Abstract
Background
Dolutegravir (DTG), a new antiretroviral drug, is being integrated into antiretroviral regimens for people with human immunodeficiency virus (PWH) in Ghana. There is little evidence of the effect of DTG on blood pressure (BP) levels in sub-Saharan Africa, especially West Africa. Our aim was to assess the incidence and predictors of hypertension (HTN) among PWH initiated on a DTG-based antiretroviral regimen in Ghana.
Methods
An observational multicenter longitudinal study was conducted among PWH in Ghana from 2020 to 2022. BPs of nonhypertensive patients with BP ≤120/80 mm Hg at baseline were measured at 3, 6, 12, and 18 months post–DTG initiation. The primary outcome of the study was incidence of HTN, defined as BP ≥140/90 mm Hg. Kaplan-Meier estimator was used to estimate risk of developing HTN. Cox proportional hazards model with robust standard errors was used to estimate hazard ratios (HRs).
Results
HTN prevalence among PWH screened was 37.3% (1366/3664). The incidence of de novo HTN among nonhypertensive PWH at 72 weeks was 598.4 per 1000 person-years (PY) (95% confidence interval [CI], 559.2–640.3) with incidence proportion of 59.90 (95% CI, 57.30–62.44). A quarter of those with de novo HTN developed it by month 6. Obesity (adjusted HR [aHR], 1.27 [95% CI, 1.05–1.54]), abnormal serum urea (aHR, 1.53 [95% CI, 1.27–1.85]), and low high-density lipoprotein (aHR, 1.45 [95% CI, 1.22–1.72]) were risk factors for HTN.
Conclusions
Incidence of HTN was high among PWH on DTG. There is a need to monitor BP for HTN in adult PWH as well as traditional risk factors to reduce the burden of HTN and its complications.
Keywords: antiretroviral therapy, dolutegravir, HIV, hypertension, incidence
Over half of adults with HIV initiated on a dolutegravir-based regimen developed hypertension, with a quarter of them developing it 6 months post–dolutegravir initiation. Incidence was higher in treatment-naive persons. Obesity and low high-density lipoprotein were risk factors for developing hypertension.
Global human immunodeficiency virus (HIV) statistics reported 76% of people with HIV (PWH) accessing antiretroviral therapy (ART) in 2022 and an increase in PWH accessing ART from 7.8 million in 2010 to 29.8 million in 2022 [1]. In sub-Saharan Africa, 25.6 million PWH were accessing ART in 2022 with 81% of them in Eastern and Southern Africa [1]. The scale-up of ART has improved management of PWH and reduced AIDS-related mortality, with a resultant increase in life expectancy [2–4].
This increased life expectancy among PWH has transformed HIV disease into a chronic condition. Thus, treatment focus has gradually shifted toward management of chronic HIV infection, ART-associated complications, and comorbidities, especially noncommunicable diseases [5]. Comorbidities of noninfectious origin accounted for >50% of mortality among PWH, with cancer and cardiovascular disease (CVD) being the commonest [6]. CVDs are major sources of morbidity and mortality among PWH [2, 3]. The burden of HIV-associated CVDs has tripled globally over the past 20 years with the greatest impact in Asia Pacific and sub-Saharan Africa (SSA) [4]. Studies have also reported that PWH are twice as likely to develop CVDs compared with HIV-negative persons [4, 7]. With SSA accounting for almost two-thirds of PWH worldwide, it is confronted with a double burden of hypertension (HTN) and HIV [8]. The HIV infection itself has been reported to be associated with a higher risk for CVD, and a pooled prevalence shows that 19% of PWH in SSA are estimated to have HTN [8]. HTN prevalence in PWH in SSA has varied widely in literature, with crude prevalence of 17% among ART-naive PWH in Nigeria [9] to 43% in PWH on ART in Tanzania [10]. A meta-analysis from South Africa also reported a prevalence of 25.5% among 123 951 PWH from 22 studies [11]. The prevalence of HTN, both in the general population and among PWH, is likely to be underrated in SSA due to complex factors such as diverse diagnostic criteria, poor surveillance, poor health-seeking behavior, and inadequate resources for screening for HTN, among others [11–13].
HTN is more common among PWH, with prevalence of 35% compared to 30% among the HIV-uninfected population [2, 14]. Traditional risk factors such as age, obesity, family history, diabetes, smoking, dyslipidemia, and a sedentary lifestyle contribute to the development of HTN [15, 16]. In addition for PWH, the HIV infection itself contributes to the pathogenesis of HTN and CVD through viral replication, decreased CD4+ T-cell count, chronic systemic inflammation and endothelial dysfunction, and lipodystrophy [17, 18]. Exposure to ART has also been significantly associated with increased risk of HTN among PWH [19–21].
In Ghana, the HIV prevalence as at 2022 was 1.66%, with the number of deaths averted due to ART increasing by 38% from 2020 to 2022 [22]. Of an estimated 354 927 PWH in 2022, 68% were female. Since 2016, Ghana has implemented the “treat all” approach in conjunction with initiation of ART on the same day as an HIV diagnosis [22]. Ghana had >715 ART units across the country as at December 2022, increasing access to ART and improving lives of PWH. The prevalence of comorbidities among PWH in a study in northern Ghana was 30.3%, with hepatitis B infection being the commonest (20.3%) comorbidity [23]. The prevalence of HTN among PWH in Ghana has been reported to be 30.8% [24]. The HTN prevalence among PWH on combination ART was 36.9% while that for PWH who were ART-naive was 23.4% [24]. Globally, the recommended ART of choice as first line in most countries, including Ghana, is a dolutegravir (DTG)–based regimen due to improved viral suppression, less resistance, and fewer side effects [21]. Ghana rolled out the DTG-based regimen as a preferred choice for PWH since 2019 [25]. With the increase in HTN prevalence reported among PWH on combination ART compared to ART-naive PWH [24], it is important to monitor the blood pressure (BP) variations or trends among PWH initiated on DTG.
