Abstract
Objective
The aim of this study is to investigate how much intermittent hypoxemia and airflow limitation contribute to cognitive impairment in overlap syndrome (OS), which is the coexistence of two common diseases, obstructive sleep apnea hypopnea syndrome (OSAHS) and chronic obstructive pulmonary disease (COPD).
Methods
We conducted a cross-sectional study of patients with OSAHS, COPD or OS, compared with normal controls, to determine the association between sleep apnea/pulmonary function-related indicators and cognitive dysfunction in individuals with OSAHS, COPD or OS.
Results
A total of 157 participants were recruited. Both OSAHS and OS presented lower adjusted Montreal cognitive assessment (MoCA) scores compared with COPD group. In addition, the MoCA score was significantly lower in COPD group compared with control group. The incidence of cognitive impairment was 57.4% in OSAHS group, and 78% in OS group, which were significantly higher than COPD group (29%) and control group (8.8%). Furthermore, a broader range of cognitive domains were affected in OS group compared with OSAHS group. Elevated levels of oxygen desaturation index (ODI) and/or apnea hypopnea index (AHI) were positively correlated with increased Epworth sleeping scale (ESS) in OSAHS and OS. Forced vital capacity (FVC), forced expiratory volume in 1 s (FEV1) and peak expiratory flow (PEF) were positively correlated with cognitive scores in OSAHS but not in OS. Serum level of hypoxia-inducible factor-1α (HIF-1α) was significantly higher in OS. Logistic regression identified ODI as an independent risk factor for cognitive impairment in OS, while severity of snoring and PEF were independent risk factors in OSAHS.
Discussion
This study revealed significant cognitive impairment in OS, OSAHS and COPD. Sleep-related indicators are warranted in OS patients for detection, differentiation and grading of cognitive impairment, whereas pulmonary functions are warranted in OSAHS patients for detection and early intervention of cognitive impairment.
Keywords: Obstructive sleep apnea hypopnea syndrome, chronic obstructive pulmonary disease, overlap syndrome, hypoxia-inducible factor-1α, brain-derived neurotrophic factor, cognitive impairment
Introduction
Obstructive sleep apnea hypopnea syndrome (OSAHS) and chronic obstructive pulmonary disease (COPD) are both common diseases, and the coexistence of these two diseases is called overlap syndrome (OS). Individuals with OS suffer more severe nocturnal oxygen desaturation and higher rates of all-cause mortality compared to those with COPD or OSAHS alone, 1 raising a need for early identification and intervention of individuals with OS.
Cognitive dysfunction has been increasingly reported as a comorbidity in both OSAHS and COPD cases.2,3 At present, decline in instantaneous memory, attention and executive function has been consistently detected in OSAHS, whereas conclusions on impairment of other cognitive domains remain to be further confirmed. Though OS may cause more severe cognitive impairments than OSAHS or COPD alone, how much intermittent hypoxemia and airflow limitation contribute to cognitive impairment in OS is less clear.
Montreal cognitive assessment (MoCA) scale is one of the main tools assessing cognitive dysfunction. It has been translated into a variety of languages including mandarin (Beijing version of Montreal Cognitive Assessment, MoCA-BJ), and validated in multiple studies. 4
Both hypoxia-inducible factor-1α (HIF-1α) and brain-derived neurotrophic factor (BDNF) play significant roles in response to hypoxia, 5 and are involved in cognitive impairment following intermittent hypoxia. 6 As the active subunit of HIF-1, HIF-1α promotes hypoxia-induced neuronal apoptosis and subsequent neurological impairment. 7 OSAHS has been previously shown to present elevated serum level of HIF-α due to associated long-term intermittent hypoxia. 8 BDNF was also elevated in serum in OSAHS patients with MoCA-measured neurocognitive impairment, suggesting a neuroprotective response to intermittent hypoxia in these patients. 9 Changes in peripheral levels of these two proteins, and their predictive and diagnostic values for cognitive dysfunction in OS patients remain unclear.
The aim of our study was to determine the incidence of cognitive impairment in OSAHS, COPD, and OS patients, as well as to determine the associations among intermittent hypoxia, airflow limitation and cognitive dysfunctions. In addition, the predictive and diagnostic values of HIF-1α and BDNF on determining cognitive impairment were also evaluated in the study.
Methods
Study design
In this cross-sectional study, we analyzed data of individuals who were admitted to the Department of Respiratory and Critical Medicine of Shanghai Putuo District Liqun Hospital between April 2020 and December 2021 (N = 157). Ethics approval was granted by the Human Research Ethics Committee of Shanghai Liqun Hospital (Reference number: JD-LK-2018-004-02).
Obstructive Sleep Apnea Hypopnea Syndrome was diagnosed according to the American Academy of Sleep Medicine (AASM) clinical practice guideline for diagnostic testing for adult obstructive sleep apnea, 10 which utilized apnea hypopnea index (AHI) ≥ 5 events/h as a threshold. COPD was diagnosed according to the Global Initiative for Chronic Obstructive Lung Disease (GOLD) Guideline, which utilized FEV1/FVC less than 0.7 to confirm persistent airflow limitation. 11 OS was diagnosed when both above-mentioned diagnostic criteria were met. Individuals were included if the following criteria were met: (1) Patients were diagnosed with OSAHS for the first time without any relevant treatment; (2) Patients were devoid of central nervous system (CNS) injury; (3) Patients were able to complete relevant tests; (4) Patients consented to participate. Individuals were excluded when the following criteria were met: (1) Patientshad pre-existed cognitive dysfunction caused by other reasons; (2) Patients presented chronic lung disease other than COPD; (3) Patients had other significant comorbidities; (4) Patients consumed drugs affecting CNS. The study was conducted per the principles of the Declaration of Helsinki, and written informed consent was obtained from all patients. Demographic information was collected, sleepiness was evaluated using Epworth Sleepiness Scale (ESS), and cognition was evaluated using MoCA-BJ prior to the sleep test. Blood was collected following the sleep test and analyzed accordingly.
All individuals were then categorized into one of the following diagnostic categories: (i) Control group (no COPD no OSAHS) (N = 34); (ii) OSAHS group (N = 54); (iii) COPD group (N = 31); (iv) OS group (N = 38).
Data collection
The sleep test was performed at the hospital. A 3-type portable monitor (PM) (Alice NightOne, Philip) was used to measure relevant parameters. Scoring of sleep and related events were based on criteria of the American Academy of Sleep Medicine (AASM). 12 All data were automatically generated, followed by manual calibration.
