Abstract
Streptococcus gordonii shows promise as a live mucosal vaccine vector for immunization against respiratory pathogens. In preparation for clinical trials to evaluate S. gordonii engineered to express group A streptococcal M protein antigens, we characterized the responses of 150 healthy volunteers to combined nasal and oral inoculation with approximately 1.5 × 109 CFU of SP204(1-1), an S. gordonii strain not bearing vaccine antigens. SP204(1-1) was selected for resistance to streptomycin and 5-fluoro-2-deoxyuridine to distinguish it from indigenous flora. In two antibiotic treatment studies, we performed serial culturing of nose, mouth, and saliva samples from 120 subjects treated with azithromycin beginning 5 days after inoculation to determine whether SP204(1-1) could be rapidly eliminated should safety concerns arise. A natural history study was performed to assess the time until spontaneous eradication in the remaining 30 subjects, who did not receive the antibiotic and who were monitored with repeated culturing for 14 weeks after inoculation. SP204(1-1) was generally well tolerated. Symptoms reported most often within 5 days of inoculation were nasal congestion (36%), headache (30%), and sore throat (19%). The strain was detected by culturing in 98% of subjects. A single dose of azithromycin eliminated colonization in 95% of subjects; all subjects receiving a 5-day course of an antibiotic showed clearance by day 11. Without the antibiotic, 82% of subjects showed spontaneous eradication of the implanted strain within 7 days, and all showed clearance by 35 days. The results of these clinical trials provide encouragement that the use of S. gordonii as a live mucosal vaccine vector is a feasible strategy.
Streptococcus gordonii has many features considered attractive for a live bacterial mucosal vaccine vector. This human oral commensal circumvents some of the safety and environmental issues posed by the common use of attenuated pathogens as vectors. The ability of S. gordonii to persistently colonize the oral cavity provides a potential means for achieving vigorous stimulation of the mucosal immune system. S. gordonii is easily manipulated genetically to produce a fusion construct that is chromosomally integrated to enhance stability (24). Unlike other live bacterial systems, in which the foreign antigen is retained in the cytoplasm, translocated to the periplasm or, in some cases, secreted, the heterologous antigen can be anchored to the cell wall. Anchoring can be accomplished by using as the attachment motif either the M6 protein from S. pyogenes (33) or the P1 antigen from S. mutans (19) and fusing the antigen to peptidoglycan, a natural adjuvant (24). Proof of this principle has been demonstrated in animal models in which local and/or systemic antibody responses have been generated by inoculating mucosal sites with S. gordonii engineered to surface express a variety of viral and bacterial antigens, such as the E7 protein of human papillomavirus type 16 (27), the pertussis toxin S1 fragment (18), the B subunit of Escherichia coli heat-labile toxin (32), Porphyromonas gingivalis FimA polypeptides (33), and tetanus toxin fragment C (23).
S. gordonii has also been evaluated as a mucosal vector for delivering group A streptococcal vaccine antigens. The candidate vaccine expresses the C repeat region of the group A streptococcal M protein (the primary virulence factor for this organism), which is conserved among group A streptococcal strains associated with rheumatic fever (8). Combined intranasal and oral vaccination of mice induces both serum and mucosal antibodies to the recombinant antigens (2, 29). The immune responses can be augmented by coexpressing cytokines, such as gamma interferon (3).
In preparation for clinical trials evaluating a recombinant strain expressing group A streptococcal or other extrinsic antigens, we assessed the clinical and microbiological responses to experimental implantation with a strain of S. gordonii that does not contain foreign antigens. Given the propensity of some commensal strains to persist even in the presence of antibiotics (34), we aimed to demonstrate that the vector strain could be rapidly eliminated either spontaneously or with an antibiotic should safety issues arise when a recombinant vector is administered in future clinical trials. Furthermore, we anticipated that phase 1 trials of an engineered S. gordonii strain expressing group A streptococcal antigens will be conducted in an inpatient setting under isolation until safety has been established; as a prerequisite to such studies, it was important to demonstrate that colonization could be eliminated to prevent transmission of the experimental strain to the community.
MATERIALS AND METHODS
Subjects.
Healthy volunteers 18 to 40 years old were recruited from the University of Maryland, College Park. Informed consent was obtained according to the guidelines of the institutional review boards of the University of Maryland, Baltimore, and the University of Maryland, College Park. Specific exclusion criteria included a history of significant heart disease; antibiotic use during the 14 days before inoculation; allergy to macrolide antibiotics; and extensive tooth loss, severe gum disease, or visible caries on oral examination. To document comprehension of the protocol, subjects were required to pass (70% correct) a written examination containing approximately 25 multiple-choice and true-false questions about the purpose, procedures, risks, and benefits of the trial.
Antibiotic treatment studies.
Two antibiotic treatment studies, each involving 60 volunteers, were conducted to determine whether experimental S. gordonii strain SP204(1-1) could be implanted in the nose and mouth of volunteers and then eradicated with an antibiotic (Table 1). Beginning 5 days after inoculation, volunteers took oral azithromycin (500 mg on day 5 followed by 250 mg daily on days 6 to 9) under the direct observation of the study team. In the first antibiotic treatment study, samples were collected from each volunteer to evaluate colonization with the test strain before implantation; on days 1, 3, and 5 after implantation but before antibiotic administration; and on days 7, 14, 21, 28, 35, and 42 after implantation and antibiotic administration. The second antibiotic treatment study was designed to derive a more precise estimate of the time until eradication during postimplantation week 2 to provide assurance that the antibiotic rapidly eliminated colonization. The design of the two studies differed in the days of sample collection, which occurred in the second study before implantation; on days 1, 3, and 5 after implantation but before antibiotic administration; and on days 6 to 14 and 21 after implantation and antibiotic administration.
