Abstract
We herein describe a straightforward protocol for the synthesis of carboxylic esters containing a gem-difluoromethylene unit. Readily available carboxylic acids can act as nucleophiles to add regioselectively to tetrasubstituted or trisubstituted β,β-difluoroacrylates (formal hydroacetoxylation) for the construction of RCO2–CF2 bonds. Thermal conditions are sufficient without the use of catalysts or additives.
An increasing level of interest in the synthesis of compounds containing the gem-difluoromethylene (-CF2-) unit has been seen in recent years.1 This is due to the ability of the CF2 group to act as a bioisostere for oxygen or carbonyl in pharmaceutical and agrochemical applications.2 Despite the existence of various difluoromethylenation methods, the addition of a nucleophile (Nuc-H) to gem-difluoroalkenes 1, a net hydrofunctionalization process, would be one of the simplest approaches for forming difluoromethylenated products 2 (Scheme 1a). It is well-known that gem-difluoroalkenes are susceptible to nucleophilic attack at the difluoro position due to the inductive effect of the two strong electron-withdrawing F atoms (Scheme 1b).3 However, the β-difluoro anion intermediate thus generated is unstable and rapidly undergoes β-fluoride elimination to form monofluoroalkenes as the major products, resulting in a net C–F bond functionalization.4
Scheme 1. Hydrofunctionalization of gem-Difluoroalkenes.
Progress has been made in tackling the challenge of shifting the reactivity toward hydrofunctionalization of gem-difluoroalkenes via the so-called “fluorine-retentive strategy”.3a Altman and co-workers successfully demonstrated base-catalyzed hydrothiophenolation5 and hydrophenolation5b of trisubstituted difluorostyrenes for the construction of S/O–CF2 bonds (Scheme 1c). Subsequent development by the same group extended the nucleophile scope to alkyl thiols5c and alcohols5d via acid catalysis and photocatalysis, respectively. Nevertheless, intermolecular hydrofunctionalization of gem-difluoroalkenes with heteroatom nucleophiles beyond thiols and alcohols under simple conditions is still very limited.6
We have a continuing interest in using gem-difluoroalkenes as building blocks for valuable fluorinated molecules. Previously, we have developed a series of palladium-catalyzed stereoselective C–F bond functionalizations of tetrasubstituted β,β-difluoroacrylates for the synthesis of monofluoroalkenes (Scheme 1d).7 Herein, we report an unprecedented hydroacetoxylation reaction of the difluoroacrylates, where carboxylic acids can act as effective nucleophiles under catalyst-free conditions. The corresponding carboxylic ester products contain the difluoromethylene unit.
Difluoroalkene 1a was used as a standard substrate in the initial studies (Scheme 2). Heating 1a in methanol at 75 °C resulted in a mixture of hydromethoxylation and monofluoroalkene products in low yields (Scheme 2a), and decomposition of the starting material was observed. In stark contrast, heating 1a in acetic acid gave the desired hydroacetoxylation product 2a in 92% isolated yield (Scheme 2b). The reaction could be scaled up to 2.0 mmol with a similar yield. Furthermore, substrates containing benzyl (2b) and heteroaromatic (2c) groups and even a trisubstituted difluoroacrylate (2d) afforded the desired products in good yields. Regioselective addition of carboxylic acids to alkenes is not a trivial task,8 and no examples of gem-difluoroalkenes are known to the best of our knowledge. We also screened other fluorinated and nonfluorinated alkenes under the same conditions for comparison, which were all unreactive (Scheme 2c). These included gem-difluoroalkenes A and B without the ester group, monofluoroalkenes C and D, and a nonfluorinated alkene as well as gem-dichloro/dibromoalkene. The results showed that both the difluoro and the ester functionalities of 1 were important for this reaction.
Scheme 2. Initial Results.
