Abstract
Mice defective in the ability to produce the tumor necrosis factor alpha p55 receptor (TNFαp55R) were orally challenged with a number of Salmonella typhimurium HWSH derivatives that differ in virulence. In comparison to TNFαp55R+/+ mice, TNFαp55R−/− mice succumbed earlier to challenge with wild-type S. typhimurium HWSH and S. typhimurium HWSH purE. In contrast, TNFαp55R−/− mice were able to control an S. typhimurium HWSH aroA challenge, although greater numbers of Salmonella organisms were present in the tissues for a longer time period than was observed with TNFαp55R+/+ mice. Vaccination of normal and TNFαp55R knockout animals with S. typhimurium HWSH aroA showed that TNFαp55R−/− mice, unlike TNFαp55R+/+ mice, were not protected against a virulent S. typhimurium HWSH challenge. Splenocytes from TNFαp55R−/− mice exhibited a reduced ability to proliferate in the presence of S. typhimurium antigen compared to TNFαp55R+/+ mice. Thus, TNFαp55R is essential for controlling Salmonella growth in tissues and for recall of immunity in murine salmonellosis.
Salmonella enterica causes a wide variety of disease syndromes in humans, many of which are associated with significant levels of fever (15). Perhaps the most dramatic example of this is typhoid, or enteric fever, caused predominantly by Salmonella typhi (15, 18, 19). Typhoid is an invasive enteric infection in which viable bacteria can often be isolated from the blood of infected individuals. Since S. typhi does not cause significant disease in animals other than higher primates, murine typhoid, caused by certain S. enterica serotypes including Salmonella typhimurium, has been used extensively as a model for systemic salmonellosis (4). Following oral infection of naive mice with virulent S. typhimurium, bacteria quickly spread from the gut, probably through the Peyer’s patches of the gut-associated lymphoid tissue to organs of the reticuloendothelial system (RES) including the liver and spleen. With a combination of virulent bacteria and a susceptible mouse strain, the rate of bacterial growth is rapid and the mice die within several days, showing signs of endotoxic shock and harboring high levels of bacteria (up to 108) in liver and spleen (3–5, 7, 25, 35). However, the relative contribution of endotoxin to the mortality associated with systemic Salmonella infection remains to be fully defined. In sublethal infections or in immunized animals, bacterial growth is suppressed by the host response, leading to a plateau phase after the early rapid growth and to eventual clearance. The mechanisms controlling bacterial growth are also not fully defined, although T cells are not thought to play a major role early in infection and production of both tumor necrosis factor alpha (TNF-α) and gamma interferon (IFN-γ) is required (17, 23, 25, 26, 29, 38). The plateau phase is followed by T-cell-dependent clearance of bacteria from the RES. Both CD4+ and CD8+ T cells contribute to clearance, although CD4+ cells apparently play the predominant role (22, 23, 26, 34).
TNF-α plays a key role in the control of infections caused by a number of different pathogens (1, 10, 11, 36). This cytokine can act synergistically with IFN-γ to enhance the bactericidal activity of macrophages and is associated in mice with the induction of nitric oxide (10, 11, 36, 38). Although TNF-α is essential for the expression of immunity, it can also contribute significantly to pathology and mortality, mediating cachexia and endotoxin-associated septic shock. The balance between the beneficial aspects of immunity and pathology vary in different host-pathogen interactions. A number of studies have investigated the role of TNF-α in immunity to Salmonella infection by using TNF-α-neutralizing antibodies (22, 23). Mice treated with anti-TNF-α were more susceptible to infection and failed to display immunity when neutralizing antibody was administered simultaneously with challenge. Mice can express at least two independent receptors for TNF-α (28). Different functions have been assigned to the receptors, although some redundancy is present. One of the receptors, with an extracellular domain of 55 kDa (TNFαp55R), is implicated in the majority of known TNF-associated effects, including cytotoxicity and synergistic interaction with IFN-γ (9). Mice that lack TNFαp55R due to gene-targeted homozygous deletion have been developed (9). These mice show increased resistance to shock with lipopolysaccharide (LPS) and vary in susceptibility to infection with different pathogens (21). These mice provide an approach to defining the role of TNFαp55R in controlling salmonellosis in the mouse. In this paper we describe the use of TNFαp55R knockout mice and their abilities to control Salmonella infections of differing virulence, along with attempts to prevent infection by vaccination with live S. typhimurium HWSH aroA mutants.
MATERIALS AND METHODS
Bacterial strains and growth conditions.
Wild-type S. typhimurium HWSH was isolated from a calf dying of salmonellosis and is highly virulent in mice and calves. S. typhimurium HWSH purE and S. typhimurium HWSH aroA are auxotrophic mutants of the wild-type strain (6, 30, 31). S. typhimurium HWSH purE is partially attenuated in mice but is able to induce pathology, including abscess formation, in the internal organs of infected mice and to cause sporadic deaths. S. typhimurium HWSH aroA is a highly attenuated strain that is unable to kill wild-type mice by the oral route of infection. S. typhimurium HWSH aroA can enter the RES but is efficiently cleared by normal mice after several weeks and is an effective single-dose oral vaccine against homologous Salmonella challenge. Bacteria were routinely cultured on Luria agar or in Luria broth. Minimal medium was supplemented with nutrients or antibiotics at the appropriate concentrations. Solid media contained 1.6% Noble agar (wt/vol) (Difco, Surrey, United Kingdom).
Infection of mice and enumeration of bacteria in murine organs.
TNFαp55R knockout mice (−/−) along with normal (+/+) controls were bred at Imperial College. They were maintained on a mixed 129Sv × C57BL/6 genetic background and have been described previously (37). Mice were tested in-house by PCR for the presence of the receptor gene by using tail tissue. Bacteria were grown static in L broth, and bacterial numbers were determined by the optical density at 650 nm and then by surface viable counting. For oral inoculation, bacteria were administered in 0.2-ml volumes to lightly halothane-anesthetized mice by gavage as described previously (30). Livers and spleens were removed and homogenized as previously described (30). Viable counts were performed on these whole organ homogenates with L agar as the growth medium and are expressed in the figures as geometric means ± 2 standard errors of the means for four mice per time point.
Isolation of spleen cells.
