Structured Abstract
Background-
Atrioventricular valve regurgitation (AVVR) is a devastating complication in children and young adults with congenital heart disease (CHD), particularly in patients with single ventricle physiology. Transcatheter edge-to-edge repair (TEER) is a rapidly expanding, minimally invasive option for the treatment of AVVR in adults that avoids the morbidity and mortality associated with open heart surgery. However, application of TEER in in CHD and in children is quite novel. We describe the development of a peri-procedural protocol including image-derived pre-intervention simulation, with successful application to four patients.
Methods-
A standardized screening and planning process was developed using cardiac magnetic resonance imaging, 3D echocardiography and both virtual and physical simulation. Procedures were performed using the MitraClip G4 system and patients were clinically followed post-intervention.
Results-
A series of 4 CHD patients with at least severe AVVR were screened for suitability for TEER with the MitraClip system: three patients had single ventricle physiology and Fontan palliation, and one had repair of a common atrioventricular canal defect. Each patient had at least severe systemic AVVR and was considered at prohibitively high risk for surgical repair. Each patient underwent a standardized pre-procedural screening protocol and image-derived modeling followed by the TEER procedure with successful clip placement at the intended location in all cases.
Conclusions-
The early results of our protocolized efforts to introduce TEER repair of severe AV valve regurgitation with MitraClip into the CHD population within our institution are encouraging. Further investigations of the use of TEER in this challenging population are warranted.
Keywords: Congenital Heart Disease, Echocardiography, Valvular Heart Disease
Graphical abstract

Condensed Abstract
Atrioventricular valve regurgitation (AVVR) is a devastating complication in children and young adults with congenital heart disease(CHD), and is particularly morbidity in those with single ventricle physiology. The application of Transcatheter-Edge-to-Edge Repair (TEER) in CHD holds promise but is in its infancy with limited description of outcomes. We describe the development of an image-derived screening and pre-interventional planning protocol followed by successful clinical application of TEER to four patients less than 21 years of age with CHD including 3 patients with palliated single ventricle physiology. Further collaborative investigations of the use of TEER in this challenging population are warranted.
Introduction
Atrioventricular valve regurgitation(AVVR) is the most prevalent form of valve disease in developed countries. Transcatheter edge-to-edge repair (TEER) is a rapidly expanding, minimally invasive option for the treatment of AVVR in adults that avoids the morbidity and mortality associated with open heart surgery.(1,2) TEER has proven to be an effective and low risk therapy in adults with acquired mitral and tricuspid valve disease who are high or moderate risk for cardiac surgery.(3–5) AVVR is also a devastating complication in children and young adults with congenital heart disease (CHD), particularly in patients with single ventricle physiology. As such, the development and application of an effective and minimally invasive therapy for AVVR would address a critical unmet need.(6,7) However, application of TEER in congenital heart disease, and in particular palliated single ventricle patients, is novel with limited description of outcomes.(8–12)
TEER therapy relies upon delivering a device to a precise location on the valve via a potentially circuitous path through the vasculature. As such, one challenge to successful application of this therapy in CHD is delivering a clip to a precise location within the constraints of highly variable and often surgically altered anatomy using “off the shelf” systems designed for structurally normal anatomy.(13,14) To date, the volume of CHD patients undergoing TEER is low, and the heterogeneity of anatomy is high, making the learning curve even more challenging than in comparatively homogeneous and high-volume populations seen in typical adult populations.
Inspired by the pathways established recently for the introduction of other novel valve therapies into the CHD population(15) we sought to establish a standardized image-based approach to patient screening and to TEER therapy in high surgical risk CHD patients at our institution.(13,14) Our long term goal is to build on this early experience and to facilitate the adaptation of TEER and other catheter-based valve therapies in CHD patients suffering from AVVR in our institution and beyond.
In this report, we describe our experience with image-derived planning and successful execution of TEER in a series of four patients with CHD, including three with single ventricle physiology at the Fontan stage, and one patient with common atrioventricular canal, along with their clinical follow-up to date.
METHODS
Subjects
The study was conducted in accordance with the Declaration of Helsinki, and was performed according to a protocol approved by the institutional review board at the Children’s Hospital of Philadelphia. Subjects were clinically identified as individuals who would benefit from reduction in atrioventricular valve regurgitation, but were determined not to be candidates for surgical valve repair.
Echo Image Acquisition and Assessment
Transesophageal 3DE images of the atrioventricular (AV) valve were acquired as per lab protocol using EKG gated, Full Volume or 3D Zoom mode. Transesophageal X8 probes were used with the Philips EPIQ 7 ultrasound systems (Philips Medical, Cambridge, MA). AVVR was qualitatively assessed by a single observer (YW) examining two-dimensional echocardiography (2DE) color-doppler images. The severity of AVVR was graded with available data based on established and proposed criteria(16,17), acknowledging the lack of a validated quantitative system of grading in congenital heart disease(18). 3D vena contracta was measured using multiplane reconstruction (YW). Tomtec Imaging Arena version 4.6 (Tomtec, Unterschleisshem, Germany) was used to quantify the longitudinal strain and estimated ejection fraction using 4-chamber images.
