Abstract
Background
The production and distribution of methamphetamine (meth) is often associated with illegal and clandestine laboratories, posing significant challenges for law enforcement and public health efforts. Global concern is growing over meth-related fatalities, as its high potential for abuse and detrimental impact on health make it an important issue in the realm of substance abuse and addiction. This concern has notably increased in Saudi Arabia, where the hot climate adds complexity to the analysis due to challenges posed by putrefaction. There is still an urgent need to enhance the screening capabilities of many toxicology laboratories to determine the cause of death, whether it be due to drug use or natural causes.
Aim
This research aimed to investigate meth concentrations in post-mortem putrefied human solid tissues in a hot climate and comparing meth metabolite concentrations in cases where signs of putrefaction were observed versus those with no signs of putrefaction. The objective is to assist criminal investigations by analyzing meth and its metabolite concentrations.
Methods
This retrospective cohort study involved postmortem samples from human subjects during autopsies conducted between 2016 and 2022. It focused on analyzing meth and its metabolite concentrations using LC-MS/MS analysis. Data on demographics, medical history, age, location, putrefaction, and other drug use were retrieved from medical records.
Results
Out of the 27 reported samples of meth and its metabolite amphetamine in both putrefied and non-putrefied biological fluids and tissues, only 8 (30%) exhibited signs of putrefaction between 2016 and 2022. Despite decomposition, detectable concentrations of meth and amphetamine were sufficient to determine the cause of death and the source of amphetamines.
Conclusion
This study found no significant difference in concentrations between putrefied and non-putrefied cases, underscoring the importance of multiple sample testing during autopsy for accurate interpretation. Each case is unique and must be considered individually.
Keywords: Postmortem, Methamphetamine, Solid tissue, LC-MS/MS
1. Introduction
Methamphetamine (meth), also known as 2-n-methyl-1-phenyl-propan-2-amine or metamfetamine, is a stimulant of the central nervous system that is commonly abused, highly addictive and illicitly used in many countries (Abbruscato and Trippier, 2018). In the early 20th century, meth was derived from its precursor drug, amphetamine, and initially was used as nasal decongestants and bronchial inhalers (Vearrier et al., 2012). Meth use has been rapidly increasing, attributed to the relatively simple and low-cost manufacturing process (Moszczynska, 2021). Global estimates suggest that more than 35 million people use meth and its analogs worldwide (Richards and Laurin, 2023). According to the 2018 National Survey on Drug Use and Health (NSDUH), around 1.9 million individuals in the United States, constituting 0.7% of the population, were estimated to be past-year users of methamphetamine, with 205,000 new initiates. These figures saw an increase in both the 2019 and 2020 NSDUH, reaching 2.5 million past-year users by 2020. However, in the 2020 NSDUH, there was a decline in new initiations of meth use to 150,000 (Ondocsin et al., 2023). The seizures of meth have surged twentyfold since 2009, with emerging cases in new areas, including the Middle East, particularly Saudi Arabia and Arab Gulf countries (Alhazmi et al., 2020). The United Nations Office on Drugs and Crime (UNODC) reports meth as the most commonly seized drug in Middle East region and 12 neighboring countries (UNODC, 2022). Specifically, the United Arab Emirates (50%), Bahrain (28%) have reported significant meth seizures between 2016 and 2020 (UNODC, 2022). This emerging trend is noteworthy in the region, where Captagon® has traditionally been the most abused drug (Al-Asmari, 2021, Al-Asmari et al., 2022a, Al-Asmari et al., 2022b). Furthermore, meth use is on the rise in Saudi Arabia (9%), mainly among amphetamine users, with a significant increase in associated fatalities. Jeddah, Saudi Arabia, witnessed a 500% surge in meth-related deaths between 2016 and 2018. The administration techniques for amphetamine and meth, specifically their impact on absorption and potency, carry clinical implications. The prevalent use of crystal meth, primarily through smoking or injection, is noteworthy (Shaerzadeh et al., 2018). This crystalline form of methamphetamine induces changes in microglial neuroimmune functions, resulting in neuroinflammation and dopaminergic neurotoxicity (Blum et al., 2021). The effects of meth use are characterized by the elevation of dopamine, serotonin, and norepinephrine levels in the brain, contributing to feelings of euphoria, increased productivity, heightened sexual drive, reduced anxiety, and elevated energy levels (Courtney and Ray, 2014). Both acute and prolonged exposure to meth are associated to cognitive impairment and cardiovascular disease (Alhazmi et al., 2020, Paknahad et al., 2021). Agitation and aggressive behavior are well-documented consequences of meth use. Additionally, there is a clear dose–response relationship between meth use and the likelihood of engaging in violent behavior (Courtney and Ray, 2014, Al-Waheeb et al., 2021). The elevated incidence of drug-related deaths (DRDs) is a prominent area of concern. Several studies have reported meth use is associated with unnatural deaths, including homicides and suicides (Bin Abdul Bin Rashid et al., 2013, McKetin et al., 2014, Liu et al., 2017, Schwarzbach et al., 2020), emphasizing the importance of toxicological testing in medicolegal investigations. Postmortem toxicology plays a crucial role in determining whether drugs contributed to death. However, interpreting toxicological data is complex, considering factors such as the circumstances of death, medical history, co-ingested drugs, and postmortem processes like autolysis, metabolism, and postmortem redistribution (PMR) (Butzbach, 2010, Karcioglu et al., 2019). The effectiveness of investigating a suspected death is significantly influenced by the quality of toxicological examinations conducted on biological materials. In instances of suspected DRDs, determining the appropriate toxicological investigations can be challenging. Even when an autopsy is conducted and the macroscopic and histological findings fail to conclusively elucidate the cause of death, a separate decision often needs to be made by the responsible authority regarding the necessity of a toxicological examination (EMCDDA, 2019). The use of blood samples in forensic postmortem investigations is considered the gold standard for investigating DRDs. Unfortunately, in some cases, blood samples may not be accessible based on the circumstances of death. In such cases, forensic investigations often depend on alternative specimens, such as tissue specimens, for examination (Wurita et al., 2016, Al-Asmari, 2021). Additional studies are urgently required to advance forensic toxicology investigations, especially in situations where neither blood nor bodily fluid specimens are accessible. While many studies have reported amphetamine and meth concentrations in various specimens (Kalasinsky et al., 2001, Alhazmi et al., 2020, Al-Asmari et al., 2022a, Al-Asmari et al., 2022b), none have explored meth concentrations in postmortem putrefied human solid tissues. Saudi Arabia, Middle East and North Africa (MENA) countries experience predominantly hot weather throughout the year, which provides conducive conditions for corpses to decompose. It is reported that the hot climate adds complexity to the analysis due to challenges posed by putrefaction. Therefore, this study aimed to test the hypothesis that putrefaction has no significant effect on postmortem meth concentrations in the biological fluids and solid tissue matrices of meth-related fatalities. The present study encompasses postmortem samples obtained from human subjects during autopsies conducted between 2016 and 2022. Meth and its metabolite amphetamine in both putrefied and non-putrefied biological fluids and tissues were examined using LC-MS/MS. The samples utilized in the study included blood, urine, vitreous humor, bile, gastric contents, liver, kidney, and brain from meth-related fatalities. The study aims to improve the understanding of postmortem meth distribution and variations in concentration to facilitate accurate case interpretation and manner of death.
