Abstract
Cardiovascular disease (CVD) represents one of the main causes of mortality worldwide and nearly a half of it is related to ischemic heart disease (IHD). The article represents a comprehensive study on the diagnostics of IHD through the targeted metabolomic profiling and machine learning techniques. A total of 112 subjects were enrolled in the study, consisting of 76 IHD patients and 36 non-CVD subjects. Metabolomic profiling was conducted, involving the quantitative analysis of 87 endogenous metabolites in plasma. A novel regression method of age-adjustment correction of metabolomics data was developed. We identified 36 significantly changed metabolites which included increased cystathionine and dimethylglycine and the decreased ADMA and arginine. Tryptophan catabolism pathways showed significant alterations with increased levels of serotonin, intermediates of the kynurenine pathway and decreased intermediates of indole pathway. Amino acid profiles indicated elevated branched-chain amino acids and increased amino acid ratios. Short-chain acylcarnitines were reduced, while long-chain acylcarnitines were elevated. Based on these metabolites data, machine learning algorithms: logistic regression, support vector machine, decision trees, random forest, and gradient boosting, were used for IHD diagnostic models. Random forest demonstrated the highest accuracy with an AUC of 0.98. The metabolites Norepinephrine; Xanthurenic acid; Anthranilic acid; Serotonin; C6-DC; C14-OH; C16; C16-OH; GSG; Phenylalanine and Methionine were found to be significant and may serve as a novel preliminary panel for IHD diagnostics. Further studies are needed to confirm these findings.
Subject terms: Biochemistry, Metabolomics, Cardiovascular diseases
Introduction
Cardiovascular disease (CVD) represents one of the leading causes of mortality worldwide1. Nearly half of the global cases of CVD are related to ischemic heart disease (IHD)2. IHD is a complex disorder, presumably resulting from metabolic dysfunction affected by different environmental and genetic impacts3. However, in many cases, IHD may be asymptomatic and occasionally remaining invisible until the onset of acute or irreversible stages of the disease4–7. To date, there are a number of clinically significant metabolic risk factors for the development of IHD including hypertension, type 2 diabetes mellitus and dyslipidemia. However, these factors do not always have a high degree of specificity and may not indicate the presence of pathology in a timely manner. Therefore, it is necessary to create more accurate and reliable approaches to predict the IHD and identify its hidden forms. It may be performed through modern in-depth diagnostic methods and comprehensive assessment of patient’s condition, particularly in the light of personalized medicine.
Metabolomic profiling represents a powerful tool for comprehensive analysis of small molecules in the considered biological fluids that may provide new biochemical insights into the IHD progression and characterize novel differences in the signaling pathways8. Unlike other OMICs technologies, metabolomic profile reflects the physiological state of the body at the current moment, therefore underlining the phenotypic changes in the body9. However, due to the nonlinearity of metabolomic data, as well as high interindividual variability, analysis of the results of metabolomic profiling requires the use of progressive bioinformatics methods of analysis.
In recent years, artificial intelligence approaches, in particular machine learning (ML) methods, have attracted special attention in metabolomics10–12. ML methods are mathematical functions applied in the optimization process using input and output data. In other words, the ML model makes a prediction based on associations between the values of its constituent features. The use of supervised ML classification methods makes it possible to build predictive models based on the training data set, which allows further stratifying patients with respect to the considered diseases. For today, there is a number of works focused on the development of ML-based diagnostic models. These models may be used in future for reliable patient stratification and timely diagnosis of IHD.
Thus, the aim of the study was to identify the key metabolites and metabolic pathways of IHD and to create on its basis the pilot ML-model for IHD diagnostics and pathogenesis.
Materials and methods
Study design
Inclusion and exclusion criteria of the study are presented in the Table 1.
Table 1.
Inclusion and exclusion criteria.
| Inclusion criteria |
Men and women aged 18 years and older Angina pectoris functional class III according to Canadian Cardiovascular Society classification Availability of signed and dated informed consent of the patient to participate in the study |
| Exclusion criteria |
Angina pectoris functional class I, II or IV Type 1 Diabetes mellitus Acetaminophen, all vitamins, minerals, amino acids, dietary supplements, including sports drinks and energy drinks, creatinine, alpha-ketoglutarate, malic acid, citric acid, maleic acid, orotic acid consumption during 4 days before blood sampling. Sweeteners (aspartame, among others), monosodium glutamate and alcohol intake 24 h before blood sampling Any other diseases or conditions that, in the opinion of the investigator, may distort the results of the study and limit the patient’s participation in the study |
In total 137 patients with IHD were screened, of whom 84 patients with IHD were initially enrolled in the study. 53 patients had exclusion criteria, the most common of them were angina pectoris functional class I (n = 12), II (n = 25) or IV (n = 11), Type 1 DM (n = 5).
62 non-CVD subjects were screened, of whom 43 subjects were enrolled in the study, other 19 subjects had IHD.
5 patients from IHD group and 4 subjects from non-CVD group were excluded from the study due to diet violation (energy drinks consumption), 3 patients from IHD group and 3 subjects from non-CVD group were excluded due to alcohol consumption.
Patients diagnosed with IHD had angina pectoris functional class III according to Canadian Cardiovascular Society classification and a combined dyslipidemia characterized by elevated triglycerides and decreased HDL cholesterol13. IHD patients used organic nitrates, β-blockers, calcium channel blockers, ACE inhibitors, ARBs and statins.
The non-CVD group consisted of adults without any clinical or laboratory signs of cardio-vascular pathology and the risk factors of IHD.
Information on demographics, medical history, biochemical analysis and patient’s treatment was provided from the hospital database.
Ethical considerations
All conducted experiments were approved by the Ethics Committee of Belgorod Regional Clinical Hospital of St. Joseph, Belgorod, Russia (protocol #10 from 16 of November, 2015) in conformity with the ethical principles for medical research involving humans stated in the Declaration of Helsinki. Written informed consent was signed by all the participants before the beginning of the study.
Anthropometric evaluation
The anthropometric evaluation included measurements of weight, height and body mass index (BMI).
