Abstract
The objective of the study is to summarize current literature on high-flow nasal cannula (HFNC) use for different indications in pediatric patient excluding acute bronchiolitis and neonatal care. The study design is a systematic scoping review. Pubmed, Scopus, and Web of Science databases were searched in February, 2023. All abstracts and full texts were screened by two independent reviewers. Randomized controlled trials focusing on HFNC use in pediatric patients (age < 18 years) were included. Studies focusing on acute bronchiolitis and neonatal respiratory conditions were excluded. Study quality was assessed by Cochrane risk of bias 2.0 tool. The main outcomes are patient groups and indications, key outcomes, and risk of bias. After screening 1276 abstracts, we included 22 full reports. Risk of bias was low in 11 and high in 5 studies. We identified three patient groups where HFNC has been studied: first, children requiring primary respiratory support for acute respiratory failure; second, perioperative use for either intraprocedural oxygenation or postoperative respiratory support; and third, post-extubation care in pediatric intensive care for other than postoperative patients. Clinical and laboratory parameters were assessed as key outcomes. None of the studies analyzed cost-effectiveness.
Conclusion: This systematic scoping review provides an overview of current evidence for HFNC use in pediatric patients. Future studies should aim for better quality and include economic evaluation with cost-effectiveness analysis.
Protocol registration: Protocol has been published https://osf.io/a3y46/.
|
What is Known: • High flow nasal cannula has been effective in acute bronchiolitis and neonatal respiratory care. • The use of HFNC on other conditions is also common and increasing, but the evidence supporting this has not been previously summarized. | |
|
What is New: • We found that HFNC has been studies in relatively few studies in children for other indication than bronchiolitis. • We indetified three main patient populations for which HFNC has been studied: perioperative patients, postintubation patients in intensive care units, and as primary support in acute respiratory failures. None of the studies have estimated possible cost-effectiveness of HFNC, compared to alternative strategies. |
Keywords: High-flow nasal cannula, Acute bronchiolitis, Neonatal care, Pediatric patients
Introduction
High-flow nasal cannula (HFNC) therapy has rapidly gained popularity as respiratory support. HFNC therapy has been proved effective in various indications in neonatal care and acute bronchiolitis in infants [1–3]. In acute bronchiolitis the HFNC has reduced treatment failure rate compared to conventional oxygen treatment (COT), but it has had similar effectiveness as continuous positive airway pressure (CPAP) [4, 5]. In adults, previous systematic reviews have found HFNC beneficial in preventing escalation to intubation in acute hypoxemic respiratory failure, in preventing extubation failure, and in improving procedural oxygenation [6–9]. Because of the favoring evidence in these patient groups, the use of HFNC has expanded beyond neonatal respiratory support and bronchiolitis treatment in pediatrics. Simultaneously, there is ongoing effort to reduce the overuse of HFNC in acute bronchiolitis [10, 11]. There are no previous systematic summaries about HFNC use as primary respiratory support for other indications in the pediatric population. A recent systematic review found that HFNC use was associated to higher likelihood of extubation failure in young children [12]. Expanding HFNC use to new patient groups without evidence could also have negative effects such as increased costs and length of hospitalization, prolonged exposure to supplementary oxygen, and delayed escalation of respiratory support.
Previous randomized studies in children have typically compared HFNC to conventional oxygen therapy (COT), and continuous positive airway pressure (CPAP) [13]. The main hypothesis has been that HFNC would be more effective and provide benefits over COT, but be non-inferior to CPAP and better tolerated [14, 15]. Intervention tolerability is especially important in younger children.
To provide better knowledge on current evidence and to guide future studies, we aim to systematically evaluate for which indications HFNC has been studied in randomized controlled trials in pediatric patients.
Methods
Study design and search process
We conducted a systematic scoping review. We searched Pubmed, Scopus, and Web of Science databases in February, 2023. The complete search strategy is provided in Supplementary Materials. Two authors independently screened each abstract and full texts. Cases with conflicting decisions were decided either by mutual consensus or third-party opinion. All authors participated in the screening process.
We have reported our scoping review according to the Scoping review extension for Preferred Reporting Items in Systematic Reviews and Meta-analyses (PRISMA-ScR) [16].
Inclusion and exclusion criteria
We used following PICOS (patients, interventions, comparator, outcome, and study design) as our inclusion criteria. Patients had to be pediatric patients, and we defined pediatric patients as children younger than 18 years. Intervention was high-flow nasal cannula therapy. HFNC was defined by the authors in the included studies. Control intervention or comparator could either be standard low flow oxygen therapy or noninvasive continuous positive airway pressure therapy or other support mode (for example, laryngeal mask airway). We did not specify any pre-selected outcome as either inclusion or exclusion criteria. Study design had to be parallel group randomized controlled trial. Crossover, quasi-experimental, or cluster randomized trials were excluded.
We decided to exclude studies only focusing on acute bronchiolitis in infants, as the evidence regarding this indication is rather solid and covered already by several systematic reviews. Similarly, we decided to exclude all studies which focused on respiratory care of preterm infants and full-term newborns during transition to extrauterine life. However, we included studies where high-flow nasal cannula was used in postoperative care as post-extubation therapy (for example, cardiothoracic surgery for congenital cardiac defects). We excluded studies that did not present original results. Furthermore, we excluded non-English literature.
Main outcomes
Our main outcome for this scoping review was to identify the current indications for which the HFNC has been studied in randomized settings [17]. As we aimed especially to analyze the potential effectiveness of the intervention, we decided to focus on parallel group randomized controlled trials. These are typically the highest standard for evidence of effectiveness. Furthermore, we aimed to analyze the control interventions and the specific design of randomized studies (non-inferiority, superiority, etc.). Finally, we aimed to analyze the most used outcomes. We expect that main outcomes can be stratified in to three themes: clinical outcomes, laboratory parameter outcomes, and cost-effectiveness outcomes.
