Abstract
Pancreatic cancer represents a formidable challenge in oncology, primarily due to its aggressive nature and limited therapeutic options. The prognosis of patients with pancreatic ductal adenocarcinoma (PDAC), the main form of pancreatic cancer, remains disappointingly poor with a 5-year overall survival of only 5%. Almost 95% of PDAC patients harbor Kirsten rat sarcoma virus (KRAS) oncogenic mutations. KRAS activates downstream intracellular pathways, most notably the rapidly accelerated fibrosarcoma (RAF)/mitogen-activated protein kinase kinase (MEK)/extracellular signal-regulated kinase (ERK) signaling axis. Dysregulation of the RAF/MEK/ERK pathway is a crucial feature of pancreatic cancer and therefore its main components, RAF, MEK and ERK kinases, have been targeted pharmacologically, largely by small-molecule inhibitors. The recent advances in the development of inhibitors not only directly targeting the RAF/MEK/ERK pathway but also indirectly through inhibition of its regulators, such as Src homology-containing protein tyrosine phosphatase 2 (SHP2) and Son of sevenless homolog 1 (SOS1), provide new therapeutic opportunities. Moreover, the discovery of allele-specific small-molecule inhibitors against mutant KRAS variants has brought excitement for successful innovations in the battle against pancreatic cancer. Herein, we review the recent advances in targeted therapy and combinatorial strategies with focus on the current preclinical and clinical approaches, providing critical insight, underscoring the potential of these efforts and supporting their promise to improve the lives of patients with PDAC.
Keywords: pancreatic cancer, RAF/MEK/ERK pathway, small-molecule inhibitors, KRAS, targeted therapy
1. Introduction
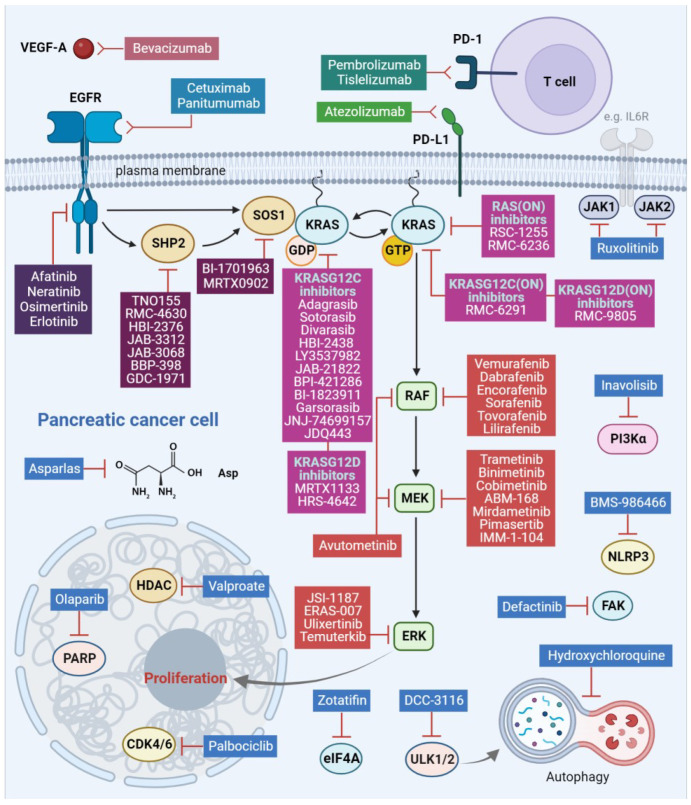
Pancreatic cancer is one of the deadliest tumors and is expected to become the second leading cause of cancer-related mortality in the US. The 5-year overall survival (OS) of patients with pancreatic ductal adenocarcinoma (PDAC), the most common form of pancreatic cancer, has only minimally improved to only 11%, presenting a modest improvement compared to other malignancies [1,2]. The role of the rapidly accelerated fibrosarcoma (RAF)/mitogen-activated protein kinase kinase (MEK)/extracellular signal-regulated kinase (ERK) pathway as the main RAS effector pathway in initiation and progression of pancreatic cancer is well established [3]. Targeted therapy using small-molecule inhibitors against components of the RAF/MEK/ERK pathway has shown significant potential for PDAC. The Kirsten rat sarcoma virus (KRAS) mutation is a hallmark of PDAC, and only recently has there been progress in drug development, with compounds that directly target the once considered “undruggable” RAS. These compounds include KRAS-mutant-specific inhibitors, that are foreseen to change the landscape in PDAC management [4] (Figure 1).
Figure 1.
RAF/MEK/ERK pathway inhibitors and combination therapies under clinical evaluation for pancreatic cancer. EGFR, epidermal growth factor receptor; SHP2, Src homology 2 domain-containing phosphatase 2; SOS1, Son of sevenless homolog 1; KRAS, Kirsten rat sarcoma viral oncogene homolog; GDP, guanosine diphosphate; GTP, guanosine triphosphate; RAF, rapidly accelerated fibrosarcoma; MEK, mitogen-activated protein kinase kinase; ERK, extracellular signal-regulated kinase; CDK4/6, cyclin-dependent kinase 4/6; HDAC, histone deacetylase; PARP, poly-adenosine diphosphate (ADP) ribose polymerase; eIF4A, eukaryotic translation initiation factor 4A; ULK1/2, unc-51-like autophagy-activating kinases 1 and 2; NLRP3, Nod-like receptor protein 3; FAK, focal adhesion kinase; JAK1/2, Janus kinase 1/2; PD-1, programmed cell death protein 1; PD-L1, programmed death ligand 1; VEGF-A, vascular endothelial growth factor A; PI3Kα, phosphoinositide 3-kinase α; IL6R, interleukin 6 receptor; Asp, asparagine. This figure was created using the tools provided by BioRender.com (accessed on 21 January 2024).
Here, we discuss rational treatment approaches with the currently available therapeutic options for PDAC patients, including novel targeting strategies using current and new compounds. We focus on the combinatorial strategies and the current clinical attempts for evaluation of the RAF/MEK/ERK pathway inhibitors that are currently in clinical development. This is significant because there is an urgent need to establish new frameworks and improve future treatments. Our aim is to contribute to understanding the complexity of the RAF/MEK/ERK pathway inhibition, which holds substantial promise for developing effective treatment modalities against this aggressive malignancy.
2. RAF/MEK/ERK Signaling Pathway in Pancreatic Cancer
The RAF/MEK/ERK pathway which controls cell growth, differentiation and survival is often upregulated in pancreatic cancer. The orchestrator of this upregulation is the small GTPase KRAS, which is mutated in 95% of patients with pancreatic cancer [2]. The most common KRAS mutations in PDAC are substitutions in position G12, with KRASG12D (41%), KRASG12V (34%) and KRASG12R (16%) being the most frequent and G12C (1–2%) the least [5]. KRAS in its active GTP-bound form promotes RAF kinase activation through dimerization and phosphorylation, resulting in phosphorylation of its substrate MEK kinase. MEK phosphorylates and activates the terminal kinase ERK. Activated ERK regulates growth-promoting transcription [2]. The RAF/MEK/ERK pathway is the key effector pathway for initiation and progression of KRAS-driven PDAC [3]. Therefore, apart from targeted efforts against the key members of the RAF/MEK/ERK pathway, several drugs, targeting different components of this pathway, including the upstream epidermal growth factor receptor (EGFR) family members and the RAF/MEK/ERK pathway regulators Src homology-containing protein tyrosine phosphatase 2 (SHP2) and Son of sevenless homolog 1 (SOS1), have been explored extensively for therapeutic intervention in PDAC [2,3,4,5] (Figure 1).
3. Targeting Strategies
3.1. EGFR Family Inhibition
Initial efforts were directed against the upstream frequently dysregulated EGFR/human epidermal growth factor receptor 2 (HER2 or ERBB2) signaling. Erlotinib, an EGFR inhibitor, combined with gemcitabine, a first-line chemotherapy, in patients with advanced PDAC, showed modest survival benefits [6]. When erlotinib was combined with gemcitabine together with nab-paclitaxel, a tubulin-polymerization stabilizer, it exhibited some clinical activity despite the observed toxicities [7]. However, combining cetuximab, a monoclonal antibody against EGFR, with gemcitabine did not show improved outcome [8]. Similarly, the combination of lapatinib, a dual tyrosine kinase inhibitor against both HER2 and EGFR, with gemcitabine or capecitabine did not demonstrate any efficacy [9,10]. To improve these results, a second-generation ERBB family inhibitor, afatinib, was used in combination with gemcitabine. Afatinib binds covalently to cysteine 797 of the EGFR and the corresponding cysteines 805 and 803 in HER2 and human epidermal growth factor receptor 4 (ErB4/HER4), respectively, inhibiting downstream signaling from all homo- and heterodimers formed by ERBB family members. However, again, this combination did not show any efficacy [11]. Subsequent clinical studies, based on preclinical synergistic evidence, assessed the EGFR inhibition in combination with components of the RAF/MEK/ERK pathway such as BRAF and MEK. These studies combined erlotinib with either sorafenib [12], a multikinase RAF inhibitor, or selumetinib [13], a MEK inhibitor, but showed modest activity. More recently, the addition of panitumumab, an EGFR monoclonal antibody, to erlotinib and gemcitabine demonstrated a small but significantly prolonged overall survival, despite the observed toxicities [14]. Overall, these studies did not show sufficient evidence of effectiveness. This agrees with the conclusions from a retrospective analysis showing that EGFR and KRAS alterations were not predictive for patient benefit from anti-EGFR therapy [15]. However, in a preclinical study, the highly selective irreversible EGFR/HER2 inhibitor neratinib suppressed KRAS mutant levels in PDAC cells [16]. The efficacy of neratinib in combination with valproate, a histone deacetylase (HDAC) inhibitor, is being evaluated in a clinical trial in patients with advanced RAS-mutated solid tumors (Table 1).
