Abstract
Background
Immunohistochemistry for p53 was a well-established method for cancer diagnosis in pathology. Aberrant cytoplasmic p53 positivity reflects the accumulation of p53 aggregates, which has been shown to be associated with chemoresistance and to be a predictive marker of a worse clinical course in ovarian cancer.
Case Report
A 65-year-old Japanese man was diagnosed with lung cancer, and surgical resection was performed. Multiple metastasis were found 21 months post-surgery. The lesions were resistant to chemotherapy, and he succumbed to the disease 29 months post-surgery. The resected primary lesion was pathologically diagnosed as squamous cell carcinoma, with notable cytoplasmic p53 positivity indicated by immunohistochemistry.
Conclusion
Notable aberrant cytoplasmic accumulation of p53 aggregate was observed in the cancer cells of this case. Chemotherapy was ineffective for the recurrent lesions, suggesting a role of p53 aggregates in chemoresistance. Pathological analysis of p53 via immunohistochemistry may be useful in predicting chemoresistance of lung squamous cell carcinoma.
Keywords: Lung, squamous cell carcinoma, p53 aggregation, aggressive
Lung cancer is one of the leading causes of cancer-related deaths worldwide, and its incidence is currently increasing (1). Lung squamous cell carcinoma (LUSC), a major histological subtype of lung cancer, is often linked to exposure to tobacco smoke, radon, and air pollution (2). While immunotherapies targeting immune checkpoint signals have become standard treatments for lung cancer, many advanced lung cancer cases remain resistant to these therapies (3). Targeted therapies against various driver mutations have been developed for lung adenocarcinoma, yet no common driver mutations have been identified in LUSC (4). As a result, treatment options for patients with advanced LUSC are more limited compared to other histological subtypes.
Tumor protein p53, which is known as p53, plays critical roles in DNA damage response and is often referred as the “guardian of the genome” (5). Mutations in the TP53 gene are thought to contribute to tumorigenesis by impairing its tumor-suppressive functions and enabling malignancy through gain-of-oncogenic functions (6). The expression of intracytoplasmic p53 has recently garnered interest in ovarian cancer research, with abnormal cytoplasmic staining of p53 observed in 2.3% of ovarian serous carcinoma (7). Our previous studies have shown that mutant p53 proteins form aggregates and that cytoplasmic p53 aggregates are associated with poor prognosis in human ovarian cancer (8). In the present case of LUSC, we observed notable aberrant p53 positivity in the cytoplasm of cancer cells during pathological analysis. The case was clinically aggressive, and p53 aggregates were suggested to play a role in this aggressive phenotype.
Case Report
A 65-year-old Japanese man was referred for surgical treatment following the detection of lung cancer during a routine health check-up. His medical history included hypertension, dyslipidemia, and diabetes. Additionally, clinical stage I laryngeal cancer was identified during the evaluation for lung cancer. He was a heavy smoker, consumed moderate amounts of alcohol daily, and had no significant history of occupational dust exposure. His family history was notable for rectal cancer in his father.
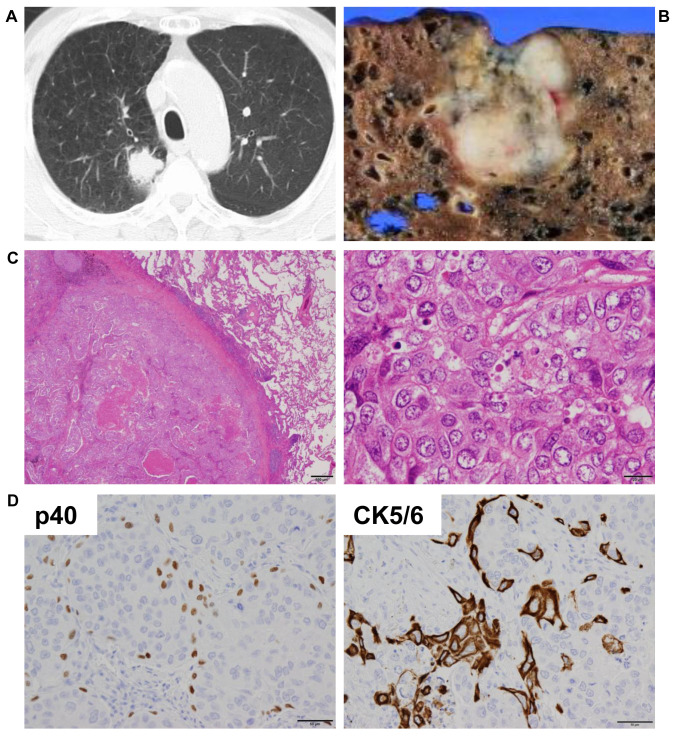
Computed tomography (CT) scans revealed a 3.3 cm mass in the upper lobe of the right lung, set against a background of emphysema and interstitial shadows (Figure 1). Squamous cell carcinoma was suspected following a transbronchial biopsy of the lesion. Imaging studies did not reveal any obvious metastatic lesions. He subsequently underwent a thoracoscopic right upper lobectomy with lymph node dissection.