Critical appraisal
We assessed the risk of bias in the each of the included study by using Cochrane risk of bias 2.0 tool [18]. As the tool is designed to be outcome specific, we decided to conduct the assessment based on the intended primary outcome. Risk of bias analysis was performed by one author with prior expertise of this method (I.K.). Risk of bias figures were generated by using the Robvis shinyapp [19].
Data extraction
Data was extracted by one author and validated by a second author to reduce potential extraction errors. For this scoping review we extracted the following information: authors, journal, title, publication year, study period, country, study setting, intervention, control interventions, inclusion criteria, exclusion criteria, main outcomes, and secondary outcomes.
Permissions and ethics
Permissions for publication were not needed due to study designs. Similarly, our study did not need ethical committee evaluation.
Protocol registration
This review protocol was registered in Open Science Framework (https://osf.io/a3y46/).
Results
Search results and study characteristics
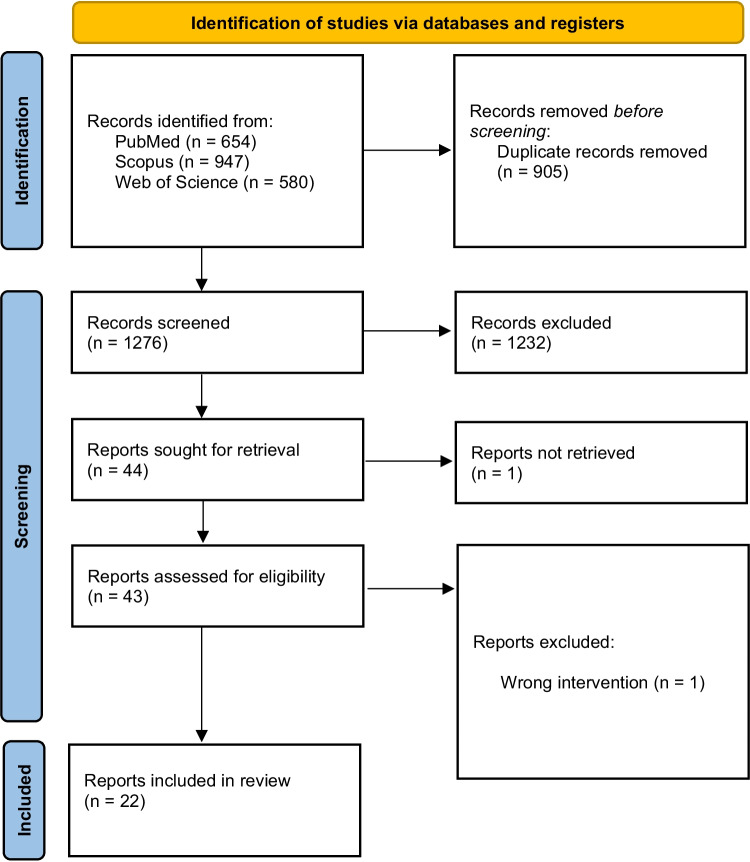
We screened 1276 abstracts and further assessed 43 full reports. Finally, 22 studies were included [20–41] (Fig. 1). Of these, 10 were conducted in Europe, 8 in Asia, 2 in Australia, 1 in South America, and 1 in Africa (Table 1). All studies were conducted in the 2010s or 2020s. Three of the included studies were single blinded and 19 were unblinded.
Fig. 1.
PRISMA flow chart of the study selection process
Table 1.
Characteristics of the included studies grouped by study setting
| Study | Country | Study period | Desing | Blinding | Setting | Patients | Intervention | Control 1 | Control 2 | Primary outcomes |
|---|---|---|---|---|---|---|---|---|---|---|
| Emergency departments or general wards | ||||||||||
| Ballestero et al. (2018) | Spain | 2012–2015 | Pilot trial | Non-blinded | ED | Moderate-to-severe asthma exacerbations | HFNC | COT | N/A | Change in asthma severity |
| Benitez et al. (2019) | Paraguay | 2017 | Superiority trial | Non-blinded | ED | Severe and moderate asthmatic crises | HFNC | COT | N/A | Change in asthma severity |
| Franklin et al. (2021) | Australia | 2016–2017 | Feasibility trial | Non-blinded | ED and GW | Acute hypoxemic respiratory failure | HFNC | COT | N/A | Proportion of children when treatment failure of the allocated oxygen therapy occurred |
| Franklin et al. (2023) | Australia and New Zealand | 2017–2020 | Superiority trial | Non-blinded | ED | Acute hypoxemic respiratory failure without bronchiolitis | HFNC | COT | N/A | Length of hospital stay |
| Maitland et al. (2021) | Kenya and Uganda | 2017–2020 | Superiority trial | Non-blinded | ED and GW | Severe WHO pneumonia with hypoxemia | HFNC | COT | Permissive hypoxemia | Mortality at 48-h post-randomization and deaths to day 28 |
| Sitthikarnkha et al. (2018) | Thailand | 2014–2015 | Superiority trial | Non-blinded | ED | Acute respiratory distress | HFNC | COT | N/A | Failure of treatment |
| Operative patients | ||||||||||
| Roncin et al. (2020) | France | 2018–2019 | Pilot trial | Single-blinded | MRI | Patients needing MRI | HFNC | COT | N/A | The ratio of the atelectasis volume to the total pulmonary volume in MRI |
| Duan et al. (2021) | China | 2018 | Superiority trial | Non-blinded | OP | Patients scheduled for percutaneous closure of a heart defect | HFNC | COT | N/A | Lowest oxygen saturation (SpO2) |
| Klotz et al. (2020) | Germany | 2016–2017 | Pilot trial | Non-blinded | OP | Gastrointestinal tract endoscopy patients | HFNC | COT | N/A | The number of events of respiratory instability defined by prespecified criteria (hypoxia, hypercapnia, apnea) |
