Abstract
The role of quorum sensing by Pseudomonas aeruginosa in producing cytotoxicity has not been fully investigated. Strains of P. aeruginosa have been characterized as having an invasive or a cytotoxic phenotype (S. M. J. Fleiszig et al., Infect. Immun. 65:579–586, 1997). We noted that the application of a large inoculum of the invasive strain 6294 caused cytotoxicity of cultured epithelial cells. To investigate this dose-related cytotoxicity, we compared the behavior of 6294 to that of another invasive strain, PAO1, and determined whether the cytotoxicity could be related to quorum sensing. Both invasive strains, 6294 and PAO1, appear to have quorum-sensing systems that were operative when large doses of bacteria were applied to cultured lung epithelial cells or instilled into the lungs of animals. Nonetheless, only 6294 was cytotoxic. Cytotoxicity induced by 6294 correlated with increased elastase production. These experiments suggest that there are multiple mechanisms for the induction of cytotoxicity, pathology, and mortality in vivo. However, in vivo cytotoxicity and mortality, but not pathology, could be predicted by quantitative in vitro cellular damage experiments utilizing a range of bacteria-to-cell ratios. It appears that quorum sensing may inversely correlate with virulence in that strains that produced PAI [N-(3-oxododecanoyl) homoserine lactone] also appeared to attract more polymorphonuclear leukocytes in vivo and were possibly eliminated more quickly. In addition, exoproduct production in bacteriological medium in vitro may differ significantly from exoproduct expression from infections in vivo or during cocultivation of bacteria with tissue culture cells.
Pseudomonas aeruginosa lung infections occur frequently in critically ill patients (8, 26, 27). Patients who are mechanically ventilated are especially at high risk for developing P. aeruginosa pneumonia (1, 4, 30). Mortality rates are higher in patients who have ventilator-associated pneumonia (VAP) than in patients who do not develop VAP (7, 34). In addition, the mortality rate of patients with VAP due to P. aeruginosa is significantly higher than the mortality rate associated with VAP secondary to other bacteria (9). P. aeruginosa pneumonia is frequently associated with the development of septic shock and multiple organ failure which are also correlated with high mortality rates (1, 22, 29).
The high mortality rate of P. aeruginosa pneumonia may be secondary to the ability of some of the P. aeruginosa strains to cause necrosis of the lung epithelium and to disseminate into the circulation rapidly (17, 32, 33). Recently, different strains of P. aeruginosa have been characterized as having either an invasive or a cytotoxic phenotype (11, 12, 25). Cytotoxic strains cause necrosis of epithelial cells in vitro and in vivo (11, 12, 25). We recently reported that the ability to induce acute cytotoxicity is associated with the expression of ExoU, a type III secretory cytotoxin regulated by the transcriptional activator ExsA (10). Invasive strains were found to lack exoU (10).
Although the expression of ExoU has been linked to cytotoxicity, P. aeruginosa synthesizes several extracellular virulence determinants which may contribute to either cytotoxicity or invasion. Many exoproducts of P. aeruginosa, such as elastase (LasB), LasA protease, alkaline protease, and exotoxin A, are not actively produced until bacterial cell density reaches a threshold concentration (15, 16). One mechanism by which bacteria respond to changes in cell density is explained as the cell density-dependent transcriptional regulation often called a quorum-sensing system (14, 23). The LasR-LasI system is one of the quorum-sensing systems found in P. aeruginosa and shows homology to the LuxR-LuxI system of V. fischeri (15, 16, 23). In this quorum-sensing system, LasI (autoinducer synthase) directs the synthesis of the autoinducer PAI-1 [N-(3-oxododecanoyl) homoserine lactone], which triggers a transcriptional activator, LasR, to induce virulence genes such as lasB, lasA, and toxA (16). In addition, P. aeruginosa possesses a second quorum-sensing system, the RhlR-RhlI system, which regulates rhamnolipid and LasB production (2, 19). PAO1, a P. aeruginosa strain classified as an invasive strain due to the lack of exoU (10, 13), has the LasR-LasI system (23) and the RhlR-RhlI system (2, 19). Cell density-dependent virulence regulatory systems also appear to be involved in the pathogenesis of bacterial dissemination and in causing the mortality associated with P. aeruginosa pneumonia (28).
In the present study, we investigated the cell density-dependent cytotoxicity and lung epithelial injury caused by two invasive P. aeruginosa strains. We found differences among the two invasive strains investigated, 6294 and PAO1, suggesting that quorum-sensing systems may be involved in the production of density-dependent cytotoxicity.
MATERIALS AND METHODS
Bacterial strains and preparation.
P. aeruginosa PA103 is a cytotoxic strain (12, 17). PAO1 was generously provided by Barbara Iglewski (University of Rochester, Rochester, N.Y.). Strain 6294 was generously provided by Suzanne Fleiszig (University of California, Berkeley, Calif.). The genotypic details of these strains were described previously (10, 12, 17); in brief, PA103 expresses ExoU and is cytotoxic at a relatively low initial bacterial density, whereas 6294 and PAO1 are not cytotoxic (do not possess exoU) at a similar bacterial density. These strains were stored as bacterial stocks at −70°C in 10% sterile skim milk solutions. Bacteria from these frozen stocks were streaked onto Trypticase soy agar plates and grown in a deferrated dialysate of Trypticase soy broth supplemented with 10 mM nitrilotriacetic acid (Sigma Chemical Co., St. Louis, Mo.), 1% glycerol, and 100 mM monosodium glutamate at 33°C for 13 h in a shaking incubator. Cultures were centrifuged at 8,500 × g for 5 min, and the bacterial pellet was washed three times in Ringer’s lactate solution and diluted into the appropriate number of CFU per milliliter in Ringer’s lactate solution as determined by spectrophotometry. The numbers of bacteria were confirmed by determining CFU of diluted aliquots on sheep blood agar plates.
In vitro cytotoxicity assay.
A human bronchial epithelial cell line, immortalized by simian virus 40 (SV40) (BEAS-2B, ATCC CRL9609) was cultured in Dulbecco’s modified Eagle’s medium H-16 with 10% heat-inactivated fetal calf serum, penicillin, and streptomycin; the cells were kept in an incubator in 5% CO2. When the cells reached confluence, 2 × 104 cells were transferred to 96-well tissue culture plates and incubated overnight. The following day, one of the three different P. aeruginosa strains (PA103, PAO1, or 6294) was mixed with medium (not containing antibiotics) and applied to the cells for a 6-h interval. Three different inocula (109, 107, and 105 CFU/ml) were applied. Cytotoxicity was quantitated by measuring the production of lactate dehydrogenase (LDH) by using a cytotoxicity assay kit (Cytotox 96; Promega, Madison, Wis.).