Biochemical analysis
Whole blood samples were collected into ethylenediaminetetraacetic acid (EDTA) tubes, immediately centrifuged (2000 rpm, 4 °C) during 20 min to receive plasma and stored at − 80 °C. Following biochemical evaluation of the samples included measurements of total cholesterol, triglycerides, high density lipoproteins (HDL), alanine aminotransferase (ALT), aspartate aminotransferase (AST), creatinine, glucose, fibrinogen, international normalised ratio (INR), activated partial thromboplastin time (APTT). Extra plasma aliquots were utilized for the metabolic analysis in the Laboratory of pharmacokinetics and metabolome analysis.
Chemicals and reagents
Standard solutions for metabolomic profiling, methanol, formic acid, bovine serum albumin (BSA) were received from Sigma-Aldrich (USA). Acetonitrile was purchased from Chromasolv® (Sigma-Aldrich Chemie GmbH, Buchs, Switzerland). Ultrapure water was received through the Millipore Milli-Q purification system (Millipore Corporation, Billerica, MA). Isotopically-labeled standard solutions for metabolic profiling Amino Acids and Acylcarnitines were received from MassChrom Non Derivatized 57000 Kit (Chromsystems, Germany), whereas isotope-labeled standard solutions for tryptophan catabolites profiling were from Toronto Research Chemicals (USA).
Metabolomic profiling
Targeted metabolomic profiling of the samples was performed in accordance to the method presented previously14 and included quantitative analysis of 87 endogenous metabolites in the patient’s plasma. Briefly, sample preparation of amino acids, intermediates of Arginine and Methionine metabolism consisted of protein precipitation with following instrumental analysis on Waters TQ-S-micro triple quadrupole mass spectrometer (Waters Corp, Milford, CT, USA). Preparation of samples for acylcarnitine and tryptophan catabolite profiling consisted of liquid–liquid extraction followed by LC–MS/MS analysis. The applied methods were validated in accordance with the guidelines for bioanalytical method validation and included assessment of selectivity, linearity, precision and accuracy, recovery, matrix effect, and stability of the methods.
Statistical analysis
To exclude the influence of age on the results of the metabolomic profiling we performed its correction using the regression analysis modeling (Python)15. The algorithm of the adjustment was following:
Select the group of non-CVD subjects and divide it into 5-year stratum.
Calculate median values of each metabolite in each stratum.
Based on the selected median values in separated stratums build linear regression model and calculate regression coefficients.
According to the received regression results calculate delta in concentration changes associated with age.
Extract the calculated delta from each absolute concentration.
All further statistical analyses for characterization of biochemical and metabolic profiling measurements were performed using the Python Stats package. Variable distribution was assessed using the Shapiro–Wilk test. According to the variable distribution, the analysis of variance was performed using parametric student t-test and ANOVA test or using non-parametric Mann–Whitney U test. The p-value less than 0.05 was considered as significant.
Development of the diagnostic model using machine learning algorithms
Further, to elucidate the best diagnostic model of IHD we applied and trained five machine learning algorithms, including: logistic regression (LR)16, support vector machine (SVM)17, decision trees (DT)18, random forest (RF)19 and gradient boosting (GB)20. LR and SVM with linear kernel relate to the class of linear classifiers that serves for categorizing a set of data point into a discrete class according to the linear combination of its explanatory variables. At the same time, DT, RF and GB are related to the non-linear class of algorithms. In DT classification procedure starts at the tree’s root node, where it assesses the attribute specified by this node, then moving down the tree branch corresponding to the attribute's value, as shown in the above figure. This procedure is repeated for each subset in a recursive partitioning manner. The RF and GB models are ensemble ML methods based on decision trees algorithms. In RF the predictions are performed by calculation the average of multiple trees’ output. As the number of trees increases, so does the precision of the output21. Contrary, GB algorithm represents an additive model which determines the impact of a poor learner by means of the gradient descent optimization. Thus, in this case the impact of each tree is assessed through the decrease of the overall error of the strong learner22.
Assessment of ML algorithms performance was performed using quality assessment metrics. For this purpose, we calculated parameters of confusion matrix, including true positive (TP), false positive (FP), true negative (TN), and false negative (FN) for actual and predicted data, based on which we further evaluated following metrics: area under the curve (AUC), accuracy, f1-score and recall.
Results
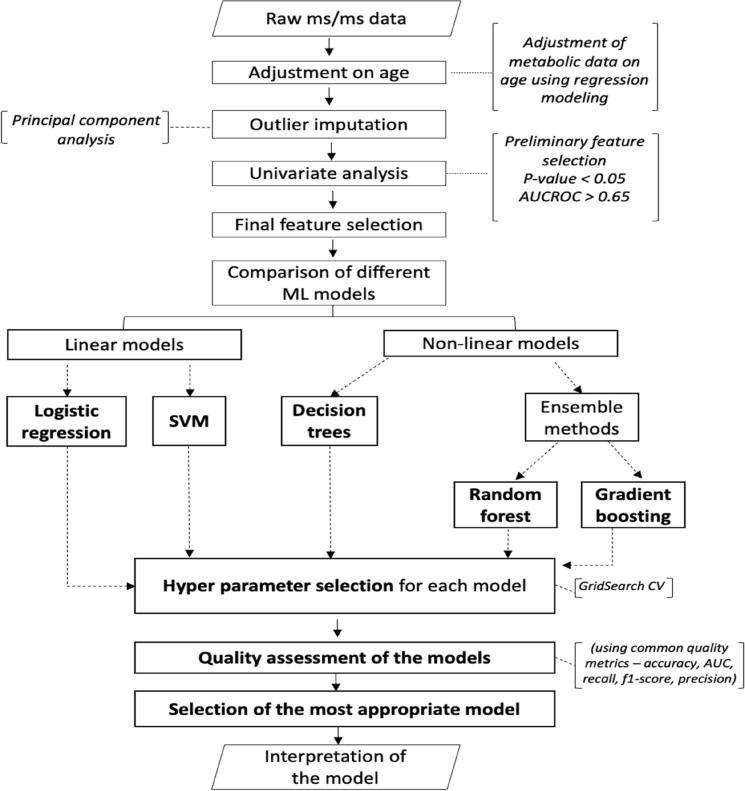
The presented study was conducted in accordance with the flowchart presented in Fig. 1.
Figure 1.
