Summary
Alkynes are widely present in natural products and pharmaceutical compounds. Here, we present a protocol for nickel-catalyzed cross-coupling of terminal alkynes with aryl iodides or bromides for constructing a C(sp2)-C(sp) bond. We describe steps for reagent preparation, reaction setup, purification process, and product characterization. We also detail procedures for obtaining a single crystal of 6-(phenylethynyl)-1-(phenylsulfonyl)-1H-indole (3b). The application of this protocol is limited to aryl bromide and iodide.
For complete details on the use and execution of this protocol, please refer to Chen et al.1
Subject areas: X-ray Crystallography, NMR, Mass Spectrometry, Chemistry, Material sciences
Graphical abstract

Highlights
-
•
Steps for Sonogashira reactions to form C(sp2)-C(sp) bonds without using a co-catalyst
-
•
Steps for Ni-catalyzed C(sp2)-C(sp) coupling of terminal alkynes and aryl halides
-
•
Steps for purification and recrystallization of product
Publisher’s note: Undertaking any experimental protocol requires adherence to local institutional guidelines for laboratory safety and ethics.
Alkynes are widely present in natural products and pharmaceutical compounds. Here, we present a protocol for nickel-catalyzed cross-coupling of terminal alkynes with aryl iodides or bromides for constructing a C(sp2)-C(sp) bond. We describe steps for reagent preparation, reaction setup, purification process, and product characterization. We also detail procedures for obtaining a single crystal of 6-(phenylethynyl)-1-(phenylsulfonyl)-1H-indole (3b). The application of this protocol is limited to aryl bromide and iodide.
Before you begin
The alkyne is a functional moiety that is important for many natural products and pharmaceutical compounds.2,3,4,5,6,7,8,9,10,11 It is widely applied for bio-orthogonal labeling, often referred to as 'click chemistry', as well as materials science.12,13,14,15,16,17,18 In addition, sp carbons of alkynes introduced via C-C(sp) cross-couplings, could be readily converted into sp3 and sp2 carbons under many known mild conditions. Accordingly, C-C(sp) cross-couplings serve as a valuable complementary approach to C-C(sp3)/(sp2) couplings for constructing the carbon-carbon backbones of products.19,20,21,22,23
The Sonogashira reaction, regarded as the most widely used C-C(sp) cross-coupling methods, traditionally combine a palladium catalyst with a copper co-catalyst to enable the coupling of terminal alkynes with electrophiles. In recent years, there has been a growing interest in the development of Sonogashira-type reactions catalyzed by a single transition metal. The state-of-the-art work reported by Buter, Feringa and co-workers presents a Pd-catalyzed cross-coupling of lithium acetylides with aryl bromides. Their innovative approach requires the preparation of air- and moisture-sensitive lithium acetylides in advance.24 The conditions for single copper-catalyzed Sonogashira reactions to form C(sp2)-C(sp) bonds can be quite harsh.25 Elevated reaction temperatures are required to facilitate oxidative addition of copper catalyst with aryl halides. In contrast, nickel owns an advantage of facile oxidative addition with C(sp2) partners, in which some coupling reactions could occur at mild conditions.26
In this protocol, we depict an efficient and practical nickel-catalyzed C(sp2)-C(sp) Sonogashira coupling reaction for unactivated terminal alkynes and aryl halides without using a co-catalyst. This protocol demonstrates good functional group tolerance since strong bases are avoided. This strategy opens up new avenues for preparing a broad array of acetylenes, including those with applications in optoelectronic materials, pharmaceuticals, and fundamental chemical biology building blocks. Additionally, this protocol can achieve selective alkynylation for multi-halide substituted arenes by careful tuning of the reaction conditions. For the details, please refer to Chen et al.1
Preparation of the reagent and equipment
A complete list of reagents and equipment can be found in the ‘‘key resources table’’ and ‘‘materials and equipment’’.
Preparation of active zinc powder
Timing: 4 h
-
1.