Infection of mice with three different strains.
All animal experiments were performed in compliance with and with the approval of the Animal Care Committee of the University of California, San Francisco. The mice were briefly anesthetized with inhaled methoxyflurane (Methofane; Pitman-Moore, Mundelein, Ill.) and were placed in a supine position at a head-up angle of approximately 30°. For each mouse, 50 μl of a bacterial inoculum was instilled slowly into the left lobe of the lung with a gavage needle (modified animal feeding needle, 24 G; Popper & Sons, Inc., New Hyde Park, N.Y.) inserted into the trachea via the mouth. The proper insertion of the needle was confirmed by observing the movement of the solution inside the syringe during the animal’s respiratory efforts. The weight of the syringe was measured prior to and after the tracheal instillation to confirm the volume instilled into each mouse.
Measurement of lung epithelial permeability and lung edema after P. aeruginosa instillation.
The lung instillate was a combination of 0.05 μCi of 125I-labeled human serum albumin (Merck-Frosst, Quebec, Canada), 5 μg of anhydrous Evan’s blue, and 5% mouse albumin in 50 μl of Ringer’s lactate solution with an appropriate quantity of the specified P. aeruginosa. The total radioactivity (cpm) in the instillate was measured with a gamma radiation counter (Auto-Gamma, model 5550; Packard, Downers Grove, Ill.). The 50-μl instillate was always instilled into the left lobe of the lung of each anesthetized mouse. After 4 h, mice were reanesthetized with pentobarbital (2.0 mg, intraperitoneal), and blood was collected by carotid arterial puncture. Sternotomies were performed after additional pentobarbital was administered. All pleural fluid was collected in sterile containers. The lungs, tracheas, oropharynxes, stomachs, and livers were harvested, and the levels of radioactivity of these samples were measured. The quantity of 125I-albumin that had entered circulation was calculated by multiplying the counts measured in the terminal blood sample (per milliliter) times the blood volume (body weight × 0.07). Lungs were homogenized and placed in preweighed aluminum pans and dried in an oven at 80°C for 3 days to calculate the wet weight to dry weight ratios of the lungs (wet/dry ratios) as previously described (17, 32, 33). The wet/dry ratio is a well-accepted index of lung edema (32).
Bacterial culture in the lungs, livers, and pleural fluid samples from infected mice.
The lungs were homogenized in sterile containers with sterile water. Lung homogenates were sequentially diluted and placed on sheep blood agar plates for a assessment of the bacterial numbers in the lungs. Similarly, the livers were homogenized and samples of the homogenate were diluted and plated on agar plates for quantitative culture. Pleural fluids were cultured directly onto blood agar plates. The lowest sensitivity limits of the bacteriological tests were 10 CFU/ml of blood and 100 CFU/g of liver.
Histopathological analysis.
Inocula (50 μl of instillate without bacteria or with PA103, PAO1, or 6294) were instilled into the lungs at three different concentrations (105, 107, 109 CFU/ml). Four hours after instillation, the lungs were perfused with 10% buffered formalin phosphate for fixation and embedded in paraffin. Mounted sections were stained with hematoxylin-eosin.
Analysis for quorum-sensing modules (autoinducer bioassay).
To measure the autoinducer production by the three different P. aeruginosa strains, bioassays for the autoinducer PAI-1 were performed as previously reported (24). Overnight cultures of Escherichia coli MG4 (a generous gift from E. P. Greenberg, University of Iowa, Iowa City) containing a plasmid with a lasR and lasB::lacZ transcriptional fusion in a conditioned medium were diluted in supplemented A medium to an optical density at 660 nm of 0.1 and stored on ice. Each bioassay consisted of 2 ml of the cell suspension plus a test sample. After 5.5 h at 30°C, β-galactosidase activity was measured as described by Miller (21). The activities of the autoinducer in the following samples were tested: (i) bacterial culture medium (a deferrated dialysate of Trypticase soy broth supplemented with 10 mM nitrilotriacetic acid [Sigma Chemical], 1% glycerol, and 100 mM monosodium glutamate) in which each of the three different P. aeruginosa strains were grown at 33°C for 13 h in a shaking incubator and (ii) cell culture medium (DME-H16 with 10% heat-inactivated fetal calf serum) in which human bronchial cells (BEAS-2B) (106 cells/ml) were cultured with the three different P. aeruginosa strains at three different bacterial concentrations (109, 107, and 105 CFU/ml). Samples were filtered (pore size, 2 μm) and mixed with 2 ml of a suspension of MG4 at the designated ratios.
Zymography and gel overlays.
Zymography was carried out with nonreducing sodium dodecyl sulfate (SDS)–8% polyacrylamide gels (Bio-Rad, Hercules, Calif.) containing gelatin (0.2%; Sigma Chemical Co.) as previously described (31). Briefly, medium conditioned with or without BEAS cells, BEAS cells plus PA103, PAO1, or 6294, or bacteria alone were applied to gels at a 1:1 ratio with sample buffer without reducing agents or boiling. Sample size was determined based on the amount of broth required to visualize the proteolytic activity without diffusion into adjacent lanes. The samples were electrophoresed under standard conditions (31). After electrophoresis, SDS was removed from the gels by using an aqueous solution containing 2.5% Triton X-100 (Sigma Chemical Co.). The gels were subsequently incubated for 18 h in 50 mM Tris buffer, pH 8.0, containing 1 mM CaCl2, 1 μM ZnCl2, and 150 mM NaCl. Proteolytic activity from separated bands was visualized by staining the gels with Coomassie brilliant blue. Elastase and alkaline proteinase were used as standards for identification of proteinases. Molecular weight standards were reduced with dithiothreitol prior to application to the gels. Apparent molecular weights were calculated relative to these standards by using the program SigmaGel (SPSS, Chicago, Ill.). All gel assays were repeated at least three times with similar results.
Western blots.
Samples were electrophoresed with 5% dithiothreitol in the sample buffer by being boiled on SDS–8% polyacrylamide gels (18). The proteins were electroblotted to nitrocellulose (Schleicher and Scheuell, Keene, N.H.), probed with a rabbit polyclonal antibody to elastase (a gift from B. Iglewski), washed, and subsequently incubated with a horseradish peroxidase-conjugated goat anti-rabbit immunoglobulin G (3). Bands were visualized with the chemiluminescence ECL kit (Amersham, Arlington Heights, Ill.).
