Abstract
People with Parkinson’s disease (PwPD) can benefit from progressive high-intensity exercise facilitated with a lower-extremity exoskeleton, but the mechanisms explaining these benefits are unknown. We explored the relationship between exercise intensity progression and memory and gait outcomes in PwPD who performed 8 weeks (2 × per week) of progressive exercise with and without a lower-extremity powered exoskeleton, as the planned exploratory endpoint analysis of an open-label, parallel, pilot randomized controlled trial. Adults 50–85 years old with a confirmed diagnosis of PD participated. Twenty-seven participants randomized to exercise with (Exo = 13) or without (Nxo = 14) the exoskeleton were included in this exploratory endpoint analysis. Detailed exercise logs were kept and actigraphy was used to measure activity count*min−1 (ACPM) during all exercise sessions. Only the Exo group were able to progressively increase their ACPM over the entire 8-week intervention, whereas the Nxo group plateaued after 4 weeks. Exercise intensity progression correlated with change in the memory sub-scale of the SCOPA-COG and change in gait endurance from the 6MWT, consistent with the prevailing hypotheses linking high-intensity interval exercise to improved muscle and brain function via angiogenic and neurotrophic mechanisms. Facilitating high-intensity exercise with advanced rehabilitation technology is warranted for improving memory and gait endurance in PwPD.
Registration: ClinicalTrials.gov, NCT 03583879 (7/10/2018).
Subject terms: Parkinson's disease, Rehabilitation
Introduction
Despite the growing body of evidence1–6 supporting exercise as a low-cost and accessible treatment to slow progression of motor and non-motor impairments in people with Parkinson’s disease (PwPD), participation in exercise programs remains a challenge for PwPD7–10. High-intensity exercise in particular holds considerable promise as a breakthrough intervention for many health ailments11–15 including Parkinson’s disease1,2. Although the exercise-induced mechanisms that work to improve body and brain function are becoming better understood in animals and humans16–18, the biggest challenge is how to deliver high-intensity exercise interventions to people who can benefit from them but whose condition is a barrier to participating in them.
There are many reasons why PwPD may find it difficult to participate in high-intensity exercise interventions, such as deconditioning, fear of falling, low self-efficacy, low expectations, and decreased motivation3,19,20. Solutions are needed for facilitating the delivery of engaging, high-intensity exercise to reduce disability in PwPD 21. Examples such as the Park-in-Shape trial1—a home-based, gamified, high-intensity aerobic exercise program, and the SPARX trial2—a high-intensity treadmill-based walking program, have both shown reduced disease progression on the UPDRS motor III subscale after 6 months of training compared to the control group, but no change in mobility and cognitive functioning was reported over the study period.
Robot-assisted gait training (RAGT) – using stationary treadmill-based robotic systems – offers another potential solution for facilitating gait training interventions in people with disability22,23. Although this approach allows for multiple training cycles with controlled levels of assistance, research thus far shows that functional gains are not superior to treadmill training24–29. It may be that this type of robot assisted therapy where the machine does all the work may not elicit a high enough metabolic cost to trigger the mechanisms that induce exercise-derived benefits; indeed, the vascular and neuroprotective benefits of exercise requires increasing metabolic demand18,30.
Wearable, overground exoskeletons are a broad class of powered orthoses that range from rigid multi-joint actuating robots that can enable standing and walking at limited speed in people with complete paralysis31–33, to rigid single-joint actuating exoskeletons34–37 to assist with mobility, to single- and multi-joint soft exoskeleton suits that can assist the user dynamically and reduce metabolic demand during walking in people with disability38–40. Although each may have their place in the continuum of rehabilitation and restoration, there are few commercial devices suitable for facilitating high-intensity exercise. Such a device must offer dynamic stability, so that users feel safe and confident while using the device during a variety of exercises (ie. beyond stationary walking), while at the same time allowing them to work harder and expend more energy during those exercises than they otherwise would be able to.
The Keeogo Rehab™ exoskeleton, which was used in our study, is one commercial device that has the capability of providing dynamic stability and minimally necessary assistance during common exercise activities such as marching in place, lunges, step ups, walking and stair climbing that can be performed in a controlled environment such as physio gym or clinic. We previously reported41 on a randomized controlled trial (RCT) to determine if 8 weeks (2 × per week) of bilateral exoskeleton (Exo) exercise (aerobic, strengthening, and functional) with the Keeogo Rehab™ exoskeleton results in positive changes in cognition, mood, gait, balance and health-related quality of life in adults with PD compared to exercising without an exoskeleton (Nxo) or wait-list control (Con). Participants in the Exo group improved significantly in the memory sub-scale of the SCOPA-COG and in the 6-min walk test (6MWT) following 8 weeks of twice-weekly exoskeleton assisted exercise compared to the Nxo and Con groups.
The planned exploratory endpoint of the trial was quantification of exercise intensity during the therapy sessions via actigraphy in the two exercise groups, and to study its relationship to trial outcomes. Although unavailable at the time of conducting this pilot RCT, Jeng et al.42 have recently published a cut-point of 1354 ACPM as the threshold between moderate and high intensity exercise for PwPD, thus allowing us to quantify the degree to which our study participants were able to exercise at an intensity greater than this threshold, and what role, if any, these outcomes play in explaining the observed effects on memory and gait endurance.
As such, this paper explores a mechanistic explanation for the positive changes in memory and gait reported in our main outcomes paper41. We specifically focus here on the relationships between exercise intensity and outcomes on the memory & learning sub-scale of the SCOPA-COG and the 6MWT for gait endurance for the two interventions that received exercise: the Exo group (exercise with the exoskeleton) and Nxo (exercised with the exoskeleton). Our prior work41 showed that changes in the memory & learning domain of the SCOPA-COG were largely responsible for changes in the overall cognition score, and that the 6MWT was much more responsive than preferred and fast gait walking trials. The following exploratory hypotheses were tested:
Did the Exo group achieve a higher exercise intensity (ACPM) during the exercise sessions compared to the Nxo group?
Did the Exo group exceed a threshold of 1354 ACPM during exercise sessions proportionally more than the Nxo group?
Does progression in exercise intensity correlate with changes in memory and gait outcomes after 8 weeks of twice-weekly exercise?