Cognitive functions were assessed with MoCA-BJ. A cutoff of 26 was set to determine existence of cognitive impairment, and a calibration was performed when the individual had an education for less than 9 years. Patients with scores of 26 or above were categorized as normal cognition, and patients with scores of 25 or below were categorized as cognitive impairment. All participants completed ESS and a scale on snoring severity (0 = no snore, 1 = mild snore; 2 = moderate snore; 3 = severe snore). 13
After inhaling bronchodilators, the pulmonary ventilation function was determined by a pulmonary function testing system (Jaeger MasterScreen, Leibnizstrasse, Hoechberg, Germany).
2 mL arterial blood was drawn for gas analysis, with PO2 and PCO2 measured in a blood gas analyzer (RAPIDPoint 500, SIEMENS), and 5 mL of fasting venous blood was drawn, processed and stored for further tests. The concentrations of HIF-1α (Cusabio, Cat# CSB-E12112h) and BDNF (R&D, Cat# MAB848R/BAM648) were measured by sandwich ABC-ELISA kits on a microplate reader (DENLEY DRAGON Wellscan MK 2, ThermoFisher, Finland), and analyzed by Ascent software for Multiskan. The detection range of the HIF-1α ELISA kit is 62.5 pg/mL – 4000 pg/mL, with intra-assay %coefficient of variation (CV) less than 8% and inter-assay %CV less than 10%. The lower detection limit of the BDNF ELISA kit is 0.6 ng/mL, with intra-assay %CV less than 10% and inter-assay %CV less than 15%.
Data analysis
Statistical analysis was performed using SPSS (IBM, version 28.0.1.1 (14)), with p value <.05 considered statistically significant. Continuous variables were calculated as mean value ± standard deviation (SD), and categorical variables were calculated as frequencies. Group differences were assessed using non-parametric analysis for continuous data, and Chi-square analysis for categorical data. Correlations were assessed using Spearman correlation. The risk factors for cognitive impairment were determined using logistic regression, and diagnostic value of HIF-1α and BDNF on cognitive dysfunction was determined by multiple receiver operating characteristic (ROC) analyses. Graphs were made using GraphPad Prism 8.4.2.
Results
Sample characteristics
A total of 157 individuals were recruited in the study, including 104 males and 53 females. The average age was 69 ± 12 (30–92) years. There was no significant difference in age (p = .075) and education level (p = .05) among groups. The male to female ratio was significantly higher in COPD group, compared with controls (p = .010). As expected, pack-years of smoking was significantly different among groups (p = .001), and BMI was significantly different among groups (p = .001). The demographics are presented in (Table 1).
Table 1.
Demographic characteristics and comparison of different parameters across four groups (total N = 157).
| Control (N = 34) | OSAHS (N = 54) | COPD (N = 31) | OS (N = 38) | p value | |
|---|---|---|---|---|---|
| Demographics | |||||
| Age (years) | 67 ± 13 | 66 ± 12 | 72 ± 9 | 72 ± 10 | .075 |
| Sex (M:F) | 15:19 bcd | 36:18 c | 25:6ab | 28:10a | .01 |
| Education (years) | 9 ± 4 | 9 ± 4 | 8 ± 3 | 7 ± 3a | .05 |
| BMI (kg/m2) | 23 ± 4b | 26 ± 4 c | 23 ± 4b | 25 ± 4 | .001** |
| Smoking (pack-years) | 242 ± 671 d | 339 ± 584c | 803 ± 942 b | 694 ± 988a | .001** |
| Alcohol (Y/N) | 0.24 ± 0.43 | 0.07 ± 0.26 | 0.23 ± 0.43 | 0.24 ± 0.44 | .101 |
| Cognitive parameters | |||||
| MoCA (points) | 28 ± 1.4 cd | 24 ± 2.2 d | 26 ± 1.8 d | 24 ± 2.3 bc | <.001** |
| Percentage of cognitive impairment (%) | 8.8 bcd | 57.4 acd | 29.0abd | 78.9 bc | <.001** |
| Visuospatial/executive | 4 ± 0.8 bd | 3 ± 0.9 ad | 4 ± 0.8 d | 3 ± 0.9 cb | <.001** |
| Naming | 3 ± 0.0 | 3 ± 0.2 | 3 ± 0.0 | 3 ± 0.3 | .194 |
| Attention | 5 ± 0.5 d | 5 ± 0.8 ad | 5 ± 0.6d | 5 ± 0.6 bc | <.001** |
| Language | 3 ± 0.0 b | 3 ± 0.4 a | 3 ± 0.0 | 3 ± 0.3 | .019* |
| Abstraction | 2 ± 0.4 d | 2 ± 0.6 d | 2 ± 0.5 d | 1 ± 0.5 bc | <.001** |
| Delayed recall | 4 ± 0.7 bcd | 3 ± 1.0 a | 3 ± 0.9 a | 3 ± 1.0a | <.001** |
| Orientation | 6 ± 0.3 | 6 ± 0.5 | 6 ± 0.3 | 6 ± 0.5a | .081 |
| Sleep parameters | |||||
| ESS | 5 ± 1.5 bcd | 9 ± 2.6 ac | 6 ± 1.9 abd | 9 ± 3.0 c | <.001** |
| Percentage of moderate-severe snoring (%) | 0 bd | 62.96 ac | 0bd | 57.89ac | <.001** |
| AHI (events/hr) | 3.28 ± 1.45 bd | 24.19 ± 15.53 c | 3.55 ± 1.94 d | 22.09 ± 17.34 c | <.001** |
