Abstract
Osteoporosis (OP) is a bone disease associated with increasing age. Currently, the most common medications used to treat OP are anabolic agents, anti-resorptive agents, and medications with other mechanisms of action. However, many of these medications have unfavorable adverse effects or are not intended for long-term use, potentially exerting a severe negative impact on a patient’s life and career and placing a heavy burden on families and society. There is an urgent need to find new drugs that can replace these and have fewer adverse effects. Quercetin (Que) is a common flavonol in nature. Numerous studies have examined the therapeutic applications of Que. However, a comprehensive review of the anti-osteoporotic effects of Que has not yet been conducted. This review aimed to describe the recent studies on the anti-osteoporotic effects of Que, including its biological, pharmacological, pharmacokinetic, and toxicological properties. The outcomes demonstrated that Que could enhance OP by increasing osteoblast differentiation and activity and reducing osteoclast differentiation and activity via the pathways of Wnt/β-catenin, BMP/SMAD/RUNX2, OPG/RANKL/RANK, ERK/JNK, oxidative stress, apoptosis, and transcription factors. Thus, Que is a promising novel drug for the treatment of OP.
Keywords: quercetin, antiosteoporosis, pharmacokinetics, toxicology, osteoblast, osteoclast
1 Introduction
Osteoporosis (OP) is the most prevalent type of systemic bone disease and is characterized by decreased bone mass and damage to the microstructure of the bone tissue, leading to increased bone fragility (Kanis et al., 2019). Patients with OP are more prone to fractures. Over 200 million people worldwide have OP (Palacios, 2022). The lifetime risk of developing the disease in Caucasian women aged >50 years is 50%, and OP-induced fractures can result in substantial medical expenses (up to $25.3 billion by 2025) and impaired physical function (Armas and Recker, 2012).
Nearly all clinical medicine preparations are derived from natural compounds found in plants, animals, insects, marine organisms, and microbes (Harvey et al., 2015). Among them, flavonoids (Rodríguez et al., 2022), alkaloids (Lin et al., 2022), polysaccharides (Lei et al., 2021), quinones (Geng et al., 2019), terpenoids (Bellavia et al., 2021), lignans (Jang et al., 2022), saponins (Siddiqi et al., 2013), polyphenols (Tao et al., 2021), and amino acids (Ling et al., 2021) have been shown to have the ability to inhibit bone resorption and promote bone formation. Quercetin (Que) is a flavonoid compound widely found in traditional Chinese medicines, such as yam bean root, Scutellaria baicalensis, and Sophora japonica. Its chemical name is 3,3′,4′,5,7 pentahydroxyflavonoid, molecular formula is C15H10O7, and relative molecular mass is 302.236. The appearance is of a yellow acicular crystalline powder, slightly soluble in water, and readily soluble in an alkaline aqueous solution (Yang et al., 2020). Numerous therapeutic effects, such as osteoprotective (Xiao et al., 2023), neuroprotective (Fideles et al., 2023), antiallergic (Mlcek et al., 2016), anti-inflammatory (Hou et al., 2019), anticancer (Rauf et al., 2018), cardiovascular protection (Patel et al., 2018), antiviral (Di Petrillo et al., 2022), antidiabetic (Eid and Haddad, 2017), immunomodulatory (Wang et al., 2021), antihypertensive (Luo et al., 2017), and gastroprotective (Martín et al., 1998), have been reported. Que inhibits bone resorption and promotes bone formation. We reviewed the anti-osteoporosis effects of quercetin and its mechanisms, and summarized its application at the cellular level and in animal experiments in Table 1 and Table 2 respectively.
TABLE 1.
Use of quercetin in the prevention and treatment of osteoporosis at the cellular level.
| Type of cell | Treatment | Findings | References |
|---|---|---|---|
| MSCs | Que (20 μM) + dasatinib (10 nM) | RAD51↑ | Wang et al. (2023) |
| SASP↓ | |||
| MC3T3-E1 induced by iron overload | Que (2.5, 5 μM) | RUNX2, Osterix, ALP, calcium nodules, Bcl-2, SOD, Nrf2, HO-1↑ | Xiao et al. (2023) |
| Bax, Caspase3, ROS, MDA↓ | |||
| BMSCs | Que (5, 25 µM) | RUNX2, ALP, OCN, BMP, β-catenin, Malat1, mineralized nodule↑ | Feng et al. (2023) |
| NF-κB, p65↓ | |||
| hBMSCs | curcumin (1 µM)+ polydatin (10 µM)+quercetin (0.5 µM) | RUNX2, COL1α1, OCN, ALP, miR-98↑ | Giordani et al. (2023) |
| IL-1β, MCP-1, miR-21, miR-146a, p38-MAPK, p-NF-κB↓ | |||
| Senescent BMSCs | Que (20 μM) | calcium nodules↑ | Xing et al. (2023) |
| SA-β-gal, P16, P21, γH2A, X protein, MCP-1, Cxcl-1, IL-1β, IL-6 ↓ | |||
| Raw 264.7 cells | Que (2 μg/mL) | TRAP, CTSK, NFATC-1, ATG5, p62, mTOR, Beclin-1↓ | Liu (2022) |
| hBMSCs | Que (0–10 μmol/L) | cell proliferation, ALP, RUNX2, BGLAP, SPP1, SATB2 ↑ | Zhang et al. (2022) |
| RANKL treated RAW 264.7 | Que (200 μM) | cell proliferation, TRAP↓ | Zheng et al. (2022) |
| BMSCs | Que (10 μmol/L) | cell proliferation, ALP, BMP–2, RUNX2, osteocalcin↑ | Bian et al. (2022) |
| DANCR↓ | |||
| MC3T3-E1 | Que (10 μmol/L) | cell proliferation, ALP, calcium deposition↑ | Ma et al. (2023) |
| MSCs | Quercetin (0.4–0.6 μg/mL) (in Ti/Gel/CaCO3-Qu-Cr) | ALP, mineralization node formation, P2X7, RUNX2, OSX, OCN, BMP2, Col I, intracellular calcium, ACP↑ | Chen et al. (2022) |
| ROS↓ | |||
| mBMSCs | Que (2, 5 μM) | SOD1, SOD2, p-AMPK, SIRT1, ALP, RUNX2, BMP2, SP7↑ | Wang et al. (2021) |
| PPARG, FABP4↓ | |||
| BMSCs | Que (10 μmol/L) | cell proliferation, ALP, RUNX2↑ | Li et al. (2020) |
| PPARγ, C/EBPα↓ | |||
| ICI ICI182780 (10 μmol/L) + Que (10 μmol/L) | PPARγ, C/EBPα, RUNX2↑ | ||
| ALP↓ | |||
| BMSCs | Que (5, 10 μmol/L) | ALP, BMP-2↑ | Bian et al. (2016) |
| MG-63 cells | Que (0.2 ppm) | cell proliferation, ALP, collagen production, OPG↑ | Ruangsuriya et al. (2020) |
| RANKL↓ | |||
| hBMSCs | quercetin 3-O-β-D-galactopyranoside (25 μM) | cell proliferation, ALP, Extracellular Mineralization calcification, RUNX2, osteorix, OCN, OPN, β-catenin, BMP2, Wnt10a, Wnt10b, Smad1/5, p-ERK↑ | Oh et al. (2020) |
| perilipin-1, PPARγ, CEBPα, SREBP1c, p-p38, p-JNK, p-c-Fos, p-c-Jun ↓ | |||
| BMSCs | Que (1 μmol/L) | calcium nodules, RUNX2, Osterix, β-catenin↑ | Yuan et al. (2018) |
| NF-κB↓ | |||
| hBMSCs | Que (10 μM) | γ-catenin↑ cell proliferation, ALPL, COL1A1, OCN, β-catenin↓ | Casado-Díaz et al. (2016) |
| BMSCs | Que (5 μM) | p-Akt↑ | Masuhara et al. (2016) |
| TRAP, GPR30↓ | |||
| MC3T3-E1 | Que (200 μM) | cell proliferation, ALP, RUNX-2, col - 1, ALP, BSP, OC, ARS↑ | Tripathi et al. (2015) |
| osteoblasts | Que aglycone (20 μM) | HO-1, GCLC, CAT, p-ERK1/2, NF-κB p65↑ | Messer et al. (2015) |
| MC3T3-E1 | Que (10 μmol/L) | cell proliferation, ALP↑ | Mu et al. (2015) |
| BMSCs | Que (4 μmol/L) | cell proliferation, p-ERK↑ | Chen et al. (2014) |
| Caco-2 cells | Que (100 μM) | VDR, VDR-LBD, TRPV6↑ | Inoue et al. (2010) |
| ROB | Que (0.1, 1, 10 mmol/L) | cell proliferation, mineralized nodule ↑ | Yang et al. (2006) |
| RAW 264.7 cells | Que (0.1–10 μM) | TRAP, OCL number, NF-κB, AP-1↓ | Wattel et al. (2004) |
| peripheral blood monocytic cells | bone resorption, OCL number↑ | ||
| MG-63 human osteoblasts | Que (1, 10, 50 μM) | ALP, p-ERK↑ | Prouillet et al. (2004) |
| Rat osteoclasts | Que (2.5, 10, 40 mmol/L) | cell apoptosis ↑ cell proliferation↓ | Chen and Wei (2002) |
MSCs, mesenchymal stem cells; SASP, senescence-associated secretory phenotype; MC3T3-E1, mouse embryo osteoblast precursor cells; Que, quercetin; RUNX2, Runt-related transcription factor 2; ALP: alkaline phosphatase calcium nodules; Bcl-2, B-cell lymphoma-2; SOD, superoxide dismutase; Nrf2, NF-E2-related factor 2; HO-1:heme oxygenase 1; Bax, Bcl-2, associated X; ROS, reactive oxygen species; MDA, malondialdehyde; BMSCs, bone mesenchymal stem cells; BMP, bone morphogenetic protein; NF-κB, nuclear factor kappa-B; hBMSCs, human bone marrow mesenchymal stem cells; COL1α1, collagen type I alpha 1; miR-98, MicroRNA-98; OCN, osteocalcin; IL-1β, Interleukin-1β; MCP-1, Monocyte Chemoattractant Protein-1; miR-21, MicroRNA-21; miR-146a, MicroRNA-146a; p38-MAPK, p38 Mitogen-Activated Protein Kinase; p-NF-κB, phosphorylated NF-κB; SA-β-gal, a senescence associated beta-galactosidase; P16: multiple tumor suppressor 1; Cxcl-1, CXC-chemokine Ligand-1; TRAP: translocon-associated protein; CTSK, Cathepsin K; NFATC-1, Nuclear Factor Of Activated T-cells, And Cytoplasmic 1; ATG5, Autophagy-related Protein 5; mTOR, mammalian target of rapamycin; BGLAP, osteocalcin; SPP1, secreted phosphoprotein 1; SATB2, Special AT-rich Sequence-binding Protein 2; RANKL, Receptor Activator of NF-KappaB ligand; DANCR, Differentiation Antagonizing Non-protein Coding RNA; P2X7, P2X Ligand-gated Ion Channel 7; Col I, Collagen I; ACP, acid phosphatase; SIRT1, Sirtuin 1; PPARG, peroxisome proliferator-activated receptor gamma; FABP4, Fatty Acid Binding Protein 4; PPARγ, Peroxisome Proliferator-activated Receptor-γ; C/EBPα, CCAAT/enhancer binding proteins alpha; Wnt10a, Wnt Family Member 10a; Wnt10b, Wnt Family Member 10b; p-ERK: Phosphorylated Extracellular Signal-regulated Kinase; SREBP1c, Sterol regulatory element binding protein 1; p-p38, Phosphorylated P38; p-JNK, Phosphorylated C-Jun N-terminal Kinase; p-c-Fos, Phosphorylated c-Fos; p-c-Jun, Phosphorylated C-Jun; hBMSCs, human bone marrow stromal cells; ALPL, alkaline phosphatase, liver; GPR30, G Protein-coupled Receptor 30; ARS, alizarin red-S; GCLC, glutamate cysteine ligase; CAT, catalase; VDR, Vitamin D Nuclear Receptor; VDR-LBD, VDR-ligand-binding Domain; ROB, rat calvarialosteoblasts; TRPV6, transient receptor potential cation channel, subfamily V, member 6; AP-1, Activator Protein-1.
TABLE 2.