There are limited studies on the prospective evaluation of the association between DTG and HTN within the in West African subregion. We carried out a prospective study to determine the prevalence, incidence, and predictors of HTN among a cohort of PWH initiated on DTG in Ghana as part of a larger prospective study.
METHODS
Study Design and Setting
A multicenter longitudinal study was conducted among PWH recruited from 5 ART facilities from 4 regions in Ghana from September 2020 to August 2022. The facilities were the Korle Bu Teaching Hospital (Greater Accra region), St Martin's de Porres hospital (Eastern region), Atua Government hospital (Eastern region), Kumasi South hospital (Ashanti region), and Kwesimintsim hospital (Western region). These facilities are designated as high-HIV-burden facilities in the regions in Ghana with a national spread and also representing the various layers of health service provision. Ghana currently has 715 ART centers nationwide where antiretroviral drugs can be accessed for PWH. The 4 selected regions have 37% of ART centers (Greater Accra region: 97 ART centers; Eastern region: 48 ART centers; Ashanti region: 90 ART centers; and Western region: 80 ART centers).
Study Participants
All adult DTG-naive PWH ≥18 years in active care being initiated or transitioned to DTG-based regimen who consented were recruited for the study. Acutely ill or pregnant women were excluded. PWH on antihypertensive medication were screened out of the study.
Patient ART Regimen
The DTG-based regimen was a combination of DTG and 2 nucleoside reverse transcriptase inhibitors such as tenofovir disoproxil fumarate, abacavir, lamivudine, or emtricitabine.
Data Collection
Baseline data on demographic, anthropometric, and clinical characteristics were collected electronically by trained research assistants after screening, consenting, and enrollment. Clinical assessments then followed with anthropometric measurements like weight (kilograms), height (meters), waist and hip circumference (centimeters), and BP (mm Hg) measurements. We measured and recorded patients’ BP using the automatic Omron HEM 7124 sphygmomanometer. Patients sat in a straight-back chair with back supported and their feet on the floor for 15 minutes [26]. The patient's arm was supported and the middle of the cuff was positioned on the patient's upper arm at the level of the right atrium (midpoint of the sternum) before BP was measured [26]. An average of 2 BP readings were taken 5 minutes apart and documented. Ten milliliters of venous blood was drawn from each patient into blood collection tubes and transported to the laboratory immediately after blood draw. The blood was analyzed for serum lipids, blood urea, electrolytes, and serum creatinine, alanine aminotransferase, and C-reactive protein. All study equipment was calibrated and standardized by the Ghana Standards Authority prior to its use in this study.
Follow-up
Each patient enrolled was followed up with repeat of the above processes at 3, 6, 12, and 18 months post–DTG initiation. Patients who missed a scheduled study visit time point but presented at the subsequent study visit time point and were still adherent on the DTG-based regimen were still included in the data analysis of that study point.
Operational Definitions
Prehypertension: All PWH who had a BP of >120/80 mm Hg to 139/89 mm Hg.
Hypertension: All PWH who had a BP of ≥140/90 mm Hg.
At-risk population: The patients who do not have the outcome of interest and initiated on DTG-based regimen.
Failure: Patients who develop the outcome of interest (HTN) during follow-up in the study.
Data Management and Analysis
Data management and analysis was conducted using Stata Statistical Software v.17 (StataCorp LLC, College Station, Texas). Incidence of HTN was the primary outcome of the study and was defined as PWH with no evidence of pre-HTN and HTN at baseline but developing BP of ≥140/90 mm Hg during follow-up in the study. BP readings were categorized as HTN or no HTN. Incidence rate of HTN was computed by dividing the total number of new cases of HTN by the total person-years (PY) at risk. The time to developing HTN was calculated using the Kaplan-Meier estimator. Survival function was assessed using the log-rank test at the various follow-up timelines. The Nelson-Aalen cumulative hazard function was provided for each specific study time point.
Association between time to development of HTN and patient characteristics was measured using the Cox proportional hazards model with robust standard errors. Hazard ratios with 95% confidence intervals (CIs) were reported.
Participants with no HTN at the end of the study or time of discontinuation were considered as censored. Missing data occurring at any time during the period of participation in the study were presumed to have no HTN. The level of significance for all statistical tests was 5%.
Patient Consent Statement
The study protocol was reviewed and approved by the Institutional Review Board of the University of Ghana College of Health Sciences (CHS:00006220), the Korle Bu Teaching Hospital (KBTH-IRB/000136/2020), and the Ghana Health Service (GHS-ERC 010/08/20). Permissions were obtained from the heads of the facilities. Written informed consent was obtained from participants. Data were collected without personal identifiers, kept confidential and secure, and were available only to the principal and co–principal investigators.
RESULTS
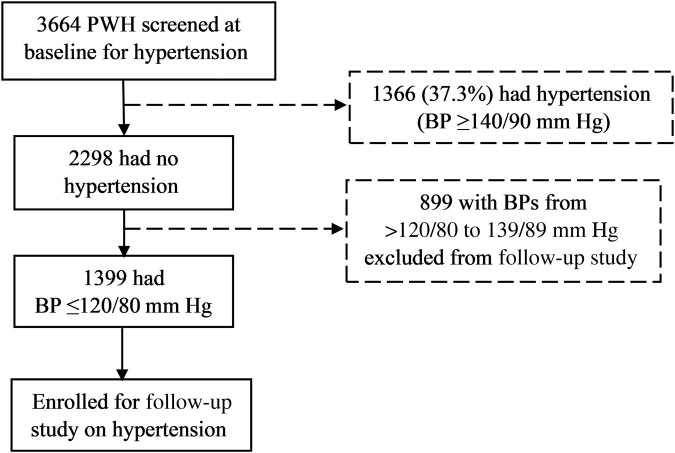
A total of 3664 PWH were screened for HTN at baseline. The prevalence of HTN was 37.28% (n = 1366) at baseline (Figure 1). All patients classified as having pre-HTN and HTN were excluded from the study. A total of 1399 patients with no prior history of HTN and with BP ≤120/80 mm Hg were recruited for the study.