Statistical analysis.
Fisher’s exact test was used for comparison of bacteriological data. The Mantel-Cox log rank test was used for survival analysis. One-way analysis of variance and the Bonferroni multiple comparison test were used for all other comparisons. Significance was accepted at P < 0.05.
RESULTS
In vitro cytotoxicity assay.
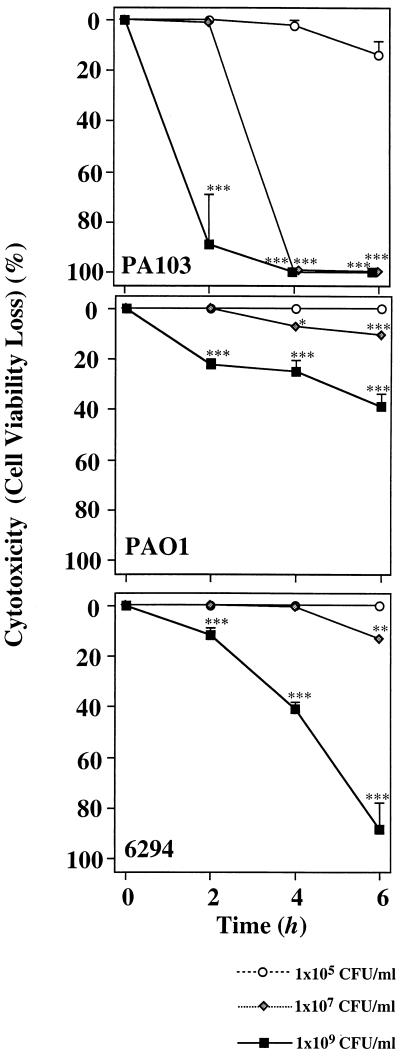
In previous studies we have correlated the ability to induce acute cytotoxicity in vitro with the expression of ExoU. Under these conditions, cytotoxicity was observed with relatively low bacterial inocula and during a short cocultivation period (3 h). To determine if ExoU-independent cytotoxicity was detectable, we compared the cytotoxic responses of three different P. aeruginosa strains, PA103 (exoU+), PAO1 (exoU), and 6294 (exoU) to that of human bronchial cells immortalized by SV40 (BEAS-2B) (Fig. 1). A low concentration (105 CFU/ml) of PA103 did not show cytotoxicity. Moderate and high concentrations of PA103 (107 and 109 CFU/ml) showed significantly higher levels of cytotoxicity after 2 h and caused a 100% loss of viability within 4 h. A low concentration (105 CFU/ml) of PAO1 did not show cytotoxicity. A moderate concentration of PAO1 (107 CFU/ml) caused a 10% loss of viability over a 6-h period. A high concentration of PAO1 (109 CFU/ml) led to moderate cytotoxicity (30% loss of viability) after 6 h of coculture. Finally, a low concentration (105 CFU/ml) of 6294 did not lead to cytotoxicity. A moderate concentration (107 CFU/ml) of 6294 caused only a 10% loss of viability, and a high concentration (109 CFU/ml) of 6294 caused a 90% loss of viability over a 6-h incubation period. These results indicated that higher bacterial inocula and longer incubation periods may be required for strains not possessing exoU to induce cytotoxicity.
FIG. 1.
Cytotoxicities of P. aeruginosa strains to lung epithelial cells. Cytotoxicities of P. aeruginosa strains to lung bronchial cells were evaluated by an assay measuring LDH release from the cells. Human bronchial cells immortalized by SV40 (BEAS-2B) (2 × 104 cells) were cocultured with one of three different P. aeruginosa strains (PA103, PAO1, or 6294) at three different concentrations (109, 107, and 105 CFU/ml) for 6 h. LDH activities were measured at 2-h intervals. Values are the averages of three assays and are shown as means ± standard deviations (indicated by error bars). ∗∗∗, P < 0.001 compared to the values at time zero.
Lung injury after the instillation of the three different P. aeruginosa strains.
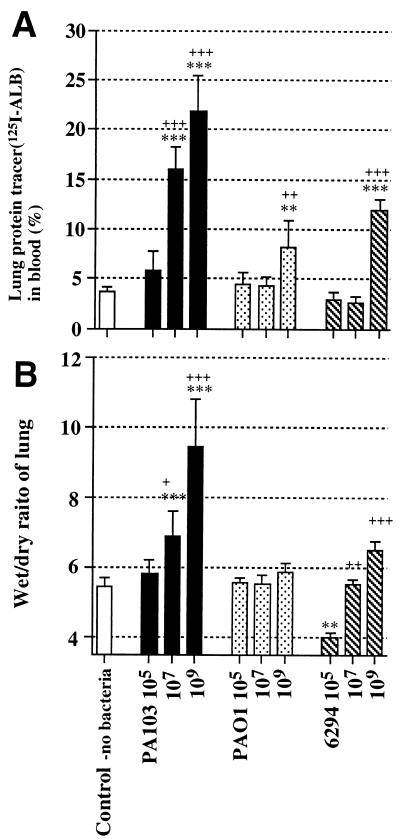
We examined the in vivo toxicities of the three P. aeruginosa strains (PA103, PAO1, and 6294) in mice by using an acute lung infection model. Three different doses of these strains were again utilized. Figure 2A shows the efflux of the airspace protein tracer into blood 4 h after the particular bacterial strain had been instilled. The efflux of the airspace protein tracer has been shown to be indicative of the quantity of lung epithelial injury (17, 32, 33). The instillation of moderate and high doses of PA103 led to significant quantities of tracer leakage from the airspaces of the instilled lungs into the circulation. The instillation of the small and moderate doses of PAO1 (105 and 107 CFU/ml) did not cause epithelial injury; only the instillation of the highest dose of PAO1 (109 CFU/ml) caused any appreciable efflux of the airspace protein tracer into the circulation. The instillation of the small and moderate doses of 6294 (105 and 107 CFU/ml) did not cause significant efflux of the airspace protein tracer into the circulation. However, the instillation of the highest dose of 6294 (109 CFU/ml) was associated with a significant efflux of the airspace protein tracer into the circulation. Therefore, only instillation of the largest dose (109 CFU/ml) of the two invasive strains led to lung epithelial injury, with 6294 causing more epithelial injury than PAO1.
FIG. 2.