Cardiac Magnetic Resonance Image Acquisition
Cardiac Magnetic Resonance (CMR) images in the single ventricle patients were acquired on a 1.5 Tesla scanner (Magnetom Avento Fit, Siemens Healthcare, Erlanden, Germany) using a cardiac-gated inversion recovery FLASH sequence with ferumoxytol (Feraheme, AMAG Pharmaceuticals, Massachusetts) as the contrast agent. Ferumoxytol was used to ensure consistent and homogeneous visualization of the vasculature.
Virtual and Physical Modeling and Simulation
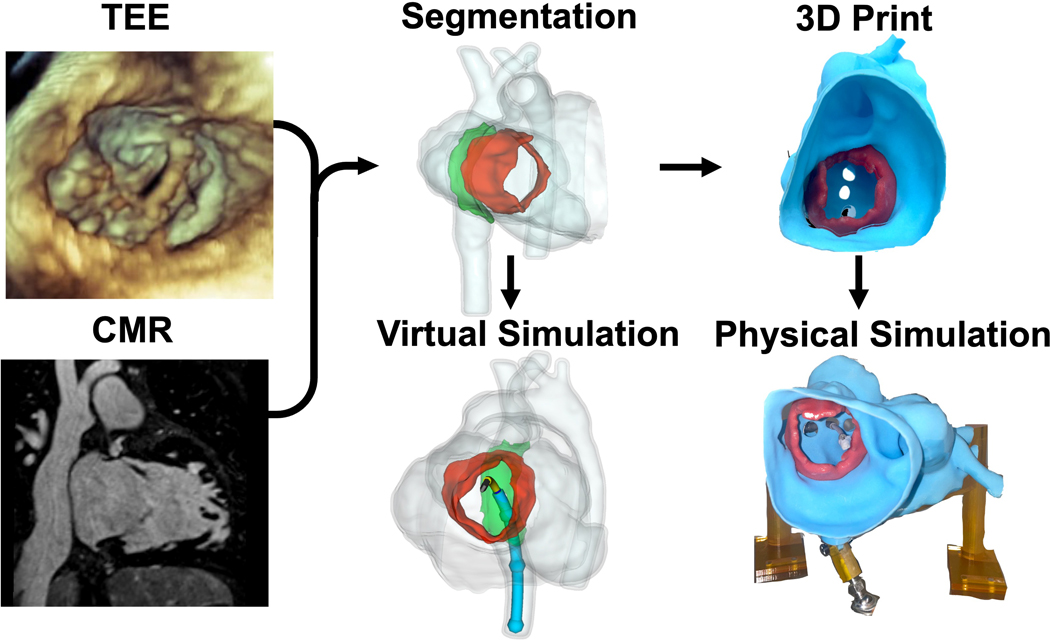
CMR images in Digital Imaging and Communications in Medicine (DICOM) format were imported into 3D Slicer (www.slicer.org).(19) Shell models of the vasculature and valves leaflets were segmented using 3D Slicer and SlicerHeart as previously described.(13,14,20) If the leaflets were not well visualized in the CMR they were segmented from 3DE images using SlicerHeart and registered within the CMR-derived model using the annulus and commissures of the valve in 3D Slicer using the Transforms module.(20) Virtual modeling of catheter trajectory was performed to investigate optimal access points and baffle puncture sites using a realistically constrained virtual TEER system in the ValveClip Device Simulator in the SlicerHeart extension as we have previously described(Video 1).(14,20) In particular, various septal and baffle puncture sites were virtually evaluated to optimize standoff of the system from the valve and delivery of the clip to area(s) of regurgitation. The patient specific segmented heart volumes were exported into Autodesk Fusion 360 (Autodesk, San Francisco, CA).The post-processed models were exported as stereolithography (STL) files that were 3D printed on a material jetting printer (J750, Stratasys, Eden Prairie, MN) in a rigid polycarbonate material (VeroVivid, Stratasys, Eden Prairie, MN) for the heart and flexible rubber material (Agilus30, Stratasys, Eden Prairie, MN) for Fontan puncture site and the valve (Figure 1). A MitraClip G4 system (Abbott Medical) was introduced into the anatomically oriented patient-specific model to simulate approaches to the atrioventricular valve as we have previously described(Figure 1, Video 1). In the Fontan patients, up to three baffle puncture sites were simulated to confirm sites previously explored on virtual modeling. (13)
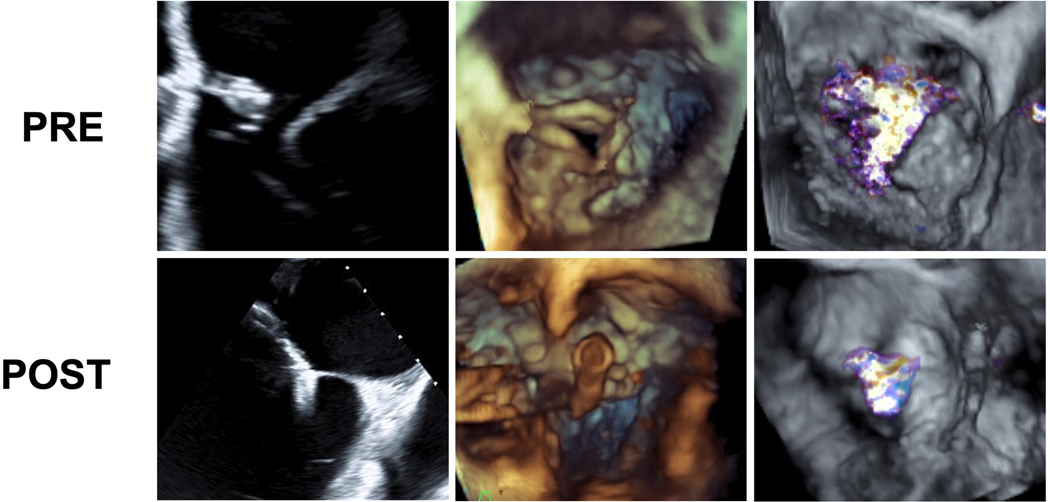
Figure 2. Case 1 with Hypoplastic Left Heart Syndrome and a Fontan Circulation.