Figure 1.
Flow diagram of hypertension screening among PWH initiated on dolutegravir based antiretroviral therapy in Ghana, 2020-2022. Abbreviations: BP, blood pressure; PWH, people with human immunodeficiency virus.
Baseline Demographic and Clinical Characteristics
Of the 1399 patients enrolled into the study, 78% (1093) were female (Table 1). Approximately one-quarter of the patients were aged ≥50 years. Eighty-seven percent (1215/1399) of patients were ART experienced and 50% (583/1161) of those on ART had been on ART for <5 years (Table 1).
Table 1.
Baseline Demographic and Clinical Characteristics of Nonhypertensive Patients Initiated on Dolutegravir-Based Antiretroviral Therapy in 5 High-Burden-HIV Sites in Ghana, 2020–2022
| Characteristic | Total | Hypertension | P Value | |
|---|---|---|---|---|
| No | Yes | |||
| Age (n = 1399) | .41 | |||
| <25 | 90 (6.43) | 36 (40) | 54 (60) | |
| 25–49 | 964 (68.91) | 381 (39.5) | 583 (60.5) | |
| 50–59 | 236 (16.87) | 105 (44.5) | 131 (55.5) | |
| ≥60 | 109 (7.79) | 39 (35.8) | 70 (64.2) | |
| Sex (n = 1399) | .31 | |||
| Male | 306 (21.87) | 115 (37.6) | 191 (62.4) | |
| Female | 1093 (78.13) | 446 (40.8) | 647 (59.2) | |
| Type of HIV (n = 1399) | .78 | |||
| HIV-1 | 1364 (97.50) | 545 (40) | 819 (60) | |
| HIV-2 | 7 (0.50) | 3 (42.9) | 4 (57.1) | |
| HIV-1/HIV-2 | 28 (2.00) | 13 (46.4) | 15 (53.6) | |
| Treatment status (n = 1399) | <.001 | |||
| ART naive | 184 (13.15) | 35 (19) | 149 (81) | |
| ART experienced | 1215 (86.85) | 526 (43.3) | 689 (56.7) | |
| Comorbidity (n = 1399)a | .60 | |||
| No | 1370 (97.93) | 548 (40) | 822 (60) | |
| Yes | 29 (2.07) | 13 (44.8) | 16 (55.2) | |
| Body mass index (n = 1399) | .002 | |||
| Underweight | 158 (11.29) | 49 (31) | 109 (69) | |
| Normal | 724 (51.75) | 292 (40.3) | 432 (59.7) | |
| Overweight | 316 (22.59) | 150 (47.5) | 166 (52.5) | |
| Obesity | 201 (14.37) | 70 (34.8) | 131 (65.2) | |
| Waist-to-hip ratio (n = 1385) | .33 | |||
| Normal | 807 (58.27) | 335 (41.5) | 472 (58.5) | |
| Abnormal | 578 (41.73) | 225 (38.9) | 353 (61.1) | |
| Duration on ART (n = 1161) | <.001 | |||
| <5 y | 583 (50.22) | 221 (37.9) | 362 (62.1) | |
| 5–10 y | 365 (31.44) | 185 (50.7) | 180 (49.3) | |
| >10 y | 213 (18.35) | 98 (46) | 115 (54) | |
| Duration of HIV diagnosis (n = 1399) | <.001 | |||
| <5 y | 718 (51.88) | 241 (33.6) | 477 (66.4) | |
| 5–10 y | 378 (27.31) | 178 (47.1) | 200 (52.9) | |
| >10 y | 288 (20.81) | 137 (47.6) | 151 (52.4) | |
| Serum urea (n = 1397) | <.001 | |||
| Normal | 1258 (90.05) | 526 (41.8) | 732 (58.2) | |
| Abnormal | 139 (9.95) | 35 (25.2) | 104 (74.8) | |
| Total serum cholesterol (n = 1398) | .85 | |||
| Normal | 1053 (75.32) | 424 (40.3) | 629 (59.7) | |
| Abnormal | 345 (24.68) | 137 (39.7) | 208 (60.3) | |
| LDL cholesterol (n = 1206) | .94 | |||
| Normal | 748 (62.02) | 276 (36.9) | 472 (63.1) | |
| Abnormal | 458 (37.98) | 170 (37.1) | 288 (62.9) | |
| HDL cholesterol (n = 1398) | <.001 | |||
| Normal | 360 (25.75) | 190 (52.8) | 170 (47.2) | |
| Abnormal | 1038 (74.25) | 371 (35.7) | 667 (64.3) | |
| Triglycerides (n = 1281) | <.001 | |||
| Normal | 1168 (91.18) | 500 (42.8) | 668 (57.2) | |
| Abnormal | 113 (8.82) | 30 (26.5) | 83 (73.5) | |
| Serum creatinine (n = 1399) | .63 | |||
| Normal | 1212 (86.63) | 489 (40.3) | 723 (59.7) | |
| Abnormal | 187 (13.37) | 72 (38.5) | 115 (61.5) | |
| Serum CRP (n = 1375) | .002 | |||
| Normal | 981 (71.35) | 419 (42.7) | 562 (57.3) | |
| Abnormal | 394 (28.65) | 132 (33.5) | 262 (66.5) | |
Data are presented as No. (%) unless otherwise indicated.
Abbreviations: ART, antiretroviral therapy; CRP, C-reactive protein; HDL, high-density lipoprotein; HIV, human immunodeficiency virus; LDL, low-density lipoprotein.
aComorbidity: Any existing chronic condition other than HIV (mainly type 2 diabetes, kidney disease).