Table 1.
RAF/MEK/ERK pathway inhibitors currently in clinical evaluation for pancreatic cancer. EGFR, epidermal growth factor receptor; ERBB2/HER2, receptor tyrosine-protein kinase erbB 2; RAF, rapidly accelerated fibrosarcoma; SHP2, Src homology 2 domain-containing phosphatase 2; SOS1, Son of sevenless homolog 1; KRAS, Kirsten rat sarcoma viral oncogene homolog; RAS, rat sarcoma viral oncogene homolog; RAF, rapidly accelerated fibrosarcoma; MEK, mitogen-activated protein kinase kinase; ERK, extracellular signal-regulated kinase; CDK4/6, cyclin-dependent kinase 4/6; HDAC, histone deacetylase; PARP, poly-adenosine diphosphate (ADP) ribose polymerase; eIF4A, eukaryotic translation initiation factor 4A; ULK1/2, unc-51-like autophagy-activating kinases 1 and 2; NLRP3, Nod-like receptor protein 3; PD-1, programmed cell death protein 1; PD-L1, programmed death ligand 1; VEGF-A, vascular endothelial growth factor A; PI3Kα, phosphoinositide 3-kinase α; FAK, focal adhesion kinase; JAK1/2, Janus kinase 1/2.
| Drug(s) | Target(s) | Second Drug(s) |
Second Target(s) |
Phase | Clinical Study Code |
|---|---|---|---|---|---|
| Neratinib | EGFR, ERBB2/HER2 | Divalproex sodium (Valproate) |
HDAC | I/II | NCT03919292 |
| Vemurafenib | BRAFV600E/K | Sorafenib | RAF | II | NCT05068752 |
| Lilirafenib | BRAF | Mirdametinib | MEK | I | NCT03905148 |
| Tovorafenib | RAF | Pimasertib | MEK | I/II | NCT04985604 |
| Avutometinib | MEK, RAF | Defactinib | FAK | I/II | NCT05669482 |
| ABM-168 | MEK | I | NCT05831995 | ||
| Binimetinib | MEK | Hydroxychloroquine | Autophagy | I | NCT04132505 |
| Binimetinib | MEK | Encorafenib | RAFV600E/K | II | NCT04390243 |
| Binimetinib | MEK | Palbociclib | CDK4/6 | II | NCT05554367 |
| Trametinib | MEK | Hydroxychloroquine | Autophagy | I | NCT03825289 |
| Trametinib | MEK | Ruxolitinib | JAK1/JAK2 | I | NCT04303403 |
| Cobimetinib | MEK | Calaspargase pegol-mnkl (Asparlas) |
Asparagine | I | NCT05034627 |
| IMM-1-104 | MEK | I/II | NCT05585320 | ||
| Temuterkib | ERK | RMC-4630 | SHP2 | I | NCT04916236 |
| Temuterkib | ERK | Hydroxychloroquine sulfate | Autophagy | II | NCT04386057 |
| Ulixertinib | ERK | Palbociclib | CDK4/6 | I | NCT03454035 |
| ERAS-007 | ERK | Encorafenib Palbociclib Cetuximab * |
BRAFV600E/K CDK4/6 EGFR |
I/II | NCT05039177 |
| BI-1701963 | SOS1 | Adagrasib | KRASG12C | I | NCT04975256 |
| BI-1701963 | SOS1 | Trametinib | MEK | I | NCT04111458 |
| HBI-2376 | SHP2 | I | NCT05163028 | ||
| JAB-3068 | SHP2 | I/II | NCT03565003 | ||
| JAB-3312 | SHP2 | I | NCT04045496 | ||
| JAB-3312 | SHP2 | Binimetinib Pembrolizumab * Sotorasib Osimertinib |
MEK PD-1 KRASG12C EGFR |
I/II | NCT04720976 |
| BBP-398 | SHP2 | Sotorasib | KRASG12C | I | NCT05480865 |
| RMC-6291 | KRASG12C | I | NCT05462717 | ||
| RMC-6291 | KRASG12C | RMC-6236 | RAS (pan-mutant and wild-type) |
I | NCT06128551 |
| HBI-2438 | KRASG12C | I | NCT05485974 | ||
| LY3537982 | KRASG12C | I | NCT04956640 | ||
| JAB-21822 | KRASG12C | II | NCT06008288 | ||
| JAB-21822 | KRASG12C | Cetuximab * | EGFR | I/II | NCT05002270 |
| JAB-21822 | KRASG12C | JAB-3312 | SHP2 | I/II | NCT05288205 |
| Adagrasib | KRASG12C | I | NCT05634525 | ||
| Adagrasib | KRASG12C | TNO155 | SHP2 | I/II | NCT04330664 |
| Adagrasib | KRASG12C | Afatinib Cetuximab * Pembrolizumab * |
EGFR/HER2 EGFR PD-1 |
I | NCT03785249 |
| Adagrasib | KRASG12C | Olaparib | PARP | I | NCT06130254 |
| Adagrasib | KRASG12C | BMS-986466 † −/+ cetuximab * |
NLRP3 EGFR |
I/II | NCT06024174 |
| Adagrasib | KRASG12C | MRTX0902 | SOS1 | I/II | NCT05578092 |
| BPI-421286 | KRASG12C | I | NCT05315180 | ||
| BI-1823911 | KRASG12C | BI-1701963 | SOS1 | I | NCT04973163 |
| Divarasib | KRASG12C | Atezolizumab * Cetuximab * Bevacizumab * Erlotinib GDC-1971 Inavolisib |
PD-L1 EGFR VEGFA EGFR SHP2 PI3Kα |
I | NCT04449874 |
| Garsorasib | KRASG12C | I | NCT04585035 | ||
| JNJ-74699157 | KRASG12C | I | NCT04006301 | ||
| JDQ443 | KRASG12C | TNO155 Tislelizumab * |
SHP2 PD-1 |
I/II | NCT04699188 |
| MK-1084 | KRASG12C | Pembrolizumab * | PD-1 | I | NCT05067283 |
| Sotorasib | KRASG12C | I/II | NCT03600883 | ||
| Sotorasib § | KRASG12C | II | NCT04185883 | ||
| Sotorasib | KRASG12C | I | NCT04380753 | ||
| Sotorasib | KRASG12C | Panitumumab * | EGFR | II | NCT05638295 |
| Sotorasib | KRASG12C | Panitumumab* | EGFR | II | NCT05993455 |
| Sotorasib | KRASG12C | DCC-3116 † | ULK1/2 | I/II | NCT04892017 |
| Sotorasib | KRASG12C | Zotatifin † | eIF4A | I/II | NCT04092673 |
| MRTX1133 | KRASG12D | I/II | NCT05737706 | ||
| RMC-9805 | KRASG12D | I | NCT06040541 | ||
| HRS-4642 | KRASG12D | I | NCT05533463 | ||
| RMC-6236 | RAS (pan-mutant and wild-type) |
I | NCT05379985 | ||
| RSC-1255 | RAS (pan-mutant and wild-type) |
I | NCT04678648 |
* Monoclonal antibody; § as monotherapy or in combination with various anti-cancer agents; † main test drug of the trial.
3.2. RAF/MEK/ERK Pathway Component Inhibition
3.2.1. RAF Inhibition
Early attempts to target BRAF in unselected patients with advanced PDAC, using the BRAF inhibitor sorafenib in combination with gemcitabine did not show any benefit [17]. This is possibly explained by the fact that sorafenib is a multikinase inhibitor and its clinical activity is generally attributed to off-target inhibition. The use of the current clinically available BRAF inhibitors (vemurafenib, dabrafenib and encorafenib) is FDA approved for BRAFV600E-mutant metastatic melanoma but not for RAS-mutant tumors. BRAFV600E oncoprotein signals as a monomer and current BRAF inhibitors target and inhibit BRAF monomers. However, this selectivity limits their effectiveness in RAS-driven tumors, where RAFs (BRAF and CRAF) signal as dimers [18]. Additionally, in RAS-mutant tumors these RAF inhibitors promote paradoxical activation of the mitogen-activated protein kinase (MAPK) signaling by inducing wild-type RAF dimerization [19]. Next-generation RAF inhibitors that inhibit both dimers and monomers are currently in clinical development. These RAF inhibitors induce minimal paradoxical activation and show preclinical activity in RAS-mutant tumors [18,20,21,22]. A clinical trial testing the efficacy of the next-generation RAF inhibitor lilirafenib, including KRAS-mutant PDAC patients, reported stable disease as best response [23]. Additionally, a second current clinical trial is assessing the combination of lilirafenib with the MEK inhibitor mirdametinib in patients with advanced or refractory solid tumors (Table 1). Another clinical trial is evaluating combined vemurafenib and sorafenib treatment in individuals with KRAS-mutant PDAC who have progressed on standard chemotherapy (Table 1).
