Abstract
Background/Aims
Mesenchymal stem cells (MSCs) are potential alternatives for liver fibrosis treatment; however, their optimal sources remain uncertain. This study compares the ex-vivo expansion characteristics of MSCs obtained from adipose tissue (AT) and umbilical cord (UC) and assesses their therapeutic potential for liver fibrosis treatment.
Methods
Since MSCs from early to mid-passage numbers (P2–P6) are preferable for cellular therapy, we investigated the growth kinetics of AT-MSCs and UC-MSCs up to P6 and evaluated their therapeutic effects in a rat model of liver fibrosis induced by diethylnitrosamine.
Results
Results from the expansion studies demonstrated that both cell types exhibited bona fide characteristics of MSCs, including surface antigens, pluripotent gene expression, and differentiation potential. However, AT-MSCs demonstrated a shorter doubling time (58.2 ± 7.3 vs. 82.3 ± 4.3 h; P < 0.01) and a higher population doubling level (10.1 ± 0.7 vs. 8.2 ± 0.3; P < 0.01) compared to UC-MSCs, resulting in more cellular yield (230 ± 9.0 vs. 175 ± 13.2 million) in less time. Animal studies demonstrated that both MSC types significantly reduced liver fibrosis (P < 0.05 vs. the control group) while also improving liver function and downregulating fibrosis-associated gene expression.
Conclusion
AT-MSCs and UC-MSCs effectively reduce liver fibrosis. However, adipose cultures display an advantage by yielding a higher number of MSCs in a shorter duration, rendering them a viable choice for scenarios requiring immediate single-dose administration, often encountered in clinical settings.
Keywords: mesenchymal stem cells, adipose tissue, umbilical cord, liver fibrosis, cellular therapy
Graphical abstract
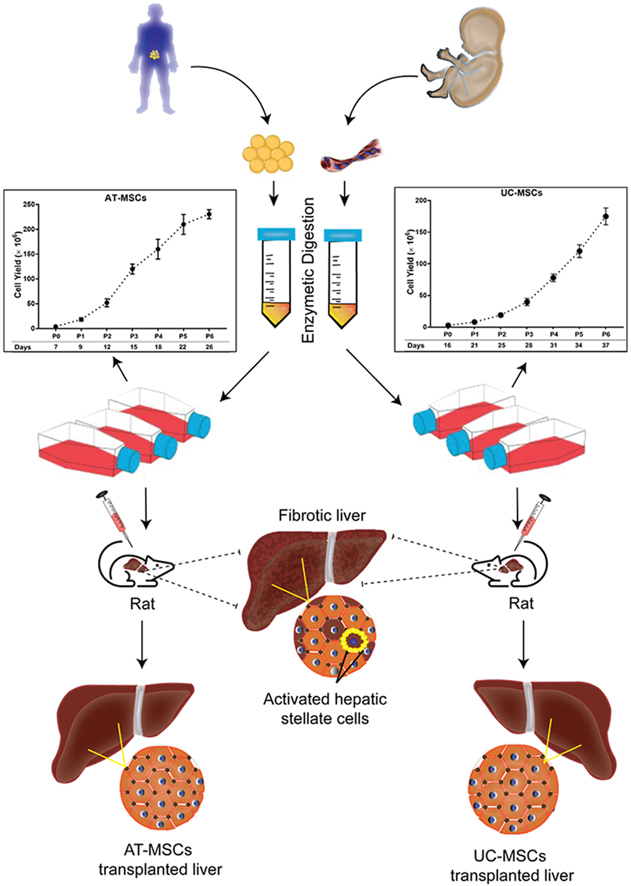
Hepatic fibrosis, which is defined as excessive deposition of extracellular matrix (ECM) proteins, is the final common pathway of nearly all chronic liver diseases. If hepatotoxic factors continue to exist, hepatic fibrosis can be a precursor to cirrhosis and hepatocellular cancer and therefore represents the first step towards a fatal outcome of liver disease.1 Although various preclinical studies are being carried out to investigate potential therapies for hepatic fibrosis, there is currently no established treatment in a clinical setting. Thus, developing a viable therapy for hepatic fibrosis is crucial.2
Advances in stem cell research over the last few decades have offered hope that stem cells may be able to treat hepatic fibrosis. Among the different types of stem cells studied over these years, such as embryonic stem cells,3 induced-pluripotent stem cells,4 hematopoietic stem cells,5 and mesenchymal stem cells (MSCs),6 the latter has gained increasing attention. This is due to its ease of acquisition from various tissues, its strong capabilities for proliferation and differentiation, as well as its immune privilege and immunomodulatory characteristics.7 Furthermore, MSCs have also been shown to inhibit hepatic stellate cell (HSC) activation, ameliorate oxidative and inflammatory stress, stimulate hepatocyte proliferation, and promote hepatic regeneration. MSCs could therefore be the best choice for hepatic fibrosis treatment.8 However, because recent studies have revealed variances in MSCs from various sources,9 there is a dearth of evidence to establish which source of MSCs is most appropriate for MSC acquisition and hepatic fibrosis treatment.
The most commonly used MSCs for the treatment of liver disease in clinical settings are derived from bone marrow (BM-MSCs). However, these MSCs have certain limitations, including an invasive isolation process, low cell yield, and early senescence during in vitro expansion.10 Moreover, BM-MSCs interventions are usually autologous, which may lead to reduced efficacy in elderly patients due to age-related declines in BM-MSCs effectiveness.11 To overcome these limitations, researchers are exploring alternative sources of MSCs. Currently, scientists are showing interest in MSCs derived from adipose tissue (AT-MSCs) and umbilical cords (UC-MSCs). These MSCs are easy to harvest without causing donor morbidity and possess excellent proliferative and differentiation properties. Additionally, because of their lower immunogenicity, allogeneic transplantation of AT-MSCs and UC-MSCs is possible, allowing for a sufficient number of MSCs with better therapeutic qualities to be obtained quickly in clinical settings.12,13
Previous research has identified several therapeutic properties of AT-MSCs and UC-MSCs, including anti-fibrogenic,14,15 anti-inflammatory,16,17 and anti-oxidative properties.18,19 However, there has been no comparative investigation of their efficacy in treating hepatic fibrosis. Considering the lack of preclinical studies, the present research was designed to achieve two objectives: (1) compare the ex-vivo expansion characteristics of AT-MSCs and UC-MSCs, and (2) examine the therapeutic potential of AT-MSCs and UC-MSCs using animal models of diethylnitrosamine (DEN)-induced hepatic fibrosis. We anticipate that our findings will aid clinicians in selecting the most suitable type of MSCs for hepatic fibrosis treatment.
Materials and Methods
Collection of Adipose and Cord Tissue
Tissue samples were collected from consenting donors following local institutional review board instructions. Adipose tissue (n = 3) was obtained from the abdominal area of healthy female donors (age: 28 ± 4) who were undergoing elective plastic surgery at Jinnah Hospital, Lahore, Pakistan. Umbilical cords (n = 3) were obtained from healthy mothers (age: 27 ± 3) who had undergone full-term caesarean sections at Sir Ganga Ram Hospital, Lahore, Pakistan. The collected samples were immediately transferred to the cell culture facility while being maintained at ambient temperature (20–25 °C) and suspended in phosphate-buffered saline (PBS).
Mesenchymal Stem Cells Isolation and Culture
Adipose Tissue-Mesenchymal Stem Cells Isolation
AT-MSCs were isolated from collected adipose tissue using the enzymatic digestion method, as previously described.20 Briefly, the sample was washed with an equal volume of PBS, incubated with a type IA collagenase solution (Sigma–Aldrich, USA; 1 mg/ml in serum-free culture medium), and then centrifuged at 1200 rpm to obtain a homogeneous tissue suspension. The obtained cell pellet (SVF: stromal vascular fraction) was seeded onto a 75 cm2 culture flask (T75 flask) containing a complete culture medium composed of Dulbecco's modified eagle's medium-low glucose (DMEM-LG; Sigma–Aldrich, USA) supplemented with 10% fetal bovine serum (FBS; Gibco, USA) and 1% antibiotic solution (100 IU/ml penicillin; Sigma–Aldrich, USA; and 100 μg/ml streptomycin; MP Biomedicals, USA).
Umbilical Cord-Mesenchymal Stem Cells Isolation
To isolate UC-MSCs, an umbilical cord was processed using the enzymatic digestion method as previously described.21 Briefly, the collected cord was cut into 2–3 cm sections, and the umbilical vessels were removed. Then, Wharton's jelly was collected, minced into 1–5 mm pieces, and incubated with a collagenase solution (3 mg/ml in serum-free culture medium) in a T75 flask at 37 °C for a 3-hour digestion period in a CO2 incubator. To neutralize the reaction, a complete culture medium containing DMEM-high glucose, 10% FBS, and 1% antibiotic solution was added.
Establishment of Mesenchymal Stem Cells Culture and Expansion Conditions
Next, the cells were cultured in a humidified CO2 incubator under standard conditions for 48 h, and then cellular debris and floating cells were removed. Subsequently, the culturale medium was refreshed every third day. When the cells reached 80–90% confluence, they were passaged multiple times using 0.25% trypsin/EDTA (Gibco, USA) and each time seeded into new T75 flasks at a density of 1 × 106 cells/flask. The primary cell culture was designated as P0, and subsequent passages were named P1–P6.
Mesenchymal Stem Cells Expansion Characteristics
The expansion characteristics of AT-MSCs and UC-MSCs were compared by determining their growth kinetics, including cell doubling time (DT), cumulative population doubling level (cPDL), and total cell yield at each passage up to P6. In addition, cellular morphology, proliferation, clonogenic potential, and cellular senescence were evaluated, particularly at P2 and P6.
