Abstract
Microscopic colitis, a diagnosis under the umbrella term of inflammatory bowel disease, is a prevalent cause of watery diarrhea, often with symptoms of urgency and bloating, typically observed in older adults aged ≥ 60 years. Its incidence has been reported to exceed those of ulcerative colitis and Crohn’s disease in some geographical areas. Although nonpathognomonic endoscopic abnormalities, including changes of the vascular mucosal pattern; mucosal erythema; edema; nodularity; or mucosal defects, e.g., “cat scratches” have been reported, a colonoscopy is typically macroscopically normal. As reliable biomarkers are unavailable, colonoscopy using random biopsies from various parts of the colon is compulsory. Based on the histological examination under a microscope, the disease is divided into collagenous (with a thickened subepithelial collagenous band) and lymphocytic (with intraepithelial lymphocytosis) colitis, although incomplete forms exist. In routine clinical settings, the disease has a high risk of being misdiagnosed as irritable bowel syndrome or even overlooked. Therefore, healthcare providers should be familiar with clinical features and rational management strategies. A 6–8-week oral budesonide treatment course (9 mg/day) is considered the first-line therapy, but patients often experience relapse when discontinued, or might become intolerant, dependent, or even fail to respond. Consequently, other therapeutic options (e.g., bismuth subsalicylate, biologics, loperamide, bile acid sequestrants, and thiopurines) recommended by available guidelines may be prescribed. Herein, clinically meaningful data is provided based on the latest evidence that may aid in reaching a diagnosis and establishing rational therapy in geriatric care to control symptoms and enhance the quality of life for those affected.
Key Points
| Microscopic colitis is a prevalent condition among older adults above the age of 60 years. |
| Patients suspected with long-standing or recurrent non-bloody watery diarrhea should always be referred for colonoscopy with three biopsies from right and left colon followed by extensive histopathological evaluations. |
| Oral budesonide is the drug of choice for microscopic colitis, but nonsteroidal medications, including bismuth subsalicylate, biologics, bile acid sequestrants, and supportive antidiarrheal drugs followed by thiopurines, may be useful alternatives. |
Introduction
Microscopic colitis, a chronic disorder of unknown etiology with a female preponderance, is a common but treatable cause of recurrent non-bloody watery diarrhea. The diarrhea, together with symptoms such as abdominal cramps, urgency, and fecal incontinence, can have a significant negative impact on quality of life and social function. The disease has been reported to have an increasing incidence, and its incidence may, in some geographical areas, exceed those of ulcerative colitis and Crohn’s disease among elderly individuals [1]. Microscopic colitis is associated with ulcerative colitis and Crohn’s disease in the umbrella diagnosis of inflammatory bowel disease (IBD) [1]. Microscopic colitis is named so because specific histological changes are identified under a microscope, whereas an endoscopic examination of the colon is usually macroscopically normal, although endoscopic findings such as ulceration of vascular mucosal pattern, a “furrowed” or “mosaic” pattern, mucosal erythema, edema, nodularity, or mucosal defects, including “cat scratches,” have been reported [2–4], which are, however, not pathognomonic. There are two major histological subtypes of microscopic colitis: collagenous and lymphocytic colitis, although incomplete forms exist [5]. The distinction between subtypes does not influence management decisions or prognosis. Due to the lack of reliable biomarkers, colonoscopy with random biopsies is always required to reach a diagnosis. Consequently, healthcare providers should be familiar with the clinical features and the most rational management of the older adults.
For the management of microscopic colitis, a treatment course with oral budesonide 9 mg/day for 6–8 weeks is considered first-line therapy [6–8]. However, patients often experience relapse when budesonide is withdrawn, and some patients may become intolerant, dependent, or even fail to respond. Accordingly, alternative therapeutics, including bismuth subsalicylate, biologics, bile acid sequestrants, and thiopurines, may be indicated to control the symptoms and enhance the quality of life of patients affected by this debilitating chronic disease, as recommended by international guidelines [9–11]. Loperamide is not included in all recommendations, but this drug is widely used for management of microscopic colitis, alone or in combination with other therapies, even though no formal, well-powered trials have been performed. Regarding the prognosis, recent data from a 5 year prospective European cohort of patients with microscopic colitis has shown that 40% had a relapsing or chronic active disease course, and that disease activity after 1 year seems to be predictive of the subsequent disease course [12].
The aim of this review is to provide the reader updated information about the diagnosis of microscopic colitis and rational management, with alternative treatment options for elderly patients not responding sufficiently to first-choice budesonide therapy.
Epidemiology
The incidence and prevalence of microscopic colitis have increased over time, emphasizing the importance of recognizing and diagnosing it in the clinic [13]. In Denmark, the overall incidence of microscopic colitis increased from 2.3 cases in 2001 to 24.3 per 100,000 patient years in 2016 (with a mean age at diagnosis of 65 years) [14], and similarly, in Switzerland, the incidence rose from 0.36 per 100,000 patient years in 1994–1997 to 6.85 in 2017, with a mean age of 63 years at diagnosis [15]. Comparable trends have been observed in the USA [16], although with subsequent stabilization of incidence [17]. In this context, it should be noted that epidemiological studies have proven that the incidence of microscopic colitis exceeds those of ulcerative colitis and Crohn’s disease in individuals aged ≥ 60 years. Thus, using nationwide health registries in Denmark, the above-mentioned incidence of microscopic colitis of 24.3 per 100,000 patient years in 2016 should be compared with 18.6 for ulcerative colitis and 9.1 Crohn’s disease per 100,000 patient years in 2013 [14, 18].
Various factors, including improved awareness of this condition among gastroenterologists and pathologists, as well as varying occurrence of risk factors (e.g., cigarette smoking, medications, intestinal infections, female sex, and concomitant diseases) at various geographical locations [1, 19, 20], may influence differences in its incidence, as a substantial difference among geographical regions has been observed [1]. Based on a recent meta-analysis, predominantly from data originating from North America and Europe, pooled worldwide incidences of the subtypes collagenous and lymphocytic colitis were 4.9 [95% confidence interval (CI) 4.2–5.7] and 5.0 [95% CI 4.0–6.1] cases, respectively, per 100,000 patient years [9].
Symptoms
The symptoms of microscopic colitis are nonspecific and may mimic those of irritable bowel syndrome [21–23], and as histology is required, the diagnosis might easily be overlooked in clinical settings [24]. The disease is characterized by a clinicopathological triad of (1) a history of chronic (i.e., for more than 4 weeks) or recurrent/intermittent non-bloody watery diarrhea, (2) a normal or almost normal colonoscopy, and (3) a distinct histological pattern in colonic biopsy specimens when examined under a microscope [1]. Apart from non-bloody watery diarrhea, which is often a key symptom in the early hours, other symptoms include urgency, fecal incontinence, fatigue, abdominal cramps or bloating, weight loss, arthralgia related to disease flares, and impaired quality of life. However, increased incidences of bile salt diarrhea [25] and celiac disease [26, 27] are associated with microscopic colitis [21]. Therefore, celiac disease should be excluded in all individuals suspected of microscopic colitis. Moreover, bile acid induced diarrhea should always be considered in those not responding properly to budesonide with continued diarrhea.
