Abstract
Objective:
Determine risk of autoimmune disease in research identified cases of ASD compared with referents using a longitudinal, population-based birth cohort.
Methods:
ASD incident cases were identified from a population-based birth cohort of 31,220 individuals. Inclusive ASD definition based on DSM-IV-TR autistic disorder, Asperger’s Disorder, and pervasive developmental disorder not otherwise specified was used to determine ASD cases. For each ASD case, two age, sex-matched referents without ASD were identified. Diagnosis codes assigned between birth and December 2017 were electronically obtained. Individuals were classified as having an autoimmune disorder if they had at least two diagnosis codes more than 30 days apart. Cox proportional hazards models were fit to estimate the hazard ratio (HR) between ASD status and autoimmune disorder.
Results:
Of 1014 ASD cases, 747 (73.7%) were male. 50 ASD cases and 59 of the 1:2 matched referents were diagnosed with first autoimmune disorder at median age of 14 years and 17.1 years, respectively. ASD cases had increased risk of autoimmune disease compared to matched referents (HR 1.74; 95% CI 1.21–2.52). The increased risk was statistically significant among males (HR 2.01; 95% CI 1.26 – 3.21) but not among the smaller number of female subjects (HR 1.38; 95% CI 0.76–2.50).
Conclusion:
This study provides evidence from a longitudinal, population-based birth cohort for co-occurrence of ASD and autoimmune disorders. Thus, children with ASD should be monitored for symptoms of autoimmune disease and appropriate workup initiated.
Keywords: Autism spectrum disorder, autoimmune disease, longitudinal population based birth cohort
INTRODUCTION
Both autism spectrum disorder (ASD) and autoimmune disease were first described in the mid-20th century.1,2 ASD is a neurodevelopmental disability defined by deficits in social communication and social interaction in conjunction with restricted and repetitive patterns of interests and behavior.3 Autoimmune diseases are defined by inappropriate immune system responses that lead to the body attacking its own cells, tissues, and organs due to failure to distinguish self from nonself.4 The prevalence of ASD has increased over time, and it has been reported to now occur in 1 in 36 children aged 8 years, with a male-to-female ratio of approximately 3.8 to 1.5 The prevalence of autoimmune disease has also increased over time; the estimated prevalence of the more than 100 documented autoimmune diseases is 3 to 5%, diagnosis generally occurs before 50 years of age and women are disproportionately affected.6
An evolving theory of a connection between immunological dysfunction and autism was introduced into the literature in the 1970s.7 Previous publications have investigated the relationships between ASD and autoimmunity, focusing on immune mediated conditions, family trends, and evidence of immune dysfunction.8–18 However, these studies have been inconsistent in establishing co-occurrence of autoimmune disease in ASD and in defining potential differences in autoimmune disease rates among males and females diagnosed with ASD. Additionally, prior studies have been limited due to a lack of comprehensive methodology in identification of ASD cases.8–11, 15–16 To address these previous limitations, the aim of this study was to test the hypothesis that autoimmune diseases are more prevalent among individuals with ASD compared with age- and sex-matched referents using rigorous methodology to ascertain all research-identified cases of ASD from a longitudinal, population-based birth cohort.
METHODS
Study Setting
Our study utilized a population-based birth cohort comprised of individuals residing in Olmsted County, Minnesota. A unique characteristic of this cohort is the ability to capture all residents in the county across age, ethnicity, sex, socioeconomic status, setting of care delivery, and insurance status. The population in this county is reflective of the United States population, according to the 2000 Census, regarding male sex (49.1% vs 49.1%) and median age percentages (49.1% vs 49.1%).19 This population is less racially diverse than the U.S. population (white, 90.3% vs 75.1 %), which decreases confounding that may be due to race and ethnicity.19
Study Resources
The study population was compiled from birth certificate records for Olmsted County, Minnesota and the resources of the Rochester Epidemiology Project (REP). The REP provides a centralized resource of longitudinal medical data that includes clinical documentation from all outpatient primary care and medical subspecialty encounters, emergency department visits, inpatient hospitalizations, laboratory results, social services reports, and birth/death certificate data from the Mayo Clinic, Olmsted Medical Center, their three affiliated hospitals, and several smaller care providers.19–21 Diagnoses assigned at each encounter are coded and maintained in continuously updated electronic files. This study was approved by the institutional review boards of Olmsted Medical Center, Mayo Clinic, and Baylor College of Medicine. Access to school records was made possible through a contractual research agreement between Mayo Clinic, the Independent School District (ISD) No. 535 school board, and equivalent authorities governing private schools to access the cumulative school records of all birth cohort members registered at any of the 41 private, parochial, or public schools in the community of Rochester, Minnesota.
