Abstract
Aim
The aim of this study was to evaluate the intra‐abdominal status related to postoperative pancreatic fistula by combining postoperative fluid collection and drain amylase levels.
Methods
We retrospectively reviewed the data of 203 patients who underwent distal pancreatectomy and classified their postoperative abdominal status into four groups based on postoperative fluid collection size and drain amylase levels. We also evaluated the incidence of clinically relevant postoperative pancreatic fistula in each group according to C‐reactive protein values.
Results
The incidence of clinically relevant postoperative pancreatic fistula in the entire cohort (n = 203) was 28.1%. Multivariate analysis revealed that postoperative fluid collection, drain amylase levels, and C‐reactive protein levels are considerable risk factors for clinically relevant postoperative pancreatic fistula. In the subgroup with large postoperative fluid collection and high drain amylase levels, 65.9% of patients developed clinically relevant postoperative pancreatic fistula. However, no significant difference was observed in C‐reactive protein levels between patients with clinically relevant postoperative pancreatic fistula and those without it. In contrast, in the subgroup with a large postoperative fluid collection size or a high amylase level alone, a significant difference was observed in C‐reactive protein values between the patients with clinically relevant postoperative pancreatic fistula and those without it.
Conclusion
Postoperative fluid collection status and the C‐reactive protein value provide a more precise assessment of intra=abdominal status related to postoperative pancreatic fistula after distal pancreatectomy. This detailed analysis may be a clinically reasonable approach to individual drain management.
Keywords: amylase, analysis, pancreatectomy, pancreatic fistula, patients
We identified PFCs, postoperative drain amylase levels, and C‐reactive protein (CRP) levels as significant risk factors for CR‐POPF after DP. Detailed assessments of the size of PFC combined with drain amylase level (PFC status) revealed potentially different clinical meanings of drain amylase level and pathogenesis of PFCs. Furthermore, we demonstrated the different clinical significances of CRP levels according to the PFC status.

1. INTRODUCTION
Postoperative pancreatic fistula (POPF) is a common complication following distal pancreatectomy (DP), with occurrence rates ranging from 12%–33%. 1 , 2 , 3 , 4 Despite advances in perioperative care and techniques, POPF continues to contribute to morbidity and mortality after DP. 5 , 6
The universally used International Study Group of Pancreatic Fistula (ISGPF) classification, updated in 2017, includes drain amylase levels as a reference to classify Grade Biochemical Leak, B or C, depending on the procedure performed in relation to the fistula. 7 Postoperative drain amylase levels have been recognized as a common predictor, with various cutoff values reported for drain removal. 8 , 9 , 10 However, whether clinical management based solely on drain amylase levels is optimal remains unclear. Yoshino et al reported that seven out of 26 cases diagnosed with clinically relevant postoperative pancreatic fistula (CR‐POPF) (grade B or C according to the ISGPF classification) had drain amylase levels below the cutoff value of 1026 IU/L on postoperative day (POD) 3, determined by receiver operating characteristic (ROC) curves. 11 In another study by Bassi et al, early drain removal was implemented in patients with amylase values in drains ≤5000 U/L on POD 1. Of the 114 eligible cases, 39 DP cases were included, and two (5.1%) required percutaneous drain insertion after drain removal owing to fluid accumulation resulting from pancreatic fistula. 8 Therefore, a clinical management policy based solely on drain amylase levels remains controversial, and further refinements are required for optimal postoperative drain management to prevent CR‐POPF development.
Recent studies have demonstrated that postoperative fluid collections (PFCs), detected by computed tomography (CT) scans, can predict the likelihood of developing POPF after DP. 11 , 12 However, several studies have also demonstrated that PFCs can be asymptomatic. 13 , 14 , 15 A consensus on the optimal means of integrating PFC assessment into clinical practice is lacking. Moreover, the presence of PFCs does not necessarily indicate POPF occurrence, as other factors, such as focal ascites or lymphatic leakage, may contribute to PFC. Therefore, the relationship between PFC and POPF remains equivocal and necessitates further investigation.
Considering the existing controversies regarding the relationship between POPF development, drain amylase, and PFCs, we hypothesized that conducting detailed analyses using drain amylase levels and PFCs could provide more precise information regarding the likelihood of CR‐POPF development, PFC nature, and other clinical conditions in the abdominal cavity. Therefore, we aimed to integrate PFCs and drain amylase levels to classify and assess the postoperative intra‐abdominal status related to POPF and identify appropriate clinical management according to these evaluations.
2. METHODS
2.1. Patients
We retrospectively analyzed the electronic medical records of 203 patients who underwent DP between January 2011 and November 2018 at the Osaka International Cancer Institute. Table S1 presents the demographic characteristics of the enrolled patients. Based on the International Study Group on Pancreatic Surgery's established criteria (Table 1), 7 the 203 patients were classified into two groups: the CR‐POPF (Grades B and C) and the non‐POPF (which included cases with no fistula and “biochemical leaks”). Postoperative complications were categorized based on the Clavien–Dindo classification. 17 This study conforms to the provisions of the Declaration of Helsinki. The study protocol of the Osaka International Cancer Institute was approved by the Ethics Committee (registration number 20230), and the enrolled patients were given the option to withdraw their participation.
TABLE 1.