Flowchart on the processing of the metabolic profiles of non-CVD patients and patients with IHD.
Baseline characteristics of the IHD patients and non-CVD subjects
Among the considered subjects, the IHD patients were older than subjects from the non-CVD group and were characterized by higher body weight and higher BMI values.
The lipid analysis showed that total cholesterol was in normal range, but high triglycerides and low HDL cholesterol were observed that are characteristics of combined dyslipidemia.
The measurements of ALT, AST and glucose were in normal range. The creatinine level was increased in IHD group vs the non-CVD nevertheless it was in normal range. The coagulogram showed the normal range of fibrinogen, INR and APTT in both groups.
More information concerning the characteristics of patients is represented in Table 2.
Table 2.
Baseline characteristics of the IHD patients and non-CVD subjects.
| Variable | Non-CVD group (n = 36) | IHD group (n = 76) | p-value |
|---|---|---|---|
| Gender, m/f (%) | 29/7 (80/20) | 65/11 (86/14) | < 0.04 |
| Age (years) | 34 [26–44] | 64 [58–71] | < 0.0001 |
| Height (m) | 1.67 [1.62–1.72] | 1.69 [1.64–1.75] | 0.21 |
| Weight (kg) | 69.0 [59.0–81.0] | 84.4 [70.0–89.0] | < 0.0001 |
| BMI (kg/m2) | 24.6 [20.7–27.4] | 29.5 [25.6–31.63] | < 0.0001 |
| Heart rate/min | 62.15 ± 10.83 | 65.81 ± 9.24 | 0.51 |
| Systolic BP (mm Hg) | 119.5 ± 5.6 | 120.3 ± 6.9 | 0.82 |
| Diastolic BP (mm Hg) | 75.9 ± 6.2 | 74.7 ± 5.9 | 0.78 |
| Total cholesterol (mmol/L) | 5.27 [4.8–5.9] | 5.55 [4.50–6.20] | 0.48 |
| HDL cholesterol (mmol/L) | 1.51 [1.22–1.78] | 1.08 [0.89–1.19] | < 0.001 |
| LDL cholesterol (mmol/L) | 3.3 [2.7–4.0] | 3.3 [2.9–4.1] | 0.61 |
| VLDL cholesterol (mmol/L) | 0.5 [0.3–0.7] | 1.0 [0.9–1.2] | 0.01 |
| Triglycerides (mmol/L) | 1.10 [0.76–1.28] | 1.73 [0.96–2.18] | < 0.01 |
| ALT (u/L) | 23.0 [20.0–24.0] | 29.0 [20.0–34.5] | 0.31 |
| AST (u/L) | 25.0 [23.0–27.0] | 36.0 [20.0–36.0] | 0.66 |
| Creatinine (µmol/L) | 89.1 [79.4–97.2] | 100.5 [85.5–111.6] | 0.01 |
| Glucose (mmol/L) | 4.88 [4.59–5.30] | 5.65 [4.70–5.88] | < 0.01 |
| Fibrinogen (g/L) | 2.25 [2.0–2.30]] | 3.28 [2.53–3.74] | < 0.05 |
| INR | 1.09 [1.05–1.14] | 1.23 [1.11–1.28] | < 0.05 |
| APTT (s) | 27.2 [25.5–28.6] | 31.5 [27.0–34.8] | 0.19 |
General and biochemical characteristics of the participants including n (%) or median and interquartile range [Q1; Q3] in the considered groups and corresponding p-values, characterizing statistically significant differences between groups.
Univariate analysis of the concentration levels of the metabolites
Due to the relationship between the metabolomic profiling and age, as well as the large difference in the age characteristics of the proposed groups of patients, we adjusted the results of the metabolomic profile using regression models. Conversion factors are presented in the Table S1. Further, identification of the metabolites that significantly altered among the considered groups of patients was performed using parametric and non-parametric comparison tests. Table 3 summarizes information on the significantly changed metabolites including class of the metabolite, direction of change and adjusted p-value, AUC score and Younden index.
Table 3.
Significant metabolites with the consequent direction of their concentration change between the non-CVD and IHD groups. Metabolites with p-value < 0.05 and AUC score > 0.65 were selected as meaningful.
| Metabolite | p-value | Direction | AUC score | Younden index |
|---|---|---|---|---|
| Acylcarnitine profiling | ||||
| Hydroxyhexadecanoylcarnitine | < 0.0001 | Increased | 0.84 | 1.69 |
| Hydroxyhexadecenoylcarnitine | < 0.05 | Increased | 0.64 | 1.68 |
| Hydroxytetradecanoylcarnitine | < 0.001 | Increased | 0.73 | 1.68 |
| Adipoylcarnitine | < 0.01 | Increased | 0.66 | 1.68 |
| Glutarylcarnitne | < 0.001 | Decreased | 0.70 | 1.71 |
| Propionylcarnitine | < 0.05 | Decreased | 0.63 | 1.78 |
| Carnitine | < 0.0001 | Decreased | 0.75 | 1.98 |
| Tryptophan metabolism pathway | ||||
| Tryptophan | < 0.05 | Decreased | 0.63 | 1.86 |
| Serotonin | < 0.0001 | Increased | 0.75 | 1.93 |
| Indole-3-propionic acid | < 0.001 | Decreased | 0.72 | 1.84 |
| Indole-3-butyric acid | < 0.01 | Decreased | 0.68 | 1.69 |
| Indole-3-carboxaldehyde | < 0.05 | Decreased | 0.62 | 1.70 |
| Kynurenic acid | < 0.01 | Increased | 0.67 | 1.69 |
| Anthranilic acid | < 0.001 | Increased | 0.73 | 1.68 |
| Xanthurenic acid | < 0.05 | Increased | 0.62 | 1.69 |
| NO/urea cycle | ||||
| Asymmetric dimethylarginine | < 0.01 | Decreased | 0.70 | 1.83 |
| Arginine | < 0.01 | Decreased | 0.68 | 1.91 |
| Citrulline | < 0.01 | Decreased | 0.67 | 1.92 |
| Amino acid profiling | ||||
| Isoleucine | < 0.01 | Increased | 0.68 | 1.95 |
| Threonine | < 0.0001 | Decreased | 0.82 | 1.99 |
| Histidine | < 0.0001 | Decreased | 0.80 | 1.98 |
| Phenylalanine | < 0.0001 | Increased | 0.79 | 1.99 |
| Proline | < 0.0001 | Decreased | 0.74 | 1.98 |
| Lysine | < 0.001 | Decreased | 0.72 | 1.96 |
| Glycine | < 0.01 | Decreased | 0.68 | 1.88 |
| Leucine | < 0.05 | Increased | 0.65 | 1.95 |
| Aspartic acid | < 0.05 | Decreased | 0.62 | 1.69 |
| Asparagine | < 0.01 | Decreased | 0.67 | 1.83 |
| 3-Aminoisobutyric acid | < 0.05 | Increased | 0.64 | 1.96 |
| Tyrosine | < 0.05 | Decreased | 0.62 | 1.82 |
| Cystathionine cycle | ||||
| Cystathionine | < 0.05 | Up | 0.64 | 1.73 |
| Methionine sulfoxide | < 0.01 | Decreased | 0.68 | 1.80 |
| Norepinephrine | < 0.0001 | Decreased | 0.76 | 1.69 |
| Dimethylglycine | < 0.01 | Increased | 0.66 | 1.93 |
| Significantly changed ratios | ||||
| Fischer ratio | < 0.05 | Increased | 0.65 | 1.92 |
| GSG ratio | < 0.05 | Increased | 0.63 | 1.92 |
The above given results showed that:
Cystathionine and dimethylglycine (DMG) were significantly increased in IHD patients. At the same time, NO/urea cycle intermediates (ADMA, arginine and citrulline), as well as methionine sulfoxide and norepinephrine were significantly decreased.