Study Population –Olmsted County Birth Cohort
This population-based birth cohort included 43,215 children who were born between January 1, 1976 and December 31, 2000 to mothers who were residents of Olmsted County at the time of the child’s birth. Of the 43,215 children included, medical record access for research purposes was available for 39,890 children in accordance with Minnesota state privacy law, statute 144.335. This study’s target population included 31,220 children who still lived in Olmsted County at or after 3 years of age. This age cut-off was used for the study population as behavioral, social, and communication characteristics of ASD are more consistently recognized at or beyond this age.22
Identification of Cases with Research-Defined Autism Spectrum Disorder
ASD cases among the study population were identified using both comprehensive medical and school records, and these case identification methods have been previously published in detail.22 ASD incident cases were based on operational research definitions and were identified by a team that consisted of two developmental-behavioral pediatricians, a senior child psychologist, an MD epidemiologist, and a speech language pathologist. Three steps were utilized when evaluating both medical and school records. First, individuals with selected relevant neurodevelopmental and psychiatric disorders based on records were identified. Next, this group was narrowed down to potential ASD cases (P-ASD). These P-ASD subject’s records were abstracted through a systematic multistage process allowing accumulation of all details related to ASD symptoms. The last step consisted of determining ASD incident case status based on DSM-IV-TR criteria for autistic disorder (AD), Asperger disorder, and pervasive developmental disorder-not otherwise specified. Although fewer people meet DSM-5 ASD diagnostic criteria than DSM-IV-TR criteria, the symptom profiles of individuals who meet DSM-5 criteria and those who meet DSM-IV-TR criteria (but not DSM-5 criteria) are very similar, and many studies suggest a high rate of agreement between the 2 sets of criteria.23–26 We created an inclusive research identified ASD case definition for the methods of this study, which includes all cases that met research criteria (signs and symptoms based on retrospective chart review) of autistic disorder, Asperger’s Disorder, and pervasive developmental disorder not otherwise specified. ASD incident cases were based on operational research definitions and were identified by a team that consisted of two developmental-behavioral pediatricians, a senior child psychologist, an MD epidemiologist, and a speech language pathologist. Additionally, in evaluation of medical and school records, clinical diagnoses were also abstracted based on HICDA and ICD-9 diagnosis codes. Of the 1056 individuals with research-identified ASD who were identified as part of this prior work, 1014 had provided research authorization at the time of this study and were included in this analysis.
Identification of Age- and Sex-Matched Referents
For each of the 1014 individuals with research-identified ASD, two age- and sex-matched referents without research-identified ASD were identified from the same birth cohort. The male to female ratio among ASD in our study is approximately 3:1, and this ratio has been shown in previous studies as a more accurate representation of the sex ratios in ASD.27 The referents were randomly selected from the pool of individuals in the birth cohort of the same sex, born within 30 days of the patients with ASD, still in the community at the time when the matched case met the research criteria for ASD, and who had research authorization at the time of this study.
Identification of Autoimmune Disease
We chose to study a group of autoimmune diseases that have been associated with circulating autoantibodies (See the 33 categories in the Supplemental Table). For each of the 1014 individuals with research-identified ASD and 2028 matched referents, clinical diagnosis codes for these diseases that were assigned by a REP-affiliated practitioner from birth through December 31, 2017 were electronically obtained. An individual was classified by the study statistician (ALW), blinded to the ASD status, as having an autoimmune disease if they had at least two diagnosis codes within the same category > 30 days apart. In the study population we are reporting on the initial autoimmune disease. The date of the later diagnosis was used as the event date.
Statistical Analysis
Analyses were performed using SAS (version 9.4; SAS Institute, Inc.; Cary, NC). The cumulative incidence of being diagnosed with an autoimmune disease, as a function of time since birth, was estimated for each group (ASD cases, referents) using the Kaplan-Meier estimator taking into account the varying duration of follow-up for each individual. Cox proportional hazards models were fit to estimate the association between ASD status and diagnosis of an autoimmune disease; associations were summarized using the hazard ratio (HR) and corresponding 95% confidence interval (CI). All calculated p-values were two-sided and p-values less than 0.05 were considered statistically significant.
RESULTS
The demographic characteristics of the 1014 research-identified ASD cases and 2028 age- and sex-matched referents are summarized in Table 1. The median age at last clinical follow-up for any reason was 25.1 (interquartile range, 20.7–29.6) years for the ASD cases and 23.7 (interquartile range, 19.8–28.0) years for the referents. Fifty-five ASD cases and 59 of the 1:2 matched referents were diagnosed with their first autoimmune disease at a median age of 14.0 (IQR 9.9–20.8) years and 17.1 (IQR 10.8–22.6) years, respectively. Among the ASD cases, the most common initial autoimmune diseases were diabetes mellitus (n=13) and alopecia areata (n=12). The most common initial autoimmune diseases identified among the referents were alopecia areata (n=11) and psoriasis (n=10) (Table 2).
Table 1.