Methods
The pilot RCT was registered prospectively at ClinicalTrials.gov (NCT 03583879, 7/10/2018). The study took place at the Assistive Technology Clinic (ATC), Baycrest Hospital, Toronto, Ontario, Canada, between Sept. 2018 and Oct. 2019, and was approved by all relevant Research Ethics Boards (Baycrest, Toronto, ON, and University of New Brunswick, Fredericton, NB). The study was conducted in accordance with the Declaration of Helsinki, and all participants provided signed informed consent prior to enrollment. Details of participants, recruitment, randomization, outcomes measures and interventions are available in the main outcomes paper41.
Assessments of memory and gait
For the present study, based on our preliminary evidence41, we focused on relationships between exercise intensity during the intervention and the observed change scores in the “Memory and Learning” sub-scale of the SCOPA-COG, and the 6MWT for gait endurance.
Scales for Outcomes in Parkinson’s-Cognition (SCOPA-COG) is a validated 10 task assessment for quantifying cognitive functioning in PwPD43,44. Domains include attention, memory, executive function, delayed recall and visuospatial impairment45. The “Memory and Learning” sub-scale has a range of 0–22, where higher scores indicate better cognitive functioning in the memory and learning domain.
The 6MWT (distance in meters) was used to evaluate gait function and endurance46, and has been reported as reliable and/or valid in many patient populations47. Participants completed the test along a 25 m walkway. The test was repeated three times, and the score was the average of three repetitions. For this test, higher scores (distance walked) indicate better gait endurance.
Actigraphy during intervention exercises
To date there is no accepted standard for assessing exercise intensity in a clinical setting during the interventions that are being administered. While indirect calorimetry is commonly employed in studies for objectively quantifying pre-post VO2 peak1,48, the cart and respirator would make it cumbersome to measure exercise intensity during overground functional exercises and would likely interfere with the delivery of the intervention. Heart rate measures are a common proxy measure of exercise intensity2,49, but may not be appropriate for accurate real-time measurement50. Actigraphy using small, unobtrusive wearable sensors is a portable and low-cost alternative to indirect calorimetry42,51,52, therefore was ideal for the purpose of quantifying exercise intensity while participants performed the exercise sessions.
Participants allocated to Exo or Nxo exercise groups visited the clinic twice-weekly for 8-weeks, for up to a total of 16 sessions. The session activities are listed in Table 1. The Exo group performed all sessions whilst wearing the exoskeleton device (fit and tuned to the user as described elsewhere41), whereas the Nxo group performed the exercise sessions in the standard of care fashion, without the exoskeleton. During the first half of the intervention (T1) all participants progressed every week to higher reps/durations and reaching their max capacity, which was then held for the remaining half of the intervention (T2). The purpose of holding the dose mid-way was to enable us to quantify progressive intensity in the 2nd half of the intervention that would be dose independent.
Table 1.
Exercise program used in the intervention.
| # | Type | Exercise | Rank | Week 1 2 sessions |
Week 2 2 sessions |
Week 3 2 sessions |
Week 4 2 sessions |
Week 5–8 2 sessions |
|---|---|---|---|---|---|---|---|---|
| T1–1st half | T2–2nd half | |||||||
| 1 | Warm up | Walking warm up | 1 | 5 min | 5 min | 5 min | 5 min | 5 min |
| 2 | Strength | Squat/ "sit to stands" | 6 | 6 reps × 3 sets | 8 reps × 3 sets | 10 reps × 3 sets | 12 reps × 3 sets | 12 reps × 3 sets |
| 3 | Static lunges | 7 | 6 reps × 3 sets, per side | 8 reps × 3 sets, per side | 10 reps × 3 sets, per side | 12 reps × 3 sets, per side | 12 reps × 3 sets, per side | |
| 4 | Heel raises | 2 | 6 reps × 3 sets | 8 reps × 3 sets | 10 reps × 3 sets | 12 reps × 3 sets | 12 reps × 3 sets | |
| 5 | Aerobic | Walking HIIT | 10 | 0.5 min fast, 0.5 min slow, 1.0 min rest, 5 sets | 1.0 min fast, 1.0 min slow, 1.0 min rest, 4 sets | 1.5 min fast, 1.5 min slow, 1.0 min rest, 3 sets | 2.0 min fast, 2.0 min slow, 1.0 min rest, 2 sets | 2.0 min fast, 2.0 min slow, 0.5 min rest, 3 sets |
| 6 | Functional Mobility | Stairs ascent and descent | 9 | 1 flight, 1 min rest | 2 flights, 1 min rest | 3 flights, 30 s rest | 4 flights, 1 min rest | 4 flights, 30 s rest |
| 7 | Backwards stepping | 4 | 10 reps × 3 sets | 12 reps × 3 sets | 14 reps × 3 sets | 16 reps × 3 sets | 16 reps × 3 sets | |
| 8 | Sidestepping (each direction) | 5 | 10 reps × 3 sets, per side | 12 reps × 3 sets, per side | 14 reps × 3 sets, per side | 16 reps × 3 sets, per side | 16 reps × 3 sets | |
| 9 | Step ups, to step downs (lead w/ each side) | 8 | 6 reps × 3 sets, per side | 8 reps × 3 sets, per side | 10 reps × 3 sets, per side | 12 reps × 3 sets, per side | 12 reps × 3 sets, per side | |
| 10 | Balance | Stride stance | 1 | 10 s × 3 sets, per side | 12 s × 3 sets, per side | 14 s × 3 sets, per side | 16 s × 3 sets, per side | 18 s × 3 sets, per side |
| 11 | Forward to reverse step, hands free | 1 | 10 reps × 3 sets, per side | 12 reps × 3 sets, per side | 14 reps × 3 sets, per side | 16 reps × 3 sets, per side | 16 reps × 3 sets, per side | |
| 12 | PNF D2 extension in stride stance | 3 | 10 reps × 3 sets, per side | 12 reps × 3 sets, per side | 14 reps × 3 sets, per side | 16 reps × 3 sets, per side | 16 reps × 3 sets, per side | |
| 13 | Downward reach to floor | 1 | 10 reps × 3 sets | 12 reps × 3 sets | 14 reps × 3 sets | 16 reps × 3 sets | 16 reps × 3 sets | |
| 14 | Forward weight shift to upward reach | 1 | 10 reps × 3 sets, per side | 12 reps × 3 sets, per side | 14 reps × 3 sets, per side | 16 reps × 3 sets, per side | 16 reps × 3 sets, per side | |
| 15 | Obstacle course | 4 | 2 min | 4 min | 5 min | 6 min | 7 min | |
HIIT High intensity interval training, PNF D2 Proprioceptive neuromuscular facilitation, “draw sword” shoulder extension.