Intermediates of tryptophan catabolism including serotonin, anthranilic acid, kynurenic acid and xanthurenic acid were significantly increased, whereas tryptophan, indole-3-carboxaldehyde, indole-3-propionic acid and indole-3-butyric acid significantly decreased.
Amino acids phenylalanine, branched-chain amino acids (BCAA) (isoleucine, leucine) and 3-aminoisobutyric acid were significantly elevated in the IHD patients. At the same time, aspartic acid, asparagine, tyrosine, glycine, lysine, proline, histidine and threonine were significantly decreased. Moreover, Fisher ratio ((Val + Ile + Leu)/(Phe + Tyr)) and GSG ratio (Glu/(Ser + Gly)) were significantly elevated in the IHD patients.
Levels of short chain acylcarnitines, including C0, C3 and C5-DC were significantly decreased in IHD patients, whereas C6-DC and hydroxylated long-chain acylcarnitines (C14-OH, C16-1-OH and C16-OH) were significantly increased.
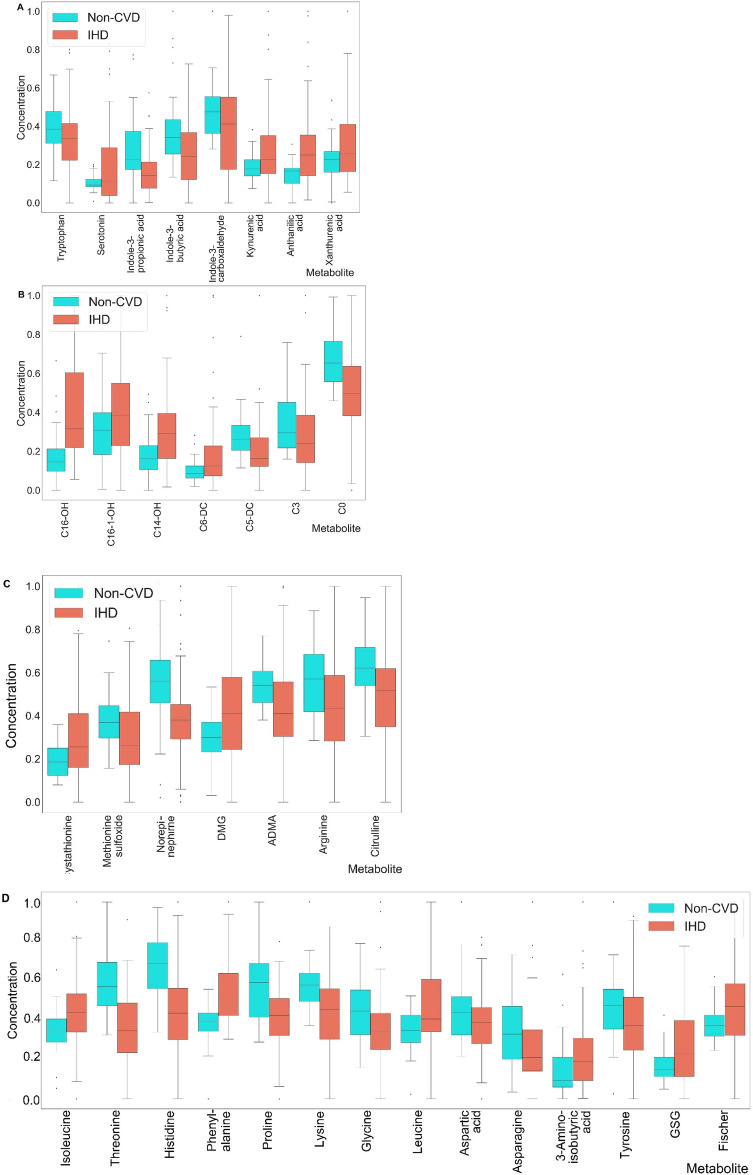
Graphical interpretation of results after min–max normalization are presented in Fig. 2A–D (A—Tryptophan metabolism intermediates; B—acylcarnitine profiling metabolites; C—cystathionine, betaine and arginine pathway intermediates; D—amino acid profiling metabolites).
Figure 2.
The box-plots of the significantly changed metabolites of: (A) tryptophan catabolism pathway; (B) acylcarnitine profiling; (C) cystathionine, betaine and arginine profiling; (D) amino acid profiling. The statistically significant metabolites were selected using parametric Student t-test (for normally distributed values) or equivalent non-parametric Mann–Whitney test (p < 0.05).
