Abstract
We conducted a multicentre hospital‐based test‐negative case–control study to measure vaccine effectiveness (VE) against PCR‐confirmed influenza in adult patients with severe acute respiratory infection (SARI) during the 2022/2023 influenza season in Europe. Among 5547 SARI patients ≥18 years, 2963 (53%) were vaccinated against influenza. Overall VE against influenza A(H1N1)pdm09 was 11% (95% CI: −23–36); 20% (95% CI: −4–39) against A(H3N2) and 56% (95% CI: 22–75) against B. During the 2022/2023 season, while VE against hospitalisation with influenza B was >55%, it was ≤20% for influenza A subtypes. While influenza vaccination should be a priority for future seasons, improved vaccines against influenza are needed.
Keywords: hospital, influenza, SARI patients, test‐negative design, vaccine effectiveness
1. BACKGROUND
In February 2022, the World Health Organization (WHO) recommendations for the northern hemisphere 2022–2023 influenza season for trivalent influenza vaccines were to include an A/Wisconsin/588/2019 (H1N1)pdm09‐like virus, an A/Darwin/6/2021 (H3N2)‐like virus and a B/Austria/1359417/2021 (B/Victoria lineage)‐like virus. Quadrivalent vaccine was recommended to contain the above three viruses plus a B/Phuket/3073/2013 (B/Yamagata lineage)‐like virus. 1 Egg‐based vaccines should contain the same last two viruses, in addition to an A/Victoria/2570/2019 (H1N1)pdm09‐like and an A/Darwin/9/2021 (H3N2)‐like virus. 1 Within the European Union/European Economic Area, an early seasonal epidemic threshold (10% positive sentinel specimens) was crossed by week 45, peaking at week 51, 2022. 2 There was co‐circulation of influenza A(H3N2), A(H1N1)pdm09 and B viruses, initially predominated by A subtypes, although patterns of dominance varied across countries. 3 There were hospitalised severe acute respiratory infection (SARI) cases (occurring mainly in older adults) caused by infection with each of these influenza types; however, ultimately, over the 2022/2023 season, influenza A(H1N1)pdm09 predominated among SARI patients. 2
We pooled data from nine study sites in eight European countries participating in the ECDC Vaccine Effectiveness, Burden and Impact Studies (VEBIS) SARI vaccine effectiveness (VE) hospital network 3 (Figure 1) with sufficient data to estimate influenza VE against circulating strains. We estimated VE against hospitalisation due to PCR‐confirmed influenza among SARI patients swabbed between 1 October 2022 and 15 May 2023.
FIGURE 1.
Countries and study sites 1 participating in the Vaccine Effectiveness, Burden and Impact Studies (VEBIS) hospital network by provision of data for this analysis, Europe, 2022–2023 influenza season. 1 Participating sites: Belgium (BE), Croatia (HR), Czechia (CZ), France (FR), Germany (DE), Hungary (HU), Ireland (IE), Lithuania (LT), Malta (MT), Portugal (PT), Romania (RO), Spain (11 regions: ES; Navarra region: NA). Included in this analysis: BE, ES, DE, HR, LT, MT, NA, PT, RO.
2. STUDY DESIGN AND DESCRIPTIVE RESULTS
Participating sites in the network use a common generic protocol 4 , 5 and the test‐negative case–control design. 6 Hospital teams collect demographic, clinical and influenza vaccination information via questionnaire, electronic medical records, vaccine registries or patient interview. We defined a SARI patient as an individual hospitalised for ≥24 h with at least one systemic symptom or sign: fever or feverishness, malaise, headache or myalgia or deterioration of general condition (asthenia, loss of weight or anorexia, confusion or dizziness) and at least one respiratory symptom or sign (cough, sore throat or shortness of breath) at admission or within 48 h after admission. Patients were excluded if their symptoms started (or clearly worsened, if chronic) more than 7 days before swabbing.
Cases were SARI patients PCR‐positive and controls PCR‐negative for influenza within 48 h of admission. We estimated VE overall, by age group (0–17, 18–64, ≥65 years), by target group for influenza vaccination according to each country's specific recommendations and by presence of at least one versus no chronic condition.
We included 5547 SARI patients aged ≥18 years (886 cases of any influenza, 4661 test‐negative controls) from 38 European hospitals, in nine of 13 participating study sites (Figure 1) providing sufficient data for this period.
