Abstract
Pneumonic plague, an often-fatal disease for which no vaccine is presently available, results from pulmonary infection by the bacterium Yersinia pestis. The Y. pestis V protein is a promising vaccine candidate, as V protein immunizations confer to mice significant protection against aerosolized Y. pestis. CD4 T cells play central roles during vaccine-primed immune responses, but their functional contributions to Y. pestis vaccines have yet to be evaluated and optimized. Toward that end, we report here the identification of three distinct epitopes within the Y. pestis V protein that activate CD4 T cells in C57BL/6 mice. To our knowledge, these are the first identified CD4 T-cell epitopes in any Y. pestis protein. The epitopes are restricted by the I-Ab class II major histocompatibility complex molecule and are fully conserved between Y. pestis, Yersinia pseudotuberculosis, and Yersinia enterocolitica. Immunizing mice with a V protein-containing vaccine or with short peptides containing the identified epitopes primes antigen-specific production of interleukin 2 and gamma interferon by CD4 T cells upon their restimulation in vitro. Consistent with prior studies documenting protective roles for CD4 T cells during Y. enterocolitica infection, vaccinating mice with a 16-amino-acid peptide encoding one of the epitopes suffices to protect against an otherwise lethal Y. enterocolitica challenge. The identification of these epitopes will permit quantitative assessments of V-specific CD4 T cells, thereby enabling researchers to evaluate and optimize the contribution of these cells to vaccine-primed protection against pneumonic plague.
Epidemics of plague, an often-fatal infectious disease, have afflicted mankind throughout recorded history (26). Plague may manifest in bubonic, septicemic, or pneumonic form. The bubonic form may be naturally acquired via the bite of an infected flea, is characterized by fever and painfully swollen lymph nodes (buboes), and can progress to the life-threatening septicemic and pneumonic forms. From a public health standpoint, pneumonic plague is particularly dangerous, as it can be transmitted directly from person to person via aerosolized respiratory droplets.
The etiologic agent of plague is Yersinia pestis, a gram-negative bacterium (26). While most isolates of Y. pestis are sensitive to antibiotics, some are not (10). Moreover, antibiotics are only marginally effective once symptoms of pneumonic plague develop. Given the potential for antibiotic-resistant Y. pestis to initiate a pneumonic plague pandemic, there is concern that Y. pestis may be exploited as a bioweapon (31).
Despite considerable research efforts, no vaccine is currently available to protect humans against pneumonic plague. A formalin-killed Y. pestis whole-cell vaccine was widely used by American soldiers during the Vietnam War (18). That vaccine induced robust humoral immunity and effectively protected against bubonic plague; however, it was significantly reactogenic and failed to protect against pneumonic plague (18). Subsequently, researchers have sought to develop subunit vaccines comprised of recombinant Y. pestis proteins (31). Such vaccines are generally safer and less reactogenic than whole-cell vaccines. Among numerous Y. pestis proteins thus far evaluated for vaccine efficacy, the fraction 1 (F1) and V proteins offer the most promise, as vaccination with their recombinant forms clearly protects mice against pneumonic plague (2, 3). Ultimately, V protein may be a better choice for vaccine development, since it protects against infection by either F1-positive or F1-negative strains (2). Nevertheless, vaccination with a combination of F1 and V affords mice with better protection than that provided by either subunit alone (33), and a recombinant F1-V fusion protein vaccine protects mice against pneumonic plague (12). However, recent studies indicate that the F1-V fusion protein vaccine is less effective in nonhuman primates, suggesting that further improvements in vaccine design are desirable (J. Adamovicz, unpublished observations).
Vaccines can potentially prime both humoral and cell-mediated immune responses. Recent Y. pestis vaccine studies have focused largely upon antibody-based humoral immunity, presumably because adoptive serotherapy can protect mice against pneumonic plague (11). CD4 T cells most certainly contribute to antibody-based immunity, as they are critically important for memory B-cell responses and the affinity maturation of antibodies (1, 16). In addition to enhancing humoral immunity, vaccine-primed CD4 T cells may also contribute to forms of protection that are directly mediated by cells of the immune system. For example, primed CD4 T cells can secrete phagocyte-activating type 1 cytokines and/or aid the expansion and persistence of cytolytic T cells (5, 15). Indeed, prior studies have revealed important protective roles for CD4 T cells, and for the type 1 cytokines that they produce, during the infection of mice by the related yersinia Y. enterocolitica (4, 6, 13, 22). With regard to Y. pestis, Williamson and colleagues have clearly established that vaccination with V protein elicits detectable CD4 T-cell responses (17, 23, 34). However, the protective capacities of Y. pestis-specific CD4 T cells have yet to be evaluated decisively.
CD4 T cells respond to processed antigens presented by class II major histocompatibility complex (MHC) molecules expressed on specialized antigen-presenting cells (APC). Identification of the precise peptide antigens (i.e., epitopes) recognized by CD4 T cells would allow the quantitative tracking, measurement, and phenotyping of CD4 T-cell responses during vaccination and challenge studies and would enable researchers to evaluate and optimize the contributions of CD4 T cells to vaccine-primed protection against pneumonic plague. Here, we report the identification of three discrete Y. pestis V protein epitopes recognized by CD4 T cells in the context of the murine I-Ab class II MHC molecule. We also establish that vaccination with peptides encoding these epitopes primes V-specific CD4 T-cell responses. These findings should pave the way for decisive evaluations of roles for V-specific CD4 T cells during vaccine-mediated protection against plague.
MATERIALS AND METHODS
Mice.
Male C57BL/6 mice, 6 to 10 weeks of age, were purchased from either Jackson Laboratory (Bar Harbor, Maine) or Taconic (Germantown, N.Y.). Animals were housed at Trudeau Institute experimental animal facility and cared for according to the Trudeau Institute Animal Care and Use Committee guidelines.