Detailed exercise logs were kept that tracked each participant’s progress. Dopamine medication timing was also tracked to ensure that all Participants were in the “On” state during intervention sessions.
All participants wore the IMU (GT9X Link, Actigraph Inc., Pensacola, FL) on their right hip during the entire exercise session. The IMU was first wiped of any existing data and then initialized with the participant’s unique ID. The therapist then placed the device on the participant’s hip using a waist-band clip immediately before the session began. After the session the therapist removed the device from the clip holder and immediately docked the device for uploading the data to the Actilife software.
Actigraphy data processing
After the conclusion of the study the raw files for the 27 participants with up to 16 sessions each were exported as activity count data to csv files and further processed with custom written Matlab algorithms to calculate activity counts per minute (ACPM) by summing the activity counts across the time interval of the session (with leading and trailing noise removed) and dividing by the total time in minutes. Consistent with the recommendation from Jeng et al.42 we included only the vertical channel accelerations. ACPM data were further normalized to body mass (ACPM/kg) to account for the mass being moved (ie. the participants’ mass plus the exoskeleton, if applicable).
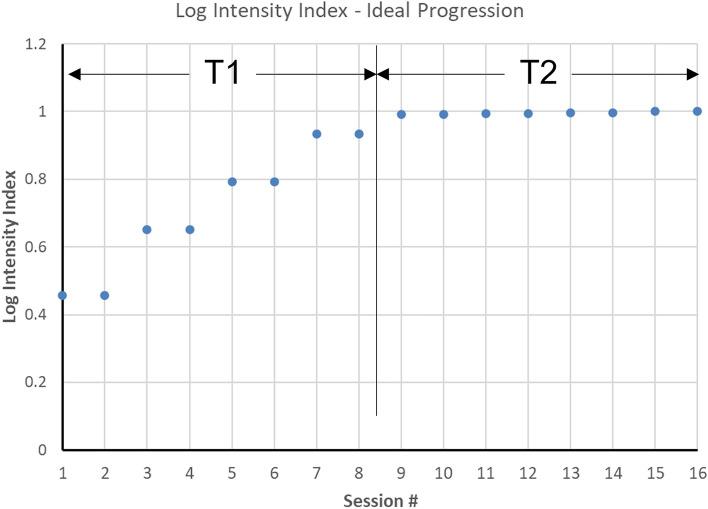
In addition to actigraphy analysis, participants’ exercise logs which contained the actual numbers of sets and intervals for each element of the intervention (Table 1) were transformed into a relative “Log intensity index” for the purpose of comparing how the exercise intervention was delivered to the two treatment groups. We first ranked by consensus all the exercise activities in the intervention by their expected level of vigor (see column 4 of Table 1), multiplied those ranks by the maximum number of minutes, repetitions and sets planned for each activity, and summed these to arrive at a prescribed maximum score if the participant performed all activities as scheduled. The algorithm was then applied to each participants’ actual session log data and total scores divided by the prescribed maximum score to arrive at an “index” representing the degree to which the participant achieved the planned dose of exercise in each session. Using the data in Table 1, the prescribed log intensity index was computed and is illustrated in Fig. 1.
Figure 1.
Log intensity index derived from the exercise progression prescription for an ideal hypothetical example where all reps and sets are completed according to the progression schedule. T1 refers to the first half (0–4 weeks) of the intervention which is the progression phase, and T2 refers to the last half (5–8 weeks) of the intervention which is the maintenance phase.
Statistical analysis
To answer the first research question, intensity metrics derived from actigraphy and from the exercise log sheet data were reduced to biweekly averages (for display) and means for intervention intervals T1 and T2, which were then compared between the Exo and Nxo groups using a 2-tailed independent samples t-test.
To answer the second research question, a 2 × 2 Chi-square cross-tabulation analysis was used to compare the frequencies of sessions for Exo and Nxo groups where the participant exceeded the ACPM threshold of 135442.
To answer the third research question, Pearson correlation analysis was conducted to test if there was any relationship between progression in exercise intensity (T2–T1 scores) and memory and gait change scores.
Descriptive statistics were used to describe the sample, which included age, sex, height and weight (as measured in the clinic), body mass index (BMI), Hoehn & Yahr PD disease stage (H&Y), and the Montreal Cognitive Assessment (MoCA).
All statistical analysis were conducted using SPSS (IBM Inc.), with significance set at α = 0.05.
Ethical approval and informed consent
The study was approved by research ethics boards of Baycrest Hospital, Toronto, Ontario (REB #18-24) and the University of New Brunswick, Fredericton, New Brunswick (REB #2018-136). All participants provided signed informed consent prior to enrollment in the study.
Results
Data were analyzed from the twenty-seven participants (Exo n = 13, Nxo n = 14) who received exercise interventions. This sample was 69.2 ± 6.7 years of age and consisted of 11 men (41%) and 16 women (59%). Average BMI was 26.6 ± 5.8 kg/m2. Participants were distributed across Hoehn & Yahr disease stages I (40.7%), II (29.6%), III (25.9%), and IV (3.7%). MoCA score for the sample was 24.2 ± 3.5 points. These data are shown in Table 2.
Table 2.
Participants in the exploratory study.
| Scale variables | Total | Group A–Exo | Group B–Nxo | |||
|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | Mean | SD | |
| Age (years) | 69.2 | 6.7 | 67.6 | 5.9 | 70.7 | 7.3 |
| Weight (kg) | 74.0 | 18.2 | 71.4 | 18.4 | 76.4 | 18.4 |
| Height (cm) | 166.4 | 8.4 | 165.4 | 9.3 | 167.4 | 7.7 |
| BMI (kg*m−2) | 26.6 | 5.8 | 26.0 | 5.8 | 27.2 | 5.9 |
| MoCA | 24.2 | 3.5 | 25.2 | 3.2 | 23.2 | 3.6 |
| Cat. variables | Count | % | Count | % | Count | % |
|---|---|---|---|---|---|---|
| Sex | ||||||
| Male | 11 | 40.7 | 4 | 30.8 | 7 | 50.0 |
| Female | 16 | 59.3 | 9 | 69.2 | 7 | 50.0 |
| H&Y | ||||||
| Stage I | 11 | 40.7 | 4 | 30.8 | 7 | 50.0 |
| Stage II | 8 | 29.6 | 4 | 30.8 | 4 | 28.6 |
| Stage III | 7 | 25.9 | 5 | 38.5 | 2 | 14.3 |
| Stage IV | 1 | 3.7 | 0 | 0.0 | 1 | 7.1 |
Exo Exoskeleton exercise training, Nxo Exercise training without exoskeleton, BMI Body mass index, MoCA Montreal Cognitive Assessment, H&Y Hoehn & Yahr PD disease stage.