Introduce commercial zinc powder (10 g), 200 mesh, obtained from Acros, into a stirring 250 mL single-neck round-bottomed flask, which contains 100 mL of 2% hydrochloric acid (with a weight ratio of HCl to H2O at 2:98).
-
2.
Stir vigorously until the surface of the zinc becomes bright and no bubbles are generated from it (10 min).
Note: Conduct stirring under nitrogen protection.
-
3.
Transfer the activated zinc powder to a suction filter. The obtained zinc powder is washed with distilled water (50 mL × 3), ethanol (50 mL × 3), acetone (50 mL × 3) and ether (50 mL × 3).
-
4.
Transfer the washed zinc powder into a single-neck round-bottomed flask and dried at 25°C under vacuum for 3 h. This process yields 8.2 g of activated zinc.
CRITICAL: Filtration and washing should be accomplished as rapidly as possible to shorten the exposure time of zinc to air (within 10 min). If necessary, the obtained zinc powder could be filtrated by 200 mesh sieve and stored under N2.
Note: Gloves are required since hydrochloric acid is corrosive.
Key resources table
| REAGENT or RESOURCE | SOURCE | IDENTIFIER |
|---|---|---|
| Chemicals, peptides, and recombinant proteins | ||
| Nickel chloride, anhydrous, powder (99.99%) | Acros | CAS: 7718-54-9 |
| 1,10-Phenanthroline, anhydrous, powder (99+%) | Acros | CAS: 66-71-7 |
| 4-Cyano pyridine N-oxide (98%) | TCI | CAS: 14906-59-3 |
| Potassium fluoride (99.99%) | Alfa Aesar | CAS: 7789-23-3 |
| Zinc powder (99.995%) | Acros | CAS: 7440-66-6 |
| N,N-dimethylacetamide, 99.5%, super dry, with molecular sieves, water ≤ 30 ppm | J&K Scientific | CAS: 127-19-5 |
| 4-Iodobiphenyl (97%) | Alfa Aesar | CAS: 1591-31-7 |
| Phenylacetylene (98+%) | Alfa Aesar | CAS: 536-74-3 |
| 6-Bromo-1-(phenylsulfonyl)-1H-indole (98%) | Adamas-Beta | CAS: 679794-03-7 |
| 1,4-Diiodobenzene (98%) | Alfa Aesar | CAS: 624-38-4 |
| (Triethylsilyl)acetylene (97%) | Adamas-Beta | CAS: 1777-03-3 |
| 4-Ethynylanisole (98%) | TCI | CAS: 768-60-5 |
| 3-Bromoiodobenzene (97%) | TCI | CAS: 591-18-4 |
| 4-Ethynyltoluene (98+%) | Adamas-Beta | CAS: 766-97-2 |
| 4-Tert-butylphenylacetylene (98%) | Adamas-Beta | CAS: 772-38-3 |
| 4-Iodophenyl acetate (99%) | Adamas-Beta | CAS: 33527-94-5 |
| Sodium hydroxide (98%) | Acros | CAS: 1310-73-2 |
| Sodium chloride (98%) | Shanghai Titan Scientific Co., Ltd. | CAS: 7647-14-5 |
| Methanol (99.5%) | J&K Scientific | CAS: 67-56-1 |
| Hydrochloric acid (37%) | Fisher Scientific | CAS: 7647-01-0 |
| Ethyl acetate | General reagent | CAS: 141-78-6 |
| Petroleum ether | General reagent | CAS: 8032-32-4 |
| Anhydrous sodium sulfate | General reagent | CAS: 7757-82-6 |
| Dichloromethane | General reagent | CAS: 75-09-2 |
| n-Hexane | General reagent | CAS: 110-54-3 |
| Deuterated chloroform, 99.8 atom % D | Energy Chemical | CAS: 865-49-6 |
| Nitrogen (>99.9999%) | Air Liquide | CAS: 7727-37-9 |