Growth Kinetics
During continuous sub-passaging, the detached cells were counted using a dual-chamber hemocytometer and 0.4% trypan blue solution (Sigma–Aldrich, USA). The numbers of cells at both seeding and harvesting times were recorded to calculate the DT and cPDL using the following equations22:
The total culture duration is denoted by CT, where N represents the number of cells harvested and N0 represents the number of cells seeded.
Cells Morphology and Size
At each passage, both types of cells were observed under an inverted microscope at 80% confluency, and images were captured at 200x magnification. In addition, at P2 and P6, 1500 cells/cm2 were cultured separately in 24-well plates and stained with crystal violet (0.5% in methanol, Sigma–Aldrich, USA) for 10 min. Morphological evaluation was performed, and changes in cell size were analyzed using the Image J software (National Institutes of Health, USA).
Colony Forming Units-Fibroblasts
At P2 and P6, colonies were grown by seeding cells at a density of 100 cells per well of a 6-well plate and allowing them to grow under standard culture conditions with medium replacement every 3 days. After 14 days, colonies (comprising >50 cells) were stained with crystal violet and counted macroscopically.
Cellular Senescence
To evaluate cellular senescence, a commercially available senescence detection kit (Abcam, USA) was utilized to detect senescence-associated β-galactosidase (SA-β-gal) activity. Cells were seeded in 24-well plates at a density of 1500 cells/cm2 and cultured until they reached 70–80% confluency. Following that, the cells were stained with the β-gal solution, and images were captured at 200x magnification. The number of β-gal-positive cells (stained blue) was then quantified.
Mesenchymal Stem Cells Characterization
Mesenchymal Stem Cells-Specific Cluster of differentiation (CD) Markers Expressions
At passage 6, MSC-specific CD markers were analyzed through an immunocytochemical staining protocol.23 Cells were seeded at a density of 30,000 cells over coverslips in a 6-well plate and allowed to reach 80% confluency. After fixation, cells were incubated with primary antibodies against CD34, CD45, CD90, and CD105 (1:50; Santa Cruz, USA), followed by corresponding secondary antibodies (1:500; Invitrogen, USA). The cells were counterstained with 4, 6-diamidino-2-phenylindole (DAPI) nuclear stain, and images were captured under a fluorescent microscope (Olympus BX61, Japan). The positive cells (green) were counted and reported as a percentage of the total number of cells (blue).
Pluripotent Genes Expressions
The expression of pluripotent genes (OCT4, SOX2, and NANOG) in passage 6 cells was analyzed using semi-quantitative Polymerase chain reaction (PCR). In brief, RNA was extracted from at least 1 million cells with TRIzol reagent (Sigma–Aldrich, USA), followed by quantification with NanoDrop 1000 (Thermo Fisher, USA) and cDNA synthesis using the Revert Aid H-Minus first-strand cDNA synthesis kit (Invitrogen, USA). The PCR reactions were run on PikoReal 96 using Maxima SYBR Green PCR Master Mix (Fermentas, USA), with glyceraldehyde-3-phosphate dehydrogenase (GAPDH) used as a normalization control. The relative expression was determined using the comparative CT method (ΔΔCT) and represented as a fold change using the formula 2−ΔΔCt. In addition, PCR products were visualized on 2% agarose gels and imaged using a gel documentation system. Table 1 lists the primers used in the study.
Table 1.
Primer Sequences Used for Semi-quantitative PCR.
| Gene | Forward primer | Reverse primer |
|---|---|---|
| OCT4 | GGAAGGTATTCAGCCAAACGAC | TTCTCCAGGTTGCCTCTCAC |
| SOX2 | AACACCAATCCCATCCACAC | TTCGTAGCTTTCTTTCCTCCAG |
| NANOG | AGATGCCTCACACGGAGACT | TCTGCTGGAGGCTGAGGTAT |
| GAPDH | GCACCACCAACTGCTTAGCA | CTTCCACGATACCAAAGTTGTCAT |
| Tgf-β1 | TGCGCCTGCAGAGATTCAAG | AGGTAACGCCAGGAATTGTTGCT |
| Col1α1 | CAAGATGGTGGCCGTTACTAC | TTAGTCCTTACCGCTCTTCCAG |
| α-SMA | AGCTCTGGTGTGTGACAATG | ATAGAAGGAGTGGTGCCAGA |
| β-Actin | GCTGTGTTGTCCCTGTATGC | GAGCGCGTAACCCTCATAGA |
α-SMA, Alpha smooth muscle actin; Col1α1, collagen type 1 alpha 1; Tgf-β1, Transforming growth factor beta 1.
Differentiation Potential
StemPro Differentiation Kits were used to differentiate AT-MSCs and UC-MSCs into osteocytes, adipocytes, and chondrocytes, as previously reported.24 Oil red O, alizarin red S, and alcian blue (all from Sigma–Aldrich, USA) were used to stain cells after the differentiation process in order to see the lipid vacuoles in adipocytes, calcium deposits in osteocytes, and proteoglycans in chondrocytes, respectively. The extent of stain uptake was also evaluated using a calorimetric assay. To accomplish this, oil red O was released with 4% Nonidet-P-40 substitute in isopropanol, alcian blue was extracted with 6 mol/L guanidine hydrochloride, and alizarin red S was extracted with 10% acetic acid. Absorbance measurements were then taken at 490 nm, 620 nm, and 405 nm, respectively.
In Vivo Experimental Design
Animals
Transplantation studies were conducted using male Sprague–Dawley rats (SD rats, n = 32) weighing 60–100 g and aged 4–6 weeks. The rats were housed in a climate-controlled animal care facility with standard rodent food and water. The Institutional Animal Ethics Committee, National Centre of Excellence in Molecular Biology, University of Punjab, Pakistan, approved all procedures of the animal experiments.
Liver Fibrosis Induction and Mesenchymal Stem Cells Transplantation
DEN (Sigma–Aldrich, USA; 30 mg/kg, twice a week) was used to chemically induce liver fibrosis in 22 rats for a period of 10 weeks, as reported previously.25 After 12 weeks, hepatic index measurements were taken from four rats treated with DEN and from four vehicle control rats. The remaining rats administered with DEN were randomly divided into three groups (n = 6 per group): sham, AT-MSCs, and UC-MSCs. To administer AT-MSCs or UC-MSCs, rats were given anesthesia using a mixture of ketamine and xylazine, and their abdomens were cut open to expose the left lateral lobe of the liver. Then, 1 × 106 cells suspended in 200 μl PBS and labeled with PKH-26 dye (Sigma–Aldrich, USA) were administered via a 26-gauge needle. Vehicle-control rats (n = 6) were given PBS.
Sample Collection
DEN administration was recommenced once a week to prevent spontaneous fibrosis recovery. On the 28th day after cell infusion, all rats in the experiment were euthanized to obtain samples. Blood samples were obtained through a cardiac puncture, followed by the removal of the liver. Each liver sample was sliced, with one piece placed in TRIzol reagent for RNA extraction and the remaining liver preserved in 4% paraformaldehyde.
Assessment of Transplantation Outcome
Liver Histopathology
Liver tissues were fixed, embedded in paraffin, and cut into 5 μm sections. These sections were then stained with DAPI, hematoxylin and eosin (H&E), Sirius red (Direct red 80; Sigma–Aldrich, USA), Masson (Sigma–Aldrich, USA), and immunohistochemical markers, following standard protocols.
To track the transplanted cells, PKH-26-labeled cells were examined in liver sections. Two sections per rat liver (12 sections per group) were stained with DAPI and examined under a fluorescent microscope. The number of PKH-26 labeled cells (red) was counted and expressed as a percentage of the total number of cells (blue) in the field.
For evaluation of liver architecture changes, two sections per rat liver (12 sections per group) were stained with H&E, while Sirius red and Masson's staining were used to assess collagen deposition in the fibrotic area. Nonoverlapping fields were captured, and stained areas were quantified using Image J software.
Alpha smooth muscle actin (α-SMA) protein was detected using routine immunohistochemical methods with diaminobenzidine (DAB) chromogen and hematoxylin counterstain. Firstly, sections were incubated with anti-α-SMA (1:100; Sigma–Aldrich, USA) overnight at 4 °C. This was followed by incubation with horseradish peroxidase-conjugated secondary antibodies (1:1000; Santa Cruz, USA) for 30 min. The immunoreactive areas were then detected using DAB solution, and sections (12 sections per group) were subsequently counterstained with hematoxylin. Photomicrographs were randomly taken, and the percent area expressing α-SMA was measured using Image J software.
Gene Expressions
Transforming growth factor beta 1 (Tgf-β1), collagen type 1 alpha 1 (Col1α1) and α-Sma mRNA expression were detected using semi-quantitative PCR, with β-Actin serving as the internal control.
Blood Biochemistry
The collected blood samples were centrifuged at 15,000 rpm for 10 min to isolate serum. The DiaSys Diagnostic Systems assay kits were used to analyze serum levels of alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), and albumin (ALB) to assess liver injury/function markers following the manufacturer's protocol.
Statistical Analysis
All the numerical data passed the normality test, and thus they were subjected to parametric analysis. The unpaired t-test was used to compare two groups, and ANOVA tests were used for more than two groups, followed by Tukey's or Sidak's post hoc test to analyze differences between the groups. All statistical analyses were performed using GraphPad Prism version 7.00 for Windows (GraphPad Software, USA), and findings were expressed as means and standard deviations of at least three independent in vitro experiments and n = 6 rats per group in animal experiments. The statistical significance level was set at P < 0.05.
