Abstract
A newborn girl had typical “blueberry muffin” skin lesions, which shows histopathologic features of monocytic leukemia cutis. The systemic leukemia was demonstrated after one month of life. She was treated by chemotherapy, including induction and three consolidation cures, according to the ELAM02 protocol, which led to complete remission. This case report with congenital form of AML5 cutaneous localization, preceding systemic involvement, with a 5‐year follow‐up and positive outcome is remarkable.
Keywords: acute leukemia, congenital leukemia, leukemia cutis
1. INTRODUCTION
Leukemia cutis (LC) is defined by cutaneous infiltration by leukemic cells, which happens at least one month before acute leukemia. The neonatal form of LC that occurs within 4 weeks of life is less frequent than adult LC [1] and leads to neonatal leukemia with a poor prognosis, usually fatal. We report an exceptional case of complete remission with a 5‐year follow‐up after aleukemic LC preceding acute myeloid leukemia (AML), with the main objectives to describe epidemiologic, clinical, biological, histological, molecular, and prognostic features of aleukemic neonatal LC.
2. CASE REPORT
In 2018, a female newborn presented at birth with a few firm red‐violaceous papulo‐nodular lesions in the face and trunk (Figure 1). Her general status was excellent and other physical examinations were strictly normal.
FIGURE 1.
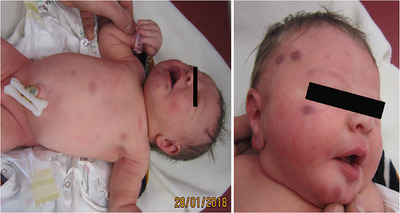
Red‐violaceous papulo‐nodular lesions in the face and the trunk. The lesions were firm on palpation, not pulsatile. The most voluminous measured 2 cm in diameter, in the middle area of the scalp.
Cutaneous biopsy on 3rd day of life showed dense dermal infiltrate composed of monomorphous undifferentiated blast cells with nuclear dust. These cells stained positively for CD15, CD68, CD4 (Figure 2), CD43, HLA‐DR, and negatively for myeloperoxidase. Immunophenotyping of skin biopsy revealed 90% of immature cells with monocytic phenotype. Cutaneous lesions were interpreted as skin involvement of the monocytic subtype of AML, and the diagnosis was confirmed on the 30th day of life by molecular testing revealing KMT2A gene rearrangement with translocation t(9;11)(p22;q23)/KMT2A::MLLT3.
FIGURE 2.
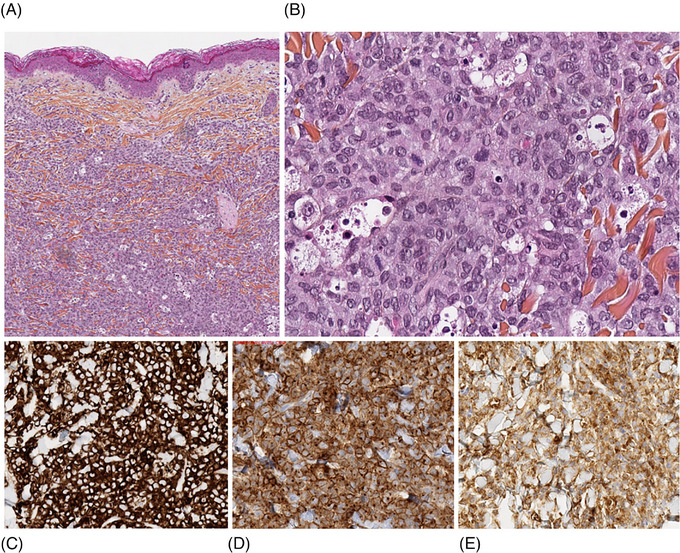
(A) Dense dermal infiltrate composed of undifferentiated blast cells with nuclear dust (HES x5). (B) Tumor cells have abundant pale cytoplasm and reniform monomorphous nuclei. Nucleoli are discrete. Nuclear dust is present. Blasts are CD15+ (C), CD68+ (D), and CD4+ (E) (IHC x20). HES, hematoxylin‐eosin saffron; IHC, immunohistochemistry.
At 16 days of life, all the laboratory tests, including complete blood count, peripheral blood smear, coagulation parameters, and biochemistry revealed no abnormality, except cholestasis with gamma‐glutamyl transferase four times higher than normal values. HIV, HBV, HCV, and HTLV1/2 serologies were negative, as well as multiplex respiratory polymerase chain reaction and blood cultures. Only Epstein‐Barr virus serology came back positive, following maternal immunization. No excess blasts were observed on bone marrow aspirate smears, and the cerebrospinal fluid examination did not show meningeal invasion. All imaging exams (echocardiography, transfontanellar and abdominopelvic ultrasound, fundus, and chest X‐ray) were normal. Given these overall results, the patient was released from hospitalization and bi‐weekly follow‐up with biological assessment was recommended.
