Abstract
Introduction
To investigate the long-term outcomes of emergency penetrating keratoplasty using cryopreserved human donor corneas in the management of actual or imminent corneal perforation.
Methods
A retrospective analysis was performed of the treatment efficacy of emergency penetrating keratoplasty using a cryopreserved human donor cornea, in 14 eyes of 14 patients with corneal ulcers of different etiology. For comparison, the medical histories of 14 patients who had undergone penetrating keratoplasty with the same indication, but received a regularly processed human corneal graft, were retrospectively analyzed. In both groups, the primary endpoint for graft failure was repeat surgery, defined as the necessity for amniotic membrane transplantation, conjunctival flap, or repeat penetrating keratoplasty, during a follow-up time of maximally 12 months.
Results
The difference in the need for repeated surgeries between the cryopreserved human donor cornea group and cultivated tissue graft group was not statistically significant (p = 0.835). Specifically, repeat complex surgery of any kind within 6 months was necessary in 50% of the cryopreserved cornea group and in 57.1% of the control group, with no further surgical interventions during the remainder of the follow-up period. However, repeat penetrating keratoplasty occurred more frequently in the cryopreserved cornea group (n = 5) than in the control group (n = 1) during the first 12 months after treatment (p = 0.048).
Conclusion
Cryopreserved corneas appear to be a viable option for promptly addressing emergencies and stabilizing the corneal situation, providing a faster solution compared to waiting for fresh tissue availability. However, repeat penetrating keratoplasty is more frequent when cryopreserved human donor corneas are used. Cryopreserved human donor corneas may be useful if surgical treatment is urgent and alternative options, such as tissue use, a conjunctival flap, or multilayer amniotic membrane transplantation, are not available.
Keywords: Penetrating keratoplasty, Corneal tissue preparation, Cryopreserved corneal graft, Corneal ulcer, Corneal perforation
Key Summary Points
| What is the purpose of this study? |
| Cryopreservation is a potential option in the context of emergency penetrating keratoplasty due to limited graft availability. |
| The aim of study was to determine whether the use of cryopreserved human corneas is effective in the emergency treatment of corneal perforations or other emergency indications compared to the use of a regularly processed human corneal graft. |
| What was learned from the study? |
| Comparable success rates in avoiding repeat surgical procedures were observed in both the cryopreservation group and the control group, emphasizing the potential benefit of cryopreserved corneas in urgent clinical situations. |
| Despite the known damage to the endothelial cells caused by cryopreservation, we found that these impairments may not have a significant impact on the long-term survival of the transplanted cornea with regard to repeat operations. |
| Patients with a cryopreserved cornea appear to undergo a notably higher frequency of subsequent penetrating keratoplasties. |
Introduction
Small perforated corneal ulcers can be treated adequately with amniotic membranes or even with only tissue adhesives [1–5], while penetrating keratoplasty may be required for large perforations or those accompanied with extensive stromal thinning [6, 7].
Corneal transplant ranks among the most frequently performed procedures globally, with approximately 180,000 surgeries conducted annually worldwide, including over 7000 in Germany [8]. However, in some regions, particularly in developing countries, the demand for donor tissue far exceeds its availability. This shortage is often attributed to factors related to the population's attitudes toward donation. Moreover, it is in these areas where the need for donor corneal tissue in cases of corneal perforations due to infectious conditions is more prevalent [3, 9–16]. In emergency situations with present or impending corneal perforation, an unavailability of fresh corneal tissue, and a risk of the eye being lost, patients may be treated with tectonic corneas that are unsuitable for penetrating keratoplasties intended for visual purposes, often with low endothelial cell counts [16]. In such cases, cryopreserved corneal tissue has occasionally been employed, yielding variable results in terms of transparency and structural preservation of the eyeball [17–21].
Although studies on penetrating keratoplasty are frequently published, there are significantly fewer publications on the use of cryopreserved corneas. Using the MeSH search terms "cryopreservation" and "corneal transplantation," we identified 183 articles on PubMed (up to 27 November 2023). However, a review of these 183 articles revealed that only 22 addressed transplantation performed on patients using cryopreserved human donor corneas. In comparison, a PubMed search using the single MeSH term "corneal transplantation" yielded 17,393 articles.
One of the first successful transplantations of a cryopreserved human donor cornea was documented by Eastcott et al. in 1954 [22]. These authors reported applying glycerol prior to the actual freezing process and then storing the corneas in a mixture of alcohol and carbon dioxide in liquid nitrogen at – 196 °C [22]. Subsequent research by O'Neil and colleagues and Capella and colleagues in the 1960s [23, 24] led independently to a number of successful transplantations that were performed using protocols that still serve as the basis for current approaches. Despite reported cryo-related damage and limited corneal endothelial regeneration capacity [25], some cryopreserved human donor grafts have achieved long-term survival [26]. To prevent cell damage during the freezing process, the cryoprotectant must fulfill the dual tasks of protecting against both intracellular ice formation and pronounced electrolyte shifts [27]. Cryoprotectants like glycerol and dimethyl sulfoxide (DMSO) permeate cells, thereby providing a certain degree of protection against freeze–thaw thermal injury [19, 24, 28–36]. Other conservation protocols include the use of Optisol GS (Bausch and Lomb, Irvine, CA, USA), which is applied for tectonic as well as therapeutic penetrating keratoplasties with or without subsequent optical keratoplasty [18, 37]. When used for cryopreservation of corneas, the endothelial cell count is presumed to be low, if present at all. Without a functioning corneal endothelium, the benefit of optical penetrating keratoplasty is questionable. However, emergency penetrating keratoplasty is first and foremost aimed at restoring the tectonic character of the cornea, eradicating any infection [38], and preserving the ocular bulb in its entirety [2].
To determine the efficacy of cryopreserved human corneas in such situations, we retrospectively compared two patient cohorts, a cryopreservation group and a control group, and analyzed the treatment results in terms of tectonic stability and complications.
Methods
The electronic (Fidus, Arztservice Wente GmbH, Darmstadt, Germany, and ORBIS, ORBIS AG, Saarbrücken, Germany) [39] and paper-based patient registries of the ophthalmology department and the cornea bank of the University Medical Center Münster were screened for the period between 2003 and 2021. Patients who had received emergency penetrating keratoplasty with an individually selected cryopreserved cornea were assigned to group A. Patients who had received a regularly processed long-term or short-term cultured cornea were included in group B. All implanted corneas had been processed according to the German Medical Association’s 2018 guidelines for the collection of donor corneas and for the management of a cornea bank [40] and in line with the medical standards of the Eye Bank Association of America [41].
