Abstract
Narrative comprehension is a linguistic ability that emerges early in life and has a critical role in language development, reading acquisition, and comprehension. According to the Simple View of Reading model, reading is acquired through word decoding and linguistic comprehension. Here, within and between networks, functional connectivity in several brain networks supporting both language and reading abilities was examined from prereading to proficient reading age in 32 healthy children, ages 5–18 years, scanned annually while listening to stories over 12 years. Functional connectivity changes within and between the networks were assessed and compared between the years using hierarchical linear regression and were related to reading abilities. At prereading age, the networks related to basic language processing accounted for 32.5% of the variation of reading ability at reading age (at 12–14 years) (R2 = 0.325, p = 0.05). At age 17, more complex cognitive networks were involved and accounted for 97.4% of the variation in reading ability (R2 = 0.974, p = 0.022). Overall, networks composing the future-reading network are highly involved in processing narratives along development; however, networks related to semantic, phonological, and syntactic processing predict reading ability earlier in life, and more complex networks predict reading proficiency later in life.
Impact statement
Functional networks composing the future-reading network, including semantic, visual, executive functions, phonology, and syntax, are highly involved in processing narratives along development. However, networks related to semantic, phonological, and syntactic processing predict reading ability earlier in life, and more complex networks predict reading proficiency later in life. These results highlight the importance of exposing children to stories before reading age, as the networks involved in this activity will facilitate future reading abilities, critical for academic achievements.
Keywords: child development, executive functions, functional connectivity, language, narrative comprehension, reading
Introduction
Narrative comprehension as the foundation for reading development
Narrative comprehension is a linguistic ability considered the most basic construct of language processing (Lynch et al., 2008; Twait et al., 2018). The ability to comprehend a narrative develops early in life and depends on several more basic aspects, including semantic, phonological, and syntactic processing (Farah et al., 2019). Narrative comprehension also relies on other subabilities not specific to language, including executive functions (EFs), visualization, consolidation, and information integration (Horowitz-Kraus et al., 2015a). The importance of narrative comprehension is demonstrated in reading development, particularly in reading comprehension.
The two main skills associated with reading ability are word decoding and language processing, which help readers match written letters to sounds and make meaning out of text (i.e., semantics) (Hoover and Gough, 1990). Decoding is defined as adequate word recognition, while language processing is the ability to extract meaning from either spoken or written text and is related to vocabulary and listening comprehension (Gough and Tunmer, 1986). These two components are interrelated as both are necessary to read efficiently and later comprehend written materials (Katz and Frost, 1992).
Previous research has shown that intact orthographical word recognition relies on several more basic linguistic processes, including phonological awareness skills, decoding abilities, rapid automatic naming, and morphological awareness (Kim, 2017). Narrative comprehension plays a prominent role in developing more complex linguistic processes, such as reading, as it involves similar basic abilities presented in the Simple View of Reading (SVR) model.
The complex process of reading
Reading is defined as recognizing words/signs and matching those with a semantic meaning. In addition to basic sensory and motor factors, reading relies on essential linguistic components such as the awareness of the basic sounds in language (phonological awareness), word recognition (orthographic abilities), and semantics (meaning of the words) (Fiez and Petersen, 1998). Moreover, reading involves decoding, fluency, vocabulary, and comprehension, which are critical for proficient reading abilities and reading comprehension (Archer et al., 2003).
Recent studies have also highlighted the involvement of several basic and higher order abilities (i.e., EFs) participating in single-word reading and contextual reading (Meri et al., 2020). EFs are a set of cognitive abilities, including higher order abilities such as problem-solving and logical thinking and more basic ones such as inhibition, basic attention, and working memory (Lynch et al., 2008). EF is the foundation for cognitive abilities, such as language comprehension, reading, and reading comprehension (Cartwright, 2012).
Neural circuits supporting narrative comprehension are well reserved along development
As previously mentioned, narrative processing is the basis for reading and reading comprehension; hence, they share several common neural circuits. Studies on narrative comprehension in 5–18-year-old participants suggest that brain regions engaged in language processing include the inferior frontal gyrus (IFG), superior temporal gyrus (STG), angular gyrus (AG), frontal regions, precuneus, and hippocampus (Farah and Horowitz-Kraus, 2019), each relate to a different ability but work in synchrony to process a narrative stream. The IFG activates syntactic processing, whereas the STG is related to phonological and language processing, the AG letter-to-sound correspondence, frontal regions are related to the involvement of cognitive processes and sequenced temporal ordering during the task, and the hippocampus is related to episodic memory consolidation (Szaflarski et al., 2012).
Neural circuits supporting reading
Horowitz-Kraus et al. (2013) suggested that better reading abilities at older ages (11 years old) are related to increased engagement of auditory, visual, and EF-related regions during story listening in prereading ages (5–7 years). This reliance on reading-related regions, even before reading is officially acquired, supports the shared neural circuits between reading and narrative comprehension. However, these brain regions need to be active and connected to achieve fluent reading, and synchronization between visual and auditory systems is critical (Breznitz and Misra, 2003). These networks likely need to be connected with the EF networks [based on Breznitz (2006)]. However, the neurobiology of these relationships is yet to be defined.