Antigens.
The F1-V fusion protein vaccine (12) was supplied by the U.S. Army Medical Research Institute of Infectious Diseases Plague Vaccine Program. Ovalbumin (OVA) was purchased from Sigma. To screen for T-cell epitopes, a set of 63 overlapping peptides was generated covering the entire length of the Y. pestis V protein. This peptide set (PepSet) was synthesized by Mimotopes (Clayton, Victoria, Australia). Each peptide contained 16 contiguous amino acids of V protein and overlapped its neighboring peptides by 11 amino acids. Each peptide also contained a carboxyl-terminal glycine residue to facilitate efficient cleavage during synthesis. To further define the V protein epitopes, a second set of peptides was generated. This set was synthesized by New England Peptide (Gardner, Mass.) and lacked the carboxyl-terminal glycine residues and included repetitions of original peptides along with amino- and carboxyl-terminal truncations that removed two, four, or six amino acids from each peptide. Finally, a third set of peptides was generated at larger scale for vaccination purposes. This set was supplied by New England Peptide at greater than 90% purity, as determined by high-pressure liquid chromatography analysis.
Vaccinations.
To measure primary T-cell responses to the F1-V vaccine, mice were immunized with 10 μg of F1-V protein in Complete Freund's adjuvant (CFA; Sigma), administered subcutaneously in the lumbar sacral region. Control mice received 200 μg of OVA in CFA. To measure primary T-cell responses to peptide vaccination, mice were immunized with 100 μg of the designated V peptide emulsified in CFA as described above for F1-V, and then boosted 30 days later with 100 μg of the same peptide emulsified in incomplete Freund's adjuvant (IFA). The peptides used for vaccination studies included V protein amino acids 71 to 86 (LKKILAYFLPEDAILK; V1), V protein amino acids 101 to 116 (VKEFLESSPNTQWELR; V2), V protein amino acids 166 to 181 (IYSVIQAEINKHLSSS; V3), and V protein amino acids 11 to 26 (HFIEDLEKVRVEQLTG; Vneg).
Measurement of CD4 T-cell responses.
At the days after immunization as indicated in the figure legends, mice were euthanized by carbon dioxide narcosis. Spleens, inguinal lymph nodes, and popliteal lymph nodes were then harvested from two to three mice per vaccination condition and pooled. After tissue disruption by passage through wire mesh, CD4 T cells were positively sorted by using CD4 monoclonal antibody (MAb)-conjugated magnetic beads (Miltenyi Biotec). Routinely, the resultant cells were >90% CD4-positive as determined by flow cytometry. CD4-positive cells (105) were cultured in 200 μl of complete medium (Dulbecco's modified Eagle medium, 10% fetal bovine serum, 2 mM glutamine, 100 μM concentrations of nonessential amino acids, 50 U of penicillin/ml, 50 μg of streptomycin/ml, 50 μM 2-mercaptoethanol, and 25 mM HEPES; all from Invitrogen) along with splenic APC (106) that had been pretreated with 50 μg of mitomycin C (Sigma)/ml for 30 min at 37°C. Cultures also contained 100 μg of F1-V/ml, 250 μg of OVA/ml, or 10 μM concentrations of individual V peptides. Supernatant samples (50 μl) were removed 24 h after the initiation of culture and assayed for levels of interleukin 2 (IL-2) by enzyme-linked immunosorbent assay (ELISA; BD Pharmingen). The removed supernatant was replaced with complete medium supplemented with recombinant human IL-2 (final concentration of 20 U/ml; Peprotech), and at 48 h after initiation of culture, an additional 50-μl supernatant sample was removed and assayed for the level of gamma interferon (IFN-γ) by ELISA (BD Pharmingen). The number of IFN-γ-producing cells was also determined by enzyme-linked immunospot assay (ELISPOT assay) using a parallel set of cultures in which CD4-positive cells were serially diluted into cellulose ester membrane plates (Millipore) coated with MAbs specific for mouse IFN-γ (BD Pharmingen). Each well also contained 106 mitomycin c-treated APC, 10 μM V peptide, and 10 U of IL-2/ml. Twenty-four hours after the initiation of ELISPOT cultures, plates were washed and developed by using biotinylated MAbs specific for mouse IFN-γ (BD Pharmingen), streptavidin-alkaline phosphatase (Sigma), and 3,3′,5,5′-tetramethylbenzidine development reagent substrate (BD Pharmingen). Where indicated, cell cultures were supplemented with the MHC-binding MAbs. Clones producing the indicated MAbs were obtained from the American Type Culture Collection, and MAb was purified by using protein A Sepharose.
Infections.
Mice were immunized with V peptides emulsified in CFA and boosted 30 days later with the same peptides emulsified in IFA as described above. On day 60 after primary immunization, mice were intraperitoneally challenged with 104 CFU of Y. enterocolitica WA (ATCC 27729). Preliminary studies established that this dose represented 10 times the 50% lethal dose (LD50), as calculated by the method of Reed and Muench (29). Challenged animals were observed and weighed daily.
Statistics.
Statistical analyses were performed by using the program Prism 4.0 (GraphPad Software, Inc.), employing analysis of variance with Tukey posttests for ELISPOT data and log rank tests for survival data.
RESULTS
Measurement of V-specific CD4 T-cell responses and identification of V peptides containing CD4 T-cell epitopes.
