Abstract
Objective
To develop a patient decision aid facilitating shared decision making for patients with potential pancreatic cancer deciding about no treatment, surgical or medical treatment.
Methods
Based on a user-centred design by Wittemann et al., we developed a shared decision making intervention in three phases: 1) Understanding decision needs 2) Development of a patient decision aid (PtDA) based on a generic template 3) Assessment of the intervention from interviews with patients (n = 11), relatives (n = 11), nurses (n = 4) and surgeons (n = 2) analysed with thematic analysis, and measuring patients' perceptions of choice of options with the Decisional Conflict Scale.
Results
Results showed varying experiences with the use of the PtDA, with surgeons not finding PtDA useful as it was impractical and constraining with patients' conversations. There was no difference in patients' perceptions in choosing options for those being presented vs those patients not being presented for the PtDA.
Conclusion
The format and structure of the PtDA was not feasible for the surgeons as fundamental users in the present clinic.
Innovation
This study highlights the urgent need to consider clinical context before introducing a predefined tool and shows the importance of a multistakeholder approach. Research should focus on finding means to successful implement shared decision making.
Keywords: Pancreatic cancer, Shared decision making, Surgery, Patient decision aid
Highlights
-
•
Novel investigation facilitating shared decision making for patients with potential pancreatic cancer deciding about their treatment.
-
•
Entail shared decision making to patients undergoing surgery for a time-critical cancer disease with limited survival and with alternative treatment options in a dichotomous irreversible choice.
-
•
The insight gained showed how the use of a generic patient decision aid template worked, not as a facilitator, but as a barrier for enhancing shared decision making.
1. Introduction
Pancreatic cancer (PC) has become one of the leading causes of cancer-related deaths world-wide. Due to its rapid progressive nature with an overall 5 year-survival rate <10% [1], diagnosis and treatment has high priority [2,3]. Patients are mainly elderly and often with more severe comorbidity than the background population, which makes the evaluation of operability complex. Long-term survival or cure can only be obtained by extensive surgical treatment, and although the postoperative mortality is low in high-volume centres, the complication rate and sequelae are considerable [1]. The postoperative outcome often leads to a reduced functional and cognitive level as well as a decreased Quality of Life (QoL) [[4], [5], [6], [7], [8], [9], [10]] and induces major psychological consequences for patients and relatives [[11], [12], [13]]. During the last decade, general healthcare has moved towards a more patient-centred approach in the decision making of the individual treatment [15]. The decision about surgical treatment has mainly been directed by resectability and operability and to a lesser extent by postoperative QoL [14]. However, there are two other options although not curable, either palliative oncologic treatment or best supportive care [1].
There are several definitions on shared decision making (SDM) [15]. We choose the definition by Elwyn et al. “a process in which decisions are made in a collaborative way, where trustworthy information is provided in accessible formats about a set of options” [16]. SDM helps patients to consider pros and cons of a treatment whether medical or surgical [16], enables patients, nurses and surgeons to agree on a treatment by sharing information and building consensus, and improves patients' satisfaction and adherence to therapy as well as long-term health related QoL [17,18]. However, patients' and surgeons' views on SDM are limited [[19], [20], [21]]. From an ethical point of view, patients' autonomy in decision making is indisputable, but their decision needs a thorough counselling by the surgeon. Surgical rates and decisional conflicts may decrease as knowledge of the patients' situation increases [22]. In the PC population, patient-centred knowledge, and perspectives on the use of SDM have only been investigated in few studies [[23], [24], [25]].
Information about the decision is provided in accessible formats, for instance as a patient decision aid (PtDA) [16]. PtDAs are tools such as personalized worksheets, take-home information sheets, online iterative formats or grids designed to support SDM, as they include information about options and associated benefits, or harms, and they evoke the patient's personal preferences and values. A review of studies dealing with a variety of decision contexts found that using a PtDA increased the patient's knowledge, decreased decisional conflict and regret, and involved the patient in the decision making. However, these studies did not entail patients, who had to undergo surgery for a time-critical cancer with a limited overall survival and with alternative treatment options in a dichotomous irreversible choice [26]. Patients with suspected PC should decide whether to receive major surgery with potential cure but with a relatively high risk of complications and recurrence or palliative oncologic treatment or best supportive care. The decision between the second options as alternative to surgery is made in an oncology setting but depends on the pathologic diagnosis, which may not be available at the time when patients are informed about their options. The aim was to develop an SDM intervention for patients with suspected PC who were to decide between intended curative surgical treatment, palliative oncologic treatment or no treatment at all apart from best supportive care.
2. Methods
This study was reported in accordance with the SUNDAE guidelines (Supplementary Fig. 3). The development of the SDM intervention was based on a user-centred design and included three phases as suggested by Witteman et al. [27]: 1) Exploring the decision needs among patients, relatives, nurses, and surgeons 2) Developing a SDM intervention, 3) Assessing patients', nurses, and surgeons' experiences of the intervention. As the intervention consisted of a PtDA, we aimed to achieve as many of the International Patient Decision Aid Standards (IPDAS) criteria as possible [28]. The results of the developing process were reported according to the IPDAS guideline [27]. The PtDA was based on a Danish generic template, “The Decision Helper ™”. This template was designed to support SDM in consultations and developed within oncological settings. The template includes a range of mandatory elements.
Elwyn et al.'s [16] three talk model for SDM was chosen because its simplicity makes it an attractive guide in clinical settings [16]. The SDM process included: 1) Team talk: the surgeon invites the patient and relatives to make a decision that suits the patient best. 2) Choice talk: the surgeon compares possible options with the patient and relative. 3) Decision talk: the surgeon finds out what matters most to the patient together with the patient and relative. “The Decision Helper ™” is also based on the three talk model. To understand decision needs among end-users we used the Ottawa Decision Support Framework to define decision needs [29]. Decision needs are: “A deficit that can adversely affect the quality of a decision”. We further defined decisional needs as uncertainty, inadequate knowledge, unrealistic expectation, unclear values, inadequate support, complex decision characteristics and personal needs [29]. (See Fig. 1.)