Animal experiments on use of quercetin to prevent osteoporosis.
| Animal models | Quercetin dose | Findings | References |
|---|---|---|---|
| OVX-induced osteoporosis mouse model | 50 mg/(kg·d) | BV/TV, BMD, Tb.Th, Tb.N↑ | Feng et al. (2023) |
| Tb.Sp↓ | |||
| the iron overload-induced osteoporosis mice model | 50, 100 mg/(kg·d) | BV/TV, Tb.Th, Tb.N↑ | Xiao et al. (2023) |
| SMI↓ | |||
| OVX rats | Dasatinib (5 mg/(kg·d)) + Que (50 mg/(kg·d)) | femur trabecular bone microarchitecture, BV/TV, trabecular number↑ | Wang et al. (2023) |
| Tb.Sp, SMI, SnC, p16, p53↓ | |||
| radiated (24Gy) C57BL/6 male mice | Dasatinib (5 mg/kg·d) + Que (50 mg/(kg·d)) | BV/TV, Conn. Dens, MS/BS,BFR/BS, P1NP↑ | Chandra et al. (2022) |
| SMI↓ | |||
| OVX rats | CaCO3-Qu-Cr (0.4 mg/mL) | BV/TV, Tb.Th, Tb.N, RUNX2, Col I, OCN↑ | Chen et al. (2022) |
| ROS, TRAP↓ | |||
| retinoic acid-induced bone loss model of rats | Que (100 mg/kg·d) +13cRA (80 mg/(kg·d)) | Ash content in femur, Ca content in femur, P content in femur, Femoral bone weights, Femur length, BMD, Glutathione content↑ | Oršolić et al. (2014) |
| MDA↓ | |||
| STZ-NA induced diabetic rats | 50 mg/(kg·d) | BMD, Tb.Bv/Tb.Tv, Tb.N, Tb.Th, Ct.Th, Tb.Sp↑ | Baş and Albeniz (2022) |
| SMI↓ | |||
| Osteoporosis in Orchiectomy Mice | 75, 150 mg/(kg·d) | bone mass, bone strength, bone microstructure, stride length and frequency, insulin-like growth factor-1, high-density lipoprotein, GPRC6A, phospho-AMPK/AMPK↑ | Sun et al. (2022) |
| phospho-mTOR/mTOR ↓ | |||
| OVX rats | 50 mg/(kg·d) | urine-Ca, urine-P, BMD, Tb.N, Tb.Th, BV/TV↑ | Min et al. (2022) |
| dexamethasone-induced zebrafish osteoporosis model | 12.5, 25 µM | BMA, IOD↑ | Hu et al. (2022) |
| TNF-α, IL-1β, p65↓ | |||
| OVX rats | 15 mg/(kg·d) | serum calcium, bone weight, bone volume, trabeculae volume, the total number of osteocytes and osteoblasts, LC3, beclin1, caspase 3 ↑ | Vakili et al. (2021) |
| total number of osteoclasts, serum osteocalcin, Bcl-2 ↓ | |||
| Disuse osteoporosis model in rats | 100, 200 mg/(kg·d) | Bone mineral density, trabecular number, trabecular thickness of femur↑ trabecular bone dispersion, ERK1/2, MAPK↓ | Wang et al. (2020) |
| Osteoporosis model in rats with low calcium and high magnesium diet | 2.64 μg/(kg·d) | serum-Ca, serum-P, BALP↑ | Deng et al. (2020) |
| serum-Mg, BGP, CTX1, tPINP↓ | |||
| GIO rats | quercetin-loaded transfersomes (10 mg/(kg·d)) | femur thickness, density, length, weight, tensile strength of femur bone, serum-Ca, serum-P, ALP↑ | Pandit et al. (2020) |
| TRAP↓ | |||
| OVX rats | 50 mg/(kg·d) | Urine-P, urine-Ca, ALP, BMD, Tb.N, Tb.Th, BV/TV↑ | Yuan et al. (2018) |
| OVX rats | 100, 200 mg/(kg·d) | BMD, the maximum fracture loading femoral midshaft, fracture loading, stiffness, energy absorption, serum-Ca, serum-P, ALP, P1NP, Osx, RUNX2↑ | Xing et al. (2017) |
| CTX-1, TRAP, intertrabecular space, p-JNK, p-P38, p-ERK↓ | |||
| OVX rats | 50, 100, 200 mg/(kg·d) | E2, BMD, U-Ca, U-P, OC, PINP, BALP, BMP2, Smad4↑ | Zheng et al. (2017) |
| CTX, TRACP-5b↓ | |||
| OVX rats | 100, 200 mg/(kg·d) | Type I collagen↑ osteocalcin ↓ | Feng et al. (2016) |
| Glucocorticoid-induced osteoporosis (GIO) rats | 50, 150 mg/(kg·d) | bone strength, osteocalcin, femoral trabecular, cortical thickness, osteoblast number↑ | Derakhshanian et al. (2013) |
| OVX rats | quercetin-6-C-A-D-glucopyranoside (1, 5 mg/(kg·d)) | B.Ar, Cs.Th, T.Ar, T.Pm, B.Pm, B.Pm-T.Ar↑ serum osteocalcin, urinary CTx↓ | Siddiqui et al. (2011) |
| 1, 5 mg/(kg·d) | B.Ar, Cs.Th, T.Ar, T.Pm, B.Pm, B.Pm-T.Ar↑ serum osteocalcin, urinary CTx↓ | ||
| OVX rats | 75, 150, 300 mg/(kg·d) | ELASTIC, M-STRESS, MLORD, OPG↑ | Wang (2009) |
| RANKL↓ | |||
| OVX rats | 75, 150, 300 mg/(kg·d) | BMD↑ | Wang (2008) |
| OVX rats | 50, 100, 200 mg/(kg·d) | BMD, BMC↑ | Zhu and Wei (2005) |
| ACP↓ |
OVX, ovariectomized; BV/TV, Bone Volume/Total Volume; BMD, bone mineral density; Tb.Th, Trabecular Thickness; Tb.N, trabecular number; Tb.Sp, Trabecular Separation/Spacing; SMI, skeletal muscle mass index; SnC, senescent cell; p16, multiple tumor suppressor 1; Conn.Dens, connectivity density; MS/BS, mineralizing surface per bone perimeter; BFR/BS, bone formation rate per bone surface; P1NP, Procollagen Type 1 N-terminal Propeptide; RUNX2, Runt-related transcription factor 2; Col I, Collagen I; OCN: osteocalcin; ROS: reactive oxygen species; TRAP, translocon-associated protein; MDA, malondialdehyde; STZ-NA, streptozotocin nicotinamide; Tb.Bv/Tb.Tv, Trabecular volume fraction; Ct.Th, Cortical bone Thickness; GPRC6A, G Protein-coupled Receptor Family C, Group 6, Subtype A; AMPK, AMP-activated Protein Kinase; mTOR:mammalian target of rapamycin; TNF-α, Tumor Necrosis Factor-α; IL-1β, Interleukin-1β; Bcl-2, B-cell lymphoma-2; ERK1/2, Extracellular Signal-Regulated Kinases 1/2; MAPK, Mitogen-Activated Protein Kinase; BALP, bone alkaline phosphatase; BGP, bone gamma-carboxyglutamic-acid-containing proteins; CTX1, type1 collagen; tPINP, N-terminal Peptide; ALP, alkaline phosphatase; TRAP, Tartrate-Resistant Acid Phosphatase; Osx:Osterix; BALP, bone alkaline phosphatase; BMP2, Bone Morphogenetic Protein 2; TRACP-5b, Tartrate-resistant Acid Phosphatase 5b; B.Ar, bone area; Cs.Th, Cortical thickness; T.Ar, trabecular area; T.Pm, Cross-sectional tissue perimeter; B.Pm, Cross-sectional bone perimeter; M-STRESS, maximum stress; M-LORD, mazimum load; BMC, bone mineral content; ACP, acid phosphatase.
2 Biological characteristics of Que
2.1 Sources of quercetin
Que exists in the flowers, leaves, and fruits of many plants (Wang et al., 2016; Kandemir et al., 2022), mostly in the form of glycosides (Crespy et al., 2001), such as rutin (rutinoside), chrysin, and other plants with high content. Que is a bioactive flavonol found mainly in frequently consumed plant foods such as onions, apples, berries, and broccoli (David et al., 2016). Many herbs, including Ginkgo biloba, mulberry leaf, cuscuta, golden buckwheat, forsythia, Panax ginseng, and Fritillaria, contain Que (Shao et al., 2023). Que exists in the skin and leaves of the Iberian oak Quercus iberica of the family Crustacea, the red octopus of the family Berberidaceae, the red drought lily (Hunan forsythia) of the family Hypericum, and the red flax leaves of the family Oleaceae.
2.2 Physicochemical properties of Que
Que is a yellow needle-like crystal (Zou et al., 2021), mostly in the form of glycosides, and can be obtained by acid hydrolysis. Que dihydrate appears as yellow needle-like crystals (dilute ethanol), becomes anhydrous at 95°C–97°C, and has a melting point of 314°C (decomposition). Que is soluble in cold ethanol (1:290), hot ethanol (1:23), methanol, ethyl acetate, glacial acetic acid, pyridine, and acetone. However, it is insoluble in water, benzene, ethyl ether, chloroform, and petroleum ether. The alkaline aqueous solution is yellow and almost insoluble in water, and the ethanol solution tastes very bitter (Wang et al., 2016). The structure of Que is shown in Figure 1.
FIGURE 1.
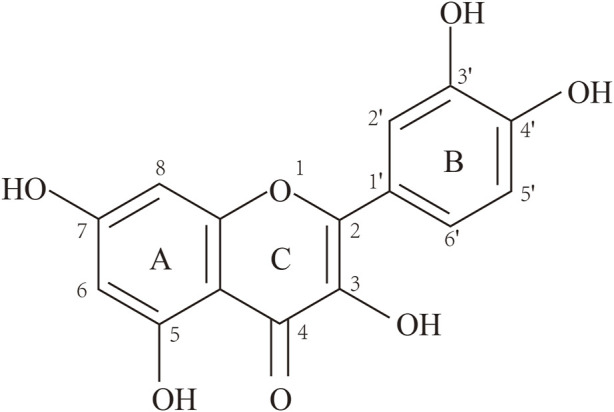
Structure of quercetin (3,3′,4′,5,7-pentahydroxyflavone).
2.3 Biosynthesis of Que
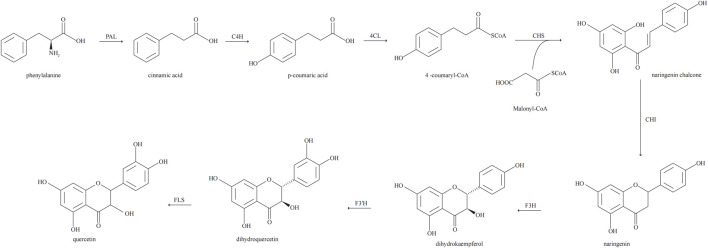
Biosynthesis of Que via the phenylpropyl metabolic pathway. In the biosynthesis of Que, phenylalanine derived from the mangiferolic acid pathway is used as the initial precursor to generate cinnamic acid by removing the amino group, catalyzed by phenylalanine ammonia-lyase. Cinnamic acid acquires a hydroxyl group through the catalytic action of cinnamate 4-hydroxylase to form p-coumaric acid. p-Coumaric acid undergoes a thioesterification reaction catalyzed by coumaryl 4-ligase to produce p-coumaroyl coenzyme A. Condensation of one molecule of p-coumaroyl coenzyme A and three molecules of malonyl coenzyme A catalyzed by chalcone synthase produces naringenin chalcone. Naringenin is catalyzed by flavanone 3-hydroxylase to form dihydrokaempferol. Flavanone 3′-hydroxylase is hydroxylated by dihydrokaempferol to form dihydroquercetin. Finally, dihydroquercetin is catalyzed by flavonol synthase for Que biosynthesis (Lakhanpal P and Rai, 2007; Alrawaiq and Abdullah, 2014; Nabavi et al., 2020). The biosynthetic pathway of Que is shown in Figure 2.
FIGURE 2.
Biosynthetic pathway of quercetin.
3 Molecular mechanisms of Que in OP
3.1 Mechanism by which quercetin promote osteoblast-mediated bone formation
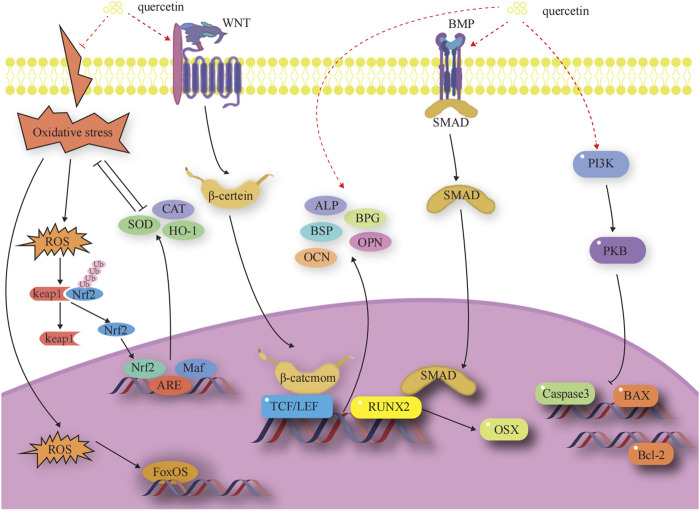
Osteoblasts (OBs) undergo four stages of bone formation: proliferation, extracellular matrix maturation, extracellular matrix mineralization, and apoptosis (Marcus and Majumder, 2001). They are affected by a variety of transcription factors and signaling pathways during various stages of development to ultimately complete normal bone formation. The factors and signaling pathways by which Que influences OB-mediated bone formation are outlined below. A diagram of the mechanisms involved in this part of the study are shown in Figure 3.
FIGURE 3.
Effects and molecular mechanisms of quercetin induction on osteoblasts. (Little et al., 2002; Nakashima et al., 2002; Steitz et al., 2002; Zhang et al., 2003; Ohri et al., 2005; Kim et al., 2007; Chen et al., 2008; Nusse et al., 2008; Clevers et al., 2014; Jin et al., 2016; An and Shang, 2018; Gao et al., 2018; Zhang et al., 2019; Kim et al., 2021; Kimball et al., 2021; Li et al., 2021; Yu et al., 2021).
