Abstract
Anaerobic digestion is a suitable technology to treat cheese whey (CW), a high-strength wastewater from cheesemaking. However, CW anaerobic digestion is limited by its high biodegradability, acidic pH, and lack of alkalinity. This publication evaluated the acidification risk of CW anaerobic digestion under psychrophilic and mesophilic conditions, aiming to improve digester design, operation, and decision-making when facing instability periods. To evaluate the acidification risk of CW anaerobic digestion, biochemical methane potential (BMP) tests were carried out at four different organic loads, each under psychrophilic (20 °C) and mesophilic (35 °C) conditions. Besides methane production, pH, soluble chemical oxygen demand, volatile fatty acid and alcohols were also monitored. Experimental results showed that CW can be successfully degraded under both temperature conditions, with methane yields of 389–436 mLCH4/gVS. The organic load had a greater impact on the accumulation of intermediate products than temperature, indicating that process inhibition by overloading is plausible under psychrophilic and mesophilic conditions. However, the degradation rate under mesophilic conditions was faster than under psychrophilic conditions. Experimental results also revealed a higher imbalance between fermentation and methanogenesis rate under psychrophilic conditions, which resulted in higher concentrations of intermediate products (volatile fatty acids and alcohols) and prolonged lower pHs. These results indicate that the degradation of intermediate products is less favourable under psychrophilic conditions compared to mesophilic conditions. This implies that psychrophilic digesters have a lower capacity to recover from process disturbances, increasing the risk of process underperformance or even failure under psychrophilic conditions.
Keywords: Biogas, Acidogenic fermentation, Dairy industry, Inhibition, Volatile fatty acids, Temperature
1. Introduction
Cheese whey (CW) is the main by-product of cheesemaking with an average production of 9 L per kilogram of cheese produced [1]. Worldwide, it is estimated that only 50% of this by-product is upcycled into human or animal products such as food, feed and antioxidants [2]. In low- and middle-income countries, cheese whey is frequently discharged into the environment without treatment representing a source of pollution with a severe environmental impact [3,4]. CW classifies as high-strength wastewater due to its high organic matter (50–102 gCOD/L), nitrogen (0.01–1.7 g/L) and phosphorus (0.006–0.5 g/L) concentration [[5], [6], [7]].
Anaerobic digestion (AD) is a biological process able to transform organic matter into renewable energy in the form of methane-rich biogas and a nutrient-rich digestate suitable for land application as fertiliser or soil conditioner. AD is a mature technology to treat and valorise several organic waste streams and represents a relatively simple technological solution to minimise the environmental impacts of CW [3,8,9]. However, CW anaerobic digestion is limited by its intrinsic properties. Specifically, by its highly biodegradable organic matter content, acidic pH and lack of alkalinity [[10], [11], [12]]. These factors increase the likelihood of AD inhibition by acidification, i.e., failure associated to pH values below 6.5 and/or acute accumulation of volatile fatty acids (VFAs).
In most applications, CW is used as a co-substrate in anaerobic co-digestion (AcoD) processes [10,[13], [14], [15]]. AcoD is the combined treatment of different residues in a single digester to maximize biogas production and/or to centralize waste treatment [11,16,17]. CW can be co-digested with cattle manure (CM) since both residues can be produced in the same facility or nearby facilities. The benefit of co-digesting CM and CW relies on (i) the CM buffer capacity to sustain circumneutral pH values and prevent process inhibition by low pH, (ii) the CM macro-nutrient and trace elements to support the metabolic activity of the AD microbiome, and (iii) the high biodegradability of CW to boost biogas yields [11,14,18]. In AcoD processes, co-substrate addition improves the digester's methane production until a certain organic loading rate (OLR) limit is surpassed [19,20].
The maximum OLR of an anaerobic digester is determined by the digester's design and its operating conditions, which determine the process kinetics and conversion efficiency [21]. Temperature is one of the most important operating factors in AD because of its impact on biochemical (e.g. metabolic rates, enzymatic activity, microorganisms growth and decay rates) and physiochemical (e.g. mass transfer rates, gas solubility, chemical equilibrium) [[22], [23], [24]]. Indeed, for soluble substrates like CW, the effluent chemical oxygen demand (COD) concentration is determined by the sludge retention time (SRT) and the kinetic coefficients for biomass growth and decay [25]. Therefore, when the CW feeding rate exceeds the AD biomass capabilities for a specific SRT, the digester becomes overloaded and accumulates intermediate products such as VFAs.
Most of the CW anaerobic digestion research has focused on the methane yield of CW and its co-digestion with CM [10,14,26,27]. However, little attention has been paid to the acidification risk associated with CW anaerobic digestion or the impact of operating temperature on acidification risk. Ghaly et al. [12], and Treu et al. [15], showed the instability of CW mono-digestion under mesophilic and thermophilic conditions caused by the accumulation of VFAs and a concomitant pH drop. Mesophilic and thermophilic anaerobic digestion have been extensively studied and compared, including the development of semi-empirical models in these temperature regimes, which study different variables (e.g. the cosubstrate and the inoculum) [28,29]. However, more research into psychrophilic conditions is required because many full-scale digesters are operated under psychrophilic conditions, including covered anaerobic lagoons and tubular bioreactors [24,30]. Research under psychrophilic conditions is also important to facilitate the implementation of full-scale digesters in the Global South, which lacks the size and economic resources/incentives to heat the digesters. Understanding how temperature affects AD capacity is critical to improving digester design, operation and return on investment for anaerobic (co-)digestion applications. Understanding the effect of temperature on AD capacity is also important to make educated decisions when facing instability periods, as knowledge gained from mesophilic and thermophilic research may not be directly transferable to digesters operating under psychrophilic conditions.
The goal of this research was to evaluate and compare the acidification risk of CW anaerobic digestion under psychrophilic and mesophilic conditions at different organic loads. The aim was to understand the inhibition behaviour at different CW concentrations, which would provide useful knowledge to optimise CW anaerobic digestion (or other highly biodegradable substrates) as well as adequately handle instability periods. To achieve this, biochemical methane potential (BMP) assays were carried out at four different CW organic loads at both 20 and 35 °C.
