Abstract
Helicobacter pylori (H. pylori) resistance is the most important risk factor for eradication failure. However, in most regions, antibiotic resistance rates of H. pylori in patients with different types of gastric mucosal lesions are still unclear. An 8-year clinical retrospective cohort study involving 2847 patients was performed. In this study, we first summarized and compared the resistance status of H. pylori in different years, ages, sexes, and gastric diseases. The resistance profiles of amoxicillin (AMX), clarithromycin (CLR), levofloxacin (LVX) and furazolidone (FR) and their changing trends in the clinic were described. Then, multiple antibiotic resistance in different gastric diseases and years were described and compared. The relationship between proton pump inhibitor (PPI) medication history and antibiotic resistance in H. pylori was also explored. Finally, an antibiotic resistance risk model was constructed for clinical resistance risk prediction. The overall resistance rates of AMX, CLR, LVX and FR in gastric diseases were 8.18%, 38.11%, 43.98%, and 13.73%, respectively. The mono resistance, double resistance, triple resistance, and quadruple resistance rates were 30.17%, 25.96%, 6.46%, and 0.63%, respectively. Compared with the period from 2014 to 2016, the rates of mono-resistance and multiple resistance all showed relatively downward trends in the past 5 years. Factors including age, sex, type of gastric lesions and recent PPI treatment history are associated with the antibiotic resistance rate of H. pylori. Atrophic gastritis is an important clinical feature of high-risk antibiotic resistance in H. pylori-infected patients. Patients with atrophic gastritis have higher risk of resistant strains infection. In this study, our data provide the association between antibiotic resistance of H. pylori and gastritis pattern, which indicate the higher risk of resistant strain infection if the patients with atrophic gastritis, PPI history and older age.
Keywords: Helicobacter pylori, Antibiotic resistance, Antimicrobial susceptibility test, Atrophic gastritis, Proton pump inhibitor
Subject terms: Clinical microbiology, Gastrointestinal diseases, Microbiota
Introduction
Helicobacter pylori (H. pylori) is a common pathogenic microorganism for digestive system diseases, infecting nearly 50% of the population worldwide1,2. H. pylori infection causes persistent damage to the gastric mucosa, leading to glandular atrophy and intestinal metaplasia in the long run3. It is very necessary to eradicate H. pylori from the stomach, as it plays an important part in the pathogenesis of some gastric diseases, such as chronic gastritis, peptic ulcers, and gastric cancer4–7.
Classic therapeutic regimens that the combination of proton pump inhibitors (PPIs) and 2 antibiotics for H. pylori eradication have been used worldwide8,9. On this basis, regimens has successively derived bismuth-containing quadruple therapies, sequential therapy, reverse sequential therapy, hybrid therapy, concomitant therapy and vonoprazan based therapeutic regimens10,11. Successful eradication treatment in these regimens mainly depends on the sensitivity of H. pylori to drugs, and its increasing resistance is the main reason for eradication failure12. So, reviewing resistance to select suitable antibiotics in therapeutic regimen is important. More importantly, the antibiotic resistance rate of H. pylori vary greatly among different populations and regions and are affected by many factors13,14. Specifically, antibiotic resistance rates of H. pylori in patients with different types of gastric mucosal lesions are still unclear. Whether the type of gastric mucosal lesion is a factor or feature associated with H. pylori resistance remains unknown.
Therefore, an 8-year clinical retrospective cohort study involving 2847 patients was performed to mainly analyze the antibiotic resistance rate of H. pylori in patients with different types of gastric mucosal lesions. In this study, we first summarized and compared the resistance status of H. pylori in different years, ages, sexes, and gastric diseases. The resistance profiles of amoxicillin (AMX), clarithromycin (CLR), levofloxacin (LVX) and furazolidone (FR) and their changing trends in the clinic were described. Then, multiple antibiotic resistance in different gastric diseases and years were described and compared. The relationship between proton pump inhibitor (PPI) medication history and antibiotic resistance in H. pylori was also explored. Finally, nomograms were constructed to predict the individual risk of antibiotic resistance.
Materials and methods
Patients and inclusion criteria
This is a clinical retrospective study, targeting clinical patients undergoing gastroscopy for H. pylori culture and antimicrobial susceptibility tests, aimed at further evaluating the relationship between types of gastric mucosal lesions and H. pylori resistance. Patients and their medical history were obtained from the First Affiliated Hospital of Ningbo University. From 2014 to 2021, a total of 2847 patients (49.21% females) with the average age of 48.04 were ultimately included in this study. The diagnosis of gastric mucosal lesion is based on endoscopic findings and biopsy pathology. The inclusion criteria in this study were as follows: (1) patients were local residents who had detailed and complete medical records; (2) patients with a history of gastroscopy and positive H. pylori culture of the gastric mucosa; (3) patients with an antimicrobial susceptibility test of H. pylori, corresponding gastroscopy and gastroscopic biopsy pathology report; (4) no antibiotic use within 3 months before gastroscopy and drug susceptibility testing. This study was approved by the Human Research Ethics Committee of the First Affiliated Hospital of Ningbo University (No. KS20227019). All clinical procedures in accordance with relevant guidelines and regulations. Informed consent was obtained from all participants. All patients’ privacy was respected and protected.
Gastroscopic biopsy and H. pylori culture
Patients in this study had same procedures and processes during past gastroscopy. Following evaluation by clinical doctors, endoscopic mucosal biopsy for pathological diagnosis was performed at the main lesion of the stomach, which collect one piece of tissue using biopsy forceps. Biopsy specimen was immersed in formalin solution for pathological examination. At the same time, two biopsy specimens for H. pylori culture were collected from the body and antrum of stomach, which were immediately stored in brain–heart infusion broth (Oxoid, Basingstoke, UK) with 5% glycerin and then transported to laboratory for culture. The biopsy method follows the “Standardized Guidelines for the Diagnosis and Treatment of Gastric Cancer” and the “Consensus on the Diagnosis and Treatment of Helicobacter pylori Infection in China”.
Antimicrobial susceptibility testing
Antimicrobial susceptibility tests were determined by the agar dilution method. Resistance breakpoints were according to the European Committee on Antibiotic Susceptibility Testing (EUCAST) guidelines.