3.1.1 OB-specific transcription factors
OB-specific Runt-associated transcription factor 2/core-binding factor alpha 1 and special protein 7 transcription factor (osterix) are required for the differentiation of mesenchymal stem cells (MSCs) to OBs and for the formation of functional OBs. The RUNX2 gene is the most critical transcription factor that regulates the differentiation of bone marrow mesenchymal stem cells (BMSCs) into OBs and the maturation of OBs in the process of bone development (Zhang et al., 2003). Expression of the RUNX2 gene is a marker for the onset of OB differentiation, making it the earliest and most specific gene in the process of bone formation. Deletion of this gene will result in hypoplasia or termination of bone development. Osterix acts as a downstream gene of RUNX2 to play an osteogenic role and is expressed exclusively in the cells of the bone tissue, which is required for the differentiation of OBs and for bone formation (Gao et al., 2018). (Figure 3)
Yang (2022) treated mouse bone marrow mesenchymal stem cells (mBMSCs) with Que and found that 5 μM Que significantly upregulates the expression levels of RUNX2 and SP7 mRNA. Xiao et al. (2023) used ferric ammonium citrate (FAC, 200 μM) to construct an iron overload environment. Iron deposition inhibits osteogenic differentiation of MC3T3-E1 cells and suppresses the expression of RUNX2 and Osterix. Iron-overloaded mice have reduced bone mass, loose trabeculae, and thinner bone marrow cavities. Que (2.5 or 5 μM) can rescue the proliferation inhibition of MC3T3-E1 cells induced by FAC and prevent the reduction of RUNX2 and Osterix expression. This action, in turn, rescues the dysfunction of osteogenic differentiation and attenuates the bone loss of MC3T3-E1 cells induced by iron overload.
3.1.2 Wnt/β-catenin signaling pathway
The classical Wnt signaling pathway is an important regulator of bone resorption and formation (Nusse et al., 2008; Clevers et al., 2014). The Wnt signaling pathway is activated when Wnt ligands bind to the heterodimeric receptor molecules of the frizzled protein family and low-density lipoprotein receptor-related protein 5 (LRP5). Subsequently, the Wnt protein binds to the cell surface receptors of the frizzled protein family, which activates the disheveled protein family and the downstream factor GSK-3β. This activation maintains β-catenin in a stable state in the cytoplasm. Eventually, β-catenin enters into the nucleus and interacts with the transcription factor Tcf/Lef to activate the expression of the target genes of Wnt signaling (Little et al., 2002; Nakashima et al., 2002). Dickkopf-1, a secreted glycoprotein, antagonizes Wnt/β-catenin signaling pathway activity by competitively binding to LRP5/6 receptors with Wnt proteins, thereby promoting bone destruction. (Figure 3).
Que promotes osteogenic differentiation of MC3T3-E1 cells by increasing β-catenin protein levels and activating the Wnt/β-catenin pathway (Guo et al., 2017). miR-625-5p expression upregulation or H19 expression downregulation suppresses β-catenin protein levels, and Que promotes the proliferation and osteogenic differentiation of BMSCs by targeting the H19/miR-625-5p axis to activate the downstream Wnt/β-catenin pathway (Bian et al., 2021). However, a high concentration of Que (10 μmol/L) inhibits the Wnt/β-catenin pathway. It downregulates the expression of the cyclin D1 gene involved in the G1/S cell cycle transition in stem cells. Additionally, it decreases the nuclear β-catenin level in undifferentiated MSCs as well as in MSCs induced to differentiate into OBs. The inhibition of the Wnt/β-catenin pathway results in the inhibition of OB formation in MSCs and promotes adipogenesis (Casado-Díaz et al., 2016).
3.1.3 BMP-2/SMADs/RUNX2 signaling pathway
The BMP-2/Smad signaling pathway affects OB differentiation and bone formation (Yu et al., 2021). After the release of BMP-2 in autocrine or paracrine forms, its monomer can form dimers through disulfide bonding. These dimers bind to BMP receptors, regulating the transcription of osteogenic genes through downstream Smad signaling. This process results in the upregulation of RUNX2 and OSX, promoting osteoclast (OC) proliferation and differentiation (Kim et al., 2021). (Figure 3).
Que (2 or 5 μM) increases the relative alkaline phosphatase (ALP) activity and matrix mineralization of mBMSCs and significantly upregulates the mRNA levels of ALP, RUNX2, BMP2, and other OB marker genes, thus promoting the osteogenic differentiation of the third-generation mBMSCs (Yang, 2022). Que enhances the activation of the BMP signaling pathway through the endoplasmic reticulum, upregulates the expression of downstream genes, such as OSX, RUNX2, and OPN, and promotes the proliferation and osteogenic differentiation of BMSCs; crosstalk between BMP-2 and estrogen receptor signaling pathways was experimentally confirmed. The upregulation of RUNX2, OSX, and OPN gene expression by Que and estrogen is inhibited after the addition of ICI182780, an estrogen receptor antagonist (Pang et al., 2018).
3.1.4 Anti-apoptosis-mediated pathways
Apoptosis is a process by which cells respond to physiological and pathological stimulation signals from the environment, changes in environmental conditions, or palliative damage. The Bcl family plays a key role in promoting or inhibiting the intrinsic apoptotic pathway triggered by mitochondrial dysfunction (Zhang et al., 2019). Bcl-2 and Bax are antagonistic proteins involved in the regulation of apoptosis. When Bcl-2 is highly expressed, it promotes the formation of Bax/Bcl-2 heterodimers and inhibits the formation of Bax/Bax homodimers, preventing the release of pro-apoptotic factors, such as cytochrome c. When Bax proteins are highly expressed, they increase the formation of Bax/Bax homodimers and inhibit the formation of Bax/Bax heterodimers. This leads to the activation of the expression of downstream caspase family proteins, promoting the release of apoptotic factors and initiating apoptosis (Chen et al., 2008). (Figure 3).
Que can reduce FAC-induced apoptosis and reactive oxygen species (ROS) production, downregulate the expression of caspase-3 and Bax, and upregulate the expression of Bcl-2 (Xiao et al., 2023; Zhu et al., 2023). Que can attenuate chondrocyte apoptosis by modulating the Bcl-2/Bax-caspase-3 signaling pathway to reduce sodium nitroprusside-induced overproduction of intracellular ROS and restore mitochondrial membrane potential (Hu et al., 2019).
3.1.5 Oxidative stress-mediated pathways
Oxidative stress is a pivotal factor that contributes to the functional uncoupling of OBs and OCs in OP. When a redox imbalance exists in cells, free Nrf2 binds to antioxidant response elements in the nucleus, thereby activating the expression of detoxification genes. Heme oxygenase 1 (HO-1), a well-known phase II detoxification enzyme, inhibits cytotoxicity originating from a variety of oxidative stresses and inflammatory responses, significantly balancing redox homeostasis (Jin et al., 2016). Under oxidative stress, cellular defense mechanisms against oxidative damage are enhanced by upregulating Nrf2 and HO-1 expression (An and Shang, 2018). Members of the forkhead box class O protein (FOXO) family are activated when intracellular reactive oxygen species are increased. FOXO binds β-catenin to shift transcription mediated by the transcription factor T cell factor/lymphocyte enhancer factor in the Wnt pathway toward FOXO transcription to increase the expression of antioxidant enzymes, such as superoxide dismutase (SOD) and catalase (CAT), to attenuate oxidative stress damage, attenuate osteogenic differentiation of BMSCs, and reduce OB bone formation (Kimball et al., 2021). (Figure 3).
Que dose-dependently upregulates HO-1 mRNA expression in BMSCs (Papiez et al., 2008). It significantly enhances Nrf2 nuclear translocation in FAC-induced MC3T3-E1 cells and attenuates FAC-induced oxidative stress injury by activating the Nrf2/HO-1 signaling pathway (Xiao et al., 2023). Que downregulates ROS levels and upregulates the expression of antioxidant genes (Nrf2, CAT, SOD-1, and SOD-2) in BMSCs stimulated by H2O2 in vitro, maintaining the viability of BMSCs and OB differentiation (Lan et al., 2022).
3.1.6 Promotion of bone matrix formation and mineralization
Three non-collagenous proteins, namely, bone salivary protein (BSP), osteopontin (OPN), and osteocalcin (OCN), are essential for the formation and maturation of mineralized tissues. BSP is an effective nucleating agent for hyaluronic acid formation in stabilized agarose gel systems (Steitz et al., 2002). The presence of aspartic acid and phosphoserine in large quantities in the OPN molecule facilitates its binding to the surface of calcium-phosphorus crystals in mineralized tissues and inhibits the calcification and growth of these crystals (Ohri et al., 2005; Li et al., 2021). OCN is involved in the regulation of bone resorption and participates in matrix mineralization processes and OB differentiation. It is associated with bone turnover, maintains the normal rate of bone mineralization, inhibits the rate of cartilage mineralization, and inhibits the formation of abnormal hydroxyapatite crystals in the bone (Kim et al., 2007). (Figure 3).
Que increases BSP transcription in OB-like cells by targeting the reversed CCAAT and FRE elements in the proximal BSP gene promoter (Hunter and Goldberg, 1993). Treatment with Que significantly upregulates the expression of OCN and OPN mRNA in BMSCs, increases the number of mineralized nodules and the accumulation of mineralized matrix in BMSCs, and promotes the osteogenic differentiation of BMSCs (Zhang et al., 2020). Pretreatment with Que significantly restores bone mineralization and OCN mRNA and protein expression levels in lipopolysaccharide (LPS)-inhibited MC3T3-E1 cells in a dose-dependent manner (Guo et al., 2017).
3.2 Mechanism by which Que inhibits OC-mediated bone resorption
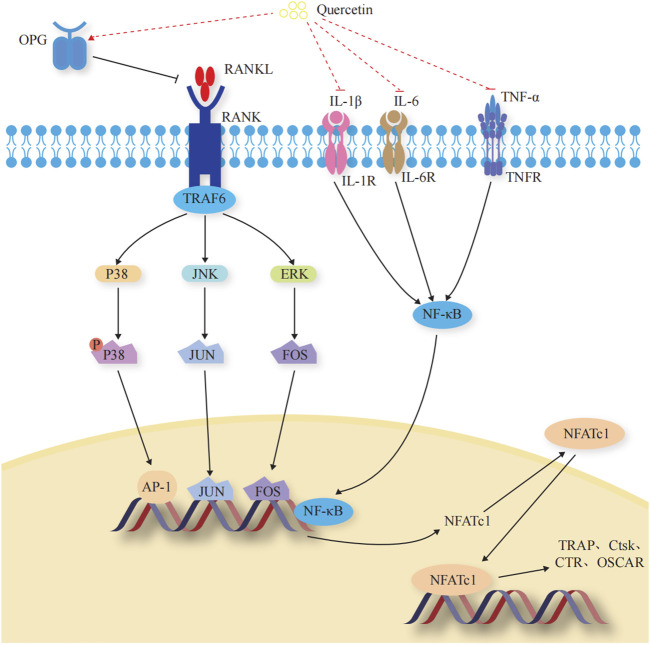
OCs are multinucleated giant cells with bone resorption functions, formed by the fusion of bone marrow monocyte precursors (Boyce, 2013). They promote bone resorption and remodeling by secreting acids and enzymes that dissolve the bone matrix. When OCs are overactive or in excessive numbers, the equilibrium between OBs and OCs is disrupted, leading to excessive bone resorption, which can also lead to OP. When exercising the function of bone resorption, OCs initially adhere to the surface of the bone matrix to form a closed area, releasing integrins ɑ and β3 to facilitate adherence to the bone matrix. Subsequently, the H+ ion pump with the help of ATP6V0d2 pumps H+ into the closed area, forming an acidic microenvironment to degrade the bone matrix. Meanwhile, OCs secrete tartrate-resistant acid phosphatase (TRAP), matrix metalloproteinase 9 (MMP-9), and cathepsin K (CtsK), among others to dissolve the bone matrix (Soysa and Alles, 2016). The mechanisms underlying the Que regulation of OC-mediated bone resorption are shown in Figure 4.
FIGURE 4.
Effects and molecular mechanisms of quercetin induction on osteoclasts. (Takayanagi et al., 2002; Teitelbaum and Ross, 2003; Duplomb et al., 2008; Ikeda et al., 2008; Belibasakis and Bostanci, 2012; Ibrahim et al., 2016; Lee et al., 2016; Pinti et al., 2016; Soysa and Alles, 2016; Amarasekara et al., 2018; Lee et al., 2018; Luo et al., 2018; Noguchi et al., 2018; Ha and Choung, 2020; Kurotaki et al., 2020; Yang et al., 2020; Udagawa et al., 2021; Velletri et al., 2021; Yao et al., 2021).
3.2.1 OC-specific transcription factors
Transcription factors regulate the differentiation and activity of OCs (Kurotaki et al., 2020). Nuclear factor of activated T cell 1 (NFATc1) is the most critical regulator of OC differentiation (Takayanagi et al., 2002). It controls OC-specific genes, such as TRAP, Ctsk, calcitonin receptor, and OC-associated receptor, through the synergistic activation of microphthalmia-associated transcription factor (Mitf), nuclear factor kappa B (NF-κB), c-Fos, and c-jun, thus affecting OC activity and regulating bone resorption (Yao et al., 2021). (Figure 4).
Treatment with Que reduces the expression of the NFATc1 gene and protein through the TRAF6/c-fos/NFATc1 signaling pathway and inhibits the differentiation of RAW264.7 cells to OCs (Liu et al., 2022). Que-3-O-β-D-glucoside inhibits OC differentiation by suppressing RANKL-induced NFATc1 expression, thereby preventing OC differentiation and bone loss (Shim et al., 2015).