Clinical factors according to CR‐POPF
| Non‐POPF (no fistula or BL) n = 146 | CR‐POPF (B or C) n = 57 | P value | |
|---|---|---|---|
| Background | |||
| Age (y) | 68 (27–82) | 66 (28–82) | 0.12 |
| Sex (male/female (%)) | 79 (54.1%)/67 (45.9%) | 43 (75.4%)/14 (24.6%) | <0.01 |
| Body mass index (kg/m2) | 21.4 (14.9–32.7) | 22.4 (18.5–30.7) | 0.01 |
| Presence of diabetes mellitus (yes (%)) | 94 (64.4%) | 41 (71.9%) | 0.33 |
| Preoperative diagnosis (pancreatic cancer: other diseases) a | 122 (83.6%)/24 (16.4%) | 42 (73.7%)/15 (26.3%) | 0.12 |
| Preoperative CRT (yes (%)) | 105 (71.9%) | 32 (56.1%) | 0.05 |
| MPD diameter (mm) | 2.0 (1.0–10.0]) | 2.00 (1.0–9.0) | 0.17 |
| Intraoperative findings | |||
| Surgical procedure (DP: DP‐CAR) b | 130/16 | 49/8 | 0.63 |
| Blood loss (ml) | 493 (10–1750) | 500 (25–2180) | 0.67 |
| Operation time (min) | 363 (184–723) | 374 (145–723) | 0.59 |
| Postoperative findings | |||
| CRP levels on POD 7 (mg/dL) | 3.5 (0.2–12.1) | 6.0 (2.2–24.0) | <0.01 |
| Drain amylase levels on POD 1 (U/L) | 337 (25–8723) | 959 (59–20 910) | <0.01 |
| Drain amylase levels on POD 3 (U/L) | 214 (17–32 185) | 851 (42–13 104) | <0.01 |
| Clavian–Dindo (IIIa≦) | 5 (3.4%) | 7 (12.4%) | 0.04 |
| Postoperative CT findings | |||
| Thickness of the pancreatic stump (mm) | 10.6 (5.3–23.2) | 11.6 (5.7–28.1) | <0.01 |
| V max (mm) | 14 (0–53) | 28 (3–62) | <0.01 |
| V ratio | 0.21 (0–0.90) | 0.35 (0.04–0.71) | <0.01 |
| H score (H≧2) | 30 (20.5%) | 41 (71.9%) | <0.01 |
Abbreviations: CRT, chemoradiation therapy; CT, computer tomography; DP, distal pancreatectomy; DP‐CAR, distal pancreatectomy with celiac axis resection 16 ; MPD, main bile duct; POD, postoperative day; POPF, postoperative pancreatic fistula.
The details are shown in Table S3.
The details are shown in Table S4.
2.2. Surgical procedure and postoperative management
The standardized procedure utilized by our hospital was followed by the same surgical team for all pancreatic resections, as described in our previous report (Figure S1). 18 The pancreas was cut using a scalpel, and the main pancreatic duct was ligated with a nonabsorbable suture. The surface of the pancreatic stump was soft, coagulated, and covered with polyglutamic acid (PGA) felt using fibrin glue. In all patients, two prophylactic closed‐suction drains were positioned in the pancreas stump and the left subphrenic space. During the postoperative phase, all patients underwent CT scans to screen for subclinical complications, such as aneurysm formation, thrombosis formation, or apparent intra‐abdominal abscess. In the absence of such complications, surgical drains were promptly removed according to the standard policy for the drain removal criteria at our institute during the study period. 18 , 19 In all participants, the decision to remove the drain was not made based on the PFC status. Our data indicated that postoperative antibiotics typically consisted of piperacillin‐tazobactam for 3 d, with a maximum prolongation of 5 d. 20 The decision to administer octreotide in the postoperative period was not standardized and depended on the surgeons' preference.
2.3. CT protocol and interpretation
Postoperative CT scans were systematically (following the protocol) obtained on the sixth or seventh POD using 64‐ and 320‐detector devices in all patients. (Aquilion CX Edition and Aquilion ONE, respectively; Canon Medical Systems, Tochigi, Japan). Two experienced surgeons retrospectively analyzed all CT findings. The thickness of the pancreatic stump was determined by measuring the dissection site on the most recent CT scan before the surgery. The PFCs in this study were defined as the radiologically evident volume of fluid collection adjacent to the pancreatic stump, as observed in CT scans. Other fluid collections separate from the pancreatic stump were not included in the PFC category and excluded from subsequent analyses. The PFCs index was determined by measuring the maximum height and width of PFCs in the axial view. The maximum vertical distance (V max) from the lower to the upper edge of the PFC was quantitatively measured (Figure 1A). The vertical ratio (V ratio) was calculated by dividing V max by the peritoneal depth, which represents the distance between the anterior surface of the aorta and the peritoneum. The maximum horizontal distance was subjectively characterized using a horizontal score (H score, Figure 1B–E): H0, an absence of persistent fluid accumulation at the pancreatic stump (Figure 1B); H1, the PFC was restricted to the periphery of the superior mesenteric artery (Figure 1C); H2, the PFC spread to the left perirenal fat (Figure 1D); H3, the PFC extended to the left subdiaphragm (Figure 1E).
FIGURE 1.

(A) The diagram illustrates the vertical assessment method employed to evaluate postoperative fluid collections (PFCs) and peritoneal depth. (B–E) Representative computed tomography images were used to evaluate the horizontal extent of continuous fluid accumulation from the pancreatic stumps. (B) The absence of consistent fluid retention at the pancreatic stump was classified as H0. (C) The confinement of PFCs to the perimeter of the superior mesenteric artery was classified as H1. (D) The spread of PFCs to the prerenal fat was classified as H2. (E) The extension of PFCs to the left subdiaphragm was classified as H3.