Information traceability and grouping criteria
Medical history data were traced from the electronic medical record system of the hospital. Patients in this study were grouped according to their age, sex, gastric mucosal lesions, and PPI treatment history. The age group was divided according to the WHO standard. The diagnosis of gastric mucosal lesions is based on endoscopic findings and biopsy pathology. Patients who met the following conditions were considered to have a recent history of PPI treatment: (1) a history of taking PPIs within 3 months; (2) continuous medication for more than 1 week.
Statistical analysis
Statistical analyses in this study were performed by using Statistical Product and Service Solutions (SPSS) 19.0 software. Chi-square (χ2) tests were used to evaluate the differences in antibiotic resistance rates among different groups. Minimal inhibitory concentration (MIC) values were assessed with Student’s t test or one-way analysis of variance (ANOVA). P < 0.05 was considered statistically significant.
Nomogram prediction models and figures were analyzed and drawn by the R software package “rms[6.4.0]”. The C-index was used to validate the predictive performance of the nomograms.
Results
Resistance rates of H. pylori strains between different years
As shown in Table 1, the overall resistance rates of AMX, CLR, LVX, and FR in gastric diseases were 8.18%, 38.11%, 43.98%, and 13.73%, respectively. From the perspective of time, the resistance rate of AMX in the last 5 years was much higher than that in 2014–2016 (P = 0.003), whereas LVX and FR showed downward trends (P < 0.001 and P < 0.001, respectively). Moreover, 36.78% of H. pylori strains were susceptible to all four tested antibiotics, and 33.05% of strains showed resistance to more than one antibiotic. There were 30.17% mono resistance, 25.96% double resistance, 6.46% triple resistance, and 0.63% quadruple resistance (Table 2). Compared with the past, multiple resistance rates in the past five years had a relatively downward trend. The no resistance strain increased from 24.06 to 41.31%, while the double resistance and triple resistance strain decreased from 32.89 and 8.96% to 23.49% and 5.57%, respectively.
Table 1.
Resistance rates of H. pylori strains between different years.
| Year | No. of patients | Antibiotics | MIC( ± s) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| AMX | CLR | LVX | FR | AMX | CLR | LVX | FR | ||
| 2014–2016 | 748 | 42 (5.61%) | 287 (38.37%) | 410 (54.81%) | 230 (30.75%) | 13.333 ± 4.647 | 14.034 ± 4.410 | 12.324 ± 10.289 | 8.265 ± 6.891 |
| 2017–2021 | 2099 | 191 (9.10%) | 798 (38.02%) | 842 (40.11%) | 161 (7.67%) | 6.658 ± 15.481 | 13.331 ± 12.021 | 12.295 ± 13.347 | 12.031 ± 12.940 |
| Total | 2847 | 233 (8.18%) | 1085 (38.11%) | 1252 (43.98%) | 391 (13.73%) | 7.862 ± 14.378 | 13.517 ± 10.558 | 12.305 ± 12.425 | 9.816 ± 10.001 |
| P | 0.003 | 0.865 | < 0.001 | < 0.001 | 0.006 | 0.333 | 0.969 | 0.001 | |
Table 2.
Multiple resistance rates between different years.
| Patterns | 2014–2016 (n = 748) | 2017–2021 (n = 2099) | Total | P |
|---|---|---|---|---|
| No resistance | 180 (24.06%) | 867 (41.31%) | 1047 (36.78%) | < 0.001 |
| Mono resistance | 248 (33.16%) | 611 (29.11%) | 859 (30.17%) | 0.495 |
| Double resistance | 246 (32.89%) | 493 (23.49%) | 739 (25.96%) | < 0.001 |
| Triple resistance | 67 (8.96%) | 117 (5.57%) | 184 (6.46%) | 0.001 |
| Quadruple resistance | 7 (0.93%) | 11 (0.52%) | 18 (0.63%) | 0.222 |
Resistance rates of H. pylori strains in patients with different gastric diseases
There were significant differences in the antibiotic resistance rates of AMX (P = 0.02), CLR (P = 0.011), LVX (P < 0.001) and FR (P < 0.001) among different gastric mucosal lesions (Table 3). Overall, the rate of antibiotic resistance in patients with atrophic gastritis was generally higher than that in other groups, which indicate patients with atrophic gastritis have higher risk of resistant strains infection (Table 3). In terms of double antibiotic resistance patterns, CLR and LVX were the most common combination (17.84%) in all gastric mucosal lesions, whereas the combination of AMX and FR (0.18%) had the lowest drug resistance rate (Table 4).
Table 3.
Resistance rates of H. pylori strains in different gastric diseases.
| Gastric diseases | No. of patients | Antibiotics | MIC ( ± s) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| AMX | CLR | LVX | FR | AMX | CLR | LVX | FR | ||
| Superficial gastritis | 558 | 47 (8.42%) | 203 (36.38%) | 179 (32.08%) | 46 (8.24%) | 8.516 ± 14.313 | 13.542 ± 10.799 | 12.413 ± 13.698 | 13.739 ± 16.014 |
| Erosive gastritis | 1166 | 90 (7.72%) | 482 (41.34%) | 549 (47.08%) | 163 (13.98%) | 8.183 ± 15.430 | 13.587 ± 10.423 | 12.172 ± 12.247 | 10.141 ± 9.967 |
| Peptic ulcer | 560 | 37 (6.61%) | 183 (32.68%) | 241 (43.04%) | 72 (12.86%) | 9.980 ± 17.321 | 13.487 ± 10.893 | 11.888 ± 12.370 | 8.681 ± 6.974 |
| Atrophic gastritis | 510 | 49 (9.61%) | 198 (38.82%) | 263 (51.57%) | 103 (20.20%) | 5.495 ± 10.881 | 13.152 ± 10.220 | 12.563 ± 11.405 | 7.495 ± 5.392 |
| Intraepithelial neoplasia | 53 | 10 (18.87%) | 19 (35.85%) | 20 (37.74%) | 7 (13.21%) | 5.650 ± 6.169 | 15.579 ± 12.357 | 16.600 ± 18.182 | 22.286 ± 20.377 |
| P | 0.02 | 0.011 | < 0.001 | < 0.001 | 0.646 | 0.912 | 0.584 | < 0.001 | |
Table 4.
