Abstract
A male Persian cat was presented with persistent fever, anorexia, weakness, hypopyon, nystagmus, and intention tremors. The hemogram showed severe neutropenia and laboratory analysis on cerebrospinal fluid (CSF) smears revealed abundant yeast cells compatible with Paracoccidioides brasiliensis. Urinalysis demonstrated persistent funguria and an increased urine protein-to-creatinine ratio (UPC) in addition to mild azotemia. Long-term therapy with oral fluconazole was effective in controlling the nervous system signs. Funguria was resolved with subcutaneous administration of diluted amphotericin B in a large volume of saline solution for a period of 12 weeks during the second year after initial diagnosis. Throughout 5 years of treatment, no adverse effects were observed and tolerance to the drugs was normal. Due to development of progressive uremic syndrome the animal was euthanased. To the best of our knowledge, this report is the first clinical case described of a nervous and urinary system infection caused by the P brasiliensis in a cat.
An 8-month-old male Persian cat was presented with persistent fever, anorexia, and loss of strength that had persisted for 3–4 days. The cat had lived indoors since the age of 6 weeks in an urbanized area of the city. The cat had history of mild nystagmus first observed at the age of 5 months and that coincided with the development of bilateral hypopyon, which was successfully treated with an ophthalmic solution of dexamethasone (Tobradex; Alcon). Clinical examination revealed generalized weakness and mild rotary nystagmus. The hemogram revealed severe neutropenia (0.528×109/l; reference range (RR) 2.5–12.5×109/l) (528/μl; RR 2500–12,500/μl) without detection of Mycoplasma haemophelis. Feline leukemia virus (FeLV) and feline immunodeficiency virus (FIV) serology results were negative. The cytochemical analysis of the cerebrospinal fluid (CSF) collected from the cerebellomedullary cistern (cisterna magna) was within normal RRs. The origin of the neutropenia could not be determined and there was no indication of septic foci in body cavities. Despite this, treatment with enrofloxacin (Baytril; Bayer) and penicillin (Benacillin; Troy Laboratories) was initiated. After 2 days, the animal was discharged with 20 mg/kg of oral amoxicillin plus clavulamic acid (Clavinex Duo Forte; Saval) twice daily. Four months later, the cat developed fever and anorexia in addition to the clinical signs observed earlier. Blood panels were normal but Mycoplasma haemofelis organisms were visualized in blood smears. Blood panels demonstrated levels of globulin and creatinine in serum of 48 g/l (RR 26–45 g/l) (4.8 g/dl; RR 2.6–4.5 g/dl) and 171 μmol/l (RR<140 μmol/l) (1.94 mg/dl; RR<1.6 mg/dl), respectively. Urinalysis showed moderate levels of protein with a density of 1.020 and abundant fungal elements. Culture of urine on Sabouraud dextrose agar (Sabouraud Dextrose 4%; Linsan Laboratories) was negative at 30 days. Abdominal and thoracic radiographs and ultrasonograms were normal. The echocardiogram was normal with no signs of vegetative growth on cardiac valves. Due to the funguria, 10 mg/dl of fluconazole (Diflucan; Pfizer-Parke Davis) was diluted in 100 ml of saline solution and administrated intravenously for a period of 60 min. With this dilution, the final concentration of the drug was 0.3 mg/ml. After 2 days the fever subsided and the cat was discharged with a prescription of 10 mg/kg of oral ciprofloxacin (Ciproval; Saval) and 10 mg of ketoconazole (Ketoconazole; Pasteur) q12 h. After 5 days of treatment the patient developed fever and intention tremors, marked rotary nystagmus, and moderate flaccid tetraparesis. Urinalysis demonstrated moderate funguria. Assay of antibodies against Toxoplasma gondii by enzyme-linked immunosorbent assay (ELISA) was negative. A second analysis of CSF showed total protein levels of 100 mg/dl (RR<30 mg/dl), 6–7 neutrophils/μl (RR<5 WBC/μl, 60–70% lymphocytes, 30–40% monocytes, <1% neutrophils), Pandy test ++, and abundant yeast cells. Sulfadiazine–trimethoprim (Salfen; Laboratorio Chile) was administered at 25 mg/kg. A new course of intravenous fluconazole was initiated. After 3 days, appetite increased and neurological signs decreased. Initially, 20 mg/kg of oral sulfamethoxazol+trimethoprim (Co-trimoxazole Forte; Laboratorio Chile) was prescribed in association with a compounded formulation of oral itraconazole at 10 mg/kg (Itraconazole; Cruz Verde Pharmaceutical). However, due to poor response to this treatment, itraconazole was replaced with a compounded formulation of 40 mg of oral fluconazole (Fluconazole; Cruz Verde Pharmaceutical) twice daily, receiving 13.3 mg/kg each time. During the following 3 weeks the animal showed signs of rapid recovery.