| Kumar et al. (2022) | India | 2018–2019 | Superiority trial | Non-blinded | OP | Elective cardiac surgery for acyanotic congenital cardiac defects under cardiopulmonary bypass | HFNC | CPAP | N/A | Arterial partial pressure of CO2 (pCO2) compared with NIV |
| Lee et al. (2021) | South-Korea | 2019 | Superiority trial | Non-blinded | OP | Children receiving general anesthesia for operation for 2 h or more | HFNC | COT | N/A | Lung ultrasound score |
| Ran et al. (2022) | China | 2020–2021 | Non-inferiority trial | Single-blinded | OP | Ambulatory, oral surgery patients | HFNC | LMA | N/A | CO2 accumulation |
| Riva et al. (2018) | Switzerland | N/A | Superiority trial | Single-blinded | OP | Elective operation patients, |
HFNC 100% FiO2 |
HFNC 30% FiO2 | COT | Apnea time until one of the termination criteria was reached |
| Sharluyan et al. (2021) | Spain | 2015–2019 | Superiority trial | Non-blinded | OP | Bronchoscopy patients | HFNC | COT | N/A | The proportion of patients experiencing oxygen desaturation during the procedure |
| Pediatric intensive care units | ||||||||||
| Akyildiz et al. (2018) | Turkey | 2014–2016 | Superiority trial | Non-blinded | PICU | All cause PICU patients after extubation | HFNC | COT | N/A | The changes of respiratory, hemodynamic, and radiologic parameters |
| Chisti et al. (2015) | Bangladesh | 2011–2013 | Superiority trial | Non-blinded | PICU | Severe WHO pneumonia with hypoxemia | HFNC | CPAP | COT | Treatment failure (i.e., clinical failure, intubation and mechanical ventilation, death) |
| Enayati et al. (2021) | Iran | 2020 | Superiority trial | Non-blinded | PICU | Cardiac surgery patients | HFNC | COT | N/A | Atelectasis and PaO2/FiO2 ratio |
| Liu et al. (2020) | China | 2018–2019 | Equivalence trial | Non-blinded | PICU | Acute mild to moderate respiratory failure due to pneumonia | HFNC | CPAP | N/A | Treatment failures and intubations, duration of hospital stay and PICU stay, mortality |
| Ramnarayan et al. (2018) | UK | 2015–2016 | Pilot trial | Non-blinded | PICU | All cause PICU patients both before and after extubation | HFNC | CPAP | N/A | Feasibility outcomes for future RCT |
| Ramnarayan et al. (2022a) | UK | 2019–2020 | Non-inferiority trial | Non-blinded | PICU | All cause PICU patients after extubation | HFNC | CPAP | N/A | Time to liberate from respiratory support, mortality, reintubation rate, hospital stay duration |
| Ramnarayan et al. (2022b) | UK | 2019–2020 | Non-inferiority trial | Non-blinded | PICU | PICU patients requiring noninvasive respiratory support | HFNC | CPAP | N/A | Time to liberate from respiratory support, mortality, reintubation rate, hospital stay duration |
| Testa et al. (2014) | Italy | 2012–2013 | Superiority trial | Non-blinded | PICU | Pediatric cardiac surgical patients | HFNC | COT | N/A | Comparison of arterial PaCO2 post-extubation |
COT conventional oxygen therapy, ED emergency department, GW general ward, HFNC high-flow nasal cannula, LMA laryngeal mask airway, MRI magnetic resonance imaging, PICU pediatric intensive care unit, OP operative patients
Risk of bias
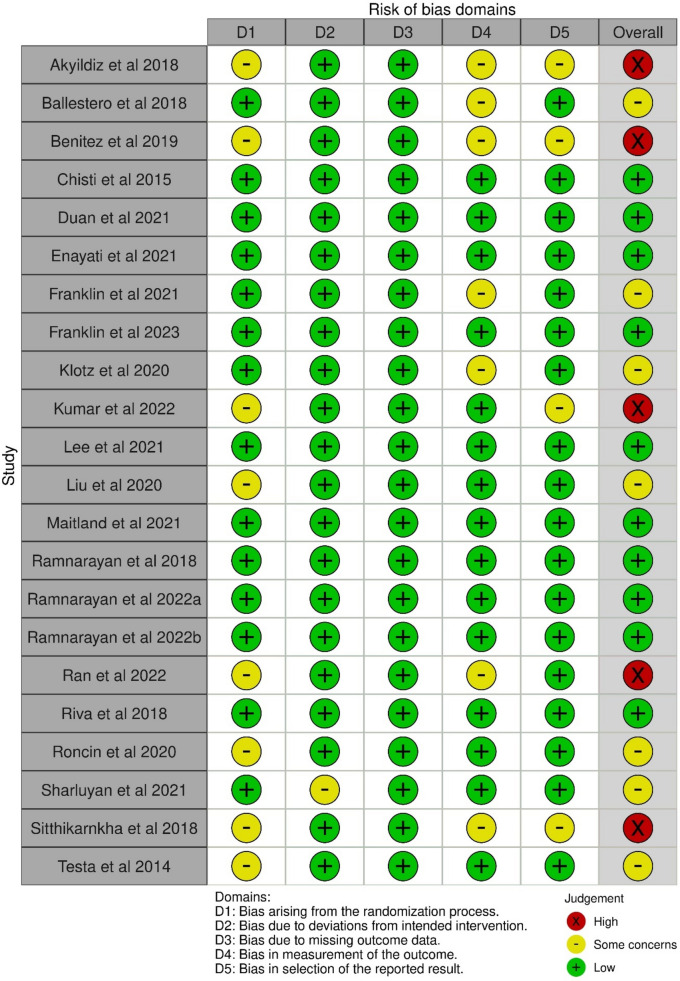
Overall risk of bias was low in 11 studies, had some concerns in 7 studies, and was high in 5 studies (Fig. 2). Most issues were due to bias in randomization process and in outcome measurement. The majority of studies were completely unblinded and caused some problems in the outcome assessment. Furthermore, some issues were seen in reporting the outcomes, as not all studies had prespecified protocol presented or referenced.