The maximum shelf life of the regularly processed corneas was 34 days after cultivation. All corneas, whether regularly processed or cryopreserved later on, were stored in culture medium I (100 ml Minimum Essential Medium Eagle [MEM], 10 ml penicillin/streptomycin, 10 ml l-glutamine [200 mM], 10 ml amphotericin B [250 µg/ml], 12.5 ml HEPES buffer solution [1 M; 50×], 29.3 ml NaHCO3, and 1000 ml distilled water [Aqua Dest.], adjusted to a pH of 7.2–7.3; Biochrom AG, Berlin, Germany), at 32 °C for a maximum of 28 days. Subsequently, either preoperatively or prior to cryopreservation, the corneas were transferred into culture medium II (culture medium I plus 60 g Dextran 500 per 1000 ml). Corneas which were unsuitable for optical penetrating keratoplasty due to endothelial cell loss were cryopreserved at – 80 °C.
The minimum postsurgical observation period was set at 3 months. The outcome was defined as "successful" if no repeat surgery was performed during the follow-up period. Treatment with a cryopreserved or a regular cornea was defined as insufficient if repeat surgery became necessary, i.e., amniotic membrane grafting, conjunctival flap, repeat keratoplasty, or enucleation. Repeat suturing or anterior chamber rinsing was defined as a microsurgical intervention due to a minor complication and was analyzed separately. Graft transparency was included in the statistically analysis.
The inclusion criteria for both groups of patients included documented histories of ocular and systemic diseases, information available on the corneal disease etiology, and detailed information on the medications received (either topically or systemically). Additional criteria included the availability of transplant-related data, such as donor tissue diameter and the performing surgeon. Postoperative data covered the status of the retina and choroid, the presence of hypopyon, and whether the cornea epithelialized during follow-up. If available, information on preoperative endothelial cell density was included in the analysis.
The size of the control group was set to the same size as group A, i.e., n = 14. The patients in group B were selected to match those in group A regarding age and gender without HLA typing.
If possible, data on corrected distance visual acuity before and 6 months after penetrating keratoplasty were also collected. For a better comparison of corrected visual acuity between the two groups, the data were converted to logMAR. For patients with poor visual acuity, the ordinal scale was converted to a numerical logMAR scale with 'counting fingers' (CF) as 2.1, 'hand movement' (HM) as 2.4, 'light perception' (LP) as 2.7 and 'no light perception' (NLP) as 3.0, based on UK National Ophthalmology Database values [42, 43].
If available, photographic documentation was retrospectively analyzed to determine whether the recipients’ corneas were vascularized at baseline, the ratio of the lesion to the total cornea, and whether the cornea cleared up during the follow-up. Corneas were considered to be vascularized if peripheral vessels were present in ≥ 1 quadrants of the recipient’s corneal stroma. A mean horizontal white-to-white diameter of 11.77 mm for males and 11.64 mm for females was used to convert the ratio of lesion to total cornea into metrics [44]. A cornea was considered to be clear if the anterior surface of the iris was well visible. The location of the ulcer/perforation was classified as lower, central or upper third of the cornea. A high-risk situation was defined by the presence of the following factors: (1) preoperative corneal vascularization in any quadrant; (2) any previous keratoplasty with or without prior graft rejection; (3) active inflammation; and (4) ocular graft-versus-host disease. The presence of inflammation was determined by either documented evidence of a microbial pathogen or, in cases where a combination of antibiotics was administered perioperatively, it was assumed retrospectively. Additionally, the presence of inflammation was considered if the conjunctiva showed significant hyperemia in perioperative images.
For patients who had missed follow-up visits in our department, their office-based ophthalmologists were contacted to find out whether and when repeat surgery had been performed and to inquire about the best-corrected visual acuity at approximately 6 months after treatment.
All procedures complied with the tenets of the Declaration of Helsinki and were approved by the local institutional review board (the ethics committee of the Westphalia–Lippe Medical Association and the University of Münster). Patients were informed verbally and in writing about the surgical procedure and consented to it.
SPSS® Statistics Version 28 (SPSS IBM Corp., Armonk, NY, USA) was used for the statistical analysis. The collected data were analyzed descriptively. Statistical significance was tested using Pearson’s Chi-square test and, if appropriate, Fisher's test. Using Spearman’s method, cell number was correlated to visual acuity. The Wilcoxon rank-sum test was used to analyze the correlation of endothelial cell density with the endpoint repeat surgery. Kaplan–Meier analysis was used to reveal repeat surgery indicative of transplant survival. A difference at a p-value of < 0.05 was defined as statistically significant.
Results
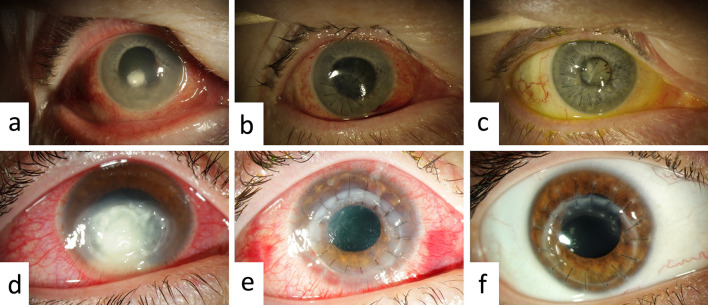
Among the 291 penetrating keratoplasties performed in our clinic between 2013 and 2020, 20 patients received a cryopreserved cornea under urgent conditions. Of these 20 patients who underwent treatment, 14 (Fig. 1a–c) fulfilled the above-mentioned inclusion criteria for inclusion in group A. Fourteen patients who had received regular corneas (Fig. 1d–f) were selected for inclusion in group B (control group).
Fig. 1.
Upper images: left eye of patient 8 (group A) at baseline (a), day 1 (b) and day 287 (c) following keratoplasty. Lower images: right eye of patient 26 (group B) at baseline (d), day 1 (e), and day 203 (f) following keratoplasty
The 14 patients in group A who fulfilled the inclusion criteria had a mean (± standard deviation [SD]) age of 66.9 ± 22.2 (range 20-86) years at baseline, and six (43%) were male and eight (57%) were female. The 14 patients in group B had a the mean age of 62.4 ± 17.6 (range 17–82) years at baseline, and seven (50%) were male and seven (50%) were female (Table 1).
Table 1.
Demographic data on all patients enrolled in the study
| Demographic data | Total study cohort | Group Aa | Group Bb |
|---|---|---|---|
| Male | 46% (n = 13) | 43% (n = 6) | 50% (n = 7) |
| Female | 54% (n = 15) | 57% (n = 8) | 50% (n = 7) |
| Age in years (mean ± SD) | 63.0 ± 20.5 | 66.9 ± 22.2 | 62.4 ± 17.6 |
SD, Standard deviation
aGroup A comprised patients who had received emergency penetrating keratoplasty with an individually selected cryopreserved cornea
bGroup B comprised patients who had obtained a regularly processed long-term or short-term culture cornea
Across both groups, the most common baseline diagnosis was graft-versus-host disease (6 cases: 14% of group A and 29% of group B), followed by neurotrophic ulcer (4 cases; 7% of group A and 21% of group B) (Table 2).