The current study aims to determine the involvement of the future reading network in processing narratives from prereading to proficient reading age. Despite the consistent evidence of the shared neural circuits for language/narrative comprehension and reading, the changes in the level of reliance on different networks supporting reading from the prereading phase until late adolescence are yet to be defined. We hypothesize that at the prereading age (5 years), the engagement of networks associated with language and visualization will be greater during story listening, decreasing to adolescence to be replaced with a greater reliance upon EF networks related to reading abilities. We also hypothesized that the involvement of these networks would later contribute to proficient reading abilities.
Methods
Participants
The current study included 32 children (17 females) with an age range of 5–18 years, enrolled at the ages of 5, 6, or 7 years, with no history of neurological or psychiatric diagnoses. All participants were native English speakers and their demographic background was previously published (Holland et al., 2001). The participants were scanned at 12 time points annually. Each parent and participant provided informed consent and assent, respectively. Several children were scanned less than 12 times due to later enrollment, metal objects in the body, or availability problems (see Table 1). Cincinnati Children's Hospital Medical Center's Institutional Review Board approved the study.
Table 1.
Detailed List of Participants with the Total Number of Scans Administered
| Participants | Year 1 | Year 2 | Year 3 | Year 4 | Year 5 | Year 6 | Year 7 | Year 8 | Year 9 | Year 10 | Year 11 | Year 12 | Number of time points for each participant |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 05F003 | x | x | x | x | x | x | x | x | 8 | ||||
| 05F004 | x | x | x | x | X | x | x | x | x | x | 10 | ||
| 05F008 | x | x | x | X | x | x | 6 | ||||||
| 05M002 | x | x | x | x | x | x | x | x | 9 | ||||
| 05M003 | x | x | x | x | x | X | x | x | x | x | x | x | 12 |
| 05M005 | x | x | x | X | x | x | x | 8 | |||||
| 05M008 | x | X | 4 | ||||||||||
| 05M019 | x | x | x | X | x | x | x | 7 | |||||
| 05M024 | x | x | x | x | 5 | ||||||||
| 06F001 | x | x | x | x | x | x | x | 7 | |||||
| 06F003 | x | x | 2 | ||||||||||
| 06F011 | x | x | x | x | X | x | x | x | x | x | 10 | ||
| 06F014 | x | x | x | 3 | |||||||||
| 06F018 | x | x | x | x | X | x | x | x | 8 | ||||
| 06M001 | x | x | x | x | X | x | x | x | x | x | x | 12 | |
| 06M005 | x | x | x | x | x | X | x | x | x | x | x | x | 12 |
| 06M012 | x | x | x | X | x | x | x | x | x | x | 10 | ||
| 07F001 | x | x | x | x | 4 | ||||||||
| 07F002 | x | x | x | x | X | x | x | x | x | x | x | 11 | |
| 07F005 | x | x | x | x | x | 5 | |||||||
| 07F007 | x | x | x | x | x | x | x | x | x | x | x | 11 | |
| 07F009 | x | x | x | x | x | x | x | x | x | x | x | 11 | |
| 07F010 | x | x | x | x | x | X | x | x | x | x | x | 11 | |
| 07F015 | x | x | x | x | x | X | x | x | 8 | ||||
| 07F021 | x | x | x | x | X | x | x | x | x | x | 10 | ||
| 07F024 | x | x | x | x | X | x | x | x | x | x | 10 | ||
| 07M001 | x | x | x | x | x | X | x | x | x | x | x | x | 12 |
| 07M004 | x | x | x | x | x | X | x | x | 8 | ||||
| 07M005 | x | x | x | x | x | x | x | x | x | 10 | |||
| 07M006 | x | x | x | x | x | x | x | x | x | x | x | 12 | |
| 07M009 | x | x | x | x | x | x | x | x | x | x | 11 | ||
| 07M012 | x | x | x | 4 | |||||||||
| Number of participants scanned at each time point | 29 | 28 | 27 | 23 | 20 | 20 | 20 | 16 | 20 | 21 | 16 | 21 |
Study procedure
In the first year of scanning, we recruited prereading children, ages 5–7 years. All of the participants were eligible for an MRI scan. Our inclusion criteria for all of the 12 years were as follows: (1) no psychiatric or neurologic diagnosis, (2) no developmental delays, (3) intact vision and hearing, and (4) intact verbal and nonverbal abilities. The information was provided by the parents or the guardian and was later verified via questionnaires.
After meeting our inclusion criteria, participants were annually scanned during a story listening task (see detailed description in the Neuroimaging Task section). Parental consent and assent were obtained from minors participating in the study. The full details of this study were reported in a previous study by Szaflarski et al. (2012). Some of the participants did not complete the 12 visits due to braces or environmental reasons such as a lack of availability. The number of children per age group is listed in Table 1. A power analysis was conducted to ensure adequate power for the study given the sample size (N = 32), alpha (0.05), and effect size based on previous literature using the same functional magnetic resonance imaging (fMRI) task in children (Hutton et al., 2020), and revealed a power of 0.86.
Behavioral measures
Children received a variety of different behavioral tests to assess their cognitive and language abilities: (1) in the first year, children performed the Oral and Written Language Scales (OWLS) (Carrow-Woolfolk, 1995) and the Wechsler Intelligence Scale for Children—III (WISC-III) (Wechsler, 1999), (2) in years 3 and 5, they performed the OWLS, (3) in the 2nd–5th-year period of the study (years 6–10), the cognitive-behavioral tests were broadened and included the following tests: the Peabody Picture Vocabulary Test, 4th edition (PPVT-4), to verify average verbal IQ; the Wisconsin Card Sorting Test (WCST) to evaluate switching abilities; the Self-Report Behavior Rating Inventory of Executive Function (BRIEF) to evaluate general EF abilities; and letter-word subtest from Woodcock–Johnson test (WJ), (4) in years 11 and 12, general EF abilities were assessed using a BRIEF questionnaire.