2.4. Clinical data collection and statistical analysis
The comparisons of continuous variables were conducted using t‐tests for variables such as age, body mass index, duration of surgery, blood loss, diameter of the main pancreatic duct, and thickness of the pancreatic stump, and using Mann–Whitney U‐tests for drain amylase levels on POD 1 and 3. A logistic regression model was applied for multivariable analysis, and statistical significance was set at P < 0.05 in two‐tailed tests. All statistical analyses were performed utilizing R v. 4.2.2, R Studio 2022.12.0 + 353, and the software EZR (Saitama Medical Center, Jichi Medical University, Saitama, Japan), a graphical user interface for R (The R Foundation for Statistical Computing, Vienna, Austria). It is a modified version of R commander designed to add statistical functions frequently used in biostatistics. 21
3. RESULTS
3.1. Patient characteristics and perioperative findings
Table S1 presents the patient characteristics, with a high proportion being patients with cancer (80.8%) and a substantial percentage undergoing preoperative chemoradiation therapy (67.5%). Of the 203 patients, 146 (71.9%) were classified into the non‐POPF group, whereas 57 patients (28.1%) were classified into the CR‐POPF group (Table 1 and Table S3). The results revealed that previously identified risk factors, including obesity and thickness of the pancreatic stump, were present at higher frequencies in the CR‐POPF group. Regarding postoperative outcomes, Table 1 demonstrates that drain amylase levels on POD 1 and 3 were significantly higher in the CR‐POPF group compared to the non‐POPF group (959 U/L versus 337 U/L, P < 0.01 and 851 U/L versus 214 U/L, P < 0.01, respectively). Similarly, C‐reactive protein (CRP) levels on POD 7 were significantly higher in the CR‐POPF group (3.5 mg/dL versus 6.0 mg/dL, P < 0.01). Furthermore, the results of postoperative CT analysis revealed significantly higher values for V max, V ratio, and H score in the CR‐POPF group (P < 0.01 for all). We also observed a positive correlation between V max and V ratio with H score (r = 0.72 and 0.71, respectively) (Figure 2A). An increase in H score was associated with a higher CR‐POPF incidence, with more patients classified in the CR‐POPF group than the non‐POPF group for H scores of 2 and 3 (Table S2). We observed no correlation between V max and other clinical factors, such as postoperative drain amylase levels and CRP on POD 7, beyond the association in CT findings (Figure S2).
FIGURE 2.
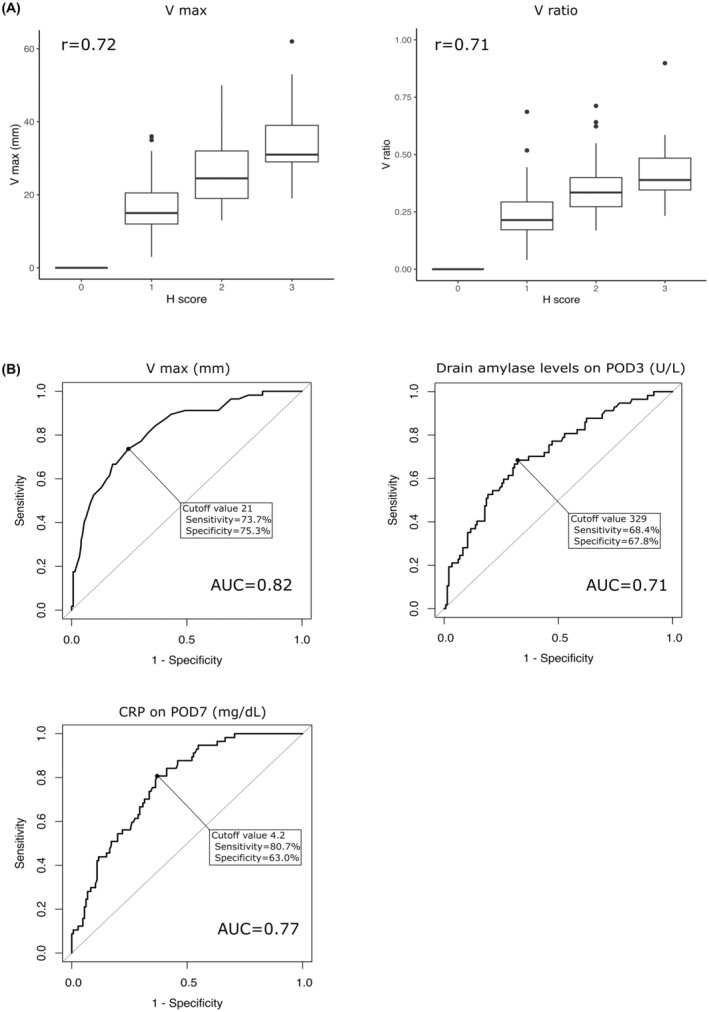
(A) The relationship between the vertical and horizontal distribution of postoperative fluid collections. (B) Receiver operating characteristic curve analysis was utilized to assess the predictive value of maximum vertical diameter (V max), drain amylase levels on postoperative day 3, and C‐reactive protein (CRP) on postoperative day 7 in detecting clinically relevant postoperative pancreatic fistula (CR‐POPF).
