Abstract
The initial host response to Mycobacterium tuberculosis is driven by innate immunity. For this study, we examined the ability of 18 recent clinical isolates and 5 reference strains to survive and replicate in the context of host innate immunity by using whole blood culture. Six healthy tuberculin-negative volunteers served as subjects. H37Ra showed the least capacity to replicate of any of the strains tested, decreasing in viability 1.3 log CFU during 72 h of whole blood culture, whereas H37Rv increased 0.32 log. Clinical isolates varied greatly in their ability to replicate in blood cells, ranging from −0.4 to +0.8 log (P < 0.001). Four showed significantly more growth than H37Rv, and one showed significantly reduced growth. Host mechanisms for restricting intracellular mycobacterial growth were more effective during the first 24 h of whole blood culture than during the 24- to 72-h period. Certain mycobacterial isolates appeared preferentially able to withstand host defenses during each of these intervals. Although there was relatively more homogeneity among subjects than among strains, one of the six subjects showed a reduced capacity to restrict intracellular mycobacterial growth due to a defect expressed during the first 24 h of culture. Our findings indicate substantial variability in the capacity of clinical tuberculosis isolates to replicate in host cells in the face of innate host immunity.
The early events following inhalation by an immunocompetent, mycobacterium-naïve host of droplet nuclei containing viable Mycobacterium tuberculosis are driven by the innate immune system. The resulting influx of neutrophils, macrophages, NK cells, and other cells to the site of infection serves as a stimulus for granuloma formation and acts directly to limit the extent of mycobacterial replication at this early stage of infection. It has been suggested that the efficiency of these early innate responses may help determine whether a latent infection is established and whether that infection ultimately will progress to active disease.
Through evolutionary selection, pathogenic mycobacteria have acquired means to evade specific host immune effector mechanisms, presumably including those of the innate response. The propensity of certain M. tuberculosis isolates to cause outbreaks, for example, has been linked to increased virulence in macrophages or mice in association with altered host cytokine expression profiles (2, 3, 6, 8, 12). However, even these virulent outbreak-associated isolates cause disease in only a small proportion of infected individuals. Our understanding of the interplay of biologic and genetic diversity in the host and in the mycobacterium is incomplete, in part because current models to examine the early events in mycobacterial pathogenesis have not been well suited to field or epidemiologic human studies.
For the present study, we assessed the capacity of mycobacterial strains to survive phagocytosis and replicate in whole blood cultures. This model permits the involvement of neutrophils, as well as monocytes and lymphocytes, in the antimycobacterial host response (Fig. 1). We and others have previously used whole blood cultures to study acquired host immune responses following vaccination with Mycobacterium bovis BCG (1, 4, 16). The model's simplicity permits the study of relatively large numbers of strains and subjects. For this project, 18 recent prospectively collected M. tuberculosis clinical isolates were compared to five reference strains by the use of whole blood cultures from six healthy, tuberculin-nonreactive donors. Care was taken to ensure that the recent clinical isolates were minimally passaged prior to study to avoid the selection of substrains that were adapted to in vitro replication. The reference strains, which had undergone extensive in vitro culture in broth, included HN878 (also known as 210), a group W-Beijing, outbreak-associated strain; CDC1551, a strain associated with an unusually large number of tuberculin skin test conversions; H37Rv, a widely studied reference isolate that is virulent in animal models; H37Ra, H37Rv's highly attenuated sibling; and MP-28, an isolate that was selected at random from a similar study conducted in Brazil in 1997 (18) and that was cultured extensively in vitro during the course of three subsequent whole-blood infection studies, in which it was identified as strain 28 (1, 16, 19). The objective of the present project was to examine the influence of strain variation in M. tuberculosis on the early events of human tuberculosis (TB) pathogenesis.
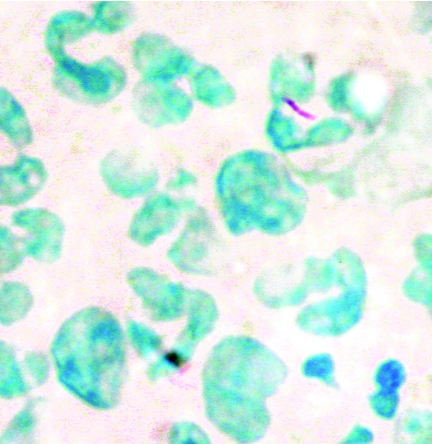
FIG. 1.