Neuroimaging measures
Neuroimaging task
Children underwent the stories listening task inside the scanner. This task was formerly described by Holland et al. (2007), and it included five stories, 30 sec each alternated with five 30-sec sessions of tones (i.e., a block design; Fig. 1). Each story was presented for 30 sec, followed by a control period of pure tones that lasted for another 30 sec. The stories consisted of 9–11 sentences with different linguistic structures. Children were informed that they would be asked several questions regarding the stories at the end of the session and were instructed to be attentive to them. Participants were asked to answer 10 multiple-choice questions at the end of the scan.
FIG. 1.
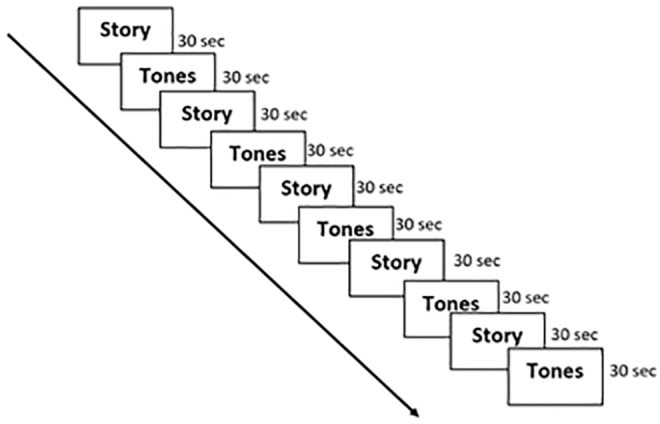
The narrative comprehension (stories listening) task. The task contains the story listening condition and the pure tones condition (i.e., control period); both are presented in blocks of 30 sec each. The task lasted 330 sec.
Neuroimaging data acquisition
In the first 8 years of the study, a 3T Bruker 30\60 scanner was used. Echo planar imaging (EPI) was used for T2*-weighted blood oxygenation level dependent (BOLD) fMRI, with the following parameters: time repetition (TR)/time echo (TE) 3000/38 msec, bandwidth 125 kHz, matrix 64 × 64, field of view (FOV) 256 × 256 mm, and slice thickness 5 mm. The scan lasted for 5 min and 30 sec and acquired 110 time frames. Only 100 frames were used for the analysis as the first 10 frames (control condition) were discarded. We used the following parameters for a 3D T1-weighted anatomical scan: TR\TE 15\4.3 msec, FOV 256 × 192 × 162 mm, and flip angle = 20°. All scans between the years 9 and 12 were acquired using a 3T Philips Achieva MRI scanner and were as close as possible to the Bruker system used.
For a whole-brain T2* scan, the following parameters were used: A gradient echo-planar sequence was used for a T2*-weighted BOLD fMRI scan with the following parameters: TR/TE = 3000/38 msec, bandwidth = 267 kHz, FOV = 256 × 256 mm, matrix = 64 × 64, and slice thickness = 5 mm. For a 3D T1-weighted whole-brain scan we used the following parameters: TR/TE = 7.8/3.7 msec, FOV 240 × 240 × 164 mm, matrix 240 × 240, flip angle = 8°, and slice thickness = 1 mm isotropic. The two different scanners used in this longitudinal study were not a concern because of the focus on interindividual effects.
Behavioral data analyses
To verify average and above-average abilities for both cognitive and reading skills, one-sample t-test analyses were conducted for all measures.
Neuroimaging data analyses
Functional MRI data preprocessing
We used SPM12 (Wellcome Department of Cognitive Neurology, London, United Kingdom) (Friston et al., 1994) to preprocess the data. The process included slice time correction, realignment, normalization, and coregistration steps. Then, we used the CONN toolbox to analyze the functional connectivity during the story-listening task (Whitfield-Gabrieli and Nieto-Castanon, 2012). We applied bandpass filtering between 0.08 and 0.9 Hz to control for motion-related artifacts. Data were scrubbed for motion. Framewise displacement (FD) for each scan and age group was exported and controlled for.
