Abstract
Inflammation-associated diseases encompass a range of infectious diseases and non-infectious inflammatory diseases, which continuously pose one of the most serious threats to human health, attributed to factors such as the emergence of new pathogens, increasing drug resistance, changes in living environments and lifestyles, and the aging population. Despite rapid advancements in mechanistic research and drug development for these diseases, current treatments often have limited efficacy and notable side effects, necessitating the development of more effective and targeted anti-inflammatory therapies. In recent years, the rapid development of nanotechnology has provided crucial technological support for the prevention, treatment, and detection of inflammation-associated diseases. Various types of nanoparticles (NPs) play significant roles, serving as vaccine vehicles to enhance immunogenicity and as drug carriers to improve targeting and bioavailability. NPs can also directly combat pathogens and inflammation. In addition, nanotechnology has facilitated the development of biosensors for pathogen detection and imaging techniques for inflammatory diseases. This review categorizes and characterizes different types of NPs, summarizes their applications in the prevention, treatment, and detection of infectious and inflammatory diseases. It also discusses the challenges associated with clinical translation in this field and explores the latest developments and prospects. In conclusion, nanotechnology opens up new possibilities for the comprehensive management of infectious and inflammatory diseases.
Subject terms: Vaccines, Biomaterials
Introduction
Inflammation is an adaptive biological response of the immune system to harmful stimuli, such as infections and tissue damage.1,2 Acute inflammation serves as the initial self-defense response of the body to pathogen infections or injuries, during which immune cells and inflammatory factors collaborate to efficiently clear pathogens, repair tissues, and restore homeostasis.3,4 If the inflammatory response is not promptly terminated, it may progress into chronic inflammation, aggravating tissue damage, and infectious diseases.5 Chronic inflammation appears to not arise directly from typical injuries or infections but more from dysfunctions in the immune system and disruptions in bodily homeostasis.3,6 In contrast to the beneficial and important role of moderate inflammation in host defense, harmful chronic inflammation results in a variety of chronic inflammatory diseases, including autoimmune diseases, allergic conditions, atherosclerosis(AS), and even an increased risk of cancer.1,7 The majority of autoimmune therapies or wide-ranging immune suppressors are supportive to slow the progression of the illness and symptoms.8 However, conventional drugs for inflammation diseases, like inflammatory bowel disease (IBD), are ineffective therapeutically and have serious side effects.9 Also, there are still no efficient or secure drugs available for clinical treatment of some inflammatory diseases, like stroke that is the leading cause of mortality and disability globally.10 Therefore, further elucidating the pathogenesis of chronic inflammation and developing more effective targeted drugs is an urgent priority for the treatment of inflammatory diseases.
In recent years, nanotechnology has emerged as a promising field with significant potential in combating infectious and inflammatory diseases. NPs with unique properties and capabilities have been explored for applications in vaccine development, antiviral drug delivery and pathogen detection. Currently, nanostructured viral vaccines based on virus-like particles (VLPs) have been widely deployed worldwide for viruses like severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), human papillomavirus (HPV), hepatitis B virus (HBV), and influenza. Notably, for the highly contagious SARS-CoV-2 virus that has caused a global pandemic, various COVID-19 vaccines have been developed using both traditional inactivated viruses and nanotechnology-based approaches, such as the BioNTech/Pfizer and Moderna messenger RNA (mRNA) vaccines, Novavax’s VLP protein vaccine.11 Some nanomaterials, such as silver nanoparticles (AgNPs), selenium nanoparticles (SeNPs), and metal NPs solutions (ND50, NK99, and TPNT1), can be prepared as environmental sanitizers or as preventive or therapeutic inhalants due to their directive antibacterial or antiviral effects in vitro.12 Moreover, NPs can be utilized for the delivery of drugs, enhancing their efficacy and reducing adverse reactions. It was shown that ethyl cellulose nanoparticles (EC-NPs) for amphotericin delivery had good stability, high bioavailability, and low cytotoxicity, providing a potential delivery vehicle for oral drugs for the treatment of fungi and parasite infections.13 Nanotechnology-based detection platforms have been developed to identify pathogens, offering rapid and sensitive diagnostics. A polyethyleneimine-assisted copper in situ growth strategy demonstrated excellent sensitivity, precision and repeatability for the detection of infectious diseases, such as E. coli and SARS-CoV-2 infections.14 These applications of nanomaterials present new opportunities to improve prevention strategies and enhance the effectiveness of therapies for infectious diseases.
In addition, nanotechnology is an effective approach to achieve therapeutic goals for inflammatory diseases, owing to its high drug loading capacity, efficient targeting, controllable sustained release, and ability to cross physiological barriers. When interferon (IFN)-β therapy was combined with NPs, like IFN-carried chitosan/sulfobutylether-cyclodextrin NPs, it was successful in intranasal administration of IFN-β into the central nervous system (CNS), boosting clinical improvement and controlling neurological inflammation in encephalomyelitis (EAE).15 Moreover, nanomaterials can also serve as molecular probes to provide support for imaging diagnosis of inflammatory diseases. Prussian blue NPs have been successfully utilized in magnetic resonance imaging (MRI) imaging to accurately concentrate and identify rheumatoid arthritis (RA).16 Therefore, nanotechnology provides the potential for treat-to-target principles, serving as the cornerstone of inflammatory disease treatment.
While nanotechnology holds tremendous potential in the fight against inflammation-associated diseases, some challenges and issues must be addressed as it progresses toward clinical applications. Further research is needed to improve the safety, stability, scalability, and efficiency of nanotechnology-based prevention and treatment approaches. Here, we provide an overview of the latest research advancements and applications of nanotechnology in infectious and inflammatory diseases, encompassing areas such as vaccine development, therapeutic drug delivery, and disease detection. Besides, we discuss the current challenges and limitations in its applications, hoping that the insights will offer valuable recommendations for the development of innovative strategies for the comprehensive prevention and treatment of infectious and inflammatory diseases.
Advanced nanotechnologies
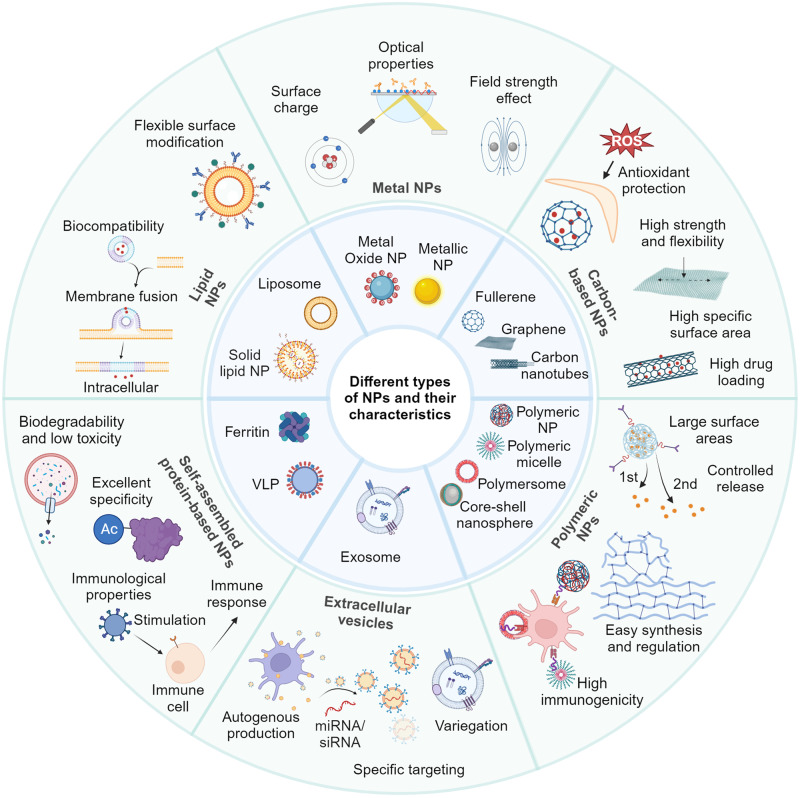
The various characteristics of NPs enable diverse applications in biomedicine. Nanomaterials can serve as adjuvants and vaccine delivery vectors to enhance vaccine-induced specific immune responses and antigen immunogenicity, and are widely used for infectious disease prevention, tumor immunotherapy, etc. Meanwhile, nanomaterials, like lipid nanoparticles (LNPs), polymeric NPs, and exosomes, can act as delivery systems for targeted drug distribution, controlled release, and effective treatment.17 By fine-tuning their surface functional groups, nanomaterials like magnetic NPs and quantum dots (QDs) can be used in biomedical imaging, providing high-sensitivity detection of specific targets and real-time monitoring of disease progression.18,19 In addition, nanomaterials with antibacterial and antiviral properties are integrated into protective equipment like masks, gloves, and disinfectants, serving as wound dressings to prevent infections. Here, we delineated the characteristics of each NP variant (Table 1), particularly focusing on their application in the prevention, treatment, and detection/diagnosis of infectious and inflammatory diseases (Fig. 1).
Table 1.
Advantages and disadvantages of existing NPs in drug delivery and vaccines
| Types of NPs | Composition | Advantages | Disadvantages | Examples of applications | Ref |
|---|---|---|---|---|---|
| Lipid NPs | Liposomes, commonly used lipids, include lecithin, triglycerides, triglycerides of palm stearate, and fatty acids | High biocompatibility; strong drug loading capacity; flexible surface modification | Limited stability under certain environmental conditions; limited control of drug release rate; costly preparation, potential toxicity at high dosages | Targeting lung therapy COVID-19 | 744 |
| Making mRNA vaccines against Zika virus infection | 745 | ||||
| Vitamin lipid nanoparticles can be used to treat septicaemia caused by drug-resistant bacteria | 746 | ||||
| Glycyrrhetinic acid-lipid framework nanocarriers improve drug loading efficiency of anti-hepatocellular carcinoma drugs | 747 | ||||
| Metal NPs | Metal and metal oxide NPs, including silver, gold, CuO, SiO2, TiO2 and various other metal oxides | Unique shape, size, structure, and local-field enhancement action | Potential toxicity; limited stability with aggregation and morphological changes; environmental pollution concerns; limited degradation in vivo | Intravaginal zinc oxide tetrapod NPs against genital herpes | 668 |
| AgNPs on H1N1 inhibit influenza A virus | 748 | ||||
| Delivery of antiviral siRNA with AuNPs inhibits dengue virus infection | 749 | ||||
| Cuprous oxide NPs against Hepatitis C Virus | 750 | ||||
| Carbon-based NPs | Carbon nanotubes | Large specific surface area and hollow structure; increasing application capability by surface modification; good chemical and physical stability | Pulmonary toxicity; complex preparation steps; varying diameters, lengths, structures | Multiwalled carbon nanotubes for the detection of zooplankton in water | 751 |
| Graphene | Excellent mechanical properties; high strength and flexibility; high specific surface area | Limitation in stability; aggregation in aqueous solution affects stabilization and release; potential biotoxicity issues | Sulfonated MNPs functionalized destroy herpes simplex virus type 1. | 752 | |
| Fullerenes | Antioxidant properties; stable structure; surface modification to obtain multiple properties | Relatively low load capacity; low solubility in water | Fullerene derivatives inhibit HIV by complexing with HIV protease. | 753 | |
| C60-β-cyclodextrin conjugate improves nuclear transport of doxorubicin | 754 | ||||
| Polymeric NPs | Natural hydrophilic polymers and synthetic hydrophobic polymers | Good drug loading capacity and controlled release capabilities; easy synthesis and regulation | Long-term toxicity from body accumulation; potentially toxic degradation products; complex preparation and functionalization | Porous PLA and PLGA NPs for pulmonary delivery of HBV vaccine | 105,755 |
| (PEG-b-PLA) NPs improve protein affinity for delivered drugs | 756 | ||||
| Protein NPs | VLP | High structural stability and resistance to degradation; immunocompatibility; biomimetic properties | Inefficient protein delivery in vivo; insufficient immunogenicity, requiring adjuvants and multiple injections for vaccination; complex preparation process | Novel virus-like particle vaccine encoding the circumsporozoite protein of plasmodium falciparum is Immunogenic. | 757 |
| Engineered VLPs for efficient delivery of therapeutic proteins. | 758 | ||||
| Proteins | Good biocompatibility and biodegradability; multifunctionality through surface modification. | Complex preparation and functionalization; high production costs; limited drug loading capacity | Dual-sensitive antibacterial peptide nanoparticles prevent dental caries. | 759 | |
| Development of spike RBD ferritin proteins vaccine against SARS-CoV-2 infection in ferrets | 760 | ||||
| Exosomes | Classification according to source | Excellent biocompatibility; targetability | Difficulty in standardized production; poor experimental reproducibility; characterization difficulties; heterogeneity; Harsh storage conditions | Recombinant SARS-CoV-2 receptor-binding structural domain-modified exosomes as inhalable COVID-19 vaccines. | 143 |
VLP virus-like particles, SARS-CoV-2 severe acute respiratory syndrome coronavirus 2, HIV human immuno-deficiency virus, siRNA small interfering RNA, AgNPs silver nanoparticles, AuNPs gold nanoparticles, MNPs magnetic nanoparticles, HBV hepatitis B virus, PLGA Poly(lactide-co-glycolic) acid, PEG polyethylene glycol, PLA Poly(lactic acid), RBD receptor-binding domain
Fig. 1.
Six common nanomaterials and their characteristics. Lipid NPs, composed of lipids such as phospholipids, exhibit good biocompatibility and flexible surface modification capabilities. Metal NPs, including metals (such as gold, silver, copper) and their metal oxides, possess excellent optical, electronic, and magnetic properties, enabling applications in biological imaging, PTT, and sensing.790 Carbon-based nanomaterials, including CNTs, graphene, and fullerenes, not only have a large surface area and high drug loading capacity but also exhibit high strength and chemical stability, allowing resistance to oxidative environments.791 Polymer NPs, composed of polymer materials, display diverse structures and properties.792 Self-assembled NPs, including ferritin family proteins and VLPs, possess good biodegradability in the case of the former and can mimic viral stimuli to initiate immune responses in the case of the latter.793 Exosomes, a type of small vesicles secreted by cells, carry abundant proteins, nucleic acids, and signaling molecules, playing vital roles in information transfer and regulation.794 These and exosomes have broad applications in the biomedical and nanotechnology fields, including drug delivery, molecular imaging, biosensing, tissue engineering, and disease diagnosis
Metal NPs
Anti-infection and anti-inflammatory medication delivery systems have been extensively investigated using metals such as silver and gold, as well as metal oxide NPs such as cupric oxide (CuO), SiO2, and TiO2. Metal NPs can be classified into pure metal NPs, alloy NPs, and core-shell NPs based on their composition and structure. Pure metal NPs are composed of a single metal element such as gold, silver, copper, etc. These NPs have a single crystalline structure, and their physical, chemical, and optical properties are mainly determined by the metal component. Alloy NPs are synthesized from two or more metal elements, forming an alloy structure with specific compositions, such as magnetic nanoparticles (MNPs), which possess superparamagnetic properties, enabling magnetic-targeted gene delivery under the influence of a magnetic field. Core-shell NPs consist of a metal core surrounded by a functional material shell. This core-shell structure allows for the control of surface properties, stability, and optical activity of the NPs, providing additional functionalities.
Metal NPs possess unique shapes and sizes. Their nanoscale dimensions give rise to size effects, resulting in distinct physical, chemical, and optical properties compared to macroscopic materials.20–22 Compared to NPs made of other materials, metal NPs exhibit unique physical and chemical properties such as light, electrical, and magnetic properties, which can be utilized in virus and bacteria detection and diagnosis.23 Scientists have explored the use of metal NPs in photodynamic therapy, where they harness the reactive oxygen species (ROS) generated by metal NPs under specific wavelengths of light to kill viruses.24 Furthermore, researchers utilize the characteristics of metal NPs to induce changes in optical signals through surface plasmon resonance (SPR), enabling the development of highly sensitive optical sensors for virus detection and diagnosis.25 Definitely, metal NPs can be used not only for pathogen detection but also for bioimaging and tumor immunotherapy.26,27
Metal NPs have been more intensively studied in the anti-infection and anti-inflammation field, especially gold nanoparticles (AuNPs) and AgNPs. AuNPs serve as vehicles for drugs and gene delivery, having excellent biocompatibility, which can be taken up by various types of cells, such as lymphocytes, macrophages, and brain micro endothelial cells.28 Moreover, AuNPs can not only generate non-enzymatic ROS to combat infections but also inhibit enzymes essential for the survival of pathogenic microorganisms.29 AgNPs are rapidly soluble and have a low potential for drug resistance due to their small size and large surface area.30–32 AgNPs have been demonstrated to have anti-HIV-1 activity and to prevent the interaction between CD4 and gp120, which prevents HIV-1 from invading host cells.33–35 However, AgNPs are cytotoxic and genotoxic due to their interactions with electron transport chain enzymes and DNA in human cells, resulting in disrupted ATP synthesis, ROS generation, and DNA damage.36 But when AgNPs are used to produce antimicrobial coatings on the surfaces of medical devices such as wound dressings, catheters, and implants, they not only exhibit significant antibacterial effects against common pathogenic bacteria, but also do not show cytotoxicity when used in vitro.37
In conclusion, research on the application of metal NPs in anti-infection treatments is continuously advancing. Among them, AuNPs and AgNPs are the most commonly studied and applied types. However, further research and clinical trials are necessary to ensure the safety and effectiveness of applying metal NPs in antiviral treatments.
Carbon-based NPs
Another type of nanomaterial is carbon-based, which includes fullerenes, carbon nanotubes (CNTs), and graphene. These various types of carbon-based NPs have multiple potential roles and applications in the field of anti-infective and anti-inflammatory research.
Carbon-based NPs exhibit superior anti-infective effects and are commonly used as anti-infection materials. Graphene oxide, which is oxidized based on graphene, exhibits antiviral activity at non-cytotoxic concentrations,38 and the viral inhibition effect was also more pronounced after Ag modification.39 Both in vivo and in vitro viral replication can be stopped by fullerenes and their derivatives, and their amino acid derivatives have also been demonstrated to stop viral replication.40 Banerjee et al. reported that protoporphyrin IX-conjugated multi-walled carbon nanotubes (PPIX-MWNT) induce RNA cleavage and protein oxidation of influenza virus (IV) under visible light, resulting in virus inactivation. Furthermore, this antiviral effect is non-specific and can be used to treat all viral infections.41 Also, nanofilms of MWCNTs combined with gelatin and chitosan have also been shown to possess antimicrobial activity.42 Ramos et al. reported for the first time the anti-Leishmania activity of fullerenes, which even reduced the liver parasite burden in the Balb/c mouse model.43 Certainly, carbon-based NPs have shown promising therapeutic effects not only for infectious diseases but also for inflammatory conditions such as diabetes. Khalid et al. reported that bacterial cellulose-functionalized multi-walled CNTs inhibit bacteria in diabetic wounds while promoting wound healing.44
Based on their excellent optical and electromagnetic properties, carbon-based NPs are ideal choices for biosensors and detection platforms. By functionalizing their surfaces, carbon-based NPs can be combined with specific biomolecules to achieve high sensitivity and selectivity in detection. For example, CNTs have been used to develop continuous sensing systems for dopamine (DA) release and ascorbic acid monitoring, and with further improvements, they can even simultaneously detect baseline levels of glucose and lactate in the rat’s brain.45 There are extensive studies for more in-depth research and comprehensive summaries on the sensing and imaging applications of carbon-based NPs.46–50
Due to their high surface area-to-volume ratio and tunable chemical properties, carbon-based NPs can enhance the solubility and stability of drugs, achieve targeted delivery, and increase therapeutic efficacy while reducing side effects. Therefore, carbon-based NPs are commonly used as drug delivery systems, where drugs can be loaded onto their surfaces or internal compartments and released in a targeted manner within the body. The drug delivery applications of carbon-based NPs have been summarized in several articles.46,51–53 It is recommended to refer to their articles for a more in-depth understanding of the topic.
Lipid NPs
LNPs are a common type of nanomaterial used for drug delivery and biomedical applications. The commonly used LNPs can be broadly classified into liposomes formed by phospholipids with amphiphilic properties and solid lipid nanoparticles (SLNs) typically prepared by nanoscale emulsification techniques. Common auxiliary ingredients, including surface functionalization agents, stabilizers, polyethylene glycol (PEG)-based polymers, cholesterol, are used to regulate the stability, targeting ability, and other characteristics of lipid NPs.
LNPs are composed of lipids that are biodegradable, biocompatible, inert, low toxic, and low immunogenic.33,54–56 They are also easily accessible and less expensive.33,57 The LNPs offer pharmaceuticals with smaller size, superior surface area, increased drug-carrying capacity, superior interfacial interactions, and even significantly enhanced delivery efficiency.58–61 In the case of hydrophobic drugs, liposomes increase their solubility and reduce their toxicity to non-specific organs.62–64 In addition, LNPs can achieve sustained, gradual, or stimulus-responsive drug release through various preparation methods and material selections.65 LNPs are highly flexible in surface modification, allowing for chemical modifications or functionalization of their outer layer using surface modifiers to impart specific properties or functions to the NPs.66,67 Therefore, medications based on LNPs have superior pharmacokinetic properties, higher bioavailability, lower toxicity, fewer adverse effects, and more accumulation at the target site in vivo.68–71
Considerations for lipid NPs include limited stability affected by storage, restricted drug loading, and challenges in controlling drug release rates due to multiple factors.72–75 LNPs may be designed for stimuli-triggered release, but accuracy in physiological conditions remains a challenge.76–79 Research and optimization are ongoing to address these limitations and enhance lipid NP performance.
Currently, there are many studies of LNPs in the anti-infection and anti-inflammation field. Among them, liposomes are more widely used than SLNs, but research in this area is still evolving and exploring. Liposomes have been used as nanocarriers for the targeted delivery of antiviral drugs and vaccines because of their high retention time but high loading capacity.80,81 Also, by virtue of their good biocompatibility, liposomes are compatible with tissues and cells in living organisms, reducing the likelihood of toxicity and immune reactions.82–84 Also, some studies have found that liposomes can neutralize inflammation or regulate and mitigate the cytokine storm against infections and their resulting inflammatory responses.85 With the action of liposomes, anti-inflammatory drugs were transported to macrophages, inhibiting signaling pathways involved in inflammation and thereby calming the cytokine storm.86 However, some studies have reported that drug-loaded liposomes can induce inflammatory responses during infection. Based on these findings, it is suggested that liposomes can enhance the effectiveness of drug therapy against infections. In addition, phospholipids may redistribute the cell surface charge, reducing the interaction between viral particles and cell surface proteoglycans, which inhibits viral entry.87 These are sufficient to demonstrate the superiority of lipid NPs as a platform for carrying anti-infection and anti-inflammatory drugs.
LNPs have also received extensive research in the field of mRNA vaccine delivery. Ionizable LNPs have demonstrated significant advantages in delivering mRNA vaccines, including the ability to efficiently deliver mRNA to antigen-presenting cells (APCs).88,89 The LNPs can also transfect neutrophils, macrophages, and dendritic cells (DCs), demonstrating that they may help transfer mRNA to a range of immune cells.90–92 In addition, there have been significant advancements in the research of delivering mRNA to the lungs via LNPs.60 An inhaled delivery lipid vector can overcome the specific cell type, mucus barrier and mucus cilia clearance system of the lung to achieve specific aggregation.93 Such nanotechnological platforms offer the advantages of a cell-free system, rapid production, high versatility, and a good safety profile over conventional vaccines.
In summary, lipid NPs are an important nanomaterial with a wide range of applications in drug delivery and biomedical fields. With the advancement of science, the design and optimization of lipid NPs will further enhance their performance and expand their application scope.
Polymeric NPs
Polymeric NPs are colloidal systems that range in size from 10 to 1000 nm and have received widespread attention due to their high immunogenicity, stability, and biocompatibility.94 Similar to metal NPs, polymeric NPs also have a large specific surface area, which gives them good drug loading capacity.95 Polymeric NPs can effectively encapsulate and present antigens/drugs.96 Employing a ROS-sensitive polymer, Wu et al. describe the creation of polymer NPs that are intended to penetrate the brain during ischemic stroke (IS) by thrombin-stimulated diameter decrease and AMD3100-regulated precise administration.97 Antigen adsorption avoids exposure to harmful chemical solvents or extreme pH values during the formulation of polymeric NPs. The encapsulation also protects antigens/drugs from exposure to metabolic enzymes and harsh gastrointestinal (GI) environment in the oral route of administration.98 Through phagocytosis or endocytosis, polymeric NPs can increase the effectiveness of antigen uptake by APCs.99,100 Furthermore, polymeric NPs can enhance the efficacy of drugs by controlling the release rate and achieving targeted delivery.101
The creation of nanovaccines can benefit from the use of both organic polymeric NPs (like chitosan and dextran) and synthetic polymeric nanomaterials (like poly(lactic acid) (PLA) and poly(lactide-co-glycolic acid) (PLGA)), on account of polymer NPs can serve as vaccine adjuvants to enhance antigen delivery and boost immune stimulation.102 A polymeric Toll-like receptor (TLR) 7 agonist NP adjuvant, developed by Sun et al., improves lymph node localization and induces long-lasting immune cell stimulation and widespread immune system reactions.103 This method improves the antibody reactivity to a SARS-CoV-2 subunit vaccination against various newly-emerging virus strains. Natural-sourced polymeric NPs are very affordable, water-soluble, and biocompatible. Chitosan (CS) or chitosan NPs can be used as adjuvants to boost the effectiveness of inactivated Rift Valley fever virus (RVFV) vaccinations. These adjuvants cause a cell-mediated immune response that is superior to that of inactivated RVFV antigens without adjuvants.99,104 Compared to natural polymers, synthetic polymer NPs typically have higher reproducibility and more controllable molecular weight composition and degradation rates. The most studied synthetic NPs include poly(glycolic acid) (PGA), PLA, and PLGA. It has been demonstrated that PLA and PLGA NPs improve humoral immunity following oral and pulmonary hepatitis B immunization.105
However, it is crucial to ensure the biodegradability of polymeric NPs to avoid their accumulation in the body. In addition, all degradation products that may be released by polymeric NPs throughout their lifecycle must be carefully considered to prevent any toxic effects on the host.
Protein-based NPs
Proteins and peptides are one of the main focuses of nanomedicine research and are mainly classified into animal proteins, plant proteins, and protein cages.106–108 Animal proteins including albumin, gelatin, collagen, milk, and silk proteins are good drug matrices. Plant proteins such as zeinolysin, wheat alginolysin, and lectins are commonly used as drug delivery carriers. Protein cages are structures derived from viruses or VLPs, which are essentially viral protein capsids without nucleic acids.109 Different viruses can produce viral cages of different shapes, uniform sizes, and good stability. Appropriate modification or modification of viral cages can achieve protein cages with multiple functions.110 In addition, ferritin/synuclein protein cages and small heat shock proteins can also be classified as protein cages.111,112
Protein NPs have several excellent features such as biocompatibility, low production cost, high cell binding capacity and targeting.113 As natural products, protein NPs have good biocompatibility, less toxicity, easy to be ingested by the body while degrading rapidly and fewer drug residues.114 Natural proteins are abundant and can be extracted directly, and the production methods of recombinant proteins are suitable for large-scale applications.107 In addition, proteins possess a variety of functional groups that can increase the amount and type of drug loading.115 The specific binding sites of protein NPs facilitate improved drug targeting.115 Different types of protein NPs each have characteristics that give them special functionality. Gelatin exhibits a rational ionic distribution with a balanced ratio of cations:anions:hydrophobic groups at 1:1:1, which makes it suitable for a wide range of pharmaceutical formulations.116,117 The reactive groups (arginine-lysine-glycine sequence) on gelatin are favorable for targeted treatment of infectious diseases such as acquired immune deficiency syndrome and malaria.118 Collagen NPs with their small size, large surface area, high absorption capacity and stable dispersion in aqueous solutions can be used as carriers for slow-release drugs, which are important in the antibacterial field.119 The protective effect of milk proteins is favorable for transporting some sensitive drugs and enhancing their stability.120 Plant proteins are mostly hydrophobic and are suitable for drug delivery of hydrophobic proteins.121 Lectins are resistant to hydrolytic degradation of proteins and have specific identification of intestinal glycosylation components and binding sites, which are beneficial for improved absorption of antiviral drugs.122,123 VLPs are a promising vaccine delivery system due to their non-infectious nature, great immunogenicity, and high biological activity.124–127 VLPs can also capture molecules such as proteins and nucleic acids, thereby acting as a vehicle to deliver these molecules to target cells and stimulate adaptive immunity.128–131
Nanoproteins have been used as important diagnostic and therapeutic agents for infectious diseases and inflammatory conditions. On the one hand, they can be used to make various biosensors to diagnose diseases, such as antibodies for detecting various viral diseases and glucose oxidase (GOx) for making glucose nanobiosensors.132 On the other hand, many proteins and peptides have been used in delivery of vaccines and drugs.
A special type of NPs-Exosomes
Exosomes, as a type of extracellular vesicle, are small vesicles secreted by cells and possess important biological functions. Based on their origins, exosomes can be classified into various types, such as tumor cell-derived vesicles, immune cell-derived vesicles, and stromal cell-derived vesicles. These vesicles play crucial roles in intercellular communication,133 modulation of antiviral immune responses,134 and participation in tissue repair.135 The small size, modifiability, compositional diversity, and heterogeneity of exosomes make them a new class of effective nanodrugs.
The size of exosomes is usually less than 200 nm, exosomes not only contain proteins involved in many basic cellular processes, such as cell adhesion, membrane fusion, metabolism, and signal transduction, but are also capable of delivering nucleic acids, including microRNAs (miRNAs), mRNAs, DNA, and other non-coding RNAs. The diverse compositions are the basis for their high biocompatibility and wide range of applications.136 In addition, exosomes can be modified by genetic or cellular engineering to introduce proteins or nucleic acids, which can increase the targeting and multifunctionality of exosome-based drugs.137,138
Currently, exosomes are mostly used as drug carriers. As nanocarriers, exosomes possess numerous advantages. Firstly, exosomes are autologous materials, exhibiting excellent biocompatibility and stability.139,140 Compared with other nanomaterials, they evoke lower immune system rejection responses. Secondly, exosomes can transport various drug molecules and enhance the bioavailability and therapeutic efficacy of drugs through specific targeting and transmembrane transport.141 In addition, exosomes exhibit greater advantages in mRNA formulations over liposomes. They not only demonstrate superior expression and safety,142 but also show enhanced lung retention time and distribution.143 The study of exosomes would be more accurate if limitations such as the complexity of the production, purification process, and difficulties in standardization could be overcome.
With intensive research on unmodified or engineered exosomes, researchers have now constructed a variety of exosome-based biotherapeutics that can be used to treat infectious diseases and inflammatory conditions. Exosomes act as delivery vehicles for existing drug molecules, nucleic acids, and proteins. Natural or modified exosomes can also be used as immunomodulators or ROS activators for the treatment of cancer or immune-related inflammation.144 In addition, exosomes’ unique nucleic acids and proteins allow them to be used as biomarkers involved in the diagnosis and prognosis of infectious diseases and inflammatory conditions.145,146
Other types of NPs
In addition to the aforementioned nanomaterials, combined NPs, biomimetic NPs, and QDs are also commonly used nanocarriers for drug delivery and diagnostics in infectious and non-infectious diseases. Various combined applications of NPs can supplement their shortcomings, produce synergistic effects, and make nanomaterials more developmental.147–149 Moreover, biomimetic nanotechnology has emerged and been used in the prevention and treatment of diseases, such as nanoenzymes and nanotoxins. Nanoenzymes are nanomaterials with enzymatic properties, characterized by high catalytic activity, stability, low cost, and scalability.150 It can be designed as a targeted delivery vehicle or simulate the catalytic generation of ROS, such as oxidases and peroxidases, which can simultaneously disrupt various essential biomolecules crucial for bacterial cell viability.151–155 Similarly, nanoenzymes can be encapsulated with antioxidants to combat oxidative stress and treat inflammatory diseases.156–162 Nanotoxins are NPs with membrane structures wrapping around bacterial toxins designed to reduce toxicity and increase biocompatibility.163,164 At present, nanotoxins have been developed as vaccines or drugs for the treatment of many diseases, such as cancer and bacterial infections.163,164 QDs are nanomaterials with unique optical properties that can be applied in bioimaging and diagnostics.165 QDs can be engineered into specific targeted probes for detecting the presence of pathogens, the expression of biomarkers,166,167 diagnostic imaging of neurodegenerative diseases,168,169 cardiovascular diseases,170,171 and more.
These different types of NP drug carriers have wide applications in the development of vaccines, delivery of anti-infective and anti-inflammatory drugs, and detection of pathogens and inflammation. They can improve the bioavailability, stability, and targeted delivery ability of drugs, contributing to improved anti-infective and anti-inflammatory efficacy and a balance between treatment safety and effectiveness. In addition, the unique optical and electrical properties of NPs enable their use in detecting viruses and pathogens, localizing, treating inflammation, and monitoring drug delivery in vivo. This not only improves the sensitivity and specificity of pathogen detection but also allows for diagnosis and treatment of diseases in a safe and non-invasive manner, without being limited by time or location. It should be noted that each NP drug carrier has its specific advantages and application scope, depending on the properties of the drug, delivery requirements, and treatment targets. Further research and evaluation are needed for the selection and design of specific diseases and drugs to ensure their safety and efficacy.
Nanotechnology’s application in infectious diseases
Infections are frequently caused by viruses, bacteria, fungi, parasites, and other microbes, which constitute a serious threat to human health. This section focused on how nanotechnology is being used to treat various infectious diseases, including the development of vaccination platforms, nanocarrier delivery systems, pharmaceuticals with direct anti-infective effects, and infectious disease diagnostic methods.172 The first part of Table 2 summarizes clinical studies of nanotechnology for infectious diseases.
Table 2.
Clinical studies of NPs for infectious and inflammatory diseases
| Role | Conditions | Interventions | Type | Primary outcome measures | Phases | Sponsor | NCT Number | Ref |
|---|---|---|---|---|---|---|---|---|
| Infectious diseases | ||||||||
| Vaccine | Coronavirus; COVID-19 | PepGNP-COVID19 | Peptide vaccine | SAEs; AESI | Phase1; Phase2 | Emergex Vaccines Holding Ltd. | NCT05633446 | |
| COVID-19 | mRNA-1273 | LNP mRNA vaccine | AEs; SAEs; MAAEs; AESIs; GMT; GMFR | Phase1; Phase2 | NIAID | NCT04889209 | ||
| COVID-19 | mRNA-1273 | LNP mRNA vaccine | MAAEs; NOCMCs); SAEs | Phase1 | NIAID | NCT04283461 | 761 | |
| COVID-19 | mRNA-1273.351 | LNP mRNA vaccine | MAAEs; NOCMCs; AESIs; SAEs; AEs | Phase1 | NIAID | NCT04785144 | ||
| COVID-19 | mRNA-1273 | LNP mRNA vaccine | Incidence; mean peak nasal viral load | Phase3 | NIAID | NCT04811664 | 762,763 | |
| COVID-19; HIV | mRNA-1273 | LNP mRNA vaccine | NAAT; ARDS | Phase2; Phase3 | COVID-19 Prevention Network | NCT05168813 | ||
| COVID-19 | HDT-301 | LNP repRNA vaccine | AEs; AESI; SAE; NOCMCs | Phase1 | HDT Bio | NCT05132907 | ||
| COVID-19 | HDT-301 | LNP repRNA vaccine | AEs; SAEs; AESI | Phase1 | SENAI CIMATEC | NCT04844268 | ||
| COVID-19 | BNT162b2 | LNP mRNA vaccine | GMTs; GMR; AEs; SAEs | Phase3 | BioNTech SE | NCT04816669 | ||
| COVID 19 | BNT162b2 | LNP mRNA vaccine | GMT; PRNT | Phase4 | The University of Hong Kong | NCT05057182 | 764 | |
| COVID-19 | PTX-COVID-19-B; Vaxzevria® | LNP mRNA vaccine | Immunogenicity response | Phase3 | Everest Medicines (Singapore) Pte., Ltd. | NCT05534035 | 765 | |
| COVID-19 | PTX-COVID-19-B | LNP mRNA vaccine | NT50; PBNA; AEs; SAEs; MAAEs; AESI; PIMMC | Phase3 | Everest Medicines (Singapore) Pte., Ltd. | NCT05534048 | ||
| COVID-19 | RNA MCTI CIMATEC HDT Vaccine | LNP; repRNA Vaccine | Ieutralizing antibody titers | Phase2 | Azidus Brasil | NCT05542693 | 766,767 | |
| COVID-19 | QTP104 | LNP repRNA vaccine | AEs; SAEs; AESI | Phase1 | Quratis Inc. | NCT05876364 | ||
| COVID-19 | AS03; BNT162b2; CoV2 preS dTM [B.1.351]; mRNA-1273 | LNP mRNA vaccine | GMFR; MSD; GMT | Phase1; Phase2 | NIAID | NCT05289037 | 768 | |
| COVID-19 | DS-5670a DAICHIRONA | LNP mRNA vaccine | AEs; GMT; GMFR | Phase1; Phase2 | Daiichi Sankyo Co. Ltd. | NCT04821674 | ||
| COVID-19 | ChulaCov19 vaccine | LNP mRNA vaccine | AEs; SAEs; GMT | Phase1; Phase2 | Chulalongkorn University | NCT04566276 | ||
| COVID-19 | Bivalent Moderna; Novavax | mRNA Vaccine; self-assembled protein NPs | IgG antibodies; solicited reactions | Phase3 | Murdoch Childrens Research Institute | NCT05658523 | ||
| COVID-19 | SARS-CoV-2 mRNA Vaccine | mRNA Vaccine | Primary efficacy endpoint | Phase3 | Walvax Biotechnology Co., Ltd. | NCT04847102 | ||
| COVID-19 | CoronaVac; Comirnaty | Inactivated vaccine; mRNA vaccine | GMT; PRNT | Phase4 | The University of Hong Kong | NCT05057169 | ||
| COVID-19 | SPFN_1B-06-PL; ALFQ | Ferritin-nanoparticle; LNP | Post-vaccination reactions | Phase1 | U.S. Army Medical Research and Development Command | NCT04784767 | 769,770 | |
| COVID-19 | COVID-19 rS | Self-assembled protein NPs | AEs; SAEs; MAAEs; AESIs; GMT; GMFR | Phase1; Phase2 | Novavax | NCT04368988 | ||
| COVID-19 | ChAdV68-S; SAM-LNP-S | Self-assembled protein NPs | AESIs; PIMMCs; MAAEs; NOCMCs | Phase1 | NIAID | NCT04776317 | 771 | |
| COVID-19 | GBP510&AS03 | Self-assembled protein NPs | AEs; SAEs; MAAEs; AESIs; GMFR | Phase1; Phase2 | SK Bioscience Co., Ltd. | NCT04750343 | 772 | |
| COVID-19 | GBP510 | Self-assembled protein NPs | AEs; SAEs; MAAEs; AESIs; GMT; GMFR | Phase1; Phase2 | SK Bioscience Co., Ltd. | NCT04742738 | 220 | |
| COVID-19 | ICC Vaccine | Self-assembled protein NPs | systemic AEs; MAAEs; AESIs; PIMMCs; SAEs | Phase1; Phase2 | Novavax | NCT04961541 | 773 | |
| COVID-19 | GBP510 | Self-assembled protein NPs | GMT; GMFR | Phase2 | Korea University Guro Hospital | NCT05175950 | ||
| COVID-19 | SARS-CoV-2 subunit protein recombinant vaccine | Self-assembled protein NPs | GMT | Phase2 | PT Bio Farma | NCT05525208 | ||
| COVID-19 | CIC Vaccine | Self-assembled protein NPs | AEs; MAAEs; AESIs; SAEs | Phase2 | Novavax | NCT05519839 | 774 | |
| COVID-19 | NVX-CoV2373 | Self-assembled protein NPs | AEs; SCR; GMT | Phase2 | Novavax | NCT05112848 | ||
| COVID-19 | SARS-CoV-2 rS/Matrix-M1 Adjuvant | Self-assembled protein NPs | (+) PCR-confirmed; AEs; MAAEs; MedDRA; GMTs; GMFRs | Phase2 | Novavax | NCT04533399 | ||
| COVID-19 | NVX-CoV2373; NVX-CoV2601 Bivalent BA.4/5 | Self-assembled protein NPs | GMTR; SRRs; NI | Phase2; Phase3 | Novavax | NCT05925127 | ||
| COVID-19 | COVID-19 vaccines | Self-assembled protein NPs | IgG antibodies | Phase3 | Murdoch Childrens Research Institute | NCT05387317 | ||
| COVID-19 | NVX-CoV2373; BBIBP-CorV vaccine | Self-assembled protein NPs | Utilizing ratio of IgG GMTs; MAAEs; AESIs; SAEs | Phase3 | Cogna Technology Solutions LLC | NCT05249816 | ||
| COVID-19 | COVID-19 Protein Subunit Recombinant Vaccine | Self-assembled protein NPs | GMT; seroconversion rate | Phase3 | PT Bio Farma | NCT05433285 | ||
| COVID-19 | Tozinameran; Elasomeran; Bivalent Pfizer; Bivalent Moderna | Self-assembled protein NPs | IgG antibodies | Phase3 | Murdoch Childrens Research Institute | NCT05543356 | ||
| COVID-19 | SARS-CoV-2 rS/Matrix M1-Adjuvant | Self-assembled protein NPs | Participants with symptomatic mild; moderate; or COVID-19 | Phase3 | Novavax | NCT04583995 | 775 | |
| COVID-19 | GBP510 adjuvanted with AS03 | Self-assembled protein NPs | GMTs | Phase3 | SK Bioscience Co., Ltd. | NCT05007951 | 776 | |
| COVID-19 | GBP510 adjuvanted with AS03 | Self-assembled protein NPs | GMFR | Phase3 | SK Bioscience Co., Ltd. | NCT05501522 | ||
| COVID-19 | NVX-CoV2515; NVX-Cov2373; NVX-CoV2540 | Self-assembled protein NPs | MN50; GMTs; SRRs; NAb | Phase3 | Novavax | NCT05372588 | ||
| COVID-19 | SARS-CoV-2 rS/Matrix-M1 Adjuvant (NVX-CoV2373) | Self-assembled protein NPs | Symptomatic; (+) PCR; MAAEs | Phase3 | Novavax | NCT04611802 | 207 | |
| COVID-19; HIV | Ad26.COV2. S Vaccine; SARS-CoV-2 rS; BNT162b2 | Self-assembled protein NPs | humoral immune responses | Phase2 | The Aurum Institute NPC | NCT05515042 | ||
| Influenza | H3 mRNA / LNP | LNP mRNA vaccine | AEs; CRF; SAEs; AESIs; HAI-Ab; GMTs | Phase1 | Sanofi Pasteur; a Sanofi Company | NCT05829356 | ||
| Influenza | DCVC H1 HA mRNA vaccine | LNP mRNA vaccine | AESIs; ILI; MAAEs; NOCMCs; SAEs; AEs | Phase1 | NIAID | NCT05945485 | ||
| Influenza | VRC H1ssF 3928 | LNP mRNA vaccine | AESIs; NOCMCs; SAEs; MAAEs; AEs | Phase1 | NIAID | NCT05755620 | ||
| Influenza | UFluA | Hemagglutinin stabilized stem nanoparticle vaccine | AEs; SAEs; AESIs; MAAEs | Phase1 | Emergent BioSolutions | NCT05155319 | 777 | |
| Influenza | Tri-NIV with NanoFlu | Self-assembled protein NPs | AEs; MAAEs; SAEs; SNMCs; MAE; SAE; HAI; GMR | Phase1; Phase2 | Novavax | NCT03293498 | ||
| Influenza | Quad-NIV | Self-assembled protein NPs | AEs; MAEs; SAEs; SNMCs; GMT | Phase2 | Novavax | NCT03658629 | ||
| Influenza | Quad-NIV with NanoFlu | Self-assembled protein NPs | GMFR; SCR; AEs; MAAEs; SAE; SNMCs | Phase3 | Novavax | NCT04120194 | 212 | |
| RSV | RSV mRNA LNP CL-0059&0137 | LNP RSV mRNA Vaccine | AEs; MAAEs; SAEs; AESIs; GMTs | Sanofi Pasteur; a Sanofi Company | Phase1; Phase2 | NCT05639894 | ||
| RSV; older adults | RSV-F Vaccine | Protein NPs | AEs; GMR; SCR; SRR | Novavax | Phase1 | NCT01709019 | ||
| RSV; Healthy volunteers | RSV-F Vaccine | Protein NPs | AEs; MAEs; SAEs; SNMCs; GMEU; GMR; SRR | Novavax | Phase1 | NCT02296463 | ||
| RSV | RSV-F Vaccine | Protein NPs | GMT; GMR; SCR; AEs | Novavax | Phase2 | NCT01704365 | ||
| RSV | RSV-F Vaccine | Protein NPs | GMEU; GMR; SRR; SCR2 and SCR4 | Novavax | Phase2 | NCT02593071 | ||
| RSV | RSV-F vaccine | Protein NPs | AEs; MAEs; SAEs; medically-attended LRTI | Novavax | Phase2 | NCT02247726 | ||
| RSV | RSV-F vaccine with adjuvant | Protein NPs | RSV LRTI or tachypnea 90 days | Novavax | Phase3 | NCT02624947 | ||
| RSV | RSV-F Vaccine | Protein NPs | Serum IgG antibody titers; GMEU; GMR; GMFR; SRR | Phase2 | Novavax | NCT01960686 | 214 | |
| EBV; Mononucleosis | EBV gp350-Ferritin Vaccine | Ferritin vaccine | Local and systemic reactogenicity; SAEs; AEs | Phase1 | NIAID | NCT04645147 | 778–780 | |
| EBV; Mononucleosis; Herpesvirus | EBV gp350-Ferritin Vaccine | Ferritin vaccine | mean EBV neutralizing antibody | Phase1; Phase2 | NIAID | NCT05683834 | 778–780 | |
| EBOV GP Vaccine | Ebola | Protein NPs | AEs; SAEs; MAEs; SNMCs; GMT; GMR; SCR; SRR | Phase1 | Novavax | NCT02370589 | 781 | |
| Drug delivery | ||||||||
| COVID-19 | Methotrexate-LDE | Lipid NPs | Lung injuries | Phase1; Phase2 | Azidus Brasil | NCT04352465 | ||
| Coronavirus; COVID-19; Inflammation | Methotrexate-LDE | Lipid NPs | Duration of hospital stay | Phase1; Phase2 | University of Sao Paulo General Hospital | NCT04610567 | ||
| COVID-19; Corona Virus | GS-5734 | Inhaled lipid NPs | AEs | Phase1 | NeuroActiva, Inc. | NCT04480333 | ||
| COVID-19 | VESTA respirator | Chitosan NPs | Incidence of laboratory-confirmed COVID-19 | NA | University of Brasilia | NCT04490200 | ||
| COVID-19 | Intranasal ivermectin spray | Aqueous nanosuspensions | Progression of COVID-19 clinical picture | Phase2; Phase3 | South Valley University | NCT04716569 | 263 | |
| Covid19 | MSC-exosomes | Exosomes | AEs | Phase1; Phase2 | AVEM HealthCare | NCT04798716 | ||
| COVID-19 | MSC-exosomes | Exosomes | Cytokine profile; inflammatory biomarkers | NA | University of Ulm | NCT05191381 | ||
| COVID-19 | Hemopurifier | Exosomes | AEs | NA | Aethlon Medical Inc. | NCT04595903 | ||
| Long COVID-19 Syndrome | UCMSC-derived exosomes | Exosomes | Cough Evaluation Test | Early-Phase1 | Huazhong University of Science and Technology | NCT05808400 | ||
| COVID-19 | Stem cell Exosomes | Exosomes | Symptom remission time; serum inflammatory markers | Early-Phase1 | First Affiliated Hospital of Wenzhou Medical University | NCT05787288 | 782,783 | |
| Severe COVID-19 | MSCs-derived exosomes | Exosomes | AEs; SAEs; TTIC | Phase1 | Ruijin Hospital | NCT04276987 | 250 | |
| Corona Virus; Pneumonia | CSTC-Exo | Exosomes | AEs; SAEs; TTCR | Phase1 | TC Erciyes University | NCT04389385 | ||
| COVID-19;ARDS | EV-Pure&WJ-Pure | Exosomes | AEs | Phase1 | Vitti Labs; LLC | NCT05387278 | ||
| COVID-19 | EXO-CD24 | Exosomes | AEs | Phase1 | Tel-Aviv Sourasky Medical Center | NCT04747574 | ||
| Corona Virus; COVID-19; SARS; ARDS | Zofin | Exosomes | AEs | Phase1; Phase2 | Organicell Regenerative Medicine | NCT04384445 | ||
| COVID-19 | Zofin | Exosomes | SAEs | Phase1; Phase2 | Organicell Regenerative Medicine | NCT05228899 | ||
| COVID-19 | EXO 1 &EXO 2 inhalation | Exosomes | AEs | Phase1; Phase2 | State-Financed Health Facility;Samara Regional Medical Center Dinasty | NCT04491240 | ||
| COVID-19 | CAP-1002 | Exosomes | Incidence of All-Cause Mortality | Phase2 | Capricor Inc. | NCT04623671 | ||
| COVID-19 | CovenD24 | Exosomes | SAEs; respiratory rate and SpO2 saturation | Phase2 | Athens Medical Society | NCT04902183 | ||
| COVID-19 | EXO-CD24 | Exosomes | Safety efficacy respiratory failure rate; death rate; PRO | Phase2 | Eli Sprecher; MD | NCT04969172 | ||
| COVID-19; ARDS | ExoFlo | Exosomes | 60-day Mortality Rate | Phase2 | Direct Biologics; LLC | NCT04493242 | 784 | |
| COVID-19 | EXO 1&EXO 2 | Exosomes | AEs | Phase2 | Olga Tyumina | NCT04602442 | ||
| COVID-19 | MSC-Exosome | Exosomes | Time to clinical improvement | Phase2; Phase3 | Dermama Bioteknologi Laboratorium | NCT05216562 | ||
| Antibiotic Resistant Infection | CIP-CS-PLGA-NPs | Chitosan coated PLGA NPs | Controlled release | Early-Phase1 | British University In Egypt | NCT05442736 | ||
| Cryptococcal Infections | Encochleated Amphotericin B | Lipid-crystal NPs | Tolerability of drug over 14 days | Phase1; Phase2 | Matinas BioPharma Nanotechnologies, Inc. | NCT03196921 | ||
| Bacterial Infections Oral | Chitosan | Coated PLGA NPs | Bacterial count | NA | British University In Egypt | NCT05475444 | ||
| Carious Lesion | Titania nanoparticle reinforced bonding agent | Titania nanoparticle | Post-restorative sensitivity | NA | Pakistan Institute of Medical Sciences | NCT05744648 | ||
| Glutathione-cyclodextrin Complex Absorption | GSH-CD | GSH-CD | GSH | Phase1 | Western University of Health Sciences | NCT05926245 | 423 | |
| Sepsis | Circulating Exosomes | Exosomes | mortality; All-cause mortality,28 days | NA | University of Kansas Medical Center | NCT04979767 | ||
| Sepsis; Critical Illness | MSC-EXO | Exosomes | The death rate of children | NA | Children’s Hospital of Fudan University | NCT04850469 | ||
| Drug-resistant | haMPC-Exos | Exosomes | Clinical cure rate,8 days | Phase1; Phase2 | Ruijin Hospital | NCT04544215 | ||
| Tinea | Oxiconazole nitrate SLNs loaded gel | SLNs | Clinical improvement; AEs | Phase1 | Minia University | NCT03823040 | ||
| Drug | ||||||||
| COVID-19 | Mouthwash and nose rinse with the AgNPs | AgNPs | Incidence of SARS-CoV-2 infection | NA | Cluster de Bioeconomia de Baja California, A.C | NCT04894409 | ||
| HIV | TLC-ART | DCNP | Cmax; Tmax | Phase1 | University of Washington | NCT05850728 | ||
| HIV | DermaVir; HAART | HIV-like particles | HIV-specific memory T cells | NA | Genetic Immunity | NCT00918840 | ||
| HIV | DermaVir; | HIV-like particles | Primary safety endpoint | Phase2 | Genetic Immunity | NCT00711230 | ||
| HIV | DermaVir; HAART | HIV-like particles | Grade 3 Adverse Event | Phase1 | Genetic Immunity | NCT00712530 | ||
| HIV | RPV | Nanosuspensions of pure drug | Cmax; AUC (last) | Phase1 | Janssen Infectious Diseases BVBA | NCT02547870 | 785,786 | |