To optimize the reaction conditions further, 1a was reacted with benzoic acid in different solvents and at different temperatures (Table 1). Several organic solvents were screened at 75 °C using 3.0 equiv of the acid, including DMSO, THF, 1,4-dioxane, and MeCN, and the yields were generally quite low (entries 1–4, respectively). A dramatic increase in the yield was observed when the reaction temperature was increased to 150 °C (entries 5 and 6). Alternatively, increasing the amount of acid to 10 equiv could also maintain a good yield at a lower temperature of 75 °C (entry 7). These two sets of conditions (entries 6 and 7) were used in the subsequent exploration of the reaction scope.
Table 1. Optimization of the Reactiona.
| entry | X | solvent | temp (°C)/time (h) | yield (%)b |
|---|---|---|---|---|
| 1 | 3.0 | DMSO | 75/12 | 27 |
| 2 | 3.0 | THF | 75/12 | 37 |
| 3 | 3.0 | 1,4-dioxane | 75/12 | 9 |
| 4 | 3.0 | MeCN | 75/12 | 26 |
| 5 | 3.0 | MeCN | 120/48 | 61 |
| 6 | 3.0 | MeCN | 150/48 | 93 |
| 7 | 10 | MeCN | 75/48 | 90 |
Unless specified otherwise, reactions were carried out using 0.1 mmol of 1a in solvent (0.2 M).
Determined by 19F NMR analysis using benzotrifluoride as an internal standard.
Carboxylic acids are inexpensive, readily available, and structurally diverse commodities. By employing various carboxylic acids as nucleophiles under the optimized conditions, tetrasubstituted gem-difluoroalkenes 1 were smoothly transformed into gem-difluoromethylenated esters 2e–t (Scheme 3). In most of the examples, a reaction temperature of 75 °C was sufficient to afford good yields (condition A).
Scheme 3. Addition of Carboxylic Acids to Tetrasubstituted gem-Difluoroalkenes.
Unless specified otherwise, reactions were carried out using 0.2 mmol of 1 for 48 h. Isolated yields. Condition A: 10 equiv of carboxylic acid at 75 °C. Condition B: 3.0 equiv of carboxylic acid at 150 °C.
Some carboxylic acids required 150 °C to increase solubility (condition B, 2f and 2g). The functional group tolerability of this reaction was excellent. Aromatic carboxylic acids containing electron-rich (2f), electron-poor (2g), and sterically hindered (2h and 2i) substituents were tolerated. Heteroaryl (2j), vinyl (2k), and alkyl (2l) carboxylic acids were also compatible. Cyclopropane ring (2m) and carbamate (2n) moieties were preserved in the reaction. However, an electron-deficient carboxylic acid gave a poor yield (2o). Substituent R1 of difluoroalkene 1 could also be tuned, as shown in products containing heteroaryl (2p) and different aryl (2q–s) groups. On the contrary, the benzyl-substituted substrate (2t) gave a yield lower than the aryl ones even at increased temperatures.
Furthermore, the reaction scope could be extended to trisubstituted gem-difluoroalkene 3a in equally good yields (Scheme 4). Various aryl (4a), heteroaryl (4b and 4c), and alkyl (4d) carboxylic acids were compatible. Drug molecules such as ibuprofen (4e), isoxepac (4f), and dehydrocholic acid (4g) were employed to synthesize the CF2-containing ester products. This late-stage functionalization strategy could be attractive in medicinal chemistry for identifying new fluorinated lead compounds.
Scheme 4. Addition of Carboxylic Acids to a Trisubstituted gem-Difluoroalkene.
Unless specified otherwise, reactions were carried out using 0.2 mmol of 3a for 24 h. Isolated yields.