2. Materials and methods
2.1. Chemical and reagents
HPLC-grade solvents such as dichloromethane, isopropanol, hexane, ethyl acetate, acetic acid and methanol were obtained from BDH (Pool, UK). Ammonium formate was purchased from Sigma-Aldrich (Steinheim, Germany). All standards for meth, amphetamine, and their internal standards, including meth-D5 and amphetamine-D5, were purchased from Lipomed (Arlesheim, Switzerland). The Clean Screen DAU (200 mg) cartridges for solid-phase extraction (Part #CSDAU203) were acquired from United Chemical Technologies (Bristol, PA, USA).
2.2. Data collection
The information used in this study was gathered from the Forensic Toxicology Reports database (FTRD). The FTRD serves as a connection between the Forensics Medicine Center in the Jeddah and Al-Madinah Al-Munawwarah regions and the Poison Control and Forensic Medical Chemistry Centers (PCFMCC) located in these two regions. It is important to highlight that FTRD is an online platform managed by Saudi Arabia's Ministry of Health.
2.3. Sample collection
Postmortem specimens were collected from human subjects during autopsies conducted between 2016 and 2022. The collection process followed the protocol described in our previous study (Al-Asmari et al., 2022a, Al-Asmari et al., 2022b). This study adhered to the Declaration of Helsinki guidelines and was approved by the Ethics Committee of Jeddah Health Affairs, Ministry of Health, Saudi Arabia (research code: IBR log no. A01307, 02/03/2021) and the Ethics Committee of Al-Madinah Al-Munawwarah Health Affairs, Ministry of Health in Saudi Arabia (IBR log no.:022-49, 31/05/2022). Data regarding background information, postmortem interval, location at the death scene, co-ingested drugs, and cadaver condition (putrefied or non-putrefied) were acquired from the FTRD. Blood samples were obtained via subclavian vein puncture using sample tubes containing a sodium fluoride solution with a concentration range of 1–2 % BNaF. The anatomical site of each blood specimen (e.g., the femoral vein or left ventricle) was documented. To prevent potential contamination, liver samples were obtained from three distinct areas within the deep right lobe. Kidney samples were collected from the central regions of both the kidneys. A sterile syringe was used to extract the urine samples directly from the bladder tissue. Whenever feasible, the gastric contents and bile in liquid form were also collected. Multiple sites (3–5) within each tissue (stomach wall, lung, brain, and small intestine) were sampled for analysis. Blood specimens were collected using EDTA tubes, whereas the heart and skin samples were preserved in cytogenetic transfer media. All specimens underwent proper labelling and sealing protocols before being stored according to established procedures. The presence of meth and its metabolites in these specimens was carefully analyzed alongside additional factors such as medical history, methodology employed during collection, storage conditions maintained throughout handling postmortem interval, and cause of death assessment prior to their inclusion in this study.
2.4. Sample preparation
Specimens of interest were extracted using in-house SPE, in line with our previous study (Al-Asmari, 2021). To prepare the bodily fluid sample (BNF, vitreous humor, urine, bile, and gastric contents), 1 mL of each sample was placed in a clean glass tube, and a mixture of internal standards containing meth-D5 and amphetamine-D5 was spiked into each tube by pipetting 50 µL of the working solution (50 μg/mL). Before starting SPE application, each sample was diluted using 3.0 mL of phosphate buffer, which was adjusted to a pH of 6 and a concentration of 0.1 M and then mixed using a shaker, and the mixture was centrifuged at 3500 rpm for at least 5 min. Solid tissues including liver, kidneys, brain, and stomach wall tissues were prepared as follows:5 g of each tissue was collected from different parts of each tissue as mentioned above, 2.0 mL of aqueous 1 % sodium fluoride (1:2, w/w) was added to each gram of each tissue, placed in Stomacher bags, and then homogenized for 5 to 10 min. One half gram of homogenized tissue was then weighed in a new test tube, 50 µL (50 ng/g) of internal standard solution (meth-D5 and amphetamine-D5) was spiked into each sample, and the mixture was vortexed and centrifuged at 3500 rpm for at least 5 min. Subsequently, 0.1 M phosphate buffer (3.0 mL) was added and mixed for 10 s. All samples prepared above were extracted using an SPE cartridge preconditioned by sequentially adding 3.0 mL of methanol, 3.0 mL of deionized water, and columns were prepared to extract specimens by adding 3.0 mL of 0.1 M phosphate buffer (pH = 6) to each SPE before applying the samples. Subsequently, the samples were loaded onto the columns using gravity. Each column was washed using D2H20 twice (1.0 mL) followed by applying two extra washing steps using 1.0 mL of acetic acid and 3.0 mL of methanol before the SPE cartridges dried with a high vacuum for at least 10.0 min. The SPE column was washed twice using 1.0 mL of n-hexane. For the initial elution, a mixture of 3.0 mL hexane and ethyl acetate in a (1:1, v/v) ratio was used. Next, the column was washed with 3.0 mL of methanol and dried under a high vacuum for 2 min. For the second elution, a mixture of 3.0 mL of mixture containing dichloromethane: isopropanol: ammonium hydroxide (78:20:2, v/v/v) was loaded. The resulting elutions were then mixed with the initial mobile phase to obtain a total volume of 100 mL Finally, only 1.0 µL of the reconstituted mixture was injected for LC-MS/MS analysis.
2.5. Toxicological testing
The toxicological testing was carried out in accordance (Al-Mousa et al., 2021) with the standard operating procedure protocol (SOPs) at the Poison Control & Forensic Medical Chemistry Center (PCFMCC). These SOPs are designed to screen for the most common drugs and toxicants typically found in post-mortem cases, with most of them have been published (Gerostamoulos et al., 2012, McIntyre et al., 2013, Narapanyakul et al., 2014).