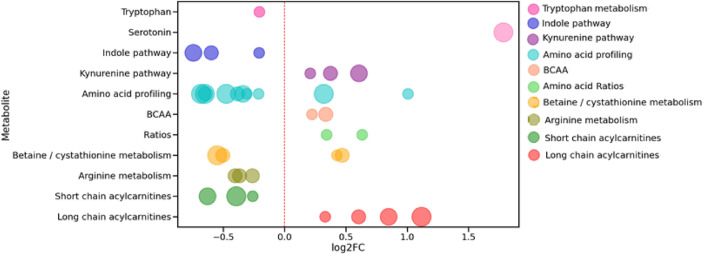
On the basis of association of significantly changed metabolites are known metabolic pathways a bubble plot was created (Fig. 3).
Figure 3.
Bubble plot of the significantly changed metabolic pathways for IHD patients compared to the non-CVD subjects. Each bubble represents the identified significantly changed metabolite, whereas its color indicates the involvement in the corresponding metabolic pathway, and its size corresponds to the magnitude of its p-value (the size of the bubble positively correlates with the p-value magnitude). The position of each bubble characterizes the value of log2FC between its concentration in IHD patients and subjects without CVD.
Development of the ML models based on the results of the metabolomic profiling
First of all, for a general overview of the received data and outlier exclusion, we performed a principal component analysis (PCA) (Supplementary Material Fig. S1). It has revealed that groups may be partly separated from each other.
Further, to identify the most appropriate prediction machine learning (ML) based model, we compared different supervised ML algorithms, including LR, SVM, DT, RF, and GB (Table 4). Each model was built based on the metabolic biomarker features using a cross-validated Python Gridsearch approach to identify of the best hyperparameters. The tuned hyperparameters of the ML models are presented in Supplementary Table S2. To determine the most precise diagnostic model of IHD there were applied common quality assessment metrics: sensitivity, specificity, AUC and confusion matrix together with the cross-validation method in splitting the working dataset. Figure 4 represents the AUC ROC of the developed ML models. The RF algorithm showed the best quality compared to the other used methods.
Table 4.
The list of the applied ML-algorithms and corresponded quality metrics (confusion matrix, sensitivity, specificity, accuracy, AUC ROC).
| ML algorithms | Confusion matrix (TP, FP, FN, TN) | Sensitivity | Specificity | Accuracy | AUC ROC | |
|---|---|---|---|---|---|---|
| Logistic regression | 28 | 8 | 0.82 | 0.89 | 0.87 | 0.94 |
| 6 | 66 | |||||
| SVM | 26 | 4 | 0.76 | 0.96 | 0.89 | 0.96 |
| 8 | 70 | |||||
| Decision trees | 24 | 10 | 0.71 | 0.86 | 0.82 | 0.86 |
| 10 | 64 | |||||
| Random forest | 26 | 0 | 0.76 | 1.00 | 0.93 | 0.98 |
| 8 | 74 | |||||
| Gradient boosting | 23 | 4 | 0.67 | 0.95 | 0.86 | 0.93 |
| 11 | 70 | |||||
Figure 4.
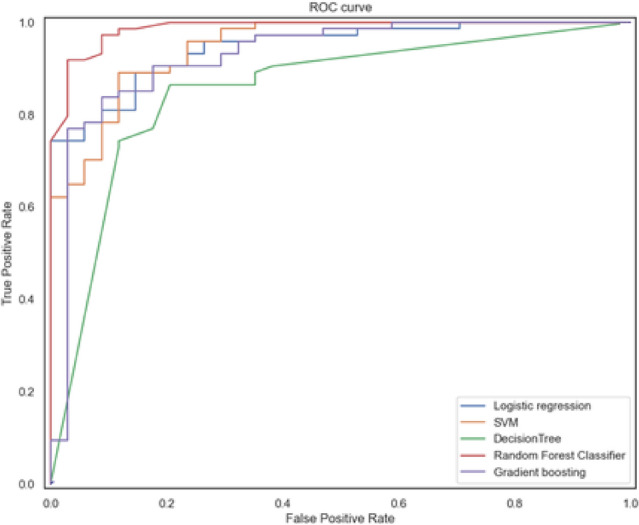
AUC ROC analysis of the applied machine learning methods. Random Forest model represents the best diagnostic accuracy.
Discussion
Univariate changes of the metabolomic profiling in IHD patients in comparison with non-CVD subjects
To the best of our knowledge it is the first complex investigation of the metabolomic profile and ML model for IHD, which comprises the main pathways of its diagnostics and pathogenesis.
In accordance with the received results, methionine metabolism was significantly affected during the CVD progression, showing increased levels of cystathionine. Elevated cystathionine plasma levels are related to endothelial dysfunction characterized by reduced nitric oxide-mediated vasodilation of arteries, therefore causing atherosclerotic lesions23. Besides this, cystathionine also affects glutathione production, which causes oxidative stress, thus inactivating nitric oxide production.
On the other hand, there was a significant decrease in the concentration of methionine sulfoxide—methionine derivative. Similarly to cystathionine, increased levels of methionine sulfoxide possess oxidative stress in the body24,25.
DMG and glycine were found to be significantly decreased in the IHD group. The glycine is known as a biomarker of cardiovascular dysregulation26, and its decreased level in IHD patients was expected, but a significant decrease in DMG level was firstly found and was unexpected.
Short-chain acylcarnitines (C0, C3, C5-DC) were significantly decreased in IHD group vs non-CVD group. Plasma concentrations of these acylcarnitines are known to reflect the gut microbiota and amino acid metabolism. C3 and C5 acylcarnitines are known as direct products of BCAA catabolism27, BCAA (leucine and isoleucine) were significantly increased in the IHD group. Hydroxylated long-chain acylcarnitines (C14-OH, C16-1-OH and C16-OH) were significantly increased in the IHD group. In general, long-chain acylcarnitines are known as markers of cardiovascular disorders28–30. Dysregulation in long chain acylcarnitines is usually associated with mitochondrial fatty acid oxidation disorders. However, little is known about the function of hydroxylated acylcarnitines in the IHD pathogenesis.
Arginine is the primary precursor for nitric oxide (NO) production in the vascular endothelium. Therefore, decreased arginine levels and its primary metabolite—ADMA—in IHD patients may indicate the lack of NO production. Additionally, there were also found significantly decreased levels of citrulline—endogenous metabolite, that is connected to arginine via the urea cycle being its end-product.