Incidence Rate of Hypertension
The total number of person-time contributed was 1400.5 years. A total of 838 patients developed HTN by end of study. The median time to development of HTN was 1.5 years, and 25% of those with HTN developed it by month 6. The overall incidence of HTN was 598.4 per 1000 PY (95% CI, 559.2–640.3) (Table 2). The cumulative hazards at months 3, 6, and 12 were 75.13, 61.83, and 50.82, respectively.
Table 2.
Cumulative Incident Proportion and Rate of Hypertension by Background and Clinical Characteristics Among Persons With HIV Initiated on a Dolutegravir-Based Regimen Over an 18-Month Period in 5 High-Burden-HIV Sites in Ghana, 2020–2022
| Characteristic | At Risk | Failure | PY | % (95% CI) | IR per 1000 PY (95% CI) |
|---|---|---|---|---|---|
| Overall | 1399 | 838 | 1400.50 | 59.90 (57.30–62.44) | 598.36 (559.19–640.27) |
| Age, y | |||||
| 18–49 | 1054 | 637 | 1054.00 | 60.44 (57.45–63.35) | 604.36 (559.21–653.17) |
| ≥50 | 345 | 201 | 346.50 | 58.26 (52.98–63.36) | 580.09 (505.19–666.09) |
| Sex | |||||
| Male | 306 | 191 | 295.75 | 62.42 (56.85–67.68) | 645.82 (560.43–744.22) |
| Female | 1093 | 647 | 1104.75 | 59.19 (56.25–62.08) | 585.65 (542.22–632.56) |
| Marital status | |||||
| Never married | 353 | 211 | 349.50 | 59.77 (54.56–64.77) | 603.72 (527.52–690.93) |
| Currently married/cohabiting | 612 | 367 | 610.75 | 59.97 (56.03–63.78) | 600.90 (542.46–665.63) |
| Previously married | 434 | 260 | 440.25 | 59.91 (55.22–64.42) | 590.57 (522.98–666.90) |
| Treatment classification | |||||
| ART naive | 184 | 149 | 130.25 | 80.98 (74.65–86.02) | 1143.95 (974.26–1343.20) |
| ART experienced | 1215 | 689 | 1270.25 | 56.71 (53.90–59.47) | 542.41 (503.39–584.46) |
| Education | |||||
| None | 260 | 161 | 261.25 | 61.92 (55.87–67.63) | 616.27 (528.06–719.21) |
| Primary | 515 | 320 | 485.00 | 62.14 (57.86–66.23) | 659.79 (591.32–736.19) |
| Secondary | 516 | 301 | 536.25 | 58.33 (54.02–62.52) | 561.31 (501.35–628.44) |
| Tertiary | 108 | 56 | 118.00 | 51.85 (42.47–61.11) | 474.58 (365.22–616.67) |
| Type of RVI | |||||
| HIV-1 | 1364 | 819 | 1361.75 | 60.04 (57.42–62.62) | 601.43 (561.62–644.07) |
| HIV-2 | 7 | 4 | 7.00 | 57.14 (22.96–85.64) | 571.43 (214.47–1522.52) |
| HIV-1/HIV-2 | 28 | 15 | 31.75 | 53.57 (35.43–70.82) | 472.44 (284.82–783.66) |
| Comorbidity | |||||
| No | 1370 | 822 | 1368.50 | 60.00 (57.38–62.57) | 600.66 (560.97–643.16) |
| Yes | 29 | 16 | 32.00 | 55.17 (37.17–71.91) | 500.00 (306.32–816.15) |
| Comorbiditya | |||||
| No | 158 | 109 | 138.75 | 68.99 (61.35–75.71) | 785.59 (651.12–947.82) |
| Yes | 724 | 432 | 730.50 | 59.67 (56.05–63.19) | 591.38 (538.16–649.86) |
| BMI, kg/m2 | 316 | 166 | 344.75 | 52.53 (47.01–57.99) | 481.51 (413.56–560.62) |
| Underweight (<18.5) | 201 | 131 | 186.50 | 65.17 (58.33–71.45) | 702.41 (591.87–833.61) |
| Normal (18.5–24.9) | |||||
| Overweight (25.0–29.9) | 807 | 472 | 819.75 | 58.49 (55.05–61.85) | 575.79 (526.12–630.14) |
| Obesity (≥30.0) | 578 | 353 | 569.00 | 61.07 (57.03–64.97) | 620.39 (558.93–688.60) |
| Serum urea | |||||
| Normal | 1258 | 732 | 1289.00 | 58.19 (55.44–60.89) | 567.88 (528.20–610.55) |
| Abnormal | 139 | 104 | 109.75 | 74.82 (66.95–81.34) | 947.61 (781.92–1148.41) |
| Serum cholesterol | |||||
| Normal | 1053 | 629 | 1051.00 | 59.73 (56.74–62.66) | 598.48 (553.49–647.12) |
| Abnormal | 345 | 208 | 349.25 | 60.29 (55.03–65.33) | 595.56 (519.88–682.26) |
| HDL cholesterol | |||||
| Normal | 360 | 170 | 398.00 | 47.22 (42.11–52.39) | 427.14 (367.52–496.42) |
| Abnormal | 1038 | 667 | 1002.25 | 64.26 (61.29–67.12) | 665.50 (616.87–717.97) |
| Serum creatinine | |||||
| Normal | 1212 | 723 | 1222.25 | 59.65 (56.86–62.38) | 591.53 (549.95–636.26) |
| Abnormal | 187 | 115 | 178.25 | 61.50 (54.33–68.20) | 645.16 (537.39–774.54) |
| C-reactive protein | |||||
| Normal | 981 | 562 | 1016.00 | 57.29 (54.17–60.35) | 553.15 (509.26–600.83) |
| Abnormal | 394 | 262 | 360.25 | 66.50 (61.68–70.99) | 727.27 (644.33–820.89) |
| Duration of HIV diagnosis | |||||
| <5 y | 718 | 477 | 665.00 | 66.43 (62.89–69.80) | 717.29 (655.73–784.64) |
| 5–10 y | 378 | 200 | 406.50 | 52.91 (47.86–57.90) | 492.00 (428.33–565.14) |
| >10 y | 288 | 151 | 314.00 | 52.43 (46.65–58.15) | 480.89 (409.99–564.05) |
| Duration on previous ART | |||||
| <5 y | 583 | 362 | 575.00 | 62.09 (58.08–65.95) | 629.57 (567.94–697.88) |
| 5–10 y | 365 | 180 | 406.50 | 49.32 (44.21–54.44) | 442.80 (382.62–512.46) |
| >10 y | 213 | 115 | 233.25 | 53.99 (47.26–60.58) | 493.03 (410.68–591.90) |
Abbreviations: ART, antiretroviral therapy; BMI, body mass index; CI, confidence interval; HDL, high-density lipoprotein; HIV, human immunodeficiency virus; IR, incidence rate; PY, person-years; RVI, Retroviral Infection.