3.2. Predictors of CR‐POPF
ROC analysis for CR‐POPF was performed for each continuous variable (Figure 2B and Figure S3). The analysis indicated that V max and V ratio had the highest area under the curve (AUC) (0.82 for both). CRP on POD 7 and the drain amylase levels on POD 3 had the next highest AUC values (AUC = 0.77 and 0.71, respectively).
After dividing each factor into two groups based on ROC analysis cutoff values, univariate analysis revealed that V max, V ratio, H score, CRP on POD 7, drain amylase levels, and other clinical factors were significantly associated with CR‐POPF (P < 0.01 for all) (Table 2). V max, V ratio, and H score were positively correlated; therefore, V max was utilized for multivariate analysis. The results of the multivariate analysis revealed that V max (P < 0.01), drain amylase levels on POD3 (P = 0.02), and CRP on d 7 (P < 0.01) were significant predictors of CR‐POPF.
TABLE 2.
Univariate and multivariate analyses for the development of CR‐POPF
| Univariate analysis | Multivariate analysis (multi logistic analysis) | ||||
|---|---|---|---|---|---|
| Non‐POPF (none or BL) n = 146 | CR‐POPF (B or C) n = 57 | P value | Odds ratio (95% CI) | P value | |
| Background | |||||
| Age (y, = < 69 / 69<) | 81/65 | 42/15 | 0.02 | 0.61 (0.27–1.40) | 0.24 |
| Sex (male/female) | 79/67 | 13/14 | <0.01 | 0.79 (0.34–1.85) | 0.59 |
| Body mass index (kg/m2, = < 23/23<) | 104/42 | 29/28 | <0.01 | 0.89 (0.39–2.05) | 0.79 |
| Presence of diabetes mellitus (yes /no) | 94/52 | 41/16 | 0.33 | ||
| Preoperative diagnosis (pancreatic cancer/other diseases) a | 122/24 | 42/15 | 0.12 | ||
| Preoperative CRT (yes /no) | 105/47 | 32/25 | 0.05 | 0.82 (0.35–1.89) | 0.64 |
| MPD diameter (mm, = < 2/2<) | 108/38 | 37/20 | 0.23 | ||
| Intraoperative findings | |||||
| Surgical procedure (DP/DP‐CAR) b | 130/16 | 41/16 | 0.63 | ||
| Blood loss (mL, = < 315/315<) | 45/101 | 12/45 | 0.22 | ||
| Operation time (min, = < 395/395<) | 89/57 | 30/27 | 0.34 | ||
| Postoperative findings | |||||
| CRP on POD7 (mg/dL, = < 4.25/4.25<) | 93/53 | 12/45 | <0.01 | 4.6 (2.04–10.4) | <0.01 |
| Drain amylase levels on POD1 (U/L, = < 389/389<) | 81/65 | 14/43 | <0.01 | — | — |
| Drain amylase levels on POD3 (U/L, = < 329/329<) | 99/47 | 19/38 | <0.01 | 2.6 (1.14–5.9) | 0.02 |
| Postoperative CT findings (POD7) | |||||
| Thickness of the pancreatic stump (mm, = < 13/13<) | 109/37 | 39/18 | 0.38 | ||
| V max (= < 21/21<) | 114/32 | 17/40 | <0.01 | 5.55 (2.52–12.2) | <0.01 |
| V ratio (= < 0.314/0.314<) | 121/25 | 16/41 | <0.01 | — | — |
| H score (0 or 1 vs. 2 or 3) | 116/30 | 16/41 | <0.01 | — | — |
Abbreviations: CRT, chemoradiation therapy; CR‐POPF, clinically relevant postoperative pancreatic fistula; CT, computer tomography; DP, distal pancreatectomy; DP‐CAR, distal pancreatectomy with celiac axis resection 16 ; MPD, main bile duct; POD, postoperative day; POPF, postoperative pancreatic fistula.
The details are shown in Table S3.
The details are shown in Table S4.
3.3. CR‐POPF incidence based on the V max and postoperative drain amylase levels
Patients were categorized into four groups based on the cutoff values of V max and drain amylase levels on POD 3, defining the PFC status as follows (Figure 3A): Group 1 had V max and drain amylase levels exceeding the cutoff value. Group 2 exhibited values surpassing the cutoff for V max only. Group 3 had levels surpassing the cutoff value for drain amylase alone. Group 4 had values lower than the cutoff for V max and drain amylase. Table 3 describes the postoperative complications related to CR‐POPF. As presented in Table 3, Group 1 (n = 41) had the highest incidence of CR‐POPF at 65.9%, whereas only 6.9% of patients in Group 4 (n = 87) experienced CR‐POPF. The proportion of patients with CR‐POPF in Group 2 (n = 31) was 41.9%, higher than that of Group 3 (n = 44, 25.0%). Approximately half of the patients in Group 1 had drain exchange (n = 19, 46.3%) and persistent drainage (n = 21, 51.2%). In addition, the incidence of intra‐abdominal abscesses was higher in Group 1 (n = 4, 9.8%) than in the other groups, and one patient required reoperation owing to the intra‐abdominal abscess. In Group 4, drain changes were performed only in two cases of suspected retrograde infections or abscess formation. We compared CRP values in the four groups shown in Figure 3A to further evaluate the clinical relevance of each PFC status (Figure 3B). In Groups 1 and 4, no significant difference was observed in CRP between the CR‐POPF and non‐POPF groups (P = 0.10 in Group 1 and P = 0.29 in Group 4). In contrast, CRP values were significantly higher in the CR‐POPF group than in the non‐POPF group in Groups 2 and 3 (P < 0.01 in Group 2 and P = 0.03 in Group 3). ROC curves were delineated for Groups 2 and 3 to predict CR‐POPF using CRP values on POD 7, with corresponding cutoff values of 4.25 and 4.27, respectively (Figure S4). Cases with CRP values higher than these cutoff values revealed a higher occurrence of drain replacement and prolonged drain placement in both groups.
