Abstract
In this report, multiple abnormalities (bilateral ovarian cysts, cystic endometrial hyperplasia and pyometra (CEH–P), mammary adenoma, fibrosarcoma and cystic-papillary adenocarcinoma) identified in a queen continually administered medroxyprogesterone acetate (MPA) for 9 years are described. An 11-year-old domestic shorthair intact queen was presented for reduced appetite, polyuria, polydipsia, abdominal distension, and mammary mass. Pyometra was diagnosed based upon clinical, ultrasonographic and radiographic examinations. Mastectomy and ovariohysterectomy (OHE) were performed for treatment. Bilateral ovarian cysts were detected during OHE. Histopathologically, follicular ovarian cysts, CEH–P, and benign and neoplastic mammary lesions were identified. We suggest that, the pathological abnormalities may have been attributed to adverse effects of prolonged administration of MPA.
Cystic endometrial hyperplasia and pyometra (CEH–P) complex is the most frequent and important endometrial disorder in cats and a sequel to progesterone stimulation of the endometrium, and by ascending uterine infection with vaginal bacteria. 1–3 In addition, mammary tumours are the third most common neoplasm in the female cat, following tumours of the skin and lymphoid tissue. 4 In the queen, ovarian cysts have been reported to be common and their frequency is reported to increase with age. 2
Ovarian steroid hormones and their synthetic derivatives, especially progestagens used to control or suppress oestrus, may enhance mammary tumour genesis and CEH–P in cats. 1 The risk of both CEH–P and mammary tumours is related to the particular progestagen used, the route of administration, the amount administered, and the duration of treatment. 5–10
An 11-year-old domestic shorthair intact queen was presented to University of Uludag, Faculty of Veterinary Medicine, Clinics of Obstetrics and Gynecology, for reduced appetite, polyuria, polydipsia, abdominal distension and enlargement of right inguinal mammary gland. The queen had delivered a litter of kittens at 2 years old and subsequent oestrus cycles were inhibited by MPA (Perlutex; Topkim, Turkey) administration, 25 mg total dose, intramuscularly, twice per year for 7 years. During the 2 years before presentation, MPA was administered once yearly, in a local clinic. The mass in the mammary gland was detected 15 days before presentation.
On physical examination, the cat was mildly febrile (rectal temperature 39.9°C; normal 38.5–39.5°C) and estimated to be 5% dehydrated. Routine haematological examination (Haemocell Counter Serono; Baker Diagnostic, England) showed leukocytosis and mild neutrophilia. All values are shown in Table 1. Right inguinal mammary gland was enlarged and had 7×4×4 cm dimensions.
Table 1.
Results of haematological examination
| Haematological parameters | The patient's values | Average limits |
|---|---|---|
| White blood cell count (k/μl) | 35×103 | 5.5–19×103 |
| Total neutrophils (%) | 85 | 35–75 |
| Red blood cell count (millions/μl) | 4.96×106 | 5–10×106 |
| Haemoglobin (g/dl) | 9.7 | 8–15 |
| Haematocrit (%) | 29.9 | 24–45 |
Vaginal discharge was not detected before and during examination. Uterus enlargement was detected with abdominal radiography and closed pyometra was diagnosed with detection of segmentally distended (approximately 6 cm diameter), fluid-filled uterine horns using abdominal sonography. Thoracic radiographs were performed to screen for possible pulmonary metastases, and no pathological changes were identified. The mammary gland was found to contain solid and cystic areas on clinical, sonographic and radiographic examination.
In general, closed cervix pyometra is treated with surgical intervention, together with antibiotics and fluid therapy. 2 This treatment should be performed as soon as possible after the diagnosis is confirmed. Therefore, according to Hedlund, 11 one of the appropriate antibiotics (Cephazolin, Sefazol; Mustafa Nevzat, Turkey; 20 mg/kg BW, IM, q12h for 7 d) and fluid therapy (5% dextrose+lactated Ringers, Polifarma, Turkey; 60–120 ml, IV, given as constant drips for 80 min, q12h for 3 d) were administered. OHE and a full mastectomy were performed for surgical treatment as described by Hedlund. 11 As an anaesthetic regime, xylazine (3 mg/kg: Alfazyn; Egevet, Turkey) was injected intramuscularly for premedication and ketamine (5 mg/kg: Alfamin; Egevet) was injected intramuscularly for induction of anaesthesia. After this initial period, isoflurane (Forane; Abbott, UK) at a vapouriser setting of 1% was delivered in the oxygen stream. The anaesthetic gas concentration was maintained at the same setting until the end of the operation. The animal was re-examined 6 and 12 months after the operation and no abnormalities were detected.