2.6. LC-MS/MS analysis
In this application, a method similar to the one used in our previous study on meth analysis in postmortem bodily fluids using LC-ESI-MS/MS was followed (Al-Asmari, 2021). The LC-MS was equipped with an automatic sampler and electrospray ionization (ESI). Amphetamine, meth, and their internal standards were separated on a Raptor Biphenyl, 2.7 um, 50 × 3.0 mm (Phenyl (L11) HPLC column from Restek (Bellefonte, PA, USA) maintained at 40 °C. Gradient elution of 3.0 mL/min flow rate was applied. The mobile phase consisted of (A) 10 mM ammonium formate adjusted to pH 3.0, and (B) methanol. Meth and its metabolites were isolated using gradient elution starting with 3% (B) for the first minute of the run. The concentration of (B) was increased to 5% over the next minute and to 95% at 15 min. Then, the concentration of (B) was maintained for one minute and reduced to 3 % until the end of the run at 20 min. The LC-MS/MS analysis was performed in the positive ESI mode for both quantification and detection of analytes. This was achieved using multiple reaction monitoring (MRM) mode. In this study, the identification criteria were based on analyte retention times and the presence of one or two product ions. The following analytes were identified: meth (precursor ion, m/z 150; quantifier ion, m/z 119; qualifier ion, m/z 91; meth-d5 (precursor ion, m/z 155; quantifier ion, m/z 119; qualifier ion, m/z 91; amphetamine (precursor ion, m/z 136; quantifier ion, m/z 119; qualifier ion, m/z 91); and amphetamine-d5 (precursor ion, m/z 141; quantifier ion, m/z 119; qualifier ion, m/z 93).
2.7. Method validation
The method was completely validated in accordance with our previous work (Al-Asmari, 2021) and ANSI/ASB standards (ASB, 2019). A negative postmortem sample of each matrix was used as the blank. The calibration curves for meth metabolites of interest were linear within the calibration range of 0.5–1000 ng/mL for bodily fluids and 0.5–1000 ng/g for tissue samples. The coefficients of determination were found to be acceptable, exceeding 0.99 in all matrices tested in this study. The method's sensitivity was evaluated using calibration curve (5 repeated calibration curve for each matrix was evaluated in accordance with ANSI/ASB standards for calculating limit of detection (LOD), which was found to be 0.3 ng/mL and 0.3 ng/g for bodily fluids and tissues specimens respectively. In the limit of quantification (LOQ) experiment a target concentration of 1.0 ng/mL and 1.0 ng/g were extracted using the described method and calculated in accordance to freshly prepared calibration curve in each matrix of interest. The precision of the described method was examined by calculating both within- and between-run precisions; both were found to be less than 10%. The accuracy value was evaluated using the same three controls tested for the method precision investigation and ranged from −2% to +4%. The matrix effect is one of the most important parameters that is mandatory for method validation using low and high concentrations (25 ng/mL (ng/g), 100 ng/mL (ng/g), and 750 ng/mL (ng/g) for bodily fluid (solid tissues)). The matrix effect was found to be acceptable at values ranging from −2.0% to +12%, and the extraction recoveries for meth metabolites were always good, ranging from 90% to 95%. In this study, all dilution controls were tested at two distinct concentrations, 1,000 and 10,000 ng/mL (ng/g), for both bodily fluid and solid tissue specimens. These concentrations were within the acceptable range for the method validation (±15%). No interference was observed from blank postmortem specimens, commonly encountered compounds, or carryover effects from previous injections.
2.8. Statistical analysis
Data processing was done using Statistical Packages for Software Sciences (SPSS) version 28.0.1.1 and Microsoft Excel 2019, obtained from IBM Corporation, Armonk, New York, and Microsoft in Redmond, WA, USA, respectively. Descriptive statistics were used to present continuous data, while frequencies and percentages were employed to express categorical data. The Shapiro-Wilk test was used to evaluate the normality of the value distribution. The Mann-Whitney U test was used to assess differences between groups, whilst the Spearman's correlation test was utilized for variable evaluation, with a P-value of less than 0.05 indicating statistical significance in all correlations.
3. Results
3.1. Case samples
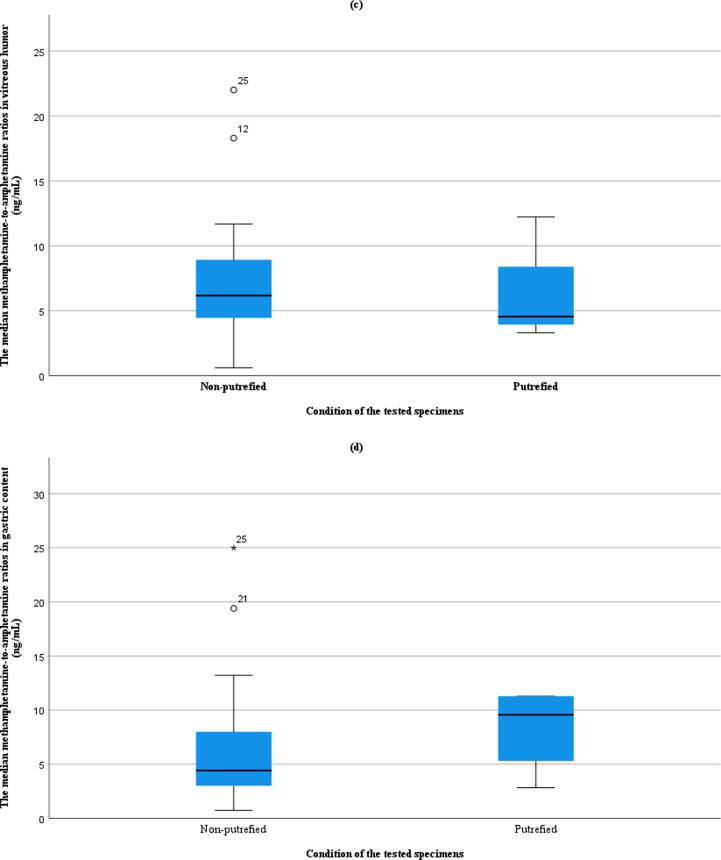
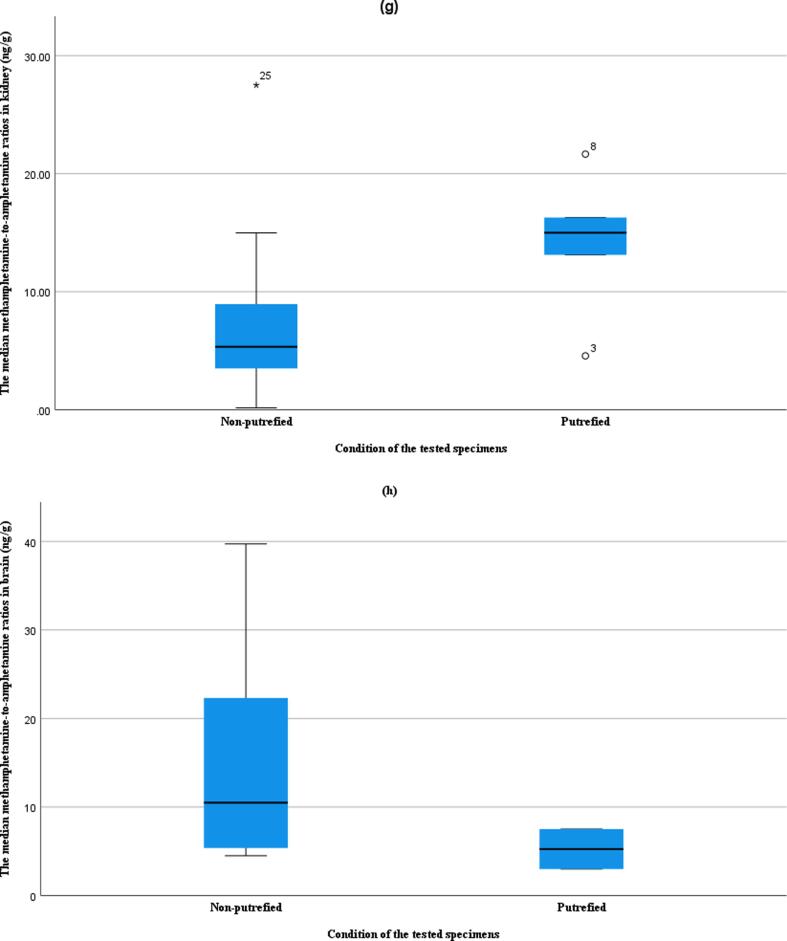