Spleen cells from infected animals, three mice per time point, were isolated by teasing out the cellular contents with a sterile needle and washing them twice in Dulbecco’s modified Eagle’s medium (Sigma, Poole, Dorset, United Kingdom) with 10% heat-inactivated fetal calf serum, l-glutamine, and penicillin-streptomycin (Sigma). Erythrocytes were lysed with ammonium chloride-potassium lysis buffer, and the cells were again washed twice and counted in a hemocytometer to give a final suspension of 5 × 106 cells/ml for splenocyte proliferation assays. Cells not excluding trypan blue were not included in the final count. Experiments were repeated at least six times.
Cell proliferation assays.
Isolated spleen cells were plated in 96-well round-bottomed plates (Costar) in 100-μl volumes containing 5 × 106 cells/ml. The antigen used was detoxified Salmonella lysate (41) to give final concentrations of antigen of 1 to 10 μg/ml per well. Detoxified Salmonella lysate had been treated with sodium hydroxide in order to eliminate the mitogenic and cell toxicity effects of LPS of the gram-negative envelope. Thus, cell proliferation is a response to protein antigens of the bacteria and not to LPS. Ten microliters of the diluted antigen was applied to appropriate wells. The positive control concanavalin A was used at a final concentration of 5 μg/ml. The cells were incubated for 72 h at 37°C in 5% carbon dioxide. Supernatants for cytokine assays were removed to a 96-well plate, covered with parafilm, and frozen at −70°C. The cells were pulsed with 10 μl of a solution of 100 μCi of [3H]thymidine per ml in RPMI to give a final concentration of 1 μCi/well. The cells were incubated for a further 6 h and then harvested with a cell harvester (Tomtec). The filters were placed in scintillation fluid and counted in a beta-plate counter (Wallac, Milton Keynes, United Kingdom).
Antibody subclass enzyme-linked immunosorbent assay (ELISA) protocol.
Costar 96-well plates were coated overnight with formalin-killed and washed S. typhimurium at a concentration of 10 μg of protein per ml at 4°C. Wells were washed three times in phosphate-buffered saline (PBS)-Tween (PBST) and blocked with 200 μl of 1% bovine serum albumin (BSA). Mouse serum was diluted in PBS in the dilution range 1/50 to 1/50,000. Fifty microliters of diluted serum was added to all wells, and the plate was incubated for 2 h at 37°C. After this time the plates were washed in PBST and a rabbit anti-mouse horseradish peroxidase conjugate (Sigma) was added at a 1/1,000 dilution for each subclass, 50 μl/well for 2 h at 37°C. The plates were washed three times with PBST, and 50 μl of substrate was added. The color was developed at 37°C or at room temperature (rt). After the reaction was judged complete, it was stopped by the addition of 50 μl of 12.5% (3 M) sulfuric acid. The absorbance was read at 492 nm in a plate reader.
Cytokine ELISA protocol.
A purified anticytokine capture monoclonal antibody was diluted to 2 μg/ml in 0.1 M sodium hydrogen carbonate, pH 8.2. Fifty microliters of this was added to a 96-well EIA/RIA plate (Costar), and the plate was left at 4°C overnight. The plates were washed twice with PBST and blocked with 3% BSA in PBS at 200 μl/well. The plates were left at rt for 2 hours and again washed with PBST. Standards and samples were diluted in PBS–3% BSA and added at 100 μl/well, and the plates were left at rt for 4 h or overnight at 4°C. The plates were washed four times with PBST, and diluted biotinylated anticytokine antibodies in PBS–3% BSA were added at 100 μl/well. Plates were left at rt for 45 min and then washed six times in PBST. Avidin-peroxidase was diluted 1:400 in PBS–3% BSA and added at 100 μl to each well. Plates were left at rt for 30 min, and then were washed eight times with PBST. One hundred microliters of substrate and hydrogen peroxide was added per well, and the color reaction was allowed to develop at rt. The color reaction was stopped, and the plates were read at 490 nm. The antibodies used were rat anti-mouse interleukin 4 (IL-4) clone BVD6-24G2, IFN-γ clones XMG1.2 and R4-6A2, IL-10 clone SXC-1, IL-5 clone TRFK4, and TNF-α clones G281-2626 and MP6-XT3 (Pharmingen, San Diego, Calif.). The sensitivity of detection for the cytokine ELISA was 15 pg/ml.
Histological sections.
Five-micrometer-thick sections stained by hematoxylin and eosin were prepared from tissues fixed in 10% (vol/vol) formal saline and embedded in paraffin wax. Sections were also cut by cryostat from livers and spleens frozen at −70°C for immunohistochemistry.
Vaccination and challenge experiments.
Normal and TNFαp55R knockout mice were vaccinated orally with 109 cells of S. typhimurium HWSH aroA vaccine strain. Counts of the vaccine strain in internal organs were performed at days 7, 14, and 21 postvaccination. After 28 days, both groups of animals and an uninfected control group were orally challenged with 108 cells of the S. typhimurium HWSH wild-type strain.
Nitric oxide determination.
Both groups of knockout mice were challenged with the S. typhimurium HWSH wild type, and serum was taken at day 2 postinfection. Serum was assayed for nitric oxide by the Griess reaction, which detects nitric oxide by determining nitrate-nitrite in the medium.
RESULTS
Susceptibility of TNFαp55R−/− and TNFαp55R+/+ mice to Salmonella infection.
TNFαp55R−/− and TNFαp55R+/+ mice were subjected to oral challenge with a number of derivatives of S. typhimurium HWSH. TNFαp55R−/− mice were significantly more susceptible than TNFαp55R+/+ to virulent S. typhimurium HWSH following oral challenge. Death occurred 3 to 5 days (average, 4 days) after oral inoculation of TNFαp55R−/− mice with a challenge of 108 S. typhimurium HWSH bacteria. In contrast, TNFαp55R+/+ mice did not start to die from infection until after 5 days, but most were dead at day 8 (average, day 7). Both TNFαp55R−/− and TNFαp55R+/+ mice succumbed to overwhelming bacterial infection, although the actual cause of mortality is unknown. Interestingly, TNFαp55R−/− mice displayed symptoms typically associated with lethal Salmonella infection of normal mice, including ruffled fur, sweating, and immobilization. The TNFαp55R−/− mice had small spleens compared to TNFαp55R+/+ mice at the time of death, and no Peyer’s patches were detectable macroscopically. Indeed, no Peyer’s patches were detected macroscopically in uninfected TNFαp55R−/− mice at the age they were used (6 to 8 weeks).