Table 2.
Etiology of corneal ulcer or perforation requiring treatment
| Etiology | Total study cohort (n = 28 eyes/patients) | Group A (n = 14 eyes/patients) | Group B (n = 14 eyes/patients) |
|---|---|---|---|
| Ocular graft-versus-host disease | 21% (n = 6) | 14% (n = 2) | 29% (n = 4) |
| Sjögren’s syndrome | 7% (n = 2) | 14% (n = 2) | 0 |
| Neurotrophic ulcer | 14% (n = 4) | 7% (n = 1) | 21% (n = 3) |
| Bacterial etiology | 10% (n = 3) | 7% (n = 1) | 14% (n = 2) |
| Candida albicans | 7% (n = 2) | 14% (n = 2) | 0 |
| Aspergillus fumigatus | 3% (n = 1) | 0 | 7% (n = 1) |
| Herpetic etiology | 7% (n = 2) | 7% (n = 1) | 7% (n = 1) |
| Lattice corneal dystrophy | 4% (n = 1) | 7% (n = 1) | 0 |
| Trauma | 11% (n = 3) | 14% (n = 2) | 7% (n = 1) |
| State after pterygium excision | 4% (n = 1) | 7% (n = 1) | 0 |
| Corneal ulcer with perforation or severe thinning of unclear etiology | 11% (n = 3) | 7% (n = 1) | 14% (n = 2) |
Ten of the 14 patients in group A had a corneal perforation at baseline, compared with six patients in group B. Twelve of the keratoplasties in each group were classified as high-risk procedures.
The average duration of follow-up was 296.5 days in group A and 365 days in group B.
Visual acuity was measured pre- and postoperatively in all patients of group B, but in group A, one patient did not undergo a sight test preoperatively and four were not tested at month 6 postoperatively.
For all patients, penetrating keratoplasty had been performed under general anesthesia by a total of seven different surgeons. Following keratoplasty, all patients received topical antibiotics and 89% were given local or systemic steroids, depending on the individual situations, e.g., antiviral, antifungal, specific topical, or systemic. The three patients in group A who did not receive topical or systemic steroids did not need repeat surgery. Systemic immunosuppressants were administered in 50% of patients (n = 7) from group A and 57% (n = 8) from group B.
Although not medically necessary, one patient (No. 11, group A) opted for enucleation 2 months post-penetrating keratoplasty to proactively alleviate the risk of future hospitalization due to persistent pain associated with phthisis bulbi. The affected eye had already lost vision. Despite the subsequent enucleation, this case was initially deemed successful (see Table 3).
Table 3.
Demographic and medical data on patients/eyes in group A
| Patient number | Age (years)/sex/eye | Cause of ulcer/perforation | Donor trephination size (mm) | Location/ corneal perforation | Procedurea | Success/failure | Follow-up time (months) | Ophthalmologically relevant secondary disease(s)b |
|---|---|---|---|---|---|---|---|---|
| 1 | 80/male/left | Graft rejection | 8.0 | Central/Yes | Re-PK | Failure | 12 | Diabetes mellitus |
| 2 | 85/female/left | Graft rejection | 8.5 | Central/Yes | Re-PK | Success | 3 | |
| 3 | 57/female/left | Sjögren's syndrome | 7.5 | Central/Yes | PK | Failure | 12 | KCS |
| 4 | 72/male/right | Sjögren's syndrome | 4.0 | Upper third/Yes | Re-PK + AMT + CF + T | Failure | 12 | Diabetes mellitus, KCS, RA |
| 5 | 20/female/right | Neurotrophic | 5.0 | Central/No | PK + AMT + T | Success | 8 | Lagophthalmos, KCS |
| 6 | 86/female/right | Bacterial | 4.0 | Central/No | PK | Failure | 12 | |
| 7 | 23/female/right | Mycotic | 7.5 | Central/Yes | PK | Failure | 8 | |
| 8 | 76/female/right | Mycotic | 5.0 | Central/No | PK | Success | 12 | Additional herpetic keratitis |
| 9 | 82/female/left | Herpetic | 4.0 | Upper third/yes | PK | Failure | 6 | RA |
| 10 | 73/male/right | Lattice endothelial dystrophy | 3.0 | Lower third/No | PK + AMT | Success | 12 | Glaucoma, diabetes mellitus, ED |
| 11 | 65/male/right | Trauma | 5.0 | Central/Yes | Re-PK + AV | Success | 2 → Enucleation | Lagophthalmos, HC |
| 12 | 82/male/right | Trauma | 4.0 | Lower third/Yes | PK | Failure | 3.5 | RA |
| 13 | 49/male/left | Status after pterygium removal | 4.0 | Central/Yes | PK | Success | 12 | |
| 14 | 86/female/right | Unknown | 3.5 | Central/Yes | PK | Success | 12 |
aAMT, amniotic membrane transplantation; AV, anterior vitrectomy; CF, conjunctival flap; PK, penetrating keratoplasty; Re-PK, Repeat penetrating keratoplasty; T, tarsorrhaphy
bED, Endothelial dystrophy; HC, Huntington's chorea; KCS, keratoconjunctivitis sicca; RA, rheumatoid arthritis
Five patients of group A needed repeat keratoplasty, due to new corneal ulcers in four cases and graft rejection in one case. In group B, one patient underwent repeat keratoplasty because of a new corneal ulcer (Table 4), and one patient received Descemet’s membrane endothelial keratoplasty due to thinning and resulting opacification.
Table 4.
Demographic and medical data on patients/eyes in group B
| Patient number | Age (years)/sex/eye | Cause of ulcer/perforation | Donor trephination size (mm) | Location/corneal perforation | Procedurea | Success/failure | Follow-up Time (mo) | Ophthalmologically relevant secondary disease(s)b |
|---|---|---|---|---|---|---|---|---|
| 1 | 57/Female/Left | Graft rejection | 8.0 | Lower third/Yes | PK | Success | 12 | KCS |
| 2 | 43/Male/Left | Graft rejection | 8.0 | Central/No | Re-PK | Success | 12 | |
| 3 | 68/Male/Left | Graft rejection | 7.5 | Central/Yes | Re-PK | Failure | 12 | Glaucoma, keratoconus, diabetes mellitus |
| 4 | 82/Female/Left | Graft rejection | 12.0 | Central/No | Re-PK | Failure | 12 | |
| 5 | 62/Female/Left | Neurotrophic | 8.0 | Central/No | PK | Failure | 12 | Glaucoma |
| 6 | 63/Male/Left | Neurotrophic | 7.5 | Lower third/Yes | PK | Success | 12 | |
| 7 | 75/Female/Left | Neurotrophic | 5.0 | Central/Yes | PK | Failure | 12 | |
| 8 | 78/Male/Left | Bacterial | 9.0 | Central/No | PK | Failure | 12 | |
| 9 | 78/Female/Right | Bacterial | 8.0 | Central/No | PK + IOL | Failure | 12 | Glaucoma |
| 10 | 61/Male/Right | Mycotic | 8.5 | Central/No | PK + IOL + AV | Success | 12 | |
| 11 | 29/Female/Left | Herpetic | 7.9 | Central/No | PK | Success | 12 | |
| 12 | 17/Male/Right | Trauma | 8.5 | Central/Yes | PK | Success | 12 | |
| 13 | 53/Female/Right | Unknown | 8.5 | Lower third/Yes | PK | Failure | 12 | |
| 14 | 61/Male/Left | Unknown | 8.0 | Central/No | PK | Failure | 12 |
aAV, anterior vitrectomy; IOL, intraocular lens extraction; PK, penetrating keratoplasty; Re-PK, repeat penetrating keratoplasty
bKCS, keratoconjunctivitis sicca
In group B, proportionally more grafts became clear than in group A (Table 5). The retina was attached in all cases in group A; in comparison, in group B there was only a slight decrease in the rate of attachment. Postoperative hypopyon was rare and was observed in only one case in each group (Table 5).