Photomicrograph showing neutrophil-associated M. tuberculosis H37Ra 1 h after inoculation into whole blood culture.
MATERIALS AND METHODS
Mycobacterial isolates.
Isolates were collected prospectively in a study of surrogate markers of the response to chemotherapy conducted in Vitória, Brazil (19). Briefly, subjects were 18- to 60-year-old human immunodeficiency virus type 1-seronegative patients with newly diagnosed initial episodes of sputum smear-positive pulmonary tuberculosis. Drug-resistant isolates were excluded. Isolates were initially propagated in BACTEC 12B medium and frozen for transport to the United States. M. tuberculosis strains H37Ra and H37Rv were obtained from the American Type Culture Collection. Strains HN878 and CDC1551 were provided by A. Kreiswirth. Strain MP-28 was previously selected at random from a study conducted in Brazil in 1997 (1, 18). The selection of strains and their analysis by whole blood culture were performed without knowledge of restriction fragment length polymorphism (RFLP) results.
Isolates were propagated in BACTEC 13A until growth indices (GI) reached 350, at which point they were frozen in aliquots. The numbers of viable organisms in these stock cultures were determined by the use of BACTEC. Serial 10-fold dilutions were inoculated into BACTEC 12B bottles to establish the relationship between the log inoculum volume and the time (days) to positivity. The inoculum used for whole blood culture was the volume that was positive in BACTEC (GI = 30) in 4.5 days, based on regression analyses of serial dilutions of stock placed into culture. In the case of M. tuberculosis H37Ra, the inoculum contained 1.2 × 103 CFU, as determined by colony counts on agar.
Whole blood culture.
Blood was collected from healthy volunteers without tuberculin skin test reactivity (0-mm induration at 48 h) or a known history of exposure to tuberculosis. The protocol was approved by the UMDNJ Institutional Review Board and followed procedures for human subject protection as outlined by the U.S. Department of Health and Human Services. Each subject underwent phlebotomy three to five times over approximately 8 to 14 months. Whole blood culture was performed as previously described (1). All cultures were performed in duplicate.
The mycobacterial inoculum was sedimented and resuspended in 300 μl of tissue culture medium. An equal volume of blood was added, resulting in an infection ratio of less than 1 CFU per 10 monocytes. Tubes were sealed and incubated at 37°C with slow constant mixing.
For some experiments, cultures were harvested after 1 h to assess mycobacterial localization. For these experiments, red blood cells were lysed in ammonium chloride. The remaining cells were resuspended in phosphate-buffered saline. Microscope slides were prepared with a cytocentrifuge, stained with Kinyoun stain, and examined by light microscopy.
After 24 or 72 h of whole blood culture, the supernatants of replicate cultures were removed, pooled, sterilized by passage through a 0.2-μm-pore-size filter, and frozen. Pelleted host cells were disrupted by hypotonic lysis. Bacilli were sedimented, resuspended in BACTEC 12B medium, and inoculated in 12B bottles. Growth indices were monitored at 24-h intervals. Changes in viability during whole blood culture were determined by comparing the time (days) to positivity of the inoculum and that of the completed whole blood culture, using the standard curve for each mycobacterial stock. The means of duplicate cultures were used as single values in subsequent calculations. Software developed by Robert S. Wallis to facilitate the calculation of Δlog CFU from daily growth indices is available online at http://tuberculosis.umdnj.edu/wba-tbrx-suppl.htm. Changes in log CFU counts from 0 to 24 and 0 to 72 h were measured directly; the change from 24 to 72 h was calculated as the difference between these values.
Assessment of mycobacterial growth rates in BACTEC.
Raw data from the standard curve for each isolate were analyzed to determine the rate of growth in broth culture. The inoculum volumes selected for analysis were those that were immediately more than and less than the volume used to inoculate whole blood cultures. The GI values used were those recorded on the day before and the day of positivity (GI > 30), representing early-log-phase growth. The growth rate for each isolate was determined as the mean of the differences in log GI values for these two cultures.
Cytokine analysis.