The current study included a total of 27 postmortem deaths related to meth in the western region of Saudi Arabia, particularly in the Jeddah and Al-Madina Al-Munawara regions. The cohort consisted of 21 males (78%) and 6 females (22%), with ages ranging from 16 to 58 years (mean, 34 years; median, 36 years). The median postmortem interval was 24 h, varying from 24 to 288 h. Postmortem details of the cases are provided in Table 1, and Table 2 lists the concentrations of analytes detected in postmortem samples. Fig. 1 illustrates the distribution of meth metabolites in the specimens tested in this study. Among the 27 cases, 8 (30%) showed signs of putrefaction, and biological fluids were not available for testing. Blood was not accessible in 6 of the 27 patients (22%), and urine was not available in 6 (22%) cases. Amphetamine, as a meth metabolite, was not detected in blood in 1 (4%) case, urine in 2 (7%) cases, liver in 2 (7%) cases, and kidney in 3 (11%) cases. However, amphetamine was found in other specimens, highlighting the importance of collecting multiple postmortem samples during autopsy, especially under abnormal conditions such as putrefaction. The majority of deaths occurred at home (56%), followed by outdoors (33%), cars (7%), and hospitals (4%). Solo ingestion of meth was observed in 10 (37%) cases, supporting previous findings that most meth-related fatalities involve polydrug intoxication (Al-Asmari, 2021). THC and THC-COOH were detected in four (15%) and seven (26%) of these cases, respectively. Heroin was found in 7 (15%) cases, while alprazolam was found in two cases (7%). Other drugs, including cocaine, tramadol, clonazepam, diazepam, midazolam, carbamazepine, olanzapine, and ethanol, were found in 1 (4%) case. This study compared the meth-to-amphetamine ratios and ratios of meth concentrations in different blood samples between putrefied and non-putrefied meth users (Table 3). The findings revealed that the meth-to-amphetamine ratios were generally higher in putrefied cases in most samples, except for the brain and vitreous humor, with significant differences observed in the liver and kidney. Additionally, the study identified a significantly higher ratio of meth concentration in bile to that in blood in putrefied cases compared to non-putrefied cases. Median, minimum, and maximum ratios of meth concentration in matrices to meth concentration in blood are presented in Table 5 (see Table 4).
Table 1.
Demographic profiles of 27 cases included in this study.
| Case no. | History | Age | Location | Putrefaction | Other drugs |
|---|---|---|---|---|---|
| 1 | Died in the hospital | 40 | Home | None | None |
| 2 | The deceased was found hanged | 36 | Home | None | None |
| 3 | Died in a car with a history of drug abuse | 25 | Outdoor | Putrefied | Heroin THC 1 Alprazolam Cocaine |
| 4 | Fell from height | 26 | Outdoor | None | THC-COOH 2 Alprazolam Tramadol |
| 5 | The deceased was shot and died in the hospital | 25 | Outdoor | None | THC-COOH |
| 6 | The deceased was stabbed and died in the hospital | 22 | Home | None | THC Clonazepam Diazepam |
| 7 | Fell from height | 29 | Outdoor | None | THC |
| 8 | Sudden death without witnesses | 23 | Outdoor | Putrefied | THC |
| 9 | The deceased was stabbed | 16 | Home | Putrefied | THC-COOH |
| 10 | Fell from height | 35 | Outdoor | None | THC-COOH |
| 11 | Found deceased with a history of drug abuse | 18 | Outdoor | None | None |
| 12 | Fell from height | 40 | Home | None | Midazolam |
| 13 | Accidental | 35 | Outdoor | Putrefied | THC-COOH |
| 14 | Found deceased with a history of drug abuse | 41 | Home | None | Meth |
| 15 | Homicide | Unknown | Outdoor | None | Meth |
| 16 | Found deceased | 40 | Home | Putrefied | Heroin THC-COOH Ethanol |
| 17 | The deceased was stabbed | 41 | Home | None | Meth Carbamazepine |
| 18 | Found deceased in a car | 42 | Car | Putrefied | Meth Heroin |
| 19 | The deceased was stabbed | 40 | Home | None | Meth |
| 20 | Sudden death– rupture uterus and peritonitis | 42 | Home | None | None |
| 21 | Fighting | 39 | Home | None | None |
| 22 | The deceased was stabbed | 30 | Home | None | None |
| 23 | Dead on arrival | 30 | Home | None | None |
| 24 | Suicide by hanging | 42 | Home | Putrefied | None |
| 25 | The deceased was stabbed | 40 | Car | None | None |
| 26 | Dead on arrival | 17 | Hospital | None | None |
| 27 | Found deceased | 58 | Home | Putrefied | Heroin Olanzapine THC-COOH |
Meth = methamphetamine.
Table 2.
Demographics of the 27 postmortem cases.
| Samples | Analytes | No of cases | Median | Minimum | Maximum |
|---|---|---|---|---|---|
| Blood with Sodium Fluoride | Meth | 21 | 340 | 50 | 1431 |
| Amphetamine | 20 | 53 | 20 | 720 | |
| Urine | Meth | 21 | 11,045 | 220 | 90,338 |
| Amphetamine | 19 | 1760 | 70 | 13,600 | |
| Vitreous Humor | Meth | 20 | 750 | 33 | 2200 |
| Amphetamine | 20 | 139 | 10 | 694 | |
| Gastric Contents | Meth | 22 | 1574 | 95 | 234,889 |
| Amphetamine | 22 | 262 | 40 | 2559 | |
| Bile | Meth | 22 | 1178 | 49 | 28,757 |
| Amphetamine | 22 | 213 | 18 | 1245 | |
| Liver | Meth | 21 | 800 | 135 | 26,120 |
| Amphetamine | 19 | 125 | 23 | 2986 | |
| Kidney | Meth | 23 | 700 | 25 | 29,378 |
| Amphetamine | 20 | 197 | 16 | 2630 | |
| Stomach Wall | Meth | 3 | 930 | 190 | 1145 |
| Amphetamine | 3 | 210 | 35 | 995 | |
| Brain | Meth | 8 | 503 | 86 | 9710 |
| Amphetamine | 8 | 27 | 16 | 435 |
Meth = methamphetamine.