Table 5.
Descriptive comparison between groups in terms of postoperative clinical course parameters
| Factors that showed up during follow-up | Group A (n = 14 eyes/patients) | Group B (n = 14 eyes/patients) | ||||
|---|---|---|---|---|---|---|
| Yes (n) | No (n) | Missing data (n) | Yes (n) | No (n) | Missing data (n) | |
| Graft cleared up | 6 | 7 | 1 | 9 | 5 | 0 |
| Graft epithelialized | 5 | 3 | 6 | 7 | 5 | 2 |
| Retina was attached | 14 | 0 | 0 | 12 | 1 | 1 |
| Choroidea was attached | 12 | 2 | 0 | 9 | 3 | 0 |
| Postoperative hypopyon | 1 | 13 | 0 | 1 | 12 | 1 |
Corneal transplants deteriorated following a mean of 183.1 days in treated eyes of group A and 139.0 days in treated eyes of group B. There was one patient in group A for whom deterioration was recorded as 1539.0 days (Table 6).
Table 6.
Comparison between groups in terms of mean chronicity and mean ratio of the lesion to the total cornea
| Mean chronicity and mean ratio of the lesion to the total cornea | Group A | Group B |
|---|---|---|
| Duration of significant pathology in the affected eye before transplantation | Mean = 183.1 days | Mean = 139.0 days |
| Minimum = 1 day | Minimum = 1 day | |
| Maximum = 1539.0 days | Maximum = 534.0 days | |
| Ratio of the lesion to the total cornea | Mean = 0.47 | Mean = 0.43 |
| Minimum = 0.18 | Minimum = 0.14 | |
| Maximum = 1.0 | Maximum = 0.65 |
Factor Analyses
The mean (± SD) endothelial cell density of the corneas used in group A before cryopreservation was 1775.4 ± 248.8 cells/mm2 (minimum 1430 cells/mm2, maximum 2668 cells/mm2). The mean preoperative endothelial cell density of the corneas used in group B was 2188.5 ± 605.5 cells/mm2 (minimum 1480 cells/mm2, maximum 3934 cells/mm2). In neither group were the differences in preoperative cell densities significantly correlated with the occurrence of repeat surgery (Wilcoxon rank-sum test, p = 0.18 in group A, p = 0.88 in group B).
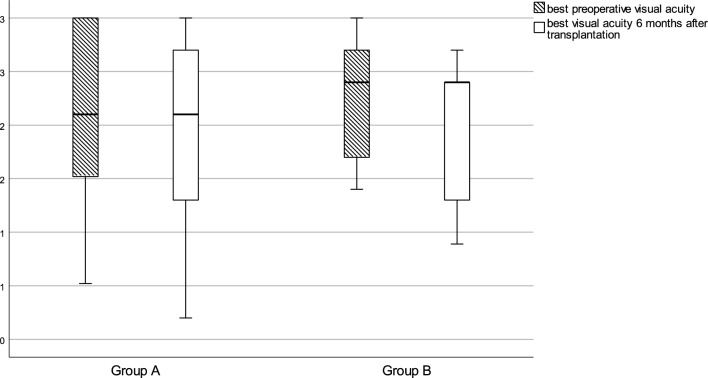
The influence of endothelial cell density on visual acuity at postoperative month 6 was not significant in either group A (Spearman’s rank correlation [rs] = 0.01, p = 0.97) or group B (Spearman’s rs = 0.33, p = 0.28) (Fig. 2). At postoperative month 12, the data were too sparse (n = 7 in group A, n = 10 in group B) for evaluation of visual acuity.
Fig. 2.
Boxplot of the best corrected visual acuity (in LogMAR) before and 6 months after keratoplasty in group A and group B. Finger counting (CF) was defined as 2.1, hand movement (HM) as 2.4, light perception (LP) as 2.7, and no light perception (NLP) as 3.0
Preoperative corneal vascularization was significantly correlated with repeat surgery in group A (Pearson’s Chi-square test, p = 0.021) but not in group B (Pearson’s Chi-square test, p = 1.0).
The results of a direct comparison of groups A and B using Pearson’s Chi-square test are provided in Table 7. The comparison was aimed at determining whether the two groups differed with regard to the following variables: preoperative corneal vascularization, postoperative corneal vascularization, donor transplant size ≥ 8 mm, preoperative synechiae, and previous penetrating keratoplasty. No statistically significant difference was found between groups A and B for any of the variables (Table 7).
Table 7.
P-values from Pearson’s Chi-square test for the comparison of groups A and B
| Parameter (primary target parameter: “repeat surgery”) | Positive test variable (yes) | Negative test variable (no) |
|---|---|---|
| Preoperative corneal vascularization | 0.335 | 0.429 |
| Postoperative corneal vascularization | 1.0 | 0.633 |
| Donor transplant size ≥ 8mm | 1.0 | 1.0 |
| Preoperative synechiae | 0.619 | 1.0 |
| Previous penetrating keratoplasty | 1.0 | 1.0 |
Seven patients (50%) from the cryopreserved group and six patients (43%) from the control group had no repeat surgery and thus experienced graft survival (Table 8). The type of repeat surgery differed according to need: there was one case of repeat keratoplasty in the control group, and five cases of repeat keratoplasty in the cryopreservation group. In contrast, amniotic membrane transplantation was performed more often in the control group (Table 8).
Table 8.