Fig. 2.
Risk of bias assessed in five domains, and overall by using Cochrane risk of bias 2.0 tool
Indications and control interventions
The indications and patient groups had high variability. Eight studies were conducted in PICU patients, five studies focused on emergency departments, and eight studies were conducted in perioperative care patients. The patients could be categorized into three main groups: first, patients requiring primary respiratory support for acute respiratory failure; second, patients in the perioperative period needing respiratory support during or after the procedure; third, PICU patients with HFNC as post-extubation respiratory support for other than perioperative use. The control intervention was conventional oxygen therapy in 15 studies, CPAP in six studies, and laryngeal mask airway in one study (Table 1).
Study design
All included studies were randomized controlled trials, of which three were single blinded. The specific designs were superiority trial (13 studies), pilot or feasibility trial (5 studies), non-inferiority trial (3 studies), and equivalence trial (1 study) (Table 1).
Most frequently reported outcomes
Most frequently reported outcomes were clinical outcomes, such as asthma severity, reintubation rate, mortality, and length of stay (PICU and overall). Laboratory outcomes were used especially in perioperative studies where the main interest was gas exchange, typically assessed by arterial pCO2, pO2, and pO2 to FiO2 ratio. Few studies assessed imaging findings, such as presence of atelectasis by lung ultrasound or magnetic resonance imaging. None of the included studies provided cost-effectiveness analysis (Table 1). Adverse effects were infrequently and incompetently reported (presence of hyperoxia or cumulative exposure to supplementary oxygen, rate of accumulation of air into intestines with effect on incidence of nausea and vomiting or on the time needed to achieve full enteral feeds).
Summary of reported results
Six studies analyzed HFNC utilization in emergency departments and general pediatric wards. Three of the studies indicated possible benefits associated with HFNC while the remaining three studies did not identify any significant difference between the HFNC and comparator interventions (Table 2). Additionally, eight studies examined the use of HFNC during procedures or in operated patients. Among these, four studies reported benefits (reduced atelectasis, improved oxygenation), and four of the studies reported no evidence of a benefit of HFNC use. Notably, none of the studies reported increased rates of adverse events (Table 2). Furthermore, eight studies analyzed HFNC use in the context of PICUs. Out these, five studies reported positive outcomes and concluded that HFNC is a feasible or non-inferior option to CPAP or superior to COT. Meanwhile, two studies did not detect differences between treatment groups, and one study found HFNC to be less effective than CPAP as post-extubation therapy (Table 2).
Table 2.
Main results and author conclusions of the included studies
| Study | Sample size | Author conclusion |
|---|---|---|
| Emergency departments or general wards | ||
| Ballestero et al. (2018) | 62 | HFNC appears to be superior to conventional oxygen therapy for reducing respiratory distress within the first 2 h of treatment in children with moderate-to-severe asthma exacerbation |
| Benitez et al. (2019) | 64 | HFNC in the treatment of asthmatic crises did not show clinical benefits nor did reduce the stay time |
| Franklin et al. (2021) | 563 | HFNC outside ICU appears to be feasible in children with acute respiratory failure and the required proportion of escalation was lower compared to standard-oxygen |
| Franklin et al. (2023) | 1567 | HFNC used as the initial primary therapy in children aged 1 to 4 years with acute hypoxemic respiratory failure did not significantly reduce the length of hospital stay compared with standard oxygen therapy |
| Maitland et al. (2021) | 1842 | Respiratory support with HFNC showing potential benefit in mortality should prompt further trials |
| Sitthikarnkha et al. (2018) | 98 | HFNC therapy revealed a potential clinical advantage in respiratory distress compared with conventional respiratory therapy. The early use of HFNC in children with moderate‐to‐severe respiratory distress may prevent endotracheal tube intubation |
| Operative patients | ||
| Roncin et al. (2020) | 39 | HFNC was associated with a lower atelectasis lung ratio compared to using a face bag-mask during anesthesia for children maintained with spontaneous ventilation |
| Duan et al. (2021) | 200 | HFNC could reduce the incidence of desaturation, the need for airway assisted ventilation and risk of carbon dioxide retention without causing hemodynamic instability or gastric distention. It is effective for pediatric patients with non-cyanotic congenital heart disease who require procedural sedation |
| Klotz et al. (2020) | 50 | HFNC did not increase respiratory stability in sedated children undergoing upper gastrointestinal tract endoscopy compared to COT |
| Kumar et al. (2022) | 127 | HFNC did not show improved CO2 washout over NIV; however, it did provide better oxygenation as measured by pO2 in arterial blood and pO2/FiO2 ratio immediate postextubation. Duration of mechanical ventilation and ICU stay were not affected by the choice of device |
| Lee et al. (2021) | 98 | Preventive use of HFNC after surgery improves the lung ultrasound score and reduces postoperative atelectasis compared to conventional oxygen therapy in infants and small children |
| Ran et al. (2022) | 120 | HFNO was not inferior to LMA for maintaining oxygenation and ventilation in patients undergoing pediatric ambulatory oral surgery under deep sedation under strict isolation from the oral cavity to the upper airway |
| Riva et al. (2018) | 60 | HFNC administered via nasal cannulas did not extend the safe apnea time for children weighing 10–20 kg compared with COT |