Demographic characteristics of study subjects
| Characteristic | Research-identified ASD cases (N=1014) | Age- and sex-matched referents (N=2028) |
|---|---|---|
|
| ||
| Birth year | ||
| 1976–1980 | 69 (6.8%) | 139 (6.9%) |
| 1981–1985 | 161 (15.9%) | 321 (15.8%) |
| 1986–1990 | 286 (28.2%) | 572 (28.2%) |
| 1991–1995 | 265 (26.1%) | 530 (26.1%) |
| 1996–2000 | 233 (23.0%) | 466 (23.0%) |
| Gender | ||
| Male | 747 (73.7%) | 1494 (73.7%) |
| Female | 267 (26.3%) | 534 (26.3%) |
| Race | ||
| American Indian or Alaskan Native | 9 (0.9%) | 7 (0.3%) |
| Asian | 16 (1.6%) | 60 (3.0%) |
| Black or African American | 26 (2.6%) | 39 (1.9%) |
| Hawaiian Pacific Islander | 4 (0.4%) | 4 (0.2%) |
| Other | 33 (3.3%) | 50 (2.5%) |
| Unknown or chose not to disclose | 24 (2.4%) | 88 (4.3%) |
| White | 902 (89.0%) | 1780 (87.8%) |
Table 2.
Type of autoimmune disease first identified among study subjects
| Research-identified ASD cases | Age- and- sex-matched referents | |||
|---|---|---|---|---|
|
|
|
|||
| Autoimmune disease | Females (N=19) | Males (N=36) | Females (N=25) | Males (N=34) |
|
| ||||
| Diabetes Mellitus | 2 | 11 | 2 | 4 |
| Crohn’s disease | 1 | 2 | 1 | 3 |
| Ulcerative colitis | 0 | 2 | 2 | 3 |
| Autoimmune hepatitis | 0 | 0 | 1 | 0 |
| Multiple sclerosis | 1 | 0 | 0 | 1 |
| Celiac disease | 2 | 5 | 2 | 2 |
| Psoriasis | 2 | 3 | 3 | 7 |
| Alopecia Areata | 5 | 7 | 3 | 8 |
| Vitiligo | 0 | 1 | 2 | 2 |
| Rheumatoid arthritis | 1 | 2 | 1 | 1 |
| Lupus | 0 | 0 | 1 | 0 |
| Scleroderma | 1 | 0 | 0 | 0 |
| Autoimmune anemia | 0 | 1 | 0 | 0 |
| Autoimmune thyroiditis | 2 | 1 | 5 | 0 |
| Graves’ disease | 1 | 0 | 1 | 2 |
| Purpura | 1 | 1 | 1 | 0 |
| Dermatomyositis | 0 | 0 | 0 | 1 |
Individuals who met research criteria for ASD were significantly more likely to have an autoimmune disease compared to the referents (HR 1.74; 95% CI 1.21–2.52; p=0.003; Figure 1). The association was stronger among males (HR 2.01; 95% CI 1.26–3.21; p=0.004) than among females (HR 1.38; 95% CI 0.76–2.50; p=0.30); however, the study did not detect a significant interaction (p=0.33) (Figure 2). By 30 years of age, the cumulative incidence of having at least one autoimmune disease was 5.3% versus 3.4% among male ASD cases versus male referents and 8.8% versus 4.8% among female ASD cases versus female referents.
Figure 1:
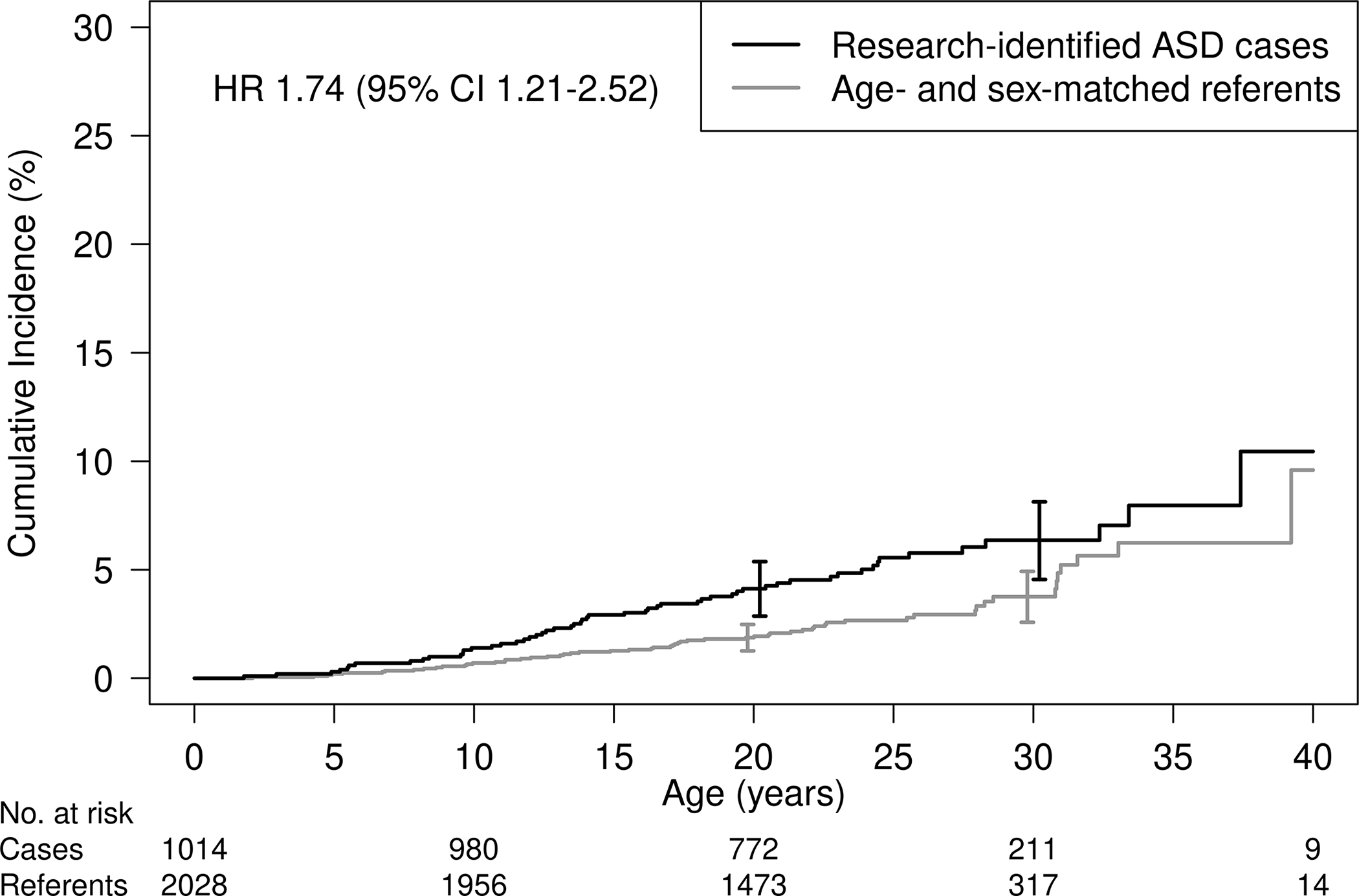
Cumulative incidence of autoimmune diseases in individuals with ASD versus age- and sex-matched referents
Figure 2:
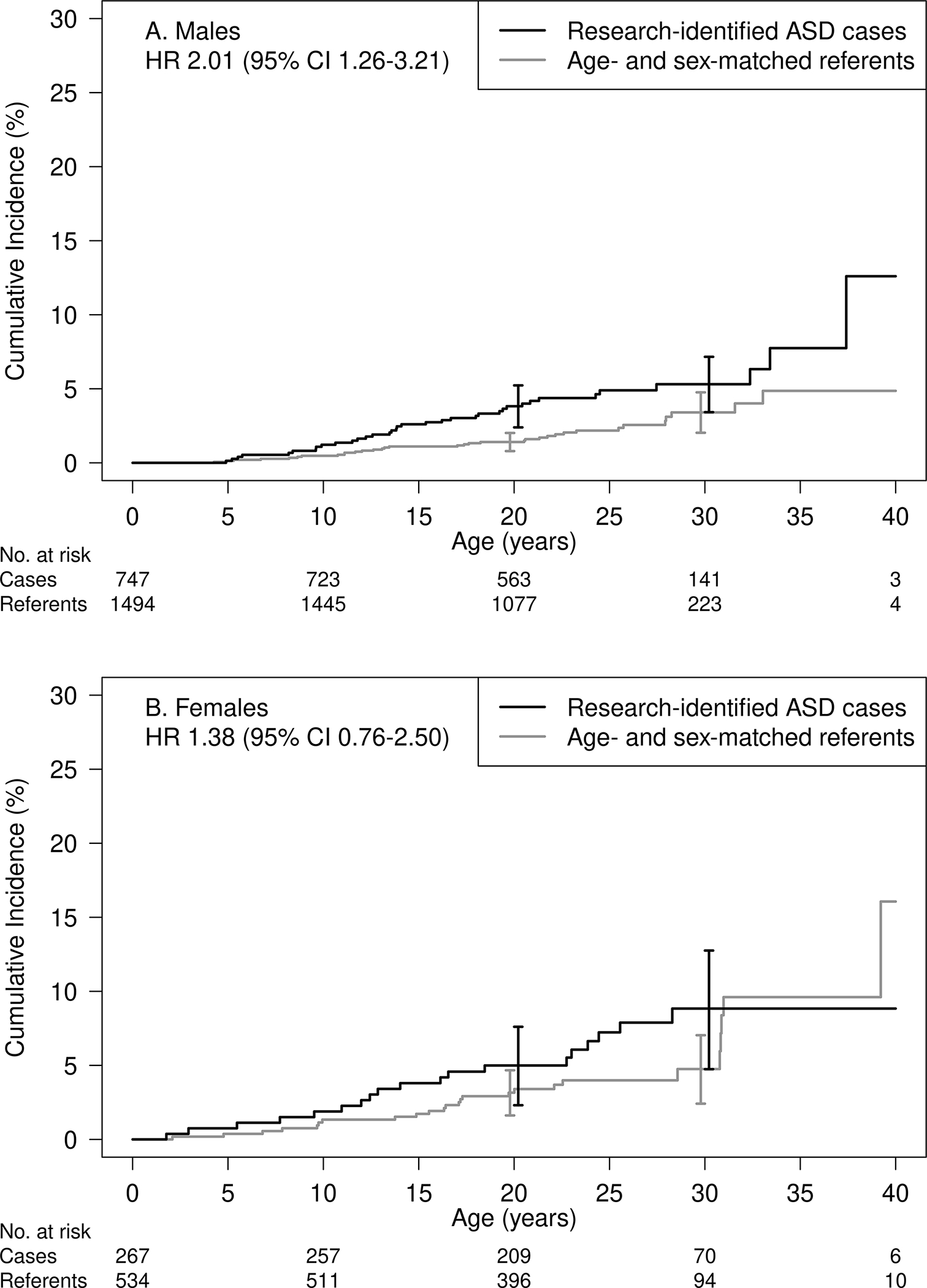