TABLE 1.
Study design
| Study | No. of subjects | Antibiotic administration | Days post implantation on which samples were obtained for culturinga |
|---|---|---|---|
| Antibiotic treatment | |||
| First | 60 | Days 5-9 | 1, 3, 5, 7, 14, 21, 28, 35, and 42 |
| Second | 60 | Days 5-9 | 1, 3, 5-14, and 21 |
| Natural history | 30 | No | 1, 3, 5, 7, 9, 11, 14, 21, 28, 35, 42, 70, and 98 |
Samples were obtained from the nares, mouth, and saliva.
Natural history study.
A natural history study was designed to delineate the time until spontaneous eradication of implanted strain SP204(1-1) in 30 subjects who did not receive an antibiotic and who were monitored for 14 weeks after inoculation. Samples were obtained from each volunteer to evaluate colonization with SP204(1-1) on days 0, 1, 3, 5, 7, 9, 11, 14, 21, 28, 3, 42, 70, and 98 postimplantation (Table 1).
Bacterial strain.
Experimental S. gordonii strain SP204(1-1) does not contain foreign antigens. SP204(1-1) expresses two biological markers to distinguish it from wild-type S. gordonii: resistance to streptomycin (Smr) by virtue of a point mutation in the S12 ribosomal protein gene and resistance to pyrimidine analog 5-fluoro-2-deoxyuridine (FudRr) by virtue of a point mutation in the thymidine kinase gene (10). SP204(1-1) was derived from strain GP204, a spontaneous Smr mutant of S. gordonii strain V288 (ATCC 35105) kindly provided by Gianni Pozzi (28). Selection of FudRr mutants was performed as described previously (10). Briefly, GP204 was plated on sheep blood agar containing 1 to 10 μg of FUdR/ml in the presence of uridine (12.5 μg/ml) and thymidine (2 μg/ml). Colonies which grew at the highest concentrations of FUdR showed both FUdRr and Smr. The point mutation in the thymidine kinase gene was verified by sequencing of the PCR-derived thymidine kinase gene fragment. Spontaneous FUdRr mutants were generated at a rate of 10−6.
Inoculum preparation.
Aliquots of a seed lot of S. gordonii strain SP204(1-1), manufactured under good manufacturing practices by Tektagen, Inc., Malvern, Pa. (lot 37225), were provided by SIGA Technologies, Inc., and stored at −70°C. All chemicals were obtained from Sigma Chemical Company, St. Louis, Mo., unless otherwise stated. In the Molecular Diagnostics and Microbiology Laboratory at the Center for Vaccine Development, a subculture was streaked onto Trypticase soy agar plates containing streptomycin at 1 mg/ml, FUdR at 50 μg/ml, and uracil at 100 μg/ml. After incubation at 37°C for 48 h in a sealed plastic bag and confirmation of uniform S. gordonii colony morphology, 50 colonies were inoculated into Trypticase soy broth (Becton Dickinson, Sparks, Md.) containing uracil at 20 μg/ml, 0.5% d-glucose, streptomycin at 1 mg/ml, and FUdR at 50 μg/ml (TSB with additives). After 24 h of incubation at 37°C without shaking, an aliquot was transferred to 5 liters of prewarmed TSB with additives and incubated at 37°C for 5 to 6 h until an optical density at 650 nm of 0.85 (18 mm) (late log phase) was attained. The suspension was cooled in an ice water bath for 20 min, divided into aliquots, and washed and centrifuged three times with sterile phosphate-buffered saline (PBS; pH 7.4). The supernatants were tested for residual antibiotic and FUdR. The final pellets were resuspended in PBS and combined into a single bottle. This suspension was concentrated to achieve ca. 5 × 109 CFU/ml (a 10-fold concentration with an optical density at 650 nm of 1.5), divided into 3.2-ml aliquots, and frozen at −70°C. Identity, purity, and colony counts were verified with five randomly selected aliquots.
One week before implantation, the appropriate number of vials for each cohort was identified for use in implantation. Without thawing of the vials, a subculture was made to verify the identity, purity, and viability of the inoculum. On the day of implantation, the selected vials of SP204(1-1) were thawed at 37°C for 20 min and vortexed vigorously, and the contents were pooled. Based on colony counts determined for a frozen vial 3 days before implantation, the bacterial suspension was diluted to provide a final concentration of 5 × 108 CFU/ml. The suspension was delivered on ice to the clinical site and used within 6 h of thawing. The accuracy of the inoculum size was verified by calculating the mean for duplicate pre- and postimplantation colony counts.
Inoculation.