Fig. 1.
Designing a patient decision aid to support patients with potential pancreatic cancer when deciding about treatment.
2.1. Setting
The study was performed at Copenhagen University Hospital, Rigshospitalet, which is a tertiary hospital in the capital region with a catchment area for pancreatic surgery of 2,500,000 residents. The average number of pancreatic operations is 250/year. The participating nurses and surgeons included the entire pancreatic team at the hospital. Given the seriousness of the pancreatic tumours there is a need for timely treatment. Therefore, eligible patients are booked for surgery prior to consultation. This booking may be cancelled after the consultation. This consultation is the first and sometimes the only physical consultation between surgeons and patients making it the best point of contact in the pathway to introduce SDM.
2.2. Phase 1: Exploration of decision needs among end-users
To explore decision needs and the process of SDM among patients with potential PC we conducted a literature search in MEDLINE and CINAHL using a two blocks search, with one block including keywords and index terms for PC and synonyms and the other block including keywords and index terms for SDM and synonyms. Studies were screened independently by two authors and were included if their focus were on patients' decision needs regarding the process of SDM. Data from the included studies were extracted and analysed narratively with a focus on patients' decision-making needs when they were involved in deciding whether or not to have pancreatic surgery.
To explore patients', relatives', nurses', and surgeons' decision needs we individually interviewed patients and relatives and held two focus groups with nurses and surgeons. The interviews took place at three occasions: within two days after the first preoperative consultation, where decision about operation was made, before discharge, and finally one week after discharge. The individual interviews were conducted using a semi-structured interview guide [30] and the focus groups using a question guide [31]. Both were based on literature regarding the practice and measurement of SDM [[32], [33], [34]]. All interviews were recorded, transcribed and analysed with thematic analysis [35]. Field observations of the consultations were performed to supplement the interviews. KD, AB and JF performed the analysis.
2.3. Phase 2: Development and refining the SDM intervention
We developed and refined the SDM intervention in four workshops with nurses, surgeons, oncologists, and patients with PC. In Workshop 1, the first iteration was settled to synthesise the evidence of treatment options into the PtDA, and in Workshop 2 the decision needs among patients, nurses and surgeons were synthesized. Afterwards, a prototype with end-uses were drafted. The second iteration was completed in Workshop 3 with patients (n = 5), and relatives (n = 2) where prototypes were discussed to achieve usability and comprehensibility. In Workshop 4, nurses (n = 5), surgeons (n = 4) and managers (n = 2) reviewed the second iteration and the SDM intervention and the PtDA were pre-tested with patients (n = 4) who were not part of the developing process. Hereafter, the third iteration of the PtDA was tested, and patients (n = 4) and relatives (n = 1) provided feedback by informal interviews leading a fourth iteration ready for assessment. All patients in this phase had been through the decision and therefore had knowledge of the choice and the aftermath. Prior to testing, each surgeon was introduced to how to use the PtDA in the consultations. This was done from one to three days before the consultations with the patients. The surgeons also had a visual one-page guide on how to structure the SDM process and using the PtDA. The guide contained pre-made sentences.
2.4. Phase 3: Assessing the end-users' experiences of the SDM intervention
To assess end-users' experiences of the SDM intervention, quantitative and qualitative data were used. Patients were included from 1. March 2021 to 1. June 2022. All patients were 18 years or older, sufficiently Danish speaking, and attended consultations regarding operation for suspected PC with surgeons and nurses involved in the treatment trajectory. The Decisional Conflict Scale (DCS) [36] was used to measure patients' self-reported uncertainty with the decision. The scale consisted of 16 questions with five subscales: informed, value clarity, support, uncertainty, and effective decision. The total DCS score equally weights all subscales. Responses to each question were reported on a 5-point Likert scale (0 = strongly agree to 4 = strongly disagree). Data obtained from the DCS were rated using a total score between 0 and 100 (0 was considered as no decisional conflict). Scores lower than 25 reflect low, 25–37,5 reflect moderate and ≥ 37,5 reflect high decisional conflict. Scores of 25 and above reflect clinically significant decisional conflicts [36]. The sub scores were calculated as follows: Total score (item 1–16) (summed, divided by 16 and multiplied by 25), Uncertainty sub score (10,11,12), Informed sub score (1,2,3), Value clarity sub score (4, 5, 6), Support sub score (7, 8, 9) (summed, divided by 3 and multiplied by 25), Effective sub score (13, 14, 15, 16) (summed, divided by 4 and multiplied by 25) [36]. Fishers' exact test was used to test the difference between patients presented for the PtDA versus patients, who had not been presented for the PtDA during the consultation. In addition to the DSC, we developed five questions (with yes/no answers) that were given to the patients after the consultation (Table 1). The patients were asked to fill in the DCS and the five questions within one week after the consultation.
Table 1.
Additional questions given to patients after the consultation.
| Did you read the preparation letter? |
| Did you fill in the preparation letter? |
| Was the preparation letter used by clinicians in the consultation? |
| Was the patient decision aid used in the consultation? |
| Are you interested in participating in an interview regarding your choice of treatment and the patient decision aid? |
Patients were interviewed using a semi-structured interview guide [30] one to five weeks after the consultation. Surgeons were interviewed in two focus groups [31] using a slightly modified question guide as portrayed in Phase 1. Data was analysed as described in Phase 1.
2.5. Ethics
Written informed consent from patients, relatives, nurses, and surgeons were obtained. The Danish Data Protection Agency approved the data management (P-2021-206). According to Danish legislation, this type of research is exempted from ethical approval. The study was performed in compliance with the Helsinki Declaration [37].