| Critical commercial assays | ||
| Structure of compound 3b | This paper; Cambridge Crystallographic Data Center | CCDC: 2068369 |
| Software and algorithms | ||
| GCMS-analysis | Shimadzu | https://www.shimadzu.com |
| MestReNova | Mestrelab Research | https://mestrelab.com |
| ChemBioDraw Ultra 14.0 | PerkinElmer | https://www.perkinelmer.com/category/chemdraw |
| Other | ||
| Magnetic stirrer | IKA | https://www.ika.com |
| Electronic balance | Mettler Toledo | Cat# AL104 |
| Magnetic stir bar | Synthware Glass | Cat# SA1758 |
| Vacuum pump | VALUE | Cat# VRD 12 |
| Syringe needle | Synthware Glass | Cat# YCKJ-KSM-2-007353 |
| Rubber plug | Shanghai Titan Scientific Co., Ltd. | Cat# 02024583 |
| Vial (2 mL) | Shanghai Titan Scientific Co., Ltd. | Cat# 02041214 |
| Tube (φ 12 × 75 mm) | Shanghai Titan Scientific Co., Ltd. | Cat# 02036870 |
| Round-bottom flask | Synthware Glass | Cat# F304250 |
| Filter funnel | Synthware Glass | Cat# F366019M |
| Chromatography column | Synthware Glass | Cat# C371720 |
| Suction filter | Synthware Glass | Cat# F664500R |
| Absorbent cotton | Innochem (Beijing) Technology | Cat# C124537-50g |
| Booster pump | Zhipu | Cat# zhipu-12L |
| Thin-layer chromatography (TLC-plates 0.25 mm) | XUBO | Cat# HSGF254 |
| Separatory funnel | Synthware Glass | Cat# F472925A |
| Oven | Keelrein | Cat# DHG-9240A |
| Rotary evaporator | EYELA | Cat# N-1100D-WB |
| 400 MHz NMR spectrometer | Bruker | Cat# AVANCE III |
| X-ray single crystal diffractometer | Rigaku | Cat# XtaLAB PRO 007HF(Mo) |
| Silica gel (200–300 mesh) | Shanghai Titan Scientific Co., Ltd. | Cat# DG72651C |
| Gloves | Ammex | Cat# APFNCHD100 |
| Gas chromatography-mass spectrometer (GC-MS) | Shimadzu | Cat# GCMS-QP2010-SE |
| Glovebox | Vigor | Cat# LG1200/750TS-F |
Materials and equipment
Materials examples
| Reagent | Final concentration | Amount |
|---|---|---|
| 4-Iodo-1,1′-biphenyl (1a) | N/A | 140 mg |
| Phenylacetylene (2a) | N/A | 77 mg |
| Nickel chloride (NiCl2) | N/A | 6.5 mg |
| 1,10-Phenanthroline | N/A | 13.5 mg |
| 4-Cyanopyridine N-oxide | N/A | 90 mg |
| Potassium fluoride (KF) | N/A | 44 mg |
| Zinc powder (Zn) | N/A | 38 mg |
| N,N-dimethylacetamide (DMAc) | N/A | 5 mL |
Store at 4°C for up to 2 years.
Step-by-step method details
Part 1: Set up Ni-catalyzed C(sp2)-C(sp) coupling of terminal alkynes and aryl iodides
Timing: 48 h
In this part, 4-(phenylethynyl)-1,1′-biphenyl (3a) is prepared with nickel catalysis (Scheme 1). Twenty-three analogs have been produced via this method. The full scope of the transformation is described in Chen et al.1
-
1.Set up reaction (Scheme 1; Figure 1):
-
a.Add nickel(II) dichloride (0.05 mmol, 10 mol %) to a solution of 1,10-phenanthroline (0.075 mmol, 15 mol %) in degassed N,N-dimethylacetamide (DMAc) (2.00 mL).Note: This results in a mixture in a 15 mL single-neck flask.
