Abstract
Hyperammonemia and liver disease are closely linked. Most of the ammonia in our body is produced by transamination and deamination activities involving amino acid, purine, pyrimidines, and biogenic amines, and from the intestine by bacterial splitting of urea. The only way of excretion from the body is by hepatic conversion of ammonia to urea. Hyperammonemia is associated with widespread toxicities such as cerebral edema, hepatic encephalopathy, immune dysfunction, promoting fibrosis, and carcinogenesis. Over the past two decades, it has been increasingly utilized for prognostication of cirrhosis, acute liver failure as well as acute on chronic liver failure. The laboratory assessment of hyperammonemia has certain limitations, despite which its value in the assessment of various forms of liver disease cannot be negated. It may soon become an important tool to make therapeutic decisions about the use of prophylactic and definitive treatment in various forms of liver disease.
Keywords: ammonia, acute liver failure, cACLD (compensated advanced chronic liver disease), MASLD (metabolic dysfunction-associated steatotic liver disease), HE (hepatic encephalopathy)
The word ‘Ammonia’ has possibly been derived from ‘Amen’ an Egyptian god, with a mythical concept that life arose from a sea of ammonia.1 The earliest scientific inquiry on the impact of portocaval shunt in dogs was described in 1893, although ammonia was not mentioned as the perpetrator.2 Behavior alterations in cirrhosis were attributed to nitrogenous substances in diet in 1952, and the first evidence linking ammonia in the pathogenesis of hepatic encephalopathy(HE) is possibly attributed to Lockwood we al.3 Since then, a huge body of evidence has accumulated to reveal the important role of ammonia in pathogenesis of various problems in chronic liver disease. This review will highlight the basic physiology of ammonia in health and chronic liver disease and some specific aspects of its clinical implications.
BASIC PHYSIOLOGY
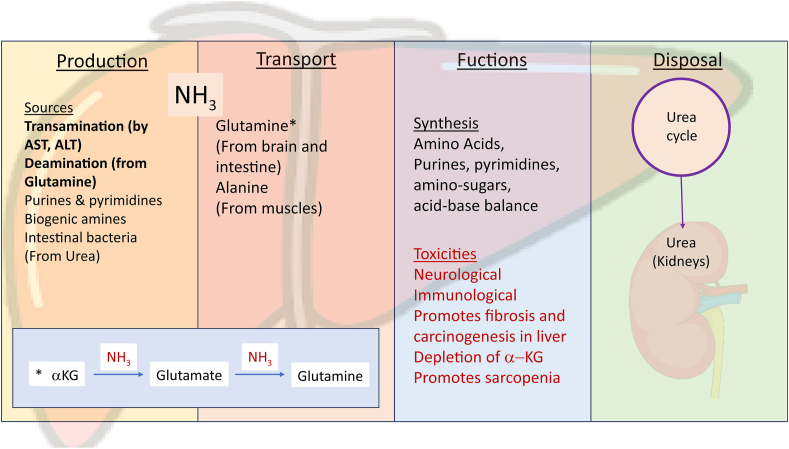
The basic physiology of ammonia is summarized in Figure 1. In the human system, ammonia exists as an ammonium ion (NH4+) at the physiological pH. Most of the body's ammonia is produced through transamination and deamination reactions from various amino acids, other biogenic amines, purines, and pyrimidines, and to some extent from intestinal bacteria that split the urea.4 The ammonia produced in the peripheral tissues is transported to the liver mostly as glutamine from the brain, intestines, and other organs and as alanine from the muscles. Glutamine is the main molecule that stores ammonia and is synthesized (glutamine synthetase is available in all tissues) by adding one ammonia molecule to glutamate.5 Ammonia is required for several vital functions such as functions synthesis of various amino acids, purines, and pyrimidines, and helps in acid-base balance regulation by the kidneys. Acidosis induces kidneys to produce and extract ammonia from glutamine via glutaminase in the proximal tubules of the kidneys. Ammonium ion (NH4+) and potassium ion (K+) have similar biophysical characteristics in aqueous solutions, and they can be effectively transported at the transport site of potassium ions.6 Excess ammonia is excreted to produce bicarbonate.7 In chronic kidney disease (CKD), an adequate quantity of ammonia cannot be produced and excreted leading to the retention of acid and the formation of metabolic acidosis.8
Figure 1.
Physiology of ammonia, showing it production, transport, functions and disposal. Major sources of circulating ammonia are deamination and transamination activities in liver and rest of the body. Over two-thirds of it is in form of glutamine (from most sources) and alanine (only from muscles). Glutamate picks mops up ammonia under the influence of glutamine synthase at brain, muscles & liver. Glutamine released ammonia under the influence of glutaminase at intestines, liver and kidneys. Ammonia is disposed off as urea through ornithine cycle (Urea cycle) in liver and urea is excreted mainly in kidneys. aKG, alpha-keto-glutarate; AST, asparate aminotransferase; ALT, alanine aminotransferase.
The final disposal of ammonia from the body is by the liver through the ornithine cycle (urea cycle) which converts it to urea to be excreted through kidneys. The gastrointestinal system is also an important organ in ammonia metabolism. Intestines play a secondary role in the excretion of urea. Besides, glutamine is an important nutrient substrate for the intestine mostly the jejunum and ileum and to some extent the large intestine.9 High glutaminase activity in small intestine mucosa produces glutamate and ammonia from glutamine. The large intestine contributes significantly to portal venous ammonia concentration by the bacterial splitting of urea and amino acids.10 (Figure 2).
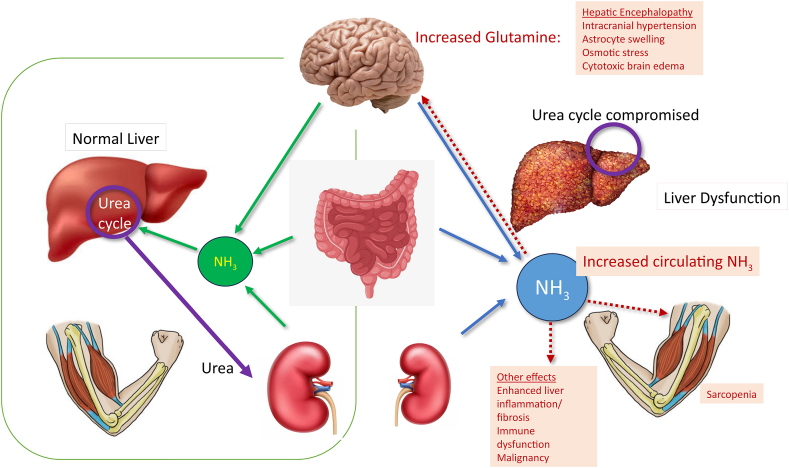
Figure 2.
A cartoon depicting dynamics of ammonia in our body. Left half of the diagram shows normal physiology enclosed in a green line, while right half shows the consequences of cirrhosis and increased circulating ammonia levels. Its untoward consequences are shown with dotted arrows in the red color.
ALTERED PHYSIOLOGY IN CIRRHOSIS
In liver disease, circulating ammonia levels are high due to three factors. First, Ammonia clearance by the urea cycle is reduced (10%–90%) as compared to normal capacity, and glutamine synthesis is also similarly reduced.11,12 Second, portosystemic circulation allows ammonia to bypass the liver.13 Hyperammonemia can be induced by a protein-rich diet in patients with the trans-jugular intrahepatic portosystemic shunt (TIPS).14 Last, Kidneys also release ammonia in circulation, more so under the influence of diuretics such as acetazolamide and chlorothiazides.15,16 Increased hyperammonemia has also been reported after hyperventilation and during the an-hepatic phase of liver transplantation.5,17 Muscles act as a sponge in situations of hyperammonemia by converting Glutamate to glutamine.18 But this capacity is reduced in cirrhosis due to associated sarcopenia.19 Muscle exercise in cirrhosis also produces greater ammonia.20
CAUSES OF HYPERAMMONEMIA
While hyperammonemia (elevated ammonia concentration in systemic circulation greater than or equal to 65 μmol) is common in chronic liver disease, it can also be seen in a variety of conditions (Table 1). The exact mechanism and management of all the conditions other than liver disease are outside the scope of this paper and hence are summarized in the table. The causal relationship of hyperammonemia and muscle wasting in cirrhosis has been suggested by many studies.21 Alcohol tends to aggravate this effect.22 Hyperammonemia of cirrhosis can also be aggravated by hemorrhagic shock. Bleeding can reduce hepatic blood flow leading to ischemia in the periportal to the centrilobular hepatocytes, and subsequent necrosis. The site of glutamine synthesis is ‘pericentral’ hepatocyte while the urea synthesis is mainly the function of ‘periportal’ hepatocyte.23
Table 1.
Causes of Hyperammonemia.
| Causes | Remarks |
|---|---|
|
The Liver is the main site where ammonia is detoxified into urea with the help of the urea cycle. Liver dysfunction is associated with compromise in its capacity to eliminate ammonia. See above in text: ‘Altered physiology in cirrhosis’ |
|
Both portal hypertension and portosystemic shunting contribute to hyperammonemia.24 |
|
Ammonia is converted to urea in the liver through a sequential enzymatic reaction (Figure 3) Inherited deficiency of any one of the enzymes involved leads to accumulation of ammonia in the body and a series of adverse effects related to it. See text above: ‘Urea cycle disorders.’ |
|
Ammonia is produced by renal epithelial cells predominantly in the proximal tubules and play an important role in renal acid base regulation.25,26 In renal failure, hyperammonemia is common and the uremic odor in breath is largely due to ammonia content.27 |
|
It is a rare metabolic disease resulting from recessive-inherited mutations in the SLC7A7 gene. It is characterized by protein-rich food intolerance with secondary urea cycle disorder, but symptoms are heterogeneous ranging from infiltrative lung disease, kidney failure to auto-immune complications. |
|
Carnitine binds fatty acyl-CoA residues and promotes their translocation from the cytoplasm into the mitochondrial matrix, where β-oxidation and generation of energy occur. Disruption of the carnitine transport system results in the cytosolic accumulation of unoxidized fatty acyl-CoA molecules. These metabolites are believed to inhibit the urea cycle, thereby impairing an important mechanism of ammonia excretion.28 |
|
A rare disorder, often presents in infants during an infection, with poor oral intake, vomiting, dehydration, lethargy, hypoglycemia, seizures, and a presentation similar to Reye syndrome, leading to death from brain edema and hyperammonemia.29 |
|
Valproate consumption may be associated with hyperammonemic encephalopathy (VHE) characterized by a decreasing level of consciousness, focal neurological deficits, cognitive slowing, vomiting, drowsiness, and lethargy. Hyperammonemia may be multifactorial, though the main reason may be the inhibition of carbamoyl phosphate synthetase-I, the first step of the urea cycle. Hyperammonemia reduces after VPA withdrawal.30 |
|
Organic acidemias are a group of disorders that lead to the detection of organic acids in the urine (or plasma). Organic acids (OAs) are intermediary products of several amino acid catabolism or degradation and classic OAs include propionic, methylmalonic isovaleric, glutaric acid, and ketogenic/ketolytic acids. Typically they present in neonates or infants with hyperammonemia and encephalopathy.31 |
|
Reye syndrome is a rare disease presenting as acute encephalopathy related to hyperammonemia and liver dysfunction possibly related to viral pathogens, fatty liver, and aspirin consumption. It is a medical emergency and patients should considered for hemodialysis if arterial ammonia levels are >150 μmol/L.32 |
|
Hyperammonemia has been described with urinary tract infection, urinary obstruction as well as septic shock due to sepsis by urea-splitting microorganisms.33,34 Even encephalopathy due to such infections has been described.35 |
|
Hyperammonemia can occur after chemotherapy in about 2.4% of cases after a few weeks of starting therapy. Pathogenesis is likely to be multifactorial. Some authors have blamed the deamination of Cytarabine or restriction of Krebs cycle by-products of 5-FU or other drugs metabolism.36 |
|
Hyperammonemia after lung transplantation (HALT) occurs in 1%–4% of cases and carries a high mortality. The exact pathophysiology of this complication has not been clearly elucidated.37 |
|
Barth syndrome is a rare X-linked genetic disorder of male infants that presents with cardiomyopathy, skeletal myopathy, neutropenia, 3-methylglutaconic aciduria, and hypercholesterolemia and hyperammonemia.38 |
|
Pyruvate carboxylase (PC) is a mitochondrial enzyme that converts pyruvate and CO2 to oxaloacetate for the Krebs cycle. Clinical presentation may occur in infancy (type A), neonates (type B), or later life (type C). The presentation may be with lactic acidosis, ketoacidosis, hyperammonemia, severe retardation, failure to thrive, pyramidal tract signs, ataxia, and convulsions.39 |
|
Primary pyruvate dehydrogenase complex deficiency (PDCD) is also a mitochondrial disorder and mostly affects the brain. Basic pathophysiology involves decreased ATP production and energy deficit and functional deficiency of carbamoyl phosphate synthetase. Affected children present often between 2 and 4 years of age with ataxia and peripheral neuropathy, growth retardation, hypotonia, microcephaly, seizures, lactic acidosis and sometimes hyperammonemia.40 |
|
The hyperinsulinism/hyperammonemia (HI/HA) syndrome is an autosomal dominant disorder. Children affected by this syndrome have both fasting and protein sensitive hypoglycemia combined with persistently elevated ammonia levels.41 |
|
In distal renal tubular acidosis, hyperammonemia is due to the increased ammonia synthesis, in response to metabolic acidosis, and the impaired ammonia excretion, typical of distal renal tubular acidosis.42 |
|
In ureterosigmoidostomy, hyperammonemia may be multifactorial with a major contributor being exposure of the colonic bacteria to urine. Intermittent hyperammonemic encephalopathy can occur decades after ureterosigmoidostomy.43 |
|
Hyperammonemia is possibly related to catabolism of glycine absorbed during the procedure. Factors other than glycine may also be operative.44 |
|
Hyperammonemia is well described with parenteral nutrition.45 A long list of drugs have been described to associated with hyperammonemia either due to increased production of ammonia or due to compromised elimination of ammonia.46 |
|
Hyperammonemia is being increasingly recognized as a result of refeeding after starvation and may even lead to encephalopathy (Food-Coma).47,48 |
ATP, adenosine triphosphate; OAs, Organic acids; PC, Pyruvate carboxylase; PDCD, Primary pyruvate dehydrogenase complex deficiency; HI/HA, hyperinsulinism/hyperammonemia; VHE, Valproate induced hepatic encephalopathy; VPA, Valproate.