Fig. 1.
Concentration ratios of (a) meth and (b) amphetamine in BNaF (ng/mL), urine (ng/mL), vitreous humor (ng/mL), gastric contents (ng/mL), bile (ng/mL), liver (ng/g), stomach wall (ng/g), and brain (ng/g) of 27 meth-related fatalities.
Table 3.
The median, minimum, and maximum meth-to-amphetamine ratios in the biological fluids and tissues.
| Specimen | No of cases | Median | Minimum | Maximum | |
|---|---|---|---|---|---|
| Blood (BNaF) 1 | Non-Putrefied | 16 | 5.5 | 1.7 | 12.7 |
| Putrefied | 4 | 8.1 | 3.0 | 18.8 | |
| Total | 20 | 5.9 | 1.7 | 18.8 | |
| Urine | Non-Putrefied | 14 | 6.3 | 0.8 | 11.0 |
| Putrefied | 5 | 8.6 | 1.9 | 11.8 | |
| Total | 19 | 6.6 | 0.8 | 11.8 | |
| Vitreous humor | Non-Putrefied | 17 | 6.2 | 0.6 | 22.0 |
| Putrefied | 3 | 4.5 | 3.3 | 12.2 | |
| Total | 20 | 5.8 | 0.6 | 22.0 | |
| Gastric Contents | Non-Putrefied | 17 | 4.4 | 0.7 | 25.0 |
| Putrefied | 5 | 9.6 | 2.8 | 91.8 | |
| Total | 22 | 6.2 | 0.7 | 91.8 | |
| Bile | Non-Putrefied | 16 | 4.5 | 0.6 | 14.0 |
| Putrefied | 6 | 8.6 | 2.7 | 31.9 | |
| Total | 22 | 4.9 | 0.6 | 31.9 | |
| Liver | Non-Putrefied | 15 | 4.6 | 0.9 | 15.7 |
| Putrefied | 4 | 10.9 | 10.2 | 50.5 | |
| Total | 19 | 6.7 | 0.9 | 50.5 | |
| Kidney | Non-Putrefied | 15 | 5.3 | 0.2 | 27.5 |
| Putrefied | 5 | 15.0 | 4.6 | 17.1 | |
| Total | 20 | 7.4 | 0.2 | 27.3 | |
| Stomach Wall | Non-Putrefied | 3 | 4.4 | 1.2 | 5.4 |
| Putrefied | 3 | 4.4 | 1.2 | 5.4 | |
| Brain | Total | 6 | 10.5 | 4.5 | 39.7 |
| Non-Putrefied | 2 | 5.3 | 3.0 | 7.5 | |
| Putrefied | 8 | 8.5 | 3.0 | 39.7 | |
Table 5.
Median ratio of meth concentration in matrices to meth concentration in blood.
| Specimen | N 1 | Median | Min 2 | Max 3 | |
|---|---|---|---|---|---|
| Urine to Blood | None 4 | 13 | 26.1 | 4.3 | 109.9 |
| Putrefied | 4 | 38.3 | 13.6 | 102.9 | |
| Total | 17 | 29.5 | 4.3 | 109.9 | |
| Vitreous humor to Blood | None | 15 | 1.4 | 0.2 | 9.0 |
| Putrefied | 2 | 2.0 | 1.6 | 2.3 | |
| Total | 17 | 1.6 | 0.2 | 9.0 | |
| Gastric Contents to Blood | None | 15 | 3.9 | 1.4 | 39.7 |
| Putrefied | 3 | 6.8 | 2.0 | 7.3 | |
| Total | 18 | 3.9 | 0.7 | 39.7 | |
| Bile to Blood | None | 15 | 2.6 | 0.7 | 8.3 |
| Putrefied | 3 | 9,6 | 4.4 | 14.8 | |
| Total | 18 | 2.6 | 0.7 | 14.8 | |
| Liver to Blood | None | 14 | 2.2 | 0.9 | 21.4 |
| Putrefied | 2 | 7.2 | 0.8 | 13.7 | |
| Total | 16 | 2.2 | 0.8 | 21.4 | |
| Kidney to Blood | None | 14 | 1.6 | 0.4 | 20.5 |
| Putrefied | 3 | 4.2 | 0.3 | 15.7 | |
| Total | 17 | 1.6 | 0.3 | 20.5 | |
| Stomach Wall to blood | None | 3 | 1.5 | 1.1 | 3.4 |
| Total | 3 | 1.5 | 1.1 | 3.4 | |
| Brain to blood | None | 6 | 2.2 | 0.5 | 6.8 |
| Putrefied | 2 | 1.2 | 0.6 | 1.7 | |
| None | 8 | 1.9 | 0.5 | 6.8 | |
Table 4.
Statistical results for the various bodily fluids and tissue.
| Specimen Statistic value | Blood (BNaF) 1 | Urine | Vitreous Humor | Gastric Contents | Bile | Liver | Kidney | Brain |
|---|---|---|---|---|---|---|---|---|
| Mann-Whitney U | 24 | 20 | 23 | 28 | 25 | 9 | 14 | 2 |
| Wilcoxon W | 160 | 125 | 29 | 181 | 161 | 129 | 134 | 5 |
| Z | −0.756 | −1.389 | −0.265 | −1.136 | −1.696 | −2.1 | −2.051 | −1.333 |
| p-value | 0.494 | 0.186 | 0.842 | 0.283 | 0.098 | 0.037* | 0.042 * | 0.286 |
3.2. Multiple samples analysis
Urine had the highest concentration of meth (median:11045 ng/mL), followed by the gastric contents of meth (median:1574 ng/mL), bile (median:1178 ng/mL), stomach wall (median:930 ng/g), liver (median:800 ng/g), vitreous humor (median:750 ng/mL), kidney (median:700 ng/g), brain (median:503 ng/g), and blood (median:340 ng/mL). The median, minimum, and maximum meth-to-amphetamine ratios in the biological fluids and tissues are listed in Table 3.
4. Discussion
Our investigation focused on examining the distribution of meth metabolites in various specimens, containing both biological fluids and solid tissues. The primary objective of the current study was to compare meth metabolite concentrations when signs of putrefaction were observed in cases with no signs of putrefaction. The use of blood concentration to assess the cause of death can depend on the route of administration, circumstances of death, time between ingestion and death, and deceased tolerance. In a fatality related to a meth body packer case, a 37-year-old male succumbed to the rupture of a package containing 25 g of meth in his mouth. Postmortem toxicological analysis revealed a subclavian blood level of meth at 36,600 ng/mL, considered a contributing factor to the cause of death (Alsaif et al., 2021).