3. Results
In Phase 1 we found from the literature three studies investigating the process of SDM and patients with potential PC deciding about surgical treatment [[23], [24], [25]], but none of the studies reported an SDM intervention for patients with potential PC making the decision about surgery [26]. Patients felt pressured by the surgeon to accept surgical treatment due to the lack of other treatment options, and only felt like “winners” if they qualified for surgery [24]. A barrier to SDM was the examination of the operability of the patient and not whether surgery could be an option [23]. Nurses and surgeons felt that they needed to get an overall picture of the patient's health and social situation in order to adequately support the patients' decision. Furthermore, nurses and surgeons called for a decision-making process over several consultations rather than just one [25]. One qualitative study explored PC patients' perceptions and preferences on information and treatment decision-making. This study found that physicians should promote an exchange of relevant preferences and values with the patients in order to have a sound basis for their recommendation [38].
Interviews with patients (n = 11) and relatives (n = 11), focus groups with clinicians (n = 6) and field observations (n = 15) (Table 2a) showed that patients had low prognostic awareness when they were informed about suspected PC and the option of surgery, and their understanding of life expectancy was overly optimistic.
Table 2a.
Demographic characteristics of patients, relatives, nurses and surgeons in Phase 1.
| Patients | (n) | 11 |
|---|---|---|
| Age | median (range) | 73 (52–89) |
| Males | (n) | 6 |
| Living alone | (n) | ˂ 3 |
| Educational level | ||
| College degree | (n) | 6 |
| Vocational education | (n) | 4 |
| Unknown | (n) | 0 |
| Relatives | (n) | 11 |
| Male | (n) | 7 |
| Spouse | (n) | 8 |
| Clinicians | (n) | 6 |
| Surgeons | (n) | 2 |
| Males | (n) | 2 |
| Years of experience | median⁎ | 16 |
| Nurses | (n) | 4 |
| Males | (n) | 0 |
| Years of experience | median | 20,5 |
Range not shown as a result of small sample size.
The focus of the consultations was primarily on the surgical procedure, the high risk of relapse after the operation, and the recommended adjuvant chemotherapy. Medical alternatives to surgery were rarely discussed because this was considered inferior regarding survival benefit. Before the consultation most patients had pronounced a wish for an operation i.e. “I just need to get through this surgical procedure and then I can get back to my normal life” and “There is no choice, if I if I don't have this operation I will die”. After the operation, most patients described a recovery longer than expected, and they did not realize that the process of regaining normal function and returning to normal life would be as hard both mentally and physically. Relatives described that focus of the consultations was the surgical procedure and explained that there was no doubt that the surgical treatment was the right one, as they were only informed about other treatments if they asked the surgeon. The surgeons were evasive when the conversation turned to the prognosis explaining that the best course of treatment was when the patients themselves decided to have surgery or not. The nurses explained that their role was to clarify any misinformation or uncertainties and explained that some patients were shocked by the extend of the operation.
In Phase 2, a PtDAs tailored for three possible surgical procedures (Whipple's, Distal and Total Pancreatectomy) were developed. They included an explanation of the decision to be made, description of the surgical procedures and pros and cons of surgery compared to oncology and palliative care. Moreover, information about possible surgical complications, short as well as long-term side effects, prognosis and finally a preparation letter for the patient to fill out upon arriving to the preoperative consultation. The preparation letter informed the patients about the SDM process. It further aimed to prepare patients for a conversation about the potential surgical treatment options and invited them into a decision-making process that made them reflect on their values and concerns regarding the surgery and their individual situation. Material was prepared for nurses and surgeons on how to use PtDA in consultations and a visual guide with two questions, one about prognosis and the other about more time for consideration. Finally, there were visual maps of the surgical procedure with illustrations of ‘before’ and ‘after’ surgery. In the third iteration, two of these maps were combined.
The PtDAs met all seven IPDAS criteria to be defined as a PtDA, and five out of eight IPDAS criteria to reduce the risk of making a biased decision. The PtDAs did not provide references to applied scientific evidence (Supplementary Fig. 4).
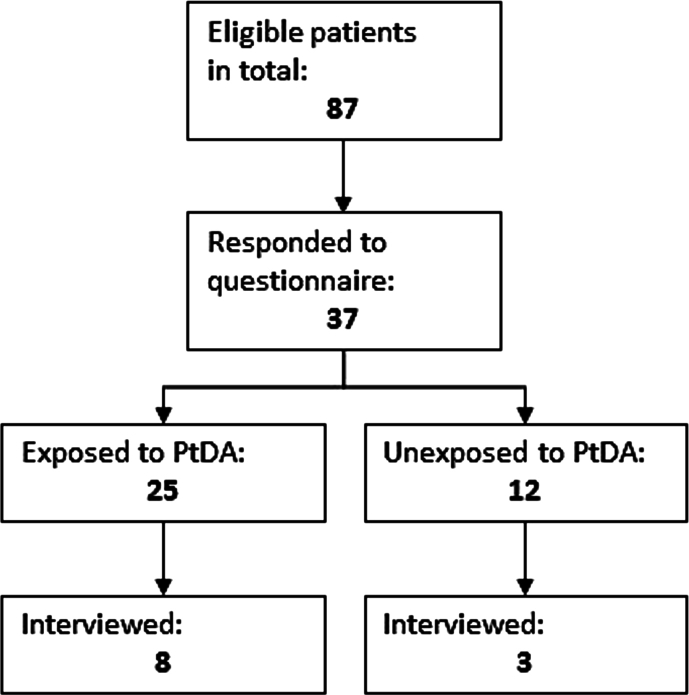
In Phase 3 a total of 87 patients with suspected PC had consultations about treatment. Out of these, 38 patients responded to the DCS questionnaire, of whom 25 patients were presented and applied the PtDA in the consultation (Fig. 2). Eleven patients participated in interviews of which eight had the PtDA applied in the consultation (Table 2b).
Fig. 2.
