2 cells following addition of IFN-γ, LPS, or aminoguanidine
| Additive (concn) | Nitrite concn (μM) in:
|
||
|---|---|---|---|
| J774 cells at 16 ha | L2 cells
|
||
| 16 h | 48 h | ||
| None | <1.6b | <1.6 | <1.6 |
| IFN-γ (100 U/ml) | <1.6 | <1.6 | <1.6 |
| IFN-γ (100 U/ml) + LPS (1 mg/ml) | 31.3 ± 12.5c | <1.6 | <1.6 |
| IFN-γ (100 U/ml) + LPS (1 mg/ml) + aminoguanidine (2 mM) | <1.6 | <1.6 | <1.6 |
Time period of treatment with additive.
<1.6 μM, below the limits of detection.
Mean ± standard deviation.
Levels of nitrite were also measured for the culture supernatants of J774 macrophage cells, bone marrow macrophages derived from either C57BL/6 or NOS2−/− mice, and L2 cells that had been infected with S. flexneri. NOS2−/− mice used in this study were derived in a 129Sv × C57BL/6 mixed background and had been backcrossed into C57BL/6 for five to six generations. Since 129Sv mice, C57BL/6 mice, and F2 129Sv × C57BL/6 mice have identical susceptibilities to infection with S. flexneri (data presented below), macrophages harvested from the bone marrow of C57BL/6 mice were used as controls in experiments involving macrophages harvested from the bone marrow of NOS2−/− mice. J774 macrophage cells were seeded at 5.0 × 105 cells/well in 24-well plates, bone marrow macrophages were seeded at 2.2 × 105 to 2.4 × 105 cells/well in 48-well plates, and L2 fibroblast cells were grown as monolayers in 35-mm-diameter dishes. For J774 macrophage cells and bone marrow macrophages, S. flexneri infections were performed at a multiplicity of infection of 1.0, as described previously (30). For L2 fibroblast cells, S. flexneri infections were performed at a multiplicity of infection of 0.05 to 0.1. Following infection with S. flexneri, increases in nitrite concentrations were consistently observed for IFN-γ-activated J774 macrophage cells and bone marrow macrophages from C57BL/6 mice but not for IFN-γ-activated L2 fibroblast cells or bone marrow macrophages derived from NOS2−/− mice (Table 2). The increases in NO production that occurred following S. flexneri infection of IFN-γ-activated J774 cells were completely inhibited by addition of the NOS inhibitor aminoguanidine (2 mM) (Table 2). In the absence of IFN-γ activation, no increases in nitrite concentration were observed for any cell type (Table 2).
TABLE 2.
Nitrite concentrations in culture supernatants of S. flexneri-infected J774 macrophage cells, L2 fibroblast cells, and bone marrow macrophages from either C57BL/6 or NOS2−/− micea
| Activator (concn) | Nitrite concn (μM)
|
|||
|---|---|---|---|---|
| J774 cells | L2 cells | Bone marrow macrophages
|
||
| C57BL/6 | NOS2−/− | |||
| None | <1.6b | <1.6 | <1.6 | <1.6 |
| IFN-γ (100 U/ml) | 1.6 | <1.6 | 4.6 | <1.6 |
| IFN-γ + aminoguanidine (2 mM) | <1.6 | <1.6 | NDc | ND |
Nitrite concentrations were evaluated 4.5, 24, and 6 h postinfection for J774 cells, L2 cells, and bone marrow macrophages, respectively.
<1.6 μM, below the limits of detection.
ND, not determined.