Results
Comparison of Expansion Characteristics of Adipose Tissue-Mesenchymal Stem Cells and Umbilical Cord-Mesenchymal Stem cells
Morphological Features
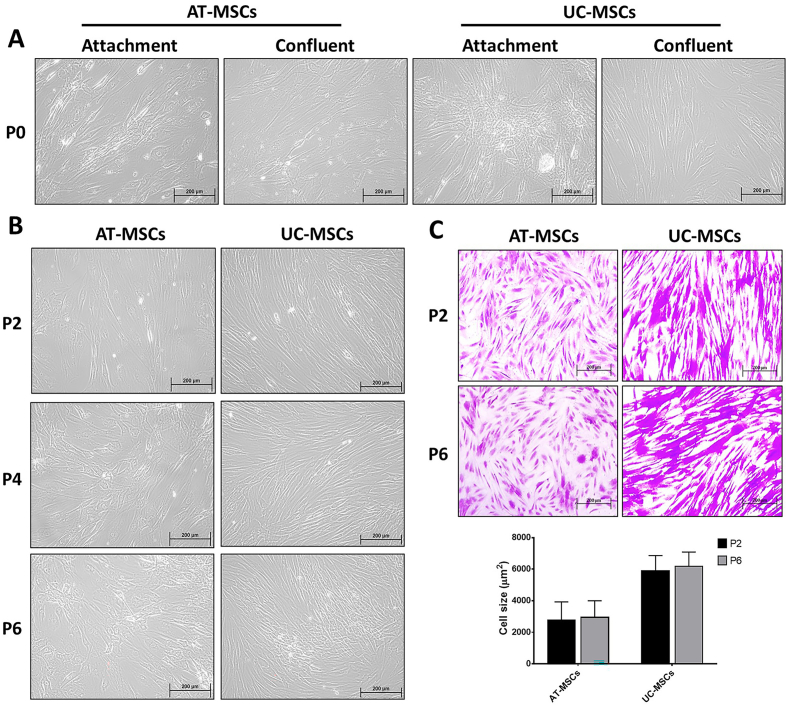
AT-MSCs and UC-MSCs were successfully isolated via plastic adherence in tissue culture flasks. AT-MSCs exhibited a fibroblast-like morphology within 1–2 days and reached confluency in 4–6 days, while UC-MSCs had a delayed attachment (8–10 days) and grew into large colonies, reaching confluency in 15–18 days. Initially, AT-MSCs exhibited varying morphology, with some cells being flat and round while others were spindle-shaped and had shorter and thinner cytoplasmic projections. On the other hand, UC-MSCs showed a homogeneous monolayer of spindle-shaped cells with longer cytoplasmic projections, as illustrated in Figure 1A. However, both MSC types displayed a uniform spindle-shaped morphology in subsequent passages, as shown in Figure 1B. Notably, no morphological changes were observed in either type of MSCs during ex-vivo expansion from P2 to P6, as depicted in Figure 1C.
Figure 1.
Morphology and size of AT-MSCs and UC-MSCs (A) morphology at P0: AT-MSCs exhibited heterogeneous morphology, whereas UC-MSCs exhibited homogeneous morphology. (B) Morphology at P2, P4, and P6: Both MSCs exhibited a uniform spindle-shaped morphology. (C) Cell size at P2 and P6: Cells were stained with crystal violet, and the average cell size was analyzed using the Image J software. No changes in cell size were observed in either type of MSCs. All micrographs were taken at 200x magnification and are representative of three AT-MSCs and UC-MSCs cultures from different donors. AT-MSCs, adipose tissue-mesenchymal stem cells; UC-MSCs, umbilical cord-mesenchymal stem cells.
Growth Expansion Characteristics
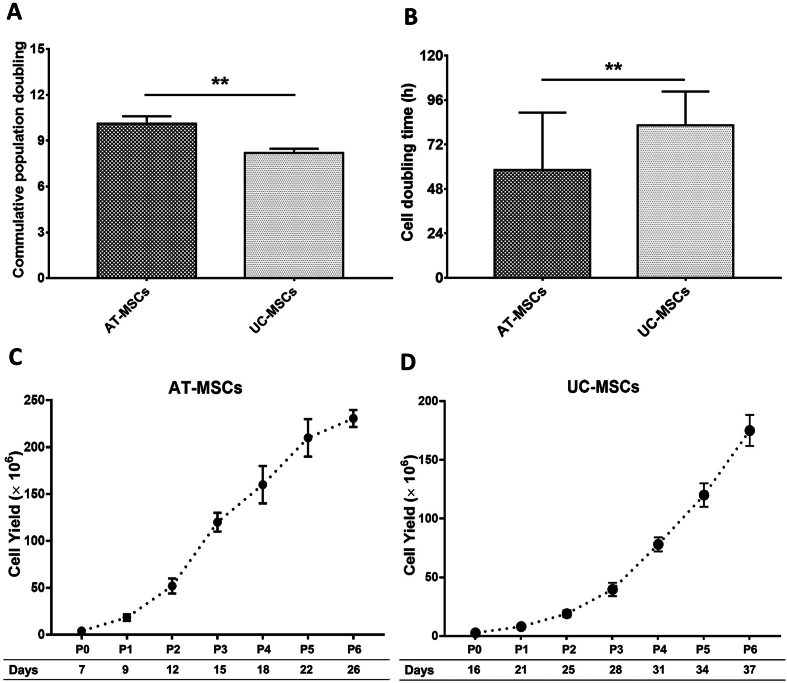
Growth kinetics studies revealed higher population doublings (10.1 ± 0.7 vs. 8.2 ± 0.3; P < 0.01) and shorter doubling time (58.2 ± 7.3 vs. 82.3 ± 4.3 h; P < 0.01) for AT-MSCs than for UC-MSCs from P1 to P6 cultivation (Figure 2A and B). In these expansion conditions, higher cellular yield (230 ± 9.0 vs. 175 ± 13.2 million) was obtained in a shorter time (∼26 vs. ∼37 days) from adipose cultures compared to cord cultures at P6 (Figure 2C and D).
Figure 2.
Growth characteristics of AT-MSCs and UC-MSCs were calculated over six passages: (A) cumulative population doubling level; (B) cell doubling time; (C) AT-MSC yield; and (D) UC-MSC yield. The data show that AT-MSCs have a shorter doubling time, a higher population doubling level, and a higher cellular yield than UC-MSCs. The data is presented as the ± SD of three independent cultures. ∗∗ indicates the statistical significance of P < 0.01. AT-MSCs, adipose tissue-mesenchymal stem cells; UC-MSCs, umbilical cord-mesenchymal stem cells.
Self-Renewal Capacity
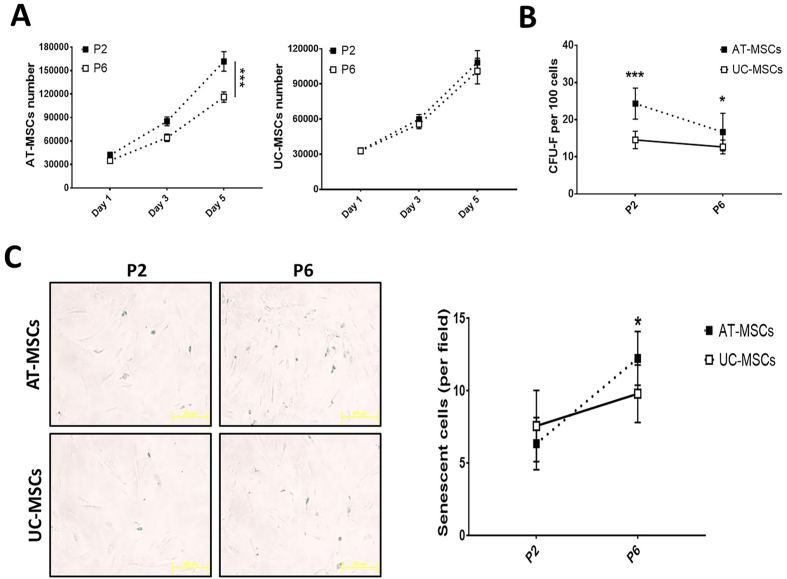
Cell proliferation and CFU-F were assessed at P2 and P6 to determine the self-renewal ability of continuously propagated MSCs. The results demonstrated that the number of AT-MSCs at P2 was considerably higher than that at P6 (P < 0.001). In contrast, there was no significant difference between P2 and P6 UC-MSCs. However, the expansion of P2 and P6 UC-MSCs was limited, with only 3.6- and 3.4-fold increases, respectively, while AT-MSCs at P2 and P6 expanded 5.4- and 3.9-fold, respectively (Figure 3A).
Figure 3.
Self-renewal capacity of AT-MSCs and UC-MSCs at different passage numbers (A) cell proliferation: Cell numbers were counted on days 1, 3, and 5 to determine the growth rate of early (P2) and late (P6) passage cells. (B) CFU–F: Bar diagram showing the number of CFU-F at P2 and P6 counted macroscopically. (C) β-galactosidase staining: In micrographs, β-galactosidase-stained positive bluish-green cells are senescent (200x magnification). Bar diagram showing the number of senescent cells at P2 and P6. Graphical data is presented as the mean ± SD of three independent cultures. ∗P < 0.05; ∗∗∗P < 0.001. AT-MSCs, adipose tissue-mesenchymal stem cells; UC-MSCs, umbilical cord-mesenchymal stem cells; CFU-F, colony-forming units-fibroblasts.
Consistent with these findings, CFU-F numbers were examined at P2, which showed that AT-MSCs had more colonies than UC-MSCs (24 ± 4 vs. 15 ± 3, P < 0.001). However, at P6, AT-MSCs displayed fewer colonies, while UC-MSCs showed similar potential to that seen at P2 (18 ± 4 vs. 14 ± 3, P < 0.05) (Figure 3B).