Cumulative incidence of autoimmune disease in individuals with ASD versus age- and sex-matched referents stratified by gender
DISCUSSION
We found that individuals who met research criteria for ASD were significantly more likely to have an autoimmune disease diagnosis by 35 years of age compared to age and sex-matched referents. By the age of 35 years, individuals with ASD were diagnosed with an autoimmune disease at approximately 1.8 times the rate in the control group. The association of ASD and co-occurring autoimmune disease was stronger among males than females, though this difference was not statistically significant. The significant relationship found among males, but not females is likely attributable to a larger male cohort with ASD among our study population. Regarding specific initial autoimmune diseases, we observed a higher number of cases of Type I DM, celiac disease, alopecia areata, and rheumatoid arthritis in individuals with ASD, compared to prevalence of these specific autoimmune diseases in referents (Table 2).
Autoimmune diseases generally occur in the young (taking prevalence into account the most common age of onset appears to be in the 20–29 year age group), are generally more common in females, and there are known associations among many autoimmune diseases.28 In the 1970’s, the first publication discussing an association between autoimmune diseases and autism was published.7 This publication was a case report in which a child with autism was noted to have an extensive family history of autoimmune conditions and was diagnosed with Addison’s disease.7 Since that time, there have been some population-based studies that have further evaluated this relationship by investigating the prevalence of comorbid autoimmune disease in individuals with ASD, but it should be noted that these studies were not consistent in detailing how ASD was diagnosed.8–11,14 One prior longitudinal population-based study in California reported that children with ASD had a higher prevalence of psoriasis compared to controls,10 but our study did not find a similar increased prevalence of psoriasis in the ASD group (Table 2). Results from studies investigating an association between ASD and Type I DM have been mixed.17 One study in Colorado found that the prevalence of ASD among children with Type I Diabetes was comparable to the general population in the state (1.16% CI 0.96–1.26).18 A systematic review and meta-analysis evaluating the association between autism and diabetes was published in 2022. This study found that when considering adjusted