To ensure a uniform inoculum for each volunteer, the tube containing the pooled bacterial cells was inverted 10 times just before implantation. An aliquot containing ca. 1.5 × 109 CFU of SP204(1-1) in 3 ml of PBS was placed into a sterile screw-top tube. Each volunteer received a single inoculation by instilling 0.5 ml of the aliquot into each nostril with a tuberculin syringe and then placing the remaining 2 ml on the anterior surface of the tongue. The volunteer swished the test strain solution over the surface of the teeth for 90 s, gargled for 30 s, and then swallowed. Volunteers did not eat, drink, or blow their nose for 90 min pre- and postinoculation. They were instructed not to brush their teeth for 12 h or use mouthwash for 5 days after inoculation and to avoid professional dental cleaning for the duration of the study. The day of inoculation was designated day 0.
Volunteer assessment.
Volunteers were observed at the study site for 30 min postinoculation. A standardized clinical evaluation was performed 30 min postinoculation by the study staff and then for the next 10 days by using a diary form that the volunteers completed. Symptoms solicited included malaise and anorexia, which were not graded for severity, and headache, sore throat, nasal congestion, and nasal burning, which the volunteer graded as mild (easily tolerated), moderate (bothersome but did not interfere with normal activity), or severe (interfered with normal activity). Volunteers recorded their oral temperature each evening in the diary. Fever was defined as an oral temperature of at least 37.8°C. At each visit for specimen collection after day 10, volunteers were asked whether they had experienced any symptoms since the last study visit, made any medical visits, or took any medication.
Detection of colonization of volunteers with SP204(1-1).
Five specimens were collected at each time point to evaluate colonization (Table 1): (i) unstimulated saliva; (ii) a swab of the mouth (buccal surface of gums, dorsal tongue, and tonsillar fossa); (iii) a swab of the teeth (buccal surface); (iv) a swab of the right and left nares; and (v) an oral specimen consisting of 2 ml of broth medium (see below) which volunteers swished around their oral cavities, gargled, and expectorated into a sterile cup. The expectorated oral specimen was added to 2 ml of broth medium containing streptomycin at 2 mg/ml, FUdR at 100 μg/ml, amphotericin at 5 μg/ml, and uracil at 40 μg/ml. These specimens were maintained at room temperature for up to 5 h during transport to the laboratory. Saliva was collected in a sterile cup and maintained on a cool pack for up to 5 h before plating. A blood sample for culturing was drawn from any patient who developed a fever of at least 38.9°C during week 1 postimplantation.
Swabs were rayon tipped and were processed as follows. Swab samples of the mouth were inoculated into the expectorated oral specimen; swab samples of the teeth were also inoculated into the expectorated specimen in the second antibiotic treatment study and the natural history study. Swabs of teeth in the first antibiotic treatment study and of nares in both studies were inoculated directly into broth medium containing streptomycin at 1 mg/ml, FUdR at 50 μg/ml, amphotericin at 2.5 μg/ml, and uracil at 20 μg/ml. The broth medium consisted of brain heart infusion (Difco, Becton Dickinson) in the first antibiotic treatment study. Thereafter, TSB was substituted for brain heart infusion (to provide a more palatable gargle) after demonstration that the exponential growth of SP204(1-1) was comparable in each medium.
Vials containing swab samples were incubated at 37°C for 48 h, and then 100 μl was spread directly onto 5% sheep blood agar with additives (Becton Dickinson). Plates were incubated at 37°C for 48 h. With a standard biochemical strip (Rapid STREP; API BioMérieux, Durham, N.C.) for identification of S. gordonii, one colony at a time was tested until a positive identification (S. sanguis) was obtained.
Statistical methods.
The sample size for each antibiotic treatment study was derived to demonstrate that, on the day on which the strain was deemed to be eradicated from all subjects, there would be a 4.6% one-sided probability that the true rate of eradication failure was as low as 5% (by binomial expansion). In other words, if the strain were eradicated from all subjects by the end of the study, then the lower one-sided 95% confidence limit of the true eradication rate would be 95%. Pooling the results of the two antibiotic treatment studies (a total of 120 subjects) would further reduce the estimate of this probability to 2.5%. The natural history study was designed to provide a qualitative assessment of the time until eradication and was not powered for precision. The incidences of loss of energy, loss of appetite, fever, headache, sore throat, nasal congestion, and nasal burning on days 0 to 4 postimplantation and on days 5 to 9 postimplantation were compared by using McNemar's test. Shedding curves for antibiotic treatment studies and the natural history study were compared by using logistic regression analysis; the dependent variable was shedding on days 5 to 28 (i.e., beginning on the day on which the antibiotic was administered in the antibiotic treatment studies), and the independent variables were day postimplantation, study, and their interaction).
RESULTS
Clinical tolerance.
The implanted bacterial inoculum was generally well tolerated. Only one volunteer reported symptoms (nasal congestion) during the standardized interview conducted 30 min postinoculation. Data from the two antibiotic treatment studies, which involved a total of 120 subjects, were pooled for analysis of the symptoms solicited in the diary. A minority of subjects in the antibiotic treatment studies reported ungraded solicited symptoms in the diary (Table 2). The most common graded solicited symptoms reported before the antibiotic was initiated (days 0 to 4) were headache (28%), nasal congestion (28%), and sore throat (22%) (Table 3). The clinical features observed among the 30 subjects participating in the natural history study were similar in frequency and severity to those reported in the antibiotic treatment study although nasal congestion occurred more often (50% of subjects during days 0 to 4) (Tables 4 and 5).