The pathogenesis of microscopic colitis is complex and multifactorial, and involves innate and adaptive immune responses to luminal antigens, genetic risk factors, and autoimmunity, as well as extracellular matrix alterations, all of which contribute by varied mechanisms to watery diarrhea [28, 29]. Moreover, similar symptoms and histologic findings may develop following therapy with checkpoint inhibitors, a therapeutic option that is increasingly used for a broad variety of oncological and hematological diseases also in older adults [30–32].
Diagnosis
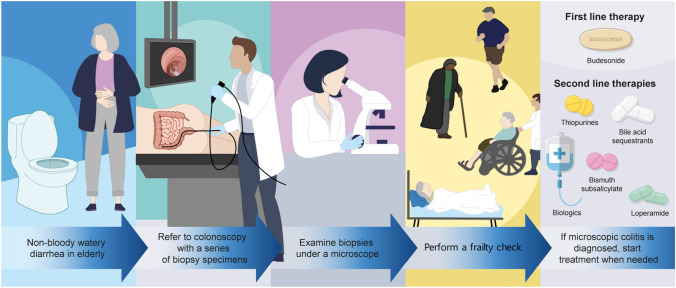
The diagnosis of microscopic colitis is based on a combination of the clinical presentation and histological changes. Therefore, all patients with chronic or recurrent non-bloody watery diarrhea should undergo colonoscopy with biopsies (Fig. 1). Thus, histological examinations are key for reaching a diagnosis, as the endoscopic findings may be normal, unlike the characteristics for flares of ulcerative colitis or Crohn’s disease.
Fig. 1.
Graphical flow chart indicating the approach to diagnosis and management of microscopic colitis
Controversy exists regarding where and how many biopsy specimens should be obtained at the time of colonoscopy. According to the American Society for Gastrointestinal Endoscopy (ASGE), two or more biopsies are suggested from each of the following locations: the right, transverse, descending, and sigmoid colons during colonoscopy; or two or more biopsies should be obtained from the transverse, sigmoid, and descending colons if flexible sigmoidoscopy is performed [33]. However, the various available guidelines are not evidence based, rely on expert opinions, and have not been prospectively validated. A recent systematic review of the optimum number of colon biopsies for diagnosing microscopic colitis recommended a minimum of six biopsies to be taken from the ascending and descending colons (three from each part) [34].Thus, a combination of clinical symptoms (watery diarrhea, urgency, and often bloating) and histological findings are needed to reach the diagnosis of microscopic colitis.
Pathology
Histological examination distinguishes collagenous and lymphocytic colitis. The collagenous form was originally described by Lindström et al. in 1976 [35], and the term microscopic colitis was introduced by Read et al. in 1980 [36], and in 1989, Lazenby et al. [37] introduced the term lymphocytic colitis. Although histologic findings are not pathognomonic or verified, they do help the pathologist reach the histological diagnosis when paired with the appropriate clinical context.
Nonetheless, in addition to the widely accepted histological criteria, an additional subtype with less prominent histological findings on hematoxylin–eosin (HE) stained specimens, known as incomplete microscopic colitis or microscopic colitis not otherwise specified, exists [38], and some patients with this condition may respond to treatment used for microscopic colitis [39] (Table 1). Due to the fact that incomplete microscopic colitis or microscopic colitis not otherwise specified exists, in clinical settings, a lower threshold should be considered in relation to the definition of the distinctive findings on a colonic biopsy in collagenous colitis, which is defined as a thickened subepithelial collagen band (> 10 µm, compared with normal ≤ 5 µm) [40], although a less prominent intraepithelial lymphocyte (IEL) infiltrate may be observed [1] (Table 1). In lymphocytic colitis, more pronounced IEL, defined as more than 20 IEL per 100 surface epithelial cells, combined with an increased inflammatory infiltrate in the lamina propria and a nonsignificantly thickened collagenous band, is a distinct feature [1] (Table 1). Additionally, a mixed infiltrate of acute and chronic inflammatory cells appears in the lamina propria during lymphocytic colitis. In collagenous colitis, inflammation of the lamina propria may be less prominent, but surface epithelial damage tends to be more pronounced than that in lymphocytic colitis. Biopsies frequently contain a neutrophilic infiltrate, even with active cryptitis [41], but acute inflammation should not predominate [1].
Table 1.
Key histological findings in microscopic colitis: Differences between collagenous, lymphocytic, and incomplete microscopic colitis are shown
| Type of disease | Subepithelial collagenous band | Intraepithelial lymphocytes | Surface epithelium |
|---|---|---|---|
| Collagenous | Thickening (≥ 10 μm) | Normal or increased number | Marked change (flattening and detachment) |
| Lymphocytic | Normal or slightly increased (< 7 μm) | 20 per 100 epithelial cells and an increased inflammatory infiltrate in the lamina propria | Slight change (vacuolization, flattening, and mucin depletion) |
| Incomplete | Thickening (> 5 μm) | 10 per 100 epithelial cells | Slight change with patchy epithelial lymphocytosis |
However, a correct diagnosis is crucial for the appropriate treatment of microscopic colitis and for differentiating this condition from other conditions with similar symptoms [25, 42].
Medications that Trigger Microscopic Colitis
Although the etiology of microscopic colitis has not yet been clarified, autoimmune diseases and medications may trigger an imbalanced mucosal immune response in the colon of genetically predisposed individuals, which may aid in the emergence of microscopic colitis [1, 29]. Thus, microscopic mucosal inflammation is likely to be the cause of the diarrhea that accompanies microscopic colitis [1].
Examples of culprit medications linked to microscopic colitis are as follows: nonsteroidal anti-inflammatory drugs (NSAIDs) [43, 44] including ibuprofen and diclofenac often used for a wide range of musculoskeletal conditions in elderly people [45]; proton-pump inhibitors [43, 44] such as lansoprazole and omeprazole, drugs reported to be used in more than 50% of individuals above 65 years [46]; histamine-2 receptor antagonists [47] such as ranitidine and famotidine; selective serotonin-reuptake inhibitors [43, 44] such as sertraline and citalopram; clozapine (an antagonist to dopamine type 2 and the serotonin type 2A receptors) [48]; acetylic salicylic acid [49] such as aspirin; statins [50] such as simvastatin and atorvastatin; and immune checkpoint inhibitors [1, 31] such as pembrolizumab. Therefore, healthcare providers should always review the medications of patients with symptomatic microscopic colitis to induce remission through optimized therapy. Nevertheless, in studies using patients with diarrhea as a control group, much less evidence of drug-induced microscopic colitis was found compared with those that used healthy controls [51, 52]. This raises the possibility that these medications may worsen diarrhea and bring cases to clinical attention, rather than actually causing microscopic colitis.
Prognosis
Although the symptoms can significantly impact quality of life, the prognosis is good, and no increased risk of colon cancer has been reported, in contrast to what has been found with other diseases under IBD [53], such as ulcerative colitis [54] and Crohn’s disease [55]. On the contrary, patients with microscopic colitis seem to have a reduced risk of colorectal cancer [56] or colonic adenoma [57] and, based on all available data, chromo-endoscopic surveillance in individuals diagnosed with microscopic colitis is not required [58]. However, a recent Swedish nationwide population-based matched cohort study revealed a 27% increased risk of major adverse cardiovascular events in microscopic colitis, which underlines the need for additional pathophysiological studies, with a focus on the mechanisms behind such complications in microscopic colitis [59]. In the interim, medical professionals should offer guidance and treatments targeted at lowering the risk of cardiovascular disease in the elderly population with this disease.