-
b.Stir the solution in a glovebox filled with N2 at 25°C for 30 min.
-
c.Add 4-iodo-1,1′-biphenyl (1a) (0.50 mmol, 1.0 equiv), phenylacetylene (2a) (0.75 mmol, 1.5 equiv), 4-cyanopyridine N-oxide (0.75 mmol, 1.5 equiv), KF (0.75 mmol, 1.5 equiv), zinc powder (0.60 mmol, 1.2 equiv) and DMAc (3.00 mL) to the mixture successively. The reaction mixture is subsequently heated under N2 at 60°C for 48 h (Figures 1A and 1B).
 CRITICAL: The reaction is sensitive to oxygen. We suggest conducting the reaction in glovebox filled with nitrogen. If Schlenk operation techniques are applied, please replace the air in tube with nitrogen for many times to ensure the inert gas atmosphere.
CRITICAL: The reaction is sensitive to oxygen. We suggest conducting the reaction in glovebox filled with nitrogen. If Schlenk operation techniques are applied, please replace the air in tube with nitrogen for many times to ensure the inert gas atmosphere.
-
a.
Scheme 1.
Ni-catalyzed Sonogashira coupling for preparation of 3a
Figure 1.
Reaction and appearance of 3a and 3b at different stages
Part 2. Purification of the crude material 4-(phenylethynyl)-1,1′-biphenyl (3a)
Timing: 1.5 h
-
2.Work up reaction (30 min):
-
a.Pour the reaction mixture into water (100 mL).
-
b.Extract the water layer with ethyl acetate (3 × 25 mL) (Figure 1C).
-
c.Wash the combined extracts with brine (1 × 25 mL).
-
d.Dry the ethyl acetate layer with anhydrous sodium sulfate (Figure 1D).
-
e.Filter off sodium sulfate and pour the filtrate into a 250 mL round-bottom flask.
-
f.Concentrate the filtrate under reduced pressure with a rotary evaporator to afford the crude product (-0.1 MPa, 35°C, 80 rmp) (Figures 1F–1H).
-
a.
-
3.Purification by column chromatography (1 h) (Figures 1F–1J).
-
a.Dilute the crude product with 10 mL dichloromethane, and add silica gel (3 gram, 200–300 mesh).
-
b.Evaporate the solvent under reduced pressure to afford a dry mixture of product with silica gel (-0.1 MPa, 35°C, 80 rmp) (Figures 1F and 1G).Note: Cotton is stuffed into the splash guard in case of silica splashing during rotary evaporation.
-
c.Add 25 g silica gel (200–300 mesh) to chromatography column (φ 17 mm, length 305 mm) and add petroleum ether subsequently.
-
d.Use air booster pump to accelerate the flow velocity to wet silica gel.
-
e.Load the crude product into chromatography column and add quartz sand (1 cm) subsequently.
-
f.Use petroleum ether (100%) to elute product (Figure 1I).
-
g.Use TLC (100% petroleum ether) combined with UV-light detection (254 nm and 365 nm) to analyze the fractions (Figure 1E).
-
h.Apply GC-MS to identify each fraction.
-
i.Collect the fractions containing product and remove the solvent by rotary evaporation (-0.1 MPa, 35°C, 80 rmp) to afford coupling product (Figure 1J).
-
j.Characterize the product by 1H-NMR,13C-NMR, MS and HRMS.
-
a.
Part 3. Set up Ni-catalyzed C(sp2)-C(sp) coupling of terminal alkynes and aryl bromides
Timing: 48 h
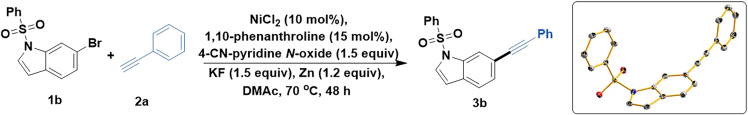
In this part, 6-(phenylethynyl)-1-(phenylsulfonyl)-1H-indole (3b) is prepared with nickel catalysis (Scheme 2). Twenty analogs have been produced via this method. The full scope of the transformation is described in Chen et al.1
Scheme 2.
