Abstract
Cellular senescence represents an irreversible state of cell-cycle arrest during which cells secrete senescence-associated secretory phenotypes, including inflammatory factors and chemokines. Additionally, these cells exhibit an apoptotic resistance phenotype. Cellular senescence serves a pivotal role not only in embryonic development, tissue regeneration, and tumor suppression but also in the pathogenesis of age-related degenerative diseases, malignancies, metabolic diseases, and kidney diseases. The senescence of renal tubular epithelial cells (RTEC) constitutes a critical cellular event in the progression of acute kidney injury (AKI). RTEC senescence inhibits renal regeneration and repair processes and, concurrently, promotes the transition of AKI to chronic kidney disease via the senescence-associated secretory phenotype. The mechanisms underlying cellular senescence are multifaceted and include telomere shortening or damage, DNA damage, mitochondrial autophagy deficiency, cellular metabolic disorders, endoplasmic reticulum stress, and epigenetic regulation. Strategies aimed at inhibiting RTEC senescence, targeting the clearance of senescent RTEC, or promoting the apoptosis of senescent RTEC hold promise for enhancing the renal prognosis of AKI. This review primarily focuses on the characteristics and mechanisms of RTEC senescence, and the impact of intervening RTEC senescence on the prognosis of AKI, aiming to provide a foundation for understanding the pathogenesis and providing potentially effective approaches for AKI treatment.
Subject terms: Ageing, Kidney
Facts
Cellular senescence represents an irreversible state of cell-cycle arrest, which has senescence-associated secretory phenotype (SASP) and apoptotic resistance phenotype.
Renal tubular epithelial cells (RTEC) senescence is a critical cellular event in the progression of acute kidney injury (AKI), which promotes the transition of AKI to chronic kidney disease.
Inhibiting RTEC senescence, targeting the clearance of senescent RTEC, or promoting the apoptosis of senescent RTEC hold promise for enhancing the renal prognosis of AKI.
Open questions
What are the differences in the characteristics and phenotypes of senescent renal tubular epithelial cells (RTEC) in distinct stressors-induced AKI?
What is the heterogeneity of the role and mechanism of senescent RTEC in distinct stressors-induced AKI?
How to improve the targeting and effectiveness of intervening RTEC senescence to further ameliorate the prognosis of AKI?
Introduction
Acute kidney injury (AKI) is a prevalent and life-threatening clinical condition characterized by a rapid decline in kidney function, with a high incidence and mortality rate [1]. However, the prognosis for surviving patients with AKI is far from optimistic. Research has shown that 30% to 70% of AKI survivors progress to chronic kidney disease (CKD), and approximately 17% of patients develop end-stage renal disease (ESRD) within one year, necessitating renal replacement therapy. This places a substantial economic burden on both society and affected families [2]. The causes of AKI encompass sepsis, nephrotoxic drugs, ischemia-reperfusion injury, and contrast-induced nephropathy. Regrettably, effective treatment strategies for AKI are currently lacking. Therefore, it is imperative to comprehensively elucidate the mechanisms underlying AKI for its prevention and treatment.
Renal tubular epithelial cells (RTECs) stand as among the most critical cells in the renal parenchyma, significantly influencing the physiological functions of the kidneys. These cells are characterized by their abundant mitochondria, signifying their high metabolic and energy demands. Situated in areas of the kidney with limited blood flow and oxygen supply, RTECs are inherently more vulnerable to damage. Additionally, they are predisposed to the accumulation and concentration of various harmful substances, rendering them the primary target cells of AKI [3]. Numerous studies have demonstrated that RTECs undergo necrosis, apoptosis, and ferroptosis following AKI. Residual RTECs possess robust proliferative and regenerative capacities that influence the prognosis of AKI. In mild and moderate AKI, RTECs quickly re-enter the cell cycle from a quiescent state and undergo proliferation and regeneration, thereby promoting the restoration of kidney structure and function. However, in severe AKI, the regenerative capacity of the residual RTEC is limited, leading to abnormal repair. Moreover, RTECs are prone to mitochondrial dysfunction, oxidative stress, and disturbances in energy metabolism, resulting in stress-induced cellular senescence. RTEC senescence not only hampers renal repair but also triggers the secretion of a multitude of senescence-associated secretory phenotypes (SASP), such as inflammatory factors and chemokines, which accelerate the progression of AKI to CKD. RTEC senescence, an early cellular event, plays a pivotal role in the development of AKI. Evidence suggests that RTEC senescence is associated with a decrease in renal recovery rate. This review primarily focuses on the characteristics and effects of cellular senescence, the mechanisms of RTEC senescence in AKI, and the impact of intervening RTEC senescence on the prognosis of AKI, aiming to provide a foundation for understanding the pathogenesis and treatment of AKI.
Cellular senescence
Characteristics and phenotypes of cellular senescence
Cellular senescence can be classified into two primary types: replicative and stress-induced premature senescence [4]. Replicative senescence primarily occurs due to telomere shortening and a decrease in telomerase activity, eventually leading to reaching the Hayflick limit [5]. Stress-induced premature senescence is independent of chronological aging and is initiated by various pathological triggers, such as DNA damage, oxidative stress, and oncogenes. Unlike quiescent cells, senescent cells experience an irreversible cell-cycle arrest at either the G1/S or G2/M phase and are incapable of proliferating or differentiating, even when subjected to specific stimuli.
Senescent cells exhibit several morphological alterations, including cellular enlargement, flattening, darker nuclear staining, and chromatin aggregation. Additionally, these cells secrete SASP and display resistance to apoptosis (Fig. 1). Recent studies employing ATAC-seq, RNA-seq, and ChIP-seq have identified NAT1, PBX1, and RRM2 as potential novel biomarkers of senescence and senescence-related diseases [4].
Fig. 1. The phenotypes and markers of cellular senescence.
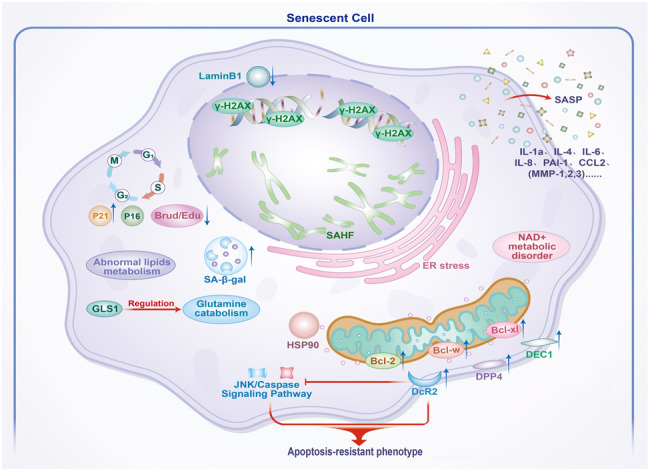
Cellular senescence is an irreversible state of cell-cycle arrest, which exhibits SASP and apoptotic resistance phenotypes. Senescent cells are enlarged and have an irregular shape. The phosphorylation of γH2AX increases following DNA damage in cellular senescence. SAHF accumulates and Lamin B1 downregulates in the nucleus of the senescent cell. Enhanced lysosomal content (SA-β-gal), ER stress, and mitochondrial dysfunction are observed in senescent cells. Moreover, lipid metabolism, glutamine catabolism, and NAD+ metabolism are abnormal after cellular senescence.
Cell morphology
Cellular senescence is characterized by irregular cell shape and cell enlargement, exceeding twice the size of normal cells, a hallmark feature of senescent cells [6]. Alterations in nuclear morphology and structure are signs of senescence, featuring enlarged multinucleated cells. The loss of nuclear membrane protein B1 (Lamin B1) contributes to nuclear membrane breakdown and the disruption of nuclear integrity, leading to condensed chromatin, the formation of heterochromatic foci, and the appearance of chromatin fragments in the cytoplasm, referred to as cytoplasmic chromatin fragments (CCFs) [7]. Aberrations in cell nucleus morphology and mutations in nuclear proteins such as MAN1, Nesprin-1, Emerin, and Torsin1, caused by premature senescence, are associated with various diseases, including cancer [8]. Additionally, cell organelles exhibit morphological abnormalities, characterized by an increased number of lysosomes and elevated β-galactosidase activity. Mitochondrial swelling and dysfunction result in the generation of excessive reactive oxygen species (ROS) [9].
Cell-cycle arrest
Cell-cycle regulation primarily relies on cell-cycle checkpoints, including the G1/S, G2/M, and spindle checkpoints. The cell cycle is influenced by various factors, such as cyclins, cyclin-dependent kinases (CDKs), and cyclin-dependent kinase inhibitors (CKIs). Cyclins form complexes with CDKs, leading to the phosphorylation of the retinoblastoma protein (RB) and the subsequent release of E2F, which facilitates the cell-cycle transition from the G1 to S phase.
Cell-cycle arrest is a primary characteristic of cellular senescence. Disruption in the regulation of cell-cycle progression can lead to cellular senescence. Unlike quiescent and terminally differentiated cells, senescence-related cycle arrest is irreversible, and senescent cells do not re-enter the cell cycle in response to any known physiological stimuli [7]. Cell-cycle arrest during the G1/S and G2/M phases induces cellular senescence [10, 11]. CKIs play a crucial role in the regulation of cellular senescence by promoting cell-cycle arrest. There are two principal families of CKIs: the Ink4 family and the KIP family. The Ink4 family includes p15, p16, p18, and p19, which inhibit the activity of CDK4/CDK6. Meanwhile, members of the KIP family, such as p21 and p27, reduce the activity of cyclin-CDK complexes, thereby promoting cell-cycle arrest [12]. The main factors driving cell-cycle arrest are CDKN2A (p16INK4, abbreviated as p16) and CKDN1A (p21CIP, abbreviated as p21). Both p21 and p16 inhibit the activity of CDK, leading to the non-phosphorylation of RB. This non-phosphorylated RB blocks the cell-cycle transition from the G1 to S phase [11]. Cell-cycle arrest during cellular senescence is primarily regulated by the p16INK4a/RB and p53/p21CIP1 pathways. Additionally, p53 induces cell-cycle arrest either at the G1/S or G2/M checkpoint, playing a pivotal role in the regulation of cellular senescence [13]. Post-translational modifications, such as ubiquitination, phosphorylation, acetylation, and SUMOylation, play crucial roles in regulating the levels and activity of p53.
Senescence-associated secretory phenotype (SASP)
Senescent cells exhibit metabolic activity and persistently secrete inflammatory factors, chemokines, matrix metalloproteinases, and other components of the SASP. The SASP fosters the senescence of neighboring cells and establishes a tissue microenvironment through autocrine and paracrine signaling. The composition and levels of the SASP vary based on the duration of senescence, senescence-induced stress, and cell type [14]. Furthermore, the composition and effects of the SASP diverge in different organs and disease types. In cases of skin trauma, the primary SASP components include CYR61 and PDGF-AA, which participate in the repair process. Tumor-induced senescence in liver cells is characterized by CCL-2, inducing immune surveillance effects [15]. In cases of aging-induced chronic senescence, IL-6, and PAI-1 contribute to chronic tissue damage and organ dysfunction [16], In CKD, the SASP predominantly consists of IL-1, 4, 6, 18, TGF-β, TNF-α, MMP-2, and PAI-1, which contribute to renal inflammation and fibrosis, thereby accelerating the deterioration of renal function [17]. In addition, SASP activation is regulated by transcription factors and epigenetic mechanisms.
Apoptosis resistance
Senescent cells generate various anti-apoptotic molecules that prevent their recognition and clearance by immune cells. The accumulation of senescent cells and the SASP exacerbate the progression of diseases [18, 19]. Multiple biological pathways, such as Bcl-2/Bcl-XL, p53/p21, and PI3K/AKT, are involved in the apoptotic resistance of senescent cells [20]. Both mRNA and protein levels of Bcl-W and Bcl-xL are upregulated in senescent cells, highlighting the pivotal role of the BCL-2 protein family in apoptosis resistance among senescent cells [21, 22]. Recent studies have revealed that the decoy receptor DcR2 is upregulated in senescent cells, and it functions to inhibit apoptosis in senescent fibroblasts and RTECs [23, 24]. Furthermore, p21 can inhibit apoptosis in senescent cells by suppressing the JNK and caspase signaling pathways [25]. The heat shock protein HSP90 emerges as a crucial factor for the survival of senescent cells, and the application of HSP90 inhibitors selectively induces apoptosis in senescent cells [20]. In a recent study, glutaminase 1 (GLS1) was found to inhibit senescent cell apoptosis by regulating glutamine metabolism [26].