Sixty‐five per cent of cases and 76% controls were aged ≥65 years, with controls having a median age of 72 years [inter‐quartile range (IQR): 56–81 years]; cases, 76 years (IQR: 65–85). Fifty‐one per cent of cases and 49% controls were female; 68% of cases and 72% of controls had at least one of the five commonly collected chronic conditions (diabetes, heart disease, lung disease, asthma, immunosuppression). Forty per cent of cases and 56% controls were vaccinated against influenza (Table 1).
TABLE 1.
Patient characteristics of cases and controls, Vaccine Effectiveness, Burden and Impact Studies (VEBIS) hospital network, 2022–2023 influenza season, Europe (N = 5547).
| Patient characteristic | Cases (n = 886) | Controls (n = 4661) | ||
|---|---|---|---|---|
| Median age in years (IQR) | 72 (56–81) | 76 (65–85) | ||
| No. | % | No. | % | |
| Age groups (years) | ||||
| 18–64 | 313 | 35.3 | 1137 | 24.4 |
| ≥65 | 573 | 64.7 | 3524 | 75.6 |
| Country‐specific vaccine target group | ||||
| No | 134 | 15.1 | 502 | 10.8 |
| Yes | 752 | 84.9 | 4159 | 89.2 |
| Sex | ||||
| Male | 431 | 48.6 | 2395 | 51.4 |
| Female | 455 | 51.4 | 2266 | 48.6 |
| At least one chronic condition a | ||||
| No | 282 | 31.8 | 1310 | 28.1 |
| Yes | 604 | 68.2 | 3351 | 71.9 |
| Influenza vaccination | ||||
| No | 535 | 60.4 | 2049 | 44.0 |
| Yes | 351 | 39.6 | 2612 | 56.0 |
| Influenza type and subtype | ||||
| Influenza A(H3N2) | 372 | 42.0 | NA | NA |
| Influenza A(H1N1)pdm09 | 245 | 27.7 | NA | NA |
| Influenza A (subtype unknown) | 162 | 18.3 | NA | NA |
| Influenza B | 100 | 11.3 | NA | NA |
| Influenza positive, type unknown | 7 | 0.8 | NA | NA |
| Site/country | ||||
| Navarra | 295 | 33.3 | 2346 | 50.3 |
| Spain | 248 | 28.0 | 1137 | 24.4 |
| Romania | 80 | 9.0 | 120 | 2.6 |
| Belgium | 72 | 8.1 | 351 | 7.5 |
| Lithuania | 67 | 7.6 | 117 | 2.5 |
| Malta | 44 | 5.0 | 288 | 6.2 |
| Croatia | 42 | 4.7 | 170 | 3.6 |
| Portugal | 22 | 2.5 | 40 | 0.9 |
| Germany | 16 | 1.8 | 92 | 2.0 |
| Vaccine product | ||||
| Influvac tetra b | 183 | 52.1 | 1638 | 62.7 |
| Vaxigrip tetra b | 24 | 6.8 | 166 | 6.4 |
| Efluelda b , c (QIV b ) | 1 | 0.3 | 5 | 0.2 |
| Unknown product | 143 | 40.7 | 803 | 30.7 |
Abbreviation: QIV, quadrivalent influenza vaccine.
At least one of five commonly collected chronic conditions: asthma, diabetes, heart disease, lung disease, immunosuppression.
Egg‐based, inactivated, split virion, tetra‐valent vaccine.
High‐dose vaccine.
3. VACCINATION DEFINITIONS AND VACCINE EFFECTIVENESS
We defined the start of the 2022/2023 influenza season in each participating site as the week number in which the first PCR‐confirmed case was reported to the study. We defined vaccination as receipt of influenza vaccine ≥14 days after the start of the current season's vaccination campaign in each participating country. Only vaccines received ≥14 days before onset were considered valid (those vaccinated 1–13 days before onset were excluded; those vaccinated on or after onset were considered unvaccinated).
SARI patients received their influenza vaccination between weeks 36, 2022 and week 3, 2023 (cases) or week 5, 2023 (controls); Figure 2. Vaccination dates coincided with the early start of this 2022/2023 influenza season (Figure 2). The median time from vaccination to onset was 74 days for cases, 88 days for controls. Where influenza vaccine type was known (n = 2017/2963; 68%), all SARI patients were vaccinated with quadrivalent vaccine (Table 1).
FIGURE 2.