2. Materials and methods
2.1. Cheese whey and inoculum origin
CW was collected in Rancho Los Álamos (Ubaté, Cundinamarca Province, Colombia), which is in the subtropical region of Colombia. The farm has 700 cows to produce cheese, yoghurt, and other dairy products. The farm produces between 7000 and 7150 L of milk per day throughout the year, of which 1000 and 1200 L are used for cheese production.
Cheese production takes place five days a week. Each day a different type of cheese is produced (e.g., provolone, mozzarella, and cheddar). As a result of cheesemaking, between 800 and 1000 L of CW are generated daily. To obtain a CW representative sample, 1 L of CW were collected and cooled (4 °C) each day for 5 consecutive days. Subsequently, the CW was freeze-dried in a lyophilizer (Labconco 753,022) and vacuum packed. The CW sample used in BMP testing was obtained by adding water to the lyophilized CW. No additional processing or pre-treatment was carried out on CW prior to anaerobic digestion. However, only half of the water initially contained in the CW was added to have a more concentrated CW sample since headspace limitations restrict the volume of substrate added in BMP tests.
The inoculum was collected from a mesophilic anaerobic digester in a municipal wastewater treatment plant (WWTP) treating sewage of approximately 400,000 population equivalents in the Barcelona metropolitan area (Spain). After collection, it was stored at 4 °C until used (6 days) as suggested by Astals et al., [31]. Because mesophilic inoculum can easily adapt to psychrophilic conditions, the same inoculum was used for both mesophilic and psychrophilic conditions to facilitate results comparison. The physicochemical properties of CW and inoculum are shown in Table 1.
Table 1.
Characterisation of the CW and inoculum (average values ± standard deviation).
| Parameter | Units | CW | Inoculum |
|---|---|---|---|
| pH | – | 5.5 ± 0.1 | 7.3 ± 0.1 |
| TS | g/L | 81.9 ± 0.3 | 26.0 ± 0.1 |
| VS | g/L | 74.4 ± 0.5 | 18.3 ± 0.2 |
| VS/TS | – | 0.91 ± 0.1 | 0.70 ± 0.1 |
| Alkalinity | g CaCO3/L | 1.3 ± 0.2 | – |
| sCOD | g/L | 21.4 ± 0.1 | 2.1 ± 0.1 |
| TAN | g N/L | – | 748 ± 24 |
Abbreviations: TS stands for total solids; VS stands for volatile solids; sCOD stands for soluble chemical oxygen demand; TAN stands for total ammoniacal nitrogen.
2.2. Biomethane potential test set up.
BMP tests were carried out in 250 mL Wheaton® serum bottles following the guidelines given in Holliger et al. [32], under psychrophilic (20 °C) and mesophilic (35 °C) conditions. Each bottle contained 110 mL of inoculum and the amount of CW needed to reach the targeted inoculum-to-substrate ratio (ISR). The ISR tested at each temperature were 1, 2, 3 and 4 in VS-basis. A positive control test containing microcrystalline cellulose and inoculum at an ISR 2 was used to validate the BMP results [32]. A blank test containing only inoculum was used to correct the background methane production from the inoculum endogenous respiration. No external buffers or trace elements were added. The headspace of each bottle was flushed with 99.9 % N2 gas for 30 s (ca. 4 L⋅min−1). The bottles were sealed with a chlorobutyl septum, retained with a screw cap, and stored in an incubator at 35 ± 1 °C (MEMMERT UF750) or 20 ± 1 °C (WTW TS606/2-I). The bottles were mixed by swirling before each sampling event. All BMP tests and blanks were carried out in quadruplicate. Cumulative methane production was measured using the gas density method [19]. At each sampling event, the biogas volume and density were measured using a 100 mL syringe connected to a bench-top water-manometer and a precision balance (FZ-500i, accuracy ±0.002 g). A qualitative factorial-isolated experimental design was used (Table 2).
Table 2.
Experimental design.
| Temperature | ISR | Organic Load (gVS/L) |
|---|---|---|
| Psychrophilic (20 °C) | 1 | 2.0 |
| 2 | 1.0 | |
| 3 | 0.66 | |
| 4 | 0.50 | |
| Mesophilic (35 °C) | 1 | 2.0 |
| 2 | 1.0 | |
| 3 | 0.66 | |
| 4 | 0.50 |
The biogas density is calculated considering the mass loss and the standardised dry biogas volume [19]. The mole fraction of CH4 in biogas is calculated from the normalised difference in density of CO2 and biogas [19]. BMP tests were run for 27 days, when daily methane production during three consecutive days was <1% of the accumulated volume of methane [32]. Cumulative volumetric gas production was calculated at standard conditions (0 °C, 1 atm, dry) using the OBA web application [33]. Three additional serum bottles were set up for each temperature (20 and 35 °C) and ISR (1, 2, 3 and 4) condition to monitor the AD process (i.e., pH, sCOD, and VFAs) without interfering with biogas measurements. These bottles were sampled each day that the biogas production was measured (12 sampling events). In each sampling event, 4 mL of the sample were withdrawn using an 18G hypodermic needle connected to a 5 mL syringe to minimise air exposure.
The BMP tests that displayed a monotonic increase in methane production were modelled using a first-order model (Eq. (1)), where B is the time-dependent cumulative methane production (mLCH4/gVS), B0 is the ultimate methane potential (mLCH4/gVS), k is the first-order degradation constant (d−1), t is time (d), and tdelay is the lag-phase.
| (Eq. 1) |
The degradation model was implemented in Matlab® (R2022a). The lsqcurvefit function using the ‘trust-region-reflective’ optimisation algorithm was used to carry out the non-linear regression of the model. The function nlparci was used to calculate the 95% confidence interval of each parameter. MiniTab® was used to conduct ANOVA and Tukey's range test to determine the influence of temperature and ISR on CW ultimate methane potential (B0) and degradation constant (k).