Flowchart of the eligible patients (n = 87) being presented/not being presented to the PtDA answering DCS (n = 38) and participating in interviews (n = 11).
Table 2b.
Demographic characteristics of patients, nurses, and surgeons in the pilot test in Phase 3.
| Patients | (n) | 38 |
|---|---|---|
| Age | median (range) | 70 (55–85) |
| Male | (n) | 15 |
| Living alone | (n) | 7 |
| Educational level | ||
| College degree | (n) | 16 |
| Vocational education | (n) | 19 |
| Unknown | (n) | ˂ 3 |
| Attended consultation alone | (n) | 8 |
| Presented for the PtDA | (n) | 25 |
| Answered the preparation letter | (n) | 24 |
| Offered Reflection time | (n) | 19 |
| Clinicians | (n) | 7 |
| Surgeons | (n) | 3 |
| Males | (n) | 3 |
| Years of experience | median⁎ | 11,5 |
| Nurses | (n) | 4 |
| Males | (n) | 0 |
| Years of experience | median⁎ | 20,5 |
Range not shown as a result of small sample size.
Patients' statements were equivocal as to whether the PtDA had the intended effect of improving SDM. Some patients found it valuable to be informed about treatment with chemotherapy as an alternative to surgery and adjuvant treatment. Other patients described that the negative outcomes in terms of implications for their QoL after surgery were much worse than they were prepared for. Patients did not recall that alternatives to surgery was discussed prior to operation. One patient stated” I thought we were to discuss the options. But he (the surgeon.) had already decided that everything had to go”. Most patients described the PtDA was handed to them at the end of the consultation without dialogue, and in these cases, patients questioned the timing of the PtDA: “If this was meant as a helping hand for me to decide, then it's far too late to hand it over for me to read at home”. The nurses did not find the PtDA to be the cornerstone of a satisfactory consultation and clarified that the consultations had changed into a more legitimate conversation about alternatives to surgery although the PtDA was not being used systematically. The surgeons explained it seemed constrained to begin the consultation asking patients in a potential existential crisis to tick a box to indicate their level of information wishes and they did not find the PtDA useful except from the cards visualising the abdominal region before and after surgery, and an overview of the treatment trajectory. A recurring argument was that the premise for the PtDA was flawed, because the prescribed treatments were not equal as surgery would always be the only potentially curative option, and oncologic treatment would only be life-extending. The surgeons found that the PtDA made the conversation rigid and dehumanizing: “The quality of the conversation declined on a human level, when you are forced to talk from a rigid structure rather than what feels right with a specific patient. What you miss is the ability to adapt your communication to the situation”.
Regarding the preparation letter the nurses described this as helpful to patients, however, patients' prepared answers or questions needed to be discussed during the consultation for the letter to be meaningful. The surgeons did not find the preparation letter useful due to the belief that patients' self-reported and the surgeon-perceived fitness for surgery often differed. Thus, to induce the risk of offering surgery to patients at high risk of complications and protracted recoveries, patient-reported information served as the sole basis for preoperative decision making. It was the surgeons' experience that most patients arrived at the consultation with a clear understanding, that surgery was the only option for them, as some fit patients had already received a booking for surgery in their digital patient app. According to the surgeons this made it difficult to openly discuss other options.
The total score of DCS was 18.23, and all sub scores were below 25. The highest sub scores were found in the Informed (23.01) and Uncertainty sub scores (20.63). The lowest sub scores were found in Support (17.85), Values Clarity (17,06), and Effective Decision (13.98). There was no difference in total or sub scores between patients guided through the PtDA and patients who were not guided through the PtDA during consultation (Table 3).
Table 3.
Results from The Decisional Conflict Scale from patients (n = 38) with potential pancreatic cancer deciding about no treatment, surgical or medical treatment.
| Low DCS scores (˂ 25) | Moderate DCS score (25–37,5) | High DCS score (>37,5) | P value * | ||
|---|---|---|---|---|---|
| Total score | |||||
| No PtDA | (n) | 6 | 7 | 0 | |
| PtDA | (n) | 15 | 10 | 0 | |
| total | (n) | 21 | 17 | 0 | P = 0.50 |
| Subscales | |||||
| Uncertainty | |||||
| No PtDA | (n) | 6 | 6 | 1 | |
| PtDA | (n) | 13 | 7 | 5 | |
| total | (n) | 19 | 13 | 6 | P = 0.38 |
| Informed | |||||
| No PtDA | (n) | 2 | 9 | 2 | |
| PtDA | (n) | 12 | 12 | 1 | |
| total | (n) | 14 | 21 | 3 | P = 0.10 |
| Value Clarity | |||||
| No PtDA | (n) | 4 | 8 | 1 | |
| PtDA | (n) | 15 | 9 | 1 | |
| total | (n) | 19 | 17 | 2 | P = 0.22 |
| Support | |||||
| No PtDA | (n) | 5 | 8 | 0 | |
| PtDA | (n) | 15 | 8 | 2 | |
| total | (n) | 20 | 16 | 2 | P = 0.26 |
| Effective decision | |||||
| No PtDA | (n) | 6 | 7 | 0 | |
| PtDA | (n) | 16 | 9 | 0 | |
| total | (n) | 22 | 16 | 0 | P = 0.32 |
| *Fisher's exact test |
The P-value refers to the difference between patients who were presented with PtDA versus patients who were not presented with PtDA in the consultations.