Physiological functions of cellular senescence
Cellular senescence serves diverse roles in various species. It has proven beneficial in mitigating pathological stress during the physiological and developmental processes of fish, amphibians, and mammals. Notably, cellular senescence not only regulates tissue remodeling in the course of embryonic development but also contributes to processes such as regeneration, repair, and tumor suppression [27].
Embryonic development
Cellular senescence constitutes a highly conserved and regulated process that unfolds during embryonic development. It actively participates in tissue remodeling, development, and morphogenesis, which are developmental processes featuring programmed senescence events [28, 29]. Senescent cells are detectable in various tissues and organs, especially in the kidney [30], inner ear [31], limbs, and nervous system, throughout embryonic development. The senescence marker p21 has been identified in the early stages of embryonic development, and its knockout results in abnormal kidney development, indicating that p21 plays a key role in embryonic development. Furthermore, p21 is not synchronized with other senescent markers such as p16 and p53, and it is regulated by the TGF-β/SMAD and P13K/FOXO pathways [28]. Cellular senescence is further associated with transient structures, such as the pronephros in salamanders and the yolk sac in zebrafish [5], both of which are contingent upon WNT and Hedgehog pathways. However, in amphibians, senescence primarily relies on the TGF-β signaling.
Regeneration and repair
Acute senescence plays a crucial physiological role in wound healing and tissue homeostasis [32]. During the early stages of trauma repair, senescent cells stimulate myofibroblast differentiation, facilitating scar formation and wound healing through paracrine signaling mechanisms, such as via platelet-derived growth factor AA (PDGF-AA). However, the removal of senescent cells results in delayed wound repair [33]. In the latter stages of trauma repair, senescent fibroblasts secrete antifibrotic factors that inhibit tissue fibrosis, thereby supporting adaptive repair processes [34]. Senescent fibroblasts and endothelial cells promote the healing of compromised skin, while the elimination of senescent cells yields opposing effects [33]. Similar roles for senescent fibroblasts have been reported in the context of myocardial infarction [35]. Moreover, senescent hepatic stellate cells decrease the secretion of extracellular matrix and enhance the production of extracellular matrix-degrading enzymes in liver injury models, thereby enhancing immune surveillance and restraining liver fibrosis [36]. In the early stages of ischemia-reperfusion-induced AKI, stress-induced senescent RTECs resist necrosis and apoptosis, consequently promoting the proliferation and regeneration of other RTECs [37]. This underscores the vital role of cellular senescence in the regeneration and repair of diverse organs following injury.
Tumor suppression
Numerous studies have shown that cellular senescence plays an important role in tumor suppression. The mechanisms underlying cellular senescence in the context of cancer primarily encompass oncogene-induced senescence (OIS), therapy-induced senescence (TIS), and the loss of tumor suppressor genes, exemplified by PTEN loss-induced cellular senescence (PICS) [38]. The p53 pathway plays a central role in both OIS and PICS. In OIS, the activation of oncogenes leads to DNA damage, subsequently activating p53 and leading to cellular senescence. In the case of PICS, p53 signaling is triggered through the mTOR pathway, even in the absence of DNA damage. Senescent cells can induce the senescence of neighboring tumor cells via the SASP, thereby inhibiting tumor progression. Remarkably, there exists a positive correlation between the low expression of senescence markers and a poor prognosis in renal cancer. The selective inhibition of cellular senescence leads to accelerated tumor progression, underscoring the protective effect of cell senescence in renal tumors. In addition, the inflammatory factors and chemokines secreted by tumor-induced senescent liver cells promote the chemotaxis of immune cells, thereby facilitating the elimination of tumor cells. Conversely, the elimination of senescent liver cells accelerates the progression of liver tumors, demonstrating that cellular senescence actively promotes and enhances the function of immune cells in clearing tumor cells.
Pathological roles of cellular senescence
Extensive evidence substantiates that senescent cells initiate the onset of diseases and accelerate the progression of various diseases [39, 40]. Cellular senescence is not only associated with age-related degenerative diseases but also plays a key role in the pathogenesis of tumors, metabolic diseases, and acute as well as chronic kidney diseases. Senescent cells, along with the SASP, represent fundamental mechanisms that drive or exacerbate disease progression. In addition, senescent cells demonstrate resistance to apoptosis, which consequently results in the accumulation of senescent cells and SASP, thereby intensifying tissue damage and dysfunction.
Neurodegenerative diseases
A large accumulation of senescent cells is evident in brain tissue associated with neurodegenerative diseases, most notably Alzheimer’s and Parkinson’s disease. Senescent astrocytes have been directly linked to the onset of these diseases, while tau protein accumulation is closely correlated with brain aging [41]. In this sense, the elimination of senescent glial cells can prevent cognitive decline caused by the accumulation of tau protein [42, 43]. These findings suggest that cellular senescence plays a crucial role in the occurrence and progression of neurodegenerative diseases.
Tumor pathogenesis
Beyond its protective effects on tumors, cellular senescence also exerts adverse impacts on tumorigenesis. Increasing evidence suggests that senescence, in conjunction with the senescent microenvironment, is associated with cancer development and metastasis [44–46]. Senescent cells secrete MMP3, which in turn promotes tumor cell invasion and angiogenesis [47]. Furthermore, the SASP facilitates the transition from epithelial to mesenchymal cells, a transformation involved in the progression of metastatic cancer [48]. SASP constituents secreted by malignant cells promote immune evasion and drug resistance in tumor cells [49]. Moreover, senescence-related genes hold significant clinical relevance in the prognosis and immunotherapy of patients with cancer. For instance, the specific clearance of p16-positive cells retards tumor progression and diminishes tumor metastasis [50].
Diabetes
Senescent cells release a large number of SASP factors, which contribute to dysfunction in pancreatic beta cells and adipose tissue, thereby amplifying insulin resistance in peripheral tissues and culminating in the development of type 2 diabetes. Metabolic abnormalities, such as high blood sugar, the presence of its byproducts, and advanced glycation end products (AGEs), foster cellular senescence, thus further fueling the progression of diabetes [51]. In diabetes mouse models induced by high glucose, pancreatic beta cell proliferation is decreased, while the expression of senescence markers is increased [52, 53]. In addition, individuals with diabetes exhibit an increased susceptibility to age-related conditions, such as renal dysfunction, Alzheimer’s disease, cardiovascular disease, and visual impairment. Numerous studies have confirmed that a large number of senescent cells can be found in diabetic nephropathy, diabetic retinopathy, and vascular lesions stemming from diabetes. These senescent cells promote dysfunction, yet the inhibition of cellular senescence significantly ameliorates these deleterious effects. These findings suggest the pivotal role of cellular senescence in the progression of diabetes and its associated complications.
Kidney diseases
The kidneys are among the organs in the body that age most rapidly. In recent years, the role of renal parenchymal cell senescence in kidney disease has gradually gained attention. Various pathogenic factors stimulate the senescence of renal parenchymal cells, including tubular, mesangial, endothelial, and other cell types. Numerous studies have confirmed that senescence of renal parenchymal cells is a pivotal pathological feature in conditions such as diabetic nephropathy (DN), IgA nephropathy (IgAN), hypertensive nephropathy, and other CKD. It plays a substantial role in the progression of CKD [54]. Consequently, CKD is considered as a clinical model of renal senescence. Cellular senescence is closely linked to glomerulosclerosis, tubular atrophy, interstitial fibrosis, and the progression of DN in mouse models of diabetes induced by streptozotocin [55, 56]. In the context of DN, the inhibition of RTEC senescence significantly alleviates kidney damage and preserves renal function, thus delaying the progression of DN [57, 58]. Among patients with IgAN, markers of RTEC senescence (p21, p16, SA-β-gal) positively correlate with renal interstitial injury [59]. Hypertensive nephropathy is associated with the susceptibility of vascular and interstitial cells to senescence [60]. In renal transplantation, it has been observed that older donors are more prone to ischemia-reperfusion injury (IRI) compared to their younger counterparts.
RTECs are particularly susceptible to cellular senescence when exposed to nephrotoxic drugs, ischemia-reperfusion injury, and other stressors associated with AKI. In addition to RTECs, podocytes, endothelial cells, immune cells, and interstitial cells also undergo stress-induced senescence [61]. Cellular senescence in AKI inhibits renal regeneration and repair, ultimately resulting in a poor prognosis. The downregulation of anti-senescence genes, such as klotho, and telomeres promotes cellular senescence in AKI and IRI mouse models [62, 63]. The key pathways driving cellular senescence in AKI are the p53/p21CIP1 and p16INK4a/Rb pathways. These pathways sequentially inhibit CDK complex formation and Rb phosphorylation, thereby suppressing E2F activity via Rb. In mild or moderate AKI, senescent cells are effectively cleared by immune cells, thereby promoting adaptive kidney repair. However, severe or persistent AKI leads to the accumulation of senescent cells that secrete the SASP through autocrine or paracrine mechanisms, causing inflammation, fibroblast proliferation, and extracellular matrix deposition. This culminates in maladaptive renal repair and fibrosis [64].
Others
Cellular senescence is also involved in various other medical conditions, including atherosclerosis, skeletal disorders, metabolic syndrome, idiopathic pulmonary fibrosis, and glaucoma. Senescent smooth muscle cells, for instance, secrete IL-6 and IL-8, which in turn promote the proliferation and migration of adjacent smooth muscle cells, ultimately leading to pulmonary arterial hypertension [65]. As individuals age, most cells in the bone microenvironment become senescent, displaying a heterogeneous SASP in both humans and mice. This phenomenon actively fosters the development of conditions such as osteoporosis and osteoarthritis [66, 67]. Furthermore, senescent cells play a crucial role in the exhaustion of stem cells and the senescence of macrophages, B lymphocytes, and T lymphocytes.
Mechanisms of cellular senescence
Cellular senescence encompasses several mechanisms, including telomere damage or shortening, DNA damage, aberrant epigenetic modifications, mitochondrial dysfunction, nutrient-sensing imbalances, stem cell exhaustion, and disrupted cell communication. Recent studies have shown that chronic inflammation and ecological imbalance are correlated with cellular senescence [68].
DNA damage
The DNA damage response (DDR) is a crucial mechanism that triggers cellular senescence. DDR is activated in response to DNA damage and serves as a self-protective mechanism. However, sustained activation of DDR can promote cell-cycle arrest, ultimately resulting in cellular senescence. DNA damage leads to an increase in the phosphorylation of histone H2AX (γH2AX). This phosphorylation, in turn, activates the cell-cycle protein kinase inhibitors p16ink4a and p21cip1 through the p53 pathway. These proteins subsequently mediate the phosphorylation of the retinoblastoma tumor suppressor protein (Rb), culminating in cell-cycle arrest and the induction of cellular senescence [69].
Telomere shortening/damage
Telomeres are essential components that stabilize the ends of eukaryotic chromosomes, safeguarding the integrity of genetic information. These telomeres consist of a highly conserved non-coding repetitive DNA sequence, TTAGGG. Both telomerase and the shelterin complex play crucial roles in maintaining telomere structure and function [70–72]. Telomerase is responsible for maintaining telomeres and fulfilling a protective role. In mice, the loss of telomerase leads to premature senescence, while pharmacological activation of telomerase delays the aging process in aging mice [73]. Telomeres gradually shorten with each cell division, thereby promoting genomic instability and contributing to cellular senescence as well as senescence-related diseases. Telomere shortening represents the primary cause of replicative senescence, a phenomenon common to cellular senescence in various species, including humans and mice. Replicative senescence is observed in a multitude of cell types such as fibroblasts, keratinocytes, endothelial cells, lymphocytes, adrenal cortex cells, and chondrocytes. When telomeres shorten to a critical length, a sustained DNA damage response (DDR) is activated, ultimately leading to cellular senescence. Additionally, cellular stress-induced senescence, caused by various harmful stressors, leads to telomere shorting-induced DDR, ultimately resulting in cellular senescence.
Mitochondrial autophagy deficiency
Autophagy is a highly conserved process in eukaryotes, serving as the fundamental mechanism for the degradation and recycling of cellular components. Cellular autophagy is crucial for maintaining cellular metabolism, homeostasis, and the quality control of organelles [74]. Previous studies have revealed that levels of autophagy-related genes decrease with senescence, whereas increased autophagy has been shown to delay the senescence process [75, 76]. Mitochondrial autophagy plays a pivotal role in maintaining mitochondrial integrity. Defects in this process in the accumulation of damaged and fragmented mitochondria, leading to elevated production of reactive oxygen species (ROS). In contrast to non-specific clearance, mitochondrial autophagy selectively removes damaged or dysfunctional mitochondria through lysosomal degradation, ensuring mitochondrial quality. Research has demonstrated that mitochondrial autophagy is involved in various physiological and pathological processes, including early embryonic development, cell differentiation, and cell apoptosis [77]. In the kidney, which ranks second only to the heart in terms of mitochondrial abundance, mitochondrial damage is a key mechanism underlying kidney disease. Elevated ROS levels caused by mitochondrial damage are particularly crucial in cases of AKI [78]. Mitochondrial autophagy plays a protective role in damaged kidney tissues, especially in IRI [79, 80]. Activated mitochondrial autophagy effectively removes damaged mitochondria, subsequently reducing inflammation and ROS levels in in vivo and in vitro AKI models, thereby exerting a protective effect [81].
