Abstract
Complementary foods and beverages (CFBs) are key components of an infant’s diet in the second 6 months of life. This article summarizes nutrition and feeding practices examined by the 2020 Dietary Guidelines Advisory Committees during the CFB life stage. Breastfeeding initiation is high (84%), but exclusive breastfeeding at 6 months (26%) is below the Healthy People 2030 goal (42%). Most infants (51%) are introduced to CFBs sometime before 6 months. The primary mode of feeding (ie, human milk fed [HMF]; infant formula or mixed formula and human milk fed [FMF]) at the initiation of CFBs is associated with the timing of introduction and types of CFBs reported. FMF infants (42%) are more likely to be introduced to CFBs before 4 months compared with HMF infants (19%). Different dietary patterns, such as higher prevalence of consumption and mean amounts, were observed, including fruit, grains, dairy, proteins, and solid fats. Compared with HMF infants of the same age, FMF infants consume more total energy (845 vs 631 kcal) and protein (22 vs 12 g) from all sources, and more energy (345 vs 204 kcal) and protein (11 vs 6 g) from CFBs alone. HMF infants have a higher prevalence of risk of inadequate intakes of iron (77% vs 7%), zinc (54% vs <3%), and protein (27% vs <3%). FMF infants are more likely to have an early introduction (<12 months) to fruit juice (45% vs 20%) and cow’s milk (36% vs 24%). Registered dietitian nutritionists and nutritional professionals should consider tailoring their advice to caregivers on dietary and complementary feeding practices, taking into account the primary mode of milk feeding during this life stage to support infants’ nutrient adequacy. National studies that address the limitations of this analysis, including small sample sizes and imputed breast milk volume, could refine findings from this analysis.
Keywords: Dietary guidelines, Birth to 24 months, Pediatrics, Nutrition policy, Breastfeeding, Complementary feeding
Growth trajectories are rapid during infancy and optimal nutritional status during this life stage is critical to support immediate and long-term health and development. The timing of the transition from exclusive human milk or infant formula feeding to the introduction of complementary foods and beverages (CFBs) coincides with this time of high nutrient needs relative to body weight compared with later periods of growth. Thus, provision of nutrient-dense foods to meet nutritional requirements during this period is crucial.1 As such, the American Academy of Pediatrics (AAP) has established guidance for infants during this period based on nutrient needs, developmental readiness, and other feeding practices.1 The AAP recommends human milk or infant formula should be the sole source of nutrition until about 6 months of age, when the introduction of complementary foods and beverages is recommended.1 Transition from a diet of exclusive consumption of human milk or infant formula to a mixed diet that includes nutrient-dense foods is recommended, depending on the infant’s developmental readiness.1–4 Dietary diversity and provision of appropriate forms of foods (eg, purees and finely chopped solids) are encouraged, and fruit juice, cow’s milk, and use of added salts and sugars are not recommended during infancy.1 A complete summary of the historical and current AAP recommendations has been published elsewhere.5 More recently, federal recommendations for infant feeding were mandated for inclusion in the Dietary Guidelines for Americans, 2020–2025 (DGA) for the first time since their inception, as part of the Agriculture Act of 2014 (ie, “The Farm Bill”).3 Before this iteration, the DGA were for Americans aged 2 years and older. The purpose of this article was to summarize the available federal dietary and nutrition data examined by the 2020 Dietary Guidelines Advisory Committees (the Committee), with a focus on current dietary intakes and complementary feeding practices from 6 to 12 months. Some portions of this work are also published as part of the Committee’s scientific report to the US Government.6
DATA REVIEW PROCESS
The Committee reviewed and integrated data from the following 3 US federal monitoring and surveillance systems in the development of the report: National Immunization Survey (NIS) conducted by the National Center for Immunization and Respiratory Diseases of the Centers for Disease Control and Prevention7; National Survey of Children’s Health (NSCH) conducted by the Maternal and Child Health Bureau of the Health Resources and Services Administration8; and the National Health and Nutrition Examination Survey (NHANES) conducted by the National Center for Health Statistics of the Centers for Disease Control and Prevention.9 The Committee only evaluated dietary intake data from federal sources so that the findings would be relevant to federal policy associated with the DGA.
Data from the NIS were used to garner information on the prevalence of breastfeeding initiation and duration by maternal characteristics among infants born in 2017 (n = 20,026).7 This survey used random-digit dialing to collect data from US households via telephone to obtain a wide variety of data, including retrospective information about breastfeeding initiation, duration, and exclusivity from parents or caregivers for children 19 to 35 months of age at the time of data collection. NIS data are collected at the state level and are pooled and weighted to obtain national estimates. The sampling design and weighting system, as well as more complete details of the NIS methods, can be found elsewhere.10 Exclusive breastfeeding in this survey is defined as consumption of human milk only, with no consumption of other solids or liquids.
The NSCH protocol includes annual collection of data from parents and caregivers on various aspects of infant feeding practices and health, including practices such as the timing of introduction of CFBs for infants. The NSCH is also designed to produce nationally representative estimates, and complete details of the methods are available publicly.8 In the 2016–2018 NSCH, the sample size was adequate to stratify infants into the following 3 feeding groups based on status at age 4 months: human milk only (n = 9,085), infant formula only (n = 9,567), or mixed-fed human milk and infant formula (n = 4,863), and the Committee examined data on early introduction to CFBs from this study.11
The vast majority of data provided to the Committee were data or research briefs from the existing literature or original analyses from the Federal Data Analysis Team (a trans-agency team of scientists) of dietary data from the What We Eat in America component of NHANES. Complete details of the NHANES methodology are available publicly.9 Dietary data are collected from two, 24-hour dietary recalls (24HRs), the first collected in person at a mobile examination center, and the second via telephone. Both of the recalls were collected from a parent or proxy caregiver using the Automated Multiple-Pass Method methodology, in collaboration with the US Department of Agriculture.12,13 Age of the infant was based on age at the time of examination in the mobile examination center. Data from NHANES 2007–2016 were combined to achieve a sample size sufficient to examine the data for older infants (6 to 12 months; n = 988) and stratified by feeding status based on both 24HRs: infants receiving human milk (HMF; n = 141; mean age 8.1 months) or infants receiving infant formula, including those who were mixed-fed (FMF; n = 847; mean age 8.6 months). Mixed-fed infants (n = 92; mean age 8.4 months) were included with formula-fed infants (n = 755; mean age 8.6 months) due to small sample sizes in NHANES data.
NHANES collects more detail on dietary intakes and feeding behaviors than the NIS and NSCH, although there are still limitations, as summarized elsewhere.14 For example, the volume of breast milk that was provided to an infant by direct breastfeeding was not quantified during the dietary recall. However, if the proxy reported that an infant was fed expressed human milk, then the amount was quantified and coded as such. Otherwise, the volume of human milk consumed was estimated according to coding rules based on pre-existing data from several studies,15–18 a strategy that has been further supported in systematic review and model estimates from stable isotope studies from 12 countries in 5 continents19 and used previously with NHANES data.20 Briefly, infants whose only milk source at 6 to 12 months was human milk were assigned breast milk intakes of 600 mL/d, and mixed-fed infants were assigned human milk intakes by subtracting the amount of formula or other milks consumed from 600 mL/d.