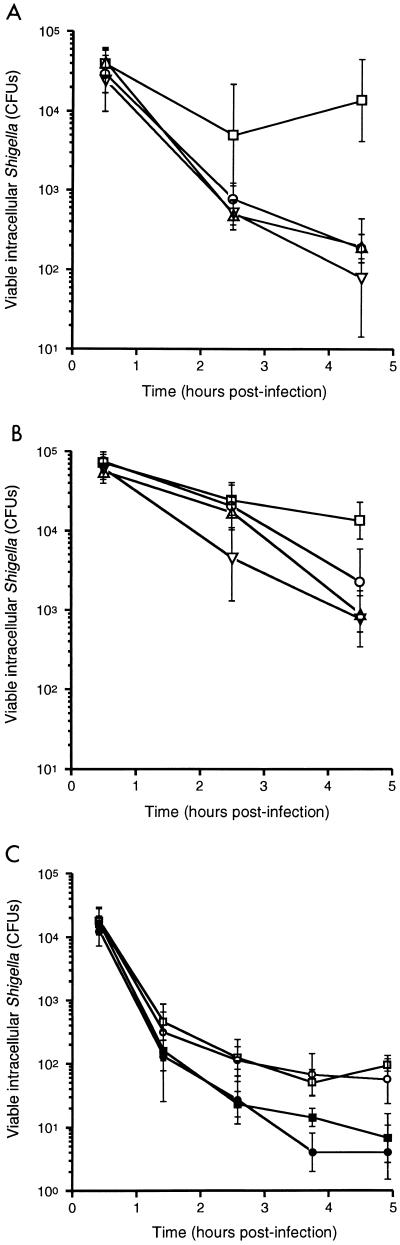
To assess whether NO and RNIs mediate the observed decreased intracellular survival of S. flexneri in IFN-γ-activated macrophages and fibroblast cells and the suppression of plaque formation by S. flexneri in fibroblast cells (30), we tested the effects of the NOS inhibitor aminoguanidine on killing of intracellular bacteria and bacterial plaque formation in S. flexneri-infected cells, as previously described (21, 30). J774 cells were activated with IFN-γ alone or IFN-γ and LPS (1 μg/ml), with and without aminoguanidine (2 mM), and infected 16 h later with the S. flexneri wild-type strain 2457T at a multiplicity of infection of 1.0. At either 2.5 or 4.5 h following infection, significantly lower numbers of intracellular S. flexneri were recovered from IFN-γ-activated cells than from cells not activated (Fig. 2A). Despite significant reductions in nitrite production in the presence of aminoguanidine (Table 2), no differences were observed in S. flexneri intracellular survival with or without aminoguanidine (Fig. 2A). In macrophage cells, measurements of S. flexneri intracellular survival were extended for only up to 4.5 to 5.0 h postinfection since maximal Shigella-induced cell death occurs between 3 and 4 h postinfection (32). We have previously shown that IFN-γ activation of L2 cells prevents the formation of plaques by S. flexneri (30). Treatment of IFN-γ-activated cells with aminoguanidine did not restore S. flexneri plaque formation (data not shown).
FIG. 2.
Intracellular survival of wild-type S. flexneri strain 2457T (A) and vacuolar escape-deficient mutant S. flexneri strain SF620 (B) within J774 macrophage cells following no activation (squares) or activation with IFN-γ (triangles), IFN-γ and LPS (circles), or IFN-γ, LPS, and aminoguanidine (inverted triangles). (C) Survival of 2457T within primary bone marrow macrophages derived from NOS2−/− (squares) or C57BL/6 (circles) mice following no activation (open symbols) or activation with IFN-γ (closed symbols). Each data point represents the mean of four to six independent determinations. Error bars, ±1 standard deviation.
S. flexneri is able to escape from the phagocytic vacuole into the cell cytoplasm (11, 25, 32, 33). Lysis of the phagocytic vacuole is mediated by the ipaB gene product (11). To determine whether the observed cellular NO-independent killing of wild-type S. flexneri was dependent upon bacterial escape into the cell cytoplasm, we evaluated the role of NO in killing of ipaB S. flexneri SF620 (20). Killing of intracellular ipaB S. flexneri in IFN-γ-activated J774 cells was also independent of NO production (Fig. 2B). Of note, IFN-γ-activated cellular killing of strain SF620 was less efficient than that of wild-type S. flexneri. This suggests that IFN-γ-activated mediators are more efficient in killing intracytoplasmic S. flexneri than intravacuolar S. flexneri.