Activating BRAF alterations make up approximately 30% of KRAS wild-type PDAC and 2% of all PDAC cases [24]. These most commonly include substitutions in position V600, most commonly BRAFV600E. BRAF mutations are mutually exclusive with KRAS mutations and are typically associated with poor prognosis [25]. Multiple preclinical BRAF-mutated models suggest that these alterations can be targeted with combination of BRAF and MEK inhibitors [20,26,27]. Furthermore, BRAFV600E expression in a genetically engineered mouse model of PDAC was sufficient to induce the formation of pancreatic intraepithelial neoplasia lesions, revealing the central role of the RAF/MEK/ERK pathway in PDAC tumorigenesis [3]. Additionally, in a patient-derived orthotopic mouse model of PDAC, treatment with the MEK inhibitors trametinib or cobimetinib resulted in tumor suppression [28]. Molecular targeting of BRAFV600E in KRAS wild-type PDAC, using BRAF and MEK inhibitor, has been reported in several case reports in which patients progressed after first lines of chemotherapy. A case report with a patient with BRAF-mutant advanced PDAC reported objective tumor response to combined vemurafenib plus trametinib treatment [29]. Li et al. reported a case of a patient with metastatic BRAFV600E-mutant PDAC who achieved almost a complete response to dabrafenib plus trametinib treatment. Notably, the patient was rechallenged successfully with the regimen after relapse [30]. Two BRAF-mutant PDAC patients showed a significant reduction in carbohydrate antigen 19-9 levels, a PDAC-associated tumor antigen, following co-treatment with dabrafenib plus trametinib [31]. Wang et al. reported a partial response in the case of advanced metastatic PDAC, after vemurafenib plus trametinib administration [32]. In a recent case report, two patients with BRAFV600E-mutant PDAC exhibited a favorable response to dabrafenib and trametinib co-treatment [33]. Ardalan et al. reported that the addition of the MEK inhibitor cobimetinib to gemcitabine and nab-paclitaxel in BRAF-mutant patients was followed by a complete response to therapy for 16 months [34]. Furthermore, a clinical trial is underway evaluating the combination of encorafenib with the MEK inhibitor binimetinib in BRAFV600E-mutant PDAC patients (Table 1).
3.2.2. MEK Inhibition
Despite the promising preclinical evidence suggesting potent MAPK pathway inhibition, using MEK inhibitors in PDAC, early clinical trials showed limited efficacy. MEK inhibitors, trametinib or pimasertib, in combination with gemcitabine did not show any benefit when compared with gemcitabine alone [35,36]. Of note, the combination of the MEK inhibitor refametinib with gemcitabine was well tolerated and resulted in an objective response rate of 23%, with improved outcomes for KRAS wild-type patients [37]. In contrast, the assessment of selumetinib versus chemotherapy with capecitabine or the dual MEK and protein kinase B (AKT) kinase inhibition with selumetinib and the AKT inhibitor MK-2206 versus oxaliplatin-5-flourouracil-based chemotherapy in patients with advanced PDAC did not show any efficacy [38,39]. Another study on the combination of trametinib with the mammalian target of rapamycin (mTOR) inhibitor everolimus showed modest clinical efficacy, although it was unable to define optimal doses for the two compounds [40]. Moreover, when trametinib was combined with the CDK4/6 inhibitor ribociclib there was no benefit, and the study was terminated [41]. Likewise, in a combination of binimetinib with either the poly-adenosine diphosphate (ADP) ribose polymerase (PARP) inhibitor talazoparib or the programmed death 1 (PD-1) ligand 1 (PD-L1) inhibitor avelumab in patients with metastatic PDAC, no objective responses were observed [42]. Interestingly, preclinical evidence suggests that pancreatic tumors with KRASG12R, the third most common KRAS mutation in PDAC (16%), are more sensitive to MEK or ERK inhibition [43]. This is supported by the documented clinical benefit for patients with KRASG12R-mutant PDAC treated with MEK inhibitors [44,45]. More recently, a phase I clinical trial evaluated ABM-168, a novel small-molecule, allosteric, highly selective MEK inhibitor in adults with advanced solid tumors, including pancreatic carcinoma, who had confirmed RAS, RAF or neurofibromatosis type 1 (NF-1) mutations (Table 1). Another ongoing clinical study is testing the MEK inhibitor cobimetinib in combination with the enzyme calaspargase pegol-mnkl (asparlas) that blocks the biosynthesis of the non-essential amino acid asparagine, leading to starvation of cancer cells (Table 1) [46].
3.2.3. ERK Inhibition
The clinical development of ERK inhibitors raised the hope that direct ERK inhibition could block the MAPK pathway oncogenic transcriptional output. However, early clinical trials using ERK inhibitors against RAS-mutant tumors, including PDAC, were unsuccessful [47,48]. In a recent study, ERK inhibition induced autophagy in KRAS-mutant PDAC and the dual ERK and autophagy inhibition, using SCH772984 and hydrochloroquine, respectively, resulted in enhanced anti-tumor activity in PDAC preclinical models [43,49]. Several clinical studies are testing the synergistic effect of combining hydroxychloroquine with binimetinb, trametinib or the ERK inhibitor temuterkib (Table 1). Cyclin-dependent kinase 4/6 (CDK4/6) is a downstream target of activated/phosphorylated ERK and there is evidence for anti-tumor activity of the dual ERK and CDK4/6 inhibition, using ulixertinib and palbociclib, respectively (Table 1) [50,51] (Figure 1).
3.3. RAF/MEK/ERK Pathway Regulator Inhibition
3.3.1. SHP2 Inhibition
The discovery of SHP2 inhibitors revealed the dependency of KRAS-mutant tumors in SHP2 [52]. In addition, several studies demonstrated that SHP2 inhibition prevents the receptor tyrosine kinase (RTK)-mediated development of adaptive resistance caused by MEK or BRAF inhibitors [53,54]. Accordingly, co-targeting SHP2 and MEK or ERK using small-molecule inhibitors has been investigated preclinically, showing promising results in various KRAS-mutant tumors, including PDAC [52,53,54,55,56]. In another strategy, SHP2 inhibitors are combined with the recently developed allele-specific KRASG12C inhibitors to overcome the development of adaptive resistance mediated by wild-type RAS [57]. This approach has been evaluated by two clinical trials testing the combination of a SHP2 inhibitor with a KRASG12C inhibitor, TNO155 with MRTX849 and JAB-3312 with JAB-21822, respectively, in KRASG12C-mutant patients with advanced solid tumors (Table 1). Another aspect of SHP2 inhibition is its reported immunomodulatory function [58]. Based on this evidence, the combination of SHP2 and KRASG12C inhibitors can promote anti-tumor immunity by disrupting MAPK-activating signals from the tumor microenvironment to cancer cells [59].
3.3.2. SOS1 Inhibition
In preclinical PDAC models, it has been shown that SOS1 is essential for the survival of RAS-mutated cancer cells [60]. Small-molecule SOS1 inhibitors that disrupt the SOS1–RAS interaction have been under development for the treatment of KRAS-mutated cancers. Recently, a selective SOS1 inhibitor, BI-3406, has been reported to reduce GTP-bound RAS levels and tumor growth across KRAS-driven cancer models [61,62]. Moreover, the combined treatment of BI-3406 with trametinib resulted in sustained RAF/MEK/ERK pathway inhibition and suppression of tumors in KRAS-mutated xenograft models, overcoming pathway feedback reactivation [62]. The corresponding clinical compound BI-1701963 was tested in a clinical trial for KRAS-mutated solid tumors, including PDAC, with preliminary data demonstrating good tolerability and modest activity [63]. The current second phase of the study is evaluating the effectiveness of the combination of BI-1701963 with trametinib (Table 1).