4. Discussion and conclusion
4.1. Discussion
The interviews with patients indicated that the PtDA made up a useful structure and provided helpful information for the decision making regarding surgery for suspected PC. On the contrary, the surgeons gave an experience of a stilted and impractical PtDA. The quantitative difference between making the choice with or without the PtDA was not significant. Combined with the finding that not all patients were presented to the PtDA, it is difficult to determine the extent to which the PtDA supports patients in their decision-making of surgery as a treatment of suspected PC. Therefore, we cannot conclude that the PtDA meets the purpose of the IPDAS criteria which is to improve the patients' criteria for making a value-based shared decision [28]. However, results from the patient interviews and from the DCS questionnaire indicate that some elements of the PtDA support the patients' decision. However, these findings have limited value when the interviews with surgeons showed that the use of the PtDA was not feasible in the present clinical context. As the format of the PtDA was validated in advance and included various mandatory elements, it was not possible to further adjust to the clinical setting as requested by surgeons. Although the generic PtDA template is designed for the consultation, it appeared that the PtDA was primarily designed for the patients and not surgeons and did not allow adjustments that made it useful for another vital user of the PtDA: the surgeons. Although we examined the patients' needs, the individual patient may have different concerns than those visualized in the PtDA.
Another challenge was that the surgeons had to assess the individual patient's needs at the start of the consultation, which the PtDA did not facilitate. That surgeons were obliged to provide balanced information, about palliative oncologic treatment and best supportive care was problematic, as these treatments were performed elsewhere. The preparation sheet was also hampered by the fact that a pathologic diagnosis was rarely available at the patient's consultation, making the information of the suspected disease by the surgeon even more delicate. Considering the relatively small patient population the time-consuming and expensive development of the disease specific PtDA appears disproportionate. It may be more meaningful using a more problem-based communication strategy such as targeted SDM described by Hargraves et al., who argue that the use of tools may never outshine the purpose of the clinical consultation to help the patient deal with a health problem [39].
The development process proposed by the IPDAS collaboration has been used, which reflects a three-step process and including four iterations. The process can be determined as a medium process of user-involvement, although for some items the process have been at a smaller level and for other items at a larger level [27]. For example, the needs assessment was not carried out with 30 participants but with 11 patients and 26 end-users in total. However, 11 participants are representative in a patient population of 250. On the other hand, we have also made observations that reflect a major process. In the development process a template was used, which has involved end-users at a major level. The surgeons did not find the PtDA useful, which may indicate that the use of a generic template has been counterproductive in meeting the context-specific decision needs of the surgeon, a vital end-user of the tool [40].
To the best of our knowledge no study has developed a PtDA for patients with suspected PC scheduled for surgery. We can therefore only compare our results with other cancer populations. Sorensen von Essen et al. [41] developed a PtDA targeting patients with high-grade glioma based on the same template and found the tool useful and acceptable in their alpha test. Further evaluation of SDM and the PtDA in a real-life decision making consultations is now being tested by Sorensen von Essen et al. However, they did not in detail describe nurses and surgeons' experiences with the tool. In our opinion this is an important aspect to investigate, as the perfect tool is useful in practice only after a development study, and if the surgeons find it attractive, meaningful, and actively use it.
Offering reflection time to the doubtful patients may be useful in some situations, but this was not possible for us to measure. As the surgeons did not favor the PtDA, it was subsequently not implemented in the clinic. Only few elements such as an overview of the treatment trajectory and abdominal illustrations of pre- and post-surgery were chosen to be a part of the future consultations. Implementation of SDM in the present clinical setting was a challenge. Steffensen et al. [42] also argue that implementing SDM in healthcare at the institutional level is challenging especially in those cases where patients have complex illness and care needs. Based on a recent review investigating situations deemed appropriate and not appropriate for SDM, the decision about surgery or not for PC is appropriate for SDM [43].
The DCS total score reflected low decisional conflict, indicating that patients were certainty with their treatment decision. The highest scores were the sub score; informed (i.e. I know which options are available to me) indicating that patients were not well informed of the available treatment options. This may be due to the inflexible use of the PtDA in the consultations. Hoesseini et al. [44] investigated decisional conflicts in patients with head and neck cancer deciding about treatment options of surgery vs. non surgery. Likewise, our study they also found the highest scores were the sub score; informed, arguing that patients did not feel they had a choice of treatment as surgery was the best treatment. The lowest sub score was found in effective decision (i.e. I am satisfied with my decision) similar to the findings of Hoesseini et al. indicating that patients with low effective decision felt they made a shared decision while patients experiencing decisional conflicts often felt the surgeon made the decision [44]. We did not find any differences in the DCS scores between patients who either were or were not presented for the PtDA. This may be due to the difference in how the individual surgeon used the PtDA in the consultation and the relatively small sample size.
A strength of this study is the assessment of the IPDAS quality criteria based on SDM concepts. Furthermore, the number of interviews performed before, during and after the implementation of the PtDA with nurses, surgeons and patients provides valuable insights into the quality of the value-based decision. There are, however, also limitations. First, we did not test the patients' uncertainty with the decision before the implementation of the PtDA, and second, the results from the DCS were not significant which might be due to the sample size. The reason for the low number of respondents may be a result of the low number of patients presented for the PtDA. We do not have long-term data on how the patients experienced the decision making after surgery. Another limitation is the lengthy and extensive development process of the PtDA, which may have caused user fatigue. It is well known that there is a need to train nurses and surgeons in communication about serious illness using a safe learning environment, explicit teaching of structured communication and preparing co-facilitators to adapt to the differing skill levels of learners [45]. However, this was not prioritized in the present study due to a combination of a reasonable reluctance to engage in further communication training with the PtDA and lack of resources given the time spent on developing the PtDA. This call was made as the small team of surgeons had all received education in the general method of SDM and all the surgeons participated in the development of the tool and as a result knew the tool in detail. Furthermore, the surgeons were given a personal review of the PtDA and how to use this in the consultation. Finally, the surgeons' approach and their presentation of the PtDA may well have influenced the patients' views of the PtDA. If the surgeon from an observation met all SDM goals, the tool is still not useful if it is not perceived or accepted as useful in the consultation.