Figure 1. Radiological and pathological observations. A) Computed tomography scans revealed a mass in the upper lobe of the right lung. B) Macroscopic observation revealed the mass was well-circumscribed, white, and exhibited areas of necrosis. C) Hematoxylin and eosin staining shows that the lung lesion is composed of polygonal cancer cells with abundant eosinophilic cytoplasm. D) Immunohistochemically, the cancer cells were focally positive for p40 (clone BC28, Nichirei, Tokyo, Japan) and CK5/6 (clone D5/16 B4, Nichirei). Scale bars: 20 μm.
Macroscopically, the resected mass measured 20×18 mm, was well-circumscribed, white, and exhibited areas of necrosis. Microscopic examination showed that the mass consisted of polygonal cancer cells with abundant eosinophilic cytoplasm and prominent nucleoli, arranged in solid nests. There was no evident keratinization, intercellular bridges, glandular structures, or intracytoplasmic mucin.
Immunohistochemically, the tumor cells were focally positive for p40 and CK5/6, but negative for TTF-1. This led to a diagnosis of non-keratinizing squamous cell carcinoma. The pathological stage of the lung cancer was determined to be pT2a, pN0, cM0, stage IB, according to the Union for International Cancer Control/American Joint Committee on Cancer 8th edition criteria, primary due to the presence of visceral pleural invasion.
The postoperative course was uneventful; however, 21 months after the surgery, he experienced an exacerbation of anorexia and dyspnea due to massive pleural effusion on the right side. CT demonstrated thickening of the right pleura, a mass causing right lung atelectasis, and enlarged mediastinal lymph nodes. Fluorodeoxyglucose (FDG)-positron emission tomography revealed high FDG accumulation in all these areas, strongly suggesting the recurrence of lung cancer. He underwent four cycles of carboplatin and pemetrexed chemotherapy within four months and then proceeded to pemetrexed maintenance therapy. However, he developed pneumonia, which was likely induced by pemetrexed. Given his decreased physical strength and the limited benefits of continuing chemotherapy, he transitioned to outpatient palliative care. He died from the lung cancer 29 months after surgery.
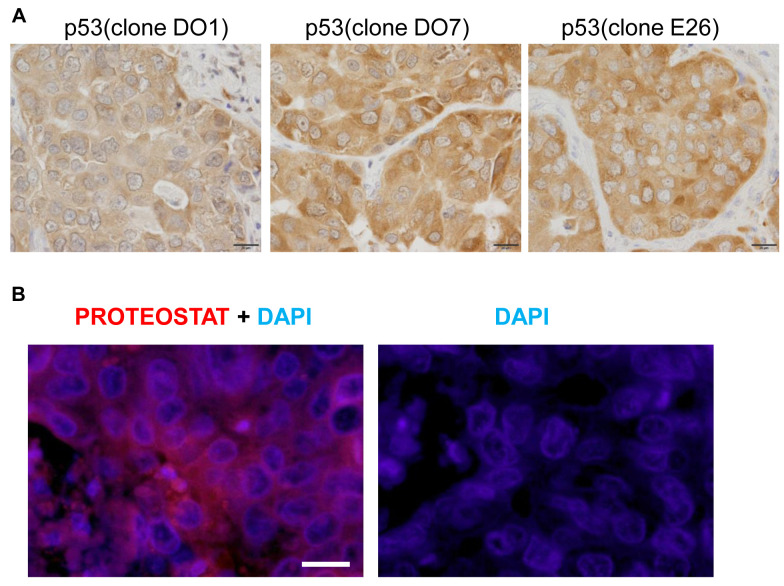
Additional pathological examinations. Notably, strong cytoplasmic p53 staining was found, whereas nuclear p53 staining was weak (Figure 2A). PROTEOSTAT Protein aggregation assay (Enzo Biochem, Farmingdale, NY, USA) was performed using the same paraffin section, and cytoplasmic positive staining was observed (Figure 2B).