| Sharluyan et al. (2021) | 104 | HFNC offers optimized oxygenation during elective bronchoscopy with a significant reduction in desaturations and can be considered for oxygen administration |
| Pediatric intensive care units | ||
| Akyildiz et al. (2018) | 100 | HFNC is better than COT, especially for the restoration of the respiratory and radiologic parameters. HFNC may have more advantages to reduce the risk of extubation failure in critically ill children |
| Chisti et al. (2015) | 255 | No difference in treatment failure was noted between patients in the bubble CPAP and HFNC groups |
| Enayati et al. (2021) | 92 | HFNC could improve the respiratory parameters and reduce postoperative pulmonary complications in infants following a congenital heart surgery |
| Liu et al. (2020) | 84 | HFNC is an effective and safe initial respiratory support treatment in children < 2 years with mild to moderate respiratory failure due to pneumonia, and the incidence of intubation and death is very low; concurrently, the comfort and tolerance of HFNC are better. To some extent, HFNC is a well-tolerated alternative to CPAP |
| Ramnarayan et al. (2018) | 113 | It is feasible to conduct a large national RCT of non-invasive respiratory support in the pediatric critical care setting in both step-up and step-down patients |
| Ramnarayan et al. (2022a) | 533 | Among critically ill children requiring noninvasive respiratory support following extubation, HFNC compared with CPAP following extubation failed to meet the criterion for noninferiority for time to liberation from respiratory support |
| Ramnarayan et al. (2022b) | 573 | Among acutely ill children clinically assessed to require noninvasive respiratory support in a pediatric critical care unit, HFNC compared with CPAP met the criterion for noninferiority for time to liberation from respiratory support |
| Testa et al. (2014) | 89 | HFNC had no impact on PaCO2 values. The use of HFNC appeared to be safe and improved PaO2 in pediatric cardiac surgical patients |
Discussion
In this systematic scoping review, we found that HFNC has been studied in a variety of pediatric patients and conditions. We identified three key patient groups: acute respiratory failure, perioperative care, and PICU post-extubation respiratory support. Key outcomes assessed were clinical and laboratory outcomes. None of the studies assessed cost-effectiveness.
The most studied patient groups and indications were patients needing primary respiratory support due to acute respiratory failure, followed by perioperative care and PICU post-extubation therapy. The indications were similar for which previous studies in adults have shown benefit or equal effectiveness of HFNC treatment compared to COT or CPAP therapies [42–44].
The most frequently used control intervention was COT. All studies comparing HFNC to COT aimed at showing the superiority of HFNC treatment. The second most used control intervention was CPAP, for which either non-inferiority or equivalence designs were used. The design choices were rational, as HFNC should provide benefit over COT and be at least non-inferior to CPAP to be a justified respiratory support mode.
Main outcomes were mostly clinical or laboratory parameters. However, the lack of adverse effect reporting and the complete missing of cost-effectiveness estimations were unfortunate, as in general novel therapies should be safe and preferably cost-effective. Previous systematic review in neonatal patients concluded that there is currently no evidence of HFNC cost-effectiveness against nCPAP in preterm patients [45]. In adult patients HFNC has shown cost-effectiveness in intubation or reintubation prevention in ICU patients, and for chronic obstructive pulmonary disease patients in chronic respiratory failure [46, 47]. A recent systematic review found that HFNC and CPAP were better than COT in preventing extubation failures in infants and young children [12]. In their review CPAP seemed to be the best performing post-extubation support, although the studies were conducted in relatively heterogenous patients.
Enhanced clarity and precision in patient population definitions within future studies would significantly contribute to the interpretability of results. For instance, the inclusiveness of a wide age range (1–14 years) within the same trial investigating acute asthma exacerbations could potentially confound findings. Physiologically, the nature of acute asthma considerably varies between a 1-year-old and a teenager [29]. Moreover, PICU studies have included both all-cause patients or cardiac surgery patients. Notably, trials focused solely on cardiac surgery patients have demonstrated outcomes that hold greater applicability in clinical settings due to the more well-defined patient population. Considering the broad spectrum of patient categories within the PICU, it is evident that HFNC is not the universal solution to all cases.
We detected issues in the risk of bias assessment in the original studies. Most of the issues came from the randomization process and outcome measurement. These issues should be remarked in future trials where the researchers should focus on proper allocation concealment and randomization process and describe those in depth in the final report. Furthermore, an attempt to blind at least outcome assessors in some parts of the studies should be made to improve the reliability. A positive sign was that we did not detect issues with missing outcome data.
This is the largest effort to gather systematic assessment of current literature on HFNC use outside of neonatal respiratory care and acute bronchiolitis infants. We performed a rigorous systematic assessment according to a pre-specified protocol and we did not have any major protocol deviations. Our scoping review provides a basis for future studies and reviews on the use of HFNC.