At 30 days of life, the pathology expressed at the hematological level, revealing anemia at 93 g/L and hyperleukocytosis at 45.4 × 109/L, with 28% of blasts and 18% of monocytes on peripheral blood smear. Blood immunophenotyping revealed 50% of monocytic cells, with 54% of immature promonocytic or monoblastic forms. The patient was urgently put under chemotherapy treatment, following the ELAM02 protocol, with doses adapted to age. At day 28 post‐induction, the bone marrow aspirate smear showed less than 5% of monoblasts, concluding in complete remission. Moreover, all the cutaneous lesions had disappeared. At 42 days post‐induction, the monitoring of residual disease showed a number of transcript copy number KMT2A::MLLT3 < 0.001. The patient was definitively released from hospitalization after induction followed by three consolidation cures, each time showing complete remission and negative minimal residual disease (Figure S1). In 2023, 5 years post‐treatment, the patient is still in remission, with normal regular biological assessments, and presents excellent psychomotor development.
3. DISCUSSION
Leukemia is the most common malignancy diagnosed in childhood, but the congenital form represents less than 1% of the leukemia in neonates [2] and is most commonly reported in infants with Down's syndrome. LC occurs in approximately 25%–30% of infants with congenital leukemia [3], but congenital LC without other clinical signs is extremely rare [4, 5, 6, 7]. Our patient appeared as a “blueberry muffin baby” with numerous red‐violaceous nodules. Skin lesions were consistent with congenital LC, but our patient did not show leukemic cells in blood or bone marrow during the first month of life. These nodules may be confused with other causes of “blueberry muffin baby”, such as congenital infections, hemolytic disease, metastatic neuroblastoma, or malignant histiocytosis [8, 9]. In our patient, none of these diagnoses was retained following the additional examinations carried out confirming the probable diagnosis of AML with KMT2A gene rearrangement. Acute myelomonocytic and monocytic leukemia (AML4 and AML5 according to the French American British classification) have the highest rate of associated LC, up to 50% [1]. Moreover, KMT2A gene rearrangement is most commonly involved in infant leukemia, particularly in M4‐M5 subtypes [10]. This abnormality at the site of the KMT2A gene carries an intermediate prognosis, with patients often refractory to treatment [11]. Few cases of congenital leukemia with spontaneous remission have been described, but none of these patients had KMT2A rearrangement at diagnosis [12, 13]. Histologic examination of skin lesions following cutaneous biopsy commonly shows a dense diffuse dermal infiltrate of leukemic cells in the reticular dermis, that extend into the subcutis. Malignant myeloid cells have round to oval monotonous nuclei, chromatin is evenly dispersed, and nucleoli are multiple and inconspicuous. Cellular debris and mitoses are generally present. The cutaneous immunohistochemistry shows in AML5 subtype, a positive expression of CD43, CD68, CD33, CD4, CD15, and CD56, and a negative expression of CD123 and CD3 [14]. Most of these characteristics were found on the histologic examination of cutaneous biopsy in our patient. LC precedes systemic leukemia development by five weeks to sixteen months according to the cases described in the literature [4]. The leukemia in our patient has been expressed at the biological level at 30 days of life. It remains unclear why some patients progress to spontaneous remission – rarely been reported [12, 13, 15] and others to systemic disease. Although some cases of remission without treatment have been described, a number of such patients presented relapses after several years [4]. Thus, the induction of treatment in this pathology remains controversial, and a close follow‐up is mandatory. The case of our patient demonstrates that the chemotherapy protocol implemented showed a real effect, with proof of complete remission and undetectable residual disease.
4. CONCLUSION
LC is common in the AML5 subtype, but its neonatal form has been very rarely described. Neonatal LC without other clinical signs has been reported in a few patients. Even with knowledge of differential diagnoses of skin lesions, cutaneous biopsy, and molecular testing are essential to make a final diagnosis and initiate appropriate treatment. This case report with the congenital form of AML5 cutaneous localization, preceding systemic involvement, with a 5‐year follow‐up and positive outcome is remarkable and highlights the importance of the transdisciplinary cooperation between biologists, pathologists, and clinicians.
CONFLICT OF INTEREST STATEMENT
The authors declare no conflict of interest.
ETHICS STATEMENT
The authors have confirmed ethical approval statement is not needed for this submission.