| ODI (events/hr) | 5.28 ± 4.29 bd | 22.85 ± 14.94 c | 3.81 ± 2.97 d | 23.82 ± 20.92 c | <.001** |
| TS90% (%) | 4.31 ± 10.21 bd | 10.28 ± 14.57 d | 7.16 ± 11.72d | 26.38 ± 30.90 bc | <.001** |
| MSaO2 (%) | 94.59 ± 1.85 bd | 92.94 ± 2.26ad | 93.77 ± 2.40d | 90.61 ± 4.08 bc | <.001** |
| LSaO2 (%) | 87.56 ± 3.58 bd | 78.74 ± 9.22 c | 84.81 ± 5.13 d | 79.55 ± 6.55 c | <.001** |
| Lung function | |||||
| %FEV1 | 96.77 ± 21.55 cd | 91.13 ± 18.87 cd | 53.34 ± 22.90 b | 56.86 ± 21.05 b | <.001** |
| FEV1 (L) | 2.23 ± 0.71 cd | 2.25 ± 0.60 cd | 1.29 ± 0.65 ab | 1.32 ± 0.51 b | <.001** |
| %FVC | 96.46 ± 21.28 cd | 91.67 ± 18.67 cd | 72.27 ± 24.62 ab | 76.21 ± 22.33 b | <.001** |
| FVC (L) | 2.80 ± 0.91 | 2.87 ± 0.77 | 2.30 ± 1.00a | 2.30 ± 0.73a | <.001** |
| FEV1/FVC (%) | 80.01 ± 6.04 cd | 78.79 ± 5.58 d | 55.16 ± 10.04 b | 56.93 ± 9.41 b | <.001** |
| PEF (L/min) | 5.84 ± 2.22 cd | 5.89 ± 1.98 cd | 3.46 ± 1.62 b | 3.45 ± 1.42 b | <.001** |
| PaO2 (mmHg) | 84.76 ± 16.39 | 81.65 ± 15.43 | 87.32 ± 19.42 | 79.84 ± 18.89 | .328 |
| PaCO2 (mmHg) | 37.27 ± 5.35 c | 39.62 ± 5.45 | 42.59 ± 7.06a | 40.60 ± 6.30a | .015* |
| Biomarkers | |||||
| HIF-1α (pg/mL) | 364.41 ± 195.96 d | 418.50 ± 271.92 d | 405.85 ± 258.43d | 597.38 ± 427.54 bc | .043* |
| BDNF (ng/mL) | 11.43 ± 8.09 | 13.59 ± 10.11 | 13.20 ± 10.81 | 14.20 ± 9.79 | .648 |
Abbreviations: BMI: body mass index; FEV1: forced expiratory volume in 1 s; FVC: forced vital capacity; PEF: peak expiratory flow; PaO2: partial pressure of oxygen; PaCO2: partial pressure of carbon dioxide; MoCA: Montreal cognitive assessment; ESS: epworth sleepiness scale; AHI: apnea hypopnea index; ODI: oxygen desaturation index; TS90%: percent nighttime with oxygen saturation less than 90%; MSaO2: mean arterial oxygen saturation (%); LSaO2: lowest arterial oxygen saturation (%); HIF-1α: hypoxia-inducible factor-1α; BDNF: brain-derived neurotrophic factor.
*, p < .05; **, p < .01; ***, p < .001.
ap < .05 compared with control group.
bp < .05 compared with OSAHS group.
cp < .05 compared with COPD group.
dp < .05 compared with OS group.
Cognitive impairment
Adjusted MoCA score was significantly different among groups (p < .001). The scores were significantly lower in OSAHS and OS groups compared with control group (both p < .001), and was significantly lower in OS group compared with OSAHS (p = .010). Interestingly, the adjusted MoCA score also decreased significantly in COPD group compared with control group (p = .007). Chi-square analysis revealed that the prevalence of cognitive impairment varied significantly across groups (p < .001), with OS group (78.95%) and OSAHS group (57.41%) presenting higher prevalence than COPD group (29.03%) and control group (8.82%) (Table 1).
The cognitive domains were affected differently across groups. Specifically, scores of visuospatial/executive function (p < .001), attention (p < .001), language (p = .019), abstraction (p < .001) and delayed recall (p < .001) were significantly different, while naming (p = .194) and orientation (p = .081) remained at similar level across groups. Both OSAHS and OS groups presented impairment in multiple cognitive domains, with OS group presenting lower scores in more cognitive domains. In addition, the scores for visual spatial and executive (p = .035), attention (p = .002) and abstraction (p < .001) were significantly lower in OS group compared with OSAHS group, indicating higher vulnerability to impairment in different cognitive domains in OS. In contrast, cognition was relatively preserved in COPD, with delayed recall being the only cognitive domain affected compared with controls (p < .001) (Table 1).
Sleep apnea-hypopnea and daytime sleepiness
As an effect from apnea/hypopnea, ESS was significantly higher in OSAHS and OS groups, compared with COPD group (both p < .001) and control group (both p < .001), but remained at a similar level between OS and OSAHS groups (p = .917) (Figure 1(a)). In addition, the ESS score in COPD group was also significantly higher than controls (p = .035), indicating that intermittent hypoxia and airflow limitation may both lead to daytime sleepiness. Furthermore, OS and OSAHS groups presented more moderate-severe snoring, compared with COPD and control groups (both p < .001) (Table 1).
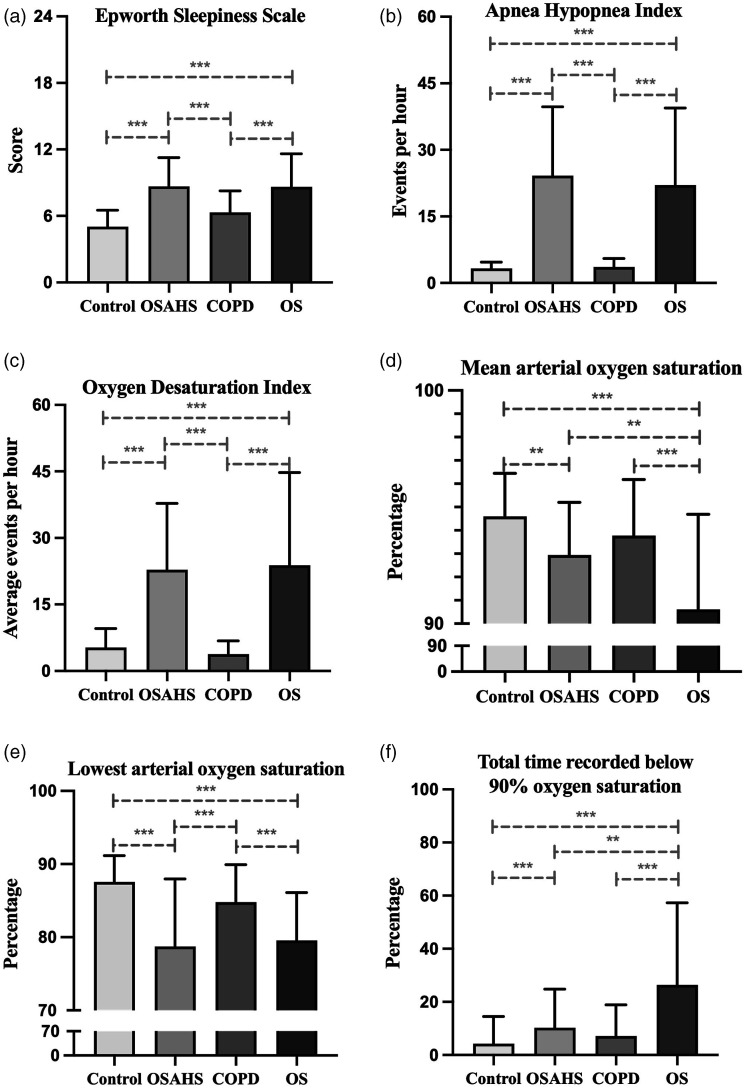
Figure 1.