Measurement of lung epithelial damage and edema. Inocula (50 μl each) were prepared with no bacteria, PA103, PAO1, or 6294 at three different concentrations (105, 107, and 109 CFU/ml). An inoculum was instilled into one lung with a protein tracer consisting of 125I-labeled human albumin. (A) Lung protein tracer leakage into blood. (B) Wet/dry ratios of lungs 4 h after bacterial instillation. Data are means ± standard deviations (indicated by error bars). Plus signs indicate statistical significances of intragroup comparisons relative to the group treated with the lowest dose of the same bacteria (+, P < 0.05; ++, P < 0.01; +++, P < 0.001). Asterisks indicate statistical significances of comparisons between the experimental groups and the control group (∗, P < 0.05; ∗∗, P < 0.01; ∗∗∗, P < 0.001). Each group consisted of three to five mice. One-way analysis of variance and the Bonferroni test were used to determine statistical significances.
The wet/dry ratios of lungs have been utilized as a measurement of lung edema. Figure 2B demonstrates the wet/dry ratios of the lungs 4 h after instillation of the three strains at the three different doses. There were no significant increases of the wet/dry ratios after the instillation of the lowest dose (105 CFU/ml) of any of the three strains; rather, the instillation of the low dose of 6294 decreased the ratio significantly. The instillation of the moderate and high doses (107 and 109 CFU/ml) of PA103 caused significant increases in the wet/dry ratios. In contrast, the instillation of the highest dose (109 CFU/ml) of PAO1 did not cause a significant increase in the wet/dry ratio. The instillation of the highest dose (109 CFU/ml) of 6294 did lead to a significant increase in the wet/dry ratios of the lungs compared with the lowest dose of 6294, but the increase was not as large as that seen after the instillation of the highest dose of PA103.
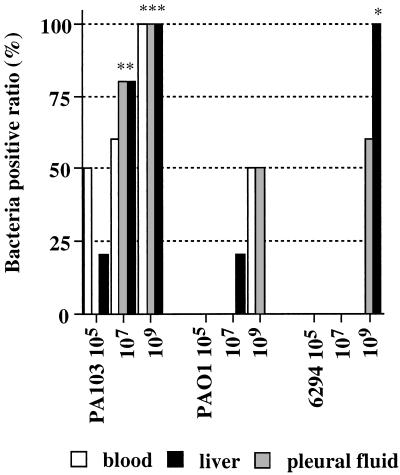
Bacterial dissemination after the instillation of the three different P. aeruginosa strains.
Bacterial cultures of blood, pleural fluids, and livers are shown in Fig. 3. The instillation of all doses of PA103 led to dissemination of bacteria into the blood and livers of the infected animals. Pleural fluids were shown to be infected when the two highest doses of PA103 were used. In contrast, only the instillation of the highest dose of PAO1 caused significant dissemination to blood, the liver, or the pleural space. Only the instillation of the highest dose of 6294 led to dissemination, but the percentages of positive blood and liver cultures were larger than those seen after the instillation of the highest dose of PAO1.
FIG. 3.
Bacterial dissemination 4 h after the instillation of P. aeruginosa. Inocula (50 μl each) containing no bacteria, PA103, PAO1, or 6294 were instilled in lungs at three different concentrations (105, 107, and 109 CFU/ml). Disseminations of bacteria into the blood, pleural fluid, and liver 4 h after instillation were compared among the three different P. aeruginosa strains. Data are ratios of quantitative bacterial cultures from three to five mice. Fisher’s exact test was used for comparison with the control group (data not shown; no positive blood, pleural fluid, or liver cultures for five mice). Asterisks indicate P values as described in the legend to Fig. 2.
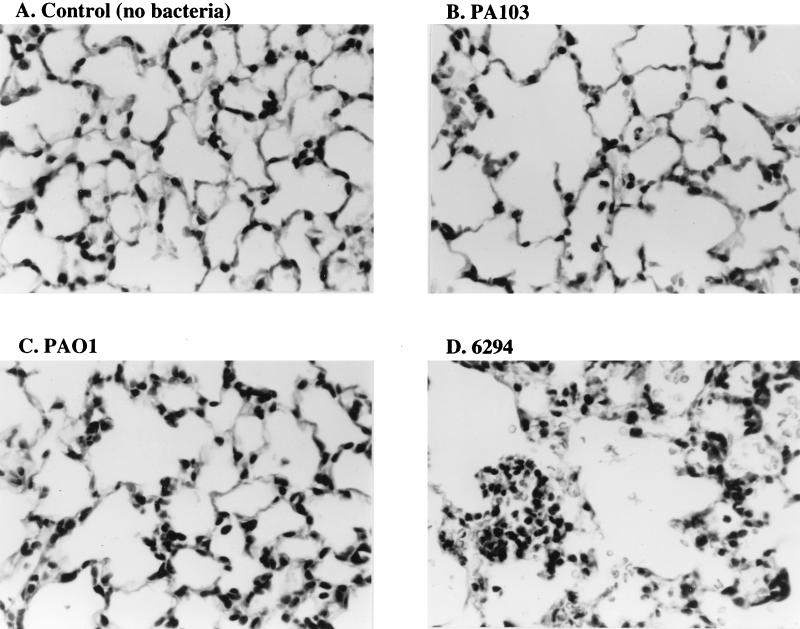
Histology of the bacterium-instilled lungs.
To obtain information on the type of tissue damage caused by each bacterial infection, histologic sections were obtained 4 h after the instillation of the three bacterial strains at three different doses. Figure 4 shows the results from the control mice that received the instillate without bacteria (Fig. 4A) and from the mice that received the highest dose (109 CFU/ml) of the three different P. aeruginosa strains (Fig. 4B, C, and D). Destruction of alveolar structures was observed in the sections obtained from the mice instilled with the highest dose of PA103. Fewer neutrophils were seen in sections from the lungs of animals which received the highest dose of PA103 compared to the number seen in sections from animals which had received PAO1 or 6294. In contrast, the lung destruction was least severe in the mice instilled with PAO1. The sections obtained from the mice instilled with the highest dose of 6294 showed large amounts of neutrophil infiltration into the airspaces and more lung destruction than that seen after the same dose of PAO1.
FIG. 4.
Lung histology 4 h after the instillation of three different P. aeruginosa strains at the highest dose (109 CFU/ml). An inoculum (50 μl) consisting of no bacteria (A), PA103 (B), PAO1 (C), or 6294 (D) was instilled into one lung. Four hours after the instillation, the lungs were perfused with a 10% buffered formalin solution for fixation and paraffin sections were made for hematoxylin-eosin staining.
Mortality in mice instilled with the three different P. aeruginosa strains.