For CFBs, the proportion of infants with reported intake of a food group or subgroup on a given day and the mean intakes of energy, nutrients, and other food components were estimated. Mean dietary intakes from all sources and from CFBs are presented separately; CFBs include all foods and beverages except human milk and infant formula. Usual intake distributions were derived using the National Cancer Institute method21 to estimate the potential risk of dietary inadequacy for protein, iron, and zinc (the only nutrients for which an Estimated Average Requirement value is available during this age range)22; usual intake estimates were not compared statistically. All statistical differences in feeding practices and dietary intakes between HMF and FMF infants were determined by t tests, with statistical significance set at P < .01 to account for multiple comparisons.
SUMMARY OF FEDERAL DATA
The national prevalence of breastfeeding initiation was 84.1% among infants born in 2017, according to the NIS.7 However, low rates of exclusive breastfeeding were observed at 3 months (49.6%) and 6 months of age (25.6%).7 The Healthy People 2030 target goal for exclusive breastfeeding through 6 months is 42.2%.23 Differences in duration of any and exclusive breastfeeding were evaluated by race and ethnicity, maternal education, maternal age, marital status, and poverty-to-income ratio.24 Figure 1 illustrates that exclusive breastfeeding rates differ by race and ethnicity; notable increases have been observed over time among non-Hispanic Black infants. Exclusive breastfeeding rates also tend to be positively associated with maternal age, education, and income. NHANES data indicate that for infants who are fed infant formula, the predominant form is cow’s milk–based (69%), with lower percentages of infants receiving soy (12%), specialty (6%), or “gentle/sensitive” or lactose-free or lactose-reduced formulas (5%).25
Figure 1.
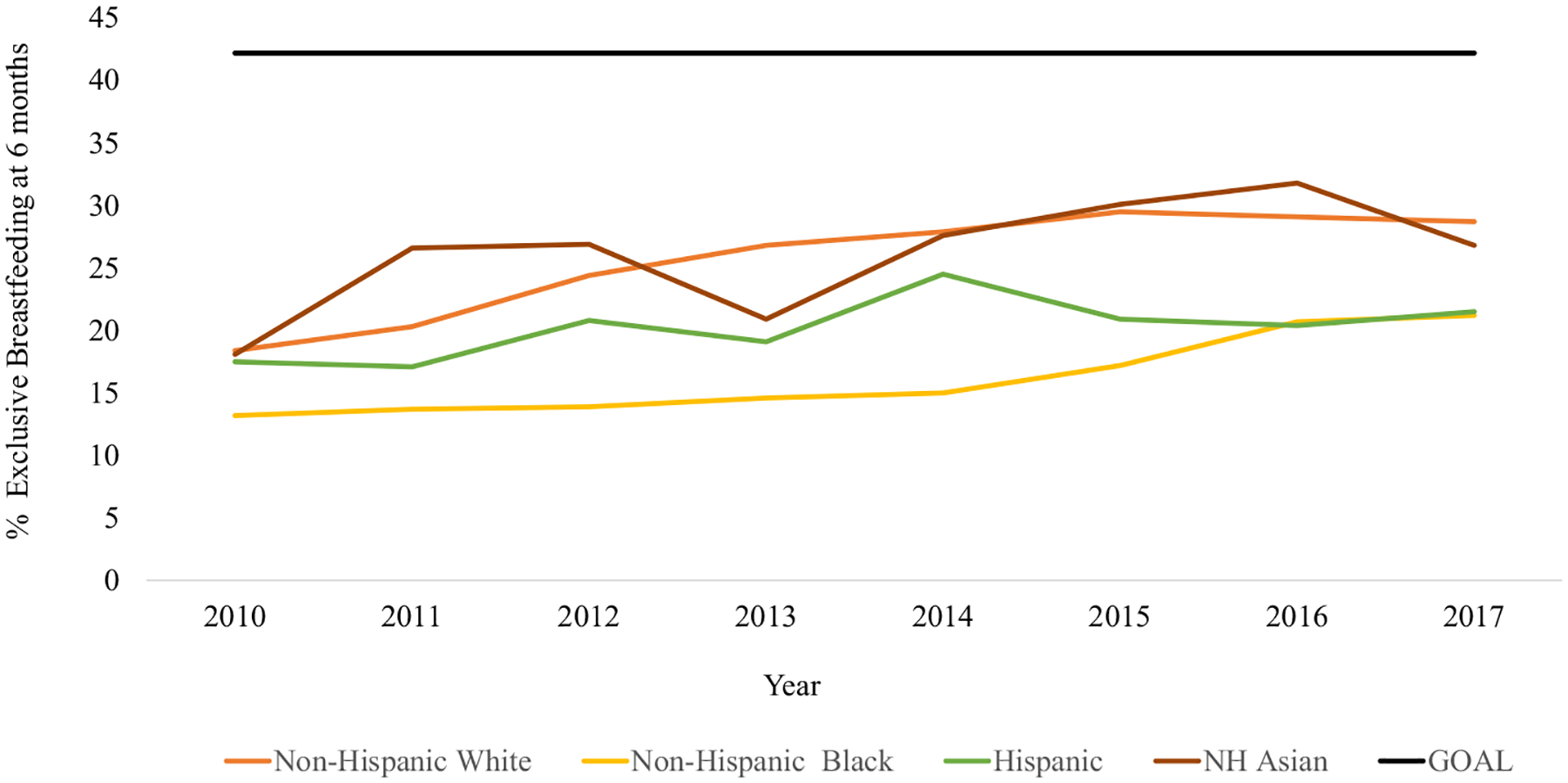
The prevalence (%) of exclusive breastfeeding at 6 months of age among US infants born in 2010–2017, by race and Hispanic origin (non-Hispanic [NH] and Hispanic), National Immunization Survey compared with the Healthy People 2030 goal. The Healthy People 2030 target goal for exclusive breastfeeding through 6 months is 42.2%.
The majority of US infants are introduced to CFBs before 6 months of age (51%). Approximately one-third (32%) of infants receive CFBs before 4 months of age, and this proportion is higher among formula-fed infants (42%) compared with those who are mixed-fed (32%) or fed human milk as the only source of milk (19%) (Figure 2).11 Non-Hispanic Black infants (40.5%) were more likely than Hispanic (29.9%), non-Hispanic White infants (31.5%), and Asian infants (23.8%) to be introduced to CFBs before 4 months. Differences in early introduction to CFBs were also observed by geographic location and other sociodemographic factors, such as maternal age and education and household income.11
Figure 2.