Our main limitation is the lack of non-English literature, as most likely we have missed some RCTs published in other languages. Furthermore, only one author performed the risk of bias assessment, which can be seen as a limitation. Furthermore, we did not proceed to meta-analysis due to substantial variation in the studies and indications and instead conducted a scoping review of current knowledge and evidence.
Conclusion
In conclusion we found that HFNC has been studied in a variety of settings and indications in children. We identified three key patient groups where HFNC was studied: acute respiratory failure, perioperative care, and post-extubation respiratory support in PICU patients. Key outcomes assessed were clinical outcomes, and none of the studies assessed cost-effectiveness. Further studies should aim to better study quality and assess cost-effectiveness alongside the clinical effectiveness and treatment-related harms or adverse events.
Authors’ contributions
HS had the original idea. IK was in charge of the study design and conducted the searched. All authors participated in the screening and study selection process. IK, EW, and SNS performed data extraction and risk of bias assessments. IK wrote the initial draft. All authors commented and revised the manuscript critically. All authors have approved the final version to be submitted.
Funding
Open access funding provided by University of Eastern Finland (including Kuopio University Hospital).
Availability of data and materials
All data generated during the review process available upon request from the corresponding author.
Declarations
Ethics approval
Not applicable to scoping review.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Lin J, Zhang Y, Xiong L, Liu S, Gong C, Dai J. High-flow nasal cannula therapy for children with bronchiolitis: a systematic review and meta-analysis. Arch Dis Child. 2019;104(6):564–576. doi: 10.1136/archdischild-2018-315846. [DOI] [PubMed] [Google Scholar]
- 2.Bruet S, Butin M, Dutheil F. Systematic review of high-flow nasal cannula versus continuous positive airway pressure for primary support in preterm infants. Arch Dis Child Fetal Neonatal Ed. 2022;107(1):56–59. doi: 10.1136/archdischild-2020-321094. [DOI] [PubMed] [Google Scholar]
- 3.de Jesus BS, Tsopanoglou SP, Galvão EL, de Deus FA, de Lima VP. Can high-flow nasal cannula reduce the risk of bronchopulmonary dysplasia compared with CPAP in preterm infants? A systematic review and meta-analysis. BMC Pediatr. 2021;21(1):407. doi: 10.1186/s12887-021-02881-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Dafydd C, Saunders BJ, Kotecha SJ, Edwards MO. Efficacy and safety of high flow nasal oxygen for children with bronchiolitis: systematic review and meta-analysis. BMJ Open Respir Res. 2021;8(1):e000844. doi: 10.1136/bmjresp-2020-000844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Buendía JA, Feliciano-Alfonso JE, Laverde MF. Systematic review and meta-analysis of efficacy and safety of continuous positive airways pressure versus high flow oxygen cannula in acute bronchiolitis. BMC Pediatr. 2022;22(1):696. doi: 10.1186/s12887-022-03754-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Rochwerg B, Granton D, Wang DX, et al. High flow nasal cannula compared with conventional oxygen therapy for acute hypoxemic respiratory failure: a systematic review and meta-analysis. Intensive Care Med. 2019;45(5):563–572. doi: 10.1007/s00134-019-05590-5. [DOI] [PubMed] [Google Scholar]
- 7.Tao Y, Sun M, Miao M, et al. High flow nasal cannula for patients undergoing bronchoscopy and gastrointestinal endoscopy: a systematic review and meta-analysis. Front Surg. 2022;9:949614. doi: 10.3389/fsurg.2022.949614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Wang Q, Peng Y, Xu S, Lin L, Chen L, Lin Y. The efficacy of high-flow nasal cannula (HFNC) versus non-invasive ventilation (NIV) in patients at high risk of extubation failure: a systematic review and meta-analysis. Eur J Med Res. 2023;28(1):120. doi: 10.1186/s40001-023-01076-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Huang HW, Sun XM, Shi ZH, et al. Effect of high-flow nasal cannula oxygen therapy versus conventional oxygen therapy and noninvasive ventilation on reintubation rate in adult patients after extubation: a systematic review and meta-analysis of randomized controlled trials. J Intensive Care Med. 2018;33(11):609–623. doi: 10.1177/0885066617705118. [DOI] [PubMed] [Google Scholar]
- 10.Huang JX, Colwell B, Vadlaputi P, et al. Protocol-driven initiation and weaning of high-flow nasal cannula for patients with bronchiolitis: a quality improvement initiative. Pediatr Crit Care Med. 2023;24(2):112–122. doi: 10.1097/PCC.0000000000003136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Gupta N, Port C, Jo D, et al. Acceptability of deimplementing high-flow nasal cannula in pediatric bronchiolitis. Hosp Pediatr. 2022;12(10):899–906. doi: 10.1542/hpeds.2022-006578. [DOI] [PubMed] [Google Scholar]
- 12.Iyer NP, Rotta AT, Essouri S et al (2023) Association of extubation failure rates with high-flow nasal cannula, continuous positive airway pressure, and bilevel positive airway pressure vs conventional oxygen therapy in infants and young children: a systematic review and network meta-analysis. JAMA Pediatr. Published online June 5, 2023:e231478. 10.1001/jamapediatrics.2023.1478 [DOI] [PMC free article] [PubMed]