3.2.2 OPG/RANKL/RANK signaling pathway
The OPG/RANKL/RANK signaling pathway is a key signaling axis during bone remodeling (Yang et al., 2020; Velletri et al., 2021). In the presence of M-CSF, RANK binds to the C-terminus of RANKL and recruits tumor necrosis factor receptor-associated factors to induce transcription and expression of downstream OC-specific genes. It ultimately activates key transcription factors, such as NF-κB, activator protein 1, cyclic adenosine monophosphate, cyclic adenosine monophosphate response element binding protein, and NFATc1, and induces OC markers, such as TRAP, β3 integrin, and CtsK, which regulate OC differentiation and induce bone resorption (Belibasakis and Bostanci, 2012; Udagawa et al., 2021). OPG secreted by OB is a high-affinity decoy-like receptor for RANKL. OPG reduces the binding of RANKL to RANK and competitively inhibits the blocking of signals from OBs to OCs, thereby inhibiting OC generation and maturation and reducing OC activity (Ibrahim et al., 2016). (Figure 4).
Que upregulates the expression of OPG and downregulates the expression of RANKL in the femoral bone tissues of ovariectomized rats at both high and medium doses, inhibits bone resorption, prevents OP, and improves the biomechanical properties of the femur (Wang, 2009). Que can directly stimulate OPG expression in OVX rBMSCs while concurrently inhibiting RANKL expression. This dual action leads to the indirect increase of the OPG/RANKL ratio, reestablishing the balance of the RANKL/OPG system and restoring the healing capacity of damaged bone in the state of OP (Zhou et al., 2017). When OB-OC-endothelial cells were inoculated in three cultures on Que-containing hydroxyapatite, the trend of OPG/RANKL levels was similar to the reduction in histone K levels, suggesting that the presence of Que has an inhibitory effect on OC viability (Forte et al., 2016).
3.2.3 ERK1/2/JNK signaling pathway
Among the OC precursors, two ERK forms (ERK1/2) and three JNK isoforms (JNK1/2/3) are mainly involved in OC precursor proliferation and OC apoptosis, respectively (Teitelbaum and Ross, 2003; Ikeda et al., 2008; Lee et al., 2016). ERKs have a typical protein kinase structure and control osteoclastogenesis by phosphorylating c-Fos, NFATc1, MITF, TFE3, hedgehog-Gli, Egr2, RSK2, and MMP-9 (Lee et al., 2018). JNK signaling regulates downstream c-Jun, CaMK, c-Fos, NFATc1, and semaphorin 3D to control OC metabolism (Noguchi et al., 2018). (Figure 4).
Que-3-O-β-D-glucuronide significantly attenuates the activation of JNK and ERK in LPS-stimulated RAW264.7 macrophages. It inhibits the secretion of plasmatic NO and PGE, as well as the expression of iNOS and COX-2, thus exerting an anti-inflammatory activity in a concentration-dependent manner (Park et al., 2016). Que exerts its anti-apoptotic effects through the JNK-c-Jun/AP-1 and ERK-c-Fos/AP-1 pathways (Ishikawa and Kitamura, 2000).
3.2.4 Regulated inflammatory factors
Dynamic regulation of osteoclastogenic and anti-osteoclastogenic cytokines is essential for maintaining bone homeostasis (Amarasekara et al., 2018). During macrophage polarization, many inflammatory factors are involved in osteoclastogenesis through different pro-inflammatory and anti-inflammatory roles, further affecting the process of bone resorption and the development of OP (Pinti et al., 2016). Tumor necrosis factor-α (TNF-α) stimulates OC differentiation by upregulating RANK pro-inflammatory target genes through activating and inducing NF-κB nuclear translocation, disrupting the balance of the RANK-RANKL bio-axis, and increasing OC activity (Luo et al., 2018). Interleukin—1β (IL-1β) and IL-6 promote OC differentiation and maturation through a RANKL-independent mechanism, resulting in the occurrence of bone resorption (Duplomb et al., 2008; Ha and Choung, 2020). (Figure 4).
Que (2 or 5 μM) significantly reduces the accumulation of TNF-α and IL-6 in LPS-induced mouse RAW264.7 macrophages (Jung and Sung, 2004). Tsai et al. (2021) found that Que inhibits M1 polarization and significantly reduces the expression levels of M1 markers, such as IL-6, TNF-α, and IL-1β, in macrophages and microglia. Que significantly reduces the levels of TNF-α and IL-1β, inhibits OC activation, and attenuates bone destruction (Li et al., 2018).
4 Effects of Que in combination with other phytochemicals
Phytochemical treatments have been used to induce OB differentiation in in vitro models (Pan et al., 2005; Shakibaei et al., 2012; Zhou et al., 2015; Martiniakova et al., 2022; Shen et al., 2022; Zhou et al., 2023). The combination of Que with other bioactive ingredients has shown synergistic anti-osteoporotic effects (Rayalam et al., 2011). Giordani et al. (2023) found that a mixture of curcumin (1 M), polydatin (10 M), and Que (0.5 M) is a safe bioactive compound. This mixture has significant synergistic effects in promoting OB differentiation of MSCs and inhibiting inflammatory phenotypes associated with cellular senescence. It significantly reduces the expression of miR-21 and miR-146a, decreases the release of IL-8 and MCP-1, increases the expression of ALP mRNA, and efficiently reduces p38-MAPK and phosphorylated NF-κB. Lai et al. (2011) found that a combination of 2400 IU/kg of vitamin D, 400 mg/kg of resveratrol, 2,000 mg/kg of Que, and 1,040 mg/kg of genistein improves bone mineral density and trabecular structure and reduces postmenopausal bone loss in de-ovulated female rats. However, Ambati et al. (2018) found that the dietary intake of a combination of vitamin D, resveratrol, Que, and genistein, owing to their relatively low dose and synergistic properties, is not as effective as zoledronic acid in a postmenopausal rat model of OP. They reported that relatively low doses of phytochemicals may not have produced a sufficiently potent effect to prevent or reverse the dramatic loss of bone trabeculae induced by ovarian hormone deficiency. This lack of efficacy may be related to the prolonged duration of the study (16 weeks) and the dynamics of bone loss after estrogen withdrawal.
5 Pharmacokinetics of Que
Researchers have used high-performance liquid chromatography to determine the concentration of Que in various organ tissues. They found that Que is mainly distributed in the gastrointestinal tract by gastrointestinal ease of diffusion and absorption, followed by the blood, liver, kidney, heart, lungs, and spleen, and has a very low distribution in the brain and muscle tissues of rats (Meng and Wang, 2000; Wu et al., 2008). Que metabolites are excreted through the kidneys, feces, and the respiratory system (Moon et al., 2000; Russo et al., 2012). Que is rapidly metabolized in the blood and has a short half-life. Que metabolites are detected in plasma 30 min after ingestion, with the major metabolites being Que-30-sulfate, Que-3-glucuronide, and Que-3-sulfate. The highest concentrations are observed at 0.8 and 0.6 h, but they are excreted in large quantities within 24 h (Mullen et al., 2006; Moon et al., 2008). Que-30-glucuronide, Que-diuronate, isorhamnetin-sulfate glucuronate, isorhamnetin-methylquercetin, and isorhamnetin-diuronate are the major urinary metabolites, with the highest concentrations observed at 4 h (Moon et al., 2008).
Que exhibits low in vitro bioavailability (5.9%) and demonstrates solubility of 235.5 g/mL in water and 2.3 × 104 g/mL in chloroform. It reaches a maximum concentration (Cmax) of 4.143 g/mL, an area under the curve of 12.015 g h/mL, and is encapsulated at a rate of 61%, with a drug loading capacity of 13 g/mg (Chen et al., 2019; Shao et al., 2019). Modern pharmaceutical scientists have improved the in vitro bioavailability of Que by applying delivery system technologies, such as particulate delivery systems, solid dispersions, encapsulation, phospholipid complexes, and hydrogels (Zhao et al., 2022). The amorphous solubility of Que-2-hydroxypropyl-β-cyclodextrin complex at small intestinal pH is at least 31 g/mL, with a Cmax value of 78.3 g/mL (Manta et al., 2020). The whey protein isolate-Que-lotus root branched chain amylose (LRA) hydrogel (whey protein isolate-Que-LRA) encapsulation rate was up to 92.4%, and Que was stable in the stomach and effectively released into the small intestine (Liu et al., 2020). The in vitro cumulative release of Que nanohybrids reached more than 65% at 30 min, which was significantly better than the cumulative release of Que APIs and physical mixtures at the same time (25.79% and 31.53%, respectively) (Liu et al., 2017). Shi et al. (2020) found a significant increase in the amount of Que passing through an artificial membrane after placing a Que/F68/HPMC 1/4/3 asd solution into the donor pool. Que shows a 12-fold increase in water solubility in the Que-phospholipid complex (from 3.44 μg/mL to 36.81 μg/mL) (Singh et al., 2012). Lee et al. (2020) loaded Que onto graphene planes by π-π stacking and weak hydrogen bonding. This loading method may result in a pH-responsive drug release mechanism in acidic environments, making it more suitable for targeted drug delivery in oncology therapy, with a maximum drug loading of 11 wt%. The oral administration of Que nanocapsules with triphenylphosphine cations as a matrix component with mitochondrial specificity results in high brain uptake and significant mitochondrial localization after cerebral ischemia/reperfusion. This effectively counteracts cerebral ischemia/reperfusion-induced cell death and neurodegenerative lesions in both young and aged rats (Ghosh et al., 2017).
6 Toxicology of Que
While some controversy exists regarding the safety of Que, the majority of views support the idea that Que is not toxic. Experimental studies of chronic toxicity have shown that after 2 years of administration of 0.1%, 1%, and 4% Que (equivalent to 40, 400, and 1900 mg/(kg·d)) in experimental rats, focal epithelial hyperplasia of the renal tubules was observed in male rats. The incidence of chronic kidney disease slightly increased with an increase in Que intake, and the incidence of renal adenomas was increased by 1% and 4% Que doses (Dunnick and Hailey, 1992). A reduction in body mass, an increased incidence of non-neoplastic polyp hyperplasia of the cecum, hyperplasia of the parathyroid glands in male rats, and the detection of calcium oxalate crystals in the urine were observed in rats following Que ingestion (Divi and Doerge, 1996; Andres et al., 2018). Que shows significant mutagenicity in mammalian cells and is potentially genotoxic (Huan et al., 2010). However, a 2-year rodent carcinogenicity bioassay conducted by the National Toxicology Program did not reveal any adverse effects relevant to the safety assessment of orally administered Que in humans (NTP, 1992; Harwood et al., 2007). Feng (2013) found that Que is free of acute toxicity, mutagenicity, and chronic or subchronic toxicity in vivo and in vitro. This finding was based on the acute toxicity test, Ames test, mouse bone marrow cell micronucleus test, mouse spermatozoa aberration test, the 30-day feeding test in rats, and the 56-day feeding test in laying hens. The State Food and Drug Administration (2017) published the World Health Organization’s International Agency for Research on Cancer list of carcinogens, classifying Que as a Group 3 carcinogen due to its suspected carcinogenicity to humans, but with insufficient human or animal data. The in vitro test lacks a well-established regulatory mechanism in the animal body. Therefore, no accurate animal test data have been established to support the conclusion that there is insufficient evidence to suggest that Que poses a safety risk. Therefore, based on the current animal safety evaluation tests within a reasonable dose range, the safety of Que is considered high.
7 Future perspective
Que, as a polyphenolic compound, does have some limitations in in vitro studies. One of the important limitations is its interfering effect on many assays, leading to false positive results (Baell and Holloway, 2010). Since Que has strong antioxidant, anti-inflammatory, anticancer and many other biological activities (Singh et al., 2021), it may affect certain biochemical reactions and assay results in in vitro experiments. Because of these interfering effects of Que, the reliability of data in in vitro studies is somewhat compromised. In order to reduce this interference, researchers should fully consider the effects of Que when conducting in vitro experiments and take appropriate measures to avoid or reduce the interference. For example, choosing appropriate experimental conditions, optimising experimental methods and reagents, setting up control groups, adding blocking agents, etc. In addition, clinical and in vivo studies can provide a more realistic evaluation of drug action effects and safety. Therefore, the results of clinical studies and in vivo studies should be combined when assessing the pharmacological effects and safety of Que to obtain a more comprehensive and accurate understanding. Also, when studying the interaction of Que with other drugs or compounds, attention needs to be paid to this interfering effect in order to avoid misleading conclusions.
The clinical applications of Que in the treatment of other diseases, such as type 2 diabetes (Mazloom et al., 2014) and obesity (Pfeuffer et al., 2013), have been explored and evaluated. Therefore, more high-quality large-sample clinical observations are warranted to discover and validate a more detailed mechanism of action for Que in the treatment of OP and to determine the optimal dosage, which will help promote its development as a functional food and drug.
However, several aspects must be considered during the development process. As the low solubility and bioavailability of Que limit its applications, researchers can improve its bioavailability through chemical modifications and composite carriers. In addition, intestinal flora was found to be involved in the development of OP (Hao et al., 2019; Zhang et al., 2021). How Que participates in bone metabolism by regulating the intestinal flora is not yet clear and will provide a new avenue for Que research.
8 Conclusion
This article reviews advances in the mechanisms, pharmacokinetics, and toxicology of Que for the treatment of OP. Que promotes OB-mediated bone formation mainly by regulating transcription factors, the Wnt/β-catenin signaling pathway, the BMP-2/SMADs/RUNX2 signaling pathway, anti-apoptosis-mediated pathways, and oxidative stress-mediated pathways, and by promoting bone matrix formation and mineralization. It also inhibits OC-mediated bone resorption by regulating transcription factors, inflammatory factors, the OPG/RANKL/RANK signaling pathway, and the ERK1/2/JNK signaling pathway. Thus, Que is a potential drug for the prevention and treatment of OP.
Funding Statement
The author(s) declare financial support was received for the research, authorship, and/or publication of this article. This study was financially supported by the Natural Science Foundation of China (No. 82004212), the Shandong Province Medical Health Science and Technology Development Plan Project (No. 202204070951) and the TCM Science and Technology Project of Shandong Province (No. 2021M175).