To investigate whether the reduced cell proliferation or CFU-F in AT-MSCs at a late passage was related to cellular aging, we examined the presence of senescent cells. As shown in Figure 3C, no significant difference in the number of senescent cells was observed in either AT-MSCs or UC-MSCs at early passage. However, at a later passage, AT-MSCs exhibited a significantly higher number of senescent cells compared to UC-MSCs (P < 0.05).
Comparison of Mesenchymal Stem Cells Bonafide Characteristics in Adipose Tissue-Mesenchymal Stem Cells and Umbilical Cord-Mesenchymal Stem Cells
Multipotency CD Markers
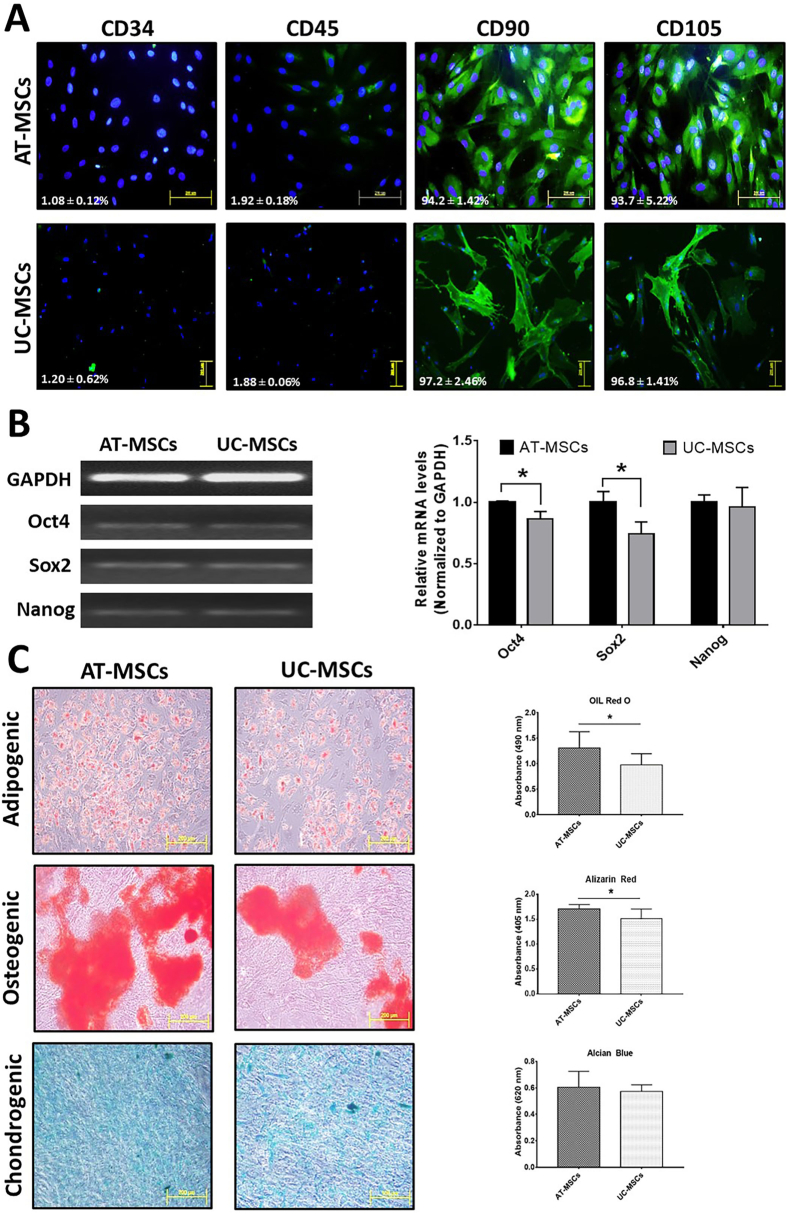
We evaluated whether the cultured AT-MSCs and UC-MSCs at P6 exhibited the characteristic MSC phenotype using a panel of MSC-specific positive and negative cell surface markers. As shown in Figure 4A, both cultures displayed >90% expression of CD90 and CD105 and <2% expression of CD34 and CD45, indicating their MSC identities.
Figure 4.
Characterizations of AT-MSCs and UC-MSCs at the 6th passage (A) MSC-specific surface marker expressions: Immunolabeled fluorescence micrographs showed >90% expressions for CD90 and CD105 and <2% expressions for CD34 and CD45 markers. (B) Pluripotent gene expressions: Bar diagrams show the relative mRNA levels of OCT4, SOX2, and NANOG genes normalized to GAPDH, and agarose gels show PCR product bands. (C) Trilineage differentiation potential: Adipogenic differentiation is shown by oil-red O-stained lipid-rich vesicles; osteogenic differentiation is shown by alizarin red S-stained mineralized matrix deposition; and chondrogenic differentiation is shown by alcian blue-stained glycosaminoglycan matrix deposition. Bar diagrams compare stain uptake levels in AT-MSCs and UC-MSCs. All micrographs were taken at 200x magnification and are representative of three independent cultures from different donors. ∗P < 0.05. AT-MSCs, adipose tissue-mesenchymal stem cells; UC-MSCs, umbilical cord-mesenchymal stem cells.
Pluripotent Genes Expression
To further observe the effect of long-term cultivation on the pluripotency of MSCs, the relative expression of pluripotent genes (OCT4, SOX2, and NANOG) was measured by performing semi-quantitative PCR. Our results revealed positive expression of pluripotent genes in both cell populations, but with some differences in level of expression between them. Specifically, OCT4 and SOX2 expression were slightly higher in AT-MSCs than in UC-MSCs, as shown in Figure 4B.
Multilineage Potential
Finally, to confirm their MSC identities, AT-MSCs and UC-MSCs were cultured in adipogenic, osteogenic, and chondrogenic mediums to assess their multilineage differentiation efficiency. Morphological changes, including vesicle formation due to differentiation into adipocytes and increased cell size due to osteogenic and chondrogenic differentiations, were observed in both MSC cultures. Oil-red O and alizarin red S staining revealed that AT-MSCs had more lipid-rich vesicles and mineralized matrix deposition compared to UC-MSCs. Alcian blue staining revealed that both cultures exhibited similar levels of glycosaminoglycan matrix deposition. Likewise, quantitative analysis demonstrated that AT-MSCs differentiated into adipocytes and osteocytes to a greater degree than UC-MSCs (P < 0.05), while both cultures exhibited similar chondrogenic differentiation capacity (Figure 4C).
Comparison of Antifibrotic Effects of Adipose Tissue-Mesenchymal Stem Cells and Umbilical Cord-Mesenchymal Stem Cells
The ultimate goal of this study was to assess the efficacy of AT-MSCs with UC-MSCs in reducing liver fibrosis. To do so, we transplanted P6 cells into a rat model of liver fibrosis and evaluated histological restoration using histomorphological, molecular, and serum analyses.
Induction of Liver Fibrosis
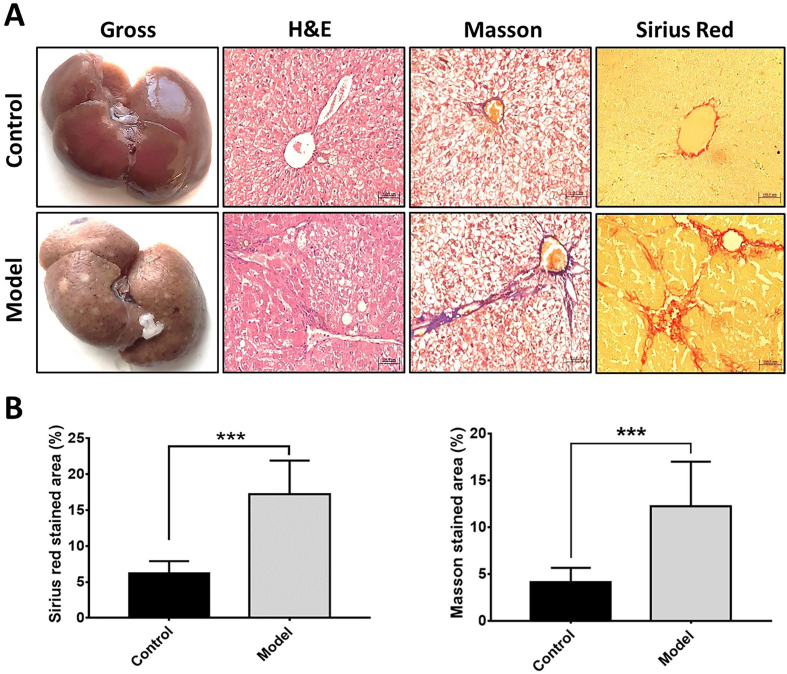
We validated the induction of fibrosis in rats via DEN administration before commencing therapeutic evaluation. Compared to normal control rats, DEN-administered model rats exhibited typical pathological features of liver inflammation and fibrosis, such as disorganization of hepatic cords, inflammatory cell infiltration, and deposition of thin collagen fibers in portal tracts or around the central vein (Figure 5A). Quantitative analysis showed significantly higher Masson and Sirius-red-stained regions in the model rats compared to the control rats (P < 0.001), indicating successful liver fibrosis induction (Figure 5B).
Figure 5.
Rat model of liver fibrosis (A) histopathological changes in the model rats: Gross images show a coating of yellowish fibrous material; H&E stain micrographs show degeneration of liver cells; and Masson and Sirius red stain micrographs show the formation of a fibrous septum in model rat liver tissue. (B) Bar graphs show percentages of fibrotic areas measured using Image J software. All micrographs were taken at 200x magnification. Graphical data is expressed as mean ± SD (n = 4 rats per group). ∗∗∗P < 0.001. H&E, hematoxylin and eosin.