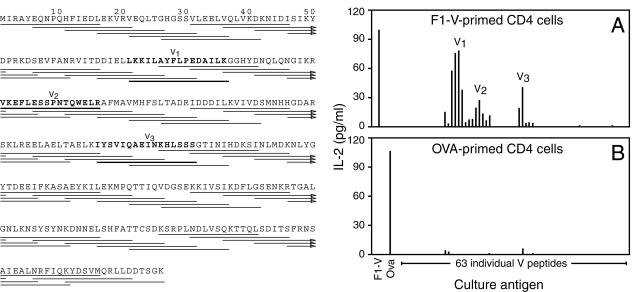
Prior to defining V protein epitopes recognized by CD4 T cells, we first established that vaccination with the F1-V fusion protein evoked detectable T-cell responses. Toward that end, we immunized C57BL/6 mice with F1-V emulsified in CFA, isolated CD4-positive cells 6 days later, and cultured those cells along with syngeneic splenic APC and either F1-V or OVA, an irrelevant control protein. As an additional specificity control, we measured responses of CD4 T cells isolated from mice that had been immunized with OVA rather than F1-V. We quantified T-cell activation by measuring cytokine release into the supernatant at 24 h after the initiation of culture. We found that vaccination evoked antigen-specific CD4 T-cell responses. CD4 T cells isolated from F1-V-vaccinated mice produced IL-2 in response to F1-V, but not OVA (Fig. 1A), whereas CD4 T cells isolated from mice vaccinated with OVA responded to OVA, but not F1-V (Fig. 1B). In multiple experiments, antigen-specific IL-2 production was always more than 100-fold above the detection limit of our assay. Thus, vaccination with F1-V primed robust antigen-specific CD4 T-cell responses that could be detected readily in vitro.
FIG. 1.
Measurement of V-specific CD4 T-cell responses and identification of V peptides containing CD4 T-cell epitopes. C57BL/6 mice were primed with 10 μg of F1-V fusion protein (A) or 200 μg of OVA (B) emulsified in CFA. Six days later, CD4-positive cells were isolated and cultured (5 × 105/ml) along with mitomycin c-treated splenic APC (5 × 106/ml) and F1-V (100 μg/ml), OVA (250 μg/ml), or individual V peptides (10 μM). The left panel depicts the sequences of the 63 individual peptides that were evaluated. After 24 h of culture, the levels of IL-2 in the culture supernatants were assayed by ELISA. Data are the average measurements for duplicate samples from two independent experiments. Similar results were obtained by using [3H]thymidine incorporation as readout (data not shown). The responses of 63 individual V peptides are shown, beginning at the left with the amino-terminal peptide and then moving sequentially through the protein. V1, V2, and V3 refer to the individual peptides that were chosen for further characterization.
To define the epitopes within V protein that are recognized by CD4 T cells, we isolated CD4-positive cells from C57BL/6 mice that had been immunized 6 days prior with the F1-V fusion protein vaccine and cultured those cells along with syngeneic splenic APC and each of 63 individual peptides encoding short segments of V protein. Each peptide was 16 amino acids in length and overlapped its neighboring peptides by 11 amino acids. We found that CD4-positive cells isolated from F1-V-vaccinated mice responded to peptides encoding multiple regions of V protein. Specifically, we identified three distinct regions of V protein in which overlapping peptides evoked significant CD4 T-cell responses (Fig. 1A). Importantly, these responses were antigen specific, as they were primed by vaccination with F1-V, but not by vaccination with OVA (Fig. 1A and B). We denoted the three most highly reactive peptides from each V protein region V1, V2, and V3 (Fig. 1A).
Identification of the precise V protein epitopes recognized by CD4 T cells.
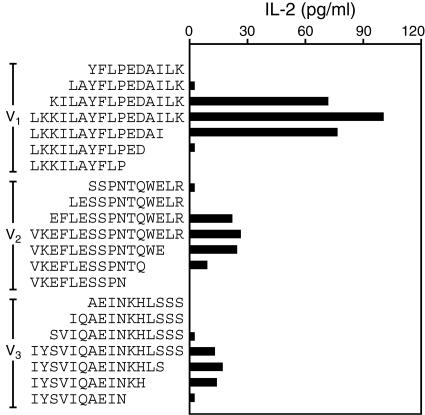
To better define the CD4 T-cell epitopes within V protein, we synthesized additional sets of peptides in which peptides V1, V2, and V3 were truncated at their amino- or carboxyl-terminus by two, four, or six amino acids. We then measured the capacity of CD4-positive cells isolated from F1-V-immunized mice to respond to these peptides. As shown in Fig. 2, these analyses established that V protein amino acids 73 to 84 (KILAYFLPEDAI), 103 to 114 (EFLESSPNTQWE), and 166 to 177 (IYSVIQAEINKH) encode the core sequences of epitopes V1, V2, and V3, respectively. Figure 3 depicts an alignment of the amino acid sequences of the Y. pestis, Y. enterocolitica, and Yersinia pseudotuberculosis V proteins. As shown in Fig. 3, the core epitopes of peptides V1, V2, and V3 are fully conserved among these three yersinia species.
FIG. 2.
Identification of the V protein epitopes recognized by CD4 T cells. C57BL/6 mice were immunized with F1-V fusion protein, and the responses of primed CD4 T cells were measured in vitro, as in Fig. 1. Cultures were supplemented with the indicated peptides (10 μM). As in Fig. 1, data are the average measurements for duplicate samples from two independent experiments.
FIG. 3.
Conservation of V protein CD4 T-cell epitopes among multiple yersinia species. Alignment of the amino acid sequences of the Y. pestis (Y. pes.; strains KIM and CO92), Y. enterocolitica (Y. ent.; type 0:8, strain WA-314), and Y. pseudotuberculosis (Y. pse.; strain IP32953) V proteins (8, 25, 27, 30). Hyphens depict residues that are conserved relative to Y. pestis, whereas dots depict residues that are absent in one species relative to another. All amino acid differences relative to the Y. pestis sequence are shown. Peptides V1, V2, and V3 are indicated by boldface type, and the core CD4 T-cell epitopes identified in Fig. 2 are boxed. Note that the core epitopes are 100% conserved among the three yersiniae.