Defining the networks
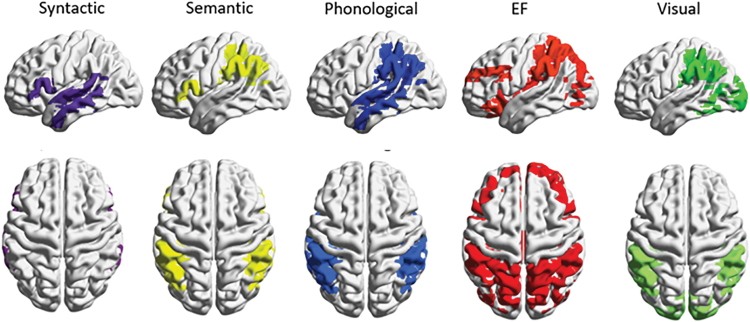
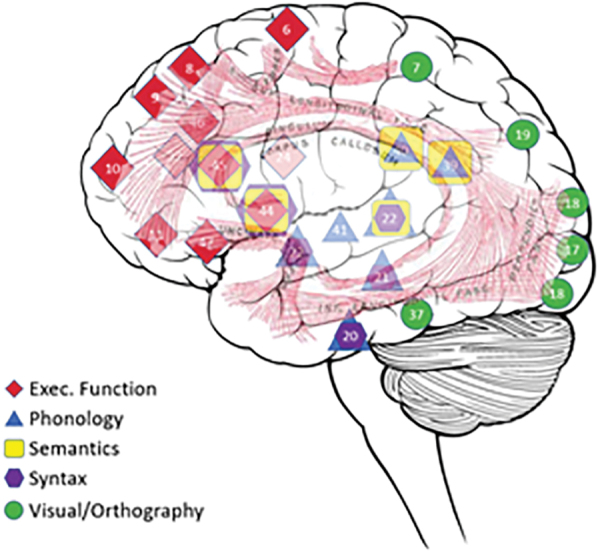
The networks composing the reading networks were defined based on literature in the field. They included the following networks supporting reading: visual (Horowitz-Kraus et al., 2016), syntactic, semantic, phonological [coordinates reported at Farah et al. (2021)], and EF (Horowitz-Kraus et al., 2015b) networks. All networks were defined based on coordinates relating these networks to reading ability (Farah et al., 2021; Horowitz-Kraus et al., 2015b, 2016). See Figures 2 and 3 and Table 2 for the network coordinates.
FIG. 2.
Visual, syntactic, semantic, phonological processing, and EF networks. The reading network used in the current study includes the EF network (in red), the phonological (in blue), the semantic (in yellow), the syntactic network (in purple), and the visual processing network (in green). EF, executive function. Color images are available online.
FIG. 3.
A glass whole-brain view of the reading related networks. A transverse view of the maps of the reading networks (syntactic in brown, semantic in purple, phonological in red, EF in blue, and the visual processing in yellow). Top: axial view; bottom: sagittal view. Color images are available online.
Table 2.
Anatomical Regions and Coordinates of the Regions Within the Language-Related Networks
| Network | Anatomical region | Coordinates | Associated Brodman areas |
|---|---|---|---|
| EF network | aPFC (L) | L (−28, 51, 15) R (27, 50, 23) |
10 |
| Lateral aI/ fO (L) | L (−51, 18, 13) R (45, 23, −4) |
47 44 |
|
| Medial aI/ fO (L) | L (−33, 24, 1) R (33, 25, −1) |
13 | |
| aI fO (L) | L (−35, 14, 5) R (36, 16, 4) |
13 | |
| dACC msFC | L (−1, 10, 46) | 32 | |
| dlPFC | L (−43, 22, 34) R (43, 22, 34 |
9 | |
| IPL | L (−51, −51, 36) R (51, 47, 42) |
40 | |
| IPS | L (−31,−59,42) R (30,−61,39) |
7 19 |
|
| Left precuneus [precuneus] | L (−9, −72, 37) R (10, −69, 39) |
7 | |
| mCC | (0, −29, 30) | 23 | |
| Phonological network | |||
| TG/Heschel's gyrus | L (−50, −27, 10) R (54, −6, 7) |
41 | |
| STG/Wernicke's | L (−61, −22, 2) R (62, −19, −5) |
22 | |
| Lateral TG/MTG | L (−63, −17, −20) R (63, −14, −26) |
21 | |
| aITG | L (−52, −20, −37) R (49, −24, 32) |
20 | |
| SMG/IPL | L (−51, −40, 46) R (53, −43, 45) |
40 | |
| AG | L (−47, −66, 31) R (49, −65, 29) |
39 | |
| Semantic network | |||
| STG/Wernicke's | L (−61, −22, 2) R (62, −19, −5) |
22 | |
| AG | L (−47, −66, 31) R (49, −65, 29) |
39 | |
| SMG\IPL | L (−51, −40, 46) R (53, −43, 45) |
40 | |
| Inferior frontal gyrus | L (−56, 15, 6) R (61, 16, 2) |
44 | |
| Triangular part of inferior frontal gyrus | L (−52, 29, 3) R (55, 29, −2) |
45 | |
| Syntactic network | |||
| aITG | L (−52, −20, −37) R (49, −24, 32) |
20 | |
| Lateral TG\MTG | L (−63, −17, −20) R (63, −14, −26) |
21 | |
| STG\Wernicke's | L (−61, −22, 2) R (62,−19,−5) |
22 | |
| Inferior frontal gyrus | L (−56, 15, 6) R (61, 16, 2) |
44 | |
| Triangular part of inferior frontal gyrus | L (−52, 29, 3) R (55, 29, −2) |
45 | |
| Visual/orthographic network | |||
| V1/primary visual | L (−33, −93, −12) R (−24, −105, −3) |
17 | |
| Visual association area | L (−33, −93, −12) R (−24, −105, −3) |
18 | |
| OTG/fusiform | L (−48, −70, −2) R (52, −68, 2) |
37 | |
| AG | L (−48, −78, 36) R (33, −84, 41) |
39 | |
| Supermarginal gyrus | L (−33, −58, 52) R (38, −42, 41) |
40 | |
AG, angular gyrus; aI/fO, anterior insula/frontal operculum; aPFC, anterior prefrontal cortex; dACC/msFC, dorsal anterior cingulate/medial superior frontal cortex; dlPFC, dorsolateral prefrontal cortex; EF, executive function; IPL, inferior parietal lobule; IPS, intraparietal sulcus; lateral aI/fO, lateral anterior insula/frontal operculum; mCC, midcingulate cortex; medial aI fO, medial anterior insula/frontal operculum; MTG, medial temporal gyrus; OTG, occipital temporal gyrus; SMG, supra marginal gyrus; STG, superior temporal gyrus; TG, temporal gyrus.