Supernatants of 24-h cultures were analyzed for interleukin-1β (IL-1β), tumor necrosis factor (TNF), TNF receptor II (TNFRII), intercellular adhesion molecule (ICAM), monocyte chemotactic protein 1 (MCP-1), macrophage inflammatory protein 1α (MIP-1α), IL-10, transforming growth factor β1 (TGF-β1), and IL-12 p40 by use of a FastQuant protein microarray (Schleicher & Schuell, Keene, N.H.). The analysis was performed by the manufacturer. Briefly, FAST slides were arrayed in triplicate with anticytokine antibodies. After blocking nonspecific binding, specimens were added and incubated for 3 h at room temperature. Arrays were washed and incubated with an antibody cocktail containing one biotinylated antibody corresponding to each of the capture antibodies. Arrays were incubated, washed, incubated with a streptavidin-Cy5 conjugate, and then imaged in a Perkin-Elmer ScanArry 4000 confocal fluorescence imaging system. Array images were saved as 16-bit TIFF files with a 10-μm resolution. Images were then analyzed with ArrayVision (Imaging Research). Each specimen was analyzed by using the standard curve for that cytokine. The sensitivity of the assays ranged from 1 pg/ml (IL-1β and TNF-α) to 100 pg/ml (TNFRII).
Supernatants of 24-h cultures were analyzed for alpha interferon (IFN-α) by an enzyme-linked immunosorbent assay (R&D Systems) according to the manufacturer's instructions. In addition, supernatants of 72-h cultures were analyzed for IFN-γ by another enzyme-linked immunosorbent assay (R&D Systems) according to the manufacturer's instructions.
Strain typing.
Each clinical M. tuberculosis isolate was genetically characterized by IS6110 DNA fingerprinting (14). The resulting RFLP patterns were compared to each other and to a database of isolates established for this purpose (9, 10).
Statistical analysis.
Multiple comparisons were examined by the Friedman repeated-measures analysis of variance on ranks (RM ANOVA). RM ANOVA permits the accurate assessment of differences when several treatments or experimental conditions are tested on all members of a single group of subjects, as it accounts for differences among the subjects in its analysis of the effects of the experimental treatments. Post hoc testing to identify differences between pairs was then performed by the Student-Newman-Keuls method to minimize the risk of a type 1 error (finding a difference when one does not exist) due to multiple comparisons. Statistical testing was performed with SigmaStat (Systat).
RESULTS
Strain typing.
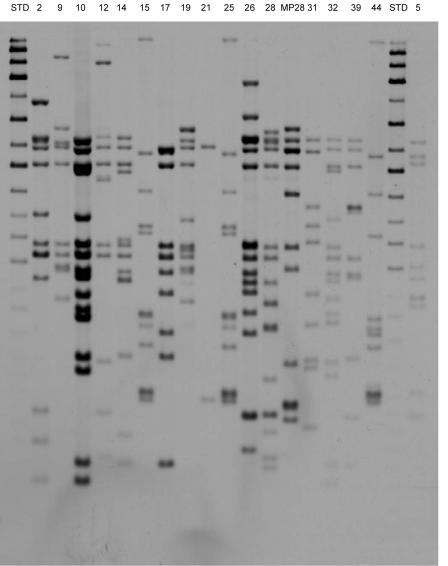
Strain typing by restriction length fragment polymorphism analysis using IS6110 was performed after the selection, propagation, and testing of the isolates in whole blood culture had been completed. The results are shown in Fig. 2. None of the clinical isolates belonged to the W-Beijing or Harlem family. They were genetically diverse, with two exceptions: strains 15 and 25 had the same IS6110 fingerprint (CH12) and were closely related to strain 44 (cluster C-1). Similarly, isolates 9 and 19 were closely related (cluster C-2). The typing of strain 8 was unsuccessful due to contamination.
FIG. 2.
RFLP typing of clinical strains using IS6110. Strains 15 and 25 showed identical fingerprints, to which the strain 44 fingerprint was closely related (cluster C-1). Similarly, isolates 9 and 19 were closely related (cluster C-2).
Mycobacterial growth in broth.
Mycobacterial replication rates in broth were assessed in duplicate BACTEC 12B cultures. We have previously documented that a change in the log BACTEC GI correlates with a change in the log CFU (17). The isolates in the present study showed a median increase in log GI during early growth in BACTEC 12B cultures of 0.45 over 24 h, corresponding to a doubling time of 17 h. The range between the most and the least growth was 0.39 log (2.45-fold difference), as indicated on the horizontal axis in Fig. 3. Reference isolates (solid circles) tended to grow more rapidly in broth than recent clinical isolates (open circles, 0.521 versus 0.428 log GI, P = 0.048), possibly indicating selection during repeated in vitro passage. Reference strains also showed somewhat less variability in replicate broth cultures than recent clinical isolates (median interquartile range [IQR], 0.023 versus 0.079 log GI, P = 0.068), possibly indicating reduced clumping or reduced genetic variation among individual CFU.