ORs, significant associations among unadjusted ORs became insignificant, which highlights that existing evidence across studies may be confounded by various clinical factors.17 This supports the notion that caution that should be taken when associations among ASD and diabetes are claimed as there is a lack of strong evidence and further large population-based studies are needed.17 An epidemiological study from Taiwan assessing the prevalence of allergic and autoimmune diseases in individuals with ASD found a statistically significant increase in prevalence of Type 1 DM in individuals with ASD,11 and a population-based study from Boston reported an increased prevalence of Type 1 DM and inflammatory bowel disease (IBD) in individuals with ASD compared to the general population.8 In contrast to the Boston study, a study from Denmark21 and a previous study using REP data22 to evaluate GI disorders in children and adolescents with ASD did not demonstrate an increased prevalence of IBD in the ASD group, and in the present study, we again did not observe any difference in the prevalence of IBD in ASD cases versus referents (Table 2). It is unclear why variation in findings have been noted between studies, however one variable to note is the racial variation included in the patient populations of each study.29 Variations in prevalence of autoimmune diseases by race may be due to different environmental exposures, genetics, social determinants of health including effects of racism, or a combination of all of these.29
Prior studies have been limited by a lack of comprehensiveness in methodology for identifying ASD cases, while a strength of our study was our review of comprehensive data present in all medical and school records, rather than exclusive reliance on ICD codes.22 Our study was also unique in examining the differential prevalence of initial autoimmune diseases among males and females. We observed that the association of higher prevalence of autoimmune diseases in ASD was stronger among the males than females; however, the study did not detect a statistically significant interaction (Figure 2). This was a particularly interesting finding, since ASD is about 4 times as common in males,5 but autoimmune diseases are known to be more common among females.6 In the United States, about 80% of the 8.5 million individuals with autoimmune diseases are women.6 However, we might not have observed a difference in the prevalence of autoimmune disease in females with ASD due to the lower number of females in the ASD group.
