Abstract
The photochemical properties of Electron Donor-Acceptor (EDA) complexes present exciting opportunities for synthetic chemistry. However, these strategies often require an inert atmosphere to maintain high efficiency. Herein, we develop an EDA complex photocatalytic system through rational design, which overcomes the oxygen-sensitive limitation of traditional EDA photocatalytic systems and enables aerobic oxygenation reactions through dioxygen activation. The mild oxidation system transfers electrons from the donor to the effective catalytic acceptor upon visible light irradiation, which are subsequently captured by molecular oxygen to form the superoxide radical ion, as demonstrated by the specific fluorescent probe, dihydroethidine (DHE). Furthermore, this visible-light mediated oxidative EDA protocol is successfully applied in the aerobic oxygenation of boronic acids. We believe that this photochemical dioxygen activation strategy enabled by EDA complex not only provides a practical approach to aerobic oxygenation but also promotes the design and application of EDA photocatalysis under ambient conditions.
Subject terms: Photocatalysis, Organocatalysis, Synthetic chemistry methodology, Catalyst synthesis
The photochemical properties of Electron Donor-Acceptor (EDA) complexes present exciting opportunities, but often require an inert atmosphere to maintain high efficiency. Here, the authors develop a photocatalytic system through rational design, which overcomes the oxygen-sensitive limitation of traditional EDA photocatalytic systems.
Introduction
Visible-light photocatalysis has emerged as an important strategy for energy transfer and redox process, which has significantly contributed to the development of synthetic chemistry1,2. Under the assistance of photocatalysts, photocatalytic reactions typically occur under mild reaction conditions and employ simple oxidants or reductants like oxygen, air, or amines to replace stoichiometric reagents3. Traditionally, the π-conjugated aromatic dyes and transition metal complex chromophores which can harness visible light energy for chemical reactions with high efficiency, have been widely used in photochemistry (Fig. 1a)4. In recent decades, electron donor-acceptor (EDA)5 complex photochemistry has garnered increased attention, which did not require any additional photocatalyst for successful execution and provided new opportunities for synthetic chemistry (Fig. 1a)6. Upon excitation of the EDA complex, an intra-complex single electron transfer (SET) occurs from the donor (D) to the acceptor (A), resulting in the formation of reactive radicals7,8. This strategy has been further utilized in valuable transformations including borylation9, amination10, alkenylation11, alkylation12, acylation13, annulation14,15 and coupling16 (Fig. 1a)17–19.
Fig. 1. EDA complex photochemistry and dioxygen activation.
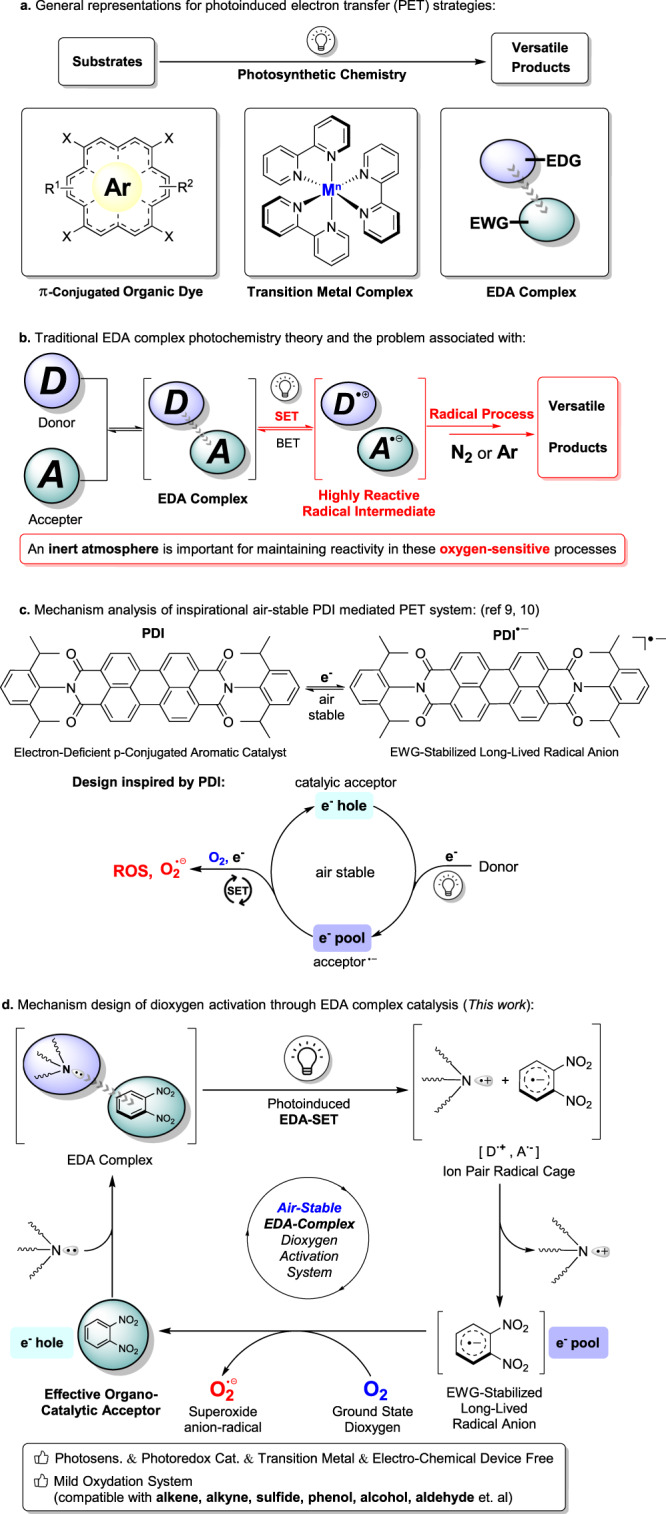
a Overview of photochemistry. b Traditional EDA complex photochemistry theory and the problem associated with. c Research inspiration. d Mechanism design.
In general, EDA complex photochemistry requires stoichiometric amounts of specifically tailored donor and acceptor substrates, limiting the overall scope of these transformations20. Recently, employing a catalytic approach where either the donor21–25 or acceptor26 is used catalytically to form EDA complex, has gained attention as a satisfactory strategy. Despite the fantastic developments of EDA complex photochemistry, compared to other photocatalysts, this strategy is often incompatible with dioxygen. As electron-rich donors are usually air-sensitive and the biradical dioxygen can disrupt the desired radical and SET processes, this approach must be performed under an inert gas atmosphere (Fig. 1b)27–32.