3.4. KRAS Inhibition
Sotorasib is a first-in-class small-molecule inhibitor developed to selectively target KRASG12C, providing evidence for in vivo activity [64]. Adagrasib, another KRASG12C inhibitor, has shown clinical activity in KRASG12C-mutated tumors, including PDAC [65]. Both inhibitors, FDA-approved for KRASG12C-mutant non-small cell lung cancer (NSCLC), trap KRASG12C in its inactive GDP-bound state and are now listed in the national comprehensive cancer network (NCCN) clinical practice guidelines as for additional KRASG12C-mutant histologies, including pancreatic and colorectal cancers [66]. Another more potent GDP-bound KRASG12C inhibitor, divarasib, in combination with various anti-cancer therapies (Table 1), has shown promising clinical benefit in a small cohort of patients with pancreatic adenocarcinoma harboring the KRASG12C mutation [67]. However, the low prevalence of KRASG12C mutation in PDAC (1–2%) limits the applicability of this approach. Luckily, MRTX1133, a “game-changer” compound, has been developed selectively targeting KRASG12D [68]. MRTX1133 is currently under clinical evaluation, while other novel compounds targeting KRASG12D as well (HRS-4642, RMC-9805) are being assessed in phase I clinical trials (Table 1). A novel non-covalent pan-KRAS inhibitor prevents the activation of wild-type KRAS and a range of KRAS mutants, excluding G12R and Q61L/K/R while sparing NRAS and HRAS isoforms (Kim). This pan-KRAS inhibitor showed preclinical anti-tumor activity in various models, indicating broad therapeutic implications in patients with KRAS-driven cancers, including pancreatic cancer [69]. ADT-007, another pan-KRAS inhibitor, that inhibits GTP binding to both mutated and wild-type KRAS, blocks oncogenic KRAS signaling and modulates T cell activation in preclinical PDAC in vitro and in vivo models [70]. Recently, tricomplex inhibitors that target the active GTP-bound state RAS(ON) for both mutant and wild-type RAS have shown promising results for KRASG12V-mutant cancers [71]. The first in class of these inhibitors, RMC-6232, forms a tricomplex with RAS(ON) and an abundant intracellular chaperon protein cyclophilin A, sterically inhibiting RAS binding to its effectors [72,73]. RMC-6232 is being assessed in a phase I clinical trial for KRASG12-mutant tumors (Table 1) and appears effective against KRAS position 12 (G12X) mutants, including G12D, G12V and G12R, inducing durable suppression of the RAS pathway activation in preclinical cellular in vitro and in vivo xenograft PDAC models [66,73].
3.5. Toxicity Challenges
As researchers explore the dynamic space of targeted therapy combinations in pancreatic cancer, the optimism of the recent advancements is tempered by the potential for overlapping toxicities in regimens incorporating two inhibitors targeting within the RAF/MEK/ERK pathway or combined with other targets, like in the case of dual inhibition with afatinib and trametinib [74]. Targeting the upstream regulators of the RAF/MEK/ERK pathway, SHP2 and SOS1, holds promise but at the same time raises concerns about unanticipated on-target toxicities, as evidenced by clear dose-associated cytopenias [75,76]. The clinical trial combining the KRASG12C inhibitor, sotorasib, with the anti-PD1 and anti-PD-L1 monoclonal antibodies pembrolizumab and atezolizumab, respectively, revealed increased liver toxicities [77]. The mechanisms driving these toxicities remain elusive, prompting hypotheses ranging from enhanced immune-mediated effects triggered by targeted therapies to potential off-target covalent protein–drug conjugates causing liver damage, exacerbated by systemic immune activation. Interestingly, Genentech’s GDC-6036, a KRASG12C inhibitor administered at lower doses, has shown reduced liver toxicities in phase I testing, suggesting that dosage adjustments may play a crucial role in mitigating adverse effects [78]. Preclinical studies of the RMC-6236 tricomplex have shown success in inhibiting active RAS(ON), including cases of acquired resistance by KRASG12C inhibitors, but the ubiquitous nature of cyclophilin A introduces uncertainties about the therapeutic window and potential off-target activity [76,79]. Amidst the hope for breakthroughs in treating pancreatic cancer, the toxicity challenges underscore the critical need for meticulous exploration of treatment schedules, adjustments and a deep understanding of the intricate interplay within the complex signaling pathways [76].
4. Discussion—Future Perspectives
The presence of KRAS mutations in pancreatic cancer has significant prognostic implications, influencing both overall survival (OS) and treatment response. Patients with KRAS-mutated PDAC generally exhibit a poorer prognosis [80]. Furthermore, recent findings indicate distinct survival outcomes related to specific KRAS mutations. For instance, patients with KRASG12D-mutated PDAC demonstrated a significantly shorter median overall survival compared to those with KRASG12R mutations, indicating a prognostic value for KRASG12D mutation [81,82]. Notably, the type of KRAS mutation may also impact the response to first-line chemotherapy. Thus, FOLFIRINOX showed improved survival in patients with KRASG12D and KRASG12V mutations, while the patients with KRASG12C-mutated tumors exhibited longer overall survival when treated with gemcitabine plus nab-paclitaxel [83]. Additionally, the variant allele frequency (VAF) and allelic imbalance of KRAS further contribute to prognosis. Higher KRAS VAF is associated with shorter survival, and allelic imbalance, leading to increased mutant KRAS dosage, correlates with a more aggressive clinical behavior [84,85]. Beyond KRAS, BRAF mutational status seems to have prognostic value. In a case report, two PDAC patients who had not responded to initial systemic chemotherapy, after identification of BRAFV600E mutation through next generation sequencing, were treated with combined dabrafenib and trametinib and sustained a favorable response [33]. These findings underscore the importance of molecular profiling, specifically KRAS mutation characterization, in guiding prognosis and tailoring therapeutic strategies for pancreatic cancer patients.
The omnipresent KRAS mutation in PDAC and the progress in drug development of small-molecule inhibitors led the early therapeutic efforts targeting the main components downstream of the RAF/MEK/ERK pathway. Although preclinical studies demonstrated promising findings, the clinical attempts were unsuccessful due to low efficacy and dose-limiting toxicities [86]. However, when a precision medicine approach was followed the paradigm was shifted. Case reports indicate benefits when the BRAF mutational status was confirmed, in a wild-type KRAS context, before therapeutic intervention [29,30,31,32,33,34]. Pharmacological targeting of the canonical components of RAF/MEK/ERK signaling in RAS-dependent tumors is often limited by the development of adaptive resistance, which is usually mediated by feedback activation of RTK signaling, resulting in reactivation of the RAF/MEK/ERK pathway activity [53]. Thus, the strategy of targeting additional effectors downstream of RTKs and upstream of RAS, such as SHP2 and SOS1, is attractive. Therapies that directly target the mutated components of the pathway such as KRAS or BRAF could be combined with inhibitors against upstream regulators such as SHP2 and SOS1 and downstream pathway components such as MEK or ERK for a sustained inhibition (Table 1). The concept of dual pathway inhibition has been successfully tested in the case of BRAFV600E-mutated melanoma, where vertical double BRAF/MEK inhibition has gained FDA approval. Furthermore, in the context of BRAFV600E-mutant tumors there is evidence for effectiveness of a triple inhibitory strategy within the RAF/MEK/ERK pathway [87]. A recent clinical trial, based on promising preclinical data, tested the combination of avutometinib, a first-in-class RAF/MEK clamp and a compound designed to inhibit MEK and block RAF-mediated phosphorylation of MEK in combination with the focal adhesion kinase (FAK) inhibitor defactinib (Table 1, Figure 1). The recent approval of the KRASG12C inhibitor sotorasib for KRASG12C-mutated non-small cell lung cancer (NSCLC) allowed the enrolment of low-frequency KRASG12C-PDAC cases in clinical trials for evaluation (Table 1). Currently, direct inhibition of mutant RAS through allele-specific inhibitors provides a therapeutic opportunity. Interestingly, inhibition of KRASG12D, using MRTX1133, in immunocompetent PDAC models resulted in tumor suppression by increasing tumor-associated macrophages (TAMs) and tumor-infiltrating cytotoxic T cells [88]. This argues that KRASG12D inhibition has a potential immunomodulatory function, which may be beneficial especially for patients with pancreatic cancer, an immunologically “cold” malignancy.
5. Conclusions
Despite the progress in drug discovery, there is an additional need to develop novel, more potent and broader RAF/MEK/ERK pathway inhibitors, including KRAS-mutant inhibitors, for improved tailored targeted therapy [89]. Developing effective and mechanism-based combination therapy regimens is essential to maximizing the efficacy of RAF/MEK/ERK pathway inhibition, which holds great promise for pancreatic cancer control.
Acknowledgments
D.D.C. was supported by Fondazione Umberto Veronesi (FUV) and Fondazione Italiana per la ricerca sulle Malattie del Pancreas (FIMP).
Author Contributions
Conceptualization, C.A., D.D.C. and A.G.P.; writing—original draft preparation, C.A.; literature search, C.A.; supervision, D.D.C. and A.G.P.; writing—review and editing, A.G.P. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Data are contained within the article.
Conflicts of Interest
The authors declare no conflicts of interest.
Funding Statement
This research received no external funding.
Footnotes
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.