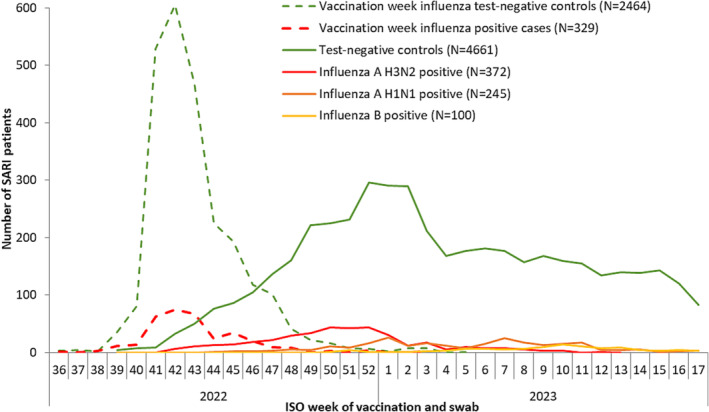
Number of severe acute respiratory infection (SARI) patients by case status and week of vaccination or swab, Vaccine Effectiveness, Burden and Impact Studies (VEBIS) in the hospital setting, influenza 2022/2023 season, Europe (N = 5547).
We compared the odds of vaccination between cases and controls using logistic regression, adjusting the odds ratio (OR) of vaccination among cases and controls by age, sex and presence of at least one of the five chronic conditions listed above. We calculated VE as 1 minus the adjusted OR (expressed as a percentage). We included study site (as a fixed effect) and date of onset (as a restricted cubic spline or categorical variable using onset month) in all analyses. We excluded sites with <10 cases or controls for each influenza (sub)type‐specific analysis. We estimated interaction terms between vaccination and (1) age group and (2) chronic condition status.
Where the number of cases or controls per parameter was <10, we conducted sensitivity analysis using Firth's method of penalised logistic regression (PLR) to assess small sample bias. 7 , 8 We considered estimates having an absolute difference >10% between the PLR and original VE estimate indicative of small sample bias and do not show these estimates. Similarly, we do not show estimates for analyses with <20 vaccinated cases or controls. As we expected most SARI patients to have been systematically tested for both influenza and SARS‐CoV‐2, we performed sensitivity analysis excluding all controls infected with SARS‐CoV‐2.
The VE against hospitalisation with any influenza (all adults) was 27% (95% CI: 13–39), 11% (95% CI: −23–36) against influenza A(H1N1)pdm09 and 20% (95% CI: −4–39) against influenza A(H3N2). Against influenza B, VE was 56% (95% CI: 22–75) overall. Against both influenza A subtypes in patients aged ≥65 years, VE point estimates were 25%. Among those in the influenza vaccine target group, VE point estimates were 20%–22% against influenza A subtypes and 66% against influenza B. Against influenza A(H3N2), VE point estimates were 15% among those with at least one, and 32% among those without any chronic condition (p‐value for interaction: 0.08). Vaccinated case numbers were too small for valid estimates by age group or chronic condition status against influenza B (Table 2).
TABLE 2.
Vaccine effectiveness against influenza hospitalisation, Vaccine Effectiveness, Burden and Impact Studies (VEBIS) in the hospital setting, influenza 2022/2023 season, Europe (N = 5547).