Top: 2D, 3D and 3D color images before intervention. Mid: Bottom: 2D, 3D and 3D color ages after delivery of first clip near the anterior-septal commissure with reduction in regurgitation. Bottom: 2D, 3D and 3D color ages after delivery of second clip centrally between the anterior and septal leaflets with significant reduction in regurgitation.
RESULTS
A series of 4 CHD patients with at least severe AVVR were screened for TEER with the MitraClip G4 system: three with single ventricle physiology who had undergone Fontan palliation, and one who had repair of a common atrioventricular canal defect. Each patients had severe or greater systemic AVVR, and each was considered at prohibitively high risk for surgical repair. A standardized pre-procedural screening protocol and image-derived modeling were applied followed by the TEER procedure with successful clip placement at the intended location in all cases. All patients were intubated and had general anesthesia for the procedure. The patient clinical course is outlined below, in Table 1, Supplementary Tables 1-3 and Videos 2–4.
Table 1.
Comparison of Echocardiographic and Hemodynamic Metrics Pre and Post TEER Procedure
| Variable | Case #1 (HLHS-Fontan) | Case #2 (HLHS-Fontan) | Case #3 (CAVC-Left AV Valve) | Case #4 (Unbalanced CAVC-Fontan) |
|---|---|---|---|---|
|
| ||||
| Vena Contracta Area (cm2) | ||||
| Pre-Procedure | 2.5 | 0.7 | 1.1 | 3.9 |
| Post Clip 1 | 1.6 | 0.2 | 0.5 | 3.1 |
| Post Clip 2 | 0.3 | N/A | N/A | 1.5 |
| Mean Inflow gradients (mmHg) | ||||
| Pre-Procedure | 3 | 1 | 7 | 2 |
| Post Clip 1 | 4 | 1 | 7 | 3 |
| Post Clip 2 | 3 | N/A | N/A | 3 |
| Atrial Pressures (mmHg) | ||||
| Pre-Procedure (mean, v-wave) | 15, 20 | 9, 13 | 24, 50 | 11, 22 |
| Post Clip 1 (mean, v-wave) | N/A | 7, 9 | 22, 28 | N/A |
| Post Clip 2 (mean, v-wave) | 13, 18 | N/A | N/A | 12, 22 |
| AVVR Qualitative Grading | ||||
| Pre-Procedure | Massive-Torrential | Severe-Massive | Severe | Torrential |
| Post Clip 1 | Severe | Mild | Moderate | Severe |
| Post Clip 2 | Mild-Moderate | N/A | N/A | Severe |
| Estimated Ventricular Ejection Fraction (%) | ||||
| Pre-Procedure | 49 | 32 | 42 | 39 |
| Post Clip 1 | 44 | 43 | 31 | 39 |
| Post Clip 2 | 38 | N/A | N/A | 40 |
| Last follow-up | 58 | 40 | 33 | 45 |
TEER= Transcatheter Edge-to-Edge-Repair; HLHS= Hypoplastic left heart syndrome; CAVC= Common Atrioventricular Canal; AV= Atrioventricular; AVVR=Atrioventricular Valve Regurgitation
Case presentation 1 (Hypoplastic Left Heart Syndrome with Fontan Circulation):
The patient is a 12-year-old boy with hypoplastic left heart syndrome (HLHS) consisting of severe mitral stenosis and aortic atresia who underwent staged palliation, culminating in placement of an 18mm extracardiac fenestrated Fontan conduit nine years prior. He had since developed severe tricuspid regurgitation with clinical symptoms of extremity and facial edema, fatigue, palpitations and early protein losing enteropathy (PLE).. He had several hospitalizations for heart failure exacerbations requiring milrinone therapy and IV diuretics. He underwent transplant evaluation and was referred for consideration of surgical tricuspid valve repair prior to listing for transplant. TEER was offered as a potential alternative to surgery.
The patient was evaluated using the imaging, modeling, and simulation protocol described above (Figure 1, Video 3). At the time of his TEER procedure, pre-clip TEE demonstrated massive to torrential tricuspid regurgitation through a large regurgitant jet spanning most of the anterior-septal coaptation zone. 3D vena contracta of the regurgitant jet was 2.5 cm2 (Table 1). The mean inflow gradient was 3 mmHg with low normal RV systolic function. Invasive hemodynamics were notable for elevated Fontan pressure of 18 mmHg and RA pressure of mean 15 mmHg (v-wave 20 mmHg).