Comparison of sleep-related parameters across groups. (a) Epworth sleepiness scale (score); (b) apnea hypopnea index (events/hr); (c) oxygen desaturation index (events/hr); (d) mean arterial oxygen saturation (%); (e) lowest arterial oxygen saturation (%); (f) TS90% (%) of each group was indicated as mean ± SD. *, p < .05; **, p < .01, ***, p < .001.
As expected, apnea hypopnea index (AHI) and oxygen desaturation index (ODI) varied significantly among groups (p < .001 for both) (Figure 1(b) and (c), Table 1). AHI and ODI were significantly higher in OSAHS and OS groups (p < .001 for all), but did not differ between the two groups (p = .458; p = .454), indicating a similar amount of apnea/hypopnea episodes. Similarly, mean arterial oxygen saturation (MSaO2) and lowest arterial oxygen saturation (LSaO2) were significantly lower in OSAHS (p = .002; p < .001) and OS (p < .001 for both) groups (Figure 1(d) and (e), Table 1). However, MSaO2 was significantly lower, and the percentage of time spent in sleep below 90% oxygen saturation (TS90%) was significantly higher in OS group compared with OSAHS group (p = .004; p = .003) (Figure 1(f), Table 1), indicating a more severe and longer nocturnal hypoxia in OS. In contrast, COPD group did not suffer from significant hypopnea events or nocturnal hypoxia, with no elevation of AHI and ODI (p = .819; p = .411) or decrease of LSaO2 or MSaO2 (p = .056; p = .105) compared with controls (Figure 1).
As expected, AHI was positively correlated with ESS in OSAHS (R = 0.626, p < .001) and OS (R = 0.428, p = .007) groups, showing that the frequencies of nocturnal hypoxia and apnea were associated with daytime sleepiness.
Pulmonary function
As expected, lung functions such as forced vital capacity (FVC), forced expiratory volume in 1s (FEV1), FEV1/FVC, and peak expiratory flow (PEF) were significantly lower in COPD compared with controls (p = .022; p < .001; p < .001; p < .001) (Table 1). Similarly, the OS group also presented a significant decrease in these parameters compared with controls (p = .026; p < .001; p < .001; p < .001) (Table 1) as well as OSAHS group (p < .001 for all). In addition, the changes in lung functions in OS group were similar to those observed in COPD group (p = .854; p = .915; p = .756; p = .881), and did not differ between OSAHS and controls (p = .410; p = .604; p = .542; p = .728).
Correlation between apnea/hypopnea and cognitive impairment
In OS group, ODI was negatively correlated with adjusted MoCA score (R = −0.625, p < .001), indicating that when OSAHS overlapped with COPD, the frequency of nocturnal hypoxemia was still the leading factor for cognitive impairment (Table 2). In addition, LSaO2 was positively correlated with overall adjusted MoCA score (R = 0.462, p = .003) and visuospatial/executive domain (R = 0.378, p = .019) in OS group, while was positively correlated with attention (R = 0.369, p = .006) in OSAHS group (Table 3), suggesting that the lowest oxygen saturation may be associated with cognitive impairment in specific domains, and the effects may vary among different disease types.
Table 2.
Association of sleep parameters, lung functions and serum biomarkers with cognition in OS groups.
| MoCA score | Visuospatial/executive | Naming | Attention | Language | Abstraction | Delayed recall | Orientation | ||
|---|---|---|---|---|---|---|---|---|---|
| Sleep parameters | ESS | R = −0.114 p = .497 |
R = 0.031 p = .853 |
R = −0.014 p = .936 |
R = −0.016 p = .925 |
R = −0.171 p = .305 |
R = −0.237 p = .151 |
R = −0.032 p = .850 |
R = −0.203 p = .221 |
| Severity of snoring | R = −0.248 p = .134 |
R = −0.011 p = .948 |
R = 0.164 p = .324 |
R = −0.117 p = .485 |
R = 0.01 p = .954 |
R = −0.334 p = .040* |
R = −0.037 p = .827 |
R = −0.32 p = .050 |
|
| AHI | R = −0.419 p = .009** |
R = −0.32 p = .050 |
R = 0.156 p = .350 |
R = −0.074 p = .657 |
R = 0.076 p = .652 |
R = −0.193 p = .246 |
R = −0.401 p = .013* |
R = −0.237 p = .152 |
|
| ODI | R = −0.625 p < .001*** |
R = −0.446 p = .008** |
R = −0.037 p = .835 |
R = −0.193 p = .274 |
R = 0.009 p = .960 |
R = −0.336 p = .052 |
R = −0.426 p = .012* |
R = −0.408 p = .017* |
|
| TS90% | R = −0.284 p = .084 |
R = −0.268 p = .104 |
R = −0.298 p = .069 |
R = −0.099 p = .552 |
R = −0.111 p = .506 |
R = −0.013 p = .937 |
R = −0.162 p = .331 |
R = −0.081 p = .627 |
|
| MSaO2 | R = 0.326 p = .046* |
R = 0.323 p = .048* |
R = 0.314 p = .055 |
R = 0.217 p = .191 |
R = −0.081 p = .630 |
R = 0.05 p = .764 |
R = 0.201 p = .226 |
R = −0.002 p = .990 |
|
| LSaO2 | R = 0.462 p = .003** |
R = 0.378 p = .019* |
R = 0.339 p = .037* |
R = 0.071 p = .674 |
R = 0.268 p = .104 |
R = 0.261 p = .113 |
R = 0.208 p = .209 |
R = 0.267 p = .106 |
|
| Lung functions | %FEV1 | R = −0.10 p = .549 |
R = −0.131 p = .433 |
R = 0.182 p = .273 |
R = −0.245 p = .139 |
R = 0.263 p = .111 |
R = 0.118 p = .479 |
R = −0.335 p = .040* |
R = −0.108 p = .519 |
| FEV1 | R = 0.09 p = .593 |
R = 0.055 p = .743 |
R = 0.316 p = .053 |
R = −0.135 p = .420 |
R = 0.374 p = .021* |
R = 0.223 p = .178 |
R = −0.101 p = .545 |
R = −0.112 p = .504 |