Author contributions
T-TD: Writing–original draft. W-YD: Writing–review and editing. X-XL: Writing–review and editing. Q-HZ: Writing–review and editing. J-XD: Writing–review and editing. L-JW: Writing–review and editing. M-NY: Writing–review and editing. YY: Writing–review and editing. F-JL: Writing–review and editing.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
- Alrawaiq N. S., Abdullah A. (2014). A review of flavonoid quercetin: metabolism, bioactivity and antioxidant properties. Int. J. PharmTech Res. 6 (3), 933–941. [Google Scholar]
- Amarasekara D. S., Yun H., Kim S., Lee N., Kim H., Rho J. (2018). Regulation of osteoclast differentiation by cytokine networks. Immune Netw. 18 (1), e8. 10.4110/in.2018.18.e8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ambati S., Miller C. N., Bass E. F., Hohos N. M., Hartzell D. L., Kelso E. W., et al. (2018). Synergistic phytochemicals fail to protect AgainstOvariectomy induced bone loss in rats. J. Med. Food 21 (10), 1044–1052. 10.1089/jmf.2017.0113 [DOI] [PubMed] [Google Scholar]
- An X., Shang F. (2018). RA-XII exerts anti-oxidant and anti-inflammatory activities on lipopolysaccharide-induced acute renal injury by suppressing NF-κB and MAPKs regulated by HO-1/Nrf2 pathway. Biochem. Biophys. Res. Commun. 495 (3), 2317–2323. 10.1016/j.bbrc.2017.12.131 [DOI] [PubMed] [Google Scholar]
- Andres S., Pevny S., Ziegenhagen R., Bakhiya N., Schäfer B., Hirsch-Ernst K. I., et al. (2018). Safety aspects of the use of quercetin as a dietary supplement. Mol. Nutr. Food Res. 62 (1), 1700447. 10.1002/mnfr.201700447 [DOI] [PubMed] [Google Scholar]
- Armas L. A., Recker R. R. (2012). Pathophysiology of osteoporosis: new mechanistic insights. Endocrinol. Metab. Clin. North Am. 41 (3), 475–486. 10.1016/j.ecl.2012.04.006 [DOI] [PubMed] [Google Scholar]
- Baell J. B., Holloway G. A. (2010). New substructure filters for removal of pan assay interference compounds (PAINS) from screening libraries and for their exclusion in bioassays. J. Med. Chem. 53 (7), 2719–2740. 10.1021/jm901137j [DOI] [PubMed] [Google Scholar]
- Baş A., Albeniz I. (2022). Investigation of the effects of eugenol and quercetin on bone loss in STZ-NA induced diabetic rats utilizing micro CT. J. Diabetes Metab. Disord. 21 (1), 637–646. 10.1007/s40200-022-01026-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- Belibasakis G. N., Bostanci N. (2012). The RANKL-OPG system in clinical periodontology. J. Clin. Periodontol. 39 (3), 239–248. 10.1111/j.1600-051X.2011.01810.x [DOI] [PubMed] [Google Scholar]
- Bellavia D., Caradonna F., Dimarco E., Costa V., Carina V., De Luca A., et al. (2021). Terpenoid treatment in osteoporosis: this is where we have come in research. Trends Endocrinol. Metab. 32 (11), 846–861. 10.1016/j.tem.2021.07.011 [DOI] [PubMed] [Google Scholar]
- Bian W., Xiao S., Yang L., Chen J., Deng S. (2021). Quercetin promotes bone marrow mesenchymal stem cell proliferation and osteogenic differentiation through the H19/miR-625-5p axis to activate the Wnt/β-catenin pathway. BMC Complement. Med. Ther. 21 (1), 243. 10.1186/s12906-021-03418-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bian W., Yang L., Sun H. (2016). Effects of quercetin on proliferation and osteogenic differentiation of bone marrow mesenchymal stem cells. Pharmacol. Clin. Chin. Materia Medica 32 (05), 27–30. 10.13412/j.cnki.zyyl.2016.05.007 [DOI] [Google Scholar]
- Bian W., Zhu H. Z., Xiao S. Q. (2022). Quercetin promotes bone marrow mesenchymal stem cell proliferation and osteogenic differentiation through inhibiting lncRNA DANCR expression. Shenzhen J. Integr. Traditional Chin. West. Med. 32 (11), 1–5. 10.16458/j.cnki.1007-0893.2022.11.001 [DOI] [Google Scholar]
- Boyce B. F. (2013). Advances in the regulation of osteoclasts and osteoclast functions. J. Dent. Res. 92 (10), 860–867. 10.1177/0022034513500306 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Casado-Díaz A., Anter J., Dorado G., Quesada-Gómez J. M. (2016). Effects of quercetin, a natural phenolic compound, in the differentiation of human mesenchymal stem cells (MSC) into adipocytes and osteoblasts. J. Nutr. Biochem. 32, 151–162. 10.1016/j.jnutbio.2016.03.005 [DOI] [PubMed] [Google Scholar]
- Chandra A., Lagnado A. B., Farr J. N., Schleusner M., Monroe D. G., Saul D., et al. (2022). Bone marrow adiposity in models of radiation- and aging-related bone loss is dependent on cellular senescence. J. Bone Min. Res. 37 (5), 997–1011. 10.1002/jbmr.4537 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen D. C., Wei S. Q. (2002). Effects of quercetin on proliferation and apotosis of SD rat osteoclasts in vitro . West China J. Pharm. Sci. (01), 16–18. 10.13375/j.cnki.wcjps.2002.01.006 [DOI] [Google Scholar]
- Chen J. H., Cao J. L., Chu Y. L., Wang Z. l., Yang Z. t., Wang H. l. (2008). T-2 toxin-induced apoptosis involving Fas, p53, Bcl-xL, Bcl-2, Bax and caspase-3 signaling pathways in human chondrocytes. J. Zhejiang Univ. Sci. B 9 (6), 455–463. 10.1631/jzus.B0820013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen L. P., Deng M. T., Du C. (2014). A study of quercetin extracted from eucommia leaf promoting the proliferation of bone marrow derived mesenchymal stem cells through the phosphorylation of ERK. Lishizhen Med. Materia Medica Res. 25 (12), 2845–2847. [Google Scholar]
- Chen M., Li M., Wei Y., Xue C., Fei Y. (2022). ROS-activatable biomimetic interface mediates in-situ bioenergetic remodeling of osteogenic cells for osteoporotic bone repair. Biomaterials 291, 121878. 10.1016/j.biomaterials.2022.121878 [DOI] [PubMed] [Google Scholar]
- Chen W., Zou M., Ma X., Lv R., Ding T., Liu D. (2019). Co-encapsulation of EGCG and quercetin in liposomes for optimum antioxidant activity. J. Food Sci. 84 (1), 111–120. 10.1111/1750-3841.14405 [DOI] [PubMed] [Google Scholar]
- Clevers H., Loh K. M., Nusse R. (2014). Stem cell signaling. An integral program for tissue renewal and regeneration: Wnt signaling and stem cell control. Science 346, 1248012. 10.1126/science.1248012 [DOI] [PubMed] [Google Scholar]
- Crespy V., Morand C., Besson C., Manach C., Démigné C., Rémésy C. (2001). Comparison of the intestinal absorption of quercetin, phloretin and their glucosides in rats. J. Nutr. 131 (8), 2109–2114. 10.1093/jn/131.8.2109 [DOI] [PubMed] [Google Scholar]
- David A. V. A., Arulmoli R., Parasuraman S. (2016). Overviews of biological importance of quercetin: a bioactive flavonoid. Pharmacogn. Rev. 10 (20), 84–89. 10.4103/0973-7847.194044 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deng J., Zhang J. L., Zhou G. P. (2020). Effect of quercetin on serum mineral content and bone metabolism in rats fed with low calcium and high magnesium diet. Chin. J. Osteoporos. 26 (07), 1039–1043. [Google Scholar]
- Derakhshanian H., Djalali M., Djazayery A., Nourijelyani K., Ghadbeigi S., Pishva H., et al. (2013). Quercetin prevents experimental glucocorticoid-induced osteoporosis: a comparative study with alendronate. Can. J. Physiol. Pharmacol. 91 (5), 380–385. 10.1139/cjpp-2012-0190 [DOI] [PubMed] [Google Scholar]
- Di Petrillo A., Orrù G., Fais A., Fantini M. C. (2022). Quercetin and its derivates as antiviral potentials: a comprehensive review. Phytother. Res. 36 (1), 266–278. 10.1002/ptr.7309 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Divi R. L., Doerge D. R. (1996). Inhibition of thyroid peroxidase by dietary flavonoids. Chem. Res. Toxicol. 9 (1), 16–23. 10.1021/tx950076m [DOI] [PubMed] [Google Scholar]
- Dunnick J. K., Hailey J. R. (1992). Toxicity and carcinogenicity studies of quercetin, a natural component of foods. Fundam. Appl. Toxicol. 19 (3), 423–431. 10.1016/0272-0590(92)90181-g [DOI] [PubMed] [Google Scholar]
- Duplomb L., Baud'huin M., Charrier C., Berreur M., Trichet V., Blanchard F., et al. (2008). Interleukin-6 inhibits receptor activator of nuclear factor kappaB ligand-induced osteoclastogenesis by diverting cells into the macrophage lineage: key role of Serine727 phosphorylation of signal transducer and activator of transcription 3. Endocrinology 149 (7), 3688–3697. 10.1210/en.2007-1719 [DOI] [PubMed] [Google Scholar]
- Eid H. M., Haddad P. S. (2017). The antidiabetic potential of quercetin: underlying mechanisms. Curr. Med. Chem. 24 (4), 355–364. 10.2174/0929867323666160909153707 [DOI] [PubMed] [Google Scholar]
- Feng J. J., Zou H., Zhu H. Y. (2016). Effect of quercetin on serum osteocalcin levels and femur typeⅠcollagen protein levels in the ovariectomized rats. J. Traditional Chin. Orthop. Traumatology 28 (07), 10–13. [Google Scholar]
- Feng L., Yang Z., Hou N., Wang M., Lu X., Li Y., et al. (2023). Long non-coding RNA Malat1 increases the rescuing effect of quercetin on tnfα-impaired bone marrow stem cell osteogenesis and ovariectomy-induced osteoporosis. Int. J. Mol. Sci. 24 (6), 5965. 10.3390/ijms24065965 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feng X. A. (2013). Safety evaluation of quercetin as a feed additive. China: Northeast Agricultural University. [Google Scholar]
- Fideles S. O. M., de Cássia Ortiz A., Buchaim D. V., de Souza Bastos Mazuqueli Pereira E., Parreira M. J. B. M., de Oliveira Rossi J., et al. (2023). Influence of the neuroprotective properties of quercetin on regeneration and functional recovery of the nervous system. Antioxidants (Basel) 12 (1), 149. 10.3390/antiox12010149 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Forte L., Torricelli P., Boanini E., Gazzano M., Rubini K., Fini M., et al. (2016). Antioxidant and bone repair properties of quercetin-functionalized hydroxyapatite: an in vitro osteoblast-osteoclast-endothelial cell co-culture study. Acta Biomater. 32, 298–308. 10.1016/j.actbio.2015.12.013 [DOI] [PubMed] [Google Scholar]
- Gao Y., Xiao F., Wang C., Cui P., Zhang X. (2018). Long noncoding RNA MALAT1 promotes osterix expression to regulate osteogenic differentiation by targeting miRNA-143 in human bone marrow-derived mesenchymal stem cells. J. Cell Biochem. 119 (8), 6986–6996. 10.1002/jcb.26907 [DOI] [PubMed] [Google Scholar]
- Geng Q., Gao H., Yang R., Guo K., Miao D. (2019). Pyrroloquinoline quinone prevents estrogen deficiency-induced osteoporosis by inhibiting oxidative stress and osteocyte senescence. Int. J. Biol. Sci. 15 (1), 58–68. 10.7150/ijbs.25783 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ghosh S., Sarkar S., Choudhury S. T., Ghosh T., Das N. (2017). Triphenyl phosphonium coated nano-quercetin for oral delivery: neuroprotective effects in attenuating age related global moderate cerebral ischemia reperfusion injury in rats. Nanomedicine 13 (8), 2439–2450. 10.1016/j.nano.2017.08.002 [DOI] [PubMed] [Google Scholar]
- Giordani C., Matacchione G., Giuliani A., Valli D., Scarpa E. S., Antonelli A., et al. (2023). Pro-osteogenic and anti-inflammatory synergistic effect of orthosilicic acid, vitamin K2, curcumin, polydatin and quercetin combination in young and senescent bone marrow-derived mesenchymal stromal cells. Int. J. Mol. Sci. 24 (10), 8820. 10.3390/ijms24108820 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guo C., Yang R. J., Jang K., Zhou X. L., Liu Y. Z. (2017). Protective effects of pretreatment with quercetin against lipopolysaccharide-induced apoptosis and the inhibition of osteoblast differentiation via the MAPK and wnt/β-catenin pathways in mc3t3-E1 cells. Cell Physiol. Biochem. 43 (4), 1547–1561. 10.1159/000481978 [DOI] [PubMed] [Google Scholar]
- Ha S. H., Choung P. H. (2020). MSM promotes human periodontal ligament stem cells differentiation to osteoblast and bone regeneration. Biochem. Biophys. Res. Commun. 528 (1), 160–167. 10.1016/j.bbrc.2020.05.097 [DOI] [PubMed] [Google Scholar]