| HIV | RPV | Nanosuspensions of pure drug | AEs | Phase1 | Janssen Research & Development, LLC | NCT01656018 | 265 | |
| HIV | V3G CH848 Pr-NP1; 3M-052-AF | Ferritin NPs; LNP | AEs; SAEs; MAAEs; AESIs | Phase1 | NIAID | NCT05903339 | ||
| Candida Infection | Titanium dioxide NPs | Titanium dioxide NPs | Bacterial colony-forming units | NA | Cairo University | NCT03666195 | ||
| Nosocomial Infections | AgNPS; copper NPs | Mental NPs | The inhibition zone; antibiofilm Activity | NA | Sohag University | NCT04775238 | ||
| Caries Class Ii | Nano Care Gold | Gold & AgNPS | Marginal adaptation | Phase1 | Cairo University | NCT03669224 | ||
| Fungal Foot Infection | Whitfield; zinc oxide NPs | Zinc oxide NPs solution | KOH test | Phase4 | Mahidol University | NCT05901961 | ||
| Foot Infection Fungal | AgNPS | AgNPS | The antimicrobial activity | Phase1 | Ahmed A. H. Abdellatif | NCT03752424 | ||
| Cutaneous Leishmaniasis | Sm29 Protein | AuNPS | Cure, 90 days | Phase1; Phase2 | Hospital Universitário Professor Edgard Santos | NCT06000514 | ||
| Dietary supplement | Recurrent Urinary Tract Infection | Magnalife | Nanotechnology Structured water | Urinalysis by GUE | NA | University of Sulaimani | NCT04306731 | |
| Sensors | Tuberculosis | Nanodisk-MS assay | Silicon NPs | Correlation, Sensitivity, specificity, positive predictive value, and negative predictive value | NA | Chinese University of Hong Kong | NCT03271567 | 430 |
| Biomarker | Sepsis With MOD | NTA double markers | Fluorescent NPs | Ubiquitination-autophagy-apoptosis biomarkers | NA | Taipei Tzu Chi Hospital, Buddhist Tzu Chi Medical Foundation | NCT03222986 | |
| Sepsis complicated with ARDS | Diagnostic test | Exosomes | Differential miRNAs | NA | Tianjin Nankai Hospital | NCT05476029 | ||
| Sepsis | Antibiotics | Exosomes | Amount of dendritic cell-derived exosomes | NA | Jinling Hospital, China | NCT02957279 | ||
| Inflammatory diseases | ||||||||
| Drug delivery | Rheumatoid Arthritis | Nanoparticulated rebamipide | LNEs | Change in the Clinical improvement of oral ulcers | Phase3 | Cairo University | NCT04649697 | |
| Atherosclerosis; coronary artery disease; inflammation | Methotrexate-LDE | LDL Like NPs | LAPV coronary | Phase2; Phase3 | University of Sao Paulo General Hospital | NCT04616872 | ||
| Coronary artery aisease; atherosclerosis; inflammation | LDE-Paclitaxel | LDL Like NPs | LAPV coronary | Phase2; Phase3 | University of Sao Paulo General Hospital | NCT04148833 | ||
| Atherosclerosis | Iron-bearing NPs | Iron-bearing NPs | TAV; QCA; IVUS | NA | Ural State Medical University | NCT01270139 | ||
| Coronary artery disease; atherosclerosis | Iron-bearing NPs | Iron-bearing NPs | TAV; IVUS | Phase1 | Ural State Medical University | NCT01436123 | ||
| Painful diabetic neuropathy | 0.75% capsaicin nanoparticle cream | SLNs | Pain relief | Phase2; Phase3 | Mahidol University | NCT01125215 | ||
| PreDiabetes | Zein nanocapsules | Zein NPs | Change in Fructosamine | NA | Clinica Universidad de Navarra, Universidad de Navarra | NCT05560412 | ||
| Chronic diabetic foot ulcer | Stem cell product | Chitosan NPs | Complete healing, full epithelization of chronic diabetic foot ulcer, 6 months | Phase1 | Assiut University | NCT03259217 | ||
| Plaque Psoriasis | SOR007 Ointment | LNEs | Change in the thickness of the ELB, 12 days | Phase1 | DFB Soria, LLC | NCT03004339 | ||
| Dementia; alzheimer Disease | APH-1105 | LNEs | ADAS-Cog | Phase2 | Aphios | NCT03806478 | 787 | |
| Tubular breast cancer; inflammatory breast cancer | Nab-Paclitaxel (Abraxane®) | Protein-NPs | Pathological complete Response (pCR=ypT0 ypN0) rates | Phase3 | German Breast Group | NCT01583426 | ||
| Inflammatory breast cancer | Carboplatin; paclitaxel albumin-stabilized nanoparticle formulation | Protein-NPs | Pathological complete response (pCR=ypT0 ypN0) rates | Phase2 | City of Hope Medical Center | NCT01525966 | 788 | |
| Breast cancer | capecitabine; paclitaxel albumin-stabilized nanoparticle formulation; neoadjuvant therapy | Protein-NPs | Pathological complete response rate | Phase2; Phase3 | Medstar Health Research Institute | NCT00397761 | ||
| Breast cancer; HER2-negative breast cancer | Carboplatin; paclitaxel albumin-stabilized nanoparticle formulation | Protein-NPs | Progression free survival | Phase2 | University of California, Irvine | NCT00618657 | ||
| Irritable bowel disease | Ginger exosomes | Exosomes | Change in inflammation on Colonoscopy | NA | University of Louisville | NCT04879810 | ||
| Ankylosing Spondylitis | Nanocurcumin | Nanomicelles spherical water | BASDI | Phase2 | Tabriz University of Medical Sciences | NCT03140657 | ||
| Drug | Chronic rhinosinusitis | Colloidal AgNPS | AgNPS | SNOT-22 | Phase1 | Washington University School of Medicine | NCT03243201 | |
| Knee arthritis; rheumatoid arthritis | Gold factor | AuNPs | KOOS | NA | 4Life Research, LLC | NCT05347602 | ||
| Type 1 Diabetes | C19-A3 GNP | Peptide-AuNPS | General safety and induction of hypersensitivity | Phase1 | Cardiff University | NCT02837094 | ||
| Inflammatory disease | Inhaled AgNPS | AgNPS | Ex vivo inflammatory response | NA | NIEHS | NCT02408874 | ||
| Crohn’s Disease | Placenal MSC derived exosomes | Exosomes | Safety of injected exosomes | Phase1; Phase2 | Tehran University of Medical Sciences | NCT05499156 | ||
| Multiple Sclerosis | Gold nanocrystals | AuNPs | CNS metabolic changes | Phase2 | Clene Nanomedicine | NCT03993171 | ||
| Dietary supplement | Type2 diabetes | Magnalife | Nanotechnology structured water | HbA1c | NA | ALI KAMAL M. SAMI | NCT04082351 | |
| Sensors | ||||||||
| Multiple sclerosis | NA-NOSE artificial olfactory system | Carbon nanotubes and AuNPS | Identification of volatile compounds in exhaled breath | complete | Carmel Medical Center | NCT01465087 | ||
| Multiple sclerosis | NA-NOSE artificial olfactory system | Carbon nanotubes and AuNPS | Successful discrimination | NA | Carmel Medical Center | NCT01206023 | ||
| Metabolic syndrome; diabetes | Breath analysis and blood analysis | nano-chemical sensors | Development of diabetes or complication | Phase1 | Rambam Health Care Campus | NCT01268813 | ||
| Neurodegenerative diseases (AD, PD) | ™NA-NOSE | Carbon nanotubes and AuNPS | NA | NA | Rambam Health Care Campus | NCT01291550 | 594 | |
| Imaging marker of MRI | ||||||||
| Multiple sclerosis | USPIO nanoparticle | USPIO nanoparticle | Signal change on T1-weighted and 3D UTE MRI brain | Early-Phase1 | University of Utah | NCT05357833 | ||
| Multiple sclerosis | Ferumoxytol | Iron oxide NPs | Brain signal intensity | Phase1 | NINDS | NCT02511028 | ||
| Myocardial infarction; inflammation | Cardiac magnetic resonance imaging | Iron-bearing NPs | Cardiac MRI signal intensity | NA | University of Edinburgh | NCT01127113 | ||
| Diabetes mellitus, type 1 | Ferumoxtran-10 | Iron oxide NPs | Changes in the pancreas associated with autoimmune diabetes | NA | Joslin Diabetes Center | NCT00585936 | ||
| Biomarker | Pleomorphic adenoma of salivary glands | CD24-Gold Nanocomposite | AuNPS | Non-conjugated CD24 | NA | Amina Fouad Farag | NCT04907422 | 789 |
| Hemodynamic instability; autophagy | Hemodynamic parameters | Exosomes | Change of hemodynamic parameters | NA | Taipei Tzu Chi Hospital, Buddhist Tzu Chi Medical Foundation | NCT03267160 | ||
Data available as of 15 September 2023. Data obtained from https://clinicaltrials.gov/. The first column highlights nanoparticles’ roles in diagnosing and treating diseases, such as their applications as vaccines, delivery vehicles, drugs, sensors, and diagnostic markers while the fourth column lists the various types of NPs
COVID-19 Corona Virus Disease 2019, CSTC-Exo COVID-19 Specific T Cell derived exosomes, AEs adverse reaction, SAE severe adverse reaction, TTIC time to clinical improvement, LNP lipid nanoparticle, NA not applicable, TTCR time to clinical recovery, PRO patient-reported outcome measure score, rep RNA representations of RNA sequences, SF-36 the 36 item Short Form Health Survey, GMT geometric mean titer, GMTR geometric mean titer ratio, GMFR geometric mean fold rise, SRRs seroresponse rates, NI non-inferiorto, NIAID National Institute of Allergy and Infectious Diseases, PRNT plaque reduction neutralization test, AESIs adverse events of special interest, PIMMCs potentially immune-mediated medical conditions, MAAEs medically attended adverse events, NOCMCs new onset chronic medical conditions, GMEU geometric mean EU, RBD receptor-binding domain, GMR geometric mean ratio, SRR seroresponse rate, RSV Respiratory Syncytial Virus, qNIV quadrivalent hemagglutinin (HA) nanoparticle influenza vaccine, CIC vaccine in-clinic mix of various doses of qNIV, SARS-CoV-2 rS, and 50 μg Matrix-M1 Adjuvant, ICC Vaccine qNIV and SARS-CoV-2 rS nanoparticle combination vaccine with Matrix-M1 adjuvant, SCR2 and SCR4 proportion of subjects with two 2- and 4-fold seroconversion rates, MedDRA Medical Dictionary for Regulatory Activities, NINDS National Institute of Neurological Disorders and Stroke, ALFQ army liposomal formulation QS21, AgNPs silver nanoparticles, pNT50 geometric mean neutralizing antibody titers against D614G pseudovirus strain, MN50 inhibitory concentration of 50%, LRTI medically-attended RSV lower respiratory tract infection, MSD Multiplex Meso Scale Discovery; ILI influenza like illnesses, EBV Epstein-Barr Virus, DLCD diffusing capacity of the lungs for carbon monoxide, CRF case report form, HAI-Ab hemagglutination inhibition HAI antibody Ab, PCR Polymerase Chain Reaction, NT50 the 50% neutralizing antibody titers, PBNA pseudovirusbased neutralization assay, IgG Immunoglobulin G, SARS CoV-2 spike RBD protein-specific binding antibody bAb IgG and SARS CoV-2-specific neutralizing antibody nAb, Cmax peak TLC-101 drug substance concentrations, AUC area under the plasma concentration, MSCs mesenchymal stem cells, UCMSCs umbilical cord mesenchymal stem cells, PLGA poly-lactic-co-glycolic acid, SNMC significant new medical condition, Tmax time to maximum TLC-101 concentration, NAAT nucleic acid amplification testing, MOD multiple organ dysfunction, NTA nanoparticle tracking analysis, ARDS Acute Respiratory Distress Syndrome, HIV Human Immunodeficiency Virus, RPV Rilpivirine, MTX-LDE lipid NPs carried methotrexate, DCNP drug combination nanoparticle, LNEs lipid nanoemulsions, ELB change in the thickness of the echolucent band, SNOT-22 Sino-Nasal Outcome Test, KOOS knee injury and osteoarthritis outcome score; LAPV low attenuation plaque volume coronary; LDL low-density lipoprotein; SLNs solid lipid nanoparticles, TAV total atheroma volume, QCA quantitative coronary angiography, CD24 cluster of differentiation 24, IVUS intravascular ultrasound, NA-NOSE nanoparticle nose ADAS-Cog, the Alzheimer’s Disease assessment scale-cognitive subscale test, ELB echolucent band, BASDI assessments of ankylosing spondylitis signs and symptoms, USPIO ultrasmall superparamagnetic iron oxide, GSH-CD glutathione–cyclodextrin nanoparticle complex
NPs in viral infection
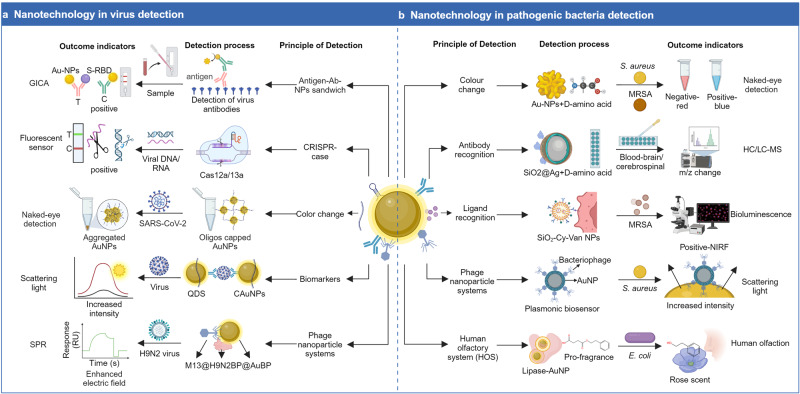
Since the beginning of the 21st century, there have been several global pandemics caused by viral infections, including Severe Acute Respiratory Syndrome Coronavirus (SARS-CoV) in 2003,173 H1N1 influenza in 2009,174 Middle East Respiratory Syndrome Coronavirus (MERS-CoV) in 2012,175 Ebola virus in West Africa from 2013 to 2016,176 Zika virus in 2015,177 and the SARS-CoV-2 pandemic in 2020.178 These outbreaks have resulted in significant morbidity and mortality, particularly the COVID-19 pandemic, which has had profound and devastating effects on individuals and societies worldwide.179 In the past few decades, numerous effective vaccines have been developed to control the spread of viruses such as smallpox, polio, measles, rabies, rubella, and tetanus globally or in specific regions.180 Traditional vaccines often produce low titers of neutralizing antibodies and may struggle to combat mutant pathogens. Previous treatments for viral infections have often been ineffective and associated with significant adverse reactions.181 Detection methods for pathogens have also been time-consuming, labor-intensive, and lacking in sensitivity and accuracy.182 The development of nanotechnology improves traditional methods for the prevention, detection, and treatment of infectious diseases.183
The application of NPs-based vaccine for virus
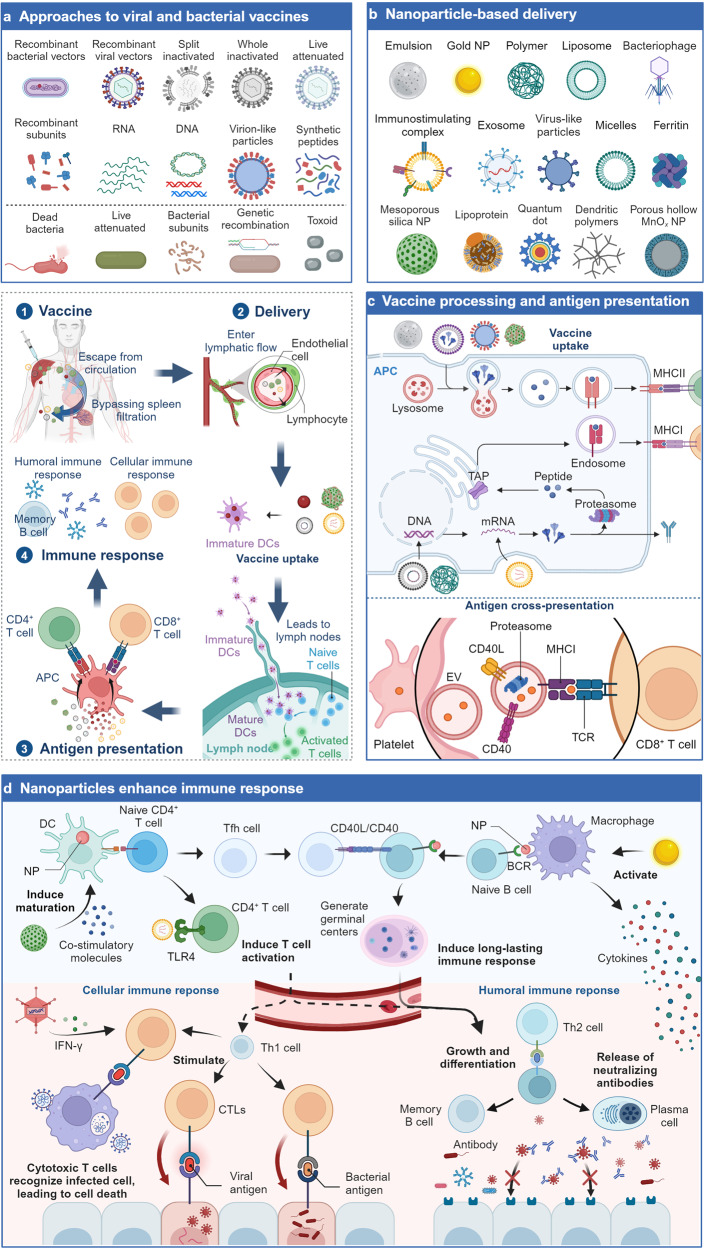
Nanotechnology has been applied to the development of vaccines as adjuvants and delivery vehicles to overcome the shortcomings of traditional vaccines, such as long development time, low immunogenicity, and antibody dependence. There have been many comprehensive reviews published on the application of nanomaterials as adjuvants,184 so we didn’t repeat here again. This section primarily focused on the application of NPs as vaccine delivery systems, with an emphasis on the most promising delivery platforms, including LNPs, self-assembled protein-based NPs, and exosomes.185 These nanotechnological strategies make immunizations more effective by improving vaccine stability, providing precise antigen presentation, and enhancing immune stimulation (Fig. 2).185–188
Fig. 2.
The application of NPs-based vaccines for pathogen prevention. After vaccination, due to the small size of the NP carrier, the nano-vaccines are more likely to escape from the bloodstream, bypass splenic filtration, enter the lymphatic flow, and then be absorbed by immature DCs, together with DCs, enter the lymph nodes, and initiate a series of immune reactions. a Types and methods for the production of viral and antibacterial vaccines. b Various nanomaterials used for antigen delivery. c Mechanisms of NP entry into cells and antigen presentation. d Mechanisms by which NPs enhance immune responses. NPs have diverse stimulating effects on the immune system, including inducing the production of co-stimulatory molecules to induce DCs maturation;795,796 promoting strong T cell activation;276 facilitating germinal center formation to induce long-lasting effective immune responses;797 and stimulating macrophages to produce cytokines to enhance immune responses.798 ①–④. The immune process of NP-based vaccines. TAP: transporter associated with antigen processing; Tfh: T follicular helper; Th2: T helper-2 cell; CTLs: cytotoxic T lymphocytes
LNP-mediated vaccines
The efficacy of nucleic acid-based vaccines depends mainly on the delivery of RNA or DNA molecules that express target-encoded antigens to trigger specific and strong immune responses in target immune cells.189 DNA vaccines have significant potential for the management of infectious diseases since they are easy, stable, and affordable to mass produce.189–191 mRNA vaccines have high antigen expression and quick clearance rates by directly expressing antigens in the cytoplasm without crossing the nuclear membrane.192,193 Nanotechnology-based delivery of plasmid DNA (pDNA) or mRNA molecules allows for the creation of precisely targeted nucleic acid vaccines. LNPs, as the delivery system for mRNA vaccines, can overcome the problem of naked mRNA transfection by stabilizing and successfully delivering it into cells.194 In phase I clinical trials, an mRNA vaccine that encodes the SARS-CoV-2 spike-in protein was reported to lower disease incidence, and viral replication was not seen in the lungs of rhesus monkeys exposed to large doses of the virus. The approved COVID-19 mRNA vaccines from Moderna and Pfizer/BioNTech are nanovaccines made from a cationic polymer/lipid complexed with negatively charged nucleic acids, which helps protect the mRNA from immune recognition and degradation.195,196 More importantly, the nano formulation may be effective for all current mutant strains including the Omicron variant.197 Recently, LNPs have been used to deliver the mRNA encoding SARS-CoV-2 S protein with incorporated ESCRT-I recruitment motif, enabling ESCRT-mediated secretion of viral spike protein VLPs from the cells. These VLPs displayed native, membrane-bound spike trimers on their surface, resulting in higher levels of neutralizing antibody titers 10 to 100-fold compared to soluble spike or commercial mRNA vaccines, and eliciting cellular immunity not achieved with mRNA vaccines alone. Notably, this VLP platform can be adapted to other viral antigens or mRNA cargoes, providing a promising direction for new vaccine development.198 Over the past few years, a number of mRNA vaccines against COVID-19 have been clinically studied and approved for use, the most representative lineage being mRNA-1273 and BNT162b2. Several clinical studies have demonstrated that mRNA-1273 provides strong protection (94.1%) in various age groups for more than 6 months (NCT04889209).199 However, despite the significant successful application of LNP-mediated mRNA in the COVID-19 vaccine, one limitation is that their stability requires freezing storage.200 For example, the vaccines developed by Moderna and BioNTech/Pfizer need to be stored at temperatures of −15 to −25 °C and −60 to −90 °C, respectively.200
In addition to COVID-19 vaccines, LNPs have potential in delivering mRNA of other viruses, enabling the development and application of vaccines for various viral diseases such as influenza, respiratory syncytial virus (RSV) and EBV. For instance, there have been reports on the design of an mRNA vaccine encapsulated in LNPs that expresses a variant of the RSV F protein. This vaccine successfully encoded multiple forms of RSV F protein in animal models and exhibited immunogenicity, providing protection against RSV infection.201 An LNP-mediated HIV-1 mRNA vaccine (gag mRNA/LNP) effectively enhanced the humoral and cellular responses previously induced by the DNA vaccine as a heterologous prime-boost regimen targeting monkeys.202 Furthermore, Peng et al. reported an effective LNP-mRNA vaccine targeting multiple pathogenic coronaviruses.203 These researches highlight the potential of LNPs in the development and application of vaccines for viral diseases. Three influenza mRNA vaccines are already in Phase I clinical studies, H3 mRNA/LNP, DCVC H1 hemagglutinin (HA) mRNA vaccine and VRC H1ssF 3928 (NCT05829356, NCT05945485, NCT05755620), and an RSV vaccine, RSV mRNA LNP CL-0059&0137 is in Phase II clinical trials (NCT05639894).
Self-assembled protein-based NP vaccines
Common self-assembling protein NPs, including VLPs, ferritin, and viral capsid proteins, have broad prospects in vaccine research and applications, demonstrating advantages in enhancing immune stimulation, antigen presentation, and physical stability.204,205 A recombinant SARS-CoV-2 spike protein vaccine developed by Novavax (NVX-CoV2373) produces full-length spike proteins that spontaneously form native trimeric conformations due to beneficial point mutations. This authorized vaccine exhibited robust immunogenicity and protection in baboon and mouse models, as well as demonstrated safety and efficacy in clinical trials (NCT04368988, NCT04611802).206,207 In another vaccine, an engineered protein combining the receptor-binding domain (RBD) domain of the SARS-CoV-2 spike protein with an HR motif self-assembles into a trimeric structure to mimic its natural conformation. In mouse and rhesus macaque models, this vaccine induced potent neutralizing antibody responses against both wildtype and variant SARS-CoV-2 strains, which led to its emergent approval in China.208 The VLPs that are made by the self-assembly of viral structural proteins have also been successfully applied in vaccines for various viruses, including HBV vaccines,209 HPV vaccines,210 and IV vaccines.211 A phase III clinical study of Quad-NIV with NanoFlu demonstrated that the qNIV vaccine was no less protective than the quadrivalent inactivated influenza vaccine (IIV4) in the elderly (NCT04120194).212 An anthrax vaccine used the coat protein of tobacco mosaic virus to deliver protective antigenic peptides of Bacillus anthracis.213 In addition, Novavax’s RSV vaccine has been shown to be well tolerated in clinical studies, with no adverse effects and a 52% reduction in infection rates in subjects overall (p = 0.009 overall) (NCT01960686).214
In addition to VLPs, other self-assembling proteins such as ferritin can also present antigens and stimulate immune responses. The spherical protein complex of ferritin forms a stable central cavity, which can be used to encapsulate target antigens and display them on the surface of ferritin. A SARS-CoV-2 vaccine made by conjugating the RBD of the viral spike protein to ferritin showed a higher affinity for the ACE2 receptor and neutralizing antibody CB6.215 Similarly, the safety and immunogenicity of a ferritin-based H2 influenza vaccine have been reported in a phase I trial, showing a safe, well tolerated and immunogenic potent in healthy adults.216
In addition, there are also proteins and peptides that have been designed as nanocarriers for viral antigens. For instance, a dengue virus E glycoprotein vaccine has been designed based on a polymeric IgG scaffold.217 Moreover, with the tremendous development of computational science, scientists can design ideal NPs based on experimental needs. A nanocarrier can display two different antigens by synthesizing two orthogonal reactive split proteins through the formation of heteropeptide bonds.218 A designed self-assembling protein NP I53-50 platform can display trimeric SARS-CoV-2 spike proteins on their surface, which elicited potent neutralizing antibody responses.219,220 Currently, NP vaccines based on self-assembling proteins against Lassa virus, HIV, HCV, and East Coast fever (ECF) virus have all shown good ability to induce neutralizing antibodies.221–224 These studies suggest that through antigen presentation by self-assembling protein vaccines, it is possible to mimic the structure and epitopes of pathogens, thereby activating the immune system to generate an immune response specifically targeting the desired antigens.
Exosomes-based vaccine
Exosomes, as cell-secreted products, have stronger capabilities in delivering vaccines without any side effects.225 There have been numerous studies utilizing exosomes to load with RNA or proteins for the COVID-19 vaccines. It has been discovered that the delivery capacity of exosomes is superior to LNPs, both in nucleic acids encoding antigens and protein immunogens.142 Another advantage of exosomes is their excellent affinity for target tissues. Exosomes derived from lung spheroid cells have excellent lung affinity compared to liposomes, enhancing the retention of the RBD in the mucosal lining of the respiratory tract and lung parenchyma.143 An inhalable COVID-19 vaccine that loaded recombinant SARS-CoV-2 RBD in lung-derived exosomes has a longer residence time in the respiratory tract and lung tissues after inhalation through nebulization.226 Furthermore, exosome-based vaccines have stronger immunogenicity due to their natural or immune-enhancing effects or their immune-modulating cargo, such as cytokines, nucleic acids, and lipids. Compared to Pfizer and Moderna’s mRNA vaccines and Oxford-AstraZeneca’s adenovirus vaccine, exosome-based vaccines demonstrate stronger immunogenicity, better stability, and easier storage.226 These benefits are attributed to the endogenous and natural homologous targeting ability of exosomes, demonstrating the superiority of exosomes in the field of viral vaccines.
Exosomes have been widely used in developing the multi-valent vaccine for SARS-CoV-2. It was found that exosomes loaded with two functional mRNAs induced long-term cellular and humoral immune responses against the spike protein and the nucleocapsid protein even after repeated injections. In mice experiments, this vaccine induced systemic humoral immune responses, including RBD-specific IgG antibodies and mucosal IgA responses in the lungs of mice. In addition, the vaccine activated CD4+ and CD8+ T cells with a Th1 cell cytokine expression profile, inducing a Th1-biased immune response and clearance of simulated SARS-CoV-2. Exosomes derived from milk have been used in an oral mRNA vaccine encoding the SARS-CoV-2 RBD.227 This vaccine successfully secreted RBD peptide in 293 cells and stimulated the production of neutralizing antibodies targeting RBD in mice. Furthermore, multi-valent COVID-19 vaccines containing spike proteins and nucleocapsid proteins of different SARS-CoV-2 strains have also developed based on exosomes, aiming to enhance the protective effects of the vaccines through combination strategies.228 In independent animal models, this vaccine induced potent and persistent neutralizing antibody responses at low doses and elicited strong T cell immune responses without the need for adjuvants.
Definitely, exosomes can also be used to develop other virus vaccines, such as HIV, HBV, HCV, IV, and rabies viruses. A targeted T-cell vaccine for HIV has developed with exosomes (Gag-Texo), which induced Gag-specific therapeutic immunity in a chronic adenovirus infection model.229 Exosomes derived from human monocyte cell lines hold promise as adjuvants for recombinant HBV vaccines. These exosomes could induce Th1 immune responses against HBsAg, leading to increased levels of IFN-γ in mice and promoting cellular immunity.230 Similarly, exosomes derived from umbilical cord mesenchymal stem cells (uMSC-Exo) can carry miRNAs to inhibit hepatitis C virus replication.231 Notably, outer membrane vesicles (OMV) derived from Gram-negative bacterium Burkholderia thailandensis were employed to express and package vaccine antigens derived from influenza A virus (IAV), inducing antigen-specific immune and antibody responses in mucosal tissues and systemically.232 Moreover, exosomes enhance the resistance of MRC-5 cells to rabies virus infection by delivering miRNA-423-5p between cells. Exosome-delivered miRNA-423-5p counteracts the inhibitory effect of cytokine signaling inhibitor 3 on type I IFN signaling, resulting in feedback inhibition of RABV replication.233 Overall, exosome-based viral vaccines are a promising new strategy to provide innovative immune defense against viral infections.
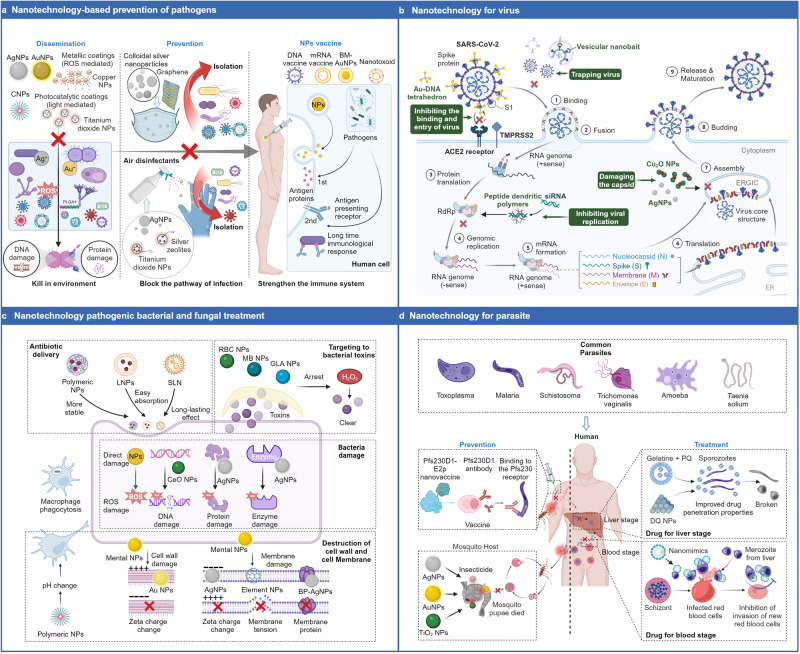
NPs in antiviral therapy
Nanotechnology with precise control over the properties and structures of nanomaterials holds tremendous potential in the field of antiviral applications. The application of nanotechnology in the antiviral field, including the efficient delivery of antiviral drugs, the blocking of viral infections, and the activation of immune responses, offers new strategies and approaches, bringing renewed hope for infectious disease treatment and prevention (Fig. 3b).
Fig. 3.
Applications of nanotechnology in the treatment of bacteria, fungal, viruses, and parasites. a Nanotechnology in the prevention of pathogenic bacterial infections, which include killing bacteria in the environment, blocking transmission routes and vaccination: AgNPs can release silver ions in the environment, generating ROS that damage the DNA and proteins of bacteria;358 AgNPs can also be applied to masks and disinfectants to prevent the invasion of pathogens and block infection pathways;799 Nanovaccines can enhance human immunity to improve resistance to infection. b Nanotechnology strategies against viruses. Taking the example of the coronavirus, nanotechnology can intervene in various processes of virus proliferation, such as capturing viruses outside nanocages, binding to spike proteins with nanocomplexes, inhibiting the binding and entry of SARS-CoV-2 into host cells. Similarly, targeting the RdRp complex can silence SARS-CoV-2 and inhibit its genome replication. AgNPs and other nanomaterials can also damage the viral envelope and disrupt the viral capsid. c Nanotechnology in antibacterial therapy. The antibacterial mechanism includes four aspects: the delivery of antibiotics,390 targeting bacterial toxins, damaging bacterial cell walls and membranes,402 and destroying bacterial DNA, proteins, and enzymes.417 d Nanotechnology applications against parasites. A variety of NPs targeting the parasite’s growth cycle have been used in the prevention and treatment of malaria: killing Plasmodium larvae in the environment and blocking the transmission pathway; generating antibodies to Plasmodium by nano-vaccination;438 and destroying Plasmodium in the liver and blood stages.437 RdRp: RNA-dependent RNA polymerase
Delivery of antiviral drugs
NPs can serve as delivery systems to effectively transport antiviral drugs to targeted cells or tissues, improving their targeting, stability, and bioavailability. Firstly, the antiviral drugs are encapsulated or adsorbed into the interior or surface of NPs, which protects the drugs from degradation factors in the external environment and enhances their stability. Due to their nanoscale size and unique surface properties, NPs can avoid excessive clearance by the immune system and exhibit prolonged circulation capability, increasing drug bioavailability and therapeutic efficacy.234–236 After reaching the target cells or tissues, NPs interact with the cell surface and promote their internalization through binding to specific receptors or the action of active targeting ligands.186,237–241 Once internalized, the NPs will release the encapsulated antiviral drugs by NP dissolution, receptor-mediated delivery, or stimuli-responsive release in response to the internal environment.242–247 In addition, the delivery system of NPs can achieve combination delivery of multiple antiviral drugs, combining different types of antiviral drugs together to enhance efficacy or combat drug resistance.248
Exosomes can effectively deliver a wide range of drug molecules such as nucleic acids, proteins, small molecules, and gene therapeutic agents, achieving high targeting and better therapeutic efficacy of antiviral drugs.
Protein drugs, such as membrane proteins, antigens, antibodies, etc., face challenges in maintaining their activity and extending their half-life when used for disease treatment. Due to the similar phospholipid bilayer structure to cell membranes, utilizing exosomes as delivery vehicles for membrane proteins can maintain their stability and achieve effective delivery. Exosomes enriched with an immune checkpoint modulator called CD24 (EXO-CD24) deliver the protein CD24 into the body through vesicular exosomes to regulate cytokine storms and combat COVID-19. In vitro and in vivo experiments have confirmed the safety and efficacy of EXO-CD24, with no drug-related adverse events reported even in a phase Ib/IIa clinical study.249 Furthermore, EXO-CD24 effectively reduces inflammatory markers and cytokines/chemokines in COVID-19 patients, establishing EXO-CD24 as a potential therapeutic strategy for inhibiting excessive lung inflammation in COVID-19 patients. Notably, several exosome-based drugs for the treatment of COVID-19 have entered clinical studies (NCT04798716, NCT04595903, NCT05787288). Amongst them, haMSC-Exos was well tolerated in the treatment of COVID-19 severe disease, with a significant remission of lung lesions after 7 days (NCT04276987).250 Zou et al. designed a method using exosomes to deliver IFN-induced transmembrane protein 3 (IFITM3) to fetuses for treating ZIKV infection.251 In their study, exosomes effectively transported IFITM3 across the placental barrier into late-stage fetal cells or lysosomes. The results showed that exosomes containing IFITM3 inhibited ZIKV in the fetus, significantly reducing viral viremia in the major organs of the fetus.
Nucleic acid drugs, especially miRNAs and small interfering RNA (siRNAs), are considered to have great potential for antiviral treatment. However, these types of drugs suffer from poor biological stability, easy degradation, and the potential to trigger immune reactions in the body, which limits their application. Using exosomes as delivery vehicles for nucleic acid drugs can overcome the aforementioned problems and facilitate the clinical application of these drugs. Exosomes delivered mRNA encoding ZFP-362, a zinc finger protein targeting the HIV-1 promoter and the active domain of DNA methyltransferase 3A, induce long-term stable HIV-1 epigenetic suppression, suppressing the HIV levels in the bone marrow, spleen, and brain of mice.252 A clinical study showed that LNP BMS-986263 could deliver siRNAs that degrade HSP47 mRNA, effectively treating pulmonary fibrosis caused by HCV infection (NCT03420768).253 Teng et al. reported that exosomes (exosomesNsp12Nsp13) released by lung epithelial cells exposed to the replicase Nsp12 and helicase Nsp13 of SARS-CoV-2 can activate nuclear factor κB (NF-κB), subsequently inducing a series of inflammatory cytokines, including tumor necrosis factor (TNF)-α, interleukin (IL)-6, and IL-1β, and leading to apoptosis of lung epithelial cells. However, they found that ginger exosome-like nanoparticles (GELNs) carrying miRNA could inhibit the activation of NF-κB and apoptosis of lung epithelial cells mediated by exosomesNsp12Nsp13, thereby treating exosomesNsp12Nsp13-mediated pulmonary inflammation. Furthermore, GELNs can also inhibit cellular pathological effects induced by SARS-CoV-2, revealing the potential of GELNs as a therapeutic agent for treating SARS-CoV-2.254 Zhang et al. designed an antiviral therapeutic system capable of crossing the placental barrier and blood brain barrier (BBB), which involves extracellular vesicles encapsulating ZIKV-specific siRNA.255 They found that this system protected pregnant AG6 mice from vertical transmission of ZIKV infection and could cross the placental barrier and BBB to inhibit ZIKV infection in the fetal brain. These examples demonstrate that exosomes are promising for a wide range of applications in the delivery of antiviral drugs.
Blocking viral infections
NPs can interfere with the binding between viruses and host cell receptors by introducing specific ligands or antibodies through surface functionalization or modification, thereby blocking the process of viral entry into host cells.
NPs can utilize their surface binding sites to interact with target molecules to inactivate viruses. Metal NPs can interact with viral surface proteins through Kazimir interactions, van der Waals forces and disulfide bonds.256,257 For example, AgNPs and AuNPs can cleave the disulfide bonds on the sulfhydryl groups of viral surface proteins, thereby preventing viral entry into cells.258,259 Cagno et al. created antiviral NPs with long, adaptable linkers that mimicked heparan sulfate proteoglycans, enabling efficient viral connection with a binding which they simulatively intended to be powerful and multivalent to the virus ligands repeating components, producing forces (190 pN) that ultimately result in permanent viral deformation.260,261 The IV is rendered inactive by the development of a gold-disulfide link between porous gold NPs (PoGNPs) and HA, which prevents membrane fusion and viral internalization.258 NPs may also bind with viral DNA or RNA to exert intracellular antiviral effects. AgNPs have been shown to inhibit by complexing S and O groups of thiols and phosphates on nucleic acids and amino acids, or by directly binding DNA or RNA to reduce the rate of viral reverse transcription.262 Glutathione (GSH)-Capped Ag2S Nanoclusters inhibit the synthesis of viral negative-strand RNA. Ivermectin can inhibit the replication of viral nucleic acids, and a clinical study has shown that topical application of ivermectin mucosal adhesion nano-suspension nasal spray is safe and effective in the treatment of patients with mild COVID-19. Respiratory manifestations (loss of olfactory sensation, coughing, and dyspnoea) can be rapidly restored in patients following treatment (NCT04716569).263 In addition, the NPs also induce the production of IFN-stimulated genes (ISGs) and pro-inflammatory cytokine, thereby potentially preventing the infection of porcine epidemic diarrhea virus.264 Nanovectors can block the interaction between the viral ligand and receptor on the host cell, thereby inhibiting the entry of viruses. This involves mechanisms such as receptor mimicry, spatial blockade, and chemical competition. Receptor mimicry is based on designing the nano carrier to have a structure or surface properties similar to the viral ligand, allowing the nano carrier to bind to the viral ligand and mimic its interaction with the cell receptor. By competitively binding to the viral ligand, the nano carrier can inhibit the normal interaction. AgNPs have been shown to block the gp120-CD4 interaction of HIV and even control infection by complexing S and O groups of thiols and phosphates on nucleic acids and amino acids, or by directly binding DNA or RNA to reduce the rate of viral reverse transcription.262 As same as nucleic acid inhibitors, long-acting ribavirin NPs demonstrate long-lasting (>4 months) inhibition of HIV replication in the rectum in a Phase 1 clinical study (p < 0.0001) (NCT01656018).265 Similarly, positively charged ZnO NPs block the interaction of SARS-CoV-2 with host cell receptors to disrupt virus-host cell binding.266,267 The presence of the nano carrier can introduce additional space, allowing it to compete with the viral ligand for binding sites on the cell receptor. As a result, the viral ligand is unable to effectively bind to the cell receptor. Mercaptoethane sulfonate-covered silver NPs (Ag-MES) inhibit herpes simplex virus-1 (HSV-1) infection by blocking viral attachment and entry into cells.268 AuNP spheres of 7.86 ± 3.3 nm size could interfere with the attachment of virus to Vero cells, inhibiting HSV-1 infection.269 Nanocarriers can competitively bind to viral ligands through mechanisms such as chemical interactions, charge interactions, hydrophilicity/hydrophobicity interactions, and other means. Fe2O3 and Fe3O4 NPs have been reported to alter the conformation of glycoproteins (E1 and E2) of and the stinging protein RBD of SARS-CoV-2.266 Boronic acid-modified lipid nanocapsules (BA-LNCs) are thought to prevent HCV from entering cells by forming a cyclic diester between the glycan on the HCV envelope protein and the BA part of the LNC.270 Highly positive-charged chitosan NPs interact electrostatically with negatively-charged viral surfaces.271 For example, the leading agent for human influenza in 2009 was the human influenza A/Puerto Rico/8//1934 (H1N1) virus (PR8), a subclass of IAV with a negative charge that is readily linked with positively charged polymers like chitosan or trimethylchitosan.272,273 In addition, targeted editing of the viral genome using the clustered regularly interspaced short palindromic repeats (CRISPR)-Cas system can also achieve the purpose of inhibiting viral replication and infection. One study used the CRISPR-Cas13 system to design and screen CRISPR RNAs (crRNA) that could target conserved viral regions and named PAC-MAN. Cas13d PAC-MAN showed excellent antiviral activity and effectively reduced H1N1 IAV load in respiratory epithelial cells.274 Subsequent work found that PAC-MAN inhibited many SARS-CoV-2 variants and multiple human coronavirus strains, reducing viral titers by >99%, as PAC-MAN can inhibit coronaviruses through cytosolic co-localization of crRNA with Cas13d and target viral RNA.236
Activating immune responses
NPs can initiate specific immune responses to facilitate the recognition and clearance of viruses by immune cells. In addition to the enhanced antigen presentation and immune stimulation mentioned earlier, NPs can also activate pattern recognition receptors (PRRs) and induce IFN production.
NPs trigger an immune response via activating PRRs. On one hand, the surface structure and composition of NPs can directly interact with PRRs, activating the signaling pathways of PRRs. For example, Montague et al. reported that surface-charged NPs can interact through charge interactions, binding to and activating platelet (PLT) glycoprotein receptors, enabling them to function as PRRs for both endogenous and exogenous charged ligands.275 On the other hand, certain NPs can be taken up by immune cells and enter the intracellular space. Within the cells, molecules released by the NPs can interact with PRRs, activating the corresponding signaling pathways, which leads to the activation of immune cells and initiation of an immune response. Zhang et al. reported that lipid-based NPs carrying mRNA vaccines can efficiently deliver mRNA to APCs while simultaneously activating TLR4 and inducing robust T cell activation.276 Notably, some NPs can activate PRRs without the need for phagocytosis. For example, Yazdi et al. reported that TiO2 NPs can trigger NLRP3 inflammasome activation signal without particle phagocytosis, leading to the release of IL-1α and IL-1β within cells, causing pulmonary inflammation.277 These examples indicate that the mechanisms of NP-induced PRR activation are complex, and different types of NPs may activate PRRs through different mechanisms. Therefore, in the design and application of NPs, it is important to consider the interaction mechanisms between NPs and PRRs in order to achieve the desired immune effects.
The NPs can also exhibit antiviral effects by inducing the expression of IFN and ISGs. Carbon dots (CDs) have been shown to significantly induce endogenous IFN and ISG production, thereby inhibiting viral replication.278,279 The antiviral activity of CDs was improved by surface chemical modification.280 In order to encapsulate 2’,3’-cyclic guanosine monophosphate-adenosine monophosphate (GAMP), an agonist of the IFN gene stimulant STING, Wang et al. created pulmonary surfactant (PS)-biomimetic liposomes and the PS-GAMP significantly enhanced influenza vaccine-induced humoral and CD8+ T cell immune reactions in mice.281 Similarly, Gly-CDs prepared by glycyrrhizic acid and CD together can also regulate the mRNA expression level of ISGs. In addition, Gly-CDs inhibit porcine reproductive and respiratory syndrome virus (PRRSV) replication by inducing the expression of host-restricted factors, such as DDX53 and NOS3, that are directly related to PRRSV proliferation.282
In addition, NPs modulate immune responses by preventing excessive formation of ROS. As the natural immunity is blocked by the viral infection-induced intracellular ROS, inhibitors that reduce ROS levels can probably stop the proliferation of viruses. Gly-CDs control the levels of intracellular ROS to prevent PRRSV replication.282 In this regard, oseltamivir-modified AgNPs (Ag@OTV) were more thoroughly investigated for their ability to inhibit virus activity. Ag@OTV significantly inhibited the levels of phosphorylated p53 and total p53 in MDCK cells, which suggests that Ag@OTV inhibits H1N1 IV-induced apoptosis in MDCK cells by activating the ROS-mediated activation of AKT and p53 phosphorylation.283
Nanotechnology in virus detection
The prevention of the epidemic and the treatment of diseases induced by viruses depend on the early diagnosis of infected microorganisms (Fig. 4a). Compared to traditional methods, nanotechnology has potential advantages in the detection and diagnosis of viruses. The size advantage of nanomaterials shortens the analysis time and improves the detection sensitivity, providing critical support for early diagnosis.284 Nanotechnology can also reduce sample consumption during the detection process, and integrate different detection modes such as optical, electrochemical, magnetic, etc., to provide complementary information and increase the reliability and accuracy of the detection results.285–288 Furthermore, the miniaturization characteristic of nanotechnology allows virus detection devices to be smaller, lighter, and even portable.289 This enables virus detection to be conducted outside the laboratory, in scenarios such as field settings, remote areas, or places with limited access to medical resources.290 In this regard, some nanomaterials, including AuNPs, ZnO/Pt-Pd, magnetic and QDs and graphene have been developed in recent years for virus detection and tracking.287,289,291–293 In recent years, there has been extensive research on applying nanotechnology to improve traditional virus detection methods, nanosensors and virus labeling. These studies have confirmed the tremendous potential of nanotechnology in the field of virus detection.
Fig. 4.
Nanotechnology in virus and pathogenic bacteria detection. This picture represents the detection principle, process and outcome indicators of nanotechnology. a Nanotechnology for virus detection: AuNPs based on the Antigen-Ab-NPs sandwich principle can be used to detect antibodies after vaccination with COVID-19,296 colloidal gold immunochromatographic assay; nanoprobes based on the CRISPR-CASE principle can be used to detect viral genes;800,801 capped by oligos AuNPs aggregation phenomenon and color change to detect SARS-CoV-2;320 AuNPs as biomarkers to track viral infections;313 NPs based on phage technology enhance SPR effect in virus detection.802 b Nanotechnology in the detection of pathogenic bacteria: AuNPs for the detection of bacteria by color change;429 SiO2@Ag+ detects infections in postoperative cerebrospinal fluid;314 SiO2-Cy-Van NPs detect MRSA by recognizing bacterial ligand MRSA;431 phage-coupled nanoparticles enhance the detection signal for S. aureus;434 Lipase-AuNP detects Escherichia coli by generating specific aroma.435 MRSA: methicillin-resistant Staphylococcus aureus
Nanotechnology can be used to improve traditional virus detection methods. Previously, semiconductor QDs were used to address the disadvantages of conventional fluorescent dyes in ELISA such as easy photobleaching, low quantum efficiency and wider fluorescence spectra. This is an advantage of QD as a marker in ELISA in place of traditional enzyme markers, and in recent years it has been found to also provide more sensitivity, stability, and multiplexing capabilities, contributing to the sensitivity and reliability of immunoassays. Using enzymatic in situ generation and immobilization of CdS QDs onto microspheres for the detection of the cancer biomarker superoxide dismutase 2, the immunoassay using enzymatically generated CdS NPs by electrochemical and fluorescence detection would have better detection limits of at least 3 orders of magnitude compared to previous studies.294 Similarly, Kurdekar et al. reported that a paper-based immunoassay platform based on carbon QDs demonstrated a higher detection range and shorter detection time for HIV antigen monitoring compared to traditional ELISA detection methods.295 A study developed a colloidal gold immunochromatographic assay (CGICA) based on the dual-antigen sandwich principle of AuNPs can detect both IgM and IgG antibodies against SARS-CoV-2 in human blood in a short period of time, which have high sensitivity and specificity.296 In comparison to ELISA and the rapid lateral flow immunoassay (LFIA), the lateral flow QIAreach anti-SARS-CoV-2 Total rapid NP fluorescence immunoassay not only shows comparable performance to ELISA but also has a shorter and simpler manual handling than LFIA.297
The CRISPR-Cas technique represents a groundbreaking gene editing technology and has emerged as a widely utilized nucleic acid assay with numerous applications.298 In contrast to traditional RT-qPCR and immunoassays, CRISPR/Cas-based methods offer significant advantages in viral detection, including scalability, rapidity, visual detection, high sensitivity, and specificity.299–302 Cas12a and Cas13a proteins can selectively target DNA and RNA, allowing them to play a crucial role in viral detection.303–307 For instance, the DNA Endonuclease-Targeted CRISPR Trans Reporter (DETECTR), based on Cas12, can detect SARS-CoV-2 in a significantly shorter time.308 Especially, the specific high-sensitivity enzymatic reporter unlocking technology (SHERLOCK) based on Cas13 demonstrated 100% specificity and 96% sensitivity when analyzing 154 clinical samples from COVID-19 patients, with sensitivity comparable to RT-PCR.309 The naked-eye detection of viruses can be carried out by combining nanotechnology with genetic detection techniques.310 The complementary cDNA sequences of viral proteins are loaded on gold nano-islands to make antisense oligonucleotide (ASO) capped plasma NPs. With the N-gene targeting NPs, SARS-CoV-2 can be selectively detected by colorimetric means without the need for complex instrumentation. These ASO-capped plasma NPs make naked-eye detection of SARS-CoV-2 possible by N-gene targeting-mediated selection.311
Nanotechnology can also be used to develop electrochemical sensors. These sensors utilize the unique properties of nanomaterials in electrochemical reactions to monitor the changes in current, potential, or impedance caused by viruses. AuNPs were employed in a dielectric electrophoresis array electrode in a study that generated a novel, low-cost electrochemical immunosensor for detecting MERS-CoV.312,313 Au3+-based ethylenediaminetetraacetic acid disodium salt (EDTA-2Na) chelators showed an increase in detection sensitivity compared to gold standard assays for HBsAg and alpha-fetoprotein by three more than three orders of magnitude.314
NPs can be used as viral markers to enable the rapid qualitative or quantitative detection of viruses by leveraging visual or fluorescence signal changes. For example, QDs are basic nanomaterials that are highly bound to fluorescent samples and can be applied for continuous fluorescence imaging and process-specific detection of cells.313 Self-assembled star-ridge plasmonic AuNPs published in 2017 were shown to help achieve the lower limit of detection values for detecting avian influenza A H5N1 virus concentrations at pmol levels, significantly improving detection sensitivity.315 In this study, self-assembled chiral QDs and AuNPs nanostructures for the detection of infectious bronchitis virus (IBV) in chicken blood samples were found to be highly effective for IBV detection.315 Metal NP probes may offer a crucial contribution to the identification of viruses via dark field microscopy.316 In addition, NPs as biomarkers can also be used to track the viral life cycle. During viral replication, streptavidin-coupled QDs labeled with self-biotinylated baculovirus nucleocapsid proteins were specific and practical, while did not affect viral infectivity.317 Duval et al. used single-particle imaging to monitor the uncoating of particular IAV virions by embedding QD-conjugated virus ribonucleoprotein combinations inside pathogenic IAV virions. Their research reveals vRNP trafficking and uncoating mechanisms, which could help researchers create novel methods to prevent IAV infection.318
Biomaterial-based nanodetection technologies have rapidly advanced. Phage-based nanoprobes have been developed and are being utilized for disease detection.319 Quantification of miRNAs through T7 phage enables effortless and precise detection of miRNAs even without the aid of laboratory equipment.320 Phage-based gold NPs may enhance the detection sensitivity significantly. Taking M13@H9N2BP@AuBP as an example, it exhibits an SPR with an electric field 40 times higher than that of traditional AuNPs. This results in a high detection sensitivity for H9N2, reaching as low as 6.3 copies/mL (approximately 1.04 × 10−5 fM).287
These applications demonstrate that nanotechnology has advantages in the field of virus detection, including high sensitivity, high specificity, rapid detection, and reliability. With the continuous development and innovation of nanotechnology, we can expect to see more virus detection methods based on nanotechnology, providing strong support for disease prevention and control.