However, in some cases, other specimens, such as stomach contents and urine, may exhibit higher drug concentrations than blood (McIntyre et al., 2013). Notably, urine samples in our study displayed the highest concentration of meth, aligning with findings from earlier reports (Bierly and Labay, 2018, Al-Asmari, 2021). Some reports highlighted the highest concentration of meth in gastric content, especially in cases involving meth injection (Takekawa et al., 2007, Kiely et al., 2009, Li et al., 2009). In our study, the second-highest concentration of meth was observed in gastric contents. While the concentrations in our study were generally higher than those previously reported (Moriya and Hashimoto, 2002, Wurita et al., 2016), a similar trend was observed in a study conducted in Saudi Arabia (Al-Asmari, 2021). A high gastric concentration of meth indicates oral administration (Wurita et al., 2016, Al-Asmari, 2020) however, the possibility of smoking meth cannot be ruled out, as reported previously (Saenz et al., 2017). It's important to note that intravenously injected meth has been reported to accumulate in gastric tissue. Therefore, relying solely on gastric content concentration may not distinguish between oral, smoking, or intravenous administration, particularly if death occurs several hours after administration (Moriya, 2010, Moriya et al., 2014). The results of meth concentration in vitreous humor were consistent with the ranges reported in earlier studies (0.3–21 mg/L) (Moriya and Hashimoto, 2002, Kiely et al., 2009, Al-Asmari, 2021).
The bile meth concentration in this study aligned with previous findings (Wurita et al., 2016, Al-Asmari, 2021). However, a lower meth concentration in bile than in blood has been reported, particularly in cases associated with massive oral ingestion without chronic use (Moriya and Hashimoto, 2002). In contrast, higher bile meth levels than blood have been reported following an overdose injection in known chronic users (Kiely et al., 2009). Another observation of elevated bile concentration was reported in cases related to meth injections (Moriya and Hashimoto, 2002, Kinoshita et al., 2019).
A study reported that blood and bile indicate enterohepatic circulation of meth due to two peaks of the analyte observed in their study in rat specimens following oral meth administration (Sakai et al., 1982). Additionally, the median ratio of meth concentration in bile to that in blood was significantly higher in putrefied cases than in non-putrefied cases. This aligns with the results of the current study. This study suggests that the degradation of meth in the blood and liver tissues during putrefaction may lead to an increase in the concentration of amphetamine, a metabolite of meth. This increase in amphetamine concentration may result in a higher median ratio of meth concentration in the bile to the blood in putrefied cases than in non-putrefied cases.
Liver specimens also exhibited one of the highest meth concentrations among tissue specimens (Kojima et al., 1984, Kiely et al., 2009, Wurita et al., 2016). Several xenobiotics, including meth, are metabolized in the liver. The concentrations of meth and amphetamine in the current study were within the ranges reported in previous studies. In earlier studies, the liver-to-blood ratio of meth concentration was found to be between 1.7 and 47, while the ratio of meth-to-amphetamine in the liver ranged from 2.0 to 60.0 (Saito et al., 2000, Takekawa et al., 2007, Kiely et al., 2009, McIntyre and I., , 2011), consistent with the ratios obtained in this study.
The kidneys, responsible for filtering blood from xenobiotics and eliminating them through urine, may exhibit high drug concentrations. In our study, Case 10 stood out with the highest meth concentration among all solid tissues (29,378 ng/g). This case also recorded the highest amounts of meth in the liver and brain; however, stomach wall tissue was unavailable for comparison. Notably, the median meth-to-amphetamine ratios in the liver and kidney were significantly higher in non-putrefied cases compared to putrefied cases (Fig. 2). The liver is involved in drug metabolism, while the kidneys play a crucial role in excreting drugs through urine. The time interval between death and postmortem testing could influence the content of amphetamine due to the degradation process of meth metabolites, such as N-hydroxyamphetamine. This process might lead to an increase in amphetamine content in liver and kidney tissues. Nagata et al. reported that the concentration of meth in liver specimens remained stable over 24 months (Nagata et al., 1990), while the concentration of amphetamine experienced a substantial increase from 37,308.2 ng/g to 159,978.896 ng/g during the same period which also support our findings. This underscores the dynamic nature of postmortem drug concentrations and the importance of considering temporal factors in the interpretation of toxicological results.
Fig. 2.
The median meth-to-amphetamine ratios between putrefied and non-putrefied (a) blood with sodium fluoride (ng/mL), (b) urine (ng/mL), (c) vitreous humor (ng/mL), (d) gastric contents (ng/mL), (e) bile (ng/mL), (f) liver (ng/g), (g) kidney (ng/g), and (h) brain (ng/g) of 27 meth-related deaths.
Brain tissue testing plays a key role in postmortem forensic toxicology, aiding in determining the cause and manner of death when other methods are inconclusive. Brain tissue testing can detect drugs or toxins that may not present in the blood or other body fluids, particularly in cases of extensive blood loss, decomposition, or embalming. Additionally, it provides insights into the chronicity and severity of drug abuse or exposure and potential interactions between substances, serving as a mirror of brain exposure in parallel with blood concentration (Rohrig and Hicks, 2015, Argo et al., 2022). Brain meth concentrations in previous reports exceeded those in blood, typically ranging from a 1.3–4.3-fold increase (Kojima et al., 1984, Wurita et al., 2016). The ratio of brain meth to brain amphetamine in these reports was consistently less than 4 %, indicating deaths resulting from meth administration, with amphetamine detected as a meth metabolite (Al-Asmari, 2021, Al-Asmari et al., 2022a, Al-Asmari et al., 2022b). The higher concentration in the brain could be attributed to the prolonged retention of the analyte of interest, while blood concentration decreased due to detoxification processes. The brain tissue in our study displayed a relatively similar median meth concentration when compared with other solid tissues, such as the liver, kidney, stomach wall, and lung. Meth, being a lipophilic substance, tends to accumulate in the highly perfused brain. In previous reports, the concentration of meth in the brain ranged from 1,254.4 to 162,400 ng/g, and amphetamine concentrations ranged from 28.1 to 3,000 ng/g. The brain-to-blood ratio of meth concentration ranged from 2.2 to 4.7, and the meth-to-amphetamine ratio in the brain ranged from 38.2 to 54.1 (Takekawa et al., 2007, Kiely et al., 2009, Wurita et al., 2016), surpassing the values reported in the current study, where the median ratio of meth concentration in the brain to blood was 1.48, and the meth-to-amphetamine ratio in the brain was 18.63.