The existing Fontan fenestration was dilated with an 8mm balloon and the MitraClip guide catheter advanced into the right atrium. The clip delivery system was introduced and a MitralClip XTW was placed in the midportion of coaptation between the anterior and septal leaflets with good capture of both leaflets. The tricuspid regurgitation remained severe with a slightly smaller 3D vena contracta of 1.57 cm2; therefore an additional clip (MitraClip XT) was positioned and deployed just inferior to the initial XTW clip within the residual more central coaptation gap between the anterior and septal leaflets (Figure 2). Tricuspid regurgitation decreased to moderate with 3D vena contracta of 0.32 cm2 and decrease of atrial pressure to mean 13 mmHg (v-wave 18 mmHg).
Figure 3. Case 2 with Hypoplastic Left Heart Syndrome and a Fontan Circulation.
Top: 2D, 3D and 3D color images before intervention. Bottom: 2D, 3D and 3D color ages after delivery of a single clip between the anterior and septal leaflets with significant reduction in regurgitation.
The procedure was well-tolerated with post-intervention junctional tachycardia managed with rate control with ivabradine and atenolol until resolution. He reported improvement in quality of life with increased energy, less significant edema and improvement to New York Heart Association (NYHA) functional class II. His most recent echocardiogram (10 months following procedure) demonstrated moderate to severe tricuspid regurgitation and normal RV systolic function nine months post-procedure (Supplemental Table 1). His albumin and brain natriuretic peptides have normalized, he has tolerated reduced diuretics, and he has avoided the need for hospital readmission. He is playing on a competitive travel baseball team, further speaking to his improved functional capacity.
Case presentation 2 (Hypoplastic Left Heart Syndrome with Fontan Circulation):
The patient is a 20 year-old woman with HLHS consisting of mitral atresia and aortic atresia who underwent staged palliation culminating in a fenestrated lateral tunnel Fontan 18 years prior. She had developed progressive, severe tricuspid regurgitation with symptoms of heart failure with fatigue, ascites and peripheral edema. She was referred for tricuspid valve repair and underwent pre-procedural imaging, modeling, and simulation (Video 4).
At the time of her TEER procedure, pre-clip TEE demonstrated severe to massive tricuspid regurgitation with 3D vena contracta of 0.65 cm2 (Table 1, Figure 3). The coaptation gap was largely central with extension into the anteroseptal commissure. The mean inflow gradient was 1 mmHg with mild to moderately diminished RV systolic function. Invasive hemodynamics were notable for Fontan pressure of 14 mmHg and atrial pressure of mean 9 mmHg (v-wave 13 mmHg). There was no patent Fontan fenestration.
Figure 4. Case 3 with Complete Atrioventricular Canal and Left Atrioventricular Valve Regurgitation).
Top: 2D, 3D and 3D color images before intervention. Bottom: 2D, 3D and 3D color images after delivery of single clip between the superior and inferior bridging leaflet (to close the open “cleft”).
A transbaffle puncture was performed with a transseptal needle and the Brockenbrough technique. The puncture was dilated with an 8mm balloon and the guide catheter advanced into the right atrium, followed by the clip delivery system. A single MitraClip XTW was positioned across the area of regurgitation between the anterior and septal leaflets and deployed. The tricuspid regurgitation improved to mild (Figure 3) with 3D vena contracta decreasing to 0.159 cm2 , inflow gradient of 1mmHg, with normal RV systolic function. Right atrial pressure decreased to mean 7 mmHg (v-wave 9 mmHg).
The procedure was well-tolerated with discharge the following morning and no complications to date in the five months since TEER. At her four-month outpatient follow-up, she reported feeling clinically well with NYHA functional class I, no documented ectopy, and no recurrence of her protein-losing enteropathy symptoms. Her most recent echocardiogram demonstrated mild to moderate tricuspid regurgitation and normal RV systolic function (Supplemental Table 2).
Case presentation 3 (Complete Atrioventricular Canal):
The patient is a 15-year-old girl with trisomy 21, complete common atrioventricular canal defect who underwent complete repair as an infant. She developed progressive left and right AVVR with heart failure symptoms. Eight years prior to TEER, she underwent surgical left and right atrioventricular valvuloplasty. Her left AVVR continued to progress after surgery with two prolonged admissions for heart failure, elevated B-natriuretic peptide, fluid retention, and renal injury. She was referred for transcatheter valve repair after pre-procedural imaging, modeling, and simulation (Video1, Video 4).
At the time of TEER, pre-clip TEE demonstrated greater than severe left AVVR with a 3D vena contracta of 1.07 cm2 (Table 1), a large coaptation gap centrally between the superior and inferior bridging leaflets (this region is commonly referred to as the “cleft”) and 7 mmHg inflow gradient. Biventricular function was mildly diminished. Invasive hemodynamics notable for elevated LA pressure of mean 24 mmHg (v-wave 50 mmHg) and LV end diastolic pressure of 20 mmHg.