NPs in pathogenic bacterial and fungal infections
Pathogenic bacterial infections, alongside viral infections, have become a significant contributor to the burden of disease. The prevalence of single or multidrug-resistant bacteria poses a significant threat to existing antimicrobial agents.321,322 Recently, nanotechnology has shown promising applications in bacterial and fungal infectious diseases. In this section, we described the application of NPs in the prevention, treatment, and detection of bacterial and fungal infections. Emerging nanovaccine technologies are contributing to the rapid development of more effective and long-lasting antimicrobial vaccines. Nanomaterials can perform multiple bactericidal pathways, including the delivery of conventional antibiotics, interaction with cell walls or cell membranes, disruption of the internal structure of the bacteria, and removal of toxins produced by the bacteria. Nanotechnology also holds new promise for the detection of bacteria.
Nanotechnology in the prevention of pathogenic bacterial infections
NPs, with their obvious advantages as vaccine adjuvants and delivery vehicles, are also playing an increasingly important role in the prevention of bacterial infections (Fig. 3a). The implementation of nanotechnology in antimicrobial vaccines may enhance the biocompatibility, immunogenicity and antigen presentation ability. In addition to pathogen-specific methods such as vaccines, directly targeting the source of infection in the environment and cutting off the transmission routes are also effective ways to prevent bacterial infections, such as novel NP fungicides and disinfectants.
Nanotechnology used for antimicrobial vaccines
The emerging nanovaccines that are currently being used for the prevention of pathogenic infections fall into the following broad categories: DNA vaccine,323,324 mRNA vaccine,325 protein vaccine,326 OMV,327,328 and nanotoxoids.329
As a third-generation vaccine, DNA vaccines have long been used to prevent foreign invaders by triggering humoral and cellular immune responses against foreign genes.330 The shortcomings of low immunogenicity of DNA vaccines are being overcome by the advent of nanotechnology.331 Various nanomaterials have been developed for DNA vaccine delivery, including polymeric NPs,332 liposome NPs,333 virus-like particles,334 and self-assembling proteins.335 PLGA has been widely used in the design and development of DNA vaccines against tuberculosis because of its immunological properties and good biocompatibility.336 Compared to naked plasmid-DNA vaccines, delivery based on liposomal nanomaterials effectively prevents extracellular nucleic acid endonucleases from degrading DNA through interactions with the cell membrane, thus improving the efficiency of plasmid-DNA transfection presentation.337 In addition, modified liposomes with appropriately targeted ligands stimulate and activate immune signal pathways via PRRs, leading to maturation of APCs and antigen processing and presentation.333
mRNA vaccine technology has rapidly advanced during the COVID-19 pandemic, with well-demonstrated safety and efficacy.338 In addition to its application in oncology and viral infections, mRNA vaccines could also prove beneficial in combating bacterial and fungal infections. Nevertheless, when compared to viruses, bacteria are known to express thousands of proteins, which presents a challenge in choosing the appropriate protein antigen to target. Despite the high potential of mRNA vaccines in treating (intracellular) bacteria, only a limited number of studies have explored this promising avenue.339,340 Mayer et al. identified 42 Listeria monocytogenes immune peptides from 68 different bacterial antigens. Subsequently, they incorporated these highly expressed antigens as vaccine candidates in mRNA vaccine formulations that significantly reduced bacterial load in the liver and spleen.341 Moreover, drug tolerance is a major problem in bacterial and fungal infections, and the rise of mRNA vaccine technology is expected to attenuate antimicrobial resistance (AMR).342 Recently, Maruggi et al. investigated the immunogenicity and efficacy of a self-amplified mRNA (sa-mRNA) vaccine against group A (GAS) and group B (GBS) streptococci.340 The sa-mRNA vaccine elicited antibody responses and provided protective effects, along with the induction of antigen-specific IgG2a (Th1) responses. This study demonstrated the potential of mRNA vaccines against AMR pathogens.
OMVs are ~20–250 nm nanostructures produced by vesiculation of the bacterial cell envelope.343 The vesicles contain a variety of membrane proteins that enhance PRR binding to PAMP on APCs, thereby activating the immune system.344 The small nanoscale size of OMVs facilitates their penetration into lymph nodes and boosts the uptake by APCs, thus enhancing immune activation. OMVs have progressed to the clinical phase as an antimicrobial vaccine, including those derived from Streptococcus meningitidis and Staphylococcus aureus.345 Besides, OMVs can also be genetically engineered and modified.346 For instance, enhancing the genetic makeup of E. coli to produce OMVs that contain modified polysaccharides offers a potential strategy to enhance the effectiveness of vaccines.347 OMVs can also be combined with NP delivery systems to enhance vaccine efficacy. The release of OMVs by Shigella fowleri bound in poly(anhydride) NPs enhances bioadhesive interactions and triggers innate immunity mediated by TLR2 and TLR4.348 Furthermore, the OMVs can be utilized as a delivery system, and if encapsulated with other NPs, the efficacy can be improved. OMV-encapsulated AuNPs can be employed to precisely regulate the size distribution of the ultimate formulation.349 This method not only improves the stability of AuNPs but also takes advantage of the multivalent antigenic properties of OMVs.
Germ infections are frequently accompanied by the discharge of harmful toxins, which can result in severe consequences like cellular damage, haemolysis, the destruction of the immune system and even sepsis. The toxoid vaccine strategy can efficiently avert the poisonous outcomes of germ infections and decrease host stress.350,351 Nanotoxins based on NP designs have been used to enhance the effectiveness of vaccines.352 Nanotoxins increase protective immunity and antibody titres in vaccinated mice with lower cytotoxicity compared to conventional inactivated toxin vaccines. Recent studies have shown that erythrocyte-based nanotoxins, are capable of neutralising a wide range of bacterial toxins, including alpha toxin,353 bee toxin, listeriolysin O and streptococcal haemolysin O.354 These nanotoxins effectively prevent toxins from binding to cell membranes. In addition, cell membranes are used to encapsulate various types of bacterial toxins, resulting in the development of nanovaccines with a broader range of targets. The cell membrane structure of these NPs allows them to be compatible with different cell membranes, enhancing their versatility.355,356 For example, NPs encapsulated with mouse macrophage membranes (MM) have demonstrated the ability to capture and neutralize endotoxin activity such as lipopolysaccharide (LPS).357 The adjustability of the cell membrane coating and the versatility of the various toxins enable the creation of a nanotoxin-based platform that can serve as a vaccine against numerous pathogenic bacteria, encompassing both Gram-negative and Gram-positive strains.
In conclusion, existing research has shown that vaccines based on various nanotechnology approaches can achieve effective prevention of pathogenic bacterial infections. This significantly reduces the risk of widespread bacterial infections and also alleviates some of the burden in treating drug-resistant bacteria.
Nanotechnology used for pathogens eradication and transmission prevention
In addition to vaccination for enhancing host immunity, using antimicrobial materials to directly inhibit or kill pathogenic microorganisms in the environment and disrupt their transmission is a straightforward and highly efficient method for source control. Currently, most of these materials are metallic NPs, including AgNPs,358,359 AuNPs,360 Carbon-modified AuNPs,361 copper NPs,362 and cerium oxide NPs.363 Besides, polymeric NPs and carbon-based NPs are also employed in the prevention of pathogenic bacteria.364,365 These NPs are often found in the form of disinfectants or other protective gear. For example, disinfectants containing AgNP can kill bacteria and fungi in a broad-spectrum manner.366,367 Tissues coated with selenium NPs effectively inhibit the growth of a wide range of Gram-positive and Gram-negative bacteria.368 Textiles functionalized with CuO NPs can be made into masks, gloves, wipes, etc. with excellent antibacterial effects.369
Pathogenic bacteria in the environment are typically found suspended in the air, present in water, or adhering to solid surfaces. Therefore, the main strategies for preventing bacterial infections include bactericidal actions, anti-biofilm measures and antiadhesive effects.370,371 Studies have shown that the small particle size of AgNPs can easily enter the interior of bacteria and destroy cell walls and membranes. Simultaneously, AgNPs can generate ROS and free radicals, destroying cell contents and leading to cell death, thereby achieving broad-spectrum antibacterial effects.372 In terms of the prevention of adhesion and biofilm formation by Candida species, a number of nanomaterials have been developed.373
In addition, nanotechnology-based antimicrobial materials can be applied in various potential infection scenarios.374 The most common situations involve infections originating from food and the oral cavity. Utilizing nanomaterials to eliminate pathogenic microorganisms within food products and preventing disease entry through the mouth is a highly effective method for preventing microbial infections.375 For example, coating palladium NPs and platinum NPs on food packaging bags can effectively kill common food-borne pathogens Salmonella enterica infantis, Escherichia coli, Listeria monocytogenes, and Staphylococcus aureus, etc.376 Implant-associated infections are also a major problem in current surgical procedures, and a variety of nanomaterials have been used for surface modification of implants, such as FeNPs, AgNPs.377,378 NPs are also interesting materials for promoting wound healing, and AgNP polymers embedded in wound dressings inhibit microbial growth and promote wound healing.379 In addition, L-arginine-containing mesoporous silica NPs were used in dental adhesives to effectively prevent dental caries.380
In conclusion, nanotechnology-based materials have been able to effectively prevent bacterial and fungal infections in multiple forms and pathways.
NPs in antibacterial therapy
Once the bacterial or fungal infection occurs, the human system will be affected by a series of inflammatory reactions, leading to various degrees of tissue damage and even systemic infections with serious consequences. Nanotechnology-based drugs have shown promising results in the treatment of such infectious diseases.381–384 Here, we summarized the application of NPs in the treatment of pathogenic bacterial infections and their mechanisms (Fig. 3c), including the delivery of drugs to overcome the drawbacks of conventional antibiotics, the direct targeting and killing of bacteria at the site of infection, or the removal of bacterial toxins to reduce the extent of damage.
Nanomaterials as antimicrobial delivery vehicles
The main advantage of NPs over traditional delivery systems is their size. The size of NPs is very small and controllable, suitable for carrying antimicrobial agents and against bacteria. NPs-based antimicrobial delivery effectively improves the bioavailability of antibiotics, reduces toxicity and side effects, overcomes drug tolerance, and enhances resistance to intracellular infection. The poor solubility, first-pass effect and inadequate targeting of traditional antibiotics contribute to their low bioavailability, which can be overcome by nanotechnology drugs.
NP delivery systems can improve the performance of prodrugs in the following ways: reducing the particle diameter to facilitate absorption,385 increasing the surface charge of particles to enhance drug binding to cells,386 and altering the oil-water partition coefficient to increase drug water-solubility or lipophilicity.387 Self-assembled chitosan NPs developed by Wu et al. can improve the dispersibility of licoricidin, effectively overcoming the poor water solubility, instability, and low bioavailability of natural flavonoids.388 In addition, the preparation of antibiotics as nanocrystals is also a means of overcoming poor bioavailability: dapsone nanocrystal suspensions produced by Nataly et al. are smaller, more soluble and more stable compared to the original drug.389 SLNs loaded with antimicrobials were found to improve the effectiveness of fluconazole (FLZ) in treating phytolacca vulgaris. Clinical studies demonstrated that FLZ-SLNs exhibited high encapsulation rate, good stability, prolonged release time, and higher cure rate compared to Candistan 1% cream.390 Nano-curcumin possesses potent antifungal properties analogous to mycobacteria. However, its therapeutic effects have slower onset compared to mycobacteria. The enhanced bioavailability of curcumin in nano form makes it a promising alternative therapy for thrush, with the potential to avoid diseases associated with mycophenolate.391
Antibiotics have prevalent adverse effects including GI irritation, damage to human commensal microorganisms, liver, and kidney toxicity, etc. Formulating multiple drugs within a nanocarrier system can enhance efficacy by increasing local concentrations while decreasing systemic concentrations, leading to fewer side effects. Gounani et al. loaded polymyxin B and vancomycin (VAN) onto NPs to achieve simultaneous local delivery of antibiotics against both Gram-positive and Gram-negative bacteria, which effectively reduced the required drug dosages.392 Fernández et al. assessed a nystatin nanoemulsion optimal for topical use, circumventing risks like systemic absorption and toxicity.393 Furthermore, NP modification can reduce drug toxicity. Ludmila et al. developed a stable chitosan-conjugated AgNPs form exhibiting non-toxicity to normal fibroblasts even at high concentrations.394
The application of NPs represents a promising approach to overcome microbial drug resistance. Simultaneously encapsulating multiple antimicrobial agents within a single NP can effectively eradicate bacteria and reduce the development of resistance.381 In addition, the intrinsic and extrinsic properties of NPs, such as their functional groups, concentration, and size, significantly influence the development of antibiotic resistance.395 Recent studies have made advances in the surface modification of nanocarriers for antimicrobial delivery.396 For instance, folic acid (FA)-modified LNPs improved the efficacy of VAN therapy.397
NP delivery systems are promising tools for the treatment of intracellular bacterial infections and for the prevention of disease recurrence caused by these bacteria. The pathogenic bacteria can escape host cell killing through various pathways.398,399 The drug-loaded NPs are recognized as foreign substances by the MPS and phagocytosed, allowing the drug to be released from the NPs to eliminate bacteria hidden inside macrophages.400 Zou et al. constructed pH-responsive NPs using cinnamaldehyde (CA)-dextran conjugates as carriers and loaded with glabridin (GLA). These NPs can target macrophages infected by MRSA. They not only reduce the risk of macrophage damage and intracellular bacterial spread but also rapidly kill intracellular MRSA with a very low probability of drug resistance.401
Destruction of cell wall and cell membrane
The cell wall and cell membrane of bacteria play a crucial role in their pathogenicity and the development of drug resistance. Therefore, exploring antimicrobial treatment approaches targeting these components holds significant promise.402 NPs can effectively impact the cell walls and cell membranes of pathogens, leading to pathogenic damage. These actions involve direct destruction of the cell wall or cell membrane, impairment of the efflux pumps on the cell membrane, and interruption of transmembrane electron transfer.403
One of the primary ways in which nanotechnology drugs interact with pathogenic cell walls is through a mismatch between the charge carried by the NPs and the surface charge of the cell wall, this imbalance can lead to cell wall damage or rupture, resulting in an antimicrobial effect. Metallic NPs are typically utilized for this purpose.404 The AgCuE nanosystem can damage the cell wall, leading to the death of planktonic Candida albicans and achieving therapeutic results for fungal keratitis.405 The intracellular pH-responsive pathway serves as a vital antimicrobial tool, often in combination with cell wall disruption techniques.406–408 Astodrimer has proven effective in inhibiting the growth of microorganisms involved in the pathogenesis of bacterial vaginosis. Its antimicrobial mechanism focuses on blocking bacterial adhesion, disrupting biofilm formation, and preventing further biofilm development. Several clinical trials have been conducted to evaluate its effectiveness.409,410 Similar to disrupting the cell wall mechanism, NPs can also act as antimicrobial agents by modifying the membrane potential and impeding cell membrane formation and maturation.411 Furthermore, multi-principal element NPs represent an emerging class of materials with potential applications in medicine and biology.412 NPs can also absorb bacteria, leading to increased membrane tension and deformation, ultimately resulting in cell rupture and death.413 In addition, a more precise method is to devise NPs that target proteins in the bacterial cell membrane. The CBD-EGFP-BP-AgNP complexes developed by Domyoung Kim and colleagues have the ability to specifically recognize the target cells and exhibit superior bactericidal effects.414 Khare et al. designed a set of multi-combination NPs named Emb-Chi-Au NPs, which can be used against multidrug-resistant bacteria by suppressing the efflux pumps on bacterial membranes.415
Production of ROS and damaging intracellular component
Affecting the production and maturation of bacterial or fungal cellular contents is also a major antimicrobial strategy. Nanomedicine can not only invade pathogens and directly kill bacteria by destroying nucleic acids and proteins inside them. It can also induce the production of ROS in microorganisms, damaging cellular components and thereby indirectly killing pathogens. In addition, it can also induce the production of ROS at the site of infection for antibacterial effects. Bacteria contribute to the dissemination of antibiotic resistance genes in the environment by ingesting and directly converting extracellular DNA (eDNA). Xu et al. treated Escherichia coli with CeO2 NPs, which inhibited the expression of DNA uptake and processing-related genes by directly binding eDNA, reducing ROS levels and cell membrane permeability. This antimicrobial strategy can inhibit bacterial growth and reproduction at the source.416 Different types of SeNPs designed by Fresneda et al. can exert their antibacterial effects by increasing the ROS content in bacteria. Among them, undefined-SeNPs perform best in inducing DNA damage (around 80% of DNA degraded).417 The multifunctional gold-silver carbon QDs nano-hybrid composite constructed by Ang and colleagues can also increase the ROS content in bacteria and play an antibacterial role under both UV irradiation and non-irradiation conditions.418
Interestingly, infection of the body by pathogens is accompanied by a series of inflammatory responses, which include the production of ROS. As powerful oxidants, ROS have the ability to completely disrupt the entire biofilm and play an important biological role in the defense against pathogens.419 How can the production of endogenous ROS act selectively as an antibacterial agent? Currently, nanoenzymes are commonly used in this antimicrobial approach. Nanoenzymes are artificial enzymes that imitate the activity of natural enzymes and generate antimicrobial effects by accelerating the production of ROS.420,421 The efficient antimicrobial activity of nanoenzymes hinges on the unification of enzyme-like activity and bacterial binding capacity within a single drug. The development of ROS nanoenzymes was reported by Gao et al. that are attached to a surface and are capable of targeting and destroying bacteria selectively in mammalian cells. In addition, these enzymes can reduce the risk of drug resistance.422 GSH is an important intracellular antioxidant that neutralizes ROS. Topical application of GSH-cyclodextrin NP complex (GSH-CD) showed beneficial therapeutic effects on mycobacterial infections.423 After three consecutive days of GSH-CD application, GSH levels increased, MDA levels decreased and the prevalence of Mycobacterium avium infection in whole blood cultures of the clinical trial participants was significantly reduced (NCT05926245).
Targeting bacterial toxins
During the procedure of bacterial infection, bacterial cells can secret toxins to damage host tissues, which will enable the penetration of bacteria into deep tissue.424 NPs can remove bacterial toxins by inhibiting the secretion of toxins from pathogens, binding and removing bacterial toxins.
A toxin-responsive NP was designed by Han et al.425 When NPs encounter bacteria within the body, the nanoreactor can capture and break down any toxins secreted by the bacteria. A biomimetic nanosponge that absorbs pore-forming toxins was reported by Hu et al. NPs that are coated with red blood cell (RBC) membranes are utilized as treatment for bacterial sepsis by neutralizing multiple kinds of bacterial toxins and killing bacteria.426 For intracellular infection, Zou et al. found that GLA-loaded pH-responsive NPs released CA downregulated the expression of cytotoxic pore-forming toxins, which reduced the risk of macrophage damage and intracellular bacterial dissemination.401 A class of reactive metal boride NPs was synthesized by Meng et al. This NP has the capability to capture LPS or PGN. This function not only inhibits excessive inflammation that is caused by dead bacteria in vitro and in vivo but also stimulates wound healing effectively.427
Nanotechnology in pathogenic bacteria detection
Nanotechnologies for targeted pathogen detection are typically categorized into three groups: antibody or aptamer-based detectors, phage-based sensors, and olfactory system-based assays. Nanotechnology detectors based on aptamers facilitate improved specificity (Fig. 4b).428 Yang et al. designed a broad-spectrum bacterial detection system based on D-amino acid-capped AuNPs. This NP has the ability to specifically target bacteria and promptly differentiate between S. aureus and MRSA by means of color changes.429 A rapid, sputum-free TB detector has been developed and is in clinical research (NCT03271567). This detector uses antibody labeling and energy-focused porous disk-shaped silica NPs (nanodisks) and high-throughput mass spectrometry to enhance sensitivity and specificity. Similar robust sensitivities were obtained in both culture-positive pulmonary tuberculosis (PTB; 91.3%) and extrapulmonary tuberculosis (EPTB; 92.3%) cases. Sensitivity in HIV-positive patients was also superior to that of routinely reported detectors.430 Modified NPs are typically detected through the recognition of bacterial ligands with the aid of optical imaging, electrochemical sensing or spectroscopy. An activatable theranostic nanoprobe was reported to the treat of MRSA infections.431 This specifically developed nanoprobe facilitates quick NIRF imaging and prolonged tracking of MRSA infection progression. The approach of recognizing antibodies specific to the bacterial surface has advantages in terms of both specificity and sensitivity. AuNPs sensitively and selectively detect and differentiate Salmonella typhimurium DT104 (103 CFU g−1 or more) from other genera and species on long leaf lettuce infected with a bacterial mixture of Escherichia coli and Salmonella typhimurium DT104 by changing the color of the NP solution from purple to gray.432 RBPs from the genus Inovirus were genetically modified for expression as phage scaffolds on M13, and the resulting chimeric phages were employed for the detection of the desired bacterial species.433 A sensor employing phage as a biorecognition element for the rapid and sensitive detection of excess Staphylococcus aureus. The procedure involves combining the sample with a Staphylococcus aureus phage S13-conjugated equipartitioned exciton scattering probe, followed by detection within 15–20 min, with a detection limit of 8 × 104 colony-forming units per milliliter.434 Nanotechnological detectors based on the olfactory system used for bacterial infections have been developed in recent years.435 The sensor platform utilizes NPs to reversibly bind and hinder lipase. When bacteria are present, these complexes are destroyed, which then restores enzyme activity and creates odor from odorless pre-scented substrate molecules. This system provides swift (15 min) sensing, and an exceedingly high sensitivity (102 CFU/mL) for bacterial detection—utilizing human olfaction as the output.435 Detection and diagnosis of complex and microscopic samples such as metabolites is heavily dependent on material design. An optimized SiO2@Ag nanoshell structure is already in clinical trials for the efficient detection of biomarkers and metabolites in patients with post-surgical brain infections.436
Other infectious diseases
In addition to viruses, bacteria and fungi, parasitic infections are an important component of infectious diseases, and their unique life histories make them widely transmissible, with an urgent need to develop new preventive diagnostic strategies to prevent their spread. In addition, transmissible spongiform encephalopathies (TSEs/prion diseases) are a group of infectious diseases that are difficult to treat. This section will discuss the application of nanotechnology to these two major groups of diseases.
NPs in parasitic infections
Owing to changes in the living environment of human beings and the rapid development of modern medicine, many parasites are on the verge of being eliminated by human beings. However, there are still parasites such as plasmodium and tapeworms that cause great harm to humans. Currently, some NPs, including liposomes, polymer NPs, SLNs, nanosuspensions, etc., have been investigated for the delivery of antiparasitic drugs. Nanomedicines combat parasitic infections through multiple pathways: inhibiting parasite entry into host cells and reproduction, directly killing parasites in the environment, prolonging drug action, and stimulating immune responses (Fig. 3d).
Najer presented a 3D nanoobjects very efficiently blocked invasion of RBCs by P. falciparum merozoites compared to soluble receptors.437 Nanovaccines for falciparum malaria are presently undergoing clinical studies. The R21 vaccine, which is based on the circumsporozoite protein and administered with adjuvant Matrix-M, has proven to be both safe and highly effective. Volunteers inoculated with the R29/Matrix-M vaccine exhibited only mild side-effects, along with a pronounced upsurge in malaria-specific anti-Asn-Ala-Asn-Pro antibodies in vivo.438 Malaria vaccines are currently in clinical trials, ChAd63-MVA ME-TRAP was used to prevent malaria infection in 22 healthy adults, which was effective in eliciting humoral immunity and T-cell responses.439 Another clinical trial also demonstrated that the vaccine prevented the progression of hepatic malaria.440 Biosynthesized CuO NPs were shown to exhibit efficient antiparasitic activity against cattle tick larvae.441 Myrrh AgNPs were first used to treat cutaneous leishmaniasis (CL) by Awad et al. Strong antiparasitic activity against Leishmania major was demonstrated in both in vivo and in vitro studies. Pfs230 TBVs are recombinant protein NPs, targeting parasite development.442 Currently, there are several Pfs230 clinical trials underway (NCT02942277, NCT03917654, NCT05135273), Pfs230D1-EPA has stronger immunogenicity than Pfs25-EPA(NCT02334462).443
The use of nanotechnology in the early stages of the development of counter parasitic diseases, but brings hope that this new field will provide a solution to the early stages of parasitic diseases, compensating for the lack of vaccines for most parasitic diseases and providing new therapeutic options for disease parasites showing increased resistance to current drugs.
NPs in TSEs/prion diseases
NPs possess exceptional features, comprising sensitivity, selectivity, and the capability to traverse the BBB when implemented in nano-sized particles. These NPs are extensively employed in imaging studies and treatments of neurological conditions.444
Thus, nanomedicine has been utilized for detecting, diagnosing and treating the Nguyen virus.445 NP-enabled biosensors have illustrated exceptional sensitivity, with apt detection of prion proteins, promoting speedy identification of prion diseases for improved treatment. Furthermore, gold nanorods have been utilized to detect amyloid fibrils in prion proteins effectively, utilizing chiral methods triggered by plasma NPs.446 Xiao, Jin et al. dual nucleic acid aptamer-enhanced magnetic particles and fluorescent QDs were applied for prion diagnostics, and were proven to be detectable in the pre-symptomatic stage.447 Sorokina et al. have reported the first instance of pH-dependent cationic pyridinylbenzene dendritic polymers being capable of breaking down amyloid aggregates, including the inclusion bodies of sheep prion protein, under physiological pH conditions.448
Nanotechnology in non-infectious inflammatory diseases
In addition to infections, dysregulated inflammatory responses and abnormal activation of the immune system can also lead to inflammatory diseases. These diseases encompass a group of chronic conditions, including autoimmune diseases like RA and multiple sclerosis (MS), allergic conditions such as asthma and contact dermatitis (CD), as well as other conditions like AS, psoriasis, and IS. Numerous natural and synthetic drugs have been developed to combat these inflammations, such as cyclosporine A for systemic lupus erythematosus (SLE),449 etoricoxib for irritant CD,450 and Rhein for ulcerative colitis (UC).451 However, their limited specificity often leads to systemic toxicity or instability in bodily fluids, restricting their application. Emerging evidence suggests that NPs have extensive prospects for treating these inflammatory conditions.9,452,453 Among the various mechanisms of NP-based anti-inflammation, the specific delivery of drugs to the specific site of inflammation is crucial to reduce systemic toxicity. NPs can also offer context-dependent selective release of drugs in response to local stimuli, such as pH, ROS, or enzymes. Some NPs also have direct anti-inflammatory effects of NPs via regulating inflammatory mediators and immune cells. Besides their therapeutic functions, NPs, owing to their unique physicochemical properties, can facilitate in vivo imaging of inflammation, enabling timely monitoring of disease progression.
NPs in the treatment of inflammatory diseases
NPs can aggregate at the lesion site through targeted mechanisms and enhance drug efficacy through multiple pathways. They primarily achieve inflammation treatment by controlling inflammatory factors and modulating immune cells. The second part of Table 2 summarizes clinical studies of nanotechnology for inflammatory diseases.
Targeted payload delivery
By delivering therapeutic cargo to sick tissues or certain organelles, targeted drug delivery methods may improve medical efficiency and lessen side effects.454,455 Nanotechnology can precisely deliver drugs to lesions to reduce systemic toxicity through passive targeting, receptor-ligand interactions, biomimetic cell membrane encapsulation, stimulus-responsive payload release, and inhalation delivery.
Receptor-ligand interactions
The nanocarriers can first passively accumulate in the targeted areas by modifying physical parameters such as charge (Fig. 5a), size, and surface modification.456 To effectively deliver the anti-inflammatory siRNA into the lungs for treating acute lung injury (ALI), DA-grafted hyaluronic acid (HA) was coated on an anti-tumor necrosis factor (anti-TNF)-α virus, which improved mucus entry of siRNA due to the electrical screen function of HA-DA and the bioadhesive properties of the grafting DA.457 In addition, negatively charged NPs were used to across the intestine epithelial barrier by causing tight junction ease and improving gut penetration, thereby facilitating oral protein administration. Specifically, the diameter and charge of the NPs affect this permeation-boosting impact, with smaller (≤200 nm) and more anionic particles (like silica) offering greater permeability.458 However, the hydrogen bonds and electrostatic forces that underlie ligand-receptor engagement only span a distance of around 0.3 to 0.5 nm,459 rendering it difficult to exert a long-distance magnetic draw on a target site. Nevertheless, active targeting has the potential to improve drug delivery to inflammatory sites.456 For example, ligand-targeting delivery can raise the preservation of drugs on target cells or tissues by conjugating cell-specific ligands to the surface of nano drug transporters.460
Fig. 5.
Overview of the mechanisms of NP treatment for inflammatory diseases. a An oral PDT that treats IBD by combining tannic acid and poly-β-cyclodextrin carrying DSP through host-guest interplay. PDT is electronegative as a result of its three parts, allowing it to electrostatically draw and target the electropositive, inflammatory colon mucosa.803 b CPHGs for regulated insulin release in the therapy of diabetes. A glucose-reactive FPBA-derived linker is used to join PVA and chitosan NPs.509 c Tris-BNPs are able to permeate solely psoriatic skin and not normal skin because of the extreme penetrability of psoriasis. Tris-BNPs gradually fade away as they spread and enter the epidermis, revealing the aldehyde chains of the BNPs, which could stick to amine chains lesional skin, enabling long-term local holding of BNPs in psoriasis skin lesions.524 d A Janus nanoplatform (Janus-CPS) for the concurrent early detection and combinatorial treatment of RA, which is made up of CeO2-Pt nanozyme on one part and PMO on the other. MCL, which has anti-osteoclastogenesis properties, is packed into the PMO’s mesopores to work in concert with nanozymes’ soothing properties to effectively manage RA. The NIR-II fluorescence imaging is employed by ICG-carried Janus-CPS to achieve the desired efficacy in the early detection of RA.162 e NPs penetrate the BBB through TfR1-mediated transcytosis to access the CNS, thereby treating diseases such as IS, MS, and Alzheimer’s disease. f SS-31 polypeptide and astaxanthin are combined in PMeTPP-MBT (a unique PA contrast agent)-loaded, smart reactive theranostic nanoplatform (PA/ASePSD). Targeting atherosclerotic lesions is enabled by PA/ASePSD’s high affinity for VCAM-1 and CD44 on impaired endothelium. Astaxanthin, SS-31 peptide, and PMeTPP-MBT are released in regulated conditions when ROS levels in acid are increased. This enables non-intrusive PA diagnostics and plaque reduction via soothing effects and lipid metabolic control.804 PCD poly-β-cyclodextrin, PDT polyphenol nanoparticle, PVA poly(vinyl alcohol), FPBA formylphenylboronic acid, CPHG chitosan NPs/PVA hybrid hydrogel, ICG indocyanine green, MCL micheliolide, ox-Dex oxidized dextran, PA photoacoustic, VCAM-1 vascular adhesion molecule-1, LOX-1 lectin-like oxidized low-density lipoprotein receptor-1, ABCA1/G1 ATP-binding cassette transporter A1/G1
The folate receptor β (FRβ)-directed administration for the therapy of inflammation relies on the increased expression of FR in hematopoietic cells of the myeloid lineage upon immunological arousal and the subnanomolar avidity between FR and folic acid (FA).461,462 Methotrexate (MTX), a derivative of FA, was carefully chosen as a disease-modifying antirheumatic medication (DMARD) due to its specific affinity to FRs.463 Moreover, to enhance its anti-inflammatory efficacy, the modification of 1,2-distearoyl-sn-glycero-3-phosphoethanolamine-N-[folate(polyethylene glycol)-2000] (FPD) was added to MV/MTX@ZIF-8 NPs because activated macrophages significantly overexpress FR.464 Research findings indicate that activated macrophages with overexpression of FR exhibit substantially higher absorption capacity for FPD/MV/MTX@ZIF-8 compared to inactive macrophages.463,464 HA encourages the adhesion of NPs via receptor targeting or integrin interaction.465 Following transcutaneous injection, a HA-NP generated by the self-package of HA-hydrophobic moiety conjugates could gather and focus on pro-inflammatory macrophages in the irritated dermis, reducing epidermal hyperplasia and inflaming reactions in rodent models of imiquimod and IL-23 induced skin inflammation.466 Scavenger receptors (SRs) are potential targets for inflammation along with oxidative damage due to their high affinity for glycoxidation end products, protein oxidation-derived compounds, and peroxidative lipid metabolites.467 The palmitic acid-modified bovine serum albumin has been created to possess outstanding SR-A targeting capacity, which would send anti-inflammatory drugs CLT to trigger macrophages in inflamed sites.468 Correspondingly, an amphiphilic polysaccharide was created by combining 5-cholanic acid to a dextran sulfate (DS) framework, which was preferentially taken up SR class A-regulated endocytosis. These DS NPs enable the targeted administration of MTX to stimulated macrophages, which are in charge of irritation and joint degeneration.469
Integrin-based NPs with issue specificity have great potential for efficacy in inflammation since integrins facilitate leukocyte aggregation in areas of irritation.470 Antheraea pernyi silk fibroin possesses a lot of arginine-glycine-aspartate (RGD) tripeptides, which could selectively attach to integrin receptors on the surface of colonic epithelial cells and macrophages in the irritated colon.471–473 The impaired colon epithelial lining was repaired by the Res-ApNP therapy, which polarized macrophages to the M2 state to minimize inflammation and the production of cytoplasmic ROS.474 In addition, following PEG fragmentation by collagenase IV, celastrol-loaded enzyme-responsive NPs (PRNPs) simultaneously target osteoclasts (OCs) and inflammatory macrophages generated from RA patients through contact with the RGD-v3 integrin. These PRNPs, which are made up of RGD transformed NPs (RNPs) coated with cleavable PEG chains, deliver CLTs to specifically cause apoptosis of OCs and macrophages in arthritic sites.475 Similarly, the secreted protein acidic and rich in cysteine (SPARC) and its ligand albumin is another receptor-ligand interaction for NP targeting. Based on the fact that SPARC is overproduced in the joint lubricating fluid of RA patients as well as animals with collagen-induced arthritis (CIA), MTX-loaded human serum albumin (MTX@HSA) NPs have been developed as biomimetic drug transport platforms for RA treatment. Combined fluorescence and MRI demonstrated larger collections and longer preservation of MTX@HSA NPs in inflammatory joints after intravenous infusion of chlorin e6-labeled MTX@HSA NPs into CIA rodents.476
There is a growing fascination with edible plant-derived exosome-like NPs, which are nanoscale particles created from edible plants (like ginger, grape, carrot, and broccoli) and have been shown to reach cells in the body via specific ligands.477,478 In a dextran sodium sulfate (DSS)-induced colitis model, oral grape exosome-mimetic NPs targeted intestinal epithelial cells (IECs) and macrophages, enhancing IEC viability, reducing TNF-α, IL-6, and IL-1 levels, and increasing IL-10 and IL-22 concentrations.477 Oral delivery of exosome-like NPs (PELNs) generated from portulaca oleracea L. (purslane, POL) specifically localized inflamed regions in a DSS-induced murine UC model.479 NPs generated from culinary plants offer an advantage of tackling restrictions associated with artificial NPs like possible toxicities and restricted industrial scale.477
Plenty of discoveries shed light on the utilization of NPs as a versatile tool for conducting thorough receptor/ligand interaction investigations and designing nanosize transport and treatment platforms.480 Nevertheless, no particular method will work in each case, and the selection of which ligand to use is entirely dependent on the application.
Cellular membrane-coated nanocarriers
Because of the inherent active-targeting capacity and superior biocompatibility, cells and associated extracellular vesicles are lately extensively used to build biomimetic techniques for the therapy of many diseases.481–483 Biomimetic camouflages made of cell membrane-coated NPs might lose their source cells’ operational capabilities while keeping just their surface characteristics.483 For the purpose of focusing on inflammation, biomimetic NPs could mimic several of the immune network’s normal inflaming reaction pathways.484 Immune cell membrane molecules may provide NPs with an array of benefits, such as longer systemic circulation, exceptional antigen recognition skills for improved targeting, stronger cell interplay, progressive drug discharge, and decreased in vivo cytotoxicity.485
The application of macrophage membrane-coated NPs offers a promising avenue for targeted therapy against inflammation. Macrophages have a key role in multiple aspects of AS’s degenerative process. Biomimetic NPs (MM/RAPNPs), which used macrophage membrane (MM), were designed by coating the exterior of rapamycin (RAP)-loaded PLGA copolymer NPs with MM, in order to mimic macrophage homing into arterial wall lesions. In vivo, the MM-coated NPs efficiently targeted and accumulated in atherosclerotic plaques.486 Similarly, biomimetic NPs (MM@Lips-SHP1i) are produced from MM covered SHP1i-carried liposome NPs. MM@Lips-SHP1i NPs would battle macrophages in the plaque site in vivo to interact with oxidized low-density lipoprotein and LPS, decrease the intake of fresh lipids by macrophages, and decrease the production of foam cells.487
Researchers also created biomimetic nanocarriers for targeted inflammation therapy inspired by natural PLTs. Because of its abrupt onset and narrow treatment window, acute IS presents a substantial obstacle for timely detection and intervention. Li et al. created a biomimetic NP based on PLT membrane envelopes stuffed with l-arginine and γ-Fe2O3 magnetic NPs (PAMNs) for thrombus-oriented administration of l-arginine and in situ generation of nitric oxide (NO), which was motivated by PLTs’ function in focusing on binding to the impaired blood vessel over thrombus development. Results show that with the supervision of an ambient magnetic field, the designed 200 nm PAMNs rapidly pinpoint IS-related sites and retain the inherent characteristics of the PLT membrane.488
The application of neutrophil membrane-coated NPs demonstrates the potential for targeted drug delivery in inflammation treatments. For precise medication administration during the therapy of RA, Yang et al. construct a biomimetic F127 polymer coiled around an altered neutrophil membrane ApoA-I mimetic peptide. The neutrophil membrane wrapping may offer the NP synovitis-orienting abilities, leading to migration to the articular fluid under the chemotactic actions of IL-8, owing to the abundance of molecular adhesives and chemotaxis receptors on neutrophils.489 As potential methods for gene silencing, ASOs have been used to treat human illness. For the specific administration of ASOs against microRNA-155 to the endothelium in atherosclerotic regions, Liu et al. developed a neutrophil membrane-covered zeolitic imidazolate framework-8 nanocarrier system. By engaging with the surface proteins of endothelium intercellular adhesion molecule-1 (ICAM-1) and neutrophil surface protein CD18, the neutrophil membrane can enhance plaque endothelial cell focusing.490 Inflammation-targeting biomimetic NPs are thought to have a promising future in developing nanomedicine.484
Stimuli-responsive drug delivery systems
The ability of stimulus-responsive materials to modulate drug transport in reaction to particular stimuli, including pH, enzyme concentrations, or redox gradients, has sparked a great deal of attention in recent years.491 Notably, stimuli-responsive NPs hold enormous potential for delivering adjuvants and antigens to specific immune cells, avoiding antigen deterioration and elimination, and boosting the intake of particular APCs, thus maintaining adaptive immune reactions and enhancing treatment for inflammation.492
According to investigations, elevated rates of metabolism in inflamed regions cause a steady rise in acidic metabolites and excessive oxidative strain, which are defined by low pH, ROS, and hypoxia.493 Healthy tissue has a pH of about 7.4, whereas inflamed tissue has a pH of about 6.4.494 In addition, irritated tissues have substantially higher ROS concentrations than healthy tissues. Scientists prefer pH and redox-responsive NPs because inflamed tissues have a special pH and redox milieu.495
When ROS levels in inflammatory sites rise, NPs with ROS-reactive devices unleash medicinal payloads at the appropriate time and location.496 Anti-rheumatic medication delivery over an extended period of time frequently results in noticeable side effects and patients’ failure to comply. Ni et al. created Dex-carried ROS-reactive NPs (Dex/Oxi-CD NPs) and FA altered Dex/Oxi-CD NPs (Dex/FA-Oxi-CD NPs) and confirmed their soothing activity in vitro and in vivo to selectively transport Dex to swollen joints.497 ALI is an inflammation condition linked to cytokine storm, which stimulates a number of ROS cascades and adversely affects patients. Utilizing an improved emulsion technique, Muhammad et al. created Dex-loaded ROS-responsive polymer NPs (PFTU@DEX NPs), which had an elevated packing content of DEX (11.61%). In ROS surroundings, DEX was liberated from the PFTU@DEX NPs more rapidly, which could effectively detoxify excess ROS in vivo.498 ROS-reactive drug administration platforms have remarkable performance in biomedical fields as their payload is exclusively delivered at inflamed areas marked by high ROS levels.499,500
pH-responsive NPs usually modify their drug discharge depending on the acidic milieu of inflammatory tissues. A precisely regulated one-step biomimetic mineralization technique was used to encapsulate insulin, GOx, and catalase (CAT) into ZIF-8 NPs, in which a powerful enzyme cascade complex (GOx/CAT group) acted as a tailored glucose-reactive component that can quickly catalyze glucose and produce gluconic acid to decrease the regional pH and efficiently devour the toxic derivative hydrogen peroxide, triggering the breakdown of pH-reactive ZIF-8 NPs for insulin discharge.501 In UC, NP-based medication transport platforms build up in the damaged epithelium of inflammatory colon mucosa. To achieve total drug protection in a gastric-like pH and precise transport of NPs to the colon, persistent medication-releasing PLGA NPs were produced and subsequently enveloped in pH-responsive Eudragit FS30D MPs.502 Because ischemia tissues exhibit a lower pH milieu than healthy ones, an acidity-responsive theranostic RAP-carried NP platform for ischemic brain tissues was developed. Accompanied by its swift dissolution in acidic conditions, treatment precision is enhanced.503 NPs with stimulation reactions can enhance drug concentration in infections, and are particularly attractive in inflammation precise treatment.495
Typically, patients undergo fingertip prick plasma glucose monitoring and insulin injections to keep up normoglycemia.504 Conventional techniques, on the other hand, not only lead to physical pain and trouble to patients, but also provide an elevated risk of complications because of erroneous insulin administration.504 As a result, the creation of a smart insulin administration device that adapts to ambient levels of glucose is critical.505 Amongst these, a frequently employed technique for designing glucose-reactive materials takes benefit of PBA and its derivatives’ capacity to interact transitorily with cis-diol groups.506,507 The glucose-sensitive insulin-liberating platform, AuNC-PBA-Ins assembly, was built with insulin transplanted on the exterior and enhanced the potency of insulin discharge in reaction to glucose level.508 Furthermore, stimuli-sensitive hydrogels (HGs) with regulated drug discharge profiles are an ongoing issue for enhanced medicinal applications. Ali et al. show that the elastic solid-like features of CS NP/poly(vinyl alcohol) hybrid hydrogels (CPHGs) are drastically diminished in low-pH and high-glucose settings. Under physiological settings, an in vitro drug release experiment revealed size-relied glucose-reactive drug delivery from the CPHGs (Fig. 5b).509
In terms of the roles of distinct tissues or organs, seeking distinct elements inside their milieu that impact the activity of NPs is also extremely novel. Colon-oriented oral medication delivery devices are attractive for the therapy of UC. In reaction to regional elevated cellulase action, two-layer Budesonide-carried SLNs with nanosized particle diameter and negative zeta activity demonstrated the desired selective drug release in the colon.510 Also, the in vitro unleashing studies demonstrated that CSO/Dex/LNPs were esterase-sensitive and would swiftly discharge the payload in synthetic intestinal fluid with esterase.511 The investigations emphasize novel drug transport techniques that provide precise regulation over drug discharge in reaction to niche conditions.
Endogenous cues concentrated in the illness microregion, including pH change, redox gradient, and particular enzymes, facilitate autonomous drug administration via activating the sensitive elements in the nano-assembly. Because of their excellent specificity, the delivered medicine in a form of endogenous stimuli-sensitive nanocomplex might accumulate at the intended areas,512 resulting in fewer adverse reactions and an increased therapeutic index.513
Inhalation delivery
Despite persistent attempts to improve chronic asthma therapy, symptomatic medications remain the sole choice for managing this common and painful condition. Inhalation is the most basic and effective means of administering drugs to the lungs.
The lung serves as a “center” for autoimmunity, via which autoreactive T cells travel before reaching illness locations. According to Saito et al., focusing on lung APCs with antigen-carried poly(lactide-co-glycolide) (PLG) particles alters lung CD4+ T cells, allowing mice to tolerate experimental autoimmune EAE. Intratracheally administered particles were connected with lung APCs and reduced costimulatory component synthesis on the APCs, inhibiting CD4+ T cell growth and decreasing their number in the CNS while boosting it in the lung.514 Small interfering RNA (siRNA), which possesses intrinsic and selective mRNA cleaving ability, has been hailed as a potential therapy for lowering the exacerbation rate of asthma by preventing airway epithelial cells (AECs) from expressing and releasing inflammation-promoting cytokines. In order to locate the intercellular ICAM-1 receptors on the apical portion of AECs, Zhang et al. developed new inhaled LNPs. With the help of this transport platform, siRNA will be delivered to AECs more effectively, reducing the production of inflammation-promoting cytokines and the accompanying symptoms.515
As an essential strategy for managing illnesses like asthma or pulmonary irritation, inhalable NPs provide a non-intrusive and effective means to administer soothing drugs precisely to the respiratory system.
Enhanced therapeutic agents
The present ways of treating inflammation mostly concentrate on pain alleviation and inflammation control. For example, nonsteroidal anti-inflammatory medicines, DMARDs, glucocorticoids, and biological substances are the primary drugs employed to manage RA.516,517 In order to attain optimal accumulations at regions of inflammatory and pathogenic states, oral or systemic delivery frequently requires larger dose levels and/or frequency, which exacerbates the unfavorable effects.518,519 Therefore, designing nanocarriers that can stably and slowly release drugs, enhance drug retention in the body, and even traverse biological barriers such as the BBB is of paramount importance.
Improved drug stability
Many drugs exhibit excellent efficacy against inflammation in vitro; however, their clinical application is limited due to their instability in the human body’s environment. NPs not only serve as carriers for these drugs, delivering them to the site of inflammation, but also protect their activity within the human body.
As a result of the poor pharmacokinetics, glucocorticoids have considerable adverse reactions, which make them tough to utilize in the therapy of RA. Nanomaterials have the potential for dispensing glucocorticoids, but the packaging of extremely crystalline and insufficiently water-soluble medicines leads to inadequate drug encapsulation and limited stability. The construction of 130 nm NPs formed entirely of dextrose palmitate and supported by phospholipids connected to PEG exhibit a negative zeta potential (55 mV), good capture efficacy, and durability for 21 days when stored at 4 °C. In a mouse collagen-triggered arthritis model, the increased therapy efficiency of the NPs was also revealed.520 In order to prevent broad exposure and immune suppression, the optimum anti-TNF treatment for IBD ought to transport the antibody precisely to the areas of intestinal irritation. The oral administration of antibodies is extremely difficult due to a number of obstacles, including limited membrane penetration and digestive enzymes in the GI tract.521 By assembling hydrogen bonding supramolecular NPs with TA and 1,2-distearoyl-sn-glycero-3-phosphoethanolamine-N-[methoxy(polyethylene glycol)-2000], Wang et al. develop a method for delivering antibody orally. Infliximab could be targeted to the region of intestine inflammation and shielded by NPs in the GI system without decomposing.522
NPs, when employed as drug carriers, exert a profound stabilizing effect by safeguarding the drugs’ chemical integrity within the complex milieu of the human body. This protective function ensures the maintenance of drug activity, overcoming the limitations posed by their inherent instability in vivo, thus enhancing their therapeutic potential for clinical applications in inflammation management.
Sustained drug release and improved internalization
Nanotechnology ensures precise drug delivery to inflamed regions for optimal therapeutic efficacy by achieving sustained drug release and improving cellular internalization.