Urea Cycle Disorders
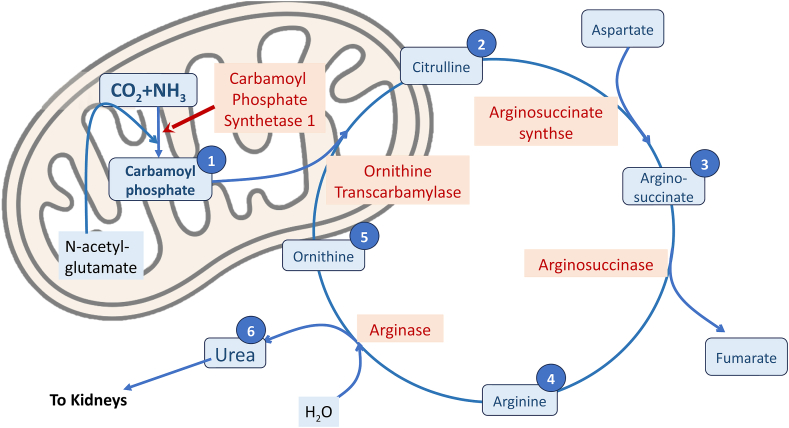
The inheritable disorders of the urea cycle are not uncommon. The urea cycle (Figure 3) in the liver is the body's main mechanism of clearing nitrogenous waste produced during the amino acid metabolism. The cycle49 consists of the sequential action of five catalytic enzymes namely Carbamoyl phosphate synthetase I (CPS1), Ornithine transcarbamylase (OTC), Argininosuccinic acid synthetase (ASS1), Argininosuccinic acid lyase (ASL) and Arginase (ARG1). It also involves two amino acid transporters (Ornithine translocase or ORNT1 and Citrin) and a co-factor-producing enzyme i.e. N-acetyl glutamate synthetase (NAGS). Inherited deficiencies of any one of the above enzymes or transporters leads to a urea cycle disorder. Clinical presentation is dependent on the degree of deficiency and the site where the cycle is interrupted.
Figure 3.
A simplified scheme of urea cycle in the liver. (1) Ammonia combines with carbon di oxide with allosteric activator ORNT-1, N-acetyl- glutamate to form Carbamoyl phosphate in the mitochondria. (2) Carbamoyl phosphate combines with ornithine and is converted to citrulline, which moves to cytosol with the help of ORNT-1. (3) Citrulline combines with aspartate to form arginosuccinate. Steps (1) and (3) are energy consuming processes requiring ATP. (4) Arginosuccinate gives off fumarate to form Arginine. (5) Arginine combines with water to form ornithine and (6) Urea that is transported to kidneys to be excreted. To complete the cycle, Ornithine moves back to mitochondria through ORNT-1 to take part in urea cycle at step (2). Five enzymes that catalyze these five steps of urea cycle are Carbamoyl phosphate synthetase-1, ornithine transcarbamylase, arginosuccinate synthetase, Argino-succinase and Arginase respectively. ORNT-1: ornithine transporter.
Infants with a urea cycle disorder appear normal at birth but within a few days/weeks develop cerebral edema and the related signs of lethargy, anorexia, hyper- or hypoventilation, hypothermia, seizures, neurologic posturing, and coma. Milder cases may present in later life and are often precipitated by a critical illness or a period of stress such as a peripartum period, surgery, or prolonged fasting. A full discussion on screening, diagnosis, and management of urea cycle defects is beyond the scope of this paper and has been reviewed elsewhere.50, 51, 52, 53, 54, 55, 56
NEUROTOXICITY OF AMMONIA
Ammonia readily traverses the blood–brain barrier (BBB) with a positive arterial–venous gradient suggesting net brain ammonia uptake. The brain contains appreciable amounts of both glutamine synthetase (GS, mostly in astrocytes) and glutaminase enzymes (mostly in neurons).57 Astrocyte GS preferentially takes up ammonia to form glutamine, which is de-aminated to form GABA and glutamate in neurons.
Acute exposure to high blood ammonia activates NMDA (N-methyl-d-aspartate) receptors in the brain. A series of consequences are set in involving depletion of brain ATP, impairment of mitochondrial function and calcium homeostasis at different levels, and increased formation of nitric oxide (NO) formation.58 This leads to Impaired bioenergetics and neurotransmission, astrocyte swelling, alteration of key astrocyte proteins, and increased oxidative and mitochondrial dysfunction. ATP depletion is responsible for the aggravation of ammonia toxicity and is the most probable cause of seizures. This stage is set for osmotic as well as cytotoxic cerebral edema. A Major component of cerebral edema is swelling of astrocytes which are the only cells involved in ammonia detoxification in the brain. The astrocyte swelling is related to altered water and K+ metabolism in the astrocytes, activation of tumor suppressor protein p53, and increased uptake of certain compounds including pyruvate, lactate, and glutamine and decreased uptake of ketone bodies, glutamate, and free glucose. The direct consequence of this event is raised intracranial pressure, which may result in brain herniation.59
Chronic rise in ammonia levels increases the transport of tryptophan, across the blood–brain barrier followed by a resultant increase in serotonin levels in the brain, which causes anorexia in these patients. There is also increased GABAergic tone due to Benzodiazepine receptor overstimulation by endogenous benzodiazepines and neurosteroids. In addition, there is a downregulation of glutamate receptors due to increased extra-synaptic glutamate accumulation. Changes in the glutamate-nitric oxide-cGMP pathway result in impaired signal transmission in the N-methyl-d-aspartate (NMDA) receptors. This contributes to the cognitive dysfunction seen in hepatic encephalopathy.60 The role of inflammation, sepsis, and other mechanisms have been reviewed elsewhere.61
Hyperammonemia, Muscles, and Liver Disease
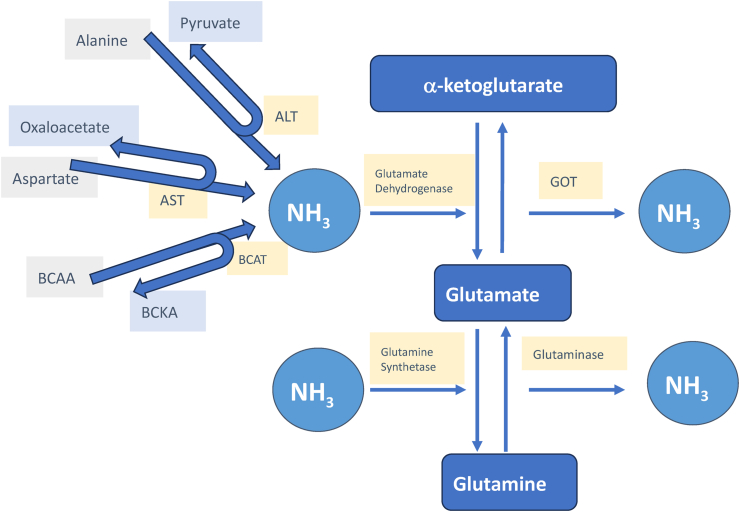
Skeletal muscles are closely associated with ammonia metabolism and this relationship becomes more important in hyperammonemia states. Muscles form the main reservoir of protein in the body. In the postprandial state, Glucose is converted into amino acid (alanine) in the liver and transported to be stored in muscles. In the fasting state, alanine is broken down to glucose to be transported to the liver for energy generation. This is called the alanine cycle or Cahill cycle.62 Muscles, one of the largest organs of the body, contain a large amount of glutamate made from alanine, aspartate, or BCAA and can soak up additional ammonia, especially in hyperammonemia states, to form glutamine63 (see Figure 4). Glutaminase activity in muscles is much lower than glutamine synthetase activity. The pathophysiological relationship between sarcopenia (loss of muscle mass) and Hepatic encephalopathy (HE) is well recognized and is likely linked to the impaired capacity of muscles to buffer hyperammonemia.
Figure 4.
Glutamate, a non-essential amino acid is the key molecule in ammonia regulation. It is formed by combining one ammonium radical with a-ketoglutarate under the influence of enzyme Glutamate Dehydrogenase. Ammonia is derived from amino acids under the influence of respective transaminases. Glutamate can accept one more ammonia molecule to form Glutamine (enzyme Glutamine Synthetase). Glutamine can be converted back to Glutamate and to a-ketoglutarate by action of glutaminase and Glutamate oxalacetate transaminase respectively. Muscles do contain some glutamine synthetase but hardly any glutaminase. Enzymes are shown in yellow background. ALT: alanine transaminase, AST: aspartate transaminase, BCAA: branched chain amino acids, BCKA: branched chain keto acids, GOT: Glutamate oxalacetate transaminase, NH3: ammonia.
Hyperammonemia and muscles have a reciprocal relationship. While hyperammonemia can result from intense muscle activity,64 hyperammonemia from other causes such as chronic liver disease also leads to loss of muscles.65 Accumulation of ammonia inhibits the translation of mRNA and protein synthesis in the skeletal muscle through inhibition of mTORC1.66 Since alpha keto-glutarate is consumed in the production of glutamate, the tricarboxylic acid cycle is compromised leading to loss of ATP, mitochondrial dysfunction, reduction of contractile function and finally to sarcopenia.67 Ammonia activates myostatin which further inhibits protein synthesis.68
And last, it enhances autophagy69 in cirrhotic patients with the ultimate effect being progressive sarcopenia in chronic liver disease. Sarcopenia is, therefore, common in cirrhosis (prevalence 30%–70%)70 and is attributed to higher ammonia levels.67 It has been demonstrated that sarcopenia increases mortality in cirrhotic patients,71 and also increases the risk of several complications of liver cirrhosis, such as ascites, spontaneous bacterial peritonitis (SBP), variceal bleeding, hepatocellular carcinoma, and infections. The relationship of sarcopenia to hepatic encephalopathy is now well accepted.72
Sarcopenia has been linked to MASLD progression and fibrosis development on liver biopsy.73,74 While hyperammonemia of chronic liver disease has been considered as a major causative factor contributing to the development of sarcopenia,75 the reverse also seems to be true. Korean sarcopenic obesity study shows that obese people with sarcopenia are more likely to develop sarcopenia.73 A meta-analysis of studies has shown that among patients with MASLD, sarcopenia is associated with enhanced fibrosis progression.74
Hyperammonemia and Immune Dysfunction
NAFLD and CLD both are characterized by increased susceptibility to infections as well as increased mortality from them.76 Animal (rat) studies have shown that innate immune response is compromised in diet-induced NASH and decreased urea synthesis.77 Ammonia-induced immune dysfunction in MASLD may be similar to that seen in cirrhosis. Thus hyperammonemia is associated with neutrophil dysfunction (reduced chemotaxis & phagocytosis)78; drop in dendritic cell count, mitochondrial dysfunction, poor antigen phagocytosis, and excessive reactive oxygen species generation ex-vivo in samples from humans as well mice with cirrhosis.79
It has been pointed out that cirrhosis patients experiencing a state of inflammation display significant deterioration in neuropsychological test scores following induced hyperammonemia.80 It was subsequently shown to be related to hyperammonemia-induced increased sensitivity to bacterial lipoprotein polysaccharides (LPS).81
AMMONIA IN MASLD
Disordered ammonia handling has emerged as a plausible hypothesis to explain the of progression in MASLD from steatosis to steatohepatitis, cirrhosis, and hepatocellular carcinoma.82 In the past, the progression in MASLD was thought to be related to related to a variety of metabolic anomalies such as Increased β oxidation of fatty acids, fatty acid lipid per-oxidation, microsomal oxidation, lipotoxicity, free radical accumulation, apoptosis, gut bacterial translocation, inflammation and accumulation of DAMPs. Recent data has highlighted that urea cycle activity is compromised in MASLD.