| Main analysis: influenza type and subtype VE, by age group, target group and by chronic condition | |||
|---|---|---|---|
| Influenza type/subtype | Vaccinated/unvaccinated cases; vaccinated/unvaccinated controls | VE a (95% CI) | |
| Influenza (all) | Nine sites b ; N = 5547 | ||
| All ages | 351/535; 2612/2049 | 27 | (13–39) |
| Age group | |||
| 18–64 | 49/264; 297/840 | 24 | (−11–47) |
| ≥65 | 302/271; 2315/1209 | 32 | (16–44) |
| Target group for vaccination | 339/413; 2525/1634 | 29 | (14–41) |
| Chronic condition c | |||
| No | 61/221; 585/725 | 44 | (20–61) |
| Yes | 290/314; 2027/1324 | 20 | (2–35) |
| Influenza A | Nine sites b ; N = 5095 | ||
| All ages | 328/451; 2460/1856 | 20 | (4–34) |
| Age group | |||
| 18–64 | 44/201; 280/777 | 10 | (−34–40) |
| ≥65 | 284/250; 2180/1079 | 29 | (12–42) |
| Target group for vaccination | 317/358; 2378/1472 | 25 | (9–39) |
| Chronic condition c | |||
| No | 54/180; 537/648 | 39 | (10–59) |
| Yes | 274/271; 1923/1208 | 13 | (−7–30) |
| Influenza A(H1N1)pdm09 | Seven sites d ; N = 4270 | ||
| All ages | 94/149; 2374/1653 | 11 | (−23–36) |
| Age group | |||
| 18–64 | 17/86; 265/710 | NA e | NA |
| ≥65 | 77/63; 2109/943 | 25 | (−12–50) |
| Target group for vaccination | 91/109; 2294/1289 | 20 | (−15–45) |
| Chronic condition c | |||
| No | 13/58; 522/598 | NA e | NA |
| Yes | 81/91; 1852/1055 | 8 | (−33–36) |
| Influenza A(H3N2) | Five sites f ; N = 3471 | ||
| All ages | 161/164; 1841/1305 | 20 | (−4–39) |
| Age group | |||
| 18–64 | 19/52; 217/540 | NA e | NA |
| ≥65 | 142/112; 1624/765 | 25 | (−1–44) |
| Target group for vaccination | 155/139; 1778/1039 | 22 | (−2–41) |
| Chronic condition c | |||
| No | 27/62; 385/447 | 32 | (−16–60) |
| Yes | 134/102; 1456/858 | 15 | (−15–38) |
| Influenza B | Four sites g ; N = 2940 | ||
| All ages | 22/63; 1863/992 | 56 | (22–75) |
| Age group | |||
| 18–64 | 5/48; 200/440 | NA e | NA |
| ≥65 | 17/15; 1663/552 | NA e | NA |
| Target group for vaccination | 21/40; 1807/765 | 66 | (31–83) |
| Chronic condition c | |||
| No | 7/34; 430/369 | NA e | NA |
| Yes | 15/29; 1433/623 | NA e | NA |
| Sensitivity analysis: influenza type and subtype VE, after excluding controls with SARS‐CoV‐2 infection, overall, for those aged ≥65 years, and by country‐specific vaccine target groups | |||
|---|---|---|---|
| Influenza type/subtype | Vaccinated/unvaccinated cases; vaccinated/unvaccinated controls | VE a (95% CI) | |
| Influenza (all) | Nine sites b ; N = 4605 | ||
| All ages | 351/535; 2174/1545 | 31 | (17–42) |
| Aged ≥65 years | 302/271; 1927/838 | 36 | (20–48) |
| Target group for vaccination | 339/413; 2105/1195 | 33 | (18–45) |
| Influenza A | Nine sites; b ; N = 4226 | ||
| All ages | 328/451; 2053/1394 | 26 | (10–39) |
| Aged ≥65 years | 284/250; 1822/744 | 33 | (17–47) |
| Target group for vaccination | 317/358; 1988/1071 | 30 | (13–43) |
| Influenza A(H1N1)pdm09 | Six sites h ; N = 3490 | ||
| All ages | 92/142; 1987/1269 | 9 | (−29–36) |
| Aged ≥ 65 years | 75/60; 1767/671 | 25 | (−15–52) |
| Target group for vaccination | 89/104; 1922/963 | 19 | (−20–46) |
| Influenza A(H3N2) | Five sites f ; N = 2876 | ||
| All ages | 161/164; 1551/1000 | 29 | (6–46) |
| Aged ≥65 years | 142/112; 1370/549 | 32 | (9–50) |
| Target group for vaccination | 155/139; 1498/777 | 30 | (7–48) |
| Influenza B | Three sites i ; N = 2502 | ||
| All ages | 20/55; 1608/819 | 62 | (31–79) |
| Aged ≥65 years | 17/14; 1431/433 | NA e | NA |
| Target group for vaccination | 20/37; 1560/616 | 66 | (32–83) |
Odds ratio adjusted (aOR) by country, time (restricted cubic spline of swab date or swab month as categorical variable, depending on model), age (restricted cubic spline or age as linear variable, depending on model), sex, presence/absence of chronic condition (immunocompromised, diabetes, heart disease, lung disease, asthma); VE = 1 − aOR.
Nine sites: Belgium, Croatia, Germany, Lithuania, Malta, Navarra, Portugal, Romania and Spain.
In analyses stratified by chronic condition, the adjustment for presence/absence of chronic condition was removed.
Seven sites: Belgium, Croatia, Lithuania, Malta, Navarra, Romania and Spain.