It is important for medical providers to be aware of the comorbid medical conditions that individuals with ASD are at an increased risk for developing. These comorbidities include mental health conditions, such as depression, anxiety, bipolar disorder, attention-deficit/ hyperactivity disorder, and substance abuse.30–31 Additional co-occurring medical conditions associated with ASD include seizures, sleep disorders, and intellectual disability.22,33 This study highlights that autoimmune disorders belong on this list of conditions for medical providers to be aware of when screening for comorbid conditions for individuals with ASD. While this study does add evidence to an association between ASD and autoimmunity, no conclusions can be drawn regarding causal association or therapeutic implications.
After the association of immune dysfunction in patients with ASD was introduced, an alarming utilization of immunotherapy for children with autism emerged in medical practice in children with ASD without any known autoimmune disease. Medications that have been used, despite a lack of controlled trials demonstrating either their safety or efficacy, include but are not limited to intravenous immunoglobulin, oral corticosteroids, and adrenocorticotrophic hormone.34,35 While our study does report an association between autism and autoimmune disease, it does not imply causation. ASD is a behavioral diagnosis, and treatment should be targeted at reducing the core deficits, maximizing functional independence, and minimizing problematic behaviors.32 Immune modulating medications should only be used when clinically indicated for specific autoimmune diseases, based on clinical and diagnostic findings.
There are some limitations to our study that should be addressed in future research. Although we utilized a large longitudinal population-based birth cohort, we found a relatively small number of patients with both ASD and an autoimmune condition. Future studies could benefit from using larger populations, perhaps spanning across multiple regions. Additionally, ASD classification was based on a comprehensive retrospective record review process, but no direct in-person evaluations were performed to verify diagnostic validity. While this method may inadvertently lead to missed ASD cases in the community, this likelihood was decreased by utilizing both comprehensive educational and medical records, extensive training of the research assistants who performed record abstraction, and use of precisely defined DSM-IV-TR based operational research criteria and an extensive data dictionary of pertinent descriptive phrases. This process also has the potential to lead to an over-identification of cases; however, when reviewing signs and symptoms, records were searched for additional occurrences, contradictory information, and situational qualifiers.22 Whenever a question arose regarding validity of signs/symptoms contributing to the ASD criteria, it was brought to the full research team for review. This method was an advantage of our manual abstraction protocol over the use of automated algorithms based on text abstracted from electronic health records.22 As our study population does not match the racial and socio-economic composition of all communities in the United States, our data might not be generalizable to all populations. While this is true, it should also be noted that the population of Olmsted County has numerous intrinsic research benefits, including that it is stable and therefore allows for consistent long term follow up, and results of studies from this population have been reflective of national data.19 While our study was unique to evaluate sex differences in the prevalence of autoimmune diseases among individuals with ASD, despite our large cohort, we were unable to gain statistical significance for our female group, and we did not detect a significant interaction. This may have been due to having too few females determined to have research identified ASD among our study population. Additionally, our study only tracked for development of the first autoimmune disease for each subject until age 35 years. These data may therefore not truly capture the full gamut of autoimmune diseases in this population, as many do not manifest until later into adulthood. Future studies would benefit from tracking autoimmune diseases through the seventh decade of life to accurately capture the majority of autoimmune diagnoses.36 It should also be noted that diagnosis codes alone were used for the determination of autoimmune disease, which could potentially lead to patient misclassification. While some patients might have been misclassified, our study intentionally included a broad list of autoimmune diseases to fully capture all patients with autoimmune diseases in our study population.
CONCLUSION
This study provides evidence from a longitudinal, population-based birth cohort for a co-occurrence of ASD and clinically diagnosed autoimmune diseases. While we noted increased co-occurrence of Type I DM, celiac disease, alopecia areata, and rheumatoid arthritis in individuals with ASD, larger studies will be needed to further investigate whether these are significant differences. This information will be helpful for future pediatric screening practices. Additionally, contrary to patterns of autoimmune diseases in the general population, we found a stronger interaction among males than females, although, this interaction was not statistically significant. This finding also warrants further study in larger populations. Based on our study findings, patients with ASD should be monitored closely for signs and symptoms of autoimmune disease and appropriate referral and workup should be initiated when indicated.
Supplementary Material
Funding/Support:
This study was funded by research Grants from the National Institutes of Health, Public Health Service (MH093522 and AG034676).
Role of Funder/Sponsor:
The NIH had no role in the design and conduct of the study.
Footnotes
Conflict of Interest Disclosures (includes financial disclosures): The authors have no disclosures.