Mice with a targeted deletion in the NOS2 gene offer a complementary approach for the analysis of the contribution of NO in host defense against pathogens (15, 23, 31). The survival of intracellular S. flexneri over time was determined in bone marrow macrophages derived from either NOS2−/− (genetic background described above) or C57BL/6 mice. Primary macrophages were seeded at 2.2 × 105 to 2.4 × 105 cells/well in 48-well plates and infected with S. flexneri at a multiplicity of infection of 1.0, as previously described (30). The same induction of S. flexneri killing by IFN-γ was observed in cells derived from NOS2−/− mice as in cells derived from C57BL/6 mice (Fig. 2C).
We have previously used a murine bronchopulmonary model of S. flexneri infection (18, 29) to assess the susceptibilities of mice with targeted deletions in specific aspects of the immune system (30). In this model, the lethal dose of S. flexneri for mice deficient in IFN-γ is at least 5 orders of magnitude less than that for immunocompetent mice, demonstrating an essential role for this cytokine in innate immunity (30). NO production in mice can be assessed by measuring the serum concentration of nitrite plus the more stable end product, nitrate (15). The concentration of nitrite plus nitrate in sera from C57BL/6 mice 24 h following intranasal infection with 107 S. flexneri organisms is 1.9-fold greater than that in sera drawn prior to infection (P = 0.004) (Table 3). In contrast, the concentration of nitrite plus nitrate in sera did not increase following S. flexneri infection in NOS2−/− mice (Table 3).
TABLE 3.
Concentration of nitrite plus nitrate in sera following intranasal infection of either C57BL/6 or NOS2−/− mice with 107 S. flexneri organismsa
| Mouse Strain | Nitrite + nitrate concn (μM)
|
Fold increase | P value | |
|---|---|---|---|---|
| Prior to infection | 24 h postinfection | |||
| C57BL/6 | 10.3 | 19.4 | 1.9 | 0.004 |
| NOS2−/− | 13.8 | 12.8 | 0.93 | 0.19 |
Data represent the mean serum concentrations of three to five mice per group.
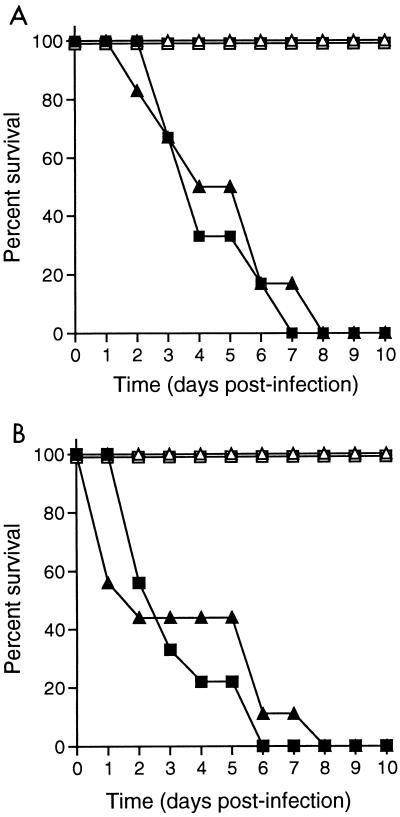
To assess the contribution of NO to the killing of S. flexneri in vivo, we examined the susceptibility of mice fed 2.5% aminoguanidine in drinking water, a dose previously demonstrated to markedly increase the susceptibility of mice to infection with M. tuberculosis or S. typhimurium (4, 5). Drinking water for groups of six C57BL/6 mice were supplemented or not supplemented with aminoguanidine for 1 week prior to challenge with 106 or 107 S. flexneri organisms and throughout the time course of infection (4). The lethal dose of S. flexneri for C57BL/6 mice has been previously shown to be 107 bacteria (30). No differences in lethal dose or time to death following infection were observed between aminoguanidine-treated mice and untreated control mice (Fig. 3A).
FIG. 3.
(A) Survival of C57BL/6 mice treated (triangles) or not treated (squares) with 2.5% aminoguanidine in drinking water after infection with 106 (open symbols) or 107 (closed symbols) S. flexneri 2457T organisms. (B) Survival of NOS2−/− (triangles) and control (F2 C57BL/6 × 129Sv) (squares) mice following infection with 106 (open symbols) or 107 (closed symbols) 2457T organisms. Each experimental group contained five to nine mice.