Immunizing mice with peptides encoding V protein CD4 T-cell epitopes evokes V-specific CD4 T-cell responses.
Having identified CD4 T-cell epitopes in V protein, we next evaluated whether immunizing mice with peptides encoding such epitopes could prime V-specific CD4 T-cell responses. For this purpose, we procured highly purified (>90% by high-pressure liquid chromatography analysis) versions of peptides V1, V2, and V3 and of an unresponsive control peptide, Vneg. We then immunized mice with these peptides emulsified in CFA, and assessed CD4 T-cell priming 6 days later by measuring the responses of CD4-positive cells in vitro, as described above. In parallel, we evaluated the responses of CD4-positive cells isolated from mice vaccinated with F1-V or OVA, as positive or negative controls, respectively. Consistent with our prior findings (Fig. 1 and 2), CD4 T cells isolated from mice immunized with F1-V specifically responded to peptides V1, V2, and V3, whereas CD4 T cells isolated from mice immunized with OVA failed to respond to these peptides (Fig. 4). Immunization with peptides V1, V2, and V3 also primed antigen-specific CD4 T-cell responses, as CD4-positive cells isolated from mice immunized with peptides V1, V2, or V3 responded specifically to peptides V1, V2, or V3, respectively.
FIG. 4.
Immunization with V peptides encoding CD4 T-cell epitopes primes antigen-specific CD4 T-cell responses. C57BL/6 mice were immunized with 10 μg of F1-V, 200 μg of OVA, 100 μg of peptide V1, 100 μg of peptide V2, 100 μg of peptide V3, or 100 μg of a control peptide (Vneg) emulsified in CFA. Six days later, CD4 T-cell priming was assessed by measuring in vitro production of IL-2, as in Fig. 1. Data are representative of results obtained in three independent experiments.
Vaccination should, ideally, prime memory cells that are capable of persisting in the long term and mounting a rapid recall response upon subsequent antigenic challenge. To assess whether immunization with V peptides primes memory CD4 T cells, we immunized mice with peptides V1, V2, V3, or Vneg emulsified in CFA, and administered secondary booster immunizations 30 days later with the same peptides emulsified in IFA. On day 60 after the initial immunization, we assayed CD4 T-cell memory by measuring antigen-stimulated cytokine production by CD4-positive cells in vitro. We found that CD4-positive cells isolated from mice vaccinated with peptides V1, V2, or V3 specifically produced IL-2 in response to in vitro stimulation with peptides V1, V2, or V3, respectively (Fig. 5A). In contrast, CD4-positive cells isolated from mice vaccinated with peptide Vneg failed to respond to any of these peptides, although they did respond when nonspecifically activated with a positive control stimulus, anti-CD3 MAb (data not shown). Thus, vaccinating with V peptides that encode CD4 T-cell epitopes specifically primes memory responses to those epitopes.
FIG. 5.
Immunization with V peptides encoding CD4 T-cell epitopes primes antigen-specific type 1 memory CD4 T-cell responses. C57BL/6 mice were immunized with peptides V1, V2, V3, or Vneg emulsified in CFA. Thirty days later, mice were reimmunized with the same peptides emulsified in IFA. On day 60 after the primary immunization, memory CD4 T-cell responses were assessed by measuring the in vitro production of IL-2 (A) or IFN-γ (B) by ELISA, as in Fig. 1. In addition, the number of IFN-γ-producing cells was measured by ELISPOT assay using triplicate determinations for each sample (C). P values were <0.01 (*) and <0.001 (**) compared with each of the other values within an immunization group. Data are representative of results obtained in three independent experiments.
Freund's adjuvant-based vaccination protocols are well recognized to prime type 1 cytokine responses. To determine whether our vaccination protocols primed V-specific type 1 responses, we assessed the capacity of V peptide-primed CD4 T cells to produce IFN-γ, a cytokine that is characteristically produced by type 1 CD4 T cells. As shown in Fig. 5B, CD4-positive cells isolated from mice vaccinated with peptides V1, V2, or V3 specifically produced IFN-γ in response to in vitro stimulation with peptides V1, V2, or V3, respectively, whereas CD4-positive cells isolated from mice vaccinated with peptide Vneg failed to produce IFN-γ in response to any peptide. In parallel with these ELISA-based measurements of total IFN-γ production, we also measured IFN-γ production by ELISPOT assay, a method that quantifies the actual number of CD4 T cells producing a given cytokine. As with the ELISA results, the ELISPOT assay also detected robust antigen-specific IFN-γ production (Fig. 5C). Together, these results document that vaccination with V peptides encoding CD4 T-cell epitopes can prime V-specific type 1 memory CD4 T-cell responses.
CD4 T cells recognize V peptides in the context of the I-Ab class II MHC molecule.