Further laboratory analyses on CSF smears stained with Giemsa and Gram stain revealed abundant thick-walled yeast cells with narrow-based budding daughter cells in the form of the spokes of a ship's steering wheel (multiple daughter cells budding from the parent cell) or ‘Mickey Mouse’ ears (two daughter cells budding from the parent cell) (Figs 1 and 2). Additionally, the presence of a small number of thin hyphae was noted. The diagnosis was infection of the nervous system by Paracoccidioides brasiliensis.
Fig 1.
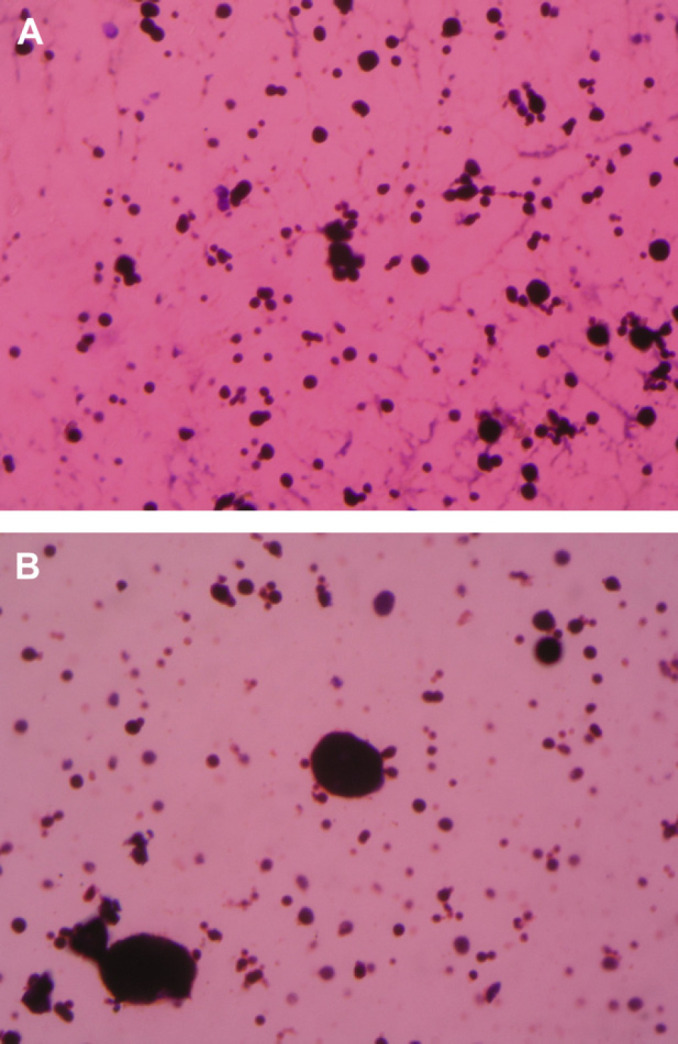
(A and B) Cerebrospinal fluid of the cat stained with Giemsa. Abundant yeast cells of different sizes and budding daughter cells in the form of the spokes of a ship's steering wheel or ‘Mickey Mouse’ ears can be observed. Note the presence of thin hyphae. 40× and 100× magnification.
Fig 2.
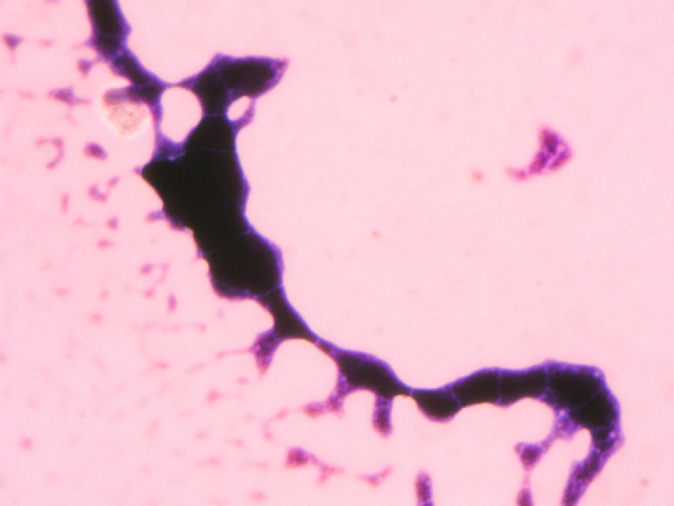
Cerebrospinal fluid of the same cat showing thick-walled yeast cells with narrow-based budding. 100× magnification.