Figure 2. p53 protein expression analyses. A) Immunohistochemistry of p53 was performed using three monoclonal antibodies (clone DO1 and DO7, DAKO, Glostrup, Denmark; clone E26, Abcam, Cambridge, UK), and the same observations were obtained. B) The PROTEOSTAT Protein aggregation assay was performed on a paraffin section. Scale bars: 20 μm.
Discussion
It is well-known that TP53 mutations are detected in many malignant tumors, and pathological observation of excessive p53 protein accumulation in the nucleus corresponds well with TP53 mutations (9). Nonsynonymous TP53 mutations suppress p53 ubiquitination and degradation, which are induced by MDM2 (10). In colorectal cancer, cytoplasmic p53 staining has been observed in cases with a high clinical stage, correlating with a worse clinical course (11,12). TP53 missense mutation have been detected in colorectal cancer cases that exhibit cytoplasmic p53 staining (13). However, there was no report on cytoplasmic p53 staining in lung cancer. Therefore, we aimed to determine whether p53 aggregates were present in lung cancers using our tissue array set, as described previously (14). We identified a case of LUSC with clear cytoplasmic p53 staining, which we report here. This case exhibited aggressive behavior and resistance to chemotherapy, consistent with observations in previous studies.
The mechanism by which p53 aggregates in the cytoplasm has not yet been clarified. One proposed mechanism is that p53 proteins released from cancer cells aggregate in the extracellular space through sulfated glycosaminoglycans, followed by cancer cell uptake of these aggregated p53 proteins (15). Cytoplasmic p53 aggregates have been suggested as a target for anti-cancer therapy. The peptide-based p53 aggregation inhibitor, ReACp53, has been shown to enhance the anti-cancer efficacy of carboplatin in ovarian cancer cells (16). A compound named ADH-6 has been shown to abrogate p53 aggregation and induce apoptosis in cancer cells with TP53 mutations (17).
In conclusion, p53 aggregation may play a role in the chemoresistance of advanced LUSC. Pathological analysis of p53 via immunohistochemistry may predict chemoresistance of LUSC. Therapies targeting p53 aggregation have been developed, and further studies are necessary to determine their effectiveness against LUSC.
Conflicts of Interest
All Authors have no conflicts of interest to declare in relation to this study.
Authors’ Contributions
HY, RY, YK, RM, and KN gathered the patient’s data and wrote the article. RY and YK were responsible for pathological diagnosis of this case. HY, YF, KI, and SS discussed the data and helped write the article. All Authors approved the final article.
Acknowledgements
Immunohistochemistry was carried out by K. I. Steinar Inc. (Kumamoto, Japan).
References
- 1.Barta JA, Powell CA, Wisnivesky JP. Global epidemiology of lung cancer. Ann Glob Health. 2019;85(1):8. doi: 10.5334/aogh.2419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Dela Cruz CS, Tanoue LT, Matthay RA. Lung cancer: epidemiology, etiology, and prevention. Clin Chest Med. 2011;32(4):605–644. doi: 10.1016/j.ccm.2011.09.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Forde PM, Spicer J, Lu S, Provencio M, Mitsudomi T, Awad MM, Felip E, Broderick SR, Brahmer JR, Swanson SJ, Kerr K, Wang C, Ciuleanu TE, Saylors GB, Tanaka F, Ito H, Chen KN, Liberman M, Vokes EE, Taube JM, Dorange C, Cai J, Fiore J, Jarkowski A, Balli D, Sausen M, Pandya D, Calvet CY, Girard N, CheckMate 816 Investigators Neoadjuvant nivolumab plus chemotherapy in resectable lung cancer. N Engl J Med. 2022;386(21):1973–1985. doi: 10.1056/NEJMoa2202170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Lau SCM, Pan Y, Velcheti V, Wong KK. Squamous cell lung cancer: Current landscape and future therapeutic options. Cancer Cell. 2022;40(11):1279–1293. doi: 10.1016/j.ccell.2022.09.018. [DOI] [PubMed] [Google Scholar]