2.2. Analytical methods
Volatile solids (VS) and total solids (TS) were measured according to the Standard Method procedure 2450G [34]. pH was measured using a semi-microelectrode (PHEL-GB3-001) coupled to a benchtop multimeter (CRIOSON, MultiMeter MM 41). The TAN was measured using a selective ammonia probe (Thermo Scientific Orion™ 9512HPBNWP) following the Standard Method procedure 4500-NH3D [34]. Alkalinity was determined using an automated titrator (Crison pH Burette 24) with a 0.1 M HCl solution and a pH endpoint of 4.30 as for the Standard Method 2320B procedure [34]. For analysis of soluble compounds, the samples were centrifuged at 16,000×g for 15 min and then filtered through a 0.45 μm nylon syringe filter. The soluble chemical oxygen demand (sCOD) was measured following the Standard Method procedure 5520B [34]. Individual VFAs (i.e., acetic, propionic, i-butyric, n-butyric, i-valeric, n-valeric, i-caproic, n-caproic, heptanoic) and alcohols (XOHs: ethanol, propanol, and butanol) were determined using a gas chromatograph (GC-2010 plus Shimadzu) equipped with a capillary column (Agilent technologies DB-FFAP, 30 m × 0.25 mm × 0.25 μm) and flame ionised detector (FID) following the Standard Method 5560D [34]. VFAs isomer (normal and iso) concentrations were summed in a single species. VFAs and alcohols concentrations (mg/L) were converted to COD equivalents using the theoretical value based on their elemental composition. The conversion factors were 1.07 for acetic acid (HAc), 1.51 for propionic acid (HPro), 1.82 for butyric acid (HBu), 2.04 for valeric acid (HVa), 2.21 for caproic acid (HCa), 2.34 for heptanoic acid (HHep), 2.09 for ethanol (EtOH), 2.40 for propanol (ProOH), and 2.59 for butanol (BuOH).
3. Results and discussion
3.1. Methane production, pH and sCOD under psychrophilic and mesophilic conditions
Fig. 1A and B shows the methane production curves of CW at different ISR under psychrophilic (20 °C) and mesophilic (35 °C) conditions, respectively. ANOVA results showed that the ISR had a significant impact on the ultimate methane potential under both temperature conditions (p < 0.001) due to process inhibition at low ISR.
Fig. 1.
BMP tests results, specific methane production curves on psychrophilic (A) and mesophilic (B) conditions, pH on psychrophilic (C) and mesophilic (D) and sCOD on psychrophilic (E) and mesophilic (F) conditions.
The experiments carried out at an ISR 3 and 4 showed a monotonic increase in methane production under both temperature conditions, indicating that the degradation process did not suffer acute inhibition despite the slight drop in pH and accumulation of VFAs during the first days of the experiment (Fig. 2). In the literature, the profile of these BMP tests is considered a “normal” shape for BMP curves [35,36]. Table 3 presents the model outputs of these experiments under both temperature conditions. Model outputs show that temperature had a significant impact on CW degradation rate, with mesophilic conditions degradation rates doubling those of psychrophilic conditions. The difference in degradation rate between both temperature conditions was also evident when comparing the degradation of the accumulated VFAs (Fig. 2). The lower degradation rate at psychrophilic conditions compared to mesophilic conditions is consistent with Martí-Herrero et al. [30], who used CM and cellulose as substrates. Nevertheless, the methane yield of CW was not notably (p = 0.087) affected by temperature, with values ranging between 389 and 436 mLCH4/gVS (Table 3). The latter indicates that CW is highly biodegradable regardless of the process temperature. The methane yield of the CW under study is consistent with values reported in the literature for CW under mesophilic conditions (423–600 mLCH4/gVS) [10,14,26].
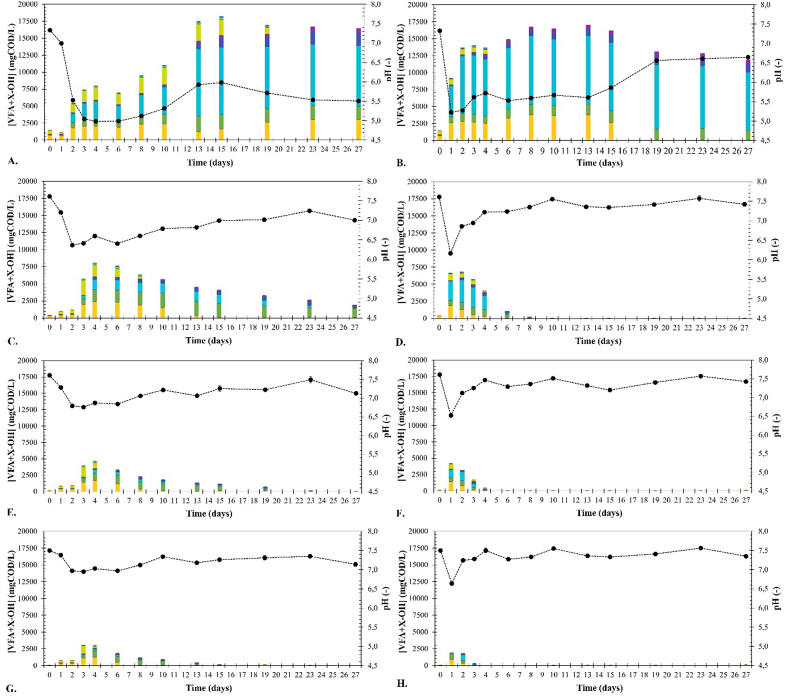
Fig. 2.
Intermediate products (VFAs and X–OH) concentrations under psychrophilic conditions for ISR 1 (A), 2 (C), 3 (E) and 4 (G) and mesophilic conditions ISR 1 (B), 2 (D), 3 (F) and 4 (H). Acetic (■), propionic (■), butyric (■), valeric (■), caproic (■), heptanoic (■), ethanol (■), propanol (■), butanol (■).
Table 3.