|
| %FVC | R = −0.011 p = .950 |
R = −0.003 p = .985 |
R = 0.20 p = .228 |
R = −0.197 p = .235 |
R = 0.263 p = .111 |
R = 0.234 p = .158 |
R = −0.242 p = .144 |
R = −0.063 p = .706 |
|
| FVC | R = 0.157 p = .348 |
R = 0.125 p = .453 |
R = 0.378 p = .019* |
R = −0.103 p = .539 |
R = 0.356 p = .028* |
R = 0.329 p = .044* |
R = −0.056 p = .737 |
R = −0.001 p = .994 |
|
| FEV1/FVC | R = −0.085 p = .610 |
R = −0.19 p = .252 |
R = 0.156 p = .350 |
R = −0.069 p = .679 |
R = 0.138 p = .409 |
R = −0.032 p = .848 |
R = −0.204 p = .220 |
R = −0.185 p = .267 |
|
| PEF | R = 0.159 p = .340 |
R = 0.083 p = .619 |
R = 0.405 p = .012* |
R = −0.007 p = .967 |
R = 0.191 p = .250 |
R = 0.247 p = .135 |
R = −0.076 p = .649 |
R = −0.063 p = .706 |
|
| PaO2 | R = −0.003 p = .985 |
R = −0.029 p = .865 |
R = 0.196 p = .239 |
R = −0.035 p = .832 |
R = 0.325 p = .047* |
R = 0.088 p = .601 |
R = −0.15 p = .367 |
R = −0.114 p = .495 |
|
| PaCO2 | R = −0.014 p = .931 |
R = 0.079 p = .637 |
R = −0.04 p = .811 |
R = 0.116 p = .489 |
R = 0.004 p = .979 |
R = 0.134 p = .421 |
R = 0.003 p = .986 |
R = −0.02 p = .906 |
|
| Serum biomarkers | HIF-1α | R = 0.024 p = .888 |
R = 0.109 p = .516 |
R = −0.12 p = .472 |
R = 0.112 p = .503 |
R = −0.129 p = .440 |
R = −0.199 p = .230 |
R = 0.069 p = .681 |
R = −0.178 p = .286 |
| BDNF | R = 0.099 p = .553 |
R = 0.14 p = .401 |
R = −0.147 p = .379 |
R = −0.032 p = .848 |
R = −0.049 p = .770 |
R = −0.109 p = .513 |
R = 0.182 p = .274 |
R = 0.158 p = .344 |
|
Abbreviations: BMI: body mass index; FEV1: forced expiratory volume in 1 s; FVC: forced vital capacity; PEF: peak expiratory flow; PaO2: partial pressure of oxygen; PaCO2: partial pressure of carbon dioxide; MoCA: Montreal cognitive assessment; ESS: epworth sleepiness scale; AHI: apnea hypopnea index; ODI: oxygen desaturation index; TS90%: percent nighttime with oxygen saturation less than 90%; MSaO2: mean arterial oxygen saturation (%); LSaO2: lowest arterial oxygen saturation (%); HIF-1α: hypoxia-inducible factor-1α; BDNF: brain-derived neurotrophic factor.
*, p < .05; **, p < .01, ***, p < .001.
Table 3.
Association of sleep parameters, lung functions and serum biomarkers with cognition in OSAHS group.
| MoCA score | Visuospatial/executive | Naming | Attention | Language | Abstraction | Delayed recall | Orientation | ||
|---|---|---|---|---|---|---|---|---|---|
| Sleep parameters | ESS | R = −0.384 p = .004** |
R = −0.068 p = .626 |
R = −0.005 p = .970 |
R = −0.24 p = .081 |
R = 0.141 p = .311 |
R = −0.175 p = .205 |
R = −0.193 p = .163 |
R = −0.114 p = .413 |
| Severity of snoring | R = −0.39 p = .004** |
R = −0.006 p = .964 |
R = −0.058 p = .679 |
R = −0.235 p = .087 |
R = −0.085 p = .542 |
R = −0.201 p = .145 |
R = −0.176 p = .203 |
R = −0.157 p = .258 |
|
| AHI | R = −0.321 p = .018* |
R = −0.167 p = .228 |
R = 0.096 p = .490 |
R = −0.225 p = .102 |
R = 0.105 p = .448 |
R = −0.146 p = .293 |
R = −0.181 p = .191 |
R = −0.073 p = .598 |
|
| ODI | R = −0.335 p = .017* |
R = −0.29 p = .041* |
R = 0.079 p = .587 |
R = −0.27 p = .058 |
R = 0.223 p = .119 |
R = 0.002 p = .990 |
R = −0.085 p = .556 |
R = −0.235 p = .101 |
|
| TS90% | R = −0.382 p = .004** |
R = −0.157 p = .257 |
R = −0.086 p = .538 |
R = −0.407 p = .002** |
R = −0.095 p = .493 |
R = −0.153 p = .270 |
R = −0.386 p = .004** |
R = −0.03 p = .827 |
|
| MSaO2 | R = 0.219 p = .111 |
R = 0.205 p = .137 |
R = −0.101 p = .469 |
R = 0.124 p = .373 |
R = 0.007 p = .961 |
R = 0.02 p = .883 |
R = 0.126 p = .364 |
R = 0.034 p = .808 |
|
| LSaO2 | R = 0.326 p = .016* |
R = 0.113 p = .414 |
R = 0.078 p = .575 |
R = 0.369 p = .006** |
R = −0.002 p = .990 |
R = 0.178 p = .199 |
R = 0.32 p = .018* |
R = −0.088 p = .529 |
|
| Lung functions | %FEV1 | R = −0.005 p = .972 |
R = −0.014 p = .922 |
R = −0.003 p = .985 |
R = 0.059 p = .670 |
R = 0.00 p = 1.000 |
R = 0.059 p = .672 |
R = 0.111 p = .424 |
R = −0.097 p = .484 |
| FEV1 | R = 0.368 p = .006** |
R = 0.234 p = .088 |
R = 0.148 p = .286 |
R = 0.348 p = .010* |
R = −0.124 p = .373 |
R = 0.205 p = .138 |
R = 0.396 p = .003** |
R = −0.065 p = .639 |
|
| %FVC | R = 0.025 p = .855 |
R = −0.06 p = .668 |
R = 0.008 p = .955 |
R = 0.047 p = .738 |
R = 0.027 p = .848 |
R = 0.067 p = .633 |
R = 0.148 p = .285 |
R = −0.10 p = .471 |
|
| FVC | R = 0.356 p = .008** |
R = 0.21 p = .127 |
R = 0.161 p = .245 |
R = 0.307 p = .024* |
R = −0.114 p = .413 |
R = 0.187 p = .175 |
R = 0.424 p = .001** |
R = −0.098 p = .482 |
|
| FEV1/FVC | R = −0.045 p = .749 |
R = 0.073 p = .599 |
R = −0.223 p = .105 |
R = 0.115 p = .410 |
R = −0.119 p = .392 |
R = −0.069 p = .620 |
R = −0.179 p = .196 |
R = 0.155 p = .263 |
|
| PEF | R = 0.418 p = .002** |
R = 0.281 p = .040* |
R = 0.195 p = .159 |
R = 0.365 p = .007** |
R = −0.114 p = .413 |
R = 0.19 p = .168 |
R = 0.435 p = .001** |
R = −0.04 p = .773 |
|
| PaO2 | R = 0.254 p = .064 |
R = 0.323 p = .017* |
R = 0.042 p = .766 |
R = 0.081 p = .562 |
R = −0.157 p = .256 |
R = 0.049 p = .722 |
R = 0.086 p = .538 |
R = −0.113 p = .416 |
|
| PaCO2 | R = 0.103 p = .458 |
R = 0.043 p = .760 |
R = 0.099 p = .478 |