Considering the physically enclosed payloads with drug explosive release and/or drug embedding, a novel prodrug technique is required to improve the construction of customizable nanotherapeutics. Zeng et al. developed a dendrimer-based drug delivery system with a benzoboroxole payload that is pH and glucose responsive. The dynamic interaction of benzoboroxole and glucose could efficiently modulate micelle disintegration and insulin secretion pace, hence stabilizing plasma glucose swings.523 Mai et al. synthesized tris(hydroxymethyl)aminomethane-modified bioadhesive NPs (Tris-BNPs). Tris molecules gradually disperse away as Tris-BNPs spread and permeate into the epidermis, disclosing the aldehyde groups of BNPs, which may attach to amine groups existing inside affected skin, resulting in lengthy regional preservation of BNPs in psoriatic cutaneous abnormalities (Fig. 5c).524 Uveitis frequently needs corticosteroid medication to avoid inflammation-associated ocular consequences. Nevertheless, utilizing traditional embedding technologies, it is challenging to pack extremely water-soluble medicines, including Dex sodium phosphate (DSP), and produce prolonged drug discharge. When carboxyl-terminated PLGA was employed as an electrical “bridge” between the PLGA and DSP, it was possible to encapsulate DSP into biodegradable NPs with a rather substantial drug load (6% w/w). DSP-Zn-NP subconjunctival infusion yielded measurable DSP concentrations in both the anterior and vitreous chambers of the eye for a minimum of 3 weeks.525
In addition to achieving sustained release, NPs also facilitate the internalization of drugs by cells. In comparison to the unbound drugs, retinoic acid against MS in 70 nm consistent-sized lipid nanocapsules, coupled with superparamagnetic iron oxide NPs, and altered with transferrin-receptor targeting peptide demonstrated a 3-fold improvement in uptake by endothelium.526 To treat the effects of UC, a CS-integrated poly(lactic-co-glycolic acid) NP platform was designed. The bioadhesive nanotechnology increased medication durability in the GI tract by increasing the efficacy of intestinal epithelium absorption.527 Polyamidoamine dendrimer is investigated as a nucleic acid transporter and has anti-inflammatory abilities. Histidine and arginine were concatenated to the main amines of polyamidoamine generation 2 (PG2) (yielding PG2HR) to increase DNA transport effectiveness. PG2HR transported pDNA into cells primarily via clathrin-independent endocytosis and partially via macropinocytosis. The gene transport effectiveness of PG2HR proved greater than that of PG2 or PG2 coupled with simply arginine (PG2R), which could be attributed to the pDNA/PG2HR complex’s better cell absorption and endosomal escape.528
NPs can exhibit good tissue penetration. Rachamalla et al. produced novel lithocholic acid analogs that would naturally generate LNPs in the aquatic environment, infiltrate the skin, enter the deeper dermal layers, and exhibit soothing actions against psoriasis-like persistent skin irritations.529 Similarly, deep eutectic solvents (DES) were shown to improve the skin permeability of stiff NPs. Li et al. described a practical and straightforward transdermal administration approach for topical RA therapy employing mesoporous silica NPs embedded in DES hydrogels. The hydrogel solvent pushed the stiff NPs over the cutaneous barrier in a non-invasive way following administration to the skin, causing prolonged infiltration and collection of MSNs at SC inflammatory areas.530 The Fc/FcRn transportation route was designed as an approach for increasing medication delivery across the epithelium.531 The Fc on the exterior of the NPs-fedratinib engaged with the FcRn displayed on the epithelium in the lungs, circumventing the lysosome and supporting NPs passing the epithelial barrier via transcytosis and aggregating at the lung parenchyma with an inflamed milieu.532
Sustained release and enhanced internalization of NPs for anti-inflammatory drugs represent a promising avenue in inflammation treatment. These NPs offer controlled drug delivery, ensuring a prolonged therapeutic effect, while their enhanced internalization into target cells enhances drug efficacy.
Extended drug retention capabilities
NPs with longer drug preservation properties avoid elimination mechanisms while maintaining medical effectiveness. Attempts to prolong NP retention time in vivo sparked numerous tactics in particle interface remodeling to avoid macrophage absorption and systemic elimination.533
The enhanced lymph angiogenesis and lymphatic circulation in arthritic locations have hastened the removal of NPs in inflammatory joints. Dex-KLVFF-PSA (DKPNPs) could maintain a steady nanoscale shape in normal conditions, and the PSA shell might equip DKPNPs with extended circulation and effective localization to arthritis areas. In inflammatory joints, acidic pH-induced Dex dispersion or macrophage-induced specialized interaction with PSA could cause DKPNPs to reassemble from NPs to nanofibers, resulting in decreased lymph removal and extended potency.534 At present, sinomenine hydrochloride (SIN) formulations, a natural DMARD, are utilized to treat RA; nevertheless, SIN’s potency is severely hampered by its brief half-life, poor bioavailability, and dose-relied side effects. In vivo images revealed that the superior immune-escape characteristics of the biomimetic NPs based on Prussian blue NPs led to significantly higher circulatory half-life and drug concentrations at arthritic regions of arthritis rodents.535 Glycyrrhizic acid (GA) has powerful soothing properties and suppresses COX-2 via inhibiting 11-hydroxysteroid dehydrogenase type II.536 GA has poor oral bioavailability in humans and rodents, since it was observed that GA was not traceable in human circulation after 100–800 mg of GA was consumed orally. Aminocellulose-grafted-polycaprolactone covered gelatin NPs are used as a transporter to achieve higher treatment efficiency than free medicines in the therapy of RA by co-delivering of GA and budesonide.537
The development of an orally delivered medication delivery platform capable of extended preservation in the GI tract is a critical problem for the successful management of GI illnesses like IBD. Zhao et al. describe a bioadhesive fluid coacervate synthesizing via hydrogen bonds. The liquid NPs-based coacervate exhibits essential pH- and salt-independent structural steadiness and creates a physically adhesive covering on an extensive area of the intestines with a raised retention time of over 2 days.538 6-shogaol, a soothing medication applicant, showed remarkable efficacy in a number of in vitro tests. Nevertheless, due to its quick degradation upon oral treatment, it has low bioavailability and imperceptible in vivo pharmacokinetics. Yang et al. developed a natural-lipid (NL) NP drug administration platform to enclose 6-shogaol and transport it to the desired therapeutic target (colon). When contrasted with natural 6-shogaol, the in vitro drug-release experiment demonstrated that NL-embedded 6-shogaol (6-S-NL) has a prolonged drug-release pattern.539
Gouty arthritis (GA) is an intractable metabolic illness that needs ongoing therapy with regular drug delivery multiple times each day. IL-1 inhibiting medicines, like IL-1 receptor antagonist (IL-1Ra), offer high treatment promise in clinical research of GA in contrast to non-specific small organic drugs. Yet, IL-1Ra’s utilization is severely constrained because of its brief half-life and restricted bioaccessibility. Zhang et al. use the noncovalent construction of a designed IL-1Ra chimeric protein to show a novel kind of nanomedicine preparation. Surprisingly, the NP complex has a biological availability 7 times greater compared to that of pure IL-1Ra a remarkably lengthy half-life of 27 h, prolonging the dosage period from a few hours to over 3 days. The construction of protein-tethered nanoplatforms presents novel possibilities for studying long-lasting and safer medicines for GA therapy.540
NPs excel in drug delivery by prolonging drug retention in the body, leading to enhanced therapy and reduced toxicity. This attribute not only improves treatment outcomes but also lowers the risk of adverse effects.
Synergy effect
Inflamed disorders such as AS, RA, and psoriasis are long-term disorders caused by a variety of parameters. When seeking to control a single pathogenic variable, standard therapies frequently fail to provide beneficial clinical outcomes.541 NP injection is one of the most appealing techniques for harnessing the synergy of many co-packaged compounds for an identical target (Fig. 5d).542
The prevalence of UC is related to many factors.543 Macrophages are acknowledged as significant target cells in UC therapy.544 Meanwhile, macrophage mitochondria, which are crucial biological components, were regarded as critical facilitators in the therapy of UC.545 A novel class of sequence-targeted astaxanthin NPs for the therapy of UC has been constructed and it was exciting to observe how effectively the created sequence-targeted astaxanthin NPs were able to hit macrophages and mitochondria.546 During the course of UC medication, pro-resolving elements (such as anti-TNF antibodies), which are vital to colonic epithelium restoration were downregulated. Elevated levels of substances like IL-22 throughout the medicinal suppression of TNF-α were believed to aid in the healing process of intestinal irritation. Gal-siTNF-NP/IL-22-encapsulated hydrogel may be used to orient the inflammatory colon and simultaneously deliver siTNF and IL-22 to enhance the outcomes of either treatment.547
Psoriasis is defined by several inflammatory pathways that communicate with each other that are specifically linked to aberrant interaction between immune cells and keratinocytes. In order to implement integrated multiple targeted treatments for psoriasis, an NP-based hydrogel was developed by incorporating MTX into ZnO/Ag. ZnO-loaded mesoporous spheres were employed as antioxidant NPs and drug transporters in this hybrid hydrogel. AgNP-anchored ZnO NPs (ZnO/Ag) in an appropriate quantity were modified with an innate immunological regulation capability outcomes of either treatment outcomes of either treatment.548 Furthermore, for a coupled chemo-photodynamic treatment for psoriasis, Wang et al. created CS/hyaluronan nanogels to co-carry MTX and 5-aminoleavulinic acid (ALA), or MTX-ALA NGs. NGs increased cellular intake (p < 0.001), protoporphyrin IX transformation (p < 0.001), and ROS formation (3.93-fold), which together had a synergistic inhibitory effect on cell growth and induced apoptosis in HaCaT cells exposed to LPS with a 78.6% apoptosis ratio.549
In order to operate as an anti-atherosclerotic drug and a nanoscale delivery system with self-transportation capabilities, Wang et al. created a hydrophilic-lipophilic complex of low molecular weight heparin and unsaturated fatty acid (LMWH-uFA). Practically, the hydrophilic portion, LMWH, inhibited premature vascular irritation by preventing monocyte adhesion, and the hydrophobic section, uFA, might control plasma lipid levels. RAP, a soothing medication, was enclosed in the micellar core, improving its dissolution in water, and worked in tandem with LMWH to interrupt the vascular irritation pathway at P-selectin. RAP-carried NPs dramatically decreased the plaque size and demonstrated excellent therapeutic benefits in an AS murine model.541
By utilizing their potential to combine several therapeutic properties, NPs demonstrate great capacity in dealing with inflammation. The comprehensive strategy has the possibility to improve the therapy for long-term inflammatory diseases.
Crossing the BBB
It is still challenging to convert CNS-focused treatments into better clinical results. The BBB, which is undoubtedly the most strictly guarded barrier in the human body and consistently blocks the majority of therapies, is primarily to blame for this. New approaches are becoming possible by developments in the engineering of nanostructures and their use in biomedicine, which may help us better comprehend and manage neurologic illnesses (Fig. 5e).550
The most efficient way to transfer NPs to the brain while navigating the BBB is by receptor-regulated shipping, which works at the receptor-ligand interaction zone.551,552 A transferrin-receptor interacting peptide was added to a tiny lipid nanocarrier harboring RA and superparamagnetic iron oxide NPs. The alteration enhanced its capacity to engage with cerebral cells, leading to a five-fold improvement in its efficiency to the BBB and a three-fold improvement in endothelial cell intake.526 The transferrin receptor 1 (TfR1) on the membrane of cerebral endothelium could be recognized and bound by heavy chain ferritin (HFn) NPs. As a result, HFn NPs would effectively permeate the BBB via TfR1 without any extra alterations.553
Taking a ride with immune cells is an ingenious approach to assist drugs pass the BBB as several immune cells voluntarily travel to the brain when inflammation arises. Blood-borne Th17 cells migrate through the BBB and settle in the MS milieu. This further results in a massive influx of Th17 cells into the brain and an escalation of localized inflammation. Therefore, employing a “Trojan horse” strategy, Th17 cells could be used as powerful cell carriers for cascade-based medicinal drug trans BBB.500 In cerebral ischemia, neutrophils travel from the bloodstream and cross the BBB and gather in vast numbers at the region of irritation. The peptide cinnamyl-F-(D)L-F-(D)L-F, which may selectively attach to the formyl peptide receptor (FPR), is employed to modify a nanocomplex interface as the membrane of neutrophils includes FPR. Following intravenous administration, the synthetic NPs hitched a ride with neutrophils to increase aggregation at the inflamed location of brain ischemia, which was accomplished by successfully adhering to the membrane of neutrophils in peripheral circulation via FPR.554
Furthermore, some NPs, due to their extremely small size, can more readily traverse the BBB. Selenium quantum dots (SeQDs) are incredibly tiny and have a rapid BBB penetration rate. According to the research results, SeQDs exhibit broad-spectrum antioxidant properties, high radical quenching capability, and protection of cells from oxidative load. According to in vivo studies, which show that SeQDs can constantly aggregate in the brain following speedy crossing of the BBB, SeQDs can swiftly relieve AD, dramatically enhance the memory damage of AD rodents, and enhance their cognitive and memory capacity.555
Nanostructures make it easier to create drug transporters that may safely cross the BBB in situations involving inflammation within the CNS, opening up new treatment options for illnesses that were previously difficult to manage.
Inflammatory modulation effect of NPs
The mechanisms by which nanotechnology alleviates inflammation primarily involve two aspects: the clearance of inflammatory markers such as nucleic acids, ROS, and cytokines; and the regulation of the immune response through modulation of immune cells.
Modulation of inflammatory factors
In the realm of inflammation, nanotechnology offers a multifaceted approach to modulate inflammatory factors, including nucleic acids, ROS, and cytokines, effectively controlling the inflammatory response at various levels.
The excess of free DNA molecules in lesions is considered to be a pathogenic factor in many diseases, the mechanism of which may be that cell-free DNA further triggers chronic inflammation. Cutaneous cationic polymeric NPs were proven to be an efficient treatment approach for addressing psoriasis via capturing cell-free DNA (cfDNA). Yet, electropositive cfDNA scavengers pose a serious threat to organs once they cross the skin barrier and enter the bloodstream. A variety of poly(2-(dimethylamino)ethyl methacrylate) (PDMA) grafted hairy silica particles (cSPs) with variable PDMA length and dimension are employed to eliminate cfDNA in the dermis for improved transition to clinical deployment.556 When cfDNA from injured or destroyed cells interacts with LL37, it becomes an immune reaction enhancer that aggravates psoriasis. According to Liang et al., cationic NPs effectively battle for DNA from the DNA-LL37 immunocomplex and prevent DNA-LL37-triggered cell stimulation.557
Hypoxia and oxidative strain are two critical metabolic elements in the emergence of numerous inflammations. Various research has demonstrated that oxidative load is a vital factor in AS development.558 Gao et al. showed that gadolinium doping of CeO2 (Gd/CeO2) nanozymes produced outstanding results for anti-AS as efficient ROS eliminators. It was discovered that the chemical modification of Gd increased the exterior fraction of Ce3+ in the nanozymes, improving their general capacity to scavenge ROS.559 Yuan et al. described versatile NP-based therapy for IS generated from a bioactive oligosaccharide substance (TPCD) created by covalently combining a radical-eliminating substance (Tempol) and a hydrogen-peroxide-clearing component of cyclodextrin-modified phenylboronic acid pinacol ester.10 More significantly, a redox process transforming ROS into O2 might simultaneously reduce oxidative burden and hypoxia, regulating the inflamed milieu.560
Elevated inflammation-promoting cytokines play a key role in the development of numerous inflammations. In RA, there is often an overproduction of inflammatory cytokines including IL-1, IL-6, and TNF-α, which would be elevated by the mitogen-activated protein kinase (MAPK)/extracellular signal-regulated kinase (ERK), NF-κB, and activator protein-1 (AP-1) signaling cascades. It was found that CQDs obtained from herbal medicines greatly reduced swelling signs and impeded the production of associated inflammation cytokines (IL-1, IL-6, and TNF-α), demonstrating exceptional anti-inflammation efficacy.561 Liu et al. suggested HIF-1 siRNA-carried calcium phosphate NPs enclosed in high-density lipoprotein-modified with apolipoprotein E3 (HIF-CaP-rHDL) as a treatment for RA in order to suppress the levels of HIF-1, NF-κB, and MAPK. By blocking these pro-inflammatory signaling pathways and attenuating cytokines including TNF-α, IL-1β, and IL-6, HIF-CaP-rHDL showed promise as a targeted remedy for RA.562 Persistent neurological inflammation caused by microglia is assumed to start in the early stages of AD and is essential to the etiology of the disease. Oxytocin (OT)-carried, angiopep-2-modified CS nanogels (AOC NGs) were developed for the prevention of AD through the suppression of intrinsic inflammatory reaction. The NPs efficiently suppress microglial engagement and lower pro-inflammatory cytokine (TNF-α and IL-1β) amounts by suppressing the ERK/p38 MAPK and COX-2/iNOS NF-κB signaling cascades.563 Nano-curcumin reduces inflammation and prevents oxidative stress in the treatment of diabetic depressed patients. Among the clinical trial, a significant fall was found in the mean score of anxiety in the nano-curcumin group (from 22.4 [4.03] to 20.6 [3.4]).564
Immune response modulation
In addition to regulating inflammatory factors, NPs can control inflammation by directly modulating the immune response. Specifically, NPs can regulate the polarization of macrophages (Fig. 5f), promote immune cell tolerance to self-antigens, target immune checkpoints, promote the conversion of Th17 cells to regulatory T cells (Tregs) cells, regulate pro-inflammatory monocytes, neutrophils, and DCs, as well as antagonize TLR in macrophages, thereby achieving a favorable therapeutic effect.
An initial event that triggers tissue deterioration and malfunction is inflammation brought on by autoimmune disorders and persistent damage. Pro-inflammatory monocytes move across the body and infiltrate irritated tissue. Monocytes play a vital part in the progression of MS, particularly because most magnified pro-inflammatory monocytes penetrate the BBB, causing neuron damage and recruiting more immune cells to penetrate the CNS. Monocytes exclusively and effectively absorbed curcumin-carried high-density lipoprotein-emulating peptide-phospholipid scaffolds (Cur-HPPS) via the SR class B type I receptor. By preventing pro-inflammatory monocytes from crossing the BBB in EAE mice, limiting microglia multiplication, and preventing the penetration of other effector immune cells, this treatment decreased EAE morbidity from 100% to 30%.565
Macrophages, which show exceptional variety and polarization, are crucial immune cells in innate immunity. In pathological situations, additional macrophages, besides the local ones, migrate to the sick regions and polarize into different phenotypes (mostly M1 and M2) in response to diverse variables in the microenvironment, thus performing varied activities and functions. Generally, macrophage polarization might be separated into two forms: classically engaged M1 and optionally induced M2.566 M1 macrophages primarily present antigens and eliminate harmful microbes and are inflammation-promoting, whereas M2 macrophages have roles that include suppressing inflammation, supporting tissue reconstruction, angiogenesis, and immune control.567,568 Research involving the adoptive transplant of M2 macrophages has shown that the tactic might successfully exacerbate allergic airway inflammation in rodents.569,570 Pei et al. investigated the exosomal membranes from M2 macrophages covered with biomimetic NPs (EM-PLGA@Dnmt3aossmart silencer). It was impressive that the EM-PLGA@Dnmt3aossmart silencer efficiently suppressed M2 macrophage polarization in AA, and the biomimetic NPs effectively collected in the lungs, resulting in gene silencing, along with a decrease in the level of inflammation cell invasion of the airway.144 From beginning to end, AS entails persistent macrophage activation. Triggering outstanding anti-AS reactions in ApoE-/- mice after intraperitoneal administration, the treatment of kaempferol-macrophage-biomimetic KPF shipping complex (KPF@MM-NPs) provided a vital drop in developing macrophage inflammation along with a decrease of major inflammatory cytokines and re-polarization M1 to M2 phenotype, which was linked to the inhibition of the ROS/NF-κB signaling networks.571
The etiology of ALI is greatly assisted by the stimulation of the TLR in macrophages. The proton pump inhibitors (PPIs) and the peptide-covered AuNPs-based TLR antagonists exhibit comparable modes of action, according to transcriptome sequencing and Connectivity Map analyses. The endosomal TLR cascade and inflammation reactions in macrophages and hematological mononuclear populations are proven to be inhibited by PPIs (including omeprazole), and these PPIs also demonstrate soothing effect in a murine model of ALI triggered by LPS. Omeprazole is subsequently converted into nanoscale using liposomes to improve its capacity for attacking macrophages and medicinal effectiveness in vivo.572 Moreover, P12, a novel soothing nanodrug composed of hexapeptides and AuNPs, could reduce the inflammation reactions regulated by TLR in macrophages.573
Antibodies produced in response to a specific autoantigen are the main driver of pathologic inflammation in several autoimmune disorders, indicating that therapies to promote tolerance to the autoantigen may be beneficial. Recombinant NPs have been created that cause both T cells and B cells to become tolerant. The RAP-carried PLGA core of the NPs, which stimulates the growth of Tregs, is encased in a lipid membrane. The lipid surface exhibits the antigen protein and a ligand of the B cell inhibitory co-receptor CD22, which work synergistically to prevent B cells from getting activated upon identifying the antigen.8 The optimal treatment for type 1 diabetes (T1D) is the recovery of immunological tolerance to cellular antigens since T1D is induced by the death of pancreatic beta cells by autoantigen-selective immunity CD4+ and CD8+ T lymphocytes. The homologous diabetogenic peptides could be exterior linked with or encapsulated in carboxylated 500 nm decomposable PLG NPs, which can quickly and effectively reestablish tolerance in NOD.574 In order to manage an MS model, PLG NPs coupled with pathogenic antigens were used to establish antigen-particular tolerance. T cells obtained from mice vaccinated against myelin proteolipid protein (PLP139-151) were cultivated together with APCs that were given PLP139-151-combined NPs, resulting in decreased T cell proliferation, elevated T cell death, and enhanced soothing reaction.575
Immune checkpoint proteins regulate the evolution and management of autoimmune disorders via regulating immune reactions. Recombinant murine PD-L1 (rmPD-L1) was utilized to alleviate multi-organ inflammation by regulating T cell response. The PD-1-displaying CD4+ and CD8+ T cells in the splenocytes were successfully directed by immunosuppressive composite NP therapy.576 Changes in the proportion of effector to regulatory CD4+ T cells are a hallmark of the possibly deadly autoimmune illness SLE. Immune checkpoints PD-1 and TIGIT have been shown to be upregulated in pathologic CD4+ T cells as the illness progressed, while their ligands PD-L1 and CD155 were downregulated. Guo et al. developed Dex-carried IFN-treated MHC class I defective tumor membrane-covered NPs using biomimetic nanomaterials in order to properly exploit the immunosuppressive ability of malignant cells for the therapy of SLE.577
The pathophysiology of the majority of prevalent autoimmune disorders, including psoriasis, RA, IBD, and MS, has been linked to Th17 cells.578 Furthermore, following medicinal immunodepletion, Th cell restoration rates best predict long-term effectiveness.579 The onset and progression of MS are intimately tied to the Th17/Treg imbalance, and the transformation of Th17 cells into Treg cells might assist in the reduction of irritation, offering a therapy for MS. When ROS are produced, a transformation inducer termed (aminooxy)-acetic acid is regionally liberated and then absorbed by Th17 cells. It is shown that Th17 cells can phenotypically transdifferentiate in situ into anti-inflammatory Treg cells utilizing the Trojan horse recombinant approach.500
A current focus of autoimmune therapeutics is DCs, which have a significant impact on the initiation of the adaptive immune reaction. Targeted metabolic modifier therapy that modifies DC signaling to polarize T cells suggests potential benefits, which might be attained by designing substances with more durable release.580 Low Zn2+ concentrations in hypoxic DCs may induce immunogenic DCs (igDCs), which in turn sets off an aggressive T-cell response that accelerates the immunological development of RA. To convert igDCs into tolerogenic DCs (tDCs) and prevent further T cell stimulation, Qiao et al. developed ZnO2/Catalase@liposome-Mannose nanoparticles (ZnCM NPs), a DC-specific immune-modulating method. ZnCM NPs demonstrated pH-sensitive directed intracellular transport of Zn2+ and O2 to igDCs. Upon blocking OTUB1 deubiquitination, ZnCM NPs stimulated CCL5 breakdown through NF-κB signaling, which caused the igDC-tDC transition and subsequently inhibited CD4+ T-cell homeostasis.581
Nanotechnology orchestrates a comprehensive strategy for immune reaction regulation, including immune cell transformation and immune response management, which together promote an anti-inflammatory environment and accurate management of inflammation.
Advanced diagnostic and imaging techniques
The remarkable biocompatibility, targeting potential, and regulated release potential of NPs have positive effects on inflammations. As a result, diagnosing these disorders using non-invasive imaging technology to track disease development and monitor immunomodulatory drugs may be conceivable.582
Fluorescence imaging, an innovative diagnostic tool, has piqued the interest of researchers owing to its outstanding selectivity and responsiveness.583,584 The second near-infrared spectrum (second near-infrared window(NIR-II), 1000–1700 nm) nanotheranostics may be accurately targeted to the location of inflammation while also performing diagnostic and therapeutic modes in a single nanocomplex. A proper UC diagnosis is essential for creating an effective therapeutic strategy. Following oral treatment, BM@EP (a pH/ROS dual-sensitive nanoplatform) sends the dyad (BOD-XT-DHM) into the intestines and discharges the dyad particles upon being induced by the alkalinity in colon, subsequently after being induced by overproduced H2O2 in the swollen colon, the boronated bond within the dyad is fractured and the chromophore escape for second NIR-II fluorescence and optoacoustic visualization for UC examination and recuperation assessment.585 Theranostic platforms that incorporate FI in the NIR-II and photothermal therapy (PTT) under secure laser fluence have enormous promise in preclinical study and clinical execution. Employing semiconducting polymer NPs (L1057 NPs), Yang et al. describe a theranostic platform for NIR-II FI and PTT under 980 nm laser irradiation, with low (25 mW/cm2) and high (720 mW/cm2) laser fluence, correspondingly. The great luminosity, coupled with outstanding durability and biocompatibility, enables real-time viewing of the entire body and cerebral vasculature high-clarity as well as identification of brain IS.586 The efficacy and precision of data collection are constrained by the low photoluminescence quantum yield (PLQY) of fluorescence probes in the NIR-II area, particularly in multimodal molecular visualization in vivo. Lanthanide-infused NPs with high PLQY and adjustable PL lifespan via multi-ion loading and core-shell construction are offered as a solution to this issue. The high PLQY NPs and custom-built fluorescence lifespan visualization platform may provide quick fluorescence lifespan mapping with a high signal-to-noise ratio, paving the way for multimodal molecular visualization of AS.587
MNPs are utilized in MRI to enhance the image contrast of selected regions. To improve proton relaxation and visualization, MNPs could be targeted into the tissue location.588 MNPs are nanocrystalline particulates that may be additionally modified with biocompatible coverings and ligands, and are regarded as a future MRI element. Before thrombosis is activated and larger plaques are ruptured, causing coronary artery crises and abrupt heart failure, AS is asymptomatic for decades. In order to potentially employ MRI to image clot formation on arterial plaques, Poon et al. developed mixed metal oxide-peptide amphiphile micelles (HMO-Ms), which incorporate an organic peptide amphiphile with fibrin-Interacting properties with an inorganic, magnetic iron oxide or manganese oxide center. MRI tests performed in vitro using hybrid NPs modified with CREKA showed that they were capable of targeting with a two-fold amplification of MR signals.589 At the level of cells and tissue levels, Gd/CeO2 nanozymes against AS effectively eliminate the damaging ROS. These NPs may also be used as T1-weighted MRI contrast agents, which can produce enough contrast to identify the plaque’s position throughout live imaging.559
CT is an X-ray-based clinical imaging technology that is frequently employed for non-intrusive GI tract (GIT) imaging. In the clinic, CT contrast media containing iodine and barium are employed for GIT imaging; nevertheless, IBD imaging is difficult because iodinated and barium-based CT agents are not particular for areas of irritation. The usage of dextran as a covering substance on cerium oxide NPs was expected to promote aggregation in IBD inflamed sites in a manner similar to other inflammatory disorders. Cerium oxide nanoparticles (Dex-CeNP) bound with dextran provided a potent CT contrast and collected in the colitis region. Dex-CeNP may potentially be utilized as a viable CT contrast material for imaging the GIT in patients with IBD while preventing oxidative harm.590
Metal NPs are broadly used in nanoprobes for the identification of numerous analytes, such as metal ions, nucleic acids, proteins, small biomolecules, and ROS, due to their distinct physicochemical characteristics. The extremely sensitive identification of fibroblast-like synoviocyte cells was made possible by a PEC biosensor constructed on ZnO nanorods/CH3NH3PbI3/nitrogen-modified CQDs nanocomplex.591 The concurrent non-intrusive actual time measurement of glucose and insulin in saliva was made possible by the creation of an electrochemical aptasensor on a screen-printed carbon electrode (SPCE). The sensing interface reacted uniformly to insulin in a spectrum of 0.05–15 nM with a sensitivity threshold of 0.85 nM and uniformly to glucose in the spectrum of 0.1–50 mM with the electrochemical signal measurement on SPCE.592 Clinical research has shown that the use of CD24-Au nanocomposite as a biomarker can detect stem cells in salivary gland tumors with good diagnostic and prognostic results (NCT04907422).593 In addition, advanced in vitro detection devices can reduce the burden on patients. A sensing device based on nanomaterials (organically functionalised CNTs and AuNPs) has been able to clearly distinguish AD from healthy states, PD from healthy states, and AD from PD states, by detecting exhaled gases, with a classification accuracy of 85, 78, and 15% (NCT01291550).594
Nanotechnology leverages the distinctive physical and chemical properties of NPs to revolutionize disease detection in inflammation, providing highly sensitive and accurate diagnostic tools.
The limitations/challenges of nanotechnology application
In recent years, numerous studies have focused on the development and utilization of nano-formulations as therapeutic approaches for infectious and inflammatory diseases. While nanomedicine offers unparalleled advantages over conventional medications, it also presents numerous limitations and challenges that must be addressed (Fig. 6).
Fig. 6.
Challenges of nanotechnology. There are still various types of challenges in the development of nano-formulations. One is the challenge of drug delivery across different biological barriers. Systems such as the lung and gastrointestinal tract have their specific biological barriers that can prevent the passage of some NPs drug delivery systems, making it difficult to achieve specific aggregation. Second is the challenge of NPs prescription, characterization and preparation process. Prescribing specific nanomedicines, such as ivermectin with poor water solubility, carries challenges in development and preparation, and characterizing their physicochemical properties may also be difficult to ensure re-producibility due to differences in laboratory equipment and reagents. Third, there are cost and regulatory challenges. The time and money spent on the development of nanomedicines to market is incalculable, and regulatory issues are also a major challenge. Nanomedicines need to be gradually compared with reference drugs for efficacy, safety, and quality. NPs: nanoparticles
Toxicity and safety
The extensive applications of NPs have provided an innovative driving force for advancements in biomedicine. However, the associated issues of toxicity and safety cannot be overlooked. Research suggests that organs with rapid perfusion, such as the kidneys, liver, lungs, and spleen, are most susceptible to NP accumulation and subsequent damage.187,595 The primary mechanisms underlying this damage include the induction of oxidative stress, inflammation, genotoxicity, and cytotoxicity.596,597
There have been extensive reports on the adverse in vivo effects of metal NPs. Metal NPs may possess toxicity, leading to mitochondrial and DNA damage.598–600 They can also induce excessive ROS production, which is more likely to result in cellular dysfunction.29 AuNPs can directly penetrate kidney cells, producing toxic substances through cyanidation and oxidation.601–604 AgNPs, when inhaled, not only cause pulmonary toxicity but also exhibit dermatological toxicity upon local application. If internalized into sperm, AgNPs can lead to fertilization difficulty and impaired embryo development.605,606 Furthermore, carbon-based NPs, polymer NPs, and others have been widely reported to exhibit toxicity. CNTs have been demonstrated to induce inflammation, pleural damage, and even lead to mesothelioma originating from the pleura.607 Studies have found that MWCNT can induce lung inflammation following pulmonary exposure,608 and exacerbate myocardial ischemia-reperfusion injury.609 It was later discovered that this was due to MWCNT-provoked serum bioactivity leading to endothelial inflammation and barrier disruption.610 Reports suggest that PEG-functionalized IFN-α can induce allergic reactions and rashes in patients undergoing treatment for chronic hepatitis C.611 Oligomers of PLA NPs mediate the inactivation of matrix metalloproteinase 12, resulting in intestinal damage and acute inflammation.612
It is essential to emphasize that the long-term in vivo toxicity of NPs is of great importance because they can interact with the organs, tissues, cells, and macromolecules of individuals suffering from chronic inflammatory diseases. As NPs exhibit different dispersion pathways from their microscale counterparts due to differences in their physicochemical properties, they may accumulate in the liver or even traverse the BBB to reach the brain.613,614 It has been demonstrated that many nanocarriers can negatively affect cell membrane integrity and shape, damage lysosomes, lead to misfolded proteins, promote protein oxidative damage in the Golgi apparatus, and even enter the cell nucleus, causing alterations in DNA.615,616 Inadvertent inhalation of NPs can result in pulmonary toxicity, where NPs migrate from lung alveolar cells through internalization to various organs, including the heart, liver, and spleen.616–618
So, what optimization measures can we take to avoid the potential toxicity of NPs? Several studies have found that particle size is a crucial factor affecting nanotoxicity.619,620 Therefore, by controlling the particle size and morphology of NPs, their toxicity can be reduced, thereby regulating their biodistribution, biodegradability, and cellular uptake capabilities. In addition, high surface charge of NPs may lead to equivalent nanotoxicity.621–624 Cationic AuNPs are more toxic than neutral ones, as they can disrupt cell morphology and induce more pronounced mitochondrial stress.625 Therefore, nanotoxicity can also be reduced by altering the NP’s surface charge. Furthermore, in the field of nanotoxicology, surface area is widely regarded as a crucial determinant of nanotoxicity. However, the hemolytic response triggered by quartz particles is related to the generation of H2O2 on the particle surface, suggesting that the pulmonary toxicity of quartz NPs is not only related to surface area but also to surface activity. In fact, surface modification and functionalization can alter surface properties and interaction modes, thereby reducing toxicity to organisms. Chakravarty et al. reported that CNTs can be functionalized to enhance biodegradability and reduce toxicity.33 Yu et al. reported that GSH-coated fluorescent AuNPs can improve kidney clearance efficiency and rate.626 Ge et al. demonstrated that PEGylation and lanthanide (Gd3+ and Yb3+) modification reduces the in vivo and in vitro toxicity of AuNPs, enabling their application in in vivo imaging and PTT in tumor-bearing mice.627
Therefore, gaining an in-depth understanding of and shedding light on the linkage between nanotoxicity and its physical and chemical characteristics (e.g., material structure, surface properties, shape, hydrophobicity, and aggregation state, etc.) may pave the way for optimized design of nanomaterials with enhanced biosafety in the future.
Delivery and location
Nanoparticulate materials need to be able to be delivered precisely to the target location and achieve targeted action. However, there are various barriers to NPs in living organisms, such as blood circulation, clearance by the immune system, and cell membrane blockage, which may limit their delivery efficiency and targeting. Existing studies prove that the successful delivery of drug-carrying NPs to target organs and tissues is related to the surface conditions of the target organ and the route of drug delivery.76,628–631 However, systems such as the lungs and gastrointestinal (GI) tract have their unique biological barriers that hinder the passage of certain NP drug delivery systems, making it difficult to achieve specific accumulation. For instance, although oral delivery is the most widely used and highly accepted form of drug delivery, but acidic conditions and a large number of degradative enzymes in the GI tract system can restrict the passage of some drugs, rendering them inactive before being absorbed, which results in drugs with low stability and low bioavailability are not easily absorbed in the GI tract and have difficulty reaching therapeutic levels in the blood.632
While brain-oriented improvements or biomimetic methods may enhance the specific impact of NPs in the brain, the BBB remains a significant penetration barrier. Many NPs have not yet been able to reach inflammatory diseases in the CNS, including IS and Alzheimer’s disease (AD).633 Therefore, non-intrusive or slightly intrusive techniques ought to be researched more thoroughly. For instance, intranasal delivery is a patient-friendly mode of treatment and might be a workable strategy if cerebral drug levels through this method are increased.550 However, the intranasal amount for delivery is modest, and the motion of the cilia and mucus significantly inhibits drug entry into the brain.634 In addition, it is possible to transport drugs to the brain without passing through the BBB by using the intrathecal and retro-orbital pathways. The two ways, nevertheless, are each constrained by high injection procedures and exceedingly painful patients. Furthermore, certain NPs founded on stem cells or gene treatment are employed for extremely intrusive cerebral delivery to preserve the survival of stem cells and enhance the efficacy of RNA or DNA transfection. In light of the evidence, the best medicinal benefits may be obtained by selecting the proper distribution pathways, in accordance with the various action mechanisms of NPs and associated targeting strategies.633 In addition, pharmaceutical agents need to be researched to enhance the pharmacokinetic characteristics within the actual cerebral compartment. A more efficient approach is necessary since the sluggish rate of dispersion within the parenchyma seems to limit the distribution of CNS medications. An efficient method of aiding medication transport to and throughout the brain might be offered by outside directed magnetic targeting or possibly remotely controlled nanotechnology.550
Uncertainty surrounds NPs’ in vivo procedure. Similar to conventional small-molecule medications, more research needs to be done on how NPs work in vivo. But pharmacokinetic information of drugs is infrequently disclosed for NPs while it is required for approval of medications, which limits the development of NPs as clinical drugs. We can determine its absorption, dispersion, metabolism, and elimination thanks to that information. Furthermore, as NP-based medicines focus on cells in vivo, certain studies on cell pharmacokinetics may be utilized as guidelines.635 The study by Paunovska et al. proposed a feasible approach to link the structure of nanomaterials with in vivo delivery, using high-throughput in vivo studies to investigate the delivery behavior of NPs targeting cells within the body,636 which provides a good lesson for us to study the behavior of localization and detection of nanomedicines in vivo.
Inefficient distribution of the active ingredient of the drug at the targeted location also limits the delivery of nanodrugs. For instance, due to the highly heterogeneous distribution of the vascular system in solid tumors, their high permeability and lack of proper lymphatic drainage increase the barriers to drug delivery, resulting in large tumor areas that may be poorly perfused. Local therapeutic delivery systems can address this issue, yet fail to target metastatic or disseminated tumors.637 Besides, there are many studies that can demonstrate the ability of NPs to increase drug localization and drug distribution at the target site,638,639 but the localization of NPs in various cell types is unknown and still needs further study.638,640,641
Active targeting that relies on ligand-receptor interaction is seldom employed to control the immune response by directing immune cells. A class of immune cells, such as immunogenic and tolerogenic DCs, may have different characteristics based on various phenotypes, increasing the challenge of targeted targeting based on ligand-receptor interactions.642 The possibility for precise distribution has so far been demonstrated by mannose-binding receptors, Toll-like receptors, and FA receptors.643 There is existing literature demonstrating that mannosylated ferritin NPs can precisely target B-cell follicles.644 In order to find additional relevant receptors, greater focus ought to be paid to cascade and checkpoint investigations.635 In addition, given the intricate pathogenic processes of inflammatory diseases, NPs typically only target one or two of these processes and are unable to offer a total defense. Contrarily, combining various medicinal compounds into a single nanocarrier makes the NPs more complicated and hinders the clinical application of these NPs.633
Stability and long-term effects
The stability of nanomaterials during preparation, storage and application is a critical concern. Various factors can impact the stability of NPs including preparation methods, environmental conditions such as temperature, humidity and light, as well as in vivo aspects like pH and enzyme activity.645–647 Alterations to the stability of the nanomaterials can lead to potential reductions in efficacy, heightened toxicity and other adverse outcomes. They may aggregate or degrade in the environment, thus affecting their performance and long-term effects. Although some nano-agents have shown good antiviral potential in vitro assays, in vivo assays suffer from poor stability, which leads to poor bioavailability. Ivermectin (IVM), for example, has potent antiviral activity against the Zika virus, but the development of its nanodrug is hampered not only by the poor water solubility properties, also because the pharmacokinetic profile of IVM may be influenced by specific formulations, thus altering plasma kinetics.648,649 Similarly, liposomes encapsulating lipophilic drugs are often manufactured in processes involving organic solvents, which can lead to drug leakage.650 In response, the drug formulation needs to be changed or the production process needs to be improved. Some NPs, although not exhibiting nanotoxicity in the short term, may also exhibit toxicity upon long-term exposure. It was found that after prolonged exposure (20 days) to iron oxide nanoclusters (IONC) coated with PEG, higher amounts of PEG-coated IONC were observed in the follicular centers of the white pulp of the mouse spleen, which not only led to cellular abnormalities in the splenic red pulp, but also led to a decrease in collagen levels.651 Previous studies have speculated about changes in splenocyte nucleic acid material due to the production of ROS using PEG-coated IONC.652
How can we address the reduced efficacy and increased toxicity resulting from deteriorating stability? Altering the NPs’ design or developing a stabilizer may ameliorate these concerns.653,654 Materials available for use as NP stabilizers consist of surfactants, silica, biomolecules, polymers, and metal shells.655–658 These materials assist in maintaining the structural integrity of NPs, preventing their aggregation or dissolution, and thus providing stabilization. These issues need to be examined by us in the future.
Clinical research and application
While there is a considerable number of preclinical studies demonstrating the superior benefits of nanomaterials in the treatment of infectious and inflammatory diseases, not enough nanomedicines are currently in clinical research or on the market to meet the demand. The challenges that may exist in the short term are: our current understanding of the pathogenesis of infectious and inflammatory diseases is not sufficiently deep or does not allow us to cope with rapidly changing conditions; the differentiation between animal models and the realities of the human body poses a challenge;659–662 and the technology for clinical design and translation needs to be improved.663–665
There have been numerous instances of widespread viral outbreaks in recent times, but research on antiviral nano-agents remains largely focused on treating influenza, hepatitis, herpes, coronaviruses, and HIV infections.666,667 Most of the antiviral nanomedicines that have reached the clinical research stage are designed primarily as vaccines, without sufficient capability to respond quickly to new viral outbreaks such as recent emergence of monkeypox.668 In addition, Excessive mutation rates during microorganisms genome evolution could give rise to drug-resistant strains, greatly diminishing the therapeutic efficacy of existing drugs.669,670 While nanocarriers may lead to unsuitable combinations of drugs and harmful side effects, ultimately compromising patient adherence.671 The biocompatible nanomaterials or technologies can reduce patient compliance problems, such as LNPs,672 and microneedle technology.672
We need to be aware that the efficacy of nanomedicines in preclinical research does not ensure their usefulness in humans, despite the previously exhibited enormous potential.673 The significant discrepancies in pathobiology between human and animal models could be the cause of this mismatch. For example, unlike the pathophysiology in humans, animal models of brain ischemia were often generated using the suture-occluded technique or the photochemical approach.633 Furthermore, while arterial plaque deposits in patients form over years or even decades and are thus more complicated and varied, atherosclerotic plaques in animal models typically form in weeks.674 Furthermore, animal models are frequently genetically altered in ways that aren’t always applicable to people.675 One of the initial NPs in the creation of RA nanodrugs that reached the phase of clinical trials was the liposomes. The results of a latest randomized clinical trial show that therapy of RA outbursts with intravenous infusion of PEG-coated liposomal prednisolone is superior to intramuscular methylprednisolone.676 But after administering prednisolone in PEG liposomal form intravenously, liposomes present a specific risk of inducing hypersensitive events. Persistent allergic reactions may make treatment futile, especially in inflammatory disorders. Moreover, therapeutic dosage control is hard as a result of the difficulty in accurately quantifying local drug levels of NPs administered via intravenous injection.677
In spite of the fact that the choice of preclinical models can be diverse, there are several challenges to designing a viable patient-delivered technology such as safety, stability, scalability, and cost of production.663,678 The option of nanomaterials is an important influencing factor for clinical translation. Complex approval processes for new materials may prolong the time to clinical translation, as the safety and efficacy of NPs must undergo rigorous review.679,680 Criteria for review include NP characterization and toxicity, where characterization would include size, shape, surface charge, stability, etc. In addition, unlike the singularity of preclinical research, clinical translation requires the cooperation of multiple departments, such as R&D institutions, experimental centers, regulatory agencies, and financial support. It is only when these sectors work together that promising nanomedicines can reach the market and be applied to patients.678,681,682 Therefore, more standardized policies and regulations are needed to ensure the clinical translation of nanomedicines.
Cost and production
Nanotechnology-based drugs, often associated with high costs, present challenges in clinical adoption.683 Key factors impacting drug pricing include manufacturing, scalability, and storage. To reduce costs, exploring methods to enhance production yields and alternative, cost-effective industrial processes may be necessary. At present, the two most popular methods for increasing the scalability of nanodrugs are microfluidic systems and 3D printing technologies.684 The production of nanodrugs has been revolutionized by microfluidic systems. By regulating minute fluid droplets or volumes with extreme accuracy, they make it possible to produce nanodrugs that are precise and controllable. In addition, by combining various production steps into a single system via microfluidic technology, fewer preparation procedures and shorter manufacturing times will be required.685 Also, incremental fabrication and 3D printing innovation are currently highlighted as strategies to reduce the price of nanodrugs while enhancing clinical application. Easy steps are used in this manufacturing procedure to enable economical, easily scalable output.677,686 It’s encouraging to note that there are now some nanotechnologies available on the market for virus detection. The team at Shanghai Jiao Tong University, led by Gu et al., has developed nano-magnetic beads as the core carriers for extracting virus nucleic acids. Under the influence of a magnetic field, a large number of these magnetic NPs can precisely capture the targeted entities, transport them to specified locations, and facilitate rapid testing. However, there is currently no literature reporting a method for achieving large-scale production of this technology. Cost-cutting is the key to accomplishing industrial-scale manufacture of NPs, but it is also crucial to create constant, greatly scalable, and reproducible technology for preparing batches of nanodrug with managed size, consistent layout, colloidal durability, etc., while concurrently assessing expenses and practicality.687
Intriguingly, the complexity of synthesizing NPs industrially differs. For the treatment of developed thrombotic thrombocytopenic purpura, cablivi was developed. Also, it was the sole nano-drug (Nanobody®) that had been proven to cure autoimmune illness.688 Although its function with ordinary NP was very distinct, it is simply an immunoglobulin segment generated by the cell. Nanobodies®, in contrast to traditional monoclonal antibodies, are substantially tiny (15 kDa). It has heightened solvency, enhanced durability, better and more targeted distribution due to its tiny size.689 The effective “manufacturing” of Nanobodies® occurs in a variety of prokaryotic and eukaryotic cells.690 The authorized nano-drug was generally straightforward, lacking a target ligand, combination, or alteration. On the other hand, modern NPs study frequently employs exceedingly complex alterations to emphasize progression and uniqueness, which makes industrial manufacturing quite challenging. Nevertheless, as modified NPs are more effective than pure NPs, changes are still required for clinical efficiency. Novel additives for convenient alterations, new efficient ligands (such as tiny molecular aptamers), along with straightforward modifying procedures are also highly required.635
Regulations and ethics
The application of NP materials also faces regulatory and ethical issues. Due to their unique nature and potential risks, regulatory frameworks need to be established to ensure their safety and compliance. Considering future clinical translation efforts, drugs containing NPs must be manufactured according to specifications.626 The efficacy, safety, and quality of nanomedicines need to be progressively compared with reference drugs to achieve equivalent therapeutic effects, but some nanomedicine structures are not fully characterized and in vivo activity depends on the manufacturing process.691 Approval criteria related to generic nanomedicines are also being considered, and bioequivalence of generic and innovative nanomedicines cannot be assumed by similar results observed in general pharmacokinetic and toxicity studies or by simple comparisons of drug product components, but rather by using disease models to respond to the pharmacology of nanomedicines and by continuously testing the safety of nanomedicines on clinical trials over time.
Conclusion and perspective
With the advancement of biomedical science, the underlying mechanisms of many infectious and inflammatory diseases have been elucidated, enabling their prevention, diagnosis, and early treatment. The rapid development of nanotechnology has provided a versatile “Swiss Army knife” tool for combating infectious and inflammatory diseases. Nanomaterials can serve as adjuvants and delivery carriers for vaccines, enhancing immune responses and antigen immunogenicity, stimulating potent cellular and humoral immunity to prevent pathogens infection. Moreover, nanotechnology can directly exert anti-pathogenic and anti-inflammatory effects, acting as drug delivery systems to target medications to infection and symptom sites, achieving sustained and precise drug delivery. In addition, they can also be used as biosensors, imaging enhancers for the detection of pathogens and the diagnosis of inflammatory diseases. While challenges related to the application of nanomaterials, such as toxicity, biocompatibility, long-term safety, etc. require further research and resolution, they have already demonstrated great potential in various fields, providing powerful support for future research.
NPs-based vaccines, particularly LNP-mRNA vaccines, have made significant advancements in the field of infectious disease prevention and control. In comparison to traditional vaccines, LNP-mRNA vaccines offer three major advantages. Firstly, their production speed is exceptionally rapid. Once the genetic sequence of a pathogen is obtained, the vaccine development process can be completed swiftly. Moreover, for pathogens with rapid mutational capabilities, upgraded vaccines can be developed promptly to address new variants. Secondly, another remarkable advantage of mRNA vaccines is their safety. From a production perspective, the absence of the need for large-scale cultivation of live viruses eliminates the risk of live virus leakage during vaccine production. Furthermore, mRNA segments do not enter the cell nucleus, thus eliminating the potential risk of stable integration into the host cell’s genome. In addition, mRNA vaccines typically do not require additional adjuvants, reducing the risk of adverse reactions that may be caused by adjuvants. Thirdly, another major advantage of mRNA vaccines is their high efficacy. They can simultaneously stimulate both cellular and humoral immunity, providing stronger protection against pathogen invasion. Furthermore, mRNA vaccines exhibit excellent scalability, allowing for the combination of different antigens for a multi-purpose vaccine. For instance, different sequences of SARS-CoV-2 variants can be integrated to prevent a broader range of variants. Different pathogen target sequences can also be designed together to provide simultaneous protection against multiple infections. LNP-mRNA vaccines are primarily administered via intramuscular injection. However, inhaled and intranasal delivery are considered to elicit IgA antibodies faster to block the spread of respiratory infectious diseases. This is also a potential direction for the future development of mRNA vaccines. Notably, LNP-mRNA vaccines have also demonstrated potential in cancer immunotherapy,692,693 prevention or treatment of allergies and autoimmune diseases,694–697 and even gene replacement therapy for rare genetic diseases.698 It should be highlighted that other nanovaccine platforms have shown significant potential in preventing infectious diseases like SARS-CoV-2. For instance, self-assembling NP vaccines, including the conditionally approved NVX-CoV2373 and the emergency use authorized (EUA) Coviccine (WestVac Biopharma) for SARS-CoV-2. The highly promising I53-50 self-assembling NP delivery system, has shown promising results in the research of vaccines for SARS-CoV-2, RSV, HIV, Lassa virus, and ECF.219,221,222,224,699,700 Currently, diverse nanovaccine technologies are under active development. The key to future advancement lies in expanding the range of vaccine development, strengthening international cooperation and coordination, and especially assisting developing countries in establishing comprehensive healthcare systems and training more professional technical personnel.
Nanotechnology has made some progress in the treatment of infectious and inflammatory diseases, such as the three-dimensional nanostructures against malaria,437 and the plant-derived exosome-like NPs for UC treatment.479 However, there is still a need to enhance the target specificity of nanomaterials for precision medicine. In recent years, nanomaterials have advanced rapidly in cancer therapy. Broadly speaking, cancer can also be considered a chronic inflammatory disease, so the targeting strategies of NPs in cancer treatment are worth considering for infectious and inflammatory diseases. The complexity of the tumor microenvironment poses higher demands on delivery systems, and also provides opportunities for designing and validating various advanced nanocarriers. For instance, Song et al. proposed a novel drug delivery approach that could target bacteria existing in tumor sites, specifically addressing the unique bacteria-rich tumor microenvironment.701 Some obligate or facultative anaerobic bacteria can selectively accumulate in tumor tissues, and also serve as tumor-targeting carriers.702–705 Luo et al. found that surface-modified lanthanide NPs can be readily adjusted to achieve tumor targeting through ligand modifications.706 In addition, with the establishment of NP transport models based on micrometastasis pathophysiology, personalized treatment based on the unique biology of patient micro-metastases may become a reality.707 Therefore, the advancements of nanotechnology in cancer treatment can provide inspiration and insights for targeted therapy of infectious and inflammatory diseases, paving the way for the design of more targeted and personalized treatment strategies.