Model parameters and their 95% confidence interval of the experiments carried out at an ISR of 3 and 4 under psychrophilic and mesophilic conditions.
| ISR | Psychrophilic (20 °C) |
Mesophilic (35 °C) |
||
|---|---|---|---|---|
| B0 (mL CH4/g VS) | k (d−1) | B0 (mL CH4/g VS) | k (d−1) | |
| 3 | 433 ± 30 | 0.14 ± 0.03 | 441 ± 16 | 0.38 ± 0.06 |
| 4 | 389 ± 6 | 0.18 ± 0.01 | 434 ± 16 | 0.45 ± 0.08 |
The specific methane production curves of the experiments carried out at an ISR 2 exhibited a “retarded methane formation” curve, a clear sign of process inhibition [35,36]. However, the degree of inhibition is different between psychrophilic and mesophilic conditions. Inhibition was more severe under psychrophilic conditions despite the sharper pH drop at the beginning of experiment under mesophilic conditions (Fig. 1C and D). The sharper pH drop under mesophilic conditions was likely caused by the higher degradation rate at this temperature (Table 3). The mesophilic experiment was able to recover circumneutral pH after 2 days, concomitant with the start of the net consumption of the accumulated VFAs (Fig. 2). The pH of the psychrophilic experiment remained below 6.50 for 6 days, suboptimum pH conditions for methanogenic archaea [37]. The pH recovered once VFAs concentration started to decrease (Fig. 2), much like under mesophilic conditions. These results indicate that there is a higher imbalance between fermentation and methanogenesis rate under psychrophilic conditions than under mesophilic conditions. The higher imbalance between fermentation and methanogenesis rates under psychrophilic conditions led to higher VFA accumulation and prolonged lower pH, indicating a higher risk of AD underperformance or even failure under psychrophilic conditions.
The BMP tests carried out at an ISR 1 were severely inhibited under both psychrophilic and mesophilic conditions, as shown by the lower (p < 0.001) methane yield compared to the experiments carried out at an ISR 4 and 3 (Fig. 1A and B). The lower methane yield of these experiments can be explained by the acute accumulation of intermediate products and the concomitant drop in pH (Fig. 2), which inhibited the anaerobic digestion process (see discussion in Section 3.2). The specific methane production curves of the experiments carried out at an ISR 1 showed a monotonic increase in methane production (so-called “normal” shape). This means that if these ISR 1 BMP results had been analysed independently (only conducting this ISR) and without measuring the sCOD (the most common approach in the AD research field), incorrect methane yields would have been reported. This observation might help to explain the low methane yield that has been reported for CW in some publications. The accumulated sCOD at the end of the experiments (∼15 gCOD/L) reaffirmed that CW was not completely degraded in these experiments (Fig. 1E and F). Indeed, converting 90% of the accumulated sCOD into methane (i.e., assuming a biomass yield of 10%) and adding it to the measured methane production results in a specific methane production of about 450 mLCH4/gVS, close to the values obtained in the non-inhibited experiments.
3.2. Intermediate product concentration under psychrophilic and mesophilic conditions
Fig. 2 shows the intermediate product (i.e. VFAs and alcohols) concentrations over time for each ISR (i.e. 1, 2, 3, 4) under both psychrophilic (20 °C) and mesophilic (35 °C) conditions. The ISR had a much greater impact on the accumulation of intermediate products than temperature, indicating that process inhibition due to overloading is plausible under both psychrophilic and mesophilic conditions. Temperature also had a noticeable impact on the profile of the accumulated intermediate products. Mesophilic conditions favoured the accumulation of butyric acid (>40% of the CODVFA + XOH), while psychrophilic conditions favoured the accumulation of a variety of products, mainly acetic acid, propionic acid, butyric acid and ethanol.
The accumulation of intermediate products increased as the ISR decreased due to the higher initial CW concentration. The concentrations of intermediate products cannot be used to calculate the CW degradation nor CH4 production because changes in their concentration comprise two phenomena, i.e., fermentation (generation) and methanogenesis (consumption). However, the net change in their concentration can be used to assess the imbalance between the rates of fermentation and methanogenesis under different conditions. The accumulation of intermediate products clearly showed that CW fermentation was faster than methanogenesis under both psychrophilic and mesophilic conditions. The maximum intermediate product concentrations were reached sooner under mesophilic conditions than under psychrophilic conditions, indicating that CW fermentation was faster under mesophilic conditions than under psychrophilic conditions. This observation is consistent with the higher methane production rates under mesophilic conditions (Table 2).
At an ISR of 4 and 3, the concentration of intermediate products never exceeded 5 gCODVFA + XOH/L for both temperature conditions (Fig. 2). The accumulated intermediate products were completely degraded after reaching their maximum concentration. Under mesophilic conditions, the maximum concentration of intermediate products was reached on the 1st day of the experiment and quickly dropped to less than 50 mgCODVFA + XOH/L on the 4th day. Contrariwise, under psychrophilic conditions, the accumulated intermediate products took much longer to completely degrade (∼20 days), owing to the slower degradation rate of propionic acid. Propionic acid acetogenesis is well-known to be the least thermodynamically favourable of the VFAs reactions [38,39].
The accumulation of intermediate products was more noticeable at an ISR of 2 than at an ISR of 4 and 3, with intermediate product concentrations reaching a maximum concentration of 6.8 gCODVFA + XOH/L on the 2nd day under mesophilic conditions and of 8.1 gCODVFA + XOH/L on the 4th day under psychrophilic conditions. Under mesophilic conditions, the accumulated products were completely degraded by the 6th day. However, under psychrophilic conditions, the accumulated intermediate products were not completely degraded by the end of the experiment (27th day). Similar to the psychrophilic experiments carried out at an ISR of 4 and 3, propionic acid was the intermediate product that showed the slowest degradation rate. These findings suggest that propionic acid accumulation is the primary limitation for CW anaerobic (co-)digestion under psychrophilic conditions.