TABLE 2.
Ungraded signs and symptoms after implantation of S. gordonii strain SP204(1-1) in the nose and mouth of 120 subjects treated with azithromycin on days 5 through 9 to eradicate the implanted strain in the antibiotic treatment studies
| Ungraded sign or symptom | No. (%) of subjects with the indicated feature on the following days postinoculation:
|
|
|---|---|---|
| 0-4 | 5-9 | |
| Loss of energy | 15 (13) | 16 (10) |
| Loss of appetite | 7 (6) | 7 (6) |
| Fever | 1 (<0.01)a | 1 (<0.01)a |
The volunteer with a fever had a peak temperature of 38.5°C.
TABLE 3.
Graded signs and symptoms after implantation with S. gordonii strain SP204(1-1) in the nose and mouth of 120 subjects treated with azithromycin on days 5 through 9 to eradicate the implanted strain in the antibiotic treatment studies
| Graded sign or symptom | No. (%) of subjects with the indicated graded feature on the following days postinoculation:
|
|||||
|---|---|---|---|---|---|---|
| 0-4
|
5-9
|
|||||
| Mild | Moderate | Severe | Mild | Moderate | Severe | |
| Headache | 25 (20) | 8 (7) | 1 (<0.01) | 14 (10) | 2 (2) | 0 (0) |
| Sore throat | 20 (17) | 5 (4) | 1 (<0.01) | 10 (8) | 8 (7) | 0 (0) |
| Nasal congestion | 28 (23) | 4 (3) | 1 (<0.01) | 17 (14) | 6 (5) | 1 (<0.01) |
| Nasal burning | 2 (2) | 0 (0) | 0 (0) | 2 (2) | 0 (0) | 0 (0) |
TABLE 4.
Ungraded signs and symptoms after implantation with S. gordonii strain SP204(1-1) in the nose and mouth of 30 subjects not treated with antibiotic to eradicate the implanted strain in the natural history study
| Ungraded sign or symptom | No. (%) of subjects with the indicated feature on the following days postinoculation:
|
|
|---|---|---|
| 0-4 | 5-9 | |
| Loss of energy | 5 (17) | 16 (10) |
| Loss of appetite | 1 (3) | 7 (6) |
| Fever | 2 (7)a | 1 (<0.01)b |
The two volunteers with fevers had peak temperatures of 37.8 and 38.2°C.
The volunteer with a fever had a peak temperature of 38.5°C.
TABLE 5.
Graded signs and symptoms after implantation with S. gordonii strain SP204(1-1) in the nose and mouth of 30 subjects not treated with antibiotic to eradicate the implanted strain in the natural history study
| Graded sign or symptom | No. (%) of subjects with the indicated graded feature on the following days postinoculation:
|
|||||
|---|---|---|---|---|---|---|
| 0-4
|
5-9
|
|||||
| Mild | Moderate | Severe | Mild | Moderate | Severe | |
| Headache | 6 (20) | 3 (10) | 1 (3) | 6 (20) | 6 (20) | 0 (0) |
| Sore throat | 4 (13) | 0 (0) | 1 (3) | 2 (7) | 1 (3) | 1 (3) |
| Nasal congestion | 12 (40) | 3 (10) | 0 (0) | 6 (20) | 3 (10) | 1 (3) |
| Nasal burning | 2 (7) | 0 (0) | 0 (0) | 1 (3) | 0 (0) | 0 (0) |
Volunteers graded most symptoms as mild. Four subjects experienced symptoms graded as severe. One participant in the antibiotic treatment studies developed intermittent fevers for the first 3 days postinoculation (maximum temperature of 38.5°C), a cough that lingered for 13 days, and a stuffy nose, sore throat, and headache that he rated as severe. Three subjects in the natural history study reported severe symptoms. One subject developed a severe headache for 6 h and a temperature of 37.8°C on day 3, associated on days 3 to 6 with a sore throat that was graded as severe on day 3. A second subject experienced a severe sore throat and nasal congestion, with associated cough, malaise, and headache, on days 5 to 13. The final subject experienced 1 day of fever (maximum temperature of 38.2°C on the evening of inoculation), associated with mild nasal congestion. The first three subjects had negative cultures for SP204(1-1) after day 3, while the final subject shed the implanted strain for 11 days.
In the absence of placebo, it cannot be determined whether these signs and symptoms were related to inoculation. An exploratory analysis was performed for volunteers in the antibiotic treatment studies to compare the frequencies of signs and symptoms that began between days 0 and 4 (which would be regarded as possibly related to inoculation) and between days 5 and 9 (which would be regarded as unlikely to be inoculum related, since azithromycin was initiated) by using McNemar's test. (Graded symptoms were first dichotomized as present or absent.) Of the eight signs and symptoms recorded, only headache began significantly more often between days 0 and 4 than between days 5 and 9 (P = 0.003).
Microbiological response.
FUdRr Smr S. gordonii was not detected in any volunteer before implantation. After implantation, SP204(1-1) was detected in the nose and/or mouth of 147 subjects (98%) for at least 1 day.