Mitochondrial autophagy is closely associated with cellular senescence. Loss of cellular autophagy leads to mitochondrial dysfunction and the accumulation of ROS, both of which are typical markers of senescence [82]. Numerous studies have demonstrated that mitochondrial dysfunction can induce cellular senescence [83]. Mitochondrial autophagy is essential for the development of senescence phenotypes [84]. Various stressors can trigger deficiencies in mitochondrial autophagy, ultimately promoting cellular senescence. Restoring autophagic function may delay the appearance of senescent phenotypes [85]. In senescent cells, the mitochondria exhibit a reduced mitochondrial membrane potential, decreased oxidative phosphorylation, and elevated ROS production [86–88]. Furthermore, while normal cellular mitochondria undergo a cycle of fission and fusion, mitochondria in senescent cells undergo excessive fusion. This may be associated with the downregulation of dynamin-related protein 1 (DRP1) and mitochondrial fission protein 1 (FIS1), which inhibit mitochondrial division and autophagy [89, 90]. Currently, two main pathways are involved in mitochondrial autophagy: PARKIN/PINK1-mediated mitochondrial autophagy and PARKIN/PINK1-independent receptor-mediated mitochondrial autophagy. The PINK1/Parkin-mediated pathway plays a protective role in AKI [91, 92]. Moreover, damage to PINK1-parkin-mediated mitochondrial autophagy has been linked to lung cell senescence, while restoring mitochondrial autophagy can delay cellular senescence [91]. In a mouse model of DN induced by high glucose levels, the absence of the autophagy receptor optneurin (OPTN) accelerated the senescence of RTEC [57]. Thus, mitochondrial autophagy is indispensable in the context of cellular senescence.
Cell metabolism
While senescent cells may lack proliferative capabilities, their metabolism remains highly active, characterized by an increased ratio of AMP/ATP to ADP/ATP ratio, enhanced glycolysis, and abnormal lipid metabolism [93]. The increase in the metabolic demand of senescent cells can be attributed to factors such as increased cell volume, increased SASP production, elevated ROS levels, and endoplasmic reticulum stress [94]. Both replicative senescence and stress-induced senescence exhibit enhanced glycolysis [95, 96]. Metabolic abnormalities involving malic acid, pyruvic acid, and fatty acids are implicated in cellular senescence [95]. Enhanced glycolysis is particularly necessary in radiation-induced senescence [97]. Disorders in lipid metabolism disorders and lipotoxicity contribute to cellular senescence and the establishment of a senescent microenvironment. Additionally, cellular metabolism regulates DNA damage and repair by balancing the nucleotide pool [98]. Key metabolic pathways, including the tricarboxylic acid (TCA) cycle, glycolysis, fatty acid (FA) synthesis, and nucleotide synthesis, are integral in maintaining cellular homeostasis and promoting DNA repair. Metabolic alterations exert a significant influence on the DDR and DNA repair processes. As a result, cellular metabolism is closely associated with senescence. Moreover, p53 can regulate cellular metabolism by inhibiting glucose uptake and glycolysis, while promoting the TCA cycle, oxidative phosphorylation, and fatty acid oxidation [7, 99]. Another crucial participant in cellular cycle arrest, RB, regulates the production of deoxyribonucleotides by controlling key enzymes through the transcription factor E2F1 [100].
Endoplasmic reticulum stress
Senescence is a persistent pathological process. At the molecular level, senescent cells accumulate abnormal DNA and proteins associated with the endoplasmic reticulum (ER) [96]. Elevated ROS levels trigger ER stress, leading to an imbalance in ER homeostasis. This imbalance results in the accumulation of unfolded proteins within the ER, prompting the activation of the unfolded protein response (UPR) [101]. ER stress can determine cell state or fate, including autophagy, apoptosis, or senescence [102]. A strong correlation exists between ER dysfunction and senescence, as senescent cells exhibit increased UPR [103–106]. Overactivation of ER stress may induce senescence and the SASP [101].
The UPR signaling pathway is activated in response to disruptions in protein homeostasis within the ER and plays a role in various kidney diseases [107]. Previous studies have demonstrated a correlation between ER stress and renal fibrosis [108]. ER stress marker glucose-regulated protein 78 (GRP78) is elevated in patients with DN and shows a positive correlation with senescent markers such as SA-β-gal and p21. This further confirms that advanced glycation end products promote the premature senescence of RTEC by activating ER stress-dependent p21 signaling. However, ER stress inhibitors can suppress premature senescence in RTEC and mitigate renal fibrosis [109, 110].
Epigenetic regulation
Epigenetics is the study of non-genotoxic, reversible, and heritable modifications that influence gene expression without altering DNA sequences [111]. Epigenetic remodeling is an important driver in cellular senescence and senescence-related diseases [112, 113]. Senescence is characterized by changes in gene expression, primarily regulated by dynamic epigenetic mechanisms. Senescent cells often exhibit chromatin remodeling, post-translational modifications of histones (PTMs), DNA methylation, and altered expression of non-coding RNAs [114–116]. To uncover common features of histone epigenetic modifications, chromatin spatial changes, and the significant role of the epigenetic factor KDM4 in senescent cells, researchers have utilized high-throughput epigenetic techniques such as quantitative proteomics, chromatin accessibility sequencing (ATAC-seq), and chromatin immunoprecipitation sequencing (ChIP-seq) [117].
DNA methylation (DNAm) is a relatively stable modification catalyzed by DNA methyltransferase (DNMT) that occurs in cytosine-guanine dinucleotide regions, known as CpG islands, and can be inherited by offspring DNA during replication. DNAm is a crucial epigenetic regulator of cellular senescence [118]. During cellular senescence, DNA methylation levels decrease, serving as a marker of cell senescence. DNA methylation influences gene expression and plays a regulatory role in senescence. For example, high methylation of the anti-senescence factor klotho promoter leads to decreased klotho gene expression, promoting the progression of CKD [119, 120]. DNA methylation is influenced by both exogenous and endogenous factors associated with cellular senescence. Nutritional factors, such as diets lacking folic acid and methyl donors, can lead to DNA hypomethylation. Dnmt1, a DNA methyltransferase, gradually decreases in activity during senescence, which has been associated with replicative senescence of smooth muscle cells and atherosclerosis. In ischemia-reperfusion AKI, Dnmt plays a central role in promoting renal fibrosis and inflammation [121].
Protein translational modifications (PTMs), including acetylation, phosphorylation, succinylation, butyrylation, SUMOylation, and lactylation, play essential roles in regulating protein function and gene transcription, and are closely associated with the epigenetic regulation of senescence and related diseases [122]. PTMs exhibit heterogeneity among different types of senescent cells. A comprehensive proteomic profiling study has revealed that approximately 700 cytoplasmic and nuclear proteins undergo post-translational modifications in senescent human cells [117]. Histone modifications occur in specific gene regions, such as histone H3 lysine, during cellular senescence [123]. In senescent endothelial cells, the levels of histone H3 lysine 4 trimethylation (H3K4me3) and H3K4 methyltransferase Smyd3 increase, promoting the production of the SASP [124]. Different regions of the tau protein have shown phosphorylation and ubiquitination modifications in Alzheimer’s disease [125], with phosphorylation sites being associated with pathological damage. SUMOylation has been linked to neurodegeneration [126]. In senescent kidney disease models, a close relationship between renal function and epigenetics has been observed [127].
Role and mechanisms of RTEC senescence in AKI
RTECs constitute the most abundant cell population in the kidney, accounting for approximately 90% of the renal cortex. RTECs are the most vulnerable cells to hypoxia, proteinuria, toxins, and metabolic disorders, among others [61]. These cells are particularly abundant in mitochondria, especially within the S3 segment of proximal tubules. This abundance of mitochondria makes them highly vulnerable to injuries such as ischemia, hypoxia, and exposure to nephrotoxic drugs. These insults can lead to pathological responses, including cell necrosis, apoptosis, and senescence. Notably, the residual RTECs have an intrinsic repair capacity. They can re-enter the cell cycle from the G0 or G1 phase, undergo mitosis, and regenerate to form new RTECs. This regenerative capability plays a pivotal role in restoring kidney structure and function, ultimately determining the prognosis of renal health [128, 129]. Therefore, RTECs are not only the main instigators of AKI but are also critical in driving kidney repair and influencing the overall prognosis [128].
RTEC senescence
RTECs contain numerous mitochondria that are prone to stress-induced senescence, particularly under certain conditions, such as ischemia and hypoxia [130]. These cells are highly sensitive to oxidative stress, mitochondrial dysfunction, and abnormal energy metabolism following AKI. These factors collectively contribute to cellular senescence and hinder the proliferation of surviving RTECs, thereby inhibiting renal regeneration [131]. In the context of AKI, the binding of CDK with cyclin forms a cyclin-CDK complex that triggers cell-cycle arrest at the G1/S checkpoint, culminating in cellular senescence. Notably, in animal models of AKI caused by various factors, such as ischemia-reperfusion, folic acid, cisplatin, aristolochic acid, and contrast agents, a large number of p16- and p21-positive senescent RTECs has been observed during the early stages of AKI. The accumulation of senescent RTECs is closely associated with interstitial fibrosis and renal dysfunction. Furthermore, the secretion of proinflammatory cytokines such as IL-6 and IL-8 increases after AKI [132, 133]. Single-cell transcriptomic analysis has revealed that Vcam1 + /Ccl2+ RTECs at a late injury stage (28 days) can be distinguished by the activation of NF-κB, TNF, and AP-1 signaling pathways. These cells exhibit secretory phenotypes typical of senescent cells [134]. These findings indicate that senescent RTECs not only inhibit tubular regeneration and repair but also contribute to the progression of AKI to CKD through the SASP. Additionally, senescent cells have the ability to induce senescence in surrounding normal cells via paracrine signaling pathways, thereby significantly impacting the poor prognosis associated with AKI [135, 136].
Numerous studies have shown that RTEC senescence is primarily dependent on the P16Ink4a/Rb and/or P19Arf/P53/P21Cip1 pathway following AKI. Recent studies have uncovered the role of the Wnt9/β-catenin signaling pathway in regulating P16 and P53/P21, leading to cell-cycle arrest and senescence in IRI. The SASP, in turn, promotes fibroblast proliferation and extracellular matrix protein deposition, ultimately culminating in renal fibrosis following AKI [133]. In UIRI mice, BRG1 inhibits autophagy to induce RTEC senescence through the Wnt/β-catenin signaling pathway, ultimately contributing to renal fibrosis [137]. The ectopic expression of Klotho significantly delays fibrotic lesions and activates the Wnt/β-catenin signaling pathway by inhibiting cell senescence [138]. Myd88, a key molecule in the Toll-like receptor/interleukin (IL)-1 receptor signaling, regulates RTEC senescence via Toll-like receptor signaling [133]. Increasing evidence suggests that mitochondrial dysfunction induces cell senescence, and senescent cells accumulate numerous dysfunctional mitochondria. Mitochondrial dysfunction exacerbates RTEC senescence by activating ROS and disturbing energy metabolism. Moreover, SIRT1, vascular non-inflammatory molecule 1 (Vanin-1, VNN1), and other factors are closely associated with RTEC senescence in AKI, implying that RTEC senescence is the result of multiple biological mechanisms working in concert following AKI.