The experiments carried out at an ISR of 1 showed an acute accumulation of intermediate products (>15 gCODVFA + XOH/L) under psychrophilic and mesophilic conditions. The acute accumulation of intermediate products, mainly VFAs, reduced the pH to ∼5.0 for both temperature conditions (Fig. 2). A pH of 5.0 is much lower than the optimal pH range for methanogenic archaea (6.8–7.5) [40,41], which may have inhibited methane formation (Fig. 1). The experiment carried out under mesophilic conditions accumulated 13.7 gCODVFA + XOH/L on the 2nd day, consisting mainly of 2.8 gCODHAc/L, 1.2 gCODHPro/L, 8.5 gCODHBu/L, and 0.9 gCODEtOH/L. The highest intermediate product concentration was observed on the 8th day (16.7 gCODVFA + XOH/L). The net consumption of acetic acid between the 15th and 19th days and the concomitant pH increases from 5.9 to 6.6 indicated the occurrence of acetotrophic methanogenesis. The BMP experiments also showed methane production between these days (Fig. 1). The concentrations of the other intermediate products remained relatively constant from the 8th day until the end of the experiment, except for a slight but constant decrease in butyric acid concentration from 10.1 gCODHBu/L to 8.5 gCODHBu/L. The accumulation of intermediate products under psychrophilic conditions at an ISR of 1 was more gradual than under mesophilic conditions. The maximum intermediate product (18.2 gCODVFA + XOH/L) was reached on the 15th day, consisting mainly of 1.6 gCODHAc/L, 2.1 gCODHPro/L, 9.9 gCODHBu/L, 1.4 gCODHVa/L, and 2.3 gCODEtOH/L. The intermediate product concentration remained relatively constant until the end of the experiment (27th day), indicating that their degradation was not thermodynamically favourable. The only difference in the intermediate product profile between the 15th and 27th was the conversion of ethanol, which could have been oxidised to acetate or used as electron donor to produce valeric acid via chain elongation with propionic acid [42,43]. The intermediate products concentrations at the end of the experiment (27th day) were 3.0 gCODHAc/L, 1.9 gCODHPro/L, 8.9 gCODHBu/L, and 1.9 gCODHVa/L. Overall, these results indicate that intermediate product degradation was less favourable under psychrophilic conditions than under mesophilic conditions. This implies that psychrophilic digesters have a lower capacity to recover from process disturbances (e.g. shock loads).
3.3. CW anaerobic digestion and acidification risk under psychrophilic and mesophilic conditions
CW had a maximum methane yield of about 430 mLCH4/gVS. This high methane yield was obtained under psychrophilic and mesophilic conditions, demonstrating that CW is a suitable (co-)substrate for anaerobic digestion applications at both operating temperatures (Fig. 1). Model outputs showed that the CW degradation rate was twice as fast under mesophilic conditions than under psychrophilic conditions (Table 2). This implies that a psychrophilic digester would require a longer hydraulic retention time (i.e. more volume) than a mesophilic digester to treat a given CW flow rate. It is important to note the kinetic constants obtained from BMP tests cannot be used to design full-scale digesters, but they can be used for comparison purposes within the same batch [44].
The faster degradation rate of CW under mesophilic conditions was also evident in the faster accumulation of intermediate products, mainly VFAs, at the beginning of the BMP test. VFAs accumulation caused a pH drop that was faster and sharper under mesophilic conditions than under psychrophilic conditions (Fig. 2). However, the faster methanogenic rate under mesophilic conditions also resulted in faster degradation of the accumulated VFAs. Regardless of the operating temperature, the accumulation of VFAs and the resulting drop pH drop have implications for the digester's feeding strategy and maximum organic loading rate (OLR). Feeding the CW daily load in a single event (semi-continuous feeding) would result in a VFA peak concentration and drop in pH after the feeding event. The VFAs concentration and the pH drop after the feeding event would determine the digester's maximum OLR. Equally distributing the CW flow rate (continuous feeding) or conducting several feeding events throughout the day is a strategy to smooth out the accumulation of VFA and the resulting pH drop [45]. Continuous feeding allows for higher OLR than semi-continuous feeding, which means processing more CW per day for an already built digester (fixed volume) or building a smaller digester.
Anaerobic digester operators aim to minimise process disturbance that can reduce methane production. However, full-scale digesters are dynamic systems. In industrial applications, such as dairy factory, shock loads are likely to occur due to an increase in production, cleaning events (flushing milk), and milk rejected for commercial application (waste milk), among others [17]. A CW shock load can cause the accumulation of VFAs and a drop in pH, both of which are well-known inhibitors of methanogenic archaea [46]. A decrease in the methanogenic rate, without a corresponding decrease in acidogenic fermentation rate results in a higher accumulation of intermediate compounds, which further inhibits the methanogenic archaea in a feedback loop that can lead to process failure [47]. To avoid this cascading effect, CW dosage should be stopped or reduced to a bare minimum until the digester has degraded the majority of the accumulated VFAs and recovered a circumneutral pH. When using CW as a co-substrate, the main substrate (e.g., cow manure) can be fed to the digester as a source of alkalinity, nutrients, and microorganisms, which should accelerate process recovery. In this regard, this research has shown that there is a greater imbalance between fermentation and methanogenesis rates under psychrophilic conditions than under mesophilic conditions. This implies that there is a greater risk of AD underperformance or process failure under psychrophilic conditions. Under psychrophilic conditions, the accumulated VFAs took much longer to completely degrade than under mesophilic conditions (Fig. 2). This implies that psychrophilic digesters have a lower capacity to recover from process disturbances and that recovery processes will take longer than under mesophilic conditions. As a result, more caution and patience are required to avoid further inhibition and restore process performance under psychrophilic conditions than under mesophilic conditions.
4. Conclusions
The experimental results showed that CW can be successfully degraded under both psychrophilic (20 °C) and mesophilic (35 °C) conditions, with methane yields of 389–436 mLCH4/gVS. However, the degradation rate under mesophilic conditions was twice as fast than under psychrophilic conditions. Organic loading had a greater impact on the accumulation of intermediate products (VFAs and XOHs) than temperature, indicating that process inhibition by overloading is plausible under psychrophilic and mesophilic conditions. The experimental results showed a higher imbalance between fermentation and methanogenesis rate under psychrophilic conditions than under mesophilic conditions, which resulted in higher concentrations of intermediate products (VFAs and XOHs) and prolonged lower pHs. Propionic acid degradation was identified as the main limitation for CW anaerobic digestion under psychrophilic conditions. Overall, these results indicate that the degradation of the intermediate products was less favourable under psychrophilic conditions than under mesophilic conditions. This implies that psychrophilic digesters have a lower capacity to recover from process disturbances and that acidification is more likely in psychrophilic conditions than in mesophilic conditions.