A transseptal puncture was performed and dilated with a 7mm balloon. The MitraClip guide catheter was advanced into the left atrium. The clip delivery system was introduced. A MitraClip XTW was positioned perpendicular to the line of coaptation between the superior and inferior bridging leaflets and deployed (Figure 4). The left AV valve regurgitation decreased to moderate with 3D vena contracta 0.54 cm2 (Figure 4, Video 4). Mean inflow gradient was 7 mmHg (unchanged from pre-intervention). LV systolic function decreased slightly to mild to moderately diminished. The LA pressure decreased to mean 22mmHg (v-wave 28 mmHg).
Figure 5. Case 4 with Unbalanced Complete Atrioventricular Canal, Interrupted Inferior Vena Cava, and a Fontan Circulation.
Top: 2D, 3D and 3D color images before intervention. Mid: 2D, 3D and 3D color ages after delivery of first clip. Bottom: 2D, 3D and 3D color ages after delivery of second clip with significant reduction in regurgitation.
The procedure was well-tolerated with brief runs of non-sustained ventricular tachycardia one-week post-procedure that were treated with mexiletine and nadolol. She was weaned from milrinone and discharged to home. At her two-month outpatient follow-up, she reported improved energy level with NYHA functional class II. Her ventricular ectopy had resolved, B-natriuretic peptide had decreased, and she had not required hospital re-admission. Her most recent echocardiogram demonstrated moderate left AVVR with a mean inflow gradient of 10 mmHg and moderately diminished LV systolic function(Supplemental Table 3).
Case Presentation 4: (Complete Atrioventricular Canal)
The patient is a 21-year-old woman with heterotaxy syndrome (polysplenia type), complete common AV canal defect severely unbalanced to the right, bilateral SVC with bridging vein, and interrupted IVC with azygous continuation. She had undergone staged single ventricle palliation with a Norwood procedure, right bidirectional Glenn with left Kawashima and a hepatic vein inclusion Fontan with an extracardiac fenestrated conduit; however, the hepatic vein conduit spontaneously occluded with thrombus. She developed progressive, severe common AV valve regurgitation, symptoms of dyspnea, palpitations and peripheral edema, and worsening hypoxemia due to significant pulmonary arteriovenous malformations. She had required admission for diuresis and was deemed too high risk a transplant candidate by multiple institutions. She was referred for potential TEER. Virtual and physical modeling suggested that the atrial chamber could be accessed via the right internal jugular vein by perforating through the bottom of the right pulmonary artery and into the common atrium.
Her pre-intervention TEE demonstrated low normal ventricular systolic function, torrential common AV valve regurgitation with 3D vena contract 3.93 cm2 (Table 1), and mean inflow gradient of 2 mmHg. Invasive hemodynamics demonstrated an elevated right atrial pressure (mean 11mmHg, v-wave 22 mmHg) and a mean Fontan pressure of 13 mmHg.
A short sheath was placed in the right internal jugular vein and a 5 Fr needle was used to puncture through the right pulmonary artery floor and into the atrium(Video 8, Video 9). The puncture site was dilated with a 5mm balloon followed by a 16 Fr sheath then the MitraClip guide catheter was advanced into the atrium. The clip delivery system was introduced into the atrium over a stiff guidewire. A MitraClip XTW was positioned perpendicular to the line of coaptation between the superior and inferior bridging leaflets over the right-sided AV valve and deployed (Figure 8). There was modest improvement in the degree of regurgitation (3D vena contracta area 3.12 cm2) without a change in inflow gradient. A second MitraClip XTW was oriented between the superior and inferior bridging leaflets leftward of the first clip, over the ventricular septum, and deployed (Video 8). The total 3D vena contracta decreased to 1.57 cm2 through three distinct orifices, mean inflow gradient was remained low at 3mmHg, ventricular systolic function remained low normal and RA pressure was largely unchanged at mean 12 mmHg (v-wave 22 mmHg). The guide catheter was removed and a 6mm Amplatzer Septal Occluder was used to close the puncture site in the right pulmonary artery (Video 9).
The procedure was well-tolerated with no noted ectopy or other complications prior to discharge the following day. Three weeks post-procedure, she reported improved energy level with NYHA functional class II, less dyspnea and less abdominal distention.
DISCUSSION
There is increasing evidence supporting TEER as an atrioventricular valve therapy in high-risk adult patients, and there are further trials investigating expanding use in moderate-risk patients.(1,21–24) While there have been case reports and a series of applications of TEER in CHD, (8–12,25) to date there has not been an organized effort to expand the use of TEER technologies to the heterogenous, anatomically complex CHD population due to the inherent challenges associated with widely varying anatomies, mechanisms of valve dysfunction, and delivery systems designed for “structurally normal” hearts.