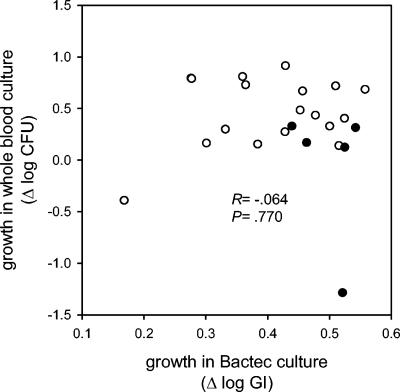
FIG. 3.
Lack of correlation between bacillary growth in 72-h whole blood cultures and that in 24-h BACTEC cultures. Each symbol represents the median of two replicate cultures in broth (horizontal axis) and two replicate whole blood cultures for each of six subjects (vertical axis). Recent clinical isolates are indicated with open circles, and reference strains are indicated with filled circles.
Mycobacterial growth in whole blood culture.
Mycobacterial growth was reduced in whole blood cultures compared to that in broth (0.110 log CFU per 24 h over 72 h, P < 0.001 compared to BACTEC); this rate corresponded to a doubling time of 37 h. However, the range between the most and the least growth, 2.2 log (158-fold difference), was substantially larger than that in broth. There was no correlation between growth in broth and growth in whole blood cultures (R = −0.064, P = 0.77) (Fig. 3). Reference strains, which grew more readily than recent clinical isolates in broth, showed somewhat reduced capacities for growth in whole blood culture than recent clinical isolates (0.17 versus 0.46 log CFU over 72 h, P = 0.044), possibly due to selection by repeated in vitro passages.
Analysis by strain.
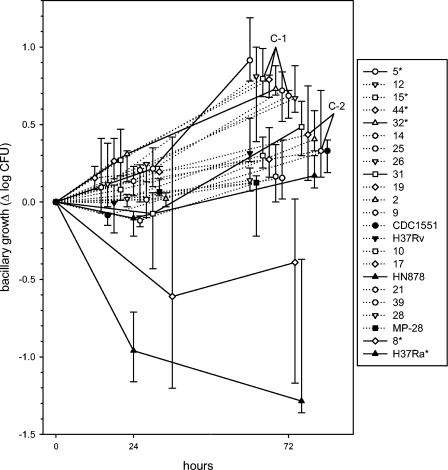
The kinetics of intracellular mycobacterial growth in whole blood culture, analyzed by strain, are shown in Fig. 4. Isolates increased by a median of 0.08 log CFU during the first 24 h, in contrast to 0.21 per 24 h during the 24- to 72-h period (P = 0.003). The attenuated strain H37Ra, for example, decreased nearly 1 log (−0.96) during the first 24 h but declined quite slowly thereafter (−0.063 per 24 h). It was the most attenuated strain tested. In contrast, counts of H37Rv remained stable during the first 24 h (−0.005) and subsequently increased by 0.223 log per 24 h. There was no overall relationship between the rates of growth of various isolates during these two intervals (R = 0.28, P = 0.19). As a result, both intervals contributed equally as independent determinants of net growth during the full 72-h period (P < 0.001 for both intervals).
FIG. 4.
Kinetics of mycobacterial growth in whole blood cultures of 24 and 72 h, analyzed according to strain. Symbols indicate medians and IQRs for six subjects. The horizontal axis has been staggered to reduce overlap. Reference strains are indicated as filled symbols, and recent clinical isolates are indicated as open symbols. The listing of strains in the graph legend indicates their rank order according to growth after 72 h. Strains differed significantly by ANOVA after both 24 and 72 h (P < 0.001 for both). Strains 5, 15, 44, 32, 8, and H37Ra (indicated with asterisks) differed from H37Rv in post hoc testing. Two clusters of genetically identical or closely related strains (C-1 and C-2) are indicated.
RM ANOVA identified significant differences among strains after both 24 and 72 h (P < 0.001 for both); these differences remained highly significant when the analysis was restricted to recent clinical isolates. The median growth rates of six clinical isolates were intermediate to those of H37Ra and H37Rv, whereas 12 (67%) rates were higher than that of H37Rv. Post hoc testing revealed that four of these clinical isolates (5, 15, 44, and 32) showed significantly more growth than H37Rv, whereas the growth of one strain (8) was significantly reduced. H37Ra differed significantly from all other isolates tested. In contrast, no statistically significant differences were found among those isolates that were identical or closely related by RFLP analysis (clusters C-1 and C-2), as indicated by the proximity of their median values in Fig. 4. All recently clustered isolates showed median growth superior to that of H37Rv.