FIGURE 3.

(A) The cases were sorted into four groups according to preestablished threshold values for V max of PFCs and drain amylase levels on postoperative day 3. Cases with CR‐POPF and those without it are depicted with black and white points, respectively. (B) The violin plot compares the CRP levels on postoperative day 7 between cases with and without CR‐POPF in the four groups classified based on the cutoff values of V max of PFCs and drain amylase levels on postoperative day 3, as shown in Figure 3(A). The interquartile range is also included.
TABLE 3.
Analysis of CR‐POPF incidence in each group categorized by cutoff values of V max and drain amylase levels on POD 3
| Group | Total number of patients (n = 203) | Number of CR‐POPF patients (n = 57) | Abscess | Pancreatic pseudocyst | Drain exchange | Persistent drainage | PTAD | Reoperation |
|---|---|---|---|---|---|---|---|---|
| 1 | 41 (20.2%) | 27 (65.9%) | 4 (9.8%) | 1 (2.4%) | 19 (46.3%) | 21 (51.2%) | 2 (4.9%) | 1 (2.4%) |
| 2 | 31 (15.2%) | 13 (41.9%) | 1 (3.2%) | 0 (0%) | 10 (32.5%) | 12 (38.7%) | 1 (3.2%) | 0 (0%) |
| 3 | 44 (21.7%) | 11 (25.0%) | 2 (4.5%) | 0 (0%) | 8 (18.1%) | 9 (20.4%) | 2 (4.5%) | 0 (0%) |
| 4 | 87 (42.9%) | 6 (6.9%) | 3 (3.4%) | 0 (0%) | 2 (2.3%) | 1 (1.1%) | 3 (3.4%) | 0 (0%) |
Abbreviations: CR‐POPF, clinically relevant postoperative pancreatic fistula; POD, postoperative day, PTAD, percutaneous abscess drainage.
4. DISCUSSION
We investigated the clinical factors associated with CR‐POPF after DP, focusing on PFCs, drain amylase levels, and CRP values. Our multivariate analysis identified PFCs, postoperative drain amylase levels, and CRP values as significant risk factors for CR‐POPF. Of note, failure to show the significance of the pancreatic thickness (which is a well‐known risk factor for CR‐POPF 22 , 23 ) in developing CR‐POPF using the multivariate analysis may have been attributed to the characteristics of the method of the pancreatic stump closure used in the patients enrolled in this study. Using this method, the pancreatic parenchyma was not compressed at the stump, thus leading to less association between the thickness of the pancreas and the incidence of CR‐POPF. 18 Among the four groups classified based on PFC status, the group characterized by a large PFC size and elevated levels of drain amylase displayed the highest incidence of CR‐POPF, followed by the group in which only the PFC size surpassed the reference value. In addition, the CRP values exhibited statistically significant variations between the presence or absence of CR‐POPF in the subgroup with a large PFC size or elevated drain amylase levels alone.
Consistent with previous research, this study confirms that PFC size and drain amylase levels are the primary factors associated with CR‐POPF. However, our study highlights that neither factor is independently sufficient to predict CR‐POPF. Specifically, low drain amylase levels do not always indicate the absence of pancreatic fistula, and large PFC size does not always indicate the presence of CR‐POPF. Uchida et al provided insights into the relationship between PFCs and CR‐POPF, as they observed that the extent of PFCs was significantly associated with elevated CRP levels at the time of CT. 12 We also identified a significant association between CRP levels and CR‐POPF. The clinical significance of CRP values varied among the four categories. These findings suggest that CR‐POPF pathogenesis is multifactorial and highlight the importance of assessment based on multiple factors (such as drain amylase level, PFC, and CRP value) rather than a single factor.