TNFαp55R−/− mice were also more susceptible to oral challenge with the partially attenuated S. typhimurium HWSH purE strain. All TNFαp55R−/− mice succumbed to oral challenge with S. typhimurium HWSH purE within 3 to 5 days. TNFαp55R+/+ mice partially controlled an S. typhimurium HWSH purE challenge, with 70% of animals surviving infection and clearing the challenge bacteria. The remaining 30% of TNFαp55R+/+ mice developed pathology, including abscess formation in the livers and spleens, and died. This is in agreement with previously published data (30). In contrast, S. typhimurium HWSH aroA was well controlled by these mice. TNFαp55R−/− mice orally challenged with S. typhimurium HWSH aroA exhibited a slight increase in spleen size and, interestingly, developed gut-associated tissue resembling abnormal looking Peyer’s patches.
Enumeration of S. typhimurium HWSH derivatives in the internal organs of orally challenged TNFαp55R−/− and TNFαp55R+/+ mice.
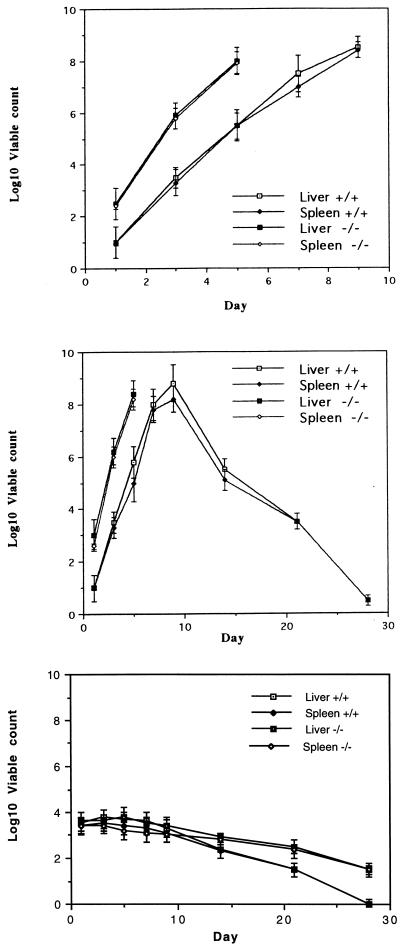
The numbers of bacteria present in selected organs of mice were determined at various times after oral challenge with different S. typhimurium HWSH derivatives. Counts of S. typhimurium HWSH in the livers and spleens of TNFαp55R−/− and TNFαp55R+/+ mice increased at about 1 log unit per day until death (Fig. 1). The same growth rate was observed in TNFαp55R−/− mice infected with S. typhimurium HWSH purE (Fig. 1). In contrast, TNFαp55R+/+ mice either controlled the infection or succumbed to death associated with abscess formation (30). TNFαp55R+/+ and TNFαp55R−/− mice orally challenged with S. typhimurium HWSH aroA all cleared the vaccine strain from internal organs, but TNFαp55R−/− mice had higher levels of the vaccine strain persisting, indicating that they did not clear the vaccine strain as efficiently as normal mice (Fig. 1).
FIG. 1.
Viable counts of the S. typhimurium HWSH wild type and purE and aroA strains in the spleens and livers of mice following oral challenge. (Top) S. typhimurium HWSH wild-type bacteria (108) administered orally to TNFαp55R+/+ and TNFαp55R−/− mice. (Middle) S. typhimurium HWSH purE bacteria (108) administered orally to TNFαp55R+/+ and TNFαp55R−/− mice. (Bottom) S. typhimurium HWSH aroA bacteria (108) administered orally to TNFαp55R+/+ and TNFαp55R−/− mice. Counts were performed on four animals per time point, with the experiment repeated three times.
Vaccination studies.
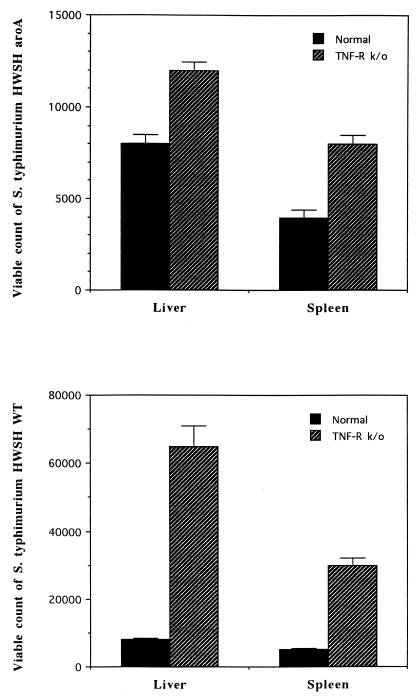
Groups of TNFαp55R−/− and TNFαp55R+/+ mice were orally vaccinated with 5 × 108 or 109 S. typhimurium HWSH aroA bacteria. At day 28 postvaccination, both groups were orally challenged with virulent S. typhimurium at a dose of 108 bacteria. All TNFαp55R+/+ mice survived challenge and were protected, but all of the TNFαp55R−/− group were dead by day 5. The experiment was performed independently three times with identical results. TNFαp55R−/− mice had larger numbers of S. typhimurium HWSH aroA organisms in their tissues at day 28 after immunization and 2 days after virulent S. typhimurium HWSH challenge (Fig. 2).
FIG. 2.
Viable counts of S. typhimurium HWSH aroA bacteria at 28 days postvaccination in the livers and spleens of TNFαp55R+/+ and TNFαp55R−/− mice. (Bottom) Viable counts of the S. typhimurium HWSH wild-type strain at 2 days postchallenge in the livers and spleens of TNFαp55R+/+ and TNFαp55R−/− mice.
Splenocyte proliferation in response to Salmonella antigen.