Prior Presentation of Study: 2019 Pediatric Academic Societies; 2019 Society for Developmental and Behavioral Pediatrics Annal Meeting
Clinical Trial Registration: None
References
- 1.Kanner L Autistic Disturbance of Affective Contact. Nervous Child: Journal of Psychopathology, Psychotherapy, Mental Hygiene, and Guidance of the Child. 1943(2):217–250. [Google Scholar]
- 2.Linklater S Defining autoimmunity: an epistemological history. In Focus Book Defining autoimmunity: an epistemological history. THE LANCET Diabetes & Endocrinology. 2015(3):321. doi: 10.1016/S2213-8587(15)00032-7. [DOI] [Google Scholar]
- 3.American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. Vol 5th Edition; 2013. 10.1176/appi.books.9780890425596. [DOI]
- 4.Wang L, Wang F-S, Gershwin & ME, et al. Human autoimmune diseases: a comprehensive update. Journal of Internal Medicine. 2015; 278: 369–395. doi: 10.1111/joim.12395. [DOI] [PubMed] [Google Scholar]
- 5.Maenner MJ, Warren Z, Williams AR, et al. Prevalence and Characteristics of Autism Spectrum Disorder Among Children Aged 8 Years — Autism and Developmental Disabilities Monitoring Network, 11 Sites, United States, 2020. MMWR Suppl 2023; 72(Suppl. 2):p1–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.McCarthy M. The “gender gap” in autoimmune disease. Lancet. 2000;356(9235):1088. doi: 10.1016/S0140-6736(05)74535-9. [DOI] [PubMed] [Google Scholar]
- 7.Money J, Bobrow NA, Clarke FC. Autism and autoimmune disease: A family study. Journal of Autism and Childhood Schizophrenia. 1971;1(2):146–160. doi: 10.1007/BF01537954. [DOI] [PubMed] [Google Scholar]
- 8.Kohane IS, McMurry A, Weber G, et al. The co-morbidity burden of children and young adults with autism spectrum disorders. PloS one. 2012;7(4). doi: 10.1371/journal.pone.0033224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Muskens JB, Velders FP, Staal WG. Medical comorbidities in children and adolescents with autism spectrum disorders and attention deficit hyperactivity disorders: a systematic review. European Child and Adolescent Psychiatry. 2017;26(9):1093–1103. doi: 10.1007/s00787-017-1020-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Zerbo O, Leong A, Barcellos L, et al. Immune mediated conditions in autism spectrum disorders. Brain, Behavior, and Immunity. 2015(46):232–236. doi: 10.1016/j.bbi.2015.02.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Chen MH, Su TP, Chen YS, et al. Comorbidity of allergic and autoimmune diseases in patients with autism spectrum disorder: A nationwide population-based study. Research in Autism Spectrum Disorders. 2013;7(2):205–212. doi: 10.1016/j.rasd.2012.08.008. [DOI] [Google Scholar]
- 12.Edmiston E; Ashwood P; Van de Water J. Autoimmunity, Autoantibodies, and Autism Spectrum Disorder. Biol Psychiatry. 2017;81(5):383–390. doi: 10.1016/j.biopsych.2016.08.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Hughes HK, Mills Ko E, Rose D, et al. Immune Dysfunction and Autoimmunity as Pathological Mechanisms in Autism Spectrum Disorders. Frontiers in Cellular Neuroscience. 2018(12). doi: 10.3389/fncel.2018.00405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Spann MN, Timonen-Soivio L, Suominen A, et al. Proband and Familial Autoimmune Diseases Are Associated With Proband Diagnosis of Autism Spectrum Disorders. Journal of the American Academy of Child and Adolescent Psychiatry. 2019; 58 (5): 496–505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Wu S, Ding Y, Wu F, et al. Family history of autoimmune diseases is associated with an increased risk of autism in children: a systematic review and meta-analysis. Neurosci Biobehav Rev. 2015(55):322–32. [DOI] [PubMed] [Google Scholar]
- 16.Lin TY, Lin PY, Su TP, et al. Autistic spectrum disorder, attention deficit hyperactivity disorder, and allergy: Is there a link? A nationwide study. Research in Autism Spectrum Disorders. 2014;8(10):1333–1338. doi: 10.1016/j.rasd.2014.07.009. [DOI] [Google Scholar]
- 17.Cortese S, Gabellone A, Marzulli L, et al. Association between autism spectrum disorder and diabetes - Systematic review and meta-analysis. Neuroscience and Biobehavioral Reviews. (2022) doi: 10.1016/j.neubiorev.2022.104592. [DOI] [PubMed] [Google Scholar]