Memory and gait outcomes were analyzed in our main outcomes paper41 across the three treatment groups (Exo, Nxo and Con). Here we show the analysis between just the two exercise groups included in this study (Exo vs Nxo). Memory sub-scale of the SCOPA-COG and 6MWT results are shown Table 3. There were no significant differences (p > 0.05) in baseline scores for either measure. Post–pre comparison showed that the Exo group had a significant improvement in memory score (p = 0.035) and 6MWT score (p < 0.001) compared to the Nxo group, consistent with our previous report that included the waitlist control (Con) group41. Also previously unreported was a significant correlation between the 6MWT and SCOPA-COG memory change scores (r = 0.675, p = 0.001) across the sample of participants in the active exercise interventions (Exo and Nxo), as shown by the overlay scatter plot in Fig. 2.
Table 3.
Memory and gait scores for trial participants in the exoskeleton exercise group (Exo) and the standard of care exercise group (Nxo).
| Exo | Nxo | Baseline | Post–Pre | |
|---|---|---|---|---|
| Mean (SD) | Mean (SD) | p-value† | ||
| SCOPA-COG memory | N = 13 | N = 14 | ||
| Baseline | 8.3 (2.8) | 9.4 (3.5) | .401 | .035* |
| Post study | 11.2 (2.4) | 9.7 (3.3) | ||
| Change | 2.9 (2.4) | 0.4 (3.1) | ||
| p-value‡ | .001** | .669 | ||
| 6-Min. Walk Test | N = 10 | N = 11 | ||
| Baseline | 374.4 (78.9) | 369.3 (122.0) | .911 | < .001* |
| Post study | 409.3 (90.8) | 367.9 (126.3) | ||
| Change | 34.8 (17.6) | -1.4 (20.4) | ||
| p-value‡ | < .001** | .822 | ||
SCOPA-COG memory = Scales for Outcomes in Parkinson’s-Cognition, memory & learning sub-scale.
†Between-groups comparisons: Baseline comparison with one-way ANOVA; Change score comparison controlling for baseline with one-way ANCOVA. *Between-groups change is significant at p < .05.
‡Within-subjects comparisons: Pre-post comparison with t-test and Bonferroni correction.
Figure 2.
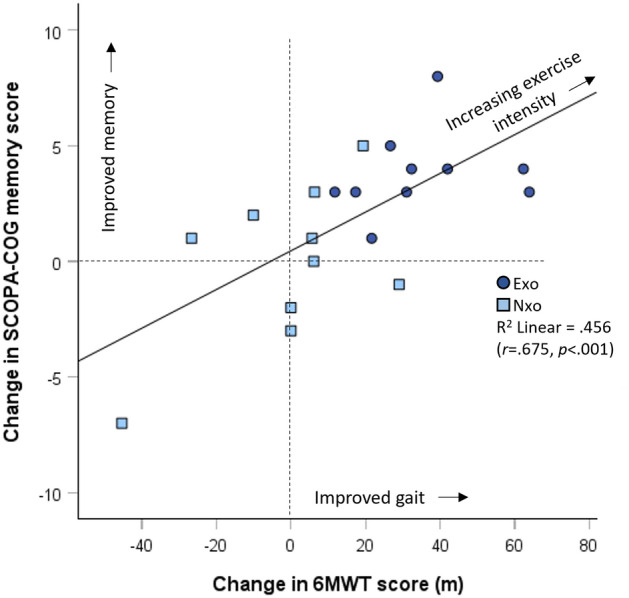
Overlay scatter plot showing the relationship between change in gait endurance as measured by the 6-min walk test (6MWT) and change in memory as measured by the SCOPA-Cog Memory and Learning sub-scale. Light blue boxes represent participants who exercised without the exoskeleton (Nxo) and the dark blue circles represent the participants who exercised with the exoskeleton (Exo). Vertical and horizontal dashed lines demark the zero-change lines.
Question 1: Did the Exo group achieve a higher exercise intensity (ACPM) during the exercise sessions compared to the Nxo group?
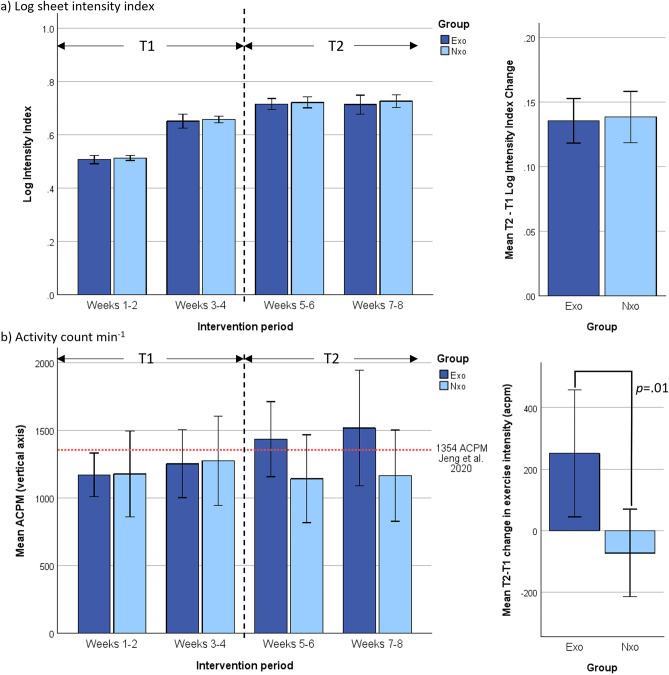
Log intensity index from log sheet data (Fig. 3a left) shows that participants in both groups progressed similarly, from about 50% to approximately 75% of the maximal prescribed intensity (ie. no. of reps and duration, etc.). Comparison of the progression from T1 and T2 (Fig. 3a right) between the groups likewise shows that both advanced the same in terms of increasing their sessional activities as prescribed (p > 0.05).