A variety of nanomaterials have been developed for the detection and diagnosis of infectious and inflammatory diseases, including metal NPs, carbon-based nanomaterials, polymer nanostructures, magnetic nanomaterials, QDs, nanowires, or nanomembrane structures.708–711 Based on different detection principles, they can be mainly divided into in vitro chemical reaction detection, sensor technology, and imaging technology. However, the stability, toxicity, and specificity of nanoparticles themselves are key limiting factors in their detection applications. These limitations can be improved through combination strategies or surface modification.712–714 For example, the combination of AuNPs with fluorescence detection can expand the detection range and improve specificity.715 A dual-mode detection method (colorimetric-fluorescence) combining PCR has been designed to achieve rapid and sensitive detection of Salmonella.716 CRISPR-based detection systems, such as SHERLOCK, DETECTR, and STOPCovid, are becoming important emerging tools for viral detection due to their speed, specificity, and ease of use.717 It should be noted that the SHERLOCK test kit and DETECTR test kit have received EUA from the U.S. FDA and can be used for the detection of the SARS-CoV-2 virus. The latest generation of SHERLOCK technology enables faster (30–60 min) and more sensitive (240 nM) assays with minimal reaction system (1ul) and lower mismatch rates than traditional PCR technologies. Furthermore, the cost per test can be as low as $0.60.718,719 The ADESSO (Accurate Detection of Evolving SARS-CoV-2 through SHERLOCK Optimization) exhibits greater sensitivity (96%) and speed (15 min) compared to SHERLOCK while detecting SARS-CoV-2 variants.720 Furthermore, the Real-Time-Multiplexed SHERLOCK assay (real-time SLK) developed by Pena et al. enables simultaneous detection of multiple samples with sensitivity down to 5 copies/μL and a time to result of under 30 min.721 The CRISPR-based detection is not limited to viruses, but has also been expanded to the detection of other microorganisms, including bacteria, fungi and parasites.722–724 This highlights its potential as an important next-generation detection technology.725,726
Most of the current detections are still in the disposable testing phase, lacking real-time monitoring capabilities, which limits their application in recurrent infections and inflammatory diseases. Some research has been devoted to the development of real-time monitoring functions of nanosensors.727,728 For instance, the implantation of nanosensors in the body can minimally invasively detect the level of adriamycin in the body in real time, which can be used to assess the clinical use of the drug.729 Wearable nanobiosensors such as microneedle skin patches are more readily available for non-invasive detection of important markers in the human body.730,731 Nanomaterials have also demonstrated excellent properties as biomarkers in imaging techniques, such as MNPs that can act as contrast agents for MRI, enhancing the image signal and improving the picture quality.732,733 The current focus should be on reducing its toxicity or improving the renal clearance of the contrast agent, which is beneficial for its long-term development and clinical introduction.734 Besides, Nano-metal-organic frameworks (nano-MOFs) labeled with radionuclides have shown great potential in the anticancer field.735 Perhaps this technique could also be adapted to detect infectious or inflammatory diseases to increase the sensitivity and specificity of the test. Non-invasive imaging technology represents the path toward reducing harm in the future. For instance, nanotechnology has been integrated with “smart capsule” technology for the purpose of monitoring intestinal health.736 These recent advancements and future directions will offer greater convenience for the detection and diagnosis of infectious and inflammatory diseases.
Currently, overcoming the toxicity of NPs and enhancing their safety remains a key challenge. There are several optimization measures that can be implemented, such as adjusting dosage and exposure times sensibly to ensure benefits outweigh risks. In-depth research into solutions for the biocompatibility, toxicity, and stability of nanomaterials is essential to ensure their safe application. In addition, improving biocompatibility and reducing potential risks to the human body can be achieved through appropriate surface modifications and designs. The development of more intelligent and precise NP drug delivery systems, as well as the creation of optical, magnetic, and environmentally responsive nanomaterials, can enhance drug delivery efficiency and targeting.737,738
In recent years, artificial intelligence (AI) has shown immense potential in predicting and assessing the toxicity of nanomaterials. A recent systematic study demonstrated that machine learning algorithms can predict the cytotoxicity of nanomaterials based on their physicochemical properties, thus guiding the optimization of nanomaterial designs.739,740 For example, researchers developed the NanoTox model, which uses features like particle size and zeta potential to predict the toxicity of metal oxide NPs.741 Another study showcased how deep learning models can enhance the interpretation of immune responses and lung burdens caused by NPs, providing support for nanosafety assessments.742 Furthermore, researchers employed machine learning-assisted single-vessel analysis techniques to evaluate the permeability of NPs in tumor vasculature, offering guidance for optimizing the targeted delivery of nanodrugs.743 In summary, AI provides crucial tools for understanding the interactions between nanomaterials and biological systems and lays the foundation for establishing efficient and cost-effective nanotoxicity assessment systems. Future research is expected to utilize cutting-edge technologies like multiscale modeling and deep learning, leveraging existing data to train predictive models for real-time forecasting of complex biological effects, thereby supporting the translation of nanomedicine applications.
In summary, nanotechnology is playing an increasingly crucial role in the prevention, diagnosis, and treatment of infectious and inflammatory diseases. Relevant research is rapidly advancing and is supported by advanced technologies such as genomics, computational science, and AI. Harnessing the advantages of nanotechnology to address safety and other challenges will contribute to an overall improvement in the treatment of infectious and inflammatory diseases. Interdisciplinary collaboration is the key to achieving this goal. With further advancements in technology and research, it is believed that nanomedicine will shine brightly in addressing infectious and inflammatory diseases.
Acknowledgements
This work was supported by grants from the National Key R&D Program of China (2020YFA0509400), Guangdong Basic and Applied Basic Research Foundation (2019B030302012), National Natural Science Foundation of China (81821002, 82130082, 82341004), and 1•3•5 project for disciplines of excellence, West China Hospital, Sichuan University (ZYGD22007, ZYJC21004), Sichuan Science and Technology Program (2021YFH0002, 2023NSFSC1878), the 1•3•5 Project of Excellent Development of Discipline of West China Hospital of Sichuan University (No. ZYYC21001). BioRender was used to create the figures.
Author contributions
Guobo Shen and Na Xie conceived, designed and edited the manuscript; Yujing Huang, Xiaohan Guo, Yi Wu, Xingyu Chen, and Lixiang Feng prepared the original draft, figures and tables; Na Xie and Guobo Shen reviewed and edited the manuscript. All authors have read and approved the article.
Competing interests
The authors declare no competing interests.
Footnotes
These authors contributed equally: Yujing Huang, Xiaohan Guo, Yi Wu
Contributor Information
Na Xie, Email: naxie@scu.edu.cn.
Guobo Shen, Email: shenguobo@126.com.
References
- 1.Chen L, et al. Inflammatory responses and inflammation-associated diseases in organs. Oncotarget. 2017;9:7204–7218. doi: 10.18632/oncotarget.23208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Leiba J, et al. Molecular actors of inflammation and their signaling pathways: mechanistic insights from zebrafish. Biology. 2023;12:153. doi: 10.3390/biology12020153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Meizlish ML, Franklin RA, Zhou X, Medzhitov R. Tissue homeostasis and inflammation. Annu. Rev. Immunol. 2021;39:557–581. doi: 10.1146/annurev-immunol-061020-053734. [DOI] [PubMed] [Google Scholar]
- 4.Sansbury BE, Spite M. Resolution of acute inflammation and the role of resolvins in immunity, thrombosis, and vascular biology. Circ. Res. 2016;119:113–130. doi: 10.1161/CIRCRESAHA.116.307308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bennett JM, Reeves G, Billman GE, Sturmberg JP. Inflammation–nature’s way to efficiently respond to all types of challenges: implications for understanding and managing “the epidemic” of chronic diseases. Front. Med. 2018;5:316. doi: 10.3389/fmed.2018.00316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Medzhitov R. Origin and physiological roles of inflammation. Nature. 2008;454:428–435. doi: 10.1038/nature07201. [DOI] [PubMed] [Google Scholar]
- 7.Herrero-Cervera A, Soehnlein O, Kenne E. Neutrophils in chronic inflammatory diseases. Cell. Mol. Immunol. 2022;19:177–191. doi: 10.1038/s41423-021-00832-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Brzezicka KA, et al. Suppression of autoimmune rheumatoid arthritis with hybrid nanoparticles that induce B and T cell tolerance to self-antigen. ACS Nano. 2022;16:20206–20221. doi: 10.1021/acsnano.2c05643. [DOI] [PubMed] [Google Scholar]
- 9.Yoo D, et al. Anti‐inflammatory glycocalyx‐mimicking nanoparticles for colitis treatment: construction and in vivo evaluation. Angew. Chem. Int. Ed. 2023;62:e202304815. doi: 10.1002/anie.202304815. [DOI] [PubMed] [Google Scholar]
- 10.Yuan J, et al. Targeted (otherapies) ACS Nano. 2021;15:16076–16094. doi: 10.1021/acsnano.1c04753. [DOI] [PubMed] [Google Scholar]
- 11.Yasamineh S, et al. An overview on nanoparticle-based strategies to fight viral infections with a focus on COVID-19. J. Nanobiotechnol. 2022;20:440. doi: 10.1186/s12951-022-01625-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Huang Y-S, et al. Metal nanoparticles and nanoparticle composites are effective against Haemophilus influenzae, Streptococcus pneumoniae, and multidrug-resistant bacteria. J. Microbiol. Immunol. Infect. 2022;55:708–715. doi: 10.1016/j.jmii.2022.05.003. [DOI] [PubMed] [Google Scholar]
- 13.Kaur K, Kumar P, Kush P. Amphotericin B loaded ethyl cellulose nanoparticles with magnified oral bioavailability for safe and effective treatment of fungal infection. Biomed. Pharmacother. 2020;128:110297. doi: 10.1016/j.biopha.2020.110297. [DOI] [PubMed] [Google Scholar]
- 14.Zhou Y, et al. Controlled copper in situ growth-amplified lateral flow sensors for sensitive, reliable, and field-deployable infectious disease diagnostics. Biosens. Bioelectron. 2021;171:112753. doi: 10.1016/j.bios.2020.112753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.González LF, et al. Intranasal delivery of interferon-β-loaded nanoparticles induces control of neuroinflammation in a preclinical model of multiple sclerosis: a promising simple, effective, non-invasive, and low-cost therapy. J. Control Release. 2021;331:443–459. doi: 10.1016/j.jconrel.2020.11.019. [DOI] [PubMed] [Google Scholar]
- 16.Zhang L, et al. Nanoenzyme engineered neutrophil-derived exosomes attenuate joint injury in advanced rheumatoid arthritis via regulating inflammatory environment. Bioact. Mater. 2022;18:1–14. doi: 10.1016/j.bioactmat.2022.02.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Lussier F, Staufer O, Platzman I, Spatz JP. Can bottom-up synthetic biology generate advanced drug-delivery systems? Trends Biotechnol. 2021;39:445–459. doi: 10.1016/j.tibtech.2020.08.002. [DOI] [PubMed] [Google Scholar]
- 18.Zeng Q, et al. Wound dressing: from nanomaterials to diagnostic dressings and healing evaluations. ACS Nano. 2022;16:1708–1733. doi: 10.1021/acsnano.1c08411. [DOI] [PubMed] [Google Scholar]
- 19.Liu T, et al. Nanomaterials and nanomaterials-based drug delivery to promote cutaneous wound healing. Adv. Drug Deliv. Rev. 2023;193:114670. doi: 10.1016/j.addr.2022.114670. [DOI] [PubMed] [Google Scholar]
- 20.Geng H, et al. Noble metal nanoparticle biosensors: from fundamental studies toward point-of-care diagnostics. Acc. Chem. Res. 2022;55:593–604. doi: 10.1021/acs.accounts.1c00598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Parsamian P, et al. Enhanced nanobubble formation: gold nanoparticle conjugation to Qβ virus-like particles. ACS Nano. 2023;17:7797–7805. doi: 10.1021/acsnano.3c00638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Avellan A, et al. Nanoparticle size and coating chemistry control foliar uptake pathways, translocation, and leaf-to-rhizosphere transport in wheat. ACS Nano. 2019;13:5291–5305. doi: 10.1021/acsnano.8b09781. [DOI] [PubMed] [Google Scholar]
- 23.Darabdhara G, et al. Ag and Au nanoparticles/reduced graphene oxide composite materials: Synthesis and application in diagnostics and therapeutics. Adv. Colloid Interface Sci. 2019;271:101991. doi: 10.1016/j.cis.2019.101991. [DOI] [PubMed] [Google Scholar]
- 24.Shabatina TI, Vernaya OI, Shimanovskiy NL, Melnikov MY. Metal and metal oxides nanoparticles and nanosystems in anticancer and antiviral theragnostic agents. Pharmaceutics. 2023;15:1181. doi: 10.3390/pharmaceutics15041181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Shrivastav AM, Cvelbar U, Abdulhalim I. A comprehensive review on plasmonic-based biosensors used in viral diagnostics. Commun. Biol. 2021;4:70. doi: 10.1038/s42003-020-01615-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Leuschner F, et al. Silencing of CCR2 in myocarditis. Eur. Heart J. 2015;36:1478–1488. doi: 10.1093/eurheartj/ehu225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Grippin AJ, et al. Dendritic cell-activating magnetic nanoparticles enable early prediction of antitumor response with magnetic resonance imaging. ACS Nano. 2019;13:13884–13898. doi: 10.1021/acsnano.9b05037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Garrido C, et al. Gold nanoparticles to improve HIV drug delivery. Future Med Chem. 2015;7:1097–1107. doi: 10.4155/fmc.15.57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Khan T, et al. Plant-based gold nanoparticles; a comprehensive review of the decade-long research on synthesis, mechanistic aspects and diverse applications. Adv. Colloid Interface Sci. 2019;272:102017. doi: 10.1016/j.cis.2019.102017. [DOI] [PubMed] [Google Scholar]
- 30.Reidy B, et al. Mechanisms of silver nanoparticle release, transformation and toxicity: a critical review of current knowledge and recommendations for future studies and applications. Materials. 2013;6:2295–2350. doi: 10.3390/ma6062295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Rudramurthy G, Swamy M, Sinniah U, Ghasemzadeh A. Nanoparticles: alternatives against drug-resistant pathogenic microbes. Molecules. 2016;21:836. doi: 10.3390/molecules21070836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Porter GC, et al. AgNP/Alginate Nanocomposite hydrogel for antimicrobial and antibiofilm applications. Carbohydr. Polym. 2021;251:117017. doi: 10.1016/j.carbpol.2020.117017. [DOI] [PubMed] [Google Scholar]
- 33.Chakravarty M, Vora A. Nanotechnology-based antiviral therapeutics. Drug Deliv. Transl. Res. 2020;11:748–787. doi: 10.1007/s13346-020-00818-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Lara HH, Ixtepan-Turrent L, Garza-Treviño EN, Rodriguez-Padilla C. PVP-coated silver nanoparticles block the transmission of cell-free and cell-associated HIV-1 in human cervical culture. J. Nanobiotechnol. 2010;8:15. doi: 10.1186/1477-3155-8-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Yang Y, et al. Targeted silver nanoparticles for rheumatoid arthritis therapy via macrophage apoptosis and Re-polarization. Biomaterials. 2021;264:120390. doi: 10.1016/j.biomaterials.2020.120390. [DOI] [PubMed] [Google Scholar]
- 36.AshaRani PV, Low Kah Mun G, Hande MP, Valiyaveettil S. Cytotoxicity and Genotoxicity of Silver Nanoparticles in Human Cells. ACS Nano. 2008;3:279–290. doi: 10.1021/nn800596w. [DOI] [PubMed] [Google Scholar]
- 37.Taheri S, et al. Substrate independent silver nanoparticle based antibacterial coatings. Biomaterials. 2014;35:4601–4609. doi: 10.1016/j.biomaterials.2014.02.033. [DOI] [PubMed] [Google Scholar]
- 38.Ye S, et al. Antiviral activity of graphene oxide: how sharp edged structure and charge matter. ACS Appl. Mater. Interfaces. 2015;7:21571–21579. doi: 10.1021/acsami.5b06876. [DOI] [PubMed] [Google Scholar]
- 39.Seyed Alian R, et al. Do nanoparticles cause hormesis? Early physiological compensatory response in house crickets to a dietary admixture of GO, Ag, and GOAg composite. Sci. Total Environ. 2021;788:147801. doi: 10.1016/j.scitotenv.2021.147801. [DOI] [PubMed] [Google Scholar]
- 40.Xu P-Y, et al. Progress in antiviral fullerene research. Nanomaterials. 2022;12:2547. doi: 10.3390/nano12152547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Banerjee I, Douaisi MP, Mondal D, Kane RS. Light-activated nanotube–porphyrin conjugates as effective antiviral agents. Nanotechnology. 2012;23:105101. doi: 10.1088/0957-4484/23/10/105101. [DOI] [PubMed] [Google Scholar]
- 42.Sharmeen S, et al. Polyethylene glycol functionalized carbon nanotubes/gelatin-chitosan nanocomposite: An approach for significant drug release. Bioact. Mater. 2018;3:236–244. doi: 10.1016/j.bioactmat.2018.03.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Ramos GS, et al. Antileishmanial activity of fullerol and its liposomal formulation in experimental models of visceral leishmaniasis. Biomed. Pharmacother. 2021;134:111120. doi: 10.1016/j.biopha.2020.111120. [DOI] [PubMed] [Google Scholar]
- 44.Khalid A, et al. Multiwalled carbon nanotubes functionalized bacterial cellulose as an efficient healing material for diabetic wounds. Int. J. Biol. Macromol. 2022;203:256–267. doi: 10.1016/j.ijbiomac.2022.01.146. [DOI] [PubMed] [Google Scholar]
- 45.Zhang M, Yu P, Mao L. Rational design of surface/interface chemistry for quantitative in vivo monitoring of brain chemistry. Acc. Chem. Res. 2012;45:533–543. doi: 10.1021/ar200196h. [DOI] [PubMed] [Google Scholar]
- 46.Panwar N, et al. Nanocarbons for biology and medicine: sensing, imaging, and drug delivery. Chem. Rev. 2019;119:9559–9656. doi: 10.1021/acs.chemrev.9b00099. [DOI] [PubMed] [Google Scholar]
- 47.Jariwala D, et al. Carbon nanomaterials for electronics, optoelectronics, photovoltaics, and sensing. Chem. Soc. Rev. 2013;42:2824–2860. doi: 10.1039/C2CS35335K. [DOI] [PubMed] [Google Scholar]
- 48.Wang H, et al. Downsized sheath–core conducting fibers for weavable superelastic wires, biosensors, supercapacitors, and strain sensors. Adv. Mater. 2016;28:4998–5007. doi: 10.1002/adma.201600405. [DOI] [PubMed] [Google Scholar]
- 49.Bartelmess J, Quinn SJ, Giordani S. Carbon nanomaterials: multi-functional agents for biomedical fluorescence and Raman imaging. Chem. Soc. Rev. 2015;44:4672–4698. doi: 10.1039/C4CS00306C. [DOI] [PubMed] [Google Scholar]
- 50.Yang Z, et al. Zero-dimensional carbon nanomaterials for fluorescent sensing and imaging. Chem. Rev. 2023;123:11047–11136. doi: 10.1021/acs.chemrev.3c00186. [DOI] [PubMed] [Google Scholar]
- 51.Chen D, Dougherty CA, Zhu K, Hong H. Theranostic applications of carbon nanomaterials in cancer: focus on imaging and cargo delivery. J. Control Release. 2015;210:230–245. doi: 10.1016/j.jconrel.2015.04.021. [DOI] [PubMed] [Google Scholar]
- 52.Kumar S, et al. Carbon nanotubes: a novel material for multifaceted applications in human healthcare. Chem. Soc. Rev. 2017;46:158–196. doi: 10.1039/C6CS00517A. [DOI] [PubMed] [Google Scholar]
- 53.Alagarsamy KN, et al. Carbon nanomaterials for cardiovascular theranostics: promises and challenges. Bioact. Mater. 2021;6:2261–2280. doi: 10.1016/j.bioactmat.2020.12.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Hou X, Zaks T, Langer R, Dong Y. Lipid nanoparticles for mRNA delivery. Nature reviews. Materials. 2021;6:1078–1094. doi: 10.1038/s41578-021-00358-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Wang C, Zhang Y, Dong Y. Lipid nanoparticle–mRNA formulations for therapeutic applications. Acc. Chem. Res. 2021;54:4283–4293. doi: 10.1021/acs.accounts.1c00550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Böttger R, et al. Lipid-based nanoparticle technologies for liver targeting. Adv. Drug Deliv. Rev. 2020;154-155:79–101. doi: 10.1016/j.addr.2020.06.017. [DOI] [PubMed] [Google Scholar]
- 57.Puri A, et al. Lipid-based nanoparticles as pharmaceutical drug carriers: from concepts to clinic. Crit. Rev. Ther. Drug Carr. Syst. 2009;26:523–580. doi: 10.1615/CritRevTherDrugCarrierSyst.v26.i6.10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Alameh M-G, et al. Lipid nanoparticles enhance the efficacy of mRNA and protein subunit vaccines by inducing robust T follicular helper cell and humoral responses. Immunity. 2021;54:2877–2892.e2877. doi: 10.1016/j.immuni.2021.11.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Tenchov R, Bird R, Curtze AE, Zhou Q. Lipid nanoparticles—from liposomes to mRNA vaccine delivery, a landscape of research diversity and advancement. ACS Nano. 2021;15:16982–17015. doi: 10.1021/acsnano.1c04996. [DOI] [PubMed] [Google Scholar]
- 60.Deng Y-Q, et al. Lipid nanoparticle-encapsulated mRNA antibody provides long-term protection against SARS-CoV-2 in mice and hamsters. Cell Res. 2022;32:375–382. doi: 10.1038/s41422-022-00630-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Swingle KL, Hamilton AG, Mitchell MJ. Lipid nanoparticle-mediated delivery of mRNA therapeutics and vaccines. Trends Mol. Med. 2021;27:616–617. doi: 10.1016/j.molmed.2021.03.003. [DOI] [PubMed] [Google Scholar]
- 62.Li T, Cipolla D, Rades T, Boyd BJ. Drug nanocrystallisation within liposomes. J. Control Release. 2018;288:96–110. doi: 10.1016/j.jconrel.2018.09.001. [DOI] [PubMed] [Google Scholar]
- 63.Yang T, et al. Liposome formulation of paclitaxel with enhanced solubility and stability. Drug Deliv. 2008;14:301–308. doi: 10.1080/10717540601098799. [DOI] [PubMed] [Google Scholar]
- 64.Mehta RT. Liposome encapsulation of clofazimine reduces toxicity in vitro and in vivo and improves therapeutic efficacy in the beige mouse model of disseminated Mycobacterium avium-M. intracellulare complex infection. Antimicrob. Agents Chemother. 1996;40:1893–1902. doi: 10.1128/AAC.40.8.1893. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Liu P, Chen G, Zhang J. A review of liposomes as a drug delivery system: current status of approved products, regulatory environments, and future perspectives. Molecules. 2022;27:1372. doi: 10.3390/molecules27041372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Bhaskaran NA, et al. Orally delivered solid lipid nanoparticles of irinotecan coupled with chitosan surface modification to treat colon cancer: Preparation, in-vitro and in-vivo evaluations. Int. J. Biol. Macromol. 2022;211:301–315. doi: 10.1016/j.ijbiomac.2022.05.060. [DOI] [PubMed] [Google Scholar]
- 67.Buzyurova DN, et al. Surface modification of pralidoxime chloride-loaded solid lipid nanoparticles for enhanced brain reactivation of organophosphorus-inhibited AChE: pharmacokinetics in rat. Toxicology. 2020;444:152578. doi: 10.1016/j.tox.2020.152578. [DOI] [PubMed] [Google Scholar]
- 68.Nabi B, Rehman S, Baboota S, Ali J. Insights on oral drug delivery of lipid nanocarriers: a win-win solution for augmenting bioavailability of antiretroviral drugs. AAPS PharmSciTech. 2019;20:60. doi: 10.1208/s12249-018-1284-9. [DOI] [PubMed] [Google Scholar]
- 69.Xu Y, et al. Surface modification of lipid-based nanoparticles. ACS Nano. 2022;16:7168–7196. doi: 10.1021/acsnano.2c02347. [DOI] [PubMed] [Google Scholar]
- 70.Freyn AW, et al. A multi-targeting, nucleoside-modified mRNA influenza virus vaccine provides broad protection in mice. Mol. Ther. 2020;28:1569–1584. doi: 10.1016/j.ymthe.2020.04.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Verma AK, Perlman S. Lipid nanoparticle-mRNA: another step in the fight against COVID-19. Cell Res. 2022;32:421–422. doi: 10.1038/s41422-022-00647-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Ball R, Bajaj P, Whitehead K. Achieving long-term stability of lipid nanoparticles: examining the effect of pH, temperature, and lyophilization. Int. J. Nanomed. 2016;12:305–315. doi: 10.2147/IJN.S123062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Li Y, Ye Z, Yang H, Xu Q. Tailoring combinatorial lipid nanoparticles for intracellular delivery of nucleic acids, proteins, and drugs. Acta Pharm. Sin. B. 2022;12:2624–2639. doi: 10.1016/j.apsb.2022.04.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Qin Y, et al. Evaluation of a DoE based approach for comprehensive modelling of the effect of lipid nanoparticle composition on nucleic acid delivery. Biomaterials. 2023;299:122158. doi: 10.1016/j.biomaterials.2023.122158. [DOI] [PubMed] [Google Scholar]
- 75.Cui L, et al. Mechanistic studies of an automated lipid nanoparticle reveal critical pharmaceutical properties associated with enhanced mRNA functional delivery in vitro and in vivo. Small. 2021;18:e2105832. doi: 10.1002/smll.202105832. [DOI] [PubMed] [Google Scholar]
- 76.Wang Z, et al. Development and applications of mRNA treatment based on lipid nanoparticles. Biotechnol. Adv. 2023;65:108130. doi: 10.1016/j.biotechadv.2023.108130. [DOI] [PubMed] [Google Scholar]
- 77.Li Z, et al. Enzyme-catalyzed one-step synthesis of ionizable cationic lipids for lipid nanoparticle-based mRNA COVID-19 vaccines. ACS Nano. 2022;16:18936–18950. doi: 10.1021/acsnano.2c07822. [DOI] [PubMed] [Google Scholar]
- 78.Samaridou E, Heyes J, Lutwyche P. Lipid nanoparticles for nucleic acid delivery: current perspectives. Adv. Drug Deliv. Rev. 2020;154-155:37–63. doi: 10.1016/j.addr.2020.06.002. [DOI] [PubMed] [Google Scholar]
- 79.Barriga HMG, et al. Coupling lipid nanoparticle structure and automated single‐particle composition analysis to design phospholipase‐responsive nanocarriers. Adv. Mater. 2022;34:e2200839. doi: 10.1002/adma.202200839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Patel S, et al. Naturally-occurring cholesterol analogues in lipid nanoparticles induce polymorphic shape and enhance intracellular delivery of mRNA. Nat. Commun. 2020;11:983. doi: 10.1038/s41467-020-14527-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Eygeris Y, Gupta M, Kim J, Sahay G. Chemistry of lipid nanoparticles for RNA delivery. Acc. Chem. Res. 2021;55:2–12. doi: 10.1021/acs.accounts.1c00544. [DOI] [PubMed] [Google Scholar]
- 82.Paoletti, A. et al. Liposomal antagomiR‐155‐5p restores anti‐inflammatory macrophages and improves arthritis in preclinical models of rheumatoid arthritis. Arthritis Rheumatol. 10.1002/art.42665 (2023). [DOI] [PubMed]
- 83.Bevilacqua Rolfsen Ferreira da Silva G, et al. Comparative effectiveness of preventive treatment with dimethyl fumarate-loaded solid lipid nanoparticles and oral dimethyl fumarate in a mouse model of multiple sclerosis. J. Autoimmun. 2022;132:102893. doi: 10.1016/j.jaut.2022.102893. [DOI] [PubMed] [Google Scholar]
- 84.Wang S, et al. Sialic acid-conjugate modified doxorubicin nanoplatform for treating neutrophil-related inflammation. J. Control Release. 2021;337:612–627. doi: 10.1016/j.jconrel.2021.07.044. [DOI] [PubMed] [Google Scholar]
- 85.Zhou A, et al. Bioengineered neutrophil extinguisher targets cascade immune pathways of macrophages for alleviating cytokine storm in pneumonia. ACS Nano. 2023;17:16461–16477. doi: 10.1021/acsnano.3c00227. [DOI] [PubMed] [Google Scholar]
- 86.Satta S, et al. An engineered nano-liposome-human ACE2 decoy neutralizes SARS-CoV-2 Spike protein-induced inflammation in both murine and human macrophages. Theranostics. 2022;12:2639–2657. doi: 10.7150/thno.66831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Kolyvushko O, et al. Differentially-charged liposomes interact with alphaherpesviruses and interfere with virus entry. Pathogens. 2020;9:359. doi: 10.3390/pathogens9050359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Kauffman KJ, et al. Optimization of lipid nanoparticle formulations for mRNA delivery in vivo with fractional factorial and definitive screening designs. Nano Lett. 2015;15:7300–7306. doi: 10.1021/acs.nanolett.5b02497. [DOI] [PubMed] [Google Scholar]
- 89.Oberli MA, et al. Lipid nanoparticle assisted mRNA delivery for potent cancer immunotherapy. Nano Lett. 2016;17:1326–1335. doi: 10.1021/acs.nanolett.6b03329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Liang F, et al. Efficient targeting and activation of antigen-presenting cells in vivo after modified mRNA vaccine administration in rhesus macaques. Mol. Ther. 2017;25:2635–2647. doi: 10.1016/j.ymthe.2017.08.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Halperin SA, et al. Final efficacy analysis, interim safety analysis, and immunogenicity of a single dose of recombinant novel coronavirus vaccine (adenovirus type 5 vector) in adults 18 years and older: an international, multicentre, randomised, double-blinded, placebo-controlled phase 3 trial. Lancet. 2022;399:237–248. doi: 10.1016/S0140-6736(21)02753-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Verbeke R, Hogan MJ, Loré K, Pardi N. Innate immune mechanisms of mRNA vaccines. Immunity. 2022;55:1993–2005. doi: 10.1016/j.immuni.2022.10.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Li B, et al. Combinatorial design of nanoparticles for pulmonary mRNA delivery and genome editing. Nat. Biotechnol. 2023;41:1410–1415. doi: 10.1038/s41587-023-01679-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Cheng CJ, Tietjen GT, Saucier-Sawyer JK, Saltzman WM. A holistic approach to targeting disease with polymeric nanoparticles. Nat. Rev. Drug Discov. 2015;14:239–247. doi: 10.1038/nrd4503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Liu B, et al. In situ formation of polymeric nanoassemblies using an efficient reversible click reaction. Angew. Chem. Int. Ed. 2020;59:15135–15140. doi: 10.1002/anie.202004017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Liu Q, et al. Use of polymeric nanoparticle platform targeting the liver to induce treg-mediated antigen-specific immune tolerance in a pulmonary allergen sensitization model. ACS Nano. 2019;13:4778–4794. doi: 10.1021/acsnano.9b01444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Wu H, et al. Brain targeting, antioxidant polymeric nanoparticles for stroke drug delivery and therapy. Small. 2022;18:e2107126. doi: 10.1002/smll.202107126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Liao R, Pon J, Chungyoun M, Nance E. Enzymatic protection and biocompatibility screening of enzyme-loaded polymeric nanoparticles for neurotherapeutic applications. Biomaterials. 2020;257:120238. doi: 10.1016/j.biomaterials.2020.120238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Cordeiro AS, et al. Nanovaccine delivery approaches and advanced delivery systems for the prevention of viral infections: from development to clinical application. Pharmaceutics. 2021;13:2091. doi: 10.3390/pharmaceutics13122091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Gutjahr A, et al. Biodegradable polymeric nanoparticles-based vaccine adjuvants for lymph nodes targeting. Vaccines. 2016;4:34. doi: 10.3390/vaccines4040034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Peviani M, et al. Biodegradable polymeric nanoparticles administered in the cerebrospinal fluid: Brain biodistribution, preferential internalization in microglia and implications for cell-selective drug release. Biomaterials. 2019;209:25–40. doi: 10.1016/j.biomaterials.2019.04.012. [DOI] [PubMed] [Google Scholar]
- 102.Wibowo D, et al. Polymeric nanoparticle vaccines to combat emerging and pandemic threats. Biomaterials. 2021;268:120597. doi: 10.1016/j.biomaterials.2020.120597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Yin Q, et al. A TLR7-nanoparticle adjuvant promotes a broad immune response against heterologous strains of influenza and SARS-CoV-2. Nat. Mater. 2023;22:380–390. doi: 10.1038/s41563-022-01464-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.El-Sissi AF, et al. Chitosan and chitosan nanoparticles as adjuvant in local Rift Valley Fever inactivated vaccine. 3 Biotech. 2020;10:88. doi: 10.1007/s13205-020-2076-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Thomas C, Rawat A, Hope-Weeks L, Ahsan F, Aerosolized PLA. Aerosolized PLA and PLGA nanoparticles enhance humoral, mucosal and cytokine responses to hepatitis B vaccine. Mol. Pharm. 2011;8:405–415. doi: 10.1021/mp100255c. [DOI] [PubMed] [Google Scholar]
- 106.Kianfar E. Protein nanoparticles in drug delivery: animal protein, plant proteins and protein cages, albumin nanoparticles. J. Nanobiotechnol. 2021;19:159. doi: 10.1186/s12951-021-00896-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Habibi N, Mauser A, Ko Y, Lahann J. Protein nanoparticles: uniting the power of proteins with engineering design approaches. Adv. Sci. 2022;9:e2104012. doi: 10.1002/advs.202104012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Cai R, Chen C. The crown and the scepter: roles of the protein corona in nanomedicine. Adv. Mater. 2018;31:e1805740. doi: 10.1002/adma.201805740. [DOI] [PubMed] [Google Scholar]
- 109.Stupka I. & Heddle, J. G. Artificial protein cages—inspiration, construction, and observation. Curr. Opin. Struct. Biol. 2020;64:66–73. doi: 10.1016/j.sbi.2020.05.014. [DOI] [PubMed] [Google Scholar]
- 110.Vázquez E, Villaverde A. Engineering building blocks for self-assembling protein nanoparticles. Microb. Cell Factories. 2010;9:101. doi: 10.1186/1475-2859-9-101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Theil ECFerritin. The protein nanocage and iron biomineral in health and in disease. Inorg. Chem. 2013;52:12223–12233. doi: 10.1021/ic400484n. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Waters ER, Vierling E. Plant small heat shock proteins—evolutionary and functional diversity. N. Phytol. 2020;227:24–37. doi: 10.1111/nph.16536. [DOI] [PubMed] [Google Scholar]
- 113.DeFrates K, et al. Protein polymer-based nanoparticles: fabrication and medical applications. Int. J. Mol. Sci. 2018;19:1717. doi: 10.3390/ijms19061717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.de Pinho Favaro MT, et al. Protein nanoparticles are nontoxic, tuneable cell stressors. Nanomedicine. 2018;13:255–268. doi: 10.2217/nnm-2017-0294. [DOI] [PubMed] [Google Scholar]
- 115.Bertrand N, et al. Mechanistic understanding of in vivo protein corona formation on polymeric nanoparticles and impact on pharmacokinetics. Nat. Commun. 2017;8:777. doi: 10.1038/s41467-017-00600-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Nayak D, et al. Stavudine loaded gelatin liposomes for HIV therapy: preparation, characterization and in vitro cytotoxic evaluation. Mater. Sci. Eng. C. 2017;73:406–416. doi: 10.1016/j.msec.2016.12.073. [DOI] [PubMed] [Google Scholar]
- 117.Kang MG, et al. Nanogels derived from fish gelatin: application to drug delivery system. Mar. Drugs. 2019;17:246. doi: 10.3390/md17040246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Salvati A, et al. Transferrin-functionalized nanoparticles lose their targeting capabilities when a biomolecule corona adsorbs on the surface. Nat. Nanotechnol. 2013;8:137–143. doi: 10.1038/nnano.2012.237. [DOI] [PubMed] [Google Scholar]
- 119.Doan VS, Chun S, Feng J, Shin S. Confinement-dependent diffusiophoretic transport of nanoparticles in collagen hydrogels. Nano Lett. 2021;21:7625–7630. doi: 10.1021/acs.nanolett.1c02251. [DOI] [PubMed] [Google Scholar]
- 120.McColman S, et al. Serum proteins on nanoparticles: early stages of the “protein corona”. Nanoscale. 2021;13:20550–20563. doi: 10.1039/D1NR06137B. [DOI] [PubMed] [Google Scholar]
- 121.Müller J, et al. Denaturation via surfactants changes composition of protein corona. Biomacromolecules. 2018;19:2657–2664. doi: 10.1021/acs.biomac.8b00278. [DOI] [PubMed] [Google Scholar]
- 122.Tobola F, Wiltschi B. One, two, many: strategies to alter the number of carbohydrate binding sites of lectins. Biotechnol. Adv. 2022;60:108020. doi: 10.1016/j.biotechadv.2022.108020. [DOI] [PubMed] [Google Scholar]
- 123.Dan X, Liu W, Ng TB. Development and applications of lectins as biological tools in biomedical research. Med. Res Rev. 2015;36:221–247. doi: 10.1002/med.21363. [DOI] [PubMed] [Google Scholar]
- 124.Mohsen MO, Bachmann MF. Virus-like particle vaccinology, from bench to bedside. Cell. Mol. Immunol. 2022;19:993–1011. doi: 10.1038/s41423-022-00897-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Mohsen M, Gomes A, Vogel M, Bachmann M. Interaction of viral capsid-derived virus-like particles (VLPs) with the innate immune system. Vaccines. 2018;6:37. doi: 10.3390/vaccines6030037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Mohsen MO, et al. A scalable and highly immunogenic virus‐like particle‐based vaccine against SARS‐CoV‐2. Allergy. 2021;77:243–257. doi: 10.1111/all.15080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Krug PW, et al. EV-D68 virus-like particle vaccines elicit cross-clade neutralizing antibodies that inhibit infection and block dissemination. Sci. Adv. 2023;9:eadg6076. doi: 10.1126/sciadv.adg6076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Kong J, et al. Rationally designed peptidyl virus‐like particles enable targeted delivery of genetic cargo. Angew. Chem. Int. Ed. 2018;57:14032–14036. doi: 10.1002/anie.201805868. [DOI] [PubMed] [Google Scholar]
- 129.Suffian IFBM, Al-Jamal KT. Bioengineering of virus-like particles as dynamic nanocarriers for in vivo delivery and targeting to solid tumours. Adv. Drug Deliv. Rev. 2022;180:114030. doi: 10.1016/j.addr.2021.114030. [DOI] [PubMed] [Google Scholar]
- 130.Fiebiger E, et al. Cytokines regulate proteolysis in major histocompatibility complex class II-dependent antigen presentation by dendritic cells. J. Exp. Med. 2001;193:881–892. doi: 10.1084/jem.193.8.881. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Nooraei S, et al. Virus-like particles: preparation, immunogenicity and their roles as nanovaccines and drug nanocarriers. J. Nanobiotechnol. 2021;19:59. doi: 10.1186/s12951-021-00806-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Krizkova S, et al. Nanotechnologies in protein microarrays. Nanomedicine. 2015;10:2743–2755. doi: 10.2217/nnm.15.81. [DOI] [PubMed] [Google Scholar]
- 133.Jiang C, Zhang N, Hu X, Wang H. Tumor-associated exosomes promote lung cancer metastasis through multiple mechanisms. Mol. Cancer. 2021;20:117. doi: 10.1186/s12943-021-01411-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Peng Y, et al. Exosome and virus infection. Front. Immunol. 2023;14:1154217. doi: 10.3389/fimmu.2023.1154217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Zou J, et al. Therapeutic potential and mechanisms of mesenchymal stem cell-derived exosomes as bioactive materials in tendon–bone healing. J. Nanobiotechnol. 2023;21:14. doi: 10.1186/s12951-023-01778-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Pegtel DM, Gould SJ. Exosomes. Annu. Rev. Biochem. 2019;88:487–514. doi: 10.1146/annurev-biochem-013118-111902. [DOI] [PubMed] [Google Scholar]
- 137.Gunassekaran GR, Poongkavithai Vadevoo SM, Baek M-C, Lee B. M1 macrophage exosomes engineered to foster M1 polarization and target the IL-4 receptor inhibit tumor growth by reprogramming tumor-associated macrophages into M1-like macrophages. Biomaterials. 2021;278:121137. doi: 10.1016/j.biomaterials.2021.121137. [DOI] [PubMed] [Google Scholar]
- 138.Tao S-C, et al. Exosomes derived from miR-140-5p-overexpressing human synovial mesenchymal stem cells enhance cartilage tissue regeneration and prevent osteoarthritis of the knee in a rat model. Theranostics. 2017;7:180–195. doi: 10.7150/thno.17133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Cheng Z, Li M, Dey R, Chen Y. Nanomaterials for cancer therapy: current progress and perspectives. J. Hematol. Oncol. 2021;14:85. doi: 10.1186/s13045-021-01096-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Xiao, Y. et al. Mesenchymal stem cell-derived exosomes: versatile nanomaterials for skin wound treatment. Nano Res. 10.1007/s12274-023-6080-5 (2023).