Ni-catalyzed Sonogashira coupling for preparation of 3b
The method on nickel-catalyzed coupling of 6-bromo-1-(phenylsulfonyl)-1H-indole (1b) with phenylacetylene (2a) is same to the coupling of 4-(phenylethynyl)-1,1′-biphenyl (3a) with phenylacetylene (2a). The only difference is the temperature is elevated to 70°C (refer to Part 1).
Compound 3b is synthesized following the procedure outlined for compound 3a (vide supra, Part 1). However, the reaction temperature is increased to 70°C instead of 60°C.
Part 4. Purification of the crude material 6-(phenylethynyl)-1-(phenylsulfonyl)-1H-indole (3b)
Timing: 1.5 h
The operational procedure for purification of 3b is the same as for 3a (refer to Part 2). The difference is elution solvents are petroleum ether and ethyl acetate (30:1).
Part 5. Recrystallization
Timing: 4 days
In this section, a procedure for obtaining crystals of 6-(phenylethynyl)-1-(phenylsulfonyl)-1H-indole (3b) is described.
-
4.Set-up for recrystallization.
-
a.Weigh 3b (8 mg) on the electronic balance.
-
b.Dissolve 3b with dichloromethane (200 μL) in a 2 mL vial.
-
c.Add 1.5 mL n-hexane to the vial and ensure no 3b precipitate.Note: If 3b is precipitated when adding n-hexane, add as much DCM until 3b is again dissolved completely.
-
d.Fill another tube (φ 12 × 75 mm) with 2 mL n-hexane.
-
e.Transfer the vial with 3b into the tube (Figure 1K).
-
f.Seal the tube and keep them standing still in a shelf at 25°C.Note: The sample should be placed on a flat surface where it is not moved.
-
g.After evaporation slowly for 4 days, the colorless crystals are obtained (Figure 1L).
-
a.
Expected outcomes
4-(phenylethynyl)-1,1′-biphenyl (3a) is obtained as white powder in 65% yield. 6-(phenylethynyl)-1-(phenylsulfonyl)-1H-indole (3b) is obtained as pale yellow powder in 74% yield. 3b is recrystallized to afford the colorless solid (Figure 1L).
Quantification and statistical analysis
Analytical data
4-(phenylethynyl)-1,1′-biphenyl (3a).
1H NMR (400 MHz, CDCl3) δ 7.61–7.53 (m, 8H), 7.45 (t, J = 8.0 Hz, 2H), 7.36–7.33 (m, 4H).
13C NMR (100 MHz, CDCl3) δ 141.1, 140.5, 132.2, 131.8, 129.0, 128.5, 128.4, 127.8, 127.1, 123.4, 122.3, 90.2, 89.5.
EI-MS m/z calculated for [M]+ 254.11, found 254.15.
6-(phenylethynyl)-1-(phenylsulfonyl)-1H-indole (3b).
1H NMR (400 MHz, CDCl3) δ 8.26 (s, 1H), 7.93 (d, J = 8.0 Hz, 2H), 7.62–7.59 (m, 3H), 7.53–7.49 (m, 2H), 7.46–7.37 (m, 6H), 6.67 (d, J = 3.6 Hz, 1H).
13C NMR (100 MHz, CDCl3) δ 138.2, 134.7, 134.1, 131.7, 130.7, 129.5, 128.5, 128.4, 127.6, 127.1, 126.8, 123.3, 121.5, 119.6, 116.8, 109.4, 90.1, 89.5.
EI-MS m/z calculated for [M]+ 357.08, found 357.00.
HRMS m/z calculated for [M + H]+ 358.0902, found 358.0902.