Intervening RTEC senescence in AKI
Inhibiting RTEC senescence
Cellular senescence is a crucial event in the progression of AKI, and several strategies have been explored to delay or mitigate cellular senescence (Fig. 2), including calorie restriction and increased exercise. These methods extend the lifespan and reduce oxidative stress levels and age-related inflammation [139, 140]. Dietary interventions, including the consumption of nutrients such as betaine, choline, and folic acid, can influence age-related diseases by modulating epigenetic processes and the gut microbiota. Conversely, excessive glucose intake has been observed to reduce microbial diversity in animal models, contributing to the advancement of age-related diseases [141]. Furthermore, the quality of sleep and regular exercise have been shown to influence senescence and the levels of SASP [142]. Strategies to neutralize SASP have become vital for inhibiting senescence, encompassing the inhibition of signal cascades associated with SASP in senescent cells, interference with SASP secretion, or inhibition of the components comprising the SASP [143]. Several anti-senescence drugs (Fig. 2), including metformin, rapamycin, sirtuin activators, and NAD+ precursors, have been developed in animal models. Metformin and rapamycin, for instance, activate autophagy, improve mitochondrial function, regulate SASP secretion, inhibit mTOR, and contribute to lifespan extension [144, 145]. Resveratrol activates SIRT, thereby exerting antioxidant and anti-inflammatory effects. Other substances, such as melatonin, estrogen, and estradiol, are known to inhibit SASP production and regulate the release of inflammatory factors [146–148]. Recent studies have unveiled the anti-senescence properties of stem cells and their extracellular vesicles, with potential applications in retarding cellular senescence and the progression of senescence-related diseases. Multiple studies have explored these effects in the context of heart, blood vessels, cartilage, skin, and kidney damage. Furthermore, interventions targeting hypertension and the renin-angiotensin-aldosterone system have been found to reduce the number of senescent cells, thereby delaying renal senescence.
Fig. 2. Senescence interventions.
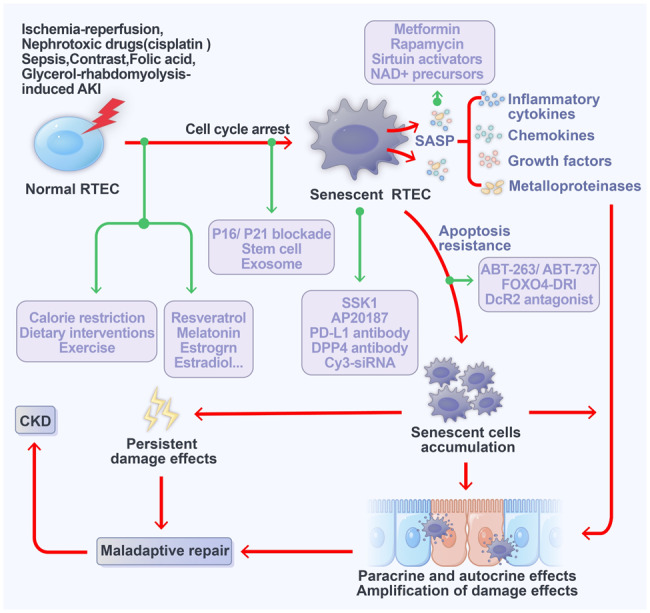
Inhibiting cell senescence, targeted clearance of senescent cells, promoting apoptosis of senescent cells, and inhibiting SASP release could be potential methods for improving renal prognosis following AKI.
When considering the mechanisms of cell senescence in AKI, one potential intervention target is cell-cycle arrest (Table 1). For example, in IRI models, p21 knockout has been shown to promote the proliferation of RTECs, but it had no discernible effect on renal function or mortality rates [149]. In a cisplatin-induced AKI model, p21 gene knockout led to increased necrosis and apoptosis of RTECs and exacerbated renal tissue damage [150]. Conversely, p16 gene knockout has been found to promote RTEC proliferation and facilitate the recovery of renal function after AKI, thereby ameliorating renal tubular atrophy, interstitial fibrosis, and collagen deposition [151, 152]. Furthermore, Smad7 has been shown to improve G1 cell-cycle arrest via the TGF-β/Smad3-p21/p27 pathway, thus inhibiting AKI progression [153]. Mesenchymal stem cells (MSCs) and their extracellular vesicles, exosomes, also play protective roles in mitigating RTEC senescence. For instance, human umbilical cord-derived MSCs have been demonstrated to protect rat kidneys from senescence induced by AKI [154]. Moreover, Klotho gene-modified bone marrow-derived MSCs promote RTEC proliferation and repair by inhibiting the Wnt/β-catenin pathway in AKI [155]. Extracellular vesicles containing miRNA let-7b-5p have been found to alleviate RTEC senescence by inhibiting p53, thereby delaying the progression of AKI to CKD [156]. G protein-coupled receptor (GPCR) family member free fatty acid receptor 4 (FFAR4) is a potential drug target against AKI. The expression of FFAR4 was abnormally decreased in RTECs of cisplatin, CLP, and IRI-induced AKI. Activation of FFAR4 by agonist TUG891 alleviated AKI by regulating cellular senescence [157]. A ligand-activated transcription factor aryl hydrocarbon receptor (AhR) was abnormally increased in the kidneys of cisplatin-induced AKI mice. Notably, inhibition of AhR could improve cellular senescence of injured kidneys, which was indicated by SA-β-gal activity, biomarker p53, p21, p16 expression, and SASP [158].
Table 1.
Effects of inhibiting cell senescence on renal outcomes in AKI models.
| Animals | Renal model | Mechanism of inhibiting cell senescence | Evidence of inhibiting cell senescence | Renal outcome | Reference |
|---|---|---|---|---|---|
| p16 KO mice | UUO | Impaired cell-cycle arrest |
SA-β-gal positivity ↓ SASP (IL-6, TGF-β) ↑ PCNA ↑ |
Interstitial fibrosis ↑ Blood urea nitrogen ↑ |
[159] |
| p16 KO mice | Renal transplant | Impaired cell-cycle arrest | Ki-67 ↑ |
Interstitial fibrosis and tubular Atrophy ↓ Creatinine clearances ↑ Renal survival ↑ |
[151] |
| p16 KO mice | Glycerol-rhabdomyolysis-induced AKI |
Impaired cell-cycle arrest Inhibiting NF-κB Downregulating ROS signaling |
SASP (IL-1β, IL-6, TNF-a) ↓ ROS ↑ |
Renal damage ↓ Serum urea ↓ Serum creatinine ↓ |
[152] |
| p16 /p19 double KO mice | Renal IRI | Impaired cell-cycle arrest |
p16 ↓ p19 ↓ Ki-67 ↑ |
Kidney regeneration ↑ Microvascular repair ↑ Serum creatinine ↓ |
[132] |
| p21 KO mice | Renal IRI | Impaired cell-cycle arrest | PCNA ↑ |
Mortality ↑ Blood urea nitrogen ↑ |
[149] |
| p21 KO mice | Cisplatin-induced AKI | Impaired cell-cycle arrest |
Brdu ↑ PCNA ↑ |
Renal damage ↑ Blood urea nitrogen ↑ Mortality ↑ |
[150] |
| p21 KO mice | Renal IRI | Impaired transient cell-cycle arrest | Ki-67↑ |
Renal survival ↓ Blood urea nitrogen ↑ Renal damage and fibrosis ↑ |
[160] |
| Renal ischemic-preconditioned IRI | Impaired transient cell-cycle arrest |
Ki-67↓ MCP-1, IL-6↑ Oxidative stress↑ |
Targeted clearance of senescent RTECs
The targeted clearance of senescent cells represents a significant advancement in the prevention and treatment of age-related diseases (Fig. 2) [7]. Current methods for the targeted clearance of senescent cells primarily encompass transgenic approaches, the use of specific drugs or interference peptides, and immune cell-targeted clearance. β-gal is widely recognized as a biomarker for cellular senescence and is often the focus of drug-based interventions. SSK1 is specifically activated by β-galactosidase, enabling the targeted elimination of senescent cells. This highlights the utility of lysosomal β-gal as an effective tool for the selective removal of senescent cells [159]. Programmed cell death ligand 1 (PD-L1) is expressed and accumulates in senescent cells and is closely linked to the SASP. The use of PD-1 antibodies has been shown to reduce the number of p16-positive cells in the body and improve senescence-related phenotypes [160]. A variety of strategies for the targeted clearance of senescent cells is presented in Table 2. In a mouse model of premature senescence, which presented numerous p16-positive senescent cells in the kidneys and heart, transgenic technology facilitated the selective elimination of p16-positive senescent cells using AP20187, which targets the senescence marker p16. This approach significantly improves kidney fibrosis and myocardial hypertrophy, ultimately extending the lifespan of the mice [161, 162]. In a mouse model of chronic ischemic renal injury, intraperitoneal injection of AP20187 significantly inhibited renal interstitial fibrosis, improved renal function, and increased renal tissue oxygen content by specifically targeting the clearance of p16-positive senescent cells [163]. These findings indicate that the targeted clearance of p16-positive senescent cells not only alleviates kidney damage and enhances renal function but also has protective effects on other organs. Furthermore, ganciclovir has been found to induce apoptosis in p16-positive senescent cells in p16-3MR transgenic mice, resulting in improved renal fibrosis and function, and the inhibition of SASP production [161]. However, it is important to note that p16 is not a specific marker for senescent RTECs. p16 is also expressed in all senescent cells within the renal parenchyma, including vascular endothelial cells, podocytes, and interstitial fibroblasts. Targeted clearance of p16-positive senescent cells may lead to structural damage and renal dysfunction. Previous studies have utilized proteomic screening to identify the specific marker DPP4 on the membranes of senescent fibroblasts. This marker inhibits the immune clearance ability of NK cells. Treatment with a DPP4 antibody has been shown to promote the clearance of senescent NK cells [161, 164]. Furthermore, proximal RTECs are the primary site for renal uptake of siRNA. Following ischemic injury, intravenous administration of Cy3-siRNA rapidly delivers the substance to proximal RTECs, significantly reducing p53 expression. This approach effectively mitigates damage to tubular cells [165]. Nano Drug Delivery (Nano-DDS) represent a highly promising drug delivery strategy that has been extensively studied and documented. This nanotechnology enables efficient drug targeting to diseased tissues, resulting in reduced dosage, minimal damage to healthy cells, and enhanced drug bioavailability. Nanotechnology offers a new therapeutic approach for kidney diseases, including AKI [166]. Research has shown that medium-sized nanoparticles (400 nm) can selectively target RTECs [167]. Nanotechnology enables the sustained or controlled release of drugs within the body. Importantly, the kidney exhibits 26–94 times higher uptake efficiency of mid-sized nanoparticles compared to other organs [168]. Thus, nanotechnology holds promise as a novel approach for the targeted removal of senescent RETCs. These studies suggest that the targeted elimination of senescent cells has the potential to serve as a new strategy for preventing and treating the progression of kidney diseases.
Table 2.
Effects of targeted clearance of senescent cell on renal outcomes in renal models.
| Animals | Renal model | Methods for clearing senescent cell | Evidence of senescent cell clearance | Renal outcome | Reference |
|---|---|---|---|---|---|
| INK-ATTAC mice | Natural aging | AP20187 administration to deplete p16- positive cells | SA-β-gal positivity ↓ |
Glomerulosclerosis ↓ Blood urea nitrogen levels ↓ |
[50] |
| INK-ATTAC mice | Renal artery stenosis-induced ischemic Nephropathy | AP20187 administration to deplete p16- positive cells |
SA-β-gal positivity ↓ P16 positivity ↓ SASP (IL1a, IL6, Ccl2, Mmp3, TNF-a) ↓ |
Interstitial fibrosis ↓ Renal perfusion ↑ |
[163] |
| XpdTTD/TTD Mice | Fast-aging | FOXO4-DRI induces targeted apoptosis of senescent cells |
Lamin B1 ↑ SASP (IL1a, IL1β, IL6) ↓ |
Serum urea ↓ Serum creatinine ↓ |
[170] |
| p16-3MR | Natural-aging | Ganciclovir induces p16-positive-restricted cell apoptosis |
Lamin B1 ↑ SASP (IL6) ↓ |
Serum urea ↓ Serum creatinine ↓ |
[170] |
| p16-3MR | Folic acid-induced AKI | Ganciclovir induces p16-positive senescent cell apoptosis |
SA-β-gal positivity ↓ Lamin B1↑ Ki-67 ↑ |
Interstitial fibrosis ↓ No effects on tubular damage |
[133] |
| Wild-type mice (C57BL/6J) | Folic acid-induced AKI | FOXO4-DRI induces apoptosis of senescent cells |
SA-β-gal positivity ↓ Lamin B1↑ Ki-67 ↑ |
No effects on kidney damage and interstitial fibrosis | [133] |
Promoting senescent RTECs apoptosis
Resistance to apoptosis is one of the main characteristics of senescent cells. Recent studies have revealed that Bcl-2/Bcl-xL specific inhibitors (Fig. 2), such as ABT-263/ABT-737, can selectively induce apoptosis in senescent cells, thereby contributing to the rejuvenation of stem cells in aged tissues [22, 169]. ABT-263 has progressed to clinical trials for the treatment of hematological malignancies and solid organ tumors. The synthesis of a small-molecule interference peptide known as FOXO4-DRI is aimed at disrupting the interaction between FOXO4 and the p53 apoptotic signaling pathway. This selective induction of apoptosis in senescent cells by FOXO4-DRI has shown promise in improving renal function, hair density, and the maintenance of tissue and organ homeostasis [170]. However, it is important to note that in the early stages of FA-induced AKI, administering FOXO4-DRI selectively induces senescence in RTECs, thereby improving renal inflammation and interstitial fibrosis [133]. Recent studies have found that senescent RTECs and fibroblasts express the decoy receptor DcR2, which is closely associated with the resistance to apoptosis observed in senescent cells [171]. Elevated DcR2 expression in fibroblasts during senescence serves to antagonize immune cell clearance. Conversely, downregulation of DcR2 expression promotes NK cell clearance of senescent cells, thereby inhibiting liver fibrosis [171]. In the context of senescent RTECs, inhibiting DcR2 expression has been observed to promote apoptosis, reduce SASP secretion, and inhibit renal interstitial fibrosis [24]. These findings suggest that promoting apoptosis in senescent RTEC may represent an effective approach for improving AKI prognosis.