Clinical Studies Investigating Therapy for Microscopic Colitis
A general problem exists when assessing clinical data from various trials on the management of microscopic colitis. In contrast to clinical trials addressing the effectiveness of therapies for the two classical IBDs, i.e., ulcerative colitis and Crohn’s disease, where well-established and validated clinical indices [e.g., Harvey Bradshaw [60] and Crohn’s Disease Activity Index (CDAI) [61] for Crohn’s disease, and the Mayo score [62] or Simple Clinical Colitis Activity Index [63] for ulcerative colitis] are widely adapted, variable and not well-defined activity scores for treatment responses (e.g., stool frequency complemented by stool weight in some studies, or time to a histological response in some other studies) are used in clinical trials for microscopic colitis. Thus, validated instruments for assessing response and remission are seldom used in microscopic colitis trials. As a result, it is difficult to make firm conclusions about the comparative effectiveness of various treatment options in microscopic colitis due to the heterogeneity in definitions of treatment response.
Chande et al. [64] has previously paid attention to the need for more accurate and uniform outcome measures in clinical trials studying the effectiveness of various therapies for microscopic colitis in an effort to define the disease activity of this disease, and thus, reliable assessments of clinical response and remission. In this context, Hjortswang et al. [65] proposed clinical criteria, including bowel movements and stool consistency, where clinical remission in collagenous colitis was defined as a mean of less than three stools per day (and with less than one watery stool per day) within the previous 7 days. In a recent systematic review of 25 clinical studies dealing with microscopic colitis therapy, this definition was identified in only four of the trials [66]. This may cause marked variance when assessing and comparing data from different clinical trials of therapeutics for this condition, and head-to-head trials between different drugs are scarce. Moreover, the Hjortswang criteria were solely derived for collagenous colitis and not for lymphocytic colitis [65]. Nonetheless, patients with microscopic colitis often complain of other troublesome symptoms, including abdominal discomfort, urgency, and fecal incontinence, which impair their health-related quality of life. These other symptoms are still unaccounted for using these criteria. As no standards have been developed to define disease activity in microscopic colitis, this matter may preclude the evaluation of responsiveness to the therapies investigated, as well as any risk stratification of patients in clinical studies.
Based on the limitations described, Cotter et al. [67] aimed to identify clinical features of microscopic colitis associated with disease severity in elderly patients (median age in this study of 66 years) in an effort to create a Microscopic Colitis Disease Activity Index (MCDAI) comparable to the CDAI scores utilized to assess disease activity in clinical trials [61]. The amount of daily unformed stools, nocturnal stools, stomach pain, fecal urgency and incontinence, and weight loss are all included in the MCDAI scores [67], which is still awaiting external validation.
Therapy for Microscopic Colitis
First-Line Therapy with Budesonide
No therapeutic differences exist in the management of collagenous and lymphocytic colitis, and based on evidence from several clinical trials, budesonide is considered the drug of choice for the management of microscopic colitis [6–8], including its incomplete form [39]. Budesonide is a second-generation glucocorticoid with low systemic bioavailability due to a 80–90% first-pass hepatic metabolism [61] and with less dysfunction of the hypothalamic–pituitary–adrenal axis compared with therapeutically equivalent doses of oral prednisone [68]. Compared with other corticosteroids, budesonide has a markedly reduced incidence of systemic adverse events, with the most frequent adverse effect reported being cushingoid features and hypokalemia [69]. Moreover, adrenal insufficiency has never been reported when budesonide is discontinued without tapering after a 6–8 week induction treatment course of 9 mg daily.
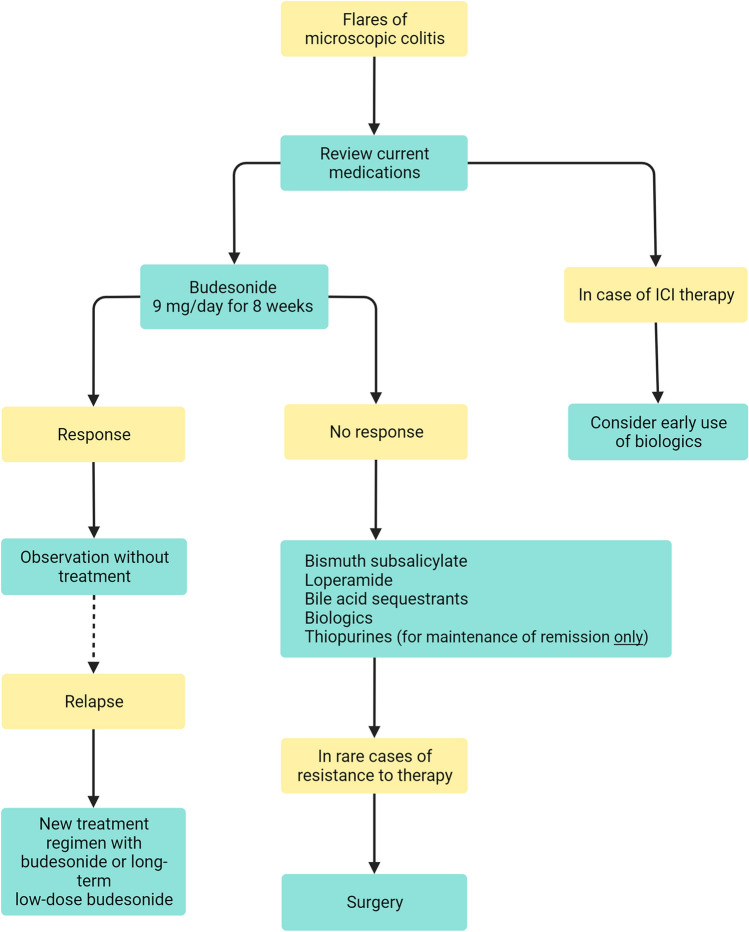
Although the vast majority of patients in a population-based cohort respond to budesonide induction therapy [70], some patients may only experience a partial response, others may have no response, and some patients may even be intolerant (Fig. 2). As recurrence shortly after cessation of a treatment course is a frequent phenomenon [8, 71–73], budesonide may be prescribed as maintenance therapy at the lowest possible dose, for example 3 mg/day or every second day, together with calcium and vitamin D supplementation [1]. In a recent systematic review and meta-analysis of 35 studies (i.e., 11 randomized controlled trails and 24 observational studies) with 1657 patients diagnosed with microscopic colitis treated with budesonide induction therapy, the overall pooled clinical remission rate on budesonide maintenance therapy for a median duration of 39 weeks (range, 24–62 weeks) was 84% [73]. In this study, the pooled recurrence rate after the discontinuation of budesonide induction therapy was 50%, with a median time to recurrence that ranged from 2 to 61 weeks, whereas the pooled recurrence rate after discontinuation of maintenance therapy was 58%, with a median time to recurrence of 6–48 weeks [73]. In another recent population-based study, 162 patients with microscopic colitis received budesonide induction therapy, of whom 96 had recurrence after discontinuation. Of these patients, 35% continued with long-term budesonide maintenance with a median duration of follow-up of 5.6 years (0.3–18.9 years) [74]. However, the incidence of metabolic bone disease (i.e., osteopenia/osteoporosis), diabetes mellitus, hypertension, glaucoma, and cataracts, which are all predisposed comorbid conditions in elderly individuals, did not increase among those on long-term budesonide maintenance [46% (95% CI, 0.35–0.56) I2 = 0%] versus those who received induction therapy or placebo [33% (95% CI, 0.23–0.44) I2 = 79%], and none stopped long-term maintenance therapy due to adverse events [73, 74]. Nevertheless, larger, more robust prospective trials are required to fully assess the effectiveness and safety of long-term maintenance budesonide for microscopic colitis.