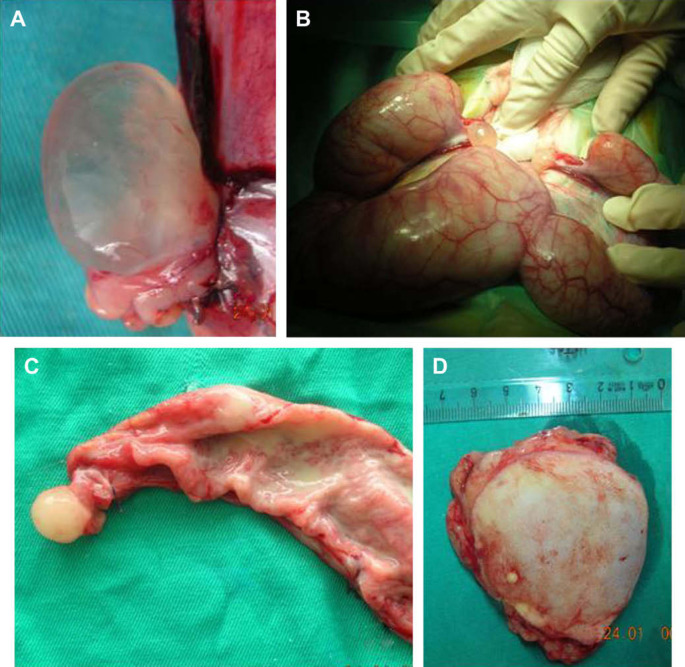
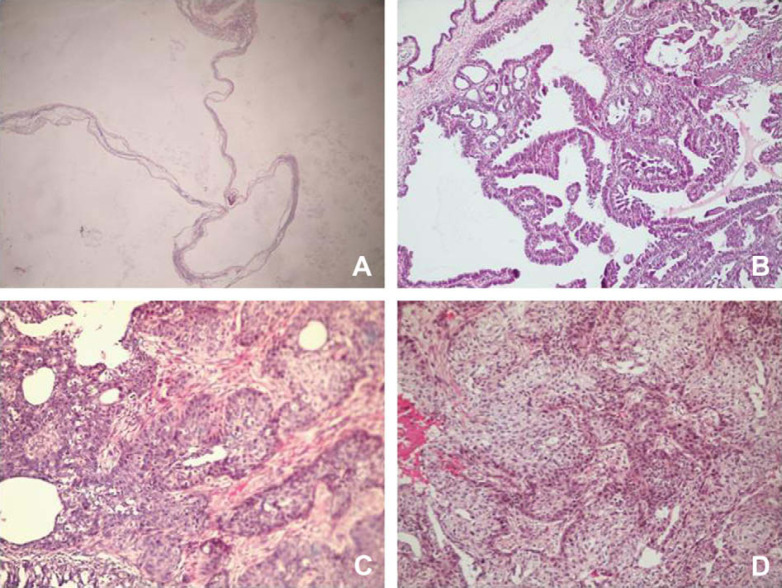
On macroscopic examination, bilateral ovarian cysts, 1.2 cm in size and filled with serous fluid, were observed (Fig 1A). Both uterine horns were distended, congested, oedematous, and filled with pus (Fig 1B). Incision of the uterus revealed the thickened mucosa (Fig 1C). The right mammary gland contained a soft whitish mass with cystic and solid areas (Fig 1D). On microscopic examination, both ovaries had cystic follicles (Fig 2A) consisting of granulosa and theca cells. The epithelial lining of the uterus lumen was vacuolated and degenerate neutrophils were observed in the lumen. Glands in the propria were enlarged and cystic. They were filled with mononuclear cells and seromucinous material. Epithelial hyperplasia was observed in the epithelium of some glands (Fig 2B). Similar inflammatory cell infiltrations were seen among the glands.
Fig 1.
Follicular cyst in right ovary (A) and fluid-filled uterus (B) and pus in left cornu uteri (C) and mammary tumour in the right inguinal gland (D).
Fig 2.
Follicular cyst (A) in ovary. Cystic-papillar areas (B) in uterus. Fibrosarcomatous (C) and hyperplasic areas in alveolar epithelium (D) in mammary glands.
In mammary tissue, three different lesions were identified. In some areas regular proliferations of the mammary epithelium was seen (Fig 2C), while other areas consisted of tumoural proliferations of fibroblasts and fibrocytes with occasional multinuclear giant cells (Fig 2D). Papillary proliferations of epithelial cells with clear nuclei and cytoplasm and prominent nucleoli, together with large cystic areas were also seen. The diagnoses were made as mammary adenoma, fibrosarcoma and cystic-papillary adenocarcinoma.