In vitro experiments have shown that long chain fatty acids and triglycerides impact gene expression of enzymes involved in urea cycle, inhibit urea genesis and increase plasma ammonia concentration.83,84 In vivo animal studies show that reduction in gene expression for ornithine transcarbamylase (OTC) reduces urea production and leads to hyperammonemia and fibrosis progression.85,86 Similar results have been shown in humans.87,88 These changes are attributed to DNA hypermethylation of the promoter regions of urea cycle enzyme genes. xxviii These epigenetic changes were more pronounced when MASLD was associated with more severe steatohepatitis and fibrosis and were reversible with weight loss indicating remodeling of epigenetic signatures.89,90 An alternate hypothesis is that ammonia may directly induce senescence in hepatocytes (and also astrocytes) which is evidenced by demonstration of overexpression of the tumor-suppressor gene p53 in human biopsies.91, 92, 93 Therefore it appears very likely that epigenetic alterations in the expression of urea cycle enzymes lead to accumulation of ammonia which in turn contributes to the progression of NAFLD.
Can this knowledge be used to prevent the progression of NAFLD? Interestingly, the ability to increase the urea cycle's capacity to process nitrogen in response to increased protein intake is intact in cirrhosis though weakened.94 It can be augmented further by the use of glucagon in normal individuals but not so much in cirrhosis.95,96 Besides it may worsen associated diabetes. Beta-blockers too are known to enhance urea synthesis capacity both in normal as well as cirrhotic subjects.97 Similarly, zinc as a co-factor of OTC enzyme can improve urea synthesis in cirrhosis.98 However, the impacts of these treatments in MASLD have not been adequately studied and remain a gap in our knowledge. Ammonia-lowering therapies have been shown to reduce the severity of fibrosis and reduce the deposition of collagen in steatotic rat liver slices (using phenyl acetate).86 But no clinical trials have been conducted so far to validate this strategy to prevent MASLD progression.
In vitro studies have shown that increased ammonia levels can activate hepatic stellate cells (HSC), which are instrumental in converting the extracellular matrix to its fibrogenic mode. It has also been shown that removing ammonia from cell cultures can reverse this process towards normalcy.99,100 Even in vivo studies in bile duct ligated rats have shown that HSC activation as well as portal hypertension can be reduced by ammonia-lowering measures.
Susceptibility to developing malignancy in MASLD, even before the development of cirrhosis widely known,101 and is possibly related to hyperammonemia. The MASLD microenvironment specifically favors cells that use ammonia as a nitrogen source and ammonia has been shown to increase the proliferation rate of cancer cells.102 Ammonia accumulation in the liver is out of proportion to the severity of liver disease in patients with metastatic cancer in the liver.103 In vitro studies have also shown enhanced growth of breast cancer cells as well as cells that lacked tumor suppressor gene p53.104,105 It has also been shown that targeting a heat-shock protein(DNAJC24) interferes with ammonia utilization and consequently affects proliferation, autophagy, and tumor progression in hepatocellular carcinoma.106
AMMONIA AND CLD
It is well known that ammonia levels may be raised in cirrhosis patients. Ammonia levels may be raised due to Loss of Functional hepatic reserve related to urea cycle dysfunction, variceal bleeding with added protein load in gut, gut microbiome change with more urease producing bacteria, portosystemic shunts allowing ammonia to bypass and sarcopenia with poor capacity of muscles to mop up ammonia.107,108 So far there were not many studies stressing on prognostic significance of plasma ammonia levels.
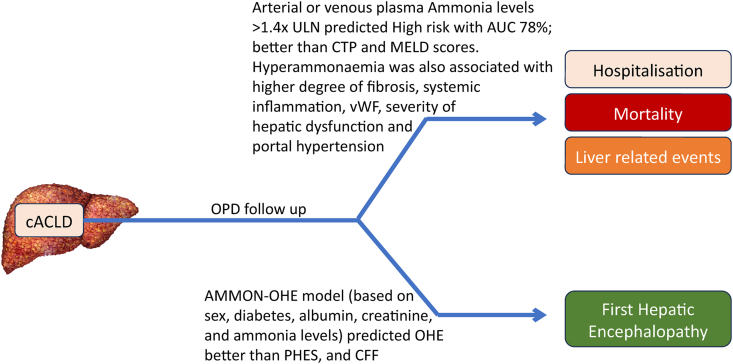
AMMON consortium has recently published an interesting data from four independent liver units in Europe.109 They reported that high ammonia levels in clinically stable outpatients were an independent predictor of hospitalization as well as mortality due to liver-related complications (namely (sepsis, variceal bleeding, overt hepatic encephalopathy, acute onset, or worsening ascites) in these patients. Not only that, but ammonia levels did also better than traditional liver disease severity scores in prognosticating these patients. AUC for hospitalization was 78% for ammonia levels, 72% for CTP scores, and 66% for MELD score (P < 0.001) The high risk of hospitalization as well as death was predicted if the ammonia levels were found to be higher than 1.4 times higher than the upper limit of normal in their labs. Either arterial or venous ammonia levels were measured in the patients in the training cohort, while only venous ammonia levels were measured in the validation cohort. The authors in this study did not adjust their multi-variable models for systemic inflammation or portal hypertension severity.
Another study from Austria110 went a step further and studied the prognostic value of venous ammonia to other important liver-related complications. Patient groups studied here were again those with ACLD and definitions of cACLD and ACLF were prevalent for that region.111,112 This study supported the findings described earlier but also brought out some additional points. The authors reported that the presence of diabetes mellitus was associated with high ammonia levels. They explained it by attributing it to autonomic dysfunction, extended gastrointestinal transit times, and bacterial overgrowth as well as increased protein catabolism and accelerated muscle breakdown.113
Hyperammonemia directly causes immune dysfunction and activation of HSCs. These associations were supported by this study which found positive correlations of the high ammonia levels with ELF-test (reflecting fibrogenesis), and with systemic inflammation, vWF severity hepatic dysfunction, and portal hypertension. Still, ammonia levels could predict liver-related outcomes independently from other established prognostic indicators such as MELD, HVPG, VWF, IL-6, CRP, ELF, and renin levels. While it was independent, the prognostic significance of ammonia level was found to be similar to MELD and HVPG. Since the value of ammonia was independent of hepatic dysfunction, portal hypertension severity or inflammation, the authors have suggested direct toxicity of ammonia to explain this effect.
Around the same time, a paper from Germany reviewed various scores to predict first episode of hepatic encephalopathy in patients with cirrhosis.114 They analyzed the evidence or lack of it available for Clinical Covert HE (CCHE) score; critical flicker frequency (CFF) score, a composite score including bilirubin, albumin, nonselective beta-blocker, and statin use (BABS score); MELD-Na-Activity-Chair stands-Quality of Life Hepatic Encephalopathy (MASQ-HE) Score; overt HE (OHE) score; Psychometric Hepatic Encephalopathy (PHES) Score; Sickness Impact Profile Clinical Covert HE (SIP CHE) score, Stroop EncephalApp, and Animal naming test score (S-ANT1). They stressed on the need to identify identifying high-risk populations with the highest need for subsequent primary prophylaxis.
Soon enough, the AMMON consortium came up with the predictive importance of serum ammonia levels in predicting first episode overt hepatic encephalopathy (OHE) in patients with cirrhosis.115 They have developed an AMMON-OHE model after studying 426 outpatients with cirrhosis from three different liver units. The model is based on patient information about sex, diabetes, albumin, creatinine, and ammonia levels and have shown which has shown excellent predictive ability. Venous ammonia was measured, and the result was normalized to the upper limit of normal (AMM-ULN) at the respective reference laboratory (Figure 5).
Figure 5.
Prognostic value of plasma ammonia levels in cACLD. Recent studies have shown that ammonia levels >1.4 times ULN can predict liver related complications requiring hospitalization, hepatic encephalopathy and mortality in patients with cACLD. cACLD, compensated aldvanced chronic liver disease; CFF, Critical flickering frequency test; CTP, Child-Pugh-Turcott score; MELD, model for end-stage liver disease; OHE, overt hepatic encephalopathy; PHES, psychometric hepatic encephalopathy score; ULN, upper limit of normal.
The study found that the AMMON-OHE model performed better than existing predictors such as psychometric hepatic encephalopathy score (PHES) and the critical flicker frequency (CFF) test. The results were validated in two validation cohorts consisting of 267 and 381 patients at two liver units.
AMMONIA AND AD/ACLF
Prognostic value of serum ammonia in patients with cirrhosis and acute decompensation (AD) or acute on chronic liver failure (ACLF)116 has also been studied. A multicenter study from India and UK analyzed 498 patients with cirrhosis and AD and found that baseline ammonia levels correlated with severity of HE, and overall mortality (P < 0.001).117 It was found to be an independent predictor of 28-day mortality. Absolute ammonia levels more of 79.5 μmol/L could be associated with a higher frequency of organ failures and could predict 28-day mortality with a sensitivity of 68.1% and specificity of 67.4%. Ammonia levels were repeated on day 5 of hospitalization and patients with persistently high ammonia levels had higher 28-day mortality (70.6%) in comparison to those where ammonia levels had improved (35.7%).
In ACLF too, similar observations have been made.118 Serial arterial ammonia was measured in 229 patients with ACLF If the levers were more than ≥79.5 μmol/L on day 3, patients were classified as having persistent or incident hyperammonemia. This group of patients had significantly high organ failures and mortality (HR for 28-day mortality was 3.174).
In yet another study by the APASL-ACLF consortium (AARC) 3009 patients were followed up for 30 days.119 Of these 43.7% had HE at presentation and its presence was significantly associated with higher age, systemic inflammatory response, elevated ammonia levels, serum protein, sepsis, and MELD score. New onset HE or progressive worsening of the level of HE was significantly associated with AARC score (≥9) and ammonia levels (≥85 μmol/L) at baseline. The study noted that ammonia levels were a significant predictor of HE occurrence, higher HE grades, and 30-day mortality. When a dynamic increase in the ammonia levels by about 60% over 7 days was recorded, it seemed to predict non-survivors and worsening of HE. Several similar reports indicate that hyperammonemia translates into poor outcomes in patients with ACLF.120, 121, 122, 123
AMMONIA AND ACUTE LIVER FAILURE
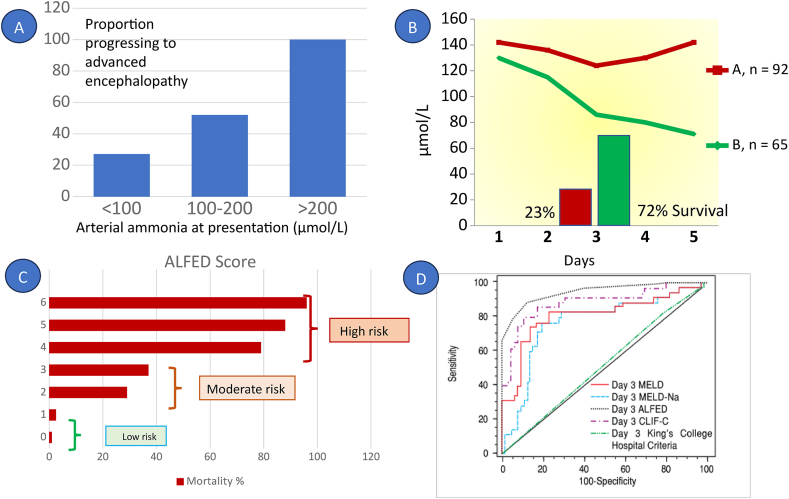
The Prognostic role of ammonia in acute liver failure (ALF) was studied in eighty consecutive patients in New Delhi from 2001 to 2003.124 High arterial ammonia level (>123 μmol/L) could predict mortality with 78.6% sensitivity, 76.3% specificity, and 77.5% diagnostic accuracy. This study also showed that patients with higher ammonia levels had higher grades of HE, cerebral edema, and seizures. This paper laid the foundation for developing a mathematical model for predicting poor outcomes in ALF. Similar results were subsequently shown from UK.125 (Figure 6A).