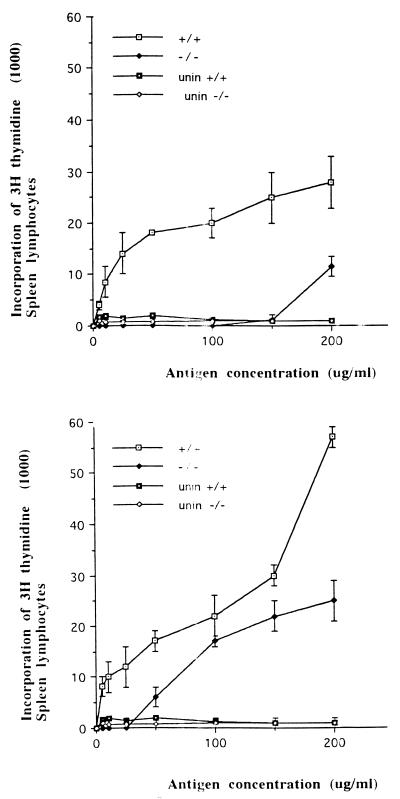
Splenocyte proliferation experiments were performed with spleens from TNFαp55R−/− and TNFαp55R+/+ mice following vaccination with S. typhimurium HWSH aroA and subsequent challenge with virulent S. typhimurium HWSH. Splenocytes prepared from TNFαp55R+/+ mice at 28 days postvaccination with S. typhimurium HWSH aroA proliferated in response to detoxified Salmonella lysate (detoxified LPS) (Fig. 3). Splenocytes prepared from TNFαp55R−/− mice at 28 days postvaccination proliferated only in response to high concentrations of Salmonella antigen and at lower levels than equivalent splenocytes prepared from the TNFαp55R+/+ mice (Fig. 3). Two days after S. typhimurium HWSH wild-type challenge, previously vaccinated mice were sacrificed and the ability of their splenocytes to proliferate in the presence of Salmonella antigen was assessed. As expected, splenocytes prepared from S. typhimurium HWSH-challenged TNFαp55R+/+ mice demonstrated a stronger proliferative response than did similar mice immunized with S. typhimurium HWSH aroA. Interestingly, splenocytes prepared from TNFαp55R−/− mice previously vaccinated with S. typhimurium HWSH aroA and challenged with the S. typhimurium HWSH wild type proliferated well in response to Salmonella antigen. However, the proliferative response was not as vigorous as in the TNFαp55R+/+ mice. This response was ineffective in controlling infection, as all TNFαp55R−/− mice died.
FIG. 3.
Proliferation of spleen cells isolated from Salmonella-infected mice. (Top) Spleen cells isolated from TNFαp55R+/+ and TNFαp55R−/− mice at 28 days after vaccination with S. typhimurium HWSH aroA proliferate after stimulation with detoxified Salmonella lysate. (Bottom) Spleen cells isolated from TNFαp55R+/+ and TNFαp55R−/− mice at 2 days after challenge with the S. typhimurium HWSH wild-type strain. The results of both experiments are averages of spleen cells from six mice per time point. unin, uninfected mice.
Measurement of cytokines in supernatants of splenocytes prepared from TNFαp55R−/− and TNFαp55R+/+ mice and stimulated with Salmonella antigen.
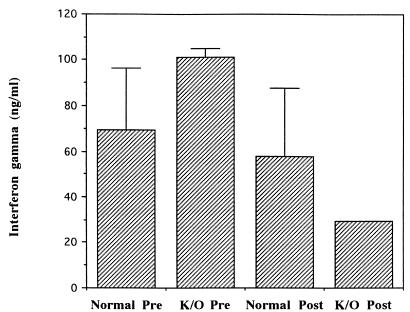
High levels of IFN-γ were detected in supernatants from Salmonella antigen-stimulated splenocytes prepared from S. typhimurium HWSH aroA-challenged TNFαp55R−/− and TNFαp55R+/+ mice. IFN-γ was also detected in supernatants of splenocytes prepared from TNFαp55R−/− mice 2 days after challenge with wild-type S. typhimurium HWSH (Fig. 4). IFN-γ production was decreased upon secondary challenge in knockout mice compared to normal animals. At the time points assayed, TNF-α, IL-4, IL-5, and IL-10 were not detected in supernatants from either TNFαp55R−/− or TNFαp55R+/+ mice (days 7 and 21). Splenocytes from control, uninfected animals did not proliferate in the presence of Salmonella antigens, whereas concanavalin A-stimulated cells produced all of the above cytokines.
FIG. 4.
Splenocytes from both TNFαp55R+/+ (Normal) and TNFαp55R−/− (K/O) mice vaccinated with S. typhimurium HWSH aroA (Pre) taken at day 28 postvaccination and then challenged with the S. typhimurium HWSH wild-type strain (Post) produce IFN-γ in the presence of Salmonella antigen.
Serum nitric oxide and antibody subclass postvaccination and postchallenge.
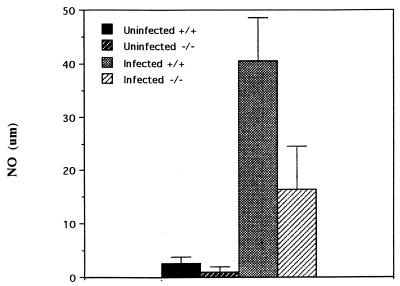
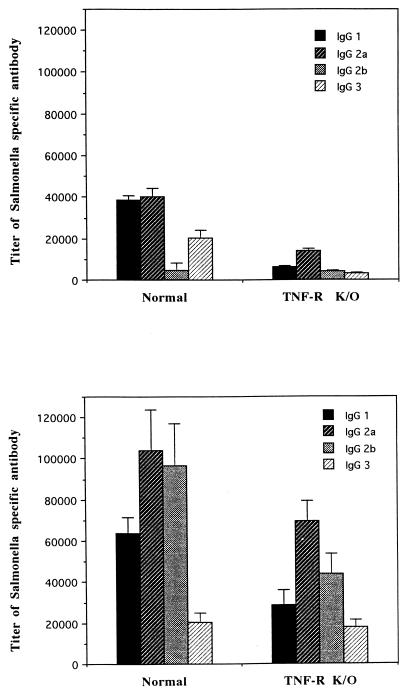
The levels of nitric oxide in the sera of TNFαp55R+/+ mice orally challenged with the S. typhimurium HWSH wild type were more than double those present in similarly infected TNFαp55R−/− mice (Fig. 5). Uninfected control animals had very low levels of nitric oxide in their sera. TNFαp55R+/+ mice orally challenged with S. typhimurium HWSH aroA mounted an anti-Salmonella immunoglobulin G (IgG) antibody response that was predominantly IgG2a, indicative of a strong Th1 response in these animals. IgG1 was also detected in normal mice. However, similarly challenged TNFαp55R−/− mice were also able to mount smaller IgG2a and IgG1 responses. Sixty percent of these mice demonstrated an IgG2a response, with only 12% producing anti-Salmonella IgG3. Postchallenge, all animals exhibited boosted responses in both groups, with the TNFαp55R−/− mice exhibiting lower titers than the TNFαp55R+/+ mice (Fig. 6). The TNFαp55R−/− mice had lower titers of antibody to Salmonella than did normal mice postvaccination.
FIG. 5.