- 141.Perocheau D, et al. Clinical applications for exosomes: are we there yet? Br. J. Pharmacol. 2021;178:2375–2392. doi: 10.1111/bph.15432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Tsai SJ, et al. Exosome-mediated mRNA delivery in vivo is safe and can be used to induce SARS-CoV-2 immunity. J. Biol. Chem. 2021;297:101266. doi: 10.1016/j.jbc.2021.101266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Wang Z, et al. Exosomes decorated with a recombinant SARS-CoV-2 receptor-binding domain as an inhalable COVID-19 vaccine. Nat. Biomed. Eng. 2022;6:791–805. doi: 10.1038/s41551-022-00902-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Pei W, et al. Exosome membrane-modified M2 macrophages targeted nanomedicine: treatment for allergic asthma. J. Control Release. 2021;338:253–267. doi: 10.1016/j.jconrel.2021.08.024. [DOI] [PubMed] [Google Scholar]
- 145.He C, Zheng S, Luo Y, Wang B. Exosome theranostics: biology and translational medicine. Theranostics. 2018;8:237–255. doi: 10.7150/thno.21945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Xu Y-X, et al. Exosomal ncRNAs: Novel therapeutic target and biomarker for diabetic complications. Pharmacol. Res. 2022;178:106135. doi: 10.1016/j.phrs.2022.106135. [DOI] [PubMed] [Google Scholar]
- 147.Zhu X, et al. Alone and combined toxicity of ZnO nanoparticles and graphene quantum dots on microalgae Gymnodinium. Environ. Sci. Pollut. Res. 2022;29:47310–47322. doi: 10.1007/s11356-022-19267-y. [DOI] [PubMed] [Google Scholar]
- 148.Zhou Y, et al. PLLA microcapsules combined with silver nanoparticles and chlorhexidine acetate showing improved antibacterial effect. Mater. Sci. Eng. C. 2017;78:349–353. doi: 10.1016/j.msec.2017.04.100. [DOI] [PubMed] [Google Scholar]
- 149.Xu Z, Shi L, Yang M, Zhu L. Preparation and biomedical applications of silk fibroin-nanoparticles composites with enhanced properties—a review. Mater. Sci. Eng. C. 2019;95:302–311. doi: 10.1016/j.msec.2018.11.010. [DOI] [PubMed] [Google Scholar]
- 150.Jiang D, et al. Nanozyme: new horizons for responsive biomedical applications. Chem. Soc. Rev. 2019;48:3683–3704. doi: 10.1039/C8CS00718G. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Sharma G, et al. Advances in nanozymes as a paradigm for viral diagnostics and therapy. Pharmacol. Rev. 2023;75:739–757. doi: 10.1124/pharmrev.122.000719. [DOI] [PubMed] [Google Scholar]
- 152.Wang J, et al. Utilizing nanozymes for combating COVID-19: advancements in diagnostics, treatments, and preventative measures. J. Nanobiotechnol. 2023;21:200. doi: 10.1186/s12951-023-01945-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Hou J, Xianyu Y. Tailoring the surface and composition of nanozymes for enhanced bacterial binding and antibacterial activity. Small. 2023;19:e2302640. doi: 10.1002/smll.202302640. [DOI] [PubMed] [Google Scholar]
- 154.Kumar V, et al. Recent technological advances in mechanism, toxicity, and food perspectives of enzyme-mediated aflatoxin degradation. Crit. Rev. Food Sci. Nutr. 2021;62:5395–5412. doi: 10.1080/10408398.2021.2010647. [DOI] [PubMed] [Google Scholar]
- 155.Songca SP. Applications of nanozymology in the detection and identification of viral, bacterial and fungal pathogens. Int. J. Mol. Sci. 2022;23:4638. doi: 10.3390/ijms23094638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Jiang Y, et al. Nanomaterials alleviating redox stress in neurological diseases: mechanisms and applications. J. Nanobiotechnol. 2022;20:265. doi: 10.1186/s12951-022-01434-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Salvador-Morales C, Grodzinski P. Nanotechnology Tools Enabling Biological Discovery. ACS Nano. 2022;16:5062–5084. doi: 10.1021/acsnano.1c10635. [DOI] [PubMed] [Google Scholar]
- 158.Yuan Y, et al. Histidine modulates amyloid-like assembly of peptide nanomaterials and confers enzyme-like activity. Nat. Commun. 2023;14:5808. doi: 10.1038/s41467-023-41591-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Ma M, et al. Self-protecting biomimetic nanozyme for selective and synergistic clearance of peripheral amyloid-β in an Alzheimer’s disease model. J. Am. Chem. Soc. 2020;142:21702–21711. doi: 10.1021/jacs.0c08395. [DOI] [PubMed] [Google Scholar]
- 160.Ji W, et al. Self‐catalytic small interfering RNA nanocarriers for synergistic treatment of neurodegenerative diseases. Adv. Mater. 2021;34:e2105711. doi: 10.1002/adma.202105711. [DOI] [PubMed] [Google Scholar]
- 161.Wang, Z. et al. A thrombin‐activated peptide‐templated nanozyme for remedying ischemic stroke via thrombolytic and neuroprotective actions. Adv. Mater. 10.1002/adma.202210144 (2023). [DOI] [PubMed]
- 162.Liu Y, et al. Multifunctional Janus nanoplatform for efficiently synergistic theranostics of rheumatoid arthritis. ACS Nano. 2023;17:8167–8182. doi: 10.1021/acsnano.2c11777. [DOI] [PubMed] [Google Scholar]
- 163.Pudineh Moarref M, Alimolaei M, Emami T, Koohi MK. Development and evaluation of cell membrane-based biomimetic nanoparticles loaded by Clostridium perfringens epsilon toxin: a novel vaccine delivery platform for Clostridial-associated diseases. Nanotoxicology. 2023;17:420–431. doi: 10.1080/17435390.2023.2252899. [DOI] [PubMed] [Google Scholar]
- 164.Jeong Y, et al. Stimuli-responsive adaptive nanotoxin to directly penetrate the cellular membrane by molecular folding and unfolding. J. Am. Chem. Soc. 2022;144:5503–5516. doi: 10.1021/jacs.2c00084. [DOI] [PubMed] [Google Scholar]
- 165.Wu P, Yan X-P. Doped quantum dots for chemo/biosensing and bioimaging. Chem. Soc. Rev. 2013;42:5489–5521. doi: 10.1039/c3cs60017c. [DOI] [PubMed] [Google Scholar]
- 166.Wang Z-G, Liu S-L, Pang D-W. Quantum dots: a promising fluorescent label for probing virus trafficking. Acc. Chem. Res. 2021;54:2991–3002. doi: 10.1021/acs.accounts.1c00276. [DOI] [PubMed] [Google Scholar]
- 167.Liu S-L, Wang Z-G, Zhang Z-L, Pang D-W. Tracking single viruses infecting their host cells using quantum dots. Chem. Soc. Rev. 2016;45:1211–1224. doi: 10.1039/C5CS00657K. [DOI] [PubMed] [Google Scholar]
- 168.Ashrafizadeh M, et al. Carbon dots as versatile nanoarchitectures for the treatment of neurological disorders and their theranostic applications: A review. Adv. Colloid Interface Sci. 2020;278:102123. doi: 10.1016/j.cis.2020.102123. [DOI] [PubMed] [Google Scholar]
- 169.Gopalan D, Pandey A, Udupa N, Mutalik S. Receptor specific, stimuli responsive and subcellular targeted approaches for effective therapy of Alzheimer: Role of surface engineered nanocarriers. J. Control Release. 2020;319:183–200. doi: 10.1016/j.jconrel.2019.12.034. [DOI] [PubMed] [Google Scholar]
- 170.Ma Z, et al. Near‐infrared IIb fluorescence imaging of vascular regeneration with dynamic tissue perfusion measurement and high spatial resolution. Adv. Funct. Mater. 2018;28:1803417. doi: 10.1002/adfm.201803417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Wu G, et al. Enhanced proliferation of visualizable mesenchymal stem cell–platelet hybrid cell for versatile intracerebral hemorrhage treatment. ACS Nano. 2023;17:7352–7365. doi: 10.1021/acsnano.2c11329. [DOI] [PubMed] [Google Scholar]
- 172.Kirtane AR, et al. Nanotechnology approaches for global infectious diseases. Nat. Nanotechnol. 2021;16:369–384. doi: 10.1038/s41565-021-00866-8. [DOI] [PubMed] [Google Scholar]
- 173.Zhong NS, et al. Epidemiology and cause of severe acute respiratory syndrome (SARS) in Guangdong, People’s Republic of China, in February, 2003. Lancet. 2003;362:1353–1358. doi: 10.1016/S0140-6736(03)14630-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Gerberding JL. Influenza in 2009. Jama. 2009;302:1907–1908,. doi: 10.1001/jama.2009.1617. [DOI] [PubMed] [Google Scholar]
- 175.Zumla A, Hui DS, Perlman S. Middle East respiratory syndrome. Lancet. 2015;386:995–1007. doi: 10.1016/S0140-6736(15)60454-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Malvy D, et al. Ebola virus disease. Lancet. 2019;393:936–948. doi: 10.1016/S0140-6736(18)33132-5. [DOI] [PubMed] [Google Scholar]
- 177.Pacheco O, et al. Zika virus disease in colombia—preliminary report. N. Engl. J. Med. 2020;383:e44. doi: 10.1056/NEJMoa1604037. [DOI] [PubMed] [Google Scholar]
- 178.Hu B, Guo H, Zhou P, Shi Z-L. Characteristics of SARS-CoV-2 and COVID-19. Nat. Rev. Microbiol. 2020;19:141–154. doi: 10.1038/s41579-020-00459-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Baker RE, et al. Infectious disease in an era of global change. Nat. Rev. Microbiol. 2021;20:193–205. doi: 10.1038/s41579-021-00639-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Excler J-L, Saville M, Berkley S, Kim JH. Vaccine development for emerging infectious diseases. Nat. Med. 2021;27:591–600. doi: 10.1038/s41591-021-01301-0. [DOI] [PubMed] [Google Scholar]
- 181.Graham BS, Sullivan NJ. Emerging viral diseases from a vaccinology perspective: preparing for the next pandemic. Nat. Immunol. 2017;19:20–28. doi: 10.1038/s41590-017-0007-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Gu W, Miller S, Chiu CY. Clinical metagenomic next-generation sequencing for pathogen detection. Annu. Rev. Pathol. 2019;14:319–338. doi: 10.1146/annurev-pathmechdis-012418-012751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Goharshadi EK, Goharshadi K, Moghayedi M. The use of nanotechnology in the fight against viruses: a critical review. Coord. Chem. Rev. 2022;464:214559. doi: 10.1016/j.ccr.2022.214559. [DOI] [Google Scholar]
- 184.Ren H, et al. Adjuvant physiochemistry and advanced nanotechnology for vaccine development. Chem. Soc. Rev. 2023;52:5172–5254. doi: 10.1039/D2CS00848C. [DOI] [PubMed] [Google Scholar]
- 185.Akinc A, et al. The Onpattro story and the clinical translation of nanomedicines containing nucleic acid-based drugs. Nat. Nanotechnol. 2019;14:1084–1087. doi: 10.1038/s41565-019-0591-y. [DOI] [PubMed] [Google Scholar]
- 186.Blanco E, Shen H, Ferrari M. Principles of nanoparticle design for overcoming biological barriers to drug delivery. Nat. Biotechnol. 2015;33:941–951. doi: 10.1038/nbt.3330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Mitchell MJ, et al. Engineering precision nanoparticles for drug delivery. Nat. Rev. Drug Discov. 2020;20:101–124. doi: 10.1038/s41573-020-0090-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Singh L, et al. The role of nanotechnology in the treatment of viral infections. Ther. Adv. Infect. Dis. 2017;4:105–131. doi: 10.1177/2049936117713593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Feng C, et al. Emerging vaccine nanotechnology: from defense against infection to sniping cancer. Acta Pharm. Sin. B. 2022;12:2206–2223. doi: 10.1016/j.apsb.2021.12.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Xu Z, et al. In vivo assembly of nanoparticles achieved through synergy of structure‐based protein engineering and synthetic DNA generates enhanced adaptive immunity. Adv. Sci. 2020;7:1902802. doi: 10.1002/advs.201902802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Park KS, Sun X, Aikins ME, Moon JJ. Non-viral COVID-19 vaccine delivery systems. Adv. Drug Deliv. Rev. 2021;169:137–151. doi: 10.1016/j.addr.2020.12.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Islam MA, et al. Adjuvant-pulsed mRNA vaccine nanoparticle for immunoprophylactic and therapeutic tumor suppression in mice. Biomaterials. 2021;266:120431. doi: 10.1016/j.biomaterials.2020.120431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Pardi N, Hogan MJ, Porter FW, Weissman D. mRNA vaccines—a new era in vaccinology. Nat. Rev. Drug Discov. 2018;17:261–279. doi: 10.1038/nrd.2017.243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Maugeri M, et al. Linkage between endosomal escape of LNP-mRNA and loading into EVs for transport to other cells. Nat. Commun. 2019;10:4333. doi: 10.1038/s41467-019-12275-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Zhang Y-Q, et al. Ionizable drug delivery systems for efficient and selective gene therapy. Mil. Med. Res. 2023;10:9. doi: 10.1186/s40779-023-00445-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196.Khurana A, et al. Role of nanotechnology behind the success of mRNA vaccines for COVID-19. Nano Today. 2021;38:101142. doi: 10.1016/j.nantod.2021.101142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Zagorski K, et al. Modular nanoarray vaccine for SARS-CoV-2. Nanomedicine. 2022;46:102604. doi: 10.1016/j.nano.2022.102604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Hoffmann MAG, et al. ESCRT recruitment to SARS-CoV-2 spike induces virus-like particles that improve mRNA vaccines. Cell. 2023;186:2380–2391.e2389. doi: 10.1016/j.cell.2023.04.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Atmar RL, et al. Homologous and heterologous Covid-19 booster vaccinations. N. Engl. J. Med. 2022;386:1046–1057. doi: 10.1056/NEJMoa2116414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.Jain S, Venkataraman A, Wechsler ME, Peppas NA. Messenger RNA-based vaccines: Past, present, and future directions in the context of the COVID-19 pandemic. Adv. Drug Deliv. Rev. 2021;179:114000. doi: 10.1016/j.addr.2021.114000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Espeseth AS, et al. Modified mRNA/lipid nanoparticle-based vaccines expressing respiratory syncytial virus F protein variants are immunogenic and protective in rodent models of RSV infection. NPJ Vaccines. 2020;5:16. doi: 10.1038/s41541-020-0163-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.Valentin A, et al. Comparative immunogenicity of an mRNA/LNP and a DNA vaccine targeting HIV gag conserved elements in macaques. Front. Immunol. 2022;13:945706. doi: 10.3389/fimmu.2022.945706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Peng L, et al. Multiplexed LNP-mRNA vaccination against pathogenic coronavirus species. Cell Rep. 2022;40:111160. doi: 10.1016/j.celrep.2022.111160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Kanekiyo M, et al. Self-assembling influenza nanoparticle vaccines elicit broadly neutralizing H1N1 antibodies. Nature. 2013;499:102–106. doi: 10.1038/nature12202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Frietze KM, Peabody DS, Chackerian B. Engineering virus-like particles as vaccine platforms. Curr. Opin. Virol. 2016;18:44–49. doi: 10.1016/j.coviro.2016.03.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Tian J-H, et al. SARS-CoV-2 spike glycoprotein vaccine candidate NVX-CoV2373 immunogenicity in baboons and protection in mice. Nat. Commun. 2021;12:372. doi: 10.1038/s41467-020-20653-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Dunkle LM, et al. Efficacy and safety of NVX-CoV2373 in adults in the United States and Mexico. N. Engl. J. Med. 2022;386:531–543. doi: 10.1056/NEJMoa2116185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208.He C, et al. A self-assembled trimeric protein vaccine induces protective immunity against Omicron variant. Nat. Commun. 2022;13:5459. doi: 10.1038/s41467-022-33209-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.Mobini S, et al. Computational design of a novel VLP-based vaccine for hepatitis B virus. Front. Immunol. 2020;11:2074. doi: 10.3389/fimmu.2020.02074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Bei L, et al. Immunogenicity correlation in cynomolgus monkeys between Luminex‐based total IgG immunoassay and pseudovirion‐based neutralization assay for a 14‐valent recombinant human papillomavirus vaccine. J. Med. Virol. 2022;94:3946–3955. doi: 10.1002/jmv.27763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211.Hui X, et al. PSMD12-mediated M1 ubiquitination of influenza A virus at K102 regulates viral replication. J. Virol. 2022;96:e0078622. doi: 10.1128/jvi.00786-22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 212.Shinde V, et al. Comparison of the safety and immunogenicity of a novel Matrix-M-adjuvanted nanoparticle influenza vaccine with a quadrivalent seasonal influenza vaccine in older adults: a phase 3 randomised controlled trial. Lancet Infect. Dis. 2022;22:73–84. doi: 10.1016/S1473-3099(21)00192-4. [DOI] [PubMed] [Google Scholar]
- 213.McComb RC, et al. Presentation of peptides from Bacillus anthracis protective antigen on Tobacco Mosaic Virus as an epitope targeted anthrax vaccine. Vaccine. 2015;33:6745–6751. doi: 10.1016/j.vaccine.2015.10.075. [DOI] [PubMed] [Google Scholar]
- 214.August A, et al. A Phase 2 randomized, observer-blind, placebo-controlled, dose-ranging trial of aluminum-adjuvanted respiratory syncytial virus F particle vaccine formulations in healthy women of childbearing age. Vaccine. 2017;35:3749–3759. doi: 10.1016/j.vaccine.2017.05.045. [DOI] [PubMed] [Google Scholar]
- 215.Kang Y-F, et al. Rapid development of SARS-CoV-2 spike protein receptor-binding domain self-assembled nanoparticle vaccine candidates. ACS Nano. 2021;15:2738–2752. doi: 10.1021/acsnano.0c08379. [DOI] [PubMed] [Google Scholar]
- 216.Houser KV, et al. Safety and immunogenicity of a ferritin nanoparticle H2 influenza vaccine in healthy adults: a phase 1 trial. Nat. Med. 2022;28:383–391. doi: 10.1038/s41591-021-01660-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 217.Kim MY, et al. Molecular engineering and plant expression of an immunoglobulin heavy chain scaffold for delivery of a dengue vaccine candidate. Plant Biotechnol. J. 2017;15:1590–1601. doi: 10.1111/pbi.12741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 218.Wen Y, Waltman A, Han H, Collier JH. Switching the immunogenicity of peptide assemblies using surface properties. ACS Nano. 2016;10:9274–9286. doi: 10.1021/acsnano.6b03409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 219.Walls AC, et al. Elicitation of potent neutralizing antibody responses by designed protein nanoparticle vaccines for SARS-CoV-2. Cell. 2020;183:1367–1382.e1317. doi: 10.1016/j.cell.2020.10.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 220.Arunachalam PS, et al. Adjuvanting a subunit COVID-19 vaccine to induce protective immunity. Nature. 2021;594:253–258. doi: 10.1038/s41586-021-03530-2. [DOI] [PubMed] [Google Scholar]
- 221.Brouwer PJM, et al. Lassa virus glycoprotein nanoparticles elicit neutralizing antibody responses and protection. Cell Host Microbe. 2022;30:1759–1772.e1712. doi: 10.1016/j.chom.2022.10.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 222.Brouwer PJM, et al. Enhancing and shaping the immunogenicity of native-like HIV-1 envelope trimers with a two-component protein nanoparticle. Nat. Commun. 2019;10:4272. doi: 10.1038/s41467-019-12080-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 223.Sliepen K, et al. Induction of cross-neutralizing antibodies by a permuted hepatitis C virus glycoprotein nanoparticle vaccine candidate. Nat. Commun. 2022;13:7271. doi: 10.1038/s41467-022-34961-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 224.Lacasta A, et al. Design and immunological evaluation of two-component protein nanoparticle vaccines for East Coast fever. Front. Immunol. 2023;13:1015840. doi: 10.3389/fimmu.2022.1015840. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 225.Santos P, Almeida F. Exosome-based vaccines: history, current state, and clinical trials. Front. Immunol. 2021;12:711565. doi: 10.3389/fimmu.2021.711565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 226.Popowski KD, et al. Inhalable dry powder mRNA vaccines based on extracellular vesicles. Matter. 2022;5:2960–2974. doi: 10.1016/j.matt.2022.06.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 227.Zhang Q, et al. Intraduodenal delivery of exosome-loaded SARS-CoV-2 RBD mRNA induces a neutralizing antibody response in mice. Vaccines. 2023;11:673. doi: 10.3390/vaccines11030673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 228.Cacciottolo M, et al. Exosome-based multivalent vaccine: achieving potent immunization, broadened reactivity, and strong T-cell responses with nanograms of proteins. Microbiol. Spectr. 2023;11:e0050323. doi: 10.1128/spectrum.00503-23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 229.Wang R, et al. Novel exosome-targeted T-cell-based vaccine counteracts T-cell anergy and converts CTL exhaustion in chronic infection via CD40L signaling through the mTORC1 pathway. Cell. Mol. Immunol. 2016;14:529–545. doi: 10.1038/cmi.2016.23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 230.Jesus S, Soares E, Cruz MT, Borges O. Exosomes as adjuvants for the recombinant hepatitis B antigen: First report. Eur. J. Pharm. Biopharm. 2018;133:1–11. doi: 10.1016/j.ejpb.2018.09.029. [DOI] [PubMed] [Google Scholar]
- 231.Qian X, et al. Exosomal microRNAs derived from umbilical mesenchymal stem cells inhibit hepatitis C virus infection. Stem Cells Transl. Med. 2016;5:1190–1203. doi: 10.5966/sctm.2015-0348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 232.Carvalho AL, et al. Bioengineering commensal bacteria‐derived outer membrane vesicles for delivery of biologics to the gastrointestinal and respiratory tract. J. Extracell. Vesicles. 2019;8:1632100. doi: 10.1080/20013078.2019.1632100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 233.Wang J, et al. Exosome-mediated delivery of inducible miR-423-5p enhances resistance of MRC-5 cells to rabies virus infection. Int. J. Mol. Sci. 2019;20:1537. doi: 10.3390/ijms20071537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 234.Ma M, et al. Quasi-amorphous and hierarchical Fe2O3 supraparticles: active T1-weighted magnetic resonance imaging in vivo and renal clearance. ACS Nano. 2020;14:4036–4044. doi: 10.1021/acsnano.9b08570. [DOI] [PubMed] [Google Scholar]
- 235.Kulkarni JA, et al. Lipid nanoparticle technology for clinical translation of siRNA therapeutics. Acc. Chem. Res. 2019;52:2435–2444. doi: 10.1021/acs.accounts.9b00368. [DOI] [PubMed] [Google Scholar]
- 236.Zeng L, et al. Broad-spectrum CRISPR-mediated inhibition of SARS-CoV-2 variants and endemic coronaviruses in vitro. Nat. Commun. 2022;13:2766. doi: 10.1038/s41467-022-30546-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 237.Verma A, et al. Surface-structure-regulated cell-membrane penetration by monolayer-protected nanoparticles. Nat. Mater. 2008;7:588–595. doi: 10.1038/nmat2202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 238.Xin X, et al. Rod‐shaped active drug particles enable efficient and safe gene delivery. Adv. Sci. 2017;4:1700324. doi: 10.1002/advs.201700324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 239.Cullis J, et al. Macropinocytosis of Nab-paclitaxel drives macrophage activation in pancreatic cancer. Cancer Immunol. Res. 2017;5:182–190. doi: 10.1158/2326-6066.CIR-16-0125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 240.Donahue ND, Acar H, Wilhelm S. Concepts of nanoparticle cellular uptake, intracellular trafficking, and kinetics in nanomedicine. Adv. Drug Deliv. Rev. 2019;143:68–96. doi: 10.1016/j.addr.2019.04.008. [DOI] [PubMed] [Google Scholar]
- 241.Lara S, et al. Identification of receptor binding to the biomolecular corona of nanoparticles. ACS Nano. 2017;11:1884–1893. doi: 10.1021/acsnano.6b07933. [DOI] [PubMed] [Google Scholar]
- 242.Uva M, et al. On the mechanism of drug release from polysaccharide hydrogels cross-linked with magnetite nanoparticles by applying alternating magnetic fields: the case of DOXO delivery. Gels. 2015;1:24–43. doi: 10.3390/gels1010024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 243.Chen Y, Feng X. Gold nanoparticles for skin drug delivery. Int. J. Pharm. 2022;625:122122. doi: 10.1016/j.ijpharm.2022.122122. [DOI] [PubMed] [Google Scholar]
- 244.Ghorbani, M. & Hamishehkar, H. Decoration of gold nanoparticles with thiolated pH-responsive polymeric (PEG-b-p(2-dimethylamio ethyl methacrylate-co-itaconic acid) shell: a novel platform for targeting of anticancer agent. Mater. Sci. Eng. C 81, 561–570 (2017). [DOI] [PubMed]
- 245.Dadfar SM, et al. Iron oxide nanoparticles: diagnostic, therapeutic and theranostic applications. Adv. Drug Deliv. Rev. 2019;138:302–325. doi: 10.1016/j.addr.2019.01.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 246.Vaughan HJ, Green JJ, Tzeng SY. Cancer‐targeting nanoparticles for combinatorial nucleic acid delivery. Adv. Mater. 2019;32:e1901081. doi: 10.1002/adma.201901081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 247.Islam Y, et al. Physiological and pathological factors affecting drug delivery to the brain by nanoparticles. Adv. Sci. 2021;8:e2002085. doi: 10.1002/advs.202002085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 248.Li Y, et al. Co‐delivery of precisely prescribed multi‐prodrug combination by an engineered nanocarrier enables efficient individualized cancer chemotherapy. Adv. Mater. 2022;34:e2110490. doi: 10.1002/adma.202110490. [DOI] [PubMed] [Google Scholar]
- 249.Shapira S, et al. A novel platform for attenuating immune hyperactivity using EXO‐CD24 in COVID‐19 and beyond. EMBO Mol. Med. 2022;14:e15997. doi: 10.15252/emmm.202215997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 250.Zhu Y-G, et al. Nebulized exosomes derived from allogenic adipose tissue mesenchymal stromal cells in patients with severe COVID-19: a pilot study. Stem Cell Res. Ther. 2022;13:220. doi: 10.1186/s13287-022-02900-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 251.Zou X, et al. EVs containing host restriction factor IFITM3 inhibited ZIKV infection of fetuses in pregnant mice through trans-placenta delivery. Mol. Ther. 2021;29:176–190. doi: 10.1016/j.ymthe.2020.09.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 252.Shrivastava S, et al. Exosome-mediated stable epigenetic repression of HIV-1. Nat. Commun. 2021;12:5541. doi: 10.1038/s41467-021-25839-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 253.Lawitz EJ, et al. BMS‐986263 in patients with advanced hepatic fibrosis: 36‐week results from a randomized, placebo‐controlled phase 2 trial. Hepatology. 2021;75:912–923. doi: 10.1002/hep.32181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 254.Teng Y, et al. Plant-derived exosomal microRNAs inhibit lung inflammation induced by exosomes SARS-CoV-2 Nsp12. Mol. Ther. 2021;29:2424–2440. doi: 10.1016/j.ymthe.2021.05.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 255.Zhang R, et al. sEVsRVG selectively delivers antiviral siRNA to fetus brain, inhibits ZIKV infection and mitigates ZIKV-induced microcephaly in mouse model. Mol. Ther. 2022;30:2078–2091. doi: 10.1016/j.ymthe.2021.10.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 256.Yadavalli T, Shukla D. Role of metal and metal oxide nanoparticles as diagnostic and therapeutic tools for highly prevalent viral infections. Nanomedicine. 2017;13:219–230. doi: 10.1016/j.nano.2016.08.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 257.Demchenko HO, Rusinchuk NM. Evaluation of the efficiency of interparticle interactions in nanosystems. J. Nanotechnol. 2019;2019:1–8. doi: 10.1155/2019/4270454. [DOI] [Google Scholar]
- 258.Kim J, et al. Porous gold nanoparticles for attenuating infectivity of influenza A virus. J. Nanobiotechnol. 2020;18:54. doi: 10.1186/s12951-020-00611-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 259.Jeremiah SS, et al. Potent antiviral effect of silver nanoparticles on SARS-CoV-2. Biochem. Biophys. Res. Commun. 2020;533:195–200. doi: 10.1016/j.bbrc.2020.09.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 260.Reina G, et al. Hard nanomaterials in time of viral pandemics. ACS Nano. 2020;14:9364–9388. doi: 10.1021/acsnano.0c04117. [DOI] [PubMed] [Google Scholar]
- 261.Cagno V, et al. Broad-spectrum non-toxic antiviral nanoparticles with a virucidal inhibition mechanism. Nat. Mater. 2017;17:195–203. doi: 10.1038/nmat5053. [DOI] [PubMed] [Google Scholar]
- 262.Galdiero S, et al. Silver nanoparticles as potential antiviral agents. Molecules. 2011;16:8894–8918. doi: 10.3390/molecules16108894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 263.Aref ZF, et al. Clinical, biochemical and molecular evaluations of ivermectin mucoadhesive nanosuspension nasal spray in reducing upper respiratory symptoms of mild COVID-19. Int. J. Nanomed. 2021;16:4063–4072. doi: 10.2147/IJN.S313093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 264.Du T, et al. Glutathione-capped Ag2S nanoclusters inhibit coronavirus proliferation through blockage of viral RNA synthesis and budding. ACS Appl. Mater. Interfaces. 2018;10:4369–4378. doi: 10.1021/acsami.7b13811. [DOI] [PubMed] [Google Scholar]
- 265.McGowan I, et al. Long-acting rilpivirine as potential pre-exposure prophylaxis for HIV-1 prevention (the MWRI-01 study): an open-label, phase 1, compartmental, pharmacokinetic and pharmacodynamic assessment. Lancet HIV. 2016;3:e569–e578. doi: 10.1016/S2352-3018(16)30113-8. [DOI] [PubMed] [Google Scholar]
- 266.Alavi M, Kamarasu P, McClements DJ, Moore MD. Metal and metal oxide-based antiviral nanoparticles: Properties, mechanisms of action, and applications. Adv. Colloid Interface Sci. 2022;306:102726. doi: 10.1016/j.cis.2022.102726. [DOI] [PubMed] [Google Scholar]
- 267.Hamdi M, et al. Investigating the internalization and COVID-19 antiviral computational analysis of optimized nanoscale zinc oxide. ACS Omega. 2021;6:6848–6860. doi: 10.1021/acsomega.0c06046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 268.Baram-Pinto D, et al. Inhibition of herpes simplex virus type 1 infection by silver nanoparticles capped with mercaptoethane sulfonate. Bioconjug. Chem. 2009;20:1497–1502. doi: 10.1021/bc900215b. [DOI] [PubMed] [Google Scholar]
- 269.Halder A, et al. Highly monodispersed gold nanoparticles synthesis and inhibition of herpes simplex virus infections. Mater. Sci. Eng. C. 2018;89:413–421. doi: 10.1016/j.msec.2018.04.005. [DOI] [PubMed] [Google Scholar]
- 270.Khanal M, et al. Boronic acid-modified lipid nanocapsules: a novel platform for the highly efficient inhibition of hepatitis C viral entry. Nanoscale. 2015;7:1392–1402. doi: 10.1039/C4NR03875D. [DOI] [PubMed] [Google Scholar]
- 271.Joonaki E, Hassanpouryouzband A, Heldt CL, Areo O. Surface chemistry can unlock drivers of surface stability of SARS-CoV-2 in a variety of environmental conditions. Chem. 2020;6:2135–2146. doi: 10.1016/j.chempr.2020.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 272.Mosafer J, et al. Preparation, characterization and in vivo evaluation of alginate-coated chitosan and trimethylchitosan nanoparticles loaded with PR8 influenza virus for nasal immunization. Asian J. Pharm. 2019;14:216–221. doi: 10.1016/j.ajps.2018.04.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 273.Liu H, et al. Preclinical evaluation of the saponin derivative GPI-0100 as an immunostimulating and dose-sparing adjuvant for pandemic influenza vaccines. Vaccine. 2011;29:2037–2043. doi: 10.1016/j.vaccine.2011.01.012. [DOI] [PubMed] [Google Scholar]
- 274.Abbott TR, et al. Development of CRISPR as an antiviral strategy to combat SARS-CoV-2 and influenza. Cell. 2020;181:865–876. doi: 10.1016/j.cell.2020.04.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 275.Montague SJ, et al. Platelet activation by charged ligands and nanoparticles: platelet glycoprotein receptors as pattern recognition receptors. Platelets. 2021;32:1018–1030. doi: 10.1080/09537104.2021.1945571. [DOI] [PubMed] [Google Scholar]
- 276.Zhang H, et al. Delivery of mRNA vaccine with a lipid-like material potentiates antitumor efficacy through Toll-like receptor 4 signaling. Proc. Natl Acad. Sci. USA. 2021;118:e2005191118. doi: 10.1073/pnas.2005191118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 277.Yazdi AS, et al. Nanoparticles activate the NLR pyrin domain containing 3 (Nlrp3) inflammasome and cause pulmonary inflammation through release of IL-1α and IL-1β. Proc. Natl Acad. Sci. USA. 2010;107:19449–19454. doi: 10.1073/pnas.1008155107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 278.Du T, et al. Carbon dots as inhibitors of virus by activation of type I interferon response. Carbon. 2016;110:278–285. doi: 10.1016/j.carbon.2016.09.032. [DOI] [Google Scholar]
- 279.Dong X, et al. Carbon dots as potent antimicrobial agents. Theranostics. 2020;10:671–686. doi: 10.7150/thno.39863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 280.Barras A, et al. High efficiency of functional carbon nanodots as entry inhibitors of herpes simplex virus type 1. ACS Appl. Mater. Interfaces. 2016;8:9004–9013. doi: 10.1021/acsami.6b01681. [DOI] [PubMed] [Google Scholar]
- 281.Wang J, et al. Pulmonary surfactant–biomimetic nanoparticles potentiate heterosubtypic influenza immunity. Science. 2020;367:eaau0810. doi: 10.1126/science.aau0810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 282.Tong T, et al. Glycyrrhizic‐acid‐based carbon dots with high antiviral activity by multisite inhibition mechanisms. Small. 2020;16:e1906206. doi: 10.1002/smll.201906206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 283.Li Y, et al. Silver nanoparticle based codelivery of oseltamivir to inhibit the activity of the H1N1 influenza virus through ROS-mediated signaling pathways. ACS Appl. Mater. Interfaces. 2016;8:24385–24393. doi: 10.1021/acsami.6b06613. [DOI] [PubMed] [Google Scholar]
- 284.Kang J, Tahir A, Wang H, Chang J. Applications of nanotechnology in virus detection, tracking, and infection mechanisms. Wiley Interdiscip. Rev.-Nanomed. Nanobiotechnol. 2021;13:e1700. doi: 10.1002/wnan.1700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 285.Cheng R, et al. “Hepatitis virus indicator”––the simultaneous detection of hepatitis B and hepatitis C viruses based on the automatic particle enumeration. Biosens. Bioelectron. 2022;202:114001. doi: 10.1016/j.bios.2022.114001. [DOI] [PubMed] [Google Scholar]
- 286.Chen H, et al. SERS imaging-based aptasensor for ultrasensitive and reproducible detection of influenza virus A. Biosens. Bioelectron. 2020;167:112496. doi: 10.1016/j.bios.2020.112496. [DOI] [PubMed] [Google Scholar]
- 287.Hou J, et al. Rapid and reliable ultrasensitive detection of pathogenic H9N2 viruses through virus-binding phage nanofibers decorated with gold nanoparticles. Biosens. Bioelectron. 2023;237:115423. doi: 10.1016/j.bios.2023.115423. [DOI] [PubMed] [Google Scholar]
- 288.Ganganboina AB, et al. Hollow magnetic-fluorescent nanoparticles for dual-modality virus detection. Biosens. Bioelectron. 2020;170:112680. doi: 10.1016/j.bios.2020.112680. [DOI] [PubMed] [Google Scholar]
- 289.Ruiz‐Hitzky E, et al. Nanotechnology responses to COVID‐19. Adv. Healthc. 2020;9:e2000979. doi: 10.1002/adhm.202000979. [DOI] [PubMed] [Google Scholar]
- 290.Draz MS, et al. Motion-based immunological detection of Zika virus using Pt-nanomotors and a cellphone. ACS Nano. 2018;12:5709–5718. doi: 10.1021/acsnano.8b01515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 291.Behrouzi K, Lin L. Gold nanoparticle based plasmonic sensing for the detection of SARS-CoV-2 nucleocapsid proteins. Biosens. Bioelectron. 2022;195:113669. doi: 10.1016/j.bios.2021.113669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 292.Ali MA, et al. Ultrarapid and ultrasensitive detection of SARS‐CoV‐2 antibodies in COVID‐19 patients via a 3D‐printed nanomaterial‐based biosensing platform. J. Med. Virol. 2022;94:5808–5826. doi: 10.1002/jmv.28075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 293.Vermisoglou E, et al. Human virus detection with graphene-based materials. Biosens. Bioelectron. 2020;166:112436. doi: 10.1016/j.bios.2020.112436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 294.Grinyte R, et al. Microbead QD-ELISA: microbead ELISA using biocatalytic formation of quantum dots for ultra high sensitive optical and electrochemical detection. ACS Appl. Mater. Interfaces. 2016;8:29252–29260. doi: 10.1021/acsami.6b08362. [DOI] [PubMed] [Google Scholar]
- 295.Kurdekar A, et al. Comparative performance evaluation of carbon dot-based paper immunoassay on Whatman filter paper and nitrocellulose paper in the detection of HIV infection. Microfluid Nanofluidics. 2016;20:99. doi: 10.1007/s10404-016-1763-9. [DOI] [Google Scholar]
- 296.Ye L, et al. Rapid colloidal gold immunochromatographic assay for the detection of SARS-CoV-2 total antibodies after vaccination. J. Mater. Chem. B. 2022;10:1786–1794. doi: 10.1039/D1TB02521J. [DOI] [PubMed] [Google Scholar]
- 297.Sibai M, et al. Evaluation of SARS-CoV-2 total antibody detection via a lateral flow nanoparticle fluorescence immunoassay. J. Clin. Virol. 2021;139:104818. doi: 10.1016/j.jcv.2021.104818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 298.Wang M, Zhang R, Li J. CRISPR/cas systems redefine nucleic acid detection: Principles and methods. Biosens. Bioelectron. 2020;165:112430. doi: 10.1016/j.bios.2020.112430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 299.Fozouni P, et al. Amplification-free detection of SARS-CoV-2 with CRISPR-Cas13a and mobile phone microscopy. Cell. 2021;184:323–333.e329. doi: 10.1016/j.cell.2020.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 300.Ding X, et al. Ultrasensitive and visual detection of SARS-CoV-2 using all-in-one dual CRISPR-Cas12a assay. Nat. Commun. 2020;11:4711. doi: 10.1038/s41467-020-18575-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 301.van Beljouw SPB, Sanders J, Rodríguez-Molina A, Brouns SJJ. RNA-targeting CRISPR–Cas systems. Nat. Rev. Microbiol. 2022;21:21–34. doi: 10.1038/s41579-022-00793-y. [DOI] [PubMed] [Google Scholar]
- 302.Kaminski MM, et al. CRISPR-based diagnostics. Nat. Biomed. Eng. 2021;5:643–656. doi: 10.1038/s41551-021-00760-7. [DOI] [PubMed] [Google Scholar]
- 303.Ackerman CM, et al. Massively multiplexed nucleic acid detection with Cas13. Nature. 2020;582:277–282. doi: 10.1038/s41586-020-2279-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 304.Lu S, et al. Fast and sensitive detection of SARS-CoV-2 RNA using suboptimal protospacer adjacent motifs for Cas12a. Nat. Biomed. Eng. 2022;6:286–297. doi: 10.1038/s41551-022-00861-x. [DOI] [PubMed] [Google Scholar]
- 305.Freije CA, et al. Programmable inhibition and detection of RNA viruses using Cas13. Mol. Cell. 2019;76:826–837.e811. doi: 10.1016/j.molcel.2019.09.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 306.Tian Y, et al. CRISPR/Cas13a-assisted rapid and portable HBV DNA detection for low-level viremia patients. Emerg. Microbes Infect. 2023;12:e2177088. doi: 10.1080/22221751.2023.2177088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 307.Freije CA, Sabeti PC. Detect and destroy: CRISPR-based technologies for the response against viruses. Cell Host Microbe. 2021;29:689–703. doi: 10.1016/j.chom.2021.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 308.Broughton JP, et al. CRISPR–Cas12-based detection of SARS-CoV-2. Nat. Biotechnol. 2020;38:870–874. doi: 10.1038/s41587-020-0513-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 309.Patchsung M, et al. Clinical validation of a Cas13-based assay for the detection of SARS-CoV-2 RNA. Nat. Biomed. Eng. 2020;4:1140–1149. doi: 10.1038/s41551-020-00603-x. [DOI] [PubMed] [Google Scholar]
- 310.Zhang J, et al. The construction of a novel nucleic acids detection microplatform based on the NSET for one-step detecting TK1-DNA and microRNA-21. Biosens. Bioelectron. 2017;97:26–33. doi: 10.1016/j.bios.2017.05.039. [DOI] [PubMed] [Google Scholar]
- 311.Moitra P, et al. Selective naked-eye detection of SARS-CoV-2 mediated by N GEne Targeted Antisense Oligonucleotide Capped Plasmonic Nanoparticles. ACS Nano. 2020;14:7617–7627. doi: 10.1021/acsnano.0c03822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 312.Layqah LA, Eissa S. An electrochemical immunosensor for the corona virus associated with the Middle East respiratory syndrome using an array of gold nanoparticle-modified carbon electrodes. Mikrochim Acta. 2019;186:224. doi: 10.1007/s00604-019-3345-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 313.Muhammad W, Zhai Z, Gao C. Antiviral activity of nanomaterials against coronaviruses. Macromol. Biosci. 2020;20:e2000196. doi: 10.1002/mabi.202000196. [DOI] [PubMed] [Google Scholar]
- 314.Zhao Q, et al. A metal chelator as a plasmonic signal‐generation superregulator for ultrasensitive colorimetric bioassays of disease biomarkers. Adv. Sci. 2018;5:1800295. doi: 10.1002/advs.201800295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 315.Ahmeda SR, Nagyb É, Neethirajan S. Self-assembled star-shaped chiroplasmonic gold nanoparticles for an ultrasensitive chiro-immunosensor for viruses. RSC Adv. 2017;7:40849–40857. doi: 10.1039/C7RA07175B. [DOI] [Google Scholar]
- 316.Yuan J, et al. Artificial intelligence-assisted enumeration of ultra-small viruses with dual dark-field plasmon resonance probes. Biosens. Bioelectron. 2022;199:113893. doi: 10.1016/j.bios.2021.113893. [DOI] [PubMed] [Google Scholar]
- 317.Wen L, et al. Labeling the nucleocapsid of enveloped baculovirus with quantum dots for single-virus tracking. Biomaterials. 2014;35:2295–2301. doi: 10.1016/j.biomaterials.2013.11.069. [DOI] [PubMed] [Google Scholar]
- 318.Duval JFL, van Leeuwen HP, Norde W, Town RM. Chemodynamic features of nanoparticles: application to understanding the dynamic life cycle of SARS-CoV-2 in aerosols and aqueous biointerfacial zones. Adv. Colloid Interface Sci. 2021;290:102400. doi: 10.1016/j.cis.2021.102400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 319.Hou J, et al. Detection of a single circulating tumor cell using a genetically engineered antibody-like phage nanofiber probe. Mater. Today Adv. 2021;12:100168. doi: 10.1016/j.mtadv.2021.100168. [DOI] [Google Scholar]
- 320.Zhou X, et al. Phage-mediated counting by the naked eye of miRNA molecules at attomolar concentrations in a Petri dish. Nat. Mater. 2015;14:1058–1064. doi: 10.1038/nmat4377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 321.Gow NAR, et al. The importance of antimicrobial resistance in medical mycology. Nat. Commun. 2022;13:5352. doi: 10.1038/s41467-022-32249-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 322.Fernández J, Piano S, Bartoletti M, Wey EQ. Management of bacterial and fungal infections in cirrhosis: The MDRO challenge. J. Hepatol. 2021;75:S101–S117. doi: 10.1016/j.jhep.2020.11.010. [DOI] [PubMed] [Google Scholar]
- 323.Franck CO, et al. Biopolymer-based carriers for DNA vaccine design. Angew. Chem. Int Ed. Engl. 2021;60:13225–13243. doi: 10.1002/anie.202010282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 324.Lee J, Arun Kumar S, Jhan YY, Bishop CJ. Engineering DNA vaccines against infectious diseases. Acta Biomater. 2018;80:31–47. doi: 10.1016/j.actbio.2018.08.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 325.Wang Y, et al. mRNA vaccine: a potential therapeutic strategy. Mol. Cancer. 2021;20:33. doi: 10.1186/s12943-021-01311-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 326.Muhammad A, et al. Bacterial ghosts as carriers of protein subunit and DNA-encoded antigens for vaccine applications. Expert Rev. Vaccines. 2014;11:97–116. doi: 10.1586/erv.11.149. [DOI] [PubMed] [Google Scholar]
- 327.Micoli F, MacLennan CA. Outer membrane vesicle vaccines. Semin. Immunol. 2020;50:101433. doi: 10.1016/j.smim.2020.101433. [DOI] [PubMed] [Google Scholar]
- 328.Meza-Segura M, et al. SepA enhances Shigella invasion of epithelial cells by degrading alpha-1 antitrypsin and producing a neutrophil chemoattractant. mBio. 2021;12:e0283321. doi: 10.1128/mBio.02833-21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 329.Mellbye B, Schuster M. The sociomicrobiology of antivirulence drug resistance: a proof of concept. mBio. 2011;2:e00131–00111. doi: 10.1128/mBio.00131-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 330.Pereira VB, et al. DNA vaccines approach: from concepts to applications. World J. Vaccines. 2014;04:50–71. doi: 10.4236/wjv.2014.42008. [DOI] [Google Scholar]
- 331.Simmons JD, et al. Immunological mechanisms of human resistance to persistent Mycobacterium tuberculosis infection. Nat. Rev. Immunol. 2018;18:575–589. doi: 10.1038/s41577-018-0025-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 332.Dalirfardouei R, et al. A novel formulation of Mtb72F DNA vaccine for immunization against tuberculosis. Iran. J. Basic Med Sci. 2020;23:826–832. doi: 10.22038/ijbms.2020.41806.9881. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 333.Chatzikleanthous D, O’Hagan DT, Adamo R. Lipid-based nanoparticles for delivery of vaccine adjuvants and antigens: toward multicomponent vaccines. Mol. Pharm. 2021;18:2867–2888. doi: 10.1021/acs.molpharmaceut.1c00447. [DOI] [PubMed] [Google Scholar]
- 334.Chung YH, Cai H, Steinmetz NF. Viral nanoparticles for drug delivery, imaging, immunotherapy, and theranostic applications. Adv. Drug Deliv. Rev. 2020;156:214–235. doi: 10.1016/j.addr.2020.06.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 335.Li M, Yu D-H, Cai H. The synthetic antimicrobial peptide KLKL5KLK enhances the protection and efficacy of the combined DNA vaccine against Mycobacterium tuberculosis. DNA Cell Biol. 2008;27:405–413. doi: 10.1089/dna.2007.0693. [DOI] [PubMed] [Google Scholar]
- 336.Mikušová V, Mikuš P. Advances in chitosan-based nanoparticles for drug delivery. Int. J. Mol. Sci. 2021;22:9652. doi: 10.3390/ijms22179652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 337.Schwendener RA, Ludewig B, Cerny A, Engler O. Liposome-based vaccines. Methods Mol. Biol. 2010;605:163–175. doi: 10.1007/978-1-60327-360-2_11. [DOI] [PubMed] [Google Scholar]
- 338.Baden LR, et al. Efficacy and Safety of the mRNA-1273 SARS-CoV-2 Vaccine. N. Engl. J. Med. 2021;384:403–416. doi: 10.1056/NEJMoa2035389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 339.Lorenzi JCC, et al. Intranasal vaccination with messenger RNA as a new approach in gene therapy: Use against tuberculosis. BMC Biotechnol. 2010;10:77. doi: 10.1186/1472-6750-10-77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 340.Maruggi G, et al. Immunogenicity and protective efficacy induced by self-amplifying mRNA vaccines encoding bacterial antigens. Vaccine. 2017;35:361–368. doi: 10.1016/j.vaccine.2016.11.040. [DOI] [PubMed] [Google Scholar]
- 341.Mayer RL, et al. Immunopeptidomics-based design of mRNA vaccine formulations against Listeria monocytogenes. Nat. Commun. 2022;13:6075. doi: 10.1038/s41467-022-33721-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 342.Fan X-Y, Lowrie DB. Where are the RNA vaccines for TB? Emerg. Microbes Infect. 2021;10:1217–1218. doi: 10.1080/22221751.2021.1935328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 343.Schwechheimer C, Kuehn MJ. Outer-membrane vesicles from Gram-negative bacteria: biogenesis and functions. Nat. Rev. Microbiol. 2015;13:605–619. doi: 10.1038/nrmicro3525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 344.Tan K, Li R, Huang X, Liu Q. Outer membrane vesicles: current status and future direction of these novel vaccine adjuvants. Front. Microbiol. 2018;9:783. doi: 10.3389/fmicb.2018.00783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 345.Wang X, Thompson CD, Weidenmaier C, Lee JC. Release of Staphylococcus aureus extracellular vesicles and their application as a vaccine platform. Nat. Commun. 2018;9:1379. doi: 10.1038/s41467-018-03847-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 346.Chen L, et al. Outer membrane vesicles displaying engineered glycotopes elicit protective antibodies. Proc. Natl Acad. Sci. USA. 2016;113:E3609–E3618. doi: 10.1073/pnas.1518311113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 347.Chen DJ, et al. Delivery of foreign antigens by engineered outer membrane vesicle vaccines. Proc. Natl Acad. Sci. USA. 2010;107:3099–3104. doi: 10.1073/pnas.0805532107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 348.Camacho AI, et al. Nanoparticle-based vaccine for mucosal protection against Shigella flexneri in mice. Vaccine. 2013;31:3288–3294. doi: 10.1016/j.vaccine.2013.05.020. [DOI] [PubMed] [Google Scholar]
- 349.Gao W, et al. Modulating antibacterial immunity via bacterial membrane-coated nanoparticles. Nano Lett. 2015;15:1403–1409. doi: 10.1021/nl504798g. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 350.Dickey SW, Cheung GYC, Otto M. Different drugs for bad bugs: antivirulence strategies in the age of antibiotic resistance. Nat. Rev. Drug Discov. 2017;16:457–471. doi: 10.1038/nrd.2017.23. [DOI] [PubMed] [Google Scholar]
- 351.Angsantikul P, Fang RH, Zhang L. Toxoid vaccination against bacterial infection using cell membrane-coated nanoparticles. Bioconjug. Chem. 2017;29:604–612. doi: 10.1021/acs.bioconjchem.7b00692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 352.Hu C-MJ, Fang RH, Luk BT, Zhang L. Nanoparticle-detained toxins for safe and effective vaccination. Nat. Nanotechnol. 2013;8:933–938. doi: 10.1038/nnano.2013.254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 353.Wang F, et al. Nanoparticle‐based antivirulence vaccine for the management of methicillin‐resistant Staphylococcus aureus skin infection. Adv. Funct. Mater. 2016;26:1628–1635. doi: 10.1002/adfm.201505231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 354.Chen Y, et al. Broad‐spectrum neutralization of pore‐forming toxins with human erythrocyte membrane‐coated nanosponges. Adv. Healthc. 2018;7:e1701366. doi: 10.1002/adhm.201701366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 355.Hu C-MJ, et al. Nanoparticle biointerfacing by platelet membrane cloaking. Nature. 2015;526:118–121. doi: 10.1038/nature15373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 356.Chen W, et al. Coating nanofiber scaffolds with beta cell membrane to promote cell proliferation and function. Nanoscale. 2016;8:10364–10370. doi: 10.1039/C6NR00535G. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 357.Thamphiwatana S, et al. Macrophage-like nanoparticles concurrently absorbing endotoxins and proinflammatory cytokines for sepsis management. Proc. Natl Acad. Sci. USA. 2017;114:11488–11493. doi: 10.1073/pnas.1714267114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 358.Bruna T, Maldonado-Bravo F, Jara P, Caro N. Silver nanoparticles and their antibacterial applications. Int. J. Mol. Sci. 2021;22:7202. doi: 10.3390/ijms22137202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 359.Deshmukh SP, Patil SM, Mullani SB, Delekar SD. Silver nanoparticles as an effective disinfectant: a review. Mater. Sci. Eng. C. 2019;97:954–965. doi: 10.1016/j.msec.2018.12.102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 360.Ali S, et al. The avenue of fruit wastes to worth for synthesis of silver and gold nanoparticles and their antimicrobial application against foodborne pathogens: A review. Food Chem. 2021;359:129912. doi: 10.1016/j.foodchem.2021.129912. [DOI] [PubMed] [Google Scholar]
- 361.Wang Z, et al. Activation of biologically relevant levels of reactive oxygen species by Au/g-C3N4 hybrid nanozyme for bacteria killing and wound disinfection. Biomaterials. 2017;113:145–157. doi: 10.1016/j.biomaterials.2016.10.041. [DOI] [PubMed] [Google Scholar]
- 362.van Hengel IAJ, et al. Self-defending additively manufactured bone implants bearing silver and copper nanoparticles. J. Mater. Chem. B. 2020;8:1589–1602. doi: 10.1039/C9TB02434D. [DOI] [PubMed] [Google Scholar]
- 363.Estes LM, et al. Characterization of a nitric oxide (NO) donor molecule and cerium oxide nanoparticle (CNP) interactions and their synergistic antimicrobial potential for biomedical applications. J. Colloid Interface Sci. 2021;586:163–177. doi: 10.1016/j.jcis.2020.10.081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 364.Nguyen T-K, et al. Rational design of single-chain polymeric nanoparticles that kill planktonic and biofilm bacteria. ACS Infect. Dis. 2017;3:237–248. doi: 10.1021/acsinfecdis.6b00203. [DOI] [PubMed] [Google Scholar]
- 365.Bakhshandeh B, et al. New analytical methods using carbon-based nanomaterials for detection of Salmonella species as a major food poisoning organism in water and soil resources. Chemosphere. 2022;287:132243. doi: 10.1016/j.chemosphere.2021.132243. [DOI] [PubMed] [Google Scholar]
- 366.Wang Y, Shukla A. Bacteria-responsive biopolymer-coated nanoparticles for biofilm penetration and eradication. Biomater. Sci. 2022;10:2831–2843. doi: 10.1039/D2BM00361A. [DOI] [PubMed] [Google Scholar]