Summary
RTEC senescence is a key cellular event in the progression of AKI. It not only hinders the proliferation and regeneration of RTEC but also contributes to the release of SASP, which promotes inflammation and fibrosis. This can be referred to as the “Quagmire effect”. The unique SASP and anti-apoptotic capabilities of senescent cells make them promote the transformation of surrounding cells into senescent cells. Senescent cells continuously accumulate, forming something akin to a quagmire, and continually dragging normal cells to expand their size. As a result, a large number of senescent cells can lead to the occurrence of chronic inflammation, with some chronic inflammation spreading to the entire kidney, ultimately leading to kidney dysfunction.
Currently, strategies aimed at inhibiting RTEC senescence, clearing senescent RTECs, and promoting apoptosis of senescent cells show promise in improving AKI prognosis. These approaches hold substantial potential for application in clinical settings. However, the challenge of achieving precise and targeted intervention for RTEC senescence to enhance renal outcomes in AKI remains an unresolved issue. Further exploration of senescent cell-specific biomarkers and underlying mechanisms may yield novel insights and strategies to promote the regeneration and repair of AKI.
Author contributions
Juan Chen wrote the manuscript, Huhai Zhang and Xiangling Yi drew the figures, Qian Dou, Xin Yang and Yani He collected relevant references and participated in the discussion. Jia Chen and Kehong Chen designed this review and revised the manuscript. All authors have contributed to the manuscript and approved the submitted version.
Funding
This study was supported by the Open Project of the State Key Laboratory of Trauma, Burn and Combined Injury Third Military Medical University (No. SKLKF202202); Chongqing Natural Science Foundation (No. CSTB2022NSCQ-MSX0860 and No.2023NSCQ-MSX2510), National Natural Science Foundation of China (No. 82270768).
Competing interests
All authors declare no conflict of interest. They declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Jia Chen, Email: chenjiasnk@163.com.
Kehong Chen, Email: kehong_chen@126.com.
References
- 1.Wang Y, Zhang M, Bi R, Su Y, Quan F, Lin Y, et al. ACSL4 deficiency confers protection against ferroptosis-mediated acute kidney injury. Redox Biol. 2022;51:102262. doi: 10.1016/j.redox.2022.102262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Gameiro J, Jorge S, Lopes JA. Acute kidney injury. Ann Intern Med. 2018;168:836–7. doi: 10.7326/l18-0015. [DOI] [PubMed] [Google Scholar]
- 3.Doke T, Susztak K. The multifaceted role of kidney tubule mitochondrial dysfunction in kidney disease development. Trends Cell Biol. 2022;32:841–53. doi: 10.1016/j.tcb.2022.03.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Song Q, Hou Y, Zhang Y, Liu J, Wang Y, Fu J, et al. Integrated multi-omics approach revealed cellular senescence landscape. Nucleic Acids Res. 2022;50:10947–63. doi: 10.1093/nar/gkac885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Villiard É, Denis JF, Hashemi FS, Igelmann S, Ferbeyre G, Roy S. Senescence gives insights into the morphogenetic evolution of anamniotes. Biol Open. 2017;6:891–6. doi: 10.1242/bio.025809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ghanam AR, Xu Q, Ke S, Azhar M, Cheng Q, Song X. Shining the light on senescence associated LncRNAs. Aging Dis. 2017;8:149–61. doi: 10.14336/ad.2016.0810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hernandez-Segura A, Nehme J, Demaria M. Hallmarks of cellular senescence. Trends Cell Biol. 2018;28:436–53. doi: 10.1016/j.tcb.2018.02.001. [DOI] [PubMed] [Google Scholar]
- 8.Deolal P, Mishra K. Regulation of diverse nuclear shapes: pathways working independently, together. Commun Integr Biol. 2021;14:158–75. doi: 10.1080/19420889.2021.1939942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Bourdens M, Jeanson Y, Taurand M, Juin N, Carrière A, Clément F, et al. Short exposure to cold atmospheric plasma induces senescence in human skin fibroblasts and adipose mesenchymal stromal cells. Sci Rep. 2019;9:8671. doi: 10.1038/s41598-019-45191-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Gire V, Dulic V. Senescence from G2 arrest, revisited. Cell Cycle. 2015;14:297–304. doi: 10.1080/15384101.2014.1000134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hume S, Dianov GL, Ramadan K. A unified model for the G1/S cell cycle transition. Nucleic Acids Res. 2020;48:12483–501. doi: 10.1093/nar/gkaa1002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Moonen L, D’Haese PC, Vervaet BA. epithelial cell cycle behaviour in the injured kidney. Int J Mol Sci. 19. 10.3390/ijms19072038 (2018). [DOI] [PMC free article] [PubMed]
- 13.Kastenhuber ER, Lowe SW. Putting p53 in context. Cell. 2017;170:1062–78. doi: 10.1016/j.cell.2017.08.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Hernandez-Segura A, de Jong TV, Melov S, Guryev V, Campisi J, Demaria M. Unmasking transcriptional heterogeneity in senescent cells. Curr Biol. 2017;27:2652–60.e2654. doi: 10.1016/j.cub.2017.07.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Acosta JC, Banito A, Wuestefeld T, Georgilis A, Janich P, Morton JP, et al. A complex secretory program orchestrated by the inflammasome controls paracrine senescence. Nat Cell Biol. 2013;15:978–90. doi: 10.1038/ncb2784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Xu P, Wang M, Song WM, Wang Q, Yuan GC, Sudmant PH, et al. The landscape of human tissue and cell type specific expression and co-regulation of senescence genes. Mol Neurodegener. 2022;17:5. doi: 10.1186/s13024-021-00507-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Bao H, Cao J, Chen M, Chen M, Chen W, Chen X, et al. Biomarkers of aging. Sci China Life Sci. 2023;66:893–1066. doi: 10.1007/s11427-023-2305-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Childs BG, Baker DJ, Kirkland JL, Campisi J, van Deursen JM. Senescence and apoptosis: dueling or complementary cell fates? EMBO Rep. 2014;15:1139–53. doi: 10.15252/embr.201439245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Deryabin PI, Shatrova AN, Borodkina AV. Apoptosis resistance of senescent cells is an intrinsic barrier for senolysis induced by cardiac glycosides. Cell Mol Life Sci. 2021;78:7757–76. doi: 10.1007/s00018-021-03980-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Fuhrmann-Stroissnigg H, Ling YY, Zhao J, McGowan SJ, Zhu Y, Brooks RW, et al. Identification of HSP90 inhibitors as a novel class of senolytics. Nat Commun. 2017;8:422. doi: 10.1038/s41467-017-00314-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Hu L, Li H, Zi M, Li W, Liu J, Yang Y, et al. Why senescent cells are resistant to apoptosis: an insight for senolytic development. Front Cell Dev Biol. 2022;10:822816. doi: 10.3389/fcell.2022.822816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Yosef R, Pilpel N, Tokarsky-Amiel R, Biran A, Ovadya Y, Cohen S, et al. Directed elimination of senescent cells by inhibition of BCL-W and BCL-XL. Nat Commun. 2016;7:11190.. doi: 10.1038/ncomms11190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Chen J, Hu W, Xiao F, Lin L, Chen K, Wang L, et al. DCR2, a cellular senescent molecule, is a novel marker for assessing tubulointerstitial fibrosis in patients with immunoglobulin a nephropathy. Kidney Blood Press Res. 2019;44:1063–74. doi: 10.1159/000502233. [DOI] [PubMed] [Google Scholar]
- 24.Chen J, Chen KH, Wang LM, Luo J, Zheng QY, He YN. Decoy receptor 2 mediates the apoptosis-resistant phenotype of senescent renal tubular cells and accelerates renal fibrosis in diabetic nephropathy. Cell Death Dis. 2022;13:522.. doi: 10.1038/s41419-022-04972-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Yosef R, Pilpel N, Papismadov N, Gal H, Ovadya Y, Vadai E, et al. p21 maintains senescent cell viability under persistent DNA damage response by restraining JNK and caspase signaling. EMBO J. 2017;36:2280–95. doi: 10.15252/embj.201695553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Johmura Y, Yamanaka T, Omori S, Wang TW, Sugiura Y, Matsumoto M, et al. Senolysis by glutaminolysis inhibition ameliorates various age-associated disorders. Science. 2021;371:265–70. doi: 10.1126/science.abb5916. [DOI] [PubMed] [Google Scholar]
- 27.Calcinotto A, Kohli J, Zagato E, Pellegrini L, Demaria M, Alimonti A. Cellular senescence: aging, cancer, and injury. Physiol Rev. 2019;99:1047–78. doi: 10.1152/physrev.00020.2018. [DOI] [PubMed] [Google Scholar]
- 28.Storer M, Mas A, Robert-Moreno A, Pecoraro M, Ortells MC, Di Giacomo V, et al. Senescence is a developmental mechanism that contributes to embryonic growth and patterning. Cell. 2013;155:1119–30. doi: 10.1016/j.cell.2013.10.041. [DOI] [PubMed] [Google Scholar]
- 29.Da Silva-Alvarez S, Picallos-Rabina P, Antelo-Iglesias L, Triana-Martinez F, Barreiro-Iglesias A, Sanchez L, et al. The development of cell senescence. Exp Gerontol. 2019;128:110742. doi: 10.1016/j.exger.2019.110742. [DOI] [PubMed] [Google Scholar]
- 30.Davaapil H, Brockes JP, Yun MH. Conserved and novel functions of programmed cellular senescence during vertebrate development. Development. 2017;144:106–14. doi: 10.1242/dev.138222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Gibaja A, Aburto MR, Pulido S, Collado M, Hurle JM, Varela-Nieto I, et al. TGFbeta2-induced senescence during early inner ear development. Sci Rep. 2019;9:5912.. doi: 10.1038/s41598-019-42040-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Walter LD, Orton JL, Fong EHH, Maymi VI, Rudd BD, Elisseeff JH, et al. Single-cell transcriptomic analysis of skeletal muscle regeneration across mouse lifespan identifies altered stem cell states associated with senescence. bioRxiv. 10.1101/2023.05.25.542370 (2023).