A subcutaneous dose of 0.3 mg/kg and a subconjunctival injection of 1 mg of betamethasone (Betamethasone; Biosano) were administered due to the appearance of moderate progressive bilateral hypopyon and anterior uveitis after 6 weeks of treatment with fluconazole. Five days after application of the glucocorticoid, the hypopyon was significantly reduced but recurrence of fever and neurological signs with stationary and intention tremors, nystagmus, severe bilateral facial paralysis, and decreased oculovestibular reflexes were noted. The pupils were symmetric, mydriatic, and reactive to light. At fundic examination, the optic disc was observed small and pale with mild retinal arterial attenuation. In addition, the cat presented marked left mandibular lymphoadenopathy. The oral cavity was normal with moderate nasal discharge. The hemogram was abnormal with marked neutrophilia and shift to the left. Urinalysis showed abundant funguria. Assay for FeLV nucleic acids in blood by polymerase chain reaction (PCR) (Clinical Laboratory, University of Zurich, Switzerland) was negative. The dose of fluconazole was increased to 100 mg/day for 7 days. In the following 6 days the lymph node enlargement was significantly reduced and the neurological signs progressively improved. The facial paralysis (as assessed by palpebral reflex) was partially reversed with a Schirmer's test result of 14 mm in each eye. Intraocular pressure was 4 mmHg in each eye measured by an applanation technique. The CSF showed 3.2 neutrophils/μl (RR<5 WBC/μl, 60–70% lymphocytes, 30–40% monocytes, <1% neutrophils), 94 mg/dl of total protein (RR<30 mg/dl), Pandy's test ++, and demonstrated abundant P brasiliensis. Intravenous fluid therapy with fluconazole was administered. A bone marrow specimen obtained from the trochanteric fossa did not reveal presence of yeast or other significant findings. At discharge, fluconazole at a dose of 40 mgq12 h and oral ketoprofen (Ketofen; Merial) at 1 mg/kg/day for 3–4 days were prescribed. For control of uveitis, an ophthalmic solution of dexamethasone and atropine (Atropine; Saval) was prescribed. In the following weeks, the neurological and ocular signs disappeared with the exception of mild nystagmus.
The cat received fluconazole for 3 years without recurrence of neurological and ocular signs, anorexia, or fever. During this period, abdominal sonograms demonstrated progressive reduction of renal size, loss of corticomedullary contrast, and mixed echogenicity of the renal parenchyma with homogeneous distribution. The concentration of creatinine in serum during this period ranged between 159 and 265 μmol/l (RR<140 μmol/l) (1.8–3.0 mg/dl; RR<1.6 mg/dl). Urinalyses demonstrated a progressive decrease of funguria and complete absence of fungi in the last 3 years. To accomplish this, in addition to fluconazole, amphotericin B (Fungizon; Bristol-Myers Squibb) was administered parenterally over 12 consecutive weeks during the second year after initial diagnosis. The drug was administered subcutaneously at a dose of 0.6 mg/kg diluted in 400 ml of saline solution using standard butterfly catheters in two different application points without sedation and twice a week. During this period of treatment no adverse effects were observed and tolerance to the drugs was normal. The concentration of creatinine in serum did not increase significantly. The protein-to-creatinine ratio (UPC) remained steady above 2.0 (RR<0.4). Only corticomedullary tissue could be obtained at the ultrasound-guided renal biopsy, showing abundant degenerating nephrons and disseminated pyogranulomatous inflammation. Glomeruli were not observed and the samples were negative for the presence of fungi by the method of Grocott. In addition to fluconazole, enalapril (Enalten; Saval) at 0.25 mg/kg daily and a commercial prescription diet (Hill's Prescription Diet® k/d® Feline; Hill's Pet Nutrition) for patients with renal insufficiency were prescribed. By the end of the third year of treatment, the dose of fluconazole was reduced to 10 mg/kg/day and subsequently suspended, maintaining only the enalapril and diet. By the middle of the fifth year, the cat developed progressive azotemia with a concentration of creatinine in serum of 618 μmol/l (RR<140 μmol/l) (7 mg/dl; RR<1.6 mg/dl) and severe uremic syndrome in absence of funguria and neurological signs. For these reasons, the animal was euthanased. A confirmatory diagnosis by post-mortem examination was not possible due to lack of consent from the owner.