Interestingly, König and coworkers reported a consecutive visible light photoinduced electron transfer (conPET) using an air-stable electron-deficient π-conjugated aromatic molecule PDI (Fig. 1c)33. The intermediate generated during photoexcitation is stabilized by electron-withdrawing groups and π-conjugation effects. Inspired by this chemistry and the generating e- pool and hole moieties34,35 we hypothesized that the discovery of an appropriate acceptor catalyst capable of producing an air-stable e- pool would offer us an opportunity to develop an EDA system compatible with dioxygen (Fig. 1c).
Furthermore, this air stable e- pool could transfer its free electron to dioxygen to generate an active superoxide radical anion, and enable exciting EDA complex catalyzed oxygenation reactions (Fig. 1c). Over the past few decades, organic chemists have taken a keen interest in the use of environmentally friendly molecular oxygen as the preferred oxidant for oxygenation reactions36–49. Although there have been considerable advancements in dioxygen activation assisted by photocatalysts50–56, the dioxygen activation and oxygenation via an EDA photocatalysis remains unknown. Herein, we report an air-stable EDA complex system that employs commercially available nitroaromatic compounds as effective organocatalytic acceptors and tertiary amines as electron donors (Fig. 1d). By using the catalytic amount of electron acceptor, the resulting dinitrobenzene radical anion is stabilized by the nitro groups, effectively inhibiting unproductive back-electron transfer (BET)57,58. This protocol efficiently initiated the generation of highly reactive superoxide radical anion under visible light excitation, thereby facilitating the aerobic oxygenation of boronic acids and producing hydroxylation products (Fig. 1d). Moreover, to the best of our knowledge, this is one of the few catalytic acceptor systems in the reported EDA complex photochemistry.
Results and discussion
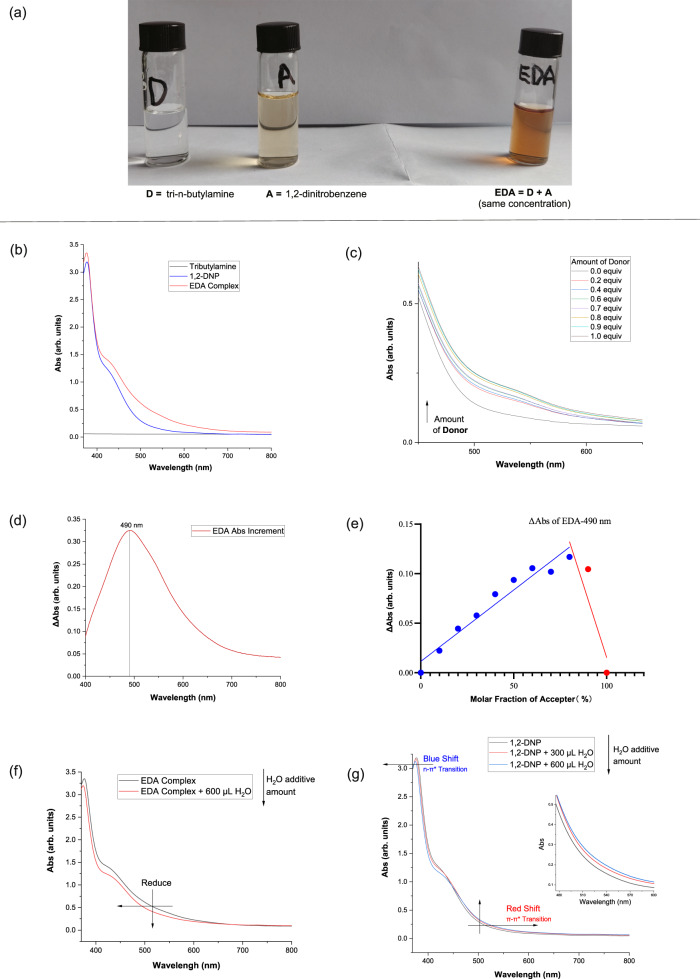
Based on the hypothesis, we examined the feasibility and properties of this proposed system. Further examination demonstrated the formation of a ground state EDA complex by UV/Vis spectroscopy (Fig. 2). Equimolar amounts of pale yellow 1,2-dinitrobenzene and colorless tri-n-butylamine were combined in acetonitrile, resulting in a darker solution (Fig. 2a). Subsequent measurement of the UV-Vis spectrum indicated a significant red shift and increased absorption of visible light (Fig. 2b). Commercially available 1,2-dinitrobenzene might not be pure, leading to a darker solution that affects light absorption. To resolve this, conduct simple column chromatography to get a pure, pale-yellow reagent. The observed absorption of visible light corresponds to the charge-transfer absorption of the EDA complex. This phenomenon is consistent with the expected effects of intermolecular interactions. While ensuring a constant final concentration of the acceptor, as the proportion of the Donor increases, the absorbance of the entire system gradually increases, displaying an initially rapid and then slower rate of increase. Once the Donor and Acceptor reach an equimolar ratio, the absorbance no longer exhibits a significant increase (Fig. 2c).
Fig. 2. Mechanistic experiments.
a Visual photograph of donor, acceptor and EDA complex. b Optical absorption spectra, the separate reaction components and appearance of the colored EDA complex. c UV-Vis absorption spectra of acceptor and that with different molar ratio of donor. d EDA Complex absorbance increment. e Job’s Plot. f Water hydrolysis of the EDA Complex. g The impact of water on the UV-visible absorption spectra of acceptor. a.u. arbitrary units.
It can be observed that the maximum increment in absorbance of the EDA complex occurs at 490 nm (Fig. 2d). Using this reference wavelength, a Job’s Plot experiment was conducted to investigate the absorbance increment of the EDA complex at different ratios. Surprisingly, the optimal binding ratio between the Acceptor and Donor was found to be 4:1 (Fig. 2e), which deviated from the commonly reported 1:1 binding ratio observed in most Acceptor and Donor pairs.
Furthermore, the addition of water (600 μL) to the EDA complex solution resulted in a significant reduction in system absorption and weakened the previously observed red shift phenomenon (Fig. 2f). When considering the dilution effect caused by the water addition (as demonstrated by a slight red shift in visible absorption upon adding water to the acceptor solution due to increased solvent polarity in the π-π* transition of nitroaromatics) (Fig. 2g), it was speculated that water acted as both a strong hydrogen bond donor and acceptor, thereby disrupting the formation of the EDA complex. This observation also provided an explanation for why the addition of 600 μL of water completely suppressed the subsequent oxygenation reaction involving superoxide radical species.