Patterns of multiple antibiotic resistance in different gastric diseases.
| Antibiotics | Superficial gastritis (n = 558) | Erosive gastritis (n = 1166) | Peptic ulcer (n = 560) |
Atrophic gastritis (n = 510) | Intraepithelial neoplasia (n = 53) | Total (n = 2847) |
|---|---|---|---|---|---|---|
| AMX + CLR | 7 (1.25%) | 17 (1.46%) | 6 (1.07%) | 6 (1.18%) | 2 (3.77%) | 38 (1.33%) |
| AMX + LVX | 10 (1.79%) | 14 (1.20%) | 6 (1.07%) | 5 (0.98%) | 0 (0.00%) | 35 (1.23%) |
| AMX + FR | 3 (0.54%) | 2 (0.17%) | 0 (0.00%) | 0 (0.00%) | 0 (0.00%) | 5 (0.18%) |
| CLR + LVX | 77 (13.80%) | 250 (21.44%) | 86 (15.36%) | 88 (17.25%) | 7 (13.21%) | 508 (17.84%) |
| CLR + FR | 5 (0.90%) | 28 (2.40%) | 2 (0.36%) | 9 (1.76%) | 0 (0.00%) | 44 (1.55%) |
| LVX + FR | 11 (1.97%) | 27 (2.32%) | 25 (4.46%) | 46 (9.02%) | 0 (0.00%) | 109 (3.83%) |
| AMX + CLR + LVX | 11 (1.97%) | 28 (2.40%) | 9 (1.61%) | 21 (4.12%) | 2 (3.77%) | 71 (2.49%) |
| AMX + CLR + FR | 0 (0.00%) | 4 (0.34%) | 2 (0.36%) | 1 (0.20%) | 3 (5.66%) | 10 (0.35%) |
| AMX + LVX + FR | 2 (0.36%) | 6 (0.51%) | 6 (1.07%) | 3 (0.59%) | 0 (0.00%) | 17 (0.60%) |
| CLR + LVX + FR | 8 (1.43%) | 25 (2.14%) | 20 (3.57%) | 32 (6.27%) | 1 (1.89%) | 86 (3.02%) |
| AMX + CLR + LVX + FR | 4 (0.72%) | 5 (0.43%) | 2 (0.36%) | 6 (1.18%) | 1 (1.89%) | 18 (0.63%) |
Resistance rates between different age groups and gender groups in different gastric diseases
We evaluated the effect of age and sex on the H. pylori strain resistance rate in different gastric diseases. LVX and AMX resistance rates were significantly different among different age groups (Table 5). After further subdivision of the data, we found that the LVX resistance rate in patients with superficial gastritis, erosive gastritis, peptic ulcer and atrophic gastritis was affected by age (P < 0.05), showing a high degree of consistency in different gastric diseases (Table 5). However, the resistance rates of AMX, CLR and FR only varied in some gastric mucosal lesions. Similar to the results in different age groups, CLR and LVX resistance rates in female group were significantly higher than those in male group (Table 6).
Table 5.
Resistance rates of H. pylori strains between different age groups in different gastric diseases.
| Antibiotics | Diseases | Age | P | MIC ( ± s) | P | ||||
|---|---|---|---|---|---|---|---|---|---|
| < 45 | 45–59 | ≥ 60 | < 45 | 45–59 | ≥ 60 | ||||
| AMX | Superficial gastritis | 18/270 (6.67%) | 17/206 (8.25%) | 12/82 (14.63%) | 0.075 | 5.306 ± 8.365 | 13.338 ± 20.986 | 6.500 ± 7.125 | 0.218 |
| Erosive gastritis | 41/481 (8.52%) | 32/480 (6.67%) | 17/205 (8.29%) | 0.528 | 8.787 ± 17.435 | 6.992 ± 12.787 | 8.971 ± 15.591 | 0.864 | |
| Peptic ulcer | 9/236 (3.81%) | 12/216 (5.56%) | 16/108 (14.81%) | 0.001 | 5.917 ± 6.457 | 15.667 ± 23.513 | 8.000 ± 15.888 | 0.379 | |
| Atrophic gastritis | 5/97 (5.15%) | 28/238 (11.76%) | 16/175 (9.14%) | 0.171 | 10.000 ± 6.000 | 6.938 ± 13.711 | 1.563 ± 1.940 | 0.181 | |
| Intraepithelial neoplasia | 0/9 (0%) | 4/17 (23.53%) | 6/27 (22.22%) | 0.282 | / | 8.375 ± 8.807 | 3.833 ± 3.474 | 0.279 | |
| Total | 73/1093 (6.68%) | 93/1157 (8.04%) | 67/597 (11.22%) | 0.005 | 7.658 ± 13.961 | 9.315 ± 16.352 | 6.067 ± 11.619 | 0.368 | |
| CLR | Superficial gastritis | 84/270 (31.11%) | 90/206 (43.69%) | 29/82 (35.37%) | 0.018 | 11.810 ± 10.001 | 14.456 ± 11.207 | 15.724 ± 11.361 | 0.136 |
| Erosive gastritis | 186/481 (38.67%) | 209/480 (43.54%) | 87/205 (42.44%) | 0.290 | 12.844 ± 10.334 | 14.234 ± 10.767 | 13.621 ± 9.681 | 0.417 | |