Serum nitric oxide (NO) levels in TNFαp55R+/+ and TNFαp55R−/− mice infected with wild-type S. typhimurium HWSH (day 2 after infection).
FIG. 6.
Subclass-specific antibodies in murine serum. (Top) Subclass-specific titers of antibody against S. typhimurium HWSH at 28 days after vaccination with S. typhimurium HWSH aroA. (Bottom) Subclass-specific titers of antibody against S. typhimurium HWSH at 2 days after wild-type challenge of vaccinated mice. K/O, knockout.
Histopathology of infected mice.
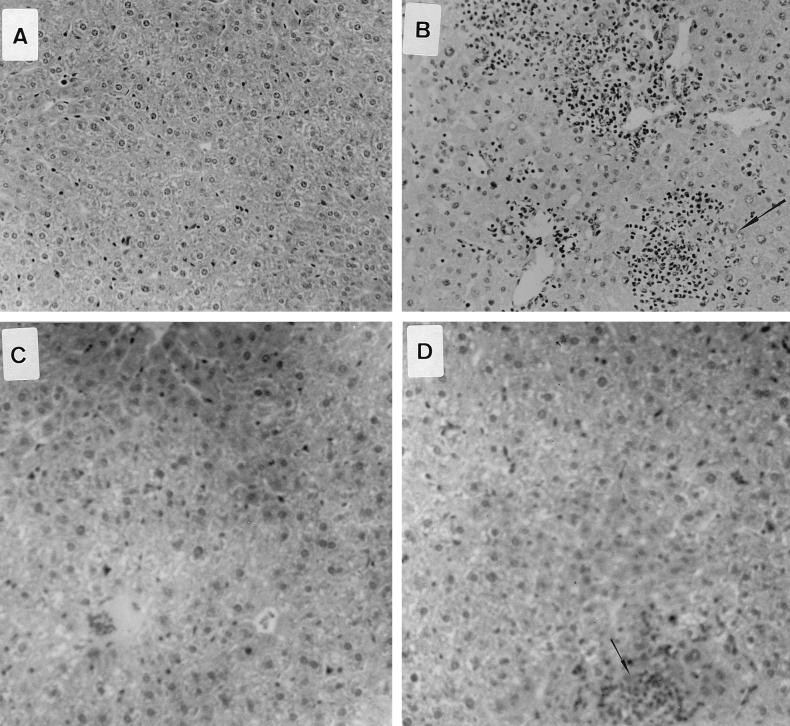
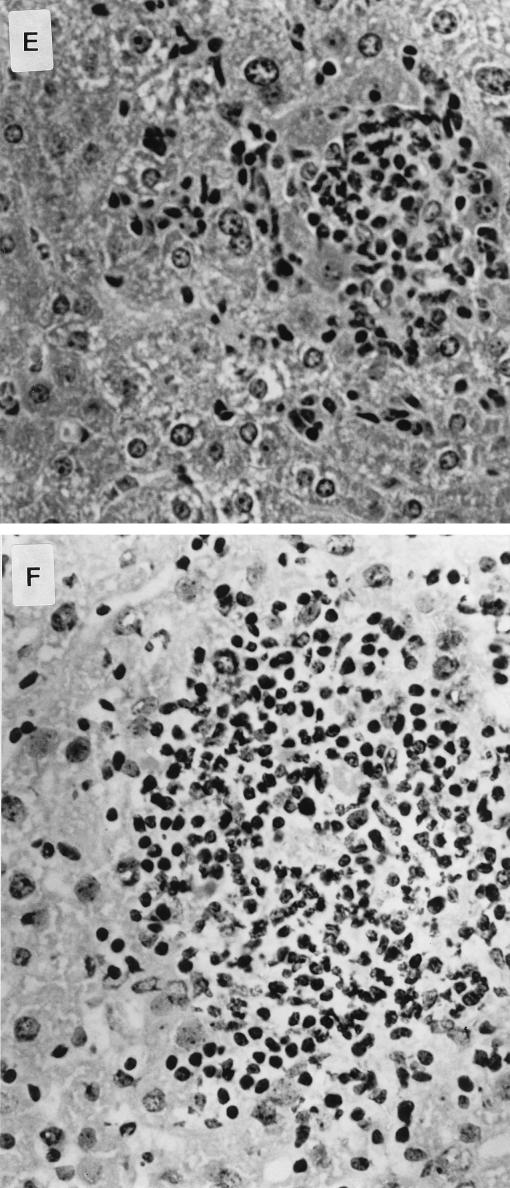
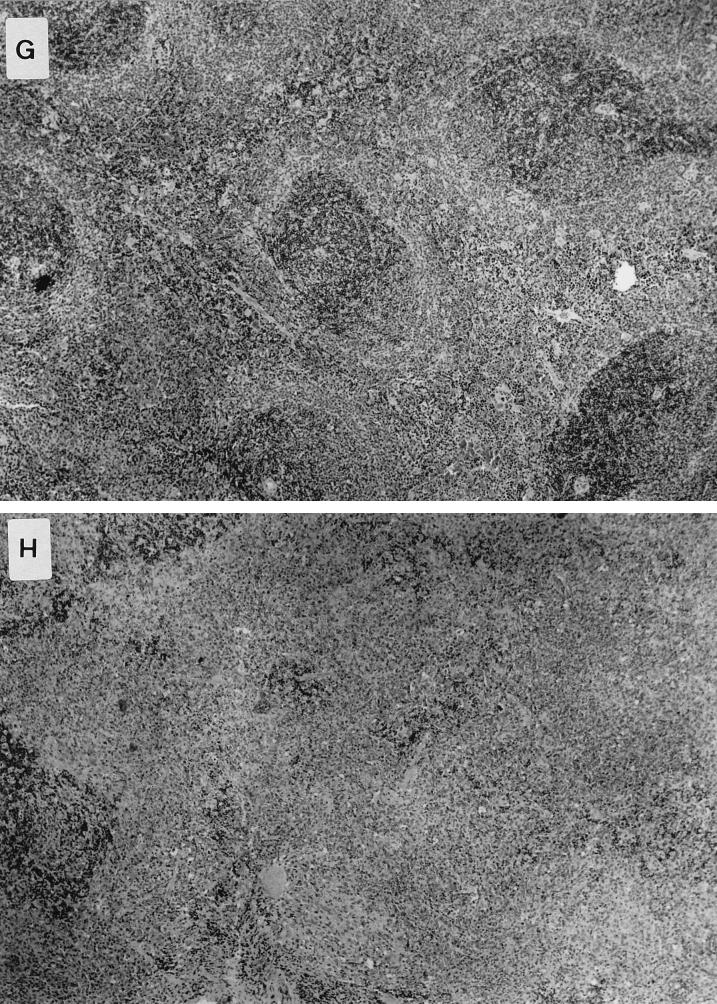
Histological examination of livers and spleens from infected normal and knockout animals was undertaken 4 days after oral inoculation of the S. typhimurium HWSH wild type. In TNFαp55R−/− mice, the livers showed focal areas of inflammatory cells consisting of predominantly neutrophils (polymorphonuclear leukocytes [PMNs]), with some mononuclear cells (Fig. 7). These were scattered throughout the entire section and were numerous and extensive. The spleens of TNFαp55R−/− mice showed large areas of inflammatory cells, with the typical tissue architecture of the spleen disrupted by predominantly PMNs and mononuclear cells. In contrast, normal mice showed similar areas but they were smaller and less numerous in both livers and spleens (Fig. 7).