Fewer than 10 cases per parameter in the model.
Five sites: Belgium, Germany, Malta, Navarra and Spain.
Four sites: Belgium, Lithuania, Navarra and Spain.
Six sites: Belgium, Croatia, Lithuania, Navarra, Romania and Spain.
Three sites: Belgium, Navarra and Spain.
The difference between VE estimates from the main and the sensitivity analysis excluding controls with SARS‐CoV‐2 infection was ≤9%, depending on influenza (sub)type (Table 2).
4. DISCUSSION AND CONCLUSION
Our results from this multi‐country European hospital study suggest that, for the 2022/2023 influenza season, adult SARI patients had the highest all‐age VE against influenza B (56%). The VE was lower against influenza A(H3N2) (20%) and against influenza A(H1N1)pdm09 (11%). Point estimates were highest in older adults (≥65 years; 29% against influenza A) and those in the vaccine target group (66% against influenza B).
Despite the early season start, the vaccine coverage among controls (66% in the age group ≥65 years; data not shown) was similar to previous seasons. 9 Our point estimates for VE against any influenza A and against A(H3N2) were similar (20%) to those found in our interim study (24%) 3 and to our 2017/2018 end‐of‐season VE estimates for influenza A(H3N2) in a hospital setting (24%). 9 Results from a test‐negative design case–control study in the United States and one in Italy for the 2022/2023 season reported higher overall VE results against influenza A and against A(H3N2) than ours, at 35% (95% CI: 27%–43%) 10 and 38% (95% CI: −34–74), 11 respectively. However, the VE in those aged ≥65 years in the Italian study was 24% (95% CI: −86–72), very close to the 25% (95% CI: −1–44) observed in our study. Although the US study included only SARS‐CoV‐2‐negative controls, their VE for influenza A was still higher than those in our sensitivity analysis including only SARS‐CoV‐2‐negative controls, although 95% CIs overlapped (26%; 95% CI: 10–39). 10 In Europe, most circulating influenza A(H3N2) viruses belonged to haemagglutinin clade 2.2. 12 In our study, from five sites sequencing viruses, 46/82 (56%) influenza A(H3N2) virus samples sequenced were clade 2b, 27/82 (33%) clade 2a.1b, 5/82 (6%) clade 2a.3 and 4/82 (5%) clade 2a.1 (data not shown).
Although influenza A(H3N2) was predominant among patients in primary care in Europe, influenza A(H1N1)pdm09 dominated among SARI patients. 2 The European Centre for Disease Prevention and Control (ECDC) reported that most sequenced A(H1N1)pdm09 viruses early on in the 2022/2023 season had haemagglutinin genes in clade 5a.2, which were poorly recognised by human sera. 12 Later circulating viruses contained several amino acid substitutions, and, indeed, most A(H1N1)pdm09 virus samples sequenced from five of our study sites were 5a.2a (55/73; 75%) or 5a.2a.1 (17/73; 23%) (data not shown). This could explain the surprisingly low VE against influenza A(H1N1)pdm09 among hospitalised SARI patients found in our study.
All 19 sequenced influenza B/Victoria viruses in our study were B/Austria/1359417/2021, matching the 2022/2023 season's vaccine strain for the northern hemisphere and likely explaining the 54% overall VE for SARI hospitalised patients against influenza B in our study.
Limitations include small sample size for younger adults (18–64 years), which, together with low vaccination coverage in this age group, led to no estimates by subtype (or for influenza B) being shown in this age group and lower precision for influenza B estimates. Multi‐country studies are inherently heterogenous, but use of a common protocol and study design, and collection of individual‐level data among our study sites, permits a larger sample size to provide robust results for continued monitoring of VE against influenza across Europe.
During the 2022/2023 season, while VE against hospitalisation with influenza B was >55%, it was ≤20% for influenza A subtypes. While better coverage for influenza vaccination should be a priority for future influenza seasons, efforts to develop improved vaccines against influenza are needed. As most hospitalised influenza patients are older adults (≥65 years), improvement of influenza A vaccines in particular is critical to help prevent severe illness among older patients.