To determine the functional connectivity of these networks during the story listening task, between and within functional connectivity values of each network were calculated. To predict reading ability based on the contribution of each network longitudinaly, connectivity values of each network were fed into a hierarchical model.
All analyses were corrected for multiple comparisons using a false-discovery rate (FDR) correction p < 0.05.
Twelve correlation coefficient matrices (one for each year) were created between the networks listed in Figure 3 and Table 2 to identify developmental changes in the functional connectivity of the reading related networks. The correlation coefficients of the within and between functional connectivity between each possible pair of the networks were estimated.
Hierarchical linear regression analysis
To see how each network contributes to future reading skills, two behavioral tests examining reading ability were used: The WJ, which examined the reading abilities of children at ages 11–13 years, and the American college testing (ACT) that examines reading abilities of children at ages 17–18 years. A hierarchical linear regression model was used to investigate which neural network contributes to the variance in future reading abilities over the years. At each level in the hierarchical model, within and between functional connectivity was added, while the variance in reading skills was tested.
Results
Behavioral data analysis
Children were within the normal range of cognitive and reading abilities and close to the normal range of verbal abilities, as presented in Table 3.
Table 3.
One-Sample t-Test Analysis of Cognitive and Reading Measures for the Whole Group as Measured at Proficient Reading Age (Ages 11–13)
| Ability | Measures | Mean (SD) | T (p) | Average range |
|---|---|---|---|---|
| Verbal abilities | PPVT (standard score) | 115.36 (13.26) | 45.15 (0.00) | 85–115 |
| Word reading | Woodcock–Johnson, letter word (standard score) | 112.66 (11.25) | 52.01 (0.00) | 85–115 |
| EFs | WCST, switching (scaled score) | 106.81 (31.37) | 17.68 (0.00) | 85–115 |
| BRIEF GEC (T-score) | 49.52 (9.30) | 27.65 (0.00) | 0–100 |
BRIEF GEC, Behavior Rating Inventory of Executive Function General Cognition; PPVT, Peabody Picture Vocabulary Test; SD, standard devitaion; WCST, Wisconsin Card Sorting Test.
Neuroimaging data analysis
Functional connectivity correlation matrices
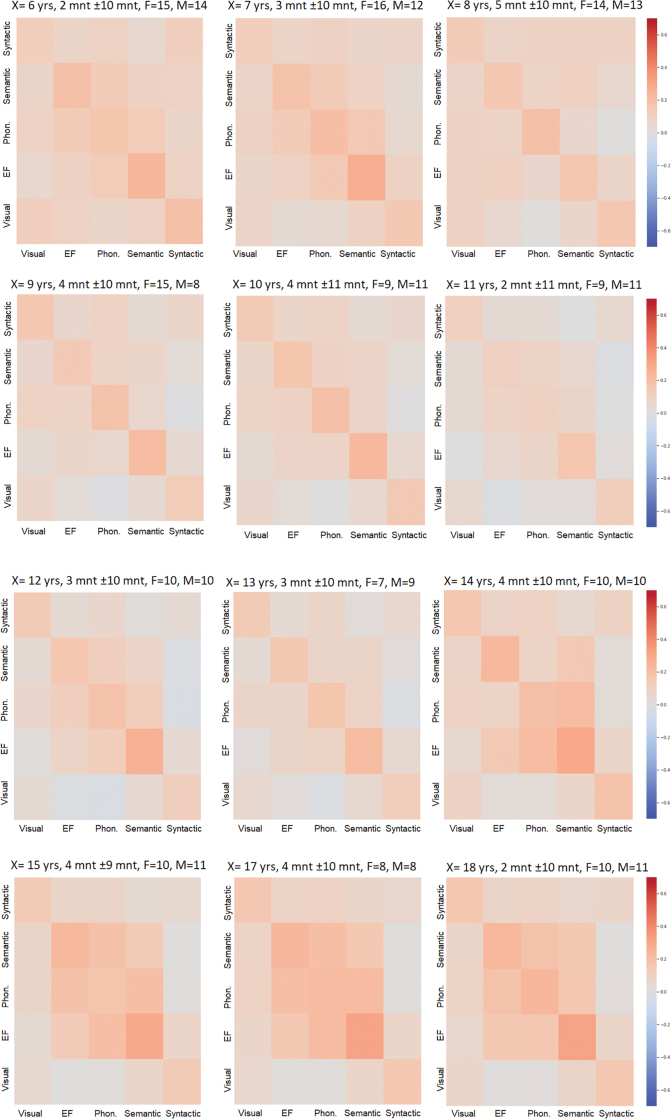
Correlation matrices of the functional connectivity data showed that at early ages (5–9 years), positive correlations were mainly observed within the semantic and phonological networks. At an older age (ages 14–16), correlations within the EF network and between the EF and language networks (i.e., semantics, phonological) increased (see Fig. 4).
FIG. 4.