Among the four groups of the PFC status (Figure 3A), the clinical management in Groups 1 and 4 was straightforward. Group 1 was characterized by a high drain amylase levels and large PFC with high CRP level, regardless of the presence of CR‐POPF. In this group, the incidence of CR‐POPF and other intra‐abdominal complications was the highest (Table 3 and Figure 3B). These observations likely indicated the presence of inadequately drained pancreatic fluid, with possible infection and inflammation, consequently leading to more severe complications. In this context, cases in this group required prompt intervention to drain the residual fluid collection (surgical intervention for redrainage or placement of additional drains along with appropriate antibiotic therapy) to prevent developing more severe complications. Group 4, which exhibited lower drain amylase levels and small PFC with low CRP levels, had the lowest incidence of CR‐POPF and other intra‐abdominal complications (Table 3 and Figure 3B). Based on these findings, timely drain removal is advised to avoid the increased likelihood of developing complications possibly due to the prolonged drain accumulation, such as retrograde infection. Contrarily, more cautious attention should be paid to the assessment of the intra‐abdominal status and the drain management in cases in Groups 2 and 3. This is because a more complicated pathogenesis of PFC was assumed in these groups and the incidence of CR‐POPF in them was moderately high (Table 3). Considering the nonelevated drain amylase levels in Group 2, the possible pathogenesis of large PFC was (1) the presence of inadequately drained POPF or (2) fluid collection due to other factors than POPF: intra‐abdominal abscess not due to POPF, chylous/residual ascites, and lymphatic leakage (Table 4). Elevated CRP levels in cases in Group 2 likely indicated the presence of inadequately drained POPF (indeed, the incidence of CR‐POPF was significantly higher in cases of an elevated CRP level than in cases without an elevated CRP level in Group 2, Figure 3A) or other reasons contributing to the inflammatory process (intra‐abdominal abscess). In such cases, the patient should be promptly treated with appropriate antibiotic therapy and/or a new drainage tube should be inserted for adequate drainage of PFC. In Group 2, in cases of low CRP levels, the likelihood of developing CR‐POPF was lower (Figure 3B) and PFC may have been formed by other nothreatening causes (residual ascites). In this context, timely drain removal may be feasible in these cases. In Group 3, the modest PFC size with elevated drain amylase level indicated the presence of POPF with relatively adequate drainage (Table 4). Drain replacement, fistulization of a residual cavity, and subsequent drain removal would be feasible, particularly in Group 3 including cases of low CRP levels. However, high CRP levels in Group 3 might indicate persistent inflammation/infection caused by POPF or other pathogeneses (Figure 3B). In these cases, more aggressive drain management (insertion of additional drains) for better drainage of residual PFC with antibiotic therapy would be required to avoid the further deterioration in postoperative outcomes. Additionally, evaluation of possible alternative intra−/extra‐ abdominal source of inflammation/infection would be needed, as clinically indicated. These findings highlighted the importance of careful postoperative management of patients according to the PFC status: the combination of the PFC size and drain amylase levels. Moreover, the assessment of CRP levels in combination with PFC status could facilitate our further understanding of the intra‐abdominal status in each individual patient, thus leading to optimal postoperative management.
TABLE 4.
Expected postoperative intra‐abdominal conditions based on pancreatic fluid collections and drain amylase levels on postoperative day 3 depicted in Figure 3A
| Group | PFC size | Drain amylase levels | Anticipated postoperative intra‐abdominal conditions |
|---|---|---|---|
| 1 | Large | High | The pancreatic fluid has not been adequately drained, resulting in the presence of residual fluid in the peritoneal cavity, etc. |
| 2 | Large | Low | Accumulation of fluid in the abdominal cavity can occur due to insufficient drainage or the presence of chylous ascites, etc. |
| 3 | Small | High | Presence of pancreatic fistula despite adequate drainage, etc. |
| 4 | Small | Low | No pancreatic fistula occurred, and postoperative ascites and other fluids were effectively drained. |
Another issue to be addressed in this study was how we selected the indicators of PFC size, such as V max, V ratio, and H score. Previous reports assessing the association between PFC and CR‐POPF evaluated the “size of the PFC” by various fashions. 11 , 12 , 24 , 25 Theoretically, the 3D evaluation of PFC would provide a more accurate PFC volume than other measurements, such as the maximum diameter measurement. Uchida et al proposed their own 3D measurement system for the size of PFC based on the observation that PFC often assume a flat and ellipsoidal shape, which may result in inappropriate evaluation of the PFC status by PFC estimation with a simple measurement (the maximum diameter of PFC). 12 In this study, we evaluated the vertical spread of PFC by V max and V ratio and horizontal spread by H score, and found that the horizontal spread of PFC (H score) was significantly correlated with vertical spread (V max and V ratio, Figure 2A). Both V max and V ratio could provide similar predictive significance for the development of POPF (AUC: 0.82 for both V max and V ratio, Figure 2B and Figure S3). Moreover, even the combination index creating the V max and the H score (multiplying the V max by the H score) offered a comparable AUC value to that of the V max alone (AUC: 0.85 vs. 0.82, P = 0.12, Figure 2B and Figure S3). Although we used V max, which is the simplest one, as the indicator of PFC size in the further analyses, the selection of which indicator to use for PFC size estimation would depend on the balance between ease of clinical implementation and accuracy of assessment.
This study had some limitations. First, routine CT imaging may not be commonly performed post‐DP owing to its radiation exposure and cost, which may limit the generalizability of these results in some institutions. Therefore, substituting with other minimally invasive tests, such as transabdominal ultrasound, may be advantageous. For instance, CT may be reserved for cases with evident fluid retention in the abdominal cavity based on the transabdominal ultrasound findings, elevated amylase levels, or elevated CRP levels. Second, this study employed a delayed timing for CT scans performed on POD 6 or 7. Considering the current trend of earlier drain removal in the field of pancreatic surgery, 26 the evaluation of PFC status during an earlier postoperative period would be more clinically reasonable. In this regard, an earlier assessment of fluid accumulation, such as through a less complex echographic examination, may be necessary before drain removal in selected cases. Third, the inclusion of the patients who received more invasive procedures, such as DP‐CAR, 16 and those who received radiation therapy preoperative CRT may have introduced unexpected biases on the results of our current study (Table S4). 27 Although the incidence of CR‐POPF and the significance of CRP values on POD 7 were similar, even in the subset of the patients, excluding DP‐CAR cases (n = 24) from the entire cohort (Figure S5A and B), a prospective study, stratifying these potential clinical confounders, is required to provide more robust conclusion of the significance of intra‐abdominal status assessments for CR‐POPF using PFC, drain amylase levels, and CRP values. Finally, the data were retrospectively collected from a single facility, and the criteria for intervention in cases of CR‐POPF relied on the treating surgeons' discretion. Therefore, validation through future multicenter prospective studies with meticulously regulated postoperative management (drain removal policy) is needed for obtaining more conclusive results, specifically the cutoff values for PFC size, drain amylase levels, and CRP levels.