Data availability statement
-
-
Has data associated with your study been deposited into a publicly available repository?
No.
Data will be made available on request.
Ethics declarations
Review and/or approval by an ethics committee was not needed for this study because did not involve live human subjects and/or anatomical and/or biological human and/or animal material.
Informed consent was not required for this study because no interviews were conducted and no information from an individual was compromised.
Funding disclosure
The authors thank the University of La Sabana for funding the project “Codigestión Anaerobia de Residuos Agroindustriales con Fines de Valorización Energética y Agrícola” (INGPHD-50-2022). Miguel Casallas-Ojeda is grateful to the Universidad de La Sabana for the funding his PhD studies through the Carlos Jordana distinction (2020-II) and the Asociación Iberoamericana de Posgrados (AUIP) for the mobility grant to develop an internship at University of Barcelona (Spain). Noemi Perez-Esteban is grateful to the Spanish Ministry of Science and Innovation for her FPI fellowship (PRE2020-092,325). Carlos M. Olaya-Rincón is grateful to the Generalitat de Catalunya for his predoctoral FI grant (2023 FI-1 00121). Sergi Astals is thankful for the funding support provided by the Spanish Ministry of Science, Innovation and Universities (RYC-2017-22372, PID2019-111284RB-I00). The authors would like to thank the Catalan Government for the quality accreditation given to the Environmental Biotechnology research group of the University of Barcelona (2021 SGR 00234). The authors would like to thank the company Agropecuaria Romero Junca and its work team for allowing access to their facilities, facilitating the samples, and providing the necessary information for the development of the research.
CRediT authorship contribution statement
Miguel Casallas-Ojeda: Investigation, Visualization, Formal analysis, Validation, Writing - original draft. Noemi Perez-Esteban: Investigation, Visualization, Writing – review & editing. Iván Cabeza: Conceptualization, Methodology, Supervision, Writing – review & editing, Funding acquisition. Martha Cobo: Writing – review & editing, Funding acquisition. Mario Olaya-Rincón: Conceptualization, Visualization, Writing – review & editing. Diana M. Caicedo-Concha: Resources, Writing – review & editing. Sergi Astals: Conceptualization, Methodology, Resources, Supervision, Writing – review & editing, Funding acquisition.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgments
The authors thank the University of La Sabana for funding the project “Codigestión Anaerobia de Residuos Agroindustriales con Fines de Valorización Energética y Agrícola” (INGPHD-50-2022). Miguel Casallas-Ojeda is grateful to the Universidad de La Sabana for the funding his PhD studies through the Carlos Jordana distinction (2020-II) and the Asociación Iberoamericana de Posgrados (AUIP) for the mobility grant to develop an internship at University of Barcelona (Spain). Noemi Perez-Esteban is grateful to the Spanish Ministry of Science and Innovation for her FPI fellowship (PRE2020-092325). Carlos M. Olaya-Rincón is grateful to the Generalitat de Catalunya for his predoctoral FI grant (2023 FI-1 00121). Sergi Astals is thankful for the funding support provided by the Spanish Ministry of Science, Innovation and Universities (RYC-2017-22372, PID2019-111284RB-I00). The authors would like to thank the Catalan Government for the quality accreditation given to the Environmental Biotechnology research group of the University of Barcelona (2021 SGR 00234). The authors would like to thank the company Agropecuaria Romero Junca and its work team for allowing access to their facilities, facilitating the samples, and providing the necessary information for the development of the research.
Contributor Information
Iván Cabeza, Email: ivan.cabeza@unisabana.edu.co.
Sergi Astals, Email: sastals@ub.edu.
References
- 1.Prazeres A.R., Carvalho F., Rivas J. Cheese whey management: a review. J. Environ. Manag. 2012;110:48–68. doi: 10.1016/j.jenvman.2012.05.018. [DOI] [PubMed] [Google Scholar]
- 2.Rocha J.M., Guerra A. On the valorization of lactose and its derivatives from cheese whey as a dairy industry by-product: an overview. Eur. Food Res. Technol. 2020;246:2161–2174. doi: 10.1007/s00217-020-03580-2. [DOI] [Google Scholar]
- 3.Osorio-González C.S., Gómez-Falcon N., Brar S.K., Ramírez A.A. Cheese whey as a potential feedstock for producing renewable biofuels: a review. Energies. 2022;15 doi: 10.3390/en15186828. [DOI] [Google Scholar]
- 4.Fernández-Gutiérrez D., Veillette M., Giroir-Fendler A., Ramirez A.A., Faucheux N., Heitz M. Biovalorization of saccharides derived from industrial wastes such as whey: a review. Rev. Environ. Sci. Biotechnol. 2017;16:147–174. doi: 10.1007/s11157-016-9417-7. [DOI] [Google Scholar]
- 5.Asunis F., De Gioannis G., Dessì P., Isipato M., Lens P.N.L., Muntoni A., Polettini A., Pomi R., Rossi A., Spiga D. The dairy biorefinery: integrating treatment processes for cheese whey valorisation. J. Environ. Manag. 2020;276 doi: 10.1016/j.jenvman.2020.111240. [DOI] [PubMed] [Google Scholar]