FIG. 7.
Pathology in livers and spleens of mice infected with S. typhimurium HWSH derivatives and of uninfected controls. (A) Uninfected liver of a TNFαp55R−/− mouse (magnification, ×13.6 [low power]). (B) Liver of a TNFαp55R−/− mouse infected with wild-type S. typhimurium HWSH. Livers of these mice have microabscess formations with many neutrophils and macrophages scattered widely within the tissue (arrow) (magnification, ×13.6 [low power]). (C) Uninfected liver of a TNFαp55R+/+ mouse (magnification, ×13.6 [low power]). (D) Liver of a TNFαp55R+/+ mouse infected with wild-type S. typhimurium HWSH. Focal granulomas consisting mainly of macrophages and some PMNs were smaller than in TNFαp55R−/− animals and less numerous throughout the tissues (arrow) (magnification, ×13.6 [low power]). (E) High-power magnification (×40) of focal inflammation in the liver of a TNFαp55R+/+ mouse infected with wild-type S. typhimurium HWSH. (F) High-power magnification (×40) of a large inflammatory lesion within the liver of a TNFαp55R−/− mouse showing many inflammatory cells, consisting mainly of PMNs and macrophages. (G) Spleen of a TNFαp55R+/+ mouse infected with wild-type S. typhimurium HWSH, showing normal tissue organization within an infected spleen (magnification, ×16 [low power]). (H) Spleen of a TNFαp55R−/− mouse infected with wild-type S. typhimurium HWSH, showing large necrotic areas of poorly organized aggregates of neutrophils and macrophages (magnification, ×16 [low power]).
DISCUSSION
In this study we demonstrate, by using gene knockout mice combined with Salmonella HWSH derivatives of differing virulence, the importance of TNFαp55R to both innate and acquired immunity to salmonellosis. Several interesting observations have emerged from these studies. We have shown that TNFαp55R is essential for controlling infections by fully virulent S. typhimurium HWSH as well as partially attenuated strains such as S. typhimurium HWSH purE. TNFαp55R−/− mice die of overwhelming infection before any appreciable immune cell recruitment occurs in the spleen, as judged by the small spleen size at the time of death and the disorganized tissue architecture. Mice differ in the number of organisms present on day 1 of infection, with the knockout mice having higher counts in the livers and spleens. It is not known why this difference in bacterial numbers between the two groups of mice is distinct so early in infection. TNF-α may be needed for PMN recruitment to control Salmonella at this early time point. TNF-α is required to efficiently clear the S. typhimurium HWSH aroA vaccine strain from internal organs. However, the S. typhimurium HWSH aroA strain did not kill orally challenged TNFαp55R−/− mice, and the bacterial growth was partially controlled. Others have shown that IFN-γ gene knockout mice are hypersusceptible to S. typhimurium aroA challenge (14, 40). We have confirmed that IFN-γ gene knockout mice can be killed by oral challenge with S. typhimurium HWSH aroA (our results not shown), and thus gene knockout mice exhibit significant differences in their abilities to control the growth even of highly attenuated Salmonella strains in normal mice. From these observations, it is evident that the expression of TNF-α in IFN-γ gene knockout mice is not sufficient alone to protect mice from overwhelming infection by Salmonella strains. TNFαp55R−/− mice produce IFN-γ in response to challenge and vaccination. However, the IFN-γ production is not sufficient to protect the TNFαp55R−/− mice from wild-type S. typhimurium HWSH challenge, demonstrating the importance of TNF-α alone or more probably the synergistic effects of IFN-γ and TNF-α in recall of immunity and bacterial killing (9, 24, 32). Antibody does not protect knockout animals from lethal infection. This observation comes from the boosted antibody levels seen in knockout animals by prevaccination and then challenge. Boosted animals still died from wild-type challenge.
Postmortem, the TNFαp55R−/− mice orally challenged with the virulent S. typhimurium HWSH wild type or unchallenged naive mice had no obvious Peyer’s patch structures upon macroscopic observation. Others have reported the lack of Peyer’s patch-like structures in TNFαp55R−/− mice (27). Significantly, tissues resembling Peyer’s patches were observed associated with the guts of TNFαp55R−/− mice orally challenged with the highly attenuated S. typhimurium HWSH aroA vaccine strain. Interestingly, however, the Peyer’s patches look macroscopically different from the Peyer’s patches of TNFαp55R+/+ mice (our unpublished observations). Pasparakis et al. (33) observed the formation of Peyer’s patches in TNFαp55R−/− mice. In their animals, which are from the same source as ours, they observed small, flat Peyer’s patches, the numbers being reduced to two to four per mouse. They also observed defective formation of B-cell follicles. The ages of the mice they used for their experiments are not stated, and for our experiments we used only mice between 6 and 8 weeks old. It seems probable that our uninfected mice would have developed Peyer’s patches such as those observed by Pasparakis et al., but the influence of infection with a Salmonella aroA mutant upon morphology is not known.
The bacterial product most frequently implicated in TNF-α induction in vivo is LPS. TNF-α induction is involved in the generation of reactive nitrogen intermediates in mice that can lead to bacterial killing (12–14). This pathway involves LPS-mediated induction of TNF-α and IFN-γ in macrophages linked to the induction of inducible nitric oxide synthase, which generates nitric oxide. In TNFαp55R−/− infected mice, there were significantly reduced detectable serum nitric oxide levels compared to TNFαp55R+/+ infected mice, where double the amount of nitric oxide was detected during infection. The defect in bacterial killing exhibited in TNFαp55R−/− mice, and thus the inability of these animals to control infection, may be attributable in part to their inability to kill via the nitric oxide pathway. Flynn et al. (9) found that macrophages from TNFαp55R−/− mice were unable to synthesize large amounts of reactive nitrogen intermediates following mycobacterial infection. It was postulated that this effect was exerted either directly on macrophages or through a blockade of TNF-α-mediated activation of IFN-γ.