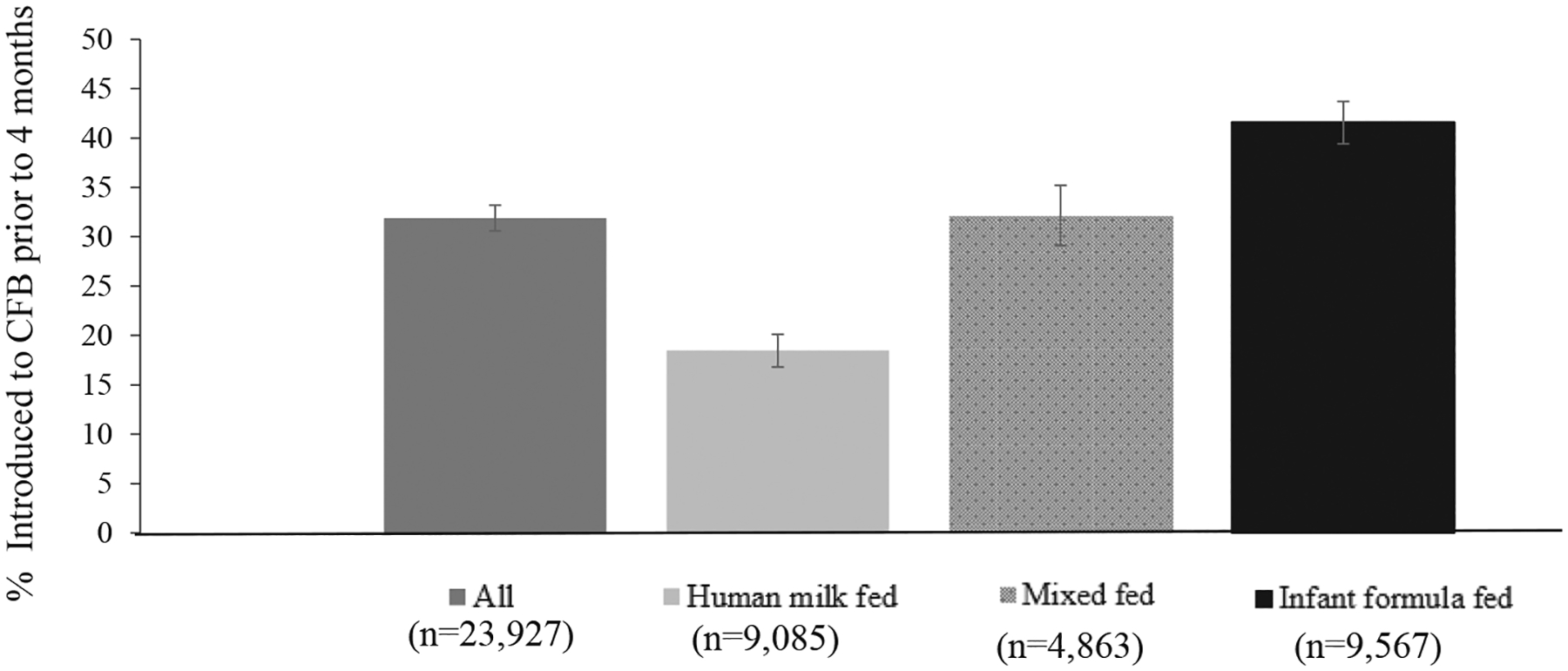
The prevalence (%) of introduction before 4 months of age of complementary foods and beverages (CFBs) among US infants, overall and stratified by mode of milk feeding, National Survey of Children’s Health, 2016–2018. The error bars represent the 95% CIs.
Dietary data from NHANES suggest that FMF infants are more likely to receive cow’s milk (36% vs 24%) and fruit juice (45% vs 20%) between 6 and 12 months of age than are HMF infants (Table 1). FMF infants are also more likely to consume fruits (86% vs 75%), grains (91% vs 81%), protein foods (51% vs 35%), and solid fats (a proxy for saturated fat in the diet; 62% vs 49%) on a given day than are HMF infants (Table 2). For total fruit, this difference is driven by higher intake of fruit juice and fruits other than citrus, melons, and berries among FMF infants compared with their HMF counterparts (Table 3; available online at www.jandonline.org). In addition, compared with HMF infants, FMF infants have a higher intake of refined grains relative to whole grains. These patterns of dietary intake are associated with differences in intakes of energy, macronutrients, total sugars, and fiber (Table 1), as well as micronutrients (Table 4) between groups of infants categorized by primary feeding mode. HMF infants have a high prevalence of inadequate intakes of protein (27% HMF vs <3% FMF), iron (77% HMF vs 7% FMF), and zinc (54% HMF vs <3% FMF) compared with FMF infants. Although <3% of all infants (regardless of primary mode of feeding) were at risk of potentially excessive iron intakes from the diet, more than three-quarters (77%) of FMF infants had zinc intakes that exceeded the Tolerable Upper Intake Level; this was much less common among HMF infants (3%) (data not shown).
Table 1.
A summary of feeding practices and intakes of macronutrient and dietary components among US older infants (6 to 12 months) by primary milk feeding mode, National Health and Nutrition Examination Survey, 2007–2016a
| Variable | HMFb infants (n = 141) | FMFc infants (n = 847) | P value |
|---|---|---|---|
| mean ± standard error | |||
| AAPd guidelines | |||
| Early-introduction fruit juice <12 mo, % | 20 ± 4.4 | 45 ± 2.2 | <.0001 |
| Early-introduction cow’s milk <12 mo, % | 24 ± 3.2 | 36 ± 1.4 | .0028 |
| Intake of macronutrients and dietary components | |||
| Energy (all sourcese), kcal | 631 ± 12.1 | 845 ± 12.5 | <.0001 |
| Energy (CFBsf only), kcal | 204 ± 12.0 | 345 ± 11.9 | <.0001 |
| Protein (all sourcese), g | 12.2 ± 0.4 | 21.7 ± 0.5 | <.0001 |
| Protein (CFBs only), g | 5.9 ± 0.4 | 10.8 ± 0.5 | <.0001 |
| Proportion at risk of protein inadequacy,g% | 27 ± 2.8 | <3 | — |
| Carbohydrate (all sourcese), g | 78 ± 2.2 | 112 ± 1.9 | <.0001 |
| Carbohydrate (CFBs only), g | 36 ± 2.2 | 58 ± 1.8 | <.0001 |
| Total sugars (All sourcese), g | 57 ± 1.0 | 78 ± 1.1 | <.0001 |
| Total sugars (CFBs only), g | 15 ± 1.0 | 26 ± 0.9 | <.0001 |
| Dietary fiber (All sourcese), g | 3.8 ± 0.3 | 4.8 ± 0.2 | .0195 |
| Dietary fiber (CFBs only), g | 3.8 ± 0.3 | 4.7 ± 0.2 | .0229 |
| Total fat (all sourcese), g | 31.6 ± 0.5 | 35.3 ± 0.5 | <.0001 |
| Total fat (CFBs only), g | 4.8 ± 0.5 | 8.3 ± 0.5 | <.0001 |
Milk reporting status determined by the report of human milk on either day 1 or day 2 of the 24-h dietary recall. Complementary foods include all foods and beverages except human milk and infant formula.
HMF = human milk fed.
FMF = formula or mixed fed.
AAP = American Academy of Pediatrics.
Includes energy and nutrients obtained from human milk or infant formula.
CFBs = complementary foods and beverages.
Usual intake distributions were used to estimate the proportion of infants at risk for dietary inadequacy.
Table 2.
Proportion (%) and mean (standard error) daily intake of older infants (6 to 12 months) reporting food group consumption by primary milk feeding mode, NHANES,a 2007–2016b
| Food group consumption | HMFc Infants (n = 141) | FMFd Infants (n = 847) | ||
|---|---|---|---|---|
| Proportion of consumers,% | Intake | Proportion of consumers, % | Intake | |
| mean ± standard error | ||||
| Fruits (cup equivalent) | 75 ± 3.7 | 0.38 ± 0.02 | 86 ± 1.1e | 0.67 ± 0.03f |
| Grains (ounce equivalent) | 81 ± 3.1 | 0.71 ± 0.07 | 91 ± 1.1e | 1.14 ± 0.05f |
| Vegetables (cup equivalent) | 76 ± 3.8 | 0.35 ± 0.04 | 80 ± 1.7 | 0.41 ± 0.02 |
| Protein foods (ounce equivalent) | 35 ± 3.4 | 0.32 ± 0.04 | 51 ± 2.4e | 0.58 ± 0.05f |
| Dairy (cup equivalent) | 40 ± 4.2 | 0.08 ± 0.01 | 46 ± 1.7 | 0.30 ± 0.03f |
| Oils (g) | 46 ± 4.5 | 1.7 ± 0.40 | 59 ± 2.4 | 1.7 ± 0.18 |
| Solid fats (g) | 49 ± 3.9 | 1.3 ± 0.23 | 62 ± 1.7e | 3.7 ± 0.25f |
| Added sugars (teaspoon equivalent) | 55 ± 4.3 | 0.7 ± 0.10 | 64 ± 2.0 | 1.1 ± 0.08 |
NHANES = National Health and Nutrition Examination Survey.