Figure 6.
Hyperammonemia and acute liver failure. A. Data showing that higher ammonia level is associated with higher risk of progressing to advanced encephalopathy127 B. Ammonia levels that remain persistently high till day 3 after admission are associated with significantly higher mortality (ref) C. ALFED score 0-1 indicates low risk, 2–3 moderate risk and >4 suggest high risk mortality128 D. ALFED model is better than othe rprevalent prognostic scores.134
Apart from arterial ammonia levels on admission, the changes in these levels over ensuing days were also found to be very important. Follow-up studies showed that persistent arterial hyperammonemia for 3 days after admission was more closely related to adverse outcomes much worse than those who had a decreasing trend (Figure 6 B). Presence of Infection, renal failure, and acidosis were found to be independent predictors of persistent hyperammonemia.126 It was speculated that ammonia could be used as a prognostic tool in acute liver failure.127
The speculation was set aside by another prospective study128 where a dynamic prognostic model was evolved to predict outcomes in ALF. The model was derived after studying a cohort of 244 patients with ALF. Scoring points were allotted to four parameters that, if showed persistently or increasing trend, significantly and independently affected mortality in ALF. Their parameters were measured on day 1 and day 3 and if they had persistent or increasing levels of
-
(a)
Hepatic encephalopathy grade >2: 2 points,
-
(b)
Arterial ammonia >123 μmol/L: 2 points.
-
(c)
INR>5: 1 point
-
(d)
Serum bilirubin >15 mg/dl: 1 point
The derivation cohort showed and was later confirmed even in a validation cohort of132 patients with ALF, that prognosis worsened with rising numbers of points in each patient (Figure 6C). Patients who were classified as having 0–1 points, had good outcomes with nearly all of them recovering. These were called low-risk patients. Accumulated points of 2–3 classified them into moderate risk and those having 4 or more points were classified as high-risk patients. Patients with 4 or more points were shown to have a high positive predictive value (PPV) of 85% and a negative predictive value (NPV) of 87% for death in the validation cohort and could be used as selection criteria for transplantation.
In ALF, the selection of patients for liver transplantation is a challenging proposition. There are several prognostic scores prevalent and validated to select ALF patients for liver transplantation, namely, King's College Criteria (KCC),129 Clichy Criteria,130 and MELDNa score.133 One study from Chandigarh described a clinical prognostic indicator (CPI) score.132 Possibly King's College criteria (KCC) have been the most popular for this purpose. Unfortunately, several independent reports have suggested that KCC do have a PPV of 70%–100% but the main limitation is that NPV has been reported to be 25%–94% often much lower than that suggested in the original study. xcii,133 While KCC remains very useful for paracetamol overdose-induced ALF, (further improved by adding post-resuscitation lactate) but for viral hepatitis-induced ALF, which accounts for most cases in South Asia, it may not select many patients that are destined to die.
Another study from New Delhi134 compared the ALFED score with some other prognostic scores in ALF related to viral hepatitis including the MELD score, MELD-Na score, ALFED model, CLIF consortium ACLF score,135 and KCC. When calculated at admission, the baseline values of prognostic scores (MELD, MELD-Na, ALFED, CLIF-C ACLF, and KCH) had modest (AUROC: 0.65–0.77) discriminatory capacity. However, the AUROC increased when the day 3 values of these scores were considered with the exception of KCC. At this point, ALFED score had the highest AUROC of 0.95, followed by CLIF-C ACLF (0.88), MELD (0.81), MELD-Na (0.77), and KCH (0.52) (Figure 6D). Thus, the ALFED score seems to be ideal for the selection of ALF patients for liver transplantation and has been recommended for this purpose by INASL.
TREATMENT OF HYPERAMMONEMIA
There are several strategies used for the treatment of hyperammonemia caused by various diseases and the topic has been reviewed elsewhere.136,137 One may reduce ammonia production by manipulating gut bacteria or by altering amino acid metabolism. Alternatively, one may boost urea cycle activity by supplementing alternative substrates. Finally, one may help in eliminating ammonia from the body by artificial means or by boosting liver functions. Detailed discussion of the treatment is beyond the scope of this paper and a summary of ammonia elimination strategies has been placed at Table 2.
Table 2.
A Summary of Ammonia-lowering Therapies.
| Therapy | Remarks | Dose |
|---|---|---|
| Therapies targeting Intestinal bacteria to reduce ammonia production | ||
| Rifaximin138 | acidification of the colonic contents, increase in osmotic pressure, cathartic effect. |
Customized drug dosage to achieve 2–3 semiformed motions.a |
| Rifaximin139 | inhibition of RNA synthesis in intestinal bacteria | 550 mg twice a daya |
| Neomycin140 | inhibition of protein synthesis in intestinal bacteria | 1 gm every 6 h for up to6 daysb |
| Metronidazole141 | inhibition of nucleic acid synthesis in intestinal bacteria |
400 mg twice dailyb |
| Sodium benzoate142 | decrease glycine degradation, Increase glycine elimination | 180 to 650 mg/kg- per daya |
| Probiotics and synbiotics | modulate the gut microbiota–short-chain fatty acid (SCFA) butyrate hormone axis. | Variablec |
| Reduce ammonia production by altering amino acid degradation | ||
| Sodium phenyl acetate/phenylbutyrate143 | decreases glutamine degradation, increases glutamine elimination Sodium Phenylbutyrate is a prodrug and converts to phenyl acetate. It combines with glutamine to form phenyl acetyl glutamine, which is rapidly excreted by the kidneys. | Weight: Less than 20 kg: 450–600 mg/kg/day Weight: 20 kg or Greater: 9.9–13 g/m2/day Sodium Phenylbutyrate tablets (500 mg) are given 3–12 tablets three times a daya |
| Glycerol phenylbutyrate144 | decreases glutamine degradation, increases glutamine elimination |
Available as liquid 1.1 g/mL Dose: 4.5–11.2 mL/m2/day If switching from Phenylbutyrate: Total daily dose of ‘Ravicti’ (mL) = total daily dosage of sodium phenyl butyrate (g) x 0.86b |
| Branched-chain amino acids (BCAA)145 | decrease glutamine degradation, increase glutamine elimination |
May increase ammonia levels146 Variable dose usedb |
| Activation of urea cycle | ||
| Carglumic acid147 | activation of UC through N-acetyl glutamate restorement | 80–100 mg/kg/daya |
| l-arginine/L-citrullin148 | activation of UC | Variable. l-arginine (0.8 mmol/kg of body weight) or of l-citrulline (1.0 mmol/kg of body weight) a |
| l-ornithine/L-aspartate149 | activation of UC | Intravenous 30 g over 24 h for 5 daysb |
| l-ornithine phenylacetate150 | activation of UC, activation of glycine and glutamine synthesis, increases glycine and glutamine elimination | Intravenous 500 ml/24 h for ≤5 daysb |
| l-carnitine151 | activation of UC | 100 mg/kg dose (max 3 gm/24 h) divided 6–8 hourlyb |
| Liver cell transplantation152 | activation of UC | For liver metabolic defects, 200–400 million hepatocytes per kilogram of body weight are injected, theoretically to achieve 5%–10% of the recipient hepatic mass. The infusion can be scheduled over one or several sessions. A defined range of 30–100 million cells per kilogram and an infusion rate of ≤8 ml/kg/hr should be respected per infusion session.c |
| Liver transplantation153 | Activation of UC | Not applicableb |
| Stem cell transplantation | activation of UC | Not applicablec |
| Adenovirus associated gene delivery154 |
activation of UC | Not applicablec |
| Increase elimination of ammonia | ||
| Albumin-based dialysis155 | elimination of albumin-bound substances |
Variable number (1–10) of sessionsa |
| Peritoneal dialysis156 and CRRT157 | decrease of blood ammonia by transporting ammonia from vascular system to peritoneal cavity |
Variable number of sessionsb |
| Bioartificial liver support systems158 | support for liver metabolic activity |
Variable number of sessionsc |
CRRT: continuous renal replacement therapy, RNA: ribo-nucleic acid, UC: urea cycle; SCFA, short-chain fatty acid.
Accepted therapy.
alternative therapy.
under investigations.
PROBLEMS WITH AMMONIA RESEARCH
-
(a)
Method of sample collection and testing
The way ammonia is tested today leaves a lot to be desired. The results show substantial laboratory variability.159 Arterial ammonia has been preferred over venous ammonia by several workers, and sample collection is inconvenient, especially as an outpatient.160,161 Some reports suggest that venous ammonia may be as good as arterial ammonia in patients with cirrhosis162 and venous sampling is significantly more convenient. Most workers insist that the sample must be transported immediately in dry ice to be processed as delay may mean inaccurate results. This requires a dedicated laboratory and staff which is only feasible in centers that focus on this type of research. Venous ammonia also has the disadvantage of being affected by the activity and condition of peripheral tissues including muscles, intestine, and kidneys. There is a strong need for point-of-care tests which can be performed at the patient's bedside to make ammonia testing a practical tool. Blood sample analyses are notoriously sensitive to disturbances and particular care and diligence are required throughout collection, transportation, and analysis, which poses challenges in clinical settings and even in clinical research protocols. xxviii
-
(b)
Limitations in published research
A bulk of work related to ammonia is in vitro, and in animals and it is difficult to interpret this data in human terms. There is significant evidence in terms of compromised urea synthesis, but similar data on ammonia dynamics in various situations especially follow up tests is lacking. Similarly, direct proof of sustained effect of ammonia lowering therapies is not robust and is a major requirement before we can fully understand ammonia dynamics.
-
(c)
Normal levels
While a lot of work has been done on ammonia in various situations, there is no standardization of normal ammonia levels in various situations. Some labs mention it as 11–32 μmol/L.163 It has been variously described as <45 μmol/L, 150–343 μmol/L in acute liver failure (ALF), 90–120 μmol/L in acute on chronic liver disease, 80–100 μmol/L after trans-jugular intrahepatic portosystemic shunt (TIPS), and 46–60 μmol/L in compensated advanced chronic liver disease (cACLD).164, 165, 166, 167 Impact of hypothermia, brain activity, sarcopenia and renal disease has never been qualified. So minor degrees of hyperammonemia will be difficult to recognize. Many laboratories may calibrate ammonia for various neurological syndromes, there is no universal upper limit of normal. xxviii Recent articles have resorted to using levels standardized to upper limit of normal worked out by local laboratories. lxix
-
(d)
Impact of critical illness
Ammonia levels are altered in several critical illnesses, not involving primary liver disease.168,169 There are myriads of causes of hyperammonemia apart from liver disease that too need to be considered. Many drugs and diseases as a cause of raised ammonia levels have been reviewed elsewhere.170 We just beginning to understand how sepsis may affect ammonia levels in the body.171 How an intestinal disease or microbiota affects ammonia levels in health and disease is not yet fully understood.9 A lot is required to be done before this test can be introduced in normal clinical practice.
Ammonia levels in plasma may be raised in a variety of diseases and situations, but its importance in the pathogenesis and prognostication cannot be overstated. In cACLD, it can predict the risk of liver-related complications and the first episode of hepatic encephalopathy and may help clinicians in the decision to institute prophylactic therapies. Hyperammonemia has direct prognostic implications in acute liver failure (ALF) and acute on chronic liver failure (ACLF). Acute liver failure early dynamic score (ALFED) score has evolved to predict poor prognosis in ALF, which has been adopted by several centers to select patients for liver transplantation in this disease. The current method of testing and interpretation of ammonia dynamics does have some limitations, but its usefulness for the clinician cannot be denied. The knowledge about ammonia handling in the body in various situations is still evolving and its newer uses continue to unravel.
CREDIT AUTHORSHIP CONTRIBUTION STATEMENT
SKA; Conceptualization; Data curation; Formal analysis; Investigation; Methodology; Supervision; review & editing.
ACA: Data curation; Formal analysis; Software; Supervision; Validation; Visualization; figure drawing, Writing - review & editing.