Correlation coefficient matrices of the within and between functional connectivity reading related networks. A representation of the within- and between-network functional connectivity correlation matrices with development. Blue and red represent negative and positive within and between functional connectivity values, respectively (the hot color represents more positive functional connectivity values; cooler colors represent more negative functional connectivity values). Color images are available online.
Hierarchical linear regression analysis
A hierarchical linear regression analysis revealed that at prereading age (at age 6), the networks related to basic language processing (i.e., visual, phonological, syntactic) accounted for 32% of the variation of reading ability at reading age (at 12–14 years) (R2 = 0.320, p = 0.05). However, at age 13, more complex networks (i.e., EF) accounted for 91.4% of the variation in reading ability at reading age (R2 = 0.914, p = 0.020). At age 9, all examined networks accounted for 70.3% of the variation in reading ability at age 17 (R2 = 0.703, p = 0.047). At age 16, 92.2% of the variation in reading ability mainly accounted for the functional connectivity within and between EF networks (R2 = 0.922, p = 0.026). The results of the model are shown in Table 4.
Table 4.
Hierarchical Linear Regression Model Results
| Behavioral reading test | Children's age (years) | Predictors | p | R 2 |
|---|---|---|---|---|
| Letter-word test (WJ) scores (at age 11 years) | Age 6 | Between Phonological-Visual Between Syntactic Visual |
0.05 | 0.320 |
| ACT scores (at age 17–18 years) | Age 9 | Between Phonological-Visual Between Syntactic-Visual Between Syntactic-Phonological Between Syntactic-EF Between Semantic-Syntactic |
0.047 | 0.703 |
| Age 11 | Between EF-Phonological Between EF-Syntactic Between Semantic-Phonological Between Semantic-Visual |
0.05 | 0.835 | |
| Age 16 | Between EF-Visual, Phonological, Semantic, Syntactic Between Semantic-Phonological Between Semantic-Visual Between Syntactic-Visual Between Syntactic-Phonological |
0.026 | 0.922 |
ACT, American college testing; WJ, Woodcock–Johnson test.
Discussion
The current study examines how much the subcomponents that comprise the reading network in older readers participate in story listening in children who do not yet read and whether the reliance on these networks changes as reading becomes automatic in late adolescence. We hypothesized that the involvement of basic linguistic networks (i.e., semantics, phonological networks) at a younger age would predict reading abilities during reading acquisition age. In contrast, higher order networks should contribute to predicting reading acquisition later in life. We also hypothesized that the involvement of these networks would later contribute to proficient reading abilities. To test our hypotheses, we used fMRI to examine the involvement of the different reading networks during story listening tasks (phonological, semantic, syntactic, visual, and EF).
In line with our hypotheses, Figure 4 shows that more basic language/sensory networks were involved earlier in processing the narratives. Higher order cognitive networks (i.e., EF) contributed later to the reading process, which contributed to proficient future reading abilities.
The contribution of EF and linguistic components to reading ability: The SVR model
The basic linguistic and sensory networks, including semantic, syntactic, visual, and phonological networks, contribute to early reading processes. The connectivity within and between these networks becomes more significant as the participants acquire reading. These results are consistent with previous findings showing that these networks' functional connectivity differs between children and adults. A study of language representation and development suggested that children show lower functional connectivity. In contrast, adults show significantly higher functional connectivity between the networks, indicating that they contribute to language development with increasing interaction among these components (Youssofzadeh et al., 2018). In another study, adults showed more robust inter-regional connectivity and a nodal degree in occipital regions during rhyming and meaning judgment tasks. At the same time, children had stronger inter-regional connectivity in temporal regions (Liu et al., 2018).
These findings suggest that there is a shift in the reliance on the different networks involved in reading development. This study supports our findings regarding the different functional connectivity patterns between the networks in younger and older ages.
EFs are involved in language processes from an early age
Our results support the findings that EFs are involved in language processes early during narrative comprehension (Farah and Horowitz-Kraus, 2019) and predict later reading proficiency. This involvement has become stronger over the years. Our results support previous literature regarding the early development of EF, and the fact that it extends across a wide range of ages, from infancy to adulthood (Zelazo and Müller, 2010). Cartwright (2012) accentuated the critical role of EF in developing reading skills, including prereading skills, word reading, and reading comprehension. This finding is significant because it emphasizes how training EF can contribute and accelerate reading acquisition.
These results highlight the gradual involvement of EF networks within the language processing domain in the SVR along development. We can cautiously suggest that the role of the EF networks is as a “synchronizer” between the auditory and visual-related neurons (Horowitz-Kraus et al., 2023; Horowitz-Kraus, 2023). These networks may exert increasing control at older ages to contribute to reading automaticity and proficiency. These results may also have implications for the design of new interventions aimed at reinforcing the participation of EF in reading to improve reading skills.
The formation of the reading network: Granular at younger ages, segregated later in life
The human brain is formed into distinct neural networks responsible for essential cognitive and linguistic abilities (Grayson and Fair, 2017). A substantial amount of research indicates that several cognitive processes, such as working memory and inhibition, improve with age and neurobiological changes (Davidson et al., 2006). Following these discoveries, Fair et al. (2012) demonstrated that the structure and organization of the cognitive control networks differed between children and adults in numerous integral ways. First, the networks were not fully separated in children but rather integrated by several central regions compared with adults. Second, the connections between the areas that comprise these networks were weaker in children than in adults. These connections were strengthened with age. These findings suggest age-related increased segregation between networks' connectivity. Moreover, previous studies utilizing this data set demonstrated an increased left-lateralized utilization of regions within language-related networks (Karunanayaka et al., 2007).