It is important to note that the very high meth concentration, high ratio of meth in solid tissue to blood, and the elevated meth/amphetamine ratio observed in previous studies were associated with body packing cases. When a drug package ruptures inside the body, it can lead to acute toxicity and death. Consequently, massive quantities of meth are distributed to adjacent organs, resulting in high meth concentrations in these solid tissues. The high meth/amphetamine ratio can be explained by death occurring immediately before metabolism and production of the amphetamine metabolite. However, it is necessary to emphasize that none of the cases in our study involved body packing or the ingestion of massive amounts of meth. This underscores the significance of interpreting toxicological data in the context of the circumstances of death, as each case may present unique characteristics.
5. Conclusion
This study contributes to a better understanding of the extent to which concentrations of amphetamine and meth change after death. Various factors, such as severe weather conditions, storage conditions, the length of the postmortem interval, and postmortem redistribution, can influence these changes. Despite these challenging conditions, both meth and its metabolite can still be detected at concentrations sufficient to ascertain the cause of death and identify the source of amphetamines used. Although determining the route of administration can be challenging due to significant decomposition, high meth concentrations in gastric contents are often anticipated in cases involving body packers and self-oral administration compared to injection routes. The analysis of multiple samples during autopsy can help to mitigate these variations and offer a more accurate interpretation. In instances where blood or body fluid samples are unavailable, tissue specimens such as liver and brain tissue can serve as reliable alternatives. Bile can be an effective tool for screening both chronic use and sudden death due to overdose. However, it is crucial to recognize the uniqueness of each case, and these findings may not universally apply. Therefore, the interpretation of postmortem toxicology results should always consider the individual circumstances of each case.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Abbruscato T.J., Trippier P.C. DARK classics in chemical neuroscience: methamphetamine. ACS Chem. Nerosci. 2018;9:2373–2378. doi: 10.1021/acschemneuro.8b00123. [DOI] [PubMed] [Google Scholar]
- Al-Asmari A.I. Method for postmortem tissue quantification of Δ9-tetrahydrocannabinol and metabolites using LC-MS-MS. J. Anal. Toxicol. 2020;44:718–733. doi: 10.1093/jat/bkaa087. [DOI] [PubMed] [Google Scholar]
- Al-Asmari A.I. Methamphetamine-related postmortem cases in Jeddah, Saudi Arabia. Forensic Sci. Int. 2021;321 doi: 10.1016/j.forsciint.2021.110746. [DOI] [PubMed] [Google Scholar]
- Al-Asmari A.I., Al-Solami F.D., Al-Zahrani A.E., et al. Post-mortem quantitation of amphetamine in cadaveric fluids in Saudi Arabia. Forensic Sci. 2022;2:222–237. doi: 10.3390/forensicsci2010017. [DOI] [Google Scholar]
- Al-Asmari A.I., Alharbi H., Zughaibi T.A. Post-mortem analysis of heroin biomarkers, morphine and codeine in stomach wall tissue in heroin-related deaths. Toxics. 2022;10:473. doi: 10.3390/toxics10080473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alhazmi H.A., Ahsan W., Al Bratty M., et al. Analysis of amphetamine and methamphetamine contents in seized tablets from Jazan, Saudi Arabia by liquid chromatography-mass spectroscopy (LC-MS/MS) and chemometric techniques. Saudi Pharm. J. 2020;28:703–709. doi: 10.1016/j.jsps.2020.04.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Al-Mousa F.A., Gado A.M., Attia A.M., et al. Medical toxicology experience: Poisoning consultations cases registry in Saudi Ministry of Health -2020 annual report. Toxicol. Rep. 2021;8:1558–1564. doi: 10.1016/j.toxrep.2021.07.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alsaif D., Almoghannam S.A., Almadani O.M., et al. Fatality in a body packer: the first reported case in Saudi Arabia, Egypt. J. Forensic Sci. 2021;11:43. doi: 10.1186/s41935-021-00257-z. [DOI] [Google Scholar]
- Al-Waheeb S., Al-Omair N., Mahdi A. Patterns of drug overdose deaths in Kuwait from 2014 to 2018. Public Health Pract (oxf). 2021;2 doi: 10.1016/j.puhip.2021.100181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Argo A., Zerbo S., Buscemi R., et al. A forensic diagnostic algorithm for drug-related deaths: a case series. Toxics. 2022;10:152. doi: 10.3390/toxics10040152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- ASB, 2019. ANSI/ASB STANDARD 036; Standard Practices for Method Validation in Forensic Toxicology. American Academy of Foreign Sciences. Available online: https://www.aafs.org/academy-standards-board.
- Bierly J., Labay L.M. The utility of bile in postmortem forensic toxicology. Acad. Forensic Pathol. 2018;8:324–327. doi: 10.1177/1925362118782073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bin Abdul Rashid, S. N., A. S. A. Rahim, M. J. Thali, et al., 2013. Death by 'ice': fatal methamphetamine intoxication of a body packer case detected by postmortem computed tomography (PMCT) and validated by autopsy. Forensic Sci. Med. Pathol. 9, 82-87. 10.1007/s12024-012-9395-1. [DOI] [PubMed]
- Blum K., Cadet J.L., Gold M.S. Psychostimulant use disorder emphasizing methamphetamine and the opioid -dopamine connection: Digging out of a hypodopaminergic ditch. J. Neurol. Sci. 2021;420 doi: 10.1016/j.jns.2020.117252. [DOI] [PubMed] [Google Scholar]
- Butzbach D.M. The influence of putrefaction and sample storage on post-mortem toxicology results. Forensic Sci. Med. Pathol. 2010;6:35–45. doi: 10.1007/s12024-009-9130-8. [DOI] [PubMed] [Google Scholar]
- Courtney K.E., Ray L.A. Methamphetamine: an update on epidemiology, pharmacology, clinical phenomenology, and treatment literature. Drug Alcohol Depend. 2014;143:11–21. doi: 10.1016/j.drugalcdep.2014.08.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- EMCDDA . Publications Office of the European Union; Luxembourg: 2019. European Monitoring Centre for Drugs and Drug Addiction. An analysis of post-mortem toxicology practices in drug-related death cases in Europe, Technical report. [Google Scholar]
- Gerostamoulos D., Beyer J., Staikos V., et al. The effect of the postmortem interval on the redistribution of drugs: a comparison of mortuary admission and autopsy blood specimens. Forensic Sci. Med. Pathol. 2012;8:373–379. doi: 10.1007/s12024-012-9341-2. [DOI] [PubMed] [Google Scholar]
- Kalasinsky K.S., Bosy T.Z., Schmunk G.A., et al. Regional distribution of methamphetamine in autopsied brain of chronic human methamphetamine users. Forensic Sci. Int. 2001;116:163–169. doi: 10.1016/s0379-0738(00)00368-6. [DOI] [PubMed] [Google Scholar]
- Karcioglu, O., B. Arslan, O. Karcioglu, et al., 2019. Poisoning in the Modern World - New Tricks for an Old Dog?