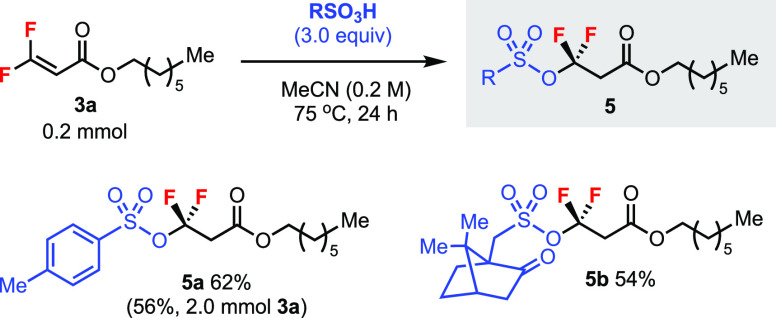
Intriguingly, trisubstituted gem-difluoroalkene 3a could react with sulfonic acids to generate products 5 containing the RSO3–CF2 bond (Scheme 5). Thus, p-toluenesulfonic acid and camphor-10-sulfonic acid led to products 5a (even at 2.0 mmol scale) and 5b, respectively, in moderate yields. In comparison, a tetrasubstituted substrate such as 1a did not react with these sulfonic acids. We also tested a BINOL-derived phosphoric acid with 3a and 1a but found no reaction.
Scheme 5. Addition of Sulfonic Acids to a Trisubstituted gem-Difluoroalkene.
In conclusion, we have discovered a straightforward method for the synthesis of a novel class of gem-difluoromethylenated compounds containing RCO2–CF2 or RSO3–CF2 bonds. Readily available carboxylic acids can undergo regioselective addition to tetrasubstituted and trisubstituted difluoroacrylates with simple heating. No catalysts or additives were required, and the reaction scope could be extended to sulfonic acids. The reaction mechanism is not completely clear at the moment. Both gem-difluoro and ester moieties of 1 were required for the reaction (cf. Scheme 2). We proposed a concerted mechanism (see the Supporting Information) in which the ester group of 1 is protonated by the carboxylic acid while the oxygen of the acid attacks the β-carbon, which is made partially positive due to the two strongly electron-withdrawing F atoms. The resulting enol intermediate tautomerizes to final product 2. This pathway would also avoid the β-F elimination side product. Further exploration of other types of heteroatom nucleophiles is ongoing in our laboratory.
Acknowledgments
This work was supported by the Research Grants Council of Hong Kong (CUHK 14303823) and the Chinese University of Hong Kong (Faculty of Science - Direct Grant for Research). The authors also thank the Key Laboratory of Organofluorine Chemistry, Shanghai Institute of Organic Chemistry, Chinese Academy of Sciences, for funding.
Data Availability Statement
The data underlying this study are available in the published article and its Supporting Information.
Supporting Information Available
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acs.orglett.4c00095.
Experimental procedures, optimization data, characterization data, and spectral data (PDF)
The authors declare no competing financial interest.
Supplementary Material
References
- a Krishnamoorthy S.; Prakash G. K. S. Silicon-Based Reagents for Difluoromethylation and Difluoromethylenation Reactions. Synthesis 2017, 49, 3394–3406. 10.1055/s-0036-1588489. [DOI] [Google Scholar]; b Yerien D. E.; Barata-Vallejo S.; Postigo A. Difluoromethylation Reactions of Organic Compounds. Chem. - Eur. J. 2017, 23, 14676–14701. 10.1002/chem.201702311. [DOI] [PubMed] [Google Scholar]; c Sap J. B. I.; Meyer C. F.; Straathof N. J. W.; Iwumene N.; am Ende C. W.; Trabanco A. S.; Gouverneur V. Late-Stage Difluoromethylation: Concepts, Developments and Perspective. Chem. Soc. Rev. 2021, 50, 8214–8247. 10.1039/D1CS00360G. [DOI] [PubMed] [Google Scholar]