Figure 3.
Exercise dose measured two ways: (a) Log intensity index showing how well participants were on average able to progress according to the ideal prescription.; and (b) Mean activity counts per minute (ACPM) from actigraphy shows how intensely participants were on average able to exercise during the interventions. The left panels show the results averaged over 2-week intervals and the panels on the right show the mean difference between T1 and T2 phases.
In contrast, the actigraphy data (Fig. 3b left) shows that although participants were progressing similarly in prescribed sessional intensity, the activity count rate for the Exo group continued to increase for each bi-weekly interval, whereas exercise intensity for the Nxo group remained relatively unchanged during the trial. Comparison of the progression from T1 and T2 (Fig. 3b right) between the groups shows that Exo group increased their exercise intensity significantly (p = 0.01) compared to the Nxo group.
Question 2: Did the Exo group exceed a threshold of 1354 ACPM during exercise sessions proportionally more than the Nxo group?
As indicated by Fig. 3b (left), only the Exo group’s mean exceeded an exercise intensity above the cut-point threshold of 1354 ACPM, during the T2 period. Analysis of the sessional data showed that in 83 of the 180 total sessions (46%) received by the Exo group, and 64 of the 190 total sessions (34%) received by the Nxo group, participants exceeded the 1354 ACPM threshold during their session. Chi-square analysis showed a significant difference in these proportions (p = 0.015) where the Exo group had a higher-than-expected number of sessions that exceeded the 1354 ACPM threshold while the Nxo group had a lower-than-expected number of sessions that exceeded the threshold.
Question 3: Does progression in exercise intensity (T2-T1) correlate with changes in memory and gait outcomes after 8 weeks of twice-weekly exercise?
Figure 4 shows correlation analysis results of SCOPA-COG memory and 6MWT change scores versus the change in body mass normalized exercise intensity achieved between T1 and T2 of the intervention as measured by actigraphy. Change in exercise intensity between T1 and T2 correlated significantly with change in 6MWT scores and (r = 0.612, p = 0.004) and change in SCOPA-COG memory score (r = 0.388, p = 0.050).
Figure 4.
Scatter plots showing the relationship between body mass normalized change in exercise intensity (ACPM/kg) between T1 and T2 phases, with change in gait endurance from 6-min walk test (6MWT) (left panel) and change in SCOPA-Cog Memory and Learning sub-scale score (right panel). Light blue boxes represent participants who exercised without the exoskeleton (Nxo) and the dark blue circles represent the participants who exercised with the exoskeleton (Exo). Vertical and horizontal dashed lines demark the zero-change lines.
Given the significant correlations between change scores in 6MWT and the memory sub-scale of the SCOPA-COG, an ad-hoc partial correlation analysis was conducted to test if the change in exercise intensity was driving this relationship. The partial correlation between 6MWT and SCOPA-COG memory when controlling for ACPM/kg remained significant (r = 0.597, p = 0.007), meaning that exercise intensity did not explain this effect.
Discussion
The main objective of this paper was to explore the relationship between exercise intensity and memory and gait outcomes in PwPD who participated in 8 weeks (2 × per week) of progressive functional exercise with and without a lower extremity powered exoskeleton, with the goal of providing a plausible mechanistic explanation for the improvement in memory and gait endurance in the group that exercised with the exoskeleton device, compared to the group that exercised without the exoskeleton41.
We demonstrated that PwPD, who have moderate levels of disability, and exercised with the exoskeleton, can feasibly achieve high-intensity exercise (exceed 1354 ACPM) after a relatively short (4 weeks) progression period, and can continue to increase their intensity for at least another 4 weeks, as illustrated in Fig. 3. Achieving those levels of intensity were less frequent with the group that did not wear the exoskeleton, whose intensity levels tended to plateau at moderate-intensity levels. Importantly, our preliminary evidence also suggests that these effects can translate into improvement in important rehabilitation outcomes—memory and gait—which, as illustrated in Figs. 2 and 4, are interconnected. The implications of our findings, and the potential actions they warrant, will next be discussed.
How can exercising with an overground exoskeleton improve memory and gait?
Past research into robot-assisted gait training (RAGT) systems, which are typically stationary exoskeletons integrated with a treadmill or stepping mechanism, have for the most part failed to show functional gains in PwPD beyond what is achievable using the treadmill alone or other forms of manual gait training therapies for neurological conditions24–28. Why then would an overground exoskeleton be expected to yield results different from a stationary one?
If we consider the multi-system integration required for independent human overground locomotion, compared to being walked in a body weight supporting and laterally constraining mechanical apparatus, the non-body weight supporting overground exoskeleton (ie. does not support vertical load) should stress the biological system in ways the stationary robot cannot, which may elicit neurophysiological responses more appropriate for triggering the cascade of mechanisms that can lead to improved muscle and brain function16–18.
Our data suggests that exercising with an overground, non-bodyweight supporting, exoskeleton may facilitate achieving this goal by providing movement stability while preserving the need to work against gravity53. Because our actigraphy analysis focused on the vertical acceleration counts, we can conclude that participants wearing the device were able to increase their intensity of anti-gravity movements during the exercises, compared to those who exercised without the device.
Our data also show that these effects were not immediate, indicating that one month of progression was required before users of the exoskeleton were able to start achieving consistent exercise intensity levels defined (for the PD population) as “high-intensity”. To elaborate, Fig. 3b (left) provides some key observations of how the participants in our study responded to the two different modes of exercise delivery. During the progression (T1) phase of the intervention both groups exercised at similar sub-threshold intensity levels, and in fact the Nxo group had slightly better (though non-significant) levels of intensity. Participants in the Exo group would have been learning how the exoskeleton interacts with them. After the 4-week progression period (during T2), only the Exo group was able to continue to increase their exercise intensity level. Indeed, the slope of the bi-weekly intensity curve would suggest that, had the intervention been longer, the Exo participants may have continued to increase their levels of intensity beyond the 8-week period.
Indeed, the difference between T2 and T1 mean exercise intensities (in this case, monthly estimates) appeared to be an important indicator of benefits gained from the intervention. Data in Fig. 4 suggests that increasing exercise intensity between the T2 and T1 periods explained about 37% of the variance in 6MWT change scores, and about 15% of the variance in SCOPA-COG memory and learning change scores, when all exercising participants were included. Figure 4 also shows that members of the Exo group appear more frequently in the upper-right quadrant of the scatter plots, again illustrating that wearing the exoskeleton during the exercise interventions had an impact on trial outcomes.