Intermediates of aspartate metabolism—aspartate and asparagine were significantly decreased in IHD patients. Asparagine is known as a glucogenic amino acid. Previously, asparagine was shown to be associated with high risks of cardiometabolic disease31. Along the aspartate metabolic pathway, asparagine is converted to aspartate and further through transamination to glutamate. Glutamate, glycine (also significantly decreased in IHD group) and cysteine represent the basis for the formation of tripeptide glutathione, which was also decreased. Glutathione is one of the major antioxidant in the body and its decreased level plays the main role in the atheroprogression in the smooth muscle and the endothelial cells32.
Amino acid ratios (Fisher and GSG) were increased in IHD group. The Fisher ratio represents the sum of BCAA divided by the sum of aromatic amino acids (Tyr, Phe). Its elevated levels were previously found in people with insulin resistance and pre-diabetes33. The GSG ratio contains amino acids involved in glutathione synthesis—the glutamine divided by the sum of serine and glycine.
Tryptophan catabolism consists of three main pathways: kynurenine, serotonin, and indole34. In the presented study, whereas tryptophan itself was significantly decreased in the IHD group, the kynurenine and serotonin pathways were significantly increased. The kynurenine pathway (KP) represents the major degradation route of tryptophan catabolism. Recently, plenty of studies indicated an association of the KP with the progression of CVD, which may be explained by its pathogenetic involvement in cardiovascular risk factors, including hypertension, diabetes mellitus, dyslipidemia, and obesity, as well as in vascular inflammation and atherosclerosis35. The presented study identified significant increased levels of three intermediates of KP—anthranilic acid, kynurenic acid, and xanthurenic acid.
Serotonin was significantly increased. Serotonin is a potent vasoconstrictor and enhances the hypertensive effects of several vasoconstrictors, such as angiotensin and endothelin36. In the previous studies, serotonin was found to be increased in patients with primary hypertension and certain types of secondary hypertension37,38.
In contrast to increased serotonin level, we found that intermediates of the indole tryptophan catabolic pathway, consisting of indole-3-propionic acid, indole-3-butyric acid, and indole-3-carboxaldehyde, were decreased in IHD patients. These metabolites are presumably generated through the gut microbiota's direct or indirect metabolism39,40.
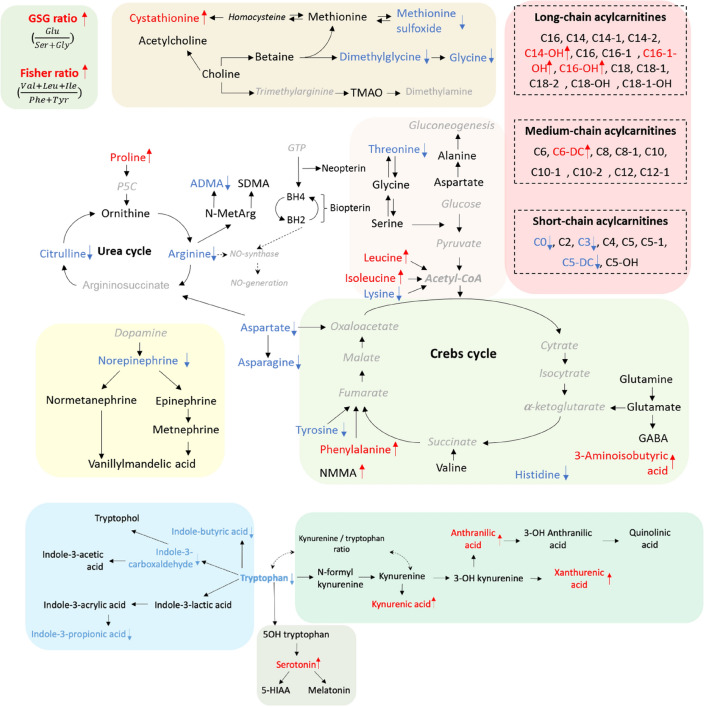
Figure 5 summarizes the scheme of the significantly altered metabolic pathways associated with IHD.
Figure 5.
Significantly changed metabolites and metabolic pathways in the IHD pathogenesis and diagnostics.
ML model
The introduction of machine learning methods to clinical diagnostics represents a promising healthcare approach. In the presented study, to find out the best model for IHD diagnostics, we compared five supervised ML algorithms, among which the best diagnostic accuracy was shown by the random forest model with an AUC value equal to 0.98.
However, it should be mentioned that all applied algorithms except for the decision trees model provided slightly the same prediction quality. In this regard, we analyzed and compared the utilized in each model set of metabolites to elucidate those whose concentration level provided the highest impact on the diagnostics of IHD patients (Table S3). Figure 6 represents features utilized in each ML method, having p-value < 0.05 and AUC score > 0.65.
Figure 6.
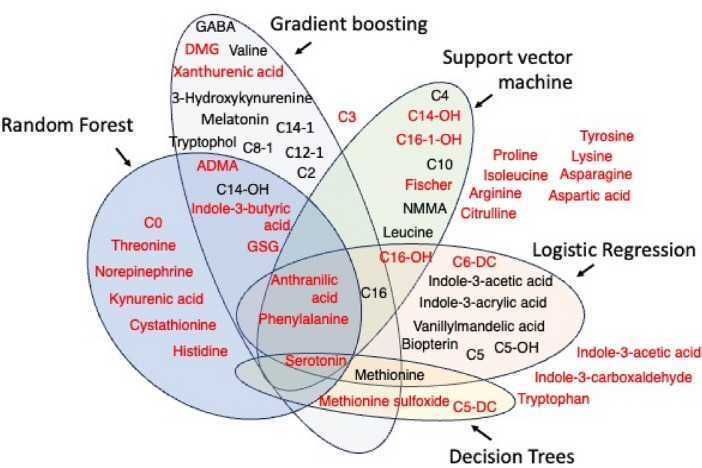
Plot of the features selected from the applied ML models. Metabolites marked in red had Mann–Whitney p-value < 0.05 and AUC < 0.65.