Mesenchymal Stem Cells Transplantation Reverses Pathological Changes of Liver Fibrosis
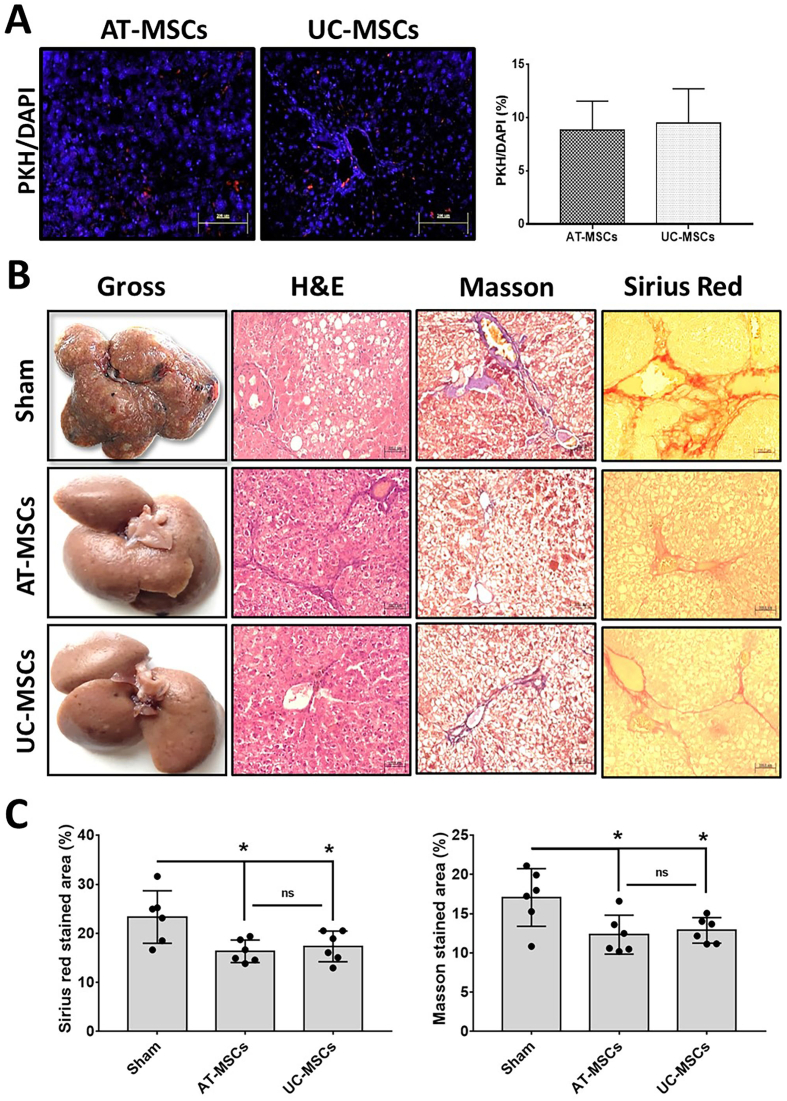
The liver sections were examined four weeks after MSC administration to determine the localization of the transplanted MSCs. As depicted in Figure 6A, PKH-labeled MSCs were detected in both groups, indicating that the transplanted MSCs had been engrafted into the injured liver. Nonetheless, no significant difference in the engraftment ratio between AT-MSCs and UC-MSCs was observed.
Figure 6.
Histopathological examination (A) homing of transplanted MSCs: DAPI (blue) represented cell nuclei and was counted as the total number of cells; PKH-26 (red) represented labeled cells and was counted as PKH-26-positive cells (200x magnifications). Bar graph compares the percentage of PKH-26-labeled cells. (B) Gross images, H&E, Masson, and Sirius red micrographs: Both types of MSCs recover hepatic architecture and reduce collagen-positive areas, as illustrated in H&E and collagen-stained micrographs, respectively (200x magnification). (C) Bar graphs compare Sirius- and Masson-stained collagen areas in experimental groups. All graphical data is expressed as mean ± SD (n = 6 rats per group). ∗P < 0.05; ∗∗P < 0.01. MSCs, mesenchymal stem cells; DAPI, 6-diamidino-2-phenylindole; H&E, hematoxylin and eosin.
Next, a histomorphological examination was conducted to determine whether engrafted MSCs (AT-MSCs or UC-MSCs) could reverse the pathological changes observed in DEN-intoxicated rats. Interestingly, in both MSCs-treated groups, there was an improvement in the pathological features of liver fibrosis, with only mild vacuolar degeneration and fewer fibrotic areas observed. In contrast, the sham-treated group showed an aggravation of these features, with thick collagen fibers invading the hepatic lobules, extensive steatosis, and bridging necrosis observed in their livers (Figure 6B). Although the reduction in collagen staining areas in the AT-MSCs-treated group compared to the UC-MSCs-treated group was not statistically significant, it is noteworthy that both MSCs-treated groups displayed a significant reduction in collagen staining areas in contrast to the sham-treated group (P < 0.05), as illustrated in Figure 6C.
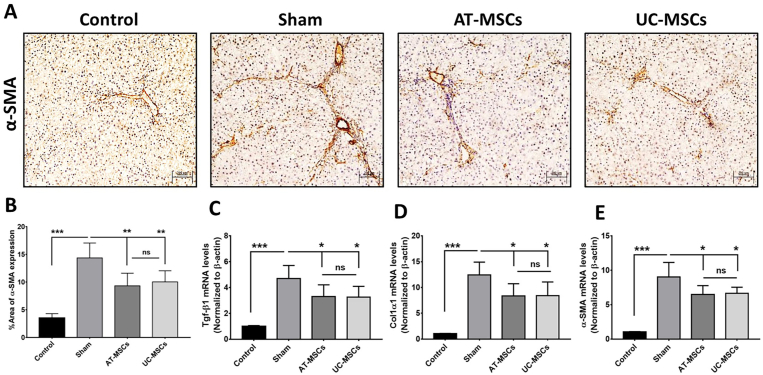
In addition, DEN administration caused a considerable increase in α-SMA expression in perisinusoidal areas, indicating HSC activation in the affected hepatic lobuli of the sham-treated group (P < 0.001 vs. the control group). However, both AT-MSCs and UC-MSCs treatments significantly reduced α-SMA-positive regions (P < 0.01 vs. sham treatment), with no significant difference between the two groups (Figures 7A and 7B). Real-time PCR analysis further confirmed the histological findings, demonstrating a significant up-regulation of fibrogenic markers (Tgf-β1, Col1α1, and α-Sma) in the sham-treated group (P < 0.001 vs. the control group). In contrast, the expression of these markers decreased in the MSC-treated groups (P < 0.05 vs. the sham-treated group) (Figure 7C–E), indicating the potential alleviation of HSC activity after MSC therapy. Taken together, these histomorphological and molecular findings suggest that both AT-MSCs and UC-MSCs were similarly effective in restoring the histological characteristics of fibrotic liver tissue.
Figure 7.
Expression of fibrogenic markers in rat liver tissues (A) Immunohistochemical staining: Representative micrographs show the α-SMA expression in liver paraffin-embedded sections (200x magnification). (B) Bar graph compares α-SMA-stained areas quantified by Image J. (C–E) Semi-quantitative PCR: relative mRNA levels of Tgf-β1, Col1α1, and α-Sma genes normalized to β-Actin and expressed as fold change versus the control. Data are expressed as mean ± SD (n = 6 rats per group). ∗P < 0.05, ∗∗P < 0.01, ∗∗∗P < 0.001. α-SMA, alpha smooth muscle actin.
Adipose Tissue-Mesenchymal Stem Cells and Umbilical Cord-Mesenchymal Stem Cells Transplantation Improves Liver Functions
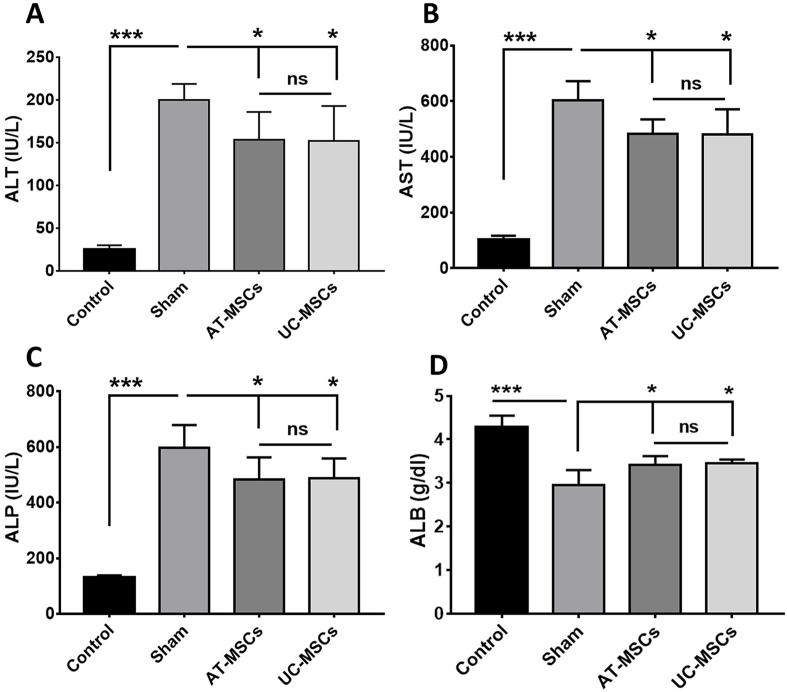
To further validate whether transplanted MSCs improve liver functions, biochemical indicators of liver injury, including hepatoenzymes (ALT, AST, and ALP), and liver function, including albumin, were assessed in serum. The results showed a significant increase in hepatoenzymes and a decrease in albumin levels in the sham-treated group (P < 0.001 vs. the control group), indicating hepatic damage after DEN intoxication (Figure 8A–D). Conversely, MSC treatment significantly restored their levels (P < 0.05 vs. sham treatment), indicating improvement in function after MSC transplantation. However, consistent with the histological findings, no discernible difference was observed between the AT-MSCs and UC-MSCs treatment groups.