PATIENT CONSENT STATEMENT
The authors have confirmed patient consent statement is not needed for this submission
CLINICAL TRIAL REGISTRATION
The authors have confirmed clinical trial registration is not needed for this submission.
Supporting information
Supporting information
ACKNOWLEDGMENTS
Caroline Lo Presti, Gautier Szymanski, Christine Lefebvre, Marie‐Christine Jacob, and Pascal Mossuz contributed to analyzing and interpreting biological data. Hugo Gil contributed to analyzing and interpreting histopathologic data. Caroline Lo Presti wrote the manuscript and all authors approved the final version.
Lo Presti C, Szymanski G, Lefebvre C, Jacob M‐C, Mossuz P, Gil H. Aleukemic congenital leukemia cutis preceding monocytic leukemia with favorable outcome: A case report. eJHaem. 2024;5:162–165. 10.1002/jha2.861
DATA AVAILABILITY STATEMENT
The data that support the findings of this study are available from the corresponding author upon reasonable request.
REFERENCES
- 1. Cho‐Vega JH, Medeiros LJ, Prieto VG, Vega F. Leukemia cutis. Am J Clin Pathol. 2008;129(1):130–142. [DOI] [PubMed] [Google Scholar]
- 2. Isaacs H. Fetal and neonatal leukemia. J Pediatr Hematol Oncol. 2003;25(5):348–361. [DOI] [PubMed] [Google Scholar]
- 3. Resnik KS, Brod BB. Leukemia cutis in congenital leukemia. Analysis and review of the world literature with report of an additional case. Arch Dermatol. 1993;129(10):1301–1306. [DOI] [PubMed] [Google Scholar]
- 4. Torrelo A, Madero L, Mediero IG, Baño A, Zambrano A. Aleukemic congenital leukemia cutis. Pediatr Dermatol. 2004;21(4):458–461. [DOI] [PubMed] [Google Scholar]
- 5. Francis JS, Sybert VP, Benjamin DR. Congenital monocytic leukemia: report of a case with cutaneous involvement, and review of the literature. Pediatr Dermatol. 1989;6(4):306–311. [DOI] [PubMed] [Google Scholar]
- 6. Lee EG, Kim TH, Yoon MS, Lee HJ. Congenital leukemia cutis preceding acute myeloid leukemia with t(9;11)(p22;q23), MLL‐MLLT3. J Dermatol. 2013;40(7):570–571. [DOI] [PubMed] [Google Scholar]
- 7. Zhang IH, Zane LT, Braun BS, Maize J, Zoger S, Loh ML. Congenital leukemia cutis with subsequent development of leukemia. J Am Acad Dermatol. 2006;54(2 Suppl):S22–27. [DOI] [PubMed] [Google Scholar]
- 8. Handler MZ, Schwartz RA. Neonatal leukaemia cutis. J Eur Acad Dermatol Venereol. 2015;29(10):1884–1889. [DOI] [PubMed] [Google Scholar]
- 9. Mehta V, Balachandran C, Lonikar V. Blueberry muffin baby: a pictorial differential diagnosis. Dermatol Online J. 2008;14(2):8. [PubMed] [Google Scholar]
- 10. Pui CH, Kane JR, Crist WM. Biology and treatment of infant leukemias. Leukemia. 1995;9(5):762–769. [PubMed] [Google Scholar]
- 11. Blais S, Boutroux H, Pasquet M, Leblanc T, Fenneteau O, Gandemer V, et al. Is acute myeloblastic leukemia in children under 2 years of age a specific entity? A report from the FRENCH ELAM02 Study Group. Hemasphere. 2019;3(6):e316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Grundy RG, Martinez A, Kempski H, Malone M, Atherton D. Spontaneous remission of congenital leukemia: a case for conservative treatment. J Pediatr Hematol Oncol. 2000;22(3):252–255. [DOI] [PubMed] [Google Scholar]
- 13. Mora J, Dobrenis AM, Bussel JB, Aledo A. Spontaneous remission of congenital acute nonlymphoblastic leukemia with normal karyotype in twins. Med Pediatr Oncol. 2000;35(2):110–113. [DOI] [PubMed] [Google Scholar]
- 14. Jin X, Li F, Li X, Zhu W, Mou Y, Huang Y, et al. Cutaneous presentation preceding acute monocytic leukemia: a CARE‐compliant article. Medicine. 2017;96(10):e6269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Bresters D, Reus ACW, Veerman AJP, van Wering ER, van der Does‐van den Berg A, Kaspers GJL. Congenital leukaemia: the Dutch experience and review of the literature. Br J Haematol. 2002;117(3):513–524. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supporting information
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.