Fig. 2.
Proposed therapeutic algorithm to control microscopic colitis in older adults. ICI, immune checkpoint inhibitors
Alternatives to Budesonide in the Management of Microscopic Colitis
Alternatives to budesonide with antidiarrheal or immunomodulatory drugs (including biologics), such as bismuth subsalicylate, biologics, loperamide, bile acid sequestrants, and thiopurines, may be trialed for patients with troublesome microscopic colitis (those who are intolerant or fail to respond to budesonide and those who develop marked adverse events to this therapy), or as maintenance therapy to avoid any side effects of glucocorticoids. In these circumstances a frailty check is of importance in geriatric practice when considering the various therapeutic options based on side effects in older patients (Fig. 1). In this context, prednisolone has been found to be inferior to budesonide [1, 75]; methotrexate showed effectiveness in only one study where 75% received combined budesonide, thus limiting the conclusions [76]; and mesalazine, a cornerstone in therapy of ulcerative colitis, was not been found to be superior to placebo [77, 78]. Moreover, no formal and well-powered data exist for probiotics or newer small molecules, such as Janus Kinase inhibitors, for the management of microscopic colitis.
Based on a recent systematic review and meta-analysis conducted until April 2023, alternatives to budesonide are comparable to those reported in clinical trials [66] (Table 2), as explained below.
Table 2.
Response rates of alternatives to budesonide for management of microscopic colitis
| Drug | Response rate (%) | 95% CI |
|---|---|---|
| Bismuth subsalicylate | 75 | 0.65–0.83 |
| TNF inhibitors | 73 | 0.63–0.83 |
| Vedolizumab | 73 | 0.57–0.87 |
| Loperamide | 62 | 0.43–0.80 |
| Bile acid sequestrants | 60 | 0.51–0.68 |
| Thiopurines | 49 | 0.27–0.71 |
Bismuth Subsalicylate
Based on results from clinical studies, bismuth subsalicylate, a substance with antisecretory and anti-inflammatory properties, has been shown to reduce diarrhea in various diseases, including microscopic colitis [79], although its antidiarrheal mechanism is still not clarified [80]. The recommended dose of bismuth subsalicylate for microscopic colitis is nine tablets [262 mg each] per day in three divided doses. Based on a meta-analysis of seven studies with 377 participants, 75% of patients showed a response to treatment with bismuth subsalicylate (95% CI, 0.65–0.83; I2 = 70.12%), and 50% achieved remission (95% CI, 0.35–0.65; I2 = 71.06%) [66]. However, robust, prospective, placebo-controlled studies stratified by disease severity and subtype are needed to ascertain the genuine advantages of bismuth subsalicylate. That said, prolonged usage of bismuth subsalicylate is not recommended as it may increase the risk of neurotoxicity and accelerate weakness in older patients [81, 82].
Biologics
In recent years, tumor necrosis factor (TNF) inhibitors (infliximab and adalimumab), together with the α4β7 integrin inhibitor, vedolizumab, and more recently the anti-IL-12/IL-23 antibody, ustekinumab, have all been investigated in the management of microscopic colitis. A recent meta-analysis identified 22 studies that used biologics for microscopic colitis [66]. However, the majority (17/22) were case reports/series, with only four cohort studies. Only two studies reported the use of ustekinumab (an IL-12/IL-23 inhibitor), while 13 studies examined the effects of TNF inhibitors (infliximab or adalimumab), and 11 studies included patients who received vedolizumab, an anti-integrin. Because the majority of the studies had insufficient participant numbers (i.e., fewer than five participants), they did not meet the inclusion criteria. Eight of the studies, comprising 156 patients, were included in the meta-analysis [66] (89 patients on TNF inhibitors and 67 patients on anti-integrin). A response rate of 73% (95% CI, 0.63–0.83; I2 = 0.00%) and a remission rate of 44% (95% CI, 0.32–0.56; I2 = 0.00%) were recorded when using a TNF inhibitor. Using an anti-integrin, a response was found in 73% (95% CI, 0.57–0.87; I2 = 35.93%) with a remission rate of 56% (95% CI, 0.36–0.75; I2 = 46.30%). Owing to sparse data on the effectiveness of ustekinumab in microscopic colitis, more well-powered trials are still warranted. Another recent systematic review and meta-analysis of TNF inhibitors and vedolizumab in 14 studies involving 164 patients additionally identified these biologics to be of clinical benefit for the management of steroid-refractory microscopic colitis and suggested that vedolizumab may be superior to TNF inhibitors [83]. In the context of elderly patients, vedolizumab seems to have a preferred safety profile of the available biologics among these patients [84], although no head-to-head studies have been performed for microscopic colitis [85]. This observation was also noticed in a recent multicenter cohort study [86]. However, cheaper therapeutic alternatives to budesonide should be attempted first, as the expense of biologics may be a potential barrier to their widespread use in the management of microscopic colitis.
Loperamide
Loperamide, a synthetic opioid that primarily affects intestinal opiate receptors to control diarrhea [87], has been studied for microscopic colitis in seven minor studies, five of which were retrospective cohorts. In these studies with 2 mg tablets (up to eight tablets daily), a meta-analysis showed that 62% of the patients responded to loperamide therapy (95% CI, 0.43–0.80; I2 = 92.99%) [66]. For the treatment of microscopic colitis, formal, prospective, and well-powered investigations on loperamide are still required. Until then, this medication may be used as a symptomatic treatment option for elderly individuals with urgency and mild microscopic colitis, or as part of a combination therapy when symptoms do not entirely subside in response to the prescription medication, such as budesonide, although this treatment is not recommended by available guidelines.
Bile Acid Sequestrants
Cholestyramine (a bile acid-binding resin indicated to reduce diarrhea caused by bile acid malabsorption) may be used as a therapy for microscopic colitis [88] or to decrease budesonide dependence in microscopic colitis [89]. Cholestyramine is prescribed at a starting dose of 4 g once or twice per day, and subsequently increased slowly depending on the clinical response to a maximum dose of 24 g/day.
In a meta-analysis, nine studies with 408 patients were identified to examine the effects of bile acid sequestrants on microscopic colitis [66]. Overall, 60% of patients responded to this treatment (95% CI, 0.51–0.68; I2 = 61.65%) and 29% (95% CI, 0.12–0.55) achieved remission. However, in a recent study, no differences were reported in the effectiveness of bile acid sequestrants for microscopic colitis between patients with and without stool or blood tests suggesting bile acid malabsorption [88].