| Peptic ulcer | 70/236 (29.66%) | 75/216 (34.72%) | 38/108 (35.19%) | 0.428 | 10.871 ± 8.614 | 14.842 ± 11.804 | 15.632 ± 12.053 | 0.035 | |
| Atrophic gastritis | 45/97 (46.39%) | 89/238 (37.39%) | 64/175 (36.57%) | 0.233 | 13.978 ± 9.631 | 13.079 ± 9.714 | 12.672 ± 11.370 | 0.062 | |
| Intraepithelial neoplasia | 5/9 (55.56%) | 7/17 (41.18%) | 7/27 (25.93%) | 0.236 | 9.600 ± 12.522 | 20.000 ± 12.000 | 15.429 ± 12.528 | 0.377 | |
| Total | 390/1093 (35.68%) | 470/1157 (40.62%) | 225/597 (37.68%) | 0.053 | 12.357 ± 9.918 | 14.241 ± 10.857 | 14.018 ± 10.869 | 0.024 | |
| LVX | Superficial gastritis | 66/270 (32.35%) | 83/206 (40.29%) | 30/82 (36.59%) | < 0.001 | 10.212 ± 12.180 | 12.651 ± 14.730 | 16.600 ± 13.278 | 0.103 |
| Erosive gastritis | 190/481 (39.5%) | 252/480 (52.50%) | 107/205 (52.20%) | < 0.001 | 11.353 ± 12.658 | 11.569 ± 11.322 | 15.047 ± 13.259 | 0.025 | |
| Peptic ulcer | 87/236 (36.86%) | 102/216 (47.22%) | 52/108 (48.15%) | 0.042 | 10.552 ± 10.391 | 12.990 ± 13.157 | 11.962 ± 13.774 | 0.403 | |
| Atrophic gastritis | 58/97 (59.79%) | 129/238 (54.20%) | 76/175 (43.43%) | 0.019 | 11.378 ± 8.858 | 12.946 ± 12.021 | 13.000 ± 12.087 | 0.561 | |
| Intraepithelial neoplasia | 4/9 (44.44%) | 7/17 (41.18%) | 9/27 (33.33%) | 0.786 | 21.000 ± 28.729 | 8.571 ± 5.381 | 20.889 ± 19.160 | 0.370 | |
| Total | 405/1093 (37.05%) | 573/1157 (49.52%) | 274/597 (45.90%) | < 0.001 | 11.059 ± 11.841 | 12.252 ± 12.295 | 14.255 ± 13.306 | 0.004 | |
| FR | Superficial gastritis | 23/270 (8.52%) | 16/206 (7.77%) | 7/82 (8.54%) | 0.952 | 14.783 ± 18.268 | 11.250 ± 9.147 | 16.000 ± 21.541 | 0.741 |
| Erosive gastritis | 68/481 (14.14%) | 62/480 (12.92%) | 33/205 (16.01%) | 0.542 | 9.074 ± 6.991 | 11.161 ± 12.443 | 10.424 ± 10.047 | 0.486 | |
| Peptic ulcer | 30/236 (12.71%) | 33/216 (15.28%) | 9/108 (8.33%) | 0.212 | 10.367 ± 8.475 | 7.697 ± 5.834 | 6.667 ± 4.000 | 0.208 | |
| Atrophic gastritis | 36/97 (37.11%) | 50/238 (21.01%) | 17/175 (9.71%) | < 0.001 | 7.889 ± 5.533 | 7.280 ± 4.895 | 7.294 ± 6.669 | 0.865 | |
| Intraepithelial neoplasia | 3/9 (33.33%) | 2/17 (11.76%) | 2/27 (7.41%) | 0.135 | 12.000 ± 6.928 | 36.000 ± 39.598 | 24.000 ± 11.314 | 0.517 | |
| Total | 160/1093 (14.64%) | 163/1157 (14.09%) | 68/597 (11.39%) | 0.162 | 9.925 ± 9.591 | 9.583 ± 10.095 | 10.118 ± 10.828 | 0.919 | |
Table 6.
Resistance rates of H. pylori strains between male and female in different gastric diseases.
| Antibiotics | Diseases | Gender | P | MIC ( ± s) | P | |||
|---|---|---|---|---|---|---|---|---|
| Male | Female | Male | Female | Total | ||||
| AMX | Superficial gastritis | 25/238 (10.5%) | 22/320 (6.88%) | 0.127 | 6.443 ± 13.913 | 10.340 ± 14.694 | 8.516 ± 14.313 | 0.357 |
| Erosive gastritis | 40/539 (7.42%) | 50/627(7.97%) | 0.724 | 7.331 ± 14.699 | 8.865 ± 16.105 | 8.183 ± 15.430 | 0.642 | |
| Peptic ulcer | 28/372 (7.53%) | 9/188 (4.79%) | 0.218 | 7.446 ± 16.444 | 17.861 ± 18.578 | 9.980 ± 17.321 | 0.118 | |
| Atrophic gastritis | 12/219 (5.48%) | 37/291 (12.71%) | 0.006 | 8.479 ± 19.682 | 4.527 ± 5.963 | 5.495 ± 10.881 | 0.279 | |
| Intraepithelial neoplasia | 4/33 (12.12%) | 6/20 (30.00%) | 0.107 | 2.875 ± 3.473 | 7.500 ± 7.141 | 5.650 ± 6.169 | 0.269 | |
| Total | 109/1401 (7.78%) | 124/1446 (8.58%) | 0.439 | 7.728 ± 13.518 | 8.014 ± 15.359 | 7.862 ± 14.378 | 0.439 | |
| CLR | Superficial gastritis | 78/238 (32.77%) | 125/320 (39.06%) | 0.127 | 14.718 ± 10.534 | 12.808 ± 10.939 | 13.542 ± 10.799 | 0.221 |
| Erosive gastritis | 209/539 (38.78%) | 273/627 (43.54%) | 0.099 | 14.077 ± 11.039 | 13.212 ± 9.929 | 13.587 ± 10.423 | 0.367 | |
| Peptic ulcer | 111/372 (29.84%) | 72/188 (38.3%) | 0.044 | 12.307 ± 10.842 | 15.306 ± 10.795 | 13.487 ± 10.893 | 0.069 | |