The extent of variation in mycobacterial replication in duplicate whole blood cultures (0.051 log CFU) was similar to that in BACTEC cultures. Analyses by strain revealed more differences among subjects for attenuated strains than for virulent strains, as indicated by the inverse correlation between the log growth rate and the IQR (R = −0.629, P = 0.001). The fact that this was reproducibly observed for whole blood culture but not broth culture suggests that it has a biologic basis which is unrelated to colonial heterogeneity or clumping. Variability was also somewhat higher for recent clinical isolates than for reference strains (0.46 versus 0.17 log CFU, P = 0.044), but multiple regression analysis indicated that statistical significance was lost after controlling for the effects of attenuation.
Analysis by subject.
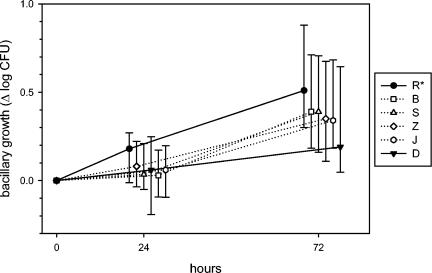
There was more homogeneity among subjects than among strains (Fig. 5). Nonetheless, one of the six subjects (R) had a diminished capacity to restrict intracellular mycobacterial growth (P = 0.014) due to a defect expressed during the initial 24 h of culture. A second subject (D) tended to show a superior ability to restrict intracellular growth that approached but did not reach statistical significance. In contrast to the defect of subject R, this ability was expressed solely during the second culture interval (24 to 72 h).
FIG. 5.
Kinetics of mycobacterial growth in whole blood culture, analyzed according to subject. Symbols indicate median values for 23 strains which were tested in duplicate. Subjects differed significantly at 72 h by ANOVA (P = 0.014). Post hoc testing revealed that subject R differed from all other subjects; a trend for subject D approached but did not reach statistical significance.
Potential surrogate markers.
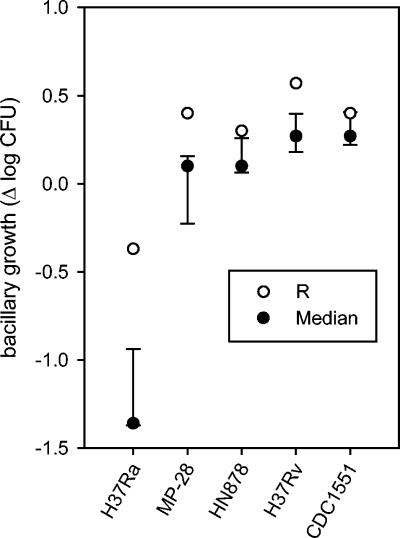
Field studies to examine the relationship between innate immune function and protection from tuberculosis would be facilitated if one mycobacterial strain could serve as an indicator of the ability to control the growth of many strains or if other surrogate markers for intracellular killing could be identified. To address this possibility, we compared subject R to all other subjects with respect to the ability to control the growth of each of the reference isolates. As indicated in Fig. 6, the difference between subject R and the remaining subjects was largest when tested with the attenuated strain H37Ra. Based on this observation, we then analyzed supernatants from 24-h whole blood cultures inoculated with H37Ra and H37Rv to identify a cytokine profile associated with inadequate initial control of mycobacterial growth. A preliminary analysis determined that subject R showed higher than anticipated levels of IFN-α and IL-10 compared to all other subjects but did not differ with respect to IL-1β, MIP-1α, IL-12 p40, MCP-1, TNFRII, TGF-β1, TNF, and ICAM (Table 1). An analysis of 72-h supernatants for IFN-γ revealed slightly lower levels for subject R than anticipated, although the levels in all subjects were substantially reduced compared to the expected levels in tuberculin skin test reactors (4).
FIG. 6.
Extent of growth of reference M. tuberculosis strains in 72-h whole blood cultures from subject R (open circles) compared to those from other subjects (filled circles).
TABLE 1.