AUTHOR CONTRIBUTIONS
Angela M C Rose: Conceptualization; formal analysis; funding acquisition; methodology; project administration; software; supervision; validation; visualization; writing—original draft; writing—review and editing. Francisco Pozo: Data curation; investigation; methodology; project administration; resources; supervision; visualization; writing—review and editing. Iván Martínez‐Baz: Data curation; formal analysis; investigation; methodology; resources; writing—review and editing. Clara Mazagatos: Data curation; formal analysis; investigation; methodology; resources; writing—review and editing. Nathalie Bossuyt: Data curation; formal analysis; investigation; methodology; resources; writing—review and editing. John Paul Cauchi: Data curation; formal analysis; investigation; methodology; resources; writing—review and editing. Goranka Petrovic: Data curation; formal analysis; investigation; methodology; resources; writing—review and editing. Isabela I. Loghin: Data curation; formal analysis; investigation; methodology; resources; writing—review and editing. Roberta Vaikutyte: Data curation; formal analysis; investigation; methodology; resources; writing—review and editing. Silke Buda: Data curation; formal analysis; investigation; methodology; resources; writing—review and editing. Ausenda Machado: Data curation; formal analysis; investigation; methodology; resources; writing—review and editing. Roisin Duffy: Data curation; formal analysis; investigation; methodology; resources; writing—review and editing. Beatrix Oroszi: Data curation; formal analysis; investigation; methodology; resources; writing—review and editing. Jennifer Howard: Data curation; formal analysis; methodology; project administration; software; validation; visualization; writing—review and editing. Aitziber Echeverria: Data curation; formal analysis; investigation; methodology; resources; writing—review and editing. Cristina Andreu Salete: Data curation; formal analysis; investigation; methodology; resources; writing—review and editing. Cyril Barbezange: Data curation; formal analysis; investigation; methodology; resources; writing—review and editing. Aušra Džiugytė: Data curation; formal analysis; investigation; methodology; resources; writing—review and editing. Diana Nonkovic: Data curation; formal analysis; investigation; methodology; resources; writing—review and editing. Corneliu Petru Popescu: Data curation; formal analysis; investigation; methodology; resources; writing—review and editing. Fausta Majauskaite: Data curation; formal analysis; investigation; methodology; resources; writing—review and editing. Kristin Tolksdorf: Data curation; formal analysis; investigation; methodology; resources; writing—review and editing. Verónica Gómez: Data curation; formal analysis; investigation; methodology; resources; writing—review and editing. Lisa Domegan: Data curation; formal analysis; investigation; methodology; resources; writing—review and editing. Judit Krisztina Horvath: Data curation; formal analysis; investigation; methodology; resources; writing—review and editing. Jesus Castilla: Data curation; formal analysis; investigation; methodology; resources; writing—review and editing. Miriam García‐Vázquez: Data curation; formal analysis; investigation; methodology; resources; writing—review and editing. Thomas Demuyser: Data curation; formal analysis; investigation; methodology; resources; writing—review and editing. Maria‐Louise Borg: Data curation; formal analysis; investigation; methodology; resources; writing—review and editing. Irena Tabain: Data curation; formal analysis; investigation; methodology; resources; writing—review and editing. Mihaela Lazar: Data curation; formal analysis; investigation; methodology; resources; writing—review and editing. Ieva Kubiliute: Data curation; formal analysis; investigation; methodology; resources; writing—review and editing. Ralf Dürrwald: Data curation; formal analysis; investigation; methodology; resources; writing—review and editing. Raquel Guiomar: Data curation; formal analysis; investigation; methodology; resources; writing—review and editing. Joan O'Donnell: Data curation; formal analysis; investigation; methodology; resources; writing—review and editing. Katalin Krisztalovics: Data curation; formal analysis; investigation; methodology; resources; writing—review and editing. Nathalie Nicolay: Conceptualization; visualization; writing—review and editing. Sabrina Bacci: Conceptualization; visualization; writing—review and editing. Esther Kissling: Conceptualization; formal analysis; funding acquisition; methodology; project administration; software; supervision; validation; visualization; writing—original draft; writing—review and editing. VEBIS SARI VE network team: Data curation; investigation; methodology; resources; validation; writing—review and editing.
CONFLICT OF INTEREST STATEMENT
No conflict of interest declared.
PEER REVIEW
The peer review history for this article is available at https://www.webofscience.com/api/gateway/wos/peer-review/10.1111/irv.13255.