References
- 1.Nevala-Plagemann C., Hidalgo M., Garrido-Laguna I. From state-of-the-art treatments to novel therapies for advanced-stage pancreatic cancer. Nat. Rev. Clin. Oncol. 2020;17:108–123. doi: 10.1038/s41571-019-0281-6. [DOI] [PubMed] [Google Scholar]
- 2.Mollinedo F., Gajate C. Novel therapeutic approaches for pancreatic cancer by combined targeting of RAF→MEK→ERK signaling and autophagy survival response. Ann. Transl. Med. 2019;7:S153. doi: 10.21037/atm.2019.06.40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Collisson E.A., Trejo C.L., Silva J.M., Gu S., Korkola J.E., Heiser L.M., Charles R.P., Rabinovich B.A., Hann B., Dankort D., et al. A central role for RAF→MEK→ERK signaling in the genesis of pancreatic ductal adenocarcinoma. Cancer Discov. 2012;2:685–693. doi: 10.1158/2159-8290.CD-11-0347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Rudloff U. Emerging kinase inhibitors for the treatment of pancreatic ductal adenocarcinoma. Expert Opin. Emerg. Drugs. 2022;27:345–368. doi: 10.1080/14728214.2022.2134346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Waters A.M., Der C.J. KRAS: The Critical Driver and Therapeutic Target for Pancreatic Cancer. Cold Spring Harb. Perspect. Med. 2018;8:a031435. doi: 10.1101/cshperspect.a031435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Moore M.J., Goldstein D., Hamm J., Figer A., Hecht J.R., Gallinger S., Au H.J., Murawa P., Walde D., Wolff R.A., et al. Erlotinib plus gemcitabine compared with gemcitabine alone in patients with advanced pancreatic cancer: A phase III trial of the National Cancer Institute of Canada Clinical Trials Group. J. Clin. Oncol. 2007;41:1960–1966. doi: 10.1200/JCO.2006.07.9525. [DOI] [PubMed] [Google Scholar]
- 7.Cohen S.J., O’Neil B.H., Berlin J., Ames P., McKinley M., Horan J., Catalano P.M., Davies A., Weekes C.D., Leichman L. A phase 1b study of erlotinib in combination with gemcitabine and nab-paclitaxel in patients with previously untreated advanced pancreatic cancer: An Academic Oncology GI Cancer Consortium study. Cancer Chemother. Pharmacol. 2016;77:693–701. doi: 10.1007/s00280-016-2981-2. [DOI] [PubMed] [Google Scholar]
- 8.Philip P.A., Benedetti J., Corless C.L., Wong R., O’Reilly E.M., Flynn P.J., Rowland K.M., Atkins J.N., Mirtsching B.C., Rivkin S.E., et al. Phase III study comparing gemcitabine plus cetuximab versus gemcitabine in patients with advanced pancreatic adenocarcinoma: Southwest Oncology Group-directed intergroup trial S0205. J. Clin. Oncol. 2010;28:3605. doi: 10.1200/JCO.2009.25.7550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Safran H., Miner T., Bahary N., Whiting S., Lopez C.D., Sun W., Charpentier K., Shipley J., Anderson E., McNulty B., et al. Lapatinib and gemcitabine for metastatic pancreatic cancer. A phase II study. Am. J. Clin. Oncol. 2011;34:50–52. doi: 10.1097/COC.0b013e3181d26b01. [DOI] [PubMed] [Google Scholar]
- 10.Wu Z., Gabrielson A., Hwang J.J., Pishvaian M.J., Weiner L.M., Zhuang T., Ley L., Marshall J.L., He A.R. Phase II study of lapatinib and capecitabine in second-line treatment for metastatic pancreatic cancer. Cancer Chemother. Pharmacol. 2015;76:1309–1314. doi: 10.1007/s00280-015-2855-z. [DOI] [PubMed] [Google Scholar]
- 11.Haas M., Waldschmidt D.T., Stahl M., Reinacher-Schick A., Freiberg-Richter J., Fischer von Weikersthal L., Kaiser F., Kanzler S., Frickhofen N., Seufferlein T., et al. Afatinib plus gemcitabine versus gemcitabine alone as first-line treatment of metastatic pancreatic cancer: The randomised, open-label phase II ACCEPT study of the Arbeitsgemeinschaft Internistische Onkologie with an integrated analysis of the ‘burden of therapy’ method. Eur. J. Cancer. 2021;146:95–106. doi: 10.1016/j.ejca.2020.12.029. [DOI] [PubMed] [Google Scholar]
- 12.Cardin D.B., Goff L., Li C.I., Shyr Y., Winkler C., DeVore R., Schlabach L., Holloway M., McClanahan P., Meyer K., et al. Phase II trial of sorafenib and erlotinib in advanced pancreatic cancer. Cancer Med. 2014;3:572–579. doi: 10.1002/cam4.208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ko A.H., Bekaii-Saab T., Van Ziffle J., Mirzoeva O.M., Joseph N.M., Talasaz A., Kuhn P., Tempero M.A., Collisson E.A., Kelley R.K., et al. A Multicenter, Open-Label Phase II Clinical Trial of Combined MEK plus EGFR Inhibition for Chemotherapy-Refractory Advanced Pancreatic Adenocarcinoma. Clin. Cancer Res. 2016;22:61–68. doi: 10.1158/1078-0432.CCR-15-0979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Halfdanarson T.R., Foster N.R., Kim G.P., Meyers J.P., Smyrk T.C., McCullough A.E., Ames M.M., Jaffe J.P., Alberts S.R. A Phase II Randomized Trial of Panitumumab, Erlotinib, and Gemcitabine versus Erlotinib and Gemcitabine in Patients with Untreated, Metastatic Pancreatic Adenocarcinoma: North Central Cancer Treatment Group Trial N064B. Oncologist. 2019;24:589-e160. doi: 10.1634/theoncologist.2018-0878. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Vickers M.M., Powell E.D., Asmis T.R., Jonker D.J., Hilton J.F., O’Callaghan C.J., Tu D., Parulekar W., Moore M.J. Comorbidity, age and overall survival in patients with advanced pancreatic cancer—Results from NCIC CTG PA.3: A phase III trial of gemcitabine plus erlotinib or placebo. Eur. J. Cancer. 2012;48:1434–1442. doi: 10.1016/j.ejca.2011.10.035. [DOI] [PubMed] [Google Scholar]
- 16.Dent P., Booth L., Roberts J.L., Liu J., Poklepovic A., Lalani A.S., Tuveson D., Martinez J., Hancock J.F. Neratinib inhibits Hippo/YAP signaling, reduces mutant K-RAS expression, and kills pancreatic and blood cancer cells. Oncogene. 2019;38:5890–5904. doi: 10.1038/s41388-019-0849-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Gonçalves A., Gilabert M., François E., Dahan L., Perrier H., Lamy R., Re D., Largillier R., Gasmi M., Tchiknavorian X., et al. BAYPAN study: A double-blind phase III randomized trial comparing gemcitabine plus sorafenib and gemcitabine plus placebo in patients with advanced pancreatic cancer. Ann. Oncol. 2012;23:2799–2805. doi: 10.1093/annonc/mds135. [DOI] [PubMed] [Google Scholar]
- 18.Moore A.R., Rosenberg S.C., McCormick F., Malek S. RAS-targeted therapies: Is the undruggable drugged? Nat. Rev. Drug Discov. 2020;19:533–552. doi: 10.1038/s41573-020-0068-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Poulikakos P.I., Zhang C., Bollag G., Shokat K.M., Rosen N. RAF inhibitors transactivate RAF dimers and ERK signalling in cells with wild-type BRAF. Nature. 2010;464:427–430. doi: 10.1038/nature08902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Karoulia Z., Wu Y., Ahmed T.A., Xin Q., Bollard J., Krepler C., Wu X., Zhang C., Bollag G., Herlyn M., et al. An Integrated Model of RAF Inhibitor Action Predicts Inhibitor Activity against Oncogenic BRAF Signaling. Cancer Cell. 2016;30:485–498. doi: 10.1016/j.ccell.2016.06.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Peng S.B., Henry J.R., Kaufman M.D., Lu W.P., Smith B.D., Vogeti S., Rutkoski T.J., Wise S., Chun L., Zhang Y., et al. Inhibition of RAF Isoforms and Active Dimers by LY3009120 Leads to Anti-tumor Activities in RAS or BRAF Mutant Cancers. Cancer Cell. 2015;28:384–398. doi: 10.1016/j.ccell.2015.08.002. [DOI] [PubMed] [Google Scholar]