- 6.Bosco F., Carletto R.A., Marmo L. An integrated cheese whey valorization process. Chem. Eng. Trans. 2018;64:379–384. doi: 10.3303/CET1864064. [DOI] [Google Scholar]
- 7.Carvalho F., Prazeres A.R., Rivas J. Cheese whey wastewater: characterization and treatment. Sci. Total Environ. 2013;385(396):445–446. doi: 10.1016/J.SCITOTENV.2012.12.038. [DOI] [PubMed] [Google Scholar]
- 8.Casallas-Ojeda M., Marmolejo-Rebellón L., Torres-Lozada P. 2020. Identification of Factors and Variables that Influence the Anaerobic Digestion of Municipal Biowaste and Food Waste, Waste and Biomass Valorization; pp. 1–16. [DOI] [Google Scholar]
- 9.Carvalho F., Prazeres A.R., Rivas J. Cheese whey wastewater: characterization and treatment. Sci. Total Environ. 2013:385–396. doi: 10.1016/j.scitotenv.2012.12.038. [DOI] [PubMed] [Google Scholar]
- 10.Escalante H., Castro L., Amaya M.P., Jaimes L., Jaimes-Estévez J. Anaerobic digestion of cheese whey: energetic and nutritional potential for the dairy sector in developing countries. Waste Manag. 2018;71:711–718. doi: 10.1016/j.wasman.2017.09.026. [DOI] [PubMed] [Google Scholar]
- 11.Mata-Alvarez J., Dosta J., Romero-Güiza M.S., Fonoll X., Peces M., Astals S. A critical review on anaerobic co-digestion achievements between 2010 and 2013. Renew. Sustain. Energy Rev. 2014;36:412–427. doi: 10.1016/j.rser.2014.04.039. [DOI] [Google Scholar]
- 12.Ghaly A.E. A comparative study of anaerobic digestion of acid cheese whey and dairy manure in a two-stage reactor. Bioresour. Technol. 1996;58:61–72. doi: 10.1016/S0960-8524(96)00105-8. [DOI] [Google Scholar]
- 13.Iglesias-Iglesias R., Kennes C., Veiga M.C. Valorization of sewage sludge in co-digestion with cheese whey to produce volatile fatty acids. Waste Manag. 2020;118:541–551. doi: 10.1016/j.wasman.2020.09.002. [DOI] [PubMed] [Google Scholar]
- 14.Jaimes-Estévez J., Mercado E.V., Jaramillo J.G., Rodríguez P., Martí-Herrero J., Escalante H., Castro L. From laboratory to farm-scale psychrophilic anaerobic co-digestion of cheese whey and cattle manure. Bioresour. Technol. Rep. 2022;19 doi: 10.1016/J.BITEB.2022.101168. [DOI] [Google Scholar]
- 15.Treu L., Tsapekos P., Peprah M., Campanaro S., Giacomini A., Corich V., Kougias P.G., Angelidaki I. Microbial profiling during anaerobic digestion of cheese whey in reactors operated at different conditions. Bioresour. Technol. 2019;275:375–385. doi: 10.1016/j.biortech.2018.12.084. [DOI] [PubMed] [Google Scholar]
- 16.Kunatsa T., Xia X. A review on anaerobic digestion with focus on the role of biomass co-digestion, modelling and optimisation on biogas production and enhancement. Bioresour. Technol. 2022;344 doi: 10.1016/J.BIORTECH.2021.126311. [DOI] [PubMed] [Google Scholar]
- 17.Sembera C., Macintosh C., Astals S., Koch K. Benefits and drawbacks of food and dairy waste co-digestion at a high organic loading rate: a Moosburg WWTP case study. Waste Manag. 2019;95:217–226. doi: 10.1016/J.WASMAN.2019.06.008. [DOI] [PubMed] [Google Scholar]
- 18.Romero-Güiza M.S., Vila J., Mata-Alvarez J., Chimenos J.M., Astals S. The role of additives on anaerobic digestion: a review. Renew. Sustain. Energy Rev. 2016;58:1486–1499. doi: 10.1016/J.RSER.2015.12.094. [DOI] [Google Scholar]
- 19.Justesen C.G., Astals S., Mortensen J.R., Thorsen R., Koch K., Weinrich S., Triolo J.M., Hafner S.D. BMP); 2019. Development and Validation of a Low-Cost Gas Density Method for Measuring Biochemical Methane Potential. [DOI] [Google Scholar]
- 20.Astals S., Nolla-Ardèvol V., Mata-Alvarez J. Anaerobic co-digestion of pig manure and crude glycerol at mesophilic conditions: biogas and digestate. Bioresour. Technol. 2012;110:63–70. doi: 10.1016/j.biortech.2012.01.080. [DOI] [PubMed] [Google Scholar]
- 21.Jensen P.D., Astals S., Bai X., Nieradzik L., Wardrop P., Batstone D.J., Clarke W.P. Established full-scale applications for energy recovery from water: anaerobic digestion. Resour. Recover. from Water. 2022:99–139. doi: 10.2166/9781780409566_0099. [DOI] [Google Scholar]
- 22.Akindolire M.A., Rama H., Roopnarain A. Psychrophilic anaerobic digestion: a critical evaluation of microorganisms and enzymes to drive the process. Renew. Sustain. Energy Rev. 2022;161 doi: 10.1016/J.RSER.2022.112394. [DOI] [Google Scholar]
- 23.Fernández-Domínguez D., Astals S., Peces M., Frison N., Bolzonella D., Mata-Alvarez J., Dosta J. Volatile fatty acids production from biowaste at mechanical-biological treatment plants: focusing on fermentation temperature. Bioresour. Technol. 2020;314 doi: 10.1016/J.BIORTECH.2020.123729. [DOI] [PubMed] [Google Scholar]
- 24.Nie E., He P., Zhang H., Hao L., Shao L., Lü F. How does temperature regulate anaerobic digestion? Renew. Sustain. Energy Rev. 2021;150 doi: 10.1016/J.RSER.2021.111453. [DOI] [Google Scholar]
- 25.Metcalf & Eddy . 2003. Wastewater Engineering : Treatment and Reuse, Boston. [Google Scholar]
- 26.Labatut R.A., Angenent L.T., Scott N.R. Biochemical methane potential and biodegradability of complex organic substrates. Bioresour. Technol. 2011;102:2255–2264. doi: 10.1016/j.biortech.2010.10.035. [DOI] [PubMed] [Google Scholar]