Figure 8.
Liver function tests. Blood serum profiles of (A) ALT, (B) AST, (C) ALP, and (D) ALB. Data are expressed as mean ± SD (n = 6 rats per group). ∗P < 0.05, ∗∗P < 0.01, ∗∗∗P < 0.001. ALT, alanine aminotransferase; AST, aspartate aminotransferase; ALP, alkaline phosphatase; ALB, albumin.
Discussion
The shortage of organ donors and other limitations of liver transplantation have prompted researchers to investigate alternative treatments, such as MSC transplantation.26 MSCs possess therapeutic properties, including antiinflammatory, antioxidative, and immunomodulatory effects, making them attractive candidates for treating liver diseases.27,28 Despite the effectiveness of MSC transplantation demonstrated in preclinical and clinical studies,29 there is still no consensus on the optimal cell source, transplantation route, frequency, or dosage of MSCs for treating liver diseases.30 Addressing these gaps, this study focuses on determining the optimal cell source, as it is a prerequisite step in MSC transplantation studies. Although bone marrow is commonly used as a source of MSCs, the limitations presented by its invasive isolation procedure and lower cell yield have led to investigations of other readily available sources of MSCs. This study compares two such sources, adipose and cord tissues, for their ex vivo growth characteristics and their anti-fibrotic potential in liver fibrosis models, and is the first to do so to the best of our knowledge.
Several techniques have been used to isolate MSCs from adipose and cord tissues, utilizing both enzymatic (collagenase or trypsin) and nonenzymatic (manual dissociation or density gradient centrifugation) methods. Notably, enzymatic methods have demonstrated higher cellular yields compared to non-enzymatic approaches.31,32 Therefore, we utilized a collagenase-based digestion approach to isolate AT-MSCs from adipose (lipoaspirates) and UC-MSCs from cord (Wharton's jelly) tissues. These MSCs were subsequently expanded up to P6 since early to mid-passage numbers (P2–P6) are generally considered optimal for MSCs employed in clinical investigations.33 Nevertheless, ex vivo MSCs expansion is critical for the development of MSCs-based therapeutics; it may affect the MSCs stemness, transcriptomic, and secretome profiles, resulting in a decrease in viable cell yield and therapeutic characteristics.34 Therefore, it is crucial to study these parameters meticulously to optimize their clinical utility.
Monitoring cell morphology is a vital approach for assessing the state of propagated MSCs. In this study, no morphological changes were seen in AT-MSCs and UC-MSCs from P1 to P6, which suggests that their state remains stable from the early to mid-passages. These findings not only support the effectiveness of the utilized culture conditions in preserving the state and properties of the MSCs but also align with previous studies highlighting the consistent maintenance of morphology in AT-MSCs and UC-MSCs across passages.35,36
Previous research has highlighted varying proliferation capacities among MSCs sourced from different origins. While past studies suggested that UC-MSCs proliferate faster than AT-MSCs, the current research contradicts this by revealing that AT-MSCs, in fact, display a greater proliferation rate than UC-MSCs.22,37, 38, 39 To address this discrepancy, several variables affecting MSC proliferation rates, including donor characteristics (age, gender, and tissue source), culture protocols (enzymatic or nonenzymatic), and culture conditions (digestion time, seeding density, and medium composition), must be considered.40 Notably, it has been demonstrated that enzymatic methods used for UC-MSCs can reduce their proliferative capability.41 Therefore, it is crucial to acknowledge that the collagenase-based digestion technique employed in the current study may have had an impact on the observed proliferation rates of UC-MSCs. Nonetheless, in order to gain a definitive understanding, further studies comparing these MSCs under different culturale protocols are necessary. Interestingly, the present findings reveal an intriguing pattern: initially, AT-MSCs exhibit a higher proliferation rate than UC-MSCs. However, this capability diminishes in later passages. This is evidenced by lower colony numbers and increased cellular senescence activity in P6 AT-MSCs compared to UC-MSCs, which aligns with previous studies.42 These findings demonstrate that AT-MSCs yield more cells in less time than UC-MSCs, but the ability of AT-MSCs to self-renew decreases with consecutive passage, most likely due to cellular maturation or senescence. In contrast, UC-MSCs can retain their self-renewal ability even at higher passage numbers.
Furthermore, the study investigated the impact of prolonged cultivation on AT-MSCs and UC-MSCs, specifically focusing on their adherence to the minimal criteria established by the International Society for Cellular Therapy (ISCT) for categorizing cells as mesenchymal.43 Negative surface markers CD34 and CD45 were chosen to distinguish MSCs from hematopoietic stem cells; conversely, positive surface markers CD90 and CD105 were selected for their association with the mesenchymal lineage. Our findings indicate that both types of MSCs exhibited mesenchymal surface markers without expressing hematopoietic stem cell markers, aligning with prior research that reported the association of high CD90 and CD105 expression with the undifferentiated status of MSCs in vitro.44,45 Additionally, we investigated the expression of pluripotency factors (NANOG, OCT4, and SOX2) in the studied MSCs. Previous reports have highlighted the pivotal role of these factors in the self-renewal and differentiation regulation of MSCs, establishing them as important markers of MSCs.46 Herein, despite slight differences, both types of MSCs retained the capacity to express these pluripotent genes, in accordance with previous research.47 As MSCs can differentiate into various cell types, both AT-MSCs and UC-MSCs demonstrated their ability to differentiate into adipocytes, osteocytes, and chondrocytes. Collectively, our findings suggest that the cells isolated from adipose tissue and the umbilical cord are MSCs and further suggest the feasibility of cultivating both types of MSCs up to P6 while preserving their stemness, crucial for clinical scenarios requiring multiple cell infusions, as they meet the stem cell criteria defined by the ISCT.
The second and final objective of our study was to examine the antifibrotic effect of AT-MSCs and UC-MSCs in rat models of DEN-induced liver fibrosis. By employing DEN, we aimed to develop a rat model mirroring the transition from chronic inflammation to fibrosis or cirrhosis, a key factor in most cases of human Hepatocellular carcinoma (HCC). Among the various rodent models for liver fibrosis, chemical injury-induced models are widely accepted, involving drug-induced hepatocyte damage, toxic metabolite generation, persistent liver injury, and subsequent fibrosis. Our focus was on the DEN-induced liver fibrosis model, which closely mimics human liver fibrosis pathophysiology.48 Furthermore, acknowledging the variability in fibrosis induction across animal species and genders, past studies,49 including our own,25 meticulously characterized the DEN-induced liver fibrosis model by employing male SD rats. Herein, we ensured robustness by replicating the established fibrotic conditions, such as hepatocyte degeneration, inflammatory cell infiltration, and collagen fiber deposition, using identical conditions. This underscores the effective induction of liver fibrosis by DEN, reinforcing the model's reliability and our study's findings.
Following MSC treatment, both AT-MSCs and UC-MSCs groups showed a considerable decrease in fibrotic regions, likely due to paracrine effects as demonstrated in earlier studies.50,51 Although we observed a lower level of collagen staining in the AT-MSCs group than in the UC-MSCs group, the statistical analysis did not reveal a substantial difference between the two groups. This observation demonstrates that both AT-MSCs and UC-MSCs exhibit comparable therapeutic effects in mitigating the pathological features, suggesting their potential as promising treatments for liver fibrosis.
After liver injury, resident liver macrophages release pro-inflammatory cytokines such as TNF-α, IL-1, and IL-6, attracting immune cells to the injury site and fostering an environment conducive to HSC activation. The direct interaction between immune cells and quiescent HSCs prompts their transformation into myofibroblast-like cells expressing α-Sma, the primary contributors to fibrosis, through the synthesis of ECM proteins, including Col1α1.52 Additionally, activated HSCs release fibrogenic factors, including TGF-β1, critical in liver fibrosis, by activating quiescent HSCs.53 MSCs have been observed to create an immunologically-tolerant milieu in the liver by promoting antiinflammatory macrophage polarization.54 In our study, both AT-MSCs and UC-MSCs suppressed key fibrosis-related genes, including Tgf-β1, α-SMA, and Col1α1, indicating attenuation of liver fibrosis by inhibiting HSC activation following transplantation. While the inactivation of HSCs suggests an immune modulatory effect of MSCs, further detailed investigations in this aspect will be crucial for a comprehensive understanding.
The liver performs various metabolic functions, including the synthesis, detoxification, and elimination of compounds. A liver function test, including measurement of ALB and hepatoenzymes (ALT, AST, and ALP) in blood serum, is used to monitor liver or liver cell (hepatocyte) injury. In this study, DEN administration caused significant liver injury, as evidenced by increased hepatoenzymes and decreased ALB levels in serum. Both AT-MSCs and UC-MSCs improved liver function indices, specifically increasing albumin levels and decreasing hepatoenzyme levels, which suggests they may regenerate injured hepatocytes and provide protection. This aligns with previous studies and could be related to the presence of MSCs in the liver parenchyma, although the study did not explore how MSCs differentiate into liver cells or protect them.55,56 Notably, the liver function index was similar in both the AT-MSCs and UC-MSCs treatment groups, consistent with the histological and molecular findings.