We described diagnosis, treatment, and clinical course of a case of P brasiliensis in a cat. This report should be considered as an example of natural infection of both the central nervous system and the urinary system in the feline host. In humans, this fungal infection is reported at higher frequencies in countries of Central and South America. 1 Most cases described in the world correspond to rural workers or tourists visiting endemic areas. 2 Cases in other species such as bats, armadillos, and dogs have also been described. 3 Transmission is likely by inhalation of conidiospores that can induce acute illness with severe pulmonary and systemic dysfunction, mainly compromising the monocyte–macrophage system.
In children and young adults, acute infection is usually manifest as ulcerative granulomas at the mucocutaneous junctions and in the oral cavity, accompanied of local adenopathy. This is known as the ‘juvenile form’. However, in most cases the infection remains latent and asymptomatic for weeks to decades. Infection can then reactivate and compromise organs such as liver, lungs, gastrointestinal tract, and central nervous system. Chronic presentation of the disease is most common in adults and older adults and it is known as the ‘adult form’. 4,5 Extremely limited resources are currently available for definitive diagnosis of this disease. These tests include, immunoelectrophoresis, ELISA, double immunodiffusion, and immunoblotting for detection of antibodies against the glycoprotein gp43, and an intradermic test. 6,7 Because these tests are not readily available in veterinary diagnostic laboratories and are not validated in the feline (or in any other species), diagnosis is based only on cytopathological assessment of visceral and encephalic lesions and body fluids. As previously described, the morphological features observed in this cat are characteristic of this agent. Similar to coccidiomycosis, blastomycosis, and histoplasmosis, the P brasiliensis is dimorphic and can reproduce through narrow-based budding. In the host, the fungus can be either found as hyphae and also as abundant double birefringent thick-walled yeast with predilection for macrophages. 8,9
The cat described in this report was likely undergoing a latent P brasiliensis infection with recurrence at the time of puberty, and in a second instance, during treatment with glucocorticoids. The presentation of the disease described in human populations of Mediterranean regions could differ from that of the tropical climates as local forms of the Paracoccidioides species can adapt to different conditions of temperature and humidity. 8 The cat primarily displayed the neurological form of the disease likely as a consequence of multiplication of yeast and formation of multiple granulomas in the rhomboid fossa (pontobulbar region) causing rotary nystagmus, tetraparesis, bilateral facial paralysis, and tremors. In humans, the neurological form occurs in up to 27% of cases and has two clinical presentations: the meningeal form, less frequent and that leads to leptomeningitis, and the granulomatous or pseudotumoral form, more common involving primarily the supratentorial regions (cerebral hemispheres) and secondarily the cerebellum and the brain stem. 10–12
In this report, early clinical signs including early nystagmus and hypopon, and subsequent funguria suggested a disseminated form of the disease. The absence of positive urine culture is in agreement with numerous reports indicating that P brasiliensis is difficult to grow in vitro, even in enriched medium.10
In addition to the permanent funguria during the initial course of fluconazole, glomerular damage reflected by the elevated UPC ratio and the progressive azotemia indicated a process of fungal infection of the kidneys. Absence of fungus in the renal biopsy could be attributed to either efficacy of treatment, the small size of the tissue sample, or to lack of cortical tissue in the sample. Additionally, the azotemia and proteinuria could be explained by natural progression of renal damage in absence of the fungus.13
Fluconazole is the treatment of choice for yeast infections of the nervous system due to a better passage through the blood-brain barrier, 14 and a fungistatic effect, 15 effectively controlling the infection by the P brasiliensis. 10 The cat in this report received parenteral and oral co-trimoxazole for a short period and this could have contributed to a partial alleviation of the clinical signs. The treatment with amphotericin B was similar to the protocol reported by Malik et al for treatment of feline encephalic cryptococcosis, 16 and could have effectively contributed to reduction and subsequent elimination of the P brasiliensis from urine. It is unlikely that the dosage and short-course of therapy, in addition to the use of large volumes of saline solution, had contributed to the observed increase in azotemia. In previously reported in cases in dogs, diluted doses of amphotericin B by slow infusion did not result in either a significant decrease in the rate of glomerular filtration or presence of tubular damage when compared to dogs that received a concentrated dose by rapid infusion. 17 Although it is possible to speculate that conidiospores may have been inadvertedly transmitted by this patient's sire and dam (both of which were originally from Central America) to the patient as a kitten, the origin of the infection in this cat could not be precisely confirmed.