- Kiely E., Lee C.J., Marinetti L. A fatality from an oral ingestion of methamphetamine. J. Anal. Toxicol. 2009;33:557–560. doi: 10.1093/jat/33.8.557. [DOI] [PubMed] [Google Scholar]
- Kinoshita H.T., A. Mumihashi M. Bile as a complementary matrix for methamphetamine testing, an autopsy case of methamphetamine poisoning. Rom. J. Legal Med. 2019;379–381 doi: 10.4323/RJLM.2019.379. [DOI] [Google Scholar]
- Kojima T., Une I., Yashiki M., et al. A fatal methamphetamine poisoning associated with hyperpyrexia. Forensic Sci. Int. 1984;24:87–93. doi: 10.1016/0379-0738(84)90156-7. [DOI] [PubMed] [Google Scholar]
- Li R.-B., Guan D.-W., Zhu B.-L., et al. Death from accidental poisoning of methamphetamine by leaking into alimentary tract in drug traffic: a case report. Leg. Med. (Tokyo) 2009;11(Suppl 1):S491–S493. doi: 10.1016/j.legalmed.2009.02.028. [DOI] [PubMed] [Google Scholar]
- Liu Y., Hao B., Shi Y., et al. Violent offences of methamphetamine users and dilemmas of forensic psychiatric assessment. Forensic Sci. Res. 2017;2:11–17. doi: 10.1080/20961790.2017.1287155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- M. McIntyre I. Postmortem Methamphetamine Distribution. J. Forensic Res. 2011;02 doi: 10.4172/2157-7145.1000122. [DOI] [Google Scholar]
- McIntyre I.M., Nelson C.L., Schaber B., et al. Antemortem and postmortem methamphetamine blood concentrations: three case reports. J. Anal. Toxicol. 2013;37:386–389. doi: 10.1093/jat/bkt040. [DOI] [PubMed] [Google Scholar]
- McKetin R., Lubman D.I., Najman J.M., et al. Does methamphetamine use increase violent behaviour? Evidence from a prospective longitudinal study. Addiction. 2014;109:798–806. doi: 10.1111/add.12474. [DOI] [PubMed] [Google Scholar]
- Moriya F. Accumulation of intravenously administered methamphetamine in stomach contents. Forensic Toxicol. 2010;28:43–46. doi: 10.1007/s11419-009-0084-z. [DOI] [Google Scholar]
- Moriya F., Hashimoto Y. A case of fatal hemorrhage in the cerebral ventricles following intravenous use of methamphetamine. Forensic Sci. Int. 2002;129:104–109. doi: 10.1016/s0379-0738(02)00233-5. [DOI] [PubMed] [Google Scholar]
- Moriya F., Yoshitome K., Miyaishi S. A large proportion of intravenously administered methamphetamine is excreted into the stomach. Forensic Toxicol. 2014;32:186–188. doi: 10.1007/s11419-013-0207-4. [DOI] [Google Scholar]
- Moszczynska A. Current and Emerging Treatments for Methamphetamine Use Disorder. Curr. Neuropharmacol. 2021;19:2077–2091. doi: 10.2174/1570159X19666210803091637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nagata T., Kimura K., Hara K., et al. Methamphetamine and amphetamine concentrations in postmortem rabbit tissues. Forensic Sci. Int. 1990;48:39–47. doi: 10.1016/0379-0738(90)90270-9. [DOI] [PubMed] [Google Scholar]
- Narapanyakul R., Tungtananuwat W., Yongpanich P., et al. Comparative study of postmortem blood, urine and vitreous humor methamphetamine. The Thai Journal of Pharmaceutical Sciences. 2014;38:5–13. [Google Scholar]
- Ondocsin J., Holm N., Mars S.G., et al. The motives and methods of methamphetamine and ‘heroin’ co-use in West Virginia. Harm Reduct. J. 2023;20:88. doi: 10.1186/s12954-023-00816-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paknahad S., Akhgari M., Ghadipasha M. An alarming rise in the prevalence of deaths with methamphetamine involved in Tehran, Iran 2011–2018. Forensic Sci. Med. Pathol. 2021;17:208–215. doi: 10.1007/s12024-020-00339-9. [DOI] [PubMed] [Google Scholar]
- Richards, J.R., Laurin, E.G., 2023. Methamphetamine Toxicity. StatPearls. Treasure Island (FL), StatPearls Publishing. [PubMed]
- Rohrig T.P., Hicks C.A. Brain tissue: a viable postmortem toxicological specimen. J. Anal. Toxicol. 2015;39:137–139. doi: 10.1093/jat/bku139. [DOI] [PubMed] [Google Scholar]
- Saenz S.R., Lewis R.J., Angier M.K., et al. Postmortem Fluid and Tissue Concentrations of THC, 11-OH-THC and THC-COOH. J. Anal. Toxicol. 2017;41:508–516. doi: 10.1093/jat/bkx033. [DOI] [PubMed] [Google Scholar]
- Saito T., Yamamoto I., Kusakabe T., et al. Determination of chronic methamphetamine abuse by hair analysis. Forensic Sci. Int. 2000;112:65–71. doi: 10.1016/s0379-0738(00)00165-1. [DOI] [PubMed] [Google Scholar]
- Sakai T., Niwaguchi T., Murata T. Distribution and excretion of methamphetamine and its metabolites in rats. I. Time-course of concentrations in blood and bile after oral administration. Xenobiotica. 1982;12:233–239. doi: 10.3109/00498258209052461. [DOI] [PubMed] [Google Scholar]
- Schwarzbach V., Lenk K., Laufs U. Methamphetamine-related cardiovascular diseases. ESC. Heart Fail. 2020;7:407–414. doi: 10.1002/ehf2.12572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shaerzadeh F., Streit W.J., Heysieattalab S., et al. Methamphetamine neurotoxicity, microglia, and neuroinflammation. J. Neuroinflammation. 2018;15:341. doi: 10.1186/s12974-018-1385-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takekawa K., Ohmori T., Kido A., et al. Methamphetamine body packer: acute poisoning death due to massive leaking of methamphetamine. J. Forensic Sci. 2007;52:1219–1222. doi: 10.1111/j.1556-4029.2007.00518.x. [DOI] [PubMed] [Google Scholar]
- UNODC, 2022. United Nation Office on Drugs and Crime, World Drug Report 2022, Vienna, Austria.
- Vearrier D., Greenberg M.I., Miller S.N., et al. Methamphetamine: history, pathophysiology, adverse health effects, current trends, and hazards associated with the clandestine manufacture of methamphetamine. Dis. Mon. 2012;58:38–89. doi: 10.1016/j.disamonth.2011.09.004. [DOI] [PubMed] [Google Scholar]
- Wurita A., Hasegawa K., Minakata K., et al. Postmortem redistribution of methamphetamine and amphetamine in blood specimens from various blood vessels and in the specimens from pericardial fluid, bile, stomach contents and various solid tissues collected from a human cadaver. Forensic Toxicol. 2016;34:191–198. doi: 10.1007/s11419-015-0303-8. [DOI] [Google Scholar]