Patients who received at least one repeat microsurgery in both groups after 12 months
| Type of repeat surgery | Total study cohort (n = 28 eyes/patients) | Group A (n = 14 eyes/patients) | Group B (n = 14 eyes/patients) |
|---|---|---|---|
| Amniotic membrane transplantation | 10 (33%) | 3 (21%) | 7 (50%) |
| Conjunctival flap | 2 (11%) | 1 (7%) | 1 (7%) |
| Repeat penetrating keratoplasty | 6 (14%) | 5 (36%) | 1 (7%) |
| No repeat surgery | 13 (42%) | 7 (50%) | 6 (43%) |
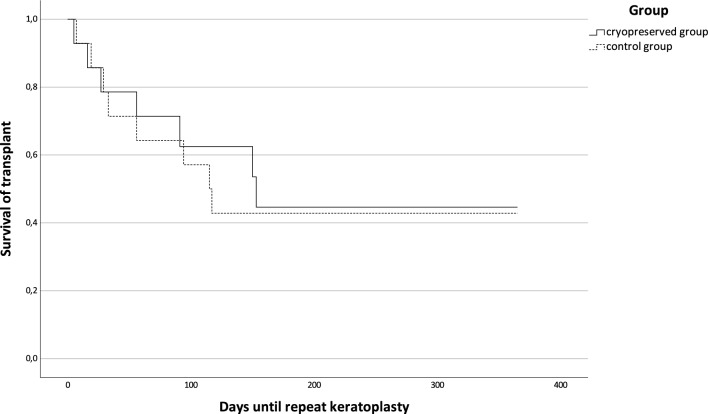
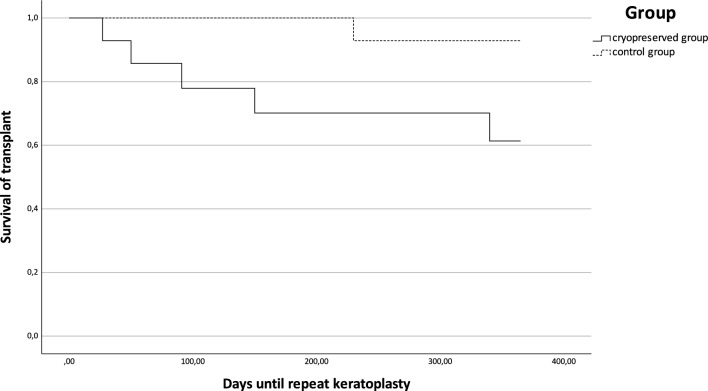
Kaplan–Meier analysis showed no significant difference between groups A and B in relation to transplant survival, i.e., repeat surgery (Kaplan–Meier analysis, p = 0.835) (Fig. 3), but did reveal a significant difference for repeat keratoplasty (Kaplan–Meier analysis, p = 0.048) (Fig. 4).
Fig. 3.
Kaplan–Meier analysis of days of transplant survival, defined as days without repeat surgery, for groups A and B (log-rank test, p = 0.835)
Fig. 4.
Kaplan–Meier analysis of days of transplant survival, defined as days without repeat keratoplasty, for groups A and B (log-rank test, p = 0.048)
The numbers of microsurgical interventions, also including multiple procedures in the same patient, were similar in groups A and B (see Table 9). On average, 0.79 minor procedures were required per patient in group A and 0.71 in group B. The incidence of patients who underwent at least one microsurgical intervention was lower in group A than in group B (28.6% vs. 50%) (Table 9).
Table 9.
Incidence of patients who had suffered at least one minor complication during the 12-month follow-up, with donor trephination size
| Repeat microsurgical intervention | Donor trephination size | ||
|---|---|---|---|
| Yes/No | Total n eyes/patients | Group A | Group B |
| Yes | 11 | n = 4 (28.6%) | n = 7 (50%) |
| Mean = 6.4 mm | Mean = 8.1mm | ||
| Median = 6.7 mm | Median = 8.0 mm | ||
| Minimum = 4.0 mm | Minimum = 7.5 mm | ||
| Maximum = 8.0 mm | Maximum = 9.0 mm | ||
| No | 10 | n = 10 (71.4%) | n = 7 (50%) |
| Mean = 5.3 mm | Mean = 8.3 mm | ||
| Median = 5.0 mm | Median = 8.0 mm | ||
| Minimum = 4.0 mm | Minimum = 5.0 mm | ||
| Maximum = 8.5 mm | Maximum = 12.0 mm | ||
Minor complications were, for example, repeat suturing or anterior chamber irrigation
Discussion
The survival rates of penetrating keratoplasties in high-risk patients vary between 30% and 80% over a period of 12 months after treatment [45–48], depending on the specific risk factors and etiologies [49, 50]. Penetrating keratoplasty in patients with keratoconus generally has a good survival rate [51], but this was not observed in group A nor in group B in the present study. Therefore, both of the patient cohorts described here would appear to have a relatively high risk of transplant failure.
Various cryoprotectants can be used for the cryopreservation of human donor corneas. Using the cryoprotectant DMSO is an elaborate process, but it offers a certain degree of protection against freeze–thaw injury [19, 24, 28, 31, 32, 52], leading to results for corneal transparency of up to 75.1%, which is comparable with results obtains with fresh tissue (81.4%) [52]. In addition to a partial preservation of the endothelium (approximately 35% after 1 year) [52], a low number of high-risk patients may have contributed to the treatment successes reported in this study. Cryopreservation with dextran as a cryoprotectant has been shown to have the capability of regaining structural integrity after thawing but also variability in terms of endothelial cell loss [53]. Cryopreservation with glycerol results in the lack of a viable endothelium. In the context of emergency corneal transplantation, Gupta et al. demonstrated comparable therapeutic success rates for cryopreserved and fresh corneal tissue [36]. Approaches to overcoming the problem of endothelial cell damage in cryopreservation with glycerol include deep anterior lamellar keratoplasty (DALK) [34, 35] or treatment with small-diameter grafts [33], depending on the etiology and extent of corneal damage.
Similar to freezing with glycerol or a balanced saline solution [54, 55], the culture medium used in our study has limited or no cryoprotective qualities. Therefore, we hypothesize that cell density of the transplants was low, if cells were still present at all, in group A. Nevertheless, these transplants did not fail significantly more often than the regularly processed corneas of group B in terms of overall repeat surgery.
Similar to our cryopreservation technique, Kim et al. and Lim et al. did not use a cryoprotectant, but stored the corneas in a culture medium (Optisol GS in both studies) at – 80 °C [18, 37]. Kim et al. examined therapeutic and tectonic keratoplasties over a total period of 11 years [18], but DALK was performed in the majority of the cases included; the average graft size of 3.7 mm was also rather small compared to the size of the transplants used in our study. Anatomical integrity could be maintained in 28 of 32 cases of tectonic keratoplasty, but these results can only be compared to ours to a limited extent, as different endpoints (enucleation vs. repeat surgery) were defined.
One might hypothesize that the cryopreserved corneas used in our study induced weaker or fewer graft rejections and less microbial contamination due to their small diameter [56].
There was a discrepancy between the two groups regarding corneal perforation, which occurred more frequently in group A. This may have influenced the microsurgical and general results.