Interestingly, although there was clearly a strong relationship between the 6MWT distance and SCOPA-COG memory and learning change scores, and both were related to T2-T1 ACPM change in exercise intensity, the partial correlation test failed: the relationship observed between change in gait endurance and change in cognition was not moderated by exercise intensity level. This suggests that mobility and cognition are linked via other neurophysiological mechanisms—ie. the mechanism exists despite exercise or exoskeletons—but that exercising at a sufficiently high intensity allows that mechanism to be exploited. In other words, the intensity of the exercise progression determines to some degree where one is positioned on the regression line in Fig. 2. What this means to practice is that it may not be the exoskeleton itself that is explaining the beneficial mechanisms at play, but what the exoskeleton facilitates (ie. novel learning experience plus high intensity interval training) that may be of particular importance to its place in rehabilitation.
More detailed dosing studies are needed that include traditional metabolics, blood biomarkers, and brain imaging, to properly discern how this, and possibly other rehabilitation technologies, interact with proangiogenic and neurotrophic mechanisms that are known to improve muscle and brain function. The role of learning to use a novel robotic technology to improve functioning, and the agency it may bring54, is also an area of future inquiry that could provide insights into cognitive benefits of novel training paradigms.
Research and practice limitations of robots for high-intensity exercise
Recent studies have demonstrated that 6-months of high-intensity treadmill walking (SPARX trial2) and similar length home-based exercise using simple, inexpensive, tools and interactive apps (Park-in-Shape trial1) can improve clinical (UPDRS) features of PD at long-term follow-up. Such interventions are considerably more accessible than what is proposed here, but it is also important to draw a distinction between the primary endpoints of these long-term studies, which was to slow disease progression in the long-term (as measured by UPDRS), compared to the endpoints of our study which was focused on shorter-term outcomes in mood, memory and gait. These observations are not necessarily at odds, nor are the different approaches incompatible with one another. A possible recommendation stemming from the available data would be short-term high-intensity robot training to “pre-condition” or “top-up” patients prior to and during more accessible and less expensive longer-term interventions.
Nevertheless, overground exoskeletons of the type studied here still represent a highly inaccessible technology, which poses an immediate barrier for translating research like ours into practice. Most importantly, practice guidelines must be developed that are evidence based. Exoskeletons vary in their construction (ie. passive vs. active, rigid vs. soft) and are expensive and difficult to study. In addition, skilled clinicians with expertise in using these devices must be resident in the clinic to conduct research like this, let alone deliver services with the technology. This study is exemplary and perhaps unique in this way, as the research was initiated and driven by practicing therapists at the ATC who had the technology and wanted to know how to best utilize it. Generally speaking, studies like this in advanced rehabilitation technology are rare. Developers need to employ better knowledge translation practices so that their technology’s place in routine clinical practice is driven by the users and recipients.
Conclusion
We conclude that high-intensity rehabilitation with an overground exoskeleton can translate into improvement in important rehabilitation outcomes, such as memory and gait, in PwPD. The take home message is that technologies suitable for rehabilitation must be able to increase metabolic demand, not reduce it, while at the same time providing enough stability and constraint to build confidence and self-efficacy, that feeds back to engagement and motivation to participate in high intensity exercise. Our data from this study and others suggest that overground powered lower-extremity exoskeletons may possess this capability and could represent a paradigm shift for delivering short, high-intensity functional training programs to PwPD and other movement disorders.
Acknowledgements
The authors are indebted to Albie Law and Lauren Binstock for their assistance in recruiting, training and assessments, and to the study participants who volunteered their time to make this study possible.
Author contributions
P.G., C.M. and A.S. acquired the funding, co-designed the study protocol, and conceived the research questions. P.G. managed the intervention delivery. A.S. managed the project operations. C.M. conducted the statistical analysis and wrote the first draft of the manuscript. All authors reviewed, edited and approved the final version of the manuscript.
Funding
This work was funded by the Centre for Aging and Brain Health Innovation (CABHI) under the Industry Innovation Partnership Program (I2P2). The industry partner (BTemia Inc.) provided $50,000 cash and in-kind (exoskeleton rental) support.
Availability of data material
All data are provided in this manuscript and tables.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.van der Kolk NM, et al. Effectiveness of home-based and remotely supervised aerobic exercise in Parkinson’s disease: A double-blind, randomised controlled trial. Lancet Neurol. 2019;18:998–1008. doi: 10.1016/S1474-4422(19)30285-6. [DOI] [PubMed] [Google Scholar]
- 2.Schenkman M, et al. Effect of high-intensity treadmill exercise on motor symptoms in patients with de novo Parki:nson disease: A phase 2 randomized clinical trial. JAMA Neurol. 2018;75:219–226. doi: 10.1001/jamaneurol.2017.3517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Shulman LM, et al. Randomized clinical trial of 3 types of physical exercise for patients with Parkinson disease. JAMA Neurol. 2013;70:183–190. doi: 10.1001/jamaneurol.2013.646. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Tomlinson CL, et al. Physiotherapy versus placebo or no intervention in Parkinson’s disease. Cochrane Database Syst. Rev. 2013 doi: 10.1002/14651858.CD002817.pub4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Altmann LJP, et al. Aerobic exercise improves mood, cognition, and language function in Parkinson’s disease: Results of a controlled study. J. Int. Neuropsychol. Soc. JINS. 2016;22:878–889. doi: 10.1017/S135561771600076X. [DOI] [PubMed] [Google Scholar]