4.2. Innovation
Patient-centered care and SDM are agendas being promoted internationally. Many resources are used promoting this important agenda. However, this study highlights the urgent need to consider the clinical context before introducing a predefined tool. In light of the current health care crisis and shortage of personal, innovation for greater SDM can only happen when all users find a given tool or practice feasible. Therefore, we strongly propose that clinicians' voices be heard even if a certain tool is validated in a different but similar context. Such involving practice will ensure that the resources used to promote true SDM will not end up in a drawer but instead help the people we want to help: the patients.
4.3. Conclusion
The limited knowledge of PtDA and SDM for patients with suspicion of PC, and the complexity of assessing the applicability of the specific PtDA in the clinical context highlights the need for further research within the field of purposeful SDM in cancer surgical settings. Overall, the PtDA seems to fill in a gap of patients' needs, but the format and structure of the PtDA was not feasible for the surgeons in the present clinical context.
The following are the supplementary data related to this article.
SUNDAE Checklist for evaluation studies of patient decision aids.
Assessment of the International Patient Decision Aids Standards.
Formatting of funding sources
This study was funded by Danish national government funds to promote the use of SDM tools within cancer care. The funders had no role in the design or the conduct of the study: collection, management, analysis, interpretation of the data, preparation, review of approval of the manuscript or finally the decision to submit the manuscript for publication.
Author contributions
KD, AB, JH, BMC were responsible for the conception of the study design. KD, CPH, and AB drafted the final manuscript and JF provided academic supervision. All authors were involved in editing of the manuscript and have read and approved the final paper.
Informed content statement
Written informed consent has been obtain form all participants to publish this paper.
CRediT authorship contribution statement
Kristine Elberg Dengsø: Writing – review & editing, Writing – original draft, Visualization, Validation, Resources, Project administration, Methodology, Investigation, Formal analysis, Data curation. Anne Berg: Writing – review & editing, Visualization, Project administration, Methodology, Investigation, Formal analysis, Data curation, Conceptualization. Carsten Palnæs Hansen: Writing – review & editing. Stefan K. Burgdorf: Writing – review & editing. Paul S. Krohn: Writing – review & editing. Martin Sillesen: Writing – review & editing. Nina Spiegelhauer: Resources. Mette Tholstrup Bach: Investigation. Marianne Melton: Resources. Betina Nielsen: Resources. Bo Marcel Christensen: Writing – review & editing, Resources. Jeanette Finderup: Writing – review & editing, Supervision, Methodology, Conceptualization. Jens Hillingsø: Writing – review & editing, Supervision.
Declaration of competing interest
None.
Acknowledgements
We would like to thank all the patients and relatives who took their time to contribute to this study.
Contributor Information
Kristine Elberg Dengsø, Email: kristine.elberg.dengsoe@regionh.dk.
Anne Berg, Email: anne.berg.villumsen@regionh.dk.
Carsten Palnæs Hansen, Email: Carsten.Palnaes.Hansen@regionh.dk.
Stefan K. Burgdorf, Email: stefan.kobbelgaard.burgdorf.02@regionh.dk.
Paul S. Krohn, Email: paul.suno.krohn@regionh.dk.
Martin Sillesen, Email: martin.hylleholt.sillesen@regionh.dk.
Nina Spiegelhauer, Email: Nina.Spiegelhauer@regionh.dk.
Mette Tholstrup Bach, Email: Mette.Tholstrup.Bach@regionh.dk.
Marianne Melton, Email: Marianne.Melton@regionh.dk.
Betina Nielsen, Email: betina.marie.foght.nielsen@regionh.dk.
Bo Marcel Christensen, Email: Bo.Marcel.Christensen@regionh.dk.
Jeanette Finderup, Email: Jeanette.Finderup@rm.dk.
Jens Hillingsø, Email: Jens.Hillingsoe@regionh.dk.
Data availability
Data cannot be made open because of the relatively small sample size.
References
- 1.Ratnayake B., Pendharkar S.A., Connor S., Koea J., Sarfati D., Dennett E., et al. Patient volume and clinical outcome after pancreatic cancer resection: a contemporary systematic review and meta-analysis. Surgery. 2022;172(1):273–283. doi: 10.1016/j.surg.2021.11.029. [DOI] [PubMed] [Google Scholar]
- 2.Michl P., Lohr M., Neoptolemos J.P., Capurso G., Rebours V., Malats N., et al. UEG position paper on pancreatic cancer. Bringing pancreatic cancer to the 21st century: prevent, detect, and treat the disease earlier and better. United European Gastroenterol J. 2021;9(7):860–871. doi: 10.1002/ueg2.12123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.McGuigan A., Kelly P., Turkington R.C., Jones C., Coleman H.G., McCain R.S. Pancreatic cancer: a review of clinical diagnosis, epidemiology, treatment and outcomes. World J Gastroenterol. 2018;24(43):4846–4861. doi: 10.3748/wjg.v24.i43.4846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Schniewind B., Bestmann B., Henne-Bruns D., Faendrich F., Kremer B., Kuechler T. Quality of life after pancreaticoduodenectomy for ductal adenocarcinoma of the pancreatic head. Br J Surg. 2006;93(9):1099–1107. doi: 10.1002/bjs.5371. [DOI] [PubMed] [Google Scholar]