Cytokines induced by M. tuberculosis strains H37Ra and H37Rv in whole blood cultures from subject R compared to those from all other subjects
| Cytokine | Amt of cytokine (pg/ml) induced by H37Rva
|
Amt of cytokine (pg/ml) induced by H37Raa
|
||
|---|---|---|---|---|
| Subject R | All other subjects | Subject R | All other subjects | |
| IFN-α | 32 | 1 (1-10) | 26 | 7 (5-14) |
| IL-1β | <1 | 1 (1-93) | <1 | 1 (1-117) |
| IL-10 | 758 | 246 (116-632) | 1,336 | 108 (80-585) |
| MIP-1α | 92 | 30 (15-618) | 82 | 32 (6-2,293) |
| IL-12 p40 | 31 | 42 (26-75) | 191 | 27 (10-266) |
| MCP-1 | 358 | 498 (254-908) | 1828 | 908 (734-3,905) |
| TNFRII | 17,915 | 25,788 (13120-31249) | 30,858 | 34,299 (24,883-36,708) |
| TGF-β1 | <20 | <20 (20-20) | <20 | <20 (20-63) |
| TNF-α | 14 | 22 (12-37) | 22 | 32 (17-74) |
| ICAM | 70,437 | 73,768 (56,693-89,990) | 62,914 | 75,902 (63,197-87,173) |
| IFN-γ | <1 | 1 (1-155) | <1 | 6 (1-48) |
Data are medians and IQRs (in parentheses).
DISCUSSION
A growing body of literature indicates that significant biologic variation exists among clinical M. tuberculosis isolates. Dormans et al., for example, found differences in survival, lung pathology, bacterial load, and delayed-type hypersensitivity when BALB/c mice were infected with 19 genetically distinct TB strains (2). Li et al. (6), Hoal-van Helden et al. (3), Manca et al. (7), and Theus et al. (12) have found similar variations with respect to replication and cytokine induction in human monocytes, monocyte-derived macrophages, dendritic cells, or macrophage-like tumor cell lines. These reports mainly have examined unique characteristics of certain group W strains, in which differential monocyte activation regulated by mycobacterial lipids has been implicated as a determinant of virulence (11).
The present study complements these reports in several key respects. It represents the largest study to date of the biologic diversity in M. tuberculosis using in vitro or animal infection models. Its main findings are that randomly selected recent M. tuberculosis clinical isolates differ substantially in their capacity to replicate within human blood cells and that a large proportion show superior growth to that of the reference strain H37Rv, even though none were associated with an outbreak-associated genotype such as group W-Beijing. These findings indicate that significant gaps exist in our current understanding of virulence in clinical isolates of M. tuberculosis.
This is also the first report in which a whole blood model was used to examine differential mycobacterial survival and replication as surrogates for virulence. Neutrophils participate in the phagocytosis of M. tuberculosis added to whole blood cultures. Kisich et al. have observed that neutrophils may contribute to host defenses against M. tuberculosis by the expression of defensins and other antibiotic peptides (5). Their research also indicates that the control of intracellular M. tuberculosis by macrophages may be superior when bacilli are ingested in the form of neutrophil apoptotic bodies. The present report indicates that the survival of M. tuberculosis in whole blood cultures of TB-naïve donors is reduced compared to that in broth culture and to values reported for isolated macrophages (6). The substantial reduction in viability during the first 24 h of whole blood culture is consistent with the effects of neutrophils or other short-lived cells. Further studies to examine the role of neutrophils in host defenses against M. tuberculosis in whole blood culture are therefore warranted. However, even during the later phase of culture, when neutrophils presumably are no longer viable, mycobacterial growth was restricted to less than half the rate observed in broth culture. This indicates that other mechanisms involved in innate defenses against M. tuberculosis are expressed as well. Our finding that certain mycobacterial isolates seem preferentially able to withstand host defenses during the first or subsequent periods of culture is consistent with the expression of distinct effector mechanisms during these intervals.
This study is also notable for its characterization of recent clinical isolates after minimal in vitro passage. This is in contrast to the study of Li et al., for example, which examined virulence using isolates from a strain collection (6). None of the clinical isolates in that study showed growth which was statistically superior to that of H37Rv, whereas three showed significantly reduced growth. In contrast, we found four isolates that were significantly more virulent than H37Rv and only one that was less so. The isolates in the present study were passaged in vitro only twice prior to their introduction into cell culture to avoid the attenuation we had previously observed with isolate MP-28. When studied in 2000, that isolate showed nearly 1-log intracellular growth in whole blood culture (1, 16); by the following year, it had become markedly attenuated (19). Similar attenuation may have occurred with CDC1551, an outbreak-associated isolate that was highly virulent when first studied in mice in 1998 (13). The present study supports the findings of Li et al. that CDC1551 currently does not differ significantly from H37Rv in its capacity for intracellular growth. Selection for mycobacterial substrains with enhanced growth in broth at the expense of their capacity for intracellular growth may be most likely to occur shortly after a clinical isolate is first introduced into broth culture. This phenomenon may complicate future studies of the relationship between genotype and phenotype for recent clinical TB isolates.