- 22.Vakana E., Pratt S., Blosser W., Dowless M., Simpson N., Yuan X.J., Jaken S., Manro J., Stephens J., Zhang Y., et al. LY3009120, a panRAF inhibitor, has significant anti-tumor activity in BRAF and KRAS mutant preclinical models of colorectal cancer. Oncotarget. 2017;8:9251–9266. doi: 10.18632/oncotarget.14002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Desai J., Gan H., Barrow C., Jameson M., Atkinson V., Haydon A., Millward M., Begbie S., Brown M., Markman B., et al. Phase I, Open-Label, Dose-Escalation/Dose-Expansion Study of Lifirafenib (BGB-283), an RAF Family Kinase Inhibitor, in Patients with Solid Tumors. J. Clin. Oncol. 2020;38:2140–2150. doi: 10.1200/JCO.19.02654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Singhi A.D., George B., Greenbowe J.R., Chung J., Suh J., Maitra A., Klempner S.J., Hendifar A., Milind J.M., Golan T., et al. Real-Time Targeted Genome Profile Analysis of Pancreatic Ductal Adenocarcinomas Identifies Genetic Alterations That Might Be Targeted with Existing Drugs or Used as Biomarkers. Gastroenterology. 2019;156:2242–2253.e4. doi: 10.1053/j.gastro.2019.02.037. [DOI] [PubMed] [Google Scholar]
- 25.Hyman D.M., Puzanov I., Subbiah V., Faris J.E., Chau I., Blay J.Y., Wolf J., Raje N.S., Diamond E.L., Hollebecque A., et al. Vemurafenib in Multiple Nonmelanoma Cancers with BRAF V600 Mutations. N. Engl. J. Med. 2015;373:726–736. doi: 10.1056/NEJMoa1502309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Aguirre A.J., Nowak J.A., Camarda N.D., Moffitt R.A., Ghazani A.A., Hazar-Rethinam M., Raghavan S., Kim J., Brais L.K., Ragon D., et al. Real-time Genomic Characterization of Advanced Pancreatic Cancer to Enable Precision Medicine. Cancer Discov. 2018;8:1096–1111. doi: 10.1158/2159-8290.CD-18-0275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Chen S.H., Zhang Y., Van Horn R.D., Yin T., Buchanan S., Yadav V., Mochalkin I., Wong S.S., Yue Y.G., Huber L., et al. Oncogenic BRAF Deletions That Function as Homodimers and Are Sensitive to Inhibition by RAF Dimer Inhibitor LY3009120. Cancer Discov. 2016;6:300–315. doi: 10.1158/2159-8290.CD-15-0896. [DOI] [PubMed] [Google Scholar]
- 28.Kawaguchi K., Igarashi K., Murakami T., Kiyuna T., Lwin T.M., Hwang H.K., Delong J.C., Clary B.M., Bouvet M., Unno M., et al. MEK inhibitors cobimetinib and trametinib, regressed a gemcitabine-resistant pancreatic-cancer patient-derived orthotopic xenograft (PDOX) Oncotarget. 2017;8:47490–47496. doi: 10.18632/oncotarget.17667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Seghers A.K., Cuyle P.J., Van Cutsem E. Molecular Targeting of a BRAF Mutation in Pancreatic Ductal Adenocarcinoma: Case Report and Literature Review. Target. Oncol. 2020;15:407–410. doi: 10.1007/s11523-020-00727-9. [DOI] [PubMed] [Google Scholar]
- 30.Li H.S., Yang K., Wang Y. Remarkable response of BRAF (V600E)-mutated metastatic pancreatic cancer to BRAF/MEK inhibition: A case report. Gastroenterol. Rep. 2021;10:goab031. doi: 10.1093/gastro/goab031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Grinshpun A., Zarbiv Y., Roszik J., Subbiah V., Hubert A. Beyond KRAS: Practical molecular targets in pancreatic adenocarcinoma. Case Rep. Oncol. 2019;12:7–13. doi: 10.1159/000496018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Wang Z., He D., Chen C., Liu X., Ke N. Vemurafenib Combined with Trametinib Significantly Benefits the Survival of a Patient with Stage IV Pancreatic Ductal Adenocarcinoma with BRAF V600E Mutation: A Case Report. Front. Oncol. 2022;11:801320. doi: 10.3389/fonc.2021.801320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Shah S., Rana T., Kancharla P., Monga D. Targeted Therapy for BRAF V600E Positive Pancreatic Adenocarcinoma: Two Case Reports. Cancer Genom. Proteom. 2023;20:398–403. doi: 10.21873/cgp.20391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Ardalan B., Azqueta J.I., England J., Eatz T.A. Potential benefit of treatment with MEK inhibitors and chemotherapy in BRAF-mutated KRAS wild-type pancreatic ductal adenocarcinoma patients: A case report. Cold Spring Harb. Mol. Case Stud. 2021;7:a006108. doi: 10.1101/mcs.a006108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Infante J.R., Somer B.G., Park J.O., Li C.P., Scheulen M.E., Kasubhai S.M., Oh D.Y., Liu Y., Redhu S., Steplewski K., et al. A randomised, double-blind, placebo-controlled trial of trametinib, an oral MEK inhibitor, in combination with gemcitabine for patients with untreated metastatic adenocarcinoma of the pancreas. Eur. J. Cancer. 2014;50:2072–2081. doi: 10.1016/j.ejca.2014.04.024. [DOI] [PubMed] [Google Scholar]
- 36.Van Cutsem E., Hidalgo M., Canon J.L., Macarulla T., Bazin I., Poddubskaya E., Manojlovic N., Radenkovic D., Verslype C., Raymond E., et al. Phase I/II trial of pimasertib plus gemcitabine in patients with metastatic pancreatic cancer. Int. J. Cancer. 2018;143:2053–2064. doi: 10.1002/ijc.31603. [DOI] [PubMed] [Google Scholar]
- 37.Van Laethem J.L., Riess H., Jassem J., Haas M., Martens U.M., Weekes C., Peeters M., Ross P., Bridgewater J., Melichar B., et al. Phase I/II Study of Refametinib (BAY 86-9766) in Combination with Gemcitabine in Advanced Pancreatic cancer. Target. Oncol. 2017;12:97–109. doi: 10.1007/s11523-016-0469-y. [DOI] [PubMed] [Google Scholar]
- 38.Bodoky G., Timcheva C., Spigel D.R., La Stella P.J., Ciuleanu T.E., Pover G., Tebbutt N.C. A phase II open-label randomized study to assess the efficacy and safety of selumetinib (AZD6244 [ARRY-142886]) versus capecitabine in patients with advanced or metastatic pancreatic cancer who have failed first-line gemcitabine therapy. Investig. New Drugs. 2012;30:1216–1223. doi: 10.1007/s10637-011-9687-4. [DOI] [PubMed] [Google Scholar]
- 39.Chung V., McDonough S., Philip P.A., Cardin D., Wang-Gillam A., Hui L., Tejani M.A., Seery T.E., Dy I.A., Al Baghdadi T., et al. Effect of Selumetinib and MK-2206 vs Oxaliplatin and Fluorouracil in Patients with Metastatic Pancreatic Cancer after Prior Therapy: SWOG S1115 Study Randomized Clinical Trial. JAMA Oncol. 2017;3:516–522. doi: 10.1001/jamaoncol.2016.5383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Tolcher A.W., Bendell J.C., Papadopoulos K.P., Burris H.A., 3rd, Patnaik A., Jones S.F., Rasco D., Cox D.S., Durante M., Bellew K.M., et al. A phase IB trial of the oral MEK inhibitor trametinib (GSK1120212) in combination with everolimus in patients with advanced solid tumors. Ann. Oncol. 2015;26:58–64. doi: 10.1093/annonc/mdu482. [DOI] [PubMed] [Google Scholar]
- 41.LoRusso P., Fakih M., De Vos F.Y.F.L., Beck J.T., Merchan J., Shapiro G., Lin C.-C., Spratlin J., Cascella T., Sandalic L., et al. Phase Ib study of ribociclib (R) + trametinib (T) in patients (pts) with metastatic/advanced solid tumours. Ann. Oncol. 2020;31:S484. doi: 10.1016/j.annonc.2020.08.675. [DOI] [Google Scholar]