The different survival rates in our two groups lead us to assume that cryopreserved corneas are probably less viable in the long term. Nevertheless, we observed that the use of cryopreserved corneas necessitated minor surgery comparatively rarely, possibly due to the absence of resident dendritic cells. These antigen-presenting cells are damaged by cryopreservation [34, 57–59]. Consequently, corneal freezing may be advantageous when performing small and peripherally located penetrating keratoplasties even if postoperative vascularization is present [57, 59].
There are alternative approaches to extending the preservation period of donor corneas before transplantation. One example is the utilization of methods such as irradiation or lyophilization. In most studies involving irradiated corneas, the procedure has been limited to lamellar transplantation due to the predominantly non-viable endothelium after the procedure [60]. Another option for preserving corneas is lyophilization, a method that requires a significant amount of time for both storage and reprocessing [61, 62] due to the necessity to rehydrate the tissue before clinical use. Again, studies have been done primarily for anterior stromal keratoplasty [61].
The limitations of this study include the relatively small sample size; the heterogeneity of the patients, with differing medical histories including ulcer etiology; the relatively large number of surgeons; and the different defect sizes, graft diameters and numbers of vascularizations. However, because there have been only few reports on the medical use of cryopreserved human corneal grafts and because human corneal grafts are rare, our results may be useful.
Conclusion
Cryopreserved tissue can be stored wherever there is a simple freezer, and as such it may represent a useful option for urgent rescue of a patient’s eyesight in situations where regularly processed corneas and further treatment options, such as histoacrylic tissue glue, corneal flap, or amniotic membrane transplantation, are unavailable. Nevertheless, further investigations are necessary for better specification of the medical use of cryopreserved corneas, such as choice of cryopreservation method, trephination diameter, lesion location, as well as identification of the ocular diseases most likely to benefit from such treatment.
Acknowledgements
We thank Dr. Carsten Szardenings (Institute for Biometry and Clinical Research Münster) for statistical advice.
Medical Writing and Editorial Assistance.
David Roseveare was paid as a linguist for correcting spelling and grammatical errors.
Author Contribution
Malik Bidzan was responsible for data collection, analysis, and writing the original draft, including preparing tables and figures. Nicole Eter contributed to logistical and financial support, as well as manuscript revision. Constantin E. Uhlig contributed to the methodology, data collection, analysis, drafting, editing of the article, and manuscript review.
Funding
We acknowledge support from the Open Access Publication Fund of the University of Münster for this study and the journal’s Rapid Service fee.
Data Availability
The datasets generated during and analyzed during the current study are available from the corresponding author on reasonable request.
Declarations
Conflict of Interest
Malik Bidzan, Nicole Eter, and Constantin E. Uhlig declare that they have no conflict of interest.
Ethical Approval
All procedures complied with the tenets of the Declaration of Helsinki and were approved by the local institutional review board (the ethics committee of the Westphalia–Lippe Medical Association and the University of Münster). Patients were informed verbally and in writing about the surgical procedures and provided written consent.
References
- 1.Deshmukh R, Stevenson LJ, Vajpayee R. Management of corneal perforations: an update. Indian J Ophthalmol. 2020;68:7–14. doi: 10.4103/ijo.IJO_1151_19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Stamate A-C, Tătaru CP, Zemba M. Update on surgical management of corneal ulceration and perforation. Rom J Ophthalmol. 2019;63:166–173. [PMC free article] [PubMed] [Google Scholar]
- 3.Uhlig CE, Böhringer D, Hirschfeld G, Seitz B, Schmidt H. Attitudes concerning postmortem organ donation: a multicenter survey in various German cohorts. Ann Transplant. 2015;20:614–621. doi: 10.12659/AOT.894385. [DOI] [PubMed] [Google Scholar]
- 4.Uhlig CE, Busse H, Groppe M. Use of fibrin glue in fixation of amniotic membranes in sterile corneal ulceration. Am J Ophthalmol. 2006;142:189–191. doi: 10.1016/j.ajo.2006.02.043. [DOI] [PubMed] [Google Scholar]
- 5.Hoffmann S, Szentmáry N, Seitz B. Amniotic membrane transplantation for the treatment of infectious ulcerative keratitis before elective penetrating keratoplasty. Cornea. 2013;32:1321–1325. doi: 10.1097/ICO.0b013e318298de10. [DOI] [PubMed] [Google Scholar]
- 6.Jhanji V, Young AL, Mehta JS, Sharma N, Agarwal T, Vajpayee RB. Management of corneal perforation. Surv Ophthalmol. 2011;56:522–538. doi: 10.1016/j.survophthal.2011.06.003. [DOI] [PubMed] [Google Scholar]
- 7.Krysik K, Dobrowolski D, Lyssek-Boron A, Jankowska-Szmul J, Wylegala EA. Differences in surgical management of corneal perforations, measured over six years. J Ophthalmol. 2017;2017:1582532. doi: 10.1155/2017/1582532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Reinshagen H, Maier PC, Böhringer D. Aktivitäten der Sektion Gewebetransplantation und Biotechnologie der Deutschen Ophthalmologischen Gesellschaft: Leistungsbericht 2018. Klin Monbl Augenheilkd. 2021;238:186–190. doi: 10.1055/a-1098-8779. [DOI] [PubMed] [Google Scholar]
- 9.Mihailovic N, Bartel CC, Rosenberger F, et al. Attitudes and knowledge regarding postmortem cornea donation among young and elderly people in germany: sufficient for decision making? Ophthalmic Res. 2019;62:173–184. doi: 10.1159/000501642. [DOI] [PubMed] [Google Scholar]
- 10.Uhlig CE, Koch R, Promesberger J, et al. Attitudes toward postmortem cornea donation in Germany: a multicenter survey. Graefes Arch Clin Exp Ophthalmol. 2014;252:1955–1962. doi: 10.1007/s00417-014-2796-y. [DOI] [PubMed] [Google Scholar]
- 11.Uhlig CE, Promesberger J, Hirschfeld G, Koch R, Reinhard T, Seitz B. Ergebnisse einer internetbasierten Umfrage zur Hornhautspendebereitschaft bei DOG-Mitgliedern. Ophthalmologe. 2012;109:1198–1206. doi: 10.1007/s00347-012-2626-8. [DOI] [PubMed] [Google Scholar]
- 12.Gain P, Jullienne R, He Z, Aldossary M, et al. Global survey of corneal transplantation and eye banking. JAMA Ophthalmol. 2016;134:167–173. doi: 10.1001/jamaophthalmol.2015.4776. [DOI] [PubMed] [Google Scholar]