| Atrophic gastritis | 83/219 (37.90%) | 115/291 (39.52%) | 0.710 | 12.843 ± 9.653 | 13.374 ± 10.646 | 13.152 ± 10.220 | 0.720 | |
| Intraepithelial neoplasia | 9/33 (27.27%) | 10/20 (50.00%) | 0.094 | 20.889 ± 13.679 | 10.800 ± 9.247 | 15.579 ± 12.357 | 0.074 | |
| Total | 490/1401 (34.98%) | 595/1446 (41.15%) | 0.001 | 13.371 ± 10.380 | 13.694 ± 10.778 | 13.517 ± 10.558 | 0.616 | |
| LVX | Superficial gastritis | 64/238 (26.89%) | 115/320 (35.94%) | < 0.001 | 12.078 ± 14.142 | 12.600 ± 13.504 | 12.413 ± 13.698 | 0.808 |
| Erosive gastritis | 232/539 (43.04%) | 317/627 (50.56%) | 0.010 | 11.795 ± 11.880 | 12.448 ± 12.531 | 12.172 ± 12.247 | 0.538 | |
| Peptic ulcer | 154/372 (41.40%) | 87/188 (46.28%) | 0.271 | 10.279 ± 10.520 | 14.736 ± 14.742 | 11.888 ± 12.370 | 0.007 | |
| Atrophic gastritis | 97/219 (44.29%) | 166/291 (57.04%) | 0.004 | 14.144 ± 11.609 | 11.639 ± 11.215 | 12.563 ± 11.405 | 0.086 | |
| Intraepithelial neoplasia | 10/33 (30.30%) | 10/20 (50.00%) | 0.152 | 12.400 ± 10.906 | 20.800 ± 23.232 | 16.600 ± 18.182 | 0.314 | |
| Total | 557/1401 (39.76%) | 695/1446 (48.06%) | < 0.001 | 12.686 ± 12.920 | 11.829 ± 11.771 | 12.305 ± 12.425 | 0.225 | |
| FR | Superficial gastritis | 17/238 (7.14%) | 29/320 (9.06%) | 0.415 | 12.235 ± 15.262 | 14.621 ± 16.640 | 13.739 ± 16.014 | 0.631 |
| Erosive gastritis | 80/539 (14.84%) | 83 (13.24%) | 0.431 | 12.750 ± 9.580 | 9.554 ± 10.350 | 10.141 ± 9.967 | 0.446 | |
| Peptic ulcer | 46/372 (12.37%) | 26/188 (13.83%) | 0.625 | 8.826 ± 7.523 | 8.423 ± 6.014 | 8.681 ± 6.974 | 0.816 | |
| Atrophic gastritis | 48/219 (21.92%) | 55/291 (18.90%) | 0.401 | 7.750 ± 5.973 | 7.272 ± 4.874 | 7.495 ± 5.392 | 0.656 | |
| Intraepithelial neoplasia | 2/33 (6.06%) | 5/20 (25.00%) | 0.048 | 16.000 ± 0 | 24.800 ± 24.397 | 22.286 ± 20.377 | 0.650 | |
| Total | 193/1401 (13.78%) | 198/1446 (13.69%) | 0.949 | 9.899 ± 10.877 | 9.731 ± 9.042 | 9.816 ± 10.001 | 0.868 | |
Effect of PPI treatment history on antibiotic resistance rate
PPI treatment history was also a factor affecting H. pylori resistance rates. As shown in Table 7, the resistance rates of AMX, CLR and LVX in patients who recently took PPI in three months were 11.28%, 47.18% and 47.86%, respectively, which were all significantly higher than those in patients without PPI treatment history. However, the opposite trend was observed in FR under the same circumstances. It is observed that a recent PPI treatment history will increase the antibiotic resistance rate by 4%-12%, except for FR.
Table 7.
Effect of PPI treatment history on antibiotic resistance rate.
| History of PPI treatment | No. of patients | Antibiotics | MIC ( ± s) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| AMX | CLR | LVX | FR | AMX | CLR | LVX | FR | ||
| No PPI history | 2262 | 167 (7.38%) | 809 (35.76%) | 972 (42.97%) | 329 | 7.488 ± 13.613 | 12.872 ± 9.845 | 12.498 ± 12.311 | 9.693 ± 9.641 |
| PPI history | 585 | 66 (11.28%) | 276 (47.18%) | 542 (47.86%) | 62 (10.06%) | 8.807 ± 16.227 | 15.410 ± 12.240 | 11.634 ± 12.810 | 10.468 ± 11.796 |
| P | 0.002 | < 0.001 | 0.034 | 0.013 | 0.529 | < 0.001 | 0.305 | 0.576 | |
Nomogram development for antibiotic resistance risk
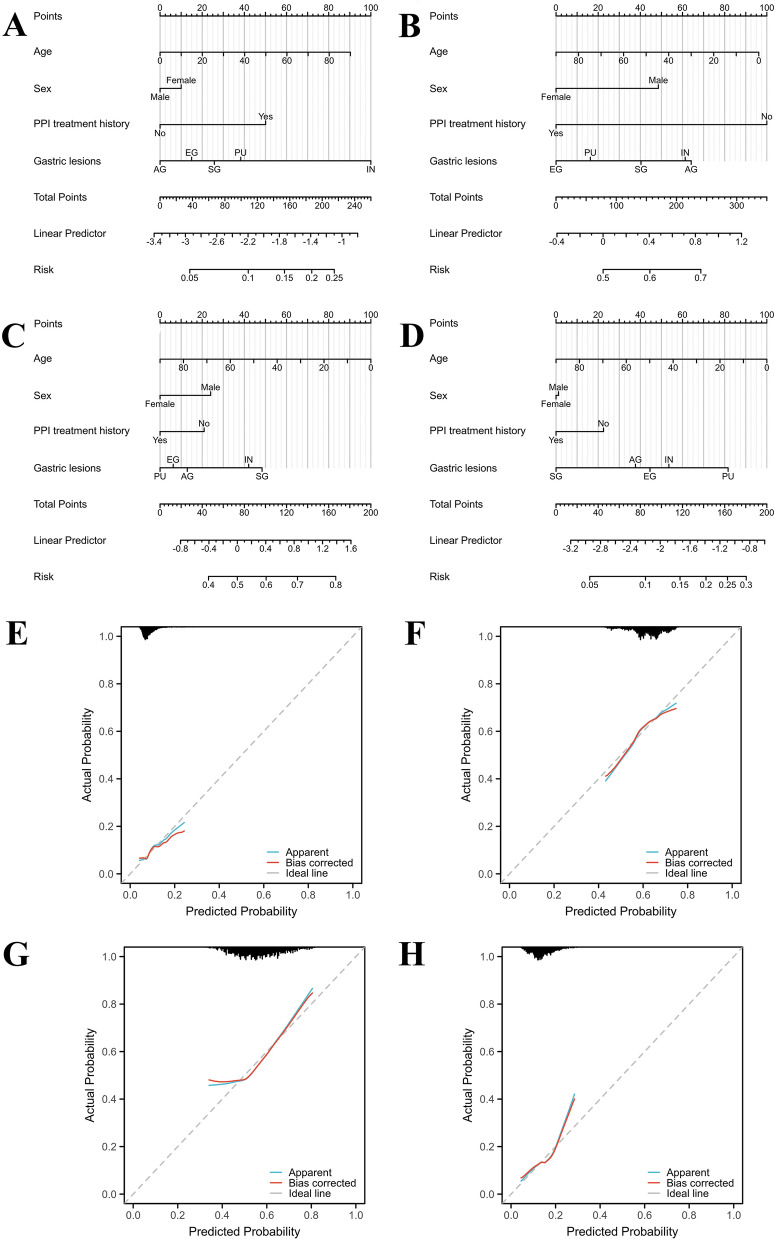
Age, sex, PPI treatment history and gastric disease were all incorporated into nomograms to predict the individual risk of antibiotic resistance (Fig. 1). The C-index of the nomograms for AMX, CLR, LVX and FR was 0.592, 0.578, 0.609, and 0.601, respectively. Calibration curves reflected good discriminative ability (Fig. 1).