In conclusion, our study identified PFCs, postoperative drain amylase levels, and CRP levels as significant risk factors for CR‐POPF after DP. Detailed assessments of the size of PFC and drain amylase level (PFC status) revealed a potentially different pathogenesis of PFCs. Furthermore, we demonstrated the different clinical significances of the PFC based on the drain amylase level and CRP levels. The PFC status, incorporating the assessment of drain amylase and CRP levels, may aid in predicting the incidence of complications. These findings suggest that individualized postoperative management strategies may be optimized by assessing the PFC status of each patient.
AUTHOR CONTRIBUTIONS
YM and HT designed the study and drafted the article. YM, HT, and KA collected and analyzed the data. HA, MK, SH, and HW contributed to data interpretation and critically reviewed the article. HM, MO, and KS contributed to the study's conception and provided significant intellectual input. All authors have reviewed and approved the final version of the article.
FUNDING INFORMATION
This work was partly supported by a grant from the Osaka Foundation for the Prevention of Cancer and Life‐style related Diseases.
CONFLICT OF INTEREST STATEMENT
The authors declare no conflicts of interest for this article.
ETHICS STATEMENT
The protocol of the Osaka International Cancer Institute was approved by the Ethics Committee, and it conforms to the provisions of the Declaration of Helsinki. Research Ethics Committee of Osaka International Cancer Institute, Approval No. 20230. All informed consent was obtained from the subjects. Confidentiality and anonymity were ensured throughout the study, and all data were collected and analyzed securely and confidentially. The study protocol was registered in the Osaka International Cancer Institute, and all data were managed in accordance with their guidelines.
Supporting information
Tables S1–S4.
Figure S1.
Figure S2.
Figure S3.
Figure S4.
Figure S5.
ACKNOWLEDGMENTS
We thank the staff members of the Gastroenterological Surgery Department at Osaka International Cancer Institute for their invaluable support and assistance throughout this project. Their dedication, hard work, and expertise have been indispensable to the success of this research.
Mukai Y, Asukai K, Akita H, Kubo M, Hasegawa S, Wada H, et al. Assessing Intra‐abdominal status for clinically relevant postoperative pancreatic fistula based on postoperative fluid collection and drain amylase levels after distal pancreatectomy. Ann Gastroenterol Surg. 2024;8:321–331. 10.1002/ags3.12741
DATA AVAILABILITY STATEMENT
The data supporting the findings of this study can be obtained upon reasonable request from the corresponding author, HT. The data cannot be made publicly available as they contain sensitive information that could potentially jeopardize the privacy of the research participants.
REFERENCES
- 1. Rodríguez JR, Germes SS, Pandharipande PV, Gazelle GS, Thayer SP, Warshaw AL, et al. Implications and cost of pancreatic leak following distal pancreatic resection. Arch Surg. 2006;141(4):361–365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Pannegeon V, Pessaux P, Sauvanet A, Vullierme MP, Kianmanesh R, Belghiti J. Pancreatic fistula after distal pancreatectomy: predictive risk factors and value of conservative treatment. Arch Surg. 2006;141(11):1071–1076. [DOI] [PubMed] [Google Scholar]
- 3. Ferrone CR, Warshaw AL, Rattner DW, Berger D, Zheng H, Rawal B, et al. Pancreatic fistula rates after 462 distal pancreatectomies: staplers do not decrease fistula rates. J Gastrointest Surg. 2008;12(10):1691–1698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Hackert T, Werner J, Büchler MW. Postoperative pancreatic fistula. Surgeon. 2011;9(4):211–217. [DOI] [PubMed] [Google Scholar]
- 5. Schwarz L, Bruno M, Parker NH, Prakash L, Mise Y, Lee JE, et al. Active surveillance for adverse events within 90 days: the standard for reporting surgical outcomes after pancreatectomy. Ann Surg Oncol. 2015;22(11):3522–3529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Winter JM, Cameron JL, Campbell KA, et al. 1423 pancreaticoduodenectomies for pancreatic cancer: a single‐institution experience. J Gastrointest Surg. 2006;10(9):1199–1210. [DOI] [PubMed] [Google Scholar]
- 7. Bassi C, Marchegiani G, Dervenis C, Sarr M, Abu Hilal M, Adham M, et al. The 2016 update of the international study group (ISGPS) definition and grading of postoperative pancreatic fistula: 11 years after. Surgery. 2017;161(3):584–591. [DOI] [PubMed] [Google Scholar]
- 8. Bassi C, Molinari E, Malleo G, Crippa S, Butturini G, Salvia R, et al. Early versus late drain removal after standard pancreatic resections: results of a prospective randomized trial. Ann Surg. 2010;252(2):207–214. [DOI] [PubMed] [Google Scholar]