To demonstrate the presence of superoxide radicals, a highly reactive form of reactive oxygen species (ROS), a validation experiment was conducted. To monitor the fluorescence signal, the ROS fluorescent probe 2′,7′-Dichlorodihydrofluorescein (DCFH)59 was introduced into the system, and an incremental increase in its positive fluorescence signal was observed with prolonged illumination time (Fig. 3a). This observation strongly supports the generation of ROS by the current dioxygen activation system. To further support our hypothesis, we utilized the superoxide-specific fluorescent probe dihydroethidium (DHE) to monitor the superoxide species, as previously reported in the literature60. Upon illumination in the present protocol, we clearly detected a positive fluorescence signal from the DHE probe (Fig. 3b), and the oxygenation product of DHE was successfully identified through mass spectrometry, providing conclusive evidence for the presence of superoxide species generated through dioxygen activation.
Fig. 3. Detection of ROS.
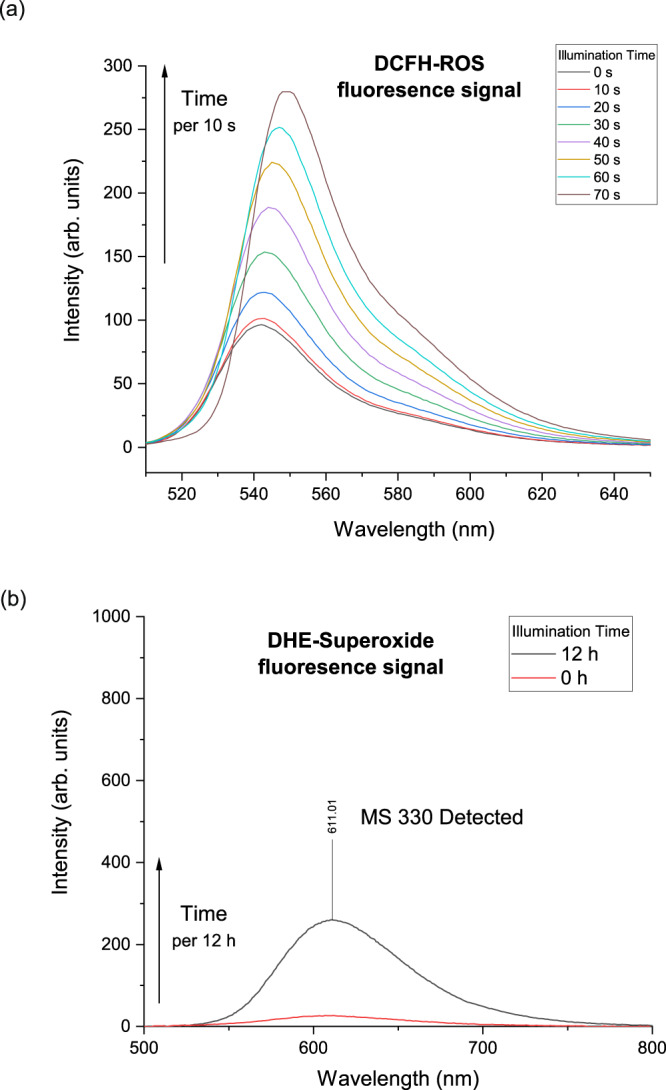
a ROS detection using DCFH fluorescence probe. b Superoxide detection using DHE fluorescence probe.
Having successfully demonstrated the compatibility and powerful capacity for the activation of oxygen to generate superoxide radical anions through this EDA complex photocatalytic system, we proceeded to employ organic boronic acid as substrates for the capture of the generated superoxide intermediates. This pivotal step provided further compelling evidence of the operability and practical value inherent in the entire oxygen activation process of this strategy. In contrast to the reported stoichiometric peroxide oxidants61–64, the EDA complex with 1,2-dinitrobenzene and tertiary amines was found very efficient to catalyzed the oxygenation process with O2 as the oxidant and oxygen-source, yielding corresponding phenol product (Table 1). Acetonitrile was the most effective solvent for promoting the oxidation-oxygenation reaction, while other commonly used solvents gave unfavorable results (entries 2–8). Solvents with strong hydrogen bond donating properties, like methanol, could hinder the formation of the EDA complex and deactivate the desired catalytic process. We further identified 1,2-dinitrobenzene as the most effective acceptor, and tri-n-butylamine as the optimal donor to avoid the undesirable SNAr reaction (entries 12–18)65,66. Control experiments indicated that the presence of light, dioxygen, donor, and acceptor was essential for this dioxygen activation methodology, providing preliminary confirmation of the rationality and feasibility of the designed mechanism (entries 19–22). Under standard conditions, by simply changing the oxygen atmosphere to ambient air, the reaction proceeded smoothly and yielded the target product but with a little bit lower efficiency (entry 23). It is noteworthy that only catalytic amount of electron acceptor (5 mol%) was employed for this highly efficient aerobic oxygenation (Table 1).
Table 1.
Screening of reaction conditions.a
 | ||
|---|---|---|
| Entry | Changed conditions | Isolated yield |
| 1 | None | 99% |
| 2 | Toluene as solvent | Trace |
| 3 | DMSO as solvent | ND |
| 4 | MeOH as solvent | Trace |
| 5 | THF as solvent | Trace |
| 6 | DCM as solvent | Trace |
| 7 | DMF as solvent | 17% |
| 8 | EtOAc as solvent | Trace |
| 9 | 0.5 eq amine Donor | 56% |
| 10 | 1.5 eq amine Donor | 98% |
| 11 | 2.0 eq amine Donor | 97% |
| 12 | Tri-n-octylamine as Donor | 29% |
| 13 | DABCO as Donor | Trace |
| 14 | Et3N as Donor | 28% |
| 15 | DIPEA as Donor | Trace |
| 16 | NPh3 as Donor | Trace |
| 17 | 1,3-DNB as Acceptor | 34% |
| 18 | 1,4-DNB as Acceptor | 17% |
| 19 | No Light | ND |
| 20 | No Donor | ND |
| 21 | No Acceptor | ND |
| 22 | Under Ar | ND |
| 23 | Under Air | 69% |
| 24 | 600 uL H2O as additive | Trace |
aReaction conditions: 1a (0.5 mmol), amine donor (0.5 mmol), 1,2-DNB acceptor (0.025 mmol, 5 mol%) in solvent (4.0 mL) under blue LED irradiation and oxygen atmosphere (1 atm).
After determining the optimized reaction conditions, we carried out experiments to evaluate the process of dioxygen activation using various boronic acid substrates (Fig. 4). The optimized conditions enabled us to achieve high yields of the oxygenation products. Notably, the reactivity of the dioxygen activation system displayed minimal sensitivity to the positional and electronic properties of the functional groups. Both electron-donating and electron-withdrawing groups were effectively accommodated by the system (Fig. 4). Additionally, functional groups that are typically easily oxidized by oxidants, such as alkynes (2b), sulfide groups (2c), alkenes (2d), aldehydes (2f), and benzyl position (2y), could be preserved under the optimized reaction conditions. These strong oxidant sensitive functional groups exhibited excellent compatibility within this oxidation-oxygenation system, allowing their preservation without any oxidative damage. This mild oxidation system demonstrated a wide range of substrates, making it promising for selectively oxidizing boryl groups in complex drug molecules or bioactive compounds containing multiple sensitive functional groups. The high tolerance of the reaction system towards functional groups suggested a gradual and suitable activation of oxygen, resulting in the generation of superoxide anion radicals during photocatalysis.