- 33.Demaria M, Ohtani N, Youssef SA, Rodier F, Toussaint W, Mitchell JR, et al. An essential role for senescent cells in optimal wound healing through secretion of PDGF-AA. Dev Cell. 2014;31:722–33. doi: 10.1016/j.devcel.2014.11.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Elder SS, Emmerson E. Senescent cells and macrophages: key players for regeneration. Open Biol. 2020;10:200309. doi: 10.1098/rsob.200309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Osorio JM, Espinoza-Pérez C, Rimassa-Taré C, Machuca V, Bustos JO, Vallejos M, et al. Senescent cardiac fibroblasts: a key role in cardiac fibrosis. Biochim Biophys Acta Mol Basis Dis. 2023;1869:166642. doi: 10.1016/j.bbadis.2023.166642. [DOI] [PubMed] [Google Scholar]
- 36.Krizhanovsky V, Yon M, Dickins RA, Hearn S, Simon J, Miething C, et al. Senescence of activated stellate cells limits liver fibrosis. Cell. 2008;134:657–67. doi: 10.1016/j.cell.2008.06.049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Sturmlechner I, Durik M, Sieben CJ, Baker DJ, van Deursen JM. Cellular senescence in renal ageing and disease. Nat Rev Nephrol. 2017;13:77–89. doi: 10.1038/nrneph.2016.183. [DOI] [PubMed] [Google Scholar]
- 38.Wang L, Lankhorst L, Bernards R. Exploiting senescence for the treatment of cancer. Nat Rev Cancer. 2022;22:340–55. doi: 10.1038/s41568-022-00450-9. [DOI] [PubMed] [Google Scholar]
- 39.Baker DJ, Wijshake T, Tchkonia T, LeBrasseur NK, Childs BG, van de Sluis B, et al. Clearance of p16Ink4a-positive senescent cells delays ageing-associated disorders. Nature. 2011;479:232–6. doi: 10.1038/nature10600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Strzyz P. Bend it like glycocalyx. Nat Rev Mol Cell Biol. 2019;20:388. doi: 10.1038/s41580-019-0142-2. [DOI] [PubMed] [Google Scholar]
- 41.Musi N, Valentine JM, Sickora KR, Baeuerle E, Thompson CS, Shen Q, et al. Tau protein aggregation is associated with cellular senescence in the brain. Aging Cell. 2018;17:e12840. doi: 10.1111/acel.12840. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Bussian TJ, Aziz A, Meyer CF, Swenson BL, van Deursen JM, Baker DJ. Clearance of senescent glial cells prevents tau-dependent pathology and cognitive decline. Nature. 2018;562:578–82. doi: 10.1038/s41586-018-0543-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Salminen A, Ojala J, Kaarniranta K, Haapasalo A, Hiltunen M, Soininen H. Astrocytes in the aging brain express characteristics of senescence-associated secretory phenotype. Eur J Neurosci. 2011;34:3–11. doi: 10.1111/j.1460-9568.2011.07738.x. [DOI] [PubMed] [Google Scholar]
- 44.Wang X, Ma L, Pei X, Wang H, Tang X, Pei JF, et al. Comprehensive assessment of cellular senescence in the tumor microenvironment. Brief Bioinform. 23. 10.1093/bib/bbac118 (2022). [DOI] [PMC free article] [PubMed]
- 45.Takasugi M, Yoshida Y, Ohtani N. Cellular senescence and the tumour microenvironment. Mol Oncol. 2022;16:3333–51. doi: 10.1002/1878-0261.13268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Liu D, Hornsby PJ. Senescent human fibroblasts increase the early growth of xenograft tumors via matrix metalloproteinase secretion. Cancer Res. 2007;67:3117–26. doi: 10.1158/0008-5472.Can-06-3452. [DOI] [PubMed] [Google Scholar]
- 47.Coppé JP, Kauser K, Campisi J, Beauséjour CM. Secretion of vascular endothelial growth factor by primary human fibroblasts at senescence. J Biol Chem. 2006;281:29568–74. doi: 10.1074/jbc.M603307200. [DOI] [PubMed] [Google Scholar]
- 48.Laberge RM, Awad P, Campisi J, Desprez PY. Epithelial-mesenchymal transition induced by senescent fibroblasts. Cancer Microenviron. 2012;5:39–44. doi: 10.1007/s12307-011-0069-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Sun Y, Campisi J, Higano C, Beer TM, Porter P, Coleman I, et al. Treatment-induced damage to the tumor microenvironment promotes prostate cancer therapy resistance through WNT16B. Nat Med. 2012;18:1359–68. doi: 10.1038/nm.2890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Baker DJ, Childs BG, Durik M, Wijers ME, Sieben CJ, Zhong J, et al. Naturally occurring p16(Ink4a)-positive cells shorten healthy lifespan. Nature. 2016;530:184–9. doi: 10.1038/nature16932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Nowotny K, Jung T, Höhn A, Weber D, Grune T. Advanced glycation end products and oxidative stress in type 2 diabetes mellitus. Biomolecules. 2015;5:194–222. doi: 10.3390/biom5010194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Bian X, Griffin TP, Zhu X, Islam MN, Conley SM, Eirin A, et al. Senescence marker activin A is increased in human diabetic kidney disease: association with kidney function and potential implications for therapy. BMJ Open Diabetes Res Care. 2019;7:e000720. doi: 10.1136/bmjdrc-2019-000720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Sone H, Kagawa Y. Pancreatic beta cell senescence contributes to the pathogenesis of type 2 diabetes in high-fat diet-induced diabetic mice. Diabetologia. 2005;48:58–67. doi: 10.1007/s00125-004-1605-2. [DOI] [PubMed] [Google Scholar]
- 54.Huang W, Hickson LJ, Eirin A, Kirkland JL, Lerman LO. Cellular senescence: the good, the bad and the unknown. Nat Rev Nephrol. 2022;18:611–27. doi: 10.1038/s41581-022-00601-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Kitada K, Nakano D, Ohsaki H, Hitomi H, Minamino T, Yatabe J, et al. Hyperglycemia causes cellular senescence via a SGLT2- and p21-dependent pathway in proximal tubules in the early stage of diabetic nephropathy. J Diabetes Complications. 2014;28:604–11. doi: 10.1016/j.jdiacomp.2014.05.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Tung CW, Hsu YC, Shih YH, Chang PJ, Lin CL. Glomerular mesangial cell and podocyte injuries in diabetic nephropathy. Nephrol (Carlton) 2018;23:32–37. doi: 10.1111/nep.13451. [DOI] [PubMed] [Google Scholar]
- 57.Chen K, Dai H, Yuan J, Chen J, Lin L, Zhang W, et al. Optineurin-mediated mitophagy protects renal tubular epithelial cells against accelerated senescence in diabetic nephropathy. Cell Death Dis. 2018;9:105.. doi: 10.1038/s41419-017-0127-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Chen K, Chen J, Wang L, Yang J, Xiao F, Wang X, et al. Parkin ubiquitinates GATA4 and attenuates the GATA4/GAS1 signaling and detrimental effects on diabetic nephropathy. FASEB J. 2020;34:8858–75. doi: 10.1096/fj.202000053R. [DOI] [PubMed] [Google Scholar]
- 59.Liu J, Yang JR, He YN, Cai GY, Zhang JG, Lin LR, et al. Accelerated senescence of renal tubular epithelial cells is associated with disease progression of patients with immunoglobulin A (IgA) nephropathy. Transl Res. 2012;159:454–63. doi: 10.1016/j.trsl.2011.11.008. [DOI] [PubMed] [Google Scholar]
- 60.McCarthy CG, Wenceslau CF, Webb RC, Joe B. Novel contributors and mechanisms of cellular senescence in hypertension-associated premature vascular aging. Am J Hypertens. 2019;32:709–19. doi: 10.1093/ajh/hpz052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Li Y, Lerman LO. Cellular senescence: a new player in kidney injury. Hypertension. 2020;76:1069–75. doi: 10.1161/hypertensionaha.120.14594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Westhoff JH, Schildhorn C, Jacobi C, Hömme M, Hartner A, Braun H, et al. Telomere shortening reduces regenerative capacity after acute kidney injury. J Am Soc Nephrol. 2010;21:327–36. doi: 10.1681/asn.2009010072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Kimura T, Shiizaki K, Akimoto T, Shinzato T, Shimizu T, Kurosawa A, et al. The impact of preserved Klotho gene expression on antioxidative stress activity in healthy kidney. Am J Physiol Ren Physiol. 2018;315:F345–52. doi: 10.1152/ajprenal.00486.2017. [DOI] [PubMed] [Google Scholar]
- 64.Li C, Shen Y, Huang L, Liu C, Wang J. Senolytic therapy ameliorates renal fibrosis postacute kidney injury by alleviating renal senescence. FASEB J. 2021;35:e21229. doi: 10.1096/fj.202001855RR. [DOI] [PubMed] [Google Scholar]
- 65.Gorenne I, Kavurma M, Scott S, Bennett M. Vascular smooth muscle cell senescence in atherosclerosis. Cardiovasc Res. 2006;72:9–17. doi: 10.1016/j.cardiores.2006.06.004. [DOI] [PubMed] [Google Scholar]
- 66.Farr JN, Khosla S. Cellular senescence in bone. Bone. 2019;121:121–33. doi: 10.1016/j.bone.2019.01.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Coryell PR, Diekman BO, Loeser RF. Mechanisms and therapeutic implications of cellular senescence in osteoarthritis. Nat Rev Rheumatol. 2021;17:47–57. doi: 10.1038/s41584-020-00533-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.López-Otín C, Blasco MA, Partridge L, Serrano M, Kroemer G. Hallmarks of aging: An expanding universe. Cell. 2023;186:243–78. doi: 10.1016/j.cell.2022.11.001. [DOI] [PubMed] [Google Scholar]
- 69.Rayego-Mateos S, Marquez-Expósito L, Rodrigues-Diez R, Sanz AB, Guiteras R, Doladé N, et al. Molecular mechanisms of kidney injury and repair. Int J Mol Sci. 23. 10.3390/ijms23031542 (2022). [DOI] [PMC free article] [PubMed]
- 70.Palm W, de Lange T. How shelterin protects mammalian telomeres. Annu Rev Genet. 2008;42:301–34. doi: 10.1146/annurev.genet.41.110306.130350. [DOI] [PubMed] [Google Scholar]
- 71.de Lange T. Shelterin: the protein complex that shapes and safeguards human telomeres. Genes Dev. 2005;19:2100–10. doi: 10.1101/gad.1346005. [DOI] [PubMed] [Google Scholar]
- 72.Denchi EL, de Lange T. Protection of telomeres through independent control of ATM and ATR by TRF2 and POT1. Nature. 2007;448:1068–71. doi: 10.1038/nature06065. [DOI] [PubMed] [Google Scholar]
- 73.Jaskelioff M, Muller FL, Paik JH, Thomas E, Jiang S, Adams AC, et al. Telomerase reactivation reverses tissue degeneration in aged telomerase-deficient mice. Nature. 2011;469:102–6. doi: 10.1038/nature09603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Gatica D, Lahiri V, Klionsky DJ. Cargo recognition and degradation by selective autophagy. Nat Cell Biol. 2018;20:233–42. doi: 10.1038/s41556-018-0037-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Hansen M, Rubinsztein DC, Walker DW. Autophagy as a promoter of longevity: insights from model organisms. Nat Rev Mol Cell Biol. 2018;19:579–93. doi: 10.1038/s41580-018-0033-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Leidal AM, Levine B, Debnath J. Autophagy and the cell biology of age-related disease. Nat Cell Biol. 2018;20:1338–48. doi: 10.1038/s41556-018-0235-8. [DOI] [PubMed] [Google Scholar]
- 77.Mortensen M, Ferguson DJ, Edelmann M, Kessler B, Morten KJ, Komatsu M, et al. Loss of autophagy in erythroid cells leads to defective removal of mitochondria and severe anemia in vivo. Proc Natl Acad Sci USA. 2010;107:832–7. doi: 10.1073/pnas.0913170107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Zhong X, He J, Zhang X, Li C, Tian X, Xia W, et al. UCP2 alleviates tubular epithelial cell apoptosis in lipopolysaccharide-induced acute kidney injury by decreasing ROS production. Biomed Pharmacother. 2019;115:108914. doi: 10.1016/j.biopha.2019.108914. [DOI] [PubMed] [Google Scholar]
- 79.Yuan Y, Zheng Y, Zhang X, Chen Y, Wu X, Wu J, et al. BNIP3L/NIX-mediated mitophagy protects against ischemic brain injury independent of PARK2. Autophagy. 2017;13:1754–66. doi: 10.1080/15548627.2017.1357792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Zhang W, Ren H, Xu C, Zhu C, Wu H, Liu D, et al. Hypoxic mitophagy regulates mitochondrial quality and platelet activation and determines severity of I/R heart injury. Elife. 5. 10.7554/eLife.21407 (2016). [DOI] [PMC free article] [PubMed]