aComorbidity: Any existing chronic condition other than HIV (mainly type 2 diabetes, kidney disease).
Survival Analysis
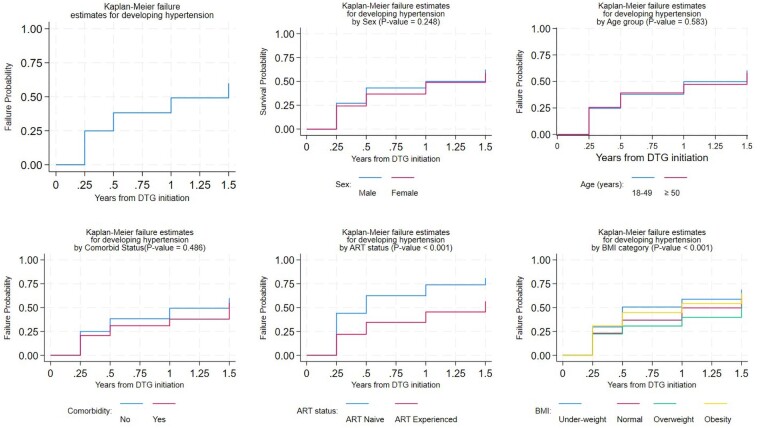
Patients who were ART experienced had a 32% lower risk of developing HTN compared to those who were ART naive (Figure 2, Table 3).
Figure 2.
Kaplan-Meier estimates for developing hypertension for selected demographic and clinical factors among people with human immunodeficiency virus on dolutegravir-based antiretroviral therapy in Ghana, 2020–2022. Abbreviations: ART, antiretroviral therapy; BMI, body mass index; DTG, dolutegravir.
Table 3.
Cox Regression Model for Time to Develop Hypertension Among Persons With HIV Initiated on a Dolutegravir-Based Regimen in Ghana, 2020–2022
| Characteristic | Unadjusted Model | Adjusted Model | ||
|---|---|---|---|---|
| HR (95% CI) | P Value | aHR (95% CI) | P Value | |
| Age, y | ||||
| 18–49 | 1 | 1 | ||
| ≥50 | 0.96 (.83–1.11) | .587 | 1.02 (.87–1.20) | .807 |
| Sex | ||||
| Male | 1.09 (.94–1.26) | .247 | 1.14 (.97–1.35) | .116 |
| Female | 1 | 1 | ||
| Treatment status | ||||
| ART naive | 1 | 1 | ||
| ART experienced | 0.53 (.45–.61) | <.001 | 0.68 (.56–.83) | <.001 |
| HIV type | ||||
| HIV-1 | 1.23 (.79–1.91) | .361 | 1.17 (.76–1.80) | .483 |
| HIV-2 | 1.18 (.43–3.21) | .745 | 1.38 (.39–4.85) | .62 |
| HIV-1/HIV-2 | 1 | 1 | ||
| Comorbiditya | ||||
| No | 1 | 1 | ||
| Yes | 0.86 (.56–1.32) | .479 | 0.82 (.54–1.24) | .346 |
| Body mass index | ||||
| Underweight | 1.27 (1.03–1.57) | .024 | 1.21 (1.00–1.46) | .046 |
| Normal | 1 | 1 | ||
| Overweight | 0.83 (.70–1.00) | .047 | 0.89 (.75–1.05) | .17 |
| Obesity | 1.16 (.96–1.42) | .128 | 1.27 (1.05–1.54) | .015 |
| Waist-to-hip ratio | ||||
| Normal | 1 | 1 | ||
| Abnormal | 1.07 (.95–1.21) | .283 | 1.02 (.88–1.17) | .829 |
| Serum urea | ||||
| Normal | 1 | 1 | ||
| Abnormal | 1.56 (1.30–1.87) | <.001 | 1.53 (1.27–1.85) | <.001 |
| Total serum cholesterol | ||||
| Normal | 1 | 1 | ||
| Abnormal | 1.00 (.87–1.15) | .994 | 1.15 (.99–1.33) | .074 |
| HDL cholesterol | ||||
| Normal | 1 | 1 | ||
| Abnormal | 1.50 (1.28–1.75) | <.001 | 1.45 (1.22–1.72) | <.001 |
| Serum creatinine | ||||
| Normal | 1 | 1 | ||
| Abnormal | 1.08 (.90–1.29) | .411 | 0.92 (.76–1.11) | .372 |
| C-reactive protein | ||||
| Normal | 1 | 1 | ||
| Abnormal | 1.27 (1.12–1.45) | <.001 | 1.19 (1.04–1.36) | .013 |
| Duration of HIV diagnosis | ||||
| <5 y | 1 | 1 | ||
| 5–10 y | 0.71 (.61–.83) | <.001 | 0.86 (.71–1.06) | .156 |
| >10 y | 0.70 (.59–.82) | <.001 | 0.99 (.72–1.37) | .959 |
| Duration on ART | 0.95 (.94–.97) | <.001 | 0.98 (.95–1.01) | .168 |
Abbreviations: aHR, adjusted odds ratio; ART, antiretroviral therapy; CI, confidence interval; HDL, high-density lipoprotein; HIV, human immunodeficiency virus.
aComorbidity: Any existing chronic condition other than HIV (mainly type 2 diabetes, kidney disease).