- 6.David FJ, et al. Exercise improves cognition in Parkinson’s disease: The PRET-PD randomized, clinical trial. Mov. Disord. Off. J. Mov. Disord. Soc. 2015;30:1657–1663. doi: 10.1002/mds.26291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Schootemeijer S, et al. Barriers and motivators to engage in exercise for persons with Parkinson’s disease. J. Park. Dis. 2020;10:1293–1299. doi: 10.3233/JPD-202247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Afshari M, Yang A, Bega D. Motivators and barriers to exercise in Parkinson’s disease. J. Park. Dis. 2017;7:703–711. doi: 10.3233/JPD-171173. [DOI] [PubMed] [Google Scholar]
- 9.Ellis T, et al. Barriers to exercise in people with Parkinson disease. Phys. Ther. 2013;93:628–636. doi: 10.2522/ptj.20120279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Prakash P, Scott TF, Baser SM, Leichliter T, Schramke CJ. Self-reported barriers to exercise and factors impacting participation in exercise in patients with Parkinson’s disease. Mov. Disord. Clin. Pract. 2021;8:631–633. doi: 10.1002/mdc3.13197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Yeh S-W, et al. High-intensity functional exercise in older adults with dementia: A systematic review and meta-analysis. Clin. Rehabil. 2021;35:169–181. doi: 10.1177/0269215520961637. [DOI] [PubMed] [Google Scholar]
- 12.Verbrugghe J, et al. High Intensity training is an effective modality to improve long-term disability and exercise capacity in chronic nonspecific low back pain: A randomized controlled trial. Int. J. Environ. Res. Public. Health. 2021;18:10779. doi: 10.3390/ijerph182010779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Solinsky R, Draghici A, Hamner JW, Goldstein R, Taylor JA. High-intensity, whole-body exercise improves blood pressure control in individuals with spinal cord injury: A prospective randomized controlled trial. PloS One. 2021;16:e0247576. doi: 10.1371/journal.pone.0247576. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Langeskov-Christensen M, et al. Efficacy of high-intensity aerobic exercise on brain MRI measures in multiple sclerosis. Neurology. 2021;96:e203–e213. doi: 10.1212/WNL.0000000000011241. [DOI] [PubMed] [Google Scholar]
- 15.Luo L, et al. Effect of high-intensity exercise on cardiorespiratory fitness in stroke survivors: A systematic review and meta-analysis. Ann. Phys. Rehabil. Med. 2020;63:59–68. doi: 10.1016/j.rehab.2019.07.006. [DOI] [PubMed] [Google Scholar]
- 16.Ben-Zeev T, Okun E. High-intensity functional training: Molecular mechanisms and benefits. Neuromolecular Med. 2021;23:335–338. doi: 10.1007/s12017-020-08638-8. [DOI] [PubMed] [Google Scholar]
- 17.Calverley TA, et al. HIITing the brain with exercise: Mechanisms, consequences and practical recommendations. J. Physiol. 2020;598:2513–2530. doi: 10.1113/JP275021. [DOI] [PubMed] [Google Scholar]
- 18.Ross RE, Saladin ME, George MS, Gregory CM. High-intensity aerobic exercise acutely increases brain-derived neurotrophic factor. Med. Sci. Sports Exerc. 2019;51:1698–1709. doi: 10.1249/MSS.0000000000001969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Chang HC, et al. An 8-week low-intensity progressive cycling training improves motor functions in patients with early-stage Parkinson’s disease. J. Clin. Neurol. Seoul Korea. 2018;14:225–233. doi: 10.3988/jcn.2018.14.2.225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lauhoff P, Murphy N, Doherty C, Horgan NF. A controlled clinical trial investigating the effects of cycle ergometry training on exercise tolerance, balance and quality of life in patients with Parkinson’s disease. Disabil. Rehabil. 2013;35:382–387. doi: 10.3109/09638288.2012.694962. [DOI] [PubMed] [Google Scholar]
- 21.Heinrich KM, Patel PM, O’Neal JL, Heinrich BS. High-intensity compared to moderate-intensity training for exercise initiation, enjoyment, adherence, and intentions: An intervention study. BMC Public Health. 2014;14:789. doi: 10.1186/1471-2458-14-789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Picelli A, et al. Robot-assisted gait training in patients with Parkinson disease: A randomized controlled trial. Neurorehabil. Neural Repair. 2012;26:353–361. doi: 10.1177/1545968311424417. [DOI] [PubMed] [Google Scholar]
- 23.Schwartz I, et al. Robot-assisted gait training in multiple sclerosis patients: A randomized trial. Mult. Scler. Houndmills Basingstoke Engl. 2012;18:881–890. doi: 10.1177/1352458511431075. [DOI] [PubMed] [Google Scholar]
- 24.Furnari A, et al. Robotic-assisted gait training in Parkinson’s disease: A three-month follow-up randomized clinical trial. Int. J. Neurosci. 2017;127:996–1004. doi: 10.1080/00207454.2017.1288623. [DOI] [PubMed] [Google Scholar]
- 25.Alwardat M, et al. Effectiveness of robot-assisted gait training on motor impairments in people with Parkinsons disease: A systematic review and meta-analysis. Int. J. Rehabil. Res. Int. Z. Rehabil. Rev. Int. Rech. Readaptation. 2018;41:287–296. doi: 10.1097/MRR.0000000000000312. [DOI] [PubMed] [Google Scholar]
- 26.Capecci M, et al. Clinical effects of robot-assisted gait training and treadmill training for Parkinson’s disease. A randomized controlled trial. Ann. Phys. Rehabil. Med. 2019;62:303–312. doi: 10.1016/j.rehab.2019.06.016. [DOI] [PubMed] [Google Scholar]
- 27.Picelli A, et al. Robot-assisted gait training versus equal intensity treadmill training in patients with mild to moderate Parkinson’s disease: A randomized controlled trial. Parkinsonism Relat. Disord. 2013;19:605–610. doi: 10.1016/j.parkreldis.2013.02.010. [DOI] [PubMed] [Google Scholar]
- 28.Carda S, et al. Robotic gait training is not superior to conventional treadmill training in parkinson disease: A single-blind randomized controlled Trial. Neurorehabil. Neural Repair. 2012;26:1027–1034. doi: 10.1177/1545968312446753. [DOI] [PubMed] [Google Scholar]
- 29.Kim H, et al. Robot-assisted gait training with auditory and visual cues in Parkinson’s disease: A randomized controlled trial. Ann. Phys. Rehabil. Med. 2022;65:101620. doi: 10.1016/j.rehab.2021.101620. [DOI] [PubMed] [Google Scholar]
- 30.Reycraft JT, et al. Exercise intensity and recovery on circulating brain-derived neurotrophic factor. Med. Sci. Sports Exerc. 2020;52:1210–1217. doi: 10.1249/MSS.0000000000002242. [DOI] [PubMed] [Google Scholar]
- 31.Esquenazi A, Talaty M, Packel A, Saulino M. The ReWalk powered exoskeleton to restore ambulatory function to individuals with thoracic-level motor-complete spinal cord injury. Am. J. Phys. Med. Rehabil. Assoc. Acad. Physiatr. 2012;91:911–921. doi: 10.1097/PHM.0b013e318269d9a3. [DOI] [PubMed] [Google Scholar]
- 32.Kolakowsky-Hayner, S. A., Crew, J., Moran, S. & Shah, A. Safety and feasibility of using the Ekso bionic exoskeleton to aid ambulation after spinal cord injury. J SpineS4, (2013).