- 81.Tang C, Han H, Yan M, Zhu S, Liu J, Liu Z, et al. PINK1-PRKN/PARK2 pathway of mitophagy is activated to protect against renal ischemia-reperfusion injury. Autophagy. 2018;14:880–97. doi: 10.1080/15548627.2017.1405880. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.García-Prat L, Martínez-Vicente M, Perdiguero E, Ortet L, Rodríguez-Ubreva J, Rebollo E, et al. Autophagy maintains stemness by preventing senescence. Nature. 2016;529:37–42. doi: 10.1038/nature16187. [DOI] [PubMed] [Google Scholar]
- 83.Korolchuk VI, Miwa S, Carroll B, von Zglinicki T. Mitochondria in cell senescence: is mitophagy the weakest link. EBioMedicine. 2017;21:7–13. doi: 10.1016/j.ebiom.2017.03.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Correia-Melo C, Marques FD, Anderson R, Hewitt G, Hewitt R, Cole J, et al. Mitochondria are required for pro-ageing features of the senescent phenotype. EMBO J. 2016;35:724–42. doi: 10.15252/embj.201592862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Ghosh-Choudhary SK, Liu J, Finkel T. The role of mitochondria in cellular senescence. FASEB J. 2021;35:e21991. doi: 10.1096/fj.202101462R. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Miwa S, Jow H, Baty K, Johnson A, Czapiewski R, Saretzki G, et al. Low abundance of the matrix arm of complex I in mitochondria predicts longevity in mice. Nat Commun. 2014;5:3837.. doi: 10.1038/ncomms4837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Passos JF, Nelson G, Wang C, Richter T, Simillion C, Proctor CJ, et al. Feedback between p21 and reactive oxygen production is necessary for cell senescence. Mol Syst Biol. 2010;6:347. doi: 10.1038/msb.2010.5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Tsagarakis S, Navarra P, Rees LH, Besser M, Grossman A, Navara P. Morphine directly modulates the release of stimulated corticotrophin-releasing factor-41 from rat hypothalamus in vitro. Endocrinology. 1989;124:2330–5. doi: 10.1210/endo-124-5-2330. [DOI] [PubMed] [Google Scholar]
- 89.Dalle Pezze P, Nelson G, Otten EG, Korolchuk VI, Kirkwood TB, von Zglinicki T, et al. Dynamic modelling of pathways to cellular senescence reveals strategies for targeted interventions. PLoS Comput Biol. 2014;10:e1003728. doi: 10.1371/journal.pcbi.1003728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Guo J, Chiang WC. Mitophagy in aging and longevity. IUBMB Life. 2022;74:296–316. doi: 10.1002/iub.2585. [DOI] [PubMed] [Google Scholar]
- 91.Lin Q, Li S, Jiang N, Shao X, Zhang M, Jin H, et al. PINK1-parkin pathway of mitophagy protects against contrast-induced acute kidney injury via decreasing mitochondrial ROS and NLRP3 inflammasome activation. Redox Biol. 2019;26:101254. doi: 10.1016/j.redox.2019.101254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Livingston MJ, Wang J, Zhou J, Wu G, Ganley IG, Hill JA, et al. Clearance of damaged mitochondria via mitophagy is important to the protective effect of ischemic preconditioning in kidneys. Autophagy. 2019;15:2142–62. doi: 10.1080/15548627.2019.1615822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Zwerschke W, Mazurek S, Stöckl P, Hütter E, Eigenbrodt E, Jansen-Dürr P. Metabolic analysis of senescent human fibroblasts reveals a role for AMP in cellular senescence. Biochem J. 2003;376:403–11. doi: 10.1042/bj20030816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Sharpless NE, Sherr CJ. Forging a signature of in vivo senescence. Nat Rev Cancer. 2015;15:397–408. doi: 10.1038/nrc3960. [DOI] [PubMed] [Google Scholar]
- 95.James EL, Michalek RD, Pitiyage GN, de Castro AM, Vignola KS, Jones J, et al. Senescent human fibroblasts show increased glycolysis and redox homeostasis with extracellular metabolomes that overlap with those of irreparable DNA damage, aging, and disease. J Proteome Res. 2015;14:1854–71. doi: 10.1021/pr501221g. [DOI] [PubMed] [Google Scholar]
- 96.Wiley CD, Campisi J. From ancient pathways to aging cells-connecting metabolism and cellular senescence. Cell Metab. 2016;23:1013–21. doi: 10.1016/j.cmet.2016.05.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Liao EC, Hsu YT, Chuah QY, Lee YJ, Hu JY, Huang TC, et al. Radiation induces senescence and a bystander effect through metabolic alterations. Cell Death Dis. 2014;5:e1255.. doi: 10.1038/cddis.2014.220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Turgeon MO, Perry NJS, Poulogiannis G. DNA damage, repair, and cancer metabolism. Front Oncol. 2018;8:15. doi: 10.3389/fonc.2018.00015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Shmulevich R, Krizhanovsky V. Cell senescence, DNA damage, and metabolism. Antioxid Redox Signal. 2021;34:324–34. doi: 10.1089/ars.2020.8043. [DOI] [PubMed] [Google Scholar]
- 100.Helman A, Klochendler A, Azazmeh N, Gabai Y, Horwitz E, Anzi S, et al. p16(Ink4a)-induced senescence of pancreatic beta cells enhances insulin secretion. Nat Med. 2016;22:412–20. doi: 10.1038/nm.4054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Pluquet O, Pourtier A, Abbadie C. The unfolded protein response and cellular senescence. A review in the theme: cellular mechanisms of endoplasmic reticulum stress signaling in health and disease. Am J Physiol Cell Physiol. 2015;308:C415–25. doi: 10.1152/ajpcell.00334.2014. [DOI] [PubMed] [Google Scholar]
- 102.Lee YS, Lee DH, Choudry HA, Bartlett DL, Lee YJ. Ferroptosis-induced endoplasmic reticulum stress: cross-talk between ferroptosis and apoptosis. Mol Cancer Res. 2018;16:1073–6. doi: 10.1158/1541-7786.Mcr-18-0055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Lyublinskaya O, Kornienko J, Ivanova J, Pugovkina N, Alekseenko L, Lyublinskaya E, et al. Induction of premature cell senescence stimulated by high doses of antioxidants is mediated by endoplasmic reticulum stress. Int J Mol Sci. 22. 10.3390/ijms222111851 (2021). [DOI] [PMC free article] [PubMed]
- 104.Ziegler DV, Vindrieux D, Goehrig D, Jaber S, Collin G, Griveau A, et al. Calcium channel ITPR2 and mitochondria-ER contacts promote cellular senescence and aging. Nat Commun. 2021;12:720.. doi: 10.1038/s41467-021-20993-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Qiao X, Zhang Y, Ye A, Zhang Y, Xie T, Lv Z, et al. ER reductive stress caused by Ero1α S-nitrosation accelerates senescence. Free Radic Biol Med. 2022;180:165–78. doi: 10.1016/j.freeradbiomed.2022.01.006. [DOI] [PubMed] [Google Scholar]
- 106.Fatima S, Ambreen S, Mathew A, Elwakiel A, Gupta A, Singh K, et al. ER-stress and senescence coordinately promote endothelial barrier dysfunction in diabetes-induced atherosclerosis. Nutrients. 14. 10.3390/nu14142786 (2022). [DOI] [PMC free article] [PubMed]
- 107.Inagi R, Ishimoto Y, Nangaku M. Proteostasis in endoplasmic reticulum-new mechanisms in kidney disease. Nat Rev Nephrol. 2014;10:369–78. doi: 10.1038/nrneph.2014.67. [DOI] [PubMed] [Google Scholar]
- 108.Chiang CK, Hsu SP, Wu CT, Huang JW, Cheng HT, Chang YW, et al. Endoplasmic reticulum stress implicated in the development of renal fibrosis. Mol Med. 2011;17:1295–305. doi: 10.2119/molmed.2011.00131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Liu J, Yang JR, Chen XM, Cai GY, Lin LR, He YN. Impact of ER stress-regulated ATF4/p16 signaling on the premature senescence of renal tubular epithelial cells in diabetic nephropathy. Am J Physiol Cell Physiol. 2015;308:C621–30. doi: 10.1152/ajpcell.00096.2014. [DOI] [PubMed] [Google Scholar]
- 110.Liu J, Huang K, Cai GY, Chen XM, Yang JR, Lin LR, et al. Receptor for advanced glycation end-products promotes premature senescence of proximal tubular epithelial cells via activation of endoplasmic reticulum stress-dependent p21 signaling. Cell Signal. 2014;26:110–21. doi: 10.1016/j.cellsig.2013.10.002. [DOI] [PubMed] [Google Scholar]
- 111.Jaenisch R, Bird A. Epigenetic regulation of gene expression: how the genome integrates intrinsic and environmental signals. Nat Genet. 2003;33:245–54. doi: 10.1038/ng1089. [DOI] [PubMed] [Google Scholar]
- 112.Gensous N, Bacalini MG, Pirazzini C, Marasco E, Giuliani C, Ravaioli F, et al. The epigenetic landscape of age-related diseases: the geroscience perspective. Biogerontology. 2017;18:549–59. doi: 10.1007/s10522-017-9695-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Zhang W, Qu J, Liu GH, Belmonte JCI. The ageing epigenome and its rejuvenation. Nat Rev Mol Cell Biol. 2020;21:137–50. doi: 10.1038/s41580-019-0204-5. [DOI] [PubMed] [Google Scholar]
- 114.Pal S, Tyler JK. Epigenetics and aging. Sci Adv. 2016;2:e1600584. doi: 10.1126/sciadv.1600584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Field AE, Robertson NA, Wang T, Havas A, Ideker T, Adams PD. DNA methylation clocks in aging: categories, causes, and consequences. Mol Cell. 2018;71:882–95. doi: 10.1016/j.molcel.2018.08.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Yang JH, Hayano M, Griffin PT, Amorim JA, Bonkowski MS, Apostolides JK, et al. Loss of epigenetic information as a cause of mammalian aging. Cell. 2023;186:305–26.e327. doi: 10.1016/j.cell.2022.12.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Zhang B, Long Q, Wu S, Xu Q, Song S, Han L, et al. KDM4 orchestrates epigenomic remodeling of senescent cells and potentiates the senescence-associated secretory phenotype. Nat Aging. 2021;1:454–72. doi: 10.1038/s43587-021-00063-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Zampieri M, Ciccarone F, Calabrese R, Franceschi C, Bürkle A, Caiafa P. Reconfiguration of DNA methylation in aging. Mech Ageing Dev. 2015;151:60–70. doi: 10.1016/j.mad.2015.02.002. [DOI] [PubMed] [Google Scholar]
- 119.Castellano G, Franzin R, Sallustio F, Stasi A, Banelli B, Romani M, et al. Complement component C5a induces aberrant epigenetic modifications in renal tubular epithelial cells accelerating senescence by Wnt4/βcatenin signaling after ischemia/reperfusion injury. Aging (Albany NY) 2019;11:4382–406. doi: 10.18632/aging.102059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Bechtel W, McGoohan S, Zeisberg EM, Müller GA, Kalbacher H, Salant DJ, et al. Methylation determines fibroblast activation and fibrogenesis in the kidney. Nat Med. 2010;16:544–50. doi: 10.1038/nm.2135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Zhao Y, Ding C, Xue W, Ding X, Zheng J, Gao Y, et al. Genome-wide DNA methylation analysis in renal ischemia reperfusion injury. Gene. 2017;610:32–43. doi: 10.1016/j.gene.2017.02.005. [DOI] [PubMed] [Google Scholar]
- 122.Ebert T, Tran N, Schurgers L, Stenvinkel P, Shiels PG. Ageing—oxidative stress, PTMs and disease. Mol Asp Med. 2022;86:101099. doi: 10.1016/j.mam.2022.101099. [DOI] [PubMed] [Google Scholar]
- 123.López-Otín C, Blasco MA, Partridge L, Serrano M, Kroemer G. The hallmarks of aging. Cell. 2013;153:1194–217. doi: 10.1016/j.cell.2013.05.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Yang D, Wei G, Long F, Nie H, Tian X, Qu L, et al. Histone methyltransferase Smyd3 is a new regulator for vascular senescence. Aging Cell. 2020;19:e13212. doi: 10.1111/acel.13212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Wesseling H, Mair W, Kumar M, Schlaffner CN, Tang S, Beerepoot P, et al. Tau PTM profiles identify patient heterogeneity and stages of Alzheimer’s disease. Cell. 2020;183:1699–713.e1613. doi: 10.1016/j.cell.2020.10.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Vijayakumaran S, Pountney DL. SUMOylation, aging and autophagy in neurodegeneration. Neurotoxicology. 2018;66:53–7. doi: 10.1016/j.neuro.2018.02.015. [DOI] [PubMed] [Google Scholar]
- 127.Shiels PG, McGuinness D, Eriksson M, Kooman JP, Stenvinkel P. The role of epigenetics in renal ageing. Nat Rev Nephrol. 2017;13:471–82. doi: 10.1038/nrneph.2017.78. [DOI] [PubMed] [Google Scholar]