- 13.Zhao H, Wang H, Sun F, Lyu S, An Y. High-flow nasal cannula oxygen therapy is superior to conventional oxygen therapy but not to noninvasive mechanical ventilation on intubation rate: a systematic review and meta-analysis. Crit Care. 2017;21(1):184. doi: 10.1186/s13054-017-1760-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Mauri T, Turrini C, Eronia N, et al. Physiologic effects of high-flow nasal cannula in acute hypoxemic respiratory failure. Am J Respir Crit Care Med. 2017;195(9):1207–1215. doi: 10.1164/rccm.201605-0916OC. [DOI] [PubMed] [Google Scholar]
- 15.Vieira F, Bezerra FS, Coudroy R et al (1985) High flow nasal cannula compared to continuous positive airway pressure: a bench and physiological study. J Appl Physiol. Published online May 5, 2022. 10.1152/japplphysiol.00416.2021 [DOI] [PubMed]
- 16.Tricco AC, Lillie E, Zarin W, et al. PRISMA Extension for Scoping Reviews (PRISMA-ScR): checklist and explanation. Ann Intern Med. 2018;169(7):467–473. doi: 10.7326/M18-0850. [DOI] [PubMed] [Google Scholar]
- 17.Munn Z, Peters MDJ, Stern C, Tufanaru C, McArthur A, Aromataris E. Systematic review or scoping review? Guidance for authors when choosing between a systematic or scoping review approach. BMC Med Res Methodol. 2018;18(1):143. doi: 10.1186/s12874-018-0611-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Sterne JAC, Savović J, Page MJ, et al. RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ. 2019;366:l4898. doi: 10.1136/bmj.l4898. [DOI] [PubMed] [Google Scholar]
- 19.McGuinness LA, Higgins JPT. Risk-of-bias VISualization (robvis): An R package and Shiny web app for visualizing risk-of-bias assessments. Res Synth Methods. 2021;12(1):55–61. doi: 10.1002/jrsm.1411. [DOI] [PubMed] [Google Scholar]
- 20.Akyıldız B, Öztürk S, Ülgen-Tekerek N, Doğanay S, Görkem SB. Comparison between high-flow nasal oxygen cannula and conventional oxygen therapy after extubation in pediatric intensive care unit. Turk J Pediatr. 2018;60(2):126–133. doi: 10.24953/turkjped.2018.02.002. [DOI] [PubMed] [Google Scholar]
- 21.Ballestero Y, De Pedro J, Portillo N, Martinez-Mugica O, Arana-Arri E, Benito J. Pilot clinical trial of high-flow oxygen therapy in children with asthma in the emergency service. J Pediatr. 2018;194:204–210.e3. doi: 10.1016/j.jpeds.2017.10.075. [DOI] [PubMed] [Google Scholar]
- 22.Chisti MJ, Salam MA, Smith JH, et al. Bubble continuous positive airway pressure for children with severe pneumonia and hypoxaemia in Bangladesh: an open, randomised controlled trial. Lancet. 2015;386(9998):1057–1065. doi: 10.1016/S0140-6736(15)60249-5. [DOI] [PubMed] [Google Scholar]
- 23.Duan X, Wei N, Wei J, et al. Effect of high-flow nasal cannula oxygen therapy on pediatric patients with congenital heart disease in procedural sedation: a prospective, randomized trial. J Cardiothorac Vasc Anesth. 2021;35(10):2913–2919. doi: 10.1053/j.jvca.2021.03.031. [DOI] [PubMed] [Google Scholar]
- 24.Enayati F, Amini S, Gerdrodbari MG, Jarahi L, Ansari M (2021) Effect of high-flow nasal oxygen on respiratory parameters and pulmonary complications after early extubation following pediatric heart surgery. J Comp Pediatr 12(3). 10.5812/compreped.116104
- 25.FIRST-ABC Step-Down RCT Investigat, Paediat Critical Care Soc Study Gr, Ramnarayan P et al (2022) Effect of high-flow nasal cannula therapy vs continuous positive airway pressure following extubation on liberation from respiratory support in critically III children a randomized clinical trial. JAMA J Am Med Assoc 327(16):1555–1565. 10.1001/jama.2022.3367 [DOI] [PMC free article] [PubMed]
- 26.FIRST-ABC Step-Up RCT Investigator, Paediat Critical Care Soc Study Gr, Ramnarayan P et al (2022) Effect of high-flow nasal cannula therapy vs continuous positive airway pressure therapy on liberation from respiratory support in acutely III children admitted to pediatric critical care units a randomized clinical trial. Jama J Am Med Assoc 328(2):162–172. 10.1001/jama.2022.9615 [DOI] [PMC free article] [PubMed]
- 27.Franklin D, Babl FE, George S, et al. Effect of early high-flow nasal oxygen vs standard oxygen therapy on length of hospital stay in hospitalized children with acute hypoxemic respiratory failure: the PARIS-2 randomized clinical trial. JAMA. 2023;329(3):224–234. doi: 10.1001/jama.2022.21805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Franklin D, Shellshear D, Babl FE, et al. High flow in children with respiratory failure: a randomised controlled pilot trial – A paediatric acute respiratory intervention study. J Paediatr Child Health. 2021;57(2):273–281. doi: 10.1111/jpc.15259. [DOI] [PubMed] [Google Scholar]
- 29.Gauto Benítez R, Morilla Sanabria LP, Pavlicich V, Mesquita M. High flow nasal cannula oxygen therapy in patients with asthmatic crisis in the pediatric emergency department. Rev Chil Pediatr. 2019;90(6):642–648. doi: 10.32641/rchped.v90i6.1145. [DOI] [PubMed] [Google Scholar]
- 30.Klotz D, Seifert V, Baumgartner J, Teufel U, Fuchs H. High-flow nasal cannula vs standard respiratory care in pediatric procedural sedation: a randomized controlled pilot trial. Pediatr Pulmonol. 2020;55(10):2706–2712. doi: 10.1002/ppul.24975. [DOI] [PubMed] [Google Scholar]