Figure 1.
Nomogram development for antibiotic resistance risk. Nomogram models were constructed to predict the risk of amoxicillin (A), clarithromycin (B), levofloxacin (C) and furazolidone (D). Calibration curves reflected good discriminative ability of models for amoxicillin (E), clarithromycin (F), levofloxacin (G) and furazolidone (H). Individual factors such as age, sex, PPI treatment history and gastric disease are all incorporated into antibiotic resistance prediction models. In clinical practice, users can calculate total points of the model based on individual factors, and then conveniently obtain the probability data of specific antibiotic resistance to predict the individual risk. For example, a 45 year-old man with PPI history and his endoscopic finding was atrophic gastritis, the risk of clarithromycin resistance will exceed 60%. Therefore, clarithromycin should not be prioritized for use, and bismuth-containing quadruple therapy should be considered as the first line empirical treatment for this patient according to Maastricht consensus. SG, superficial gastritis; EG, erosive gastritis; PU, peptic ulcer; AG, atrophic gastritis; IN, intraepithelial neoplasia.
Discussion
Currently, eradication of H. pylori faces a series of challenges. The increasing proportion of antibiotic-resistant strains and the emergence of multidrug-resistant strains have increased the difficulty of antibiotic selection15–17. For example, AMX is widely used as a first-line treatment in current therapeutic regimens for H. pylori eradication due to its affordability and safety11. However, due to the widespread emergence of AMX-resistance strains, amoxicillin in Africa has lost its clinical application value, in where the overall rate of resistance to AMX have reached as high as 72.6%18. In addition, the lack of sufficient retrospective studies to provide guidance for eradication therapy and the differences in resistance among different populations and regions also make the success rate of primary eradication unsatisfactory19,20. In the absence of an antibiotic sensitivity test, rational selection of antibiotics has become the key to H. pylori eradication therapy.
To change this situation and increase the rationality of antibiotic selection, we need to conduct a retrospective study on the resistance of commonly used antibiotics. Since AMX, CLR, LVX, and FR are commonly used antibiotics in H. pylori eradication therapy in China21,22, in this study, an 8-year clinical retrospective study was performed to mainly analyze the antibiotic resistance rate of H. pylori in patients with different types of gastric mucosal lesions. Our statistical results showed that the overall resistance rates of AMX, CLR, LVX, and FR were 8.18%, 38.11%, 43.98% and 13.73%, respectively. The resistance rates of CLR and LVX were greater than 30%, which implies that they were not suitable for empirical treatment unless under the guidance of an antimicrobial sensitivity test. Among all the types of double resistance patterns, CLR and LVX were the most common combination in all gastric mucosal lesions, whereas the combination of AMX and FR had the lowest drug resistance rate, which suggests that AMX + FR is the best combination for H. pylori eradication therapy, especially in the first empirical treatment. Moreover, compared with the period from 2014 to 2016, the rates of mono-resistance and multiple resistance all showed relatively downward trends in recent 5 years (Table 2). Within the region, the no resistance strain increased from 24.06 to 41.31%, while the double resistance and triple resistance strain decreased from 32.89% and 8.96% to 23.49% and 5.57%, respectively. This situation may be owing to the relatively strict management of antibiotic application in China in recent years23. However, it is worth noting that compared with neighboring countries, our H. pylori antibiotic resistance rate is still at a relatively high level24–26.
H. pylori infection causes different types of gastric mucosal lesions, and it remains unclear whether the type of gastric mucosal lesion a factor or feature associated with H. pylori resistance. There is no relevant research report on this issue at present. Therefore, the relationship between the type of gastric lesions and the H. pylori resistance rate is the theme of this study. Remarkably, according to our data, we confirmed that there are significant differences in the resistance rates of these four antibiotics among different gastric mucosal lesions. More specifically, patients with atrophic gastritis have more serious H. pylori resistance than other groups. In the atrophic gastritis group, the resistance rates of the four antibiotics were all higher than the average level. Patients with atrophic gastritis have higher risk of resistant strains infection. Altogether, our results indicated that atrophic gastritis is an important clinical feature of high-risk antibiotic resistance in H. pylori-infected patients. This phenomenon may be related to bacterial-specific virulence factors such as cytotoxin-associated gene A (CagA) and vacuolating cytotoxin A (VacA). For example, CagA is an important effector protein of H. pylori, which was previously demonstrated to not only activate cell senescence and cause gastric mucosal atrophy but also promote H. pylori strains to further form biofilms, thus causing multidrug resistance27–29.
The antibiotic resistance rate of H. pylori vary greatly among different populations and regions and are affected by many factors, such as age, sex and medication history13,30. In our study, we found that the resistance rates of CLR and LVX in females were significantly higher than those in males. The resistance rates of AMX and LVX in different age groups were significantly different. Our results suggest that the distribution of antibiotic resistance was associated with age and sex. Therefore, we should consider the patient's age and sex when choosing antibiotics for H. pylori eradication.
It is worth mentioning that it remains unclear whether PPI treatment history before eradication therapy will affect the resistance rate of H. pylori. Previous studies have suggested that PPIs cause H. pylori to migrate from the antrum to the gastric body and transform into a spherical shape, thereby reducing the sensitivity of H. pylori to antibiotics31,32. However, some meta-analyses revealed that preadministration of PPIs did not affect the final eradication rate of H. pylori infection33,34, suggesting that PPI treatment history before eradication therapy may not affect H. pylori resistance. Thus, we assessed the effect of PPI treatment history on the antibiotic resistance rate in this study. We observed that a recent PPI treatment history will increase the antibiotic resistance rate by 4–12%, except for FR, hinting at multiple mechanisms for PPIs in response to antibiotic resistance.