Fig. 4. Substrate scopea.
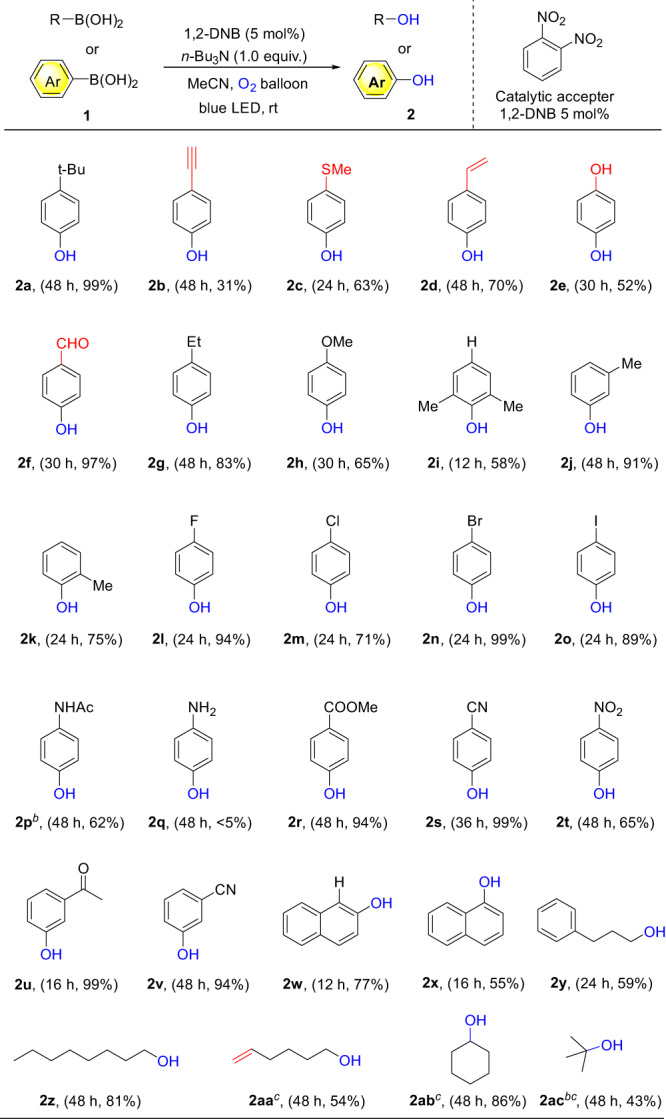
a Conditions: Reactions were conducted on 0.5 mmol scale using 1.0 equiv. amine, 5 mol% 1,2-DNB at 0.125 M. Isolated yields after chromatographic purification. b boronic acid pinacol ester was used as starting compound. c H-NMR Yield in CD3CN due to its low boiling point.
Moreover, besides conventional aromatic boronic compounds, alkyl-boronic acids also underwent efficient oxygenation reactions in this system, yielding the corresponding alkyl alcohol products. It is noteworthy that, apart from conventional phenyl-boronic acid, boronic esters could also serve as substrates, leading to the desired oxygenation products with good efficiency (2p), thereby expanding the range of substrates. However, unprotected amino groups posed a challenge in obtaining the desired products by this protocol. This was due to the electron-rich nature of amino groups, which competed with the donor and hindered the formation of the EDA complex. Nevertheless, this issue could be easily resolved by implementing a simple functional group protection strategy. In terms of alkyl substrates, we also attempted using secondary and tertiary alkylboron compounds as substrates, and achieved moderate to good yields of the target products.
Interestingly, the structurally complex boron-containing pharmaceutical molecule, bortezomib, worked well under the present EDA Complex photochemical oxidation-oxygenation reaction system (Fig. 5), showing its application in the late-stage modification of complex chemicals. Furthermore, to highlight the flexibility and application potential of this work, we also attempted using other types of substrates to capture the generated superoxide radicals. Interestingly, we have discovered that widely prevalent organic phosphorus compounds and organosilicon compounds can also be transformed into oxygenated products (Fig. 5).
Fig. 5. Late-stage modification of drug molecule and diversifying the applicationsa.
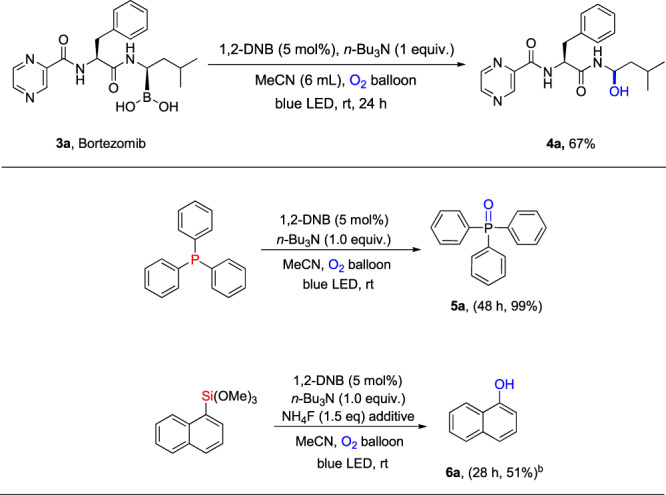
aConditions: Reactions were conducted on 0.5 mmol scale using 1.0 equiv. amine, 5 mol% 1,2-DNB at 0.125 M. Isolated yields after chromatographic purification. b1 equivalent of NH4F as additive.
Stern-Volmer quenching experiment gives the constant KSV = 94.1 (see Supplementary Figs. 14 and 15). As the concentration of the donor increases, the saturation of coordination leads to a decrease in the Stern-Volmer constant (KSV) slope, which also evidences the existence of the EDA Complex. Furthermore, the results from Cyclic Voltammetry (CV) experiments also support the interaction of Doner and Acceptor (see Supplementary Figs. 17–20). To investigate the possibility of a chain reaction occurring within this system, we conducted tests and calculations for the quantum yield, ultimately determining that the quantum yield for the standard reaction is 2.39. Additionally, the results of the light on/off experiments are presented in Fig. 6. Combining the experimental results and related literature67, we can conclude that the reaction is likely a light-driven chain reaction. The illumination provides the necessary energy for initiating and maintain the chain reaction. The experimental findings, coupled with studies on the mechanism in related literature, hint at the existence of short chain reactions. Specifically, this involves the formation of oxidizing peroxide or radical intermediates through the reaction of certain amine radical intermediates with oxygen68,69.