Survival studies were performed for mice instilled with the three strains, PA103, PAO1 and 6294, at the three different doses (Table 1). Mice infected with PA103 had the highest mortality rate; all the mice died after the instillation of the moderate or the high dose. In contrast, none of the mice died after the instillation of the moderate dose of 6294 or PAO1. However, mice that received the highest dose of 6294 died sooner than did mice that received the highest dose of PAO1.
TABLE 1.
Survival of mice after the instillation of P. aeruginosa strains
| Strain | Concn of bacteria (CFU/ml) | No. of mice surviving at indicated times postinstillation (h)a
|
|||
|---|---|---|---|---|---|
| 0 | <24 | <48 | <72 | ||
| Control | No bacteria | 10 | 10 | 10 | 10 |
| PA103 | 105 | 10 | 10 | 10 | 10 |
| 107 | 10 | 1 | 0* | ||
| 109 | 10 | 0a | |||
| PAO1 | 105 | 10 | 10 | 10 | 10 |
| 107 | 10 | 10 | 10 | 10 | |
| 109 | 10 | 8 | 2 | 0* | |
| 6294 | 105 | 10 | 10 | 10 | 10 |
| 107 | 10 | 10 | 10 | 10 | |
| 109 | 10 | 0* | |||
A total of 10 mice was used for each experimental group. Each mouse received a 50-μl inoculum. The Mantel-Cox log rank test was used to analyze the statistical significance of differences between the control and experimental groups. *, P < 0.05 compared with the control group.
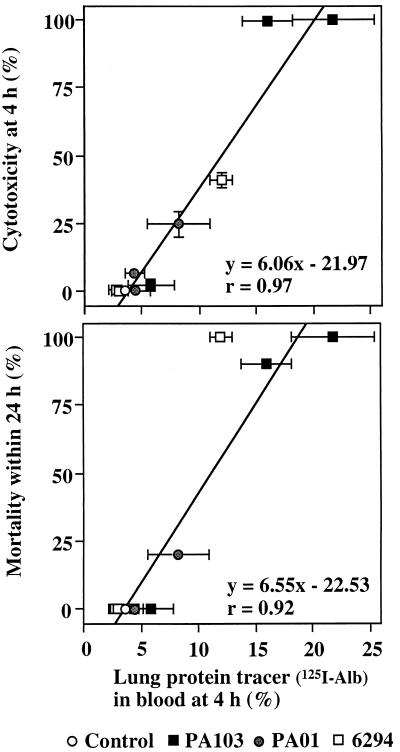
Correlation between lung epithelial injury and both cytotoxicity and mortality due to infection with P. aeruginosa.
Figure 5A shows the correlation between cytotoxicity (Fig. 1) and lung epithelial injury (as measured by protein tracer leakage) (Fig. 2). Conditions which led to high cytotoxicity in vitro also showed large quantities of lung protein tracer leakage in vivo. We concluded that cytotoxicity in vitro correlated with the production of severe lung epithelial injury. Figure 5B shows the correlation between mortality of the mice within 24 h (Table 1) and lung epithelial injury as measured by the protein tracer leakage (Fig. 2). These data showed a positive correlation between mortality and epithelial injury. Thus, cytotoxicity in vitro appears to be a good predictor of the capability of bacterial strains to injure the epithelium in vivo, which in turn correlates with an increased probability of a fatal infection.
FIG. 5.
Correlation between lung epithelial injury and both cytotoxicity and mortality. (A) Correlation between lung protein tracer leakage (data from Fig. 2A) and cytotoxicity (data from Fig. 1) was calculated. (B) Correlation between lung protein tracer leakage (data from Fig. 2A) and mortality of mice within 24 h (data from Table 1) was calculated. Data are means ± standard deviations (indicated by error bars). The linear regressions were calculated with the regression coefficients.
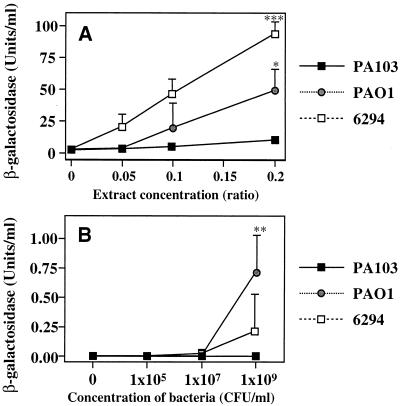
Production of the autoinducer PAI-1.
We reasoned that density-induced cytotoxicity may result from the expression of virulence determinants coordinately regulated by the P. aeruginosa quorum-sensing systems. To examine this hypothesis, the different levels of production of the autoinducer PAI-1 from the overnight cultures of the three different P. aeruginosa strains were compared utilizing the lasR lasB::lacZ plasmids in E. coli (Fig. 6A). β-Galactosidase activities induced by the autoinducer were the highest in culture medium from 6294; PAO1 cultures expressed less autoinducer as measured by β-galactosidase activity, and culture medium from the cytotoxic strain PA103 did not induce any β-galactosidase activity. In contrast to the results obtained when bacterial culture medium was assayed, cocultivation of the three different P. aeruginosa strains with human bronchial cells showed a different pattern. After 4 h, the cell culture medium with PAO1 expressed the highest level of autoinducer activity (Fig. 6B). Cocultivation of PA103 with BEAS cells over the 4-h period did not result in the production of autoinducer activity.
FIG. 6.
Production of the autoinducer PAI-1 in culture. (A) The bioactivity of PAI-1 in the culture medium from each P. aeruginosa strain was measured as the ability to activate LasR in a β-galactosidase reporter construct with the lasB promoter. Supernatants from bacterial cultures of PA103, PAO1, or 6294 were added in the relative amounts shown on the horizontal axis. (B) Bioactivities of PAI-1 in the culture medium of human bronchial cells exposed to three different P. aeruginosa strains at three different concentrations for 4 h. Data are means ± standard deviations (indicated by error bars). ∗, P < 0.05; ∗∗, P < 0.01; ∗∗∗, P < 0.001 (relative to the negative-control group [no extract for experiment in panel A, no bacteria for experiment in panel B]). One-way analysis of variance and the Bonferroni test were used to determine statistical significances.
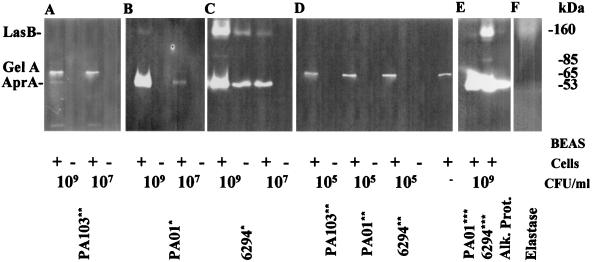
Proteinase production.