To further assess whether in vivo resistance to S. flexneri is an NO-independent process, the susceptibility of NOS2−/− mice to intranasal S. flexneri infection was examined. Groups of five to nine NOS2−/−, F2 129Sv × C57BL/6, 129Sv, and C57BL/6 mice were infected with either 106 or 107 S. flexneri organisms. No differences in lethal dose or time to death following infection were observed among these mouse strains (Fig. 3B) (30).
In conclusion, these data demonstrate that S. flexneri is sensitive to NO generated in cell-free conditions, yet both the killing of S. flexneri in infected cells in vitro and the resistance of mice are NO-independent processes. This independence from cellular NO-mediated killing is unusual for intracellular pathogens and has only recently been described for other pathogens under specific experimental conditions. For example, for some clinical isolates of M. tuberculosis, NOS inhibitors did not significantly reverse the effects of activated macrophage cell killing (24); for Chlamydia trachomatis, NOS2−/− mice and mice treated with NOS inhibitors demonstrated no difference in ability to clear infection (23); following Mycobacterium avium infection, immunocompetent and NOS2−/− mice demonstrate the same susceptibilities to infection (7); and for T. gondii, NO is not required to control acute murine infections but is essential for the prevention of persistent infection (26).
Due to the intrinsic limitations of infections in vitro and in the mouse bronchopulmonary model, we cannot rule out the possibility that NO contributes to killing of S. flexneri in intestinal infections. Of note, during human intestinal infection with Shigella, the expression of inducible NOS has been shown to increase in the rectal mucosa (12).
Data presented here suggest that for S. flexneri infection, either (i) IFN-γ-activated mediators other than NO or other RNIs are responsible for clearance of the organism or (ii) the mechanisms of S. flexneri resistance to NO are activated only after cellular invasion. The observation that S. flexneri with disruption of the superoxide dismutase gene (sodB) is attenuated in both in vitro and in vivo infection assays suggests that reactive oxygen intermediates may contribute to bacterial clearance during infection (10). However, since reactive oxygen intermediates react with RNIs to form compounds with more potent antibacterial activity (8, 22), this observation does not rule out a role for RNIs in the clearance of S. flexneri. If the mechanism of resistance of intracellular S. flexneri to NO is one that is activated only after cellular invasion, it might involve enhanced scavenging by low-molecular-weight thiols (e.g., glutathione or homocysteine), up-regulation of repair enzymes following injury induced by RNIs (e.g., RecBCD exonuclease or endonuclease IV), reduction of intracellular peroxynitrite formation though the superoxide dismutases (e.g., SodA or SodC) (8), or as-yet-undescribed factors. Moreover, identification of the cellular mediators responsible for killing intracellular S. flexneri will require further study.
Acknowledgments
We are indebted to J. Chan, L. Weiss, A. Sikora, and J. McKinney for helpful discussions and critical review of this manuscript; J. Stamler and F. Fang for useful advice; J. Mudgett, C. Nathan, and L. Weiss for providing NOS2−/− mice; and A. Zychlinsky for providing S. flexneri SF620.
This work was supported by NIH grants T32 GM07288 (S.S.W.) and AI35817 (M.B.G.), a Pew Scholars Award in the Biomedical Sciences (M.B.G.), and Established Investigator (M.B.G.) and Grant-in-Aid (M.B.G.) Awards from the American Heart Association.