Based on this finding, we may conclude that metabolites Norepinephrine, Xanthurenic acid, Anthranilic acid, Serotonin, C6-DC, C14-OH, C16, C16-OH, GSG, Phenylalanine, and Methionine were found significant in most of the ML models. So, each of the ML model (RF; GB; SVH; LR) can be used separately as the preliminary diagnostic panel in patients with IHD. We hypothesize that these metabolites and ML model can be used for screening of IHD.
Advantages and limitations of the study
The main advantage of the study is that the presented approach provides new insights into the development of IHD from the metabolic point of view and the selected metabolic panel may be applied in the diagnostics of IHD in clinical practice.
Limitations of this study must be addressed. We acknowledge that a larger cohort studies are recommended which would confirm the presented findings. At the same time, we identified unexpected changes in concentration levels of several endogenous metabolites in IHD patients’ compared to non-CVD subjects, that were previously unknown or disagreed with already published data.
Conclusion
In conclusion, the presented study has successfully applied plasma metabolite-based ML modeling in screening IHD patients from non-CVD subjects, showing its efficacy in diagnostics of IHD with high levels of accuracy. Thus, even though this study was pilot, the presented results may facilitate future combination of ML-modeling and clinical metabolomics profiling for up-to-date diagnostics. Moreover, the suggested regression method for age-adjustment correction of metabolic data may be helpful in future metabolic studies with cohorts of non-balanced on-age participants. In addition, the identified, through the univariate analysis, significantly changed metabolites may also serve for the interpret of the molecular pathogenesis of IHD.
Supplementary Information
Author contributions
S.S.M.: Conceptualization, Writing—review & editing; E.A.P.: Writing—review & editing, Supervision; Y.A.R.: Data curation, Supervision; T.O.P.: Data curation, Supervision; S.V.I.: Writing—original draft; F.N.B.: Writing—review & editing; S.L.K.: Data collection, Patient supervision; A.A.N.: Data collection, Patient supervision; A.G.K.: Data collection, Patient supervision; A.I.G.: Data collection, Patient supervision; K.M.S.: Writing—original draft, Bioinformatics; P.A.M.: Laboratory analysis; N.E.M.: Laboratory analysis; M.V.K.: Data collection, Patient supervision; Z.Y.C.: Data collection, Patient supervision; S.A.A.: Writing—original draft.
Funding
This work was financed by the Ministry of Science and Higher Education of the Russian Federation within the framework of state support for the creation and development of World-Class Research Centers ‘Digital Biodesign and Personalized Healthcare’ (No 075-15-2022-305).
Data availability
All data generated and analysed during this study are included in this published article and its Supplementary Information files.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
The online version contains supplementary material available at 10.1038/s41598-024-53215-9.
References
- 1.World Health Organization. Cardiovascular Diseases. https://www.who.int/health-topics/cardiovascular-diseases#tab=tab_1 (2023).
- 2.Khan AB, et al. Global epidemiology of ischemic heart disease: Results from the global burden of disease study. Cureus. 2020;12(7):e9349. doi: 10.7759/cureus.9349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bertuzzi M, et al. Family history of ischemic heart disease and risk of acute myocardial infarction. Prev. Med. 2003;37(3):183–187. doi: 10.1016/S0091-7435(03)00094-X. [DOI] [PubMed] [Google Scholar]
- 4.Moran AE, et al. Assessing the global burden of ischemic heart disease: Part 1: Methods for a systematic review of the global epidemiology of ischemic heart disease in 1990 and 2010. Glob. Heart. 2012;7(4):315–329. doi: 10.1016/j.gheart.2012.10.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kones R, Fesc F, Rumana U. Stable ischemic heart disease. Heart Fail. Clin. 2016;12:11–29. doi: 10.1016/j.hfc.2015.08.003. [DOI] [PubMed] [Google Scholar]
- 6.McDonagh TA, et al. Symptomatic and asymptomatic left-ventricular systolic dysfunction in an urban population. The Lancet. 1997;350(9081):829–833. doi: 10.1016/S0140-6736(97)03033-X. [DOI] [PubMed] [Google Scholar]
- 7.Scheidt-Nave Ch, Barrett-Connor E, Wingard DL. Resting electrocardiographic abnormalities suggestive of asymptomatic ischemic heart disease associated with non-insulin-dependent diabetes mellitus in a defined population. Circulation. 1990;81(3):899–906. doi: 10.1161/01.CIR.81.3.899. [DOI] [PubMed] [Google Scholar]
- 8.Marchev AS, et al. Metabolomics and health: From nutritional crops and plant-based pharmaceuticals to profiling of human biofluids. Cell Mol. Life Sci. 2021;78(19–20):6487–6503. doi: 10.1007/s00018-021-03918-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Markin PA, et al. Plasma sarcosine measured by gas chromatography–mass spectrometry distinguishes prostatic intraepithelial neoplasia and prostate cancer from benign prostate hyperplasia. Lab. Med. 2020;51(6):566–573. doi: 10.1093/labmed/lmaa008. [DOI] [PubMed] [Google Scholar]
- 10.Galal A, Talal M, Moustafa A. Applications of machine learning in metabolomics: Disease modeling and classification. Front. Genet. 2022;13:1017340. doi: 10.3389/fgene.2022.1017340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Pomyen Y, Wanichthanarak K, Poungsombat P, Fahrmann J, Grapov D, Khoomrung S. Deep metabolome: Applications of deep learning in metabolomics. Comput. Struct. Biotechnol. J. 2020;18:2818–2825. doi: 10.1016/j.csbj.2020.09.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Shestakova KM, et al. Targeted metabolomic profiling as a tool for diagnostics of patients with non-small-cell lung cancer. Sci. Rep. 2023;13(1):11072. doi: 10.1038/s41598-023-38140-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Campeau L. The Canadian Cardiovascular Society grading of angina pectoris revisited 30 years later. Can. J. Cardiol. 2002;18:371–379. [PubMed] [Google Scholar]