PA103 at a concentration of 109 CFU/ml in the presence of BEAS cells synthesizes alkaline protease (AprA) but not elastase (LasB) (Fig. 7A). In addition, gelatinase A (MMP-2, 72 kDa), a proteinase constitutively synthesized by BEAS cells (Fig. 7D) was detected. PAO1 required the presence of the BEAS cells to synthesize alkaline protease and elastase. Elastase was detected only when 109 CFU of PAO1/ml was added (Fig. 7B). At both 109 and 107 CFU/ml, 6294 synthesized more alkaline protease and elastase in the presence of the BEAS cells than in medium alone (Fig. 7C). Both proteases were detected when 109 CFU of 6294/ml was incubated in medium alone but not when 107 CFU/ml was used alone. An unknown protease with a molecular mass of 85 kDa was detected in the medium containing 109 CFU of 6294/ml plus BEAS cells.
FIG. 7.
Proteinase production by BEAS cells and P. aeruginosa. Proteinase activities present in culture medium containing of 109, 107, or 105 CFU of PA103/ml (A and D), PAO1 (B, D, and E), and 6294 (C, D, and E) with (+) and without (−) BEAS cells were visualized by gelatin zymography. Samples containing 1.5 μl (∗), 15 μl (∗∗), and 4 μl (∗∗∗) of medium were electrophoresed on SDS–8% polyacrylamide gels containing 0.2% gelatin. Following electrophoresis, the gels were incubated overnight in cleavage buffer, and the cleaved bands were visualized by negative staining with Coomassie brilliant blue. LasB, P. aeruginosa-encoded elastase; AprA, P. aeruginosa-encoded alkaline protease; GelA, gelatinase A (MMP2 encoded by BEAS cells). (F) Elastase standard.
In medium taken from the BEAS cells exposed to bacteria, the largest amount of 6294 (109 CFU/ml) produced about 25-fold more elastase than was produced in the medium from these cells exposed to the same concentration of PAO1 organisms. The enhancement of elastase production was measured in Western blots (498 ± 65 pg/109 CFU of 6294 versus 21 ± 7 pg/109 CFU of PAO1) and by zymographic analysis (Fig. 7B, C, and E). At the lowest concentration, 105 CFU/ml of PA103, PAO1, or 6294, the only protease detected was gelatinase A synthesized by the BEAS cells (Fig. 7D). This proteinase was present in all samples but is not visible in Fig. 7B or C because of the amount of sample that was loaded.
DISCUSSION
In pneumonia, the integrity of the epithelial barrier of the lung is critical for protection from dissemination of airspace bacteria. Experimentally, only a few agents have been found that cause severe lung epithelial injury (32). We reported that lung epithelial injury was caused by the airspace instillation of live P. aeruginosa (4 × 109 to 10 × 109 CFU of the clinical isolate Fisher immunotype I) in sheep but not by the airspace instillation of large doses of E. coli endotoxin (32, 33). The severities of lung epithelial injury caused by the various P. aeruginosa strains have been shown to be different (17); we have previously shown that the production of ExoU by PA103 and other cytotoxic strains was responsible for cytotoxicity and lung injury (10). The present experiments again verify that the cytotoxicity of PA103 is independent of proteinase production.
In the present study, we investigated the cytotoxicity seen after the application or instillation of two invasive strains, 6294 and PAO1, and compared the results to that seen after the application or instillation of the same dose of a cytotoxic strain, PA103. We replicated our previous results in that the instillation of low and moderate doses of these two invasive strains did not cause either significant cytotoxicity in vitro or lung injury in vivo (13). However, the instillation of large doses of the invasive strains, especially 6294, caused cytotoxicity and lung injury and the demise of the animals from dissemination of bacteria.
P. aeruginosa 6294 has been classified as an invasive strain due to the lack of exoU. This strain, however, appears to possess the LasR-LasI quorum-sensing modules, similar to strain PAO1. The production of PAI-1 in the overnight bacterial culture medium containing 6294 was greater than that in the culture medium containing PAO1. Notably, the application of these bacteria onto epithelial cells altered their production of the autoinducers; PAO1 produced more PAI-1 than 6294 did in the presence of the epithelial cells. Increases in LasB did not correlate with the production of PAI-1 by 6294. Therefore, the production of PAI-1 did not explain the increase in the production of elastase by strain 6294 compared to that by PAO1.
Recently, several quorum-sensing systems including the RhlR-RhlI system have been described; the amount of LasB produced by 6294 may be influenced by this second system (2, 19). On the other hand, the increased production of elastase by 6294 compared to that produced by PAO1 could be due to differences in other regulatory or sensory components in 6294. Although we previously demonstrated that elastase production was not associated with lung epithelial injury (17), the present results indicate that large doses of bacteria correlate with the enhanced production of virulence determinants regulated by quorum sensing (LasB and LasI) and are therefore markers of other virulence factors. We have also shown that the levels of LasB production correlate with increased cytotoxicity and lung epithelial injury associated with a clinical isolate. Taken together these observations may explain the wide variations in exoproduct production observed when different P. aeruginosa strains are used in different in vitro and in vivo model systems. Thus, exoproduct production is a function of the genetic makeup of the particular P. aeruginosa strain and changes in expression may be due to direct and indirect differences in gene expression and secretion.
Despite the complexity of factors involved in P. aeruginosa-induced lung pathology, we found that the quantitative experiments measuring cytotoxicity in vitro accurately predicted the in vivo lung injury and mortality results (Fig. 5). However, a large range of bacterial concentrations and incubation periods had to be utilized to obtain accurate correlations. Notably, cytotoxic factors and virulence in the lung were inversely correlated with autoinducer PAI-1 production in vitro, particularly when PAI-1 was measured in the supernatants of bacteria exposed to cultured cells (Fig. 6). PAI-1 also appeared to increase the recruitment of polymorphonuclear leukocytes to the infected airspaces (Fig. 4) and, therefore, increase the elimination of bacteria from the lung. PAI-1 may promote cytokine production (interleukin-8 or other chemoattractants, see below) that produced the leukocyte recruitment to the infected lungs and thereby may cause the difference in pathology noted between these three infections.