Additional studies report increased within-networks connectivity between EF networks, suggesting an overall increased between-networks functional connections along development (Fair et al., 2007). However, based on previous studies suggesting a better integration between visual and auditory modalities in relation to reading proficiency (Breznitz and Misra, 2003), we suggest that the findings of the current study of greater functional connections between the networks compose the reading network in relation to reading proficiency represent the neurobiological maturation of the reading network.
The current study results support the claim that functional connectivity within and between reading-related networks becomes greater along the years.
Study's limitation
Although our study provides a strong base for future studies, it has several limitations. First, missing data for several participants did not allow us to conduct a complete, longitudinal study. Future studies should consider recruiting more participants to improve statistical power and account for missing data sets. Moreover, the current study used the same fMRI narrative comprehension task for the 12 years of the study, which may have a test–retest effect. As this was done to not add confounder to the study with different types of stories, future research should use different age-appropriate stories every year. In addition, some results reported in this study were marginally significant, which might be attributed to the relatively small sample size.
An additional study with a larger sample size is warranted. Also, following the procedure reported in a study that utilized the same data set (Szaflarski et al., 2012), a similar anatomical template was used for the participants in the current study due to a minimal difference in brain anatomy from age 5 years and up. Moreover, Power's atlas was successfully used in young children [as young as 18 months (Farah and Horowitz-Kraus, 2019)] as well as in older children at 12 years (Meri et al., 2023) and hence using this atlas is sufficient. However, a future analysis to determine the correspondence between the Power's atlas and a data-driven method for functional connectivity (i.e., independent component analysis) and the changes along development should be conducted.
Lastly, the absence of information regarding the home literacy environment does not allow us to account for the household's exposure to storytelling or reading at a prereading age. This factor is known to influence the engagement of EF abilities [see Hutton et al. (2015)].
Conclusion
This unique study examined 32 healthy children ages 5–18 annually for 12 years using different behavioral batteries and fMRI scans. We found that basic networks, that is, the phonological, semantic, and syntactic networks, and complex networks, that is, the EF network, are already involved in the prereading age. We also showed that functional connectivity within and between these networks becomes stronger with development. We also found that these networks contribute to proficient reading abilities. Future studies can reveal how EF training may accelerate the reading network's maturation while considering functional and behavioral measures.
Authors' Contributions
T.H.-K.: Data analysis, writing—original draft, writing—review and editing, and supervision. R.M.: Writing—review and editing. S.K.H.: Data collection and writing—review and editing. R.F.: Data anlaysis and writing—review and editing. N.H.: Writing—original draft and data anlaysis. T.R.: Writing—original draft and data anlaysis.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
This study was supported by the National Institute of Child Health and Human Development and the Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD) (PI: T.H.-K.; 1R01HD086011-01A1, PI S.K.H., 2RO1 HD38578-11).
References
- Archer AL, Gleason MM, Vachon VL. Decoding and fluency: Foundation skills for struggling older readers. Learn Disabil Quart 2003;26:89–101. [Google Scholar]
- Breznitz Z. Fluency in Reading: Synchronization of Processes. Lawrence Erlbaum Associates: Mahwah, NJ, USA; 2006. [Google Scholar]
- Breznitz Z, Misra M. Speed of processing of the visual-orthographic and auditory-phonological systems in adult dyslexics: The contribution of “asynchrony” to word recognition deficits. Brain Lang 2003;85:486–502. [DOI] [PubMed] [Google Scholar]
- Carrow-Woolfolk E. Oral and written language scales (OWLS). American Guidance Service: Circle Pines, MN; 1995. [Google Scholar]
- Cartwright KB Insights from cognitive neuroscience: The importance of executive function for early reading development and education. Early Educ Dev 2012;23:24–36. [Google Scholar]
- Davidson MC, Amso D, Anderson LC, et al. Development of cognitive control and executive functions from 4 to 13 years: Evidence from manipulations of memory, inhibition, and task switching. Neuropsychologia 2006;44:2037–2078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fair DA, Dosenbach NU, Petersen SE, et al. Resting state studies on the development of control systems. Cogn Neurosci Attent 2012;2:291–311. [Google Scholar]
- Fair DM, Dosenbach NUF, Church JA, et al. 2007. Development of distinct control networks through segregation and integration. PNAS 2007;104:13507–13512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Farah R, Dudley J, Hutton JS, et al. 2021. Maternal depression is associated with decreased functional connectivity within semantics and phonology networks in preschool children. Depress Anxiety 2021;38(8):826–835. [DOI] [PubMed] [Google Scholar]