As the proportion of antibiotic-resistant H. pylori strains continues to rise and the emergence of multidrug resistance strains, eradication therapy are constantly evolving11. Due to the declining efficacy of legacy triple therapies, bismuth-containing quadruple therapies have been recommend as the best initial empiric treatment35. Some bismuth-free quadruple options such as concomitant, sequential and hybrid therapies are only recommended for areas with low clarithromycin and metronidazole resistance35. Moreover, vonoprazan-containing regimens show high efficacy in terms of H. pylori eradication compared with PPI-containing therapy, which indicate future therapies may be influenced by adding the novel potassium-competitive acid blocker11,36,37. However, comprehensive consideration of local antibiotic resistance and individual factors is necessary to propose more effective individualized therapeutic regimens. In this study, we established antibiotic resistance prediction models based on individual factors such as age, sex, PPI treatment history and gastric disease, which all closely related to H. pylori resistance. Our models are presented in the form of nomograms, which will be better used by clinical doctors for predicting patients’ individual risk of antibiotic resistance.
Admittedly, our research has some limitations. The major limitations of the present research is that it is a single center study with a single source of patient population. Moreover, our study is a retrospective study. Although most patients in this study have no previous history of H. pylori eradication treatment, there are still a very small number of patients who have undergone eradication failure. In addition, our study is based on the results of antimicrobial susceptibility testing in vitro, and the potential risk of heteroresistance of H. pylori should not be ignored38,39. However, these limitations have extremely limited influence and do not affect the credibility of our study.
In summary, our data provide the association between antibiotic resistance of H. pylori and gastritis pattern, which indicate factors including age, sex, type of gastric lesions and recent PPI treatment history are associated with H. pylori resistance rate. Atrophic gastritis is an important clinical feature of high-risk antibiotic resistance in H. pylori-infected patients. Patients with atrophic gastritis, PPI history and older age have higher risk of resistant strains infection.
Acknowledgements
Ye and Shao made substantial contributions to conception and design of this manuscript. Lin, Fang and Shao were involved in drafting the manuscript and revising it critically for important intellectual content. Yan drew the figure in this manuscript. Ye and Zheng reviewed and revised the final manuscript. All authors contributed to the figure discussions and approved the final manuscript submitted.
Author contributions
G.Y. and Y.S. made substantial contributions to conception and design of this manuscript. Y.L., Z.F. and Y.S. were involved in drafting the manuscript and revising it critically for important intellectual content. J.Y. drew the figure in this manuscript. G.Y. and T.Z. reviewed and revised the final manuscript. All authors contributed to the figure discussions and approved the final manuscript submitted.
Funding
This study was supported by grants from the Key Scientific and Technological Projects of Ningbo (No. 2021Z133), National Natural Science Foundation of China (No.81702367) and the Youth Medical Backbone Talents Training Program of Ningbo.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
These authors contributed equally: Yongfu Shao, Yifan Lin and Ziyi Fang.
Contributor Information
Tuo Zheng, Email: ztuo1209@sina.com.
Guoliang Ye, Email: yeguoliang@nbu.edu.cn, Email: ndfyygl@126.com.
References
- 1.Yang H, Hu B. Diagnosis of Helicobacter pylori infection and recent advances. Diagn. Basel. 2021;11:1305. doi: 10.3390/diagnostics11081305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Yang H, Hu B. Immunological perspective: Helicobacter pylori infection and gastritis. Med. Inflamm. 2022;8:2944156. doi: 10.1155/2022/2944156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Deng L, et al. An improved quantitative real-time polymerase chain reaction technology for Helicobacter pylori detection in stomach tissue and its application value in clinical precision testing. BMC Biotechnol. 2020;20:33. doi: 10.1186/s12896-020-00624-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Dabbebi H, et al. Effect of Helicobacter pylori eradication therapy on the response of MALT-Gastric lymphoma. Tunis Med. 2022;100:37–43. [PMC free article] [PubMed] [Google Scholar]
- 5.Rupp S, et al. Diagnostic approach to Helicobacter pylori-related gastric oncogenesis. Ann. Gastroenterol. 2022;35:333–344. doi: 10.20524/aog.2022.0725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Vu TB, Tran TNQ, Tran TQA, Vu DL, Hoang VT. Antibiotic resistance of Helicobacter pylori in patients with peptic ulcer. Med. Kaunas. 2022;59:6. doi: 10.3390/medicina59010006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Argueta EA, Moss SF. The prevention of gastric cancer by Helicobacter pylori eradication. Curr. Opin. Gastroenterol. 2021;37:625–630. doi: 10.1097/MOG.0000000000000777. [DOI] [PubMed] [Google Scholar]
- 8.Argueta EA, Moss SF. Treatment of Helicobacter pylori. Curr. Opin. Gastroenterol. 2019;35:544–550. doi: 10.1097/MOG.0000000000000578. [DOI] [PubMed] [Google Scholar]
- 9.Kim SY, et al. Antibiotic treatment for Helicobacter pylori: Is the end coming? World J. Gastrointest. Pharmacol. Ther. 2015;6:183–198. doi: 10.4292/wjgpt.v6.i4.183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Abadi ATB, Ierardi E. Vonoprazan and Helicobacter pylori treatment: A lesson from Japan or a limited geographic phenomenon? Front. Pharmacol. 2019;10:316. doi: 10.3389/fphar.2019.00316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lin YF, et al. Antibiotic resistance in Helicobacter pylori: From potential biomolecular mechanisms to clinical practice. J. Clin. Lab. Anal. 2023;37:e24885. doi: 10.1002/jcla.24885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Boyanova L, et al. Evolution of Helicobacter pylori resistance to antibiotics: A topic of increasing concern. Antibiot. (Basel) 2023;12:56–62. doi: 10.3390/antibiotics12020332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Wang J, et al. Prevalence of antibiotic resistance of Helicobacter pylori isolates in Shanghai, China. Am. J. Transl. Res. 2022;14:7831–7841. [PMC free article] [PubMed] [Google Scholar]