There are reports that P. aeruginosa exoproducts (20) and the P. aeruginosa autoinducer PAI-1 (6) stimulate interleukin-8 production in human respiratory epithelial cells. The histological results from the present study suggest that neutrophil infiltration was most significant in the lungs of the animals which had received the highest dose of 6294. Neutrophils might have contributed to the lung injury in these animals. In contrast, there were very few neutrophils in the sections obtained from the animals exposed to the highest dose of PA103; these results suggest that the exoproducts from PA103 are directly responsible for the lung epithelial damage and that either the resultant edema dilutes the neutrophil infiltration or PA103 inhibits neutrophil infiltration in some manner.
The lung injury assessments suggest that PA103 caused the most severe lung injury and the most dissemination compared to the comparable doses of PAO1 and 6294. The instillation of the highest dose of 6294 did lead to bacterial dissemination to the liver that was similar to that seen with the same dose of PA103. However, the instillation of the highest dose of PAO1 led to only 50% of the bacterial dissemination of the other strains. Although the instillation of the largest dose of any of these bacterial strains led to the demise of the mice within 3 days, a high dose of PA103 or 6294 led to much faster animal death than did such a dose of PAO1. As the lung edema and lung epithelial injury seen in the mice instilled with PAO1 were not severe even after exposure to the highest dose, these results suggest mortality is determined by the ability of the bacteria to damage the lung epithelium enough to disseminate.
In conclusion, although P. aeruginosa strains have been recently characterized as to their cytotoxicities or invasiveness based on the presence of a functional exoU allele, we found that the application or instillation of an invasive strain, especially 6294, led to cytotoxicity and severe lung epithelial injury depending on bacterial density. Although elastase does not cause cytotoxicity of the lung epithelium, its elaboration by the invasive 6294 strain was a marker for other virulence factors. Our results suggest that strains that produced the most PAI-1 were the least virulent in vitro as well as in vivo and that the production of PAI-1 may increase neutrophil recruitment to the infected lungs. LasI and autoinducer have been recently shown to be required for biofilm production (5). As the requirements for biofilm production are markedly different than the requirements for bacterial dissemination, it is reasonable that the LasI pathway may be inversely regulated relative to the pathways that regulate dissemination (i.e., ExsA). Although there are clearly multiple mechanisms for the induction of cytotoxicity and mortality in vivo, quantitative measurements of cytotoxicity in vitro were accurate predictors of cytotoxicity and mortality in vivo when a wide range of concentrations of bacteria were utilized.
ACKNOWLEDGMENTS
This work was supported by National Heart and Lung Institute award HL49810 (J.P.W.-K.), National Institute of Allergy and Infectious Disease awards AI31665 and AI01289 (D.W.F.) and EY06663 (S.S.T.).
We thank Richard Shanks for his technical assistance and E. P. Greenberg, Department of Microbiology, University of Iowa, Iowa City, for providing E. coli MG4 and for suggestions concerning the autoinducer assay.
REFERENCES
- 1.Brewer S C, Wunderink R G, Jones C B, Leeper K V., Jr Ventilator-associated pneumonia due to Pseudomonas aeruginosa. Chest. 1996;109:1019–1029. doi: 10.1378/chest.109.4.1019. [DOI] [PubMed] [Google Scholar]
- 2.Brint J M, Ohman D E. Synthesis of multiple exoproducts in Pseudomonas aeruginosa is under the control of RhlR-RhlI, another set of regulators in strain PAO1 with homology to the autoinducer-responsive LuxR-LuxI family. J Bacteriol. 1995;177:7155–7163. doi: 10.1128/jb.177.24.7155-7163.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Burnette W N. “Western blotting”: electrophoretic transfer of proteins from sodium dodecyl sulfate–polyacrylamide gels to unmodified nitrocellulose and radiographic detection with antibody and radioiodinated protein A. Anal Biochem. 1981;112:195–203. doi: 10.1016/0003-2697(81)90281-5. [DOI] [PubMed] [Google Scholar]
- 4.Craven D E, Steger K. Nosocomial pneumonia in mechanically ventilated adult patients: epidemiology and prevention in 1996. Semin Respir Infect. 1996;11:32–53. [PubMed] [Google Scholar]
- 5.Davies D G, Parsek M R, Pearson J P, Iglewski B H, Costerton J W, Greenberg E P. The involvement of cell-to-cell signals in the development of a bacterial biofilm. Science. 1998;280:295–298. doi: 10.1126/science.280.5361.295. [DOI] [PubMed] [Google Scholar]
- 6.Dimango E, Zar H J, Bryan R, Prince A. Diverse Pseudomonas aeruginosa gene products stimulate respiratory epithelial cells to produce interleukin-8. J Clin Invest. 1995;96:2204–2210. doi: 10.1172/JCI118275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Dunn M, Richard G, Wundderink G. Ventilator-associated pneumonia caused by Pseudomonas infection. Clin Chest Med. 1995;16:95–109. [PubMed] [Google Scholar]
- 8.Fabregas N, Torres A, El-Ebiary M, Ramfrez J, Hernandez C, Gonzalez J, de la Bellacasa J P, de Anta J, Rodriguez-Roisin R. Histopathologic and microbiologic aspects of ventilator-associated pneumonia. Anesthesiology. 1996;84:760–771. doi: 10.1097/00000542-199604000-00002. [DOI] [PubMed] [Google Scholar]
- 9.Fagon J Y, Chastre J, Hance A J, Montravers P, Novara A, Gilbert C. Nosocomial pneumonia in ventilated patients: a cohort study evaluating attributable mortality and hospital stay. Am J Med. 1993;94:281–288. doi: 10.1016/0002-9343(93)90060-3. [DOI] [PubMed] [Google Scholar]
- 10.Finck-Barbancon V, Goranson J, Zhu L, Sawa T, Wiener-Kronish J P, Fleiszig S M J, Wu C, Mende-Mueller L, Frank D W. ExoU expression by Pseudomonas aeruginosa correlates with acute cytotoxicity and epithelial injury. Mol Microbiol. 1997;25:547–557. doi: 10.1046/j.1365-2958.1997.4891851.x. [DOI] [PubMed] [Google Scholar]