Fig. 6. Light on/off experiments.
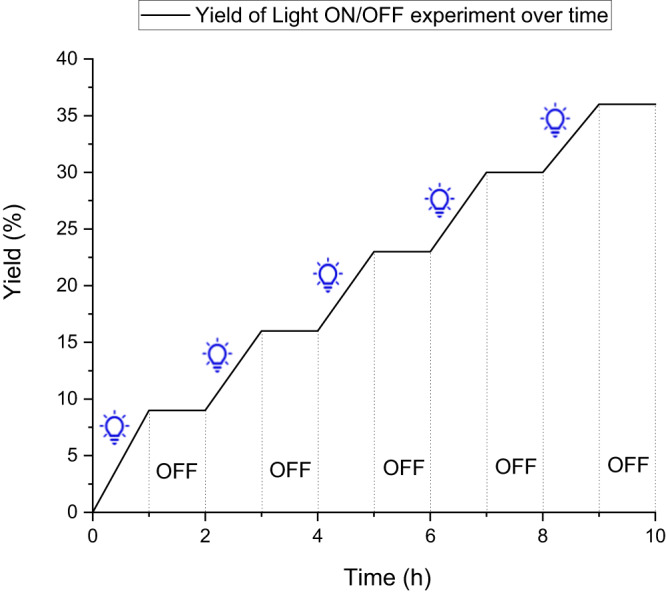
Under the symbol of a light bulb corresponds to the period of light-induced reactions, above the term 'OFF' corresponds to dark reactions, and the vertical axis represents the reaction yield.
Based on the aforementioned experimental results and relevant mechanistic validation experiments, the reaction mechanism is proposed in Fig. 7. Initially, tri-n-butylamine as the electron donor interacts with a catalytic amount of 1,2-dinitrobenzene, which served as the electron acceptor in solution, leading to the formation of the EDA complex. This EDA complex has a visible light absorption charge-transfer band. Under excitation by visible light, a single electron transfer (SET) process takes place, where an electron from the donor is transferred to the acceptor, resulting in the formation of a diradical cage in acetonitrile solution. The electron-accepting intermediate, 1,2-dinitrobenzene radical anion A, is stabilized by the presence of two strongly electron-withdrawing nitro groups, preventing unproductive back electron transfer (BET). Subsequently, the dioxygen molecule captures the electron from the dinitrobenzene radical anion A, generating a superoxide radical anion, which is a kind of classical reactive oxygen species (ROS) with strong oxidizing capability. Simultaneously, the acceptor is regenerated, completing the catalytic cycle. The superoxide anion radical is then trapped by boronic acid, forming a peroxide radical intermediate C, which undergoes hydrogen atom transfer (HAT) with the generated cationic amine radical intermediate B to form the peroxyl alcohol species D. Subsequently, the rearrangement of intermediate D occurs to produce intermediate E, which is further hydrolyzed to yield the final oxygenation product.
Fig. 7. Proposed EDA catalytic aerobic oxygenation mechanism.
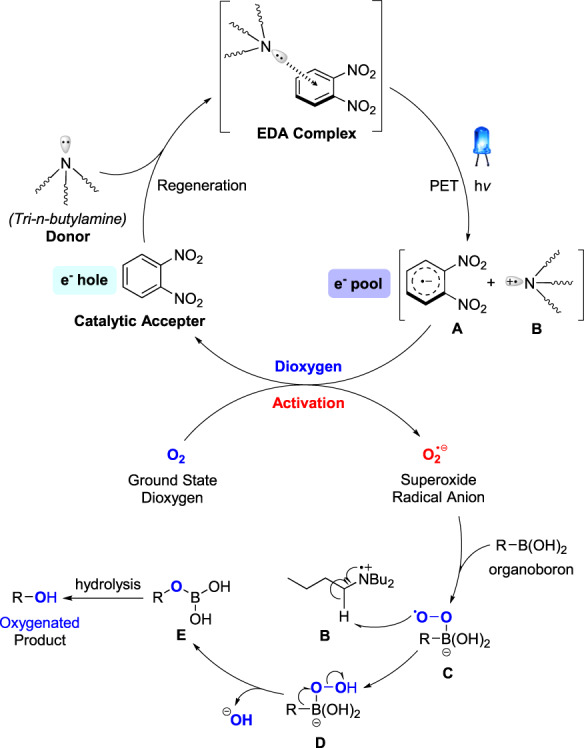
The reaction mechanism is divided into two main parts: the upper half involves the catalytic cycle of oxygen activation leading to the formation of superoxide, while the lower half describes the oxygenation process of organic boron substrates.
In conclusion, through rational mechanism design, we have developed an EDA complex photocatalytic system that overcomes the oxygen-sensitive limitation of traditional EDA photocatalytic systems. This system innovatively achieves oxygen activation under mild conditions without the participation of transition metals, photoredox catalysts, and electrochemical equipment. The present dioxygen activation and oxidation systems is mild, and highly compatible with different sensitive functional groups, such as thioethers, and aldehydes et. al. As a highly promising approach for dioxygen activation, this protocol is expected to have tremendous potential for a wide range of applications in oxygenation reactions.
Methods
General procedure for aerobic oxygenation of boric acid
To a dry 25 mL Schlenk tube, 0.5 mmol of boronic acid (if solid) and 4.3 mg (5 mol%) of 1,2-dinitrobenzene (1,2-DNB) were added. The tube was evacuated under reduced pressure and refilled with oxygen (3 times). Next, 120 μL of tri-n-butylamine (0.5 mmol, 1.0 eq) was added as a donor to the Schlenk tube. Anhydrous MeCN (4.0 mL) was then added using a syringe. The mixture was stirred at room temperature for 48 h under irradiation with blue LED (450 nm). After completion of the reaction, the solvent was removed under vacuum and the crude products were purified by column chromatography. For complete experimental details, including Photochemical instrumentation, related detection, procedures and full characterization (1H and 13C NMR, HRMS spectrometry) of all new compounds, see Supplementary Information.
Supplementary information
Acknowledgements
The authors acknowledge the National Key R&D Program of China (No. 2021YFA1501700), the NSFC (Nos. 22293014, 22131002, 22161142019), Changping Laboratory, the New Cornerstone Science Foundation through the New Cornerstone Investigator Program and the XPLORER PRIZE for financial support. We thank Licheng Yang in this group for reproducing the results of 2c, 2f and 2r.
Author contributions
J.W., and N.J., conceived and designed the experiments; J.W. and J.M. carried out most of experiments; J.W., J.M., C.Z., Y. L., and N.J. analyzed data; J.W., Y. L. and N.J., wrote the paper; N.J. directed the project.