Thiopurines
When symptoms are refractory and considerably affect the quality of life, thiopurines (azathioprine 2–2.5 mg/kg per day or mercaptopurine 1–1.5 mg/kg per day), which are antimetabolites of purines and immunomodulators [90], may be considered for the maintenance of clinical remission as a last resort both based on effectiveness [66], but also due to the risk of complications. Thus, the risk of potential drug interactions, and the development of non-melanoma skin cancer, lymphoma, and infections need to be carefully considered before prescribing this drug to individuals above the age of 60 years [91]. Moreover, owing to the delayed onset of action of approximately 3 months, thiopurines are not ideal for induction therapy [92]. Based on five studies included in a meta-analysis for long-term therapy, an overall response rate to thiopurine exposure was reported in 49% of patients (95% CI, 0.27–0.71; I2 = 81.45%), and remission was achieved in 38% (95% CI, 0.23–0.54; I2 = 50.05%) after a median of 15 months of therapy [66]. Nonetheless, several studies have reported high rates of intolerance to these drugs, and it still remains unknown how long thiopurines should be prescribed in cases of beneficial effects. If this therapy is tolerated, patients should be monitored regularly with routine blood samples, including complete blood cell count and liver and pancreatic function tests [92].
Conclusions
Symptoms related to flares of chronic microscopic colitis are frequent among older adults and include non-bloody watery diarrhea, urgency, and fecal incontinence, which may easily be clinically misdiagnosed as irritable bowel syndrome due to the similarity of symptoms. However, microscopic colitis should always be taken into consideration in geriatric settings in cases of unexplained non-bloody watery diarrhea in elderly individuals aged 60 years or older.
As no biomarkers have yet been identified, patients with long-standing or recurrent watery diarrhea should be referred for colonoscopy with six biopsies in total: three from the ascending and three from the descending colon, even if no macroscopic endoscopic signs are visible. Such biopsies require extensive histopathological evaluation under a microscope to reach a definitive diagnosis and to initiate rational therapy, as medicines are available to control symptoms of microscopic colitis.
All patients diagnosed with microscopic colitis should be recommended to stop smoking and any culprit medication should be discontinued. Although the current treatment options for microscopic colitis are affiliated with limitations, the management of microscopic colitis aims to resolve symptoms and, in this way, improve the quality of life of these patients. Oral budesonide is the drug of choice, but nonsteroidal medications with reasonable response rates, such as bismuth subsalicylate, biologics, bile acid sequestrants, and the supportive antidiarrheal drug loperamide followed by thiopurines (for maintenance therapy only), may be indicated as alternatives for those who are dependent, intolerant, or even fail to respond to this medication [66]. Nevertheless, even if a standard treatment regimen with budesonide (9 mg daily for 6–8 weeks) is usually prescribed, it is still unclear as to how long patients with microscopic colitis on the alternative treatment options should be treated, or if any prophylactic therapy exists.
Finally, as microscopic colitis—a chronic disease with recurrent non-bloody, secretory diarrhea—can be a disabling life experience with impacts on every aspect of patients’ lives, including urgency and often unpredictable fecal incontinence, patients with symptoms should be offered regular follow-ups, and all elderly individuals with microscopic colitis should have easy access to a gastroenterologist in case of flaring disease. In this way, microscopic colitis deserves the same attention as the classical entities of inflammatory bowel diseases.
Acknowledgements
The authors would like to thank Mr. Alexander Hammerhøj and Ms. Sandra Myrtue for assistance with the artwork and flowchart.
Declarations
Funding
Open access funding provided by Copenhagen University. The Memorial Foundation of Solveig Høymann Jacobsen was sponsor of the artwork. The charitable funding source had no role in the design of the study or in the analysis or interpretation of data.
Conflicts of interest
OHN: None, DSP: None.
Availability of data and material
N/A for this type of paper.
Author contributions
OHN wrote original draft, both authors subsequently contributed to, read, and approved the final version of the manuscript.
Ethics approval (appropriate approvals or waivers)
N/A.
Consent to participate
N/A.
Consent for publication
N/A.
Code availability (software application or custom code)
N/A.
References
- 1.Nielsen OH, Fernandez-Banares F, Sato T, et al. Microscopic colitis: etiopathology, diagnosis, and rational management. Elife. 2022;11:e79397. doi: 10.7554/eLife.79397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Aggarwal A, Helper D. Macroscopic findings in lymphocytic colitis. Clin Gastroenterol Hepatol. 2014;12:e65–e66. doi: 10.1016/j.cgh.2013.10.030. [DOI] [PubMed] [Google Scholar]
- 3.Koulaouzidis A, Yung DE, Nemeth A, et al. Macroscopic findings in collagenous colitis: a multi-center, retrospective, observational cohort study. Ann Gastroenterol. 2017;30:309–314. doi: 10.20524/aog.2017.0131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Marlicz W, Skonieczna-Zydecka K, Yung DE, et al. Endoscopic findings and colonic perforation in microscopic colitis: a systematic review. Dig Liver Dis. 2017;49:1073–1085. doi: 10.1016/j.dld.2017.07.015. [DOI] [PubMed] [Google Scholar]
- 5.Yuan L, Wu TT, Zhang L. Microscopic colitis: lymphocytic colitis, collagenous colitis, and beyond. Hum Pathol. 2023;132:89–101. doi: 10.1016/j.humpath.2022.06.027. [DOI] [PubMed] [Google Scholar]
- 6.Chande N, Al Yatama N, Bhanji T, et al. Interventions for treating lymphocytic colitis. Cochrane Database Syst Rev. 2017;7:CD006096. doi: 10.1002/14651858.CD006096.pub4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kafil TS, Nguyen TM, Patton PH, et al. Interventions for treating collagenous colitis. Cochrane Database Syst Rev. 2017;11:CD003575. doi: 10.1002/14651858.CD003575.pub6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Sebastian S, Wilhelm A, Jessica L, et al. Budesonide treatment for microscopic colitis: systematic review and meta-analysis. Eur J Gastroenterol Hepatol. 2019;31:919–927. doi: 10.1097/MEG.0000000000001456. [DOI] [PubMed] [Google Scholar]