R = 0.256 p = .062 |
R = −0.038 p = .782 |
R = 0.073 p = .598 |
R = −0.019 p = .892 |
R = 0.154 p = .267 |
|
| Serum biomarkers | HIF-1α | R = −0.199 p = .150 |
R = −0.353 p = .009** |
R = −0.086 p = .538 |
R = 0.061 p = .659 |
R = 0.112 p = .420 |
R = −0.187 p = .175 |
R = −0.063 p = .649 |
R = 0.101 p = .470 |
| BDNF | R = −0.253 p = .065 |
R = −0.389 p = .004** |
R = 0.008 p = .955 |
R = −0.081 p = .559 |
R = 0.067 p = .631 |
R = −0.142 p = .307 |
R = −0.041 p = .768 |
R = −0.036 p = .793 |
|
Abbreviations: BMI: body mass index; FEV1: forced expiratory volume in 1 s; FVC: forced vital capacity; PEF: peak expiratory flow; PaO2: partial pressure of oxygen; PaCO2: partial pressure of carbon dioxide; MoCA: Montreal cognitive assessment; ESS: epworth sleepiness scale; AHI: apnea hypopnea index; ODI: oxygen desaturation index; TS90%: percent nighttime with oxygen saturation less than 90%; MSaO2: mean arterial oxygen saturation (%); LSaO2: lowest arterial oxygen saturation (%); HIF-1α: hypoxia-inducible factor-1α; BDNF: brain-derived neurotrophic factor.
*, p < .05; **, p < .01, ***, p < .001.
The severity of snoring was associated with AHI (R = 0.617, p < .001; R = 0.542, p < .001) in both OS and OSAHS groups) (Tables 2 and 3), but not in COPD or control group. These results suggest that snoring may present closer relationships with nocturnal apnea and hypoxemia under conditions where intermittent hypoxemia becomes more obvious.
Correlation between pulmonary function and cognitive impairment
In OS and COPD group, neither FVC nor FEV1 was correlated with overall cognitive scores (p > .05). (Table 2), indicating that decrease of pulmonary functions alone was not sufficient to affect cognitive functions in these diseases. Interestingly, in OSAHS group, FVC, FEV1 and PEF were all positively correlated with adjusted MoCA score (R = 0.356, p = .008; R = 0.368, p = .006; R = 0.418, p = .002) as well as cognitive domains including attention (R = 0.307, p = .024; R = 0.348, p = .010; R = 0.365, p = .007), and delayed recall (R = 0.424, p = .001; R = 0.396, p = .003; R = 0.435, p = .001 (Table 3). These results showed that when presenting within normal ranges, pulmonary functions were more closely associated with overall cognition as well as individual cognitive domains.
Serum levels of HIF-1α and BDNF
The serum level of HIF-1α was significantly higher in OS compared with controls (p = .010) (Table 1), OSAHS group (p = .026) and COPD group (p = .037) (Figure 2(a)), suggesting that OS patients may suffer from severer hypoxemia. Though the serum level of HIF-1α was not correlated with adjusted MoCA score (R = −0.199, p = .150), it was negatively correlated with visuospatial/executive function (R = −0.353, p = .009) in OSAHS group (Table 3), indicating an association of increased hypoxemia with cognitive impairment in specific domains within the group.
Figure 2.
Comparison of biomarkers across groups. (a) HIF-1α (pg/mL); (b) BDNF (ng/mL) of each group was indicated as mean ± SD. *, p < .05.
The serum level of BDNF did not vary among groups (p = .648) (Figure 2(b), Table 1). In OSAHS group, the serum level of BDNF was negatively correlated with visuospatial/executive function (R = −0.389, p = .004) (Table 3), possibly as a reaction to cognitive impairment of the domain.
The serum level of HIF-1α was positively correlated with BDNF in OS (R = 0.496, p = .002), OSAHS (R = 0.650, p < .001), COPD (R = 0.839, p < .001) and control groups (R = 0.594, p < .001), suggesting that the neurotrophic factor may increase in response to hypoxemia.
Factors associated with cognitive impairment
Logistic regression analysis revealed that in the total cohort, ESS, ODI and PEF were all independent factors for cognitive impairment (p < .001, OR = 1.518; p < .001, OR = 1.087; p < .001, OR = 0.593), indicating that both nocturnal hypoxemia and airflow limitation contribute to decline of cognition. Subgroup analysis revealed that in OSAHS group, ESS and PEF were both independent factors for cognitive impairment (p = .003, OR = 1.756; p = .001, OR = 0.363). In OS group, ODI was identified as an independent factor associated with cognitive impairment (p = .022, OR = 1.388), further supported that ODI was a leading factor for cognitive impairment in OS. While in COPD, ESS was the only factor associated with cognitive impairment (p = .034, OR = 1.923).
Diagnostic value of HIF-1α and BDNF in cognitive impairment in OSAHS and OS
Analysis of receiver operating characteristic (ROC) curve revealed that in the total cohort, HIF-1α presented predictive value for cognitive impairment, with an area under curve (AUC) of 0.599 (95% CI: 0.510∼0.688, p = .033) (Figure 3). A cutoff value as 282.14 pg/mL presented a sensitivity of 76.7% and a specificity of 46.4%. In contrast, BDNF did not show a significant predictive value for cognitive impairment in the total cohort.
Figure 3.
ROC curve for predicting cognitive impairment with two biomarkers in total cohort. Curve in blue indicates HIF-1α, curve in green indicates BDNF, and curve in red is reference.