In the pursuit of assessing the antifibrotic potential of ex vivo expanded human-derived MSCs through transplantation into a rat model of liver fibrosis, the effectiveness of this strategy critically relies on the immune privilege of MSCs. The elimination of MSCs could potentially compromise the robustness of the evidence presented, thereby challenging the rationale for clinical trials. Encouragingly, our study demonstrates the presence of transplanted MSCs even four weeks posttransplantation, aligning with previous studies.50,51 This sustained engraftment likely stems from intrinsic MSC mechanisms, including hypoimmunogenicity, modulation of T-cell functions, and the establishment of an immunosuppressive microenvironment.57 However, it's crucial to underscore that translating results from animal studies to clinical applications in humans demands careful consideration of species-specific differences and potential challenges.
The study concluded that both AT-MSCs and UC-MSCs hold promise for cellular therapy in the treatment of liver fibrosis. Notably, adipose cultures yield a greater number of MSCs in a shorter period of time during the early to mid-passages, making them a practical choice for immediate single-dose infusion, which is often the case in clinical scenarios. Conversely, UC-MSCs can be effectively cultured in long-term passages, making them more suitable for cases requiring multiple infusions over an extended period. However, it is important to acknowledge that the study had certain limitations. For instance, it did not investigate the exosome profiles of MSCs in vitro or delve into the mechanisms of action of MSCs in vivo. Therefore, further research is warranted to enhance our understanding of AT- or UC-MSC therapy in liver fibrosis. Furthermore, while MSC transplantation is deemed safe and promising for liver diseases, caution is essential due to concerns like ectopic cell differentiation, tumorigenicity, and immune responses. To ensure safety and efficacy, additional screening, including comprehensive MSC characterization, genetic stability assessment, and Human Leukocyte Antigens (HLA) typing, is crucial before proceeding to clinical trials. Addressing these considerations is imperative for optimizing the therapeutic potential of MSC transplantation in the treatment of liver diseases.
Credit Authorship Contribution Statement
H.G. and A.M. conceived the idea and reviewed the manuscript; M.U. and H.B performed the in vitro experiments; H.G and M.A performed the in vivo experiments; S.T and M.T.B analyzed and interpreted the data; H.G drafted the manuscript, prepared the tables and figures; A.M revised the manuscript; S.R edited and finalized the manuscript.
All authors have read the journal's authorship agreement and the manuscript has been reviewed by and approved by all named authors.
Conflicts of interest
All authors have none to declare.
Acknowledgments
The authors would like to express their gratitude to Dr. Amin Yousaf for providing lipoaspirates from Jinnah Hospital, Lahore, Pakistan, and to Prof. Dr. Shamsa Humayon for providing cord tissue samples from Sir Ganga Ram Hospital, Lahore. Notably, the authors are deeply indebted to the patient, who gave their written consent for research use of their tissue material.
Funding
This study was conducted with funds provided by our institute, National Centre of Excellence in Molecular Biology, University of the Punjab, Lahore, Pakistan.
Ethical statement
The patient provided written informed consent for the use of their tissue material in this research study. Additionally, all procedures involving the handling of animals in this study received prior approval from the Institutional Animal Ethics Committee at the National Centre of Excellence in Molecular Biology, University of the Punjab, Pakistan.
Contributor Information
Azra Mehmood, Email: azra_mehmood@hotmail.com.
Sheikh Riazuddin, Email: riazuddin@aimrc.org.
References
- 1.Dhar D., Baglieri J., Kisseleva T., Brenner D.A. Mechanisms of liver fibrosis and its role in liver cancer. Exp Biol Med. 2020;245:96–108. doi: 10.1177/1535370219898141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Khanam A., Saleeb P.G., Kottilil S. Pathophysiology and treatment options for hepatic fibrosis: can it be completely cured? Cells. 2021;10:1097. doi: 10.3390/cells10051097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Moriya K., Yoshikawa M., Ouji Y., et al. Embryonic stem cells reduce liver fibrosis in CCl4-treated mice. Int J Exp Pathol. 2008;89:401–409. doi: 10.1111/j.1365-2613.2008.00607.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Pouyanfard S., Meshgin N., Cruz L.S., et al. Human induced pluripotent stem cell-derived macrophages ameliorate liver fibrosis. Stem Cell. 2021;39:1701–1717. doi: 10.1002/stem.3449. [DOI] [PubMed] [Google Scholar]
- 5.Zhan Y., Wang Y., Wei L., et al., editors. Transplantation Proceedings. Elsevier; 2006. Differentiation of hematopoietic stem cells into hepatocytes in liver fibrosis in rats. [DOI] [PubMed] [Google Scholar]
- 6.Huang B., Cheng X., Wang H., et al. Mesenchymal stem cells and their secreted molecules predominantly ameliorate fulminant hepatic failure and chronic liver fibrosis in mice respectively. J Transl Med. 2016;14:1–12. doi: 10.1186/s12967-016-0792-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Liu Y., Dong Y., Wu X., Xu X., Niu J. The assessment of mesenchymal stem cells therapy in acute on chronic liver failure and chronic liver disease: a systematic review and meta-analysis of randomized controlled clinical trials. Stem Cell Res Ther. 2022;13:1–16. doi: 10.1186/s13287-022-02882-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Lee C., Kim M., Han J., Yoon M., Jung Y. Mesenchymal stem cells influence activation of hepatic stellate cells, and constitute a promising therapy for liver fibrosis. Biomedicines. 2021;9:1598. doi: 10.3390/biomedicines9111598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Du W.J., Chi Y., Yang Z.X., et al. Heterogeneity of proangiogenic features in mesenchymal stem cells derived from bone marrow, adipose tissue, umbilical cord, and placenta. Stem Cell Res Ther. 2016;7:1–11. doi: 10.1186/s13287-016-0418-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Baxter M.A., Wynn R.F., Jowitt S.N., Wraith J.E., Fairbairn L.J., Bellantuono I. Study of telomere length reveals rapid aging of human marrow stromal cells following in vitro expansion. Stem Cell. 2004;22:675–682. doi: 10.1634/stemcells.22-5-675. [DOI] [PubMed] [Google Scholar]
- 11.Stolzing A., Jones E., Mcgonagle D., Scutt A. Age-related changes in human bone marrow-derived mesenchymal stem cells: consequences for cell therapies. Mech Ageing Develop. 2008;129:163–173. doi: 10.1016/j.mad.2007.12.002. [DOI] [PubMed] [Google Scholar]
- 12.Hu C., Zhao L., Li L. Current understanding of adipose-derived mesenchymal stem cell-based therapies in liver diseases. Stem Cell Res Ther. 2019;10:1–13. doi: 10.1186/s13287-019-1310-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Xie Q., Liu R., Jiang J., et al. What is the impact of human umbilical cord mesenchymal stem cell transplantation on clinical treatment? Stem Cell Res Ther. 2020;11:1–13. doi: 10.1186/s13287-020-02011-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Yu F., Ji S., Su L., et al. Adipose-derived mesenchymal stem cells inhibit activation of hepatic stellate cells in vitro and ameliorate rat liver fibrosis in vivo. J Formos Med Assoc. 2015;114:130–138. doi: 10.1016/j.jfma.2012.12.002. [DOI] [PubMed] [Google Scholar]
- 15.Zhang L.-T., Peng X.-B., Fang X.-Q., Li J.-F., Chen H., Mao X.-R. Human umbilical cord mesenchymal stem cells inhibit proliferation of hepatic stellate cells in vitro. Int J Mol Med. 2018;41:2545–2552. doi: 10.3892/ijmm.2018.3500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Yoshizumi Y., Yukawa H., Iwaki R., et al. Immunomodulatory effects of adipose tissue-derived stem cells on concanavalin A-induced acute liver injury in mice. Cell Med. 2017;9:21–33. doi: 10.3727/215517916X693159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Yun J.-W., Ahn J.H., Kwon E., et al. Human umbilical cord-derived mesenchymal stem cells in acute liver injury: hepatoprotective efficacy, subchronic toxicity, tumorigenicity, and biodistribution. Regul Toxicol Pharmacol. 2016;81:437–447. doi: 10.1016/j.yrtph.2016.09.029. [DOI] [PubMed] [Google Scholar]
- 18.Salomone F., Barbagallo I., Puzzo L., Piazza C., Volti G.L. Efficacy of adipose tissue-mesenchymal stem cell transplantation in rats with acetaminophen liver injury. Stem Cell Res. 2013;11:1037–1044. doi: 10.1016/j.scr.2013.07.003. [DOI] [PubMed] [Google Scholar]
- 19.Yao J., Zheng J., Cai J., et al. Extracellular vesicles derived from human umbilical cord mesenchymal stem cells alleviate rat hepatic ischemia-reperfusion injury by suppressing oxidative stress and neutrophil inflammatory response. Faseb J. 2019;33:1695–1710. doi: 10.1096/fj.201800131RR. [DOI] [PubMed] [Google Scholar]
- 20.Zhu M., Heydarkhan-Hagvall S., Hedrick M., Benhaim P., Zuk P. Manual isolation of adipose-derived stem cells from human lipoaspirates. JoVE. 2013 doi: 10.3791/50585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Wajid N., Mehmood A., Bhatti F-u-R, Khan S.N., Riazuddin S. Lovastatin protects chondrocytes derived from Wharton's jelly of human cord against hydrogen-peroxide-induced in vitro injury. Cell Tissue Res. 2013;351:433–443. doi: 10.1007/s00441-012-1540-3. [DOI] [PubMed] [Google Scholar]