- 367.Kheiri S, Liu X, Thompson M. Nanoparticles at biointerfaces: antibacterial activity and nanotoxicology. Colloids Surf. B. 2019;184:110550. doi: 10.1016/j.colsurfb.2019.110550. [DOI] [PubMed] [Google Scholar]
- 368.Webster T, Wang Q, Larese-Casanova P. Inhibition of various gram-positive and gram-negative bacteria growth on selenium nanoparticle coated paper towels. Int. J. Nanomed. 2015;10:2885–2894. doi: 10.2147/IJN.S78466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 369.Román LE, Gomez ED, Solís JL, Gómez MM. Antibacterial cotton fabric functionalized with copper oxide nanoparticles. Molecules. 2020;25:5802. doi: 10.3390/molecules25245802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 370.Costa EM, et al. Chitosan nanoparticles as alternative anti-staphylococci agents: Bactericidal, antibiofilm and antiadhesive effects. Mater. Sci. Eng. C. 2017;79:221–226. doi: 10.1016/j.msec.2017.05.047. [DOI] [PubMed] [Google Scholar]
- 371.Gallo G, Schillaci D. Bacterial metal nanoparticles to develop new weapons against bacterial biofilms and infections. Appl. Microbiol. Biotechnol. 2021;105:5357–5366. doi: 10.1007/s00253-021-11418-4. [DOI] [PubMed] [Google Scholar]
- 372.Zhang M, et al. Antimicrobial copper ion‐graphene oxide composite to inhibit Pseudomonas Aeruginosa keratitis in rabbits. MedComm – Biomater. Appl. 2023;2:e56. [Google Scholar]
- 373.Tornero-Gutiérrez F, Ortiz-Ramírez JA, López-Romero E, Cuéllar-Cruz M. Materials used to prevent adhesion, growth, and biofilm formation of Candidaspecies. Med. Mycol. 2023;61:myad065. doi: 10.1093/mmy/myad065. [DOI] [PubMed] [Google Scholar]
- 374.Naha PC, et al. Dextran-coated iron oxide nanoparticles as biomimetic catalysts for localized and pH-activated biofilm disruption. ACS Nano. 2019;13:4960–4971. doi: 10.1021/acsnano.8b08702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 375.dos Santos CA, Ingle AP, Rai M. The emerging role of metallic nanoparticles in food. Appl. Microbiol. Biotechnol. 2020;104:2373–2383. doi: 10.1007/s00253-020-10372-x. [DOI] [PubMed] [Google Scholar]
- 376.Chlumsky O, et al. Antimicrobial properties of palladium and platinum nanoparticles: a new tool for combating food-borne pathogens. Int. J. Mol. Sci. 2021;22:7892. doi: 10.3390/ijms22157892. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 377.Putra NE, et al. Preventing antibiotic-resistant infections: additively manufactured porous Ti6Al4V biofunctionalized with Ag and Fe nanoparticles. Int. J. Mol. Sci. 2022;23:13239. doi: 10.3390/ijms232113239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 378.Brennan SA, et al. Silver nanoparticles and their orthopaedic applications. Bone Jt. J. 2015;97-B:582–589. doi: 10.1302/0301-620X.97B5.33336. [DOI] [PubMed] [Google Scholar]
- 379.Kalantari K, et al. Wound dressings functionalized with silver nanoparticles: promises and pitfalls. Nanoscale. 2020;12:2268–2291. doi: 10.1039/C9NR08234D. [DOI] [PubMed] [Google Scholar]
- 380.López-Ruiz M, et al. L-arginine-containing mesoporous silica nanoparticles embedded in dental adhesive (Arg@MSN@DAdh) for targeting cariogenic bacteria. J. Nanobiotechnol. 2022;20:502. doi: 10.1186/s12951-022-01714-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 381.Pelgrift RY, Friedman AJ. Nanotechnology as a therapeutic tool to combat microbial resistance. Adv. Drug Deliv. Rev. 2013;65:1803–1815. doi: 10.1016/j.addr.2013.07.011. [DOI] [PubMed] [Google Scholar]
- 382.Forier K, et al. Lipid and polymer nanoparticles for drug delivery to bacterial biofilms. J. Control Release. 2014;190:607–623. doi: 10.1016/j.jconrel.2014.03.055. [DOI] [PubMed] [Google Scholar]
- 383.Agreles MAA, Cavalcanti IDL, Cavalcanti IMF. Synergism between metallic nanoparticles and antibiotics. Appl. Microbiol. Biotechnol. 2022;106:3973–3984. doi: 10.1007/s00253-022-12001-1. [DOI] [PubMed] [Google Scholar]
- 384.Alavi M, Rai M. Recent advances in antibacterial applications of metal nanoparticles (MNPs) and metal nanocomposites (MNCs) against multidrug-resistant (MDR) bacteria. Expert Rev. Anti Infect. Ther. 2019;17:419–428. doi: 10.1080/14787210.2019.1614914. [DOI] [PubMed] [Google Scholar]
- 385.Zheng T, et al. Co-assembled nanocomplexes comprising epigallocatechin gallate and berberine for enhanced antibacterial activity against multidrug resistant Staphylococcus aureus. Biomed. Pharmacother. 2023;163:114856. doi: 10.1016/j.biopha.2023.114856. [DOI] [PubMed] [Google Scholar]
- 386.Alsakhawy SA, et al. Encapsulation of thymus vulgaris essential oil in caseinate/gelatin nanocomposite hydrogel: In vitro antibacterial activity and in vivo wound healing potential. Int. J. Pharm. 2022;628:122280. doi: 10.1016/j.ijpharm.2022.122280. [DOI] [PubMed] [Google Scholar]
- 387.Wu J, et al. Mucus-permeable polymyxin B-hyaluronic acid/poly (lactic-co-glycolic acid) nanoparticle platform for the nebulized treatment of lung infections. J. Colloid Interface Sci. 2022;624:307–319. doi: 10.1016/j.jcis.2022.05.121. [DOI] [PubMed] [Google Scholar]
- 388.Wu Y, Gao H, Liu J, Liang H. Chitosan nanoparticles efficiently enhance the dispersibility, stability and selective antibacterial activity of insoluble isoflavonoids. Int. J. Biol. Macromol. 2023;232:123420. doi: 10.1016/j.ijbiomac.2023.123420. [DOI] [PubMed] [Google Scholar]
- 389.Paredes da Rocha N, et al. Highly water-soluble dapsone nanocrystals: towards innovative preparations for an undermined drug. Int. J. Pharm. 2023;630:122428. doi: 10.1016/j.ijpharm.2022.122428. [DOI] [PubMed] [Google Scholar]
- 390.El-Housiny S, et al. Fluconazole-loaded solid lipid nanoparticles topical gel for treatment of pityriasis versicolor: formulation and clinical study. Drug Deliv. 2017;25:78–90. doi: 10.1080/10717544.2017.1413444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 391.Anwar SK, et al. Curcumin nanoparticles: the topical antimycotic suspension treating oral candidiasis. Odontology. 2022;111:350–359. doi: 10.1007/s10266-022-00742-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 392.Gounani Z, et al. Mesoporous silica nanoparticles carrying multiple antibiotics provide enhanced synergistic effect and improved biocompatibility. Colloids Surf. B. 2019;175:498–508. doi: 10.1016/j.colsurfb.2018.12.035. [DOI] [PubMed] [Google Scholar]
- 393.Fernández‐Campos F, et al. Evaluation of novel nystatin nanoemulsion for skin candidosis infections. Mycoses. 2012;56:70–81. doi: 10.1111/j.1439-0507.2012.02202.x. [DOI] [PubMed] [Google Scholar]
- 394.Cinteza L, et al. Chitosan-stabilized Ag nanoparticles with superior biocompatibility and their synergistic antibacterial effect in mixtures with essential oils. Nanomaterials. 2018;8:826. doi: 10.3390/nano8100826. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 395.Xu Y, Li H, Li X, Liu W. What happens when nanoparticles encounter bacterial antibiotic resistance? Sci. Total Environ. 2023;876:162856. doi: 10.1016/j.scitotenv.2023.162856. [DOI] [PubMed] [Google Scholar]
- 396.Osman N, et al. Surface modification of nano‐drug delivery systems for enhancing antibiotic delivery and activity. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2021;14:e1758. doi: 10.1002/wnan.1758. [DOI] [PubMed] [Google Scholar]
- 397.Vanamala K, et al. Folate functionalized lipid nanoparticles for targeted therapy of methicillin-resistant Staphylococcus aureus. Pharmaceutics. 2021;13:1791. doi: 10.3390/pharmaceutics13111791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 398.Muppidi K, Wang J, Betageri G, Pumerantz AS. PEGylated liposome encapsulation increases the lung tissue concentration of vancomycin. Antimicrob. Agents Chemother. 2011;55:4537–4542. doi: 10.1128/AAC.00713-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 399.Peacock SJ, et al. Induction of mouse melioidosis with meningitis by CD11b+ phagocytic cells harboring intracellular B. pseudomallei as a Trojan Horse. PLoS Negl. Trop. Dis. 2013;7:e2363. doi: 10.1371/journal.pntd.0002363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 400.Nguyen L, Pieters J. The Trojan Horse: survival tactics of pathogenic mycobacteria in macrophages. Trends Cell Biol. 2005;15:269–276. doi: 10.1016/j.tcb.2005.03.009. [DOI] [PubMed] [Google Scholar]
- 401.Zou X, et al. Natural antibacterial agent-based nanoparticles for effective treatment of intracellular MRSA infection. Acta Biomater. 2023;169:410–421. doi: 10.1016/j.actbio.2023.08.004. [DOI] [PubMed] [Google Scholar]
- 402.Li X, Chen D, Xie S. Current progress and prospects of organic nanoparticles against bacterial biofilm. Adv. Colloid Interface Sci. 2021;294:102475. doi: 10.1016/j.cis.2021.102475. [DOI] [PubMed] [Google Scholar]
- 403.Dey N, et al. Role of nanomaterials in deactivating multiple drug resistance efflux pumps – a review. Environ. Res. 2022;204:111968. doi: 10.1016/j.envres.2021.111968. [DOI] [PubMed] [Google Scholar]
- 404.Chwalibog, et al. Visualization of interaction between inorganic nanoparticles and bacteria or fungi. Int. J. Nanomed. 2010;5:1085–1094. doi: 10.2147/IJN.S13532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 405.Ye Y, et al. Cell wall destruction and internal cascade synergistic antifungal strategy for fungal keratitis. ACS Nano. 2022;16:18729–18745. doi: 10.1021/acsnano.2c07444. [DOI] [PubMed] [Google Scholar]
- 406.Radovic-Moreno AF, et al. Surface charge-switching polymeric nanoparticles for bacterial cell wall-targeted delivery of antibiotics. ACS Nano. 2012;6:4279–4287. doi: 10.1021/nn3008383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 407.Yang L, et al. pH-Responsive hyperbranched polymer nanoparticles to combat intracellular infection by disrupting bacterial wall and regulating macrophage polarization. Biomacromolecules. 2022;23:4370–4378. doi: 10.1021/acs.biomac.2c00823. [DOI] [PubMed] [Google Scholar]
- 408.Hou S, et al. Metabolic labeling mediated targeting and thermal killing of gram‐positive bacteria by self‐reporting janus magnetic nanoparticles. Small. 2020;17:e2006357. doi: 10.1002/smll.202006357. [DOI] [PubMed] [Google Scholar]
- 409.Castellarnau A, et al. Astodrimer sodium antiviral nasal spray for reducing respiratory infections is safe and well tolerated in a randomized controlled trial. Sci. Rep. 2022;12:10210. doi: 10.1038/s41598-022-14601-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 410.Mendling W, et al. A phase 2, double-blind, multicenter, randomized, placebo-controlled, dose‑ranging study of the efficacy and safety of Astodrimer Gel for the treatment of bacterial vaginosis. PloS ONE. 2020;15:e0232394. doi: 10.1371/journal.pone.0232394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 411.Thompson RN, Brooks-Pollock E. Detection, forecasting and control of infectious disease epidemics: modelling outbreaks in humans, animals and plants. Philos. Trans. R. Soc. Lond., B, Biol. Sci. 2019;374:20190038. doi: 10.1098/rstb.2019.0038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 412.Phakatkar AH, et al. In situ microscopic studies on the interaction of multi-principal element nanoparticles and bacteria. ACS Nano. 2023;17:5880–5893. doi: 10.1021/acsnano.2c12799. [DOI] [PubMed] [Google Scholar]
- 413.Linklater DP, et al. Antibacterial action of nanoparticles by lethal stretching of bacterial cell membranes. Adv. Mater. 2020;32:e2005679. doi: 10.1002/adma.202005679. [DOI] [PubMed] [Google Scholar]
- 414.Kim D, et al. Selective killing of pathogenic bacteria by antimicrobial silver nanoparticle—cell wall binding domain conjugates. ACS Appl. Mater. Interfaces. 2018;10:13317–13324. doi: 10.1021/acsami.8b00181. [DOI] [PubMed] [Google Scholar]
- 415.Khare T, et al. Embelin-loaded chitosan gold nanoparticles interact synergistically with ciprofloxacin by inhibiting efflux pumps in multidrug-resistant Pseudomonas aeruginosa and Escherichia coli. Environ. Res. 2021;199:111321. doi: 10.1016/j.envres.2021.111321. [DOI] [PubMed] [Google Scholar]
- 416.Xu Y, et al. CeO2 nanoparticles-regulated plasmid uptake and bioavailability for reducing transformation of extracellular antibiotic resistance genes. Nanomaterials. 2023;13:969. doi: 10.3390/nano13060969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 417.Ruiz-Fresneda MA, et al. Exploring antibacterial activity and bacterial-mediated allotropic transition of differentially coated selenium nanoparticles. ACS Appl. Mater. Interfaces. 2023;15:29958–29970. doi: 10.1021/acsami.3c05100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 418.Wang Z, et al. Multifunctional gold–silver–carbon quantum dots nano-hybrid composite: advancing antibacterial wound healing and cell proliferation. ACS Appl. Mater. Interfaces. 2023;15:40241–40254. doi: 10.1021/acsami.3c07625. [DOI] [PubMed] [Google Scholar]
- 419.Ezraty B, Gennaris A, Barras F, Collet J-F. Oxidative stress, protein damage and repair in bacteria. Nat. Rev. Microbiol. 2017;15:385–396. doi: 10.1038/nrmicro.2017.26. [DOI] [PubMed] [Google Scholar]
- 420.Yang D, et al. Nanozymes used for antimicrobials and their applications. Colloids Surf. B. 2020;195:111252. doi: 10.1016/j.colsurfb.2020.111252. [DOI] [PubMed] [Google Scholar]
- 421.Mo F, et al. Recent advances in nanozymes for bacteria-infected wound therapy. Int. J. Nanomed. 2022;17:5947–5990. doi: 10.2147/IJN.S382796. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 422.Gao F, et al. Surface-bound reactive oxygen species generating nanozymes for selective antibacterial action. Nat. Commun. 2021;12:745. doi: 10.1038/s41467-021-20965-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 423.Sasaninia K, et al. Topical absorption of glutathione–cyclodextrin nanoparticle complex in healthy human subjects improves immune response against Mycobacterium avium infection. Antioxidants. 2023;12:1375. doi: 10.3390/antiox12071375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 424.Huang Y, et al. Stimuli‐responsive nanoplatforms for antibacterial applications. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2022;14:e1775. doi: 10.1002/wnan.1775. [DOI] [PubMed] [Google Scholar]
- 425.Albayaty YN, et al. Enzyme responsive copolymer micelles enhance the anti-biofilm efficacy of the antiseptic chlorhexidine. Int. J. Pharm. 2019;566:329–341. doi: 10.1016/j.ijpharm.2019.05.069. [DOI] [PubMed] [Google Scholar]
- 426.Hu C-MJ, et al. A biomimetic nanosponge that absorbs pore-forming toxins. Nat. Nanotechnol. 2013;8:336–340. doi: 10.1038/nnano.2013.54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 427.Meng Y, et al. Reactive metal boride nanoparticles trap lipopolysaccharide and peptidoglycan for bacteria-infected wound healing. Nat. Commun. 2022;13:7353. doi: 10.1038/s41467-022-35050-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 428.Hajipour MJ, et al. Nanotechnology for targeted detection and removal of bacteria: opportunities and challenges. Adv. Sci. 2021;8:e2100556. doi: 10.1002/advs.202100556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 429.Yang X, et al. D-alanyl-D-alanine-modified gold nanoparticles form a broad-spectrum sensor for bacteria. Theranostics. 2018;8:1449–1457. doi: 10.7150/thno.22540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 430.Liu C, et al. Quantification of circulating Mycobacterium tuberculosis antigen peptides allows rapid diagnosis of active disease and treatment monitoring. Proc. Natl Acad. Sci. USA. 2017;114:3969–3974. doi: 10.1073/pnas.1621360114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 431.Zhao Z, et al. Bacteria-activated theranostic nanoprobes against methicillin-resistant Staphylococcus aureus infection. ACS nano. 2017;11:4428–4438. doi: 10.1021/acsnano.7b00041. [DOI] [PubMed] [Google Scholar]
- 432.Khan SA, et al. Targeted highly sensitive detection of multi-drug resistant salmonella DT104 using gold nanoparticles. ChemComm. 2011;47:9444–9446. doi: 10.1039/c1cc13199k. [DOI] [PubMed] [Google Scholar]
- 433.Naidoo R, et al. Surface-immobilization of chromatographically purified bacteriophages for the optimized capture of bacteria. Bacteriophage. 2014;2:15–24. doi: 10.4161/bact.19079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 434.Imai M, et al. Dark-field microscopic detection of bacteria using bacteriophage-immobilized SiO2@AuNP core–shell nanoparticles. Anal. Chem. 2019;91:12352–12357. doi: 10.1021/acs.analchem.9b02715. [DOI] [PubMed] [Google Scholar]
- 435.Duncan B, et al. Sensing by smell: nanoparticle–enzyme sensors for rapid and sensitive detection of bacteria with olfactory output. ACS Nano. 2017;11:5339–5343. doi: 10.1021/acsnano.7b00822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 436.Huang L, et al. Plasmonic silver nanoshells for drug and metabolite detection. Nat. Commun. 2017;8:220. doi: 10.1038/s41467-017-00220-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 437.Najer A, et al. Nanomimics of host cell membranes block invasion and expose invasive malaria parasites. ACS Nano. 2014;8:12560–12571. doi: 10.1021/nn5054206. [DOI] [PubMed] [Google Scholar]
- 438.Datoo MS, et al. Efficacy of a low-dose candidate malaria vaccine, R21 in adjuvant Matrix-M, with seasonal administration to children in Burkina Faso: a randomised controlled trial. Lancet. 2021;397:1809–1818. doi: 10.1016/S0140-6736(21)00943-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 439.Venkatraman N, et al. Safety and immunogenicity of heterologous prime-boost immunization with viral-vectored malaria vaccines adjuvanted with Matrix-M™. Vaccine. 2017;35:6208–6217. doi: 10.1016/j.vaccine.2017.09.028. [DOI] [PubMed] [Google Scholar]
- 440.Gola A, et al. Prime and target immunization protects against liver-stage malaria in mice. Sci. Transl. Med. 2018;10:eaap9128. doi: 10.1126/scitranslmed.aap9128. [DOI] [PubMed] [Google Scholar]
- 441.Kamaraj C, et al. Biosynthesis and extrinsic toxicity of copper oxide nanoparticles against cattle parasites: an eco-friendly approach. Environ. Res. 2022;214:114009. doi: 10.1016/j.envres.2022.114009. [DOI] [PubMed] [Google Scholar]
- 442.Salinas ND, et al. A potent and durable malaria transmission-blocking vaccine designed from a single-component 60-copy Pfs230D1 nanoparticle. NPJ Vaccines. 2023;8:124. doi: 10.1038/s41541-023-00709-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 443.Healy SA, et al. Pfs230 yields higher malaria transmission–blocking vaccine activity than Pfs25 in humans but not mice. J. Clin. Investig. 2021;131:e146221. doi: 10.1172/JCI146221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 444.Waris A, et al. Applications of various types of nanomaterials for the treatment of neurological disorders. Nanomaterials. 2022;12:2140. doi: 10.3390/nano12132140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 445.Murugesan C, Manivannan P, Gangatharan M. Pros and cons in prion diseases abatement: Insights from nanomedicine and transmissibility patterns. Int. J. Biol. Macromol. 2020;145:21–27. doi: 10.1016/j.ijbiomac.2019.12.150. [DOI] [PubMed] [Google Scholar]
- 446.Kumar J, et al. Detection of amyloid fibrils in Parkinson’s disease using plasmonic chirality. Proc. Natl Acad. Sci. USA. 2018;115:3225–3230. doi: 10.1073/pnas.1721690115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 447.Lasmezas CI, et al. A visual dual-aptamer logic gate for sensitive discrimination of prion diseases-associated isoform with reusable magnetic microparticles and fluorescence quantum dots. PLoS ONE. 2013;8:e53935. doi: 10.1371/journal.pone.0053935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 448.Sorokina SA, Stroylova YY, Shifrina ZB, Muronetz VI. Disruption of amyloid prion protein aggregates by cationic pyridylphenylene dendrimers. Macromol. Biosci. 2015;16:266–275. doi: 10.1002/mabi.201500268. [DOI] [PubMed] [Google Scholar]
- 449.Ganugula R, et al. A highly potent lymphatic system–targeting nanoparticle cyclosporine prevents glomerulonephritis in mouse model of lupus. Sci. Adv. 2020;6:eabb3900. doi: 10.1126/sciadv.abb3900. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 450.Abuelella KE, Abd-Allah H, Soliman SM, Abdel-Mottaleb MMA. Skin targeting by chitosan/hyaluronate hybrid nanoparticles for the management of irritant contact dermatitis: In vivo therapeutic efficiency in mouse-ear dermatitis model. Int. J. Biol. Macromol. 2023;232:123458. doi: 10.1016/j.ijbiomac.2023.123458. [DOI] [PubMed] [Google Scholar]
- 451.Wu J, et al. Rhein modulates host purine metabolism in intestine through gut microbiota and ameliorates experimental colitis. Theranostics. 2020;10:10665–10679. doi: 10.7150/thno.43528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 452.Park J, et al. Tolerogenic nanovaccine for prevention and treatment of autoimmune encephalomyelitis. Adv. Mater. 2022;35:e2202670. doi: 10.1002/adma.202202670. [DOI] [PubMed] [Google Scholar]
- 453.Wang P, et al. Hyaluronic acid-based M1 macrophage targeting and environmental responsive drug releasing nanoparticle for enhanced treatment of rheumatoid arthritis. Carbohydr. Polym. 2023;316:121018. doi: 10.1016/j.carbpol.2023.121018. [DOI] [PubMed] [Google Scholar]
- 454.Jain P, Kathuria H, Momin M. Clinical therapies and nano drug delivery systems for urinary bladder cancer. Pharmacol. Ther. 2021;226:107871. doi: 10.1016/j.pharmthera.2021.107871. [DOI] [PubMed] [Google Scholar]
- 455.Ma X, et al. Bioengineered nanogels for cancer immunotherapy. Chem. Soc. Rev. 2022;51:5136–5174. doi: 10.1039/D2CS00247G. [DOI] [PubMed] [Google Scholar]
- 456.Li S, et al. Anti-ICAM-1 antibody-modified nanostructured lipid carriers: a pulmonary vascular endothelium-targeted device for acute lung injury therapy. J. Nanobiotechnol. 2018;16:105. doi: 10.1186/s12951-018-0431-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 457.Zhu J, et al. Surface coating of pulmonary siRNA delivery vectors enabling mucus penetration, cell targeting, and intracellular radical scavenging for enhanced acute lung injury therapy. ACS Appl. Mater. Interfaces. 2022;14:5090–5100. doi: 10.1021/acsami.1c23069. [DOI] [PubMed] [Google Scholar]
- 458.Lamson NG, Berger A, Fein KC, Whitehead KA. Anionic nanoparticles enable the oral delivery of proteins by enhancing intestinal permeability. Nat. Biomed. Eng. 2019;4:84–96. doi: 10.1038/s41551-019-0465-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 459.Du X, et al. Insights into protein–ligand interactions: mechanisms, models, and methods. Int. J. Mol. Sci. 2016;17:144. doi: 10.3390/ijms17020144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 460.Tietjen GT, Bracaglia LG, Saltzman WM, Pober JS. Focus on Fundamentals: Achieving Effective Nanoparticle Targeting. Trends Mol. Med. 2018;24:598–606. doi: 10.1016/j.molmed.2018.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 461.Chandrupatla DMSH, et al. The folate receptor β as a macrophage-mediated imaging and therapeutic target in rheumatoid arthritis. Drug Deliv. Transl. Res. 2018;9:366–378. doi: 10.1007/s13346-018-0589-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 462.Lu YJ, et al. Targeting folate receptor beta on monocytes/macrophages renders rapid inflammation resolution independent of root causes. Cell Rep. Med. 2021;2:100422. doi: 10.1016/j.xcrm.2021.100422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 463.Wang Y, et al. Microvesicle-camouflaged biomimetic nanoparticles encapsulating a metal-organic framework for targeted rheumatoid arthritis therapy. J. Nanobiotechnol. 2022;20:253. doi: 10.1186/s12951-022-01447-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 464.Thomas TP, et al. Folate‐targeted nanoparticles show efficacy in the treatment of inflammatory arthritis. Arthritis Rheumatol. 2011;63:2671–2680. doi: 10.1002/art.30459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 465.Feng Y, et al. Immune-microenvironment modulatory polyurethane-hyaluronic acid hybrid hydrogel scaffolds for diabetic wound treatment. Carbohydr. Polym. 2023;320:121238. doi: 10.1016/j.carbpol.2023.121238. [DOI] [PubMed] [Google Scholar]
- 466.Lee WH, et al. Hyaluronic acid nanoparticles as a topical agent for treating psoriasis. ACS Nano. 2022;16:20057–20074. doi: 10.1021/acsnano.2c07843. [DOI] [PubMed] [Google Scholar]
- 467.Bayarsaikhan G, Bayarsaikhan D, Lee J, Lee B. Targeting scavenger receptors in inflammatory disorders and oxidative stress. Antioxidants. 2022;11:936. doi: 10.3390/antiox11050936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 468.Gong T, et al. Palmitic acid-modified bovine serum albumin nanoparticles target scavenger receptor-A on activated macrophages to treat rheumatoid arthritis. Biomaterials. 2020;258:120296. doi: 10.1016/j.biomaterials.2020.120296. [DOI] [PubMed] [Google Scholar]
- 469.Heo R, et al. Dextran sulfate nanoparticles as a theranostic nanomedicine for rheumatoid arthritis. Biomaterials. 2017;131:15–26. doi: 10.1016/j.biomaterials.2017.03.044. [DOI] [PubMed] [Google Scholar]
- 470.Park EJ, Yuki Y, Kiyono H, Shimaoka M. Structural basis of blocking integrin activation and deactivation for anti-inflammation. J. Biomed. Sci. 2015;22:51. doi: 10.1186/s12929-015-0159-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 471.Fu C, et al. Understanding the mechanical properties of Antheraea Pernyi silk—from primary structure to condensed structure of the protein. Adv. Funct. Mater. 2011;21:729–737. doi: 10.1002/adfm.201001046. [DOI] [Google Scholar]
- 472.Rodriguez-Nogales A, et al. Intestinal anti-inflammatory effects of RGD-functionalized silk fibroin nanoparticles in trinitrobenzenesulfonic acid-induced experimental colitis in rats. Int. J. Nanomed. 2016;11:5945–5958. doi: 10.2147/IJN.S116479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 473.Kuwada T, et al. Identification of an anti-integrin αvβ6 autoantibody in patients with ulcerative colitis. Gastroenterology. 2021;160:2383–2394.e2321. doi: 10.1053/j.gastro.2021.02.019. [DOI] [PubMed] [Google Scholar]
- 474.Ma Y, et al. Oral nanotherapeutics based on Antheraea pernyi silk fibroin for synergistic treatment of ulcerative colitis. Biomaterials. 2022;282:121410. doi: 10.1016/j.biomaterials.2022.121410. [DOI] [PubMed] [Google Scholar]
- 475.Deng C, et al. Targeted apoptosis of macrophages and osteoclasts in arthritic joints is effective against advanced inflammatory arthritis. Nat. Commun. 2021;12:2174. doi: 10.1038/s41467-021-22454-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 476.Liu L, et al. Secreted protein acidic and rich in cysteine mediated biomimetic delivery of methotrexate by albumin-based nanomedicines for rheumatoid arthritis therapy. ACS Nano. 2019;13:5036–5048. doi: 10.1021/acsnano.9b01710. [DOI] [PubMed] [Google Scholar]
- 477.Zhang M, et al. Edible ginger-derived nanoparticles: a novel therapeutic approach for the prevention and treatment of inflammatory bowel disease and colitis-associated cancer. Biomaterials. 2016;101:321–340. doi: 10.1016/j.biomaterials.2016.06.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 478.Ju S, et al. Grape exosome-like nanoparticles induce intestinal stem cells and protect mice from DSS-induced colitis. Mol. Ther. 2013;21:1345–1357. doi: 10.1038/mt.2013.64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 479.Zhu M-z, et al. Edible exosome-like nanoparticles from Portulaca oleracea L mitigate DSS-induced colitis via facilitating double-positive CD4+CD8+T cells expansion. J. Nanobiotechnol. 2023;21:309. doi: 10.1186/s12951-023-02065-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 480.Huang Y-F, et al. Nanoparticle-mediated IgE−receptor aggregation and signaling in RBL mast cells. J. Am. Chem. Soc. 2009;131:17328–17334. doi: 10.1021/ja907125t. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 481.Mendanha D, Vieira de Castro J, Ferreira H, Neves NM. Biomimetic and cell-based nanocarriers – new strategies for brain tumor targeting. J. Control Release. 2021;337:482–493. doi: 10.1016/j.jconrel.2021.07.047. [DOI] [PubMed] [Google Scholar]
- 482.Ma Q, et al. Calming cytokine storm in pneumonia by targeted delivery of TPCA-1 using platelet-derived extracellular vesicles. Matter. 2020;3:287–301. doi: 10.1016/j.matt.2020.05.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 483.Gao, M. et al. Erythrocyte‐membrane‐enveloped perfluorocarbon as nanoscale artificial red blood cells to relieve tumor hypoxia and enhance cancer radiotherapy. Adv. Mater. 29, 10.1002/adma.201701429 (2017). [DOI] [PubMed]
- 484.Jin K, Luo Z, Zhang B, Pang Z. Biomimetic nanoparticles for inflammation targeting. Acta Pharm. Sin. B. 2018;8:23–33. doi: 10.1016/j.apsb.2017.12.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 485.Oroojalian F, et al. Immune cell membrane‐coated biomimetic nanoparticles for targeted cancer therapy. Small. 2021;17:e2006484. doi: 10.1002/smll.202006484. [DOI] [PubMed] [Google Scholar]
- 486.Wang Y, et al. Macrophage membrane functionalized biomimetic nanoparticles for targeted anti-atherosclerosis applications. Theranostics. 2021;11:164–180. doi: 10.7150/thno.47841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 487.Sha X, et al. Pro-efferocytic macrophage membrane biomimetic nanoparticles for the synergistic treatment of atherosclerosis via competition effect. J. Nanobiotechnol. 2022;20:506. doi: 10.1186/s12951-022-01720-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 488.Li M, et al. Platelet membrane biomimetic magnetic nanocarriers for targeted delivery and in situ generation of nitric oxide in early ischemic stroke. ACS Nano. 2020;14:2024–2035. doi: 10.1021/acsnano.9b08587. [DOI] [PubMed] [Google Scholar]
- 489.Yang N, et al. Peptide-anchored neutrophil membrane-coated biomimetic nanodrug for targeted treatment of rheumatoid arthritis. J. Nanobiotechnol. 2023;21:13. doi: 10.1186/s12951-023-01773-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 490.Liu Y, et al. Neutrophil-membrane-coated biomineralized metal–organic framework nanoparticles for atherosclerosis treatment by targeting gene silencing. ACS Nano. 2023;17:7721–7732. doi: 10.1021/acsnano.3c00288. [DOI] [PubMed] [Google Scholar]
- 491.Nelson A. Engineering interactions. Nat. Mater. 2008;7:523–525. doi: 10.1038/nmat2214. [DOI] [PubMed] [Google Scholar]
- 492.Mao L, et al. Stimuli-responsive polymeric nanovaccines toward next-generation immunotherapy. ACS Nano. 2023;17:9826–9849. doi: 10.1021/acsnano.3c02273. [DOI] [PubMed] [Google Scholar]
- 493.Tan J, et al. Anti-inflammatory polymersomes of redox-responsive polyprodrug amphiphiles with inflammation-triggered indomethacin release characteristics. Biomaterials. 2018;178:608–619. doi: 10.1016/j.biomaterials.2018.03.035. [DOI] [PubMed] [Google Scholar]
- 494.Pu H-L, et al. Nanoparticles with dual responses to oxidative stress and reduced pH for drug release and anti-inflammatory applications. ACS Nano. 2014;8:1213–1221. doi: 10.1021/nn4058787. [DOI] [PubMed] [Google Scholar]
- 495.Xu Y, et al. Glycogen-based pH and redox sensitive nanoparticles with ginsenoside Rh2 for effective treatment of ulcerative colitis. Biomaterials. 2022;280:121077. doi: 10.1016/j.biomaterials.2021.121077. [DOI] [PubMed] [Google Scholar]
- 496.Li F, et al. Stimuli-responsive nano-assemblies for remotely controlled drug delivery. J. Control Release. 2020;322:566–592. doi: 10.1016/j.jconrel.2020.03.051. [DOI] [PubMed] [Google Scholar]
- 497.Ni R, et al. Reactive oxygen species-responsive dexamethasone-loaded nanoparticles for targeted treatment of rheumatoid arthritis via suppressing the iRhom2/TNF-α/BAFF signaling pathway. Biomaterials. 2020;232:119730. doi: 10.1016/j.biomaterials.2019.119730. [DOI] [PubMed] [Google Scholar]
- 498.Muhammad W, et al. ROS-responsive polymer nanoparticles with enhanced loading of dexamethasone effectively modulate the lung injury microenvironment. Acta Biomater. 2022;148:258–270. doi: 10.1016/j.actbio.2022.06.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 499.Pu Y, et al. Sono‐controllable and ROS‐sensitive CRISPR‐Cas9 genome editing for augmented/synergistic ultrasound tumor nanotherapy. Adv. Mater. 2021;33:e2104641. doi: 10.1002/adma.202104641. [DOI] [PubMed] [Google Scholar]
- 500.Shi C, et al. Trojan horse nanocapsule enabled in situ modulation of the phenotypic conversion of Th17 cells to treg cells for the treatment of multiple sclerosis in mice. Adv. Mater. 2023;35:e2210262. doi: 10.1002/adma.202210262. [DOI] [PubMed] [Google Scholar]
- 501.He M, et al. Erythrocyte-membrane-enveloped biomineralized metal–organic framework nanoparticles enable intravenous glucose-responsive insulin delivery. ACS Appl. Mater. Interfaces. 2021;13:19648–19659. doi: 10.1021/acsami.1c01943. [DOI] [PubMed] [Google Scholar]
- 502.Naeem M, et al. Colitis-targeted hybrid nanoparticles-in-microparticles system for the treatment of ulcerative colitis. Acta Biomater. 2020;116:368–382. doi: 10.1016/j.actbio.2020.09.017. [DOI] [PubMed] [Google Scholar]
- 503.Cheng Y, et al. pH-Responsive multifunctional theranostic rapamycin-loaded nanoparticles for imaging and treatment of acute ischemic stroke. ACS Appl. Mater. Interfaces. 2021;13:56909–56922. doi: 10.1021/acsami.1c16530. [DOI] [PubMed] [Google Scholar]
- 504.Nathan DM. The Diabetes control and complications trial/epidemiology of diabetes interventions and complications study at 30 years: overview. Diabetes Care. 2014;37:9–16. doi: 10.2337/dc13-2112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 505.Yang J, Cao Z. Glucose-responsive insulin release: analysis of mechanisms, formulations, and evaluation criteria. J. Control Release. 2017;263:231–239. doi: 10.1016/j.jconrel.2017.01.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 506.Zhao Y, Trewyn BG, Slowing II, Lin VSY. Mesoporous silica nanoparticle-based double drug delivery system for glucose-responsive controlled release of insulin and cyclic AMP. J. Am. Chem. Soc. 2009;131:8398–8400. doi: 10.1021/ja901831u. [DOI] [PubMed] [Google Scholar]
- 507.Chou DH-C, et al. Glucose-responsive insulin activity by covalent modification with aliphatic phenylboronic acid conjugates. Proc. Natl Acad. Sci. USA. 2015;112:2401–2406. doi: 10.1073/pnas.1424684112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 508.Zhang Y, et al. High drug-loading gold nanoclusters for responsive glucose control in type 1 diabetes. J. Nanobiotechnol. 2019;17:74. doi: 10.1186/s12951-019-0505-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 509.Ali A, et al. Glucose-responsive chitosan nanoparticle/poly(vinyl alcohol) hydrogels for sustained insulin release in vivo. ACS Appl. Mater. Interfaces. 2023;15:32240–32250. doi: 10.1021/acsami.3c05031. [DOI] [PubMed] [Google Scholar]
- 510.Zhang Y, et al. Surface-anchored microbial enzyme-responsive solid lipid nanoparticles enabling colonic budesonide release for ulcerative colitis treatment. J. Nanobiotechnol. 2023;21:145. doi: 10.1186/s12951-023-01889-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 511.Chen S-Q, et al. Chitosan-modified lipid nanodrug delivery system for the targeted and responsive treatment of ulcerative colitis. Carbohydr. Polym. 2020;230:115613. doi: 10.1016/j.carbpol.2019.115613. [DOI] [PubMed] [Google Scholar]
- 512.Luo Z, et al. Intracellular redox-activated anticancer drug delivery by functionalized hollow mesoporous silica nanoreservoirs with tumor specificity. Biomaterials. 2014;35:7951–7962. doi: 10.1016/j.biomaterials.2014.05.058. [DOI] [PubMed] [Google Scholar]
- 513.Vazzana M, et al. Tramadol hydrochloride: pharmacokinetics, pharmacodynamics, adverse side effects, co-administration of drugs and new drug delivery systems. Biomed. Pharmacother. 2015;70:234–238. doi: 10.1016/j.biopha.2015.01.022. [DOI] [PubMed] [Google Scholar]
- 514.Saito E, et al. Modulating lung immune cells by pulmonary delivery of antigen-specific nanoparticles to treat autoimmune disease. Sci. Adv. 2020;6:eabc9317. doi: 10.1126/sciadv.abc9317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 515.Zhang M, et al. Airway epithelial cell-specific delivery of lipid nanoparticles loading siRNA for asthma treatment. J. Control Release. 2022;352:422–437. doi: 10.1016/j.jconrel.2022.10.020. [DOI] [PubMed] [Google Scholar]
- 516.Khan ZA, Tripathi R, Mishra B. Methotrexate: a detailed review on drug delivery and clinical aspects. Expert Opin. Drug Deliv. 2012;9:151–169. doi: 10.1517/17425247.2012.642362. [DOI] [PubMed] [Google Scholar]
- 517.Hoes JN, Jacobs JWG, Buttgereit F, Bijlsma JWJ. Current view of glucocorticoid co-therapy with DMARDs in rheumatoid arthritis. Nat. Rev. Rheumatol. 2010;6:693–702. doi: 10.1038/nrrheum.2010.179. [DOI] [PubMed] [Google Scholar]
- 518.Jia Z, et al. Structural optimization of HPMA copolymer-based dexamethasone prodrug for improved treatment of inflammatory arthritis. J. Control Release. 2020;324:560–573. doi: 10.1016/j.jconrel.2020.05.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 519.Ren S, et al. Acupoint nanocomposite hydrogel for simulation of acupuncture and targeted delivery of triptolide against rheumatoid arthritis. J. Nanobiotechnol. 2021;19:409. doi: 10.1186/s12951-021-01157-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 520.Lorscheider M, et al. Dexamethasone palmitate nanoparticles: an efficient treatment for rheumatoid arthritis. J. Control Release. 2019;296:179–189. doi: 10.1016/j.jconrel.2019.01.015. [DOI] [PubMed] [Google Scholar]
- 521.Yun Y, Cho YW, Park K. Nanoparticles for oral delivery: targeted nanoparticles with peptidic ligands for oral protein delivery. Adv. Drug Deliv. Rev. 2013;65:822–832. doi: 10.1016/j.addr.2012.10.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 522.Wang X, et al. Oral delivery of anti-TNF antibody shielded by natural polyphenol-mediated supramolecular assembly for inflammatory bowel disease therapy. Theranostics. 2020;10:10808–10822. doi: 10.7150/thno.47601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 523.Zeng Z, et al. Stimuli-responsive self-assembled dendrimers for oral protein delivery. J. Control Release. 2019;315:206–213. doi: 10.1016/j.jconrel.2019.10.049. [DOI] [PubMed] [Google Scholar]
- 524.Mai Y, et al. Topical formulation based on disease-specific nanoparticles for single-dose cure of psoriasis. J. Control Release. 2022;349:354–366. doi: 10.1016/j.jconrel.2022.07.006. [DOI] [PubMed] [Google Scholar]
- 525.Luo L, et al. Controlled release of corticosteroid with biodegradable nanoparticles for treating experimental autoimmune uveitis. J. Control Release. 2019;296:68–80. doi: 10.1016/j.jconrel.2019.01.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 526.Moura RP, et al. Functionalized retinoic acid lipid nanocapsules promotes a two-front attack on inflammation and lack of demyelination on neurodegenerative disorders. J. Control Release. 2023;358:43–58. doi: 10.1016/j.jconrel.2023.04.034. [DOI] [PubMed] [Google Scholar]
- 527.Zhang J, et al. “Two-birds-one-stone” colon-targeted nanomedicine treats ulcerative colitis via remodeling immune microenvironment and anti-fibrosis. J. Nanobiotechnol. 2022;20:389. doi: 10.1186/s12951-022-01598-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 528.Lee Y, et al. Brain gene delivery using histidine and arginine-modified dendrimers for ischemic stroke therapy. J. Control Release. 2021;330:907–919. doi: 10.1016/j.jconrel.2020.10.064. [DOI] [PubMed] [Google Scholar]
- 529.Rachamalla HK, et al. Skin-permeable nano-lithocholic lipidoid efficiently alleviates psoriasis-like chronic skin inflammations. ACS Appl. Mater. Interfaces. 2022;14:14859–14870. doi: 10.1021/acsami.1c19180. [DOI] [PubMed] [Google Scholar]
- 530.Li M, et al. Deep eutectic solvents—Hydrogels for the topical management of rheumatoid arthritis. J. Control Release. 2023;354:664–679. doi: 10.1016/j.jconrel.2023.01.050. [DOI] [PubMed] [Google Scholar]
- 531.Martins JP, et al. Engineered multifunctional albumin‐decorated porous silicon nanoparticles for FcRn translocation of insulin. Small. 2018;14:e1800462. doi: 10.1002/smll.201800462. [DOI] [PubMed] [Google Scholar]
- 532.Sun W, et al. FcRn‐targeting and ROS‐responsive Fedratinib‐incorporated nanoparticles alleviate asthma by inducing eosinophil apoptosis. Allergy. 2022;78:1659–1663. doi: 10.1111/all.15575. [DOI] [PubMed] [Google Scholar]
- 533.Hu C-MJ, et al. Erythrocyte membrane-camouflaged polymeric nanoparticles as a biomimetic delivery platform. Proc. Natl Acad. Sci. USA. 2011;108:10980–10985. doi: 10.1073/pnas.1106634108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 534.Qin X, et al. Inflammation-responsive nanoparticles suppress lymphatic clearance for prolonged arthritis therapy. J. Control Release. 2022;352:700–711. doi: 10.1016/j.jconrel.2022.11.005. [DOI] [PubMed] [Google Scholar]
- 535.Lin Y, et al. Multifunctional nanoparticles of sinomenine hydrochloride for treat-to-target therapy of rheumatoid arthritis via modulation of proinflammatory cytokines. J. Control Release. 2022;348:42–56. doi: 10.1016/j.jconrel.2022.05.016. [DOI] [PubMed] [Google Scholar]
- 536.Zhang M-Z, et al. Inhibition of 11β–hydroxysteroid dehydrogenase type II selectively blocks the tumor COX-2 pathway and suppresses colon carcinogenesis in mice and humans. J. Clin. Investig. 2009;119:876–885. doi: 10.1172/JCI37398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 537.Ansari MM, et al. Aminocellulose-grafted-polycaprolactone coated gelatin nanoparticles alleviate inflammation in rheumatoid arthritis: a combinational therapeutic approach. Carbohydr. Polym. 2021;258:117600. doi: 10.1016/j.carbpol.2020.117600. [DOI] [PubMed] [Google Scholar]
- 538.Zhao P, et al. Nanoparticle-assembled bioadhesive coacervate coating with prolonged gastrointestinal retention for inflammatory bowel disease therapy. Nat. Commun. 2021;12:7162. doi: 10.1038/s41467-021-27463-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 539.Yang C, et al. Natural-lipid nanoparticle-based therapeutic approach to deliver 6-shogaol and its metabolites M2 and M13 to the colon to treat ulcerative colitis. J. Control Release. 2020;323:293–310. doi: 10.1016/j.jconrel.2020.04.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 540.Zhang J, et al. Engineering non-covalently assembled protein nanoparticles for long-acting gouty arthritis therapy. J. Mater. Chem. B. 2021;9:9923–9931. doi: 10.1039/D1TB01760H. [DOI] [PubMed] [Google Scholar]
- 541.Wang Q, et al. Inhibition of atherosclerosis progression by modular micelles. J. Control Release. 2023;354:294–304. doi: 10.1016/j.jconrel.2023.01.020. [DOI] [PubMed] [Google Scholar]
- 542.Dubreil C, et al. Tolerogenic iron oxide nanoparticles in type 1 diabetes: biodistribution and pharmacokinetics studies in nonobese diabetic mice. Small. 2018;14:e1802053. doi: 10.1002/smll.201802053. [DOI] [PubMed] [Google Scholar]
- 543.Ungaro R, et al. Ulcerative colitis. Lancet. 2017;389:1756–1770. doi: 10.1016/S0140-6736(16)32126-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 544.Bauché D, et al. LAG3+ regulatory T cells restrain interleukin-23-Producing CX3CR1+ Gut-resident macrophages during group 3 innate lymphoid cell-driven colitis. Immunity. 2018;49:342–352.e345. doi: 10.1016/j.immuni.2018.07.007. [DOI] [PubMed] [Google Scholar]
- 545.Matondo A, Kim SS. Targeted-mitochondria antioxidants therapeutic implications in inflammatory bowel disease. J. Drug Target. 2017;26:1–8. doi: 10.1080/1061186X.2017.1339196. [DOI] [PubMed] [Google Scholar]
- 546.Chen Y, et al. Orally deliverable sequence-targeted astaxanthin nanoparticles for colitis alleviation. Biomaterials. 2023;293:121976. doi: 10.1016/j.biomaterials.2022.121976. [DOI] [PubMed] [Google Scholar]
- 547.Xiao B, et al. TNFα gene silencing mediated by orally targeted nanoparticles combined with interleukin-22 for synergistic combination therapy of ulcerative colitis. J. Control Release. 2018;287:235–246. doi: 10.1016/j.jconrel.2018.08.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 548.Xu J, et al. A multifunctional composite hydrogel as an intrinsic and extrinsic coregulator for enhanced therapeutic efficacy for psoriasis. J. Nanobiotechnol. 2022;20:155. doi: 10.1186/s12951-022-01368-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 549.Wang Y, et al. Chitosan/hyaluronan nanogels co-delivering methotrexate and 5-aminolevulinic acid: a combined chemo-photodynamic therapy for psoriasis. Carbohydr. Polym. 2022;277:118819. doi: 10.1016/j.carbpol.2021.118819. [DOI] [PubMed] [Google Scholar]
- 550.Furtado D, et al. Overcoming the Blood–Brain Barrier: the role of nanomaterials in treating neurological diseases. Adv. Mater. 2018;30:e1801362. doi: 10.1002/adma.201801362. [DOI] [PubMed] [Google Scholar]
- 551.Wang B, et al. Targeted delivery of a STING agonist to brain tumors using bioengineered protein nanoparticles for enhanced immunotherapy. Bioact. Mater. 2022;16:232–248. doi: 10.1016/j.bioactmat.2022.02.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 552.Parvez S, et al. Dodging blood brain barrier with “nano” warriors: Novel strategy against ischemic stroke. Theranostics. 2022;12:689–719. doi: 10.7150/thno.64806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 553.Liu D, et al. Cyclosporine A loaded brain targeting nanoparticle to treat cerebral ischemia/reperfusion injury in mice. J. Nanobiotechnol. 2022;20:256. doi: 10.1186/s12951-022-01474-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 554.Mu Q, et al. Ligustrazine nanoparticle hitchhiking on neutrophils for enhanced therapy of cerebral ischemia‐reperfusion injury. Adv. Sci. 2023;10:e2301348. doi: 10.1002/advs.202301348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 555.Guo X, et al. Multifunctional selenium quantum dots for the treatment of Alzheimer’s disease by reducing Aβ-neurotoxicity and oxidative stress and alleviate neuroinflammation. ACS Appl. Mater. Interfaces. 2021;13:30261–30273. doi: 10.1021/acsami.1c00690. [DOI] [PubMed] [Google Scholar]
- 556.Yan Y, et al. Topical cationic hairy particles targeting cell free DNA in dermis enhance treatment of psoriasis. Biomaterials. 2021;276:121027. doi: 10.1016/j.biomaterials.2021.121027. [DOI] [PubMed] [Google Scholar]
- 557.Liang H, et al. Topical nanoparticles interfering with the DNA-LL37 complex to alleviate psoriatic inflammation in mice and monkeys. Sci. Adv. 2020;6:eabb5274. doi: 10.1126/sciadv.abb5274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 558.Wu C, et al. Advances in treatment strategies based on scavenging reactive oxygen species of nanoparticles for atherosclerosis. J. Nanobiotechnol. 2023;21:271. doi: 10.1186/s12951-023-02058-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 559.Gao Y, et al. Reduction of reactive oxygen species accumulation using gadolinium-doped ceria for the alleviation of atherosclerosis. ACS Appl. Mater. Interfaces. 2023;15:10414–10425. doi: 10.1021/acsami.2c20492. [DOI] [PubMed] [Google Scholar]
- 560.Yang B, Shi J. Ferrihydrite nanoparticles alleviate rheumatoid arthritis by nanocatalytic antioxidation and oxygenation. Nano Lett. 2023;23:8355–8362. doi: 10.1021/acs.nanolett.3c02743. [DOI] [PubMed] [Google Scholar]
- 561.Qiang R, et al. Carbon quantum dots derived from herbal medicine as therapeutic nanoagents for rheumatoid arthritis with ultrahigh lubrication and anti-inflammation. ACS Appl. Mater. Interfaces. 2023;15:38653–38664. doi: 10.1021/acsami.3c06188. [DOI] [PubMed] [Google Scholar]
- 562.Liu X, et al. CaP-based anti-inflammatory HIF-1α siRNA-encapsulating nanoparticle for rheumatoid arthritis therapy. J. Control Release. 2022;343:314–325. doi: 10.1016/j.jconrel.2022.01.029. [DOI] [PubMed] [Google Scholar]
- 563.Ye C, et al. Oxytocin nanogels inhibit innate inflammatory response for early intervention in Alzheimer’s disease. ACS Appl. Mater. Interfaces. 2022;14:21822–21835. doi: 10.1021/acsami.2c00007. [DOI] [PubMed] [Google Scholar]
- 564.Asadi S, et al. Beneficial effects of nano‐curcumin supplement on depression and anxiety in diabetic patients with peripheral neuropathy: A randomized, double‐blind, placebo‐controlled clinical trial. Phytother. Res. 2019;34:896–903. doi: 10.1002/ptr.6571. [DOI] [PubMed] [Google Scholar]
- 565.Lu L, et al. Targeted immunomodulation of inflammatory monocytes across the blood-brain barrier by curcumin-loaded nanoparticles delays the progression of experimental autoimmune encephalomyelitis. Biomaterials. 2020;245:119987. doi: 10.1016/j.biomaterials.2020.119987. [DOI] [PubMed] [Google Scholar]
- 566.Yunna C, Mengru H, Lei W, Weidong C. Macrophage M1/M2 polarization. Eur. J. Pharm. Biopharm. 2020;877:173090. doi: 10.1016/j.ejphar.2020.173090. [DOI] [PubMed] [Google Scholar]
- 567.Mantovani A, et al. Macrophage plasticity and polarization in tissue repair and remodelling. J. Pathol. 2012;229:176–185. doi: 10.1002/path.4133. [DOI] [PubMed] [Google Scholar]
- 568.Wang C, et al. Macrophage polarization and its role in liver disease. Front. Immunol. 2021;12:803037. doi: 10.3389/fimmu.2021.803037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 569.Qian F, et al. The transcription factor PU.1 promotes alternative macrophage polarization and asthmatic airway inflammation. J. Mol. Cell Biol. 2015;7:557–567. doi: 10.1093/jmcb/mjv042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 570.Lee YG, et al. Recruited alveolar macrophages, in response to airway epithelial–derived monocyte chemoattractant protein 1/CCL2, regulate airway inflammation and remodeling in allergic asthma. Am. J. Respir. Cell Mol. Biol. 2015;52:772–784. doi: 10.1165/rcmb.2014-0255OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 571.Zhao J, et al. M1/M2 re-polarization of kaempferol biomimetic NPs in anti-inflammatory therapy of atherosclerosis. J. Control Release. 2023;353:1068–1083. doi: 10.1016/j.jconrel.2022.12.041. [DOI] [PubMed] [Google Scholar]
- 572.Sun L, et al. Nano‐enabled reposition of proton pump inhibitors for TLR inhibition: toward a new targeted nanotherapy for acute lung injury. Adv. Sci. 2021;9:e2104051. doi: 10.1002/advs.202104051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 573.Wang L, et al. Administration route governs the therapeutic efficacy, biodistribution and macrophage targeting of anti-inflammatory nanoparticles in the lung. J. Nanobiotechnol. 2021;19:56. doi: 10.1186/s12951-021-00803-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 574.Prasad S, et al. Tolerogenic Ag-PLG nanoparticles induce tregs to suppress activated diabetogenic CD4 and CD8 T cells. J. Autoimmun. 2018;89:112–124. doi: 10.1016/j.jaut.2017.12.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 575.Kuo R, Saito E, Miller SD, Shea LD. Peptide-conjugated nanoparticles reduce positive co-stimulatory expression and T cell activity to induce tolerance. Mol. Ther. 2017;25:1676–1685. doi: 10.1016/j.ymthe.2017.03.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 576.An E-K, et al. Immunosuppressive nanoparticles containing recombinant PD-L1 and methotrexate alleviate multi-organ inflammation. Biomaterials. 2023;301:122233. doi: 10.1016/j.biomaterials.2023.122233. [DOI] [PubMed] [Google Scholar]
- 577.Guo Q, et al. Engineered PD-1/TIGIT dual-activating cell-membrane nanoparticles with dexamethasone act synergistically to shape the effector T cell/Treg balance and alleviate systemic lupus erythematosus. Biomaterials. 2022;285:121517. doi: 10.1016/j.biomaterials.2022.121517. [DOI] [PubMed] [Google Scholar]
- 578.Yang J, Sundrud MS, Skepner J, Yamagata T. Targeting Th17 cells in autoimmune diseases. Trends Pharmacol. Sci. 2014;35:493–500. doi: 10.1016/j.tips.2014.07.006. [DOI] [PubMed] [Google Scholar]
- 579.Moser T, et al. The role of TH17 cells in multiple sclerosis: therapeutic implications. Autoimmun. Rev. 2020;19:102647. doi: 10.1016/j.autrev.2020.102647. [DOI] [PubMed] [Google Scholar]
- 580.Gammon JM, et al. Controlled delivery of a metabolic modulator promotes regulatory T cells and restrains autoimmunity. J. Control Release. 2015;210:169–178. doi: 10.1016/j.jconrel.2015.05.277. [DOI] [PubMed] [Google Scholar]