Discussion
The study revealed that cognitive impairment was present in OSAHS, COPD and OS, with OS group presenting the highest prevalence of cognitive impairment and lowest adjusted MoCA score, which was consistent with previous findings 14 and added valuable information on the difference in cognitive impairment among OS, OSAHS and COPD. One possible explanation is that in addition to the intermittent hypoxia presented in OSAHS, airflow limitation presented in COPD may lead to continuous hypoxia. In addition, the present study also detailed changes in specific cognitive domains in OS, suggesting a more generalized damage. A broad range of cognitive deficits have been previously observed in both OSAHS and COPD. 15 With respect to the impairments in specific cognitive domains, the executive function-processing speed domain, but not memory, language and visuospatial skills, was found to be affected in OS patients compared with COPD-alone individuals, which was likely to be associated with carotid artery stiffness. 16 Our study revealed that visuospatial/executive function, attention and delayed recall were affected in both OSAHS and OS, while only abstraction was affected in COPD. This may be due to the heterogeneity, disease duration, severity, and interventions of COPD, which requires further investigation.
The increase of AHI and ODI are key features in OSAHS. 17 The present study revealed a significant increase in AHI and ODI among individuals with OSAHS and OS. Though no difference has been observed in these parameters between the two groups, MSaO2 was significantly lower while TS90% was significantly higher in OS compared with OSAHS, which was similarly reported by Bednarek et al., 18 indicating that the two parameters may better reflect degree and duration of oxygen desaturation than LSaO2.
In OS group, ODI was negatively correlated with adjusted MoCA score, and logistic regression analysis also showed that ODI was an independent factor for cognitive impairment in OS, indicating that nocturnal hypoxemia is a leading factor driving cognitive impairment when OSAHS and COPD are overlapped. Previous studies showed that the MoCA score was significantly lower and the risk of dementia was dramatically higher in OS patients compared with COPD-only patients.14,19 Additionally, the level of dementia in OS patients was significantly correlated with the severity of OSA. 19 Therefore, it is of great importance to screen cognitive impairment in OS patients at early stage by using appropriate indicators.
Though airflow limitation was less severe in OSAHS group compared with OS and COPD groups, FVC, FEV1 and PEF were positively correlated with scores of adjusted MoCA and multiple domains in OSAHS group. In contrast, pulmonary functions were not associated with overall cognitive performance in COPD, suggesting that pulmonary functions were more crucial in accelerating cognitive impairment where nocturnal hypoxemia is obvious but pulmonary dysfunction is subtle, and that attention should be paid to assess pulmonary functions at baseline for OSAHS patients. Interestingly, PEF has been identified as a contributor in OSAHS group, suggesting that airflow limitation and respiratory muscle weakness may lead to vulnerability for cognitive function in OSAHS patients, and airflow limitation may act as an add-on in OS patients that are already vulnerable to cognitive impairment due to nocturnal hypoxemia. Previous studies on the correlation between pulmonary and cognitive functions among individuals with OS or COPD-alone have yielded conflicting results. For instance, MMSE was observed to be inversely associated with the severity of airflow obstruction in one study, 20 whereas no association was detected between lung function parameters and risk of dementia in COPD patients with or without OSA. 19 Therefore, additional evidence is required to confirm the association between lung injury and cognitive decline.
Animal study has previously shown that HIF-1α participated in cognitive impairment induced by intermittent hypoxia.6,21,22 Human studies further proved that serum level of HIF-1α was elevated in OSAHS patients, 23 especially in severe OSAHS patients.24,25 The present study revealed elevation of HIF-1α in OS group compared with control group, indicating more robust hypoxia within the group. In addition, serum level of HIF-1α was correlated with visualspatial/executive domain in OSAHS, showing a potential contribution of hypoxemia to impairment of specific cognitive domain. ROC analysis revealed diagnostic value of HIF-1α for cognitive impairment in the total population, with relatively high sensitivity and low specificity, suggesting its potential role as a screening marker for differentiation of subtle cognitive impairment in general population. In contrast, BDNF neither showed any significant change among groups nor presented diagnostic value for cognitive impairment. Whether BDNF would serve as a marker for specific cognitive domains require further investigation.
The present study had several limitations. Firstly, due to the use of portable devices, it was not possible to collect full range of sleep-related data, which may underestimate the severity of the condition. Secondly, sex ratio, which is a risk factor for cognitive impairment, has not been matched among groups, which may potentially affect evaluation of group differences. Thirdly, in addition to weight gain and cigarette smoking, other medical factors such as certain medications for COPD and higher diaphragmatic position may also predispose to OSAHS. 1 Cohorts matched for known risk factors and utilization of polysomnography are required to evaluate the roles hypoxemia and airflow limitation play in these diseases, as well as the predictive value for HIF-1α and BDNF for such diseases in future studies.
Conclusion
This study revealed significant cognitive impairment in OS, affecting multiple cognitive domains. This adds novel knowledge to the fields and raises attention to the disease. The frequency of intermittent hypoxemia plays a leading role in the pathogenesis of cognitive impairment in OS, while airflow limitation and respiratory muscle weakness play a more crucial role driving cognitive impairment in OSAHS, suggesting the necessasity of screening airflow limitation in OSAHS patients. Our results suggested that clinicians managing these patients should be aware of their increased risk of cognitive impairment and that family doctor should screen COPD patients for OSAHS and vice versa to improve their prognosis and quality of life.
Appendix.
Abbreviations
- OHAHS
Sleep apnea hypopnea syndrome
- COPD
Chronic obstructive pulmonary disease
- OS
Overlap syndrome
- ESS
Epworth sleeping scale
- MoCA
Montreal cognitive assessment
- HIF-1α
Hypoxia-inducible factor-1α
- BDNF
Brain-derived neurotrophic factor
- ELISA
Enzyme-linked immunosorbent assay
- ODI
Oxygen desaturation index
- AHI
Apnea hypopnea index
- FVC
Forced vital capacity
- FEV1
Forced expiratory volume in 1 s
- PEF
Peak expiratory flow
- MCI
Mild cognitive impairment
- MoCA-BJ
Beijing version of Montreal cognitive assessment
- ROS
Reactive oxygen species
- CNS
Central nervous system
- CPAP
Continuous positive airway pressure
- TS90%
Total time recorded below 90% oxygen saturation
- LSaO2
Lowest arterial oxygen saturation
- MSaO2
Mean arterial oxygen saturation
- LAT
Longest apnea time
- %FVC
Percent predicted value of FVC
- %FEV1
Percent predicted value of FEV1
- FEV1/FVC
Percent of forced vital capacity exhaled in the first second
- ROC
Receiver operating characteristic
- AUC
Area under the curve
- AECOPD
Acute exacerbation of COPD
Footnotes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding: The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Science and Technology Innovation Project of Shanghai Putuo District Health System (ptkwws201917).