ETHICS STATEMENT
The planning, conduct and reporting of the studies was in line with the Declaration of Helsinki. Official ethical approval was not required if studies were classified as being part of routine care/surveillance (Spain, Malta). In Belgium and Germany, VE is included in SARI surveillance. For Belgium, the study protocol was approved by the central Ethical Committee (CHU ST Pierre, Bruxelles) and each participating hospital's local ethical committees in 2011 (AK/12‐02‐11/4111), updated in 2014 (B.U.N. 143201215671). The German SARI surveillance was approved by the Charité‐Universitätsmedizin Berlin Ethical Board (Reference EA2/218/19). Other study sites obtained local ethical approval from a national review board (Croatia: approved 24 May 2021 and 26 January 2022, Ethics committee of the Croatian Institute of Public Health, Klasa:030‐02/21‐01/1, Ur.broj:381‐15‐21‐7; Klasa:030‐02/21‐01/1, Ur.broj:381‐15‐22‐14; Lithuania: approved by Lithuanian Bioethics Committee on 27th May 2020, and later permission extended for the study period, No. L‐20‐3/1; Navarra: PI2020/45; Portugal: approved 19 January 2021 by the Ethics Committee of Instituto Nacional de Saúde Dr Ricardo Jorge, no registration number given); Romania: approved by the Ethics Committee of the Ministerul Apărării Naionale Institutul Naional de Cercetare pentru Dezvoltare Medico‐Militară ‘Cantacuzino’ for the period 2022–2023, No. CE199/2022.
ACKNOWLEDGEMENTS
Study teams are very grateful to all patients, physicians, laboratory teams and national and regional epidemiologists who have contributed to the studies.
Participating laboratories submitted their sequences to GISAID (www.gisaid.org) for easy sharing with the central laboratory in Madrid.
The Hungarian study team works as part of the National Laboratory for Health Security Hungary (RRF‐2.3.1‐21‐2022‐00006) supported by the National Research, Development and Innovation Office (NKFIH).
Rose AMC, Pozo F, Martínez‐Baz I, et al. Vaccine effectiveness against influenza hospitalisation in adults during the 2022/2023 mixed season of influenza A(H1N1)pdm09, A(H3N2) and B circulation, Europe: VEBIS SARI VE hospital network. Influenza Other Respi Viruses. 2024;18(2):e13255. doi: 10.1111/irv.13255
Contributor Information
Angela M. C. Rose, Email: a.rose@epiconcept.fr.
VEBIS SARI VE network team:
Itziar Casado, Camino Trobajo‐Sanmartín, Manuel García Cenoz, Nerea Egüés, Guillermo Ezpeleta, Ana Navascués, Miguel Fernández‐Huerta, Ana Miqueleiz, and Carmen Ezpeleta
Belgium SARI Surveillance Network (BelsariNet):
Lucie Seyler, Arne Witdouck, Caroline Wylock, Els Van Nedervelde, Svea Geeroms, Virgini Van Buggenhout, Nathalie Bossuyt, Sarah Denayer, Cyril Barbezange, Benédicte Lissoir, Xavier Holemans, Marc Hainaut, Nicolas Dauby, Benedicte Delaere, Marc Bourgeois, Evelyn Petit, Marijke Reynders, Door Jouck, Koen Magerman, Marieke Bleyen, Melissa Vermeulen, François Dufrasne, Tanya Melillo, Stephen Abela, Gerd Xuereb, Maja Ilić, Ivan Mlinarić, Iva Pem Novosel, Petra Tomaš Petrić, Ivana Bočina, Svjetlana Karabuva, Mihaela Čikeš Šimunković, Suzana Mladinov, Mateo Ćurin, Željka Čuljak Jurić, Joško Markić, Ivana Jukić, Ina Tomas, Marija Tonkić, Odette Popovici, Catalina Pascu, Alina Ivanciuc, Iulia Bistriceanu, Sorin Dinu, Mihaela Oprea, Maria Elena Mihai, Alexandru Marin, Gratiela Tardei, Alma‐Gabriela Tudor, Emonoil Ceausu, Simin Aysel Florescu, Elena Duca, Catalina Mihaela Luca, Carmen Mihaela Dorobat, Aukse Mickiene, Monika Kuliese, Ligita Jancoriene, Birute Zablockiene, Luise Goerlitz, Ute Preuss, Barbara Biere, Dschin‐Je Oh, Janine Reiche, Marianne Wedde, Irina Kislaya, Ana Paula Rodrigues, Camila Henriques, Aryse Mello, Gergő Túri, Katalin Krisztalovics, Annamária Ferenczi, and Krisztina Mucsányiné Juhász
DATA AVAILABILITY STATEMENT
Data subject to third party restrictions. The data that support the findings of this study are available from ECDC. Restrictions apply to the availability of these data, which were used under licence for this study. Data are available from the authors with the permission of ECDC and from all sites whose data are included.