Data are from NHANES, What We Eat In America 2007–2016 Day 1 and Food Patterns Equivalents Databases. Milk reporting status determined by the report of human milk on either day 1 or day 2. The mean amount represents the feeding group-level average for consumers and nonconsumers. Legumes are included in the Protein Foods food group.
HMF = human milk fed.
FMF = formula or mixed fed.
Statistically significant at P < .01 compared with the proportion of HMF infants who are consumers.
Statistically significant differences when the mean intake of HMF infants is compared with the mean intake of FMF infants at P < .01.
Table 3.
Proportion (%) and mean (standard error) daily intake of older infants (6 to 12 months) reporting food subgroup consumption by primary milk feeding mode, NHANES,a 2007–2016b
| HMFc Infants (n = 141) | FMFd Infants (n = 847) | |||
|---|---|---|---|---|
| Food subgroup consumption | Proportion of consumers, % | Intake, g | Proportion of consumers, % | Intake, g |
| mean ± standard error | ||||
| Fruits (cup equivalent) | ||||
| Citrus, melon, berry | 18 ± 2.4 | 0.03 ± 0.01 | 17 ± 1.7 | 0.03 ± 0.01 |
| Other fruit | 70 ± 3.6 | 0.31 ± 0.02 | 75 ± 1.9 | 0.41 ± 0.02e |
| Juice | 20 ± 4.4 | 0.04 ± 0.01 | 45 ± 2.2f | 0.22 ± 0.01e |
| Grains (ounce equivalent) | ||||
| Whole | 51 ± 4.1 | 0.24 ± 0.05 | 61 ± 2.7 | 0.34 ± 0.02 |
| Refined | 68 ± 4.5 | 0.46 ± 0.04 | 83 ± 1.7f | 0.81 ± 0.04e |
| Vegetables (cup equivalent) | ||||
| Total starchy | 37 ± 4.6 | 0.07 ± 0.01 | 43 ± 2.1 | 0.11 ± 0.01 |
| Potatoes | 18 ± 3.7 | 0.03 ± 0.01 | 29 ± 1.5 | 0.06 ± 0.00 |
| Other starchy | 28 ± 4.3 | 0.04 ± 0.01 | 31 ± 2.0 | 0.05 ± 0.01 |
| Total red/orange | 59 ± 4.6 | 0.17 ± 0.02 | 66 ± 2.1 | 0.21 ± 0.01 |
| Tomatoes | 12 ± 2.7 | 0.01g ± 0.00 | 19 ± 1.9 | 0.03 ± 0.00e |
| Other red/orange | 55 ± 5.3 | 0.16 ± 0.02 | 57 ± 2.2 | 0.18 ± 0.02 |
| Dark green | 6g ± 1.9 | 0.01g ± 0.01 | 6 ± 1.5 | 0.01g ± 0.00 |
| Other | 36 ± 5.3 | 0.08 ± 0.02 | 28 ± 2.4 | 0.06 ± 0.01 |
| Legumes | 6g ± 1.6 | 0.01g ± 0.00 | 6 ± 0.9 | 0.01 ± 0.00 |
| Protein foods (ounce equivalent) | ||||
| Total meat, poultry, seafood | 25 ± 3.7 | 0.21 ± 0.03 | 44 ± 2.1f | 0.43 ± 0.04e |
| Meat (eg, beef, veal, or pork) | 7g ± 2.4 | 0.06g ± 0.02 | 16 ± 1.3f | 0.13 ± 0.02e |
| Poultry | 18 ± 3.6 | 0.13g ± 0.02 | 30 ± 2.0 | 0.22 ± 0.03 |
| Cured meat | 3g ± 0.9 | 0.01g ± 0.01 | 8 ± 1.3f | 0.07 ± 0.02 |
| Total fish and seafood | — h | — h | 1g ± 0.4 | 0.01g ± 0.00 |
| Eggs | 17 ± 3.1 | 0.04g ± 0.01 | 19 ± 1.7 | 0.08 ± 0.01 |
| Peanuts, nuts, seeds | 3g ± 1.3 | 0.02g ± 0.01 | 2 ± 0.7 | 0.01g ± 0.01 |
| Soy products | 2g ± 1.2 | 0.01g ± 0.01 | 3 ± 0.8 | — h |
| Dairy (cup equivalent) | ||||
| Fluid milk | 24 ± 3.2 | 0.02g ± 0.01 | 36 ± 1.4f | 0.20 ± 0.02e |
| Cheese | 10 ± 2.5 | 0.01g ± 0.01 | 21 ± 1.4f | 0.06 ± 0.01e |
| Yogurt | 17 ± 3.1 | 0.04 ± 0.01 | 10 ± 1.4 | 0.04 ± 0.01 |
NHANES = National Health and Nutrition Examination Survey.
Data are from NHANES What We Eat In America 2007–2016 Day 1 and Food Patterns Equivalents Databases. Milk reporting status determined by the report of human milk on either day 1 or day 2. The mean amount represents the feeding group-level average for consumers and nonconsumers.
HMF = human milk fed.
FMF = formula or mixed fed.
Statistically significant difference when the mean intake of HMF infants is compared with the mean intake of FMF infants, P < .01.
Statistically significant at P < .01 when compared with the proportion of HMF infants who are consumers.
Less stable estimate due to small sample size or a large relative standard error.
Nonzero value that is too small to present.
Table 4.