- 27.Jaimes-Estévez J., Zafra G., Martí-Herrero J., Pelaz G., Morán A., Puentes A., Gomez C., Castro L.D.P., Hernández H.E. Psychrophilic full scale tubular digester operating over eight years: complete performance evaluation and microbiological population. Energies. 2021;14:151. doi: 10.3390/EN14010151. [DOI] [Google Scholar]
- 28.Cordoba V.E., Mussi J., De Paula M., Acosta G.G. Prediction of biomethane production of cheese whey by using artificial neural networks. IEEE Lat. Am. Trans. 2023;21:1032–1039. doi: 10.1109/TLA.2023.10251810. [DOI] [Google Scholar]
- 29.Bella K., Venkateswara Rao P. Anaerobic co-digestion of cheese whey and septage: effect of substrate and inoculum on biogas production. J. Environ. Manag. 2022;308 doi: 10.1016/J.JENVMAN.2022.114581. [DOI] [PubMed] [Google Scholar]
- 30.Martí-Herrero J., Castro L., Jaimes-Estévez J., Grijalva M., Gualatoña M., Aldás M.B., Escalante H. Biomethane potential test applied to psychrophilic conditions: three issues about inoculum temperature adaptation. Bioresour. Technol. Rep. 2022;20 doi: 10.1016/J.BITEB.2022.101279. [DOI] [Google Scholar]
- 31.Astals S., Koch K., Weinrich S., Hafner S.D., Tait S., Peces M. Impact of storage conditions on the methanogenic activity of anaerobic digestion inocula. Water. 2020;12 doi: 10.3390/w12051321. [DOI] [Google Scholar]
- 32.Holliger C., Astals S., Fruteau De Laclos H., Hafner S.D., Koch K., Weinrich S. 2021. Towards a Standardization of Biomethane Potential Tests: a Commentary. [DOI] [PubMed] [Google Scholar]
- 33.Hafner S.D., Koch K., Carrere H., Astals S., Weinrich S., Rennuit C. Software for biogas research: tools for measurement and prediction of methane production. SoftwareX. 2018;7:205–210. doi: 10.1016/J.SOFTX.2018.06.005. [DOI] [Google Scholar]
- 34.APHA . 23rd edition. 2017. Standard Methods for the Examination of Water and Wasterwater. [Google Scholar]
- 35.Koch K., Hafner S.D., Weinrich S., Astals S. Identification of critical problems in biochemical methane potential (BMP) tests from methane production curves. Front. Environ. Sci. 2019;7 doi: 10.3389/fenvs.2019.00178. [DOI] [Google Scholar]
- 36.VDI 4630 . 2016. Fermentation of Organic Materials: Characterization of the Substrate, Sampling, Collection of Material Data, Fermentation Tests. [Google Scholar]
- 37.Latif M.A., Mehta C.M., Batstone D.J. Low pH anaerobic digestion of waste activated sludge for enhanced phosphorous release. Water Res. 2015;81:288–293. doi: 10.1016/j.watres.2015.05.062. [DOI] [PubMed] [Google Scholar]
- 38.Batstone D.J., Keller J., Angelidaki I., Kalyuzhnyi S.V., Pavlostathis S.G., Rozzi A., Sanders W.T., Siegrist H., Vavilin V.A. The IWA anaerobic digestion model No 1 (ADM1) Water Sci. Technol. 2002;45:65–73. doi: 10.2166/wst.2002.0292. [DOI] [PubMed] [Google Scholar]
- 39.Zhang Y., Li J., Liu F., Yan H., Li J. Mediative mechanism of bicarbonate on anaerobic propionate degradation revealed by microbial community and thermodynamics. Environ. Sci. Pollut. Res. 2018;25:12434–12443. doi: 10.1007/s11356-018-1430-7. [DOI] [PubMed] [Google Scholar]
- 40.Jankowska E., Chwiałkowska J., Stodolny M., Oleskowicz-Popiel P. Effect of pH and retention time on volatile fatty acids production during mixed culture fermentation. Bioresour. Technol. 2015;190:274–280. doi: 10.1016/j.biortech.2015.04.096. [DOI] [PubMed] [Google Scholar]
- 41.Liu Y., Whitman W.B. Metabolic, phylogenetic, and ecological diversity of the methanogenic archaea. Ann. N. Y. Acad. Sci. 2008;1125:171–189. doi: 10.1196/ANNALS.1419.019. [DOI] [PubMed] [Google Scholar]
- 42.Cavalcante W.D.A., Leitão R.C., Gehring T.A., Angenent L.T., Santaella S.T. Anaerobic fermentation for n-caproic acid production: a review. Process Biochem. 2017;54:106–119. doi: 10.1016/j.procbio.2016.12.024. [DOI] [Google Scholar]
- 43.Coma M., Vilchez-Vargas R., Roume H., Jauregui R., Pieper D.H., Rabaey K. Product diversity linked to substrate usage in chain elongation by mixed-culture fermentation. Environ. Sci. Technol. 2016;50:6467–6476. doi: 10.1021/acs.est.5b06021. [DOI] [PubMed] [Google Scholar]
- 44.Hafner S.D., Astals S., Holliger C., Koch K., Nielsen L., Refsahl L., Weinrich S. Assessing the value of kinetic results from biochemical methane potential tests: reproducibility from a large inter-laboratory study. Clean. Chem. Eng. 2022;4 doi: 10.1016/J.CLCE.2022.100065. [DOI] [Google Scholar]
- 45.Jensen P.D., Astals S., Lu Y., Devadas M., Batstone D.J. Anaerobic codigestion of sewage sludge and glycerol, focusing on process kinetics, microbial dynamics and sludge dewaterability. Water Res. 2014;67:355–366. doi: 10.1016/J.WATRES.2014.09.024. [DOI] [PubMed] [Google Scholar]
- 46.Wang K., Yin J., Shen D., Li N. Anaerobic digestion of food waste for volatile fatty acids (VFAs) production with different types of inoculum: effect of pH. Bioresour. Technol. 2014;161:395–401. doi: 10.1016/J.BIORTECH.2014.03.088. [DOI] [PubMed] [Google Scholar]
- 47.Astals S., Batstone D.J., Tait S., Jensen P.D. Development and validation of a rapid test for anaerobic inhibition and toxicity. Water Res. 2015;81:208–215. doi: 10.1016/J.WATRES.2015.05.063. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
-
-
Has data associated with your study been deposited into a publicly available repository?
No.
Data will be made available on request.