- 128.Liu BC, Tang TT, Lv LL, Lan HY. Renal tubule injury: a driving force toward chronic kidney disease. Kidney Int. 2018;93:568–79. doi: 10.1016/j.kint.2017.09.033. [DOI] [PubMed] [Google Scholar]
- 129.Chang-Panesso M, Humphreys BD. Cellular plasticity in kidney injury and repair. Nat Rev Nephrol. 2017;13:39–46. doi: 10.1038/nrneph.2016.169. [DOI] [PubMed] [Google Scholar]
- 130.Melk A, Schmidt BM, Takeuchi O, Sawitzki B, Rayner DC, Halloran PF. Expression of p16INK4a and other cell cycle regulator and senescence associated genes in aging human kidney. Kidney Int. 2004;65:510–20. doi: 10.1111/j.1523-1755.2004.00438.x. [DOI] [PubMed] [Google Scholar]
- 131.Fang Y, Gong AY, Haller ST, Dworkin LD, Liu Z, Gong R. The ageing kidney: Molecular mechanisms and clinical implications. Ageing Res Rev. 2020;63:101151. doi: 10.1016/j.arr.2020.101151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Lee DH, Wolstein JM, Pudasaini B, Plotkin M. INK4a deletion results in improved kidney regeneration and decreased capillary rarefaction after ischemia-reperfusion injury. Am J Physiol Ren Physiol. 2012;302:F183–91. doi: 10.1152/ajprenal.00407.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Jin H, Zhang Y, Ding Q, Wang SS, Rastogi P, Dai DF, et al. Epithelial innate immunity mediates tubular cell senescence after kidney injury. JCI Insight. 4. 10.1172/jci.insight.125490 (2019). [DOI] [PMC free article] [PubMed]
- 134.Gerhardt LMS, Liu J, Koppitch K, Cippà PE, McMahon AP. Single-nuclear transcriptomics reveals diversity of proximal tubule cell states in a dynamic response to acute kidney injury. Proc Natl Acad Sci USA. 118. 10.1073/pnas.2026684118 (2021). [DOI] [PMC free article] [PubMed]
- 135.Ferenbach DA, Bonventre JV. Mechanisms of maladaptive repair after AKI leading to accelerated kidney ageing and CKD. Nat Rev Nephrol. 2015;11:264–76. doi: 10.1038/nrneph.2015.3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Luo C, Zhou S, Zhou Z, Liu Y, Yang L, Liu J, et al. Wnt9a promotes renal fibrosis by accelerating cellular senescence in tubular epithelial cells. J Am Soc Nephrol. 2018;29:1238–56. doi: 10.1681/asn.2017050574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Gong W, Luo C, Peng F, Xiao J, Zeng Y, Yin B, et al. Brahma-related gene-1 promotes tubular senescence and renal fibrosis through Wnt/β-catenin/autophagy axis. Clin Sci (Lond) 2021;135:1873–95. doi: 10.1042/cs20210447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Miao J, Huang J, Luo C, Ye H, Ling X, Wu Q, et al. Klotho retards renal fibrosis through targeting mitochondrial dysfunction and cellular senescence in renal tubular cells. Physiol Rep. 2021;9:e14696. doi: 10.14814/phy2.14696. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Yzydorczyk C, Li N, Chehade H, Mosig D, Bidho M, Keshavjee B, et al. Transient postnatal overfeeding causes liver stress-induced premature senescence in adult mice. Sci Rep. 2017;7:12911. doi: 10.1038/s41598-017-11756-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Greeley EH, Spitznagel E, Lawler DF, Kealy RD, Segre M. Modulation of canine immunosenescence by life-long caloric restriction. Vet Immunol Immunopathol. 2006;111:287–99. doi: 10.1016/j.vetimm.2006.02.002. [DOI] [PubMed] [Google Scholar]
- 141.Mafra D, Borges NA, Lindholm B, Shiels PG, Evenepoel P, Stenvinkel P. Food as medicine: targeting the uraemic phenotype in chronic kidney disease. Nat Rev Nephrol. 2021;17:153–71. doi: 10.1038/s41581-020-00345-8. [DOI] [PubMed] [Google Scholar]
- 142.Carroll JE, Cole SW, Seeman TE, Breen EC, Witarama T, Arevalo JMG, et al. Partial sleep deprivation activates the DNA damage response (DDR) and the senescence-associated secretory phenotype (SASP) in aged adult humans. Brain Behav Immun. 2016;51:223–9. doi: 10.1016/j.bbi.2015.08.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Birch J, Gil J. Senescence and the SASP: many therapeutic avenues. Genes Dev. 2020;34:1565–76. doi: 10.1101/gad.343129.120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Moiseeva O, Deschênes-Simard X, St-Germain E, Igelmann S, Huot G, Cadar AE, et al. Metformin inhibits the senescence-associated secretory phenotype by interfering with IKK/NF-κB activation. Aging Cell. 2013;12:489–98. doi: 10.1111/acel.12075. [DOI] [PubMed] [Google Scholar]
- 145.Zhang E, Guo Q, Gao H, Xu R, Teng S, Wu Y. Metformin and resveratrol inhibited high glucose-induced metabolic memory of endothelial senescence through SIRT1/p300/p53/p21 Pathway. PLoS One. 2015;10:e0143814. doi: 10.1371/journal.pone.0143814. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Zhang Q, Shi J, Yuan F, Wang H, Fu W, Pan J, et al. Higher expression of XPF is a critical factor in intrinsic chemotherapy resistance of human renal cell carcinoma. Int J Cancer. 2016;139:2827–37. doi: 10.1002/ijc.30396. [DOI] [PubMed] [Google Scholar]
- 147.Kamal Y, Cheng C, Frost HR, Amos CI. Predictors of disease aggressiveness influence outcome from immunotherapy treatment in renal clear cell carcinoma. Oncoimmunology. 2019;8:e1500106. doi: 10.1080/2162402x.2018.1500106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Sarkisian CJ, Keister BA, Stairs DB, Boxer RB, Moody SE, Chodosh LA. Dose-dependent oncogene-induced senescence in vivo and its evasion during mammary tumorigenesis. Nat Cell Biol. 2007;9:493–505. doi: 10.1038/ncb1567. [DOI] [PubMed] [Google Scholar]
- 149.Megyesi J, Andrade L, Vieira JM, Jr, Safirstein RL, Price PM. Positive effect of the induction of p21WAF1/CIP1 on the course of ischemic acute renal failure. Kidney Int. 2001;60:2164–72. doi: 10.1046/j.1523-1755.2001.00044.x. [DOI] [PubMed] [Google Scholar]
- 150.Megyesi J, Safirstein RL, Price PM. Induction of p21WAF1/CIP1/SDI1 in kidney tubule cells affects the course of cisplatin-induced acute renal failure. J Clin Invest. 1998;101:777–82. doi: 10.1172/jci1497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Braun H, Schmidt BM, Raiss M, Baisantry A, Mircea-Constantin D, Wang S, et al. Cellular senescence limits regenerative capacity and allograft survival. J Am Soc Nephrol. 2012;23:1467–73. doi: 10.1681/asn.2011100967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Gu X, Peng CY, Lin SY, Qin ZY, Liang JL, Chen HJ, et al. P16(INK4a) played a critical role in exacerbating acute tubular necrosis in acute kidney injury. Am J Transl Res. 2019;11:3850–61. [PMC free article] [PubMed] [Google Scholar]
- 153.Fu S, Tang Y, Huang XR, Feng M, Xu AP, Lan HY. Smad7 protects against acute kidney injury by rescuing tubular epithelial cells from the G1 cell cycle arrest. Clin Sci (Lond) 2017;131:1955–69. doi: 10.1042/cs20170127. [DOI] [PubMed] [Google Scholar]
- 154.Rodrigues CE, Capcha JM, de Bragança AC, Sanches TR, Gouveia PQ, de Oliveira PA, et al. Human umbilical cord-derived mesenchymal stromal cells protect against premature renal senescence resulting from oxidative stress in rats with acute kidney injury. Stem Cell Res Ther. 2017;8:19. doi: 10.1186/s13287-017-0475-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Zhang F, Wan X, Cao YZ, Sun D, Cao CC. Klotho gene-modified BMSCs showed elevated antifibrotic effects by inhibiting the Wnt/β-catenin pathway in kidneys after acute injury. Cell Biol Int. 2018;42:1670–9. doi: 10.1002/cbin.11068. [DOI] [PubMed] [Google Scholar]
- 156.Wang SY, Xu Y, Hong Q, Chen XM, Cai GY. Mesenchymal stem cells ameliorate cisplatin-induced acute kidney injury via let-7b-5p. Cell Tissue Res. 2023;392:517–33. doi: 10.1007/s00441-022-03729-3. [DOI] [PubMed] [Google Scholar]
- 157.Yang L, Wang B, Guo F, Huang R, Liang Y, Li L, et al. FFAR4 improves the senescence of tubular epithelial cells by AMPK/SirT3 signaling in acute kidney injury. Signal Transduct Target Ther. 2022;7:384. doi: 10.1038/s41392-022-01254-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Wen L, Ren Q, Guo F, Du X, Yang H, Fu P, et al. Tubular aryl hydratocarbon receptor upregulates EZH2 to promote cellular senescence in cisplatin-induced acute kidney injury. Cell Death Dis. 2023;14:18. doi: 10.1038/s41419-022-05492-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Cai Y, Zhou H, Zhu Y, Sun Q, Ji Y, Xue A, et al. Elimination of senescent cells by β-galactosidase-targeted prodrug attenuates inflammation and restores physical function in aged mice. Cell Res. 2020;30:574–89. doi: 10.1038/s41422-020-0314-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Wang TW, Johmura Y, Suzuki N, Omori S, Migita T, Yamaguchi K, et al. Blocking PD-L1-PD-1 improves senescence surveillance and ageing phenotypes. Nature. 2022;611:358–64. doi: 10.1038/s41586-022-05388-4. [DOI] [PubMed] [Google Scholar]
- 161.Al-Douahji M, Brugarolas J, Brown PA, Stehman-Breen CO, Alpers CE, Shankland SJ. The cyclin kinase inhibitor p21WAF1/CIP1 is required for glomerular hypertrophy in experimental diabetic nephropathy. Kidney Int. 1999;56:1691–9. doi: 10.1046/j.1523-1755.1999.00728.x. [DOI] [PubMed] [Google Scholar]
- 162.Wolstein JM, Lee DH, Michaud J, Buot V, Stefanchik B, Plotkin MD. INK4a knockout mice exhibit increased fibrosis under normal conditions and in response to unilateral ureteral obstruction. Am J Physiol Ren Physiol. 2010;299:F1486–95. doi: 10.1152/ajprenal.00378.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Kim SR, Puranik AS, Jiang K, Chen X, Zhu XY, Taylor I, et al. Progressive cellular senescence mediates renal dysfunction in ischemic nephropathy. J Am Soc Nephrol. 2021;32:1987–2004. doi: 10.1681/asn.2020091373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Kim KM, Noh JH, Bodogai M, Martindale JL, Yang X, Indig FE, et al. Identification of senescent cell surface targetable protein DPP4. Genes Dev. 2017;31:1529–34. doi: 10.1101/gad.302570.117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Molitoris BA, Dagher PC, Sandoval RM, Campos SB, Ashush H, Fridman E, et al. siRNA targeted to p53 attenuates ischemic and cisplatin-induced acute kidney injury. J Am Soc Nephrol. 2009;20:1754–64. doi: 10.1681/asn.2008111204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Bishop B, Sharma S, Scott EA. Nanomedicine in kidney disease. Curr Opin Nephrol Hypertens. 2023;32:366–76. doi: 10.1097/mnh.0000000000000897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Deng X, Zeng T, Li J, Huang C, Yu M, Wang X, et al. Kidney-targeted triptolide-encapsulated mesoscale nanoparticles for high-efficiency treatment of kidney injury. Biomater Sci. 2019;7:5312–23. doi: 10.1039/c9bm01290g. [DOI] [PubMed] [Google Scholar]
- 168.Williams RM, Shah J, Tian HS, Chen X, Geissmann F, Jaimes EA, et al. Selective nanoparticle targeting of the renal tubules. Hypertension. 2018;71:87–94. doi: 10.1161/hypertensionaha.117.09843. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Chang J, Wang Y, Shao L, Laberge RM, Demaria M, Campisi J, et al. Clearance of senescent cells by ABT263 rejuvenates aged hematopoietic stem cells in mice. Nat Med. 2016;22:78–83. doi: 10.1038/nm.4010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Baar MP, Brandt RMC, Putavet DA, Klein JDD, Derks KWJ, Bourgeois BRM, et al. Targeted apoptosis of senescent cells restores tissue homeostasis in response to chemotoxicity and aging. Cell. 2017;169:132–47.e116. doi: 10.1016/j.cell.2017.02.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Chen Z, Yu J, Fu M, Dong R, Yang Y, Luo J, et al. Dipeptidyl peptidase-4 inhibition improves endothelial senescence by activating AMPK/SIRT1/Nrf2 signaling pathway. Biochem Pharm. 2020;177:113951. doi: 10.1016/j.bcp.2020.113951. [DOI] [PubMed] [Google Scholar]