Micronutrient intakes among older US infants (6 to 12 months) by primary milk feeding mode, NHANES,a 2007–2016b
| Micronutrient intake data | HMFc infants (n = 141) | FMFd infants (n = 847) | P value |
|---|---|---|---|
| mean ± standard error | |||
| Vitamin A, RAEe (all sourcesf), μg | 588 ± 28.0 | 690 ± 16 | .0027 |
| Vitamin A, RAE (CFBsg only), μg | 215 ± 28.1 | 242 ± 13.2 | .3911 |
| Retinol (all sourcesf), μg | 393 ± 5.5 | 506 ± 6.8 | <.0001 |
| Retinol (CFBs only), μg | 26 ± 5.4 | 58 ± 4.3 | <.0001 |
| Thiamin (all sourcesf), mg | 0.39 ± 0.04 | 0.91 ± 0.02 | <.0001 |
| Thiamin (CFBs only), mg | 0.31 ± 0.04 | 0.48 ± 0.02 | .0010 |
| Riboflavin (all sourcesf), mg | 0.59 ± 0.04 | 1.29 ± 0.02 | <.0001 |
| Riboflavin (CFBs only), mg | 0.37 ± 0.04 | 0.62 ± 0.02 | <.0001 |
| Niacin (all sourcesf), mg | 5.2 ± 0.4 | 11.4 ± 0.3 | <.0001 |
| Niacin (CFBs only), mg | 4.1 ± 0.4 | 6.4 ± 0.2 | .0002 |
| Vitamin B-6 (all sourcesf), μg | 0.36 ± 0.02 | 0.74 ± 0.02 | <.0001 |
| Vitamin B-6 (CFBs only), μg | 0.29 ± 0.02 | 0.44 ± 0.02 | <.0001 |
| Folate, DFEh (all sourcesf), μg | 93 ± 5.6 | 214 ± 4.4 | <.0001 |
| Folate, DFE (CFBs only), μg | 62 ± 5.6 | 91 ± 4.6 | .0016 |
| Choline (all sourcesf), mg | 127 ± 2.6 | 135 ± 3.4 | .0943 |
| Choline (CFBs only), mg | 29 ± 2.6 | 52 ± 2.8 | <.0001 |
| Vitamin B-12 (all sourcesf), mg | 0.67 ± 0.04 | 2.14 ± 0.05 | <.0001 |
| Vitamin B-12 (CFBs only), mg | 0.36 ± 0.04 | 0.77 ± 0.06 | <.0001 |
| Vitamin C (all sourcesf), mg | 52.6 ± 1.9 | 98.7 ± 2.4 | <.0001 |
| Vitamin C (CFBs only), mg | 21.9 ± 1.9 | 47.7 ± 2.1 | <.0001 |
| Vitamin D (all sourcesf), μg | 1.08 ± 0.07 | 8.36 ± 0.13 | <.0001 |
| Vitamin D (CFBs only), μg | 0.41 ± 0.07 | 1.18 ± 0.09 | <.0001 |
| Alpha-tocopherol (all sourcesf), mg | 2.00 ± 0.13 | 7.72 ± 0.14 | <.0001 |
| Alpha-tocopherol (CFBs only), mg | 1.50 ± 0.13 | 2.06 ± 0.08 | .0045 |
| Calcium (all sourcesf), mg | 329 ± 12.9 | 684 ± 10.8 | <.0001 |
| Calcium (CFBs only), mg | 133 ± 12.9 | 258 ± 10.0 | <.0001 |
| Magnesium (all sourcesf), mg | 59 ± 3.2 | 105 ± 2.1 | <.0001 |
| Magnesium (CFBs only), mg | 41 ± 3.2 | 63 ± 1.9 | <.0001 |
| Iron (all sourcesf), mg | 5.8 ± 0.7 | 17.0 ± 0.4 | <0.0001 |
| Iron (CFBs only), mg | 5.5 ± 0.7 | 8.5 ± 0.3 | .0013 |
| Proportion at risk of iron inadequacy,i % | 77 ± 4.7 | 7 ± 1.3 | — |
| Zinc (all sourcesf), mg | 2.6 ± 0.2 | 6.7 ± 0.1 | <.0001 |
| Zinc (CFBs only), mg | 1.6 ± 0.2 | 2.4 ± 0.1 | .0010 |
| Proportion at risk of zinc inadequacy,i % | 54 ± 4.8 | <3 | — |
| Potassium (all sourcesf), mg | 700 ± 24 | 1141 ± 20 | <.0001 |
| Potassium (CFBs only), mg | 388 ± 23.8 | 613 ± 20.0 | <.0001 |
| Sodium (all sourcesf), mg | 305 ± 21 | 508 ± 23 | <.0001 |
| Sodium (CFBs only), mg | 201 ± 20.4 | 344 ± 24.1 | <.0001 |
NHANES = National Health and Nutrition Examination Survey.
Data are from NHANES What We Eat In America 2007–2016 Day 1 and Food Patterns Equivalents Databases. Milk reporting status determined by the report of human milk on either day 1 or day 2.
HMF = human milk fed.
FMF = formula or mixed fed.
RAE = retinol activity equivalents.
Includes energy and nutrients obtained from human milk or infant formula.
CFBs = complementary foods and beverages.
DFE = dietary folate equivalent.
Usual intake distributions were used to estimate the proportion of infants at risk for dietary inadequacy using the Estimated Average Requirement and were not compared statistically.
DISCUSSION
For the first time, the 2020 Dietary Guidelines Advisory Committee extensively reviewed data on breastfeeding, the introduction of CFBs, and the diets of infants in order to inform the development of the DGA for infants and toddlers. Although rates of breastfeeding initiation are high, exclusive breastfeeding at 6 months (26%) was markedly lower than the Healthy People 2030 target goal (42.2%),23 with notably lower rates of initiation and duration observed among non-Hispanic Black and lower-resource caregivers, consistent with previous reports.26,27 This is concerning because breastfeeding has positive health benefits for both the mother and her offspring.28–36 Nutritional exposures and feeding practices in infancy have been related to risk of overweight and obesity and associated cardiometabolic conditions in childhood and subsequently later in life.37 Through the systematic review process, the Committee concluded that “moderate evidence suggested that ever, compared with never, consuming human milk is associated with a lower risk of overweight and obesity at ages 2 y and older, particularly if the duration of human milk consumption is >6 mo.”38 However, the Committee noted a need for stronger research designs to better address the complex relationship between infant feeding practices and risk of overweight and obesity. Nevertheless, policies and strategies can support breastfeeding across multiple sectors, including immediate support and education after childbirth, community-level programs, as well as workplace-related practices, including length of maternity leave,39 as well other as known barriers.40
The AAP Committee on Nutrition recommends against introduction of CFBs before 4 months, with appropriate introduction at about 6 months.1 Approximately one-third of US infants are fed CFBs before 4 months11; these estimates are higher than those reported in other studies, such as Project Viva (19%)41 and the Feeding Infants and Toddlers Study (FITS; 17%).42 Although the Committee did not have information on early introduction of CFBs and demographic characteristics from NHANES, data from FITS 201642 and the NSCH11 suggest that non-Hispanic Black infants are more likely than other reported racial and ethnic groups to receive early CFBs.42 Data are not reported in these studies on Native Hawaiians, Pacific Islanders, American Indian, or Alaska Native groups. Infants receiving infant formula are also more likely to have early introduction of CFBs.11 Regardless of primary milk feeding mode, some studies suggest that early introduction of CFBs before 4 months of age has been associated with markers of childhood adiposity.41
The timing of introduction and the types and amounts of CFBs provided to infants and toddlers potentially influence nutritional status, growth and body composition, neuro-cognitive development, healthy feeding behaviors, and both short-term and long-term health outcomes, including bone health and risk of food allergies and other atopic diseases.43–48 Introduction of fruit juice49–51 or cow’s milk52 is not recommended before 12 months of age. National data suggest that approximately 1 in 10 US infants consume cow’s milk before 12 months of age,25 signaling a need for caregiver education surrounding early introduction of cow’s milk, given that it does not optimally support nutrient requirements at this age, has been associated with gastrointestinal blood loss in infants,53,54 and has low iron and high protein content. Overall, 40% of older infants consume fruit juice before 12 months of age. This is of potential concern because the energy provided by fruit juice may displace energy from more nutrient-rich CFBs, which may compromise the infant’s ability to meet requirements for essential nutrients or may lead to excess energy intake.