Application of TEER may be particularly important in populations of CHD at high or moderate risk of complication from traditional surgical valve repair like those in this report. Patients with heart failure, those who are not currently candidates for transplant, and those who are awaiting transplant may all benefit from reduction in AVVR, without associated insult of cardiopulmonary bypass.(26) For example, left AVVR after complete atrioventricular canal repair is prevalent, difficult to repair, and typically found in populations with significant comorbidities.(7,27,28) Further, patients with single ventricle physiology, are at extremely high risk of atrioventricular valve dysfunction, with subsequent failure of Fontan physiology.(6,29) AVVR typically presents in childhood with nearly half of single right ventricle patients and two-thirds of single ventricle patients with common atrioventricular canal valves progressing to frank valve failure by their mid-twenties. It is clear that early reduction or elimination of AVVR is physiologically beneficial in these patients, however, current surgical repair strategies are often unsuccessful, and associated with at least transient ventricular dysfunction.(6,29,30) In addition, atrioventricular valve replacement is highly associated with need for postoperative extracorporeal membrane oxygenation support, death or transplant.(30,31) Given these limitations, TEER, which conceptually represents a lower risk therapy given the avoidance of sternotomy and cardiopulmonary bypass, may be uniquely beneficial in patients with failing single ventricular physiology.
The potential of TEER technologies in these types of CHD patients is intriguing, and yet to be fully realized. In order to maximize the potential benefit in this challenging and heterogeneous groups of patients, we assert that TEER therapy could be offered in childhood or adolescence, before symptomatic heart failure develops. Notably most of these young patients are cared for in pediatric hospitals where TEER therapy is not currently available. To address the challenge of introducing an “off the shelf” technology intended for structurally normal adult patients into a younger, diverse CHD population, an organized, patient-specific approach to screening and procedural planning is required.
In this setting we present the initial experience with preoperative modeling and application of TEER within a free-standing pediatric hospital. The imaging, modeling, and simulation-based approach that we developed was successfully applied to 4 patients younger than 21 years of age. Subjectively, this approach (13,14) enhanced procedural planning, allowed for “procedural rehearsal”; including testing compatibility of the Mitra Clip delivery system in the specific anatomies, and facilitated effective peri-procedural communication between the interventional cardiologist and the imaging cardiologist. This resulted in a relatively rapid and safe execution of TEER in all 4 patients despite heterogeneous anatomy, and the relative inexperience of the team involved. In our series both HLHS Fontan patients had significant reductions in AVVR and durable improvement in symptoms after TEER. The patient with CAVC had significant reduction in left AVVR after TEER which resulted in the rapid de-escalation in care including the weaning of inotropic support and eventual discharge to home. In all cases, systemic ventricular function decreased acutely, and two patients developed transient arrythmia. However, with time ventricular function recovered or improved from pre-intervention(Supplemental Table 1-3).
Limitations
This report details the mid-term outcomes of four patients with complex atrioventricular valve failure associated with congenital heart disease, who underwent TEER. Further application and reporting of outcomes are needed to determine long term risks and benefits of TEER in the diverse population of patients with CHD.
CONCLUSIONS
The early results of our protocolized efforts to introduce TEER of severe AVVR with MitraClip into the CHD population within our institution are encouraging. Further investigations of the use of TEER in this challenging population are warranted.
Supplementary Material
Video 3: Intraprocedural Fluoroscopic Imaging of TEER in Hypoplastic Left Heart Syndrome (Case 1)
Video 5: Intraprocedural Fluoroscopic Imaging of TEER in Hypoplastic Left Heart Syndrome (Case 2)
Video 7: Intraprocedural Fluoroscopic Imaging of TEER in Complete Atrioventricular Canal Left Atrioventricular Valve (Case 3)
Video 1: Overview of Virtual and Physical Image-Derived Modeling Pre-Intervention Workflow
Video 2: Intraprocedural Echo Imaging of TEER in Hypoplastic Left Heart Syndrome (Case 1)
Video 4: Intraprocedural Echo Imaging of TEER in Hypoplastic Left Heart Syndrome (Case 2)
Video 6: Intraprocedural Echo Imaging of TEER in Complete Atrioventricular Canal Left Atrioventricular Valve (Case 3)
Video 8: Intraprocedural Echo Imaging of TEER in Unbalanced Complete Atrioventricular Canal Valve (Case 4)
Video 9: Intraprocedural Fluoroscopic Imaging of TEER in Unbalanced Complete Atrioventricular Canal Valve (Case 4)
Figure 1. Overview of Preoperative Modeling.
Cardiac Magnetic resonance images and 3D echocardiographic images were registered to create a combined model. These models were then used for virtual simulation using SlicerHeart, and physical simulation with the actual MitraClip Delivery System.
CLINICAL PERSPECTIVE.
What is Known?
Atrioventricular valve regurgitation (AVVR) is a devastating complication in children and young adults with congenital heart disease (CHD), particularly in patients with single ventricle physiology. Transcatheter edge-to-edge repair (TEER) is a rapidly expanding, minimally invasive option for the treatment of AVVR that avoids the morbidity and mortality associated with open heart surgery. AVVR in CHD patients is a complex problem. An organized approach is required if the potential of TEER therapy is ever to be realized in this heterogenous population.
What is New?
We demonstrate an image-derived modeling protocol and clinically successful application of TEER in 4 patients with CHD including three patients with palliated single ventricle physiology and a Fontan circulation.
What is Next?
The protocolized approach to TEER in CHD described in this report may serve as a foundation for the safe initiation of TEER therapy at other free-standing pediatric hospitals. Further collaborative investigations of the use of TEER in this challenging population are warranted. Ideally, a multicenter study will ensue.