- 14.Zhang Y, et al. Antibiotic resistance of Helicobacter pylori and related risk factors in Yangzhou, China: A cross-sectional study. J Clin Med. 2023;12:816. doi: 10.3390/jcm12030816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Shao YF, Lin YF, Wang BJ, Miao M, Ye GL. Antibiotic resistance status of Helicobacter pylori strains isolated from initial eradication patients in Ningbo, China, from 2017 to 2021. Helicobacter. 2022;27:e1292. doi: 10.1111/hel.12920. [DOI] [PubMed] [Google Scholar]
- 16.Tshibangu-Kabamba E, Yamaoka Y. Helicobacter pylori infection and antibiotic resistance—from biology to clinical implications. Gastroenterol. Hepatol. 2021;18:613–629. doi: 10.1038/s41575-021-00449-x. [DOI] [PubMed] [Google Scholar]
- 17.Kuo CJ, et al. Multidrug resistance: The clinical dilemma of refractory Helicobacter pylori infection. J. Microbiol. Immunol. Infect. 2021;54:1184–1187. doi: 10.1016/j.jmii.2021.03.006. [DOI] [PubMed] [Google Scholar]
- 18.Jaka H, et al. The magnitude of antibiotic resistance to Helicobacter pylori in Africa and identified mutations which confer resistance to antibiotics: Systematic review and meta-analysis. BMC Infect. 2018;18:193. doi: 10.1186/s12879-018-3099-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Yan TL, et al. Current status of Helicobacter pylori eradication and risk factors for eradication failure. World J. Gastroenterol. 2020;26:4846–4856. doi: 10.3748/wjg.v26.i32.4846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Xu X, He C, Zhu Y. Treatment of refractory Helicobacter pylori infection: A new challenge for clinicians. Front. Microbiol. 2022;13:998240. doi: 10.3389/fmicb.2022.998240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Zhong ZS, et al. A retrospective study of the antibiotic-resistant phenotypes and genotypes of Helicobacter pylori strains in China. Am. J. Cancer Res. 2021;11:5027–5037. [PMC free article] [PubMed] [Google Scholar]
- 22.Hu Y, Zhu Y, Lu NH. Primary antibiotic resistance of Helicobacter pylori in China. Dig. Dis. Sci. 2017;62:1146–1154. doi: 10.1007/s10620-017-4536-8. [DOI] [PubMed] [Google Scholar]
- 23.Du Y, et al. Consensus on eradication of Helicobacter pylori and prevention and control of gastric cancer in China (2019, Shanghai) J. Gastroenterol. Hepatol. 2020;35:624–629. doi: 10.1111/jgh.14947. [DOI] [PubMed] [Google Scholar]
- 24.Kageyama C, et al. Increase in antibiotic resistant Helicobacter pylori in a University Hospital in Japan. Infect. Drug Resist. 2019;12:597–602. doi: 10.2147/IDR.S196452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Park JY, et al. The prevalence of multidrug resistance of Helicobacter pylori and Its impact on eradication in Korea from 2017 to 2019: A single-center study. Antibiot. Basel. 2020;9:646. doi: 10.3390/antibiotics9100646. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Schubert JP, et al. Antibiotic resistance of Helicobacter pylori in Australia and New Zealand: A systematic review and meta-analysis. J. Gastroen. Hepatol. 2021;36:1450–1456. doi: 10.1111/jgh.15352. [DOI] [PubMed] [Google Scholar]
- 27.Cai Q, et al. Inflammation-associated senescence promotes Helicobacter pylori-induced atrophic gastritis. Cell Mol. Gastroenterol. Hepatol. 2021;11:857–880. doi: 10.1016/j.jcmgh.2020.10.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Takahashi-Kanemitsu A, Knight CT, Hatakeyama M. Molecular anatomy and pathogenic actions of Helicobacter pylori CagA that underpin gastric carcinogenesis. Cell Mol. Immunol. 2020;17:50–63. doi: 10.1038/s41423-019-0339-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Jeyamani L, et al. CagA and VacA genes of Helicobacter pylori and their clinical relevance. Indian J. Pathol. Microbiol. 2018;61:66–69. doi: 10.4103/IJPM.IJPM_234_17. [DOI] [PubMed] [Google Scholar]
- 30.Goldstein NS. Chronic inactive gastritis and coccoid Helicobacter pylori in patients treated for gastroesophageal reflux disease or with H. pylori eradication therapy. Am. J. Clin. Pathol. 2002;118:719–726. doi: 10.1309/LJ4D-E2LX-7UMR-YMTH. [DOI] [PubMed] [Google Scholar]
- 31.Zhang M. High antibiotic resistance rate: A difficult issue for Helicobacter pylori eradication treatment. World J. Gastroenterol. 2015;21:13432–13437. doi: 10.3748/wjg.v21.i48.13432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Gasparetto M, Pescarin M, Guariso G. Helicobacter pylori eradication therapy: Current availabilities. ISRN Gastroenterol. 2012;2012:186734. doi: 10.5402/2012/186734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Kuang S, et al. The influence of pretreatment with PPI on Helicobacter pylori eradication: A systematic review and meta-analysis. Med. Baltim. 2021;100:e27944. doi: 10.1097/MD.0000000000027944. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Yoon SB, et al. Long-term pretreatment with proton pump inhibitor and Helicobacter pylori eradication rates. World J. Gastroenterol. 2014;20:1061–1066. doi: 10.3748/wjg.v20.i4.1061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Georgopoulos S, Papastergiou V. An update on current and advancing pharmacotherapy options for the treatment of H. pylori infection. Expert Opin. Pharmacother. 2021;22:729–741. doi: 10.1080/14656566.2020.1845649. [DOI] [PubMed] [Google Scholar]
- 36.Abadi TBA, et al. Helicobacter pylori therapy and clinical perspective. J. Glob. Antimicrob. Resist. 2018;14:111–117. doi: 10.1016/j.jgar.2018.03.005. [DOI] [PubMed] [Google Scholar]
- 37.Suzuki S, Esaki M, Kusano C, Ikehara H, Gotoda T. Development of Helicobacter pylori treatment: How do we manage antimicrobial resistance? World J. Gastroenterol. 2019;25:1907–1912. doi: 10.3748/wjg.v25.i16.1907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Rizvanov AA, et al. Helicobacter pylori and its antibiotic heteroresistance: A neglected issue in published guidelines. Front. Microbiol. 2019;10:1796. doi: 10.3389/fmicb.2019.01796. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Mascellino MT, et al. Antibiotic susceptibility, heteroresistance, and updated treatment strategies in Helicobacter pylori infection. Drug Des. Dev. Ther. 2017;11:2209–2220. doi: 10.2147/DDDT.S136240. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.