- Farah R, Horowitz-Kraus T. Increased functional connectivity within and between cognitive-control networks from early infancy to nine years during story listening. Brain Connect 2019;9(3):285–295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Farah R, Meri R, Kadis DS, et al. Hyperconnectivity during screen-based stories listening is associated with lower narrative comprehension in preschool children exposed to screens vs dialogic reading: An EEG study. PLoS One 2019;14:e0225445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fiez JA, Petersen SE. Neuroimaging studies of word reading. Proc Natl Acad Sci 1998;95:914–921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Friston K, Jezzard P, Turner R. Analysis of functional MRI time-series. Hum Brain Mapp 1994;1:153–171. [Google Scholar]
- Gough PB, Tunmer WE. Decoding, reading, and reading disability. Remed Spec Educ 1986;7:6–10. [Google Scholar]
- Grayson DS, Fair DA. Development of large-scale functional networks from birth to adulthood: A guide to the neuroimaging literature. Neuroimage 2017;160:15–31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holland SK, Plante E, Weber Byars A, et al. Normal fMRI brain activation patterns in children performing a verb generation task. Neuroimage 2001;14:837–843. [DOI] [PubMed] [Google Scholar]
- Holland SK, Vannest J, Mecoli M, et al. Functional MRI of language lateralization during development in children. Int J Audiol 2007;46:533–551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Horowitz-Kraus T. The role of executive functions in fluent reading: Lessons from reading acquisition and remediation. Mind Brain Educ 2023. [Google Scholar]
- Horowitz-Kraus T, Rosch K, Fotang J, et al. Fluent contextual reading is associated with greater synchronization of the visual and auditory networks, fluent reading and better speed of processing in children with dyslexia. Cortex 2023;168:62–75. [DOI] [PubMed] [Google Scholar]
- Hoover WA, Gough PB. The simple view of reading. Reading Writing 1990;2:127–160. [Google Scholar]
- Horowitz-Kraus T, Buck C, Dorrmann D. Altered neural circuits accompany lower performance during narrative comprehension in children with reading difficulties: An fMRI study. Ann Dyslexia 2016;66:301–318. [DOI] [PubMed] [Google Scholar]
- Horowitz-Kraus T, Eaton K, Farah R, et al. Predicting better performance on a college preparedness test from narrative comprehension at the age of 6 years: An fMRI study. Brain Res 2015a;1629:54–62. [DOI] [PubMed] [Google Scholar]
- Horowitz-Kraus T, Toro-Serey C, Difrancesco M. Increased resting-state functional connectivity in the cingulo-opercular cognitive-control network after intervention in children with reading difficulties. PLoS One 2015b;10:e0133762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Horowitz-Kraus T, Vannest JJ, Holland SK. Overlapping neural circuitry for narrative comprehension and proficient reading in children and adolescents. Neuropsychologia 2013;51:2651–2662. [DOI] [PubMed] [Google Scholar]
- Hutton JS, Dudley J, Horowitz-Kraus T, et al. Differences in functional brain network connectivity during stories presented in audio, illustrated, and animated format in preschool-age children. Brain Imaging Behav 2020;14:130–141. [DOI] [PubMed] [Google Scholar]
- Hutton JS, Horowitz-Kraus T, Mendelsohn AL, et al. Home reading environment and brain activation in preschool children listening to stories. Pediatrics 2015;136(3):466–478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karunanayaka PR, Holland SK, Schmithorst VJ, et al. Age-related connectivity changes in fMRI data from children listening to stories. Neuroimage 2007;34:349–360. [DOI] [PubMed] [Google Scholar]
- Katz L, Frost R.. The Reading Process Is Different for Different Orthographies: The Orthographic Depth Hypothesis. Haskins Laboratories Status Report on Speech Research. SR-111/112, 1992; pp. 147–160. [Google Scholar]
- Kim Y-SG. Why the simple view of reading is not simplistic: Unpacking component skills of reading using a direct and indirect effect model of reading (DIER). Sci Stud Reading 2017;21:310–333. [Google Scholar]
- Liu X, Gao Y, Di Q, et al. Differences between child and adult large-scale functional brain networks for reading tasks. Hum Brain Mapp 2018;39:662–679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lynch JS, Van Den Broek P, Kremer KE, et al. The development of narrative comprehension and its relation to other early reading skills. Read Psychol 2008;29:327–365. [Google Scholar]
- Meri R, Farah R, Horowitz-Kraus T. Children with dyslexia utilize both top-down and bottom-up networks equally in contextual and isolated word reading. Neuropsychologia 2020;147:107574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meri R, Hutton J, Farah R, et al. Higher access to screens is related to decreased functional connectivity between neural networks associated with basic attention skills and cognitive control in children. Child Neuropsychol 2023;29:666–685. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Szaflarski JP, Altaye M, Rajagopal A, et al. A 10-year longitudinal fMRI study of narrative comprehension in children and adolescents. Neuroimage 2012;63:1188–1195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Twait E, Farah R, Horowitz-Kraus T. Decreased functional connectivity of the salience network during narrative comprehension in children with reading difficulties: An fMRI study. Neuroimage Clin 2018;20:987–992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wechsler D. Wechsler Abbreviated Scale of Intelligence; 1999. [Google Scholar]
- Whitfield-Gabrieli S, Nieto-Castanon A. Conn: A functional connectivity toolbox for correlated and anticorrelated brain networks. Brain Connect 2012;2(3):125–141. [DOI] [PubMed] [Google Scholar]
- Youssofzadeh V, Vannest J, Kadis DS. fMRI connectivity of expressive language in young children and adolescents. Hum Brain Mapp 2018;39:3586–3596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zelazo P, Müller U.. Executive Functioning in Typical and Atypical Children. In: Blackwell Handbook of Childhood Cognitive Development. (Goswami U. ed.) 2nd ed., 2010; pp. 574–603. [Google Scholar]