- 5.Crippa S., Dominguez I., Rodriguez J.R., Razo O., Thayer S.P., Ryan D.P., et al. Quality of life in pancreatic cancer: analysis by stage and treatment. J Gastrointest Surg. 2008;12(5):783–793. doi: 10.1007/s11605-007-0391-9. discussion 793–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Nieveen van Dijkum E.J., Kuhlmann K.F., Terwee C.B., Obertop H., de Haes J.C., Gouma D.J. Quality of life after curative or palliative surgical treatment of pancreatic and periampullary carcinoma. Br J Surg. 2005;92(4):471–477. doi: 10.1002/bjs.4887. [DOI] [PubMed] [Google Scholar]
- 7.Heerkens H.D., Tseng D.S., Lips I.M., van Santvoort H.C., Vriens M.R., Hagendoorn J., et al. Health-related quality of life after pancreatic resection for malignancy. Br J Surg. 2016;103(3):257–266. doi: 10.1002/bjs.10032. [DOI] [PubMed] [Google Scholar]
- 8.Eaton A.A., Gonen M., Karanicolas P., Jarnagin W.R., D’Angelica M.I., DeMatteo R., et al. Health-related quality of life after pancreatectomy: results from a randomized controlled trial. Ann Surg Oncol. 2016;23(7):2137–2145. doi: 10.1245/s10434-015-5077-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Brown S.R., Mathew R., Keding A., Marshall H.C., Brown J.M., Jayne D.G. The impact of postoperative complications on long-term quality of life after curative colorectal cancer surgery. Ann Surg. 2014;259(5):916–923. doi: 10.1097/SLA.0000000000000407. [DOI] [PubMed] [Google Scholar]
- 10.Elberg Dengso K., Tjornhoj-Thomsen T., Oksbjerg Dalton S., Marcel Christensen B., Hillingso J., Thomsen T. It's all about the CA-19-9. A longitudinal qualitative study of patients' experiences and perspectives on follow-up after curative surgery for cancer in the pancreas, duodenum or bile-duct. Acta Oncol. 2019;58(5):642–649. doi: 10.1080/0284186X.2018.1562212. [DOI] [PubMed] [Google Scholar]
- 11.Dengso K.E., Andersen E.W., Thomsen T., Hansen C.P., Christensen B.M., Hillingso J., et al. Increased psychological symptom burden in patients with pancreatic cancer: a population-based cohort study. Pancreatology. 2020;20(3):511–521. doi: 10.1016/j.pan.2020.01.001. [DOI] [PubMed] [Google Scholar]
- 12.Dengso K.E., Thomsen T., Andersen E.W., Hansen C.P., Christensen B.M., Hillingso J., et al. The psychological symptom burden in partners of pancreatic cancer patients: a population-based cohort study. Support Care Cancer. 2021;29(11):6689–6699. doi: 10.1007/s00520-021-06251-4. [DOI] [PubMed] [Google Scholar]
- 13.Dengso K.E., Tjornhoj-Thomsen T., Dalton S.O., Christensen B.M., Hillingso J., Thomsen T. Gut disruption impairs rehabilitation in patients curatively operated for pancreaticoduodenal cancer - a qualitative study. BMC Cancer. 2018;18(1):1017. doi: 10.1186/s12885-018-4933-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Adham M., Bredt L.C., Robert M., Perinel J., Lombard-Bohas C., Ponchon T., et al. Pancreatic resection in elderly patients: should it be denied? Langenbecks Arch Surg. 2014;399(4):449–459. doi: 10.1007/s00423-014-1183-9. [DOI] [PubMed] [Google Scholar]
- 15.Makoul G., Clayman M.L. An integrative model of shared decision making in medical encounters. Patient Educ Couns. 2006;60(3):301–312. doi: 10.1016/j.pec.2005.06.010. [DOI] [PubMed] [Google Scholar]
- 16.Elwyn G., Frosch D., Thomson R., Joseph-Williams N., Lloyd A., Kinnersley P., et al. Shared decision making: a model for clinical practice. J Gen Intern Med. 2012;27(10):1361–1367. doi: 10.1007/s11606-012-2077-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Shay L.A., Lafata J.E. Where is the evidence? A systematic review of shared decision making and patient outcomes. Med Decis Making. 2015;35(1):114–131. doi: 10.1177/0272989X14551638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Joosten E.A., DeFuentes-Merillas L., de Weert G.H., Sensky T., van der Staak C.P., de Jong C.A. Systematic review of the effects of shared decision-making on patient satisfaction, treatment adherence and health status. Psychother Psychosom. 2008;77(4):219–226. doi: 10.1159/000126073. [DOI] [PubMed] [Google Scholar]
- 19.Shinkunas L.A., Klipowicz C.J., Carlisle E.M. Shared decision making in surgery: a scoping review of patient and surgeon preferences. BMC Med Inform Decis Mak. 2020;20(1):190. doi: 10.1186/s12911-020-01211-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.de Mik S.M.L., Stubenrouch F.E., Balm R., Ubbink D.T. Systematic review of shared decision-making in surgery. Br J Surg. 2018;105(13):1721–1730. doi: 10.1002/bjs.11009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Legare F., Ratte S., Gravel K., Graham I.D. Barriers and facilitators to implementing shared decision-making in clinical practice: update of a systematic review of health professionals’ perceptions. Patient Educ Couns. 2008;73(3):526–535. doi: 10.1016/j.pec.2008.07.018. [DOI] [PubMed] [Google Scholar]
- 22.Niburski K., Guadagno E., Abbasgholizadeh-Rahimi S., Poenaru D. Shared decision making in surgery: a Meta-analysis of existing literature. Patient. 2020;13(6):667–681. doi: 10.1007/s40271-020-00443-6. [DOI] [PubMed] [Google Scholar]
- 23.Ziebland S., Chapple A., Evans J. Barriers to shared decisions in the most serious of cancers: a qualitative study of patients with pancreatic cancer treated in the UK. Health Expect. 2015;18(6):3302–3312. doi: 10.1111/hex.12319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Geessink N.H., Ofstad E.H., Olde Rikkert M.G.M., van Goor H., Kasper J., Schoon Y. Shared decision-making in older patients with colorectal or pancreatic cancer: determinants of patients’ and observers’ perceptions. Patient Educ Couns. 2018;101(10):1767–1774. doi: 10.1016/j.pec.2018.06.005. [DOI] [PubMed] [Google Scholar]