- Hao M., Wang G., Zuo X., Qu C. J., Yao B. C., Wang D. L. (2019). Gut microbiota: an overlooked factor that plays a significant role in osteoporosis. J. Int. Med. Res. 47 (9), 4095–4103. 10.1177/0300060519860027 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harvey A. L., Edrada-Ebel R., Quinn R. J. (2015). The re-emergence of natural products for drug discovery in the genomics era. Nat. Rev. Drug Discov. 14 (2), 111–129. 10.1038/nrd4510 [DOI] [PubMed] [Google Scholar]
- Harwood M., Danielewska-Nikiel B., Borzelleca J. F., Flamm G. W., Williams G. M., Lines T. C. (2007). A critical review of the data related to the safety of quercetin and lack of evidence of in vivo toxicity, including lack of genotoxic/carcinogenic properties. Food Chem. Toxicol. 45 (11), 2179–2205. 10.1016/j.fct.2007.05.015 [DOI] [PubMed] [Google Scholar]
- Hou D. D., Zhang W., Gao Y. L., Sun Y. Z., Wang H. X., Qi R. Q., et al. (2019). Anti-inflammatory effects of quercetin in a mouse model of MC903-induced atopic dermatitis. Int. Immunopharmacol. 74, 105676. 10.1016/j.intimp.2019.105676 [DOI] [PubMed] [Google Scholar]
- Hu Y., Gui Z., Zhou Y., Xia L., Lin K., Xu Y. (2019). Quercetin alleviates rat osteoarthritis by inhibiting inflammation and apoptosis of chondrocytes, modulating synovial macrophages polarization to M2 macrophages. Free Radic. Biol. Med. 145, 146–160. 10.1016/j.freeradbiomed.2019.09.024 [DOI] [PubMed] [Google Scholar]
- Hu Y., Yuan W., Cai N., Jia K., Meng Y., Wang F., et al. (2022). Exploring quercetin anti-osteoporosis pharmacological mechanisms with in silico and in vivo models. Life (Basel) 12 (7), 980. 10.3390/life12070980 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huan F., Cheng J., Jin S. X. (2010). Genotoxicity effects of quercetin on mammalian cells in vitro . Chin. Prev. Med. 11 (08), 797–800. 10.16506/j.1009-6639.2010.08.011 [DOI] [Google Scholar]
- Hunter G. K., Goldberg H. A. (1993). Nucleation of hydroxyapatite by bone sialoprotein. Proc. Natl. Acad. Sci. U. S. A. 90 (18), 8562–8565. 10.1073/pnas.90.18.8562 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ibrahim T., Ricci M., Scarpi E., Bongiovanni A., Ricci R., Riva N., et al. (2016). RANKL: a promising circulating marker for bone metastasis response. Oncol. Lett. 12 (4), 2970–2975. 10.3892/ol.2016.4977 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ikeda F., Matsubara T., Tsurukai T., Hata K., Nishimura R., Yoneda T. (2008). JNK/c-Jun signaling mediates an anti-apoptotic effect of RANKL in osteoclasts. J. Bone Min. Res. 23 (6), 907–914. 10.1359/jbmr.080211 [DOI] [PubMed] [Google Scholar]
- Inoue J., Choi J. M., Yoshidomi T., Yashiro T., Sato R. (2010). Quercetin enhances VDR activity, leading to stimulation of its target gene expression in Caco-2 cells. J. Nutr. Sci. Vitaminol. (Tokyo) 56 (5), 326–330. 10.3177/jnsv.56.326 [DOI] [PubMed] [Google Scholar]
- Ishikawa Y., Kitamura M. (2000). Anti-apoptotic effect of quercetin: intervention in the JNK- and ERK-mediated apoptotic pathways. Kidney Int. 58 (3), 1078–1087. 10.1046/j.1523-1755.2000.00265.x [DOI] [PubMed] [Google Scholar]
- Jang W. Y., Kim M. Y., Cho J. Y. (2022). Antioxidant, anti-inflammatory, anti-menopausal, and anti-cancer effects of lignans and their metabolites. Int. J. Mol. Sci. 23 (24), 15482. 10.3390/ijms232415482 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jin C. H., So Y. K., Han S. N., Kim J. B. (2016). Isoegomaketone upregulates heme oxygenase-1 in RAW264.7 cells via ROS/p38 MAPK/Nrf2 pathway. Biomol. Ther. Seoul. 24 (5), 510–516. 10.4062/biomolther.2015.194 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jung W. J., Sung M. K. (2004). Effects of major dietary antioxidants on inflammatory markers of RAW 264.7 macrophages. Biofactors 21 (1-4), 113–117. 10.1002/biof.552210122 [DOI] [PubMed] [Google Scholar]
- Kandemir K., Tomas M., McClements D. J., Capanoglu E. (2022). Recent advances on the improvement of quercetin bioavailability. Trends Food Sci. Technol. 119, 192–200. 10.1016/j.tifs.2021.11.032 [DOI] [Google Scholar]
- Kanis J. A., Cooper C., Rizzoli R., Reginster J. Y. (2019). European guidance for the diagnosis and management of osteoporosis in postmenopausal women. Osteoporos. Int. 30 (1), 3–44. 10.1007/s00198-018-4704-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim D. S., Takai H., Arai M., Araki S., Mezawa M., Kawai Y., et al. (2007). Effects of quercetin and quercetin 3-glucuronide on the expression of bone sialoprotein gene. J. Cell Biochem. 101 (3), 790–800. 10.1002/jcb.21233 [DOI] [PubMed] [Google Scholar]
- Kim J. H., Kim M., Hong S., Kim E. Y., Lee H., Jung H. S., et al. (2021). Albiflorin promotes osteoblast differentiation and healing of rat femoral fractures through enhancing BMP-2/smad and wnt/β-catenin signaling. Front. Pharmacol. 12, 690113. 10.3389/fphar.2021.690113 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kimball J. S., Johnson J. P., Carlson D. A. (2021). Oxidative stress and osteoporosis. J. Bone Jt. Surg. Am. 103 (15), 1451–1461. 10.2106/JBJS.20.00989 [DOI] [PubMed] [Google Scholar]
- Kurotaki D., Yoshida H., Tamura T. (2020). Epigenetic and transcriptional regulation of osteoclast differentiation. Bone 138, 115471. 10.1016/j.bone.2020.115471 [DOI] [PubMed] [Google Scholar]
- Lai C. Y., Yang J. Y., Rayalam S., Della-Fera M. A., Ambati S., Lewis R. D., et al. (2011). Preventing bone loss and weight gain with combinations of vitamin D and phytochemicals. J. Med. Food 14 (11), 1352–1362. 10.1089/jmf.2010.0232 [DOI] [PubMed] [Google Scholar]
- Lakhanpal P., Rai D. K. (2007). Quercetin: a versatile flavonoid. Internet J. Med. Update 2 (2), 22–37. 10.4314/ijmu.v2i2.39851 [DOI] [Google Scholar]
- Lan D., Qi S., Yao C., Li X., Liu H., Wang D., et al. (2022). Quercetin protects rat BMSCs from oxidative stress via ferroptosis. J. Mol. Endocrinol. 69 (3), 401–413. 10.1530/JME-22-0086 [DOI] [PubMed] [Google Scholar]
- Lee K., Chung Y. H., Ahn H., Kim H., Rho J., Jeong D. (2016). Selective regulation of MAPK signaling mediates RANKL-dependent osteoclast differentiation. Int. J. Biol. Sci. 12 (2), 235–245. 10.7150/ijbs.13814 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee K., Seo I., Choi M. H., Jeong D. (2018). Roles of mitogen-activated protein kinases in osteoclast biology. Int. J. Mol. Sci. 19 (10), 3004. 10.3390/ijms19103004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee X. J., Lim H. N., Gowthaman N. S. K., Rahman M. B. A., Che Abdullah C. A., Muthoosamy K. (2020). In-situ surface functionalization of superparamagnetic reduced graphene oxide–Fe3O4 nanocomposite via Ganoderma lucidum extract for targeted cancer therapy application. Appl. Surf. Sci. 512, 145738. 10.1016/j.apsusc.2020.145738 [DOI] [Google Scholar]
- Lei S. S., Su J., Zhang Y., Huang X. W., Wang X. P., Huang M. C., et al. (2021). Benefits and mechanisms of polysaccharides from Chinese medicinal herbs for anti-osteoporosis therapy: a review. Int. J. Biol. Macromol. 193 (Pt B), 1996–2005. 10.1016/j.ijbiomac.2021.11.030 [DOI] [PubMed] [Google Scholar]
- Li R., Zhu X., Zhang M., Zong G., Zhang K. (2021). Association of serum periostin level with classical bone turnover markers and bone mineral density in Shanghai Chinese postmenopausal women with osteoporosis. Int. J. Gen. Med. 14, 7639–7646. 10.2147/IJGM.S335296 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li X. Y., Yang L., Zhang R. H. (2020). Research on quercetin regulating osteogenic and adipogenic differentiation of bone marrow mesenchymal stem cells through the estrogen receptor signaling pathway. China J. Traditional Chin. Med. Pharm. 35 (12), 6011–6014. [Google Scholar]
- Li Z., Zhang J., Ren X., Liu Q., Yang X. (2018). The mechanism of quercetin in regulating osteoclast activation and the PAR2/TRPV1 signaling pathway in the treatment of bone cancer pain. Int. J. Clin. Exp. Pathol. 11 (11), 5149–5156. [PMC free article] [PubMed] [Google Scholar]
- Lin B., Xu P., Zheng J., Deng X., Ye Q., Huang Z., et al. (2022). Effects and mechanisms of natural alkaloids for prevention and treatment of osteoporosis. Front. Pharmacol. 13, 1014173. 10.3389/fphar.2022.1014173 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ling C. W., Miao Z., Xiao M. L., Zhou H., Jiang Z., Fu Y., et al. (2021). The association of gut microbiota with osteoporosis is mediated by amino acid metabolism: multiomics in a large cohort. J. Clin. Endocrinol. Metab. 106 (10), e3852–e3864. 10.1210/clinem/dgab492 [DOI] [PubMed] [Google Scholar]
- Little R. D., Recker R. R., Johnson M. L. (2002). High bone density due to a mutation in LDL-receptor-related protein 5. N. Engl. J. Med. 347 (12), 943–944. 10.1056/NEJM200209193471216 [DOI] [PubMed] [Google Scholar]
- Liu J., Liu F., Yan C. (2020). Effect of quercetin supplementation on bone metabolism in elderly osteoporosis patients treated with bisphosphonates. Chin. J. Osteoporos. 26 (07), 1044–1048. [Google Scholar]
- Liu K., Zha X. Q., Shen W. D., Li Q. M., Pan L. H., Luo J. P. (2020). The hydrogel of whey protein isolate coated by lotus root amylopectin enhance the stability and bioavailability of quercetin. Carbohydr. Polym. 236, 116009. 10.1016/j.carbpol.2020.116009 [DOI] [PubMed] [Google Scholar]
- Liu X., Liu J., Pang J. Y., Shen B. d., Shen C. Y., Lian W. Q., et al. (2017). Preparation of nanosuspension of quercetin with a miniaturized milling method. China J. Chin. Materia Medica 42 (15), 2984–2988. 10.19540/j.cnki.cjcmm.2017.0120 [DOI] [PubMed] [Google Scholar]
- Liu Z. M., Guan Z. Y., Jiang T. P. (2022). To explore quercetin in Panax Notoginseng on autophagy regulation mechanism of osteoclast differentiation based on the theory of “treatment from stasis”. Chin. J. Osteoporos. 28 (12), 1740–1744. [Google Scholar]
- Liu Z. Q. (2022). Quercetin inhibits osteoclast formation and its mechanism. China: Anhui Medical University. [Google Scholar]
- Luo G., Li F., Li X., Wang Z. G., Zhang B. (2018). TNF-α and RANKL promote osteoclastogenesis by upregulating RANK via the NF-κB pathway. Mol. Med. Rep. 17 (5), 6605–6611. 10.3892/mmr.2018.8698 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luo J., Zhang C., Liu Q., Ou S., Zhang L., Peng X. (2017). Combinative effect of sardine peptides and quercetin alleviates hypertension through inhibition of angiotensin I converting enzyme activity and inflammation. Food Res. Int. 100 (Pt 1), 579–585. 10.1016/j.foodres.2017.07.019 [DOI] [PubMed] [Google Scholar]
- Ma Z. Y., Zhang B., Zhang Y. T. (2023). Effects of quercetin sustained release system on osteogenic properties of MC3T3-E1 cells. Chin. J. Tissue Eng. Res. 27 (12), 1870–1876. [Google Scholar]
- Manta K., Papakyriakopoulou P., Chountoulesi M., Diamantis D. A., Spaneas D., Vakali V., et al. (2020). Preparation and biophysical characterization of quercetin inclusion complexes with β-cyclodextrin derivatives to be formulated as possible nose-to-brain quercetin delivery systems. Mol. Pharm. 17 (11), 4241–4255. 10.1021/acs.molpharmaceut.0c00672 [DOI] [PubMed] [Google Scholar]
- Marcus R., Majumder S. (2001). The nature of osteoporosis. Osteoporosis, 3–17. 10.1016/b978-012470862-4/50036-2 [DOI] [Google Scholar]
- Martín M. J., La-Casa C., Alarcón-de-la-Lastra C., Cabeza J., Villegas I., Motilva V. (1998). Anti-oxidant mechanisms involved in gastroprotective effects of quercetin. Z Naturforsch C J. Biosci. 53 (1-2), 82–88. 10.1515/znc-1998-1-215 [DOI] [PubMed] [Google Scholar]