- 13.Seitz B, Blüthner K, Kruse FE, Reinhard T, Sundmacher R. Hornhauttransplantation: Zu geringe Spendenbereitschaft. Dtsch Ärztebl. 2004;101:A3326–3330. [Google Scholar]
- 14.Galvis V, Tello A, Laiton AN, Salcedo SLL. Indications and techniques of corneal transplantation in a referral center in Colombia, South America (2012–2016) Int Ophthalmol. 2019;39:1723–1733. doi: 10.1007/s10792-018-0994-z. [DOI] [PubMed] [Google Scholar]
- 15.Qureshi S, Dohlman TH. Penetrating keratoplasty: indications and graft survival by geographic region. Semin Ophthalmol. 2023;38:31–43. doi: 10.1080/08820538.2022.2152710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Doğan C, Arslan OŞ. Outcomes of therapeutic and tectonic penetrating keratoplasty in eyes with perforated infectious corneal ulcer. Turk J Ophthalmol. 2019;49:55–60. doi: 10.4274/tjo.galenos.2018.06937. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Gupta N, Upadhyay P. Use of glycerol-preserved corneas for corneal transplants. Indian J Ophthalmol. 2017;65:569–573. doi: 10.4103/ijo.IJO_56_17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kim J-G, Jun JH. Therapeutic and tectonic keratoplasty with simple cryopreserved remnants of donor corneas: an 11 year retrospective case series. Sci Rep. 2022;12:7331. doi: 10.1038/s41598-022-10994-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Corydon C, Hjortdal J, Ehlers N. Re-examination of organ-cultured, cryopreserved human corneal grafts after 27 years. Acta Ophthalmol. 2009;87:173–175. doi: 10.1111/j.1755-3768.2008.01204.x. [DOI] [PubMed] [Google Scholar]
- 20.Mane SK, Patwardhan SD. Use of cryopreserved donor corneal tissues first time in India for therapeutic penetrating keratoplasty during COVID-19 pandemic—a case series. Indian J Ophthalmol. 2021;69:2527–2530. doi: 10.4103/ijo.IJO_627_21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Wang CF. Penetrating keratoplasty with long-term cryopreserved corneas. Zhonghua Yan Ke Za Zhi. 1990;26:17–20. [PubMed] [Google Scholar]
- 22.Eastcott HHG, Holt L, Peacock JH, Rob CG. Preservation of arterial grafts by freeze-drying: a simplified method. Lancet. 1954;266:1311–1314. doi: 10.1016/s0140-6736(54)92206-0. [DOI] [PubMed] [Google Scholar]
- 23.O’Neill P, Mueller FO, Trevor-Roper PD. On the preservation of corneae at -196 degrees C. for full-thickness homografts in man and dog. Br J Ophthalmol. 1967;51:13–30. doi: 10.1136/bjo.51.1.13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Capella JA, Kaufman HE, Robbins JE. Preservation of viable corneal tissue. Cryobiology. 1965;2:116–121. doi: 10.1016/s0011-2240(65)80096-7. [DOI] [PubMed] [Google Scholar]
- 25.Kaufman HE, Capella JA, Robbins JE. The human corneal endothelium. Am J Ophthalmol. 1966;61:835–841. doi: 10.1016/0002-9394(66)90921-4. [DOI] [PubMed] [Google Scholar]
- 26.Schultz RO, Matsuda M, Yee RW, Glasser DB, Sabin SM, Edelhauser HF. Long-term survival of cryopreserved corneal endothelium. Ophthalmology. 1985;92:1663–1667. doi: 10.1016/s0161-6420(85)34088-5. [DOI] [PubMed] [Google Scholar]
- 27.Shao Y, Chen C, Zhou Q, et al. Cryopreservation in Ophthalmology. In: Quain M, editor. Cryopreservation—current advances and evaluations. London: IntechOpen; 2020. 10.5772/intechopen.91312.
- 28.Ehlers N, Sperling S, Olsen T. Post-operative thickness and endothelial cell density in cultivated, cryopreserved human corneal grafts. Acta Ophthalmol (Copenh) 1982;60:935–944. doi: 10.1111/j.1755-3768.1982.tb00624.x. [DOI] [PubMed] [Google Scholar]
- 29.Ehlers N, Sperling S. Ultrastructure of cryopreserved, functioning human corneal endothelium. Acta Ophthalmol (Copenh) 1983;61:245–253. doi: 10.1111/j.1755-3768.1983.tb01418.x. [DOI] [PubMed] [Google Scholar]
- 30.Erdmann L, Ehlers N. Long-term results with organ cultured, cryopreserved human corneal grafts. Re-examination of 17 patients. Acta Ophthalmol (Copenh). 1993;71:703–706. doi: 10.1111/j.1755-3768.1993.tb04666.x. [DOI] [PubMed] [Google Scholar]
- 31.Bourne WM, Nelson LR, Hodge DO. Comparison of three methods for human corneal cryopreservation that utilize dimethyl sulfoxide. Cryobiology. 1999;39:47–57. doi: 10.1006/cryo.1999.2182. [DOI] [PubMed] [Google Scholar]
- 32.Rodríguez-Fernández S, Álvarez-Portela M, Rendal-Vázquez E, et al. Analysis of cryopreservation protocols and their harmful effects on the endothelial integrity of human corneas. Int J Mol Sci. 2021;22:12564. doi: 10.3390/ijms222212564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Shi W, Liu M, Gao H, Li S, Wang T, Xie L. Penetrating keratoplasty with small-diameter and glycerin-cryopreserved grafts for eccentric corneal perforations. Cornea. 2009;28:631–637. doi: 10.1097/ICO.0b013e318191b857. [DOI] [PubMed] [Google Scholar]
- 34.Chen W, Lin Y, Zhang X, et al. Comparison of fresh corneal tissue versus glycerin-cryopreserved corneal tissue in deep anterior lamellar keratoplasty. Invest Ophthalmol Vis Sci. 2010;51:775–781. doi: 10.1167/iovs.09-3422. [DOI] [PubMed] [Google Scholar]
- 35.Liu X, Zhou Q, Huang X, Liu Z, Bi Y. Clinical evaluation of deep anterior lamellar keratoplasty using glycerol-cryopreserved corneal tissues for refractory herpetic stromal keratitis: an observational study. Medicine (Baltimore) 2016;95:e4892. doi: 10.1097/MD.0000000000004892. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Gupta N, Dhasmana R, Maitreya A, Badahur H. Glycerol-preserved corneal tissue in emergency corneal transplantation: an alternative for fresh corneal tissue in COVID-19 crisis. Indian J Ophthalmol. 2020;68:1412–1416. doi: 10.4103/ijo.IJO_1198_20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Lim LS, Arundhati A, Tan DTH. Sequential therapeutic penetrating keratoplasty with cryopreserved and fresh corneal tissue for severe infectious keratitis: a case-control study. Cornea. 2011;30:739–743. doi: 10.1097/ICO.0b013e318203217c. [DOI] [PubMed] [Google Scholar]
- 38.Stamate A-C, Tătaru CP, Zemba M. Emergency penetrating keratoplasty in corneal perforations. Rom J Ophthalmol. 2018;62:253–259. [PMC free article] [PubMed] [Google Scholar]
- 39.Maamri A, Fries FN, Spira-Eppig C, Eppig T, Seitz B. Mitarbeiterbefragung nach Einführung der elektronischen Patientenakte FIDUS an der Universitätsaugenklinik des Saarlandes. Ophthalmologe. 2021;119:471–480. doi: 10.1007/s00347-021-01514-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Bundesärztekammer. Richtlinie zur Gewinnung von Spenderhornhäuten und zum Führen einer Augenhornhautbank, Erste Fortschreibung. Dtsch Arztebl. 2018. 10.3238/arztebl.2018.rl_augenhornhautbank_02.