Limitations
Aryl halides are limited to aryl iodide and bromide. When aryl chloride is employed as substrate, the reaction temperature is required to be elevated to 120°C, generating coupling product in an extremely low yield (< 5% yield). Although aryl triflates are tolerated, coupling products are obtained in low yield (< 20% yield).
Troubleshooting
Problem 1
Nickel chloride (anhydrous) is sensitive to water (step 1).
Potential solution
-
•
Store NiCl2 in a vacuum desiccator with allochroic silica gel as the desiccant. Use a vacuum pump to keep the desiccator under vacuum before use.
-
•
Store NiCl2 in nitrogen-filled glovebox if available.
Problem 2
Zinc powder is sensitive to air (step 1).
Potential solution
-
•
Store Zn in a vacuum desiccator with allochroic silica gel as the desiccant. Use a vacuum pump to keep the desiccator under vacuum before use.
-
•
Store Zn in nitrogen-filled glovebox if available.
-
•
Store Zn in a flame dried Schlenk flask under argon or nitrogen.
Problem 3
N,N-dimethylacetamide (DMAc) is sensitive to water (step 1).
Potential solution
-
•
Add activated 3Å molecular sieve to the super dry DMAc and store the solvent in a sealed bottle.
-
•
Store DMAc in nitrogen-filled glovebox if available.
Problem 4
N,N-dimethylacetamide (DMAc) is need to be degassed (step 1).
Potential solution
-
•
A 250 mL single-neck flask is charged with 230 mL of DMAc. The solvent can be degassed by repeated sonication under vacuum. Subject the flask to vacuum conditions while sonicating for 5 min and then replenishing the flask with N2. After 5–10 cycles, degassed DMAc is achieved. Transfer and store the solvent in a sealed bottle.
Problem 5
N,N-dimethylacetamide cannot be removed completely (step 2).
Potential solution
-
•
When the reaction is quenched, pour the mixture into ethyl acetate (100 mL). Use water (25 mL × 2) and brine (25 mL × 2) to wash the organic layer, respectively.
Problem 6
Yields are lower than expected (step 3).
Potential solution
-
•
This protocol is water and air sensitive; therefore, ensure the solvent is extra dry (water < 50 ppm).
-
•
When the reaction is conducted in glovebox, ensure the content of water and oxygen of it is lower than 10 ppm.
Resource availability
Lead contact
Further information and requests for resources and reagents should be directed to and will be fulfilled by the lead contact, Xuebin Liao (liaoxuebin@mail.tsinghua.edu.cn).
Technical contact
Further information and requests for technical support will be fulfilled by technical contact, Xuebin Liao (liaoxuebin@mail.tsinghua.edu.cn).
Materials availability
All other data supporting the finding of this study are available within the article reported by Chen et al.1 or from the lead contact upon reasonable request.
Data and code availability
All data reported in this paper will be shared by the lead contact upon request.
This paper does not report the original code.
Any additional information required to analyze the data reported in this paper is available from the lead contact upon reasonable request.
Acknowledgments
We are grateful for financial support from the National Natural Science Foundation of China (NSFC) (No. 21877067), China Postdoctoral Science Foundation (BX20220163) (2023M732005), and Postdoctoral Foundation of Tsinghua-Peking Center for Life Science. We thank Dr. Bei Zhang at Nanjing University of Information Science and Technology for helpful discussion.
Author contributions
H.C. designed and wrote the protocol with inputs from all the authors. H.C. and Z.A. performed the experiments and collected data. X.L. supervised the whole project.
Declaration of interests
The authors declare no competing interests.