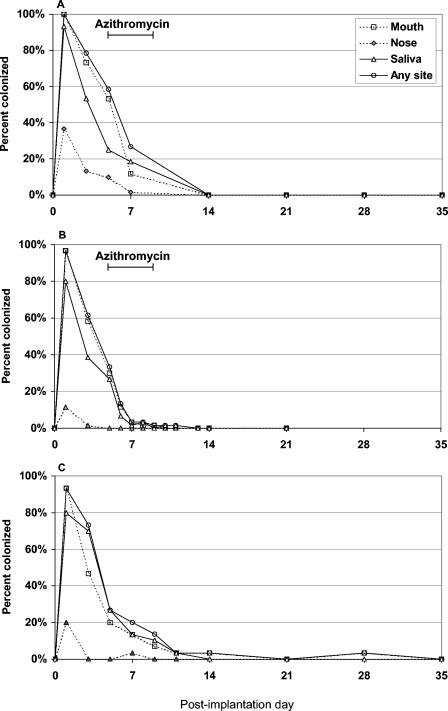
The number of subjects in each study in whom the implanted strain was detected by culturing according to the day postinoculation on which sampling was performed is shown in Fig. 1. In the first antibiotic treatment study, SP204(1-1) was last detected on day 7; however, no data were collected again until day 14 (Fig. 1A). In the second antibiotic treatment study, all but three subjects had negative cultures after day 6 (following only a single dose of the antibiotic) (Fig. 1B). The remaining three subjects shed the implanted strain erratically between days 7 and 11. Eradication thus was achieved in 100% of subjects who were treated with azithromycin within 11 days of implantation.
FIG. 1.
Percentages of subjects with positive cultures for experimental S. gordonii strain SP204(1-1) during 35 days postimplantation according to sampling site. (A and B) In two separate studies (n = 60 in each), subjects were inoculated with SP204(1-1) in the nose and mouth and then treated with oral azithromycin on days 5 to 9 (A and B). These studies differed with respect to the sampling schedule, as shown. (C) A third study was done to assess the time until spontaneous eradication of SP204(1-1) in 30 subjects who did not receive the antibiotic. No positive cultures were detected after day 28.
All participants in the natural history study spontaneously cleared the strain by day 35 without the need for an antibiotic (Fig. 1C). In fact, 23 of the 28 colonized subjects (82%) had negative cultures after day 7. The remaining subjects stopped shedding the implanted strain on day 9 (n = 2), day 11 (n = 1), day 14 (n = 1), and day 28 (n = 1). The last subject had interim negative cultures from days 9 to 21.
The mouth was the most common site from which SP204(1-1) was cultivated, yielding positive results for 147 of the 150 subjects examined (98%). In comparison, saliva cultures were positive for 134 subjects (89%), and nasal swab samples were positive for 37 subjects (25%). As shown in Fig. 1, colonization of the nares was both less frequent and less enduring than that of other sites.
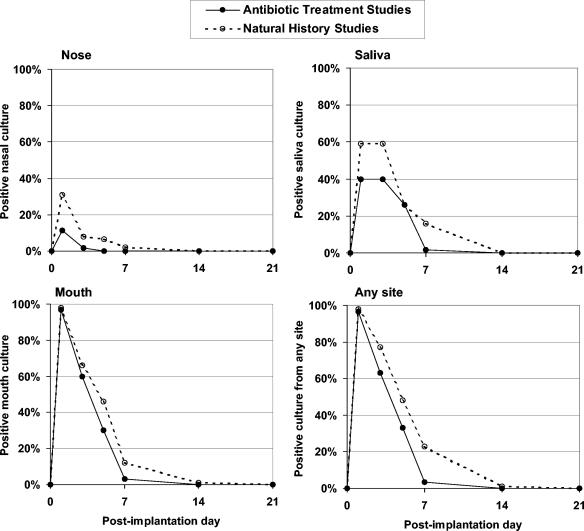
To assess whether the antibiotic had a significant impact on eradication of the implanted strain, we compared the proportions of subjects who had positive cultures for SP204(1-1) at each site according to day postvaccination in the two antibiotic treatment studies (combined) and the natural history study. As shown in Fig. 2, subjects in the antibiotic treatment studies were significantly less likely to shed SP204(1-1) from the mouth (P < 0.001) or from any site (nares, mouth, and saliva combined) (P < 0.001) after the antibiotic was initiated on day 5, as measured by logistic regression analysis of the interaction between study and time.
FIG. 2.
Percentages of subjects with positive cultures for experimental S. gordonii strain SP204(1-1) during 21 days postimplantation according to sampling site and whether or not the subjects received an antibiotic. Subjects in the two antibiotic treatment studies (total of 120) received azithromycin on days 5 through 9, whereas subjects in the natural history study (total of 30) did not receive the antibiotic. Subjects in the antibiotic treatment studies were less likely to shed SP204(1-1) after the antibiotic was initiated. This difference reached statistical significance in a logistic regression analysis for subjects with positive cultures from the mouth (P < 0.001) or from any site (nares, mouth, and saliva combined) (P < 0.001), as measured by the interaction between study and time. Data are from time points shared by either of the antibiotic treatment studies and the natural history study, but logistic regression analysis compared only points that were shared by all three studies.