This study is also significant for its observation that an increased expression of IFN-α may be a pathogenic mechanism in human tuberculosis. Previous studies have examined this possibility in mice, in which IFN-α, like IL-10, down-regulates protective IFN-γ responses and may interfere with granuloma formation (8). Further in vitro and population-based studies will be required to examine the relationship of IFN-α expression to human TB risk.
Lastly, this study revealed the shortcomings of the whole blood model. One question that remains unanswered is the precise cellular localization of M. tuberculosis bacilli during the initial phase of whole blood culture. The model was originally developed by using the smallest inoculum (as low as 103 CFU) that would still permit an accurate assessment of growth or killing (16). This approach favors phagocytosis but hinders the detection of ingested mycobacteria by light microscopy. Experiments in which green fluorescent protein-labeled bacilli are detected by flow cytometry and confocal microscopy may be required to definitively determine the proportion of mycobacteria that are initially ingested by neutrophils and to elucidate the fate of these bacilli.
We also observed that small inocula provided submaximal stimuli for cytokine expression compared to lipopolysaccharide or phytohemagglutinin A (15). Unlike mycobacterial killing, which is a proportional process, cytokine induction is highly stimulus dependent. Future studies of differential cytokine induction will require that strains be tested across a range of inoculum sizes to more clearly elucidate the dose-response relationship.
Acknowledgments
This work was partially supported by the Fogarty International Central/Eastern European HIV Research Program grant 3 D43 TW00233 (Jack DeHovitz, principal investigator).
Editor: F. C. Fang
REFERENCES
- 1.Cheon, S. H., B. Kampmann, A. G. Hise, M. Phillips, H. Y. Song, K. Landen, Q. Li, R. Larkin, J. J. Ellner, R. F. Silver, D. F. Hoft, and R. S. Wallis. 2002. Bactericidal activity in whole blood as a potential surrogate marker of immunity after vaccination against tuberculosis. Clin. Diagn. Lab. Immunol. 9:901-907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Dormans, J., M. Burger, D. Aguilar, R. Hernandez-Pando, K. Kremer, P. Roholl, S. M. Arend, and D. Van Soolingen. 2004. Correlation of virulence, lung pathology, bacterial load and delayed type hypersensitivity responses after infection with different Mycobacterium tuberculosis genotypes in a BALB/c mouse model. Clin. Exp. Immunol. 137:460-468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Hoal-van Helden, E. G., L. A. Stanton, R. Warren, M. Richardson, and P. D. van Helden. 2001. Diversity of in vitro cytokine responses by human macrophages to infection by Mycobacterium tuberculosis strains. Cell Biol. Int. 25:83-90. [DOI] [PubMed] [Google Scholar]
- 4.Hoft, D. F., S. Worku, B. Kampmann, C. C. Whalen, J. J. Ellner, C. S. Hirsch, R. B. Brown, R. Larkin, Q. Li, H. Yun, and R. F. Silver. 2002. Investigation of the relationships between immune-mediated inhibition of mycobacterial growth and other potential surrogate markers of protective Mycobacterium tuberculosis immunity. J. Infect. Dis. 186:1448-1457. [DOI] [PubMed] [Google Scholar]
- 5.Kisich, K. O., M. Higgins, G. Diamond, and L. Heifets. 2002. Tumor necrosis factor alpha stimulates killing of Mycobacterium tuberculosis by human neutrophils. Infect. Immun. 70:4591-4599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Li, Q., C. C. Whalen, J. M. Albert, R. Larkin, L. Zukowski, M. D. Cave, and R. F. Silver. 2002. Differences in rate and variability of intracellular growth of a panel of Mycobacterium tuberculosis clinical isolates within a human monocyte model. Infect. Immun. 70:6489-6493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Manca, C., M. B. Reed, S. Freeman, B. Mathema, B. Kreiswirth, C. E. Barry III, and G. Kaplan. 