- 41.Eye Banking Committee of the American Academy of Ophthalmology. Medical standards. Int J Eye Bank. 2021;9:1–39. https://eyebankingjournal.org/article/medical-standards/.
- 42.Day AC, Donachie PHJ, Sparrow JM, Johnston RL. The Royal College of Ophthalmologists’ National Ophthalmology Database study of cataract surgery: report 1, visual outcomes and complications. Eye (Lond) 2015;29:552–560. doi: 10.1038/eye.2015.3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Moussa G, Bassilious K, Mathews N. A novel excel sheet conversion tool from Snellen fraction to LogMAR including ‘counting fingers’, ‘hand movement’, ‘light perception’ and ‘no light perception’ and focused review of literature of low visual acuity reference values. Acta Ophthalmol. 2021;99:e963–e965. doi: 10.1111/aos.14659. [DOI] [PubMed] [Google Scholar]
- 44.Rüfer F, Schröder A, Erb C. White-to-white corneal diameter: normal values in healthy humans obtained with the orbscan II topography system. Cornea. 2005;24:259–261. doi: 10.1097/01.ico.0000148312.01805.53. [DOI] [PubMed] [Google Scholar]
- 45.Maier P, Böhringer D, Reinhard T. Clear graft survival and immune reactions following emergency keratoplasty. Graefes Arch Clin Exp Ophthalmol. 2007;245:351–359. doi: 10.1007/s00417-006-0410-7. [DOI] [PubMed] [Google Scholar]
- 46.Ünal M, Yücel I. Evaluation of topical ciclosporin 0.05% for prevention of rejection in high-risk corneal grafts. Br J Ophthalmol. 2008;92:1411–1414. doi: 10.1136/bjo.2008.143024. [DOI] [PubMed] [Google Scholar]
- 47.Zhang X, Shen L, Jin Y, Saban DR, Chauhan SK, Dana R. Depletion of passenger leukocytes from corneal grafts: an effective means of promoting transplant survival? Invest Ophthalmol Vis Sci. 2009;50:3137–3144. doi: 10.1167/iovs.08-1899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Szaflik JP, Major J, Izdebska J, Lao M, Szaflik J. Systemic immunosuppression with mycophenolate mofetil to prevent corneal graft rejection after high-risk penetrating keratoplasty: a 2-year follow-up study. Graefes Arch Clin Exp Ophthalmol. 2016;254:307–314. doi: 10.1007/s00417-015-3200-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Weisbrod DJ, Sit M, Naor J, Slomovic AR. Outcomes of repeat penetrating keratoplasty and risk factors for graft failure. Cornea. 2003;22:429–434. doi: 10.1097/00003226-200307000-00008. [DOI] [PubMed] [Google Scholar]
- 50.Al-Mezaine H, Wagoner MD. Repeat penetrating keratoplasty: indications, graft survival, and visual outcome. Br J Ophthalmol. 2006;90:324–327. doi: 10.1136/bjo.2005.079624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Barraquer RI, Pareja-Aricò L, Gómez-Benlloch A, Michael R. Risk factors for graft failure after penetrating keratoplasty. Medicine (Baltimore) 2019;98:e15274. doi: 10.1097/MD.0000000000015274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Brunette I, Le François M, Tremblay M-C, Guertin MC. Corneal transplant tolerance of cryopreservation. Cornea. 2001;20:590–596. doi: 10.1097/00003226-200108000-00007. [DOI] [PubMed] [Google Scholar]
- 53.Halberstadt M, Böhnke M, Athmann S, Hagenah M. Cryopreservation of human donor corneas with dextran. Invest Ophthalmol Vis Sci. 2003;44:5110–5115. doi: 10.1167/iovs.03-0370. [DOI] [PubMed] [Google Scholar]
- 54.Yao Y-F, Zhang Y-M, Zhou P, Zhang B, Qiu W-Y, Tseng SCG. Therapeutic penetrating keratoplasty in severe fungal keratitis using cryopreserved donor corneas. Br J Ophthalmol. 2003;87:543–547. doi: 10.1136/bjo.87.5.543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Ying L-Y, Qiu W-Y, Wang B-H, Zhou P, Zhang B, Yao Y-F. Corneal endothelial regeneration in human eyes using endothelium-free grafts. BMC Ophthalmol. 2022;22:32. doi: 10.1186/s12886-022-02260-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Niu G, Zhou Q, Huang X, et al. Individualized penetrating keratoplasty using edge-trimmed glycerol-preserved donor corneas for perforated corneal ulcers. BMC Ophthalmol. 2019;19:85. doi: 10.1186/s12886-019-1091-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Amouzegar A, Chauhan SK, Dana R. Alloimmunity and tolerance in corneal transplantation. J Immunol. 1950;2016(196):3983–3991. doi: 10.4049/jimmunol.1600251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Qazi Y, Hamrah P. Corneal allograft rejection: immunopathogenesis to therapeutics. J Clin Cell Immunol. 2013;2013:006. doi: 10.4172/2155-9899.S9-006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Amouzegar A, Chauhan SK. Effector and regulatory T cell trafficking in corneal allograft rejection. Mediators Inflamm. 2017;2017:e8670280. doi: 10.1155/2017/8670280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Mathews PM, Fogla R, Samayoa E, VanCourt S, Akpek EK. Long-term clinical outcomes of keratoplasty using gamma-irradiated corneal lenticules. BMJ Open Ophthalmol. 2019;4:e000396. doi: 10.1136/bmjophth-2019-000396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Rovere M-R, Ouilhon C, Salmon D, Haftek M, Damour O, Auxenfans C. Development and characterization of Lyophilized Transparized Decellularized stroma as a replacement for living cornea in deep anterior lamellar keratoplasty. Cell Tissue Bank. 2019;20:49–59. doi: 10.1007/s10561-018-9742-x. [DOI] [PubMed] [Google Scholar]
- 62.Farias R, Barbosa L, Lima A, et al. Deep anterior lamellar transplant using lyophilized and Optisol corneas in patients with keratoconus. Cornea. 2008;27:1030–1036. doi: 10.1097/ICO.0b013e31817e903a. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets generated during and analyzed during the current study are available from the corresponding author on reasonable request.