- Holovach S.; Melnykov K. P.; Skreminskiy A.; Herasymchuk M.; Tavlui O.; Aloshyn D.; Borysko P.; Rozhenko A. B.; Ryabukhin S. V.; Volochnyuk D. M.; Grygorenko O. O. Effect of gem-Difluorination on the Key Physicochemical Properties Relevant to Medicinal Chemistry: The Case of Functionalized Cycloalkanes. Chem. - Eur. J. 2022, 28, e20220033 10.1002/chem.202200331. [DOI] [PubMed] [Google Scholar]
- a Sorrentino J. P.; Altman R. A. Fluorine-Retentive Strategies for the Functionalization of gem-Difluoroalkenes. Synthesis 2021, 53, 3935–3950. 10.1055/a-1547-9270. [DOI] [PMC free article] [PubMed] [Google Scholar]; b Liu C.; Zeng H.; Zhu C.; Jiang H. Recent Advances in Three-Component Difunctionalization of gem-Difluoroalkenes. Chem. Commun. 2020, 56, 10442–10452. 10.1039/D0CC04318D. [DOI] [PubMed] [Google Scholar]; c Nguyen B. V.; Burton D. J. A New Route for the Preparation of Substituted 2,2-Difluorostyrenes and A Convenient Route to Substituted (2,2,2-Trifluoroethyl)benzenes. J. Org. Chem. 1997, 62, 7758–7764. 10.1021/jo971019w. [DOI] [Google Scholar]; d Riss P. J.; Aigbirhio F. I. A simple, Rapid Procedure for Nucleophilic Radiosynthesis of Aliphatic [18F]Trifluoromethyl Groups. Chem. Commun. 2011, 47, 11873–11875. 10.1039/c1cc15342k. [DOI] [PubMed] [Google Scholar]
- a Fujita T.; Fuchibe K.; Ichikawa J. Transition-Metal-Mediated and -Catalyzed C-F Bond Activation by Fluorine Elimination. Angew. Chem., Int. Ed. 2019, 58, 390–402. 10.1002/anie.201805292. [DOI] [PubMed] [Google Scholar]; b Zhang X.; Cao S. Recent Advances in the Synthesis and C-F Functionalization of gem-Difluoroalkenes. Tetrahedron Lett. 2017, 58, 375–392. 10.1016/j.tetlet.2016.12.054. [DOI] [Google Scholar]; c Amii H.; Uneyama K. C-F. Bond Activation in Organic Synthesis. Chem. Rev. 2009, 109, 2119–2183. 10.1021/cr800388c. [DOI] [PubMed] [Google Scholar]
- a Orsi D. L.; Easley B. J.; Lick A. M.; Altman R. A. Base Catalysis Enables Access to α. α-Difluoroalkylthioethers. Org. Lett. 2017, 19, 1570–1573. 10.1021/acs.orglett.7b00386. [DOI] [PMC free article] [PubMed] [Google Scholar]; b Orsi D. L.; Yadav M. R.; Altman R. A. Organocatalytic Strategy for Hydrophenolation of gem-Difluoroalkenes. Tetrahedron 2019, 75, 4325–4336. 10.1016/j.tet.2019.04.016. [DOI] [PMC free article] [PubMed] [Google Scholar]; c Sorrentino J. P.; Orsi D. L.; Altman R. A. Acid-Catalyzed Hydrothiolation of gem-Difluorostyrenes to Access α,α-Difluoroalkylthioethers. J. Org. Chem. 2021, 86, 2297–2311. 10.1021/acs.joc.0c02440. [DOI] [PMC free article] [PubMed] [Google Scholar]; d Herrick R. M.; Abd El-Gaber M. K.; Coy G.; Altman R. A. A Diselenide Additive Enables Photocatalytic Hydroalkoxylation of gem-Difluoroalkenes. Chem. Commun. 2023, 59, 5623–5626. 10.1039/D3CC01012K. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Related examples using S and O nucleophiles:; a Li J.; Xu C.; Wei N.; Wang M. Synthesis of 2,2-Difluorinated 4-Isoflavanols/4-Thioisoflavanols via a Base-Catalyzed [4 + 2] Annulation Reaction of gem-Difluoroolefins. J. Org. Chem. 2017, 82, 11348–11357. 