Similar to other studies, we observed many differences in the patterns of dietary intake and, importantly, in the amounts of CFBs that were related to an infant’s primary mode of milk feeding, that is, between HMF and FMF infants. For example, FMF infants are more likely to consume fruit juice and saturated fats by 1 year of age; they also consume more total energy and larger amounts of most other food components compared with HMF infants, who had a higher prevalence of risk for dietary inadequacy for iron. These observations suggest the need to introduce foods that are good sources of bioavailable iron during the transition from milk-based to table food feedings, particularly if human milk is the primary milk source, given its low iron content.55 Data across the years from FITS studies (2002, 2008, and 2016) suggest that dietary iron inadequacy has increased over time.56,57 Although the Committee did not specifically examine trends in the use of iron-fortified cereals, FITS data suggest that consumption has decreased over time. Thus, the specific guidance to provide foods that are intrinsically rich in iron (eg, meats) or fortified with iron (eg, iron-fortified infant cereal), particularly during the period of 6 to 12 months of age among infants receiving human milk, is warranted. The identification of iron as a nutrient of public health concern among HMF infants was based largely on dietary intake data58; more information is needed to interpret these dietary estimates, including maternal iron status, timing of umbilical cord clamping at birth, and biomarkers of iron status in infants in this age group.59 Recent data from Abrams and colleagues60 also suggest that less net iron is absorbed by HMF infants compared with formula-fed infants, but that overall the estimated iron from typical CFBs is below that needed and recommended for optimal growth and erythropoiesis in a large proportion of infants in the CFB life stage. Although updated Dietary Reference Intake values for infants and toddlers are needed to better characterize potential risk of dietary inadequacy and excess,61 the iron data examined by the Committee and those of FITS60 together signal a concern for iron adequacy during this life stage; more extensive biomarker data of the nutritional status of infants in this life stage are also needed.
Research Gaps and Limitations
Many gaps exist and more research is needed to better characterize the dietary landscape of older infants. The findings presented in this article should be considered with several considerations in mind. First, although the mean ages of the 2 feeding groups in NHANES were similar (8.1 months in HMF; 8.6 months in FMF), energy intakes are largely influenced by the age of the infant, and our findings should be interpreted with the caveat that each month represents a dynamic period of growth and development. The NHANES data are weighted to be reflective of the US population. The limited sample size from which national data are derived has precluded additional analysis of food and nutrient intake related to primary milk feeding mode and other important characteristics, such as race and ethnicity, family income, food security, and participation in federal food assistance programs. Mixed-fed infants were combined with formula-fed infants into a single group in the NHANES, although they may have different food consumption patterns. We also know that chrononutrition is an emerging area of importance,62 and very little is known about the timing of feeding both milk sources and CFBs throughout the day and how that may relate to growth and weight status.63 Although the AAP recommends offering fruits and vegetables to young children at each feeding occasion,64 the Committee did not have these data to compare contemporary dietary patterns with this guideline.
Energy and nutrient values from breastmilk are based on very limited data that may not be representative of a diverse range of factors that influence human milk nutrients and other bioactive components; there are ongoing federal efforts to update the human milk composition database. Our analysis and the published literature from 24HR data suggest that reported energy intakes at this age exceed estimated needs.20,65 If assumptions of over-reporting energy intakes in infants are true,66 the current reported estimates of inadequacy may be biased; however, to our knowledge the magnitude of this potential bias is largely unknown, and it is unknown whether this bias is different based on primary mode of milk feeding. What may be considered “usual” intakes is difficult to define in such a dynamic segment of the population. For example, the Committee categorized infants participating in NHANES into 2 feeding groups based on a reporting of 24HRs, which may or may not be representative of usual milk feeding. Proxy and retrospective recall bias of other child feeding behaviors may also exist, the extent to which is largely unknown.67,68 Another limitation of the data is a lack of information about those infants who receive cereal that is used to thicken milk feedings before 4 months of age, as this is a common recommendation for infants with symptoms of gastroesophageal reflux.
CONTEXT AND CONCLUSIONS
We found significant differences in dietary exposures by primary mode of milk feeding (human milk vs infant formula). CFBs are needed to provide more than one-half of the infant’s requirements for vitamins B-6, D, E, thiamin, and niacin as well as iron, zinc, magnesium, phosphorus, manganese, and fluoride at 6 to 8 months of age,4 but this varies by the amounts and combinations of human milk and infant formula. Given the estimated calorie requirements during this life stage, it can be quite challenging to meet these nutrient requirements without careful consideration of choices for CFBs. Through extensive food pattern modeling exercises, described elsewhere, the Committee developed a number of potential dietary patterns to meet dietary recommendations in this age group.69 This led to one of the hallmark recommendations of the DGA to “make every bite count.”3
This work of the Committee identified opportunities to improve current infant feeding practices and demonstrates that a “one-size fits all approach” to CFBs may not be ideal, given the differences observed between subgroups of infants categorized by primary milk feeding mode. Registered dietitian nutritionists and nutritional professionals across all sectors should educate caregivers on the recommended timing of introduction of CFBs and tailor their advice to caregivers to support needs for growth and development during this life stage. Because food preferences and patterns are beginning to form during this developmental stage, it is important that caregivers encourage consumption of nutrient-rich foods and avoidance of energy-dense but nutrient-poor CFBs.
RESEARCH SNAPSHOT.
Research Question:
What was the existing evidence base to inform the 2020 Dietary Guidelines Advisory Committee with regard to the dietary intakes and feeding practices during the complementary feeding period among US infants, 6 to 12 months of age?
Key Findings:
Intake of complementary foods and beverages differs by primary mode of infant milk feeding; health care professionals must help caregivers make appropriate nutrient-dense complementary food choices.
ACKNOWLEDGEMENTS
The findings and conclusions are those of the authors and do not represent the views of their respective universities or any entity of the US Government. This work was completed in accordance with the Federal Advisory Committee Act.
FUNDING/SUPPORT
There is no funding to disclose.
Footnotes
Supplementary materials:
Table 3 is available at www.jandonline.org
STATEMENT OF POTENTIAL CONFLICT OF INTEREST
No potential conflict of interest was reported by the authors.