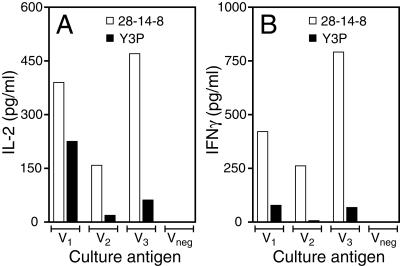
CD4 T cells recognize peptides presented by class II MHC molecules. In C57BL/6 mice, CD4 T-cell responses are primarily restricted by the I-Ab class II MHC molecule, and each of the core epitopes within V1, V2, and V3 contains sequences that are predicted to bind I-Ab by the computer program RANKPEP (28). To formally assess whether I-Ab restricts the presentation of these epitopes, we assessed whether MAbs known to suppress antigen presentation by I-Ab suppressed presentation of the identified V epitopes. In the experiment depicted in Fig. 6, we immunized mice with a combination of all three V epitope-containing peptides, and we analyzed CD4 responses in vitro 6 days later. We supplemented the in vitro CD4 T-cell cultures with Y3P, a MAb that binds I-Ab and inhibits its capacity to present antigens (14), or with 28-14-8, an isotype-matched control MAb that binds the Db class I MHC molecules expressed by C57BL/6 APC (24). As shown in Fig. 6, inclusion of MAb Y3P specifically suppressed the activation of V-specific CD4 T cells; IL-2 and IFN-γ production were reduced in peptide-stimulated cultures supplemented with Y3P, compared to cultures supplemented with 28-14-8. We conclude that the I-Ab class II MHC molecule presents epitopes V1, V2, and V3 to CD4 T cells.
FIG. 6.
Immunization with V peptides encoding CD4 T-cell epitopes primes antigen-specific CD4 T-cell responses that are restricted by the I-Ab class II MHC molecule. C57BL/6 mice were immunized with a combination of peptides V1, V2, and V3 emulsified in CFA. Six days later, CD4 T-cell priming was assessed by measuring in vitro production of IL-2 (A) and IFN-γ (B) by ELISA, as in Fig. 5. Peptide-specific responses were suppressed when in vitro cultures were supplemented with 200 μg of Y3P (a MAb that binds I-Ab)/ml, but not when supplemented with 28-14-8, a MAb that binds Db. The Y3P-induced suppression was specific, as polyclonal responses evoked by stimulation with anti-CD3 MAb were not suppressed by Y3P. IL-2 levels were 1,050 and 920 pg/ml and IFN-γ levels were 6,270 and 8,850 pg/ml for 28-14-8 and Y3P, respectively. Data are representative of results obtained in three independent experiments.
Vaccinating mice with peptides encoding V protein CD4 T-cell epitopes protects against lethal infection by Y. enterocolitica.
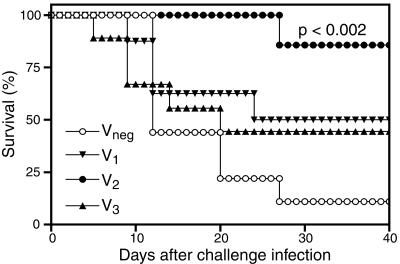
To assess whether vaccination with V peptides encoding CD4 T-cell epitopes suffices to protect against infection, we performed infectious challenge experiments. We employed Y. enterocolitica for these studies, as the V epitopes that we identified are fully conserved between Y. pestis and Y. enterocolitica (Fig. 3), and prior studies established that CD4 T cells and the type 1 cytokines that they produce can function protectively during Y. enterocolitica infection (4, 6, 13, 22). As shown in Fig. 5, mice were immunized with peptides V1, V2, V3, or Vneg emulsified in CFA, and then boosted on day 30 with the same peptides emulsified in IFA. On day 60 after the initial immunization, mice were challenged intraperitoneally with 10 times the LD50 of Y. enterocolitica (Fig. 7). Mice vaccinated with the Vneg largely succumbed, with only one of nine mice surviving to day 40 postinfection. By comparison, mice vaccinated with peptides V1, V2, or V3 survived the challenge infection in greater numbers. Vaccination with peptide V2 generated the most robust protection, with six of seven mice surviving to day 40. Statistical analyses revealed that the 86% survival rate of the V2-vaccinated group was highly significant in comparison to the 11% survival rate of the Vneg-vaccinated control group (P < 0.002 by log rank test). Although not statistically significant, some degree of protection appeared to have been provided against lethal Y. enterocolitica challenge also by vaccination with peptides V1 and V3, as demonstrated by 50 and 44% survival rates, respectively.
FIG. 7.
Protection against yersinia infection by vaccination with V peptides containing CD4 T-cell epitopes. C57BL/6 mice were immunized and boosted, as described for Fig. 5. On day 60 after the primary immunization, the mice (n = 7 to 10 per group) were intraperitoneally inoculated with 10 times the LD50 of Y. enterocolitica and monitored for survival.
DISCUSSION
CD4 T cells recognize processed antigenic epitopes in the context of class II MHC molecules expressed on specialized APC. Upon their subsequent activation, CD4 T cells play central roles in the priming and maintenance of acquired immune responses, including those induced by vaccination (1, 5, 15, 16). Prior studies established that F1-V-based vaccines provide mice with significant protection against aerosolized plague (2, 3, 12, 33) and demonstrated that F1-V-based vaccines evoke detectable CD4 T-cell responses (17, 23, 34). In this report, we described our identification of three distinct CD4 T-cell epitopes within the Y. pestis V protein. Specifically, we demonstrated that immunizing mice with a V protein-containing vaccine, or short peptides encoding the identified V protein epitopes, primes antigen-specific production of IL-2 and IFN-γ by CD4 T cells upon their restimulation in vitro (Fig. 1, 2, 4, 5, and 6). In addition, we demonstrated that I-Ab class II MHC molecules present these epitopes to CD4 T cells, as a MAb specific for I-Ab suppresses the capacity of CD4 T cells to respond to these epitopes (Fig. 6). To our knowledge, these epitopes constitute the first identified CD4 T-cell epitopes in the Y. pestis V protein and the only Y. pestis CD4 T-cell epitopes identified thus far.