REFERENCES
- 1. World Health Organization (WHO) . Recommended composition of influenza virus vaccines for use in the 2022‐2023 northern hemisphere influenza season [Internet]. 2022. Available at: https://cdn.who.int/media/docs/default-source/influenza/who-influenza-recommendations/vcm-northern-hemisphere-recommendation-2022-2023/202202_recommendation.pdf
- 2. World Health Organisation and European Centre for Disease Prevention and Control . Flu News Europe season overview. 2023. Available at: https://flunewseurope.org/SeasonOverview
- 3. Kissling E, Maurel M, Emborg HD, et al. Interim 2022/23 influenza vaccine effectiveness: six European studies, October 2022 to January 2023. Eurosurveillance. 2023;28(21):2300116. doi: 10.2807/1560-7917.ES.2023.28.21.2300116 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. I‐MOVE influenza and COVID‐19 networks . I‐MOVE‐COVID‐19 hospital network VE generic protocol [internet]. Paris, France: Epiconcept; 2021. Available at: https://www.imoveflu.org/wp-content/uploads/2021/03/08feb2021_draft_generic_VE_protocol_hospital-based_COVID-19_v07.pdf
- 5. European Centre for Disease Prevention and Control . Core protocol for ECDC VEBIS studies of COVID‐19 vaccine effectiveness against hospitalisation with severe acute respiratory infection laboratory‐confirmed with SARS‐CoV‐2 or seasonal influenza ‐ version 2.0 [Internet]. Stockholm: ECDC; 2023. Available at: https://www.ecdc.europa.eu/sites/default/files/documents/covid-19-vaccine-effectiveness-sari-protocol-version-2.pdf
- 6. Jackson ML, Nelson JC. The test‐negative design for estimating influenza vaccine effectiveness. Vaccine. 2013;31(17):2165‐2168. doi: 10.1016/j.vaccine.2013.02.053 [DOI] [PubMed] [Google Scholar]
- 7. Peduzzi P, Concato J, Feinstein AR, Holford TR. Importance of events per independent variable in proportional hazards regression analysis. II. Accuracy and precision of regression estimates. J Clin Epidemiol. 1995;48(12):1503‐1510. doi: 10.1016/0895-4356(95)00048-8 [DOI] [PubMed] [Google Scholar]
- 8. Covenay J. FIRTHLOGIT: Stata module to calculate bias reduction in logistic regression [Internet]. Boston: Boston College Department of Economics; 2008. [cited 2020 Feb 3]. Available at: https://www.google.com/search?client=firefox-b-d&q=FIRTHLOGIT%3A+Stata+module+to+calculate+bias+reduction+in+logistic+regression
- 9. Rose AMC, Kissling E, Gherasim A, et al. Vaccine effectiveness against influenza A(H3N2) and B among laboratory‐confirmed, hospitalised older adults, Europe, 2017‐18: a season of B lineage mismatched to the trivalent vaccine. Influenza Other Respi Viruses. 2020;14(3):302‐310. doi: 10.1111/irv.12714 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Tenforde MW, Weber ZA, Yang DH, et al. Influenza vaccine effectiveness against influenza‐A‐associated emergency department, urgent care, and hospitalization encounters among U.S. adults, 2022‐2023. J Infect Dis. 2023;jiad542. doi: 10.1093/infdis/jiad542 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Domnich A, Orsi A, Ogliastro M, et al. Influenza vaccine effectiveness in preventing hospital encounters for laboratory‐confirmed infection among Italian adults, 2022/23 season. Vaccine. 2023;41(33):4861‐4866. doi: 10.1016/j.vaccine.2023.06.072 [DOI] [PubMed] [Google Scholar]
- 12. European Centre Diseases Prevention and Control (ECDC) . Influenza virus characterization ‐ summary Europe, February 2023 [Internet]. 2023. [cited 2023 Sep 1]. Available at: https://www.ecdc.europa.eu/en/publications-data/influenza-virus-characterization-summary-europe-february-2023
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Data subject to third party restrictions. The data that support the findings of this study are available from ECDC. Restrictions apply to the availability of these data, which were used under licence for this study. Data are available from the authors with the permission of ECDC and from all sites whose data are included.