Our data, taken together with that of Mastroeni et al. (22, 23), who used anti-TNF-α antibody to assess the contribution of TNF-α to the control of Salmonella infection, suggests that neutralization of TNF-α or deletion of TNFαp55R increases murine susceptibility to salmonellosis. For example, a normally sublethal infection is lethal after anti-TNF-α administration, and treatment with antibodies to TNF-α or IFN-γ abolishes the plateau phase in ityr mice, allowing bacteria to grow unchecked.
In summary, we have demonstrated a central role of TNFαp55R in controlling infection by S. typhimurium. It would be of interest to see if TNF-α plays a role in humans in controlling S. typhi growth associated with typhoid fever. We are currently undertaking a study to investigate the nature of TNF-α production in this disease and to screen for potential genetic polymorphisms associated with the human TNF-α and TNFp55R genes.
ACKNOWLEDGMENT
This work was supported by a program of the Wellcome Trust.
REFERENCES
- 1.Bancroft G J, Sheehan K C F, Schreiber R D, Unanue E. Tumour necrosis factor is involved in the T-cell independent pathway of macrophage activation in SCID mice. J Immunol. 1989;143:127–132. [PubMed] [Google Scholar]
- 2.Bermudez L E M, Young L S. Tumour necrosis factor alone or in combination with IL-2, but not IFN-γ, is associated with macrophage killing of Mycobacterium avium complex. J Immunol. 1988;140:3006–3013. [PubMed] [Google Scholar]
- 3.Blackwell J M. Bacterial infections. In: Wakelin D M, Blackwell J M, editors. Genetics of resistance to bacterial and parasitic infections. London, United Kingdom: Taylor and Francis; 1988. pp. 63–98. [Google Scholar]
- 4.Collins F M, Mackaness G B, Blanden R V. Infection and immunity in experimental salmonellosis. J Exp Med. 1966;124:601–619. doi: 10.1084/jem.124.4.601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Collins F M. Vaccines and cell-mediated immunity. Bacteriol Rev. 1974;38:371–402. doi: 10.1128/br.38.4.371-402.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Dougan G. Genetics as a route toward mucosal vaccine development. In: Miller V L, Kaper J B, Portnoy D A, Isberg R R, editors. Molecular genetics of bacterial pathogenesis. Washington, D.C: ASM Press; 1994. pp. 491–506. [Google Scholar]
- 7.Eisenstein T K, Killar L, Sultzer B M. Immunity and infection in Salmonella typhimurium: mouse-strain differences in vaccine and serum induced protection. J Infect Dis. 1985;150:425–435. doi: 10.1093/infdis/150.3.425. [DOI] [PubMed] [Google Scholar]
- 8.Finlay B B. Molecular and cellular mechanisms of Salmonella pathogenesis. Curr Top Microbiol. 1994;192:163–185. doi: 10.1007/978-3-642-78624-2_8. [DOI] [PubMed] [Google Scholar]
- 9.Flynn J L, Goldstein M M, Chan J, Triebold K J, Pfeffer K, Lowenstein C J, Schreiber R, Mak T W, Bloom B B. Tumour necrosis factor α is required in the protective immune response against Mycobacterium tuberculosis in mice. Immunity. 1995;2:561–566. doi: 10.1016/1074-7613(95)90001-2. [DOI] [PubMed] [Google Scholar]
- 10.Green S J, Nacy C A, Schreiber R D, Granger D L, Crawford R M, Meltzer M S, Fortier A H. Neutralization of gamma interferon and tumor necrosis factor alpha blocks in vivo synthesis of nitrogen oxides from l-arginine and protection against Francisella tularensis infection in Mycobacterium bovis BCG-treated mice. Infect Immun. 1991;61:689–698. doi: 10.1128/iai.61.2.689-698.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Green S J, Nacy C A. Antimicrobial and immunopathologic effects of cytokine-induced nitric oxide synthesis. Curr Opin Infect Dis. 1993;6:384–396. [Google Scholar]
- 12.Havell E A. Production of tumour necrosis factor during murine listeriosis. J Immunol. 1987;139:4225–4231. [PubMed] [Google Scholar]
- 13.Havell E A. Evidence that tumour necrosis factor has an important role in antibacterial resistance. J Immunol. 1989;143:2894–2899. [PubMed] [Google Scholar]
- 14.Hess J, Ladel C, Miko D, Kaufmann S H. Salmonella typhimurium aroA− infection in gene targetted immunodeficient mice: major role of CD4+ TCR-alpha beta cells and IFN-gamma in bacterial clearance independent of intracellular location. J Immunol. 1996;156:3321–3326. [PubMed] [Google Scholar]
- 15.Hook E W. Salmonella species (including typhoid fever) In: Mandell G L, Douglas R G, Bennett J E, editors. Principles and practice of infectious diseases. New York, N.Y: Churchill Livingstone; 1985. pp. 1256–1266. [Google Scholar]
- 16.Hormaeche C E. Natural resistance to Salmonella typhimurium in different inbred mouse strains. Immunology. 1979;37:311–318. [PMC free article] [PubMed] [Google Scholar]
- 17.Hormaeche C E, Mastroeni P, Arena A, Uddin J, Joysey H S. T-cells do not mediate the initial suppression of a Salmonella infection in the R. E S Immunology. 1990;70:247–250. [PMC free article] [PubMed] [Google Scholar]
- 18.Hornick R B, Greisman S E, Woodward T E, DuPont H L, Dawkins A T, Snyder M J. Typhoid fever: pathogenesis and control. N Engl J Med. 1970;283:686–691. doi: 10.1056/NEJM197009242831306. [DOI] [PubMed] [Google Scholar]
- 19.Hornick R B, Greisman S E, Woodward T E, DuPont H L, Dawkins A T, Snyder M J. Typhoid fever: pathogenesis and control. N Engl J Med. 1970;283:739–746. doi: 10.1056/NEJM197010012831406. [DOI] [PubMed] [Google Scholar]
- 20.Kaufmann S H E. Immunity to intracellular bacteria. Annu Rev Immunol. 1993;11:129–163. doi: 10.1146/annurev.iy.11.040193.001021. [DOI] [PubMed] [Google Scholar]