- 22.Choudhery M.S., Badowski M., Muise A., Harris D.T. Comparison of human mesenchymal stem cells derived from adipose and cord tissue. Cytotherapy. 2013;15:330–343. doi: 10.1016/j.jcyt.2012.11.010. [DOI] [PubMed] [Google Scholar]
- 23.Ghufran H., Azam M., Mehmood A., et al. Tumoricidal effects of unprimed and curcumin-primed adipose-derived stem cells on human hepatoma HepG2 cells under oxidative conditions. Tissue Cell. 2022;79 doi: 10.1016/j.tice.2022.101968. [DOI] [PubMed] [Google Scholar]
- 24.Azam M., Ghufran H., Butt H., et al. Curcumin preconditioning enhances the efficacy of adipose-derived mesenchymal stem cells to accelerate healing of burn wounds. Burns & Trauma. 2021;9 doi: 10.1093/burnst/tkab021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ghufran H., Azam M., Mehmood A., Butt H., Riazuddin S. Standardization of diethylnitrosamine-induced hepatocellular carcinoma rat model with time based molecular assessment. Exp Mol Pathol. 2021;123 doi: 10.1016/j.yexmp.2021.104715. [DOI] [PubMed] [Google Scholar]
- 26.Cao Y., Ji C., Lu L. Mesenchymal stem cell therapy for liver fibrosis/cirrhosis. Ann Transl Med. 2020;8 doi: 10.21037/atm.2020.02.119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Lee S.M., Lee S.D., Wang S.Z., et al. Effect of mesenchymal stem cell in liver regeneration and clinical applications. Hepatoma Res. 2021;7:53. [Google Scholar]
- 28.Zhu M., Hua T., Ouyang T., Qian H., Yu B. Applications of mesenchymal stem cells in liver fibrosis: novel strategies, mechanisms, and clinical practice. Stem Cell Int. 2021;2021 doi: 10.1155/2021/6546780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Liu P., Mao Y., Xie Y., Wei J., Yao J. Stem cells for treatment of liver fibrosis/cirrhosis: clinical progress and therapeutic potential. Stem Cell Res Ther. 2022;13:1–20. doi: 10.1186/s13287-022-03041-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Kang S.H., Kim M.Y., Eom Y.W., Baik S.K. Mesenchymal stem cells for the treatment of liver disease: present and perspectives. Gut Liver. 2020;14:306. doi: 10.5009/gnl18412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Winnier G.E., Valenzuela N., Peters-Hall J., Kellner J., Alt C., Alt E.U. Isolation of adipose tissue derived regenerative cells from human subcutaneous tissue with or without the use of an enzymatic reagent. PLoS One. 2019;14 doi: 10.1371/journal.pone.0221457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Salehinejad P., Alitheen N.B., Ali A.M., et al. Comparison of different methods for the isolation of mesenchymal stem cells from human umbilical cord Wharton's jelly. In Vitro Cell Dev Biol Anim. 2012;48:75–83. doi: 10.1007/s11626-011-9480-x. [DOI] [PubMed] [Google Scholar]
- 33.Binato R., de Souza Fernandez T., Lazzarotto-Silva C., et al. Stability of human mesenchymal stem cells during in vitro culture: considerations for cell therapy. Cell Prolif. 2013;46:10–22. doi: 10.1111/cpr.12002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Yang Y.-H.K., Ogando C.R., Wang See C., Chang T.-Y., Barabino G.A. Changes in phenotype and differentiation potential of human mesenchymal stem cells aging in vitro. Stem Cell Res Ther. 2018;9:1–14. doi: 10.1186/s13287-018-0876-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Widowati W., Noverina R., Ayuningtyas W., et al. Proliferation, characterization and differentiation potency of adipose tissue-derived mesenchymal stem cells (AT-MSCs) cultured in fresh frozen and non-fresh frozen plasma. Int J Molecular Cell Med. 2019;8:283. doi: 10.22088/IJMCM.BUMS.8.4.283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Zhao Q., Zhang L., Wei Y., et al. Systematic comparison of hUC-MSCs at various passages reveals the variations of signatures and therapeutic effect on acute graft-versus-host disease. Stem Cell Res Ther. 2019;10:1–14. doi: 10.1186/s13287-019-1478-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Kim J.-H., Jo C.H., Kim H.-R., Hwang Y-i. Comparison of immunological characteristics of mesenchymal stem cells from the periodontal ligament, umbilical cord, and adipose tissue. Stem Cell Int. 2018;2018 doi: 10.1155/2018/8429042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Chen J.-Y., Mou X.-Z., Du X.-C., Xiang C. Comparative analysis of biological characteristics of adult mesenchymal stem cells with different tissue origins. Asian Pac J Tropical Med. 2015;8:739–746. doi: 10.1016/j.apjtm.2015.07.022. [DOI] [PubMed] [Google Scholar]
- 39.Trivanović D., Jauković A., Popović B., et al. Mesenchymal stem cells of different origin: comparative evaluation of proliferative capacity, telomere length and pluripotency marker expression. Life Sci. 2015;141:61–73. doi: 10.1016/j.lfs.2015.09.019. [DOI] [PubMed] [Google Scholar]
- 40.Mastrolia I., Foppiani E.M., Murgia A., et al. Challenges in clinical development of mesenchymal stromal/stem cells: concise review. Stem Cells Translation Med. 2019;8:1135–1148. doi: 10.1002/sctm.19-0044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Hendijani F. Explant culture: an advantageous method for isolation of mesenchymal stem cells from human tissues. Cell Prolif. 2017;50 doi: 10.1111/cpr.12334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Jeske R., Yuan X., Fu Q., Bunnell B.A., Logan T.M., Li Y. In vitro culture expansion shifts the immune phenotype of human adipose-derived mesenchymal stem cells. Front Immunol. 2021;12 doi: 10.3389/fimmu.2021.621744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Dominici M., Le Blanc K., Mueller I., et al. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy. 2006;8:315–317. doi: 10.1080/14653240600855905. [DOI] [PubMed] [Google Scholar]
- 44.Moraes D.A., Sibov T.T., Pavon L.F., et al. A reduction in CD90 (THY-1) expression results in increased differentiation of mesenchymal stromal cells. Stem Cell Res Ther. 2016;7:1–14. doi: 10.1186/s13287-016-0359-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Jin H.J., Park S.K., Oh W., Yang Y.S., Kim S.W., Choi S.J. Down-regulation of CD105 is associated with multi-lineage differentiation in human umbilical cord blood-derived mesenchymal stem cells. Biochem Biophys Res Commun. 2009;381:676–681. doi: 10.1016/j.bbrc.2009.02.118. [DOI] [PubMed] [Google Scholar]
- 46.Tsai C.-C., Hung S.-C. Functional roles of pluripotency transcription factors in mesenchymal stem cells. Cell Cycle. 2012;11:3711–3712. doi: 10.4161/cc.22048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Secunda R., Vennila R., Mohanashankar A., Rajasundari M., Jeswanth S., Surendran R. Isolation, expansion and characterisation of mesenchymal stem cells from human bone marrow, adipose tissue, umbilical cord blood and matrix: a comparative study. Cytotechnology. 2015;67:793–807. doi: 10.1007/s10616-014-9718-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Wu S., Wang X., Xing W., et al. An update on animal models of liver fibrosis. Front Med. 2023;10 doi: 10.3389/fmed.2023.1160053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Ding Y-f, Wu Z-h, Wei Y-j, Shu L., Peng Y-r. Hepatic inflammation-fibrosis-cancer axis in the rat hepatocellular carcinoma induced by diethylnitrosamine. J Cancer Res Clin Oncol. 2017;143:821–834. doi: 10.1007/s00432-017-2364-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Nomura M., George J., Hashizume C., et al. Surgical implantation of human adipose derived stem cells attenuates experimentally induced hepatic fibrosis in rats. Mol Med. 2022;28:1–18. doi: 10.1186/s10020-022-00566-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Chai N.-L., Zhang X.-B., Chen S.-W., Fan K.-X., Linghu E.-Q. Umbilical cord-derived mesenchymal stem cells alleviate liver fibrosis in rats. World J Gastroenterol. 2016;22:6036. doi: 10.3748/wjg.v22.i26.6036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Liu P., Qian Y., Liu X., et al. Immunomodulatory role of mesenchymal stem cell therapy in liver fibrosis. Front Immunol. 2023;13 doi: 10.3389/fimmu.2022.1096402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Fallowfield J.A. Therapeutic targets in liver fibrosis. Am J Physiol Gastrointest Liver Physiol. 2011;300:G709–G715. doi: 10.1152/ajpgi.00451.2010. [DOI] [PubMed] [Google Scholar]
- 54.Tian S., Zhou X., Zhang M., et al. Mesenchymal stem cell-derived exosomes protect against liver fibrosis via delivering miR-148a to target KLF6/STAT3 pathway in macrophages. Stem Cell Res Ther. 2022;13:1–20. doi: 10.1186/s13287-022-03010-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Baig M.T., Ghufran H., Mehmood A., Azam M., Humayun S., Riazuddin S. Vitamin E pretreated Wharton's jelly-derived mesenchymal stem cells attenuate CCl4-induced hepatocyte injury in vitro and liver fibrosis in vivo. Biochem Pharmacol. 2021;186 doi: 10.1016/j.bcp.2021.114480. [DOI] [PubMed] [Google Scholar]
- 56.Zhang G.-Z., Sun H.-C., Zheng L.-B., Guo J.-B., Zhang X.-L. In vivo hepatic differentiation potential of human umbilical cord-derived mesenchymal stem cells: therapeutic effect on liver fibrosis/cirrhosis. World J Gastroenterol. 2017;23:8152. doi: 10.3748/wjg.v23.i46.8152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Li J., Ezzelarab M.B., Cooper D.K. Do mesenchymal stem cells function across species barriers? Relevance for xenotransplantation. Xenotransplantation. 2012;19:273–285. doi: 10.1111/xen.12000. [DOI] [PMC free article] [PubMed] [Google Scholar]