MPA is a synthetic derivative of progesterone and is commonly used for contraceptive therapy in queens in some countries. 1,2,9 In general, a treatment period of 6 months is considered adequate in most individuals, although longer treatments can also be safe provided that the female is given a rest of 1–2 months every 4–6 months. 9 While most bitches and queens may tolerate treatment periods of more than 6 months, animals with a pre-existing disease such as subclinical diabetes, microscopic mammary lesions or CEH may become clinically ill. 9
CEH–P complex is the most frequent and important endometrial disorder in cats. A general agreement is that hormonal factors play a predisposing role in the development of disease. 2 In previous studies, the administration of progesterone to healthy bitches, over a prolonged period of time, has resulted in the development of uterine lesions that are comparable with spontaneously occurring CEH–P. 3,5 CEH–P complex in cats has been considered to be roughly comparable with the disease in dogs. 1
Regular administration of progestagens is associated with an increased risk of both benign and malignant mammary tumours in cats. 6,7,8,12 Studies reveal history of previous progestational contraceptive therapy in cats with mammary masses. 6,13 This fact supports the strong predisposition of development of benign and malignant mammary lesions. The risk of both CEH–P and mammary tumours is related to the particular progestagen used, the route of administration, the amount administered, and the duration of treatment. 5,10,12
Ovarian follicular cysts can develop in many mammals, but the physiological mechanism leading to this condition remains undefined. However, in the queen, follicular cysts that arise from mature or atretic follicles are reported to be most common type. Affected queens may be asymptomatic or may exhibit prolonged oestrus if cells lining the cyst secrete oestrogen. 2 In the case reported here, the queen did not show oestrus behaviour and according to the histopathological examination, the structures were defined as follicular cysts. Application of subcutaneous or intramuscular repositol forms of synthetic progestins, which are potent anti-ovulatory compounds with a progestational activity 25 times greater than that of endogenous progesterone, 7 may have inhibited the luteinising hormone surge necessary to induce ovulation during oestrus and may have caused formation of these follicular cysts.
In this case, administration of MPA continuously for 9 years may have led to the development of malignant mammary tumours, CEH–P, and ovarian cysts.
References
- 1.Feldman E.C., Nelson R.W. Feline Reproduction. Canine and Feline Endocrinology and Reproduction, 3rd edn, 2004, Saunders: Missouri, 1016–1044. [Google Scholar]
- 2.Johnston S.D., Kustritz M.V.R., Olson P.N. Disorders of the Mammary Glands of Queens. Canine and Feline Theriogenology, 2001, WB Saunders: Philadelphia, 474–476. [Google Scholar]
- 3.Noakes D.A., Dhaliwal G.K., England G.C. Cystic endometrial hyperplasia/pyometra in dogs: A review of the causes and pathogenesis, J Reprod Fertil 57 (suppl.), 2001, 395–406. [PubMed] [Google Scholar]
- 4.MacLachlan N.J., Kennedy P.C. Tumors of the genital system. Meuten D.J. Tumors in Domestic Animals, 4th edn, 2002, Iowa State University Press, 509–546. [Google Scholar]
- 5.Arora N., Sandford J., Browing G.F., Sandy J.R., Wright P.J. A model for cystic endometrial hyperplasia/pyometra complex in the bitch, Theriogenology 66, 2006, 1530–1536. [DOI] [PubMed] [Google Scholar]
- 6.Hayden D.W., Johnston S.D., Kiang D.T., Johnson K.H., Barnes D.M. Feline mammary hypertrophy/fibroadenoma complex: Clinical and hormonal aspects, Am J Vet Res 42, 1981, 1699–1703. [PubMed] [Google Scholar]
- 7.Hayden D.W., Barnes D.M., Johnson K.H. Morphologic changes in the mammary gland of megestrol acetate treated and untreated cats: A retrospective study, Vet Pathol 26, 1989, 104–113. [DOI] [PubMed] [Google Scholar]
- 8.Misdorp W., Romijn A., Hart A.A. Feline mammary tumors: A case-control study of hormonal factors, Anticancer Res 11, 1991, 1793–1797. [PubMed] [Google Scholar]
- 9.Romagnoli S. Clinical use of hormones in the control of reproduction in bitches and queens, Proceedings of the Veterinary Sciences Congress (SPCV); 2002 Oct 10–12; Oeiras, Portugal, 2002, 162–166.
- 10.Verstegen J.P. Pharmacological control of reproduction in the cat. Simpson G. Manual of Small Animal Reproduction and Neonatology, 1st edn, 1998, British Small Animal Veterinary Association (BSAVA): Cheltenham, UK, 219–227. [Google Scholar]
- 11.Hedlund C.S. Surgery of the reproductive and genital systems. Fossum T.W. Small Animal Surgery, 2nd edn, 2002, Mosby, 632–644. [Google Scholar]
- 12.Misdorp W. Progestagens and mammary tumors in dogs and cats, Acta Endocrinol 125, 1991, 27–31. [PubMed] [Google Scholar]
- 13.Amorim V.F., Souza H.J.M., Ferrerira A.M.R., Fonseca A.B.M. Clinical, cytological and histopathological evaluation of mammary masses in cats from Rio de Janeiro, Brazil, J Feline Med Surg 8, 2006, 379–388. [DOI] [PMC free article] [PubMed] [Google Scholar]