- 9.Miehlke S, Guagnozzi D, Zabana Y, et al. European guidelines on microscopic colitis: United European Gastroenterology and European Microscopic Colitis Group statements and recommendations. United European Gastroenterol J. 2021;9:13–37. doi: 10.1177/2050640620951905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Nguyen GC, Smalley WE, Vege SS, et al. American Gastroenterological Association Institute Guideline on the medical management of microscopic colitis. Gastroenterology. 2016;150:242–246. doi: 10.1053/j.gastro.2015.11.008. [DOI] [PubMed] [Google Scholar]
- 11.Pardi DS, Tremaine WJ, Carrasco-Labra A. American Gastroenterological Association Institute Technical Review on the medical management of microscopic colitis. Gastroenterology. 2016;150:247–274. doi: 10.1053/j.gastro.2015.11.006. [DOI] [PubMed] [Google Scholar]
- 12.Verhaegh B, Münch A, Wildt S, et al. The disease course of microscopic colitis – A 5-year prospective European incidence cohort (PRO-MC) United European Gastroenterol J. 2023;11:925–926. [Google Scholar]
- 13.Tong J, Zheng Q, Zhang C, et al. Incidence, prevalence, and temporal trends of microscopic colitis: a systematic review and meta-analysis. Am J Gastroenterol. 2015;110:265–276. doi: 10.1038/ajg.2014.431. [DOI] [PubMed] [Google Scholar]
- 14.Weimers P, Ankersen DV, Lophaven S, et al. Incidence and prevalence of microscopic colitis between 2001 and 2016: a Danish nationwide cohort study. J Crohns Colitis. 2020;14:1717–1723. doi: 10.1093/ecco-jcc/jjaa108. [DOI] [PubMed] [Google Scholar]
- 15.Maye H, Safroneeva E, Godat S, et al. Increasing incidence of microscopic colitis in a population-based cohort study in Switzerland. Clin Gastroenterol Hepatol. 2021;19:2205–2206. doi: 10.1016/j.cgh.2020.10.015. [DOI] [PubMed] [Google Scholar]
- 16.Gentile NM, Khanna S, Loftus EV, Jr, et al. The epidemiology of microscopic colitis in Olmsted County from 2002 to 2010: a population-based study. Clin Gastroenterol Hepatol. 2014;12:838–842. doi: 10.1016/j.cgh.2013.09.066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Tome J, Sehgal K, Kamboj AK, et al. The epidemiology of microscopic colitis in Olmsted County, Minnesota: population-based study from 2011 to 2019. Clin Gastroenterol Hepatol. 2022;20:1085–1094. doi: 10.1016/j.cgh.2021.06.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Lophaven SN, Lynge E, Burisch J. The incidence of inflammatory bowel disease in Denmark 1980–2013: a nationwide cohort study. Aliment Pharmacol Ther. 2017;45:961–972. doi: 10.1111/apt.13971. [DOI] [PubMed] [Google Scholar]
- 19.Fernandez-Banares F, de Sousa MR, Salas A, et al. Epidemiological risk factors in microscopic colitis: a prospective case-control study. Inflamm Bowel Dis. 2013;19:411–417. doi: 10.1002/ibd.23009. [DOI] [PubMed] [Google Scholar]
- 20.Roth B, Manjer J, Ohlsson B. Microscopic colitis is associated with several concomitant diseases. Drug Target Insights. 2013;7:19–25. doi: 10.4137/DTI.S12109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Yadav YS, Eslick GD, Talley NJ. Review article: irritable bowel syndrome: natural history, bowel habit stability and overlap with other gastrointestinal disorders. Aliment Pharmacol Ther. 2021;54(Suppl 1):S24–S32. doi: 10.1111/apt.16624. [DOI] [PubMed] [Google Scholar]
- 22.Abboud R, Pardi DS, Tremaine WJ, et al. Symptomatic overlap between microscopic colitis and irritable bowel syndrome: a prospective study. Inflamm Bowel Dis. 2013;19:550–553. doi: 10.1097/MIB.0b013e31827febfd. [DOI] [PubMed] [Google Scholar]
- 23.Limsui D, Pardi DS, Camilleri M, et al. Symptomatic overlap between irritable bowel syndrome and microscopic colitis. Inflamm Bowel Dis. 2007;13:175–181. doi: 10.1002/ibd.20059. [DOI] [PubMed] [Google Scholar]
- 24.Nielsen OH, Vainer B, Schaffalitzky-de-Muckadell OB. Microscopic colitis: a missed diagnosis? Lancet. 2004;364:2055–2057. doi: 10.1016/S0140-6736(04)17518-1. [DOI] [PubMed] [Google Scholar]
- 25.Fernandez-Banares F, Esteve M, Salas A, et al. Bile acid malabsorption in microscopic colitis and in previously unexplained functional chronic diarrhea. Dig Dis Sci. 2001;46:2231–2238. doi: 10.1023/a:1011927302076. [DOI] [PubMed] [Google Scholar]
- 26.Nimri FM, Muhanna A, Almomani Z, et al. The association between microscopic colitis and celiac disease: a systematic review and meta-analysis. Ann Gastroenterol. 2022;35:281–289. doi: 10.20524/aog.2022.0714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Bergman D, Khalili H, Lebwohl B, et al. Celiac disease and risk of microscopic colitis: a nationwide population-based matched cohort study. United European Gastroenterol J. 2023;11:189–201. doi: 10.1002/ueg2.12374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Zabana Y, Tontini G, Hultgren-Hornquist E, et al. Pathogenesis of microscopic colitis: a systematic review. J Crohns Colitis. 2022;16:143–161. doi: 10.1093/ecco-jcc/jjab123. [DOI] [PubMed] [Google Scholar]
- 29.Miehlke S, Verhaegh B, Tontini GE, et al. Microscopic colitis: pathophysiology and clinical management. Lancet Gastroenterol Hepatol. 2019;4:305–314. doi: 10.1016/S2468-1253(19)30048-2. [DOI] [PubMed] [Google Scholar]
- 30.Nielsen DL, Juhl CB, Chen IM, et al. Immune checkpoint inhibitor-induced diarrhea and colitis: incidence and management a systematic review and meta-analysis. Cancer Treat Rev. 2022;109:102440. doi: 10.1016/j.ctrv.2022.102440. [DOI] [PubMed] [Google Scholar]
- 31.Hughes MS, Molina GE, Chen ST, et al. Budesonide treatment for microscopic colitis from immune checkpoint inhibitors. J Immunother Cancer. 2019;7:292. doi: 10.1186/s40425-019-0756-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Shirwaikar Thomas A, Hanauer S, Wang Y. Immune checkpoint inhibitor enterocolitis vs idiopathic inflammatory bowel disease. Clin Gastroenterol Hepatol. 2023;21:878–890. doi: 10.1016/j.cgh.2022.10.004. [DOI] [PubMed] [Google Scholar]
- 33.ASGE Standards of Practice Committee. Sharaf RN, Shergill AK, et al. Endoscopic mucosal tissue sampling. Gastrointest Endosc. 2013;78:216–224. doi: 10.1016/j.gie.2013.04.167. [DOI] [PubMed] [Google Scholar]
- 34.Malik A, Nadeem M, Javaid S, et al. Estimating the optimum number of colon biopsies for diagnosing microscopic colitis: a systematic review. Eur J Gastroenterol Hepatol. 2022;34:733–738. doi: 10.1097/MEG.0000000000002355. [DOI] [PubMed] [Google Scholar]
- 35.Lindstrom CG. 'Collagenous colitis' with watery diarrhea—a new entity? Pathol Eur. 1976;11:87–89. [PubMed] [Google Scholar]