Funding:
This work was supported NIH R01HL153166, an Additional Ventures Expansion Award, Additional Ventures Single Ventricle Research Fund, The Topolewski Pediatric Valve Center at CHOP, and the Topolewski Endowed Chair at the Children’s Hospital of Philadelphia.
Abbreviations:
- 2D
Two Dimensional
- 2DE
Two-Dimensional Echocardiography
- 3D
Three Dimensional
- 3DE
Three-Dimensional Echocardiography
- AVVR
Atrioventricular valve regurgitation
- CHD
Congenital Heart Disease
- CMR
Cardiac Magnetic Resonance
- DICOM
Digital Imaging and Communications in Medicine
- Echo
Echocardiography
- HLHS
Hypoplastic Left Heart Syndrome
- RV
Right Ventricle
- TEER
Transcatheter Edge-to-Edge Repair
- TR
Tricuspid Regurgitation
- TV
Tricuspid Valve
Footnotes
Disclosures: Matthew Gillespie is a consultant for Abbott. Jonathan Edelson is a consultant for Abbott and Abiomed. The other authors have no disclosures.
REFERENCES
- 1.Stone GW, Lindenfeld J, Abraham WT et al. Transcatheter Mitral-Valve Repair in Patients with Heart Failure. N Engl J Med 2018;379:2307–2318. [DOI] [PubMed] [Google Scholar]
- 2.Nickenig G, Kowalski M, Hausleiter J et al. Transcatheter Treatment of Severe Tricuspid Regurgitation With the Edge-to-Edge MitraClip Technique. Circulation 2017;135:1802–1814. [DOI] [PubMed] [Google Scholar]
- 3.Orban M, Braun D, Deseive S et al. Transcatheter Edge-to-Edge Repair for Tricuspid Regurgitation Is Associated With Right Ventricular Reverse Remodeling in Patients With Right-Sided Heart Failure. JACC Cardiovasc Imaging 2019;12:559–560. [DOI] [PubMed] [Google Scholar]
- 4.Orban M, Lusebrink E, Braun D et al. Recent advances in patient selection and devices for transcatheter edge-to-edge mitral valve repair in heart failure. Expert Rev Med Devices 2020;17:93–102. [DOI] [PubMed] [Google Scholar]
- 5.Sorajja P, Whisenant B, Hamid N et al. Transcatheter Repair for Patients with Tricuspid Regurgitation. N Engl J Med 2023;388:1833–1842. [DOI] [PubMed] [Google Scholar]
- 6.King G, Ayer J, Celermajer D et al. Atrioventricular Valve Failure in Fontan Palliation. J Am Coll Cardiol 2019;73:810–822. [DOI] [PubMed] [Google Scholar]
- 7.Ho DY, Katcoff H, Griffis HM, Mercer-Rosa L, Fuller SM, Cohen MS. Left Valvar Morphology Is Associated With Late Regurgitation in Atrioventricular Canal Defect. Ann Thorac Surg 2020;110:969–978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Alshawabkeh L, Mahmud E, Reeves R. Percutaneous mitral valve repair in adults with congenital heart disease: Report of the first case-series. Catheter Cardiovasc Interv 2021;97:542–548. [DOI] [PubMed] [Google Scholar]
- 9.Blusztein D, Moore P, Qasim A, Mantri N, Mahadevan VS. Transcatheter Edge-to-Edge Repair of Systemic Tricuspid Valve in Extracardiac Fontan Circulation: First in Human. JACC Case Rep 2022;4:221–225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Guerin P, Jalal Z, Le Ruz R et al. Percutaneous Edge-to-Edge Repair for Systemic Atrioventricular Valve Regurgitation in Patients With Congenital Heart Disease: The First Descriptive Cohort. J Am Heart Assoc 2022;11:e025628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Iriart X, Guerin P, Jalal Z, Thambo JB. Edge to edge repair using a MitraClip for severe tricuspid valve regurgitation after a Mustard operation. Catheter Cardiovasc Interv 2021;98:E108–E114. [DOI] [PubMed] [Google Scholar]
- 12.Mital R, Ramakrishna S, Judson GL, Watt C, Tolstrup K, Mahadevan VS. Transcatheter Edge-to-Edge Repair for Left Atrioventricular Valve Cleft After Previously Repaired Complete Atrioventricular Canal Defect in Down Syndrome. CASE (Phila) 2023;7:35–38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ching S, Cianciulli AR, Flynn M et al. Physical Simulation of Transcatheter Edge-to-Edge Repair using Image-Derived 3D Printed Heart Models. Ann Thorac Surg Short Rep 2023;1:40–45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Cianciulli A, Lasso A, Pinter C et al. Simulation of Delivery of Clip-Based Therapies Within Multimodality Images to Facilitate Preprocedural Planning. J Am Soc Echocardiogr 2021;34:1111–1114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Gillespie MJ, Benson LN, Bergersen L et al. Patient Selection Process for the Harmony Transcatheter Pulmonary Valve Early Feasibility Study. Am J Cardiol 2017;120:1387–1392. [DOI] [PubMed] [Google Scholar]
- 16.Hahn RT, Badano LP, Bartko PE et al. Tricuspid regurgitation: recent advances in understanding pathophysiology, severity grading and outcome. Eur Heart J Cardiovasc Imaging 2022;23:913–929. [DOI] [PubMed] [Google Scholar]