Fine mapping of the three V protein CD4 T-cell epitopes established that each epitope is distinct (Fig. 2) and evolutionarily conserved among Y. pestis, Y. pseudotuberculosis, and Y. enterocolitica (Fig. 3). At present, it is unclear whether the conservation of these epitopes is biologically meaningful or coincidental. Regardless, this conservation suggests that these epitopes will facilitate studies of CD4 T-cell responses in the context of vaccination and/or infection by each of the pathogenic Yersinia species. Indeed, we found that vaccination with a peptide encoding the V2 epitope sufficed to protect against an otherwise lethal Y. enterocolitica challenge infection (Fig. 7), consistent with prior studies documenting that CD4 T cells and type 1 cytokines combat infection by Y. enterocolitica (4, 6, 13, 22). Our data do not formally exclude the possibility that antibodies also contributed to the protection observed in our model, but such a scenario seems unlikely given that antibodies raised against Y. pestis V protein do not cross-protect against the strain of Y. enterocolitica chosen for our studies (19, 21, 30). Regardless, our data establish that vaccination with a short peptide encoding a V protein epitope recognized by CD4 T cells is sufficient to protect against Y. enterocolitica infection.
In striking contrast to the abovementioned studies with Y. enterocolitica, we have thus far failed to observe significant protection when mice are vaccinated with V epitopes and then challenged with Y. pestis KIM D27, a pigmentation-negative strain (data not shown) (32). Ongoing studies are aimed at further optimizing vaccine-primed V-specific CD4 T-cell responses.
Interestingly, the three V epitopes that we identified seem to differentially stimulate CD4 T cells. Upon immunization with either the F1-V fusion protein or the epitope-containing peptides, epitope V1 or V3 always primed for maximal IL-2 and IFN-γ production by effector CD4 T cells (i.e., when responses were measured at 6 days after immunization) (Fig. 1, 2, 4, and 6). Likewise, whenever mice were immunized and boosted, epitope V1 or V3 also evoked maximal IL-2 production by memory CD4 T cells (i.e., when responses were measured at 60 days after immunization) (Fig. 5). However, in multiple independent experiments, peptides containing epitope V2 always primed maximal IFN-γ production by memory CD4 T cells (Fig. 5 and data not shown). Further studies are required to assess whether these observations reflect intrinsic differences between these epitopes or whether they reflect variables that can be modified, for example, by changing immunization dosages and/or adjuvants. Regardless, our studies thus far suggest that vaccination with epitope V2, which primes maximal IFN-γ production, best protects against Y. enterocolitica infection. That observation is consistent with prior literature that indicated that IFN-γ is an important mediator of protection against Y. enterocolitica (4, 6, 13, 22).
In recent years, Y. pestis vaccine studies have aimed primarily to stimulate robust humoral immunity, presumably reflecting solid evidence that antibodies can protect mice against pneumonic plague. For example, the passive transfer of V-specific antisera protects immunodeficient SCID/Beige mice against pneumonic plague (11), indicating that V-specific antibodies can suffice to protect against pulmonary Y. pestis infection, at least in a murine model. CD4 T cells are critically important for the affinity maturation of antibodies (1, 16), and thus, they most certainly participate in the priming of vaccine-mediated humoral immunity. CD4 T cells also function in the long-term maintenance of memory humoral responses and, thus, in the longevity of vaccine efficacy (1, 16). Our identification of the precise epitopes recognized by V-specific CD4 T cells will enable the specific priming and monitoring of such cells, thereby permitting researchers to evaluate whether optimizing vaccine protocols to maximally expand V-specific CD4 T-cell populations will enhance antibody-mediated protection against pneumonic plague.
While humoral immunity certainly contributes to protection against plague, full protection may also benefit from robust priming of cellular immunity. Several unpublished, but publicly disclosed, studies by the U.S. Army Medical Research Institute of Infectious Diseases found that immunization of nonhuman primates with the F1-V fusion protein provided early protection against lethal challenge with aerosolized Y. pestis (G. P. Andrews, Medical Defense Against Bioterrorism—Workshop, 6 to 7 December 2000 [http://www.fda.gov/cber/summaries/120600bio09.htm]; M. L. Pitt, Public Workshop on Animal Models and Correlates of Protection for Plague Vaccines, Gaithersburg, Md., 13 to 14 October 2004 [http://www.fda.gov/cber/minutes/workshop-min.htm]). Nevertheless, some challenged animals succumbed to Y. pestis infection, despite their possession of high titer F1-V-specific antibody at the time of challenge (Adamovicz, unpublished). These studies suggest that antibody titers may not suffice as predictors of pneumonic plague vaccine efficacy and that future studies should evaluate whether levels of vaccine-elicited cellular immunity can be used to predict vaccine efficacy.
In addition to their well-recognized roles during humoral immune responses, CD4 T cells also play important roles during cellular immune responses. Specifically, CD4 T cells may secrete phagocyte-activating type 1 cytokines, such as IFN-γ and tumor necrosis factor alpha, and can play critical roles in the development and maintenance of memory CD8 T-cell responses (5, 15). While explicit roles for CD8 T cells during Y. pestis infection have yet to be described, it appears that Y. pestis maintains virulence, at least in part, by suppressing the production of type 1 cytokines (7, 20). Moreover, the parenteral administration of IFN-γ and tumor necrosis factor alpha protects mice against lethal Y. pestis infection (20), and the efficacy of an F1- and V-based vaccine is reduced in Stat 4-deficient mice, which are diminished in their capacity to mount type 1 cytokine responses (9). Together, these observations strongly suggest that cytokine-mediated cellular immunity is detrimental to Y. pestis and that priming yersinia-specific type 1 CD4 T cells may enhance vaccine-mediated protection against pneumonic plague. Ongoing studies are aimed at evaluating whether cellular immunity orchestrated by V-specific CD4 T cells can synergize with humoral immunity to protect against Y. pestis infection.