- 33.Murray SA, Ha KH, Hartigan C, Goldfarb M. An assistive control approach for a lower-limb exoskeleton to facilitate recovery of walking following stroke. IEEE Trans. Neural Syst. Rehabil. Eng. Publ. IEEE Eng. Med. Biol. Soc. 2015;23:441–449. doi: 10.1109/TNSRE.2014.2346193. [DOI] [PubMed] [Google Scholar]
- 34.Martini E, et al. Gait training using a robotic hip exoskeleton improves metabolic gait efficiency in the elderly. Sci. Rep. 2019;9:7157. doi: 10.1038/s41598-019-43628-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.McGibbon C, et al. Evaluation of a lower-extremity robotic exoskeleton for people with knee osteoarthritis. Assist. Technol. Off. J. RESNA. 2021 doi: 10.1080/10400435.2021.1887400. [DOI] [PubMed] [Google Scholar]
- 36.McGibbon CA, et al. Evaluation of the Keeogo exoskeleton for assisting ambulatory activities in people with multiple sclerosis: An open-label, randomized, cross-over trial. J. Neuroengineering Rehabil. 2018;15:117. doi: 10.1186/s12984-018-0468-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.McCain EM, et al. Mechanics and energetics of post-stroke walking aided by a powered ankle exoskeleton with speed-adaptive myoelectric control. J. Neuroengineering Rehabil. 2019;16:57. doi: 10.1186/s12984-019-0523-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Di Natali C, et al. Design and evaluation of a soft assistive lower limb exoskeleton. Robotica. 2019;37:2014–2034. doi: 10.1017/S0263574719000067. [DOI] [Google Scholar]
- 39.Ding Y, et al. Biomechanical and physiological evaluation of multi-joint assistance with soft exosuits. IEEE Trans Neural Syst Rehabil Eng. 2017;25:119–130. doi: 10.1109/TNSRE.2016.2523250. [DOI] [PubMed] [Google Scholar]
- 40.Panizzolo, F. A. et al. A biologically-inspired multi-joint soft exosuit that can reduce the energy cost of loaded walking. J. Neuroeng. Rehabil.13, 43-016-0150-9 (2016). [DOI] [PMC free article] [PubMed]
- 41.McGibbon, C. A., Sexton, A. & Gryfe, P. Exercising with a robotic exoskeleton can improve memory and gait in people with Parkinson’s disease by facilitating progressive exercise intensity. Sci. Rep.In revision. (2023). [DOI] [PMC free article] [PubMed]
- 42.Jeng B, et al. Accelerometer output and its association with energy expenditure in persons with mild-to-moderate Parkinson’s disease. PLOS ONE. 2020;15:e0242136. doi: 10.1371/journal.pone.0242136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Serrano-Duenas M, Calero B, Serrano S, Serrano M, Coronel P. Metric properties of the mini-mental Parkinson and SCOPA-COG scales for rating cognitive deterioration in Parkinson’s disease. Mov. Disord. Off. J. Mov. Disord. Soc. 2010;25:2555–2562. doi: 10.1002/mds.23322. [DOI] [PubMed] [Google Scholar]
- 44.Marras C, Troster AI, Kulisevsky J, Stebbins GT. The tools of the trade: A state of the art ‘How to Assess Cognition’ in the patient with Parkinson’s disease. Mov. Disord. Off. J. Mov. Disord. Soc. 2014;29:584–596. doi: 10.1002/mds.25874. [DOI] [PubMed] [Google Scholar]
- 45.Marinus J, et al. Assessment of cognition in Parkinson’s disease. Neurology. 2003;61:1222–1228. doi: 10.1212/01.WNL.0000091864.39702.1C. [DOI] [PubMed] [Google Scholar]
- 46.Harada ND, Chiu V, Stewart AL. Mobility-related function in older adults: Assessment with a 6-minute walk test. Arch. Phys. Med. Rehabil. 1999;80:837–841. doi: 10.1016/S0003-9993(99)90236-8. [DOI] [PubMed] [Google Scholar]
- 47.Wetzel JL, Fry DK, Pfalzer LA. Six-minute walk test for persons with mild or moderate disability from multiple sclerosis: Performance and explanatory factors. Physiother. CanadaPhysiotherapie Can. 2011;63:166–180. doi: 10.3138/ptc.2009-62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Munari D, et al. High-intensity treadmill training improves gait ability, VO2peak and cost of walking in stroke survivors: Preliminary results of a pilot randomized controlled trial. Eur. J. Phys. Rehabil. Med. 2018;54:408–418. doi: 10.23736/S1973-9087.16.04224-6. [DOI] [PubMed] [Google Scholar]
- 49.Moore CG, et al. Study in Parkinson disease of exercise (SPARX): Translating high-intensity exercise from animals to humans. Contemp. Clin. Trials. 2013;36:90–98. doi: 10.1016/j.cct.2013.06.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Chow H-W, Yang C-C. Accuracy of optical heart rate sensing technology in wearable fitness trackers for young and older adults: Validation and comparison study. JMIR MHealth UHealth. 2020;8:e14707. doi: 10.2196/14707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Block VAJ, et al. Remote physical activity monitoring in neurological disease: A systematic review. PloS One. 2016;11:e0154335. doi: 10.1371/journal.pone.0154335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Feito Y, Bassett DR, Thompson DL. Evaluation of activity monitors in controlled and free-living environments. Med. Sci. Sports Exerc. 2012;44:733–741. doi: 10.1249/MSS.0b013e3182351913. [DOI] [PubMed] [Google Scholar]
- 53.Malling ASB, Jensen BR. Motor intensive anti-gravity training improves performance in dynamic balance related tasks in persons with Parkinson’s disease. Gait Posture. 2016;43:141–147. doi: 10.1016/j.gaitpost.2015.09.013. [DOI] [PubMed] [Google Scholar]
- 54.Wilkenfeld JN, et al. Sensemaking, adaptation and agency in human-exoskeleton synchrony. Front. Robot. AI. 2023;10:1207052. doi: 10.3389/frobt.2023.1207052. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data are provided in this manuscript and tables.