References
- 1.Feeding the infant. In: Kleinman RE, Greer FR, eds. The Pediatric Nutrition Handbook. 8th ed. American Academy of Pediatrics; 2019: 17–188. [Google Scholar]
- 2.Section on Breastfeeding. Breastfeeding and the use of human milk. Pediatrics. 2012;129(3):e827–e841. [DOI] [PubMed] [Google Scholar]
- 3.Dietary Guidelines for Americans, 2020–2025. 9th edition. US Department of Agriculture, US Department of Health and Human Services. Published December 2020. Accessed October 24, 2021. https://www.dietaryguidelines.gov/sites/default/files/2020-12/Dietary_Guidelines_for_Americans_2020-2025.pdf [Google Scholar]
- 4.Butte N, Cobb K, Dwyer J, et al. The Start Healthy Feeding Guidelines for Infants and Toddlers. J Am Diet Assoc. 2004;104(3):442–454. [DOI] [PubMed] [Google Scholar]
- 5.Kleinman RE, Coletta FA. Historical overview of transitional feeding recommendations and vegetable feeding practices for infants and young children. Nutr Today. 2016;51(1):7–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.The 2020 Dietary Guidelines Advisory Committee. Scientific Report of the 2020 Dietary Guidelines Advisory Committee. US Department of Agriculture, US Department of Health and Human Services; 2020. [Google Scholar]
- 7.Breastfeeding Rates: National Immunization Survey. Centers for Disease Control and Prevention. Accessed April 1, 2020, https://www.cdc.gov/breastfeeding/data/nis_data/results.html
- 8.Ghandour RM, Jones JR, Lebrun-Harris LA, et al. The design and implementation of the 2016 National Survey of Children’s Health. Matern Child Health J. 2018;22(8):1093–1102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.About the National Health and Nutrition Examination Survey. National Center for Health Statistics. Accessed August 17, 2008, http://www.cdc.gov/nchs/nhanes/about_nhanes.htm
- 10.Wolter KK, Smith PJ, Khare M, et al. Statistical methodology of the National Immunization Survey, 2005–2014. Vital Health Stat. 2017;1(61):1–107. [PubMed] [Google Scholar]
- 11.Chiang KV, Hamner HC, Li R, Perrine CG. Timing of introduction of complementary foods - United States, 2016–2018. MMWR Morb Mortal Wkly Rep. 2020;69(47):1787–1791. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 12.Blanton CA, Moshfegh AJ, Baer DJ, Kretsch MJ. The USDA Automated Multiple-Pass Method accurately estimates group total energy and nutrient intake. J Nutr. 2006;136(10):2594–2599. [DOI] [PubMed] [Google Scholar]
- 13.Moshfegh AJ, Rhodes DG, Baer DJ, et al. The US Department of Agriculture Automated Multiple-Pass Method reduces bias in the collection of energy intakes. Am J Clin Nutr. 2008;88(2):324–332. [DOI] [PubMed] [Google Scholar]
- 14.Ahluwalia N Nutrition monitoring of children aged birth to 24 Mo (B-24): Data collection and findings from the NHANES. Adv Nutr. 2020;11(1):113–127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Dewey K, Finlay D, Lonnerdal B. Breast milk volume and composition during late lactation (7–20 months). J Pediatr Gastroenterol Nutr. 1984;3(5):713–720. [DOI] [PubMed] [Google Scholar]
- 16.Dewey K, Lonnerdal B. Milk and nutrient intake of breast-fed infants from 1 to 6 months: Relation to growth and fatness. J Pediatr Gastroenterol Nut. 1983;2(3):497–506. [DOI] [PubMed] [Google Scholar]
- 17.Kent K Breast volume and milk production during extended lactation in women. Exp Physiol. 1999;84(2):435–447. [PubMed] [Google Scholar]
- 18.Ponza M, Devaney B, Ziegler P, Reidy K, Squatrito C. Nutrient intakes and food choices of infants and toddlers participating in WIC. J Am Diet Assoc. 2004;104(1 suppl 1):71–79. [DOI] [PubMed] [Google Scholar]
- 19.da Costa TH, Haisma H, Wells JC, Mander AP, Whitehead RG, Bluck LJ. How much human milk do infants consume? Data from 12 countries using a standardized stable isotope methodology. J Nutr. 2010;140(12):2227–2232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ahluwalia N, Herrick KA, Rossen LM, et al. Usual nutrient intakes of US infants and toddlers generally meet or exceed Dietary Reference Intakes: Findings from NHANES 2009–2012. Am J Clin Nutr. 2016;104(4):1167–1174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Tooze J, Midthune D, Dodd K, et al. A new statistical method for estimating the usual intake of episodically consumed foods with application to their distribution. J Am Diet Assoc. 2006;106(10):1575–1587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Food and Nutrition Board. Dietary Reference Intakes: Applications in Dietary Assessment. Institute of Medicine; 2000. [Google Scholar]
- 23.National Academies of Sciences, Engineering, and Medicine. Criteria for Selecting the Leading Health Indicators for Healthy People 2030. National Academies Press; 2019. [PubMed] [Google Scholar]
- 24.Rates of any and exclusive breastfeeding by sociodemographics among children born in 2017. National Immunization Survey. Centers for Disease Control and Prevention, Department of Health and Human Services. Accessed October 24, 2021, https://www.cdc.gov/breastfeeding/data/nis_data/rates-any-exclusive-bf-socio-dem-2017.html
- 25.Rossen LM, Simon AE, Herrick KA. Types of infant formulas consumed in the United States. Clin Pediatr. 2016;55(3):278–285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Jones K, Power M, Queenan J, Schulkin J. Racial and ethnic disparities in breastfeeding. Breastfeed Med. 2015;10(4):186–196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Sparks P Racial/ethnic differences in breastfeeding duration among WIC-eligible families. Womens Health Issues. 2011;21(5):374–382. [DOI] [PubMed] [Google Scholar]
- 28.Smithers LG, Kramer MS, Lynch JW. Effects of breastfeeding on obesity and intelligence: Causal insights from different study designs. JAMA Pediatr. 2015;169(8):707–708. [DOI] [PubMed] [Google Scholar]
- 29.Güngör D, Nadaud P, LaPergola CC, et al. Infant milk-feeding practices and food allergies, allergic rhinitis, atopic dermatitis, and asthma throughout the life span: A systematic review. Am J Clin Nutr. 2019;109(suppl 1):772S–799S. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Güngör D, Nadaud P, LaPergola CC, et al. Infant milk-feeding practices and diabetes outcomes in offspring: A systematic review. Am J Clin Nutr. 2019;109(suppl 1):817S–837S. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Gunderson EP, Lewis CE, Lin Y, et al. Lactation duration and progression to diabetes in women across the childbearing years: The 30-year CARDIA study. JAMA Intern Med. 2018;178(3):328–337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Collaborative Group on Hormonal Factors in Breast Cancer. Breast cancer and breastfeeding: Collaborative reanalysis of individual data from 47 epidemiological studies in 30 countries, including 50 302 women with breast cancer and 96 973 women without the disease. Lancet. 2002;360(9328):187–195. [DOI] [PubMed] [Google Scholar]
- 33.Cramer DW. The epidemiology of endometrial and ovarian cancer. Hematol Oncol Clin North Am. 2012;26(1):1–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Kirkegaard H, Bliddal M, Støvring H, et al. Breastfeeding and later maternal risk of hypertension and cardiovascular disease—The role of overall and abdominal obesity. Prev Med. 2018;114:140–148. [DOI] [PubMed] [Google Scholar]
- 35.Ajmera VH, Terrault NA, VanWagner LB, et al. Longer lactation duration is associated with decreased prevalence of non-alcoholic fatty liver disease in women. J Hepatol. 2019;70(1): 126–132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Feltner C, Weber RP, Stuebe A, Grodensky CA, Orr C, Viswanathan M. Breastfeeding programs and policies, breastfeeding uptake, and maternal health outcomes in developed countries. In: Comparative Effectiveness Review, No. 210. AHRQ Publication No. 18-EHC014-EF. Agency for Healthcare Research and Quality; 2018. [PubMed] [Google Scholar]