REFERENCES
- 1.Beckerman K P, Rogers H W, Corbett J A, Schreiber R D, McDaniel M L, Unanue E R. Release of nitric oxide during the T cell-independent pathway of macrophage activation. J Immunol. 1993;150:888–895. [PubMed] [Google Scholar]
- 2.Boockvar K S, Granger D L, Poston R M, Maybodi M, Washington M K, Hibbs J B, Jr, Kurlander R L. Nitric oxide produced during murine listeriosis is protective. Infect Immun. 1994;62:1089–1100. doi: 10.1128/iai.62.3.1089-1100.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Chan J, Xing Y, Magliozzo R S, Bloom B R. Killing of virulent Mycobacterium tuberculosis by reactive nitrogen intermediates produced by activated murine macrophages. J Exp Med. 1992;175:1111–1122. doi: 10.1084/jem.175.4.1111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Chan J, Tanaka K, Carroll D, Flynn J, Bloom B R. Effects of nitric oxide synthase inhibitors on murine infection with Mycobacterium tuberculosis. Infect Immun. 1995;63:736–740. doi: 10.1128/iai.63.2.736-740.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.De Groote M A, Testerman T, Xu Y, Stauffer G, Fang F C. Homocysteine antagonism of nitric oxide-related cytostasis in Salmonella typhimurium. Science. 1996;272:414–417. doi: 10.1126/science.272.5260.414. [DOI] [PubMed] [Google Scholar]
- 6.Ding A H, Nathan C F, Stuehr D J. Release of reactive nitrogen intermediates and reactive oxygen intermediates from mouse peritoneal macrophages. J Immunol. 1988;141:2407–2412. [PubMed] [Google Scholar]
- 7.Doherty T M, Sher A. Defects in cell-mediated immunity affect chronic, but not innate, resistance of mice to Mycobacterium avium infection. J Immunol. 1997;158:4822–4831. [PubMed] [Google Scholar]
- 8.Fang F C. Mechanisms of nitric-oxide-related antimicrobial activity. J Clin Invest. 1997;99:2818–2825. doi: 10.1172/JCI119473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Fernandez-Prada C M, Hoover D L, Tall B D, Venkatesan M M. Human monocyte-derived macrophages infected with virulent Shigella flexneri in vitro undergo a rapid cytolytic event similar to oncosis but not apoptosis. Infect Immun. 1997;65:1486–1496. doi: 10.1128/iai.65.4.1486-1496.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Franzon V L, Arondel J, Sansonetti P J. Contribution of superoxide dismutase and catalase activities to Shigella flexneri pathogenesis. Infect Immun. 1990;58:529–535. doi: 10.1128/iai.58.2.529-535.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.High N, Mounier J, Prevost M C, Sansonetti P J. IpaB of Shigella flexneri causes entry into epithelial cells and escape from the phagocytic vacuole. EMBO J. 1992;11:1991–1999. doi: 10.1002/j.1460-2075.1992.tb05253.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Islam D, Veress B, Bardhan P K, Lindberg A A, Christensson B. In situ characterization of inflammatory responses in the rectal mucosae of patients with shigellosis. Infect Immun. 1997;65:739–749. doi: 10.1128/iai.65.2.739-749.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.James S L. Role of nitric oxide in parasitic infections. Microbiol Rev. 1995;59:533–547. doi: 10.1128/mr.59.4.533-547.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Katz S. New vaccine development establishing priorities: diseases of importance in developing countries. Washington, D.C: National Academy Press; 1986. [PubMed] [Google Scholar]
- 15.MacMicking J D, Nathan C, Hom G, Chartrain N, Fletcher D S, Trumbauer M, Stevens K, Xie Q, Sokol K, Hutchinson N, Chen H, Mudgett J S. Altered responses to bacterial infection and endotoxic shock in mice lacking inducible nitric oxide synthase. Cell. 1995;81:641–650. doi: 10.1016/0092-8674(95)90085-3. [DOI] [PubMed] [Google Scholar]
- 16.MacMicking J D, North R J, LaCourse R, Mudgett J S, Shah S K, Nathan C F. Identification of nitric oxide synthase as a protective locus against tuberculosis. Proc Natl Acad Sci USA. 1997;94:5243–5248. doi: 10.1073/pnas.94.10.5243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.MacMicking J D, Xie Q, Nathan C. Nitric oxide and macrophage function. Annu Rev Immunol. 1997;15:323–350. doi: 10.1146/annurev.immunol.15.1.323. [DOI] [PubMed] [Google Scholar]