CONFLICTS OF INTEREST
There is no conflict of interest to declare.
FUNDING
No Funding was received for this paper.
References
- 1.Shawcross D.L., Olde Damink S.W., Butterworth R.F., Jalan R. Ammonia and hepatic encephalopathy: the more things change, the more they remain the same. Metab Brain Dis. 2005 Sep;20:169–179. doi: 10.1007/s11011-005-7205-0. PMID: 16167195. [DOI] [PubMed] [Google Scholar]
- 2.Hahn M., Massen O., Nencki M., Pavlov I. Die Eck’sche fistel zwischen der unteren hohlvene und der pfortader und ihre folgen Fur den organismus. Arch. Exp. Pathol. 1893;32:161–210. Pharm. (Quoted by 1 ibid) [Google Scholar]
- 3.Lockwood A., Yap E., Wong W. Cerebral ammonia metabolism in patients with severe liver disease and minimal hepatic encephalopathy. J Cerebr Blood Flow Metabol. 1991;11:337–341. doi: 10.1038/jcbfm.1991.67. [DOI] [PubMed] [Google Scholar]
- 4.Mohiuddin S.S., Khattar D. StatPearls Publishing; 2023 Jan. Biochemistry, Ammonia.https://www.ncbi.nlm.nih.gov/books/NBK541039/ [Updated 2023 Feb 20]. In: StatPearls [Internet]. Treasure Island (FL) [PubMed] [Google Scholar]
- 5.Adeva M.M., Souto G., Blanco N., Donapetry C. Ammonium metabolism in humans. Metabolism. 2012 Nov;61:1495–1511. doi: 10.1016/j.metabol.2012.07.007. Epub 2012 Aug 24. PMID: 22921946. [DOI] [PubMed] [Google Scholar]
- 6.Weiner I.D., Verlander J.W. Renal ammonia metabolism and transport. Compr Physiol. 2013 Jan;3:201–220. doi: 10.1002/cphy.c120010. [PMC free article: PMC4319187] [PubMed: 23720285] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.van Assendelft O.W., Zijlstra W.G. Extinction coefficients for use in equations for thespectrophotometric analysis of haemoglobin mixtures. Anal Biochem. 1975 Nov;69:43–48. doi: 10.1016/0003-2697(75)90563-1. [PubMed: 2033] [DOI] [PubMed] [Google Scholar]
- 8.Ruscák M., Hager H., Orlický J. Alanine formation and alanine aminotransferase activity inthe nerve tissue with proliferating macroglia. Acta Neuropathol. 1976 Mar 15;34:149–155. doi: 10.1007/BF00684665. [PubMed: 3940] [DOI] [PubMed] [Google Scholar]
- 9.Kim M.H., Kim H. The roles of glutamine in the intestine and its implication in intestinal diseases. Int J Mol Sci. 2017 May 12;18:1051. doi: 10.3390/ijms18051051. PMID: 28498331; PMCID: PMC5454963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Weber F.L., Jr., Veach G.L. The importance of the small intestine in gut ammonium production in the fasting dog. Gastroenterology. 1979 Aug;77:235–240. PMID: 447037. [PubMed] [Google Scholar]
- 11.Olde Damink S.W., Jalan R., Redhead D.N., et al. Interorgan ammonia and amino acid metabolism in metabolically stable patients with cirrhosis and a TIPSS. Hepatology. 2002;36:1163–1171. doi: 10.1053/jhep.2002.36497. [DOI] [PubMed] [Google Scholar]
- 12.Kaiser S., Gerok W., Haussinger D. Ammonia and glutamine metabolism in human liver slices: new aspects on the pathogenesis of hyperammonaemia in chronic liver disease. Eur J Clin Invest. 1988;18:535–542. doi: 10.1111/j.1365-2362.1988.tb01053.x. [DOI] [PubMed] [Google Scholar]
- 13.Conn H.O. Studies of the source and significance of blood ammonia. IV. Early ammonia peaks after ingestion of ammonium salts. Yale J Biol Med. 1972;45:543–549. [PMC free article] [PubMed] [Google Scholar]
- 14.Plauth M., Roske A.E., Romaniuk P., et al. Post-feeding hyperammonaemia in patients with transjugular intrahepatic portosystemic shunt and liver cirrhosis: role of small intestinal ammonia release and route of nutrient administration. Gut. 2000;46:849–855. doi: 10.1136/gut.46.6.849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Owen E.E., Tyor M.P., Giordano D. The effect of acute alkalosis on renal metabolism of ammonia in cirrhotics. J Clin Invest. 1962;41:1139–1144. doi: 10.1172/JCI104566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Owen E.E., Flanagan J.F., Tyor M.P. Kidney as a source of blood ammonia: effect of chlorothiazide. Proc Soc Exp Biol Med. 1959;102:696–697. doi: 10.3181/00379727-102-25366. [DOI] [PubMed] [Google Scholar]
- 17.Berry J.N., Owen E.E., Flanagan J.F., et al. The effect of acute hyperventilation on the blood ammonia concenration of patients with liver disease. The relative role of the kidney, liver, and extremity. J Lab Clin Med. 1960;55:849–854. [PubMed] [Google Scholar]
- 18.Tyor M.P., Owen E.E., Berry J.N., et al. The relative role of extremity, liver, and kidney as ammonia receivers and donors in patients with liver disease. Gastroenterology. 1960;39:420–424. [PubMed] [Google Scholar]
- 19.Ganda O.P., Ruderman N.B. Muscle nitrogen metabolism in chronic hepatic insufficiency. Metabolism. 1976;25:427–435. doi: 10.1016/0026-0495(76)90075-5. [DOI] [PubMed] [Google Scholar]
- 20.Sinniah D., Fulton T.T., McCullough H. The effect of exercise on the venous blood ammonium concentration in man. J Clin Pathol. 1970;23:715–719. doi: 10.1136/jcp.23.8.715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Anand A.C. Nutrition and muscle in cirrhosis. J Clin Exp Hepatol. 2017 Dec;7:340–357. doi: 10.1016/j.jceh.2017.11.001. Epub 2017 Nov 8. PMID: 29234200; PMCID: PMC5719462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Crossland H., Smith K., Atherton P.J., Wilkinson D.J. The metabolic and molecular mechanismsof hyperammonaemia- and hyperethanolaemia-induced protein catabolism in skeletal musclecells. J Cell Physiol. 2018 Dec;233:9663–9673. doi: 10.1002/jcp.26881. [PMC free article: PMC6220958][PubMed: 30144060] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Hagiwara A., Sakamoto T. Clinical significance of plasma ammonia in patients with traumatic hemorrhage. J Trauma. 2009 Jul;67:115–120. doi: 10.1097/TA.0b013e3181a5e63e. [PubMed: 19590319] [DOI] [PubMed] [Google Scholar]
- 24.Schindler P., Heinzow H., Trebicka J., Wildgruber M. Shunt-induced hepatic encephalopathy in TIPS: current approaches and clinical challenges. J Clin Med. 2020 Nov 23;9:3784. doi: 10.3390/jcm9113784. PMID: 33238576; PMCID: PMC7700586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Weiner I. David, Mitch William E., Sands Jeff M. Urea and ammonia metabolism and the control of renal nitrogen excretion. Clin J Am Soc Nephrol. August 2015;10:1444–1458. doi: 10.2215/CJN.10311013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Harris A.N., Skankar M., Melanmed M., Batlle D. An update on kidney ammonium transport along the nephron. Adv Kidney Dis Health. 2023 Mar;30:189–196. doi: 10.1053/j.akdh.2022.12.005. PMID: 36868733. [DOI] [PubMed] [Google Scholar]
- 27.Vaziri N.D., Khazaeli M., Nunes A.C.F., et al. Effects of end-stage renal disease and dialysis modalities on blood ammonia level. Hemodial Int. 2017 Jul;21:343–347. doi: 10.1111/hdi.12510. Epub 2016 Nov 1. PMID: 27804262; PMCID: PMC5812671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Maldonado C., Guevara N., Queijo C., González R., Fagiolino P., Vázquez M. Carnitine and/or acetylcarnitine deficiency as a cause of higher levels of ammonia. BioMed Res Int. 2016;2016 doi: 10.1155/2016/2920108. Epub 2016 Feb 21. PMID: 26998483; PMCID: PMC4779505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Gong Z., Liang L., Qiu W., et al. Clinical, biochemical, and molecular analyses of medium-chain acyl-CoA dehydrogenase deficiency in Chinese patients. Front Genet. 2021;12 doi: 10.3389/fgene.2021.577046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Mehta S., Tayabali S., Lachmann R. Valproate-induced hyperammonemia - uncovering an underlying inherited metabolic disorder: a case report. J Med Case Rep. 2018;12:134. doi: 10.1186/s13256-018-1666-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Häberle J., Chakrapani A., Ah Mew N., Longo N. Hyperammonaemia in classic organic acidaemias: a review of the literature and two case histories. Orphanet J Rare Dis. 2018 Dec 6;13:219. doi: 10.1186/s13023-018-0963-7. PMID: 30522498; PMCID: PMC6282273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Rare Disease Database: Reye’s syndrome. https://rarediseases.org/rare-diseases/reye-syndrome/(accessed on 3 January 2024).
- 33.Kenzaka T., Kato K., Kitao A., et al. Hyperammonemia in urinary tract infections. PLoS One. 2015 Aug 20;10 doi: 10.1371/journal.pone.0136220. PMID: 26292215; PMCID: PMC4546155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Hassan A.A.I., Ibrahim W., Subahi A., Mohamed A. 'All that glitters is not gold': when hyperammonaemia is not from hepatic aetiology. BMJ Case Rep. 2017 May 22;2017 doi: 10.1136/bcr-2017-219441. PMID: 28536219; PMCID: PMC5753707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Gorantla A., Kishore A., Zaman A., et al. Hyperammonemic encephalopathy secondary to urinary tract infection. Cureus. 2022 Nov 21;14 doi: 10.7759/cureus.31754. PMID: 36569707; PMCID: PMC9771524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Neves Briard J., Lezaic N., Keezer M.R. Pearls & oy-sters: chemotherapy-associated hyperammonemic encephalopathy. Neurology. 2020 Feb 25;94:e874–e877. doi: 10.1212/WNL.0000000000009004. Epub 2020 Jan 28. PMID: 31992681. [DOI] [PubMed] [Google Scholar]
- 37.Kamel A.Y., Emtiazjoo A.M., Adkins L., et al. Hyperammonemia after lung transplantation: systematic review and a mini case series. Transpl Int. 2022 May 3;35 doi: 10.3389/ti.2022.10433. Erratum. Transpl Int. 2022 Jul 08;35:10670. PMID: 35620675; PMCID: PMC9128545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Raja V., Reynolds C.A., Greenberg M.L. Barth syndrome: a life-threatening disorder caused by abnormal cardiolipin remodeling. J Rare Dis Res Treat. 2017;2:58–62. doi: 10.29245/2572-9411/2017/2.1087. Epub 2017 Mar 21. PMID: 31032491; PMCID: PMC6482962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Rare disease database: pyruvate carboxylase deficiency. https://rarediseases.org/rare-diseases/pyruvate-carboxylase-deficiency/(accessed on 3 January 2024).
- 40.Gene Reviews, National Library of Medicine. Primary Pyruvate Dehydrogenase Complex Deficiency Overview. https://www.ncbi.nlm.nih.gov/books/NBK571223/(accessed on 3 January 2024).