DISCUSSION
The current study assessed the incidence of HTN among a cohort of PWH initiated on DTG in Ghana. The prevalence of HTN was 37.2%. The overall incidence of HTN among nonhypertensive PWH was 598.4 per 1000 PY. The median time to development of HTN was 1.5 years, and one-fourth of those with HTN developed it by month 6. Obesity and having abnormal serum urea and C-reactive protein levels were associated with developing HTN.
Baseline screening of PWH in our study revealed HTN prevalence of 37.2%. Our finding was higher than the 17% reported in Nigeria [9] and 27% from a meta-analysis from South Africa [11], but was lower than the prevalence of 43% reported in Tanzania [10]. The higher prevalence from our study compared to the one from Nigeria may be due to our patients being mostly ART experienced compared to ART-naive patients from the Nigerian study as ART has been linked to development of CVDs [19–21].
Our study finding of overall incidence of de novo HTN of 598.4 per 1000 PY was higher than the incidence rates of 403 and 363 per 1000 PY at 48 and 96 weeks reported in the European Network for AIDS and Treatment 022 (NEAT-022) randomized trial of persons initiated on DTG [27]. This high incidence is significant as our participant numbers were larger in size (n = 1399) compared to the NEAT-022 trial (n = 197), which also looked at HTN prevalence in ART-experienced PWH. Another study by Brennan et al in South Africa also reported a 14.2 percentage point increase in the risk of HTN in patients exposed to DTG for 12 months compared to those that remained on efavirenz [28]. Similarly, a cross-sectional study conducted in Uganda reported a prevalence of 27.2% among patients initiated on DTG for a median duration of 28 months [16]. Results of the New Antiretroviral and Monitoring Strategies in HIV-Infected Adults in Low-Income countries (NAMSAL) study showed a significant increase in BP comparing baseline versus week 192 [29]. The incidence of HTN reported in our study is very high compared to most studies, which may be the true incidence among our study population. However, this could be an outlier and could be attributed to various reasons including possible errors arising from the methods in measurement of BP of participants. The digital sphygmomanometers were calibrated at the biomedical engineering department of the Korle Bu Teaching Hospital. We however concede that despite all efforts to standardize measurements across the 5 sites, there could have been some interobserver differences across sites accounting for high measurements [30, 31]. Second, since the study involved drawing of fasting blood samples, participants reported very early in the morning without having had breakfast. We know that BP measurements tend to be higher in the mornings than the evenings, and this could also have contributed to the high incidence. However, the Cross-sectional, Observational Study to Characterize the Transition to Dolutegravir-based Regimen in South Africa in Terms of the Emergence of Obesity, Viral Re-suppression and Integration Into Routine Programme Care (CHARACTERISE) study conducted in South Africa did not find any significant increase in BP of clients [32] and this may possibly be due to the cross-sectional nature of the study over a short period of 6 months. These findings reveal a higher risk of increase in BP in patients on DTG compared to other ART.
This study showed higher risk of developing HTN among male PWH. This finding is consistent with previous studies [15, 28, 33]. A potential biological mechanism explaining the sex difference in the incidence of HTN could be due to hormonal differences that protect women from developing HTN. It is widely believed that estrogen exerts a beneficial influence on the vasodilation of blood vessels, thus contributing to the regulation of HTN. This biological protection among women is more evident in adolescence and adulthood and becomes insignificant during the menopausal stage [16, 34]. Poor health-seeking behavior exhibited by men could also account for these findings. A study in urban Ghana revealed that men with HIV have poor health-seeking behavior, contributing to higher probability of developing noncommunicable diseases, including HTN, as observed in our findings [35].
Our study again revealed that older adults (aged ≥50 years) on DTG-based regimens have slightly higher risk (2%) of developing HTN, even though this difference is not clinically significant. These findings are consistent with what was found in a study in Uganda to determine factors associated with HTN among adults on DTG-based regimen [16, 36]. This could be associated with the aging process, which leads to various alterations in the arterial vasculature’s structure and function and reduced blood supply to the heart [37].
In this study, persons who were ART exposed had a lower risk of developing HTN, in contrast to findings by others [19–21]. We postulate that having been already exposed to the health system, they may have benefited from interventions that could have resulted in a reduced risk.
Higher body mass index as a behavioral factor has been reported as a risk factor for developing HTN among PWH on ART. Our findings showed a significant association of body mass index with the incidence of HTN. Obese PWH on DTG had 27% higher risk of being hypertensive. In assessing the correlates of HTN among PWH in Livingstone Province, Zambia, body mass index was reported as a significant risk factor for HTN among PWH on DTG [21]. Similar findings have been documented in Ethiopia [33].
While studies have shown that PWH, irrespective of whether they receive ART or not, exhibit a significant incidence of cardiovascular complications and other comorbidities [33], our study, however, revealed a reduced odds of developing HTN among clients with comorbidities. The observed outcome may be attributed to lifestyle modifications and improved health-seeking behavior among individuals with other comorbidities.
Our study had some limitations. There is a possibility of potential bias in this study as 87% of our patients initiated on DTG were already ART experienced and therefore might have developed some ART-associated risks for HTN prior to DTG initiation. Therefore, the unavailability of a control or comparative group in this study affects the internal validity of the study results and potentially impacts data interpretation. We also did not evaluate other HTN risk factors such as diet, family history of HTN, or physical inactivity among the study cohort. Despite this limitation, the study had a large cohort for which it was established that they had no HTN prior to DTG initiation. Though it was an observational study, BP measurements were standardized for all patients and all clinical data were routinely collected from heterogenous populations, including elderly patients at significant CVD risk.