- 18.Mallet C, VanDeVerg L, Collins H, Hale T. Evaluation of Shigella vaccine safety and efficacy in an intranasal challenged mouse model. Vaccine. 1993;11:190–196. doi: 10.1016/0264-410x(93)90016-q. [DOI] [PubMed] [Google Scholar]
- 19.Mantis N, Prevost M C, Sansonetti P J. Analysis of epithelial cell stress response during infection by Shigella flexneri. Infect Immun. 1996;64:2474–2482. doi: 10.1128/iai.64.7.2474-2482.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Menard R, Sansonetti P J, Parsot C. Nonpolar mutagenesis of the ipa genes defines IpaB, IpaC, and IpaD as effectors of Shigella flexneri entry into epithelial cells. J Bacteriol. 1993;175:5899–5906. doi: 10.1128/jb.175.18.5899-5906.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Oaks E V, Wingfield M E, Formal S B. Plaque formation by virulent Shigella flexneri. Infect Immun. 1985;48:124–129. doi: 10.1128/iai.48.1.124-129.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Pacelli R, Wink D A, Cook J A, Krishna M C, DeGraff W, Friedman N, Tsokos M, Samuni A, Mitchell J B. Nitric oxide potentiates hydrogen peroxide-induced killing of Escherichia coli. J Exp Med. 1995;182:1469–1479. doi: 10.1084/jem.182.5.1469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Ramsey K H, Miranpuri G S, Poulsen C E, Marthakis N B, Braune L M, Bryne G I. Inducible nitric oxide synthase does not affect resolution of murine chlamydial genital tract infections or eradication of chlamydiae in primary murine cell culture. Infect Immun. 1998;66:835–838. doi: 10.1128/iai.66.2.835-838.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Rhoades E R, Orme I M. Susceptibility of a panel of virulent strains of Mycobacterium tuberculosis to reactive nitrogen intermediates. Infect Immun. 1997;65:1189–1195. doi: 10.1128/iai.65.4.1189-1195.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Sansonetti P J, Ryter A, Clerc P, Maurelli A T, Mounier J. Multiplication of Shigella flexneri within HeLa cells: lysis of the phagocytic vacuole and plasmid-mediated contact hemolysis. Infect Immun. 1986;51:461–469. doi: 10.1128/iai.51.2.461-469.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Scharton-Kersten T M, Yap G, Magma J, Sher A. Inducible nitric oxide is essential for host control of persistent but not acute infection with the intracellular pathogen Toxoplasma gondii. J Exp Med. 1997;185:1261–1273. doi: 10.1084/jem.185.7.1261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Stuehr D J, Nathan C F. Nitric oxide. A macrophage product responsible for the cytostasis and respiratory inhibition in tumor target cells. J Exp Med. 1989;169:1543–1555. doi: 10.1084/jem.169.5.1543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Umezawa K, Akaike T, Fujii S, Suga M, Setoguchi K, Ozawa A, Maeda H. Induction of nitric oxide synthesis and xanthine oxidase and their roles in the antimicrobial mechanism against Salmonella typhimurium infection in mice. Infect Immun. 1997;65:2932–2940. doi: 10.1128/iai.65.7.2932-2940.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Van De Verg L L, Mallet C, Collins H H, Larsen T, Hammack C, Hale T L. Antibody and cytokine responses in a mouse pulmonary model of Shigella flexneri serotype 2a infection. Infect Immun. 1995;63:1947–1954. doi: 10.1128/iai.63.5.1947-1954.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Way S S, Borczuk A C, Dominitz R, Goldberg M B. An essential role for gamma interferon in innate resistance to Shigella flexneri infection. Infect Immun. 1998;66:1342–1348. doi: 10.1128/iai.66.4.1342-1348.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Wei X, Charles I G, Smith A, Ure J, Feng G, Huang F, Xu D, Muller W, Moncada S, Liew F Y. Altered immune responses in mice lacking inducible nitric oxide synthase. Nature. 1995;375:408–411. doi: 10.1038/375408a0. [DOI] [PubMed] [Google Scholar]
- 32.Zychlinsky A, Prevost M C, Sansonetti P J. Shigella flexneri induces apoptosis in infected macrophages. Nature. 1992;358:167–169. doi: 10.1038/358167a0. [DOI] [PubMed] [Google Scholar]
- 33.Zychlinsky A, Kenny B, Menard R, Prevost M C, Holland I B, Sansonetti P J. IpaB mediates macrophage apoptosis induced by Shigella flexneri. Mol Microbiol. 1994;11:619–627. doi: 10.1111/j.1365-2958.1994.tb00341.x. [DOI] [PubMed] [Google Scholar]