DISCUSSION
Our experience with 150 volunteers demonstrates that an experimental strain of S. gordonii bearing Smr and FUdRr can be implanted into the nose and mouth of healthy adults and detected by culturing for at least 1 day in nearly all subjects (98%). The presence of the strain for 3 or more days in the mouth of most subjects (ca. 60%) provides evidence for local replication. Nonetheless, colonization was self-limited and generally short term; the majority (82%) of subjects who did not receive antibiotic treatment spontaneously cleared the implanted strain within 1 week, and all 30 subjects did so within 35 days. The transient nature of colonization with SP204(1-1) can be considered a desirable feature for a group A streptococcal vaccine strain. It minimizes the risk of transmission to susceptible contacts and assuages concerns that persistent low-level antigenic stimulation might lead to immunologic tolerance or induce low-avidity antibodies (5), albeit an unlikely phenomenon for a replicating mucosal vaccine strain (7, 25).
We cannot determine with certainty whether inadvertent mutations occurred in SP204(1-1) and affected its ability to establish durable colonization; however, published studies might predict that experimentally implanted S. gordonii would not readily colonize the mouth of a healthy adult. S. gordonii normally joins the oral microbiota following tooth eruption during early childhood. Acquisition and persistence of S. gordonii and other indigenous flora in the oral cavity involve a complex interaction among the bacterium, other resident flora, and the host (14-17, 20, 21, 35). The success of colonization appears to be influenced in part by the ability of commensal streptococci to evade immune surveillance, for example, by becoming embedded in protective habitats such as biofilm (11) and maintaining a high degree of antigenic variability (9, 13). Considerable genetic diversity has been observed among commensal strains and has raised the possibility that colonization actually is maintained by a succession of clones (13). Whether such factors will impede the use of S. gordonii as a live vector will be addressed in future studies.
The microbiological features considered desirable for S. gordonii strains that are targeted for use as vaccine carriers and those intended for the delivery of biologically active molecules may differ. Concerns about transmissibility have led to a more favorable view of the live attenuated vaccine strains that can stimulate the immune system after a limited period of colonization. In contrast, prolonged colonization of an immunologically protected niche may be optimal for the delivery of some bioactive agents. For example, the ability of S. gordonii to achieve enduring gastrointestinal delivery of a human interleukin 1 receptor antagonist has been demonstrated in mice as a potential therapy for acute and chronic inflammatory diseases (31). It is envisioned that similar systems would be capable of delivering directly to the vaginal mucosa microbicidal agents such as cyanovirin-N, a human immunodeficiency virus-inactivating protein (12), or candidacidal molecule H6 (1). Our experience with SP204(1-1) suggests that if protracted delivery of biologically active substances to targeted sites is needed, then a strain of S. gordonii that is a more robust colonizer must be identified, and serial inoculations might be required.
Although this study was not placebo controlled, several observations about clinical tolerance nonetheless can be made. For one, the inoculation was generally well tolerated, and there was no evidence that unacceptable reactogenicity had occurred. Volunteers considered most of their graded solicited symptoms to be mild. Although three subjects reported severe symptoms, only two subjects (one with a headache and low-grade fever and the other with multiple symptoms suggestive of an upper respiratory tract infection) were shedding SP204(1-1) at the time. Finally, the temporal association that we observed between headache and implantation is noteworthy and warrants careful evaluation in future placebo-controlled studies.
An advantage of constructing a group A streptococcal vaccine that delivers antigens directly to the mucosal surface (the primary site of natural infection) is that antigen-specific secretory immunoglobulin A can be maximally stimulated. Human challenge studies conducted in the 1970s suggested that purified M protein vaccines elicit higher levels of protection against both illness and infection when delivered mucosally (intranasally plus orally) rather than parenterally (6, 26). Nonetheless, a live mucosal group A streptococcal vaccine poses certain safety concerns that stem from the observation that acute rheumatic fever follows ca. 1 to 3% of untreated episodes of group A streptococcal pharyngitis (22, 30, 36). Invoking the experience with wild-type group A streptococcal infection that penicillin therapy is highly efficacious in preventing acute rheumatic fever when initiated within 9 days after the onset of pharyngitis (4), we aimed to demonstrate with a high degree of certainty that SP204(1-1) can be eradicated with alacrity when an antibiotic is initiated 5 days after implantation. We showed that this is indeed the case. When shedding was compared by logistic regression analysis for antibiotic treatment and natural history studies, subjects were significantly less likely to shed the implanted strain once an antibiotic was initiated. After just a single dose of azithromycin, SP204(1-1) was no longer detected in 95% of subjects, and eradication was achieved in 100% of subjects 11 days after implantation. Furthermore, these data suggest that a phase 1 inpatient study in which subjects are treated with azithromycin to eliminate carriage of the vaccine strain before discharge to home is feasible.
Acknowledgments
We thank the volunteers who participated in this study; Fran Rubin, Myron Levine, and James Nataro for helpful suggestions; Gail Lee for assistance in the clinical care of volunteers; JoAnna Becker and Eugenia Harper-Jones for performing recruitment and data collection; and Elisa Sindall for regulatory support.
This work was supported by U.S. Public Health Service contract N01-AI-45251 (to Myron Levine) and AI11822 (to Vincent A. Fischetti).