- 11.Fleiszig S M J, Zaidi T S, Pier G B. Pseudomonas aeruginosa invasion of and multiplication within corneal epithelial cells in vitro. Infect Immun. 1995;63:4072–4077. doi: 10.1128/iai.63.10.4072-4077.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Fleiszig S M J, Zaidi T S, Preston M J, Grout M, Evans D J, Pier G B. Relationship between cytotoxicity and corneal epithelial cell invasion by clinical isolates of Pseudomonas aeruginosa. Infect Immun. 1996;64:2288–2294. doi: 10.1128/iai.64.6.2288-2294.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Fleiszig S M J, Wiener-Kronish J P, Miyazaki H, Vallas V, Mostov K E, Kanada D, Sawa T, Yen T S B, Frank D W. Cytotoxic and invasive strains of Pseudomonas aeruginosa are genotypically distinct at the loci encoding exoenzyme S. Infect Immun. 1997;65:579–586. doi: 10.1128/iai.65.2.579-586.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Fuqua C, Winans S C, Greenberg E P. Census and consensus in bacteria ecosystems: LuxR-LuxI family of quorum-sensing transcriptional regulators. Annu Rev Microbiol. 1996;50:727–751. doi: 10.1146/annurev.micro.50.1.727. [DOI] [PubMed] [Google Scholar]
- 15.Gambello M J, Iglewski B H. Cloning and characterization of Pseudomonas aeruginosa lasR gene. A transcriptional activator of elastase expression. J Bacteriol. 1991;173:3000–3009. doi: 10.1128/jb.173.9.3000-3009.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Gambello M J, Kaye S, Iglewski B H. LasR of Pseudomonas aeruginosa is a transcriptional activator of the alkaline protease gene (apr) and an enhancer of exotoxin A expression. Infect Immun. 1993;61:1180–1184. doi: 10.1128/iai.61.4.1180-1184.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kudoh I, Wiener-Kronish J P, Hashimoto S, Pittet J F, Frank D. Exoproduct secretions of Pseudomonas aeruginosa strains influence severity of alveolar epithelial injury. Am J Physiol. 1994;267:L551–L556. doi: 10.1152/ajplung.1994.267.5.L551. [DOI] [PubMed] [Google Scholar]
- 18.Laemmli U K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970;227:680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- 19.Latifi A, Winson M K, Foglino M, Bycroft B W, Stewart G S A B, Lazdunski A, Williams P. Multiple homologues of LuxR and LuxI control expression of virulence determinants and secondary metabolites through quorum sensing in Pseudomonas aeruginosa PAO1. Mol Microbiol. 1995;17:333–343. doi: 10.1111/j.1365-2958.1995.mmi_17020333.x. [DOI] [PubMed] [Google Scholar]
- 20.Massion P, Inoue H, Richman-Eisenstat J, Grunberger D, Jorens P G, Houssel B, Wiener-Kronish J P, Nadel J. Novel Pseudomonas product stimulates interleukin-8 production in airway epithelial cells in vitro. J Clin Invest. 1994;93:26–32. doi: 10.1172/JCI116954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Miller J A. Experiments in molecular genetics. Cold Spring Harbor, N.Y: Cold Spring Harbor Laboratory Press; 1976. pp. 352–355. [Google Scholar]
- 22.Parrillo J E, Parker M M, Nathanson C, Suffredini A F, Danner R L, Cunnion R E, Ognibene F P. Septic shock in humans. Advances in the understanding of pathogenesis, cardiovascular dysfunction and therapy. Ann Intern Med. 1990;113:227–237. doi: 10.7326/0003-4819-113-3-227. [DOI] [PubMed] [Google Scholar]
- 23.Passador L, Cook J M, Gambello M J, Rust L, Iglewski B H. Expression of Pseudomonas aeruginosa virulence genes requires cell-to-cell communication. Science. 1993;260:1127–1130. doi: 10.1126/science.8493556. [DOI] [PubMed] [Google Scholar]
- 24.Pearson J P, Gray K M, Passador L, Tucker K D, Eberhard A, Igelwski B H, Greenberg E P. Structure of the autoinducer required for expression of Pseudomonas aeruginosa virulence genes. Proc Natl Acad Sci USA. 1994;91:197–201. doi: 10.1073/pnas.91.1.197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Preston M J, Fleiszig S M J, Zaidi T S, Goldberg J B, Shortridge V D, Vasil M L, Pier G B. Rapid and sensitive method for evaluating Pseudomonas aeruginosa virulence factors during corneal infections in mice. Infect Immun. 1995;63:3497–3501. doi: 10.1128/iai.63.9.3497-3501.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Rouby J J. Nosocomial infection in the critically ill. The lung as a target organ. Anesthesiology. 1996;84:757–758. doi: 10.1097/00000542-199604000-00001. [DOI] [PubMed] [Google Scholar]
- 27.Rouby J J. Histology and microbiology of ventilator-associated pneumonias. Semin Respir Infect. 1996;11:54–61. [PubMed] [Google Scholar]
- 28.Tang H B, Dimango E, Bryan R, Gambello M J, Igelwski B H, Goldberg J B, Prince A. Contribution of specific Pseudomonas aeruginosa virulence factors to pathogenesis of pneumonia in a neonatal mouse model of infection. Infect Immun. 1996;64:37–43. doi: 10.1128/iai.64.1.37-43.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Taylor G D, Buchanan-Chell M, Kirkland T, McKenzie M, Wiens R. Bacteremic nosocomial pneumonia. A 7-year experience in one institution. Chest. 1994;108:786–788. doi: 10.1378/chest.108.3.786. [DOI] [PubMed] [Google Scholar]
- 30.Torres A, Aznar R, Gatell J M, Jimenez P, Gonzalez J, Ferrer A, Celis R, Rodriguez-Roisin R. Incidence, risk, and prognosis factors of nosocomial pneumonia in mechanically ventilated patients. Am Rev Respir Dis. 1990;142:523–528. doi: 10.1164/ajrccm/142.3.523. [DOI] [PubMed] [Google Scholar]
- 31.Twining S S, Zhou X, Schulte D P, Wilson P M, Fish B, Moulder J. Effect of vitamin A deficiency on the early response to experimental Pseudomonas keratitis. Investig Ophthalmol Vis Sci. 1996;37:511–522. [PubMed] [Google Scholar]
- 32.Wiener-Kronish J P, Albertine K H, Matthay M A. Differential responses of the endothelial and epithelial barriers of the lung in sheep to Escherichia coli endotoxin. J Clin Invest. 1991;88:864–875. doi: 10.1172/JCI115388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Wiener-Kronish J P, Sakuma T, Kudoh I, Pittet J F, Frank D, Dobbs L, Vasil M L, Matthay M. Alveolar epithelial injury and pleural empyema in acute P. aeruginosa pneumonia in anesthetized rabbits. J Appl Physiol. 1993;75:1661–1669. doi: 10.1152/jappl.1993.75.4.1661. [DOI] [PubMed] [Google Scholar]
- 34.Wunderink R G. Ventilator-associated pneumonia. Failure to respond to antibiotics therapy. Clin Chest Med. 1995;16:173–193. [PubMed] [Google Scholar]