Our study findings suggest that initiating patients on DTG is a risk factor for HTN and therefore, such patients require regular BP monitoring at each clinic visit. This is important and must be incorporated in national HIV guidelines given that integrase strand transfer inhibitors have become the preferred ART option in many developing countries, including Ghana, in addition to CVD prevention and management guidelines for adults. The high incidence of HTN in our study among treatment-naive persons calls for more research into the role of DTG. All patients should be screened for risk factors at baseline prior to DTG initiation, and those found with traditional risk factors for HTN need to be routinely monitored for HTN.
In summary, incidence of HTN was high among PWH on DTG. There is a need to monitor BP and traditional risk factors for HTN in adult PWH to reduce the burden of HTN and its complications. Future studies to investigate the role of DTG and its effect on epithelial sodium channel regulation in the pathogenesis of HTN are recommended.
Contributor Information
Margaret Lartey, Department of Medicine and Therapeutics, University of Ghana Medical School, Accra, Ghana; Department of Medicine, Korle Bu Teaching Hospital, Accra, Ghana.
Kwasi Torpey, Department of Population, Family, and Reproductive Health, School of Public Health, University of Ghana, Accra, Ghana.
Vincent Ganu, Department of Medicine, Korle Bu Teaching Hospital, Accra, Ghana.
Stephen Ayisi Addo, National AIDS/STI Control Programme, Ghana Health Service, Accra, Ghana.
Delia Bandoh, Ghana Field Epidemiology and Laboratory Training Programme, Department of Epidemiology and Disease Control, School of Public Health, University of Ghana, Accra, Ghana.
Marijanatu Abdulai, National AIDS/STI Control Programme, Ghana Health Service, Accra, Ghana.
Golda Akuffo, Ghana Field Epidemiology and Laboratory Training Programme, Department of Epidemiology and Disease Control, School of Public Health, University of Ghana, Accra, Ghana.
Ernest Kenu, Ghana Field Epidemiology and Laboratory Training Programme, Department of Epidemiology and Disease Control, School of Public Health, University of Ghana, Accra, Ghana.
Notes
Acknowledgments. We thank all the following for their various valuable contributions to ensure the success of the study: the study participants and study teams from Korle Bu Teaching Hospital, Kwesimintsim hospital, Kumasi South hospital, St Martins hospital, and Atua Government hospital; MDS Lancet Laboratories, Ghana; the Ghana National AIDS/STI Control Programme; the Ghana Health Service; and the Global Fund to Fight AIDS, Tuberculosis and Malaria.
Author contributions. M. L., K. T., and E. K.: Conceptualization, funding acquisition, methodology, supervision, formal analysis, writing of original draft, reviewing and editing draft, finalization of manuscript. V. G.: Conceptualization, methodology, supervision, formal analysis, writing of original draft, reviewing and editing draft, finalization of manuscript. S. A. A.: Funding acquisition, methodology, supervision, writing of original draft, reviewing and editing draft, finalization of manuscript. D. B. and M. A.: Formal analysis, writing of original draft, reviewing and editing draft, finalization of manuscript. G. A.: Supervision, writing of original draft, reviewing and editing draft, finalization of manuscript.
Financial support. This work was supported by the Global Fund to Fight AIDS, Tuberculosis and Malaria through the Ghana Health Service.
References
- 1. Joint United Nations Programme on HIV/AIDS . Global HIV and AIDS statistics—fact sheet. 2023. Available at: https://www.unaids.org/en/resources/fact-sheet?. Accessed 25 September 2023.
- 2. Xu Y, Chen X, Wang K. Global prevalence of hypertension among people living with HIV: a systematic review and meta-analysis. J Am Soc Hypertens 2017; 11:530–40. [DOI] [PubMed] [Google Scholar]
- 3. Kämpfen F, Wijemunige N, Evangelista B. Aging, non-communicable diseases, and old-age disability in low- and middle-income countries: a challenge for global health. Int J Public Health 2018; 63:1011–2. [DOI] [PubMed] [Google Scholar]
- 4. Shah ASV, Stelzle D, Ken Lee K, et al. Global burden of atherosclerotic cardiovascular disease in people living with HIV: systematic review and meta-analysis. Circulation 2018; 138:1100–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Rodríguez-Arbolí E, Mwamelo K, Kalinjuma AV, et al. Incidence and risk factors for hypertension among HIV patients in rural Tanzania—a prospective cohort study. PLoS One 2017; 12:e0172713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Pourcher V, Gourmelen J, Bureau I, Bouee S. Comorbidities in people living with HIV: an epidemiologic and economic analysis using a claims database in France. PLoS One 2020; 15:e0243529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Ruamtawee W, Tipayamongkholgul M, Aimyong N, Manosuthi W. Prevalence and risk factors of cardiovascular disease among people living with HIV in the Asia-Pacific region: a systematic review. BMC Public Health 2023; 23:1–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Derick KI, Khan Z. Prevalence, awareness, treatment, control of hypertension, and availability of hypertension services for patients living with human immunodeficiency virus (HIV) in sub-Saharan Africa (SSA): a systematic review and meta-analysis. Cureus 2023; 15:e37422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Odubela O, Odunukwe N, Peer N, Musa AZ, Lawal Salako B, Kengne AP. Prevalence of hypertension among antiretroviral therapy naïve patients in Lagos, Nigeria. Clin Hypertens 2023; 29:29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Memiah P, Nkinda L, Majigo M, et al. Hypertension and associated inflammatory markers among HIV-infected patients in Tanzania. J Interferon Cytokine Res 2021; 41:291–301. [DOI] [PubMed] [Google Scholar]