Editor: J. N. Weiser
REFERENCES
- 1.Beninati, C., M. R. Oggioni, M. Boccanera, M. R. Spinosa, T. Maggi, S. Conti, W. Magliani, F. De Bernardis, G. Teti, A. Cassone, G. Pozzi, and L. Polonelli. 2000. Therapy of mucosal candidiasis by expression of an anti-idiotype in human commensal bacteria. Nat. Biotechnol. 18:1060-1064. [DOI] [PubMed] [Google Scholar]
- 2.Bolken, T. C., C. A. Franke, K. F. Jones, R. H. Bell, R. M. Swanson, D. S. King, V. A. Fischetti, and D. E. Hruby. 2002. Analysis of factors affecting surface expression and immunogenicity of recombinant proteins expressed by gram-positive commensal vectors. Infect. Immun. 70:2487-2491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Byrd, C. M., T. C. Bolken, K. F. Jones, T. K. Warren, A. T. Vella, J. McDonald, D. King, Z. Blackwood, and D. E. Hruby. 2002. Biological consequences of antigen and cytokine co-expression by recombinant Streptococcus gordonii vaccine vectors. Vaccine 20:2197-2205. [DOI] [PubMed] [Google Scholar]
- 4.Catanzaro, F. J., C. A. Stetson, A. J. Morris, R. Chamovitz, J. C. H. Rammelkamp, B. L. Stolzer, and W. D. Perry. 1954. The role of streptococcus in the pathogenesis of rheumatic fever. Am. J. Med. 17:749-756. [DOI] [PubMed] [Google Scholar]
- 5.Cole, M. F., S. P. Fitzsimmons, M. J. Sheridan, and Y. Xu. 1995. Humoral immunity to commensal oral bacteria: quantitation, specificity and avidity of serum IgG and IgM antibodies reactive with Actinobacillus actinomycetemcomitans in children. Microbiol. Immunol. 39:591-598. [DOI] [PubMed] [Google Scholar]
- 6.D'Alessandri, R., G. Plotkin, R. M. Kluge, M. K. Wittner, E. N. Fox, A. Dorfman, and R. H. Waldman. 1978. Protective studies with group A streptococcal M protein vaccine. III. Challenge of volunteers after systemic or intranasal immunization with Type 3 or Type 12 group A Streptococcus. J. Infect. Dis. 138:712-718. [DOI] [PubMed] [Google Scholar]
- 7.Duchmann, R., I. Kaiser, E. Hermann, W. Mayet, K. Ewe, and K. H. Meyer zum Buschenfelde. 1995. Tolerance exists toward resident intestinal flora but is broken in active inflammatory bowel disease (IBD). Clin. Exp. Immunol. 102:448-455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Fischetti, V. A. 1991. Streptococcal M protein. Sci. Am. 264:58-65. [DOI] [PubMed] [Google Scholar]
- 9.Fitzsimmons, S., M. Evans, C. Pearce, M. J. Sheridan, R. Wientzen, G. Bowden, and M. F. Cole. 1996. Clonal diversity of Streptococcus mitis biovar 1 isolates from the oral cavity of human neonates. Clin. Diagn. Lab. Immunol. 3:517-522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Franke, C. A., T. M. Bolman, S. A. Ottum, K. F. Jones, and D. E. Hruby. 2000. Streptococcus gordonii strains resistant to fluorodeoxyuridine contain mutations in the thymidine kinase gene and are deficient in thymidine kinase activity. Antimicrob. Agents Chemother. 44:787-789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Gilmore, K. S., P. Srinivas, D. R. Akins, K. L. Hatter, and M. S. Gilmore. 2003. Growth, development, and gene expression in a persistent Streptococcus gordonii biofilm. Infect. Immun. 71:4759-4766. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Giomarelli, B., R. Provvedi, F. Meacci, T. Maggi, D. Medaglini, G. Pozzi, T. Mori, J. B. McMahon, R. Gardella, and M. R. Boyd. 2002. The microbicide cyanovirin-N expressed on the surface of commensal bacterium Streptococcus gordonii captures HIV-1. AIDS 16:1351-1356. [DOI] [PubMed] [Google Scholar]
- 13.Hohwy, J., J. Reinholdt, and M. Kilian. 2001. Population dynamics of Streptococcus mitis in its natural habitat. Infect. Immun. 69:6055-6063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kilian, M., J. Reinholdt, B. Nyvad, E. V. Frandsen, and L. Mikkelsen. 1989. IgA1 proteases of oral streptococci: ecological aspects. Immunol. Investig. 18:161-170. [DOI] [PubMed] [Google Scholar]
- 15.Kolenbrander, P. E., and J. London. 1993. Adhere today, here tomorrow: oral bacterial adherence. J. Bacteriol. 175:3247-3252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kononen, E., H. Jousimies-Somer, A. Bryk, T. Kilp, and M. Kilian. 2002. Establishment of streptococci in the upper respiratory tract: longitudinal changes in the mouth and nasopharynx up to 2 years of age. J. Med. Microbiol. 51:723-730. [DOI] [PubMed] [Google Scholar]
- 17.Lee, S. F. 1995. Active release of bound antibody by Streptococcus mutans. Infect. Immun. 63:1940-1946. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Lee, S. F., S. A. Halperin, H. Wang, and A. MacArthur. 2002. Oral colonization and immune responses to Streptococcus gordonii expressing a pertussis toxin S1 fragment in mice. FEMS Microbiol. Lett. 208:175-178. [DOI] [PubMed] [Google Scholar]