References
- 1.Chen H., Yao L., Guo L., Liu Y.A., Tian B., Liao X. Coupling of alkynes and aryl halides with nickel-catalyzed Sonogashira reactions. Cell Reports Physical Science. 2023;4 [Google Scholar]
- 2.Myers A.G., Glatthar R., Hammond M., Harrington P.M., Kuo E.Y., Liang J., Schaus S.E., Wu Y., Xiang J.N. Development of an enantioselective synthetic route to neocarzinostatin chromophore and its use for multiple radioisotopic incorporation. J. Am. Chem. Soc. 2002;124:5380–5401. doi: 10.1021/ja012487x. [DOI] [PubMed] [Google Scholar]
- 3.Shear N.H., Villars V.V., Marsolais C. Terbinafine: an oral and topical antifungal agent. Clin. Dermatol. 1991;9:487–495. doi: 10.1016/0738-081x(91)90077-x. [DOI] [PubMed] [Google Scholar]
- 4.Gung B.W. Total synthesis of polyyne natural products. C. R. Chim. 2009;12:489–505. [Google Scholar]
- 5.Zhou T., Commodore L., Huang W.S., Wang Y., Thomas M., Keats J., Xu Q., Rivera V.M., Shakespeare W.C., Clackson T., et al. Structural mechanism of the pan-BCR-ABL inhibitor ponatinib (AP24534): lessons for overcoming kinase inhibitor resistance. Chem. Biol. Drug Des. 2011;77:1–11. doi: 10.1111/j.1747-0285.2010.01054.x. [DOI] [PubMed] [Google Scholar]
- 6.Huang W.S., Metcalf C.A., Sundaramoorthi R., Wang Y., Zou D., Thomas R.M., Zhu X., Cai L., Wen D., Liu S., et al. Discovery of 3-[2-(imidazo [1, 2-b] pyridazin-3-yl) ethynyl]-4-methyl-N-{4-[(4-methylpiperazin-1-yl) methyl]-3-(trifluoromethyl) phenyl} benzamide (AP24534), a potent, orally active pan-inhibitor of breakpoint cluster region-abelson (BCR-ABL) kinase including the T315I gatekeeper mutant. J. Med. Chem. 2010;53:4701–4719. doi: 10.1021/jm100395q. [DOI] [PubMed] [Google Scholar]
- 7.Zhu X., Liu J., Zhang W. De novo biosynthesis of terminal alkyne-labeled natural products. Nat. Chem. Biol. 2015;11:115–120. doi: 10.1038/nchembio.1718. [DOI] [PubMed] [Google Scholar]
- 8.Ren X., Pan X., Zhang Z., Wang D., Lu X., Li Y., Wen D., Long H., Luo J., Feng Y., et al. Identification of GZD824 as an orally bioavailable inhibitor that targets phosphorylated and nonphosphorylated breakpoint cluster region–abelson (Bcr-Abl) kinase and overcomes clinically acquired mutation-induced resistance against imatinib. J. Med. Chem. 2013;56:879–894. doi: 10.1021/jm301581y. [DOI] [PubMed] [Google Scholar]
- 9.Bastos M.M., Costa C.C.P., Bezerra T.C., da Silva F.d.C., Boechat N. Efavirenz a nonnucleoside reverse transcriptase inhibitor of first-generation: approaches based on its medicinal chemistry. Eur. J. Med. Chem. 2016;108:455–465. doi: 10.1016/j.ejmech.2015.11.025. [DOI] [PubMed] [Google Scholar]
- 10.Li X., Lv J.M., Hu D., Abe I. Biosynthesis of alkyne-containing natural products. RSC Chem. Biol. 2021;2:166–180. doi: 10.1039/d0cb00190b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Marchand J.A., Neugebauer M.E., Ing M.C., Lin C.I., Pelton J.G., Chang M.C.Y. Discovery of a pathway for terminal-alkyne amino acid biosynthesis. Nature. 2019;567:420–424. doi: 10.1038/s41586-019-1020-y. [DOI] [PubMed] [Google Scholar]
- 12.Kolb H.C., Sharpless K.B. The growing impact of click chemistry on drug discovery. Drug Discov. Today. 2003;8:1128–1137. doi: 10.1016/s1359-6446(03)02933-7. [DOI] [PubMed] [Google Scholar]