In conclusion, a number of prior studies support the notion that V-specific CD4 T cells, if preprimed by vaccination, may enhance both humoral and cell-mediated defense against pneumonic plague. Moreover, optimizing the expansion and persistence of vaccine-primed CD4 T cells should improve the longevity of protection after vaccination, as CD4 T cells are important regulators of both humoral and cellular memory responses. Our identification of precise V protein epitopes recognized by CD4 T cells will permit the quantitative monitoring of V-specific CD4 T-cell numbers, phenotypes, and cytokine-secreting capacities during vaccination and challenge experiments. Knowledge of these epitopes will also permit the specific priming of such cells, thereby facilitating studies of their functional attributes. Acquiring direct information about the activation, expansion, and persistence of Y. pestis-specific CD4 T cells, and optimizing vaccines to harness their protective capacities, should certainly aid the development of effective pneumonic plague vaccines.
Acknowledgments
This work was supported by PHS grants AI054595, AI057158, and AI061577 (S.T.S.) and by funds from Trudeau Institute.
We thank Lawrence Johnson and Andrea Cooper for critical reading of the manuscript. We are indebted to the employees of the Trudeau Institute Animal Breeding and Maintenance Facilities for dedicated care of the mice used in these studies. We also thank Robert Brubaker for providing access to Y. pestis KIM D27.
Editor: D. L. Burns
REFERENCES
- 1.Ahmed, R., and D. Gray. 1996. Immunological memory and protective immunity: understanding their relation. Science 272:54-60. [DOI] [PubMed] [Google Scholar]
- 2.Anderson, G. W., Jr., S. E. Leary, E. D. Williamson, R. W. Titball, S. L. Welkos, P. L. Worsham, and A. M. Friedlander. 1996. Recombinant V antigen protects mice against pneumonic and bubonic plague caused by F1-capsule-positive and -negative strains of Yersinia pestis. Infect. Immun. 64:4580-4585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Andrews, G. P., D. G. Heath, G. W. Anderson, Jr., S. L. Welkos, and A. M. Friedlander. 1996. Fraction 1 capsular antigen (F1) purification from Yersinia pestis CO92 and from an Escherichia coli recombinant strain and efficacy against lethal plague challenge. Infect. Immun. 64:2180-2187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Autenrieth, I. B., M. Beer, E. Bohn, S. H. Kaufmann, and J. Heesemann. 1994. Immune responses to Yersinia enterocolitica in susceptible BALB/c and resistant C57BL/6 mice: an essential role for gamma interferon. Infect. Immun. 62:2590-2599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bevan, M. J. 2004. Helping the CD8+ T-cell response. Nat. Rev. Immunol. 4:595-602. [DOI] [PubMed] [Google Scholar]
- 6.Bohn, E., and I. B. Autenrieth. 1996. IL-12 is essential for resistance against Yersinia enterocolitica by triggering IFN-γ production in NK cells and CD4+ T cells. J. Immunol. 156:1458-1468. [PubMed] [Google Scholar]
- 7.Brubaker, R. R. 2003. Interleukin-10 and inhibition of innate immunity to yersiniae: roles of Yops and LcrV (V antigen). Infect. Immun. 71:3673-3681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Chain, P. S., E. Carniel, F. W. Larimer, J. Lamerdin, P. O. Stoutland, W. M. Regala, A. M. Georgescu, L. M. Vergez, M. L. Land, V. L. Motin, R. R. Brubaker, J. Fowler, J. Hinnebusch, M. Marceau, C. Medigue, M. Simonet, V. Chenal-Francisque, B. Souza, D. Dacheux, J. M. Elliott, A. Derbise, L. J. Hauser, and E. Garcia. 2004. Insights into the evolution of Yersinia pestis through whole-genome comparison with Yersinia pseudotuberculosis. Proc. Natl. Acad. Sci. USA 101:13826-13831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Elvin, S. J., and E. D. Williamson. 2004. Stat 4 but not Stat 6 mediated immune mechanisms are essential in protection against plague. Microb. Pathog. 37:177-184. [DOI] [PubMed] [Google Scholar]
- 10.Galimand, M., A. Guiyoule, G. Gerbaud, B. Rasoamanana, S. Chanteau, E. Carniel, and P. Courvalin. 1997. Multidrug resistance in Yersinia pestis mediated by a transferable plasmid. N. Engl. J. Med. 337:677-680. [DOI] [PubMed] [Google Scholar]
- 11.Green, M., D. Rogers, P. Russell, A. J. Stagg, D. L. Bell, S. M. Eley, R. W. Titball, and E. D. Williamson. 1999. The SCID/Beige mouse as a model to investigate protection against Yersinia pestis. FEMS Immunol. Med. Microbiol. 23:107-113. [DOI] [PubMed] [Google Scholar]
- 12.Heath, D. G., G. W. Anderson, Jr., J. M. Mauro, S. L. Welkos, G. P. Andrews, J. Adamovicz, and A. M. Friedlander. 1998. Protection against experimental bubonic and pneumonic plague by a recombinant capsular F1-V antigen fusion protein vaccine. Vaccine 16:1131-1137. [DOI] [PubMed] [Google Scholar]
- 13.Heesemann, J., K. Gaede, and I. B. Autenrieth. 1993. Experimental Yersinia enterocolitica infection in rodents: a model for human yersiniosis. APMIS 101:417-429. [PubMed] [Google Scholar]