ORCID iD
References
- 1.Ioachimescu OC, McNicholas WT. Chronic obstructive pulmonary disease-obstructive sleep apnea overlap: more than a casual acquaintance. Am J Respir Crit Care Med 2022; 206: 139–141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Villeneuve S, Pepin V, Rahayel S, et al. Mild cognitive impairment in moderate to severe COPD: a preliminary study. Chest 2012; 142(6): 1516–1523. [DOI] [PubMed] [Google Scholar]
- 3.Campman CA, Sitskoorn MM. Better care for patients with COPD and cognitive impairment. Lancet Respir Med 2013; 1(7): 504–506. [DOI] [PubMed] [Google Scholar]
- 4.Mu L, Peng L, Zhang Z, et al. Memory and executive screening for the detection of cognitive impairment in obstructive sleep apnea. Am J Med Sci 2017; 354(4): 399–407. [DOI] [PubMed] [Google Scholar]
- 5.Iyer NV, Kotch LE, Agani F, et al. Cellular and developmental control of O2 homeostasis by hypoxia-inducible factor 1 alpha. Genes Dev 1998; 12(2): 149–162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Arias-Cavieres A, Khuu MA, Nwakudu CU, et al. A HIF1a-dependent pro-oxidant state disrupts synaptic plasticity and impairs spatial memory in response to intermittent hypoxia. eNeuro. 2020; 7(3). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Althaus J, Bernaudin M, Petit E, et al. Expression of the gene encoding the pro-apoptotic BNIP3 protein and stimulation of hypoxia-inducible factor-1alpha (HIF-1alpha) protein following focal cerebral ischemia in rats. Neurochem Int 2006; 48(8): 687–695. [DOI] [PubMed] [Google Scholar]
- 8.Belaidi E, Morand J, Gras E, et al. Targeting the ROS-HIF-1-endothelin axis as a therapeutic approach for the treatment of obstructive sleep apnea-related cardiovascular complications. Pharmacol Ther 2016; 168: 1–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Flores KR, Viccaro F, Aquilini M, et al. Protective role of brain derived neurotrophic factor (BDNF) in obstructive sleep apnea syndrome (OSAS) patients. PLoS One 2020; 15(1): e0227834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kapur VK, Auckley DH, Chowdhuri S, et al. Clinical practice guideline for diagnostic testing for adult obstructive sleep apnea: an American academy of sleep medicine clinical practice guideline. J Clin Sleep Med 2017; 13(3): 479–504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Vogelmeier CF, Criner GJ, Martinez FJ, et al. Global strategy for the diagnosis, management, and prevention of chronic obstructive lung disease 2017 report. GOLD executive summary. Am J Respir Crit Care Med 2017; 195(5): 557–582. [DOI] [PubMed] [Google Scholar]
- 12.Berry RB, Budhiraja R, Gottlieb DJ, American Academy of Sleep Medicine , et al. Rules for scoring respiratory events in sleep: update of the 2007 AASM manual for the scoring of sleep and associated events. Deliberations of the sleep apnea definitions task force of the American Academy of sleep medicine. J Clin Sleep Med 2012; 8(5): 597–619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Westreich R, Gozlan-Talmor A, Geva-Robinson S, et al. The presence of snoring as well as its intensity is underreported by women. J Clin Sleep Med 2019; 15(3): 471–476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Wang Y, Li B, Li P, et al. Severe obstructive sleep apnea in patients with chronic obstructive pulmonary disease is associated with an increased prevalence of mild cognitive impairment. Sleep Med 2020; 75: 522–530. [DOI] [PubMed] [Google Scholar]
- 15.Olaithe M, Bucks RS, Hillman DR, et al. Cognitive deficits in obstructive sleep apnea: insights from a meta-review and comparison with deficits observed in COPD, insomnia, and sleep deprivation. Sleep Med Rev 2018; 38: 39–49. [DOI] [PubMed] [Google Scholar]
- 16.Luehrs RE, Moreau KL, Pierce GL, et al. Cognitive performance is lower among individuals with overlap syndrome than in individuals with COPD or obstructive sleep apnea alone: association with carotid artery stiffness. J Appl Physiol (1985) 2021; 131(1): 131–141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kong D, Hu C, Zhu H. Oxygen desaturation index, lowest arterial oxygen saturation and time spent below 90% oxygen saturation as diagnostic markers for obstructive sleep apnea. Am J Transl Res 2023; 15(5): 3597–3606. [PMC free article] [PubMed] [Google Scholar]
- 18.Bednarek M, Plywaczewski R, Jonczak L, et al. There is no relationship between chronic obstructive pulmonary disease and obstructive sleep apnea syndrome: a population study. Respiration 2005; 72(2): 142–149. [DOI] [PubMed] [Google Scholar]
- 19.Zhang XL, Gao B, Han T, et al. Moderate-to-severe obstructive sleep apnea and cognitive function impairment in patients with COPD. Int J Chronic Obstr Pulm Dis 2020; 15: 1813–1822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Hu X, Wang H, Tu Y, et al. Alterations of the default mode network and cognitive impairments in patients with chronic obstructive pulmonary disease. Int J Chronic Obstr Pulm Dis 2018; 13: 519–528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Gozal D, Row BW, Gozal E, et al. Temporal aspects of spatial task performance during intermittent hypoxia in the rat: evidence for neurogenesis. Eur J Neurosci 2003; 18(8): 2335–2342. [DOI] [PubMed] [Google Scholar]
- 22.Row BW, Kheirandish L, Neville JJ, et al. Impaired spatial learning and hyperactivity in developing rats exposed to intermittent hypoxia. Pediatr Res 2002; 52(3): 449–453. [DOI] [PubMed] [Google Scholar]
- 23.Gabryelska A, Szmyd B, Szemraj J, et al. Patients with obstructive sleep apnea present with chronic upregulation of serum HIF-1α protein. J Clin Sleep Med 2020; 16(10): 1761–1768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Liu C, Wang H, Zhu C, et al. Plasma expression of HIF-1α as novel biomarker for the diagnosis of obstructive sleep apnea-hypopnea syndrome. J Clin Lab Anal 2020; 34(12): e23545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Lu D, Li N, Yao X, et al. Potential inflammatory markers in obstructive sleep apnea-hypopnea syndrome. Bosn J Basic Med Sci 2017; 17(1): 47–53. [DOI] [PMC free article] [PubMed] [Google Scholar]