- 41.Palladino A.A., Stanley C.A. The hyperinsulinism/hyperammonemia syndrome. Rev Endocr Metab Disord. 2010 Sep;11:171–178. doi: 10.1007/s11154-010-9146-0. PMID: 20936362. [DOI] [PubMed] [Google Scholar]
- 42.Clericetti C.M., Milani G.P., Lava S.A.G., Bianchetti M.G., Simonetti G.D., Giannini O. Hyperammonemia associated with distal renal tubular acidosis or urinary tract infection: a systematic review. Pediatr Nephrol. 2018 Mar;33:485–491. doi: 10.1007/s00467-017-3829-7. Epub 2017 Nov 13. PMID: 29134448. [DOI] [PubMed] [Google Scholar]
- 43.Jäger W., Viertmann A.O., Janßen C., Birklein F., Thüroff J.W., Stein R. Intermittent hyperammonemic encephalopathy after ureterosigmoidostomy: spontaneous onset in the absence of hepatic failure. Cent European J Urol. 2015;68:121–124. doi: 10.5173/ceju.2015.01.459. Epub 2015 Mar 13. PMID: 25914851; PMCID: PMC4408387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Ryder K.W., Olson J.F., Kahnoski R.J., Karn R.C., Oei T.O. Hyperammonemia after transurethral resection of the prostate: a report of 2 cases. J Urol. 1984 Nov;132:995–997. doi: 10.1016/s0022-5347(17)49984-8. PMID: 6492293. [DOI] [PubMed] [Google Scholar]
- 45.Devasahayam J.V.M., John S.G., Assar S., et al. In: Diet and Nutrition in Critical Care. Rajendram R., Preedy V., Patel V., editors. Springer; New York, NY: 2014. Hyperammonemia as an adverse effect in parenteral nutrition. [DOI] [Google Scholar]
- 46.Kalra A., Norvell J.P. Cause for confusion: noncirrhotic hyperammonemic encephalopathy. Clin Liver Dis. 2020 Jun 30;15:223–227. doi: 10.1002/cld.929. PMID: 32617154; PMCID: PMC7326631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Stergachis A.B., Mogensen K.M., Khoury C.C., et al. A retrospective study of adult patients with noncirrhotic hyperammonemia. J Inherit Metab Dis. 2020;43:1165–1172. doi: 10.1002/jimd.12292. [DOI] [PubMed] [Google Scholar]
- 48.Khoory J., Rupal A., Jani C., Singh H., Hu K. Food coma: hyperammonemic encephalopathy from refeeding syndrome. Cureus. 2021 Oct 19;13 doi: 10.7759/cureus.18898. PMID: 34820220; PMCID: PMC8601258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Stone W.L., Basit H., Jaishankar G.B. StatPearls [Internet]. Treasure Island (FL) StatPearls Publishing; 2023 Aug 7. Urea cycle disorders. 2023 Jan–. PMID: 29493985. [Google Scholar]
- 50.Redant S., Empain A., Mugisha A., et al. Management of late onset urea cycle disorders—a remaining challenge for the intensivist? Ann Intensive Care. 2021;11:2. doi: 10.1186/s13613-020-00797-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Long M.T., Kruser J.M., Quinonez S.C. Urea cycle disorders in critically Ill adults. Curr Opin Clin Nutr Metab Care. 2024 Mar 1;27:184–191. doi: 10.1097/MCO.0000000000000992. Epub 2023 Nov 6. PMID: 37938118. [DOI] [PubMed] [Google Scholar]
- 52.Bijarnia-Mahay S., Häberle J., Jalan A.B., et al. Urea cycle disorders in India: clinical course, biochemical and genetic investigations, and prenatal testing. Orphanet J Rare Dis. 2018;13:174. doi: 10.1186/s13023-018-0908-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Rüfenacht V., Häberle J. Mini-review: challenges in newborn screening for urea cycle disorders. International Journal of Neonatal Screening. 2015;1:27–35. doi: 10.3390/ijns1010027. [DOI] [Google Scholar]
- 54.Duff C., Alexander I.E., Baruteau J. Gene therapy for urea cycle defects: an update from historical perspectives to future prospects. J Inherit Metab Dis. 2023;47:1–13. doi: 10.1002/jimd.12609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.García Vega M., Andrade J.D., Morais A., et al. Urea cycle disorders and indications for liver transplantation. Frontiers in Pediatrics. 2023;11 doi: 10.3389/fped.2023.1103757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Raven K., Poole C., Chronopoulou E., Pierre G. Think hyperammonaemia: the importance of early clinical management in urea cycle disorders. Paediatr Child Health. 2023;33:57–64. doi: 10.1016/j.paed.2022.12.006. [DOI] [Google Scholar]
- 57.Cooper A.J., Plum F. Biochemistry and physiology of brain ammonia. Physiol Rev. 1987 Apr;67:440–519. doi: 10.1152/physrev.1987.67.2.440. PMID: 2882529. [DOI] [PubMed] [Google Scholar]
- 58.Monfort P., Kosenko E., Erceg S., Canales J.J., Felipo V. Molecular mechanism of acute ammonia toxicity: role of NMDA receptors. Neurochem Int. 2002 Aug-Sep;41:95–102. doi: 10.1016/s0197-0186(02)00029-3. PMID: 12020609. [DOI] [PubMed] [Google Scholar]
- 59.Sepehrinezhad A., Zarifkar A., Namvar G., Shahbazi A., Williams R. Astrocyte swelling in hepatic encephalopathy: molecular perspective of cytotoxic edema. Metab Brain Dis. 2020 Apr;35:559–578. doi: 10.1007/s11011-020-00549-8. [DOI] [PubMed] [Google Scholar]
- 60.Llansola M., Rodrigo R., Monfort P., et al. NMDA receptors in hyperammonemia and hepatic encephalopathy. Metab Brain Dis. 2007 Dec;22:321–335. doi: 10.1007/s11011-007-9067-0. [PubMed:17701332] [DOI] [PubMed] [Google Scholar]
- 61.Jayakumar A.R., Norenberg M.D. Hyperammonemia in hepatic encephalopathy. J Clin Exp Hepatol. 2018 Sep;8:272–280. doi: 10.1016/j.jceh.2018.06.007. Epub 2018 Jun 20. PMID: 30302044; PMCID: PMC6175739. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Sarabhai T., Roden M. Hungry for your alanine: when liver depends on muscle proteolysis. J Clin Invest. 2019 Nov 1;129:4563–4566. doi: 10.1172/JCI131931. PMID: 31545302; PMCID: PMC6819091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Cynober L. Metabolism of dietary glutamate in adults. Ann Nutr Metab. 3 December 2018;73(suppl 5):5–14. doi: 10.1159/000494776. [DOI] [PubMed] [Google Scholar]
- 64.Taneja V., Jasuja H. Severe hyperammonemia from intense skeletal muscle activity: a rare case report and literature review. Medicine (Baltim) 2019 Nov;98 doi: 10.1097/MD.0000000000017981. PMID: 31764807; PMCID: PMC6882587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.McDaniel J., Davuluri G., Hill E.A., et al. Hyperammonemia results in reduced muscle function independent of muscle mass. Am J Physiol Gastrointest Liver Physiol. 2016 Feb 1;310:G163–G170. doi: 10.1152/ajpgi.00322.2015. Epub 2015 Dec 3. PMID: 26635319; PMCID: PMC4971815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Anda S., Zach R., Grallert B. Activation of Gcn2 in response to different stresses. PLoS One. 2017;12 doi: 10.1371/journal.pone.0182143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Di Cola S., Nardelli S., Ridola L., Gioia S., Riggio O., Merli M. Ammonia and the muscle: an emerging point of view on hepatic encephalopathy. J Clin Med. 2022 Jan 26;11:611. doi: 10.3390/jcm11030611. PMID: 35160063; PMCID: PMC8836353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Nishikawa H., Enomoto H., Ishii A., et al. Elevated serum myostatin level is associated with worse survival in patients with liver cirrhosis. J. Cachex-Sarcopenia Muscle. 2017;8:915–925. doi: 10.1002/jcsm.12212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Qiu J., Tsien C., Thapalaya S., et al. Hyperammonemia-mediated autophagy in skeletal muscle contributes to sarcopenia of cirrhosis. Am. J. Physiol. Metab. 2012;303:E983–E993. doi: 10.1152/ajpendo.00183.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Kim G., Kang S.H., Kim M.Y., Baik S.K. Prognostic value of sarcopenia in patients with liver cirrhosis: a systematic review and meta-analysis. PLoS One. 2017;12 doi: 10.1371/journal.pone.0186990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Tantai X., Liu Y., Yeo Y.H., et al. Effect of sarcopenia on survival in patients with cirrhosis: a systematic review and meta-analysis. J Hepatol. 2021;42:205–215. [Google Scholar]
- 72.Nardelli S., Lattanzi B., Merli M., et al. Muscle alterations are associated with minimal and overt hepatic encephalopathy in patients with liver cirrhosis. Hepatology. 2019;70:1704–1713. doi: 10.1002/hep.30692. [DOI] [PubMed] [Google Scholar]
- 73.Hong H.C., Hwang S.Y., Choi H.Y., et al. Relationship between sarcopenia and nonalcoholic fatty liver disease: the Korean Sarcopenic Obesity Study. Hepatology. 2014 May;59:1772–1778. doi: 10.1002/hep.26716. Epub 2014 Mar 24. PMID: 23996808. [DOI] [PubMed] [Google Scholar]
- 74.Pan X., Han Y., Zou T., et al. Sarcopenia contributes to the progression of nonalcoholic fatty liver disease- related fibrosis: a meta-analysis. Dig Dis. 2018;36:427–436. doi: 10.1159/000491015. Epub 2018 Jul 26. PMID: 30048963. [DOI] [PubMed] [Google Scholar]
- 75.Dasarathy S., Hatzoglou M. Hyperammonemia and proteostasis in cirrhosis. Curr Opin Clin Nutr Metab Care. 2018;21:30–36. doi: 10.1097/MCO.0000000000000426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Jepsen P., Vilstrup H., Mellemkjaer L., et al. Prognosis of patients with a diagnosis of fatty liver–a registry-based cohort study. Hepato-Gastroenterology. 2003;50:2101–2104. [PubMed] [Google Scholar]
- 77.Thomsen K.L., Hebbard L., Glavind E., et al. Non-alcoholic steatohepatitis weakens the acute phase response to endotoxin in rats. Liver Int. 2014;34:1584–1592. doi: 10.1111/liv.12547. [DOI] [PubMed] [Google Scholar]
- 78.Shawcross D.L., Wright G.A., Stadlbauer V., et al. Ammonia impairs neutrophil phagocytic function in liver disease. Hepatology. 2008;48:1202–1212. doi: 10.1002/hep.22474. [DOI] [PubMed] [Google Scholar]
- 79.Luo C., Shen G., Liu N., et al. Ammonia drives dendritic cells into dysfunction. J Immunol. 2014;193:1080. doi: 10.4049/jimmunol.1303218. [DOI] [PubMed] [Google Scholar]
- 80.Shawcross D.L., Davies N.A., Williams R., Jalan R. Systemic inflammatory response exacerbates the neuropsychological efects of induced hyperammonemia in cirrhosis. J Hepatol. 2004;40:247–254. doi: 10.1016/j.jhep.2003.10.016. [DOI] [PubMed] [Google Scholar]
- 81.Marini J.C., Broussard S.R. Hyperammonemia increases sensitivity to LPS. Mol Genet Metabol. 2006 Jun;88:131–137. doi: 10.1016/j.ymgme.2005.12.013. Epub 2006 Feb 23. PMID: 16497529. [DOI] [PubMed] [Google Scholar]
- 82.Thomsen K.L., Eriksen P.L., Kerbert A.J., De Chiara F., Jalan R., Vilstrup H. Role of ammonia in NAFLD: an unusual suspect. JHEP Rep. 2023 Apr 25;5 doi: 10.1016/j.jhepr.2023.100780. PMID: 37425212; PMCID: PMC10326708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Tomomura M., Tomomura A., Dewan M.A., Saheki T. Long-chain fatty acids suppress the induction of urea cycle enzyme genes by glucocorticoid action. FEBS Lett. 1996;399:310–312. doi: 10.1016/s0014-5793(96)01344-0. [DOI] [PubMed] [Google Scholar]
- 84.Zhu L.H., Armentano L.E., Bremmer D.R., Grummer R.R., Bertics S.J. Plasma concentration of urea, ammonia, glutamine around calving, and the relation of hepatic triglyceride, to plasma ammonia removal and blood acid-base balance. J Dairy Sci. 2000;83:734–740. doi: 10.3168/jds.S0022-0302(00)74935-6. [DOI] [PubMed] [Google Scholar]
- 85.Thomsen K.L., Gronbaek H., Glavind E., et al. Experimental nonalcoholic steatohepatitis compromises ureagenesis, an essential hepatic metabolic function. Am J Physiol Gastrointest Liver Physiol. 2014;307:G295–G301. doi: 10.1152/ajpgi.00036.2014. [DOI] [PubMed] [Google Scholar]