10.1021/acs.joc.7b01635. [DOI] [PubMed] [Google Scholar]; b Fang X.; Wu Y.-M.; Deng J.; Wang S.-W. The Nucleophilic Addition Reactions of Difluorovinyl Tosylates with O- and S-nucleophiles. J. Fluor. Chem. 2004, 125, 1481–1484. 10.1016/j.jfluchem.2004.05.014. [DOI] [Google Scholar]; c Liu C.; Zhu C.; Cai Y.; Jiang H. Solvent-Switched Oxidation Selectivities with O2: Controlled Synthesis of α-Difluoro(thio)methylated Alcohols and Ketones. Angew. Chem., Int. Ed. 2021, 60, 12038–12045. 10.1002/anie.202017271. [DOI] [PubMed] [Google Scholar]; d Yang E.; Reese M. R.; Humphrey J. M. Synthesis of α,α-Difluoroethyl Aryl and Heteroaryl Ethers. Org. Lett. 2012, 14, 3944–3947. 10.1021/ol301696x. [DOI] [PubMed] [Google Scholar]; e Han X.; Liu X.; Len C.; Liu L.; Wang D.; Zhang Y.; Duan X. H.; Hu M. Photoredox-Catalyzed gem-Difluoromethylenation of Aliphatic Alcohols with 1,1-Difluoroalkenes to Access α,α-Difluoromethylene Ethers. J. Org. Chem. 2023, 88, 12744–12754. 10.1021/acs.joc.3c01428. [DOI] [PubMed] [Google Scholar]; f Timperley C. M.; Waters M. J.; Greenall J. A. Fluoroalkene Chemistry: Part 3. Reactions of Arylthiols with Perfluoroisobutene, Perfluoropropene and Chlorotrifluoroethene. J. Fluor. Chem. 2006, 127, 249–256. 10.1016/j.jfluchem.2005.11.008. [DOI] [Google Scholar]
- a Ma Q.; Wang Y.; Tsui G. C. Stereoselective Palladium-Catalyzed C-F Bond Alkynylation of Tetrasubstituted gem-Difluoroalkenes. Angew. Chem., Int. Ed. 2020, 59, 11293–11297. 10.1002/anie.202002219. [DOI] [PubMed] [Google Scholar]; b Wang Y.; Qi X.; Ma Q.; Liu P.; Tsui G. C. Stereoselective Palladium-Catalyzed Base-Free Suzuki-Miyaura Cross-Coupling of Tetrasubstituted gem-Difluoroalkenes: An Experimental and Computational Study. ACS Catal. 2021, 11, 4799–4809. 10.1021/acscatal.0c05141. [DOI] [Google Scholar]; c Wang Y.; Tsui G. C. Stereoselective Palladium-Catalyzed C–F Bond Alkenylation of Tetrasubstituted gem-Difluoroalkenes via Mizoroki–Heck Reaction. Org. Lett. 2023, 25, 6217–6221. 10.1021/acs.orglett.3c02452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- a Perkowski A. J.; Nicewicz D. A. Direct Catalytic Anti-Markovnikov Addition of Carboxylic Acids to Alkenes. J. Am. Chem. Soc. 2013, 135, 10334–10337. 10.1021/ja4057294. [DOI] [PMC free article] [PubMed] [Google Scholar]; b Nicewicz D.; Hamilton D. Organic Photoredox Catalysis as a General Strategy for Anti-Markovnikov Alkene Hydrofunctionalization. Synlett 2014, 25, 1191–1196. 10.1055/s-0033-1340738. [DOI] [PMC free article] [PubMed] [Google Scholar]; c Petrosyan A.; Zach L.; Taeufer T.; Mayer T. S.; Rabeah J.; Pospech J. Pyrimidopteridine-Catalyzed Photo-Mediated Hydroacetoxylation. Chem. - Eur. J. 2022, 28, e202201761 10.1002/chem.202201761. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The data underlying this study are available in the published article and its Supporting Information.