Peer review
Peer review information
Nature Communications thanks Tian Qin and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Data availability
The authors declare that the data supporting the findings of this study are available within the paper or its Supplementary Information files and from the corresponding author upon request.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
The online version contains supplementary material available at 10.1038/s41467-024-45866-z.
References
- 1.Großkopf J, Kratz T, Rigotti T, Bach T. Enantioselective Photochemical Reactions Enabled by Triplet Energy Transfer. Chem. Rev. 2022;122:1626–1653. doi: 10.1021/acs.chemrev.1c00272. [DOI] [PubMed] [Google Scholar]
- 2.Prier CK, Rankic DA, MacMillan DWC. Visible Light Photoredox Catalysis with Transition Metal Complexes: Applications in Organic Synthesis. Chem. Rev. 2013;113:5322–5363. doi: 10.1021/cr300503r. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Stephenson, C. R. J., Yoon, T. P. & MacMillan, D. W. C., (eds.) Visible Light Photocatalysis in Organic Chemistry (Wiley-VCH: Weinheim, 2018).
- 4.Schultz DM, Yoon TP. Solar Synthesis: Prospects in Visible Light Photocatalysis. Science. 2014;343:1239176. doi: 10.1126/science.1239176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Mulliken RS. Molecular Compounds and their Spectra. II. J. Am. Chem. Soc. 1952;74:811–824. doi: 10.1021/ja01123a067. [DOI] [Google Scholar]
- 6.Crisenza GE, Mazzarella D, Melchiorre P. Synthetic Methods Driven by the Photoactivity of Electron Donor-Acceptor Complexes. J. Am. Chem. Soc. 2020;142:5461–5476. doi: 10.1021/jacs.0c01416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Arceo E, Jurberg ID, Álvarez-Fernández A, Melchiorre P. Photochemical Activity of a Key Donor−Acceptor Complex can Drive Stereoselective Catalytic α-Alkylation of Aldehydes. Nat. Chem. 2013;5:750–756. doi: 10.1038/nchem.1727. [DOI] [PubMed] [Google Scholar]
- 8.Tobisu M, Furukawa T, Chatani N. Visible Light-mediated Direct Arylation of Arenes and Heteroarenes Using Diaryliodonium Salts in the Presence and Absence of a Photocatalyst. Chem. Lett. 2013;42:1203–1205. doi: 10.1246/cl.130547. [DOI] [Google Scholar]
- 9.Wu J, Bar RM, Guo L, Noble A, Aggarwal VK. Photoinduced Deoxygenative Borylations of Aliphatic Alcohols. Angew. Chem. Int. Ed. 2019;58:18830–18834. doi: 10.1002/anie.201910051. [DOI] [PubMed] [Google Scholar]
- 10.Davies J, Booth SG, Essafi S, Dryfe RAW, Leonori D. Visible-Light-Mediated Generation of Nitrogen-Centered Radicals: Metal-Free Hydroimination and Iminohydroxylation Cyclization Reactions. Angew. Chem. Int. Ed. 2015;54:14017–14021. doi: 10.1002/anie.201507641. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Xie S, Li D, Huang H, Zhang F, Chen Y. Intermolecular Radical Addition to Ketoacids Enabled by Boron Activation. J. Am. Chem. Soc. 2019;141:16237–16242. doi: 10.1021/jacs.9b09099. [DOI] [PubMed] [Google Scholar]
- 12.Jung S, Shin S, Park S, Hong S. Visible-Light-Driven C4-Selective Alkylation of Pyridinium Derivatives with Alkyl Bromides. J. Am. Chem. Soc. 2020;142:11370–11375. doi: 10.1021/jacs.0c04499. [DOI] [PubMed] [Google Scholar]
- 13.Morack T, Mück-Lichtenfeld C, Gilmour R. Bioinspired Radical Stetter Reaction: Radical Umpolung Enabled by Ion-Pair Photocatalysis. Angew. Chem. Int. Ed. 2019;58:1208–1212. doi: 10.1002/anie.201809601. [DOI] [PubMed] [Google Scholar]
- 14.Hsu C-W, Sundén H. Aerobic Oxidative EDA Catalysis: Synthesis of Tetrahydro-quinolines Using an Organocatalytic EDA Active Acceptor. J. Org. Chem. 2022;87:1457–1469. doi: 10.1021/acs.joc.1c02776. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Guo W, et al. Synthesis of Purine Analogues: Photocatalyst-Free Visible-Light-Enhanced Annulation Approach to Pyrazolo[1,5-a][1,3,5]triazine-2,4-diamines. J. Org. Chem. 2021;86:8365–8380. doi: 10.1021/acs.joc.1c00783. [DOI] [PubMed] [Google Scholar]
- 16.Liu B, Lim C-H, Miyake GM. Visible-Light-Promoted C−S Cross-Coupling Via Intermolecular Charge Transfer. J. Am. Chem. Soc. 2017;139:13616–13619. doi: 10.1021/jacs.7b07390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Lima CGS, et al. Organic Synthesis Enabled by Light-Irradiation of EDA Complexes: Theoretical Background and Synthetic Applications. ACS Catal. 2016;6:1389–1407. doi: 10.1021/acscatal.5b02386. [DOI] [Google Scholar]
- 18.Saxena B, Patel RI, Sharma A. Recent Advances in Electron Donor-Acceptor (EDA)-Complex Reactions involving Quaternary Pyridinium Derivatives. Adv. Synth. Catal. 2023;365:1538–1564. doi: 10.1002/adsc.202300078. [DOI] [Google Scholar]
- 19.van Dalsen L, Brown RE, Rossi-Ashton JA, Procter DJ. Sulfonium Salts as Acceptors in Electron Donor-Acceptor Complexes. Angew. Chem. Int. Ed. 2023;62:e202303104. doi: 10.1002/anie.202303104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Tasnim T, Ayodele MJ, Pitre SP. Recent Advances in Employing Catalytic Donors and Acceptors in Electron Donor−Acceptor Complex Photochemistry. J. Org. Chem. 2022;87:10555–10563. doi: 10.1021/acs.joc.2c01013. [DOI] [PubMed] [Google Scholar]
- 21.Li H, Liu Y, Chiba S. Leveraging of Sulfur Anions in Photoinduced Molecular Transformations. Jacs. Au. 2021;1:2121–2129. doi: 10.1021/jacsau.1c00363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Fu M-C, Shang R, Zhao B, Wang B, Fu Y. Photocatalytic Decarboxylative Alkylations Mediated by Triphenylphosphine and Sodium Iodide. Science. 2019;363:1429–1434. doi: 10.1126/science.aav3200. [DOI] [PubMed] [Google Scholar]