- 21.Kaufmann S H E. Bacterial and protozoal infections in genetically disrupted mice. Curr Opin Immunol. 1994;6:518–525. doi: 10.1016/0952-7915(94)90135-x. [DOI] [PubMed] [Google Scholar]
- 22.Mastroeni P, Arena A, Costa G B, Liberto M C, Bonina L, Hormaeche C E. Serum TNF in mouse typhoid and enhancement of the infection by anti-TNF antibodies. Microb Pathog. 1991;11:33–38. doi: 10.1016/0882-4010(91)90091-n. [DOI] [PubMed] [Google Scholar]
- 23.Mastroeni P, Villareal B, Hormaeche C E. Role of T-cells, TNF and IFN in recall of immunity to oral challenge with virulent salmonellae in mice vaccinated with live attenuated aro Salmonella vaccines. Microb Pathog. 1992;13:477–491. doi: 10.1016/0882-4010(92)90014-f. [DOI] [PubMed] [Google Scholar]
- 24.Muotiala A, Makela P H. The role of IFN in murine Salmonella typhimurium infection. Microb Pathog. 1990;8:135–141. doi: 10.1016/0882-4010(90)90077-4. [DOI] [PubMed] [Google Scholar]
- 25.Nauciel C, Espinasse-Maes F, Matsiota-Bernad P. Role of γ interferon and tumor necrosis factor in early resistance to murine salmonellosis. In: Cabello F, Hormaeche C E, Bonina L, Mastroeni P, editors. Biology of Salmonella. NATO A. S. I. series A245. New York, N.Y: Plenum Press; 1992. pp. 255–264. [Google Scholar]
- 26.Nauciel C, Espinasse-Maes F, Matsiota-Bernad P. Role of gamma interferon and tumor necrosis factor alpha in resistance to Salmonella typhimurium infection. Infect Immun. 1992;60:450–454. doi: 10.1128/iai.60.2.450-454.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Neumann B, Luz A, Pfeffer K, Holzmann B. Defective Peyer’s patch organogenesis in mice lacking the 55-kD receptor for tumour necrosis factor. J Exp Med. 1996;184:259–264. doi: 10.1084/jem.184.1.259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Nicola N A, editor. Guidebook to cytokines and their receptors. Oxford, United Kingdom: Oxford University Press; 1995. [Google Scholar]
- 29.O’Brien A D, Metcalf E S. Control of early Salmonella typhimurium growth in innately salmonella resistant mice does not require functional T-lymphocytes. J Immunol. 1982;129:1349–1351. [PubMed] [Google Scholar]
- 30.O’Callaghan D, Maskell D, Liew F Y, Easmon C S F, Dougan G. Characterization of aromatic- and purine-dependent Salmonella typhimurium: attenuation, persistence, and ability to induce protective immunity in BALB/c mice. Infect Immun. 1988;56:419–423. doi: 10.1128/iai.56.2.419-423.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.O’Callaghan D, Maskell D, Tite J, Dougan G. Immune responses in BALB/c mice following immunisation with aromatic compound or purine dependent Salmonella typhimurium strains. Immunology. 1990;69:184–189. [PMC free article] [PubMed] [Google Scholar]
- 32.Oswald I P, Wynn T A, Sher A, James S L. IL-10 inhibits macrophage microbicidal activity by blocking the production of TNFα required as a costimulatory factor for interferon-γ induced activation. Proc Natl Acad Sci USA. 1992;89:8676–8680. doi: 10.1073/pnas.89.18.8676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Pasparakis M, Alexopolou L, Grell M, Pfizenmaier K, Bluethmann H, Kollias G. Peyer’s patch organogenesis is intact yet formation of B lymphocyte follicles is defective in peripheral lymphoid organs of mice deficient for tumor necrosis factor and its 55-kDa receptor. Proc Natl Acad Sci USA. 1997;94:6319–6323. doi: 10.1073/pnas.94.12.6319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Pie S, Matsiota-Bernard P, Truffa-Bachi P, Nauciel C. Gamma interferon and interleukin-10 gene expression in innately susceptible and resistant mice during the early phase of Salmonella typhimurium infection. Infect Immun. 1996;64:849–854. doi: 10.1128/iai.64.3.849-854.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Plant J, Glynn A A. Locating salmonella resistance genes on mouse chromosome 1. Clin Exp Immunol. 1979;37:1–6. [PMC free article] [PubMed] [Google Scholar]
- 36.Rockett K A, Awburn M M, Cowden W B, Clark I A. Killing of Plasmodium falciparum in vitro by nitric oxide derivatives. Infect Immun. 1991;59:3280–3283. doi: 10.1128/iai.59.9.3280-3283.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Rothe J, Lesslauer W, Lotscher H, Lang Y, Koebel P, Kontgen F, Althage A, Zinkernagel R, Steinmetz M, Bluethmann H. Mice lacking the tumour necrosis factor receptor 1 are resistant to TNF-mediated toxicity but highly susceptible to infection by Listeria monocytogenes. Nature. 1993;364:798–802. doi: 10.1038/364798a0. [DOI] [PubMed] [Google Scholar]
- 38.Tite J P, Dougan G, Chatfield S N. The involvement of tumour necrosis factor in immunity to Salmonella typhimurium. J Immunol. 1991;47:3161–3164. [PubMed] [Google Scholar]
- 39.Tracey K J, Cerami A. Cachectin/tumour necrosis factor and other cytokines in infectious disease. Curr Opin Immunol. 1989;1:454–461. doi: 10.1016/0952-7915(88)90026-x. [DOI] [PubMed] [Google Scholar]
- 40.Vancott J L, Yamamoto M, van Ginkel F W, Okahashi N, Pascual D W, Kiyono H, Bluthman H, McGhee J R. The host response to vaccines in the context of transgenic or targeted gene knockout mice. Mucosal Immunol Update. 1995;3(4):3,12–14. [Google Scholar]
- 41.Villareal B, Mastroeni P, DeMarco de Hormaeche R, Hormaeche C E. Proliferative and T-cell specific IL-2/IL-4 responses in spleen cells from mice vaccinated with aroA live attenuated Salmonella vaccines. Microb Pathog. 1992;13:305–315. doi: 10.1016/0882-4010(92)90040-u. [DOI] [PubMed] [Google Scholar]