- 36.Read NW, Krejs GJ, Read MG, et al. Chronic diarrhea of unknown origin. Gastroenterology. 1980;78:264–271. [PubMed] [Google Scholar]
- 37.Lazenby AJ, Yardley JH, Giardiello FM, et al. Lymphocytic ("microscopic") colitis: a comparative histopathologic study with particular reference to collagenous colitis. Hum Pathol. 1989;20:18–28. doi: 10.1016/0046-8177(89)90198-6. [DOI] [PubMed] [Google Scholar]
- 38.Bjornbak C, Engel PJ, Nielsen PL, et al. Microscopic colitis: clinical findings, topography and persistence of histopathological subgroups. Aliment Pharmacol Ther. 2011;34:1225–1234. doi: 10.1111/j.1365-2036.2011.04865.x. [DOI] [PubMed] [Google Scholar]
- 39.Munch A, Mihaly E, Nagy F, et al. Budesonide as induction therapy for incomplete microscopic colitis: A randomised, placebo-controlled multicentre trial. United European Gastroenterol J. 2021;9:837–847. doi: 10.1002/ueg2.12131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Magro F, Langner C, Driessen A, et al. European consensus on the histopathology of inflammatory bowel disease. J Crohns Colitis. 2013;7:827–851. doi: 10.1016/j.crohns.2013.06.001. [DOI] [PubMed] [Google Scholar]
- 41.Mihaly E, Patai A, Tulassay Z. Controversials of microscopic colitis. Front Med (Lausanne) 2021;8:717438. doi: 10.3389/fmed.2021.717438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Enck P, Aziz Q, Barbara G, et al. Irritable bowel syndrome. Nat Rev Dis Primers. 2016;2:16014. doi: 10.1038/nrdp.2016.14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Verhaegh BP, de Vries F, Masclee AA, et al. High risk of drug-induced microscopic colitis with concomitant use of NSAIDs and proton pump inhibitors. Aliment Pharmacol Ther. 2016;43:1004–1013. doi: 10.1111/apt.13583. [DOI] [PubMed] [Google Scholar]
- 44.Hamdeh S, Micic D, Hanauer S. Drug-induced colitis. Clin Gastroenterol Hepatol. 2021;19:1759–1779. doi: 10.1016/j.cgh.2020.04.069. [DOI] [PubMed] [Google Scholar]
- 45.Monteiro C, Silvestre S, Duarte AP, et al. Safety of non-steroidal anti-inflammatory drugs in the elderly: an analysis of published literature and reports sent to the Portuguese pharmacovigilance system. Int J Environ Res Public Health. 2022;19:3541. doi: 10.3390/ijerph19063541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Torres-Bondia F, de Batlle J, Galvan L, et al. Evolution of the consumption trend of proton pump inhibitors in the Lleida Health Region between 2002 and 2015. BMC Public Health. 2022;22:818. doi: 10.1186/s12889-022-13217-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Felicia-D'Sa F, Fernandes EZ, Kesarkar SV, et al. Use of histamine-2 receptor antagonists and risk of inflammatory bowel diseases: a systematic review and meta-analysis of observational studies. J Clin Pharm Ther. 2022;47:1103–1111. doi: 10.1111/jcpt.13662. [DOI] [PubMed] [Google Scholar]
- 48.Holz K, Martos NA, Margolis RL. Clozapine associated with microscopic colitis in the setting of biopsy-proven celiac disease. J Clin Psychopharmacol. 2018;38:150–152. doi: 10.1097/JCP.0000000000000844. [DOI] [PubMed] [Google Scholar]
- 49.Zhang SW, Xu RH, Chen D. Drug exposure and risk of microscopic colitis: a systematic review and meta-analysis. Dig Dis. 2023;41:217–226. doi: 10.1159/000526809. [DOI] [PubMed] [Google Scholar]
- 50.Tarar ZI, Farooq U, Gandhi M, et al. Are drugs associated with microscopic colitis? A systematic review and meta-analysis. Diseases. 2022;11:6. doi: 10.3390/diseases11010006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Zylberberg HM, Kamboj AK, De Cuir N, et al. Medication use and microscopic colitis: a multicentre retrospective cohort study. Aliment Pharmacol Ther. 2021;53:1209–1215. doi: 10.1111/apt.16363. [DOI] [PubMed] [Google Scholar]
- 52.Lucendo AJ. Drug exposure and the risk of microscopic colitis: a critical update. Drugs R D. 2017;17:79–89. doi: 10.1007/s40268-016-0171-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Beaugerie L, Itzkowitz SH. Cancers complicating inflammatory bowel disease. N Engl J Med. 2015;372:1441–1452. doi: 10.1056/NEJMra1403718. [DOI] [PubMed] [Google Scholar]
- 54.Le Berre C, Honap S, Peyrin-Biroulet L. Ulcerative colitis. Lancet. 2023;402:571–584. doi: 10.1016/S0140-6736(23)00966-2. [DOI] [PubMed] [Google Scholar]
- 55.Roda G, Chien Ng S, Kotze PG, et al. Crohn's disease. Nat Rev Dis Primers. 2020;6:22. doi: 10.1038/s41572-020-0156-2. [DOI] [PubMed] [Google Scholar]
- 56.Bergman D, Khalili H, Roelstraete B, et al. Microscopic colitis and risk of cancer - a population-based cohort study. J Crohns Colitis. 2021;15:212–221. doi: 10.1093/ecco-jcc/jjaa156. [DOI] [PubMed] [Google Scholar]
- 57.Kamboj AK, Zylberberg HM, Lane CM, et al. Microscopic colitis and risk of colon adenomas: a multicenter retrospective cohort study. Clin Gastroenterol Hepatol. 2022;20:e902–e904. doi: 10.1016/j.cgh.2021.05.050. [DOI] [PubMed] [Google Scholar]
- 58.Liu YH, Wu Z, Ding JY, et al. Microscopic colitis is associated with a reduced risk of colorectal adenoma and cancer: a meta-analysis. Inflamm Bowel Dis. 2022;28:1584–1591. doi: 10.1093/ibd/izab333. [DOI] [PubMed] [Google Scholar]
- 59.Forss A, Bergman D, Roelstraete B, et al. Patients with microscopic colitis are at higher risk of major adverse cardiovascular events: a matched cohort study. Clin Gastroenterol Hepatol. 2023;21:3356–3364. doi: 10.1016/j.cgh.2023.05.014. [DOI] [PubMed] [Google Scholar]
- 60.Harvey RF, Bradshaw JM. A simple index of Crohn's-disease activity. Lancet. 1980;1:514. doi: 10.1016/s0140-6736(80)92767-1. [DOI] [PubMed] [Google Scholar]
- 61.Best WR, Becktel JM, Singleton JW, et al. Development of a Crohn's disease activity index. National Cooperative Crohn's Disease Study. Gastroenterology. 1976;70:439–444. [PubMed] [Google Scholar]
- 62.Schroeder KW, Tremaine WJ, Ilstrup DM. Coated oral 5-aminosalicylic acid therapy for mildly to moderately active ulcerative colitis. A randomized study. N Engl J Med. 1987;317:1625–1629. doi: 10.1056/NEJM198712243172603. [DOI] [PubMed] [Google Scholar]
- 63.Walmsley RS, Ayres RC, Pounder RE, et al. A simple clinical colitis activity index. Gut. 1998;43:29–32. doi: 10.1136/gut.43.1.29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Chande N, MacDonald JK, McDonald JW. Interventions for treating microscopic colitis: a Cochrane Inflammatory Bowel Disease and Functional Bowel Disorders Review Group systematic review of randomized trials. Am J Gastroenterol. 2009;104:235–241. doi: 10.1038/ajg.2008.16. [DOI] [PubMed] [Google Scholar]
- 65.Hjortswang H, Tysk C, Bohr J, et al. Defining clinical criteria for clinical remission and disease activity in collagenous colitis. Inflamm Bowel Dis. 2009;15:1875–1881. doi: 10.1002/ibd.20977. [DOI] [PubMed] [Google Scholar]