- 9. Israel JS, Rettammel RJ, Leverson GE, Hanks LR, Cho CS, Winslow ER, et al. Does postoperative drain amylase predict pancreatic fistula after pancreatectomy? J Am Coll Surg. 2014;218(5):978–987. [DOI] [PubMed] [Google Scholar]
- 10. Lee CW, Pitt HA, Riall TS, Ronnekleiv‐Kelly SS, Israel JS, Leverson GE, et al. Low drain fluid amylase predicts absence of pancreatic fistula following pancreatectomy. J Gastrointest Surg. 2014;18(11):1902–1910. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Yoshino J, Ban D, Ogura T, Ogawa K, Ono H, Mitsunori Y, et al. The clinical implications of peripancreatic fluid collection after distal pancreatectomy. World J Surg. 2019;43(8):2069–2076. [DOI] [PubMed] [Google Scholar]
- 12. Uchida Y, Masui T, Sato A, Nagai K, Anazawa T, Takaori K, et al. Computer tomographic assessment of postoperative peripancreatic collections after distal pancreatectomy. Langenbecks Arch Surg. 2018;403(3):349–357. [DOI] [PubMed] [Google Scholar]
- 13. Sierzega M, Kulig P, Kolodziejczyk P, Kulig J. Natural history of intra‐abdominal fluid collections following pancreatic surgery. J Gastrointest Surg. 2013;17(8):1406–1413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Zhao N, Cui J, Yang Z, Xiong J, Wu H, Wang C, et al. Natural history and therapeutic strategies of post‐pancreatoduodenectomy abdominal fluid collections: ten‐year experience in a single institution. Medicine. 2019;98(22):e15792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Barreto G, D'Souza MA, Shukla PJ, Shrikhande SV. The gray zone between postpancreaticoduodenectomy collections and pancreatic fistula. Pancreas. 2008;37(4):422–425. [DOI] [PubMed] [Google Scholar]
- 16. Hirano S, Kondo S, Hara T, Ambo Y, Tanaka E, Shichinohe T, et al. Distal pancreatectomy with en bloc celiac axis resection for locally advanced pancreatic body cancer: long‐term results [Internet]. Ann Surg. 2007;246(1):46–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Dindo D, Demartines N, Clavien PA. Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg. 2004;240(2):205–213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Akita H, Takahashi H, Gotoh K, Kobayashi S, Sugimura K, Miyoshi N, et al. Closure method for thick pancreas stump after distal pancreatectomy: soft coagulation and polyglycolic acid felt with fibrin glue. Langenbecks Arch Surg. 2015;400(7):843–848. [DOI] [PubMed] [Google Scholar]
- 19. Imamura H, Takahashi H, Wada H, Mukai Y, Asukai K, Hasegawa S, et al. Postoperative aggressive diuresis prevents postoperative tissue edema and complications in patients undergoing distal pancreatectomy. Langenbecks Arch Surg. 2022;407(2):645–654. [DOI] [PubMed] [Google Scholar]
- 20. Asukai K, Akita H, Mukai Y, Mikamori M, Hasegawa S, Fujii Y, et al. The utility of bile juice culture analysis for the management of postoperative infection after pancreaticoduodenectomy. Surgery. 2023;173(4):1039–1044. [DOI] [PubMed] [Google Scholar]
- 21. Kanda Y. Investigation of the freely available easy‐to‐use software ‘EZR’ for medical statistics [Internet]. Bone Marrow Transplant. 2013;48(3):452–458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Eguchi H, Nagano H, Tanemura M, Takeda Y, Marubashi S, Kobayashi S, et al. A thick pancreas is a risk factor for pancreatic fistula after a distal pancreatectomy: selection of the closure technique according to the thickness. Dig Surg. 2011;28(1):50–56. [DOI] [PubMed] [Google Scholar]
- 23. Kawai M, Tani M, Okada K‐I, Hirono S, Miyazawa M, Shimizu A, et al. Stump closure of a thick pancreas using stapler closure increases pancreatic fistula after distal pancreatectomy. Am J Surg. 2013;206(3):352–359. [DOI] [PubMed] [Google Scholar]
- 24. Chang YR, Kang MJ, Kim H, Jang J‐Y, Kim S‐W. The natural course of pancreatic fistula and fluid collection after distal pancreatectomy: is drain insertion needed? Ann Surg Treat Res. 2016;91(5):247–253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Tjaden C, Hinz U, Hassenpflug M, Fritz F, Fritz S, Grenacher L, et al. Fluid collection after distal pancreatectomy: a frequent finding. HPB. 2016;18(1):35–40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Wu AGR, Mohan R, Fong KY, Chen Z, Bonney GK, Kow AWC, et al. Early vs late drain removal after pancreatic resection—a systematic review and meta‐analysis. Langenbecks Arch Surg. 2023;408(1):317. [DOI] [PubMed] [Google Scholar]
- 27. Takahashi H, Ogawa H, Ohigashi H, Gotoh K, Yamada T, Ohue M, et al. Preoperative chemoradiation reduces the risk of pancreatic fistula after distal pancreatectomy for pancreatic adenocarcinoma. Surgery. 2011;150(3):547–556. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Tables S1–S4.
Figure S1.
Figure S2.
Figure S3.
Figure S4.
Figure S5.
Data Availability Statement
The data supporting the findings of this study can be obtained upon reasonable request from the corresponding author, HT. The data cannot be made publicly available as they contain sensitive information that could potentially jeopardize the privacy of the research participants.