2004. Differential monocyte activation underlies strain-specific Mycobacterium tuberculosis pathogenesis. Infect. Immun. 72:5511-5514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Manca, C., L. Tsenova, A. Bergtold, S. Freeman, M. Tovey, J. M. Musser, C. E. Barry III, V. H. Freedman, and G. Kaplan. 2001. Virulence of a Mycobacterium tuberculosis clinical isolate in mice is determined by failure to induce Th1 type immunity and is associated with induction of IFN-alpha/beta. Proc. Natl. Acad. Sci. USA 98:5752-5757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Milan, S. J., K. A. Hauge, N. E. Kurepina, K. H. Lofy, S. V. Goldberg, M. Narita, C. M. Nolan, P. D. McElroy, B. N. Kreiswirth, and G. A. Cangelosi. 2004. Expanded geographical distribution of the N family of Mycobacterium tuberculosis strains within the United States. J. Clin. Microbiol. 42:1064-1068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Munsiff, S. S., B. Nivin, G. Sacajiu, B. Mathema, P. Bifani, and B. N. Kreiswirth. 2003. Persistence of a highly resistant strain of tuberculosis in New York City during 1990-1999. J. Infect. Dis. 188:356-363. [DOI] [PubMed] [Google Scholar]
- 11.Reed, M. B., P. Domenech, C. Manca, H. Su, A. K. Barczak, B. N. Kreiswirth, G. Kaplan, and C. E. Barry III. 2004. A glycolipid of hypervirulent tuberculosis strains that inhibits the innate immune response. Nature 431:84-87. [DOI] [PubMed] [Google Scholar]
- 12.Theus, S. A., M. D. Cave, and K. D. Eisenach. 2004. Activated THP-1 cells: an attractive model for the assessment of intracellular growth rates of Mycobacterium tuberculosis isolates. Infect. Immun. 72:1169-1173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Valway, S. E., M. P. Sanchez, T. F. Shinnick, I. Orme, T. Agerton, D. Hoy, J. S. Jones, H. Westmoreland, and I. M. Onorato. 1998. An outbreak involving extensive transmission of a virulent strain of Mycobacterium tuberculosis. N. Engl. J. Med. 338:633-639. [DOI] [PubMed] [Google Scholar]
- 14.Van Soolingen, D., P. E. de Haas, P. W. Hermans, P. M. Groenen, and J. D. Van Embden. 1993. Comparison of various repetitive DNA elements as genetic markers for strain differentiation and epidemiology of Mycobacterium tuberculosis. J. Clin. Microbiol. 31:1987-1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Wallis, R. S., H. M. Lederman, J. Spritzler, J. L. Devers, D. Georges, A. Weinberg, S. Stehn, and M. M. Lederman. 1998. Measurement of induced cytokines in AIDS clinical trials using whole blood: a preliminary report. Clin. Diagn. Lab. Immunol. 5:556-560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Wallis, R. S., M. Palaci, S. Vinhas, A. G. Hise, F. C. Ribeiro, K. Landen, S. H. Cheon, H. Y. Song, M. Phillips, R. Dietze, and J. J. Ellner. 2001. A whole blood bactericidal assay for tuberculosis. J. Infect. Dis. 183:1300-1303. [DOI] [PubMed] [Google Scholar]
- 17.Wallis, R. S., S. Patil, S. H. Cheon, K. Edmonds, M. Phillips, M. D. Perkins, M. Joloba, A. Namale, J. L. Johnson, L. Teixeira, R. Dietze, S. Siddiqi, R. D. Mugerwa, K. Eisenach, and J. J. Ellner. 1999. Drug tolerance in Mycobacterium tuberculosis. Antimicrob. Agents Chemother. 43:2600-2606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Wallis, R. S., M. Perkins, M. Phillips, M. Joloba, B. Demchuk, A. Namale, J. L. Johnson, D. Williams, K. Wolski, L. Teixeira, R. Dietze, R. D. Mugerwa, K. D. Eisenach, and J. J. Ellner. 1998. Induction of the antigen 85 complex of M. tuberculosis in sputum: a determinant of outcome in pulmonary tuberculosis. J. Infect. Dis. 178:1115-1121. [DOI] [PubMed] [Google Scholar]
- 19.Wallis, R. S., S. A. Vinhas, J. L. Johnson, F. C. Ribeiro, M. Palaci, R. L. Peres, R. T. Sa, R. Dietze, A. Chiunda, K. Eisenach, and J. J. Ellner. 2003. Whole blood bactericidal activity during treatment of pulmonary tuberculosis. J. Infect. Dis. 187:270-278. [DOI] [PubMed] [Google Scholar]