- 23.Biegasiewicz KF, et al. Photoexcitation of Flavoenzymes Enables a Stereoselective Radical Cyclization. Science. 2019;364:1166–1169. doi: 10.1126/science.aaw1143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Huang X, et al. Photoenzymatic Enantioselective Intermolecular Radical Hydroalkylation. Nature. 2020;584:69–74. doi: 10.1038/s41586-020-2406-6. [DOI] [PubMed] [Google Scholar]
- 25.de Pedro Beato E, Spinnato D, Zhou W, Melchiorre P. A General Organocatalytic System for Electron Donor−Acceptor Complex Photoactivation and Its Use in Radical Processes. J. Am. Chem. Soc. 2021;143:12304–12314. doi: 10.1021/jacs.1c05607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Wei Z, Wu S, Melchiorre P. Tetrachlorophthalimides as Organocatalytic Acceptors for Electron Donor−Acceptor Complex Photoactivation. J. Am. Chem. Soc. 2022;144:8914–8919. doi: 10.1021/jacs.2c03546. [DOI] [PubMed] [Google Scholar]
- 27.Adam W, Platsch H, Wirz J. Oxygen trapping and thermochemistry of a hydrocarbon singlet biradical: 1,3-diphenylcyclopentane-1,3-diyl. J. Am. Chem. Soc. 1989;111:6896–6898. doi: 10.1021/ja00199a091. [DOI] [Google Scholar]
- 28.Reddy AR, Bendikov M. Diels–Alder reaction of acenes with singlet and triplet oxygen-theoretical study of two-state reactivity. Chem. Commun. 2006;11:1179–1181. doi: 10.1039/b513597d. [DOI] [PubMed] [Google Scholar]
- 29.Leifert D, Studer A. The Persistent Radical Effect in Organic Synthesis. Angew. Chem. Int. Ed. 2020;59:74–108. doi: 10.1002/anie.201903726. [DOI] [PubMed] [Google Scholar]
- 30.Rosenthal J, Nocera DG. Role of Proton-Coupled Electron Transfer in O–O Bond Activation. Acc. Chem. Res. 2007;40:543–553. doi: 10.1021/ar7000638. [DOI] [PubMed] [Google Scholar]
- 31.Bahamonde A, Melchiorre P. Mechanism of the Stereoselective α-Alkylation of Aldehydes Driven by the Photochemical Activity of Enamines. J. Am. Chem. Soc. 2016;138:8019–8030. doi: 10.1021/jacs.6b04871. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Fawcett A, et al. Photoinduced decarboxylative borylation of carboxylic acids. Science. 2017;357:283–286. doi: 10.1126/science.aan3679. [DOI] [PubMed] [Google Scholar]
- 33.Ghosh I, Ghosh T, Bardagi JI, König B. Reduction of aryl halides byconsecutive visible light-induced electron transfer processes. Science. 2014;346:725–728. doi: 10.1126/science.1258232. [DOI] [PubMed] [Google Scholar]
- 34.Murphy JJ, Bastida D, Paria S, Fagnoni M, Melchiorre P. Asymmetric catalytic formation of quaternary carbons by iminium ion trapping of radicals. Nature. 2016;532:218–222. doi: 10.1038/nature17438. [DOI] [PubMed] [Google Scholar]
- 35.Sandanayaka ASD, Ito O. Photoinduced electron transfer in supramolecules composed of porphyrin/phthalocyanine and nanocarbon materials. J. Porphyr. Phthalocyanines. 2009;13:1017–1033. doi: 10.1142/S1088424609001388. [DOI] [Google Scholar]
- 36.Liang Y-F, Jiao N. Oxygenation via C−H/C−C Bond Activation with Molecular Oxygen. Acc. Chem. Res. 2017;50:1640–1653. doi: 10.1021/acs.accounts.7b00108. [DOI] [PubMed] [Google Scholar]
- 37.Zhang C, Feng P, Jiao N. Cu-Catalyzed Esterification Reaction via Aerobic Oxygenation and C−C Bond Cleavage: An Approach to α-Ketoesters. J. Am. Chem. Soc. 2013;135:15257–15262. doi: 10.1021/ja4085463. [DOI] [PubMed] [Google Scholar]
- 38.Wang T, Jiao N. TEMPO-Catalyzed Aerobic Oxygenation and Nitrogenation of Olefins via C=C Double Bond Cleavage. J. Am. Chem. Soc. 2013;135:11692–11695. doi: 10.1021/ja403824y. [DOI] [PubMed] [Google Scholar]
- 39.Liang Y-F, Jiao N. Highly Efficient C−H Hydroxylation of Carbonyl Compounds with Oxygen under Mild Conditions. Angew. Chem. Int. Ed. 2014;53:548–552. doi: 10.1002/anie.201308698. [DOI] [PubMed] [Google Scholar]
- 40.Shen T, Zhang Y, Liang Y-F, Jiao N. Direct Tryptophols Synthesis from 2-Vinylanilines and Alkynes via C-C Triple Bond Cleavage and Dioxygen Activation. J. Am. Chem. Soc. 2016;138:13147–13150. doi: 10.1021/jacs.6b08094. [DOI] [PubMed] [Google Scholar]
- 41.Liang Y, et al. Electrochemically induced Nickel Catalysis for Oxygenation Reactions with Water. Nat. Catal. 2021;4:116–123. doi: 10.1038/s41929-020-00559-w. [DOI] [Google Scholar]
- 42.Sigman MS, Jensen DR. Ligand-Modulated Palladium-Catalyzed Aerobic Alcohol Oxidations. Acc. Chem. Res. 2006;39:221–229. doi: 10.1021/ar040243m. [DOI] [PubMed] [Google Scholar]
- 43.Punniyamurthy T, Velusamy S, Iqbal J. Recent Advances in Transition Metal Catalyzed Oxidation of Organic Substrates with Molecular Oxygen. Chem. Rev. 2005;105:2329–2363. doi: 10.1021/cr050523v. [DOI] [PubMed] [Google Scholar]
- 44.Wendlandt AE, Suess AM, Stahl SS. Copper-Catalyzed Aerobic Oxidative C−H Functionalizations: Trends and Mechanistic Insights. Angew. Chem. Int. Ed. 2011;50:11062–11087. doi: 10.1002/anie.201103945. [DOI] [PubMed] [Google Scholar]
- 45.Wu W, Jiang H. Palladium-Catalyzed Oxidation of Unsaturated Hydrocarbons Using Molecular Oxygen. Acc. Chem. Res. 2012;45:1736–1748. doi: 10.1021/ar3000508. [DOI] [PubMed] [Google Scholar]