- 25.Geessink N.H., Schoon Y., van Herk H.C., van Goor H., Olde Rikkert M.G. Key elements of optimal treatment decision-making for surgeons and older patients with colorectal or pancreatic cancer: a qualitative study. Patient Educ Couns. 2017;100(3):473–479. doi: 10.1016/j.pec.2016.10.013. [DOI] [PubMed] [Google Scholar]
- 26.Stacey D., Legare F., Lewis K., Barry M.J., Bennett C.L., Eden K.B., et al. Decision aids for people facing health treatment or screening decisions. Cochrane Database Syst Rev. 2017;4:CD001431. doi: 10.1002/14651858.CD001431.pub5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Witteman H.O., Maki K.G., Vaisson G., Finderup J., Lewis K.B., Dahl Steffensen K., et al. Systematic development of patient decision aids: an update from the ipdas collaboration. Med Decis Making. 2021;41(7):736–754. doi: 10.1177/0272989X211014163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Elwyn G., O'Connor A., Stacey D., Volk R., Edwards A., Coulter A., et al. Developing a quality criteria framework for patient decision aids: online international Delphi consensus process. BMJ. 2006;333(7565):417. doi: 10.1136/bmj.38926.629329.AE. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Hoefel L., O’Connor A.M., Lewis K.B., Boland L., Sikora L., Hu J., et al. 20th anniversary update of the Ottawa decision support framework part 1: a systematic review of the decisional needs of people making health or social decisions. Med Decis Making. 2020;40(5):555–581. doi: 10.1177/0272989X20936209. [DOI] [PubMed] [Google Scholar]
- 30.Kvale S.B. 2015. S., Interview. Det kvalitative forskningsinterview somhåndværk, Gyldendal. [Google Scholar]
- 31.Halkier B. 2016. Fokusgrupper, Specialtrykkeriet Viborg A/S. [Google Scholar]
- 32.Elwyn G., Tsulukidze M., Edwards A., Legare F., Newcombe R. Using a ‘talk’ model of shared decision making to propose an observation-based measure: observer OPTION 5 item. Patient Educ Couns. 2013;93(2):265–271. doi: 10.1016/j.pec.2013.08.005. [DOI] [PubMed] [Google Scholar]
- 33.in: D.o.H.s.N.S.D.M. programme (Ed.) National Health Service, United Kingdom. 2012. Measuring shared decision making. [Google Scholar]
- 34.Dahl Steffensen K., Hjelholt Baker V., Vinter M.M. Implementing shared decision making in Denmark: first steps and future focus areas. Z Evid Fortbild Qual Gesundhwes. 2017;123–124:36–40. doi: 10.1016/j.zefq.2017.05.005. [DOI] [PubMed] [Google Scholar]
- 35.Braun V., Clarke V. Using thematic analysis in psychology. Qual Res Psychol. 2006;3(2):77–101. [Google Scholar]
- 36.O'Connor A. 1993. User manual - Decisional Conflict Scale. https://decisionaid.ohri.ca/docs/develop/User_Manuals/UM_decisional_conflict.pdf. 1993) [Google Scholar]
- 37.I. World Medical Association . 2017. WMA Declaration of Helsinki - Ethical Principles for Medical Research Involving Human Subjects. http://www.wma.net/en/30publications/10policies/b3/. (Accessed 23. march 2017) [PubMed] [Google Scholar]
- 38.Schildmann J., Ritter P., Salloch S., Uhl W., Vollmann J. ’One also needs a bit of trust in the doctor … ’: a qualitative interview study with pancreatic cancer patients about their perceptions and views on information and treatment decision-making. Ann Oncol. 2013;24(9):2444–2449. doi: 10.1093/annonc/mdt193. [DOI] [PubMed] [Google Scholar]
- 39.Hargraves I.G., Montori V.M., Brito J.P., Kunneman M., Shaw K., LaVecchia C., et al. Purposeful SDM: a problem-based approach to caring for patients with shared decision making. Patient Educ Couns. 2019;102(10):1786–1792. doi: 10.1016/j.pec.2019.07.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Stacey D., Suwalska V., Boland L., Lewis K.B., Presseau J., Thomson R. Are patient decision aids used in clinical practice after rigorous evaluation? A survey of trial authors. Med Decis Making. 2019;39(7):805–815. doi: 10.1177/0272989X19868193. [DOI] [PubMed] [Google Scholar]
- 41.Sorensen von Essen H., Poulsen F.R., Dahlrot R.H., Piil K., Steffensen K.D. Development of a patient decision aid to support shared decision making for patients with recurrent high-grade glioma. Int J Environ Res Public Health. 2022;19(12) doi: 10.3390/ijerph19127396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Steffensen K.D., Vinter M., Cruger D., Dankl K., Coulter A., Stuart B., et al. Lessons in integrating shared decision-making into cancer care. J Oncol Pract. 2018;14(4):229–235. doi: 10.1200/JOP.18.00019. [DOI] [PubMed] [Google Scholar]
- 43.van der Horst D.E.M., Garvelink M.M., Bos W.J.W., Stiggelbout A.M., Pieterse A.H. For which decisions is shared decision making considered appropriate? - a systematic review. Patient Educ Couns. 2023;106:3–16. doi: 10.1016/j.pec.2022.09.015. [DOI] [PubMed] [Google Scholar]
- 44.Hoesseini A., Dorr M.C., Dronkers E.A.C., de Jong R.J.B., Sewnaik A., Offerman M.P.J. Decisional conflict in patients with head and neck cancer. JAMA Otolaryngol Head Neck Surg. 2023;149(2):160–167. doi: 10.1001/jamaoto.2022.4269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Daubman B.R., Bernacki R., Stoltenberg M., Wilson E., Jacobsen J. Best practices for teaching clinicians to use a serious illness conversation guide. Palliat Med Rep. 2020;1(1):135–142. doi: 10.1089/pmr.2020.0066. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
SUNDAE Checklist for evaluation studies of patient decision aids.
Assessment of the International Patient Decision Aids Standards.
Data Availability Statement
Data cannot be made open because of the relatively small sample size.