- 14.Janeway, C. A., Jr., P. J. Conrad, E. A. Lerner, J. Babich, P. Wettstein, and D. B. Murphy. 1984. Monoclonal antibodies specific for Ia glycoproteins raised by immunization with activated T cells: possible role of T cellbound Ia antigens as targets of immunoregulatory T cells. J. Immunol. 132:662-667. [PubMed] [Google Scholar]
- 15.Kaech, S. M., E. J. Wherry, and R. Ahmed. 2002. Effector and memory T-cell differentiation: implications for vaccine development. Nat. Rev. Immunol. 2:251-262. [DOI] [PubMed] [Google Scholar]
- 16.Kelsoe, G. 2000. Studies of the humoral immune response. Immunol. Res. 22:199-210. [DOI] [PubMed] [Google Scholar]
- 17.Leary, S. E., E. D. Williamson, K. F. Griffin, P. Russell, S. M. Eley, and R. W. Titball. 1995. Active immunization with recombinant V antigen from Yersinia pestis protects mice against plague. Infect. Immun. 63:2854-2858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Meyer, K. F. 1970. Effectiveness of live or killed plague vaccines in man. Bull. W. H. O. 42:653-666. [PMC free article] [PubMed] [Google Scholar]
- 19.Motin, V. L., R. Nakajima, G. B. Smirnov, and R. R. Brubaker. 1994. Passive immunity to yersiniae mediated by anti-recombinant V antigen and protein A-V antigen fusion peptide. Infect. Immun. 62:4192-4201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Nakajima, R., and R. R. Brubaker. 1993. Association between virulence of Yersinia pestis and suppression of gamma interferon and tumor necrosis factor alpha. Infect. Immun. 61:23-31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Nakajima, R., V. L. Motin, and R. R. Brubaker. 1995. Suppression of cytokines in mice by protein A-V antigen fusion peptide and restoration of synthesis by active immunization. Infect. Immun. 63:3021-3029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Noll, A., A. Roggenkamp, J. Heesemann, and I. B. Autenrieth. 1994. Protective role for heat shock protein-reactive αβ T cells in murine yersiniosis. Infect. Immun. 62:2784-2791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Oyston, P. C., E. D. Williamson, S. E. Leary, S. M. Eley, K. F. Griffin, and R. W. Titball. 1995. Immunization with live recombinant Salmonella typhimurium aroA producing F1 antigen protects against plague. Infect. Immun. 63:563-568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Ozato, K., and D. H. Sachs. 1981. Monoclonal antibodies to mouse MHC antigens. III. Hybridoma antibodies reacting to antigens of the H-2b haplotype reveal genetic control of isotype expression. J. Immunol. 126:317-321. [PubMed] [Google Scholar]
- 25.Parkhill, J., B. W. Wren, N. R. Thomson, R. W. Titball, M. T. Holden, M. B. Prentice, M. Sebaihia, K. D. James, C. Churcher, K. L. Mungall, S. Baker, D. Basham, S. D. Bentley, K. Brooks, A. M. Cerdeno-Tarraga, T. Chillingworth, A. Cronin, R. M. Davies, P. Davis, G. Dougan, T. Feltwell, N. Hamlin, S. Holroyd, K. Jagels, A. V. Karlyshev, S. Leather, S. Moule, P. C. Oyston, M. Quail, K. Rutherford, M. Simmonds, J. Skelton, K. Stevens, S. Whitehead, and B. G. Barrell. 2001. Genome sequence of Yersinia pestis, the causative agent of plague. Nature 413:523-527. [DOI] [PubMed] [Google Scholar]
- 26.Perry, R. D., and J. D. Fetherston. 1997. Yersinia pestis—etiologic agent of plague. Clin. Microbiol. Rev. 10:35-66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Perry, R. D., S. C. Straley, J. D. Fetherston, D. J. Rose, J. Gregor, and F. R. Blattner. 1998. DNA sequencing and analysis of the low-Ca2+-response plasmid pCD1 of Yersinia pestis KIM5. Infect. Immun. 66:4611-4623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Reche, P. A., J. P. Glutting, H. Zhang, and E. L. Reinherz. 2004. Enhancement to the RANKPEP resource for the prediction of peptide binding to MHC molecules using profiles. Immunogenetics 56:405-419. [DOI] [PubMed] [Google Scholar]
- 29.Reed, L. J., and H. Muench. 1938. A simple method of estimating fifty per cent endpoints. Am. J. Hyg. 27:493-497. [Google Scholar]
- 30.Roggenkamp, A., A. M. Geiger, L. Leitritz, A. Kessler, and J. Heesemann. 1997. Passive immunity to infection with Yersinia spp. mediated by anti-recombinant V antigen is dependent on polymorphism of V antigen. Infect. Immun. 65:446-451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Titball, R. W., and E. D. Williamson. 2004. Yersinia pestis (plague) vaccines. Expert Opin. Biol. Ther. 4:965-973. [DOI] [PubMed] [Google Scholar]
- 32.Une, T., and R. R. Brubaker. 1984. In vivo comparison of avirulent Vwa− and Pgm− or Pstr phenotypes of yersiniae. Infect. Immun. 43:895-900. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Williamson, E. D., S. M. Eley, K. F. Griffin, M. Green, P. Russell, S. E. Leary, P. C. Oyston, T. Easterbrook, K. M. Reddin, A. Robinson, et al. 1995. A new improved sub-unit vaccine for plague: the basis of protection. FEMS Immunol. Med. Microbiol. 12:223-230. [DOI] [PubMed] [Google Scholar]
- 34.Williamson, E. D., P. M. Vesey, K. J. Gillhespy, S. M. Eley, M. Green, and R. W. Titball. 1999. An IgG1 titre to the F1 and V antigens correlates with protection against plague in the mouse model. Clin. Exp. Immunol. 116:107-114. [DOI] [PMC free article] [PubMed] [Google Scholar]