- 11. Gizamba JM, Davies J, Africa C, et al. Prevalence of obesity, hypertension and diabetes among people living with HIV in South Africa: a systematic review and meta-analysis. BMC Infect Dis 2023; 23:861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Chakraborty S, Rai RK, Biswas AK, Barik A, Gurung P, Praveen D. Health care seeking behaviour and financial protection of patients with hypertension: a cross-sectional study in rural West Bengal, India. PLoS One 2022; 17:e0264314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Mwenda AK, Kirigia C, Kamweru PK, Gitonga LK. Factors affecting health seeking behaviour in hypertensive patients of Imenti North sub county, Kenya. Int J Community Med Public Health 2020; 8:37. [Google Scholar]
- 14. Harimenshi D, Niyongabo T, Preux PM, Aboyans V, Desormais I. Hypertension and associated factors in HIV-infected patients receiving antiretroviral treatment in Burundi: a cross-sectional study. Sci Rep 2022; 12:20509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Mbuthia GW, Magutah K, McGarvey ST. The prevalence and associated factors of hypertension among HIV patients. Int J Hypertens 2021; 2021:5544916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Kasoma Mutebi R, Weil Semulimi A, Mukisa J, et al. Prevalence of and factors associated with hypertension among adults on dolutegravir-based antiretroviral therapy in Uganda: a cross sectional study. Integr Blood Press Control 2023; 16:11–21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Kovacs L, Kress TC, Belin de Chantemèle EJ. HIV, combination antiretroviral therapy, and vascular diseases in men and women. JACC Basic Transl Sci 2022; 7:410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Costa AN, Val F, Macedo ÁE, et al. Increased prevalence of hypertension among people living with HIV: where to begin? Rev Soc Bras Med Trop 2020; 53:1–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Davis K, Perez-Guzman P, Hoyer A, et al. Association between HIV infection and hypertension: a global systematic review and meta-analysis of cross-sectional studies. BMC Med 2021; 19:1–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Nduka CU, Stranges S, Sarki AM, Kimani PK, Uthman OA. Evidence of increased blood pressure and hypertension risk among people living with HIV on antiretroviral therapy: a systematic review with meta-analysis. J Hum Hypertens 2016; 30:355–62. [DOI] [PubMed] [Google Scholar]
- 21. Musekwa R, Hamooya BM, Koethe JR, Nzala S, Masenga SK. Prevalence and correlates of hypertension in HIV-positive adults from the Livingstone Central Hospital, Zambia. Pan Afr Med J 2021; 39:237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Joint United Nations Programme on HIV/AIDS . UNAIDS data 2023. Available at: https://www.unaids.org/sites/default/files/media_asset/data-book-2023_en.pdf. Accessed 27 September 2023.
- 23. Hattoh KA, Sienso BA, Kuugbee ED. Prevalence of comorbidities and associated factors among HIV patients attending antiretroviral clinics in the Tamale metropolis, Ghana. Venereology 2023; 2:1–15. [Google Scholar]
- 24. Sarfo FS, Nichols M, Singh A, et al. Characteristics of hypertension among people living with HIV in Ghana: impact of new hypertension guideline. J Clin Hypertens (Greenwich) 2019; 21:838–50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. National AIDS/STI Control Programme . NACP annual report. Accra, Ghana: National AIDS/STI Control Programme; 2019.
- 26. Muntner P, Carey RM, Gidding S, et al. Potential U.S. population impact of the 2017 American College of Cardiology/American Heart Association high blood pressure guideline. J Am Coll Cardiol 2018; 71:109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Sempere A, Assoumou L, González-Cordón A, et al. Incidence of hypertension and blood pressure changes in persons with HIV at high risk for cardiovascular disease switching from boosted protease inhibitors to dolutegravir: a post-hoc analysis of the 96-week randomised NEAT-022 trial. Clin Infect Dis 2023; 77:991–1009. [DOI] [PubMed] [Google Scholar]
- 28. Brennan AT, Jamieson L, Crowther NJ, et al. Prevalence, incidence, predictors, treatment, and control of hypertension among HIV-positive adults on antiretroviral treatment in public sector treatment programs in South Africa. PLoS One 2018; 13:e0204020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Tovar Sanchez T, Mpoudi-Etame M, Kouanfack C, et al. Risks of metabolic syndrome in the ADVANCE and NAMSAL trials. Front Reprod Health 2023; 5:1133556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Mishra B, Sinha ND, Gidwani H, Shukla SK, Kawatra A, Mehta SC. Equipment errors: a prevalent cause for fallacy in blood pressure recording—a point prevalence estimate from an Indian Health University. Indian J Community Med 2013; 38:15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Siddiqui M, Calhoun DA. Blood pressure measurement challenges in severely obese patients. Am J Hypertens 2019; 32:139–40. [DOI] [PubMed] [Google Scholar]
- 32. Bosch B, Akpomiemie G, Chandiwana N, et al. Weight and metabolic changes after switching from tenofovir alafenamide/emtricitabine (FTC)+dolutegravir (DTG), tenofovir disoproxil fumarate (TDF)/FTC + DTG, and TDF/FTC/efavirenz to TDF/lamivudine/DTG. Clin Infect Dis 2023; 76:1492–5. [DOI] [PubMed] [Google Scholar]
- 33. Mulugeta H, Afenigus AD, Haile D, et al. Incidence and predictors of hypertension among HIV patients receiving ART at public health facilities, northwest Ethiopia: a one-year multicenter prospective follow-up study. HIV AIDS (Auckl) 2021; 13:889–901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Everett B, Zajacova A. Gender differences in hypertension and hypertension awareness among young adults. Biodemography Soc Biol 2015; 61:1–17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Abdulai MA, Marable JK, Wadus A, Asante KP. A qualitative analysis of factors influencing health-seeking behavior of people living with HIV, hypertension and diabetes in an urban area of Ghana. J Multimorbidity Comorbidity 2022; 12:26335565221092664. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Fan H, Guo F, Hsieh E, et al. Incidence of hypertension among persons living with HIV in China: a multicenter cohort study. BMC Public Health 2020; 20:1–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Oliveros E, Patel H, Kyung S, et al. Hypertension in older adults: assessment, management, and challenges. Clin Cardiol 2020; 43:99–107. [DOI] [PMC free article] [PubMed] [Google Scholar]