Acknowledgements
The authors thank Dr David Hird for editorial assistance.
References
- 1.Pereira C., Finotti A., Gollner A., Neumann S., Quesado-Filgueiras M., Curzio M. Paracoccidioidomicose do sistema nervoso central, Arq Neuropsiquiatr 58, 2000, 741–747. [DOI] [PubMed] [Google Scholar]
- 2.Kamei K., Sano A., Kikuchi K., et al. The trend of imported mycoses in Japan, J Infect Chemother 9, 2003, 16–20. [DOI] [PubMed] [Google Scholar]
- 3.Ono M.A., Bracarense A.P., Morais H.S., Trapp S.M., Belitardo D.R., Camargo Z.P. Canine paracoccidioidomycosis: a seroepidemiologic study, Med Mycol 39, 2001, 277–282. [DOI] [PubMed] [Google Scholar]
- 4.Borges-Walmsley M.I., Chen D., Shu X., Walmsley A.R. The pathobiology of Paracoccidioides brasiliensis, Trends Microbiol 10, 2002, 80–87. [DOI] [PubMed] [Google Scholar]
- 5.Borgia G., Reynaud L., Cerini R., et al. A case of paracoccidioidomycosis: experience with long-term therapy, Infection 28, 2000, 119–120. [DOI] [PubMed] [Google Scholar]
- 6.Borba C.M., Vinhas E.A.L., Lopes-Bezerra L.M., Lucena-Silva N. Morphological, biochemical and molecular approaches for comparing typical and atypical Paracoccidioides brasiliensis strains, Antonie van Leeuwenhoek 88, 2005, 257–266. [DOI] [PubMed] [Google Scholar]
- 7.Marques S.H., Lopes A., Souza M.H., Lopes J., Queiroz-Telles F., de Camargo Z. Pires. Detection of circulating gp43 antigen in serum, cerebrospinal fluid, and bronchoalveolar lavage fluid of patients with paracoccidioidomycosis, J Clin Microbiol 41, 2003, 3675–3680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Gálvez J., Barraza G., Lara C. Systemic mycosis: paracoccidioidomycosis, Bol Hosp S J de Dios 50, 2003, 332–340. [Google Scholar]
- 9.Sivieri M., Sousa S., Correia D. Avaliação do exame citopatológico como método para diagnosticar a paracoccidioidomicose crônica oral, Rev Soc Brasil Med Trop 36, 2003, 427–430. [PubMed] [Google Scholar]
- 10.Lorenzoni P.J., Rodrigues M., Paniago A.M., Rippel P. Paracoccidioidomycosis meningitis: case report, Arq Neuropsiquiatr 60, 2002, 1015–1018. [DOI] [PubMed] [Google Scholar]
- 11.de Almeida S.M. Central nervous system paracoccidioidomycosis: an overview, Braz J Infect Dis 9, 2005, 126–133. [DOI] [PubMed] [Google Scholar]
- 12.de Almeida S.M., Queiroz-Telles F., Teive H.A., Ribeiro C.E., Werneck L.C. Central nervous system paracoccidioidomycosis: clinical features and laboratorial findings, J Infect 48, 2004, 193–198. [DOI] [PubMed] [Google Scholar]
- 13.Brown S.A. Management of chronic kidney disease. Elliot J., Grauer G.F. BSAVA manual of canine and feline nephrology and urology, 2nd edn, 2007, Gloucester, 223–230. [Google Scholar]
- 14.Wolf A. Antifungical agents. August J.R. Consultations in feline internal medicine, 1994, WB Saunders: Philadelphia, PA. [Google Scholar]
- 15.Pereira R.M., Tresoldi A.T., da Silva M.T., Bucaretchi F. Fatal disseminated paracoccidioidomycosis in a two-year-old child, Rev Inst Med Trop Sao Paulo 46, 2004, 37–39. [DOI] [PubMed] [Google Scholar]
- 16.Malik R., Jacobs J., Love D. Criptococosis: nuevas perspectivas sobre la etiología, patogenia, diagnóstico y manejo clínico. In: August JR, ed. Consultas en medicina interna felina, 2004, Ed Intermédica: Buenos Aires. Argentina. [Google Scholar]
- 17.Rubin S.I., Krawiec D.R., Gelberg H., Shanks R.D. Nephrotoxicity of amphotericin B in dogs: a comparison of two methods of administration, Can J Vet Res 53, 1989, 23–28. [PMC free article] [PubMed] [Google Scholar]