- 19.Lee, S. F., R. J. March, S. A. Halperin, G. Faulkner, and L. Gao. 1999. Surface expression of a protective recombinant pertussis toxin S1 subunit fragment in Streptococcus gordonii. Infect. Immun. 67:1511-1516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Loach, D. M., H. F. Jenkinson, and G. W. Tannock. 1994. Colonization of the murine oral cavity by Streptococcus gordonii. Infect. Immun. 62:2129-2131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Lucas, V. S., D. Beighton, and G. J. Roberts. 2000. Composition of the oral streptococcal flora in healthy children. J. Dent. 28:45-50. [DOI] [PubMed] [Google Scholar]
- 22.Massell, B. F., L. H. Honikman, and J. Amezcua. 1969. Rheumatic fever following streptococcal vaccination. Report of three cases. JAMA 207:1115-1119. [PubMed] [Google Scholar]
- 23.Medaglini, D., A. Ciabattini, M. R. Spinosa, T. Maggi, H. Marcotte, M. R. Oggioni, and G. Pozzi. 2001. Immunization with recombinant Streptococcus gordonii expressing tetanus toxin fragment C confers protection from lethal challenge in mice. Vaccine 19:1931-1939. [DOI] [PubMed] [Google Scholar]
- 24.Medaglini, D., G. Pozzi, T. P. King, and V. A. Fischetti. 1995. Mucosal and systemic immune responses to a recombinant protein expressed on the surface of the oral commensal bacterium Streptococcus gordonii after oral colonization. Proc. Natl. Acad. Sci. USA 92:6868-6872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ogra, P. L., H. Faden, and R. C. Welliver. 2001. Vaccination strategies for mucosal immune responses. Clin. Microbiol. Rev. 14:430-445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Polly, S. M., R. H. Waldman, P. High, M. K. Wittner, and A. Dorfman. 1975. Protective studies with a group A streptococcal M protein vaccine. II. Challenge of volunteers after local immunization in the upper respiratory tract. J. Infect. Dis. 131:217-224. [DOI] [PubMed] [Google Scholar]
- 27.Pozzi, G., M. Contorni, M. R. Oggioni, R. Manganelli, M. Tommasino, F. Cavalieri, and V. A. Fischetti. 1992. Delivery and expression of a heterologous antigen on the surface of streptococci. Infect. Immun. 60:1902-1907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Pozzi, G., R. A. Musmanno, E. A. Renzoni, M. R. Oggioni, and M. G. Cusi. 1988. Host-vector system for integration of recombinant DNA into chromosomes of transformable and nontransformable streptococci. J. Bacteriol. 170:1969-1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Pozzi, G., M. R. Oggioni, R. Manganelli, and V. A. Fischetti. 1992. Expression of M6 protein gene of Streptococcus pyogenes in Streptococcus gordonii after chromosomal integration and transcriptional fusion. Res. Microbiol. 143:449-457. [DOI] [PubMed] [Google Scholar]
- 30.Rammelkamp, C. H., F. W. Denny, and L. W. Wannamaker. 1952. Studies on epidemiology of rheumatic fever in the armed services, p. 72. In L. Thomas (ed.), Rheumatic fever. A symposium. University of Minnesota, Minneapolis.
- 31.Ricci, S., G. Macchia, P. Ruggiero, T. Maggi, P. Bossu, L. Xu, D. Medaglini, A. Tagliabue, L. Hammarstrom, G. Pozzi, and D. Boraschi. 2003. In vivo mucosal delivery of bioactive human interleukin 1 receptor antagonist produced by Streptococcus gordonii. BMC Biotechnol. 3:15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Ricci, S., D. Medaglini, C. M. Rush, A. Marcello, S. Peppoloni, R. Manganelli, G. Palu, and G. Pozzi. 2000. Immunogenicity of the B monomer of Escherichia coli heat-labile toxin expressed on the surface of Streptococcus gordonii. Infect. Immun. 68:760-766. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Sharma, A., H. Nagata, N. Hamada, H. T. Sojar, D. E. Hruby, H. K. Kuramitsu, and R. J. Genco. 1996. Expression of functional Porphyromonas gingivalis fimbrillin polypeptide domains on the surface of Streptococcus gordonii. Appl. Environ. Microbiol. 62:3933-3938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Svanberg, M., and G. Westergren. 1986. Persistence and spread of the orally-implanted bacterium Streptococcus sanguis between persons. Arch. Oral Biol. 31:1-4. [DOI] [PubMed] [Google Scholar]
- 35.Tappuni, A. R., and S. J. Challacombe. 1993. Distribution and isolation frequency of eight streptococcal species in saliva from predentate and dentate children and adults. J. Dent. Res. 72:31-36. [DOI] [PubMed] [Google Scholar]
- 36.Valkenburg, H. A., M. J. Haverkorn, W. R. Goslings, J. C. Lorrier, C. E. De Moor, and W. R. Maxted. 1971. Streptococcal pharyngitis in the general population. II. The attack rate of rheumatic fever and acute glomerulonephritis in patients. J. Infect. Dis. 124:348-358. [DOI] [PubMed] [Google Scholar]