- 37.Horta BL, Loret de Mola C, Victora CG. Long-term consequences of breastfeeding on cholesterol, obesity, systolic blood pressure and type 2 diabetes: A systematic review and meta-analysis. Acta Paediatr. 2015;104(467):30–37. [DOI] [PubMed] [Google Scholar]
- 38.Dewey KG, Güngör D, Donovan SM, et al. Breastfeeding and risk of overweight in childhood and beyond: A systematic review with emphasis on sibling-pair and intervention studies. Am J Clin Nutr. 2021;114(5):1774–1790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Guendelman S, Kosa J, Pearl M, Graham S, Goodman J, Kharrazi M. Juggling work and breastfeeding: Effects of maternity leave and occupational characteristics. Pediatrics. 2009;123(1):e38–e46. [DOI] [PubMed] [Google Scholar]
- 40.Barraza L, Lebedevitch C, Stuebe A. The Role of Law and Policy in Assisting Families to Reach Healthy People’s Maternal, Infant, and Child Health Breastfeeding Goals in the United States. Department of Health and Human Services, Office of Disease Prevention and Health Promotion; 2020. [DOI] [PubMed]
- 41.Gingras V, Aris IM, Rifas-Shiman SL, Switkowski KM, Oken E, Hivert MF. Timing of complementary feeding introduction and adiposity throughout childhood. Pediatrics. 2019;144(6): e20191320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Roess AA, Jacquier EF, Catellier DJ, et al. Food consumption patterns of infants and toddlers: Findings from the Feeding Infants and Toddlers Study (FITS) 2016. J Nutr. 2018;148(suppl 3):1525S–1535S. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.English LK, Obbagy JE, Wong YP, et al. Complementary feeding and developmental milestones: A systematic review. Am J Clin Nutr. 2019;109(suppl 7):879s–889s. [DOI] [PubMed] [Google Scholar]
- 44.English LK, Obbagy JE, Wong YP, et al. Types and amounts of complementary foods and beverages consumed and growth, size, and body composition: A systematic review. Am J Clin Nutr. 2019;109(suppl 7):956s–977s. [DOI] [PubMed] [Google Scholar]
- 45.English LK, Obbagy JE, Wong YP, et al. Timing of introduction of complementary foods and beverages and growth, size, and body composition: A systematic review. Am J Clin Nutr. 2019;109(suppl 7): 935s–955s. [DOI] [PubMed] [Google Scholar]
- 46.Obbagy JE, English LK, Psota TL, et al. Complementary feeding and micronutrient status: A systematic review. Am J Clin Nutr. 2019;109(suppl 7):852s–871s. [DOI] [PubMed] [Google Scholar]
- 47.Obbagy JE, English LK, Wong YP, et al. Complementary feeding and food allergy, atopic dermatitis/eczema, asthma, and allergic rhinitis: A systematic review. Am J Clin Nutr. 2019;109(suppl 7):890s–934s. [DOI] [PubMed] [Google Scholar]
- 48.Obbagy JE, English LK, Wong YP, et al. Complementary feeding and bone health: A systematic review. Am J Clin Nutr. 2019;109(suppl 7):872s–878s. [DOI] [PubMed] [Google Scholar]
- 49.Abrams S, Daniels S. Fruit juice and child health. Pediatrics. 2017;139(4):e20170041. [DOI] [PubMed] [Google Scholar]
- 50.Auerbach B, Wolf F, Hikida A, et al. Fruit juice and change in BMI: A meta-analysis. Pediatrics. 2017;139(4). 2017;e20162454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Heyman M, Abrams S. Fruit juice in infants, children, and adolescents: Current recommendations. Pediatrics. 2017;139(6): e20170967. [DOI] [PubMed] [Google Scholar]
- 52.American Academy of Pediatrics Committee on Nutrition. The use of whole cow’s milk in infancy. Pediatrics. 1992;89(6):1105–1109. [PubMed] [Google Scholar]
- 53.Nowak-Węgrzyn A, Katz Y, Mehr S, Koletzko S. Non-IgE-mediated gastrointestinal food allergy. J Allergy Clin Immunol. 2015;135(5): 1114–1124. [DOI] [PubMed] [Google Scholar]
- 54.Ziegler E Consumption of cow’s milk as a cause of iron deficiency in infants and toddlers. Nutr Rev. 2011;69(supp1 1):S37–S42. [DOI] [PubMed] [Google Scholar]
- 55.O’Neill LM, Dwyer JT, Bailey RL, Reidy KC, Saavedra JM. Harmonizing micronutrient intake reference ranges for dietary guidance and menu planning in complementary feeding. Curr Dev Nutr. 2020;4(3):nzaa017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Eldridge AL, Catellier DJ, Hampton JC, Dwyer JT, Bailey RL. Trends in mean nutrient intakes of US infants, toddlers, and young children from 3 Feeding Infants and Toddlers Studies (FITS). J Nutr. 2019;149(7):1230–1237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Bailey RL, Jun S, Eldridge AL. The 2016 Feeding Infants and Toddlers Study (FITS): Dietary intakes and practices of children in the United States from birth to 48 months. Nestle Nutr Inst Workshop Ser. 2019;91:99–109. [DOI] [PubMed] [Google Scholar]
- 58.Bailey RL, Ard JD, Davis TA, et al. A proposed framework for identifying nutrients and food components of public health relevance in the Dietary Guidelines for Americans. J Nutr. 2021;151(5):1197–1204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Viteri FE. Iron endowment at birth: Maternal iron status and other influences. Nut Rev. 2011;69(suppl 1):S3–S16. [DOI] [PubMed] [Google Scholar]
- 60.Abrams SA, Hampton JC, Finn KL. A substantial proportion of 6- to 12-month-old infants have calculated daily absorbed iron below recommendations, especially those who are breastfed. J Pediatr. 2021;231:36–42.e32. [DOI] [PubMed] [Google Scholar]
- 61.National Academies of Sciences, Engineering, and Medicine; Health and Medicine Division; Food and Nutrition Board; Committee on Scanning for New Evidence on the Nutrient Content of Human Milk, Vorosmarti A, Yaktine AL, Rasmussen K, eds. Scanning for New Evidence on the Nutrient Content of Human Milk: A Process Model for Determining Age-Specific Nutrient Requirements. National Academies Press; 2020. [PubMed] [Google Scholar]
- 62.Johnston JD, Ordovas JM, Scheer FA, Turek FW. Circadian rhythms, metabolism, and chrononutrition in rodents and humans. Adv Nutr. 2016;7(2):399–406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Hahn-Holbrook J, Saxbe D, Bixby C, Steele C, Glynn L. Human milk as “chrononutrition”: Implications for child health and development. Pediatr Res. 2019;85(7):936–942. [DOI] [PubMed] [Google Scholar]
- 64.About HALF: Healthy Active Living for Families (HALF) Program. American Academy of Pediatrics. Published 2012. Accessed 2017, https://www.aap.org/en-us/advocacy-and-policy/aap-health-initiatives/HALF-Implementation-Guide/Pages/About-HALF.aspx
- 65.Bailey RL, Catellier DJ, Jun S, et al. Total usual nutrient intakes of US children (<48 months): Findings from the Feeding Infants and Toddlers Study (FITS) 2016. J Nutr. 2018;148(9S):1557S–1566S. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Fisher JO, Butte NF, Mendoza PM, et al. Overestimation of infant and toddler energy intake by 24-h recall compared with weighed food records. Am J Clin Nutr. 2008;88(2):407–415. [DOI] [PubMed] [Google Scholar]
- 67.Gillespie B, d’Arcy H, Schwartz K, Bobo J, Foxman B. Recall of age of weaning and other breastfeeding variables. Int Breastfeed J. 2006;1:4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Sorce LR, Schoeny ME, Curley MAQ, Meier PP. Are mothers certain about their perceptions of recalled infant feeding history? J Pediatr Health Care. 2021;35(2):156–162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Dewey KG, Pannucci T, Casavale KO, et al. Development of food pattern recommendations for infants and toddlers 6–24 months of age to support the Dietary Guidelines for Americans, 2020–2025. J Nutr. 2021;151(10):3113–3124. [DOI] [PubMed] [Google Scholar]