- 18.Stanek KR, Youngkin EM, Pyle LL, et al. Prevalence, characteristics, and diabetes management in children with comorbid autism spectrum disorder and type 1 diabetes. Pediatr Diabetes. 2019;20(5):645–651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.St Sauver JL, Grossardt BR, Yawn BP, et al. Data resource profile: The Rochester epidemiology project (REP) medical records-linkage system. International Journal of Epidemiology. 2012;41(6):1614–1624. doi: 10.1093/ije/dys195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Melton LJ. History of the Rochester Epidemiology Project. Mayo Clinic proceedings. 1996; 71.3: 266–274.. [DOI] [PubMed] [Google Scholar]
- 21.Melton LJ, Rocca WA, Roger VL. Development of Population Research at Mayo Clinic. Mayo Clinic proceedings. 2014; 89.2: e17–e20. doi: 10.1016/j.mayocp. [DOI] [PubMed] [Google Scholar]
- 22.Myers SM, Voigt RG, Colligan RC, et al. Autism Spectrum Disorder: Incidence and Time Trends Over Two Decades in a Population-Based Birth Cohort. Journal of Autism and Developmental Disorders. 2019;49(4):1455–1474. doi: 10.1007/s10803-018-3834-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Beighley JS, Matson JL, Rieske RD, et al. Differences in Stereotypic Behavior in Adults Diagnosed with Autism Spectrum Disorders Using the DSM-IV-TR and the DSM-5. Journal of developmental and physical disabilities. 2014(26):193–202. doi: 10.1007/s10882-013-9356-y. [DOI] [Google Scholar]
- 24.Beighley JS, Matson JL, Rieske RD, et al. Comparing challenging behavior in children diagnosed with autism spectrum disorders according to the DSM-IV-TR and the proposed DSM-5. Developmental Neurorehabilitation. 2013;16(6):375–381. doi: 10.3109/17518423.2012.760119. [DOI] [PubMed] [Google Scholar]
- 25.Bennett M, Goodall E. A Meta-Analysis of DSM-5 Autism Diagnoses in Relation to DSM-IV and DSM-IV-TR. Rev J Autism Dev Disord. 2016(3):119–124. doi: 10.1007/s40489-016-0070-4. [DOI] [Google Scholar]
- 26.Kim YS, Fombonne E, Koh YJ, et al. A comparison of DSM-IV pervasive developmental disorder and DSM-5 autism spectrum disorder prevalence in an epidemiologic sample. J Am Acad Child Adolesc Psychiatry. 2014; 53(5):500–508. doi: 10.1016/j.jaac.2013.12.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Loomes R, Hull L, Mandy WPL. What Is the Male-to-Female Ratio in Autism Spectrum Disorder? A Systematic Review and Meta-Analysis. J Am Acad Child Adolesc Psychiatry. 2017;56(6):466–474. doi: 10.1016/j.jaac.2017.03.013. [DOI] [PubMed] [Google Scholar]
- 28.Hayter SM, Cook MC. Updated assessment of the prevalence, spectrum and case definition of autoimmune disease. Autoimmun Rev. 2012;11(10):754–65. [DOI] [PubMed] [Google Scholar]
- 29.McLeod DSA, Caturegli P, Cooper DS, et al. Variation in rates of autoimmune thyroid disease by race/ethnicity in US military personnel. JAMA - Journal of the American Medical Association. 2014;311(15):1563–1565. doi: 10.1001/jama.2013.285606. [DOI] [PubMed] [Google Scholar]
- 30.Kirsch AC, Huebner ARS, Mehta SQ, et al. Association of Comorbid Mood and Anxiety Disorders with Autism Spectrum Disorder. JAMA Pediatrics. 2020;174(1):63–70. doi: 10.1001/jamapediatrics.2019.4368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Hossain MM, Khan N, Sultana A, et al. Prevalence of comorbid psychiatric disorders among people with autism spectrum disorder: An umbrella review of systematic reviews and meta-analyses. Psychiatry Res. 2020;287: 112922–112922. doi: 10.1016/j.psychres.2020.112922. [DOI] [PubMed] [Google Scholar]
- 32.Hyman SL, Levy SE, Myers SM. Identification, Evaluation, and Management of Children With Autism Spectrum Disorder. Pediatrics. 2020;145(1). doi: 10.1542/peds.2019-3447. [DOI] [PubMed] [Google Scholar]
- 33.Rydzewska E, Dunn K, Cooper SA. Umbrella systematic review of systematic reviews and meta-analyses on comorbid physical conditions in people with autism spectrum disorder. Br J Psychiatry. 2021;218(1):10–19. [DOI] [PubMed] [Google Scholar]
- 34.Comi AM, Zimmerman AW, Frye VH, et al. Familial clustering of autoimmune disorders and evaluation of medical risk factors in autism. Journal of Child Neurology. 1999;14(6):388–394. doi: 10.1177/088307389901400608. [DOI] [PubMed] [Google Scholar]
- 35.Mead J, Ashwood P. Evidence supporting an altered immune response in ASD. Immunology Letters. 2015;163(1):49–55. doi: 10.1016/j.imlet.2014.11.006. [DOI] [PubMed] [Google Scholar]
- 36.Amador-Patarroyo MJ, Rodriguez-Rodriguez A, Montoya-Ortiz G. How Does Age at Onset Influence the Outcome of Autoimmune Diseases?. Autoimmune Diseases. 2012;2012: 251730–251737. doi: 10.1155/2012/251730. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