- 66.Rehde A, Hendel SK, Juhl CB, et al. Effectiveness of non-budesonide therapies in management of microscopic colitis: a systematic review and meta-analysis. Drugs. 2023;83:1027–1038. doi: 10.1007/s40265-023-01914-4. [DOI] [PubMed] [Google Scholar]
- 67.Cotter TG, Binder M, Loftus EV, Jr, et al. Development of a microscopic colitis disease activity index: a prospective cohort study. Gut. 2018;67:441–446. doi: 10.1136/gutjnl-2016-313051. [DOI] [PubMed] [Google Scholar]
- 68.Desrame J, Sabate JM, Agher R, et al. Assessment of hypothalamic-pituitary-adrenal axis function after corticosteroid therapy in inflammatory bowel disease. Am J Gastroenterol. 2002;97:1785–1791. doi: 10.1111/j.1572-0241.2002.05786.x. [DOI] [PubMed] [Google Scholar]
- 69.O'Donnell S, O'Morain CA. Therapeutic benefits of budesonide in gastroenterology. Ther Adv Chronic Dis. 2010;1:177–186. doi: 10.1177/2040622310379293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Loreau J, Duricova D, Gower-Rousseau C, et al. Long-term natural history of microscopic colitis: a population-based cohort. Clin Transl Gastroenterol. 2019;10:e00071. doi: 10.14309/ctg.0000000000000071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Fernandez-Banares F, Salas A, Esteve M, et al. Collagenous and lymphocytic colitis. Evaluation of clinical and histological features, response to treatment, and long-term follow-up. Am J Gastroenterol. 2003;98:340–347. doi: 10.1111/j.1572-0241.2003.07225.x. [DOI] [PubMed] [Google Scholar]
- 72.Miehlke S, Madisch A, Voss C, et al. Long-term follow-up of collagenous colitis after induction of clinical remission with budesonide. Aliment Pharmacol Ther. 2005;22:1115–1119. doi: 10.1111/j.1365-2036.2005.02688.x. [DOI] [PubMed] [Google Scholar]
- 73.Tome J, Tariq R, Hassett LC, et al. Effectiveness and safety profile of budesonide maintenance in microscopic colitis: a systematic review and meta-analysis. Inflamm Bowel Dis. 2024 doi: 10.1093/ibd/izad178. [DOI] [PubMed] [Google Scholar]
- 74.Tome J, Sehgal K, Kamboj AK, et al. Budesonide maintenance in microscopic colitis: clinical outcomes and safety profile from a population-based study. Am J Gastroenterol. 2022;117:1311–1315. doi: 10.14309/ajg.0000000000001774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Gentile NM, Abdalla AA, Khanna S, et al. Outcomes of patients with microscopic colitis treated with corticosteroids: a population-based study. Am J Gastroenterol. 2013;108:256–259. doi: 10.1038/ajg.2012.416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Cotter TG, Kamboj AK, Hicks SB, et al. Immune modulator therapy for microscopic colitis in a case series of 73 patients. Aliment Pharmacol Ther. 2017;46:169–174. doi: 10.1111/apt.14133. [DOI] [PubMed] [Google Scholar]
- 77.Miehlke S, Aust D, Mihaly E, et al. Efficacy and safety of budesonide, vs mesalazine or placebo, as induction therapy for lymphocytic colitis. Gastroenterology. 2018;155:1795–1804. doi: 10.1053/j.gastro.2018.08.042. [DOI] [PubMed] [Google Scholar]
- 78.Miehlke S, Madisch A, Kupcinskas L, et al. Budesonide is more effective than mesalamine or placebo in short-term treatment of collagenous colitis. Gastroenterology. 2014;146:1222–1230. doi: 10.1053/j.gastro.2014.01.019. [DOI] [PubMed] [Google Scholar]
- 79.Senderovich H, Vierhout M. Is there a role for bismuth in diarrhea management? Rambam Maimonides Med J. 2021;12:e0002. doi: 10.5041/RMMJ.10422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Ericsson CD, Tannenbaum C, Charles TT. Antisecretory and antiinflammatory properties of bismuth subsalicylate. Rev Infect Dis. 1990;12(Suppl 1):S16–20. doi: 10.1093/clinids/12.supplement_1.s16. [DOI] [PubMed] [Google Scholar]
- 81.Borbinha C, Serrazina F, Salavisa M, et al. Bismuth encephalopathy—a rare complication of long-standing use of bismuth subsalicylate. BMC Neurol. 2019;19:212. doi: 10.1186/s12883-019-1437-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Hogan DB, Harbidge C, Duncan A. Bismuth toxicity presenting as declining mobility and falls. Can Geriatr J. 2018;21:307–309. doi: 10.5770/cgj.21.323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.El Hage CN, Ghoneim S, Shah S, et al. Efficacy and safety of vedolizumab and tumor necrosis factor inhibitors in the treatment of steroid-refractory microscopic colitis: a systematic review and meta-analysis. J Clin Gastroenterol. 2024 doi: 10.1097/MCG.0000000000001914. [DOI] [PubMed] [Google Scholar]
- 84.Khan N, Pernes T, Weiss A, et al. Efficacy of vedolizumab in a nationwide cohort of elderly inflammatory bowel disease patients. Inflamm Bowel Dis. 2022;28:734–744. doi: 10.1093/ibd/izab163. [DOI] [PubMed] [Google Scholar]
- 85.Hahn GD, Golovics PA, Wetwittayakhlang P, et al. Safety of biological therapies in elderly inflammatory bowel diseases: a systematic review and meta-analysis. J Clin Med. 2022;11:4422. doi: 10.3390/jcm11154422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Boivineau G, Zallot C, Zerbib F, et al. Biologic therapy for budesonide-refractory, -dependent or -intolerant microscopic colitis: a multicentre cohort study from the GETAID. J Crohns Colitis. 2022;16:1816–1824. doi: 10.1093/ecco-jcc/jjac089. [DOI] [PubMed] [Google Scholar]
- 87.Baker DE. Loperamide: a pharmacological review. Rev Gastroenterol Disord. 2007;7(Suppl 3):S11–S18. [PubMed] [Google Scholar]
- 88.Tome J, Sehgal K, Kamboj AK, et al. Bile acid sequestrants in microscopic colitis: clinical outcomes and utility of bile acid testing. Clin Gastroenterol Hepatol. 2023;21:3125–3131. doi: 10.1016/j.cgh.2023.04.031. [DOI] [PubMed] [Google Scholar]
- 89.Northcutt MJ, Gentile NM, Goldstein JL, et al. Bile acid sequestrant therapy in microscopic colitis. J Clin Gastroenterol. 2022;56:161–165. doi: 10.1097/MCG.0000000000001496. [DOI] [PubMed] [Google Scholar]
- 90.de Boer NKH, Peyrin-Biroulet L, Jharap B, et al. Thiopurines in inflammatory bowel disease: new findings and perspectives. J Crohns Colitis. 2018;12:610–620. doi: 10.1093/ecco-jcc/jjx181. [DOI] [PubMed] [Google Scholar]
- 91.Calafat M, Manosa M, Canete F, et al. Increased risk of thiopurine-related adverse events in elderly patients with IBD. Aliment Pharmacol Ther. 2019;50:780–788. doi: 10.1111/apt.15458. [DOI] [PubMed] [Google Scholar]
- 92.Nielsen OH, Bjerrum JT, Herfarth H, et al. Recent advances using immunomodulators for inflammatory bowel disease. J Clin Pharmacol. 2013;53:575–588. doi: 10.1002/jcph.2. [DOI] [PubMed] [Google Scholar]