- 46.Allen SE, Walvoord RR, Padilla-Salinas R, Kozlowski MC. Aerobic Copper-Catalyzed Organic Reactions. Chem. Rev. 2013;113:6234–6458. doi: 10.1021/cr300527g. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Serrano-Plana J, Garcia-Bosch I, Company A, Costas M. Structural and Reactivity Models for Copper Oxygenases: Cooperative Effects and Novel Reactivities. Acc. Chem. Res. 2015;48:2397–2406. doi: 10.1021/acs.accounts.5b00187. [DOI] [PubMed] [Google Scholar]
- 48.Boisvert L, Goldberg KI. Reactions of Late Transition Metal Complexes with Molecular Oxygen. Acc. Chem. Res. 2012;45:899–910. doi: 10.1021/ar2003072. [DOI] [PubMed] [Google Scholar]
- 49.Roduner E, et al. Selective Catalytic Oxidation of C−H Bonds with Molecular Oxygen. ChemCatChem. 2013;5:82–112. doi: 10.1002/cctc.201200266. [DOI] [Google Scholar]
- 50.Bonesi SM, Fagnoni M, Albini A. Hammett Correlations in the Photosensitized Oxidation of 4-Substituted Thioanisoles. J. Org. Chem. 2004;69:928–935. doi: 10.1021/jo035679e. [DOI] [PubMed] [Google Scholar]
- 51.Ohkubo K, Kobayashi T, Fukuzumi S. Direct Oxygenation of Benzene to Phenol Using Quinoinium Ions as Homogeneous Photocatalysts. Angew. Chem. Int. Ed. 2011;50:8652–8655. doi: 10.1002/anie.201102931. [DOI] [PubMed] [Google Scholar]
- 52.Yadav AK, Yadav LDS. Visible-light-mediated difunctionalization of styrenes: an unprecedented approach to 5-aryl-2-imino-1,3-oxathiolanes. Green. Chem. 2015;17:3515–3520. doi: 10.1039/C5GC00642B. [DOI] [Google Scholar]
- 53.Li Y, et al. Selective Late-Stage Oxygenation of Sulfides with Ground-State Oxygen by Uranyl Photocatalysis. Angew. Chem. Int. Ed. 2019;58:13499–13506. doi: 10.1002/anie.201906080. [DOI] [PubMed] [Google Scholar]
- 54.Lasky MR, et al. Photochemical C(sp2)−H Pyridination via Arene−Pyridinium Electron Donor−Acceptor Complexes. Angew. Chem. Int. Ed. 2022;61:e202208741. doi: 10.1002/anie.202208741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Huang Z, et al. Oxidative Cleavage of Alkenes by O2 with a Non-Heme Manganese Catalyst. J. Am. Chem. Soc. 2021;143:10005–10013. doi: 10.1021/jacs.1c05757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Zou Y-Q, et al. Highly Efficient Aerobic Oxidative Hydroxylation of Arylboronic Acids: Photoredox Catalysis Using Visible Light. Angew. Chem. Int. Ed. 2012;51:784–788. doi: 10.1002/anie.201107028. [DOI] [PubMed] [Google Scholar]
- 57.McClain EJ, Monos TM, Mori M, Beatty JW, Stephenson CRJ. Design and Implementation of a Catalytic Electron Donor−Acceptor Complex Platform for Radical Trifluoromethylation and Alkylation. ACS Catal. 2020;10:12636–12641. doi: 10.1021/acscatal.0c03837. [DOI] [Google Scholar]
- 58.Runemark A, Sundén H. Overcoming Back Electron Transfer in the Electron Donor−Acceptor Complex-Mediated Visible Light-Driven Generation of α‑Aminoalkyl Radicals from Secondary Anilines. J. Org. Chem. 2023;88:462–474. doi: 10.1021/acs.joc.2c02448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Zhang Y, Dai M, Yuan Z. Methods for the detection of reactive oxygen species. Anal. Methods. 2018;10:4625–4638. doi: 10.1039/C8AY01339J. [DOI] [Google Scholar]
- 60.Singh SK, Husain SM. A Redox-Based Superoxide Generation System Using Quinone/Quinone Reductase. ChemBioChem. 2018;19:1657–1663. doi: 10.1002/cbic.201800071. [DOI] [PubMed] [Google Scholar]
- 61.Gogoi A, Bora U. A mild and efficient protocol for the ipso-hydroxylation of arylboronic acids. Tetrahedron Lett. 2013;54:1821–1823. doi: 10.1016/j.tetlet.2013.01.079. [DOI] [Google Scholar]
- 62.Gupta S, Chaudhary P, Srivastava V, Kandasamy J. A chemoselective ipso-hydroxylation of arylboronic acids using urea-hydrogen peroxide under catalyst free condition. Tetrahedron Lett. 2016;57:2506–2510. doi: 10.1016/j.tetlet.2016.04.099. [DOI] [Google Scholar]
- 63.Molloy JJ, et al. Chemoselective oxidation of aryl organoboron systems enabled by boronic acid-selective phase transfer. Chem. Sci. 2017;8:1551–1559. doi: 10.1039/C6SC04014D. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Molander GA, Cavalcanti LN. Oxidation of organotrifluoroborates via oxone. J. Org. Chem. 2011;76:623–630. doi: 10.1021/jo102208d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Singh JO, Anunziata JD, Silber JJ. n-π Electron Donor-Acceptor Complexes. II. Aliphatic Amines with Dinitrobenzenes. Can. J. Chem. 1985;63:903–907. doi: 10.1139/v85-150. [DOI] [Google Scholar]
- 66.Glukhovtsev MN, Bach RD, Laiter S. Single-Step and Multistep Mechanisms of Aromatic Nucleophilic Substitution of Halobenzenes and Halonitrobenzenes with Halide Anions: Ab Initio Computational Study. J. Org. Chem. 1997;62:4036–4046. doi: 10.1021/jo962096e. [DOI] [Google Scholar]
- 67.Cismesiaa, M. A. & Yoon, T. P. Characterizing chain processes in visible light photoredox catalysis. Chem. Sci.6, 5426–5434 (2015). [DOI] [PMC free article] [PubMed]
- 68.Hu, J., Wang, J., Nguyen, T. H. & Zheng, N. Beilstein J. The chemistry of amine radical cations produced by visible light photoredox catalysis. Org. Chem.9, 1977–2001 (2013). [DOI] [PMC free article] [PubMed]
- 69.Yamamoto, Y., Kodama, S., Nomotoa, A. & Ogawa, A. Innovative green oxidation of amines to imines under atmospheric oxygen. Org. Biomol. Chem.20, 9503–9521 (2022). [DOI] [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The authors declare that the data supporting the findings of this study are available within the paper or its Supplementary Information files and from the corresponding author upon request.