- 5.Huang CL, Yokomise H, Miyatake A. Clinical significance of the p53 pathway and associated gene therapy in non-small cell lung cancers. Future Oncol. 2007;3(1):83–93. doi: 10.2217/14796694.3.1.83. [DOI] [PubMed] [Google Scholar]
- 6.Komiya T, Hirashima T, Kawase I. Clinical significance of p53 in non-small-cell lung cancer. Oncol Rep. 1999;6(1):19–28. [PubMed] [Google Scholar]
- 7.Köbel M, Piskorz AM, Lee S, Lui S, LePage C, Marass F, Rosenfeld N, Mes Masson AM, Brenton JD. Optimized p53 immunohistochemistry is an accurate predictor of TP53 mutation in ovarian carcinoma. J Pathol Clin Res. 2016;2(4):247–258. doi: 10.1002/cjp2.53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Iwahashi N, Ikezaki M, Komohara Y, Fujiwara Y, Noguchi T, Nishioka K, Sakai K, Nishio K, Ueda M, Ihara Y, Uchimura K, Ino K, Nishitsuji K. Cytoplasmic p53 aggregates accumulated in p53-mutated cancer correlate with poor prognosis. PNAS Nexus. 2022;1(3):pgac128. doi: 10.1093/pnasnexus/pgac128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.McManus DT, Yap EP, Maxwell P, Russell SE, Toner PG, McGee JO. p53 Expression, mutation, and allelic deletion in ovarian cancer. J Pathol. 1994;174(3):159–168. doi: 10.1002/path.1711740304. [DOI] [PubMed] [Google Scholar]
- 10.Nenutil R, Smardova J, Pavlova S, Hanzelkova Z, Muller P, Fabian P, Hrstka R, Janotova P, Radina M, Lane DP, Coates PJ, Vojtesek B. Discriminating functional and non-functional p53 in human tumours by p53 and MDM2 immunohistochemistry. J Pathol. 2005;207(3):251–259. doi: 10.1002/path.1838. [DOI] [PubMed] [Google Scholar]
- 11.Bosari S, Viale G, Bossi P, Maggioni M, Coggi G, Murray JJ, Lee AKC. Cytoplasmic accumulation of p53 protein: an independent prognostic indicator in colorectal adenocarcinomas. J Natl Cancer Inst. 1994;86(9):681–687. doi: 10.1093/jnci/86.9.681. [DOI] [PubMed] [Google Scholar]
- 12.Sun XF, Carstensen JM, Zhang H, Stål O, Wingren S, Hatschek T, Nordenskjöld B. Prognostic significance of cytoplasmic p53 oncoprotein in colorectal adenocarcinoma. Lancet. 1992;340(8832):1369–1373. doi: 10.1016/0140-6736(92)92558-w. [DOI] [PubMed] [Google Scholar]
- 13.Jansson A, Gentile M, Sun XF. p53 Mutations are present in colorectal cancer with cytoplasmic p53 accumulation. Int J Cancer. 2001;92(3):338–341. doi: 10.1002/ijc.1189. [DOI] [PubMed] [Google Scholar]
- 14.Shinchi Y, Komohara Y, Yonemitsu K, Sato K, Ohnishi K, Saito Y, Fujiwara Y, Mori T, Shiraishi K, Ikeda K, Suzuki M. Accurate expression of PD-L1/L2 in lung adenocarcinoma cells: A retrospective study by double immunohistochemistry. Cancer Sci. 2019;110(9):2711–2721. doi: 10.1111/cas.14128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Iwahashi N, Ikezaki M, Nishikawa T, Namba N, Ohgita T, Saito H, Ihara Y, Shimanouchi T, Ino K, Uchimura K, Nishitsuji K. Sulfated glycosaminoglycans mediate prion-like behavior of p53 aggregates. Proc Natl Acad Sci USA. 2020;117(52):33225–33234. doi: 10.1073/pnas.2009931117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Neal A, Lai T, Singh T, Rahseparian N, Grogan T, Elashoff D, Scott P, Pellegrini M, Memarzadeh S. Combining ReACp53 with carboplatin to target high-grade serous ovarian cancers. Cancers (Basel) 2021;13(23):5908. doi: 10.3390/cancers13235908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Palanikumar L, Karpauskaite L, Al-Sayegh M, Chehade I, Alam M, Hassan S, Maity D, Ali L, Kalmouni M, Hunashal Y, Ahmed J, Houhou T, Karapetyan S, Falls Z, Samudrala R, Pasricha R, Esposito G, Afzal AJ, Hamilton AD, Kumar S, Magzoub M. Protein mimetic amyloid inhibitor potently abrogates cancer-associated mutant p53 aggregation and restores tumor suppressor function. Nat Commun. 2021;12(1):3962. doi: 10.1038/s41467-021-23985-1. [DOI] [PMC free article] [PubMed] [Google Scholar]