- Martiniakova M., Babikova M., Mondockova V., Blahova J., Kovacova V., Omelka R. (2022). The role of macronutrients, micronutrients and flavonoid polyphenols in the prevention and treatment of osteoporosis. Nutrients 14 (3), 523. 10.3390/nu14030523 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Masuhara M., Tsukahara T., Tomita K., Furukawa M., Miyawaki S., Sato T. (2016). A relation between osteoclastogenesis inhibition and membrane-type estrogen receptor GPR30. Biochem. Biophys. Rep. 8, 389–394. 10.1016/j.bbrep.2016.10.013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mazloom Z., Abdollah Z. S. M., Dabbaghmanesh M. H. (2014). The effect of quercetin supplementation on oxidative stress, glycemic control, lipid profile and insulin resistance in type 2 diabetes: a randomized clinical trial. J. Health Sci. Surveillance Syst. 2 (1), 8–14. [Google Scholar]
- Meng D. H., Wang S. L. (2000). Research progress of quercetin and its glycosides. China Pharm. (05), 42–43. [Google Scholar]
- Messer J. G., Hopkins R. G., Kipp D. E. (2015). Quercetin metabolites up-regulate the antioxidant response in osteoblasts isolated from fetal rat calvaria. J. Cell Biochem. 116 (9), 1857–1866. 10.1002/jcb.25141 [DOI] [PubMed] [Google Scholar]
- Min J., Cheng Q. F., Zhang Q. (2022). Effect of quercetin on osteoporosis in ovary castrated rats. Jiangxi Med. J. 57 (10), 1353–1356. [Google Scholar]
- Mlcek J., Jurikova T., Skrovankova S., Sochor J. (2016). Quercetin and its anti-allergic immune response. Molecules 21 (5), 623. 10.3390/molecules21050623 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moon J. H., Nakata R., Oshima S., Inakuma T., Terao J. (2000). Accumulation of quercetin conjugates in blood plasma after the short-term ingestion of onion by women. Am. J. Physiol. Regul. Integr. Comp. Physiol. 279 (2), R461–R467. 10.1152/ajpregu.2000.279.2.R461 [DOI] [PubMed] [Google Scholar]
- Moon Y. J., Wang L., DiCenzo R., Morris M. E. (2008). Quercetin pharmacokinetics in humans. Biopharm. Drug Dispos. 29, 205–217. 10.1002/bdd.605 [DOI] [PubMed] [Google Scholar]
- Mu L. Q., Du J., Hu Y. Y. (2015). Effect of quercetin, geniposide, and aucubin in Eucommia ulmoides on proliferation and differentiation of osteoblast MC3T3-E1 in mice. Drug Eval. Res. 38 (02), 165–169. [Google Scholar]
- Mullen W., Edwards C. A., Crozier A. (2006). Absorption, excretion and metabolite profiling of methyl-glucuronyl-glucosyl- and sulpho-conjugates of quercetin in human plasma and urine after ingestion of onions. Br. J. Nutr. 96 (1), 107–116. 10.1079/bjn20061809 [DOI] [PubMed] [Google Scholar]
- Nabavi S. M., Šamec D., Tomczyk M., Milella L., Russo D., Habtemariam S., et al. (2020). Flavonoid biosynthetic pathways in plants: versatile targets for metabolic engineering. Biotechnol. Adv. 38, 107316. 10.1016/j.biotechadv.2018.11.005 [DOI] [PubMed] [Google Scholar]
- Nakashima K., Zhou X., Kunkel G., Zhang Z., Deng J. M., Behringer R. R., et al. (2002). The novel zinc finger-containing transcription factor osterix is required for osteoblast differentiation and bone formation. Cell 108, 17–29. 10.1016/s0092-8674(01)00622-5 [DOI] [PubMed] [Google Scholar]
- Noguchi T., Ebina K., Hirao M., Otsuru S., Guess A. J., Kawase R., et al. (2018). Apolipoprotein E plays crucial roles in maintaining bone mass by promoting osteoblast differentiation via ERK1/2 pathway and by suppressing osteoclast differentiation via c-Fos, NFATc1, and NF-κB pathway. Biochem. Biophys. Res. Commun. 503 (2), 644–650. 10.1016/j.bbrc.2018.06.055 [DOI] [PubMed] [Google Scholar]
- NTP (1992). Toxicology and carcinogenesis studies of quercetin (CAS No. 117-39-5) in F344/N rats (feed study). NTP technical report series No. 409. Research Triangle Park (NC): National Toxicology Program. [PubMed] [Google Scholar]
- Nusse R., Fuerer C., Ching W., Harnish K., Logan C., Zeng A., et al. (2008). Wnt signaling and stem cell control Cold. Spring Harb. Sym 73, 59–66. 10.1101/sqb.2008.73.035 [DOI] [PubMed] [Google Scholar]
- Oh J. H., Karadeniz F., Seo Y., Kong C. S. (2020). Effect of quercetin 3-O-β-D-Galactopyranoside on the adipogenic and osteoblastogenic differentiation of human bone marrow-derived mesenchymal stromal cells. Int. J. Mol. Sci. 21 (21), 8044. 10.3390/ijms21218044 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ohri R., Tung E., Rajachar R., Giachelli C. M. (2005). Mitigation of ectopic calcification in osteopontin-deficient mice by exogenous osteopontin. Calcif. Tissue Int. 76 (4), 307–315. 10.1007/s00223-004-0071-7 [DOI] [PubMed] [Google Scholar]
- Oršolić N., Goluža E., Dikić D., Lisičić D., Sašilo K., Rođak E., et al. (2014). Role of flavonoids on oxidative stress and mineral contents in the retinoic acid-induced bone loss model of rat. Eur. J. Nutr. 53 (5), 1217–1227. 10.1007/s00394-013-0622-7 [DOI] [PubMed] [Google Scholar]
- Palacios S. (2022). Medical treatment of osteoporosis. Climacteric 25 (1), 43–49. 10.1080/13697137.2021.1951697 [DOI] [PubMed] [Google Scholar]
- Pan W., Quarles L. D., Song L. H., Yu Y. H., Jiao C., Tang H. B., et al. (2005). Genistein stimulates the osteoblastic differentiation via NO/cGMP in bone marrow culture. J. Cell Biochem. 94 (2), 307–316. 10.1002/jcb.20308 [DOI] [PubMed] [Google Scholar]
- Pandit A. P., Omase S. B., Mute V. M. (2020). A chitosan film containing quercetin-loaded transfersomes for treatment of secondary osteoporosis. Drug Deliv. Transl. Res. 10 (5), 1495–1506. 10.1007/s13346-020-00708-5 [DOI] [PubMed] [Google Scholar]
- Pang X. G., Cong Y., Bao N. R., Li Y. G., Zhao J. N. (2018). Quercetin stimulates bone marrow mesenchymal stem cell differentiation through an estrogen receptor-mediated pathway. Biomed. Res. Int. 2018, 4178021. 10.1155/2018/4178021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Papiez M. A., Cierniak A., Krzysciak W., Bzowska M., Taha H. M., Jozkowicz A., et al. (2008). The changes of antioxidant defense system caused by quercetin administration do not lead to DNA damage and apoptosis in the spleen and bone marrow cells of rats. Food Chem. Toxicol. 46 (9), 3053–3058. 10.1016/j.fct.2008.06.006 [DOI] [PubMed] [Google Scholar]
- Park J. Y., Lim M. S., Kim S. I., Lee H. J., Kwon Y. S. (2016). Quercetin-3-O-β-D-Glucuronide suppresses lipopolysaccharide-induced JNK and ERK phosphorylation in LPS-challenged RAW264.7 cells. Biomol. Ther. Seoul. 24 (6), 610–615. 10.4062/biomolther.2016.026 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Patel R. V., Mistry B. M., Shinde S. K., Syed R., Singh V., Shin H. S. (2018). Therapeutic potential of quercetin as a cardiovascular agent. Eur. J. Med. Chem. 155, 889–904. 10.1016/j.ejmech.2018.06.053 [DOI] [PubMed] [Google Scholar]
- Pfeuffer M., Auinger A., Bley U., Kraus-Stojanowic I., Laue C., Winkler P., et al. (2013). Effect of quercetin on traits of the metabolic syndrome, endothelial function and inflammation in men with different APOE isoforms. Nutr. Metab. Cardiovasc Dis. 23 (5), 403–409. 10.1016/j.numecd.2011.08.010 [DOI] [PubMed] [Google Scholar]
- Pinti M., Appay V., Campisi J., Frasca D., Fülöp T., Sauce D., et al. (2016). Aging of the immune system: focus on inflammation and vaccination. Eur. J. Immunol. 46 (10), 2286–2301. 10.1002/eji.201546178 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prouillet C., Mazière J. C., Mazière C., Wattel A., Brazier M., Kamel S. (2004). Stimulatory effect of naturally occurring flavonols quercetin and kaempferol on alkaline phosphatase activity in MG-63 human osteoblasts through ERK and estrogen receptor pathway. Biochem. Pharmacol. 67 (7), 1307–1313. 10.1016/j.bcp.2003.11.009 [DOI] [PubMed] [Google Scholar]
- Rauf A., Imran M., Khan I. A., Ur-Rehman M., Gilani S. A., Mehmood Z., et al. (2018). Anticancer potential of quercetin: a comprehensive review. Phytother. Res. 32 (11), 2109–2130. 10.1002/ptr.6155 [DOI] [PubMed] [Google Scholar]
- Rayalam S., Della-Fera M. A., Baile C. A. (2011). Synergism between resveratrol and other phytochemicals: implications for obesity and osteoporosis. Mol. Nutr. Food Res. 55 (8), 1177–1185. 10.1002/mnfr.201000616 [DOI] [PubMed] [Google Scholar]
- Rodríguez V., Rivoira M., Picotto G., de Barboza G. D., Collin A., Tolosa de Talamoni N. (2022). Analysis of the molecular mechanisms by flavonoids with potential use for osteoporosis prevention or therapy. Curr. Med. Chem. 29 (16), 2913–2936. 10.2174/0929867328666210921143644 [DOI] [PubMed] [Google Scholar]
- Ruangsuriya J., Charumanee S., Jiranusornkul S., Sirisa-Ard P., Sirithunyalug B., Sirithunyalug J., et al. (2020). Depletion of β-sitosterol and enrichment of quercetin and rutin in Cissus quadrangularis Linn fraction enhanced osteogenic but reduced osteoclastogenic marker expression. BMC Complement. Med. Ther. 20 (1), 105. 10.1186/s12906-020-02892-w [DOI] [PMC free article] [PubMed] [Google Scholar]
- Russo M., Spagnuolo C., Tedesco I., Bilotto S., Russo G. L. (2012). The flavonoid quercetin in disease prevention and therapy: facts and fancies. Biochem. Pharmacol. 83 (1), 6–15. 10.1016/j.bcp.2011.08.010 [DOI] [PubMed] [Google Scholar]
- Shakibaei M., Shayan P., Busch F., Aldinger C., Buhrmann C., Lueders C., et al. (2012). Resveratrol mediated modulation of Sirt-1/Runx2 promotes osteogenic differentiation of mesenchymal stem cells: potential role of Runx2 deacetylation. PLoS One 7 (4), e35712. 10.1371/journal.pone.0035712 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shao X. M., Li K., Feng S. (2023). Research progress on biological function and application of quercetin in animal production. Feed Ind. 44 (17), 29–35. 10.13302/j.cnki.fi.2023.17.005 [DOI] [Google Scholar]
- Shao Y., Yu H., Yang Y., Li M., Hang L., Xu X. (2019). A solid dispersion of quercetin shows enhanced Nrf2 activation and protective effects against oxidative injury in a mouse model of dry age-related macular degeneration. Oxid. Med. Cell Longev. 2019, 1479571. 10.1155/2019/1479571 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shen D., Feng Y., Zhang X., Gong L., Liu J., Li Y., et al. (2022). Antiosteoporosis studies of 20 medicine food homology plants containing quercetin, rutin, and kaempferol: TCM characteristics, in vivo and in vitro activities, potential mechanisms, and food functions. Evid. Based Complement. Altern. Med. 2022, 5902293. 10.1155/2022/5902293 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shi X., Fan N., Zhang G., Sun J., He Z., Li J. (2020). Quercetin amorphous solid dispersions prepared by hot melt extrusion with enhanced solubility and intestinal absorption. Pharm. Dev. Technol. 25 (4), 472–481. 10.1080/10837450.2019.1709502 [DOI] [PubMed] [Google Scholar]
- Shim K. S., Ha H., Kim T., Lee C. J., Ma J. Y. (2015). Orostachys japonicus suppresses osteoclast differentiation by inhibiting NFATc1 expression. Am. J. Chin. Med. 43 (05), 1013–1030. 10.1142/S0192415X15500585 [DOI] [PubMed] [Google Scholar]
- Siddiqi M. H., Siddiqi M. Z., Ahn S., Kang S., Kim Y. J., Sathishkumar N., et al. (2013). Ginseng saponins and the treatment of osteoporosis: mini literature review. J. Ginseng Res. 37 (3), 261–268. 10.5142/jgr.2013.37.261 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Siddiqui J. A., Sharan K., Swarnkar G., Rawat P., Kumar M., Manickavasagam L., et al. (2011). Quercetin-6-C-β-D-glucopyranoside isolated from Ulmus wallichiana planchon is more potent than quercetin in inhibiting osteoclastogenesis and mitigating ovariectomy-induced bone loss in rats. Menopause 18 (2), 198–207. 10.1097/gme.0b013e3181e84e67 [DOI] [PubMed] [Google Scholar]
- Singh D., Rawat M. S., Semalty A., Semalty M. (2012). Quercetin-phospholipid complex: an amorphous pharmaceutical system in herbal drug delivery. Curr. Drug Discov. Technol. 9 (1), 17–24. 10.2174/157016312799304507 [DOI] [PubMed] [Google Scholar]