- 13.Kolb H.C., Finn M.G., Sharpless K.B. Click chemistry: diverse chemical function from a few good reactions. Angew. Chem. Int. Ed. 2001;40:2004–2021. doi: 10.1002/1521-3773(20010601)40:11<2004::AID-ANIE2004>3.0.CO;2-5. [DOI] [PubMed] [Google Scholar]
- 14.Debets M.F., van Hest J.C.M., Rutjes F.P.J.T. Bioorthogonal labelling of biomolecules: new functional handles and ligation methods. Org. Biomol. Chem. 2013;11:6439–6455. doi: 10.1039/c3ob41329b. [DOI] [PubMed] [Google Scholar]
- 15.Trotuş I.T., Zimmermann T., Schüth F. Catalytic reactions of acetylene: a feedstock for the chemical industry revisited. Chem. Rev. 2014;114:1761–1782. doi: 10.1021/cr400357r. [DOI] [PubMed] [Google Scholar]
- 16.Hein C.D., Liu X.M., Wang D. Click chemistry, a powerful tool for pharmaceutical sciences. Pharm. Res. (N. Y.) 2008;25:2216–2230. doi: 10.1007/s11095-008-9616-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Yi G., Son J., Yoo J., Park C., Koo H. Application of click chemistry in nanoparticle modification and its targeted delivery. Biomater. Res. 2018;22:13–18. doi: 10.1186/s40824-018-0123-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Xi W., Scott T.F., Kloxin C.J., Bowman C.N. Click chemistry in materials science. Adv. Funct. Mater. 2014;24:2572–2590. [Google Scholar]
- 19.Nair P.P., Philip R.M., Anilkumar G. Nickel catalysts in Sonogashira coupling reactions. Org. Biomol. Chem. 2021;19:4228–4242. doi: 10.1039/d1ob00280e. [DOI] [PubMed] [Google Scholar]
- 20.Naeimi H., Kiani F. Functionalized graphene oxide anchored to Ni complex as an effective recyclable heterogeneous catalyst for Sonogashira coupling reactions. J. Organomet. Chem. 2019;885:65–72. [Google Scholar]
- 21.Zhu D.L., Xu R., Wu Q., Li H.Y., Lang J.P., Li H.X. Nickel-catalyzed Sonogashira C(sp)−C(sp2) coupling through visible-light sensitization. J. Org. Chem. 2020;85:9201–9212. doi: 10.1021/acs.joc.0c01177. [DOI] [PubMed] [Google Scholar]
- 22.Arundhathi K.V., Vaishnavi P., Aneeja T., Anilkumar G. Copper-catalyzed Sonogashira reactions: advances and perspectives since 2014. RSC Adv. 2023;13:4823–4834. doi: 10.1039/d2ra07685c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Nowrouzi N., Zarei M. NiCl2 6H2O: an efficient catalyst precursor for phosphine-free Heck and Sonogashira cross-coupling reactions. Tetrahedron. 2015;71:7847–7852. [Google Scholar]
- 24.Helbert H., Visser P., Hermens J.G.H., Buter J., Feringa B.L. Palladium-catalysed cross-coupling of lithium acetylides. Nat. Catal. 2020;3:664–671. [Google Scholar]
- 25.Monnier F., Turtaut F., Duroure L., Taillefer M. Copper-catalyzed Sonogashira-type reactions under mild palladium-free conditions. Org. Lett. 2008;10:3203–3206. doi: 10.1021/ol801025u. [DOI] [PubMed] [Google Scholar]
- 26.Tasker S.Z., Standley E.A., Jamison T.F. Recent advances in homogeneous nickel catalysis. Nature. 2014;509:299–309. doi: 10.1038/nature13274. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data reported in this paper will be shared by the lead contact upon request.
This paper does not report the original code.
Any additional information required to analyze the data reported in this paper is available from the lead contact upon reasonable request.