- 86.De Chiara F., Thomsen K.L., Habtesion A., et al. Ammonia scavenging prevents progression of fibrosis in experimental nonalcoholic fatty liver disease. Hepatology. 2020;71:874–892. doi: 10.1002/hep.30890. [DOI] [PubMed] [Google Scholar]
- 87.De Chiara F., Heeboll S., Marrone G., et al. Urea cycle dysregulation in non-alcoholic fatty liver disease. J Hepatol. 2018;69:905–915. doi: 10.1016/j.jhep.2018.06.023. [DOI] [PubMed] [Google Scholar]
- 88.Eriksen P.L., Vilstrup H., Rigbolt K., et al. Non-alcoholic fatty liver disease alters expression of genes governing hepatic nitrogen conversion. Liver Int. 2019;39:2094–2101. doi: 10.1111/liv.14205. [DOI] [PubMed] [Google Scholar]
- 89.Johnson N.D., Wu X., Still C.D., et al. Differential DNA methylation and changing cell-type proportions as fibrotic stage progresses in NAFLD. Clin epigenetics. 2021;13:152. doi: 10.1186/s13148-021-01129-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Sodum N., Kumar G., Bojja S.L., Kumar N., Rao C.M. Epigenetics in NAFLD/NASH: targets and therapy. Pharmacol Res. 2021;167 doi: 10.1016/j.phrs.2021.105484. [DOI] [PubMed] [Google Scholar]
- 91.Panasiuk A., Dzieciol J., Panasiuk B., Prokopowicz D. Expression of p53, Bax and Bcl-2 proteins in hepatocytes in non-alcoholic fatty liver disease. World J Gastroenterol. 2006;12:6198–6202. doi: 10.3748/wjg.v12.i38.6198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Gorg B., Karababa A., Schutz E., et al. O-GlcNAcylation-dependent upregulation of HO1 triggers ammonia-induced oxidative stress and senescence in hepatic encephalopathy. J Hepatol. 2019;71:930–941. doi: 10.1016/j.jhep.2019.06.020. [DOI] [PubMed] [Google Scholar]
- 93.Gao G., Yu Z., Yan J., et al. Lowering blood ammonia prevents hepatocyte injury and apoptosis. Int J Clin Exp Med. 2015;8:12347–12355. [PMC free article] [PubMed] [Google Scholar]
- 94.Hamberg O., Nielsen K., Vilstrup H. Effects of an increase in protein intake on hepatic efficacy for urea synthesis in healthy subjects and in patients with cirrhosis. JHepatol. 1992;14:237–243. doi: 10.1016/0168-8278(92)90164-k. [DOI] [PubMed] [Google Scholar]
- 95.Vilstrup H., Hansen B.A., Almdal T.P. Glucagon increases hepatic efficacy for urea synthesis. JHepatol. 1990;10:46–50. doi: 10.1016/0168-8278(90)90072-y. [DOI] [PubMed] [Google Scholar]
- 96.Fabbri A., Marchesini G., Bianchi G., et al. Unresponsiveness of hepatic nitrogen metabolism to glucagon infusion in patients with cirrhosis: dependence on liver cell failure. Hepatology. 1993;18:28–35. [PubMed] [Google Scholar]
- 97.Fabbri A., Bianchi G.P., Brizi M., et al. Effects of beta-blockade on hepatic conversion of amino acid nitrogen and on urea synthesis in cirrhosis. Metabolism. 1995;44:899–905. doi: 10.1016/0026-0495(95)90243-0. [DOI] [PubMed] [Google Scholar]
- 98.Marchesini G., Fabbri A., Bianchi G., Brizi M., Zoli M. Zinc supplementation and amino acid-nitrogen metabolism in patients with advanced cirrhosis. Hepatology. 1996;23:1084–1092. doi: 10.1053/jhep.1996.v23.pm0008621138. [DOI] [PubMed] [Google Scholar]
- 99.Jalan R., De Chiara F., Balasubramaniyan V., et al. Ammonia produces pathological changes in human hepatic stellate cells and is a target for therapy of portal hypertension. J Hepatol. 2016;64:823–833. doi: 10.1016/j.jhep.2015.11.019. [DOI] [PubMed] [Google Scholar]
- 100.Rombouts K., Marra F. Molecular mechanisms of hepatic fibrosis in nonalcoholic steatohepatitis. Dig Dis. 2010;28:229–235. doi: 10.1159/000282094. [DOI] [PubMed] [Google Scholar]
- 101.Vitale A., Svegliati-Baroni G., Ortolani A., et al. Epidemiological trends and trajectories of MAFLD associated hepatocellular carcinoma 2002-2033: the ITA.LI.CA database. Gut. 2023;74:141–152. doi: 10.1136/gutjnl-2021-324915. [DOI] [PubMed] [Google Scholar]
- 102.Lie S., Wang T., Forbes B., Proud C.G., Petersen J. The ability to utilise ammonia as nitrogen source is cell type specific and intricately linked to GDH, AMPK and mTORC1. Sci Rep. 2019;9:1461. doi: 10.1038/s41598-018-37509-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Lockwood A.H., McDonald J.M., Reiman R.E., et al. The dynamics of ammonia metabolism in man. Effects of liver disease and hyperammonemia. J Clin Invest. 1979;63:449–460. doi: 10.1172/JCI109322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Spinelli J.B., Yoon H., Ringel A.E., Jeanfavre S., Clish C.B., Haigis M.C. Metabolic recycling of ammonia via glutamate dehydrogenase supports breast cancer biomass. Science. 2017;358:941–946. doi: 10.1126/science.aam9305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Li L., Mao Y., Zhao L., et al. p53 regulation of ammonia metabolism through urea cycle controls polyamine biosynthesis. Nature. 2019;567:253–256. doi: 10.1038/s41586-019-0996-7. [DOI] [PubMed] [Google Scholar]
- 106.Li G., He Y., Liu H., et al. DNAJC24 is a potential therapeutic target in hepatocellular carcinoma through affecting ammonia metabolism. Cell Death Dis. 2022 May 24;13:490. doi: 10.1038/s41419-022-04953-z. PMID: 35606363; PMCID: PMC9127113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Deutsch-Link S., Moon A.M. The ongoing debate of serum ammonia levels in cirrhosis: the good, the bad, and the ugly. Am J Gastroenterol. 2023;118:10–13. doi: 10.14309/ajg.0000000000001966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Rai R., Saraswat V.A., Dhiman R.K. Gut microbiota: its role in hepatic encephalopathy. J Clin Exp Hepatol. 2015;5(suppl 1):S29–S36. doi: 10.1016/j.jceh.2014.12.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Tranah T.H., Ballester M.P., Carbonell-Asins J.A., et al. Plasma ammonia levels predict hospitalisation with liver-related complications and mortality in clinically stable outpatients with cirrhosis. J Hepatol. 2022 Dec;77:1554–1563. doi: 10.1016/j.jhep.2022.07.014. Epub 2022 Jul 22. PMID: 35872326. [DOI] [PubMed] [Google Scholar]
- 110.Balcar L., Krawanja J., Scheiner B., et al. Impact of ammonia levels on outcome in clinically stable outpatients with advanced chronic liver disease. JHEP Rep. 2023 Jan 23;5 doi: 10.1016/j.jhepr.2023.100682. PMID: 36873421; PMCID: PMC9976454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.D'Amico G., Morabito A., D'Amico M., et al. Clinical states of cirrhosis and competing risks. J Hepatol. 2018;68:563–576. doi: 10.1016/j.jhep.2017.10.020. [DOI] [PubMed] [Google Scholar]
- 112.Moreau R., Jalan R., Gines P., et al. Acute-on chronic liver failure is a distinct syndrome that develops in patients with acute decompensation of cirrhosis. Gastroenterology. 2013;144:1426–1437. doi: 10.1053/j.gastro.2013.02.042. [DOI] [PubMed] [Google Scholar]
- 113.Gairing S.J., Schleicher E.M., Labenz C. Diabetes mellitus–risk factor and potential future target for hepatic encephalopathy in patients with liver cirrhosis? Metab Brain Dis. 2023;38:1691–1700. doi: 10.1007/s11011-02201068-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Gairing S.J., Schleicher E.M., Galle P.R., Labenz C. Prediction and prevention of the first episode of overt hepatic encephalopathy in patients with cirrhosis. Hepatol Commun. 2023 Mar 17;7 doi: 10.1097/HC9.0000000000000096. PMID: 36930868; PMCID: PMC10027066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Ballester M.P., Tranah T.H., Balcar L., et al. Development and validation of the AMMON-OHE model to predict risk of overt hepatic encephalopathy occurrence in outpatients with cirrhosis. J Hepatol. 2023 Oct;79:967–976. doi: 10.1016/j.jhep.2023.05.022. Epub 2023 Jun 3. Erratum in: J Hepatol. 2023 Oct 31;: PMID: 37277075. [DOI] [PubMed] [Google Scholar]
- 116.Kumar R., Mehta G., Jalan R. Acute-on-chronic liver failure. Clin Med. 2020 Sep;20:501–504. doi: 10.7861/clinmed.2020-0631. PMID: 32934045; PMCID: PMC7539713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Shalimar, Sheikh M.F., Mookerjee R.P., Agarwal B., Acharya S.K., Jalan R. Prognostic role of ammonia in patients with cirrhosis. Hepatology. 2019 Sep;70:982–994. doi: 10.1002/hep.30534. Epub 2019 Mar 21. PMID: 30703853. [DOI] [PubMed] [Google Scholar]
- 118.Shalimar Rout G., Kumar R., Singh A.D., et al. Persistent or incident hyperammonemia is associated with poor outcomes in acute decompensation and acute-on-chronic liver failure. JGH Open. 2020 Feb 28;4:843–850. doi: 10.1002/jgh3.12314. PMID: 33102753; PMCID: PMC7578315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Verma N., Dhiman R.K., Choudhury A., et al. APASL ACLF Research Consortium (AARC) for APASL ACLF working Party Dynamic assessments of hepatic encephalopathy and ammonia levels predict mortality in acute-on-chronic liver failure. Hepatol Int. 2021 Aug;15:970–982. doi: 10.1007/s12072-021-10221-7. Epub 2021 Jul 18. PMID: 34275111. [DOI] [PubMed] [Google Scholar]
- 120.Hu C., Huang K., Zhao L., et al. Serum ammonia is a strong prognostic factor for patients with acute-on-chronic liver failure. Sci Rep. 2020;10 doi: 10.1038/s41598-020-73603-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Chiriac S., Stanciu C., Cojocariu C., et al. Role of ammonia in predicting the outcome of patients with acute-on-chronic liver failure. World J Clin Cases. 2021 Jan 26;9:552–564. doi: 10.12998/wjcc.v9.i3.552. PMID: 33553393; PMCID: PMC7829715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Cai Y.-J., Dong J.-J., Chen R.-C., et al. Serum ammonia variation predicts mortality in patients with hepatitis B virus-related acute-on-chronic liver failure. Front Microbiol. 2023;14 doi: 10.3389/fmicb.2023.1282106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Hu Chenxia, Huang Kaizhou, Zhao Lingfei, Zhang Fen, Wu Zhongwen, Li Lanjuan. Serum ammonia is a strong prognostic factor for patients with acute-on-chronic liver failure. Sci Rep. 2020;10 doi: 10.1038/s41598-020-73603-1. AN 16970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Bhatia V., Singh R., Acharya S.K. Predictive value of arterial ammonia for complications and outcome in acute liver failure. Gut. 2006 Jan;55:98–104. doi: 10.1136/gut.2004.061754. Epub 2005 Jul 15. PMID: 16024550; PMCID: PMC1856380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Bernal W., Hall C., Karvellas C.J., Auzinger G., Sizer E., Wendon J. Arterial ammonia and clinical risk factors for encephalopathy and intracranial hypertension in acute liver failure. Hepatology. 2007 Dec;46:1844–1852. doi: 10.1002/hep.21838. PMID: 17685471. [DOI] [PubMed] [Google Scholar]
- 126.Kumar R., Shalimar, Sharma H., et al. Persistent hyperammonemia is associated with complications and poor outcomes in patients with acute liver failure. Clin Gastroenterol Hepatol. 2012 Aug;10:925–931. doi: 10.1016/j.cgh.2012.04.011. Epub 2012 Apr 17. PMID: 22521861. [DOI] [PubMed] [Google Scholar]
- 127.Fuhrmann V., Drolz A., Jaeger B., et al. Prognostic relevance of arterial ammonia levels in different acute and acute-on-chronic liver diseases. Crit Care. 2012;16(suppl 1):P390. doi: 10.1186/cc10997. [DOI] [Google Scholar]