- 42.Rodon Ahnert J., Tan D.S., Garrido-Laguna I., Harb W., Bessudo A., Beck J.T., Rottey S., Bahary N., Kotecki N., Zhu Z., et al. Avelumab or talazoparib in combination with binimetinib in metastatic pancreatic ductal adenocarcinoma: Dose-finding results from phase Ib of the JAVELIN PARP MEKi trial. ESMO Open. 2023;8:101584. doi: 10.1016/j.esmoop.2023.101584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Hobbs G.A., Baker N.M., Miermont A.M., Thurman R.D., Pierobon M., Tran T.H., Anderson A.O., Waters A.M., Diehl J.N., Papke B., et al. Atypical KRASG12R Mutant Is Impaired in PI3K Signaling and Macropinocytosis in Pancreatic Cancer. Cancer Discov. 2020;10:104–123. doi: 10.1158/2159-8290.CD-19-1006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Kenney C., Kunst T., Webb S., Christina D., Jr., Arrowood C., Steinberg S.M., Mettu N.B., Kim E.J., Rudloff U. Phase II study of selumetinib, an orally active inhibitor of MEK1 and MEK2 kinases, in KRASG12R-mutant pancreatic ductal adenocarcinoma. Investig. New Drugs. 2021;39:821–828. doi: 10.1007/s10637-020-01044-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Ardalan B., Azqueta J., Sleeman D. Cobimetinib Plus Gemcitabine: An Active Combination in KRAS G12R-Mutated Pancreatic Ductal Adenocarcinoma Patients in Previously Treated and Failed Multiple Chemotherapies. J. Pancreat. Cancer. 2021;7:65–70. doi: 10.1089/pancan.2021.0006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Lopez C.D., Kardosh A., Chen E.Y., Pegna G.J., Goodyear S., Taber E., Rajagopalan B., Edmerson E., Vo J., Jackson A., et al. Casper: A phase I, open-label, dose finding study of calaspargase pegol-mnkl (cala) in combination with cobimetinib (cobi) in locally advanced or metastatic pancreatic ductal adenocarcinoma (PDAC) J. Clin. Oncol. 2023;41:TPS772. doi: 10.1200/JCO.2023.41.4_suppl.TPS772. [DOI] [Google Scholar]
- 47.Grierson P.M., Tan B., Pedersen K.S., Park H., Suresh R., Amin M.A., Trikalinos N.A., Knoerzer D., Kreider B., Reddy A., et al. Phase Ib Study of Ulixertinib Plus Gemcitabine and Nab-Paclitaxel in Patients with Metastatic Pancreatic Adenocarcinoma. Oncologist. 2023;28:e115–e123. doi: 10.1093/oncolo/oyac237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Wang J., Johnson M., Barve M., Pelster M., Chen X., Li Z., Gordon J., Reiss M., Pai S., Falchook G., et al. Preliminary results from HERKULES-1: A phase 1b/2, open-label, multicenter study of ERAS-007, an oral ERK1/2 inhibitor, in patients with advanced or metastatic solid tumors. Eur. J. Cancer. 2022;174:S80–S81. doi: 10.1016/S0959-8049(22)01013-9. [DOI] [Google Scholar]
- 49.Bryant K.L., Stalnecker C.A., Zeitouni D., Klomp J.E., Peng S., Tikunov A.P., Gunda V., Pierobon M., Waters A.M., George S.D., et al. Combination of ERK and autophagy inhibition as a treatment approach for pancreatic cancer. Nat. Med. 2019;25:628–640. doi: 10.1038/s41591-019-0368-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Franco J., Witkiewicz A.K., Knudsen E.S. CDK4/6 inhibitors have potent activity in combination with pathway selective therapeutic agents in models of pancreatic cancer. Oncotarget. 2014;5:6512–6525. doi: 10.18632/oncotarget.2270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Franco J., Balaji U., Freinkman E., Witkiewicz A.K., Knudsen E.S. Metabolic Reprogramming of Pancreatic Cancer Mediated by CDK4/6 Inhibition Elicits Unique Vulnerabilities. Cell Rep. 2016;14:979–990. doi: 10.1016/j.celrep.2015.12.094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Ruess D.A., Heynen G.J., Ciecielski K.J., Ai J., Berninger A., Kabacaoglu D., Görgülü K., Dantes Z., Wörmann S.M., Diakopoulos K.N., et al. Mutant KRAS-driven cancers depend on PTPN11/SHP2 phosphatase. Nat. Med. 2018;24:954–960. doi: 10.1038/s41591-018-0024-8. [DOI] [PubMed] [Google Scholar]
- 53.Ahmed T.A., Adamopoulos C., Karoulia Z., Wu X., Sachidanandam R., Aaronson S.A., Poulikakos P.I. SHP2 Drives Adaptive Resistance to ERK Signaling Inhibition in Molecularly Defined Subsets of ERK-Dependent Tumors. Cell Rep. 2019;26:65–78.e5. doi: 10.1016/j.celrep.2018.12.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Fedele C., Ran H., Diskin B., Wei W., Jen J., Geer M.J., Araki K., Ozerdem U., Simeone D.M., Miller G., et al. SHP2 Inhibition Prevents Adaptive Resistance to MEK Inhibitors in Multiple Cancer Models. Cancer Discov. 2018;8:1237–1249. doi: 10.1158/2159-8290.CD-18-0444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Mainardi S., Mulero-Sánchez A., Prahallad A., Germano G., Bosma A., Krimpenfort P., Lieftink C., Steinberg J.D., de Wit N., Gonçalves-Ribeiro S., et al. SHP2 is required for growth of KRAS-mutant non-small-cell lung cancer in vivo. Nat. Med. 2018;24:961–967. doi: 10.1038/s41591-018-0023-9. [DOI] [PubMed] [Google Scholar]
- 56.Frank K.J., Mulero-Sánchez A., Berninger A., Ruiz-Cañas L., Bosma A., Görgülü K., Wu N., Diakopoulos K.N., Kaya-Aksoy E., Ruess D.A., et al. Extensive preclinical validation of combined RMC-4550 and LY3214996 supports clinical investigation for KRAS mutant pancreatic cancer. Cell Rep. Med. 2022;3:100815. doi: 10.1016/j.xcrm.2022.100815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Ryan M.B., Fece de la Cruz F., Phat S., Myers D.T., Wong E., Shahzade H.A., Hong C.B., Corcoran R.B. Vertical Pathway Inhibition Overcomes Adaptive Feedback Resistance to KRASG12C Inhibition. Clin. Cancer Res. 2020;26:1633–1643. doi: 10.1158/1078-0432.CCR-19-3523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Fedele C., Li S., Teng K.W., Foster C.J.R., Peng D., Ran H., Mita P., Geer M.J., Hattori T., Koide A., et al. SHP2 inhibition diminishes KRASG12C cycling and promotes tumor microenvironment remodeling. J. Exp. Med. 2021;218:e20201414. doi: 10.1084/jem.20201414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Canon J., Rex K., Saiki A.Y., Mohr C., Cooke K., Bagal D., Gaida K., Holt T., Knutson C.G., Koppada N., et al. The clinical KRAS(G12C) inhibitor AMG 510 drives anti-tumour immunity. Nature. 2019;575:217–223. doi: 10.1038/s41586-019-1694-1. [DOI] [PubMed] [Google Scholar]
- 60.Jeng H.H., Taylor L.J., Bar-Sagi D. Sos-mediated cross-activation of wild-type Ras by oncogenic Ras is essential for tumorigenesis. Nat. Commun. 2012;3:1168. doi: 10.1038/ncomms2173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Ma Y., Schulz B., Trakooljul N., Al Ammar M., Sekora A., Sender S., Hadlich F., Zechner D., Weiss F.U., Lerch M.M., et al. Inhibition of KRAS, MEK and PI3K Demonstrate Synergistic Anti-Tumor Effects in Pancreatic Ductal Adenocarcinoma Cell Lines. Cancers. 2022;14:4467. doi: 10.3390/cancers14184467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Hofmann M.H., Gmachl M., Ramharter J., Savarese F., Gerlach D., Marszalek J.R., Sanderson M.P., Kessler D., Trapani F., Arnhof H., et al. BI-3406, a Potent and Selective SOS1-KRAS Interaction Inhibitor, Is Effective in KRAS-Driven Cancers through Combined MEK Inhibition. Cancer Discov. 2021;11:142–157. doi: 10.1158/2159-8290.CD-20-0142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Johnson M., Gort E., Pant S., Lolkema M., Sebastian M., Scheffler M., Hwang J., Dünzinger U., Riemann K., Kitzing T., et al. 524P A phase I, open-label, dose-escalation trial of BI 1701963 in patients (pts) with KRAS mutated solid tumours: A snapshot analysis. Ann. Oncol. 2021;32:S591–S592. doi: 10.1016/j.annonc.2021.08.1046. [DOI] [Google Scholar]
- 64.Janes M.R., Zhang J., Li L.S., Hansen R., Peters U., Guo X., Chen Y., Babbar A., Firdaus S.J., Darjania L., et al. Targeting KRAS Mutant Cancers with a Covalent G12C-Specific Inhibitor. Cell. 2018;172:578–589.e17. doi: 10.1016/j.cell.2018.01.006. [DOI] [PubMed] [Google Scholar]
- 65.Jänne P.A., Riely G.J., Gadgeel S.M., Heist R.S., Ou S.I., Pacheco J.M., Johnson M.L., Sabari J.K., Leventakos K., Yau E., et al. Adagrasib in Non-Small-Cell Lung Cancer Harboring a KRASG12C Mutation. N. Engl. J. Med. 2022;387:120–131. doi: 10.1056/NEJMoa2204619. [DOI] [PubMed] [Google Scholar]