- 31.Kumar A, Joshi S, Tiwari N, et al. Comparative evaluation of high-flow nasal cannula oxygenation vs nasal intermittent ventilation in postoperative paediatric patients operated for acyanotic congenital cardiac defects. Med J Armed Forces India. 2022;78(4):454–462. doi: 10.1016/j.mjafi.2021.07.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Lee JH, Ji SH, Jang YE, Kim EH, Kim JT, Kim HS. Application of a high-flow nasal cannula for prevention of postextubation atelectasis in children undergoing surgery: a randomized controlled trial. Anesth Analg. 2021;133(2):474–482. doi: 10.1213/ANE.0000000000005285. [DOI] [PubMed] [Google Scholar]
- 33.Liu C, Cheng WY, Li JS, Tang T, Tan PL, Yang L (2020) High-flow nasal cannula vs. continuous positive airway pressure therapy for the treatment of children <2 years with mild to moderate respiratory failure due to pneumonia. Front Pediat 8. 10.3389/fped.2020.590906 [DOI] [PMC free article] [PubMed]
- 34.Maitland K, Kiguli S, Olupot-Olupot P, et al. Randomised controlled trial of oxygen therapy and high-flow nasal therapy in African children with pneumonia. Intensive Care Med. 2021;47(5):566–576. doi: 10.1007/s00134-021-06385-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Ramnarayan P, Lister P, Dominguez T, et al. FIRST-line support for assistance in breathing in children (FIRST-ABC): a multicentre pilot randomised controlled trial of high-flow nasal cannula therapy versus continuous positive airway pressure in paediatric critical care. Crit Care. 2018;22(1):144. doi: 10.1186/s13054-018-2080-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Ran L, Huang G, Yao Y et al (2022) Efficacy of high-flow nasal oxygenation compared with laryngeal mask airway in children undergoing ambulatory oral surgery under deep sedation: a randomized controlled non-inferiority trial. Front Med 9. 10.3389/fmed.2022.1001213 [DOI] [PMC free article] [PubMed]
- 37.Riva T, Pedersen TH, Seiler S, et al. Transnasal humidified rapid insufflation ventilatory exchange for oxygenation of children during apnoea: a prospective randomised controlled trial. Br J Anaesth. 2018;120(3):592–599. doi: 10.1016/j.bja.2017.12.017. [DOI] [PubMed] [Google Scholar]
- 38.Roncin C, Scemama U, Zieleskiewicz L, Loundou A, Lesavre N, Vialet R. Atelectasis prevention during anaesthesia using high-flow nasal cannula therapy: a paediatric randomised trial using MRI images. Anaesth Crit Care Pain Med. 2020;39(6):819–824. doi: 10.1016/j.accpm.2020.08.009. [DOI] [PubMed] [Google Scholar]
- 39.Sharluyan A, Osona B, Frontera G, et al. High flow nasal cannula versus standard low flow nasal oxygen during flexible bronchoscopy in children: a randomized controlled trial. Pediatr Pulmonol. 2021;56(12):4001–4010. doi: 10.1002/ppul.25655. [DOI] [PubMed] [Google Scholar]
- 40.Sitthikarnkha P, Samransamruajkit R, Prapphal N, Deerojanawong J, Sritippayawan S. High-flow nasal cannula versus conventional oxygen therapy in children with respiratory distress. Indian Journal of Critical Care Medicine. 2018;22(5):321–325. doi: 10.4103/ijccm.IJCCM_181_17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Testa G, Iodice F, Ricci Z, et al. Comparative evaluation of high-flow nasal cannula and conventional oxygen therapy in paediatric cardiac surgical patients: a randomized controlled trial. Interact Cardiovasc Thorac Surg. 2014;19(3):456–461. doi: 10.1093/icvts/ivu171. [DOI] [PubMed] [Google Scholar]
- 42.Chaudhuri D, Trivedi V, Lewis K, Rochwerg B. High-flow nasal cannula compared with noninvasive positive pressure ventilation in acute hypoxic respiratory failure: a systematic review and meta-analysis. Crit Care Explor. 2023;5(4):e0892. doi: 10.1097/CCE.0000000000000892. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Khanna P, Haritha D, Das A, Sarkar S, Roy A. Utility of high-flow nasal oxygen in comparison to conventional oxygen therapy during upper gastrointestinal endoscopic procedures under sedation: a systematic review and meta-analyses. Indian J Gastroenterol. 2023;42(1):53–63. doi: 10.1007/s12664-022-01308-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Roy A, Khanna P, Chowdhury SR, Haritha D, Sarkar S. The impact of high-flow nasal cannula vs other oxygen delivery devices during bronchoscopy under sedation: a systematic review and meta-analyses. Indian J Crit Care Med. 2022;26(10):1131–1140. doi: 10.5005/jp-journals-10071-24339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Fleeman N, Mahon J, Bates V, et al. The clinical effectiveness and cost-effectiveness of heated humidified high-flow nasal cannula compared with usual care for preterm infants: systematic review and economic evaluation. Health Technol Assess. 2016;20(30):1–68. doi: 10.3310/hta20300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Sørensen SS, Storgaard LH, Weinreich UM. Cost-effectiveness of domiciliary high flow nasal cannula treatment in COPD patients with chronic respiratory failure. Clinicoecon Outcomes Res. 2021;13:553–564. doi: 10.2147/CEOR.S312523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Eaton Turner E, Jenks M. Cost-effectiveness analysis of the use of high-flow oxygen through nasal cannula in intensive care units in NHS England. Expert Rev Pharmacoecon Outcomes Res. 2018;18(3):331–337. doi: 10.1080/14737167.2018.1411804. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data generated during the review process available upon request from the corresponding author.