- 128.Kumar R., Shalimar, Sharma H., et al. Prospective derivation and validation of early dynamic model for predicting outcome in patients with acute liver failure. Gut. 2012 Jul;61:1068–1075. doi: 10.1136/gutjnl-2011-301762. Epub 2012 Feb 15. PMID: 22337947. [DOI] [PubMed] [Google Scholar]
- 129.O'Grady J.G., Alexander G.J., Hayllar K.M., Williams R. Early indicators of prognosis in. Liver. Gastroenterology. 1989 Aug;97:439–445. doi: 10.1016/0016-5085(89)90081-4. [DOI] [PubMed] [Google Scholar]
- 130.Bernuau J., Goudeau A., Poynard T., et al. Multivariate analysis of prognostic factors in fulminant hepatitis B. Hepatology. 1986 Jul;6:648–651. doi: 10.1002/hep.1840060417. [DOI] [PubMed] [Google Scholar]
- 131.Kim W.R., Biggins S.W., Kremers W.K., et al. Hyponatremia and mortality among patients on the liver-transplant waiting list. N Engl J Med. 2008;359:1018–1026. doi: 10.1056/NEJMoa0801209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Dhiman R.K., Jain S., Maheshwari U., et al. Early indicators of prognosis in fulminant hepatic failure: an assessment of the model for end-stage liver disease (MELD) and King's College Hospital criteria. Liver Transplant. 2007;13:814–821. doi: 10.1002/lt.21050. [DOI] [PubMed] [Google Scholar]
- 133.Anand A.C., Nightingale P., Neuberger J.M. Early indicators of prognosis in fulminant hepatic failure: an assessment of the King's criteria. J Hepatol. 1997;26:62–68. doi: 10.1016/s0168-8278(97)80010-4. [DOI] [PubMed] [Google Scholar]
- 134.Shalimar, Sonika U., Kedia S., et al. Comparison of dynamic changes among various prognostic scores in viral hepatitis-related acute liver failure. Ann Hepatol. 2018;17:403–412. doi: 10.5604/01.3001.0011.7384. [DOI] [PubMed] [Google Scholar]
- 135.Koch D.G., Speiser J.L., Durkalski V., et al. The natural history of severe acute liver injury. Am J Gastroenterol. 2017 Sep;112:1389–1396. doi: 10.1038/ajg.2017.98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Liu J., Lkhagva E., Chung H.J., Kim H.J., Hong S.T. The pharmabiotic approach to treat hyperammonemia. Nutrients. 2018 Jan 28;10:140. doi: 10.3390/nu10020140. PMID: 29382084; PMCID: PMC5852716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Rajpurohit S., Musunuri B., Shailesh, Basthi Mohan P., Shetty S. Novel drugs for the management of hepatic encephalopathy: still a long journey to travel. J Clin Exp Hepatol. 2022 Jul-Aug;12:1200–1214. doi: 10.1016/j.jceh.2022.01.012. Epub 2022 Jan 31. PMID: 35814520; PMCID: PMC9257922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Zhu Z.Y., Cui D., Gao H., et al. Efficient synthesis and activity of beneficial intestinal flora of two lactulose-derived oligosaccharides. Eur J Med Chem. 2016;114:8–13. doi: 10.1016/j.ejmech.2016.03.007. [DOI] [PubMed] [Google Scholar]
- 139.Bajaj J.S., Riggio O. Drug therapy: rifaximin. Hepatology. 2010;52:1484–1488. doi: 10.1002/hep.23866. [DOI] [PubMed] [Google Scholar]
- 140.Rose C.F. Ammonia-lowering strategies for the treatment of hepatic encephalopathy. Clin Pharmacol Ther. 2012;92:321–331. doi: 10.1038/clpt.2012.112. [DOI] [PubMed] [Google Scholar]
- 141.Morgan M.H., Read A.E., Speller D.C. Treatment of hepatic encephalopathy with metronidazole. Gut. 1982;23:1–7. doi: 10.1136/gut.23.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Barshop B.A., Breuer J., Holm J., Leslie J., Nyhan W.L. Excretion of hippuric acid during sodium benzoate therapy in patients with hyperglycinaemia or hyperammonaemia. J Inherit Metab Dis. 1989;12:72–79. doi: 10.1007/BF01805533. [DOI] [PubMed] [Google Scholar]
- 143.Pena-Quintana L., Llarena M., Reyes-Suarez D., Aldamiz-Echevarria L. Profile of sodium phenylbutyrate granules for the treatment of urea-cycle disorders: patient perspectives. Patient Prefer Adherence. 2017;11:1489–1496. doi: 10.2147/PPA.S136754. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Rockey D.C., Vierling J.M., Mantry P., et al. Randomized, double-blind, controlled study of glycerol phenylbutyrate in hepatic encephalopathy. Hepatology. 2014;59:1073–1083. doi: 10.1002/hep.26611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Scaglia F., Carter S., O'Brien W.E., Lee B. Effect of alternative pathway therapy on branched chain amino acid metabolism in urea cycle disorder patients. Mol Genet Metabol. 2004;81(suppl. 1):S79–S85. doi: 10.1016/j.ymgme.2003.11.017. [DOI] [PubMed] [Google Scholar]
- 146.Dam G., Keiding S., Munk O.L., et al. Branched-chain amino acids increase arterial blood ammonia in spite of enhanced intrinsic muscle ammonia metabolism in patients with cirrhosis and healthy subjects. Am J Physiol Gastrointest Liver Physiol. 2011;301:G269–G277. doi: 10.1152/ajpgi.00062.2011. [DOI] [PubMed] [Google Scholar]
- 147.Daniotti M., la Marca G., Fiorini P., Filippi L. New developments in the treatment of hyperammonemia: emerging use of carglumic acid. Int J Gen Med. 2011;4:21. doi: 10.2147/IJGM.S10490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Haberle J., Boddaert N., Burlina A., et al. Suggested guidelines for the diagnosis and management of urea cycle disorders. Orphanet J Rare Dis. 2012;7:32. doi: 10.1186/1750-1172-7-32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Soarez P.C., Oliveira A.C., Padovan J., Parise E.R., Ferraz M.B. A critical analysis of studies assessing L-ornithine-L-aspartate (LOLA) in hepatic encephalopathy treatment. Arq Gastroenterol. 2009;46:241–247. doi: 10.1590/s0004-28032009000300019. [DOI] [PubMed] [Google Scholar]
- 150.Jover-Cobos M., Noiret L., Lee K., et al. Ornithine phenylacetate targets alterations in the expression and activity of glutamine synthase and glutaminase to reduce ammonia levels in bile duct ligated rats. J Hepatol. 2014;60:545–553. doi: 10.1016/j.jhep.2013.10.012. [DOI] [PubMed] [Google Scholar]
- 151.Maldonado C., Guevara N., Silveira A., Fagiolino P., Vazquez M. L-carnitine supplementation to reverse hyperammonemia in a patient undergoing chronic valproic acid treatment: a case report. J Int Med Res. 2017;45:1268–1272. doi: 10.1177/0300060517703278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Najimi M., Defresne F., Sokal E.M. Concise review: updated advances and current challenges in cell therapy for inborn liver metabolic defects. Stem Cells Transl. Med. 2016;5:1117–1125. doi: 10.5966/sctm.2015-0260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Meyburg J., Hoffmann G.F. Liver, liver cell and stem cell transplantation for the treatment of urea cycle defects. Mol Genet Metabol. 2010;100(suppl. 1):S77–S83. doi: 10.1016/j.ymgme.2010.01.011. [DOI] [PubMed] [Google Scholar]
- 154.Hu C., Tai D.S., Park H., et al. Minimal ureagenesis is necessary for survival in the murine model of hyperargininemia treated by AAV-based gene therapy. Gene Ther. 2015;22:111–115. doi: 10.1038/gt.2014.106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Tsipotis E., Shuja A., Jaber B.L. Albumin dialysis for liver failure: a systematic review. Adv Chron Kidney Dis. 2015;22:382–390. doi: 10.1053/j.ackd.2015.05.004. [DOI] [PubMed] [Google Scholar]
- 156.Mehta K.P. Dialysis therapy in children. J Indian Med Assoc. 2001;99:368–373. [PubMed] [Google Scholar]
- 157.Gupta S., Fenves A.Z., Hootkins R. The role of RRT in hyperammonemic patients. Clin J Am Soc Nephrol. 2016 Oct 7;11:1872–1878. doi: 10.2215/CJN.01320216. Epub 2016 May 19. PMID: 27197910; PMCID: PMC5053785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Tsiaoussis J., Newsome P.N., Nelson L.J., Hayes P.C., Plevris J.N. Which hepatocyte will it be? Hepatocyte choice for bioartificial liver support systems. Liver Transplant. 2001;7:2–10. doi: 10.1053/jlts.2001.20845. [DOI] [PubMed] [Google Scholar]
- 159.159a Dam G., Ott P., Aagaard N.K., Vilstrup H. Branched-chain amino acids and muscle ammonia detoxification in cirrhosis. Metab Brain Dis. 2013;28:217–220. doi: 10.1007/s11011-013-9377-3. [DOI] [PubMed] [Google Scholar]; 159b da Fonseca-Wollheim F. Pre-analytical increase of ammonia in blood specimens from healthy subjects. Clin Chem. 1990;36:1483–1487. [PubMed] [Google Scholar]
- 160.Brusilow S.W., Koehler R.C., Traystman R.J., Cooper A.J. Astrocyte glutamine synthetase: importance in hyper-ammonemic syndromes and potential target for therapy. Neurotherapeutics. 2010;7:452–470. doi: 10.1016/j.nurt.2010.05.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Kramer L., Tribl B., Gendo A., et al. Partial pressure of ammonia versus ammonia in hepatic encephalopathy. Hepatology. 2000;31:30–34. doi: 10.1002/hep.510310107. [DOI] [PubMed] [Google Scholar]
- 162.Ong J.P., Aggarwa lA., Krieger D., et al. Correlation between ammonia levels and the severityof hepatic encephalopathy. Am J Med. 2003;114:188–193. doi: 10.1016/s0002-9343(02)01477-8. [DOI] [PubMed] [Google Scholar]
- 163.Mount Sinai health library. https://www.mountsinai.org/health-library/tests/ammonia-blood-test#:∼:text=The%20normal%20range%20is%2015,of%20your%20specific%20test%20results.
- 164.Clemmesen J.O., Larsen F.S., Kondrup J., Hansen B.A., Ott P. Cerebral herniation in patients with acute liver failure is correlated with arterial ammonia concentration. Hepatology. 1999 Mar;29:648–653. doi: 10.1002/hep.510290309. PMID: 10051463. [DOI] [PubMed] [Google Scholar]
- 165.Jalan R., O Damink S.W., Deutz N.E., Lee A., Hayes P.C. Moderate hypothermia for uncontrolled intracranial hypertension in acute liver failure. Lancet. 1999 Oct 2;354:1164–1168. doi: 10.1016/s0140-6736(98)12440-6. PMID: 10513710. [DOI] [PubMed] [Google Scholar]
- 166.Clemmesen J.O., Kondrup J., Ott P. Splanchnic and leg exchange of amino acids and ammonia in acute liver failure. Gastroenterology. 2000 Jun;118:1131–1139. doi: 10.1016/s0016-5085(00)70366-0. PMID: 10833488. [DOI] [PubMed] [Google Scholar]
- 167.Bhatia V., Batra Y., Acharya S.K. Prophylactic phenytoin does not improve cerebral edema or survival in ALF—a controlled clinical trial. J Hepatol. 2004;41:89–96. doi: 10.1016/j.jhep.2004.03.017. [DOI] [PubMed] [Google Scholar]
- 168.Yue Li, Yao Zhipeng, Li Yunlong, et al. 28 June 2021. Prognostic Value of Serum Ammonia in Critical Patients with Non-hepatic Disease : A Prospective, Observational, Multicenter Study. PREPRINT (Version 1) available at: Research Square. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Duarte T., Fidalgo P., Karvellas C.J., Cardoso F.S. What every Intensivist should know about the role of ammonia in liver failure. J Crit Care. 2023 Nov 7 doi: 10.1016/j.jcrc.2023.154456. Epub ahead of print. PMID: 37945461. [DOI] [PubMed] [Google Scholar]
- 170.Shakerdi L., Ryan A. Drug-induced hyperammonaemia. J Clin Pathol. 2023 Aug;76:501–509. doi: 10.1136/jcp-2022-208644. Epub 2023 May 10. PMID: 37164630. [DOI] [PubMed] [Google Scholar]
- 171.Wang P., Yan J., Shi Q., et al. Relationship between nonhepatic serum ammonia levels and sepsis-associated encephalopathy: a retrospective cohort study. Emerg Med Int. 2023 Oct 12;2023 doi: 10.1155/2023/6676033. PMID: 37869361; PMCID: PMC10590267. [DOI] [PMC free article] [PubMed] [Google Scholar]