- 66.Murciano-Goroff Y.R., Suehnholz S.P., Drilon A., Chakravarty D. Precision Oncology: 2023 in Review. Cancer Discov. 2023;13:2525–2531. doi: 10.1158/2159-8290.CD-23-1194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Sacher A., LoRusso P., Patel M.R., Miller W.H., Jr., Garralda E., Forster M.D., Santoro A., Falcon A., Kim T.W., Paz-Ares L., et al. Single-Agent Divarasib (GDC-6036) in Solid Tumors with a KRAS G12C Mutation. N. Engl. J. Med. 2023;389:710–721. doi: 10.1056/NEJMoa2303810. [DOI] [PubMed] [Google Scholar]
- 68.Wang X., Allen S., Blake J.F., Bowcut V., Briere D.M., Calinisan A., Dahlke J.R., Fell J.B., Fischer J.P., Gunn R.J., et al. Identification of MRTX1133, a Noncovalent, Potent, and Selective KRASG12D Inhibitor. J. Med. Chem. 2022;65:3123–3133. doi: 10.1021/acs.jmedchem.1c01688. [DOI] [PubMed] [Google Scholar]
- 69.Kim D., Herdeis L., Rudolph D., Zhao Y., Böttcher J., Vides A., Ayala-Santos C.I., Pourfarjam Y., Cuevas-Navarro A., Xue J.Y., et al. Pan-KRAS inhibitor disables oncogenic signalling and tumour growth. Nature. 2023;619:160–166. doi: 10.1038/s41586-023-06123-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Foote J.B., Mattox T.E., Keeton A.B., Purnachandra G.N., Maxuitenko Y., Chen X., Valiyaveettil J., Buchsbaum D.J., Piazza G.A., El-Rayes B.F. Abstract 4140: Oncogenic KRAS inhibition with ADT-007 primes T cell responses in pancreatic ductal adenocarcinoma. Cancer Res. 2023;83((Suppl. 7)):4140. doi: 10.1158/1538-7445.AM2023-4140. [DOI] [Google Scholar]
- 71.Koltun E., Cregg J., Rice M.A., Whalen D.M., Freilich R., Jiang J., Hansen R., Bermingham A., Knox D., Dinglasan J., et al. Abstract 1260: First-in-class, orally bioavailable KRASG12V(ON) tri-complex inhibitors, as single agents and in combinations, drive profound anti-tumor activity in preclinical models of KRASG12V mutant cancers. Cancer Res. 2021;13:1260. doi: 10.1158/1538-7445.AM2021-1260. [DOI] [Google Scholar]
- 72.de Jesus V.H.F., Mathias-Machado M.C., de Farias J.P.F., Aruquipa M.P.S., Jácome A.A., Peixoto R.D. Targeting KRAS in Pancreatic Ductal Adenocarcinoma: The Long Road to Cure. Cancers. 2023;15:5015. doi: 10.3390/cancers15205015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Koltun E.S., Rice M.A., Gustafson W.C., Wilds D., Jiang J., Lee B.J., Wang Z., Chang S., Flagella M., Mu Y., et al. Direct targeting of KRASG12X mutant cancers with RMC-6236, a first-in-class, RAS-selective, orally bioavailable, tri-complex RASMULTI(ON) inhibitor. Cancer Res. 2022;82((Suppl. S12)):3597. doi: 10.1158/1538-7445.AM2022-3597. [DOI] [Google Scholar]
- 74.Park S.R., Davis M., Doroshow J.H., Kummar S. Safety and feasibility of targeted agent combinations in solid tumours. Nat. Rev. Clin. Oncol. 2013;10:154–168. doi: 10.1038/nrclinonc.2012.245. [DOI] [PubMed] [Google Scholar]
- 75.Liu C., Lu H., Wang H., Loo A., Zhang X., Yang G., Kowal C., Delach S., Wang Y., Goldoni S., et al. Combinations with Allosteric SHP2 Inhibitor TNO155 to Block Receptor Tyrosine Kinase Signaling. Clin. Cancer Res. 2021;27:342–354. doi: 10.1158/1078-0432.CCR-20-2718. [DOI] [PubMed] [Google Scholar]
- 76.Akhave N.S., Biter A.B., Hong D.S. The Next Generation of KRAS Targeting: Reasons for Excitement and Concern. Mol. Cancer Ther. 2022;21:1645–1651. doi: 10.1158/1535-7163.MCT-22-0356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Li B.T., Falchook G.S., Durm G.A., Burns T.F., Skoulidis F., Ramalingam S.S., Spira A., Bestvina C.M., Goldberg S.B., Veluswamy R., et al. CodeBreaK 100/101: First report of safety/efficacy of sotorasib in combination with pembrolizumab or atezolizumab in advanced KRAS p.G12C NSCLC. J. Thorac. Oncol. 2022;17:S10–S11. doi: 10.1016/j.jtho.2022.07.025. [DOI] [Google Scholar]
- 78.Sacher A., Patel M.R., Miller W.H., Jr., Desai J., Garralda E., Bowyer S., Kim T.W., De Miguel M., Falcon A., Krebs M.G., et al. OA03.04 phase I A study to evaluate GDC-6036 monotherapy in patients with Non-small Cell Lung Cancer (NSCLC) with KRAS G12C mutation. J. Thorac. Oncol. 2022;17:S8–S9. doi: 10.1016/j.jtho.2022.07.023. [DOI] [Google Scholar]
- 79.Tanaka N., Lin J.J., Li C., Ryan M.B., Zhang J., Kiedrowski L.A., Michel A.G., Syed M.U., Fella K.A., Sakhi M., et al. Clinical Acquired Resistance to KRASG12C Inhibition through a Novel KRAS Switch-II Pocket Mutation and Polyclonal Alterations Converging on RAS-MAPK Reactivation. Cancer Discov. 2021;11:1913–1922. doi: 10.1158/2159-8290.CD-21-0365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Zhang J., Darman L., Hassan M.S., Von Holzen U., Awasthi N. Targeting KRAS for the potential treatment of pancreatic ductal adenocarcinoma: Recent advancements provide hope (Review) Oncol. Rep. 2023;50:206. doi: 10.3892/or.2023.8643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Philip P.A., Azar I., Xiu J., Hall M.J., Hendifar A.E., Lou E., Hwang J.J., Gong J., Feldman R., Ellis M., et al. Molecular Characterization of KRAS Wild-type Tumors in Patients with Pancreatic Adenocarcinoma. Clin. Cancer Res. 2022;28:2704–2714. doi: 10.1158/1078-0432.CCR-21-3581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Ardalan B., Ciner A., Baca Y., Darabi S., Kasi A., Lou E., Azqueta J.I., Xiu J., Nabhan C., Shields A.F., et al. Not all treated KRAS-mutant pancreatic adenocarcinomas are equal: KRAS G12D and survival outcome. J. Clin. Oncol. 2023;41:4020. doi: 10.1200/JCO.2023.41.16_suppl.4020. [DOI] [Google Scholar]
- 83.Ardalan B., Ciner A., Baca Y., Darabi S., Kasi A., Lou E., Azqueta J.I., Xiu J., Nabhan C., Shields A.F., et al. Prognostic indicators of KRAS G12X mutations in pancreatic cancer. J. Clin. Oncol. 2023;41:735. doi: 10.1200/JCO.2023.41.4_suppl.735. [DOI] [Google Scholar]
- 84.Ciner A., Ardalan B., Baca Y., Darabi S., Kasi A., Lou E., Azqueta J.I., Xiu J., Nabhan C., Shields A.F., et al. KRAS G12C-mutated pancreatic cancer: Clinical outcomes based on chemotherapeutic regimen. J. Clin. Oncol. 2023;41:4150. doi: 10.1200/JCO.2023.41.16_suppl.4150. [DOI] [Google Scholar]
- 85.Suzuki T., Masugi Y., Inoue Y., Hamada T., Tanaka M., Takamatsu M., Arita J., Kato T., Kawaguchi Y., Kunita A., et al. KRAS variant allele frequency, but not mutation positivity, associates with survival of patients with pancreatic cancer. Cancer Sci. 2022;113:3097–3109. doi: 10.1111/cas.15398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Mueller S., Engleitner T., Maresch R., Zukowska M., Lange S., Kaltenbacher T., Konukiewitz B., Öllinger R., Zwiebel M., Strong A., et al. Evolutionary routes and KRAS dosage define pancreatic cancer phenotypes. Nature. 2018;554:62–68. doi: 10.1038/nature25459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Adamopoulos C., Ahmed T.A., Tucker M.R., Ung P.M.U., Xiao M., Karoulia Z., Amabile A., Wu X., Aaronson S.A., Ang C., et al. Exploiting Allosteric Properties of RAF and MEK Inhibitors to Target Therapy-Resistant Tumors Driven by Oncogenic BRAF Signaling. Cancer Discov. 2021;11:1716–1735. doi: 10.1158/2159-8290.CD-20-1351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Kemp S.B., Cheng N., Markosyan N., Sor R., Kim I.K., Hallin J., Shoush J., Quinones L., Brown N.V., Bassett J.B., et al. Efficacy of a Small-Molecule Inhibitor of KrasG12D in Immunocompetent Models of Pancreatic Cancer. Cancer Discov. 2023;13:298–311. doi: 10.1158/2159-8290.CD-22-1066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Mullard A. The KRAS crowd targets its next cancer mutations. Nat. Rev. Drug Discov. 2023;22:167–171. doi: 10.1038/d41573-023-00015-x. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Data are contained within the article.