- 14.Moskaleva NE, et al. Target metabolome profiling-based machine learning as a diagnostic approach for cardiovascular diseases in adults. Metabolites. 2022;12(12):1185. doi: 10.3390/metabo12121185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Van Rossum G, Drake FL. Python 3 Reference Manual. CreateSpace; 2009. [Google Scholar]
- 16.Nusinovici S, et al. Logistic regression was as good as machine learning for predicting major chronic diseases. J. Clin. Epidemiol. 2020;122:56–69. doi: 10.1016/j.jclinepi.2020.03.002. [DOI] [PubMed] [Google Scholar]
- 17.Durgesh KS, Lekha B. Data classification using support vector machine. J. Theor. Appl. Inform. Technol. 2010;12(1):1–7. [Google Scholar]
- 18.Charbuty B, Abdulazeez A. Classification based on decision tree algorithm for machine learning. J. Theor. Appl. Inform. Technol. 2021;2(1):20–28. [Google Scholar]
- 19.Qi Y. Random forest for bioinformatics. In: Zhang C, Ma Y, editors. Ensemble Machine Learning: Methods and Applications. Springer; 2012. pp. 307–323. [Google Scholar]
- 20.Xie Y, et al. Use of gradient boosting machine learning to predict patient outcome in acute ischemic stroke on the basis of imaging, demographic, and clinical information. Am. J. Roentgenol. 2019;212(1):44–51. doi: 10.2214/AJR.18.20260. [DOI] [PubMed] [Google Scholar]
- 21.Dalal S, et al. Application of machine learning for cardiovascular disease risk prediction. Comput. Intell. Neurosci. 2023;2023:1–12. doi: 10.1155/2023/9418666. [DOI] [Google Scholar]
- 22.Prasannavenkatesan Th. Predictive analysis of cardiovascular disease using gradient boosting based learning and recursive feature elimination technique. Intell. Syst. Appl. 2022;16:200121. [Google Scholar]
- 23.Singh RB, et al. Pathogenesis of atherosclerosis: A multifactorial process. Exp. Clin. Cardiol. 2002;7(1):40–53. [PMC free article] [PubMed] [Google Scholar]
- 24.Makukhin N, Tretyachenko V, Moskovitz J, Míšek J. A ratiometric fluorescent probe for imaging of the activity of methionine sulfoxide reductase A in cells. Angew. Chem. Int. Ed. Engl. 2016;55(41):12727–12730. doi: 10.1002/anie.201605833. [DOI] [PubMed] [Google Scholar]
- 25.Moskovitz J, Herbert W, Nathan B. Cloning the expression of a mammalian gene involved in the reduction of methionine sulfoxide residues in proteins. Proc. Natl. Acad. Sci. U.S.A. 1996;93(5):2095–2099. doi: 10.1073/pnas.93.5.2095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Wittemans LBL, et al. Assessing the causal association of glycine with risk of cardio-metabolic diseases. Nat. Commun. 2019;10(1):1060. doi: 10.1038/s41467-019-08936-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Newgard CB, et al. A branched-chain amino acid-related metabolic signature that differentiates obese and lean humans and contributes to insulin resistance. Cell Metab. 2009;9(4):311–326. doi: 10.1016/j.cmet.2009.02.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Aitken-Buck HM, et al. Long-chain acylcarnitines and cardiac excitation-contraction coupling: Links to arrhythmias. Front. Physiol. 2020;11:577856. doi: 10.3389/fphys.2020.577856. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Markin PA, et al. Short-and medium-term exposures of diazepam induce metabolomic alterations associated with the serotonergic, dopaminergic, adrenergic and aspartic acid neurotransmitter systems in zebrafish (Danio rerio) embryos/larvae. Comp. Biochem. Physiol. D Genom. Proteom. 2021;38:100816. doi: 10.1016/j.cbd.2021.100816. [DOI] [PubMed] [Google Scholar]
- 30.Kukharenko A, et al. Relationship between the plasma acylcarnitine profile and cardiometabolic risk factors in adults diagnosed with cardiovascular diseases. Clin. Chim. Acta. 2020;507:250–256. doi: 10.1016/j.cca.2020.04.035. [DOI] [PubMed] [Google Scholar]
- 31.Ottosson F, Smith E, Melander O, Fernandez C. Altered asparagine and glutamate homeostasis precede coronary artery disease and type 2 diabetes. J. Clin. Endocrinol. Metab. 2018;103(8):3060–3069. doi: 10.1210/jc.2018-00546. [DOI] [PubMed] [Google Scholar]
- 32.Rebholz CM, et al. Serum metabolomic profile of incident diabetes. Diabetologia. 2018;61:1046–1054. doi: 10.1007/s00125-018-4573-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Øivind M, et al. Combined measurement of 6 fat-soluble vitamins and 26 water-soluble functional vitamin markers and amino acids in 50 μL of serum or plasma by high-throughput mass spectrometry. Anal. Chem. 2016;88(21):10427–10436. doi: 10.1021/acs.analchem.6b02325. [DOI] [PubMed] [Google Scholar]
- 34.Roth W, et al. Tryptophan metabolism and gut-brain homeostasis. Int. J. Mol. Sci. 2021;22(6):2973. doi: 10.3390/ijms22062973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Gáspár R, et al. Kynurenine pathway metabolites as potential clinical biomarkers in coronary artery disease. Front. Immun. 2022;12:768560. doi: 10.3389/fimmu.2021.768560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Watts SW. 5-HT in systemic hypertension: Foe, friend or fantasy? Clin. Sci. 2005;108(5):399–412. doi: 10.1042/CS20040364. [DOI] [PubMed] [Google Scholar]
- 37.Hervé P, et al. Increased plasma serotonin in primary pulmonary hypertension. Am. J. Med. 1995;99(3):249–254. doi: 10.1016/S0002-9343(99)80156-9. [DOI] [PubMed] [Google Scholar]
- 38.Watts SW, Morrison SF, Davis RP, Barman SM. Serotonin and blood pressure regulation. Pharmacol. Rev. 2012;64(2):359–388. doi: 10.1124/pr.111.004697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Oluwagbemigun K, et al. Longitudinal relationship of amino acids and indole metabolites with long-term body mass index and cardiometabolic risk markers in young individuals. Sci. Rep. 2020;10(1):6399. doi: 10.1038/s41598-020-63313-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Buergel T, et al. Metabolic profiles predict individual multidisease outcomes. Nat. Med. 2022;28:2903–2920. doi: 10.1038/s41591-022-01980-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
All data generated and analysed during this study are included in this published article and its Supplementary Information files.