- Singh P., Arif Y., Bajguz A., Hayat S. (2021). The role of quercetin in plants. Plant Physiol. Biochem. 166, 10–19. 10.1016/j.plaphy.2021.05.023 [DOI] [PubMed] [Google Scholar]
- Soysa N. S., Alles N. (2016). Osteoclast function and bone-resorbing activity: an overview. Biochem. Biophys. Res. Commun. 476 (3), 115–120. 10.1016/j.bbrc.2016.05.019 [DOI] [PubMed] [Google Scholar]
- Steitz S. A., Speer M. Y., McKee M. D., Liaw L., Almeida M., Yang H., et al. (2002). Osteopontin inhibits mineral deposition and promotes regression of ectopic calcification. Am. J. Pathol. 161 (6), 2035–2046. 10.1016/S0002-9440(10)64482-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sun J., Pan Y., Li X., Wang L., Liu M., Tu P., et al. (2022). Quercetin attenuates osteoporosis in orchiectomy mice by regulating glucose and lipid metabolism via the GPRC6A/AMPK/mTOR signaling pathway. Front. Endocrinol. (Lausanne) 13, 849544. 10.3389/fendo.2022.849544 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takayanagi H., Kim S., Koga T., Nishina H., Isshiki M., Yoshida H., et al. (2002). Induction and activation of the transcription factor NFATc1 (NFAT2) integrate RANKL signaling in terminal differentiation of osteoclasts. Dev. Cell 3 (6), 889–901. 10.1016/s1534-5807(02)00369-6 [DOI] [PubMed] [Google Scholar]
- Tao H., Li W., Zhang W., Yang C., Zhang C., Liang X., et al. (2021). Urolithin A suppresses RANKL-induced osteoclastogenesis and postmenopausal osteoporosis by, suppresses inflammation and downstream NF-κB activated pyroptosis pathways. Pharmacol. Res. 174, 105967. 10.1016/j.phrs.2021.105967 [DOI] [PubMed] [Google Scholar]
- Teitelbaum S. L., Ross F. P. (2003). Genetic regulation of osteoclast development and function. Nat. Rev. Genet. 4 (8), 638–649. 10.1038/nrg1122 [DOI] [PubMed] [Google Scholar]
- Tripathi G., Raja N., Yun H. S. (2015). Effect of direct loading of phytoestrogens into the calcium phosphate scaffold on osteoporotic bone tissue regeneration. J. Mater Chem. B 3 (44), 8694–8703. 10.1039/c5tb01574j [DOI] [PubMed] [Google Scholar]
- Tsai C. F., Chen G. W., Chen Y. C., Shen C. K., Lu D. Y., Yang L. Y., et al. (2021). Regulatory effects of quercetin on M1/M2 macrophage polarization and oxidative/antioxidative balance. Nutrients 14 (1), 67. 10.3390/nu14010067 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Udagawa N., Koide M., Nakamura M., Nakamichi Y., Yamashita T., Uehara S., et al. (2021). Osteoclast differentiation by RANKL and OPG signaling pathways. J. Bone Min. Metab. 39 (1), 19–26. 10.1007/s00774-020-01162-6 [DOI] [PubMed] [Google Scholar]
- Vakili S., Zal F., Mostafavi-Pour Z., Savardashtaki A., Koohpeyma F. (2021). Quercetin and vitamin E alleviate ovariectomy-induced osteoporosis by modulating autophagy and apoptosis in rat bone cells. J. Cell Physiol. 236 (5), 3495–3509. 10.1002/jcp.30087 [DOI] [PubMed] [Google Scholar]
- Velletri T., Huang Y., Wang Y., Li Q., Hu M., Xie N., et al. (2021). Loss of p53 in mesenchymal stem cells promotes alteration of bone remodeling through negative regulation of osteoprotegerin. Cell Death Differ. 28 (1), 156–169. 10.1038/s41418-020-0590-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang N., Wang L., Yang J., Wang Z., Cheng L. (2021). Quercetin promotes osteogenic differentiation and antioxidant responses of mouse bone mesenchymal stem cells through activation of the AMPK/SIRT1 signaling pathway. Phytother. Res. 35 (5), 2639–2650. 10.1002/ptr.7010 [DOI] [PubMed] [Google Scholar]
- Wang P., Xia X. F., Zuo B. (2020). Preventive effect of quercetin on disused osteoporosis in rats and its effect on ERK1/2-MAPK signaling pathway. Med. Pharm. J. Chin. People's Liberation Army 32 (11), 6–10. [Google Scholar]
- Wang W., Sun C., Mao L., Ma P., Liu F., Yang J., et al. (2016). The biological activities, chemical stability, metabolism and delivery systems of quercetin: a review. Trends Food Sci. Technol. 56 (56), 21–38. 10.1016/j.tifs.2016.07.004 [DOI] [Google Scholar]
- Wang Y. (2008). Effects of quercetin in ongenital system and bone mineral density in ovariectomized rats. China Pract. Med. (18), 22–24. [Google Scholar]
- Wang Y. (2009). Effects of quercetin on the expression of osteoprotegerin and receptor activator for NFκB ligand in femurs of ovariectomized rats. Her. Med. 28 (08), 999–1002. [Google Scholar]
- Wang Y., Che L., Chen X., He Z., Song D., Yuan Y., et al. (2023). Repurpose dasatinib and quercetin: targeting senescent cells ameliorates postmenopausal osteoporosis and rejuvenates bone regeneration. Bioact. Mater 25, 13–28. 10.1016/j.bioactmat.2023.01.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Y., Li C., Wan Y., Qi M., Chen Q., Sun Y., et al. (2021). Quercetin-loaded ceria nanocomposite potentiate dual-directional immunoregulation via macrophage polarization against periodontal inflammation. Small 17 (41), e2101505. 10.1002/smll.202101505 [DOI] [PubMed] [Google Scholar]
- Wattel A., Kamel S., Prouillet C., Petit J. P., Lorget F., Offord E., et al. (2004). Flavonoid quercetin decreases osteoclastic differentiation induced by RANKL via a mechanism involving NF kappa B and AP-1. J. Cell Biochem. 92 (2), 285–295. 10.1002/jcb.20071 [DOI] [PubMed] [Google Scholar]
- Wu D. F., Zhang L., Zhang S. P. (2008). Distribution of quercetin in plasma and tissues in rats. Chin. J. Hosp. Pharm. 28 (21), 1822–1824. [Google Scholar]
- Xiao J., Zhang G., Chen B., He Q., Mai J., Chen W., et al. (2023). Quercetin protects against iron overload-induced osteoporosis through activating the Nrf2/HO-1 pathway. Life Sci. 322, 121326. 10.1016/j.lfs.2022.121326 [DOI] [PubMed] [Google Scholar]
- Xing L. Z., Ni H. J., Wang Y. L. (2017). Quercitrin attenuates osteoporosis in ovariectomized rats by regulating mitogen-activated protein kinase (MAPK) signaling pathways. Biomed. Pharmacother. 89, 1136–1141. 10.1016/j.biopha.2017.02.073 [DOI] [PubMed] [Google Scholar]
- Xing X., Tang Q., Zou J., Huang H., Yang J., Gao X., et al. (2023). Bone-targeted delivery of senolytics to eliminate senescent cells increases bone formation in senile osteoporosis. Acta Biomater. 157, 352–366. 10.1016/j.actbio.2022.11.056 [DOI] [PubMed] [Google Scholar]
- Yang B., Li S., Chen Z., Feng F., He L., Liu B., et al. (2020). Amyloid β peptide promotes bone formation by regulating Wnt/β-catenin signaling and the OPG/RANKL/RANK system. FASEB J. 34 (3), 3583–3593. 10.1096/fj.201901550R [DOI] [PubMed] [Google Scholar]
- Yang D., Wang T., Long M. (2020). Quercetin: its main pharmacological activity and potential application in clinical medicine. Oxid. Med. Cell Longev. 2020, 8825387. 10.1155/2020/8825387 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang J. H. (2022). Evaluation on of the efficacy of open reduction and internal fixation in the treatment of femoral head fracture and the mechanism research of quercetin in promoting osteogenic differentiation. China: Zhengzhou University. [Google Scholar]
- Yang Y. J., Yang Z. L., Wang D. C., Xiao X. c., Li P. (2006). Comparative study on effects of rutin and quercetin on metabolism in osteoblast cells. J. Chin. Med. Mater. 29 (05), 467–470. 10.13863/j.issn1001-4454.2006.05.024 [DOI] [PubMed] [Google Scholar]
- Yao Z., Getting S. J., Locke I. C. (2021). Regulation of TNF-induced osteoclast differentiation. Cells 11 (1), 132. 10.3390/cells11010132 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu D., Huang C., Jiang C., Zhu H. (2021). Features of a simvastatin-loaded multi-layered co-electrospun barrier membrane for guided bone regeneration. Exp. Ther. Med. 22 (1), 713. 10.3892/etm.2021.10145 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yuan Z., Min J., Wang K. (2018). Effects of different pharmacological components of eucommia ulmoides flavonoids on postmenopausal osteoporosis. Chin. J. Osteoporos. 24 (02), 244–248. [Google Scholar]
- Zhang B., Hu L. Y., Gou L. (2022). The effect of quercetin on the proliferation and osteogenic differentiation of human bone marrow mesenchymal stem cells and its molecular mechanism. Chin. J. Osteoporos. 28 (12), 1765–1769. [Google Scholar]
- Zhang Q., Chang B., Zheng G., Du S., Li X. (2020). Quercetin stimulates osteogenic differentiation of bone marrow stromal cells through miRNA-206/connexin 43 pathway. Am. J. Transl. Res. 12 (5), 2062–2070. [PMC free article] [PubMed] [Google Scholar]
- Zhang X., Aubin J. E., Inman R. D. (2003). Molecular and cellular biology of new bone formation: insights into the ankylosis of ankylosing spondylitis. Curr. Opin. Rheumatol. 15 (4), 387–393. 10.1097/00002281-200307000-00004 [DOI] [PubMed] [Google Scholar]
- Zhang Y., Yang X., Ge X., Zhang F. (2019). Puerarin attenuates neurological deficits via Bcl-2/Bax/cleaved caspase-3 and Sirt3/SOD2 apoptotic pathways in subarachnoid hemorrhage mice. Biomed. Pharmacother. 109, 726–733. 10.1016/j.biopha.2018.10.161 [DOI] [PubMed] [Google Scholar]
- Zhang Y. W., Li Y. J., Lu P. P., Dai G. C., Chen X. X., Rui Y. F. (2021). The modulatory effect and implication of gut microbiota on osteoporosis: from the perspective of “brain–gut–bone” axis. Food and Funct. 12 (13), 5703–5718. 10.1039/d0fo03468a [DOI] [PubMed] [Google Scholar]
- Zhao X., Deng Y., Xue X., Liao L., Zhou M., Peng C., et al. (2022). Research progress of quercetin delivery systems. Curr. Pharm. Des. 28 (9), 727–742. 10.2174/1381612828666220317141923 [DOI] [PubMed] [Google Scholar]
- Zheng H., Tang W., Jiao J. L. (2017). Molecular mechanism of quercetin ameliorates on the castration osteoporosis rats by promoting osteogenetic differentiation. Pharmacol. Clin. Chin. Materia Medica 33 (05), 16–20. 10.13412/j.cnki.zyyl.2017.05.005 [DOI] [Google Scholar]
- Zheng Y., Zhao L., Yi J., Cai S. (2022). Effects and mechanisms of rhus chinensis mill. Fruits on suppressing RANKL-induced osteoclastogenesis by network pharmacology and validation in RAW264.7 cells. Nutrients 14 (5), 1020. 10.3390/nu14051020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou C., Shen S., Zhang M., Luo H., Zhang Y., Wu C., et al. (2023). Mechanisms of action and synergetic formulas of plant-based natural compounds from traditional Chinese medicine for managing osteoporosis: a literature review. Front. Med. (Lausanne) 10, 1235081. 10.3389/fmed.2023.1235081 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou Y., Wu Y., Jiang X., Zhang X., Xia L., Lin K., et al. (2015). The effect of quercetin on the osteogenesic differentiation and angiogenic factor expression of bone marrow-derived mesenchymal stem cells. PLoS One 10 (6), e0129605. 10.1371/journal.pone.0129605 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou Y., Wu Y., Ma W., Jiang X., Takemra A., Uemura M., et al. (2017). The effect of quercetin delivery system on osteogenesis and angiogenesis under osteoporotic conditions. J. Mater Chem. B 5 (3), 612–625. 10.1039/c6tb02312f [DOI] [PubMed] [Google Scholar]
- Zhu F., Li W., Wang L., Dai B., Liu Z., Wu H., et al. (2023). Study on the treatment of postmenopausal osteoporosis with quercetin in Liuwei Dihuang Pill based on network pharmacology. J. Orthop. Surg. Res. 18 (1), 21. 10.1186/s13018-022-03470-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu X. J., Wei S. Q. (2005). Protective effect of quercetin on ovariectomy-induced bone loss in rats. Chin. J. Osteoporos. (04), 96–100. [Google Scholar]
- Zou H., Ye H., Kamaraj R., Zhang T., Zhang J., Pavek P. (2021). A review on pharmacological activities and synergistic effect of quercetin with small molecule agents. Phytomedicine 92, 153736. 10.1016/j.phymed.2021.153736 [DOI] [PubMed] [Google Scholar]