- 17.Zoghbi WA, Adams D, Bonow RO et al. Recommendations for Noninvasive Evaluation of Native Valvular Regurgitation: A Report from the American Society of Echocardiography Developed in Collaboration with the Society for Cardiovascular Magnetic Resonance. J Am Soc Echocardiogr 2017;30:303–371. [DOI] [PubMed] [Google Scholar]
- 18.Prakash A, Lacro RV, Sleeper LA et al. Challenges in echocardiographic assessment of mitral regurgitation in children after repair of atrioventricular septal defect. Pediatr Cardiol 2012;33:205–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Fedorov A, Beichel R, Kalpathy-Cramer J et al. 3D Slicer as an image computing platform for the Quantitative Imaging Network. Magn Reson Imaging 2012;30:1323–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lasso A, Herz C, Nam H et al. SlicerHeart: An open-source computing platform for cardiac image analysis and modeling. Front cardiovasc med 2022;9:886549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.McCarthy PM, Whisenant B, Asgar AW et al. Percutaneous MitraClip Device or Surgical Mitral Valve Repair in Patients With Primary Mitral Regurgitation Who Are Candidates for Surgery: Design and Rationale of the REPAIR MR Trial. J Am Heart Assoc 2023;12:e027504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Baron SJ, Wang K, Arnold SV et al. Cost-Effectiveness of Transcatheter Mitral Valve Repair Versus Medical Therapy in Patients With Heart Failure and Secondary Mitral Regurgitation: Results From the COAPT Trial. Circulation 2019;140:1881–1891. [DOI] [PubMed] [Google Scholar]
- 23.Mack MJ, Lindenfeld J, Abraham WT et al. 3-Year Outcomes of Transcatheter Mitral Valve Repair in Patients With Heart Failure. J Am Coll Cardiol 2021;77:1029–1040. [DOI] [PubMed] [Google Scholar]
- 24.Song C, Madhavan MV, Lindenfeld J et al. Age-Related Outcomes After Transcatheter Mitral Valve Repair in Patients With Heart Failure: Analysis From COAPT. JACC Cardiovasc Interv 2022;15:397–407. [DOI] [PubMed] [Google Scholar]
- 25.Haeffele CL, Lui GK, Peng L, Chan F, Sharma RP. First described mitral clip in an adult extracardiac Fontan patient: a case report. Eur Heart J Case Rep 2023;7:ytac479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Lipshultz SE, Law YM, Asante-Korang A et al. Cardiomyopathy in Children: Classification and Diagnosis: A Scientific Statement From the American Heart Association. Circulation 2019;140:e9–e68. [DOI] [PubMed] [Google Scholar]
- 27.Gellis L, McGeoghegan P, Lu M et al. Left atrioventricular valve repair after primary atrioventricular canal surgery: Predictors of durability. J Thorac Cardiovasc Surg 2023;166:1168–1177. [DOI] [PubMed] [Google Scholar]
- 28.Atz AM, Hawkins JA, Lu M et al. Surgical management of complete atrioventricular septal defect: associations with surgical technique, age, and trisomy 21. J Thorac Cardiovasc Surg 2011;141:1371–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.King G, Buratto E, Celermajer DS et al. Natural and Modified History of Atrioventricular Valve Regurgitation in Patients With Fontan Circulation. J Am Coll Cardiol 2022. [DOI] [PubMed] [Google Scholar]
- 30.Stephens EH, Dearani JA, Niaz T, Arghami A, Phillips SD, Cetta F. Effect of Earlier Atrioventricular Valve Intervention on Survival After the Fontan Operation. Am J Cardiol 2020;137:103–110. [DOI] [PubMed] [Google Scholar]
- 31.Gillespie MJ, Marino BS, Cohen MS et al. Risk factors for adverse outcomes after surgery on the systemic atrioventricular valve in 109 children. Cardiol Young 2006;16 Suppl 3:35–42. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Video 3: Intraprocedural Fluoroscopic Imaging of TEER in Hypoplastic Left Heart Syndrome (Case 1)
Video 5: Intraprocedural Fluoroscopic Imaging of TEER in Hypoplastic Left Heart Syndrome (Case 2)
Video 7: Intraprocedural Fluoroscopic Imaging of TEER in Complete Atrioventricular Canal Left Atrioventricular Valve (Case 3)
Video 1: Overview of Virtual and Physical Image-Derived Modeling Pre-Intervention Workflow
Video 2: Intraprocedural Echo Imaging of TEER in Hypoplastic Left Heart Syndrome (Case 1)
Video 4: Intraprocedural Echo Imaging of TEER in Hypoplastic Left Heart Syndrome (Case 2)
Video 6: Intraprocedural Echo Imaging of TEER in Complete Atrioventricular Canal Left Atrioventricular Valve (Case 3)
Video 8: Intraprocedural Echo Imaging of TEER in Unbalanced Complete Atrioventricular Canal Valve (Case 4)
Video 9: Intraprocedural Fluoroscopic Imaging of TEER in Unbalanced Complete Atrioventricular Canal Valve (Case 4)