- 581.Qiao H, et al. Immune-regulating strategy against rheumatoid arthritis by inducing tolerogenic dendritic cells with modified zinc peroxide nanoparticles. J. Nanobiotechnol. 2022;20:323. doi: 10.1186/s12951-022-01536-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 582.Rahiman N, et al. Recent advancements in nanoparticle-mediated approaches for restoration of multiple sclerosis. J. Control Release. 2022;343:620–644. doi: 10.1016/j.jconrel.2022.02.009. [DOI] [PubMed] [Google Scholar]
- 583.Wan H, Du H, Wang F, Dai H. Molecular imaging in the second near‐infrared window. Adv. Funct. Mater. 2019;29:1900566. doi: 10.1002/adfm.201900566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 584.Xu W, Wang D, Tang BZ. NIR‐II AIEgens: a win–win integration towards bioapplications. Angew. Chem. Int. Ed. 2020;60:7476–7487. doi: 10.1002/anie.202005899. [DOI] [PubMed] [Google Scholar]
- 585.Zeng Z, et al. A biomarker‐responsive nanosystem with colon‐targeted delivery for ulcerative colitis’s detection and treatment with optoacoustic/NIR‐II fluorescence imaging. Adv. Healthc. 2022;11:e2201544. doi: 10.1002/adhm.202201544. [DOI] [PubMed] [Google Scholar]
- 586.Yang Y, et al. Semiconducting polymer nanoparticles as theranostic system for near-infrared-II fluorescence imaging and photothermal therapy under safe laser fluence. ACS Nano. 2020;14:2509–2521. doi: 10.1021/acsnano.0c00043. [DOI] [PubMed] [Google Scholar]
- 587.Guo Y, et al. In vivo NIR‐II fluorescence lifetime imaging of whole‐body vascular using high quantum yield lanthanide‐doped nanoparticles. Small. 2023;19:e2300392. doi: 10.1002/smll.202300392. [DOI] [PubMed] [Google Scholar]
- 588.Sun C, Lee JSH, Zhang M. Magnetic nanoparticles in MR imaging and drug delivery. Adv. Drug Deliv. Rev. 2008;60:1252–1265. doi: 10.1016/j.addr.2008.03.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 589.Poon C, et al. Hybrid, metal oxide-peptide amphiphile micelles for molecular magnetic resonance imaging of atherosclerosis. J. Nanobiotechnol. 2018;16:92. doi: 10.1186/s12951-018-0420-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 590.Naha PC, et al. Dextran-coated cerium oxide nanoparticles: a computed tomography contrast agent for imaging the gastrointestinal tract and inflammatory Bowel disease. ACS Nano. 2020;14:10187–10197. doi: 10.1021/acsnano.0c03457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 591.Pang X, et al. A photoelectrochemical biosensor for fibroblast-like synoviocyte cell using visible light-activated NCQDs sensitized-ZnO/CH 3 NH 3 PbI 3 heterojunction. Biosens. Bioelectron. 2016;77:330–338. doi: 10.1016/j.bios.2015.09.047. [DOI] [PubMed] [Google Scholar]
- 592.Liu S, Shen Z, Deng L, Liu G. Smartphone assisted portable biochip for non-invasive simultaneous monitoring of glucose and insulin towards precise diagnosis of prediabetes/diabetes. Biosens. Bioelectron. 2022;209:114251. doi: 10.1016/j.bios.2022.114251. [DOI] [PubMed] [Google Scholar]
- 593.Kesharwani P, et al. Gold nanoparticles and gold nanorods in the landscape of cancer therapy. Mol. Cancer. 2023;22:98. doi: 10.1186/s12943-023-01798-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 594.Tisch U, et al. Detection of Alzheimer’s and Parkinson’s disease from exhaled breath using nanomaterial-based sensors. Nanomedicine. 2013;8:43–56. doi: 10.2217/nnm.12.105. [DOI] [PubMed] [Google Scholar]
- 595.Li X, et al. Surface chemistry governs the sub-organ transfer, clearance and toxicity of functional gold nanoparticles in the liver and kidney. J. Nanobiotechnol. 2020;18:45. doi: 10.1186/s12951-020-00599-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 596.Belal R, Gad A. Zinc oxide nanoparticles induce oxidative stress, genotoxicity, and apoptosis in the hemocytes of Bombyx mori larvae. Sci. Rep. 2023;13:3520. doi: 10.1038/s41598-023-30444-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 597.Xuan L, et al. Nanoparticles‐induced potential toxicity on human health: applications, toxicity mechanisms, and evaluation models. MedComm. 2023;4:e327. doi: 10.1002/mco2.327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 598.Xiong P, et al. Cytotoxicity of metal‐based nanoparticles: from mechanisms and methods of evaluation to pathological manifestations. Adv. Sci. 2022;9:e2106049. doi: 10.1002/advs.202106049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 599.Buchman JT, Hudson-Smith NV, Landy KM, Haynes CL. Understanding nanoparticle toxicity mechanisms to inform redesign strategies to reduce environmental impact. Acc. Chem. Res. 2019;52:1632–1642. doi: 10.1021/acs.accounts.9b00053. [DOI] [PubMed] [Google Scholar]
- 600.Bhargava A, et al. Pre-coating of protein modulate patterns of corona formation, physiological stability and cytotoxicity of silver nanoparticles. Sci. Total Environ. 2021;772:144797. doi: 10.1016/j.scitotenv.2020.144797. [DOI] [PubMed] [Google Scholar]
- 601.Sereemaspun A, Rojanathanes R, Wiwanitkit V. Effect of gold nanoparticle on renal cell: an implication for exposure risk. Ren. Fail. 2009;30:323–325. doi: 10.1080/08860220701860914. [DOI] [PubMed] [Google Scholar]
- 602.Lee P, et al. Biokinetics of subacutely co-inhaled same size gold and silver nanoparticles. Part. Fibre Toxicol. 2023;20:9. doi: 10.1186/s12989-023-00515-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 603.Kreyling WG, et al. Age-dependent rat lung deposition patterns of inhaled 20 nanometer gold nanoparticles and their quantitative biokinetics in adult rats. ACS Nano. 2018;12:7771–7790. doi: 10.1021/acsnano.8b01826. [DOI] [PubMed] [Google Scholar]
- 604.Peng C, Yu M, Zheng J. In situ ligand-directed growth of gold nanoparticles in biological tissues. Nano Lett. 2019;20:1378–1382. doi: 10.1021/acs.nanolett.9b04911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 605.Antony JJ, Sivalingam P, Chen B. Toxicological effects of silver nanoparticles. Environ. Toxicol. Pharmacol. 2015;40:729–732. doi: 10.1016/j.etap.2015.09.003. [DOI] [PubMed] [Google Scholar]
- 606.Yoisungnern T, et al. Internalization of silver nanoparticles into mouse spermatozoa results in poor fertilization and compromised embryo development. Sci. Rep. 2015;5:11170. doi: 10.1038/srep11170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 607.Donaldson K, Murphy FA, Duffin R, Poland CA. Asbestos, carbon nanotubes and the pleural mesothelium: a review and the hypothesis regarding the role of long fibre retention in the parietal pleura, inflammation and mesothelioma. Part. Fibre Toxicol. 2010;7:5. doi: 10.1186/1743-8977-7-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 608.Duke KS, et al. STAT1-dependent and -independent pulmonary allergic and fibrogenic responses in mice after exposure to tangled versus rod-like multi-walled carbon nanotubes. Part. Fibre Toxicol. 2017;14:26. doi: 10.1186/s12989-017-0207-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 609.Urankar RN, et al. Expansion of cardiac ischemia/reperfusion injury after instillation of three forms of multi-walled carbon nanotubes. Part. Fibre Toxicol. 2012;9:38. doi: 10.1186/1743-8977-9-38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 610.Young TL, et al. Pulmonary delivery of the broad-spectrum matrix metalloproteinase inhibitor marimastat diminishes multiwalled carbon nanotube-induced circulating bioactivity without reducing pulmonary inflammation. Part. Fibre Toxicol. 2021;18:34. doi: 10.1186/s12989-021-00427-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 611.Meller S, et al. Allergic sensitization to pegylated interferon‐α results in drug eruptions. Allergy. 2015;70:775–783. doi: 10.1111/all.12618. [DOI] [PubMed] [Google Scholar]
- 612.Wang M, et al. Oligomer nanoparticle release from polylactic acid plastics catalysed by gut enzymes triggers acute inflammation. Nat. Nanotechnol. 2023;18:403–411. doi: 10.1038/s41565-023-01329-y. [DOI] [PubMed] [Google Scholar]
- 613.Campbell F, et al. Directing nanoparticle biodistribution through evasion and exploitation of Stab2-dependent nanoparticle uptake. ACS Nano. 2018;12:2138–2150. doi: 10.1021/acsnano.7b06995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 614.Satapathy MK, et al. Solid lipid nanoparticles (SLNs): an advanced drug delivery system targeting brain through BBB. Pharmaceutics. 2021;13:1183. doi: 10.3390/pharmaceutics13081183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 615.Elsaesser A, Howard CV. Toxicology of nanoparticles. Adv. Drug Deliv. Rev. 2012;64:129–137. doi: 10.1016/j.addr.2011.09.001. [DOI] [PubMed] [Google Scholar]
- 616.Ginzburg VV, Balijepalli S. Modeling the thermodynamics of the interaction of nanoparticles with cell membranes. Nano Lett. 2007;7:3716–3722. doi: 10.1021/nl072053l. [DOI] [PubMed] [Google Scholar]
- 617.Uppal S, Italiya KS, Chitkara D, Mittal A. Nanoparticulate-based drug delivery systems for small molecule anti-diabetic drugs: An emerging paradigm for effective therapy. Acta Biomater. 2018;81:20–42. doi: 10.1016/j.actbio.2018.09.049. [DOI] [PubMed] [Google Scholar]
- 618.Yacobi NR, et al. Mechanisms of alveolar epithelial translocation of a defined population of nanoparticles. Am. J. Respir. Cell Mol. Biol. 2010;42:604–614. doi: 10.1165/rcmb.2009-0138OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 619.Egbuna C, et al. Toxicity of nanoparticles in biomedical application: nanotoxicology. J. Toxicol. 2021;2021:1–21. doi: 10.1155/2021/9954443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 620.Liu Y, et al. Toxicity of manufactured nanomaterials. Particuology. 2022;69:31–48. doi: 10.1016/j.partic.2021.11.007. [DOI] [Google Scholar]
- 621.Mohammadpour R, et al. Subchronic and chronic toxicity evaluation of inorganic nanoparticles for delivery applications. Adv. Drug Deliv. Rev. 2019;144:112–132. doi: 10.1016/j.addr.2019.07.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 622.Trotta F, et al. Integration of cyclodextrins and associated toxicities: a roadmap for high quality biomedical applications. Carbohydr. Polym. 2022;295:119880. doi: 10.1016/j.carbpol.2022.119880. [DOI] [PubMed] [Google Scholar]
- 623.Labouta HI, Asgarian N, Rinker K, Cramb DT. Meta-analysis of nanoparticle cytotoxicity via data-mining the literature. ACS Nano. 2019;13:1583–1594. doi: 10.1021/acsnano.8b07562. [DOI] [PubMed] [Google Scholar]
- 624.Roshanzadeh A, et al. Surface charge-dependent cytotoxicity of plastic nanoparticles in alveolar cells under cyclic stretches. Nano Lett. 2020;20:7168–7176. doi: 10.1021/acs.nanolett.0c02463. [DOI] [PubMed] [Google Scholar]
- 625.Schaeublin NM, et al. Surface charge of gold nanoparticles mediates mechanism of toxicity. Nanoscale. 2011;3:410–420,. doi: 10.1039/c0nr00478b. [DOI] [PubMed] [Google Scholar]
- 626.Yu M, Xu J, Zheng J. Renal clearable luminescent gold nanoparticles: from the bench to the clinic. Angew. Chem. Int. Ed. 2019;58:4112–4128. doi: 10.1002/anie.201807847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 627.Ge X, et al. Lanthanide (Gd3+ and Yb3+) functionalized gold nanoparticles for in vivo imaging and therapy. Biomaterials. 2016;108:35–43. doi: 10.1016/j.biomaterials.2016.08.051. [DOI] [PubMed] [Google Scholar]
- 628.Desai N. Challenges in development of nanoparticle-based therapeutics. AAPS J. 2012;14:282–295. doi: 10.1208/s12248-012-9339-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 629.Ejazi SA, Louisthelmy R, Maisel K. Mechanisms of nanoparticle transport across intestinal tissue: an oral delivery perspective. ACS Nano. 2023;17:13044–13061. doi: 10.1021/acsnano.3c02403. [DOI] [PubMed] [Google Scholar]
- 630.Beatty PH, Lewis JD. Cowpea mosaic virus nanoparticles for cancer imaging and therapy. Adv. Drug Deliv. Rev. 2019;145:130–144. doi: 10.1016/j.addr.2019.04.005. [DOI] [PubMed] [Google Scholar]
- 631.Poon W, et al. Elimination pathways of nanoparticles. ACS Nano. 2019;13:5785–5798. doi: 10.1021/acsnano.9b01383. [DOI] [PubMed] [Google Scholar]
- 632.Ensign LM, Cone R, Hanes J. Oral drug delivery with polymeric nanoparticles: the gastrointestinal mucus barriers. Adv. Drug Deliv. Rev. 2012;64:557–570. doi: 10.1016/j.addr.2011.12.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 633.He W, Zhang Z, Sha X. Nanoparticles-mediated emerging approaches for effective treatment of ischemic stroke. Biomaterials. 2021;277:121111. doi: 10.1016/j.biomaterials.2021.121111. [DOI] [PubMed] [Google Scholar]
- 634.Falconer JL, Grainger DW. In vivo comparisons of silver nanoparticle and silver ion transport after intranasal delivery in mice. J. Control Release. 2018;269:1–9. doi: 10.1016/j.jconrel.2017.10.018. [DOI] [PubMed] [Google Scholar]
- 635.Wen J, et al. Synthesized nanoparticles, biomimetic nanoparticles and extracellular vesicles for treatment of autoimmune disease: comparison and prospect. Pharmacol. Res. 2021;172:105833. doi: 10.1016/j.phrs.2021.105833. [DOI] [PubMed] [Google Scholar]
- 636.Paunovska K, et al. Using large datasets to understand nanotechnology. Adv. Mater. 2019;31:e1902798. doi: 10.1002/adma.201902798. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 637.Rosenblum D, et al. Progress and challenges towards targeted delivery of cancer therapeutics. Nat. Commun. 2018;9:1410. doi: 10.1038/s41467-018-03705-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 638.Gao H, et al. Ligand modified nanoparticles increases cell uptake, alters endocytosis and elevates glioma distribution and internalization. Sci. Rep. 2013;3:2534. doi: 10.1038/srep02534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 639.Ang MJY, et al. Deciphering nanoparticle trafficking into glioblastomas uncovers an augmented antitumor effect of metronomic chemotherapy. Adv. Mater. 2021;34:e2106194. doi: 10.1002/adma.202106194. [DOI] [PubMed] [Google Scholar]
- 640.Wang C, et al. Liquid core nanoparticle with high deformability enables efficient penetration across biological barriers. Adv. Healthc. 2022;12:e2201889. doi: 10.1002/adhm.202201889. [DOI] [PubMed] [Google Scholar]
- 641.Zhang Z, et al. Mechanistic study of silica nanoparticles on the size-dependent retinal toxicity in vitro and in vivo. J. Nanobiotechnol. 2022;20:146. doi: 10.1186/s12951-022-01326-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 642.Hubo M, et al. Costimulatory molecules on immunogenic versus tolerogenic human dendritic cells. Front. Immunol. 2013;4:82. doi: 10.3389/fimmu.2013.00082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 643.Narasimhan B, Goodman JT, Vela Ramirez JE. Rational design of targeted next-generation carriers for drug and vaccine delivery. Annu Rev. Biomed. Eng. 2016;18:25–49. doi: 10.1146/annurev-bioeng-082615-030519. [DOI] [PubMed] [Google Scholar]
- 644.Tokatlian T, et al. Innate immune recognition of glycans targets HIV nanoparticle immunogens to germinal centers. Science. 2019;363:649–654. doi: 10.1126/science.aat9120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 645.Verma V, Ryan KM, Padrela L. Production and isolation of pharmaceutical drug nanoparticles. Int. J. Pharm. 2021;603:120708. doi: 10.1016/j.ijpharm.2021.120708. [DOI] [PubMed] [Google Scholar]
- 646.Ruan S, et al. Mechanical force-induced dispersion of starch nanoparticles and nanoemulsion: Size control, dispersion behaviour, and emulsified stability. Carbohydr. Polym. 2022;275:118711. doi: 10.1016/j.carbpol.2021.118711. [DOI] [PubMed] [Google Scholar]
- 647.Chen X, et al. Plant-based nanoparticles prepared from proteins and phospholipids consisting of a core–multilayer-shell structure: fabrication, stability, and foamability. J. Agric. Food Chem. 2019;67:6574–6584. doi: 10.1021/acs.jafc.9b02028. [DOI] [PubMed] [Google Scholar]
- 648.Surnar B, et al. Orally administrable therapeutic synthetic nanoparticle for Zika virus. ACS Nano. 2019;13:11034–11048. doi: 10.1021/acsnano.9b02807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 649.Caparco AA, et al. Delivery of nematicides using TMGMV-derived spherical nanoparticles. Nano Lett. 2023;23:5785–5793. doi: 10.1021/acs.nanolett.3c01684. [DOI] [PubMed] [Google Scholar]
- 650.Huynh NT, Passirani C, Saulnier P, Benoit JP. Lipid nanocapsules: a new platform for nanomedicine. Int. J. Pharm. 2009;379:201–209. doi: 10.1016/j.ijpharm.2009.04.026. [DOI] [PubMed] [Google Scholar]
- 651.Awaad A, Elkady EF, El-Mahdy SM. Time-dependent biodistribution profiles and reaction of polyethylene glycol-coated iron oxide nanoclusters in the spleen after intravenous injection in the mice. Acta Histochem. 2022;124:151907. doi: 10.1016/j.acthis.2022.151907. [DOI] [PubMed] [Google Scholar]
- 652.Feng Q, et al. Uptake, distribution, clearance, and toxicity of iron oxide nanoparticles with different sizes and coatings. Sci. Rep. 2018;8:2082. doi: 10.1038/s41598-018-19628-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 653.Tian L, et al. Melanin-like nanoparticles: advances in surface modification and tumour photothermal therapy. J. Nanobiotechnol. 2022;20:485. doi: 10.1186/s12951-022-01698-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 654.Kang H, et al. Stabilization of silver and gold nanoparticles: preservation and improvement of plasmonic functionalities. Chem. Rev. 2018;119:664–699. doi: 10.1021/acs.chemrev.8b00341. [DOI] [PubMed] [Google Scholar]
- 655.Grzelczak M, Pérez-Juste J, Mulvaney P, Liz-Marzán LM. Shape control in gold nanoparticle synthesis. Chem. Soc. Rev. 2008;37:1783–1791. doi: 10.1039/b711490g. [DOI] [PubMed] [Google Scholar]
- 656.Yang L, Wang H, Fang Y, Li Z. Polarization state of light scattered from quantum plasmonic dimer antennas. ACS Nano. 2015;10:1580–1588. doi: 10.1021/acsnano.5b07223. [DOI] [PubMed] [Google Scholar]
- 657.Gillich T, et al. PEG-stabilized core–shell nanoparticles: impact of linear versus dendritic polymer shell architecture on colloidal properties and the reversibility of temperature-induced aggregation. ACS Nano. 2012;7:316–329. doi: 10.1021/nn304045q. [DOI] [PubMed] [Google Scholar]
- 658.Höller RPM, et al. Protein-assisted assembly of modular 3D plasmonic raspberry-like core/satellite nanoclusters: correlation of structure and optical properties. ACS Nano. 2016;10:5740–5750. doi: 10.1021/acsnano.5b07533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 659.Silvestri T, et al. Investigating the effect of surface hydrophilicity on the destiny of PLGA-poloxamer nanoparticles in an in vivo animal model. Int. J. Mol. Sci. 2023;24:14523. doi: 10.3390/ijms241914523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 660.Mohammed DM, et al. Effect of nano-encapsulated food flavorings on streptozotocin-induced diabetic rats. Food Funct. 2023;14:8814–8828. doi: 10.1039/D3FO01299A. [DOI] [PubMed] [Google Scholar]
- 661.Shen Z, et al. Lipid nanoparticle‐mediated delivery of IL‐21‐encoding mRNA induces viral clearance in mouse models of hepatitis B virus persistence. J. Med. Virol. 2023;95:e29062. doi: 10.1002/jmv.29062. [DOI] [PubMed] [Google Scholar]
- 662.Srinivasan SY, et al. Arrhythmic effects evaluated on Caenorhabditis elegans: the case of polypyrrole nanoparticles. ACS Nano. 2023;17:17273–17284. doi: 10.1021/acsnano.3c05245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 663.Chehelgerdi M, et al. Progressing nanotechnology to improve targeted cancer treatment: overcoming hurdles in its clinical implementation. Mol. Cancer. 2023;22:169. doi: 10.1186/s12943-023-01865-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 664.Huang Y, et al. Reactive oxygen species-sensitive biodegradable mesoporous silica nanoparticles harboring TheraVac elicit tumor-specific immunity for colon tumor treatment. ACS Nano. 2023;17:19740–19752. doi: 10.1021/acsnano.3c03195. [DOI] [PubMed] [Google Scholar]
- 665.Ko MJ, et al. Magnetic nanoparticles for ferroptosis cancer therapy with diagnostic imaging. Bioact. Mater. 2024;32:66–97. doi: 10.1016/j.bioactmat.2023.09.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 666.Wheatley AK, Kent SJ. Strength in diversity: widening B cell recruitment for broader vaccine protection. Immunity. 2023;56:2182–2184. doi: 10.1016/j.immuni.2023.09.009. [DOI] [PubMed] [Google Scholar]
- 667.Widge AT, et al. An influenza hemagglutinin stem nanoparticle vaccine induces cross-group 1 neutralizing antibodies in healthy adults. Sci. Transl. Med. 2023;15:eade4790. doi: 10.1126/scitranslmed.ade4790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 668.Antoine TE, et al. Intravaginal zinc oxide tetrapod nanoparticles as novel immunoprotective agents against genital herpes. J. Immunol. 2016;196:4566–4575. doi: 10.4049/jimmunol.1502373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 669.Liu L, et al. Single-cell diagnosis of cancer drug resistance through the differential endocytosis of nanoparticles between drug-resistant and drug-sensitive cancer cells. ACS Nano. 2023;17:19372–19386. doi: 10.1021/acsnano.3c07030. [DOI] [PubMed] [Google Scholar]
- 670.Siraj EA, Yayehrad AT, Belete A. How combined macrolide nanomaterials are effective against resistant pathogens? A comprehensive review of the literature. Int. J. Nanomed. 2023;18:5289–5307. doi: 10.2147/IJN.S418588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 671.Komalla V, et al. Advances in soft mist inhalers. Expert Opin. Drug Deliv. 2023;20:1055–1070. doi: 10.1080/17425247.2023.2231850. [DOI] [PubMed] [Google Scholar]
- 672.Vora LK, et al. Long-acting microneedle formulations. Adv. Drug Deliv. Rev. 2023;201:115055. doi: 10.1016/j.addr.2023.115055. [DOI] [PubMed] [Google Scholar]
- 673.van der Valk FM, et al. Prednisolone-containing liposomes accumulate in human atherosclerotic macrophages upon intravenous administration. Nanomedicine. 2015;11:1039–1046. doi: 10.1016/j.nano.2015.02.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 674.Bentzon JF, Falk E. Atherosclerotic lesions in mouse and man: is it the same disease? Curr. Opin. Lipidol. 2010;21:434–440. doi: 10.1097/MOL.0b013e32833ded6a. [DOI] [PubMed] [Google Scholar]
- 675.Chen W, et al. Macrophage-targeted nanomedicine for the diagnosis and treatment of atherosclerosis. Nat. Rev. Cardiol. 2021;19:228–249. doi: 10.1038/s41569-021-00629-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 676.Metselaar JM, et al. Intravenous pegylated liposomal prednisolone outperforms intramuscular methylprednisolone in treating rheumatoid arthritis flares: a randomized controlled clinical trial. J. Control Release. 2022;341:548–554. doi: 10.1016/j.jconrel.2021.12.007. [DOI] [PubMed] [Google Scholar]
- 677.Han Y, Huang S. Nanomedicine is more than a supporting role in rheumatoid arthritis therapy. J. Control Release. 2023;356:142–161. doi: 10.1016/j.jconrel.2023.02.035. [DOI] [PubMed] [Google Scholar]
- 678.Owens TC, Anton N, Attia MF. CT and X-ray contrast agents: current clinical challenges and the future of contrast. Acta Biomater. 2023;171:19–36. doi: 10.1016/j.actbio.2023.09.027. [DOI] [PubMed] [Google Scholar]
- 679.Ma J-C, et al. Exploring the translational potential of PLGA nanoparticles for intra-articular rapamycin delivery in osteoarthritis therapy. J. Nanobiotechnol. 2023;21:361. doi: 10.1186/s12951-023-02118-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 680.Carvalho SG, et al. Multifunctional systems based on nano-in-microparticles as strategies for drug delivery: advances, challenges, and future perspectives. Expert Opin. Drug Deliv. 2023;20:1231–1249. doi: 10.1080/17425247.2023.2263360. [DOI] [PubMed] [Google Scholar]
- 681.Liu L, Tang H, Wang Y. Nanotechnology-boosted biomaterials for osteoarthritis treatment: current status and future perspectives. Int. J. Nanomed. 2023;18:4969–4983. doi: 10.2147/IJN.S423737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 682.Li B, et al. Powering mesoporous silica nanoparticles into bioactive nanoplatforms for antibacterial therapies: strategies and challenges. J. Nanobiotechnol. 2023;21:325. doi: 10.1186/s12951-023-02093-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 683.He H, et al. Survey of clinical translation of cancer nanomedicines—lessons learned from successes and failures. Acc. Chem. Res. 2019;52:2445–2461. doi: 10.1021/acs.accounts.9b00228. [DOI] [PubMed] [Google Scholar]
- 684.Younis MA, et al. Clinical translation of nanomedicines: challenges, opportunities, and keys. Adv. Drug Deliv. Rev. 2022;181:114083. doi: 10.1016/j.addr.2021.114083. [DOI] [PubMed] [Google Scholar]
- 685.Colombo S, et al. Transforming nanomedicine manufacturing toward Quality by Design and microfluidics. Adv. Drug Deliv. Rev. 2018;128:115–131. doi: 10.1016/j.addr.2018.04.004. [DOI] [PubMed] [Google Scholar]
- 686.Jain K, et al. 3D Printing in development of nanomedicines. Nanomaterials. 2021;11:420–443. doi: 10.3390/nano11020420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 687.Wang Y, et al. Recent advances of nanomedicine-based strategies in diabetes and complications management: Diagnostics, monitoring, and therapeutics. J. Control Release. 2021;330:618–640. doi: 10.1016/j.jconrel.2021.01.002. [DOI] [PubMed] [Google Scholar]
- 688.Duggan S. Caplacizumab: first global approval. Drugs. 2018;78:1639–1642. doi: 10.1007/s40265-018-0989-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 689.Bannas P, Hambach J, Koch-Nolte F. Nanobodies and nanobody-based human heavy chain antibodies as antitumor therapeutics. Front. Immunol. 2017;8:1603. doi: 10.3389/fimmu.2017.01603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 690.McMahon C, et al. Yeast surface display platform for rapid discovery of conformationally selective nanobodies. Nat. Struct. Mol. Biol. 2018;25:289–296. doi: 10.1038/s41594-018-0028-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 691.Soares S, Sousa J, Pais A, Vitorino C. Nanomedicine: principles, properties, and regulatory issues. Front Chem. 2018;6:360. doi: 10.3389/fchem.2018.00360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 692.Rojas LA, et al. Personalized RNA neoantigen vaccines stimulate T cells in pancreatic cancer. Nature. 2023;618:144–150. doi: 10.1038/s41586-023-06063-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 693.Hao Y, et al. Lipid‐based nanoparticles as drug delivery systems for cancer immunotherapy. MedComm. 2023;4:e339. doi: 10.1002/mco2.339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 694.Xu X, et al. Use of a liver-targeting immune-tolerogenic mRNA lipid nanoparticle platform to treat peanut-induced anaphylaxis by single- and multiple-epitope nucleotide sequence delivery. ACS Nano. 2023;17:4942–4957. doi: 10.1021/acsnano.2c12420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 695.Cao W, Xia T. mRNA lipid nanoparticles induce immune tolerance to treat human diseases. Med. Rev. 2023;3:180–183. doi: 10.1515/mr-2023-0008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 696.Flemming A. mRNA vaccine shows promise in autoimmunity. Nat. Rev. Immunol. 2021;21:72–72. doi: 10.1038/s41577-021-00504-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 697.Krienke C, et al. A noninflammatory mRNA vaccine for treatment of experimental autoimmune encephalomyelitis. Science. 2021;371:145–153. doi: 10.1126/science.aay3638. [DOI] [PubMed] [Google Scholar]
- 698.Conde J, Langer R, Rueff J. mRNA therapy at the convergence of genetics and nanomedicine. Nat. Nanotechnol. 2023;18:537–540. doi: 10.1038/s41565-023-01347-w. [DOI] [PubMed] [Google Scholar]
- 699.Brouwer PJM, et al. Two-component spike nanoparticle vaccine protects macaques from SARS-CoV-2 infection. Cell. 2021;184:1188–1200.e1119. doi: 10.1016/j.cell.2021.01.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 700.Marcandalli J, et al. Induction of potent neutralizing antibody responses by a designed protein nanoparticle vaccine for respiratory syncytial virus. Cell. 2019;176:1420–1431.e1417. doi: 10.1016/j.cell.2019.01.046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 701.Song W-F, et al. Targeting to tumor-harbored bacteria for precision tumor therapy. ACS Nano. 2022;16:17402–17413. doi: 10.1021/acsnano.2c08555. [DOI] [PubMed] [Google Scholar]
- 702.Wang Y, et al. Genetically engineered bacteria-mediated multi-functional nanoparticles for synergistic tumor-targeting therapy. Acta Biomater. 2022;150:337–352. doi: 10.1016/j.actbio.2022.07.056. [DOI] [PubMed] [Google Scholar]
- 703.Du Y, et al. Drug-loaded nanoparticles conjugated with genetically engineered bacteria for cancer therapy. Biochem. Biophys. Res. Commun. 2022;606:29–34. doi: 10.1016/j.bbrc.2022.03.049. [DOI] [PubMed] [Google Scholar]
- 704.Jiang F, et al. US/MR bimodal imaging-guided bio-targeting synergistic agent for tumor therapy. Int. J. Nanomed. 2022;17:2943–2960. doi: 10.2147/IJN.S363645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 705.Wang Y, et al. Dual mode imaging guided multi-functional bio-targeted oxygen production probes for tumor therapy. J. Nanobiotechnol. 2023;21:142. doi: 10.1186/s12951-023-01901-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 706.Luo Z, Yi Z, Liu X. Surface engineering of lanthanide nanoparticles for oncotherapy. Acc. Chem. Res. 2023;56:425–439. doi: 10.1021/acs.accounts.2c00681. [DOI] [PubMed] [Google Scholar]
- 707.Kingston BR, et al. Assessing micrometastases as a target for nanoparticles using 3D microscopy and machine learning. Proc. Natl Acad. Sci. USA. 2019;116:14937–14946. doi: 10.1073/pnas.1907646116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 708.Kaushal S, et al. Nano-biotechnology for bacteria identification and potent anti-bacterial properties: a review of current state of the art. Nanomaterials. 2023;13:2529. doi: 10.3390/nano13182529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 709.Yuwen L, Zhang S, Chao J. Recent advances in DNA nanotechnology-enabled biosensors for virus detection. Biosensors. 2023;13:822. doi: 10.3390/bios13080822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 710.Wei-Wen Hsiao W, et al. Nanomaterial-based biosensors for avian influenza virus: a new way forward. Talanta. 2023;265:124892. doi: 10.1016/j.talanta.2023.124892. [DOI] [PubMed] [Google Scholar]
- 711.Song N, et al. Emerging nanotechnology for Alzheimer’s disease: from detection to treatment. J. Control Release. 2023;360:392–417. doi: 10.1016/j.jconrel.2023.07.004. [DOI] [PubMed] [Google Scholar]
- 712.de Puig H, et al. Multiplexed rapid antigen tests developed using multicolored nanoparticles and cross-reactive antibody pairs: Implications for pandemic preparedness. Nano Today. 2022;47:101669. doi: 10.1016/j.nantod.2022.101669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 713.Chen AL, et al. Sandwich-type electrochemiluminescence immunosensor based on CDs@dSiO2 nanoparticles as nanoprobe and co-reactant. Biosensors. 2023;13:133. doi: 10.3390/bios13010133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 714.Huang H, et al. MXene-incorporated C60NPs and Au@Pt with dual-electric signal outputs for accurate detection of Mycobacterium tuberculosis ESAT-6 antigen. Biosens. Bioelectron. 2023;242:115734. doi: 10.1016/j.bios.2023.115734. [DOI] [PubMed] [Google Scholar]
- 715.Mordini D, et al. Luminescent gold nanoclusters for bioimaging: increasing the ligand complexity. Nanomaterials. 2023;13:648. doi: 10.3390/nano13040648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 716.Han L, et al. Development of a dual mode UCNPs-MB biosensor in combination with PCR for sensitive detection of salmonella. Biosensors. 2023;13:475. doi: 10.3390/bios13040475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 717.Tang W-F, Tran A-T, Wang L-Y, Horng J-T. SARS-CoV-2 pandemics: an update of CRISPR in diagnosis and host–virus interaction studies. Biomed. J. 2023;46:100587. doi: 10.1016/j.bj.2023.02.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 718.Mustafa MI, Makhawi AM, Kraft CS. SHERLOCK and DETECTR: CRISPR-Cas systems as potential rapid diagnostic tools for emerging infectious diseases. J. Clin. Microbiol. 2021;59:e00745–00720. doi: 10.1128/JCM.00745-20. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 719.Sima N, et al. SHERLOCK4HAT: a CRISPR-based tool kit for diagnosis of Human African Trypanosomiasis. EBioMedicine. 2022;85:104308. doi: 10.1016/j.ebiom.2022.104308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 720.Casati B, et al. Rapid, adaptable and sensitive Cas13-based COVID-19 diagnostics using ADESSO. Nat. Commun. 2022;13:3308. doi: 10.1038/s41467-022-30862-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 721.Pena JM, et al. Real-time, multiplexed SHERLOCK for in vitro diagnostics. J. Mol. Diagn. 2023;25:428–437. doi: 10.1016/j.jmoldx.2023.03.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 722.Allan-Blitz L-T, et al. Development of Cas13a-based assays for Neisseria gonorrhoeae detection and gyrase A determination. mSphere. 2023;8:e0041623. doi: 10.1128/msphere.00416-23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 723.Thakku SG, et al. Genome-wide tiled detection of circulating Mycobacterium tuberculosis cell-free DNA using Cas13. Nat. Commun. 2023;14:1803. doi: 10.1038/s41467-023-37183-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 724.MacGregor SR, et al. Development of CRISPR/Cas13a-based assays for the diagnosis of Schistosomiasis. EBioMedicine. 2023;94:104730. doi: 10.1016/j.ebiom.2023.104730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 725.Li H, et al. Strategies to improve multi-enzyme compatibility and coordination in one-pot SHERLOCK. Anal. Chem. 2023;95:10522–10531. doi: 10.1021/acs.analchem.2c05032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 726.Yarnall MTN, et al. Drag-and-drop genome insertion of large sequences without double-strand DNA cleavage using CRISPR-directed integrases. Nat. Biotechnol. 2022;41:500–512. doi: 10.1038/s41587-022-01527-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 727.El-Safty SA, Shenashen MA. Nanoscale dynamic chemical, biological sensor material designs for control monitoring and early detection of advanced diseases. Mater. Today Bio. 2020;5:100044. doi: 10.1016/j.mtbio.2020.100044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 728.Jhunjhunwala A, et al. In vivo photoacoustic monitoring of stem cell location and apoptosis with caspase-3-responsive nanosensors. ACS Nano. 2023;17:17931–17945. doi: 10.1021/acsnano.3c04161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 729.Harvey JD, et al. An in vivo nanosensor measures compartmental doxorubicin exposure. Nano Lett. 2019;19:4343–4354. doi: 10.1021/acs.nanolett.9b00956. [DOI] [PubMed] [Google Scholar]
- 730.Zhang Y, et al. A nanometallic conductive composite-hydrogel core–shell microneedle skin patch for real-time monitoring of interstitial glucose levels. Nanoscale. 2023;15:16493–16500. doi: 10.1039/D3NR01245J. [DOI] [PubMed] [Google Scholar]
- 731.Choi JH, et al. Combination of porous silk fibroin substrate and gold nanocracks as a novel SERS platform for a high-sensitivity biosensor. Biosensors. 2021;11:441. doi: 10.3390/bios11110441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 732.Chaparro CIP, et al. A promising approach: magnetic nanosystems for Alzheimer’s disease theranostics. Pharmaceutics. 2023;15:2316. doi: 10.3390/pharmaceutics15092316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 733.Fang L, et al. Hyperbranched polymer dots with strong absorption and high fluorescence quantum yield for in vivo NIR-II imaging. Nano Lett. 2023;23:8734–8742. doi: 10.1021/acs.nanolett.3c02751. [DOI] [PubMed] [Google Scholar]
- 734.Zhang, Q. et al. Renal clearable magnetic nanoparticles for magnetic resonance imaging and guided therapy. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 10.1002/wnan.1929 (2023). [DOI] [PubMed]
- 735.Liang R, et al. Multimodal imaging-guided strategy for developing 177Lu-labeled metal–organic framework nanomedicine with potential in cancer therapy. ACS Appl. Mater. Interfaces. 2023;15:45713–45724. doi: 10.1021/acsami.3c11098. [DOI] [PubMed] [Google Scholar]
- 736.Rehan M, et al. Smart capsules for sensing and sampling the gut: status, challenges and prospects. Gut. 2024;73:186–202. doi: 10.1136/gutjnl-2023-329614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 737.Devnarain N, et al. Intrinsic stimuli‐responsive nanocarriers for smart drug delivery of antibacterial agents—An in‐depth review of the last two decades. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2020;13:e1664. doi: 10.1002/wnan.1664. [DOI] [PubMed] [Google Scholar]
- 738.Gayet RV, et al. Creating CRISPR-responsive smart materials for diagnostics and programmable cargo release. Nat. Protoc. 2020;15:3030–3063. doi: 10.1038/s41596-020-0367-8. [DOI] [PubMed] [Google Scholar]
- 739.Banaye Yazdipour A, et al. Predicting the toxicity of nanoparticles using artificial intelligence tools: a systematic review. Nanotoxicology. 2023;17:62–77. doi: 10.1080/17435390.2023.2186279. [DOI] [PubMed] [Google Scholar]
- 740.Nuhn L. Artificial intelligence assists nanoparticles to enter solid tumours. Nat. Nanotechnol. 2023;18:550–551. doi: 10.1038/s41565-023-01382-7. [DOI] [PubMed] [Google Scholar]
- 741.Subramanian NA, Palaniappan A. NanoTox: development of a parsimonious in silico model for toxicity assessment of metal-oxide nanoparticles using physicochemical features. ACS Omega. 2021;6:11729–11739. doi: 10.1021/acsomega.1c01076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 742.Yu F, et al. Deep exploration of random forest model boosts the interpretability of machine learning studies of complicated immune responses and lung burden of nanoparticles. Sci. Adv. 2021;7:eabf4130. doi: 10.1126/sciadv.abf4130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 743.Zhu M, et al. Machine-learning-assisted single-vessel analysis of nanoparticle permeability in tumour vasculatures. Nat. Nanotechnol. 2023;18:657–666. doi: 10.1038/s41565-023-01323-4. [DOI] [PubMed] [Google Scholar]
- 744.Arber Raviv S, et al. Lung targeted liposomes for treating ARDS. J. Control Release. 2022;346:421–433. doi: 10.1016/j.jconrel.2022.03.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 745.Richner JM, et al. Modified mRNA vaccines protect against Zika virus infection. Cell. 2017;168:1114–1125.e1110. doi: 10.1016/j.cell.2017.02.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 746.Hou X, et al. Vitamin lipid nanoparticles enable adoptive macrophage transfer for the treatment of multidrug-resistant bacterial sepsis. Nat. Nanotechnol. 2020;15:41–46. doi: 10.1038/s41565-019-0600-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 747.Xu Z, et al. Glycyrrhizic acid-lipid framework nanovehicle loading triptolide for combined immunochemotherapy. ACS Appl. Mater. Interfaces. 2023;15:41337–41350. doi: 10.1021/acsami.3c08003. [DOI] [PubMed] [Google Scholar]
- 748.Xiang D-x, Chen Q, Pang L, Zheng C-l. Inhibitory effects of silver nanoparticles on H1N1 influenza A virus in vitro. J. Virol. Methods. 2011;178:137–142. doi: 10.1016/j.jviromet.2011.09.003. [DOI] [PubMed] [Google Scholar]
- 749.Paul AM, et al. Delivery of antiviral small interfering RNA with gold nanoparticles inhibits dengue virus infection in vitro. J. Gen. Virol. 2014;95:1712–1722. doi: 10.1099/vir.0.066084-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 750.Hang X, et al. Antiviral activity of cuprous oxide nanoparticles against Hepatitis C Virus in vitro. J. Virol. Methods. 2015;222:150–157. doi: 10.1016/j.jviromet.2015.06.010. [DOI] [PubMed] [Google Scholar]
- 751.Howarth JR, et al. Interactions between multi-walled carbon nanotubes and plankton as detected by Raman spectroscopy. Chemosphere. 2022;295:133889. doi: 10.1016/j.chemosphere.2022.133889. [DOI] [PubMed] [Google Scholar]
- 752.Deokar AR, et al. Graphene-based “hot plate” for the capture and destruction of the herpes simplex virus type 1. Bioconjug. Chem. 2017;28:1115–1122. doi: 10.1021/acs.bioconjchem.7b00030. [DOI] [PubMed] [Google Scholar]
- 753.Bakry R, et al. Medicinal applications of fullerenes. Int J. Nanomed. 2007;2:639–649. [PMC free article] [PubMed] [Google Scholar]
- 754.Biswas R, et al. C60-β-cyclodextrin conjugates for enhanced nucleus delivery of doxorubicin. Nanoscale. 2022;14:4456–4462. doi: 10.1039/D2NR00777K. [DOI] [PubMed] [Google Scholar]
- 755.Zmonarski SZ, Stojanowski J, Zmonarska J. Polymers with antiviral properties: a brief review. Polim. Med. 2021;50:79–82. doi: 10.17219/pim/131643. [DOI] [PubMed] [Google Scholar]
- 756.Cao Z-T, et al. Protein binding affinity of polymeric nanoparticles as a direct indicator of their pharmacokinetics. ACS Nano. 2020;14:3563–3575. doi: 10.1021/acsnano.9b10015. [DOI] [PubMed] [Google Scholar]
- 757.Kurtovic L, et al. Novel virus-like particle vaccine encoding the circumsporozoite protein of Plasmodium falciparum is immunogenic and induces functional antibody responses in mice. Front. Immunol. 2021;12:641421. doi: 10.3389/fimmu.2021.641421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 758.Banskota S, et al. Engineered virus-like particles for efficient in vivo delivery of therapeutic proteins. Cell. 2022;185:250–265.e216. doi: 10.1016/j.cell.2021.12.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 759.Zhang P, et al. Dual-sensitive antibacterial peptide nanoparticles prevent dental caries. Theranostics. 2022;12:4818–4833. doi: 10.7150/thno.73181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 760.Kim Y-I, et al. Development of spike receptor-binding domain nanoparticles as a vaccine candidate against SARS-CoV-2 infection in ferrets. mBio. 2021;12:e00230–00221. doi: 10.1128/mBio.00230-21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 761.Jochum S, et al. Clinical utility of Elecsys anti-SARS-CoV-2 S assay in COVID-19 vaccination: an exploratory analysis of the mRNA-1273 phase 1 trial. Front Immunol. 2021;12:798117. doi: 10.3389/fimmu.2021.798117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 762.Shabu A, Nishtala PS. Safety outcomes associated with the moderna COVID-19 vaccine (mRNA-1273): a literature review. Expert Rev. Vaccines. 2023;22:393–409. doi: 10.1080/14760584.2023.2209177. [DOI] [PubMed] [Google Scholar]
- 763.Graña C, et al. Efficacy and safety of COVID-19 vaccines. Cochrane Database Syst. Rev. 2022;2023:Cd015477. doi: 10.1002/14651858.CD015477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 764.Bergman P, et al. Safety and efficacy of the mRNA BNT162b2 vaccine against SARS-CoV-2 in five groups of immunocompromised patients and healthy controls in a prospective open-label clinical trial. EBioMedicine. 2021;74:103705. doi: 10.1016/j.ebiom.2021.103705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 765.Martin-Orozco N, et al. Phase I randomized, observer-blinded, placebo-controlled study of a SARS-CoV-2 mRNA vaccine PTX-COVID19-B. Sci. Rep. 2023;13:8557. doi: 10.1038/s41598-023-35662-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 766.Erasmus JH, et al. An Alphavirus-derived replicon RNA vaccine induces SARS-CoV-2 neutralizing antibody and T cell responses in mice and nonhuman primates. Sci. Transl. Med. 2020;12:eabc9396. doi: 10.1126/scitranslmed.abc9396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 767.O’Connor MA, et al. A single dose SARS-CoV-2 replicon RNA vaccine induces cellular and humoral immune responses in simian immunodeficiency virus infected and uninfected pigtail macaques. Front. Immunol. 2021;12:800723. doi: 10.3389/fimmu.2021.800723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 768.Branche AR, et al. Comparison of bivalent and monovalent SARS-CoV-2 variant vaccines: the phase 2 randomized open-label COVAIL trial. Nat. Med. 2023;29:2334–2346. doi: 10.1038/s41591-023-02503-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 769.Johnston SC, et al. A SARS-CoV-2 spike ferritin nanoparticle vaccine is protective and promotes a strong immunological response in the cynomolgus macaque coronavirus disease 2019 (COVID-19) model. Vaccines. 2022;10:717. doi: 10.3390/vaccines10050717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 770.Joyce MG, et al. SARS-CoV-2 ferritin nanoparticle vaccines elicit broad SARS coronavirus immunogenicity. Cell Rep. 2021;37:110143. doi: 10.1016/j.celrep.2021.110143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 771.Zhang H, et al. Adenovirus-mediated artificial MicroRNAs targeting matrix or nucleoprotein genes protect mice against lethal influenza virus challenge. Gene Ther. 2015;22:653–662. doi: 10.1038/gt.2015.31. [DOI] [PubMed] [Google Scholar]
- 772.Song JY, et al. Safety and immunogenicity of a SARS-CoV-2 recombinant protein nanoparticle vaccine (GBP510) adjuvanted with AS03: a randomised, placebo-controlled, observer-blinded phase 1/2 trial. EClinicalMedicine. 2022;51:101569. doi: 10.1016/j.eclinm.2022.101569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 773.Gong J, Gujjula KR, Ntaimo L. An integrated chance constraints approach for optimal vaccination strategies under uncertainty for COVID-19. Socio-Econ. Plan. Sci. 2023;87:101547. doi: 10.1016/j.seps.2023.101547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 774.Shinde V, et al. Induction of cross-reactive hemagglutination inhibiting antibody and polyfunctional CD4+ T-cell responses by a recombinant matrix-M–adjuvanted hemagglutinin nanoparticle influenza vaccine. Clin. Infect. Dis. 2021;73:e4278–e4287. doi: 10.1093/cid/ciaa1673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 775.Toback S, et al. Safety, immunogenicity, and efficacy of a COVID-19 vaccine (NVX-CoV2373) co-administered with seasonal influenza vaccines: an exploratory substudy of a randomised, observer-blinded, placebo-controlled, phase 3 trial. Lancet. Respir. Med. 2022;10:167–179. doi: 10.1016/S2213-2600(21)00409-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 776.Walls AC, et al. Distinct sensitivities to SARS-CoV-2 variants in vaccinated humans and mice. Cell Rep. 2022;40:111299. doi: 10.1016/j.celrep.2022.111299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 777.Pleguezuelos O, et al. Immunogenicity, safety, and efficacy of a standalone universal influenza vaccine, FLU-v, in healthy adults. Ann. Intern. Med. 2020;172:453–462,. doi: 10.7326/M19-0735. [DOI] [PubMed] [Google Scholar]
- 778.Cohen JI, Fauci AS, Varmus H, Nabel GJ. Epstein-Barr virus: an important vaccine target for cancer prevention. Sci. Transl. Med. 2011;3:107fs107. doi: 10.1126/scitranslmed.3002878. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 779.Cohen JI. Epstein–Barr virus vaccines. Clin. Transl. Immunol. 2015;4:e32. doi: 10.1038/cti.2014.27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 780.Kanekiyo M, et al. Rational design of an Epstein-Barr virus vaccine targeting the receptor-binding site. Cell. 2015;162:1090–1100. doi: 10.1016/j.cell.2015.07.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 781.Fries L, et al. Randomized, blinded, dose-ranging trial of an Ebola virus glycoprotein nanoparticle vaccine with matrix-M adjuvant in healthy adults. J. Infect. Dis. 2020;222:572–582. doi: 10.1093/infdis/jiz518. [DOI] [PubMed] [Google Scholar]
- 782.Chu M, et al. Nebulization therapy with umbilical cord mesenchymal stem cell-derived exosomes for COVID-19 pneumonia. Stem Cell Rev. Rep. 2022;18:2152–2163. doi: 10.1007/s12015-022-10398-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 783.El-Shennawy L, et al. Circulating ACE2-expressing extracellular vesicles block broad strains of SARS-CoV-2. Nat. Commun. 2022;13:405. doi: 10.1038/s41467-021-27893-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 784.Lightner AL, et al. Bone marrow mesenchymal stem cell-derived extracellular vesicle infusion for the treatment of respiratory failure from COVID-19. Chest. 2023;164:1444–1453. doi: 10.1016/j.chest.2023.06.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 785.Williams PE, Crauwels HM, Basstanie ED. Formulation and pharmacology of long-acting rilpivirine. Curr. Opin. HIV AIDS. 2015;10:233–238. doi: 10.1097/COH.0000000000000164. [DOI] [PubMed] [Google Scholar]
- 786.Jackson AGA, et al. A compartmental pharmacokinetic evaluation of long-acting rilpivirine in HIV-negative volunteers for pre-exposure prophylaxis. Clin. Pharmacol. Ther. 2014;96:314–323. doi: 10.1038/clpt.2014.118. [DOI] [PubMed] [Google Scholar]
- 787.Taléns-Visconti R, et al. Intranasal drug administration in Alzheimer-type dementia: towards clinical applications. Pharmaceutics. 2023;15:1399. doi: 10.3390/pharmaceutics15051399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 788.Gradishar WJ, et al. Significantly longer progression-free survival with nab-paclitaxel compared with docetaxel as first-line therapy for metastatic breast. Cancer J. Clin. Oncol. 2009;27:3611–3619. doi: 10.1200/JCO.2008.18.5397. [DOI] [PubMed] [Google Scholar]
- 789.Farag AF, Hassabou NF. CD24-gold nanocomposite as promising and sensitive biomarker for cancer stem cells in salivary gland tumors. Nanomedicine. 2022;46:102598. doi: 10.1016/j.nano.2022.102598. [DOI] [PubMed] [Google Scholar]
- 790.Hald Albertsen C, et al. The role of lipid components in lipid nanoparticles for vaccines and gene therapy. Adv. Drug Deliv. Rev. 2022;188:114416. doi: 10.1016/j.addr.2022.114416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 791.Díez-Pascual AM. Carbon-Based Nanomaterials. Int. J. Mol. Sci. 2021;22:7726. doi: 10.3390/ijms22147726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 792.Ferreira Soares DC, Domingues SC, Viana DB, Tebaldi ML. Polymer-hybrid nanoparticles: current advances in biomedical applications. Biomed. Pharmacother. 2020;131:110695. doi: 10.1016/j.biopha.2020.110695. [DOI] [PubMed] [Google Scholar]
- 793.Shi J, Xiao Z, Kamaly N, Farokhzad OC. Self-assembled targeted nanoparticles: evolution of technologies and bench to bedside translation. Acc. Chem. Res. 2011;44:1123–1134. doi: 10.1021/ar200054n. [DOI] [PubMed] [Google Scholar]
- 794.Liang Y, Duan L, Lu J, Xia J. Engineering exosomes for targeted drug delivery. Theranostics. 2021;11:3183–3195. doi: 10.7150/thno.52570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 795.Vallhov H, et al. Adjuvant properties of mesoporous silica particles tune the development of effector T cells. Small. 2012;8:2116–2124. doi: 10.1002/smll.201102620. [DOI] [PubMed] [Google Scholar]
- 796.Vallhov H, et al. Mesoporous silica particles induce size dependent effects on human dendritic cells. Nano Lett. 2007;7:3576–3582. doi: 10.1021/nl0714785. [DOI] [PubMed] [Google Scholar]
- 797.Singh A. Eliciting B cell immunity against infectious diseases using nanovaccines. Nat. Nanotechnol. 2020;16:16–24. doi: 10.1038/s41565-020-00790-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 798.Dykman LA, Khlebtsov NG. Immunological properties of gold nanoparticles. Chem. Sci. 2017;8:1719–1735. doi: 10.1039/C6SC03631G. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 799.Li X, et al. Changes in the microbial communities in vacuum-packaged smoked bacon during storage. Food Microbiol. 2019;77:26–37. doi: 10.1016/j.fm.2018.08.007. [DOI] [PubMed] [Google Scholar]
- 800.Gootenberg JS, et al. Multiplexed and portable nucleic acid detection platform with Cas13, Cas12a, and Csm6. Science. 2018;360:439–444. doi: 10.1126/science.aaq0179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 801.Gootenberg JS, et al. Nucleic acid detection with CRISPR-Cas13a/C2c2. Science. 2017;356:438–442. doi: 10.1126/science.aam9321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 802.Sacks D, et al. Multisociety consensus quality improvement revised consensus statement for endovascular therapy of acute ischemic stroke. Int J. Stroke. 2018;13:612–632. doi: 10.1177/1747493018778713. [DOI] [PubMed] [Google Scholar]
- 803.Zhang C, et al. An oral polyphenol host-guest nanoparticle for targeted therapy of inflammatory bowel disease. Acta Biomater. 2023;169:422–433. doi: 10.1016/j.actbio.2023.08.020. [DOI] [PubMed] [Google Scholar]
- 804.Xu H, et al. Duplex responsive nanoplatform with cascade targeting for atherosclerosis photoacoustic diagnosis and multichannel combination therapy. Adv. Mater. 2023;35:e2300439. doi: 10.1002/adma.202300439. [DOI] [PubMed] [Google Scholar]